

Global study shows widening disparity in mitral valve disease outcomes
9 DAPT de-escalation
New insight on post-PCI anticoagulation
14 Profile Dipti Itchhaporia


Degenerative mitral valve disease has doubled in prevalence globally over the last 30 years, and though improvements in surgical techniques and advances in transcatheter technologies have led to improvements in rates of mortality, these have arisen in higher income countries—leading to concern over a global disparity in care for patients with mitral valve disease.
These were among the conclusions of research presented at New York Valves 2025 (25–27 June, New York, USA), following analysis of data from the Global Burden of Disease (GBD) study—a worldwide health programme involving more than 14,000 collaborators from over 200 countries—to determine the extent of degenerative mitral valve disease from 1990 through to 2023.
Guido Ascione, a cardiac surgeon and research fellow at the Cardiovascular Research Foundation (CRF) in New York, USA, presented the data which assessed the prevalence of the disease according to population-based cohort studies, population surveys and administrative data from health facilities.
For the purposes of the study, degenerative mitral valve disease was defined as an echocardiographic finding of prolapse or myxomatous degeneration of the mitral valve with mitral regurgitation (MR) ≥2, whilst cases of secondary MR, congenital, rheumatic, or those from infectious causes were excluded from the analysis.
Mortality estimates were established using information from death certificates including vital registration and autopsy data, and only cases in which degenerative mitral valve disease was the primary cause of death were taken into consideration, with estimates presented as absolute and age-standardised rates, and countries stratified based upon sociodemographic index (SDI) to determine their relative wealth.
“Primary MR progressively grew over time; from 1990 to 2023 there was a percentage increase in absolute prevalence of
+126%, and in 2023 there were globally 16 million people with primary MR and at least moderate MR,” Ascione said, noting that prevalence was higher in males than in females.
Looking at age-standardised rates, Ascione commented that the data pointed to the prevalence of the disease being “actually pretty steady”, suggesting that the main driver of the increase in absolute prevalence can be ascribed to a globally ageing population or a lack of improvement in global diagnostic capabilities (i.e. access to healthcare, screening or echocardiography) over the timespan of the study.
“As prevalence increases in age, the rate starts to peak after 60 years of age, and it is the highest in patients that are in between 70 and 74 years,” he detailed. “This means that in 2023, globally there were 11 million people with primary MR that were older than 70 years and four million people were actually older than 80. This has dramatic consequences when
This has dramatic consequences when we think about treatment algorithms for the future”
Artificial intelligence AI predicts valvular regurgitation

HYPERTENSION
Blood pressure guidelines bump up recommendation for renal denervation
NEW GUIDELINES FROM THE American College of Cardiology (ACC) and the American Heart Association (AHA) for the prevention and management of high blood pressure have, for the first time, added renal denervation as a treatment option for patients with resistant or uncontrolled hypertension.
Published in the Journal of the American College of Cardiology (JACC), Circulation and Hypertension, the guidelines give the therapy a class IIb recommendation for patients with a systolic blood pressure of 140–180mmHg and diastolic blood pressure ≥90mmHg despite four drugs, or in patients with uncontrolled hypertension intolerant to drug escalation. All patients with hypertension who are being considered for renal denervation should be evaluated by a multidisciplinary team with expertise in resistant hypertension and in performing the technique, the guidelines state.
Renal denervation is a devicebased treatment in which energy is targeted through a catheter to the renal nerves to modulate the sympathetic signalling between the kidneys and brain to reduce blood pressure.
Two devices have been approved by the US Food and Drug Administration (FDA)—the Paradise (Recor Medical) and Symplicity Spyral (Medtronic) systems—following a review of evidence from trials assessing both technologies, evidence that also contributed to the ACC/AHA guideline change. Medtronic’s Symplicity Spyral system is a minimally invasive procedure that delivers radiofrequency energy to
Continued on page 2
Blood pressure guidelines bump up recommendation for renal denervation
nerves near the kidneys that can become overactive and contribute to high blood pressure, whilst the Paradise system is an ultrasound-based technology.
In July, the US Centers for Medicare & Medicaid Services (CMS) issued its proposed National Coverage Determination (NCD) on renal denervation, recommending coverage for the treatment of uncontrolled hypertension. A final determination is due in October.
“These updated hypertension guidelines are pivotal for physicians, as they validate renal denervation as an additional option for managing hypertension in patients who do not sufficiently respond to lifestyle modifications and pharmaceutical treatments,” said Jason Weidman, senior vice president and president of the Coronary and Renal Denervation business within the Cardiovascular Portfolio at Medtronic, in a press release issued by the company. “To date, we are seeing great interest from healthcare systems across the USA that are looking to offer the Symplicity blood pressure procedure. The updated renal denervation guidelines will enable greater patient access to this potentially life-changing intervention—not only in the USA, but globally as well.”
“As leading voices in cardiovascular health practices, the AHA and ACC strengthen support for renal denervation as a treatment option for high blood pressure,” said Lara Barghout, president and chief executive officer of Recor. “We are pleased to see that the strength of our RADIANCE Global programme formed the basis for the positive inclusion in the guidelines, which not only reinforce the credibility of this therapy—they directly align with the compelling clinical evidence behind the Paradise ultrasound renal denervation system, which has consistently demonstrated safe, effective blood pressure reduction.”
The 2025 Guideline for the Prevention, Detection, Evaluation, and Management of High Blood Pressure in
Adults replaces guidelines from 2017 and includes new or updated recommendations for blood pressure management based on the latest scientific evidence to achieve the best health outcomes for patients.
The new guideline reflects several major changes since 2017, including use of the AHA’s PREVENT (Predicting Risk of cardiovascular disease EVENTs) risk calculator to estimate cardiovascular disease risk. It also provides updated guidance on medication options, including the early treatment for high blood pressure to reduce the risk of cognitive decline and dementia; use of specific medications including the possible addition of newer therapies such as GLP-1 medications for some patients with high blood pressure and who are overweight or obese, and recommendations for managing high blood pressure before, during and after pregnancy.
“High blood pressure is the most common and most modifiable risk factor for heart disease,” said chair of the guideline writing committee Daniel W Jones, professor emeritus of the University of Mississippi School of Medicine in Jackson, USA, who was also a member of the writing committee for the 2017 high blood pressure guideline. “By addressing individual risks earlier and offering more tailored strategies across the lifespan, the 2025 guideline aims to aid clinicians in helping more people manage their blood pressure and reduce the toll of heart disease, kidney disease, type 2 diabetes and dementia.”
“This updated guideline is designed to support healthcare professionals—from primary care teams to specialists, and to all clinicians across health systems— with the diagnosis and care of people with high blood pressure. It also empowers patients with practical tools that can support their individual health needs as they manage their blood pressure, whether through lifestyle changes, medications or both,” Jones said.
The new guideline recommends that healthcare professionals use the PREVENT risk calculator to estimate a person’s risk of a heart attack, stroke or heart failure. Developed by the AHA in 2023, PREVENT is a tool to estimate 10- and 30-year risk of cardiovascular disease in people aged 30–79 years. It includes variables such as age, sex, blood pressure, cholesterol levels and other health indicators, including zip code as a proxy for social drivers of health.
These updated hypertension guidelines are pivotal for physicians, as they validate renal denervation as an additional option for managing hypertension”
Editor-in-chief: Simon Redwood | Publisher: Stephen Greenhalgh
Senior editor: Will Date will@bibamedical.com | Editorial contribution: Jamie Bell, Jocelyn Hudson
Design: Terry Hawes, Josh Lyon and David Reekie
Advertising: Hannah Shiels hannah@bibamedical.com
Subscriptions: subscriptions@bibamedical.com | News or advertising queries Tel: +44 (0)20 7736 8788
Published by: BIBA News, which is a subsidiary of BIBA Medical Ltd
BIBA Medical, Europe, 526 Fulham Road, Fulham, London, SW6 5NR, United Kingdom Tel: +44 (0) 20 7736 8788 BIBA Medical, North America, 155 North Wacker Drive, Suite 4250, Chicago, IL 60606, United States Tel: +1 708-770-7323
Printed by: Buxton Press. Reprint requests and all correspondence regarding the newspaper should be addressed to the editor at the United Kingdom address. © BIBA Medical Ltd, 2024. All rights reserved.
cardiovascularnews linkedin.com/company/cardiovascular-news @cn_publishing
NEWS IN BRIEF
n CABG GRAFT OCCLUSION:
Early bypass graft occlusion occurs significantly more in female compared to male patients after isolated coronary artery bypass graft (CABG) surgery, new research published in The Annals of Thoracic Surgery has shown. The authors of the paper have speculated on a number of the underlying demographic and anatomic factors that may have contributed to their finding.

For more on this story go to page 13.
n TAVI MARKET UPDATE:

Recent months have seen significant moves in the transcatheter aortic valve implantation (TAVI) device market, with Boston Scientific's surprise move to pull the Acurate device from commercial and clinical activities, whilst the proposed acquisition of JenaValve by Edwards Lifesciences has hit an unexpected stumbling block by market regulators in the USA due to competition concerns.
For more on this story go to page 17.
n TEER IN AFMR:
New data from Japan suggest that patients with atrial functional mitral regurgitation (AFMR) undergoing transcatheter edge-toedge-repair (TEER) have significantly lower mortality and heart failure hospitalisation rates than those receiving medical therapy alone. The researchers behind the study, published in the European Heart Journal, suggest that their findings could provide reassurance for the treatment of patients who fall into a "therapeutic grey zone".

For more on this story go to page 20.
Scan the QR code to subscribe
If you have comments on this issue or suggestions for upcoming editions write to will@bibamedical.com
Picture: Cleveland Clinic
Global study shows widening
disparity
in mitral valve disease outcomes
Continued from page 1
we think about treatment algorithms for the future.”
Furthermore, prevalence was shown to be greater in high-income countries compared with low-income countries, which he said could be attributed to greater diagnostic capabilities in these countries as well as the older average age of populations in these regions.
On mortality, Ascione detailed that there had been an increase in absolute number of deaths of around 75% from 1990 to 2023, with a similar trend expected up to 2050. However, age-standardised rates decreased, meaning that there has been a progressive improvement in the treatment of the disease.
“If we dig a little more into age standardised mortality rates, we can see that, starting from 1990, there was a first drop in mortality, and we can clearly attribute this to a progressive improvement in surgery, with standardisation of surgical techniques, and improvement in pre- and post-operative care,” Ascione commented.
“After a steady state in the late 2010s, especially after 2015, there is a second drop, and this is highly probably due to the progressive rise in transcatheter technologies, that offer the treatment option to patients that were not treated before due to high surgical risk.”
However, most of the decreases in mortality had been achieved in high SDI countries, whilst in low SDI countries rates of mortality went up. “This is highly
probably due to a progressive increase in diagnosis that was not followed by an improvement in treatment, and so it opens up huge opportunities for low-income countries to catch up and to fill this gap,” Ascione said in the concluding remarks of his presentation.
Offering a commentary on the findings at New York Valves, Mayra Guerrero (Mayo Clinic Hospital, Rochester, USA) said the finding that degenerative mitral valve disease has increased in prevalence within an ageing population was not surprising, but said that the data highlight the anticipated “surge” in the need for mitral valve interventions coming in the future.
We must be prepared to expand our capacity to treat these patients, but that is the easy part”
“We must be prepared to expand our capacity to treat these patients, but that is the easy part,” said Guerrero.
“What is truly alarming is the profound spike in mortality, significantly higher among women and in countries with low socioeconomic demographic index. While prevalence appears to be lower in these groups, mortality was not only higher, but some regions continue to increase. This raises an important concern: is that prevalence really truly lower, or is the disease under-diagnosed?”
Many patients are being under-diagnosed, under-treated, or treated too late, without access to transcatheter therapies, Guerrero said, resulting
All-female coronary IVL trial reports positive outcomes at 30 days
Thirty-day primary endpoint results of EMPOWER CAD, the first prospective, real-world percutaneous coronary intervention (PCI) study in female patients with complex, calcified coronary artery disease have been presented at EuroPCR 2025 (20–23 May, Paris, France).
THE FAVOURABLE RESULTS confirm earlier retrospective analyses demonstrating the benefits of coronary intravascular lithotripsy (IVL, Shockwave Medical) in female patients.
Women with coronary artery calcification undergoing PCI are often underrepresented in clinical trials and have historically poorer outcomes than men, including increased risk of adverse clinical outcomes and high rates of procedural complications with other calcium modification treatments, such as rotational or orbital atherectomy.
Investigators designed the EMPOWER CAD study to evaluate real-world outcomes in female patients with severely calcified coronary lesions treated with an IVL-first treatment strategy and confirm positive results from previous studies showing similar safety and effectiveness outcomes across both men and women.
EMPOWER CAD is a pivotal study representing Shockwave’s first prospective, post-market study of IVL examining an all-comers female
population with minimal exclusion criteria, the first study to encourage a Shockwave IVL-first PCI strategy for calcified coronary lesions, and the first study to follow-up with enrolled patients for three years following IVL therapy.
The study enrolled 399 participants across 45 sites spanning five countries in the European Union and USA. The

in poorer outcomes and contributing to the disproportionate rates of mortality seen in the study.
“This is not just an inconvenient truth, this is a serious and urgent problem,” she added. “Let’s treat this moment as a turning point, as a call to action to address these important healthcare disparities. In just 20 years, TAVI [transcatheter aortic valve implantation] has transformed from a bold idea into a global movement. In that spirit, I will leave that with 20 years of TEER [transcatheter edge-to-edgerepair], and almost a decade of transcatheter mitral valve replacement, we now must push to make mitral valve care a truly global movement, one that ensures accessibility, standardisation and reproducibility for all patients, regardless of sex, regardless of country of origin.”
In response to Guerrero’s comments, Ascione said that steps to reduce the disparity should begin with optimised and earlier diagnosis and identifying the patients that are most likely to benefit from treatment.
“We should come up with algorithms that account for the growing and ageing population,” he said. “Access to healthcare is a great problem, because primary MR—especially in the form of prolapse and valve disease—is clearly something that is also happening in the lower-income countries.”
Juan Granada, president and chief executive officer of the CRF and a co-author of the study, said that the data would be important to create awareness, “so that companies know what the real problem is” regarding disease prevalence.
“We have a big access problem, disparity of care, inequality in our field that needs to be addressed,” he said. Further research will be undertaken to assess the global burden of mitral annular calcification (MAC), Granada revealed.
study is led by co-principal investigators Margaret McEntegart (Columbia University Medical Center/NewYorkPresbyterian Hospital, New York, USA), who presented the results at EuroPCR, and Alexandra Lansky (Yale University School of Medicine, New Haven, USA). The study’s European lead is Nieves Gonzalo (Hospital Clinico San Carlos, Madrid, Spain).
“Women are typically underdiagnosed, underrepresented and have underwhelming outcomes in coronary artery disease studies,” said Lansky. “Our goal with the EMPOWER CAD study was to address this gap and determine whether Shockwave IVL should be considered as the front-line calcium modification approach in female patients with complex coronary artery disease. The results demonstrate the benefits of IVL use in this specific patient population and validate findings from previous studies.”
Women are typically underdiagnosed, underrepresented and have underwhelming outcomes in coronary artery disease studies”
The primary effectiveness endpoint of procedural success, defined as stent delivery with ≤30% residual stenosis for all treated target lesions without inhospital target lesion failure (TLF), was 86.9% at 30 days.
The primary safety endpoint, defined as TLF as a composite of cardiac death, myocardial infarction attributable to target vessel (TV-MI) or ischaemiadriven target lesion revascularisation (ID-TLR) within 30 days, was 12.1%. TLF was primary driven by the rate of periprocedural MI3 (10.6%); the majority of these periprocedural MIs were not associated with any clinical signs or symptoms. Cardiac death and ID-TLR rates were each 1.3%.
Procedural complications were rare with only one patient (0.2%) having a serious angiographic complication when assessed at the end of the procedure. There were also clinically and statistically significant improvements in health-related quality of life scores at 30 days compared to baselines.
“By leveraging a Shockwave IVLfirst approach, the EMPOWER CAD 30-day results demonstrated high procedural success, low complication rates and significant improvements in quality of life among real-world female patients,” said McEntegart. “While we look forward to following these patients for the next three years, these primary endpoint results confirm that excellent outcomes can be achieved with the use of Shockwave IVL in women with complex, calcified coronary artery disease.”
Margaret McEntegart

SAME SHOCK, BIGGER WAVES
Shockwave Medical is Now Part of J&J MedTech
As part of J&J MedTech, we’ll continue to deliver our same unmatched commitment to innovation, customer service and improving patient outcomes — with the added energy of the global leader in healthcare.
Stroke rates diverge among TAVI and surgery patients at five years in pragmatic trial
Long-term results of the UK TAVI trial, a pragmatic trial comparing transcatheter aortic valve implantation (TAVI) to surgical aortic valve replacement (SAVR) point to comparable rates of mortality, myocardial infarction (MI), endocarditis and reintervention between the two strategies at five years, but a higher incidence of stroke among TAVI patients.
“MOST OF THE PREVIOUSLY published major TAVI trials have focused on specific valves and were explanatory rather than pragmatic in their design,” said William Toff (University of Leicester, Leicester, UK), the study’s lead investigator, during a late-breaking trial session at the 2025 EuroPCR congress (20–23 May, Paris, France), where he reported the five-year findings of the study, which set out to compare the clinical effectiveness and cost utility of the two approaches.
Patients enrolled in the trial—which took place at TAVI centres across the UK—had severe, symptomatic aortic stenosis, were aged at least 70 years (with a median age of 81 years), and due to comorbidities or advanced age, were thought to be at increased operative risk.
“The trial had deliberately broad inclusion criteria and permitted the inclusion of patients with bicuspid valves, and those needing revascularisation,” said Toff. Unlike many of the industry-funded TAVI
trials, UK TAVI permitted the use of any CE-marked TAVI valve, with no predefined risk score threshold for inclusion. Toff commented that the Society of Thoracic Surgeons (STS) risk score among the trial’s 913 participants stood at a median of 2.6, making it more akin to a low-risk trial.
One-year results of the trial, published in the Journal of the American Medical Association (JAMA) in 2022, demonstrated the noninferiority of TAVI to SAVR for all-cause mortality, which was the primary endpoint. TAVI patients had a shorter hospital stay and fewer bleeding events, although significantly more vascular complications and conduction disturbances requiring pacemaker implantation.
At five years, Toff reported at EuroPCR 2025 that there were no differences in all-cause deaths or those attributable to cardiovascular causes, but did note that there was an excess of strokes in the TAVI arm, which he said was not a “procedural phenomenon”, with the curves having begun to
Search for suitable candidates for cerebral embolic protection continues
A pooled, individual patient-level data meta-analysis of two trials investigating the use of the Sentinel (Boston Scientific) cerebral embolic protection device has found no evidence of a reduction in stroke when the device is routinely used during transcatheter aortic valve implantation (TAVI).
THE ANALYSIS OF THE PROTECTED TAVR and BHF PROTECT TAVI trials, presented at EuroPCR 2025 (20–23 May, Paris, France), did show that severe, disabling strokes may be reduced among patients in whom the Sentinel device is fully deployed, according to investigator Rajesh Kharbanda (John Radcliffe Hospital, Oxford, UK), who said work is ongoing to establish which patients at high risk for stroke may confer a benefit from Sentinel.
“Stroke remains an issue even with modern day TAVI, and the Sentinel cerebral embolic protection device captures debris and reduces its incidence of going to the brain,” Kharbanda said at a press conference at EuroPCR.
diverge at around four months, and continuing to do so through to five years. Around half of these strokes were disabling.
Quality of life metrics showed an early advantage for TAVI persisting out to one year, although from two years onwards, there was no significant difference between the two strategies.
“UK TAVI is the first large pragmatic trial comparing a TAVI strategy with a surgical strategy to report five-year outcomes,” said Toff. “The findings are consistent with those of the explanatory trials, showing a similar treatment effect in respect of mortality, reintervention, endocarditis and MI, but we did identify an increased incidence of stroke after TAVI that clearly requires further elucidation.”
Stroke Rates
belong to the group of surgeons that think that all industry sponsored trials are inherently evil—I see them as very, very useful. However, I readily acknowledge that industry will not sponsor a trial unless they think there is a reasonable chance that they will get a new indication or an indication expansion. That is why it is very important that we have these industry independent studies confirming what we see in industry studies.”
12.9%
7.3%
Commenting on the trial’s results during discussion that followed the presentation at EuroPCR, Michael Borger (Leipzig Heart Center, Leipzig, Germany) praised the research as providing an important addition to the results garnered from industry sponsored TAVI trials, which comprise the majority of the long-term data accumulated to date.
“I am a cardiac surgeon but I don’t
Borger also praised the broad inclusion criteria of the trial, and the device agnostic approach chosen by the investigators.
“The results you very clearly stated with a higher pacemaker rate than what we have seen in the modern low-risk industry sponsored trials, and also the higher stroke rate at five years, which is a novel finding,” he added, asking if there were any protocols in the trial concerning anticoagulation. Toff responded that there was no prespecified management strategy in UK TAVI, noting that there was a slightly higher use of anticoagulation in the surgery group, which may have been a contributor to the difference in stroke rates.
The findings are consistent with those of the explanatory trials, showing a similar treatment effect in respect of mortality, reintervention, endocarditis and MI”
Taken together, PROTECTED TAVR, conducted in the USA and Europe, and BHF PROTECT TAVI, conducted in the UK, enrolled more than 10,000 patients, with both trials neutral for the primary endpoint of all-cause stroke at 24 hours.
The present study includes a primary endpoint of all-cause mortality with a secondary prespecified endpoint of disabling stroke, using a modified intention to treat approach. Investigators used a complier average causal effect (CACE) analysis to adjust for patients whose devices were not deployed for the entirety of the procedure—for example due to complex anatomy caused by advanced peripheral vascular disease—and a specified perprotocol analysis of patients who received the device as intended with both filters deployed.
patients undergoing TAVI with protection and 1% without, and 1.3% vs. 0.8% respectively within the per-protocol analysis.
“That is a complex set of data, and I want to summarise by saying that what this meta-analysis shows is that there is no evidence that, for all stroke, a routine strategy of embolic protection reduces incidence,” the presenter stated.

In the modified intention to treat analysis, Kharbanda showed that the incidence of stroke stood at 2.3% in the group undergoing TAVI with Sentinel, versus 2.2% among those without, whilst in the per-protocol analysis of patients whose filters were present for the entire duration of the procedure these figures stood at 2.3% and 1.7%, respectively. CACE analysis showed there was no difference between the groups when adjusted for non-compliance. “The summary of that is that all-cause stroke is not reduced by this device,” Kharbanda commented.
Looking at disabling stroke, the modified intentionto-treat analysis showed an incidence of 1.3% among
Any hint of an effect in a reduction in disabling stroke needs further analysis, Kharbanda stated, adding that the perprotocol analysis can be prone to postrandomisation error and selection bias.
“We need to understand what that signal means. As part of that, further work is under way to identify who is at risk of TAVI-associated stroke and therefore identify if cerebral embolic protection might be effective in that subgroup. Using this dataset, we will be able to answer some of those questions.”
Questioned by William Wijns (Lambe Institute for Translational Medicine, University of Galway, Galway, Ireland) on which subgroups of patients may be the beneficiaries of the technology, Kharbanda said that it is “still early days” in looking at the data. However, he said that factors including a high surgical risk score, prior stroke and peripheral vascular disease may be at play. “I suspect it is not one factor, and that is the problem with univariate [analysis], when you look at single factors it is a combination of factors.”
Rajesh Kharbanda
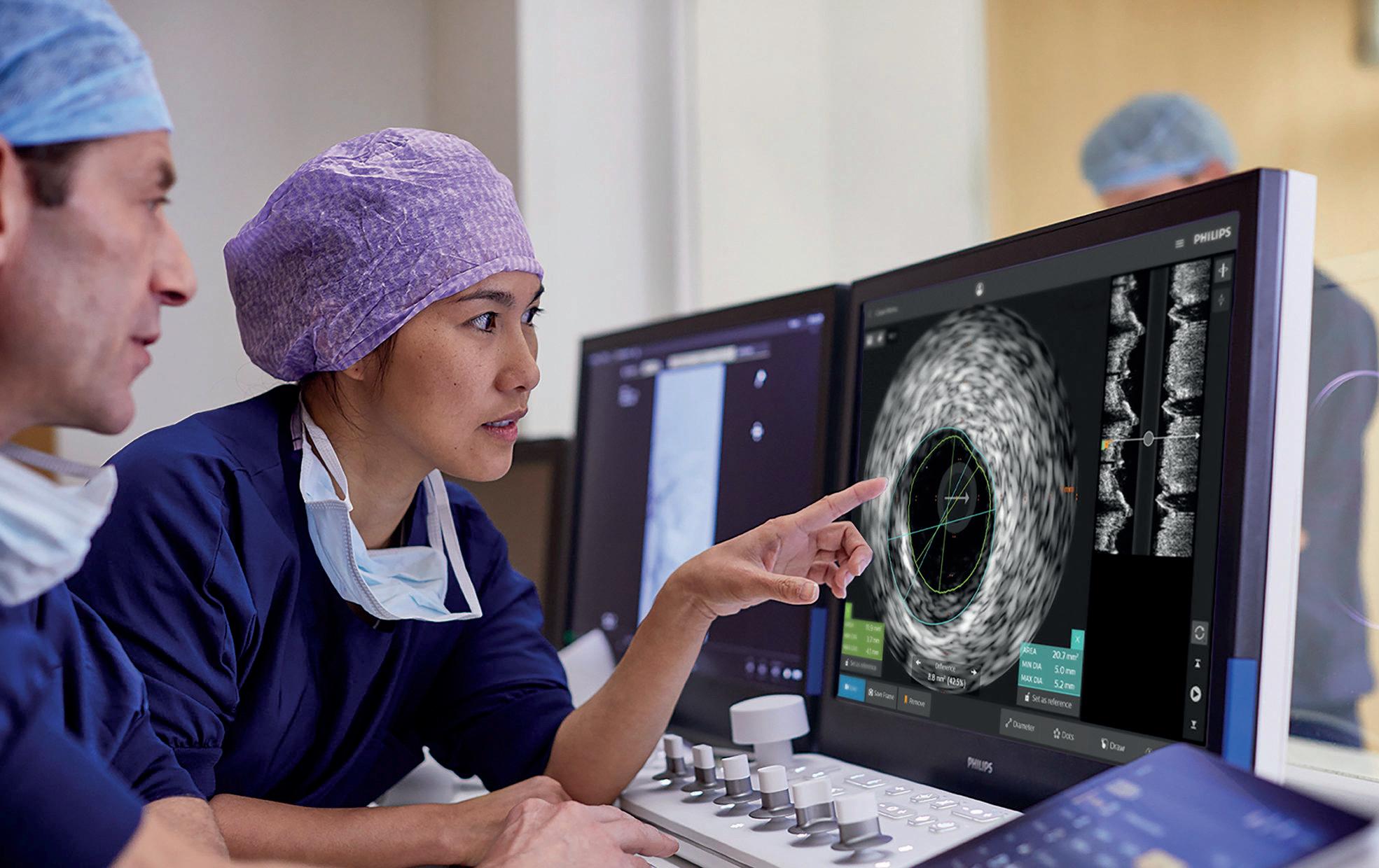
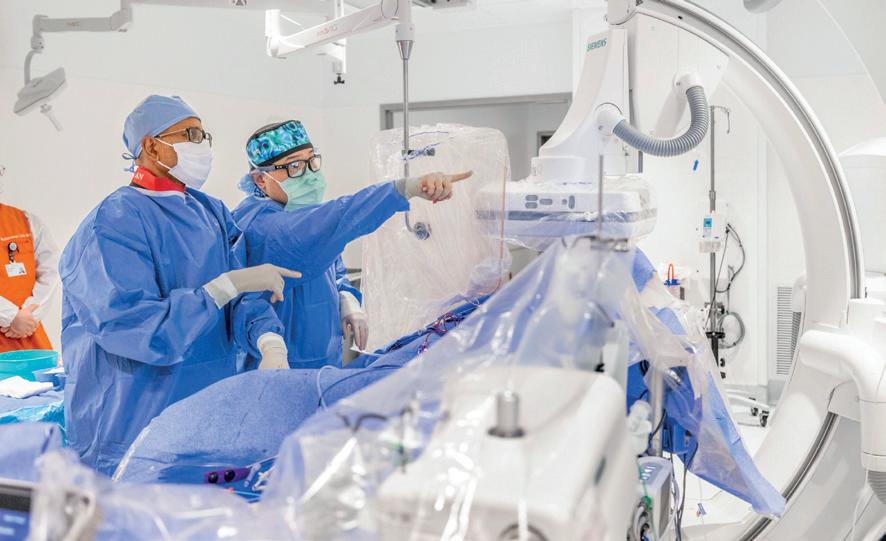
New focus on education needed to improve uptake of intracoronary imaging
Better provision of hands-on education and case-based learning models are among the actions that will help to drive greater adoption of intravascular imaging technologies to support coronary interventions.
This is according to a panel of experts who have reviewed current barriers to more widespread usage of imaging technologies—including intravascular ultrasound (IVUS) and optical coherence tomography (OCT)—which remains low despite latest clinical practice guidelines favouring their use in specific clinical and anatomical scenarios.
Last year, updated European Society of Cardiology (ESC) chronic coronary syndrome guidelines brought in a class 1, level of evidence A recommendation for imaging in complex scenarios including left main disease, bifurcations, or long lesions, with subsequent guidelines from the American College of Cardiology (ACC) and American Heart Association (AHA) also carrying a similar weight of recommendation.
Advocates of intracoronary imaging were optimistic that these guideline changes would bring about a shift in practice to see more imaging used for coronary interventions, but a wide-ranging white paper published in the Journal of the Society for Cardiovascular Angiography & Interventions (JSCAI) by authors Javier Escaned (Hospital Clínico San Carlos, Madrid, Spain) et al acknowledges that adoption has remained low in many regions with wide disparities in usage opening up across the world.
“The question is: how is it possible that something
that contributes to saving lives and has a 1A recommendation in clinical practice guidelines does not get accepted?” Escaned comments to Cardiovascular News, setting out the rationale for the discussion paper. “I was discussing this with colleagues, and we thought we didn’t need one more paper highlighting the importance of this for clinical benefit or giving a clinical consensus statement, we need to explore what is going on here.”
As well as looking at current rates of usage of intracoronary imaging and existing barriers to adoption, Escaned and colleagues set out a series of concrete actions that they believe will cement the use of imaging into clinical practice—with an aim to encourage acceptability, acceptance and adoption of imaging amongst interventionalists. Whilst recommendations cover a broad set of issues including referral, reimbursement, data collection and reporting, and engagement with cath lab teams, there is strong emphasis on education and the way that interventionalists are trained in when and how to use these technologies.
“In some regions reimbursement has been suggested as a reason. It is important to advocate for better financial incentives. After all, imaging permits better optimisation and more accurate evaluation of the
stent results with fewer stent failures and event rates,” Mirvat Alasnag (King Fahd Armed Forces Hospital, Jeddah, Saudi Arabia) tells Cardiovascular News
“Another important factor, however, is training. It is important to note that up until five years ago, the majority of operators were trained in the non-IVUS/ OCT era. They’ve grown comfortable sizing stents and identifying landing zones without imaging. As such, they didn't feel the necessity of intravascular imaging despite the plethora of evidence.
“In order to get them to that level of comfort in image acquisition and interpretation, robust training programmes and technical support were necessary in coordination with industry.”
Regional variation
Though the use of intravascular imaging is seen as a relatively new innovation in the field of coronary interventions, Escaned et al’s paper identifies that these technologies have been in circulation for several decades, noting that in Japan, for example, where as many as 84.4% of percutaneous coronary intervention (PCI) procedures involve the use of IVUS or OCT in some form, the use of intracoronary imaging has been relatively well established since the mid-1990s.
However, the East Asian country stands as an outlier globally, as nearly all other nations lag significantly behind in their uptake of intracoronary imaging—a
It is important to note that up until five years ago, the majority of operators were trained in the nonIVUS/OCT era”
Specialists at an IVUS console
fact that is attributed to a multitude of issues including availability of technologies, lack of training and inconsistencies in reimbursement, to name just a few.
In the UK—which is among the more rapid adopters of intracoronary imaging according to figures quoted in the JSCAI paper—imaging is used in around 25% of cases. “In recent years there has been considerable progress in the adoption of intracoronary imaging within the UK—the majority of left main procedures are now done with intravascular ultrasound. This is at least in part to do with continued emphasis by the British Cardiovascular Intervention Society (BCIS) on quality improvement and optimising patient outcomes,” Adrian Banning (Oxford Heart Centre, John Radcliffe Hospital, Oxford, UK), a co-author of the white paper, tells Cardiovascular News, reflecting on how practice has begun to shift in the UK. “However, it is clear that use of imaging in the UK is still patchy and there remain areas where use of imaging is infrequent, even in complex procedures.”
The USA is another more rapid adopter of intracoronary imaging techniques, with the JSCAI paper quoting usage at around 15.4%, but it is noted that there is wide variability within the country, “mainly driven by hospital and physician rather than PCI complexity or patient-level characteristics”. This is underscored by data published in The American Journal of Cardiology in July showing that despite an increase in intravascular imaging usage over time, prevalence of imaging is far lower in the country’s southern states (7.6%) than in western states (12.6%), for example—hinting at a disparity in care opening up at more local level.
outcomes are as good with angiography-guided PCI as with intravascular imaging-guided PCI, despite more than 20 randomised trials showing the opposite,” he comments.
Stone says that he remains optimistic that with class I guidelines now in place in both the USA and Europe for the routine use of intravascular imaging guidance for PCI in complex lesions, adoption rates will increase to a more acceptable level. “Such rates are being achieved in certain states such as Michigan and hospital systems such as the Veterans Affairs (VA) where clinical pathways for optimal practice are implemented and metrics are reported,” he says.
Communication breakdown
If the UK and USA are among the more rapid adopters in the western world, of the major European nations highlighted in the paper, France has the lowest overall use of intracoronary imaging as a percentage of PCI, with an overall rate of 1.7%.
According to Escaned, the reason for such wide disparities among developed nations rests with how the benefits and use of the technologies are communicated to interventionalists.

“Intravascular imaging use for guidance of PCI procedures remains low in the USA, despite indisputable evidence that such routine use substantially reduces mortality, myocardial infarction, stent thrombosis, and repeat intervention,”
Gregg W Stone (Icahn School of Medicine at Mount Sinai, New York, USA), who has contributed to several key randomised trials in this domain, tells Cardiovascular News, discussing current usage amongst US interventionalists.
According to Stone, there are a multitude of reasons why adoption continues at a slow rate in the USA, citing factors including poor training, as well as operator perception of the technologies. “I believe the major factors are lack of training and knowledge of how to implement all the information inherent in IVUS and OCT images into actionable practice (most common in older physicians); perception of increased time required; lack of reimbursement; and frankly, hubris—the mistaken belief by selected physicians (again, mostly the older generation) that their patient
“Interestingly, I’ve heard some cardiologists argue that the limited use of imaging in their countries proves it isn’t necessary. My question is: what advice would these cardiologists give to a colleague practicing in Japan, where utilisation rates are considerably higher?”
How is it possible that something that contributes to saving lives and has a 1A recommendation in clinical practice guidelines does not get accepted?”
Javier Escaned
Escaned comments to Cardiovascular News, citing this as a form of “cognitive dissonance” in effect, whereby operators who have been educated in an angiographybased culture may have their expectations challenged by the introduction of new technologies.
Escaned believes that “practical” educational models will be key to overcoming this challenge. “The way that you use imaging in clinical practice differs for
the different clinical subsets and education has to be focused on what actionable information you obtain from IVUS in each different scenario that you can apply to your intervention, so that is something that is truly important.”
Offering examples of how educational activities could be changed to demystify intracoronary imaging techniques for operators who may have reservations about using them, Escaned says that hands-on or case-based learning models may facilitate uptake more effectively than traditional evidence reviews or technical lectures, whilst activities should promote clinical benefit as well as seeking to improve imaging interpretation skills.
Additionally, Escaned says that users must be shown techniques in a way that can be easily transposed into their own practice and says that educators must avoid portraying intracoronary imaging as something that only the top experts can replicate.
“If you are showing a live case that is guided with intracoronary imaging, and you see that there is a super expert by the table performing IVUS and making the analysis, you are extending the idea that you need such a person to use IVUS in your cath lab,” he comments. “It is completely wrong. The logical idea is that you see that it is used by operators, that it is fully integrated in what they do. It is an example of how you may be sending the wrong message.”
Educational model
Escaned says that the framing of debate in educational sessions as having individuals speaking for or against the use of the technology may also be counterproductive. “With a 1A recommendation it should be crystal clear that intracoronary imaging in complex PCI saves lives,” he says. “In my opinion, there is no room for giving the microphone to someone who says that this is not really working; once the evidence and expert opinion are so well established, attention should move from debate to promoting adoption.”
Of the potential impact of the white paper, Escaned says he hopes that all stakeholders involved in medical education, including industry, will take into consideration the suggestions put forward to expand the practice of intracoronary imaging.
“Hopefully all colleagues that are involved in medical education and congresses will try to follow these recommendations on how to provide educative models that avoid the problems we have highlighted and align better. Industry also plays a very important role in education. Making sure that the first experience the interventional cardiologists have with intracoronary imaging is successful, and they realise that they are getting actionable information to improve the procedures, these are aspects that are important.”
White paper recommendations for improving intracoronary imaging uptake
Addressing
Sirolimus

A countdown to celebrate every step we’ve taken together watch the video

10. Dr. Willem Dewilde, Belgium. 9. Dr. Grzegorz Sobieszec, Poland. 8. Dr. Ander Regueiro, Spain. 7. Dr. Chung Tak Shun, Hong Kong. 6. Dr. Armando Pérez de Prado, Spain. 5. Dr. Bipin Dubey, India. 4. Dr. Alberto Bazzoni, Mexico. 3. Dr. Manel Sabaté, Spain. 2. R&D team, iVascular. 1. Lluís Duocastella, CEO iVascular. Teresa Codina, iVascular CEO’s Mother (two Angiolite’s stents saved her life).
Meta-analysis supports benefit of P2Y12 inhibitor monotherapy after short DAPT in ACS patients
A meta-analysis of trials studying dual antiplatelet therapy (DAPT) de-escalation strategies after percutaneous coronary intervention (PCI) in patients with acute coronary syndrome (ACS) has shown that outcomes vary according to whether patients receive aspirin or P2Y12 inhibitor monotherapy thereafter.
WRITING IN JACC:
Cardiovascular Interventions, Davide Capodanno (University of Catania and G. Rodolico-San Marco Hospital, Catania, Italy) and colleagues note that several studies have investigated deescalation strategies to mitigate the risk of bleeding associated with DAPT after PCI. Short DAPT followed by one of the two antiplatelet agents—P2Y12 inhibitor or aspirin monotherapy—have emerged as options to balance safety and efficacy.
However, the authors note in their JACC: Cardiovascular Interventions paper that no trial has directly compared the two approaches, with previous metaanalyses comparing the two strategies having provided mixed results.
To shed more light on this, Capodanno et al performed a pairwise meta-analysis with subgroup interaction and network meta-analysis to evaluate the safety and efficacy of short DAPT and compare the merits of subsequent aspirin or P2Y12 inhibitor monotherapy in ACS patients undergoing PCI. The
primary outcome was trial-defined net adverse clinical events (NACE), a composite of ischaemic and bleeding events, whilst secondary outcomes included single components of the primary endpoint.
Researchers included 23 studies, encompassing 45,394 patients in total, of which 14 evaluated the effects of short DAPT followed by aspirin monotherapy, and nine evaluated short DAPT followed by P2Y12 inhibitor monotherapy. Demographic and procedural characteristics were noted to be similar across DAPT de-escalation trials, with a weighted mean age of 63 years, and with women totalling 23%.
these differences resulted in a nonsignificant reduction in NACE for short DAPT compared with standard DAPT. At indirect comparison, P2Y12 inhibitor monotherapy significantly reduced the risk of NACE compared with aspirin monotherapy, and ranked first for reducing NACE events according to surface under the cumulative ranking curve (SUCRA) evaluation. The quality of evidence was deemed moderate for the pooled analysis and aspirin monotherapy subgroup, but high for the P2Y12 inhibitor monotherapy subgroup.

“The overall net benefits of short DAPT were mainly driven by P2Y12 inhibitor rather than aspirin monotherapy, with significant interaction between the two regimens for NACE and any bleeding,” Capodanno et al write in their summary of
the findings. “At indirect comparison, P2Y12 inhibitor monotherapy significantly reduced NACE and any bleeding compared with aspirin monotherapy,” they state. Furthermore, according to a trial sequential analaysis, the results of reduced bleeding without significant increase in ischaemic events appear to be conclusive for P2Y12 inhibitor monotherapy, but not aspirin monotherapy, the researchers note.
Capodanno et al reported that, compared with standard DAPT, P2Y12 inhibitor monotherapy, but not aspirin monotherapy, reduced the risk of NACE, resulting in significant statistical interaction. Pooled analysis showed that
The overall net benefits of short DAPT were mainly driven by P2Y12 inhibitor rather than aspirin monotherapy”
Davide Capodanno
DynamX coronary bioadaptor “durable” through three years
Elixir Medical has announced three-year results from the 445-patient BIOADAPTOR 1:1 randomised controlled trial (RCT), comparing the DynamX coronary bioadaptor system to Resolute Onyx (Medtronic) drug-eluting stent (DES) from 34 centres in Japan, Europe, and New Zealand.
THE RESULTS DEMONSTRATE SUSTAINED very low adverse events and durability of clinical outcomes with DynamX in target lesion failure (TLF) and cardiovascular death (CVD) compared to nonplateauing increase in adverse events in the DES arm. The data were presented at a late-breaking clinical session during the 2025 EuroPCR congress (20–23 May, Paris, France).
Clinical results show sustained significant reduction of device-oriented adverse events with DynamX bioadaptor over the drug-eluting stent at three years, including a significant reduction in TLF rate (2.7% vs. 7.2%; p=0.030) demonstrating the durability of DynamX treatment through three years of follow-up.
The significant reduction in TLF was driven by low adverse events across all components of the composite endpoint with DynamX compared to DES, respectively, including significantly lower CVD (0.5% vs. 3.2%, p=0.033), target-vessel myocardial infarction (0.9% vs. 1.8%) and ischaemia-driven target lesion revascularisation (1.4% vs. 2.7%)
The reduction in adverse events was amplified in left anterior descending (LAD) artery lesions, with a significantly lower TLF rate in LAD lesions (2.7% vs. 10.6%; p=0.019) consistent with bioadaptor mechanism of action of restoring vessel function in this haemodynamically critical coronary vessel as reported previously in the imaging subgroup at 12 months.
“These three-year results from the BIOADAPTOR RCT demonstrate that DynamX bioadaptor provides for a new class of treatment—with significant clinical benefit from six months when the bioadaptor mechanism of action is activated and sustained through long-term follow-up, including the hard clinical endpoint of cardiovascular death,” said Shigeru Saito (Shonan Kamakura General Hospital, Kamakura, Japan). “We also see the heightened importance of restoring haemodynamic modulation in the LAD vessel demonstrated by the large difference in clinical events in LAD lesions between the two treatment arms.”
The DynamX bioadaptor has a novel mechanism of
“Our study supports the increased net benefit of P2Y12 inhibitor rather than aspirin monotherapy after short DAPT. Specifically, compared with standard DAPT, significant interaction for the monotherapy administered was detected for NACE, as P2Y12 inhibitor monotherapy resulted in a 26% relative risk reduction, whereas aspirin monotherapy was associated with neutral results. Additionally, P2Y12 inhibitor monotherapy reduced NACE by 23% when compared with aspirin, with consistent findings after accounting for between-trial heterogeneity,” the authors state.
There are limitations to the study, Capodanno et al note, in particular the absence of patient-level data did not allow exploration of the effects of possible confounders on overall treatment estimate—though they note that this may have been mitigated by the performance of several subanalyses and meta-regression analyses. Additionally, the definition of NACE varied across trials, which, they state, may have introduced some heterogeneity.
action designed to return vessel health through three distinct phases of adapting in the body to restore vessel biology. In the locked phase during the implant, the bioadaptor opens the artery and restores blood flow. After six months the implant unlocks, separates into three helical strands, releasing the vessel and providing dynamic support to maintain the established blood flow lumen.
The continued adaptive dynamic support helps return vessel haemodynamic modulation through restoration of pulsatility and adaptive blood flow volume and has also shown evidence of plaque stabilisation and regression in the lesion, Elixir Medical says in a press release.
“With these results, we are pleased to demonstrate that restoring haemodynamic modulation of the artery translates to lasting clinical outcomes, elevating the level of care for patients,” said Motasim Sirhan, chief executive officer of Elixir Medical. “With the threeyear BIOADAPTOR-RCT data, and the results from the 2,400 patient INIFINITY-SWEDEHEART RCT published in The Lancet, we have shown consistently across multiple trials significantly lower, plateauing clinical events after six months with DynamX bioadaptor compared to DES, validating the benefit of our transformative technology.”
We also see the heightened importance of restoring haemodynamic modulation in the LAD vessel”
DynamX coronary bioadaptor

Surgeons complete pioneering intercontinental robotic cardiac procedure
SS INNOVATIONS
International has announced the successful completion of an intercontinental robotic cardiac telesurgery utilising the company’s SSi Mantra 3 surgical robotic system.
Sudhir Srivastava, who is the company’s chairman and chief executive officer, performed the cardiac procedure, a robotic atrial septal defect closure, remotely from the global headquarters of the Institute of Research Against Digestive Cancer (IRCAD) in Strasbourg, France, during the Society of Robotic Surgery (SRS) annual meeting (16–20 July, Strasbourg, France) at a transmission distance of more than 4,000 miles to the operating room at the Sri Aurobindo Institute of Medical Sciences (SAIMS) in Indore, India.
Patient-side support in Indore was coordinated by Lalit Malik (Manipal Hospital, Jaipur, India), Ram Krishna Shukla (SAIMS, Indore, India), Bipin Arya (SAIMS, Indore, India), and the SS Innovations and SAIMS teams.
The live telesurgery was moderated by Husam H Balkhy (University of Chicago Medicine, Chicago, USA), president of the International Society for Minimally Invasive Cardiothoracic Surgery (ISMICS).
“I would like to extend my gratitude to Dr Vipul Patel, chairman of the Society of Robotic Surgery for this opportunity at the SRS 2025 conference, Dr Mohit Bhandari, the IRCAD India team, and the entire SS Innovations team whose dedication and innovation made the world’s first intercontinental robotic cardiac telesurgery possible,” commented Srivastava. “This historic achievement not only demonstrates the advanced capabilities of our SSi Mantra 3 surgical robotic system but also further paves the way for redefining and democratising global delivery of complex cardiac care, especially in regions with limited access to surgical expertise.”
The surgical robotic technology platform, including its telesurgery capabilities, addresses traditional obstacles related to travel, time, cost, and surgical accessibility, creating a more efficient and equitable medical standard for both patients and surgeons, the company says in a press release.
To date, 35 telesurgeries including 10 cardiac procedures, and a total of 250 cardiac procedures have been completed utilising the SSi Mantra surgical robotic system.
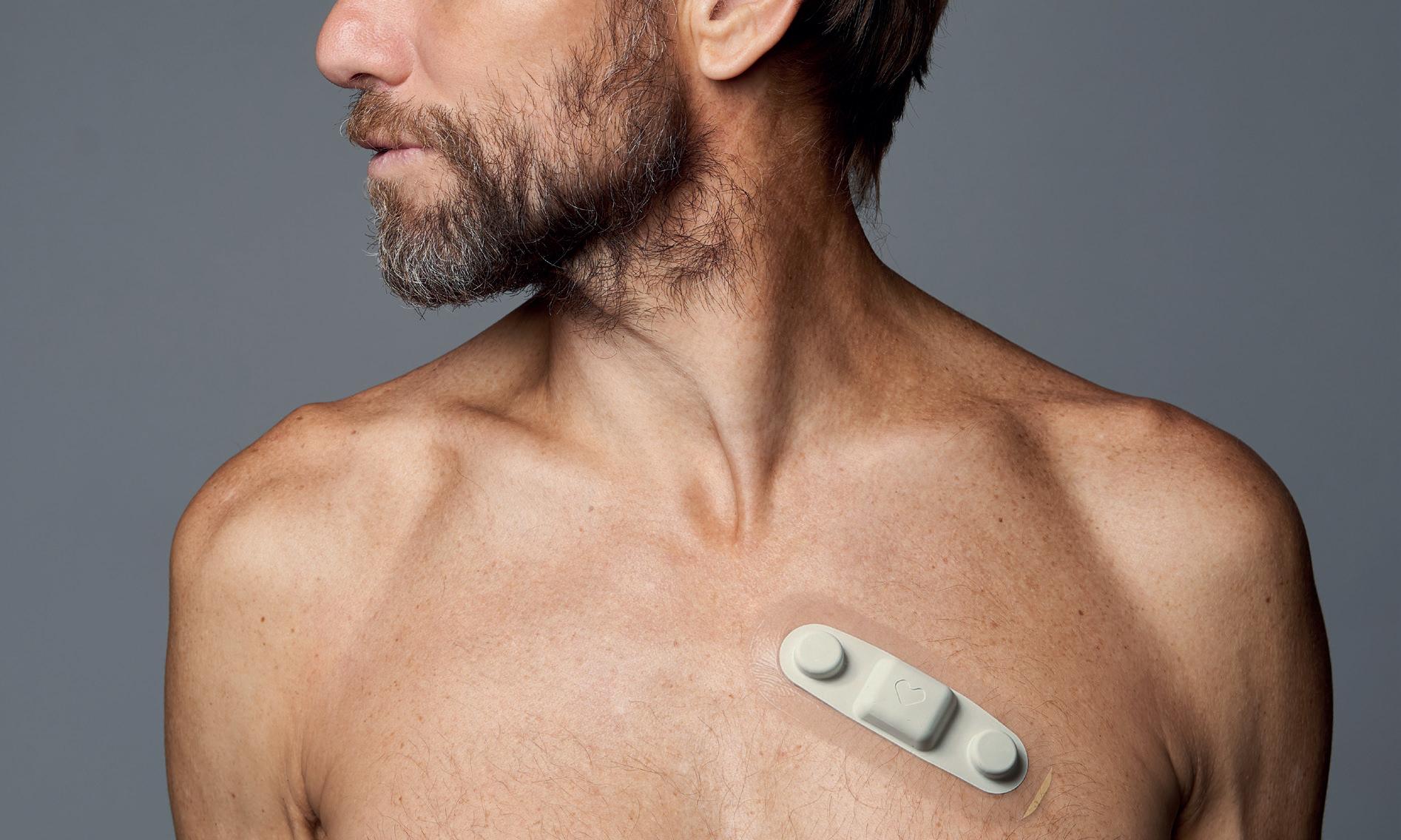
Wearable patch may speed up diagnosis of atrial fibrillation after cardiac surgery
The use of a wearable electrocardiogram (ECG) monitor for up to 14 days after cardiac surgery may improve the detection of postoperative atrial fibrillation (poAF), potentially speeding up the detection of cases which may have gone unseen until up to three months’ follow-up, researchers have shown.
PRELIMINARY RESULTS OF A 100-PATIENT
study involving the use of the Vivalink wearable ECG patch were presented at Euroanaesthesia 2025 (25–27 May, Lisbon, Portugal), in which researchers analysed data from the wearable monitor after discharge. The study followed patients undergoing cardiac surgery procedures at Brigham and Women’s Hospital (Boston, USA),
Of the included patients, 42% experienced poAF during hospitalisation, and 27% showed signs of atrial fibrillation on the patch monitor recordings. In 24% of patients with AF detected on the patch monitor, the arrhythmia was first identified on the wearable monitor and had not been detected while hospitalised. In 80% of these cases, AF was not diagnosed using standard modalities until the three-month follow-up.
“We’re pretty good in general at continuously monitoring patients so that while they are in the operating room (OR) or while they’re in the intensive care unit (ICU) there are hardly any gaps in monitoring. The real gap occurs when patients are discharged home,” Jakob Wollborn, a cardiac anaesthesiologist and intensivist at Brigham and Women’s Hospital, who is a co-author of the research, tells Cardiovascular News
“In the hospital it’s more or less simple, either they’re on continuous telemetry, if they don’t feel well and there’s enough suspicion of an irregular pulse you’ll probably do an intermittent 12-lead ECG. When you’re at home patients may just simply not feel great, but who has the opportunity to run an ECG or a smartwatch-based rhythm analysis at home? Certainly not all of our patients,” says Wollborn.
Vivalink’s wearable ECG monitor is a small, reusable, rechargeable patch that streams to a biometrics data platform in the cloud, which includes an arrhythmia detection algorithm and clinician portal.
“The device that we used from Vivalink is essentially an adhesive that is put on the chest, it’s probably a coin-sized
device and doesn't need to be recharged every day, it has continuous battery and connects to a patient’s smartphone that then transmits the data onwards,” says Wollborn.
He notes that the study remains ongoing, and results may differ when the full study has been completed, but the researchers are optimistic about the utility of this device and other wearables in detecting AF after surgery.
“What we’re seeing is certainly that patients do develop AF after hospital discharge and we’ll ask them whether they noticed, and a lot of them say no,” he comments.
“It’s valuable to detect poAF when it occurs, because it’s obviously shown to increase the risk for stroke from embolic complications and AF in general is dangerous. There are studies showing that 24 hours of even subclinical AF does increase the risk for stroke. I think we have to make all efforts to pick it up.
“We’re working in a very vulnerable patient population in cardiac surgery where complications are unfortunately still higher than in other patient fields just given the nature of it being very invasive and somewhat dangerous. It is obviously our goal to get every one of our patients safely beyond hospital discharge. I think this is where this really comes in.”
The full study results are expected to be submitted for publication later this year.
It’s valuable to detect poAF when it occurs, because it's obviously shown to increase the risk for stroke from embolic complications”
ECG Patch placement
Study points to potential markers for early post-TAVI valve deterioration
Early haemodynamic valve deterioration (HVD) occurs in around 6% of patients at one year after undergoing transcatheter aortic valve implantation (TAVI), with younger patients and those with small aortic annuli the most likely to be impacted.
These are among the findings of a single-centre analysis of more than 2,000 patients undergoing the TAVI procedure at the University of Pittsburgh (Pittsburgh, USA) between 2012–2022, details of which were published in The Annals of Thoracic Surgery
The study, which was conducted by Eishan Ashwat, a medical student at the University of Pittsburgh School of Medicine, evaluated 2,123 patients who had complete echocardiographic follow-up data out of a total population of 4,317 patients who underwent the procedure during the study period.
the aetiologic factors that precipitate HVD, with some studies citing residual aortic insufficiency in transcatheter bioprosthesis and other studies pointing to surgical valve design as a potential determinant,” they add.
The present study sought to compare post-TAVI outcomes in patients with and without the development of one-year HVD, while also identifying predictors of transcatheter valve deterioration.
Using the University of Pittsburgh institutional database, Ashwat et al assessed patients who underwent isolated TAVI and who had immediate post-procedural, 30-day and oneyear follow-ups available in order to compare the clinical and echocardiographic outcomes among HVD and non-HVD patients.
Of the 2,123 TAVI patients included in the study, 131 (6.2%) patients met echocardiographic criteria for one-year post-TAVI HVD and 1992 (93.8%) patients showing no evidence of HVD.
The study also showed that incidence of early HVD after TAVI was roughly 10% in the earlier years of the study period, with the adoption of both a higher procedural volume and newer generation devices, meaning that the yearly incidence of one-year HVD remained broadly consistent between 2015 and 2022, fluctuating between 4% and 7%.
Comparing baseline characteristics between the two groups, the researchers reported that they had broadly comparable preoperative profiles, though they found that TAVI patients with one-year HVD were younger (p=0.05) and more likely to have had a prior aortic valve replacement (p=<0.01) and stroke or transient ischaemic attack (p=0.02) compared with non-HVD patients.
Researchers used the updated Valve Academic Research Consortium-3 (VARC-3) definitions to assess bioprosthetic valve function and structural deterioration, defining HVD as an increase in mean transvalvular gradient >10mmHg resulting in a mean gradient >20mmHg or an increase of >1 grade of intraprosthetic aortic regurgitation (AR) resulting in moderate or greater AR.
“The advent of TAVI has revolutionised the treatment of aortic stenosis, expanding patient eligibility for aortic valve replacement,” Ashwat and colleagues write in their Annals paper. “Midterm outcomes from randomised trials studying TAVI have shown non-inferiority in comparing postoperative survival and reoperation rates with surgical aortic valve replacement. Nonetheless, as transcatheter heart valves are being implanted in increasingly younger patients with subsequently longer follow-up times, valve durability has become an important clinical point of discussion.”
HVD, they note—“as a harbinger of valve failure”— represents an important classification of the performance of a dysfunctional transcatheter valve.
Understanding of early HVD after TAVI remains limited in the context of updated VARC-3 criteria, they write, with studies claiming incidences ranging from 2–10%. “Moreover, there remains no clear consensus regarding
After TAVI, immediate postoperative outcomes were non-significantly different between HVD and non-HVD patients, including adverse neurologic events, permanent pacemaker placement, and major vascular complications, the study team reports.
On echocardiographic outcomes, Ashwat et al report that patients who experienced one-year post-TAVI HVD had higher aortic valve mean gradients at both follow-up time points postoperatively compared with nonHVD patients. No significant difference was observed in rates of immediate post-TAVI gradients >20mmHg between groups.
Differences in pre-TAVI versus immediate post-TAVI aortic valve mean gradients were comparable between HVD and non-HVD patients, whilst rates of moderate and severe aortic regurgitation were increased in HVD patients at one month and one year after TAVI, and, clinically significant paravalvular leak frequency was significantly higher in HVD vs. non-HVD patients at one month and one-year post-procedurally.
On multivariable logistic regression, the researchers found that larger implanted valve size (for each additional millimetre in diameter) was significantly associated with decreased odds of one-year HVD, whilst prior valve was associated with an increased hazard of one-year HVD.
The findings underscore the importance of vigilant long-term follow-up and may influence future decisionmaking regarding valve selection, particularly in patients with smaller aortic anatomy or a history of aortic valve replacement, the study’s authors have stated.
“As TAVI is increasingly used in younger and healthier patients, our findings suggest a need to carefully consider valve sizing and long-term durability,” said lead study author Ashwat.
“Early identification of haemodynamic deterioration may serve as a key predictor of future reintervention and patient outcomes.”
One-year results show positive safety and haemodynamic performance for biopolymer surgical mitral valve
FOLDAX HAS ANNOUNCED THE presentation of one-year results from the India clinical trial of the Tria mitral valve, showing a good safety profile, sustained haemodynamic performance, and statistically significant improvement in patient quality of life.
These are the first one-year outcomes presented for a multicentre clinical study of a polymer heart valve worldwide, the company says in a press release. The results were presented at New York Valves 2025 (25–27 June, New York, USA) and published in the Journal of the American College of Cardiology (JACC).
Key results from the trial showed no valve-related mortality or reinterventions, a >50% reduction in mean gradient (9.7mmHg to 4.5mmHg) and >90% increase in effective orifice area (0.9cm² to 1.5cm²), and a 24-point improvement in Kansas City Cardiomyopathy
Questionnaire (KCCQ) score (57.5 to 81.9) and 65% increase in six-minute walk test distance (298.1m to 494.8m), indicating improvement in quality of life.
The prospective, multicentre trial enrolled 67 patients aged 19 to 67 across eight sites in India, with an average age of 42 years. Notably, 64% were women, and of these, nearly half were of childbearing age, a significantly larger percentage than typically seen in studies. Seventy-three percent had rheumatic heart disease.
“It is exciting to see the positive clinical outcomes associated with this novel polymer valve,” said Isaac George (Columbia University, New York, USA). “This new type of valve sets out to reimagine how a device can address clinical needs that are underserved by current valve solutions.”
“I am proud to be the principal investigator in India for this groundbreaking trial and represent the highly esteemed clinical investigators who participated in the study,” said Kaushal Pandey (Hinduja Hospital, Mumbai, India), principal investigator of the TRIA mitral valve India clinical trial. “The Tria mitral valve provides hope for Indian patients— including younger patients and many women of childbearing age—for whom current valve options often fall short.”
“These results validate our strategy of taking heart valve therapy to a new level by bringing innovation to a platform of products that will help patients globally,” said Ken Charhut, chief executive officer of Foldax.
Aortic valve stenosis can be treated using surgical or transcatheter options
Graft occlusion after CABG seen more often in female patients
Early bypass graft occlusion occurred significantly more frequently in female compared to male patients after isolated coronary artery bypass graft (CABG) surgery, a singlecentre analysis published in The Annals of Thoracic Surgery has shown.
CONDUCTED BY LUCA KOECHLIN, ISLAM Slikhanov (both of University Hospital Basel, Basel, Switzerland) and colleagues, the analysis should stimulate research on the underlying pathophysiological mechanisms responsible for disparities in outcomes after CABG surgery, the authors say.
Recent data have demonstrated poorer outcomes in female patients following CABG, they note in their Annals paper, including more frequent graft failure at a median follow-up of one year, and a higher risk of major adverse cardiac and cerebrovascular events (MACCE) within a five-year follow-up.
Early bypass graft occlusion—a potential complication that may influence long-term outcomes after CABG—has been shown to be “substantially” underestimated when imaging is restricted to symptomatic patients, they write, whilst pilot studies evaluating bypass patency using computed tomography (CT) have shown higher occlusion rates in female patients.
All patients undergoing isolated CABG at University Hospital Basel have undergone routine CT scanning since April 2020, enabling the researchers to investigate the presence of a sex-specific difference in early bypass graft occlusion rates.
The study included a total of 589 patients who
underwent isolated CABG at the institution between April 2020 and January 2025. Patients had a mean age of 67 years, and were mostly male, with 95 female patients (16%) treated over the duration of the study. Female patients were older and more often had extracardiac arteriopathy and previous cerebrovascular events, the researchers detail, noting that the majority of the demographic data were comparable between the two sexes.
Female patients received fewer distal anastomoses compared to men (3 vs. 4), more often venous grafts and less often a second arterial graft, whilst the right internal mammary artery (RIMA) was used less often in female patients (15% vs. 25%).
Graft occlusion was detected in a total of 45 (7.6%) of the patients, the researchers report, observing

Surgical ablation during CABG carries survival advantage for patients with pre-existing AF
Medicare patients with atrial fibrillation (AF) who undergo surgical ablation during isolated coronary artery bypass grafting (CABG) live longer than those who do not, new research published in The Annals of Thoracic Surgery have shown.
RESEARCHERS EXAMINED
Medicare claims data from more than 87,000 patients with pre-existing AF who underwent CABG between 2008 and 2019. Overall, only 22% of studied patients received concomitant surgical ablation during CABG, and, although the prevalence of ablation did increase over the study period, ablation rates were only 27% in 2019—two years after a class I recommendation for ablation was published by the Society of Thoracic Surgeons (STS).
The study found that surgical ablation during CABG was associated with a risk-adjusted median survival advantage of 4.4 months compared to patients who did not receive ablation (7.82 vs. 7.46 years; p<0.001). Patients treated by surgeons who frequently performed ablation had a median
survival advantage of nearly five months (7.03 vs. 6.62 years; p<0.001) compared to those treated by surgeons who rarely performed the procedure.
“This study is one of several recent analyses suggesting that surgical ablation may meaningfully improve survival in patients with pre-existing atrial fibrillation undergoing CABG,” said Justin Schaffer, lead author of the study and medical director of surgical outcomes at Baylor Scott & White— The Heart Hospital (Plano, USA). “Our analysis found that the treatment effect of surgical ablation for AF manifested late, over two years after CABG. We hypothesise this is because ablation leads to a decreased incidence of tachycardia-related heart failure, which translates to improved late survival.”
To reduce bias related to the fact that
that this was seen significantly more often in female patients (13.7%) compared to males (6.5%). The majority of the occlusions—87%—were classed as being clinically silent, with six patients having exhibited symptoms of early in-hospital graft occlusion prompting urgent coronary angiography. Of these patients three underwent percutaneous coronary intervention (PCI) and three required redo CABG.
“The higher incidence of occlusion in female patients appears to be multifactorial and may include demographic differences, as women in our cohort undergoing CABG surgery were older and more frequently had extracardiac arteriopathy and previous cerebrovascular events,” Koechlin, Slikhanov et al document in their Annals paper. “Furthermore, women may have smaller coronary arteries, more complex coronary artery disease potentially reflected in a different SYNTAX score, and therefore undergo technically more challenging procedures; however, these factors remain speculative.”
In general, they write, the findings add to the evidence of sex-specific differences after CABG surgery, demonstrating worse outcomes for female patients. “While most studies highlighting these differences have been retrospective and thus only hypothesis-generating, future research should focus on the underlying pathophysiological mechanisms responsible for these disparities,” they add. “Whether the sex-specific differences in occlusion rates can be reduced by adapting operative strategies or postoperative treatment remains to be evaluated in future studies.”
The authors note several limitations to their research, notably that it is a single-centre study, the small sample size, and the lack of any available data concerning patients’ SYNTAX scores.
healthier patients are more likely to receive ablation, the researchers used two advanced statistical methods.
One method (overlap propensity score weighting) adjusted for measured differences between patients, while the other (a surgeon-preference instrumental variable analysis) compared outcomes among patients treated by surgeons who frequently ablate and those who rarely do.
This method simulates the effects of randomisation by leveraging variation in clinical practice among surgeons to balance unmeasured differences
Surgical ablation may meaningfully improve survival in patients with pre-existing atrial fibrillation undergoing CABG”
between patients. Under certain assumptions, patients can be thought of as being pseudo-randomly distributed among surgeons with different approaches to ablation.
“We used both as-treated and surgeon-preference analyses to provide a more complete picture of ablation’s potential benefits,” Schaffer explained.
“While each method has strengths and limitations, the consistency of findings across both approaches is encouraging, especially given that a large-scale randomised trial in this area does not appear forthcoming.”
Between 10% and 20% of patients undergoing CABG have AF before surgery, a condition that is associated with both perioperative complications and reduced long-term survival. Prior research has shown that surgical ablation can restore normal heart rhythm and improve long-term outcomes, but remains underutilised.
“These data underscore the importance of guideline adherence and hopefully will lead to a re-evaluation of surgical decision-making for patients with AF,” added study co-author John Squiers (Baylor Scott & White—The Heart Hospital, Plano, USA).
DIPTI ITCHHAPORIA
Serving as the president of the American College of Cardiology (ACC) during the height of the COVID-19 pandemic, Dipti Itchhaporia, an interventional cardiologist at the Hoag Heart and Vascular Institute (Newport, USA) who holds the Eric & Shelia Samson Endowed Chair in Cardiovascular Health, has seen firsthand the importance of digital transformation in cardiovascular care. She talks to Cardiovascular News about her research and life in medicine.
What drew you to medicine and to interventional cardiology in particular?
I have always had a strong interest in health and medicine, growing up in a family of physicians. As a child, I would often spend time at my grandmother’s house, where my uncle, a family practice doctor, ran his clinic. I would watch him work, fascinated by the interactions and the care he provided. Medicine seemed to offer the perfect blend of science, which I’ve always loved, and the ability to make tangible difference in people’s lives. That combination is what initially drew me to the field.
In high school and college, I was also heavily involved in journalism and served as the editor of the student newspaper. At one point, a friend encouraged me to pursue journalism, but the way I saw it, I could still write as a doctor, but I couldn’t practice medicine if I only wrote. Choosing medicine kept a world of options open.
Cardiology stood out for me because of its fast pace and the immediate impact you can make, particularly in interventional cardiology. I was especially drawn to treating patients with ST-elevation myocardial infarction (STEMI) where the improvement can be dramatic and immediate.
Beyond the urgency and intensity, what makes cardiology so meaningful is its broad impact—cardiovascular disease remains the leading cause of death globally. The field also offers deep intellectual and scientific challenges, and it is a privilege to earn the trust of patients during some of the most critical moments of their lives.
Who are the individuals that have helped to shape your career?
Many individuals have played a role in shaping my career and the way I practice medicine. From the beginning, my parents instilled in me the importance of hard work, compassion and dedication—values that continue to guide me every day. One of my earliest mentors outside of medicine was my high school journalism teacher. She helped me develop the communication and interpersonal skills that are so essential in healthcare.
During medical school, Michael McGuire, an orthopaedic surgeon who was a friend and mentor inspired me with his genuine love for medicine and for connecting with patients. I’ve been fortunate to train at institutions like Stanford and Georgetown, where I had the privilege of learning from many brilliant and dedicated clinicians. Each of them influenced me in different ways, making it difficult to single out just one person. Collectively, their mentorship, examples and support have shaped not only my clinical skills, but also the kind of physician I strive to be.
What has been the biggest change in the field over your career?
Transcatheter aortic valve implantation (TAVI) stands out as the most transformative advancement. The first TAVI was performed over 20 years ago by Alain Cribier—a good friend of mine. My only ACC presidential citation went to him for his profound impact on cardiovascular care. While we’ve seen remarkable progress in percutaneous therapies, increased adoption of minimally invasive procedures, and expanded use of imaging in interventional cardiology, no innovation has had a greater impact on patient outcomes across such a broad population as TAVI.
Have there been any disappointments?
My very first publication, over 20 years ago, focused on neural networks in cardiology. While we’re finally beginning to see innovations in artificial intelligence (AI) enter the field, the progress has been frustratingly slow. Even now, AI has yet to be integrated in a truly meaningful way. I hope that within my lifetime, we will move beyond simply discussing the potential of AI in cardiology and healthcare—and begin to speak confidently about its real-world applications.
What are your research priorities?
My primary focus is on digital health and innovation. During my presidential year, I spoke often about the promise of digital transformation in healthcare. While there’s growing interest in AI, data analytics, and home-based care, the true leap forward has yet to occur. Much is said about the digital divide, but I believe we need to rethink the entire model of healthcare delivery. I hope to see digital health become as seamlessly integrated into our lives as smartphones have.
Are there pitfalls to having more AI in healthcare?
Privacy is a major concern—especially when sensitive patient data is involved. Accountability is another complex issue: if an adverse event occurs following an AI-driven decision, who is ultimately responsible—the AI system, its developer, or the clinician who acted on the recommendation? We also need to be cautious about becoming over-reliant on AI; technology should augment, not replace, clinical judgment. Additionally, the risk of deepening the digital divide is real, potentially leaving certain populations behind. These challenges underscore the need for thoughtful regulation, equitable access, and robust data governance as we integrate AI into healthcare.
Which recent trials caught your eye? In 2025, two areas of cardiology have stood out for their clinical and scientific impact:
FACT FILE
APPOINTMENTS
Hoag Heart and Vascular Institute
Eric & Sheila Samson Endowed Chair in Cardiovascular Health University of California, Irvine Clinical Professor
American College of Cardiology President (2021–2022)
HealthTech Horizons: AI & Digital Innovation Summit Chair
EDUCATION & TRAINING
Saint Louis University School of Medicine, MD
Santa Clara Valley Medical Center, Internship, Internal Medicine
Stanford University Medical Center, Residency, Internal Medicine
Georgetown University Medical Center, Cardiology Fellowship
Stanford University Medical Center, Interventional Cardiology Fellowship
FELLOWSHIPS AND HONOURS
American College of Cardiology, Fellow
European Society of Cardiology, Fellow
British Cardiovascular Society, Honorary Member
TAVI and lipoprotein(a)—Lp(a)—targeted therapies. NOTION-2 broke new ground by enrolling low-risk patients under 75 years old—many with bicuspid aortic valves—into a head-to-head comparison of TAVI versus surgical aortic valve replacement (SAVR). At one year, composite outcomes were comparable between TAVI and SAVR, but nuances emerged: TAVI patients experienced higher rates of non-disabling stroke and paravalvular regurgitation, yet benefited from quicker recovery and improved quality of life. A meta-analysis of over 7,700 patients with long-term follow-up further reinforced TAVI’s safety and durability across surgical risk groups, supporting broader application in younger, lower-risk populations.
Meanwhile, the field of Lp(a)-lowering therapies has rapidly advanced, with multiple phase 3 trials underway. Agents such as lepodisiran—an siRNA therapy shown to reduce Lp(a) levels by more than 90%—hold promise for addressing a major unmet need in cardiovascular prevention. As Lp(a) becomes recognised as an independent risk factor for atherosclerotic disease, these trials may mark a turning point in personalised lipid management and long-term vascular risk reduction.
What will be the next evolution for the field of interventional cardiology?
I believe we’ll continue to see significant advances in minimally invasive techniques, with percutaneous interventions expanding beyond percutaneous coronary intervention (PCI) and even beyond cardiology. Robotics and automation, will play a larger role, potentially leading to more precise procedures, faster recovery times, and improved outcomes, AI and data-driven analytics will help streamline care and enhance safety, while augmented reality will offer better visualisation during interventions.
Another exciting frontier is phenotype detection. We perform extensive imaging, yet we only analyse a fraction of the data available. There’s enormous untapped potential to uncover connections and insights we’ve previously overlooked.
What truly excites me is the vision of creating lifelong health profiles—starting at birth—so we can continuously learn from individual data. This would allow us to predict and catch health issues early, shifting care from reactive to proactive. It’s a big dream, but one that can fundamentally change how we approach healthcare—moving away from sick care to early, personalised prevention.
What are the biggest challenges to improving health equity?
One major challenge is acknowledging that the healthcare we deliver only determines 20% of a person’s health outcomes—the remaining 80% is influenced by social determinants like income, education, housing, and access to healthy food. For cardiologists and healthcare clinicians who are already working incredibly hard, it can feel overwhelming to take on that broader 80%. But if we truly want to improve outcomes, we can’t ignore it.
We need to be strong advocates for policies that address these root causes. We also need greater transparency in the healthcare system, especially around costs, so that patients facing hardship can still receive care that aligns with their best interests. Without systemic change, equity will remain out of reach.

What is needed for interventional cardiology to attract the best talent?
Work-life balance has always been a concern, and it’s becoming more critical as women enter medicine. I recently read that for the first time, women will outnumber men in the UK National Health Service (NHS). If this continues and we want top-tier talent to enter interventional cardiology, we have to address structural challenges that disproportionately affect women and young physicians.
We are making progress in areas like radiation safety, and with advances in automation, robotics, and AI-enhanced imaging we have the potential to further reduce exposure, particularly compared to traditional angiographic procedures.
Just as important, we need to show young physicians that this work has real impact. It’s essential to inspire them—not just with the promise of a career, but with a sense of purpose. Mentorship plays a crucial role here. We must be intentional about fostering a culture that values contribution, growth, and meaning—not just financial rewards.
What is your most memorable case?
It’s hard to single out just one case. What stays with me most are the patients that we saved, it reminds me why the work matters. In a field that can be physically and emotionally demanding, those moments provide a sense of purpose. At tough times, I try to remember that any one of us could be a patient someday, and that helps me stay grounded in compassion.
What is the legacy from your ACC presidency?
I served as president during the pandemic, which shaped my tenure in unique and unprecedented ways. One challenge was finding new ways to connect with members in the absence of traditional travel and in-person meetings. I launched the Coffee with the ACC President series—a virtual initiative that created a meaningful avenue for engagement during a difficult time that has endured as a lasting platform. Even now I hear from members who found it valuable and impactful.
Maintaining optimism within the cardiovascular community was another
“I hope that within my lifetime we will move beyond simply discussing the potential of AI in cardiology and healthcare”
important focus. I emphasised continued education and championed digital transformation—recognising that as a technologically advanced specialty, cardiology must continue to evolve or risk falling behind. The pandemic also brought the issue of health equity into sharper focus. The ACC Health Equity Taskforce, which I headed in my vice presidential year, was embedded into the culture of the college and has started the conversation in a meaningful way.
What occupies you outside of medicine?
Life beyond medicine is deeply fulfilling. I make time to reflect on what else life has to offer beyond a professional career. Relationships with family and friends are important to me, and I feel fortunate that both of my parents are still with us. Travel remains a passion of mine and I continue to explore new places whenever I can. I also enjoy watching movies, they have a unique ability to transport you to different worlds and moments in time. Gran Torino is a particular favourite, I found it particularly inspirational.

Structural Heart Interventions
Regulatory and competition challenges hit TAVI market
Recent weeks have seen major developments for companies seeking to gain an edge in the burgeoning transcatheter aortic valve implantation (TAVI) market.
In May, Boston Scientific announced the surprise move to end sales of its Acurate TAVI systems—including the Acurate neo2 and Acurate Prime paltforms—following discussions with regulators.
“Boston Scientific is discontinuing worldwide sales of the Acurate neo2 and Acurate Prime aortic valve systems,” the company said in a statement. “While data continue to support the performance of the Acurate valve system when the product’s optimised instructions for use are followed, this decision was made based on recent discussions with regulators which resulted in increased clinical and regulatory requirements to maintain approvals in global markets and to obtain approval in new markets. Therefore, related commercial, clinical, research and development, and manufacturing activities will cease.”
Boston Scientific had been seeking US Food and Drug Administration (FDA) approval for the Acurate neo2 platform—which has been available and widely in use in Europe since 2020. The self-expanding, supra-annular Acurate neo2 system features an annular sealing technology designed to conform to irregular, calcified anatomies and minimise paravalvular leaking (PVL). The newer Acurate Prime platform was granted CE mark in August 2024.
A setback in the bid for FDA approval for neo2 was encountered with the release of results of the ACURATE investigational device exemption (IDE) trial in late 2024, in which the device failed to meet the prespecified 8% noninferiority margin across a combined endpoint of all-cause mortality, stroke or rehospitalisation at one year versus the Evolut (Medtronic) and Sapien 3 (Edwards Lifesciences) devices.
Observational data from implanters in Europe had appeared to validate the clinical performance of the neo2 valve, with an analysis presented at the 2025 Cardiovascular Research Technologies (CRT) meeting (8–11 March, Washington DC, USA), which emulated the conditions of the IDE trial, showing non-inferiority of Acurate neo2 against the competitor valves.
Investigators had been keen to establish an explanation for the valve’s underperformance in the IDE trial and valve under-expansion—which may have occurred in some instances due to
operator inexperience in handling the platform—had been identified as being a key contributor to the negative result.
A post-hoc review of cases from the IDE trial was presented at EuroPCR 2025 (20–23 May, Paris, France) and appeared to support this theory, showing that as many as 21.6% of patients in the trial had under-expanded valves which was associated with a higher one-year rate of death, stroke or rehospitalisation.
Attendees of EuroPCR also heard the first real-world results of the use of the newer Acurate Prime device, with findings of the Early ACURATE Prime registry presented by Andreas Rück (Karolinska University Hospital Stockholm, Stockholm, Sweden).
The results from the registry, which included data from 517 allcomers patients, demonstrated high rates of technical success and 30-day device success, as well as favourable echocardiographic results and a singledigit pacemaker rate.
“At Karolinska we have been among the largest users worldwide of the Acurate,” Rück commented to Cardiovascular News, in response to the news of the withdrawal of both devices from the market. “Clearly Acurate Prime can give excellent results, as we recently showed in the Early ACURATE Prime registry presented at EuroPCR. Also the previous Acurate neo2 generation has equivalent results as competitor valves in Swedish registry data with one year follow-up shown at CRT, and in other European datasets.
It is not just product design that matters”
“What this shows is that it not just the product design that matters, it is also how it is used and how the company communicates best practice, especially when trying to enter the US market. It just needs a little finesse. That is where the mistakes were made.”
Others have told Cardiovascular News that increased regulatory scrutiny on the device in Europe, and the challenging path ahead for approval in the USA, are likely to have ultimately

been the deciding factors in the decision to withdraw the device from the market.
Elsewhere, Edwards Lifesciences has faced its own setback in its bid to grow its TAVI stable, with the US Federal Trade Commission (FTC) has moved to block the proposed acquisition of JenaValve due to concerns that the acquisition would limit access to devices for the treatment of aortic regurgitation (AR).
JenaValve is the developer of the Trilogy TAVI system, a transfemoral TAVI system which carries a CE mark for the treatment of AR and aortic stenosis (AS), which is also seeking US FDA approval. The device is seen as meeting an unmet need for the treatment of AR, particularly among patients who are deemed to be at too high risk for surgery.
continue competing to innovate, expand treatment eligibility, and keep down costs. Americans deserve all the benefits that come from competition between medical device makers, just as they do in other markets.”

In July 2024 Edwards announced plans to acquire the company as one of several acquisitions that also saw it complete a deal for JC Medical, including the intellectual property and commercial rights to J-Valve, another TAVI system designed for the treatment of AR.
According to FTC the proposed US$945 million acquisition of JenaValve would combine the only two companies with ongoing clinical trials in the USA for a TAVI-AR device, threatening to reduce competition in the market and likely resulting in reduced innovation, diminished product quality, and potentially increased prices.
“Edwards’ attempt to buy the US market for TAVI-AR devices would eliminate the head-to-head competition that has spurred innovation for lifesaving artificial heart valves,” said Daniel Guarnera, director of the FTC’s Bureau of Competition. “The FTC is taking action to stop this anticompetitive deal and ensure that JenaValve and Edwards’ JC Medical subsidiary
The competition concerns caused by Edwards’ dual acquisition strategy are predicated on Edwards owning both JenaValve and JC Medical simultaneously, but Edwards has elected to attempt to buy JenaValve while retaining its ownership of JC Medical, the FTC statements adds. According to the complaint, Edwards has not been willing to engage on divesting JC Medical to resolve the competition concerns with the proposed JenaValve acquisition.
The federal court complaint and request for preliminary relief was filed in the US District Court for the District of Columbia to halt the transaction pending an administrative proceeding. In a statement issued following the FTC’s public challenge to the deal, Edwards strongly pushed back against the claims and maintained its intention to continue with the acquisition.
“Edwards disagrees with FTC’s decision and believes it will limit the availability of an important treatment option for patients suffering from AR,” the company stated. “The company further believes the acquisition of JenaValve will accelerate the availability, adoption and continued innovation of a life-saving treatment for patients suffering from AR.”
The company added that it intends to continue to pursue regulatory approval of the acquisition and estimates a final determination by the end of Q1 2026.
Acurate neo2 (top) and Trilogy (bottom)
AI model able to predict onset of valve regurgitation
An artificial intelligence (AI)-enhanced electrocardiography model may effectively predict the development and progression of moderate or severe mitral, tricuspid or aortic regurgitation—MR, TR, or AR—before the appearance of symptoms or physical changes that can be detected by ultrasound, research published in the European Heart Journal has shown.
THE STUDY WAS LED BY ARUNASHIS Sau and Fu Siong Ng (Imperial College London, London, UK) alongside researchers at Zhongshan Hospital (Shanghai, China), who trained AI models using nearly one million electrocardiogram (ECG) and transthoracic echocardiogram (TTE) records from over 400,000 patients in China. These were tested on a separate group of more than 34,000 patients from a secondary care dataset from Beth Israel Deaconess Medical Center (Boston, USA).
were up to 10 times more likely to develop these diseases than those at lower risk.
“AI has enormous potential for improving healthcare around the world, but it requires huge amounts of data to train and test these algorithms,” said Ng, the senior author. “Our work is an example of the benefits of international collaboration in this fast-growing area. By training the model in an almost exclusively Chinese population and then testing in a US cohort, we can show that our AI tool has the potential to be applied in various countries and settings around the world.”
Discussing the research in the European Heart Journal, the team writes that their study addresses a “notable gap in the management of regurgitant valvular heart diseases” adding that advances in AI could greatly facilitate screening of these conditions. AI is poised to streamline surveillance of patients with clinically insignificant regurgitant valve disease, they state, facilitating enhanced diagnostic capabilities and personalised treatment plans.
There are several limitations to the study, they add, including that the ECG and TTE data were retrospectively collected from the hospital-based cohorts, which may introduce some inherent selection bias, and the model for AR had reduced performance compared with those for MR and TR.

Using a deep learning approach known as a convolutional neural network (CNN) to analyse the data, the AI was trained to spot patterns in the ECG with signs of valve disease on the TTE. Classification models learned to identify if someone had moderate or severe valve disease at the time of the ECG, and survival models learned to predict if someone without significant disease at baseline may go on to develop it in the future.
The researchers showed that the AI could accurately predict who would go on to develop significant regurgitant disease, with the model able to correctly identify the risk (from high to low) in around 69–79% of cases.
“By the time symptoms and structural changes appear in the heart, it may be too late to do much about it,” said Sau. “Our work is harnessing AI to detect subtle changes at the earliest stage from a simple and common test, and we think this could be really transformative for doctors and patients.
“Rather than waiting for symptoms, or relying only on expensive and time-consuming imaging tests, we could use AI-enhanced ECGs to spot those most at risk earlier than ever before. This means that many more people could get the care they need before their hidden condition affects their quality of life or becomes life-threatening.”
People flagged as ‘high risk’ by the algorithm
“Artificial intelligence-enhanced electrocardiography can accurately diagnose prevalence and predict future development of significant MR, AR, and TR,” they conclude. “This approach may serve as the basis for the development of a regurgitant valvular heart disease prediction programme, to facilitate early detection, timely intervention, and potentially preventative therapeutics, while also optimising resource utilisation by tailoring the frequency of echocardiographic surveillance based on risk of progression.”
Rather than waiting for symptoms, or relying only on expensive and time-consuming imaging tests, we could use AI-enhanced ECGs to spot those most at risk earlier than ever before”
Germany sees “exponential” growth in tricuspid interventions
Transcatheter therapies for the treatment of tricuspid regurgitation (TR) have grown “exponentially” in Germany over the past decade, a research letter published in JACC: Cardiovascular Interventions has detailed, reflecting “very rapid” adoption of these techniques in clinical practice.
USING DATA FROM THE DRG-Statistic database of the German Federal Statistic Office, Jakob Christoph Voran and colleagues at University Hospital Schleswig-Holstein (Kiel, Germany) assessed trends in catheter-
based therapies for the treatment of TR, including transcatheter edge-to-edgerepair (TEER), from 2012 to 2023. They showed that cases increased from 74 in the four years from 2012 to 2015, to 4,298 in 2023—with 13,165
Sustained reduction in TR seen at two years with Triclip in real-world data
Two-year outcomes from the bRIGHT registry, a prospective multicentre registry detailing real-world results of tricuspid transcatheter edge-to-edge repair (TEER) using the Triclip (Abbott) system, demonstrate “sustained” reductions in tricuspid regurgitation (TR) with the device.
THESE WERE AMONG RESULTS SHARED during a late-breaking presentation at the 2025 EuroPCR meeting (20–23 May, Paris, France) by Raffi Bekeredjian (Robert-Bosch-Krankenhaus, Stuttgart, Germany).
Bekeredjian described the patients represented in the registry as being elderly, with an average age of 79 years, having multiple comorbidities and suffering from complex tricuspid pathology—with massive or torrential tricuspid regurgitation present in 88% of patients.
“The first question we tried to answer was whether the TR reduction was sustained throughout these two years. After 30 days we had a very significant reduction compared to baseline,” said Bekeredjian, reporting that 83% of the 511 patients treated within the study were observed to have moderate or less TR at the two-year timepoint.
At two years, all-cause mortality stood at 26.4%, with 14.5% deemed to be of cardiovascular causes. Major bleeding occurred in 12.7% of cases, with 4.5% due to device- or procedure-related causes.
The second question Bekeredjian and colleagues hoped to answer was whether there would be a significant improvement in heart failure symptoms after two years compared to baseline. “New York Heart Association (NYHA) functional class is significantly lower at two years compared to baseline,” he reported, with 20% of patients in NYHA class I, 49% in class II, and 29% in class III at two years.
The study team also looked at heart failure hospitalisation rates, comparing the year before the Triclip implantation to the first and second year after the implantation in order to see if the procedure would have any impact on hospitalisation rates due to heart failure, demonstrating that this was indeed significantly reduced at two years.
Gauging the impact of residual TR on two-year mortality, Bekeredjian and colleagues found no difference in two-year survival among patients with 30-day residual TR of moderate or less (24.4%), compared to a higher rate among those whose TR grade at 30 days was deemed to be severe or higher (41.7%).
cases with endovascular intervention for TR recorded over the entire period.
“This first comprehensive real-world data analysis of percutaneous tricuspid valve treatment in Germany showed an exponential increase in procedure numbers,” Voran and colleagues write. “This striking finding proves a very rapid adoption of this novel transcatheter concept.”
Researchers compared real-world data to findings from randomised trials including TRILUMINATE pivotal, TRI. FR and TRISCEND II, finding that patients were typically older outside of the trial setting, with a median age of 81
years and had a longer median length of hospital stay, at eight days, compared to a median of 1–3 days within the trials. In-hospital mortality declined from 4% in 2016 to 1.6% in 2023.
Though the researchers were unable to consistently compare real-world outcomes for each comorbidity with those reported in the trials, they did show that the presence of chronic kidney disease was reported more frequently than in TRILUMINATE, but was comparable with the rates reported in TRISCEND II, and diabetes was reported meaningfully more frequently than in TRILUMINATE and TRI.FR.
Study intensifies call for randomised trial in patients with bicuspid aortic stenosis
The results of a retrospective analysis should act as a “word of caution” against the liberal use of transcatheter aortic valve implantation (TAVI) in younger patients with bicuspid aortic valve anatomy.
THESE WERE THE COMMENTS OF THOMAS Cuisset (CHU Timone, Marseille, France), speaking at EuroPCR 2025 (20–23 May, Paris, France), where the results of the analysis which used propensity score matching to compare outcomes after TAVI and surgical aortic valve replacement (SAVR) among patients with severe aortic stenosis and bicuspid aortic valve anatomy were presented.
“We know that TAVI, as of today, is the standard of care for the majority of patients with severe aortic stenosis, but patients with bicuspid anatomy have been excluded from most of the recent TAVI trials, and mainly from low-risk trials,” commented Cuisset on the need for more data on the efficacy of TAVI in the subset of patients with bicuspid aortic valves during a press conference at EuroPCR.
To date, evidence has pointed towards differences in outcomes after TAVI amongst patients with bicuspid and tricuspid valves, Cuisset noted, highlighting results from the NOTION-2 trial, presented at the 2024 edition of EuroPCR, which suggested that in younger, low-risk patients, there were less favourable outcomes after TAVI where bicuspid aortic anatomy was present.
“As of today, we don’t have randomised trials comparing TAVI and surgery in this specific and

challenging population,” said Cuisset, “so we definitely need some evidence in this specific population.”
Cuisset and colleagues including Pierre Deharo (CHU La Timone, Marseille, France) used data from the TriNetX registry, capturing data from 16 countries between 2012–2024. Patients who had either surgery or TAVI for reasons other than isolated bicuspid aortic stenosis were excluded, leaving a total of 12,202 patients. Of these, 9,582 had undergone surgery and 2,620 TAVI. Investigators then propensity-matched patients leaving them with 1,195 in each cohort, with a
Improved survival seen following TAVI explant compared to redo procedures
Analysis of real-world US data comparing outcomes after posttranscatheter aortic valve implantation (TAVI) reinterventions points towards improved long-term survival and freedom from further valve reintervention among patients who have had their TAVI valve explanted surgically than compared to those who underwent a second—redo—TAVI procedure.
“THERE IS A PERCEPTION THAT TAVI explant is associated with really dismal outcomes, however I think that the data is really encouraging in terms of a relatively favourable long-term survival,” Shinichi Fukuhara, a cardiac surgeon at the University of Michigan (Ann Arbor, USA), who presented the data at New York Valves 2025 (25–27 June, New York, USA) commented.
Fukuhara’s analysis looked at Medicare fee-for-service beneficiaries who received TAVI between 2011–2024, excluding patients who required same-day reintervention during or after the index procedure. Researchers found a total of 4,443 patients with post-TAVI reinterventions during this timeframe, 2,553 undergoing redo TAVI and 1,890 TAVI explant.
Both procedures are increasing in prevalence, Fukuhara detailed, commenting that the clinical relevance of both interventions is “steeply rising”.
However, he noted, there is little understanding of the outcomes and trajectories of the two approaches.
Detailing baseline characteristics of the two groups, Fukuhara detailed that there were, unsurprisingly, differences among the two groups, with redo TAVI patients typically older, and with a higher prevalence of comorbidities including diabetes and kidney failure. The TAVI explant group had a significantly higher rate of endocarditis.
“Not surprisingly, 30-day mortality was significantly higher in the TAVI-explant group, reaching above 15%,” Fukuhara detailed, outlining the unadjusted survival rate after reintervention. “Despite this unfavourable short-term survival in the TAVI-explant group, the long-term survival was significantly better in the TAVI explant group, with the crossover point at 1.8 years.”
This result was consistent when the
mean follow-up of 1.9 years.
The key result reported by Cuisset was that the probability of mortality was twice as high in the TAVI patient population compared to patients treated with surgery, with a hazard ratio (HR) of 2.09 (p<0.0001). TAVI patients were almost twice as likely to experience heart failure (HR 1.96, p<0.0001). Other outcomes including pacemaker implantation and major bleeding were similar in the two groups, albeit skewing towards surgery, whilst rates of atrial fibrillation tended to favour TAVI.
“As of today, this is the largest retrospective cohort with the longest follow-up, and clearly it is a word of caution for the liberal use of TAVI in young patients with bicuspid aortic valves,” commented Cuisset. “It is not really our practice in Europe, but we know in the USA in patients less than 65 years old, more than 45% will be treated by TAVI today, and we know that a significant proportion of these patients will be patients with bicuspid anatomy.”
Cuisset said that the study raises two important points, the first being the need for careful patient selection and individualised decision-making in this anatomically and clinically complex setting, and added it is “a call for randomised controlled trials between TAVI and surgery in this population of severe aortic stenosis with bicuspid anatomy”.
Clearly it is a word of caution for the liberal use of TAVI in young patients with bicuspid aortic valves”
researchers adjusted for survival and excluded endocarditis cases.
When they adjusted the analysis to stratify outcomes by age at the time of the index procedure, they found that among those aged 65–70 years, TAVI explant demonstrated significantly improved survival after four years, with a crossover point between the two approaches at 1.3 years. Among older patients, those aged 70–80 or >80 years, this was no longer seen.
“Post-TAVI reinterventions remain very infrequent long-term,” Fukuhara said of the data, acknowledging that both are steadily increasing. “TAVI explant is associated with an improved long-term survival and freedom from second valve reintervention compared with redo TAVI,” he concluded.
“These are patients that have, for whatever reason, some sort of hostile situation towards the TAVI,” Michael Borger (Leipzig Heart Center, Leipzig, Germany), who was a discussant for Fukuhara’s presentation, commented of the findings. “I think it is not terribly surprising, if we add more hostility to an already hostile situation we are not going to have good long-term results.”
Fukuhara responded that the two approaches are not “mutually exclusive” and it is important to consider the individual characteristics of each patient. However, he said, many patients who have anatomy suitable for redo TAVI may tolerate TAVI explant well.
Speakers highlighted, however, that the surgical approach to removing valves can carry a steep learning curve, with Fukuhara commenting that it is the “modern cardiac surgeon’s duty” to improve the safety and outcomes associated with the procedure, noting that timing is also important.
“I always talk about this at the surgical meetings, the learning curve of TAVI explant is not only the technique, how to explant TAVI safely, but also timely intervention,” Fukuhara said.
“Patients who did not do well after TAVI explant are typically those who waited and waited, explored some other non-surgical options, and they end up with renal failure and heart failure— those are the patients who did not do well. Timely explant procedures I believe, will yield excellent outcomes.”
1.8YEARS
Thomas Cuisset
AF increases risk of death or hospitalisation for patients undergoing transcatheter mitral repair
Patients with heart failure and atrial fibrillation (AF) who are also undergoing mitral transcatheter edge-to-edge repair (M-TEER) for severe mitral regurgitation (MR) are more than twice as likely to die or be rehospitalised for heart failure (HF), compared to patients without AF.
THESE ARE THE FINDINGS OF a study published in the Journal of the American Heart Association (JAHA), which, according to the authors, suggest that AF may have to be treated more aggressively before patients undergo this type of transcatheter procedure in order to improve outcomes.
“We found that even when the transcatheter mitral valve procedure is successful, having atrial fibrillation at the day of the procedure doubles the risk of death or rehospitalisation for heart failure. AF is a powerful risk marker that we need to take into consideration when planning and performing these procedures,” says corresponding author Stamatios Lerakis (Icahn School of Medicine at Mount Sinai, New York, USA). “We know that mitral
regurgitation and AF are interconnected.
The one begets more of the other and vice versa. This study suggests that rhythm control ahead of time may be critical for the outcome of patients undergoing these procedures.”
AF is found in up to one-third of patients with mitral regurgitation.
The Mount Sinai researchers wanted to better understand how M-TEER impacts outcomes of patients who have AF.
They analysed 156 patients with symptomatic heart failure
and severe mitral regurgitation treated with M-TEER at Mount Sinai Fuster Heart Hospital. All these patients were evaluated by the heart team before undergoing M-TEER.
The researchers divided patients into two groups: those with AF at the time of the procedure (38%), and those with no AF at the time of the procedure (62%). After the procedure, researchers tracked patient survival, heart failure hospitalisations, and heart structure/function changes using echocardiography over a median followup of more than a year.
M-TEER was equally successful in both groups (AF and no AF). However, those with AF fared worse in the months following: they were more likely to experience a return of valve leakage and their hearts didn’t recover as well.
More specifically, within two years following the procedure, more than 52% of AF patients were either rehospitalised for heart failure or had died, compared to 33% of patients with no AF. Their hearts didn’t recover as well—they showed no improvement in heart size and additionally, severe valve leakage returned in 19% of AF patients versus 8% of patients without AF.

“Previous research has shown
Japanese registry data support TEER in atrial functional mitral regurgitation patients
Patients with atrial functional mitral regurgitation (AFMR) undergoing transcatheter edgeto-edge-repair (TEER) have significantly lower mortality and heart failure hospitalisation rates than those receiving medical therapy alone, research from Japan, published in the European Heart Journal, has shown.
THE STUDY LED BY TOMOHIRO KANEKO
and Nobuyuki Kagiyama (both Juntendo University Graduate School of Medicine, Tokyo, Japan) is described as a large-scale collaborative study involving multiple Japanese centres.
The study included a total of 1,081 patients with AFMR, with a mean age of 80.1±8.2 years, of whom 60.5% were women. Among them, 441 patients, selected from the OCEAN-Mitral registry, underwent TEER, while the remaining 640 patients, chosen from the REVEAL-AFMR registry, were the medically managed controls.
The research team used propensity score-based overlap weighting to balance the characteristics of both groups, enabling an accurate estimation of treatment outcomes. The association between TEER and clinical outcomes was evaluated based on mortality and heart failure-related hospitalisations in patients with AFMR.
hospitalisation was 44.3% in the medication group, compared to only 21% in the TEER group.
“Elderly patients with AFMR often fall into a therapeutic grey zone—too high-risk for surgery, yet poorly served by medical therapy alone,” says first author Kaneko. “TEER may help address this gap by providing a feasible, evidence-supported option that is associated with better outcomes in this vulnerable population.”

Patients who underwent TEER had a 35% lower risk of the primary endpoint—a composite of allcause mortality and hospitalisation due to heart failure (hazard ratio [HR]: 0.65 [0.43–0.99]). The risk of all-cause mortality alone, which was the secondary endpoint, was also 42% lower (HR: 0.58 [0.35–0.99]).
Survival benefit remained evident over time. Even after three years, the estimated incidence of death or
Outcomes were also determined by the degree of residual mitral regurgitation after TEER (residual AFMR). Among patients in whom residual AFMR was mild or less, the risk of death or hospitalisation was more than 50% lower compared to the medication group. However, in cases of moderate or greater residual AFMR, outcomes were no better or worse than those who received medical therapy alone. This suggests that the degree of success after surgery is a key determinant of the benefit of TEER, the researchers claim.
TEER success was notably high in the study’s Japanese cohort, with over 78% achieving mild or less residual AFMR at the time of discharge. Only 2.9% experienced some complications. Moreover, factors such as age, sex, and left atrial volume did not significantly affect the association between TEER and favourable outcomes. However, in cases of extreme
conflicting results on M-TEER and AF patients—and while this is a single-centre study from Mount Sinai Fuster Heart Hospital with a relatively small sample size, it still brings up an important problem,” says author Carlo Mannina, a cardiology fellow at Mount Sinai Morningside. “This definitely needs further investigation.”
“Our study emphasises the importance of atrial fibrillation as a major prognostic factor in patients with heart failure and severe mitral regurgitation, even after effective transcatheter edge-to-edge repair. These findings support early treatment of heart failure and mitral regurgitation before atrial fibrillation develops, in addition to considering ablation should atrial fibrillation occur,” says author Gregg W Stone (Icahn School of Medicine at Mount Sinai, New York, USA).
Rhythm control ahead of time may be critical for the outcome of patients undergoing these procedures”
left atrial enlargement (>100mL/m²), this association appeared to be attenuated or possibly absent, the researchers note.
“This was an observational study, not a randomised trial, so unmeasured confounding cannot be ruled out,” notes Kaneko. “Nevertheless, we employed multiple statistical methods to validate our findings, and the consistency across analyses strengthens our confidence in the conclusions.”
With Japan’s growing elderly population, mitral regurgitation could become more prevalent, the study’s authors suggest, noting that their research offers evidence that TEER, when performed successfully, may represent a promising and low-risk treatment option for patients with AFMR who have limited alternatives.
“In an ageing world where frailty limits surgical choices, TEER provides a much-needed bridge between risk and reward,” Kagiyama concluded. “Our findings support its use as a real and promising solution for this demographic.”
The research was co-authored by Masanori Yamamoto (Toyohashi Heart Center, Toyohashi, Japan) and Kentaro Hayashida (Keio University School of Medicine, Tokyo, Japan).
Elderly patients with AFMR often fall into a therapeutic grey zone—too high-risk for surgery, yet poorly served by medical therapy alone. TEER may help address this gap”
Stamatios Lerakis







ABSTRACTS

Accepted abstracts published in JACC Interventions
DEADLINE: November 21, 2025
CRT PRACTICE
INTERESTING CASES
CRMIC Publication Opportunity









DEADLINE: December 5, 2025
DEADLINE: Dec. 19, 2025
EARLY CAREER LEADERSHIP
FELLOWS WOMEN IN CARDIOLOGY
INNOVATORS
DEADLINE: January 16, 2026
NURSE & TECH INTERESTING CASES
Top 3 cases will be published in Cath Lab Digest

CRT EVIDENCE
















































LATE-BREAKING CLINICAL TRIALS & CLINICAL SCIENCE




Simulatenous Publication Opportunities
CRT INNOVATION
BEST INNOVATION COMPETITION
The Top Innovation will be the recipient of our CRT 2026 Innovations Award
DEADLINE: January 9, 2026
Associate Faculty
• Waived CRT 2026 registration fee
• Discounted Community Premium Access, includes all current CRTmeeting videos, PPT/PDF presentations, and archives
• Complimentary subscription to Cardiovascular Revascularization Medicine the official peer-reviewed journal of CRT
Submit Your Science Apply For Scholarships
New!
Prevail™
DCB has the broadest range of CE Mark indications, including bifurcation.†,1,2
New indications are supported by clinical evidence showing safety and efficacy in a real-world, complex patient population.†,1,3

De novo lesions
vessel disease
Bifurcation lesions New
Multivessel disease

7 new CE Mark indications
Learn more about Prevail™ DCB
™* Third-party brands are trademarks of their respective owners.
† Prevail DCB expanded indications are approved in EU and UK.
1. Medtronic data on file.
2. Information comes from key competitive DCB IFUs, or the respective websites accessed in May 2025.
Prevail™ paclitaxel-coated PTCA balloon catheter
Proven results.
Effortlessly delivered.
3. Von Koch, S. et al. Real-world Usage of Prevail paclitaxel-coated balloon compared with other contemporary drug-coated balloon | A two-year analysis from the Swedish Coronary Angiography and Angioplasty (SCAAR) Registry in over 6,000 patients. Presented at CRT 2025.
This material should not be considered the exclusive source of information, it does not replace or supersede information contained in the device manual(s). Please note that the intended use of a product may vary depending on geographical approvals.
See the device manual(s) for detailed information regarding the intended use, the implant procedure, indications, contraindications, warnings, precautions, and potential adverse events. For a MRI compatible device(s), consult the MRI information in the device manual(s) before performing a MRI. If a device is eligible for eIFU usage, instructions for use can be found at Medtronic’s website manuals.medtronic.com. Manuals can be viewed using a current version of any major internet browser. For best results, use Adobe Acrobat® Reader with the browser. Medtronic products placed on European markets bear the CE mark and the UKCA mark (if applicable). For any further information, contact your local Medtronic representative and/or consult Medtronic’s websites.
18556564-en-gb-emea © 2025 Medtronic. Medtronic, Medtronic logo, and Engineering the extraordinary are trademarks of Medtronic. All other brands are trademarks of a Medtronic company. For distribution only in markets where the Prevail paclitaxel-coated PTCA balloon catheter and the expanded indications have been approved. Not for distribution in the USA, France, Japan, or Canada.
Expanded indications for Prevail DCB broaden options for complex PCI, including bifurcations
Recent years have seen a surge of interest in the use of drug-coated balloons (DCBs) as an option to treat coronary artery disease. In clinical scenarios including in-stent restenosis (ISR) and de novo small vessel disease there is an increasing recognition that leaving nothing behind in the vessel is a favourable strategy to reduce stent burden by reducing the length and number of stents implanted.
As operators have become more proficient in using DCBs in their percutaneous coronary intervention (PCI) practice, there is a new impetus to determine whether these devices may offer an advantage in a broader set of clinical scenarios— including some of the most challenging lesion subsets.
In May, the PrevailTM paclitaxel-coated percutaneous transluminal coronary angioplasty (PTCA) balloon catheter (Medtronic) gained a series of new CE mark indications, making it the DCB with the widest range of indications in the market. Broadening its use to include scenarios such as multivessel disease, acute coronary syndrome, diabetes and, most significantly, bifurcation lesions, the approvals offer interventional cardiologists the option to use this device in more challenging clinical cases.
Discussing why it is important to have new treatment options in bifurcation lesions in particular, which remains a challenging subset, Goran Stankovic (University of Belgrade, Belgrade, Serbia), a founding member of the European Bifurcation Club (EBC)—a pan European group of interventional cardiologists who consider latest evidence on the treatment of coronary bifurcations to disseminate best practice— tells Cardiovascular News that bifurcations represent a significant proportion of the coronary artery disease cases seen in contemporary practice. Research has suggested that these comprise as many as 15–20% of PCI cases, with some papers putting this figure as high as 30%.1
“Historical data, as well as new evidence, suggest that this is one of the lesion subsets with the highest rate of periprocedural complications, as well as adverse long-term outcomes. The vessel divides at the bifurcation point and plaque usually continues in the lateral walls of both branches, so treatment of one branch compromises flow in the other branch,” Stankovic comments. “If it is not done properly, implanting a stent in the main vessel—which is usually the main goal of bifurcation PCI—shifts this part of the plaque, but also carina, which reduces the flow into the side branch.”
Over years of research and refinement, numerous techniques have emerged for the treatment of coronary bifurcations often involving the use of one or more drug-eluting stents (DESs), with factors including anatomical and lesion complexity, operator experience and preference determining the approach. A typical provisional stenting technique sees a single DES deployed in the vessel’s main branch, with a side branch treated using an additional stent only if necessary.
in the long-term because the longer the follow-up, the higher the event rate with two stents compared to a single-stent strategy,” says Stankovic. Henceforth, he explains, the emergence of DCBs in coronary interventions has opened the door to so-called “hybrid” treatment strategies, which involve using a combination of DES and DCB, or even “puristic”—DCB-only—strategies in bifurcation lesions.
4.4% all-cause mortality, 5.5% myocardial infarction and 5.8% target lesion revascularisation for Prevail DCB in the DCB-only arm (against rates of 4.7%, 5.2% and 5.6% respectively in the comparator arm).
Using Prevail DCB in a hybrid strategy resulted in a 3.2% rate of all-cause mortality compared to 3.9% in the comparator arm, 0.7% vs. 1.8% target lesion revascularisation, and 0.4% vs. 3.7% myocardial infarction.
“In general, I think Prevail DCB is one of the best, if not the best available DCB on the market at the moment,” comments Scheller on the performance of the device. “Prevail DCB has excellent deliverability, the coating method is state of the art, the drug loss during implant is reduced, the drug transfer rate is very good and the dose is also appropriate,” he comments, noting that randomised study of the device in the setting of bifurcation lesions will be necessary before it is possible to draw firm conclusions from the SCAAR results.


Bruno Scheller (University of Saarland, Homburg/Saar, Germany), a pioneer in the development and use of DCBs in coronary interventions tells Cardiovascular News that there are different ways to employ either the hybrid or puristic approach in the treatment of bifurcation lesions. “Number one is that the DCB is the tool for reducing late lumen loss and restenosis of the side branch and treating the main branch with a stent,” he explains. The operator has the option to treat the main branch with a stent and then cross the side branch, or alternatively to use the DCB first in the side branch, followed by stent implantation in the main branch.
The second approach is the puristic option, which Scheller also categorises in two ways. “Number one is to treat the main and the side branch with the DCB in a sequential way. If you already have disease in the origin of the side branch and the side branch is not very big then you can treat only the main branch and you can expect that the origin of the side branch will get better over time,” he explains.
At the recent EuroPCR meeting (20–23 May, Paris, France) new data were presented from a subgroup analysis of the Swedish Coronary Angiography and Angioplasty Registry (SCAAR) comparing the use of PrevailTM DCB to other contemporary DCB platforms in the setting of bifurcations lesions. These data, totalling 1,479 realworld PCI procedures performed in Sweden between August 2021 and 2024, including 462 using Prevail DCB and 1,017 using other DCB platforms, represent the largest analysis to date comparing different DCBs in bifurcation PCI including DCBonly and hybrid strategies.

“Treatment with two stents involves the main vessel and the side branch. Although we are seduced by the beauty of the procedural results you achieve by putting stents in both branches, there is a payoff
The study demonstrated low mortality (3.7% for Prevail DCB vs. 4.2% for other DCB platforms) and target lesion revascularisation rates (2.8% for Prevail DCB vs. 3.4% for other DCB platforms), with Prevail DCB also performing favourably in terms of new myocardial infarction (2.3% vs. 3.7%), and target lesion definite thrombosis (0% vs. 4.2%) at one year, with event rates consistently low across the different bifurcation strategies. Comparing the performance of Prevail DCB as part of a DCB-only or hybrid strategy, the data also appear favourable. Investigators report an incidence of
Stankovic comments that the SCAAR data are encouraging but agrees that randomised study will be vital. “We have large datasets, coming from one of the best systems for follow-up in the world, showing that there are at least the signals for a lower rate of myocardial infarction in the group treated with Prevail DCB compared to other available systems, and on the other side you have good safety data with an extremely low rate of reported clinical events,” he comments.
“Of course, we need to confirm that in a randomised fashion, but at least now we have approval for use of specific devices in coronary bifurcations, which really makes the thought process much simpler.”
With a desire for more evidence on the performance of DCB-PCI in the setting of bifurcation lesions, investigators are beginning enrolment in the EBCDCB trial, a 750-patient randomised trial of stepwise provisional stenting versus DCB therapy for non-left main coronary bifurcations. Follow-up will take place at one, three, five, and eight years, with a bifurcationorientated composite endpoint (BOCE) comprising cardiovascular death, target bifurcation-related MI and target bifurcation revascularisation.
“We have the feeling that this puristic DCB approach improves outcomes, but we do not know this for sure, therefore we have to do the trial,” comments Scheller. “If this trial is positive, this would change our complete understanding of how to treat bifurcations, because the DCB approach is much easier and faster to do. If both were equal it is clear I would select the simpler approach over the more complex procedure.”
References
1. von Birgelen C, Zocca P, Buiten RA, et al. Thin composite wire strut, durable polymer-coated (Resolute Onyx) versus ultrathin cobalt-chromium strut, bioresorbable polymer-coated (Orsiro) drug-eluting stents in all-comers with coronary artery disease (BIONYX): an international, single-blind, randomised non-inferiority trial. Lancet. 2018 Oct 6;392(10154):1235-45.
Prevail DCB has excellent deliverability, the coating method is state of the art, the drug loss during implant is reduced, the drug transfer rate is very good and the dose is also appropriate”
Bruno Scheller
Goran Stankovic
Evoque tricuspid valve replacement system gains Health Canada approval
Edwards Lifesciences’ Evoque tricuspid valve replacement system has received Health Canada’s approval for tricuspid regurgitation (TR) treatment.
Evoque is indicated for the improvement of health status in patients with symptomatic severe TR despite optimal medical therapy, for whom tricuspid valve replacement is deemed appropriate by a heart team.
The Evoque system is comprised of a nitinol self-expanding frame, intraannular sealing skirt and tissue leaflets made from bovine pericardial tissue. The Evoque valve will be available in four sizes, all delivered through the same low-profile transfemoral 28Fr system.
“Patients suffering from severe tricuspid regurgitation endure debilitating symptoms and poor quality of life and are desperate for effective treatment. The Evoque system is able to fully replace the tricuspid valve, eliminating tricuspid regurgitation in a wide range of anatomies,” said Neil Fam (St Michael’s Hospital, Toronto, Canada). “The significant improvements in patients’ quality-oflife are remarkable, with Evoque now offering a therapy to many patients who previously had no treatment options.”
Results from the randomised controlled pivotal trial, TRISCEND II, were presented at TCT 2024 (27–30 October, Washington, DC, USA), demonstrating the Evoque system’s superiority compared to medical therapy alone for the one-year primary endpoint. Key findings in the trial included significant reduction or elimination of tricuspid regurgitation and significant improvements in symptoms, function and quality-of-life at one year, with favourable numerical outcomes in mortality and heart failure hospitalisation.
The Evoque system received CE mark approval in October 2023, making it the world’s first transcatheter valve replacement therapy to receive regulatory approval to treat TR, and earned US Food and Drug Administration (FDA) approval in February 2024.
Prevail DCB gains expanded CE mark indications Medtronic has received CE mark approval for several expanded indications for the treatment of coronary artery disease (CAD) with the Prevail paclitaxel-coated percutaneous
transluminal coronary angioplasty (PTCA) balloon catheter, or drugcoated balloon (DCB).
The Prevail DCB now has the broadest range of CE mark indications including bifurcation and is the only DCB indicated for treatment in patients with multivessel disease, acute coronary syndrome, and diabetes, the company states in a press release.
The CE mark approval follows the release of data from a large, real-world, complex patient population from the Swedish Coronary Angiography and Angioplasty Registry (SCAAR) presented as late-breaking clinical research at Cardiovascular Research Technologies (CRT) 2025 (8–11 March, Washington DC, USA). There, it was reported that the Prevail DCB demonstrated low event rates at two years including 6.1% new myocardial infarction, 7.4% target lesion revascularisation, and 0.8% target lesion definite thrombosis. In hybrid percutaneous coronary intervention (defined as DCB and drug-eluting stent in the same vessel), the study showed the Prevail DCB demonstrated numerically lower new myocardial infarction rates versus other DCBs.
These results were confirmed for bifurcation patients in a subgroup analysis presented at EuroPCR (20–23 May, Paris, France). In bifurcation lesions, the Prevail demonstrated low mortality and revascularisation rates, with 2.3% new myocardial infarction, 2.8% target lesion revascularisation, and 0% target lesion definite thrombosis at one year. The event rates were consistently low across the different bifurcation strategies.
Total artificial heart system gains entry to US FDA advisory programme
Bivacor has announced that its total artificial heart (TAH) system has been accepted into the US Food and Drug Administration’s (FDA) total product life cycle (TPLC) advisory programme—known as the TAP programme.
The programme is designed to accelerate the development and patient access to high-impact medical technologies. Entry into the programme provides Bivacor with proactive, strategic engagement with the FDA throughout the entire product life cycle, from development to commercialisation, supporting more efficient, risk-informed decisionmaking, the company says in a press release.
“Acceptance into the TAP programme marks a major milestone not just for Bivacor, but for the field of mechanical circulatory support as a whole,” said Daniel Timms, founder and chief technology officer of Bivacor.
“The Bivacor TAH has the potential to fundamentally redefine the standard of care for patients with end-stage heart
failure. TAP access gives us a powerful framework for working hand-in-hand with the FDA to bring this technology to the patients who need it most.”
The Bivacor TAH is intended for use as a bridge to transplant in adults with severe, irreversible biventricular or univentricular heart failure, particularly for patients who cannot be treated with traditional left ventricular assist devices (LVADs). The system employs a magnetically levitated centrifugal pump, inspired by space and industrial technologies, which provides continuous, pulsatile, and physiologically responsive cardiac support.
The FDA’s acceptance letter to Bivacor emphasised that the TAP programme’s inclusion reflects the agency’s confidence in the technology’s potential to transform clinical practice. As part of the programme, Bivacor will receive more regulatory guidance, earlier identification of scientific and evidentiary gaps, and greater coordination among stakeholders, including payers and patient advocacy groups.
ShortCut leaflet splitting device granted add-on payment designation by CMS
Pi-Cardia has announced that the Centers for Medicare & Medicaid Services (CMS) has approved a New Technology Add-On Payment (NTAP) for the ShortCut leaflet splitting device, effective 1 October 2025.
The NTAP designation recognises ShortCut as a breakthrough technology that addresses a critical unmet need in valve-in-valve transcatheter aortic valve implantation (TAVI) by enabling treatment for patients at risk of coronary obstruction.
CMS will provide eligible hospitals with up to US$9,750 in additional reimbursement per case when the ShortCut procedure is performed. This payment is in addition to the Medicare Severity Diagnosis Related Group (MS-DRG) amount for TAVI. To receive the NTAP, hospitals must report ICD-10-PCS code X28F3VA, which specifically identifies leaflet splitting procedures performed with ShortCut.
“We are thrilled to get CMS NTAP for the ShortCut device,” said Erez Golan, Pi-Cardia’s chief executive officer. “We started our US limited commercial launch just a few months ago at leading TAVI centres and already see significant utilisation and enthusiasm with the device. The NTAP will now allow us to expand our commercial footprint, enabling more hospitals to adopt ShortCut with additional reimbursement support—and
ultimately helping more patients at risk of coronary obstruction gain access to this important technology.”
As bioprosthetic valves degenerate over time, many of these patients will eventually require a valve-in-valve procedure. A significant portion of them who are at risk for coronary obstruction may benefit from leaflet splitting with ShortCut.
The NTAP programme is intended to facilitate Medicare beneficiary access to new technologies that offer substantial clinical improvement over existing technologies. It provides hospitals with supplemental payments to help offset adoption costs while enabling CMS to collect cost data that will inform future inpatient payment rates.
US FDA gives approval to commercialise AI software to guide transcatheter procedures Caranx Medical has announced US Food and Drug Administration (FDA) clearance for the commercialisation of its artificial intelligence (AI) software for real-time intra-operative guidance of transcatheter valve implantation.
The FDA clearance marks a significant milestone in Caranx’s strategy towards planned market introduction of TAVIPILOT Soft by the end of 2025, the company says in a press release.
“Obtaining FDA clearance for TAVIPILOT Soft is a major achievement for the Caranx team. This clearance confirms the potential of realtime guidance in TAVI [transcatheter aortic valve implantation] procedures to enhance precision and improve patient outcomes. We are excited to now move forward with early commercialisation by end 2025 and to provide interventional teams with a solution that can support safe and predictable valve deployment,” said Jorgen Hansen, chief executive officer of Caranx.
“Allowing a broader number of cardiologists and interventional cardiology centres to perform complex procedures, such as TAVI, with the Caranx AI-driven software and autonomous robot is at the heart of our mission of entrepreneurs and business builders to save lives and revolutionise medicine,” said Philippe Pouletty, founder of Caranx and chief executive officer of Truffle Capital.
“We are assessing how to make Caranx’s future even more promising and ambitious, with potential collaborations with other cardiovascular-focused companies, in the context of an ambitious project named CarvOlympics.”
TAVIPILOT Soft is an AI-driven intra-operative software which tracks real-time anatomical and instrument landmarks. It enables precise and accurate heart valve positioning and delivery, Caranx says in a press release. TAVIPILOT Soft is compatible with all cardiac imaging systems and will be compatible with all TAVI heart valves on the market.
Pi-Cardia
Evoque
CE mark approval for Biosensors’ Imperia THV delivery system
Biosensors International has received CE mark certification for the Imperia delivery system under the EU Medical Device Regulation (MDR).
The Imperia delivery system is part of the Allegra transcatheter heart valve (THV) system and is intended for the transfemoral implantation of the already CE-marked Allegra THV, indicated for the treatment of severe aortic stenosis.
The new delivery system emphasises ease of use while maintaining maximum safety and procedural predictability, a press release from Biosensors International states. The system enables recapturability, repositioning, and retrievability of the Allegra valve, facilitating precise valve implantation. This contributes to optimised haemodynamic performance post-implantation, the company press release adds.
The Allegra THV system with the Imperia delivery system was investigated in the EMPIRE study. Thirty-day results, presented at PCR London Valves 2024 (24–26 November, London, UK), demonstrated high procedural success, user-friendliness, and excellent haemodynamic performance of the valve, the company’s press release states.
José Antonio Baz Alonso (Hospital Álvaro Cunqueiro, Vigo, Spain), principal investigator of the EMPIRE study, commented: “During the EMPIRE study, we were impressed by the seamless performance of the new delivery system across a wide range of anatomies and clinical indications. Importantly, the study reported no cases of moderate or severe patientprosthesis mismatch (PPM) at 30 days in the native cohort. This outcome highlights a valuable option for treating patients with small to medium annulus sizes, supporting the reduction of PPM and potentially improving long-term mortality outcomes.”
The Imperia delivery system is now commercially available across Europe with immediate effect.
US patent issued for algorithm to predict renal denervation response
Geneticure has announced that the US Patent and Trademark Office has granted a patent for a geneticbased weighted algorithm model for predicting response to renal denervation.
This patent protects Geneticure’s proprietary method of using a patient’s genetic profile to predict their likelihood of benefiting from renal denervation—a minimally invasive procedure used to treat hypertension.
“Renal denervation has demonstrated the ability to reduce blood pressure in certain patients, with other’s experiencing no change or an increase in blood pressure post-denervation,” said Scott Snyder, chief executive officer of Geneticure. “Prior to Geneticure’s research, a reliable
method of predicting the response had not yet emerged and regardless of the denervation method, the changes in blood pressure have been variable across patient populations. Our technology has proven to identify the individuals most likely to respond, enabling more targeted use of this emerging therapy with increased prospects for adoption and payer coverage.”
The patented method leverages specific genetic markers combined with a proprietary weighted algorithm to determine responsiveness to renal denervation, enabling physicians to make informed treatment decisions beyond traditional risk-factor assessments.
This newly issued patent expands Geneticure’s growing intellectual property portfolio in the field of pharmacogenomics and device response prediction, further positioning the company at the forefront of personalised hypertension management, a press release states.
Prevencio granted patents to protect proprietary coronary artery disease blood test platform Prevencio has been granted patents for its HART CADhs test in the USA—the second US patent for this novel test— as well as the EU and Japan.
The patents protect Prevencio’s proprietary methods and algorithms for detecting obstructive coronary artery disease (CAD) using a multi-protein, artificial intelligence (AI)-driven blood test platform.
The newly issued patents strengthen Prevencio’s intellectual property portfolio and support its long-term strategy to expand HART CADhs adoption globally, the company says in a press release. HART CADhs is a blood test that combines multiple cardiac proteins with machine learning to deliver a highly accurate, noninvasive diagnosis of obstructive CAD.
“Securing patent protection in the USA, Europe, and Japan marks a major step forward in advancing our innovation and market leadership,” said Rhonda Rhyne, chief executive officer of Prevencio. “These patents reinforce the uniqueness of our HART CADhs test and underscore our commitment to improving cardiovascular diagnostics worldwide.”
“Cardiovascular disease is a global problem. International patent protection will help Prevencio to deliver scalable solutions that meet urgent diagnostic needs in the USA as well as global markets,” said James L Januzzi (Harvard Medical School, Boston, USA), chief scientific officer at the Baim Institute for Clinical Research and principal investigator for the HART test development and validation.
Prevencio’s HART blood tests are designed to improve accessibility, accuracy, and patient outcomes while reducing costs through earlier, more accessible, and accurate blood diagnostics, the company says in a press release.
Both HART CADhs and HART CVE, a second AI-driven, multi-protein blood test that assesses a patient’s one-year risk of heart attack, stroke, or cardiovascular death, are currently available to healthcare providers and for research use.
Vivasure Medical submits premarket approval for PerQseal Elite vascular closure device Vivasure Medical has announced the submission of a premarket approval (PMA) application to the US Food and Drug Administration (FDA) for its PerQseal Elite vascular closure system for arterial procedures.

The submission builds upon the successful results of the PATCH study as well as positive clinical use in Europe, reinforcing the system’s potential safety and performance profile.
In addition, the company received European CE mark approval for an expanded indication for PerQseal Elite covering large-bore venous closure. This follows its first CE mark approval in April 2025 for arterial procedures and positions PerQseal Elite as the first fully bioresorbable, sutureless solution in Europe for both arterial and venous access closure.
“With the increasing adoption of minimally invasive therapies in structural heart and electrophysiology procedures, managing large-bore access sites remains a critical consideration,” said Azeem Latib (Montefiore Health System, New York, USA). “PerQseal Elite was designed to address the growing need with a novel, fully bioabsorbable approach and we look forward to further progress in the programme.”
The PerQseal Elite vascular closure system is designed for fully absorbable, sutureless closure following percutaneous cardiovascular procedures. Currently, there are no fully bioresorbable devices available on the market for closure following largebore procedures.
Medtronic TAVI platforms gain CE mark for expanded redo TAVI indication
Medtronic has announced it has received CE mark for the expanded redo transcatheter aortic valve implantation (TAVI) indication of the Evolut PRO+ and FX TAVI systems. This approval allows for the implantation of a new Evolut transcatheter aortic valve (TAV) inside any failed previously implanted TAV.
The redo TAVI procedure is indicated for the failure of any TAV, regardless of the manufacturer, amongst patients
with severe aortic stenosis and for those at high-risk for open-heart surgery.
“The CE mark approval for the Evolut TAVI system’s redo TAVI procedure is great news for physicians working in this field, but most importantly for patients with failing transcatheter heart valves, who now have a crucial new treatment option,” said Dan Blackman (Leeds Teaching Hospitals NHS Trust, Leeds, UK).
“This minimally invasive procedure not only offers an alternative for patients at high risk for surgery but also underscores the commitment to improving outcomes and extending the benefits of TAVI therapy.”
“Redo TAVI indication builds upon the Evolut platform’s proven valve performance and durability to offer safe and reliable lifetime management options, providing physicians exceptional patient outcomes today, while maintaining important options for the future,” said Jorie Soskin, vice president and general manager, Structural Heart, part of the Cardiovascular Portfolio at Medtronic.
In native aortic valves, Evolut TAVI systems are currently indicated for implantation in symptomatic severe aortic stenosis patients across all risk categories (extreme, high, intermediate, and low) in the European Union and the USA.
Tria polymer mitral valve gains Indian CDSCO approval
Foldax has announced that the Indian Central Drugs Standard Control Organization (CDSCO) has approved its Tria mitral valve. Dolphin Life Science India LLP will locally manufacture the Tria mitral valve in India.
Traditional valves made from animal tissue are prone to calcification and degradation, limiting their durability and often leading to repeat surgeries, especially for younger patients.
Mechanical valves, while durable, are prone to thrombosis and require lifelong anticoagulation therapy that can impact a patient’s long-term quality of life.
Foldax’s vision for its novel polymer heart valves is to address the limitations of tissue and mechanical valves by making them durable, with the goal of avoiding the requirement for lifelong anticoagulation.
The company’s portfolio of transcatheter and surgical valves in development, including robotic implantation capabilities, is based on its proprietary LifePolymer, a polymer material developed specifically for heart valve use and engineered at a molecular level to be calcium-resistant, biostable, and biocompatible.
Clinical research on the Tria mitral valve demonstrates favourable safety, stable haemodynamics, and meaningful improvements in patient quality of life, Foldax says in a press release.
The Tria mitral valve is for investigational use only and is not available for commercial sale in the USA.
PerQseal
Clinical News
First patients enrolled in MAGICAL-SV study of sirolimus DCB in small coronary vessels Concept Medical has announced the enrolment of the first patient in the MAGICAL-SV trial, a US investigational device exemption (IDE) study evaluating its MagicTouch sirolimus-coated balloon (SCB) for the treatment of small coronary vessels, following US Food and Drug Administration (FDA) IDE approval.
Samin Sharma enrolled the first patient at New York’s Mount Sinai Hospital, marking a crucial step in advancing treatment options for patients with small-vessel coronary artery disease, the company said in a press release.

“Today marks a great feat for us and for the MAGICAL-SV trial,” said Sharma. “My team at Mount Sinai is privileged to be the first to begin this study. This is a proud moment for all of us, especially knowing what the trial signifies and aims to achieve by evaluating MagicTouch as a potential alternative for treating small vessels in the USA.”
Concept Medical is now actively enrolling in three IDE trials concurrently in the USA, across coronary and peripheral interventions, underscoring its commitment to bringing innovative therapies to patients worldwide.
MAGICAL-SV is a prospective, multicentre, single-blind randomised trial (2:1 randomisation) comparing Concept Medical’s MagicTouch sirolimus-coated balloon to everolimus or zotarolimus drug-eluting stents (DESs) for the treatment of small coronary artery lesions.
Randomisation is stratified by study site and presence of medicallytreated diabetes mellitus status. The trial will enrol 1,605 patients across the USA and Europe, to evaluate the primary endpoint of target lesion failure (TLF) at 12 months and aims to support premarket approval (PMA) for MagicTouch SCB in small vessels.
Study chair Martin B Leon (Columbia University, New York, USA), with principal investigators Azeem Latib (Montefiore Health Systems, New York, USA), Ajay Kirtane (Columbia University Irving Medical Center/NewYork-Presbyterian Hospital, New York, USA), and Antonio Colombo (San Raffaele Hospital, Milan, Italy) will lead the trial.
“The initiation of the MAGICALSV trial marks an important step in evaluating sirolimus-coated balloon therapy for small coronary vessels,” commented Leon. “By addressing key limitations of stenting in this complex anatomy, the study aims to generate strong comparative data versus DES. The results could inform future treatment strategies in the USA and Europe, offering clinicians a potential alternative for managing small-vessel disease.”
Preliminary data “encouraging” for biorestorative coronary bypass conduit
Xeltis has announced preliminary data for Xabg, its coronary artery bypass graft (CABG) conduit, demonstrating positive safety and patency among patients with multi-vessel atherosclerotic coronary artery disease.
Xabg is a novel, biorestorative, polymeric conduit which enables continued blood flow in coronary artery disease patients.
Developed using Xeltis’ proprietary Endogenous Tissue Restoration (ETR) platform, Xabg has regenerative properties, enabling living tissue to replace it over time, the company says in a press release.
A clinical trial is underway at various sites in the EU with preliminary data demonstrating Xabg to be functioning well with excellent flow in patients. This underscores the potential of Xabg to eliminate the need for vein harvesting and transform the bypass surgery landscape with the goal of longer-term durability, the company adds. With no approved off-the-shelf, small-diameter vascular grafts with good patency rates available, Xabg has the potential to address this large unmet clinical need of suboptimal conduits in bypass surgery.
In an earlier trial, Xabg demonstrated patency after bypass surgery in a patient with excellent flow in a 24-month follow-up, marking the first time any artificial bypass surgery conduit has remained open and functional in humans for this duration.
Isaac George, surgical director at Columbia University Medical Center (New York, USA), said: “Xeltis is pushing the boundaries of innovation in vascular access solutions. The recent 24-month follow-up data is remarkable, the first of its kind to remain open at 24 months and demonstrate excellent flow. This, combined with the safety and patency results in Europe, underscores the potential of Xabg to improve long-term outcomes, reduce the need for reinterventions, and significantly impact patient care and healthcare costs.”
The single-arm, human feasibility study in the EU will evaluate the preliminary safety and performance of the Xabg technology in patients with multivessel atherosclerotic coronary
artery disease who are scheduled to undergo elective CABG surgery.
AtriCure completes enrolment in trial assessing stroke prevention with AtriClip device
AtriCure has announced the completion of enrolment in the LeAAPS clinical trial—a prospective, randomised, blinded, superiority investigational device exemption (IDE) trial evaluating the AtriClip left atrial appendage (LAA) exclusion system for the prevention of ischaemic stroke and systemic arterial embolism in cardiac surgery patients without a history of atrial fibrillation (AF).
The LeAAPS trial was initiated in January 2023, and has enrolled 6,500 patients at 137 centres across the globe.
“For decades, the medical community has sought to better understand the role of the LAA in stroke following cardiac surgery,” said Richard Whitlock (McMaster University, Hamilton, Canada), global principal investigator for the trial.
“LeAAPS aims to deliver definitive evidence to guide optimal care for high-risk patients without AF who may benefit from LAA exclusion.”
An AtriCure press release notes that cardiac surgery patients with no history of AF constitute “a large and often underserved population at elevated risk for these events”. It is estimated that more than one million cardiac surgery procedures occur annually and more than 70% of these patients have no history of AF before surgery, the release adds.
According to AtriCure, results from the LeAAPS trial are expected to inform clinical practice and treatment guidelines for stroke prevention using AtriClip platform technology in

patients undergoing planned cardiac surgery. The trial will continue with five years of follow-up to assess long-term outcomes. LeAAPS is being conducted in collaboration with the Population Health Research Institute (PHRI), which is affiliated with McMaster University.
Trial comparing TAVI and SAVR in aortic regurgitation enrols first patients
JenaValve has announced that patient enrolment has begun in the ARTIST study, a randomised controlled trial evaluating the safety and effectiveness of the Trilogy transcatheter heart valve system compared to surgical aortic valve replacement (SAVR) in patients with aortic regurgitation (AR).
The study is now enrolling patients in both transcatheter and surgical arms
with the first case being performed in Los Angeles, USA, at Cedars-Sinai Medical Center.
ARTIST is designed to provide evidence for the treatment of moderateto-severe and severe AR in patients that are not at high-risk for surgical aortic valve replacement and is the first ever randomised study comparing SAVR versus transcatheter aortic valve implantation (TAVI) in AR patients.
“It’s incredibly rewarding to have enrolled and treated the first randomised patient in the ARTIST trial,” said Raj Makkar (Cedars-Sinai Medical Center, Los Angeles, USA).
“In a single day, we performed multiple Trilogy cases, including as mentioned our first ARTIST case as well as implanting Trilogy in a patient with left ventricular assist device (LVAD)related AR disease who was enrolled into the JENAVAD Registry.
“These experiences highlight just how much demand exists for less invasive solutions in this complex and undertreated population; it’s a glimpse into how the Trilogy valve could reshape care for patients with AR.”
“ARTIST is a landmark study designed to answer a critical question of how to best treat patients with severe AR,” said Torsten Vahl, principal investigator of the ARTIST trial and interventional cardiologist at Columbia University Irving Medical Center (New York, USA). “Building on ALIGN-AR, which established TAVI as a solution for high-risk AR patients, ARTIST has the potential to expand TAVI to a broader population of severe AR patients. It is also the first prospective head-to-head evaluation of TAVI versus surgery ever conducted in this patient population.
“The results will provide critical data to guide future treatment decisions in the structural heart community.”
“The launch and enrolment of ARTIST marks a defining moment for both JenaValve and the structural heart field,” said John Kilcoyne, chief executive officer of JenaValve. “ARTIST reflects our commitment to generating the highest level of clinical evidence as we work to bring a less invasive solution to a larger group of patients suffering from this challenging disease.”
Image-guided crossing and reentry system used for first time in coronary CTO trial Simpson Interventions has announced that its Acolyte image-guided crossing and re-entry catheter system has been successfully used to treat the first patients in the Acolyte study, a pivotal investigational device exemption (IDE) trial, assessing the system's safety and effectiveness in treating coronary chronic total occlusions (CTOs) in patients with persistent symtpoms following medical therapy.
The technology is designed to overcome current challenges in treating CTOs and is designated by the US Food and Drug Administration (FDA) as a breakthrough device.
MagicTouch
AtriClip
CroíValve secures funding from Horizon Europe EIC Accelerator
CroíValve, developer of the Duo transcatheter system for the treatment of tricuspid regurgitation (TR), has been awarded funding from Horizon Europe’s European Innovation Council (EIC) Accelerator Programme.
Part of the EU’s Horizon Europe 2021–2027 Research and Innovation programme, the EIC Accelerator Programme provides funding to highpotential, high-risk start-ups, scale-ups and subject matter experts.
This award includes grant funding of €2.5 million, combined with an equity investment of €10 million in CroiValve’s next financing. The latest competition included 959 applications, with only 40 awards across 16 countries. Almost one third of the selected companies are led by a woman and CroiValve is incredibly proud to be one of this group, the company said in a press release.
“Securing European Innovation Council (EIC) funding is instrumental in expanding clinical validation of the Duo system, a novel minimally invasive, transcatheter device to treat severe tricuspid regurgitation. There is a significant unmet clinical need to treat the heterogeneous patient population who are not suitable for first generation transcatheter tricuspid devices. It is CroiValve’s mission to innovate a better way to help more patients suffering with this disease and we are honoured to have EIC support for this journey,” said Lucy O’Keeffe, chief executive officer of CroíValve.
Duo is a novel transcatheter heart valve that preserves the patient’s native anatomy while treating TR. It works in tandem with the native tricuspid valve to restore valve function while leaving the patient’s right heart and native
Conference calendar
17–19 September
Biennial Heart Valve Biology & Tissue Engineering Meeting London, UK hvbte.com
8–11 October
European Association for Cardio-Thoracic Surgery (EACTS) annual meeting Copenhagen, Denmark eacts.org/39th-annual-meeting
valve apparatus untouched through an innovative anchoring mechanism.
The device is currently under clinical investigation through the TANDEM II study, a multicentre, prospective early feasibility study including centres in Poland and the USA.
Teleflex completes acquisition of Biotronik’s Vascular Intervention business
Teleflex has completed the previously announced acquisition of substantially all of the Vascular Intervention business of Biotronik. The company notes in a press release that the acquisition adds a broad portfolio of therapeutic products to Teleflex’s portfolio of interventional access products.
On 27 February 2025, Teleflex announced its entry into a definitive agreement to acquire all of Biotronik’s Vascular Intervention business for a cash payment of €760 million, less certain adjustments as provided in the purchase agreement, including certain working capital not transferring and other customary adjustments.
Papyrus covered coronary stent for acute coronary artery perforations, and the Orsiro Mission drug-eluting stent (DES), an ultrathin DES with differentiated clinical features.
Teleflex shares that the acquisition will also allow the company the opportunity to invest in and expand the clinical trial programme for Freesolve, a sirolimus-eluting resorbable metallic scaffold (RMS) technology, including plans to initiate a US pivotal study.
Ceryx Medical closes latest funding round
Ceryx Medical has announced the successful close of a new funding round, bringing the company’s total raised to US$15 million.
The company’s lead product—a temporary cardiac pacing device— is currently undergoing clinical evaluation.
Early results from the ongoing study are “highly encouraging”, the company says in a press release, showing the potential to significantly enhance patient recovery following cardiac surgery.

“We are pleased to announce the completion of the acquisition of substantially all of the Vascular Intervention business of Biotronik earlier than expected,” said Liam Kelly, chairman, president and chief executive officer of Teleflex. “The acquisition will significantly enhance our global presence in the cath lab, expand our suite of innovative technologies, and improve patient care. We believe the acquisition will allow us to position this advanced coronary portfolio alongside our existing Interventional business and establish our global footprint in the fast-growing peripheral intervention market.”
The acquired Vascular Intervention business consists of a portfolio for coronary and peripheral interventions performed in the cath lab and interventional radiology suites. In coronary vascular interventions, Teleflex details that key products include the Pantera Lux drug-coated balloon catheter, the novel PK
25–28 October Transcatheter Cardiovascular Therapeutics (TCT) San Francisco, USA tctconference.com
11–14 November Cardiovascular Interventions La Jolla, USA cvinterventions.com
This new funding will accelerate development of Ceryx’s second product: a permanent, physiologically responsive pacemaker designed to provide longterm support for patients with chronic heart failure.
Unlike conventional pacemakers, Ceryx’s technology mimics the body’s natural modulation of the heart to deliver real-time, biomimetic pacing, offering a more natural and effective treatment option.
“This investment is a strong endorsement of our team, technology, and vision,” said Stuart Plant, chief executive officer of Ceryx Medical.
“The early clinical data from our temporary device has given us real momentum, and with this funding, we’re excited to advance our permanent pacing platform—an innovation that could redefine how heart failure is treated.”
The round was supported by investors including BGF, Parkwalk Advisors, Development Bank of Wales (DBW), and BBI. Ceryx Medical was spun out of research from the Universities of Bristol, Bath, and Auckland.
16–18 November PCR London Valves London, UK pcronline.com/courses
21–22 November European Society of Cardiology (ESC) Digital & Artificial Intelligence (AI) Summit Berlin, Germany escardio.org
Merit Medical names Martha Aronson as new president and CEO
Merit Medical has announced the appointment of Martha Aronson as the company’s new president and chief executive officer (CEO), effective 3 October 2025.
Fred P Lampropoulos will remain as chairman of the board, president and CEO of Merit through 3 October. Upon Aronson’s appointment, Lampropoulos will continue to serve as chairman of the board.
“I am pleased to welcome Martha to Merit,” said Lampropoulos. “I believe the company has never been in a stronger position. Martha is uniquely qualified to lead Merit into the future and drive its continued success. Her keen intellect and proven track record of empowering global teams through collaborative excellence, consistent with ‘The Merit Way,’ makes her the ideal leader for Merit’s next stage of growth.”
“I am excited to join Merit and truly honoured to take on this role,” said Aronson. “Fred has built and grown an extraordinary company over the past four decades, starting with one single idea and building it into the incredibly successful global company that it is today. Working with the talented team at Merit, I look forward to building upon his remarkable legacy and leading the company into a future of continued excellence and innovation.”
Heartflow raises more than US$364 million through initial public offering
Heartflow, a developer of artificial intelligence (AI) tools for the diagnosis of coronary artery disease, has been publicly listed on the Nasdaq stock exchange, raising more than US$364.2 million through an initial public offering (IPO).
The company offered 19,166,667 shares of its common stock at a public offering price of US$19 per share, which began trading on the Nasdaq Global Select Market on 8 August under the ticker symbol HTFL.
The Heartflow One system is a non-invasive, precision coronary care platform, that is intended to provide patient insights throughout the guideline-directed coronary computed tomography angiography (CCTA) pathway.
2026
29 January–1 February Society of Thoracic Surgeons (STS) annual meeting New Orleans, USA sts.org
5–6 February
Controversies & Advances in the Treatment of Cardiovascular Disease Beverly Hills, USA promedicacme.com/controversies
5–7 March
The Houston Aortic Symposium: Frontiers in Cardiovascular Disease Houston, USA promedicacme.com/houston-aortic
8–11 March
Cardiovascular Research Technologies (CRT) Washington, DC, USA crtmeeting.org
Duo tricuspid valve system
Biotronik



