


m a g a z i n e p o s t e r i o r s e g m e n t • i n n o v a t i o n • e n li g h t e n m e n t THE WORLD’S FIRST FUNKY OPHTHALMOLOGY MAGAZINE THE WORLD’S FIRST FUNKY OPHTHALMOLOGY MAGAZINE THE OPHTHALMOLOGY-OPTOMETRY CROSSOVER ISSUE June/July 2022 piemagazine.org 22












2 18 November 2022 | Issue #1 SHOW DAILY by DIGITAL MARKETING + ADVERTISING + VIDEO PRODUCTION + MEDICAL WRITING + EVENTS Request our 2023 Media Kit Now! Write enquiry@mediamice.com for a copy HQ Office: 6001 Beach Road, #09-09 Golden Mile Tower, Singapore 199589 Phone: +65 8186 7677 Satellite Office: 2 Nuoc Man 2 Street, Da Nang City, Vietnam 50506 Phone: +84 868 063 773 E-mail: enquiry@mediamice.com Web: www.mediamice.com






Matt Young CEO & Publisher Hannah Nguyen COO & CFO Robert Anderson Media Director Gloria D.
Chief Editor Brooke Herron Editor International Business Development
Mahajan Ranga Brandon Winkeler Writers
Joanna
Nick
Graphic
Media MICE Pte. Ltd. 6001 Beach Road, #19-06 Golden Mile Tower, Singapore 199589 Tel: +65 8186 7677 / +1 302 261 5379 Email: enquiry@mediamice.com www.mediaMICE.com Published by IN THIS ISSUE... We are looking for eye docs who can contribute articles to PIE magazine. Interested? Let's talk! Send us an email at editor@mediamice.com. To place an advertisement, advertorial, symposium highlight, video, email blast, or other promotion in PIE magazine, contact sales@mediamice.com. Posterior Segment Innovation Enlightenment
Aging
Innovations in Retinal Imaging Benefit Glaucoma Management Night of the Living Neurons:
researchers have revived light-sensing tissue in organ donor eyes Complex Vitreoretinal Surgery Cases: Highlights from Retinawesome 14, Part 2 08 16 10 18 20 32 Cover Story m a g a z i n e p o s e rio s e g m ent nnov a io n enl ghtenm ent 22 Breaking the Glass Ceiling: The Prof. Ava
Story The Effect of the
Pandemic
Babies and Other Updates in ROP The Stars and Stalwarts of Ophthalmology, Honored at AIOC 2022 24 26 Pearls from
Studies:
Gamat
Ruchi
April Ingram Hazlin Hassan
Lee Matt Herman
Eustice Tan Sher Lynn Maricel Salvador
Designer
The Gut-Eye Link to
Seeing Colors in the Dark What Lurks in the (Quantal) Shadows?
How
Hossain
COVID-19
on
Surgical Retina Case
Highlights from AOS Virtual 2022
Dr. Alay S. Banker is the director of Banker’s Retina Clinic and Laser Centre in Ahmedabad, India and his practice has served the city since 2007. He started off his career as a clinical instructor and fellow at the Department of Diseases of Retina and Vitreous, Uvea and Inflammation of Eye at University of California, San Diego, USA. He was the first Indian to receive the “International Scholar Award” from AAO in 2010 and also the youngest Indian to receive the Achievement Award by the AAO in 2006. His contributions toward his medical peers and community services have also earned him the Senior Achievement Award from AAO (2013) and the Dr. Piyush Patel Award for Service to Society and Mankind from Ahmedabad Medical Association (2013). He is the senior founding editor of the Retina Image Bank (ASRS 2012), has presented at more than 250 international and national conferences with over 40 papers published in peer-reviewed medical journals, and has published five book chapters in international book publications.


alay.banker@gmail.com
Prof. Gemmy Cheung is currently a professor at Duke-NUS Medical School, National University of Singapore, and head of the Medical Retina Department at Singapore National Eye Center (SNEC). Her research focuses on retinal diseases, including age-related macular degeneration (AMD), polypoidal choroidal vasculopathy (PCV) and myopic macular degeneration, as well as risk factors for these conditions that may be unique to Asian populations. Prof. Cheung has more than 200 peer-reviewed publications and serves on the editorial boards of several journals, including the American Journal of Ophthalmology, Retina and Eye.

gemmy.cheung.c.m@singhealth.com.sg
Dr. Hudson Nakamura is an ophthalmologist specializing in the retina and vitreous. He completed his medical degree from School of Medicine at the Federal University of Goiás, UFG and residency from the Base Hospital of the Federal District, Brasília, DF. Presently, Dr. Nakamura is a member of the AAO, Brazilian Council of Ophthalmology, Canadian Society of Ophthalmology and ARVO. He currently works as a professor in the Department of Retina and Vitreous Course of Medical Residency in Ophthalmology at the Bank of Goias Eye Foundation. Dr. Nakamura holds a vitreoretinal disease fellowship from the University of Toronto Canada and the Brazilian Center for Eye Surgery.
hudson.nakamura@gmail.com
Dr. Kenneth Fong is recognized as an ophthalmologist in the United Kingdom, Australia and Malaysia. He graduated with a medical degree from the University of Cambridge in 1998 and trained to be an eye surgeon in London. Dr. Fong then spent two more years training in the U.K. and at the Royal Perth Hospital in Australia to subspecialize in retina. After 18 years of working in the U.K. and Australia, he returned to Malaysia in 2009 to serve as associate professor, consultant ophthalmologist and retinal surgeon at the University of Malaya in Kuala Lumpur. He is currently the managing director of OasisEye Specialists in Kuala Lumpur. Dr. Fong is the president of the Malaysian Society of Ophthalmology and serves as a council member for the APVRS.
kcsfong@gmail.com

| June/July 2022 4
ADVISORY BOARD MEMBERS
Dr. Alay S. Banker
Dr. Kenneth Fong
Prof. Gemmy Cheung
Dr. Hudson Nakamura
Prof. Mark Gillies presently holds a number of positions including: director of research and director of the Macula Research Group for the Save Sight Institute; foundation fellow for the Sydney Medical School; professor in the Department of Clinical Ophthalmology at the University of Sydney; head of the Medical Retina Unit at the Sydney Eye Hospital; deputy chair for the Ophthalmic Research Institute of Australia; and director of Eye Associates in Sydney. Prof. Gillies has served as a principal investigator or associate investigator in more than 70 clinical trials, and his research regarding macular degeneration and drug safety and efficacy has been published in 188 journals. He has also received a number of grants to study treatments for age-related macular degeneration, retinal disease and Muller cell dysfunction – among other treatments and studies. Prof. Gillies is a dedicated and multi-awarded researcher.

mark.gillies@sydney.edu.au


Dr. Saad Waheeb is an associate professor of ophthalmology and a senior academic consultant. He is the ophthalmologist-in-chief at King Faisal Specialist Hospital & Research Centre in Riyadh, Saudi Arabia. He is also the CEO and founder of First Lens Eye Center. Dr. Waheeb’s specialty is on the diseases and surgery of the retina and vitreous with special interest in diabetic retinopathy and retinal vascular disorders. He has published in the field of retina and has been an invited speaker both nationally and internationally. He is a founding member of the Saudi Retina Society. He completed his residency and retinal fellowship in vitreoretinal diseases and surgery at the University of Toronto, Canada.







saadwaheeb@hotmail.com


SOCIETY FRIENDS


| June/July 2022 5
Prof. Mark Gillies
Dr. Saad Waheeb
Asia-Pacific Vitreo-retina Society ASEAN Ophthalmology Society
Arunodaya Charitable Trust (ACT)
Asia-Pacific Academy of Ophthalmology
He Eye Specialist Hospital
Ophthalmology Innovation Summit
Retinawesome Retina & Vitreous International
Orbis Singapore Subthreshold Ophthalmic Laser Society
Vitreo-Retinal Society - India
Young Ophthalmologists Society of India ( YOSI )
World Ophthalmology Congress
Russian Ophthalmology Society (ROS)
We Can Be Better Together
Dear Readers,
t Media MICE, and the publisher behind PIE, we believe in working together. Not only that — we also tend to believe that certain things are better together … including eyeball-related things. In past issues, we’ve covered the opportunities and benefits (as well as the challenges) of bringing the front and back of the eye together from a surgical perspective. But this still leaves one important component out — that of the optometrist.
So two years ago, we launched COOKIE, the world’s first “funky” optometry magazine, to complement PIE and CAKE (our anterior segment magazine). Not only did this result in further making us look like some sort of baked goods company — it also rounded out our coverage to include all aspects of the eye care journey, from general eye exams to retinal detachment repairs.
And an eye exam and retinal detachment might not seem to have too much in common — unless of course, the eye exam shows high myopia … which can lead to retinal detachment. Indeed, when it comes to this tiny and intricate organ, it’s all related. Therefore, in this issue of PIE, we wanted to look at the relationship between retinal specialists and optometrists to learn how these two can work together — and thus, be better together.
It all began innocently enough … but after a few interviews, we discovered that we had opened a proverbial can of worms. We quickly learned that there are “tensions” on both sides — and for good reason. In reality, the relationship between the two is not always rosy and aggravations were aired on both sides.
However, we also learned that these challenges are not insurmountable
Aand there are opportunities for the two specialties to work together to improve care and outcomes for patients. Indeed, there are those who have formed long-standing relationships and work together quite well — and this relationship has shown benefits on both sides, and for the patient too. Further, in a world where health care resources are already stretched thin, it might be time to drop any residual reservations for the betterment of everyone — doctors and patients both.

And fortunately, it’s not all doom and gloom and there are pathways for better collaboration and integrative care between ophthalmologists and optometrists. In this issue’s cover story, we take a look at the good, the bad and the ugly in the ophthalmologist-optometrist working relationship and discuss strategies on how the two can be better together. We would like to thank the doctors who shared their candid perspectives and personal experience on this topic — including their personal insight into how we, as an eye care community, can improve. Both specialities are, after all, working toward the same goal: preserving vision and saving sight.
To further the “better together” concept, this year Media MICE will host the CAKE & PIE Expo 2.0 in Da Nang, Vietnam. This one-of-a-kind eye care event will bring together all three eyeball specialties, including anterior and posterior segment ophthalmologists and optometrists, under one roof. It’s our hope that this event will provide an opportunity for the eye care industry to come together — and be better together.
Cheers, Brooke Herron Editor, PIE, CAKE & COOKIE magazines

| June/July 2022 6 LETTER TO READERS

The Gut-Eye Link to Aging
 by Tan Sher Lynn
by Tan Sher Lynn
Anew study on mice provides evidence about the direct involvement of gut microbes in aging and the functional decline of the eye, offering a potential solution in the form of gut microbe replacement therapy.

Aging affects the entire body. But the eye can be especially vulnerable to it as eye tissues are highly sensitive to metabolic dysregulation and the integrity of the gastrointestinal (GI) epithelial barrier. As one ages, this fragile organ may experience decline in the form of reduced visual acuity and peripheral vision, weakened eye muscles, corneal changes, lens hardening, and macular degeneration, among others.
Moreover, as an extension of the central nervous system, the eye is exposed to multiple stressors, such as oxidative stress-inducing UV light and age-related immune dysregulation,


as well as being susceptible to energy insufficiency resulting from altered circulation and metabolism in old age.1 Also, with advancing age, the outer retina of the eye exhibits widespread deposition of extracellular debris and elevated inflammation, which is involved in the pathogenesis of agerelated macular degeneration (AMD) like choroidal neovascularization and geographic atrophy.2
Thankfully, a recent study offers a glimmer of hope in addressing agerelated ocular decline by demonstrating that transplantation of fecal microbiota from young into old mice can reverse hallmarks of aging not only in the gut, but the eyes and brain as well.3
In the study, researchers exchanged the intestinal microbiota of young (3 months), old (18 months), and aged (24 months) mice. They then developed a custom analysis workflow to analyze the changes in gut microbiota composition
and metabolic potential using whole metagenomic shotgun sequencing and metabolomics. The effects of age and microbiota transfer on the gut barrier, retina and brain were assessed using protein assays, immunohistology and behavioral testing.
Reducing retinal inflammation
The elevated expression of inflammatory complement protein C3 in the retina of aging mice is associated with progressive extracellular deposits between the retinal pigment epithelium (RPE) and Bruch’s membrane (BM), which contribute to retinal degeneration.4,5 Results of the study showed that that specific proteins associated with retinal degeneration were elevated in the young mice receiving microbiota from old donors. On the other hand, old mice that received gut microbiota from young
| June/July 2022 8 VISION RESEARCH POSTERIOR SEGMENT
mice had these detrimental changes in the retina reversed.
“Chronic inflammation is associated with driving or perpetuating pathology in multiple diseases of the eye. In our recent mouse study, we found that gut microbes can regulate levels of inflammatory proteins and immune signaling molecules in the retina. In aged mice, transfer with young donor microbiota reduced retinal inflammation. This suggests that manipulation of the gut microbiota may provide a new avenue by which we may eventually be able to maintain good retinal health for longer in old age,” said lead author Aimee Parker.
Preserving photoreceptors
Photoreceptors of the retina are especially sensitive to fluctuations in systemic inflammation and metabolism in old age. Retinal pigment epithelial protein RPE65, which is critical for regeneration of retinal visual pigment in photoreceptors, was depleted in aged mice compared with young mice.
The study showed that in young mice, baseline levels of RPE65 were reduced post-transfer with an aged donor microbiota; while the transfer of a young donor microbiota into aged mice restored RPE65 expression to levels seen in young mice.
“Excitingly, we also found that transfer of young donor microbiota to aged mice increased the levels of an important functional protein (RPE65) which is normally depleted with age. This protein is involved in the turnover of visual pigment, as part of a set of chemical reactions termed the visual cycle. Our results therefore suggest that microbial modulation may prove
useful not only in ameliorating ageassociated retinal inflammation, but may also be of benefit in maintaining good vision by regulating normal photoreceptor functioning,” said Parker.
Reducing proinflammatory cytokines
In addition, the researchers assessed the cytokine levels in retinal lysates in aged mice receiving fecal microbiota transplant (FMT) through the use of a proteome profiling array. They discovered that multiple proinflammatory cytokines elevated in aged mice were reduced after
affecting the retina, but these changes can be reversed by replacement with young donor microbiota. Nevertheless, further work is required to assess the long-term beneficial effects, as well as whether FMT can benefit aged individuals.
“At the moment, we don’t know how long these beneficial effects last for, and it is still too early to say if the same results can be achieved in humans. However, we do know that the human gut microbiota also changes with age, and that changes in gut microbes are seen in people with degenerative eye conditions, so it is possible that microbial modulation could be beneficial in these cases. We are currently working hard to identify the specific microbes or molecules which may be responsible for the positive changes we saw in the mice, and which could be used therapeutically in people. Ideally, these would be delivered as a drug or dietary supplement, thus avoiding the need to do fecal transfers. It will also be important to design proper clinical trials to test any candidate therapeutics arising from further animal studies,” Parker noted.

transplantation of young microbiota including CCL11 (eotaxin), CXCL11, and IL-1ß. On the other hand, IL-13 which is a putative neuroprotective cytokine, showed the highest increase following FMT from young donors into aged mice.
What does all this mean?
These findings demonstrate that age-associated changes in murine intestinal microbiota contribute to disrupted gut barrier integrity and systemic and tissue inflammation
References:
1. Lin JB, Tsubota K, Apte RS. A glimpse at the aging eye. Npj Aging Mech Dis. 2016;2(1):1–7.
2. Tan W, Zou J, Yoshida S, et al. The Role of Inflammation in Age-Related Macular Degeneration. Int J Biol Sci. 2020; 16(15): 2989–3001.
3. Parker A, Romano S, Ansorge R, et al. Fecal microbiota transfer between young and aged mice reverses hallmarks of the aging gut, eye, and brain. Microbiome. 2022; 10: 68.
4. Balaratnasingam C, Yannuzzi LA, Curcio CA, et al. Associations between retinal pigment epithelium and drusen volume changes during the lifecycle of large drusenoid pigment epithelial detachments. Investig Ophthalmol Vis Sci. 2016;57(13):5479–5489.
5. Rutar M, Valter K, Natoli R, Provis JM. “Synthesis and propagation of complement C3 by microglia/ monocytes in the aging retina.” edited by Erica Lucy fletcher. PLoS One. 2014;9(4):e93343.
| June/July 2022 9
What Lurks in the (Quantal) Shadows?
by Nick Eustice
We need light to see. Simple enough, right?
But just how much light do we need to see clearly? Well, that probably depends on what we’re doing. We might need more light when we’re cooking in the kitchen than we do when we’re reading a book before bed, for example. If we’re taking a late night stroll, the moon is often enough, and once our eyes have adjusted, we find we can detect even subtle, creepy movements in the shadows.
Taking a step beyond these everyday practical questions, and into those shadows in the dark, we come to a harder, more scientific quandary: What is the absolute minimal amount of light our retina needs to detect light, or the lack of light?

This question is the focus of a recent study published in Current Biology 1 The


paper documents the findings of a team at Aalto University in Finland, which has conducted a series of experiments aimed at discovering how dark a shadow can be detected by the rods in the retina, and subsequently received in the brain.
In this case, that brain is that of a mouse in a very, very dark maze, the exit of which was marked by a black spot on a white background. Despite the almost total lack of light in the maze, the researchers found that the mouse was able to detect this marker, and make its way out of the maze.
OFF ganglion cells help us see in the dark
When we think about visual detection, we don’t normally start by thinking about the things which the eye doesn’t detect. But that’s exactly what’s been studied
here, and revealing a lot about how the retina processes the information it receives. This very dark marker in a near lightless maze was visible to the mouse in question not because of the light that was present, but that which was absent. In other words, the mouse’s brain was able to perceive a difference in the eye’s visual field due to the lack of light.
This absence of light is detected by one particular type of neuron, called OFF ganglion cells. These cells have the highest receptivity to minuscule differences in light, and follow their own unique pathway to the brain. The presence, number and pathways of these cells are identical in the human eye to those in a mouse.
These tiny differences in light are what the study’s director, Professor Petri AlaLaurila, refers to as “quantal shadows.” Dr. Ala-Laurila, a neuroscientist and biophysicist at the Universities of Aalto

| June/July 2022 10 VISION RESEARCH POSTERIOR SEGMENT
and Helsinki, told us that these quantal shadows are the ones that occur in insanely dim light.
“The question we need to ask in evaluating these shadows is how many photons are missing — how many photons are needed to detect a shadow? We found that only a few missing photons are required to detect a shadow. These OFF cells are doing an incredibly good performance.
“When light goes off,” Dr. Ala-Laurila continued, “they increase their firing, while ON cells decrease their firing. We then hypothesized that the most sensitive OFF-type ganglion cells are shadow detectors. Perhaps the behavior really relies on that type for shadow detection, and it doesn’t integrate across channels. This was exactly what we found in this study.”
The “ON cells” to which Dr. Ala-Laurila refers are another, quite similar type of incredibly sensitive neurons, the neural ON-ganglion cells. In a previous paper 2 published in 2019, Dr. AlaLaurila’s team talked about these ON cells, which their studies had revealed increase their firing responses depending on the amount of light they were able to detect.
The team found that a distinct correlation between these two types of cells, the most sensitive ganglion cells, drive behavioral decisions whether there is light or no light. They developed a series of techniques in the laboratory to bridge behavior to not only the input layer of the retina, but also to the output neurons which were received by the brain.
“There is a distribution of labor between these two cell types,” Dr. Ala-Lauria said. “Mice have 44 cell types, but very few have the capacity to detect very low light. ON cells react more, OFF do not. Everything we ever see comes from optical responses: It’s a binary world. You see the silencing of the off cells would have allowed an equal sensitivity, so ON cells increase firing. Then the big question was: What are these extremely sensitive OFFf cells doing, then?”
The answer, this new study shows, is reporting the lack of stimulus to the brain, providing information that drives
perception and behavior due to the lack of light.
Where this work began, and where it is going
The question of how we detect light has been one of interest in various fields for hundreds of years. While optical science is of course concerned with this topic, Dr. Ala-Laurila pointed out that his studies, and much of our understanding of optics along with them, have their origins in quantum physics.

“In the past hundred years,” he said, “since the advent of quantum physics, we have been asking what the dimmest light one can detect is. People realized that light consists of photons, and so the smallest amount of light is a single photon. In the early twentieth century, [Hendrik] Lorentz posited the question of whether a single photon could be a limit for the detection of light.
“In the 1940s we began to see psychophysics studies, and realized that only about 100 photons at the level of cornea are needed for about 60% of detection. Only about 12 or so are absorbed into the retina. They concluded at that time that a single rod within the retina has the capacity to absorb a single photon, and distinguish the presence of light.”
This sensitivity is incredibly remarkable, Dr. Ala-Laurila points out, especially compared to any man-made detectors. Since these discoveries were made, neurologists and ophthalmologists alike have wondered what neural mechanisms allow for such sensitivity. This question inspired Dr. Ala-Laurila, and led to his studies of dim light detection. This, he said, is one of the most effective ways to get from molecules all the way to behavioral diseases sometimes, by charting the transmission.
We asked Dr. Ala-Laurila what impact he expected these findings might have upon the eye care industry. He replied that there are a lot of exciting prospects for diagnosis and treatment of neuro-retinopathies due to these new understandings of photoreceptive neurons.
While many of these prospects are currently in development, Dr. Ala-Laurila assures us that many more exciting breakthroughs and applications are coming very soon. Toward the end of developing real-world applications for these newly discovered neural connections in ophthalmology and other fields, he has established a company, Quantal Vision Technologies (Finland), the purpose of which is to utilize these revolutionary findings.
New understandings about how the eye processes light could influence eye care in ways we cannot even foresee at the present moment. We look forward to seeing what new breakthroughs Dr. Ala-Lauria’s team achieves, and what applications they could have in improving people’s sight.
References:
1. Westö J, Martyniuk N, Koskela S, et al.Retinal OFF ganglion cells allow detection of quantal shadows at starlight.Curr Biol. 2022;S09609822(22)00731-X. [Online ahead of print.]
2. Smeds L, Takeshita D, Turunen T, et al. Paradoxical Rules of Spike Train Decoding Revealed at the Sensitivity Limit of Vision. Neuron. 2019;104(3):576-587.e11
Contributing Doctor
Professor Petri Ala-Laurila is a professor of biophysics (Aalto University, Finland, a professor of neurobiology (University of Helsinki) in Finland and the founder of the company Quantal Vision Technologies. His laboratories study the processing of visual signals across the neural circuits of the retina and link these directly to visuallyguided behavior - the final output of biological decision making. The goal is to break new frontiers by revealing fundamental principles of how visual information is encoded in the retina and how the brain reads this neural code to drive animal behavior. The Ala-Laurila lab combines cutting-edge optical imaging, electrophysiological recording techniques, precise manipulations of retinal circuit function, mathematical modelling and state-of-the-art and beyond behavioral assays to reach our goals.
petri.alalaurila@gmail.com
| June/July 2022 11
Longstanding tensions between ophthalmologists and optometrists hurt both patients and the profession — but it doesn’t have to be that way.

Ophthalmologist and optometrist: The words for these professions are so similar that the average Joe likely doesn’t know the difference. But what patients also might not know is that
by Matt Herman
things can get heated between them, and when that happens, everybody loses.
Tensions between professionals are nothing new, but those between ophthalmologists and optometrists (ODs) can sometimes turn bitter. With them both being eye doctors, there is quite a lot of overlap in what these two eye doctors do. Meanwhile, lobbying
groups and individual doctors on both sides in the United States and around the world continue to duke it out to delineate the scope of each profession. “I've been to many parts of the world and I've seen the animosity between these two professions spill over to the courts,” shared Dr. Carmen AbesamisDichoso, an optometrist practicing in the Philippines.
COVER STORY
Unfortunately, patients and the profession as a whole can be harmed by this increasingly frequent bickering; a divided house leads to decreased confidence in caregivers and a lower standard of care, among other things. Fortunately, there are still many things that doctors on both sides of the divide can do to work together for mutual benefit.

Where the battle lines are drawn
The chief source of bad blood between ODs and ophthalmologists has always been about what falls under the purview of each job. These tensions have risen somewhat in recent years in many countries around the world, as the regulatory framework for what procedures ODs are permitted to perform has expanded.

The definition of an ophthalmologist and the associated procedures they are allowed to perform is relatively constant around the world. They are medical doctors (MD) that have completed medical school. Following 12 years of training after high school, including a residency and a fellowship, ophthalmologists are highly specialized doctors that treat eye diseases and conditions by performing surgery and prescribing medication, among other things.
By contrast, the definition of an OD differs from country to country, and it is this ever-expanding list of what optometrists can do that serves as a point of friction. An OD typically spends 8 years in school after high school, and the job has traditionally been the correction of refractive errors using devices such as glasses or contact lenses.
However, optometrists are also considered primary care providers. They are often the first doctor a patient with eye trouble will see, and the sticky topic of what they can do when confronted with certain conditions is changing around the world. In the Philippines, for example, ODs received the right to use certain (mostly diagnostic) medications in 1998, but the time
to obtain an optometry degree increased from four to six years as a result.
In the United States, though, things are moving at a much more lively clip. An optometry degree in the United States requires the usual 4 years of training after an undergraduate degree, but what they can do with that degree is changing.
Optometrists have long pointed to the fact that their curriculum prepares them for far more than correcting refractive errors, and in the United States, they have been increasingly granted permission to go beyond their traditional roles of prescribing vision correction, screening for eye disease, and referring to ophthalmologists. In almost all states, it is now legal for optometrists to prescribe drugs from Schedules III-V, and in many, optometrists are given permission to prescribe hydrocodone.1 Similarly, a majority of states allow optometrists to administer anaphylaxis injections, with nearly half being given the green light for injections.2
A particularly contentious change is that optometrists are beginning to gain a foothold in being allowed to perform surgery, a realm regarded as the exclusive preserve of ophthalmologists. In Alaska, Wyoming, Oklahoma, Arkansas, Louisiana, Mississippi and Kentucky, optometrists can perform a wide array of surgical procedures, including lucrative LASIK surgery.3
Not just about ‘the Benjamins’
The expanding role of optometrists into ophthalmologist strongholds like surgery is a source of financial agitation to be sure, but to dismiss the rift as a simple squabble over cash is unfair. There is another far more precious commodity being feuded over: trust. And when trust is compromised, the patient is the loser.
“Sometimes the optometrist will go into detail about what’s going to happen [when they visit the ophthalmologist],” one anonymous MD complained. “If we maybe offer a different treatment than what the optometrist has alluded to … that patient becomes a little bit distrustful.” This disconnect doesn’t just go one way, either; optometrists regularly run into similar problems, and the consequences can be costly.
It’s not exactly a mystery what happens when the left hand doesn’t know what the right hand is doing, no matter whose side you see it from. Managing patient expectations and establishing faith in the doctor–patient relationship is perhaps one of the most challenging aspects of the medical profession, and ODs and MDs suffer when this relationship collapses due to miscommunication between doctors and misaligned messaging.
“Sometimes coming as the second opinion, right after they've already heard something … the patient really can be confused or even dubious about what we say,” one specialist concluded.
Too many cooks in the kitchen can undermine any doctor’s ability to do their job, and with abysmal medical e-advice becoming the first port of call for scared patients, the highwire act of balancing patient expectations with trust in their care provider is being pushed to the brink of a
Opening up lines of communication
Professional pride, occupational mistrust, increasing
overlap in the scope of professional duties, and competition in the marketplace with big bucks on the line — a heady brew, to be sure. But how to counteract the effects before things get out of hand?
Given the bitterness of the battle and the complex web of competing interests involved, getting optometrists and ophthalmologists on the same page is no easy task. But according to vitreoretinal specialist Dr. Christina Weng, it all starts with picking up the phone. “The more you can cultivate a relationship on a personal level, you get to know [the other party], not only from a professional standpoint, but a personal standpoint, you build that trust. You also build a mutual understanding of referral thresholds.”
No one expects every MD–OD pair to be best buds, of course, but knowing a colleague and building rapport with them leads to confidence in whom you are working with and the decisions they make, and that can only be a good thing. “There are certain optometrists that … when they refer something that's retinal, I feel very confident of the diagnosis that's coming my way. And that's because I've taken care of hundreds of patients with that same person,” reflected Dr. Weng.
The effects of even minimal communication on patient care levels and treatment outcomes are drastic. For one, being confident in the information passed on from a colleague is a major factor in efficiency. MDs can spend more of their time doing MD things, and ODs can spend more of their time doing OD things instead of second guessing each other.
Communication is also, crucially, a boon to levels of patient trust in both parties and helps manage patient expectations by keeping stories about diagnosis and treatment straight. Anxious patients who see a united tandem of doctors that have confidence in their counterpart’s abilities feel better taken care of, and thus are more likely to comply with treatment regimes, come in for followups, and trust diagnoses. The days of cockamamie medical help webpages and second-guessing professional decisions are likely not over, but the more trust patients have in their care
tandem, the smoother everything goes.
Presenting a united front
The fact is, optometrists and ophthalmologists are a team. Even in a world where new boundaries on what doctors can and can’t do are being drawn and redrawn. Dr. AbesamisDichoso believes the pie is more than big enough for everyone. “There’s enough space for ophthalmologists and optometrists in vision care to address the urgent care issues of a patient. It’s not just an optometrist seeing a patient or an ophthalmologist seeing a patient. Teamwork is fantastic and at the end of the day it is for the benefit of the patient.” Ultimately, ophthalmologists and optometrists have the same mutual goal — treating the patient — and this just works better when both doctors see each other as two halves of an organic whole.
Dr. Weng also thinks it’s important for everyone to remember that choosing between treatment options in the medical field is never an exact science.
“We all know that in surgical decision making, there's not always a right or wrong answer. A lot of times, there's an art to it.” If an OD’s opinion conflicts with an MD’s opinion or vice-versa, it is grossly unfair to assume that one doctor is giving the patient “bad” or “incorrect” information. Everybody approaches problems differently, and decision making in medicine is rarely cut-and-dry.
With that being said, certain
expectations are nevertheless created before a surgery consultation that may be contradicted later, and once the cat is out of the bag it’s tough to put it back in. Communicating with the other doctor is again crucial to understanding how to navigate this minefield, but in lieu of that, Dr. Weng has some thoughts for both ophthalmologists and optometrists. “For a patient being referred, I recommend staying more general when discussing options,” she suggested.
Patient questions about what’s coming next are inevitable and deserve explanations. But phrasing things neutrally before the patient sees the doctor making the final call keeps patient expectations neutral, and with properly managed patient expectations, everybody wins.
Not a zero-sum game
Besides, the smart money is that MDs and ODs have more to gain by cooperating. As the go-to primary care provider, optometrists are the foremost font of referrals for their MD counterparts: In a 2010 study in Bradford and Airedale in the United Kingdom, 72% percent of patient records showed a referral from an optometrist.4 Though this may be a bit higher than the average, it is the increase in this number as time has passed that is most eye-opening; only 39% percent of referrals in these hospitals came from MDs in 1988.4
It’s not all about quantity, either. The highest quality referrals also came from ODs as well. Another 2016 study showed a full 1.5% lower false positive rate from OD referrals over general practitioner referrals. Concordance in diagnosis was also almost 10% higher for ODs than GPs (76.1 to 67.2%, respectively).5
Optometrists naturally know the eye far better than a GP, so it follows that their OD referrals are not only going to be more detailed, saving the MD time, but are also more likely to be serious enough to warrant treatment by an ophthalmologist.
There’s no shame in working together, either. To make the relationship work, both sides need to see the other as a partner that works on two sides of the

| June/July 2022 14
COVER STORY
same issue. Optometrists see patients more regularly, know them better, and are more likely to gain the patient’s trust. The front lines of the battle against eye disease are as critical as ever, and ODs are uniquely positioned to do outsized good for patient eye health.
“While I believe that retina specialists are the best-equipped to diagnose and treat retinal diseases, when I think about patients who have been referred to me, many times the first person they have seen is an optometrist,” said Dr. Weng. “A lot of the serious conditions that we take care of, they may be able to detect early on and make a real difference in patients’ outcomes.”
Riding off into the sunset
However significant the challenges facing this relationship are, they are not insurmountable. Dr. AbesamisDichoso has already witnessed a thawing in optometrist-ophthalmologist relations. “I would like to believe the tension between optometrists and ophthalmologists in other parts of the world and in the Philippines is now loosening up because of the younger generation. They don’t care about what the tension was all about in the past; all they want to do is move forward,” she said with a grin.
But even with an air of civility, communication and mutual respect, it's not all going to be sunshine and roses. Conflicts are still inevitable from doctor’s offices to legislatures worldwide, so it’s always going to be a little hot in the eye care kitchen. But even when all else fails, remembering that everyone is on the same team in this mutually beneficial relationship might help make biting the bullet a lot easier.
References:
Contributing Doctors
Christina Y. Weng , MD, MBA is a professor of ophthalmology and the director of the Vitreoretinal Diseases & Surgery Fellowship Program at the Baylor College of Medicine in Houston, Texas (USA). She also has a faculty appointment at the Level 1 Trauma Center of Ben Taub General Hospital (Houston, TX, USA). Dr. Weng graduated cum laude from Northwestern University (Illinois, USA) and then went on to medical school at the University of Michigan (Ann Arbor, Michigan, USA) where she was elected to the Alpha Omega Alpha (AOA) Medical Society. While in Ann Arbor, she pursued an MBA degree from the University of Michigan-Ross School of Business and graduated with high distinction. Dr. Weng completed her ophthalmology residency at the Wilmer Eye Institute-Johns Hopkins University (Maryland, USA) and surgical retina fellowship at the Bascom Palmer Eye Institute-University of Miami (Florida, USA). Dr. Weng is involved with multiple clinical trials; these include the DRCR Retina Network diabetic retinopathy trials and the AGTC Phase 1/2 subretinal gene therapy study for achromatopsia. She also leads numerous research studies in her areas of interest: clinical/surgical outcomes, medical economics, healthcare quality metrics, and telemedicine. Dr. Weng serves on the Board of Directors of the American Society of Retina Specialists (ASRS), American Society of Cataract & Refractive Surgery (ASCRS), and Women in Ophthalmology (WIO) and also holds leadership roles within the Retina Society and Macula Society.

christina.weng@bcm.edu
1. Optometrists: Prescription of Controlled Substances. NCSL Scope of Practice Policy Website. Available at: https://scopeofpracticepolicy.org/practitioners/optometrists/sop/prescription-of-controlled-substances/ Accessed on June 24, 2022.
2. Optometrists: Injectable Authority. NCSL Scope of Practice Policy Website. Available at: https:// scopeofpracticepolicy.org/practitioners/optometrists/sop/injectable-authority/ Accessed on June 24, 2022.
3. Optometrists: Authority to Perform Ophthalmic Procedures. NCSL Scope of Practice Policy Website. Available at: https://scopeofpracticepolicy.org/practitioners/optometrists/sop/authority-to-performophthalmic-procedures/ Accessed on June 24, 2022.
4. Davey CJ, Green C, Elliott DB. Assessment of referrals to the hospital eye service by optometrists and GPs in Bradford and Airedale. Ophthalmic Physiol Opt. 2011;31(1):23-28.
5. Fung M, Myers P, Wasala P, Hirji N. A review of 1000 referrals to Walsall's hospital eye service. J Public Health (Oxf). 2016;38(3):599-606.
Dr. Carmen AbesamisDichoso , OD, MAT, FPCO, FIACLE, FBCLA, FAAO, received her Doctor of Optometry from the Central Colleges of the Philippines (Quezon City, Philippines) in 1989, and earned her Master of Arts in Teaching from the same institution in 2001. Her specialties include special contact lens design for keratoconus, children and high astigmatism; and visual assessment of the mentally challenged, autistic, ADHD, cerebral palsy and learning disabilities. In addition, Dr. Abesamis-Dichoso has been an orthokeratology practitioner in the Philippines since 2005. Since 1998, she has been self-employed in a private practice at Medical Plaza Makati (Manila, Philippines). She was awarded Outstanding Optometrist of the Year in 2017 by the Optometric Association of the Philippines (OAP). Dr. AbesamisDichoso served as the chair of the International Affairs Committee of the Optometric Association of the Philippines; director of the Special Olympics Opening Eyes in the Philippines; program manager of Optometric Association of the Philippines Vision Screening Program and provision of eyeglasses with the United Nations Development Program (UNDP) in 10 areas and four regions in the Philippines; and chairperson of the Special Olympics Healthy Athletes Program in the Philippines. She was also an advisor of the AsiaPacific Region for the Special Olympics Opening Eyes; served as treasurer at the Asia-Pacific Council of Optometry (APCO); and is an APCO representative for the World Council of Optometry which she represented at the World Health Organization (WHO). In addition, she also served as a member of the Legislation, Registration and Standards Committee. She is currently the education chair of Professional ODs Society (PODS) – an organization of optometrists whose advocacy is professional autonomy and independent practice. She has also authored numerous published papers and is a popular lecturer at industry meetings.
carmen.dichoso@gmail.com

| June/July 2022 15
Seeing Colors in the Dark
by Tan Sher Lynn
Advanced deep learning paves the way toward full-color night vision imagery, which could assist retinal studies and eye surgeries
Why can't humans see colors in the dark?

Sight begins when light enters the eye, triggering light-sensitive cells (photoreceptors) in the retina at the back of the eye. These cells then send signals along the optic nerve to the brain, which makes sense of them, giving us the experience of seeing.
There are two kinds of light-sensitive organs located in the backs of our eyes: rod-shaped and cone-shaped. Rods are extremely efficient — a little amount of light can trigger them. They can detect lines, movement and contrast but cannot distinguish color. Meanwhile, cones are responsible for color vision but need plenty of light to be activated. In the dark, cones lose their ability to detect colors, hence, we can’t recognize an object’s color even though other animals can recognize it.
Night-vision technology currently available
Light visible to humans falls between 400-700 nm. Hence, objects appear to
have certain colors depending on the wavelength of light that is reflected off them and enters the eye.
In order to “see” better in the dark, humans have developed night vision technology, which includes thermal imaging, low-light imaging and nearinfrared illumination. These technologies have assisted in improvements in various fields, including night driving or flying, wildlife observation, search and rescue, night surveillance and studies of biological samples. As researchers continue to work toward a technology that could provide full-color images or videos with the absence of light, they began to harness the power of machine learning.
The beauty of colors in darkness
Recently, a team of researchers at the University of California Irvine (USA) published a paper where they enabled limited color vision in the dark using deep learning.1 The study demonstrated that deep learning network can be used to “teach” an AI system how to figure out the color(s) of an object in the absence of visible light.
Their method involved the use of more than one wavelength of infrared light and data from light in the visible spectrum.
A monochrome camera capable of responding to light in both the visible and infrared spectrum was used to take multiple pictures of people’s faces. Photographs of each image under different wavelengths of illumination or combinations of single wavelength illuminated images are used to train machine-learning models to predict RGB color images. Architectures inspired by the U-Net architecture, in which a contracting path is combined with an expansive path, was used.
They then used the system to take similar photographs in the dark. In so doing, they found the system was capable of making accurate guesses to color the pictures, which were shown on an external display. Human subject comparison of model outputs with ground truth inputs was also used to determine perceived model performance.
In a nutshell, this proof-of-principle study demonstrated that deep neural networks, particularly convolutional neural networks (CNNs), are capable of producing color reconstructions starting from infrared-illuminated images, taken at different infrared wavelengths invisible to the human eye. Such systems could be the starting point of the development of technologies that colorize night-vision imagery, which would be indispensable in practical situations such as security and military operations, animal observation, handling restoration of light-sensitive artifacts, as well as studying samples sensitive to visible light. For instance, studying light-sensitive retinal tissue may require processing the sample in darkness to avoid altering its biochemistry and function,2,3 and performing eye surgery in a low light setting can avoid retinal damage,4 the authors noted.
References:
1. Browne AW, Deyneka E, Ceccarelli F, To JK, Chen S, Tang J, et al. Deep learning to enable color vision in the dark. PLoS One. 2022; 17(4): e0265185.
2. McLelland B, Lin B, Mathur A, et al. Transplanted hESC-derived retina organoid sheets differentiate, integrate, and improve visual function in retinal degenerate rats. Invest Ophthalmol Vis Sci. 2018; 59(6): 2586–2603.
3. Palczewska G, Stremplewski P, Suh S, Alexander N, et al. Others Two-photon imaging of the mammalian retina with ultrafast pulsing laser. JCI Insight. 2018;3(17): e121555.
| June/July 2022 16 INNOVATION VISION RESEARCH
4. Youssef P, Sheibani N, Albert DM. Retinal light toxicity. Eye (Lond). 2011; 25(1): 1–14.
Get Ready for Chicago
AAO 2022
Subspecialty Day
AAOE® Program
AAO 2022 Expo
Sept. 30 – Oct. 3
Sept. 30 – Oct. 1
Sept. 30 – Oct. 3
Oct. 1 – 3
Closing Session Keynote Speaker
Pulitzer Prize-winning author and presidential historian Doris Kearns Goodwin brings history alive with an uncanny sense for detail and a master storyteller’s grasp of drama and depth as she examines the leadership triumphs, trials and tribulations of the men and women who have shaped this nation, culled from her lifetime examination of the U.S. presidency.

Register Now and Save at aao.org/registration
Registration is now open to everyone! Get the best deal by registering before July 27. Registration includes:
AAO and AAOE sessions | Skills transfer lectures | Videos and posters | AAO 2022 Expo | AAO 2022 Virtual content
Where All of Ophthalmology Meets® aao.org/2022

| June/July 2022 17 5998
Scan this code to register for AAO 2022 now!
Innovations in Retinal Imaging Benefit Glaucoma Management
Singaporean ophthalmologists discussed new technologies and methods that could change disease diagnosis and management during the 5th ASEAN Ophthalmology Society Virtual Congress (AOS Virtual 2022).

Real-time intraoperative OCT
Optical coherence tomography (OCT) has revolutionized the diagnosis of eye disease and patient management since 1993 when the first OCT retinal imaging prototype instrument was created. In 2006, the spectral-domain OCT (SD-OCT) became the standard of care. Later, new technologies were born, such as the swept-source OCT
by Tan Sher Lynn
(SS-OCT) and OCT angiography (OCT-A). “With the integration of the OCT into the microscope, for the first time, real-time OCT was possible. Today, there are three FDA-approved intraoperative OCT (iOCT) systems: ZEISS Rescan 700, Haag-Streit iOCT and Leica EnFocus,” noted Dr. Tien-En Tan from Singapore.
He shared his knowledge and experience of using the Leica EnFocus iOCT, which helped alter his decision-making in surgery. “The Leica EnFocus is fully integrated into the Proveo 8 Ophthalmic Microscope. It is a spectral domain OCT and can be used on the anterior and posterior segments. It has an axial resolution of 2.4-4.0 μm, high scan density of 500-1000 B-scans and automatic image optimization,” he said.
In the DISCOVER study* of 593 posterior segment cases, it has been shown that iOCT added valuable information in about 60% cases, altered surgical decision-making in about 30% of the cases, and was most useful for membrane peeling (there was 2040% discordance between surgeon impression and iOCT findings on whether the membrane peel was complete).
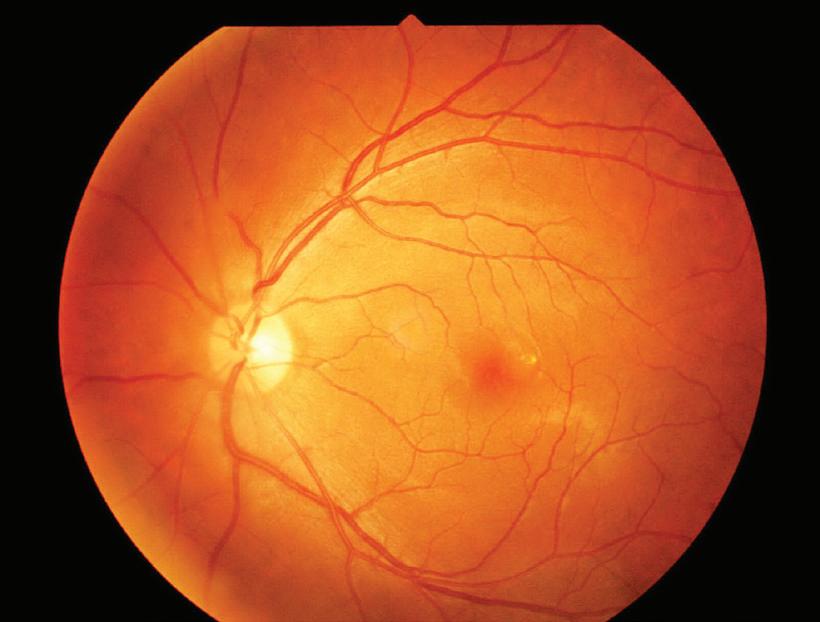
He shared a case done in the Singapore National Eye Centre where the iOCT helped to detect extrafoveal polypoidal choroidal vasculopathy (PCV), which helped the surgeon decide on performing an intraoperative endolaser. “This step would not be possible without the iOCT,” he said.
According to Dr. Tan, various applications for iOCT in vitreoretinal surgery have been described, including epiretinal membranes, membrane peeling, macular holes, retinal detachment, vitreous hemorrhage, diabetic vitrectomy, pediatric vitreoretinal surgery and subretinal surgery.
In the future, OCT-compatible surgical instruments may be available; there have been attempts in developing them to rectify the issue of shadowing on OCT caused by current surgical instruments, noted Dr. Tan. “Other possibilities in the
| June/July 2022 18 INNOVATION RETINAL IMAGING
future include automated iOCT tracking and digital staining as the software improves,” he said.
Use of the RNFL thickness to detect early glaucoma

A diagnostic challenge in using OCT to detect early glaucoma exists due to anatomical factors that can affect retinal nerve fiber layer (RNFL) measurements, according to Dr. Jacqueline Chua Yu Min from Singapore. “Clinically, there’s a need for a single, comprehensive model to account for all factors that are associated with RNFL thickness, such as ethnicity, age and refractive error, among others, to improve the diagnostic performance of OCT for early glaucoma detection,” she said.
Dr. Chua and her colleagues developed a compensation model with the aim of refining and validating an Asianspecific compensation model based on their established regression model (Caucasian-model).
“We hypothesize that the refined model will further reduce the inter-subject variability of the circumpapillary RNFL thickness,” she added.
Two thousand six hundred ninetynine (2,699) healthy participants were enrolled to construct and test a multivariate compensation model, which then was applied in 387 healthy participants and 387 patients with glaucoma. Compensated RNFL thickness was generated based on ethnicity, age, refractive error, optic disc (ratio, orientation and area), fovea (distance and angle), and retinal vessel density.
After applying the Asian-specific compensation model, the standard deviation of RNFL thickness reduced, with the greatest effect seen on Chinese participants (16.9%), followed by Malay participants (13.9%), and Indian participants (12.1%). Multivariate normative comparison outperformed measured RNFL for discrimination of early glaucoma (area under the curve [AUC], 0.90 vs. 0.85; P < 0.001), moderate glaucoma (AUC, 0.94 vs. 0.91; P < 0.001), and advanced glaucoma (AUC, 0.98 vs. 0.96; P < 0.001).
“The compensation model improves the diagnostic performance of RNFL measurements. Compared to deep learning approaches, the current technique is a comprehensible approach that can easily be implemented in clinical workflow. Of course, the compensation approach needs to be externally validated in future studies,” she said.
Novel tube shunt for refractory glaucoma
There are various indications for glaucoma drainage implants, including failed trabeculectomy, uveitic glaucoma, neovascular glaucoma, trauma and aphakic glaucoma. However, current commercially available implants have several issues, noted Dr. Victor Koh from Singapore. “In the long run, intraocular pressure control is an issue due to the encapsulation of
the conjunctiva over the plate. There can also be complications such as hypotony and shallow anterior chambers, hypertensive phase, tube exposure/ conjunctival erosion and corneal decompensation,” he said.
The Paul Glaucoma Implant (PGI), with its micro-sized tube, maximum effective surface area and flexible plate, may just be the solution to the issues listed above.
According to Dr. Koh, compared to commonly used implants like the Ahmed Glaucoma Valve and Baerveldt, the PGI has a bigger plate size, which is able to reduce encapsulation. In terms of wingspan, it is also smaller and more flexible, making it easier to implant. In addition, its smaller outer diameter tube size poses less risk of tube erosion and cornea touch, while its smaller internal diameter tube size helps to prevent hypotony.
* Ehlers JP, Modi YS, Pecen PE, et al. The DISCOVER Study 3-Year Results: Feasibility and Usefulness of Microscope-Integrated Intraoperative OCT during Ophthalmic Surgery. Ophthalmology. 2018;125(7):1014-1027.
Editor’s Note:
The 5th AOS Congress was held virtually on March 26-27, 2022. Reporting for this story took place during the event. A version of this article was first published on piemagazine.org.
| June/July 2022 19
“Clinically, there’s a need for a single, comprehensive model to account for all factors that are associated with RNFL thickness, such as ethnicity, age and refractive error, among others, to improve the diagnostic performance of OCT for early glaucoma detection.”
— Dr. Jacqueline Chua Yu Min, Singapore
On a dark and stormy night, in a castle on a mountaintop, the doctor achieved what nobody ever had before: He had restored dead tissue to life!
Actually, most of that isn’t true. This wasn’t just one doctor on one night, and it wasn’t dark or stormy. The setting was not a faraway castle, but rather at a Scripps Research facility in La Jolla, California. Getting past the classic horror tropes though, the most important and incredible line of that introduction is true: Doctors have in fact successfully restored activity to once-dead retinal cells.
In a study1 recently published in Nature, a team led by Professor Anne Henneken of Scripps Memorial Hospital and Dr. Hans Vinberg of the University of Utah’s John A. Moran Eye Center and a visiting investigator at Scripps, described their remarkable new investigations into the life of neural cells, focusing on the rod and cone receptors within the retina.
of received light impulses to the brain via the optic nerve. Once the function of these cells has ceased, blindness has been considered inevitable and irreversible.

Working up the neural food chain
Interestingly enough, though human neurons do not generally regenerate, this is not the case throughout the animal kingdom. Fish, frogs and birds have the innate ability to regenerate neurons — as well as retina cells — when these cells experience damage or mutation. Recent studies3 have begun to explore these animals’ capability to regenerate their retinal tissue, in the hope that their findings may be transferable to human tissue.

was released that showed rejuvenation of retinal cells had been achieved in lab mice.
That study,5 published in Nature, showed that a transcription factor known as ASCL1 was responsible for activating genes which prompt regeneration of Müller cells following retinal damage in the eyes of zebrafish. These fish have retinal structures which, though they come from dramatically different neural systems, resemble those of mammals quite significantly.
The study revealed that by targeting cells in a mouse whose retinal neurons had experienced cell death with that same transcription factor, the mouse’s Müller cells were able to form functioning synapses. The cells were also able to be reprogrammed by the mouse’s genetic
| June/July 2022 20 INNOVATION VISION RESEARCH
How researchers have revived light-sensing tissue in organ donor eyes
“It’s alive … alive!”
material into showing characteristics of interneurons, capable of transmitting
Reviving the human retina
Though the revival of retina cells in a mouse was a substantial leap forward in understanding and potentially treating retinal cell death, science had not yet successfully applied these techniques to the human eye. Now, with the recent Scripps and Utah study, this has been in a limited laboratory setting.
The aim of this study has been to obtain more accurate research data by conducting experiments on revived eyes from deceased human organ donors instead of lab animals. Though research is typically conducted on mice, and other mammals with substantial nervous function, the eyes — and especially the retinas — of these
restoring a supply of oxygen and a typical, static acid-alkaline (pH) balance to the removed eye, the research team was able to restore retinal function within that previously non-functioning eye. Light receptivity returned to photoreceptors, and these signals were in turn networked by the retinal ganglion cells as well.
Having achieved this already remarkable result with mouse eyes up to three hours after the test subject’s death, the researchers then took the next, pivotal step of attempting the same process using eyes from deceased human donors. Working in collaboration with the San Diego Eye Bank and the organ donor society Lifesharing, the research
team coordinated a rapid transporting operation in order to attempt to restore function to human eyes in the hours directly after their donors’ death.

Incredibly, the results were very positive. The researchers discovered that they were able to achieve similar retinal signaling from human autopsied eyes. Not only were these human retinal networks able to be restored to a functional state, but they were able to do so after a greater duration than with the mouse cells, up to five hours after death.
Breathing new life into research and treatment
The potential uses of these findings gives retinal researchers a massive new opportunity for studying and treating conditions that had until now been limited by what was considered impossible: close study of functioning human retinal networks. The potential for analysis of incurable diseases that rob countless patients of their sight each year may very well have arrived at a bold new era.
with this goal had been the lack of previous studies, and the presuppositions regarding cell death in organ donors. The commonly-held belief that neurons irreversibly cease function had until this point been taken as a given for retinal cells in donor eyes.
In this study, the researchers first confirmed that cell death does in fact occur quite rapidly in lab mice, whose retinal function ceased rapidly after the mouse was euthanized. Using a technique called electroretinography, the researchers documented the decline and cessation of transmission of light sensitivity by the subject photoreceptors.

What they did next, however, prompted a somewhat surprising result. By
References:
What’s more, the study reveals that the long-held beliefs of medical science regarding neuron cell death may not be as completely true as once had been believed. This, in itself, may be of tremendous benefit in discovering treatments for the various afflictions of the retina.
In reviving retinal tissue for study in the lab, these researchers may also have revived hopes for the future of treating numerous retinal diseases.
1. Abbas F, Becker S, Jones BW, et al. Revival of light signalling in the postmortem mouse and human retina. Nature. 2022;606(7913):351-357.
2. Restoring sight: Can the retina be regenerated? UW Medicine Newsroom Archive. Available at: https:// newsroom.uw.edu/story/restoring-sight-can-retina-be-regenerated Accessed on July 4, 2022.
3. Pitt Scientists who Regrew Retina Cells to Restore Vision in Tiny Fish set their Sights on Humans. UPMC Newsroom. Available at: https://inside.upmc.com/retina-cell-regrowth-to-restore-vision/ Accessed on July 4, 2022.
4. Helping the retina regenerate. NEI Research News. Available at: https://www.nei.nih.gov/about/news-andevents/news/helping-retina-regenerate Accessed on July 4, 2022.
5. Jorstad NL, Wilken MS, Grimes WN, et al. Stimulation of functional neuronal regeneration from Müller glia in adult mice. Nature. 2017;548(7665):103-107.
6. Light after death: scientists revive human eyes. Scripps Research News. Available at: https://www.scripps. edu/news-and-events/press-room/2022/20220511-hanneken-scientist-revive-human-eyes.html Accessed on July 4, 2022.
| June/July 2022 21
Breaking the Glass Ceiling

The Prof. Ava Hossain Story
by Joanna Lee
Dreams do come true … but without the right support, strategies and action, they might just remain in the imagination.
“I always feel their blessed hands on my head which gives me strength, ideas and inspiration to break the glass ceiling and move forward.” With this touching tribute to her late parents, President Elect of APAO (2019) as well as Chairperson of APAO-WHO Standing Committee Professor Ava Hossain began her honorary lecture at the recently held 80th Annual Conference of the All India Ophthalmological Society (AIOC 2022).
Born in Kumudini Hospital in Mirzapur, a humble region in the major state of Tangail in Bangladesh, she was also raised on the compounds of the very same charitable hospital where her father, the sole breadwinner, had worked to support her family which included her and her seven brothers.
The modest staff quarters inside the hospital’s complex was her family’s home, and the value of education and hard work were instilled in them by her
parents from an early age.
Her early education in the austere Bharateswari Homes school taught her much discipline through the 5 a.m. wake-up calls and strict supervision from the teachers there, including having meal times in total silence. School vacations were very short as well.
Seed for sight-saving planted
The moment of epiphany for her arrived one day when she noticed some patients queuing at the hospital’s verandah. “Something was odd. They were all holding the person in front of them by his shoulders and had a bewildered, lost look on their faces,” she said. “I went inside the ward and was told they came here to be treated for blindness.”
A few days later, she saw the same patients again at the hospital but this time, they wore thick glasses. She asked them if they could see. They told her that

they could now see everything and eat by themselves.
“One could clearly see the wonderment in their eyes and the unmistakable joy on their faces,” she recounted. “That day, a seed was planted in me.”
What doesn’t kill you makes you stronger
She went to Mymensingh Medical College in December 1970. In the following year, Bangladesh’s Liberation War started and her education was disrupted. Her brother lost his right hand during the war but she spoke of him with pride as he had gone on to achieve first places in several examinations using his left hand.
Her family’s experience with the 9-month war seemed to have strengthened her resolve to never give up. Her grit helped her push through one of the major setbacks in the beginning of her career in ophthalmology. In 1979, she was denied a training opportunity in the field due to her gender. “Not giving up, I made alternative plans and followed through,” she said.
Prof. Hossain strategized to complete the first part of a two-part fellowship program which would guarantee her a residency placement.
But it was a highly competitive program where even getting an admission was extremely difficult. Striding through, she ultimately succeeded in getting admitted and passed the part 1 examinations in the attempt to secure her spot for the residency.
After that, Prof. Hossain gained a place in the residency training program at the prestigious National Institute of Ophthalmology. “I took this great opportunity to learn from the very best and developed my skills,” she said.
After completing the residency and the fellowship part 2 course, Prof. Hossain finally passed the rigorous fellowship examinations in January 1985. During that period of learning, one of her mentors was Prof. AQSM Harun, the pioneer of microsurgical technique in Bangladesh
| June/July 2022 22 WOMEN IN OPHTHALMOLOGY ENLIGHTENMENT
Trailblazing the way for women
The same year of her graduation also saw Prof. Hossain become the first female fellow in ophthalmology in Bangladesh. Today, the number has grown to over 200. Beaming with joy, she mentioned that several of her students, now professors of ophthalmology themselves, were sitting among the audience during her speech
“I am proud to have inspired many women to consider ophthalmology as their specialization,” she said
These days, the residency program sees women doctors making up two-thirds of its number in each cohort, she further said.
Ever learning, ever progressing
Prof. Hossain has never looked back ever since her first publication in 1986 on retinal detachment. Besides obtaining exposure to modern technologies at the Orbis Flying Eye Hospital, she also trained at the Princess Alexandra Eye Pavilion at University of Edinburgh in the United Kingdom, as well as the Institute of Clinical Ophthalmology in Kiryu, Japan.
She had the privilege of gleaning from some of the top leaders in ophthalmology during her trainings, among whom were Prof. Momosi from Japan, Prof. M. A. Matin, Prof. Ahmad Sharif and Prof. Mustafizur Rahman.
“I always tried to learn the best things from all of my mentors.”
Light for the next generation
In the last 40 years, Prof. Hossain had found teaching as one of her callings as she had been imparting her skills and experiences to undergraduate and postgraduate students at the National Institute of Ophthalmology (NIO) for two decades.
Besides providing guidance for research protocols, she has mentored many
aspiring ophthalmologists throughout her career
She has also contributed significantly to ophthalmic policy developments at a national level and beyond, apart from playing a stewardship role in the National Eye Care (NEC) program, as well as played an advisory role in the prevention of blindness survey in Bangladesh.
Prof. Hossain also encouraged doctors to build their networks. She was involved with the Ophthalmological Society of Bangladesh (OSB) from the start of her career, having moved up the ranks from treasurer, secretary general, president in 2009 to 2010 and to an advisor from 2011 to now.
Ever the pioneer, she was also the founder and president of the Bangladesh Community Ophthalmology Society.
Outstanding secrets
Sharing from her heart, Prof. Hossain let the audience in on the qualities that have helped her to stand out apart from the crowd. One of the key qualities of her success is her dedication to regular communication with her seniors, juniors, colleagues and friends while giving prompt responses on emails. She also believes in growing relationships, taking initiative and promoting professional development, as well as developing leadership qualities.
Leadership: The world is your oyster
Beyond her involvement at the community and national levels, Prof. Hossain has also spread her wings internationally, leading several regional and global ophthalmological societies. In 2011, she became the president of SAARC Academy of Ophthalmology (South Asian Association for Regional Cooperation). She was also involved in the Asia-Pacific Academy of Ophthalmology (APAO) in various capacities, serving as Chairman of the Registration Committee in 1993 and regional secretary from 2009 to 2019. She also served as
vice president from 2015 to 2019. Prof. Hossain made history in 2019 by becoming the first ever woman president-elect in the APAO’s 62-year history. Two years later, she served as president of APAO’s Virtual Congress. Among her many illustrious awards received through the years, she was bestowed the Honorary Fellow of AllIndia Collegium of Ophthalmology (FAIO) in 2020. She is also listed as one of the top 100 women in the Ophthalmology Power List 2021.
Giving back in gratitude
Perhaps even more inspiring above her titles and accolades are her efforts in expressing her gratitude and appreciation to the place of her birth, Kumudini Hospital, where it all began. She volunteered her weekends there at the prime of her career.
There, she taught the students at the Kumudini Medical College while performing surgeries free-of-charge, as well as training the staff at its ophthalmology department.
The hospital was 60 km away from her home which meant a long 2-hour journey each way.
Despite the sacrifices of time, she said, “The work at this hospital has been one of the most fulfilling experiences of my career; in a way, my life has completed its full circle.”
Editor’s Note:
Prof. Ava Hossain was the keynote speaker in a special session on Day 1 of the recently held 80th Annual Conference of the All India Ophthalmological Society (AIOS) in Mumbai, Maharashtra (a.k.a. DREAMCON AIOC 2022), where recipients of the AIOS General Awards 2021 were honored with a tribute. Reporting for this story took place during the event.
| June/July 2022 23
The Effect of the COVID-19 Pandemic on Babies and Other Updates in ROP
Retinopathy of prematurity (ROP) is a potentially blinding eye disorder that primarily affects preterm babies. It can result in retinal detachment, myopia, strabismus, amblyopia and glaucoma.

The smaller a baby is at birth, the more likely that baby will develop ROP.

This disorder — which usually develops in both eyes — is one of the most common causes of visual loss in childhood and can lead to lifelong vision impairment and blindness.
By 2020, ROP will be the single largest
by Hazlin Hassan
cause of needless blindness. Delegates heard how the COVID-19 pandemic took a toll on the management and screening of babies with ROP during a session held on the second day of the 5th meeting of the ASEAN Ophthalmology Society (AOS Virtual 2022) on Sunday March 26, 2022.
What India and the Philippines have in common
Dr. Rachelle Anzures, of the Anzures Eye, Ear, Nose and Throat Specialty Clinic (Philippines), shared the ways in
which clinics adapted to the new norm. “The COVID-19 pandemic has led to the significant disruption of the delivery of health care, including retinopathy of prematurity,” she said. Published reports from the Boston Children’s Hospital (USA), have shown that more infants required ROP examination in 2020 compared to 2019, but fewer examinations were performed. There was an 18% increase in the number of infants screened but there was a 19% decrease in the number of examinations performed.
In India, there was a significant decrease in the number of infants screened and treated in 2020 compared to the previous year. At the All India Institute of Medical Sciences (AIIMS), there was a 73-89% decrease in the number of infants screened and a 63-88% decrease in the number of infants treated. At the Ospital Ng Makati (Philippines), the number of screenings dropped from 79 in 2019 to 30 in 2020. This rose to 50 in 2021. The number of babies treated also fell from 8 in 2019 to 3 in 2020. This rose to 8 in 2021.
| June/July 2022 24 ROP UPDATE ENLIGHTENMENT
Pandemic-related ROP blindness
During the pandemic, there was an increased incidence of ROP blindness. Because of COVID-19 restrictions, lack of public transport and travel restrictions — not to mention the fear of virus among parents — delayed presentation to ophthalmologists, who were already in short supply. Ophthalmologists have also had to adapt their practice patterns to ensure continued delivery of care without compromising the safety of patients. Infants and toddlers can be asymptomatic carriers or in the presymptomatic period of transmission. Implementing source control measures like face masks and social distancing in this age group is difficult, Dr. Anzures noted.
Crying increases the risk of aerosol generation and transmission. Proximity of these patients to caregivers, along with sustained crying, might further increase the risk and load of aerosol. Risk mitigation and infection control measures include wearing proper personal protective equipment, daily screening of staff, proper disinfection of indirect ophthalmoscope and screening instruments, as well as visitor screening prior to hospital entry. Face-to-face interactions are minimized and family counseling is done via teleconference. Disinfection is carried out in between examinations.
While the pandemic has presented many challenges, it has also led to opportunities such as improvements in virtual communications with parents, virtual meetings and lectures. “From an infection control point of view, the pandemic has exposed what we lacked in taking care of our patients. However, it has also accelerated the development of technologies needed to address the problems,” emphasized Dr. Anzures. Advances in telemedicine and artificial intelligence (AI) are seen as being able to bridge the gap in the availability of medical care.
Laser or anti-VEGF? That is the question…
While laser remains an important management option for ROP, the use of anti-VEGF may have higher treatment
success, better structural outcomes, involve less surgical intervention and less myopia, said Dr. Gavin Tan, senior consultant in surgical retina at the Singapore National Eye Centre (SNEC).
In the RAINBOW study, infants treated with ranibizumab 0.2 mg were twice as likely to achieve clinically-relevant treatment success compared with those treated with laser. RAINBOW was a randomized, multicenter, open-label, parallel-group clinical trial to compare ranibizumab with laser therapy in premature infants with ROP. The primary objective was to demonstrate superior efficacy of ranibizumab 0.2 mg to laser as measured by treatment success at week 24.
The study results showed that a treatment success rate of 80% was achieved with ranibizumab 0.2 mg versus 66% with laser. Ranibizumab 0.2 mg was well tolerated in patients with ROP and the safety profile was as expected in a preterm population. RAINBOW was the first study to report pharmacokinetic and systemic VEGF data in ROP.
A 5-year extension study is currently ongoing, with results expected in 2022. The use of anti-VEGF must however be balanced with the risk of recurrences which may occur later and require a higher follow-up burden, Dr. Tan cautioned. Screening remains the most critical tool to prevent blindness. If ROP is not detected early, this decreases the chances of treating and preventing blindness. “Always involve parents in the discussion,” he added.
The role of FFA in ROP pathogenesis
Fundus fluorescein angiography (FFA) could play a central role in better understanding the pathogenesis of ROP, said Dr. Sumalin Trichaiyaporn, a pediatric retina specialist from Queen Sirikit National Institute of Child Health, Thailand.
ROP is a vasoproliferative disorder that can lead to significant blindness in children. FFA is the gold standard technique to study retinal vasculature and a valuable tool for evaluation of the chorioretinal vasculature in
vasoproliferative disorders. It appears to be safe in children including neonates with ROP, with no adverse effects reported. Ultra-widefield multimodal imaging, including FFA, has an increasing role in the pediatric retina for diagnosis, treatment and follow-up. The use of FFA is suggested because certain components of the International Classification of Retinopathy of Prematurity (ICROP) are subjective and open to interpretation.
The introduction of anti-VEGF therapy has presented new challenges associated with the recognition of clinical features characteristic of posttreatment regression and reactivation. The pattern of ROP in regions of the world with limited resources is also not adequately described by the current classification system. FFA in ROP is useful to evaluate the position and extent of pathologic vascular proliferation (especially stage 3 disease), the activity of fibrovascular proliferation, the vascular pattern of the retina, the capillary network of the retina, and the extent of photocoagulation.
In conclusion, the supplementation of color fundus photographs with FFA may improve the accuracy for certain classifications and management of ROP. In the era of multimodal imaging, FFA is playing an emerging role in guiding the management of ROP in the anti-VEGF era. FFA may have a role in characterizing vascular maturation after intravitreal anti-VEGF therapy. Prospective multicenter studies assessing the long-term role of FA in this population are warranted, said Dr. Trichaiyaporn.
Editor’s Note:
The 5th AOS Congress was held virtually on March 26-27, 2022. Reporting for this story took place during the event. A version of this article was first published on piemagazine.org.
| June/July 2022 25
The Stars and Stalwarts of Ophthalmology, Honored at AIOC 2022
by Gloria D. Gamat
In a special session on Day 1 of the 80th Annual Conference of the All India Ophthalmological Society (AIOS) in Mumbai, Maharashtra (a.k.a. DREAMCON AIOC 2022), recipients of the AIOS General Awards 2021 were honored with a tribute. Each awardee shared their journeys, not just in life, but in ophthalmology as well. Aptly named Ophthalmology, My Life and My Dreams, the session’s theme spoke volumes of each awardees’ humble beginnings, finer qualities and noble aspirations for the future of ophthalmology.
Keynote Speaker Prof. Ava Hossain opened the session by recognizing her parents, her family, the schools she went to, and especially, her many mentors along the way, with whom she
credits her success. Without them, she said, she would not have reached the leadership status she currently has in ophthalmology, not just in her home country of Bangladesh, but also in India and the whole Asia-Pacific region.
Indeed, Prof. Hossain has come a long way: From being rejected for an ophthalmology training course in 1979 because of her gender, to becoming the first female ophthalmology fellow in Bangladesh (1985) — and then the first ever woman President-Elect (2019) in the 62-year history of the Asia-Pacific Academy of Ophthalmology (APAO). Furthermore, she has been awarded the APAO Arthur Lim Award, a prestigious award for her contribution in the prevention of blindness — just one of the
many feathers in her cap.
Today, Prof. Hossain continues to teach and enlighten young ophthalmologists through her various leadership roles in the ophthalmic community, while basking in the love of her immediate family. “I am quite fortunate in getting unconditional support from my husband and family,” she emphasized.
Why ophthalmology?


“Ophthalmology combines the best of medicine in one specialty — it is medical, and it is surgical, treating both wellness and disease,” said AIOS C. N. Shroff Award 2021 recipient, Prof. Dr. Atul Kumar, when it was his turn to take the center stage.
“Our patients span all ages, and the range of the diagnoses is broad. The technology in ophthalmology is breathtaking, and the innovation — as I have witnessed — is constant,” he added. Since 2016, Prof. Dr. Kumar has served as chief and professor of ophthalmology for the Dr. Rajendra
| June/July 2022 26 OPHTHALMIC LEADERS ENLIGHTENMENT
Prasad Centre for Ophthalmic Sciences (RPC) at the All India Institute of Medical Sciences (AIIMS) in New Delhi. He is well known for his efforts in the upliftment of ophthalmology in India, and despite his countless high-level accolades in his four-decade-long ophthalmic career, Prof. Dr. Kumar remains a passionate teacher and learner. “Learning never ceases,” he highlighted. While he started as a junior resident of RPC at AIIMS way back in the early ‘80s, he continued his professional work with focus on patient care and mentoring students. He never hesitated to face challenges along the way and his eye on innovation in ophthalmology never waned: For example, Prof. Dr. Kumar pioneered and aced the heads-up 3D VR surgery in India.
On the other hand, it was program failure and an early heart health issue that almost curtailed AIOS P. Siva Reddy Award 2021 recipient Dr. Samar Basak’s career in ophthalmology. But he has always dreamed of becoming a faculty member of the prestigious RPC at AIIMS. However, it seemed that in every moment, there was an obstacle. But he never gave up.
According to Dr. Basak, the one thing that can prevent you from achieving your dreams is when the conflict between job and career starts. “A job is often boring and has no scope to grow further,” he said. “A career on the other hand is a journey that includes your job experience, learning, training, teaching, and most importantly, your attitude toward fulfilling your long-term dreams or goals,” Dr. Basak explained.
So when an opportunity knocked and he was called to join and build a hospital for a cause, Dr. Basak took a leap of faith. At present, he is the director of DISHA Eye Hospitals & Research Centre and dreams of having 1,000 cornea surgeons, each of whom could perform 100 grafts per year. He believes that 100,000 grafts per year can support the Vision 2020 India Program. “Your career is a journey, not a job. So dream and enjoy the climb,” Dr. Basak added.
The Evolving Ophthalmic Technology
Similar to Prof. Dr. Atul, AIOS P. Siva
Reddy International Award 2021 recipient, Prof. Jeewan Singh Titiyal, MD, always had an eye for the evolving technology in ophthalmology. Sharing his evolution as an anterior segment surgeon, Prof. Tityal was very nostalgic when he shared his triumph over modern day cataract surgery and overcoming the learning curve in SMILE (small incision lenticule extraction) — a far cry from his first intracapsular cataract extraction in the early ‘80s. Although he said, “all that, are not my true achievements.”
“My true achievement lies not in the performing of thousands of surgeries with the latest technology or optimal costs to the patient or hospital,” noted Prof. Titiyal. He said that his real triumphs include training the next generation of ophthalmologists in cutting-edge technology, for one. “That, and bringing them at par with the global platform,” he emphasized. “And then when my students become my teacher and together we promote research and innovative thinking for the betterment of society,” he explained.
Uveitis and Eye Banking: Eye Care Dilemmas as Old as Time
AIOS B. K. Narayan Award 2021 recipient, Dr. Jyotirmay Biswas’s journey in “battling” uveitis goes back some sweet 28 years, which basically started with India’s battle against tuberculosis. “Two million people develop TB every year and every 4th TB patient is an Indian,” said Dr. Biswas. Tubercular uveitis is the protean manifestation, he said. Having been involved for more than two decades in various national and international studies involving ocular tuberculosis, Dr. Biswas and his co-investigators found that the most common presentation of the condition is asymptomatic choroidal tubercle — this occurs in more than 50% of patients with ocular TB. Throughout the years of investigating this ancient disease, newer therapeutic strategies now focus on even more novel modes of immunomodulation.
Efforts in eye banking, like for uveitis, is an eye condition and dilemma that continues to be in progress, and is not
fully addressed or treated. It’s not that experts are failing, but the condition itself is evolving to higher levels as time goes by.
“While prevention is the most desirable way to control corneal blindness, once a cornea loses its transparency, a corneal transplant is the patient’s best chance to regain vision in the affected eye,” stated Dr. Sujata Das, AIOS K.R. Dutta Award 2021 recipient.
Easier said than done as it’s a known fact that the biggest limiting factor is the shortage of donor corneas, Dr. Das reminded the audience. “There is a huge need for transplantable donor corneas in order to reduce the rate of corneal blindness worldwide,” she added.
While she presented the data and known statistics on the global issue of blindness, Dr. Das focused more on the vision for India and how efforts on putting up eye banks will impact the eradication of blindness. Moreover, Dr. Das shared that the vision is that there’ll be no more waiting lists for cornea blinds in India and that the country should export corneas to other countries in need. “Eye banking activities are in full swing in all states, so that donor families can fulfill the wishes of their loved ones,” she concluded. Eye bank training efforts, as well as training corneal surgeons and general ophthalmologists are crucial to this noble cause, and India, in general, is a major player.
Editor’s Note:
The DREAMCON AIOC 2022 was held as a physical show on 2-5 June at the JIO World Centre in Mumbai, India. Reporting for this story took place during the conference. Media MICE is the Official Media Partner of AIOC 2022. A version of this article was first published on piemagazine.org.
| June/July 2022 27
Pearls from Surgical Retina Case Studies

Highlights from AOS Virtual 2022
 by Joanna Lee
by Joanna Lee
Vulnerable to many possible types of diseases and injuries, the retina is a hotbed of potential issues. Anything from genetic factors to even cosmetic procedures could serve as a risk. Learning from case studies is vital to prevention (where possible), but also for better diagnoses and subsequent treatment decisions. This was demonstrated by the following cases presented at the recently held 5th ASEAN Ophthalmology Society Virtual Congress (AOS Virtual 2022).
FAF helps in Stargardt’s disease diagnosis
No one could’ve suspected anything was amiss when a 7-year-old patient came in for a regular eye check-up. Dr. Thanaporn Kritfuangfoo from Ramathibodi Hospital in Bangkok, Thailand, presented the case of a juvenile macular atrophy patient.
The girl’s visual acuity was 20/40 in the right eye and 20/50 in the left. With no previous medical history or any medical complaint, the fundus photo of her right eye was clear and the vasculature was normal without any macular edema. Her left eye also appeared normal. The OCT showed nothing remarkable.
However, the fundus auto-fluorescence (FAF) revealed a subtle increase in foveal autofluorescence while maintaining a small central zone. An infrared autofluorescence (IRAF) showed speckled hypo-autofluorescence lesions in the macula area, Dr. Kritfuangfoo said.
Differential diagnoses considered
Congenital infections such as rubella
were suspected, and toxic or laser maculopathy were considered, but a deeper probe quickly ruled out these diagnoses. In the end, retinal dystrophy became the chief suspect of her subnormal vision.
“We found that her older sister also had a vision problem in both eyes which improved with eyeglasses. Her sister’s fundus photo showed bilateral yellowish flakes at the posterior pole,” said Dr. Kritfuangfoo.
Widefield FAF showed disseminated speckled hypo- and hyperautofluorescence corresponding with the chorioretinal atrophy and the flakes; the initial foveal atrophy presented as well. The OCT scan showed the outer retinal lamination was thinning at the central area. Finally, the disruption of the ellipsoid zone was more pronounced in this patient.
“All of the above are the typical features of Stargardt disease,” she said.
Stargardt disease is an inherited condition where a slow loss of central vision occurs in both eyes.1 It is a type of macular atrophy that affects juveniles and youths. There is currently no cure for it.2
Dr. Kritfuangfoo learned that patients with a suspected early onset of Stargardt disease should undergo FAF or OCT.3 “These diagnoses should also be confirmed by screening for the presence of the ANCA4 gene mutation.”
Occlusive retinal vasculitis in SLE
Ocular manifestations are a marker for systemic lupus erythematosus (SLE),
a chronic, auto-immune, connective tissue disorder that affects multiple organ systems, often with a relapsing and remitting clinical course. The manifestations can occur in up to onethird of all SLE patients.
In a case study of a male patient, Dr. Carlo Ladores from the University of Santo Tomas Hospital, Manila, Philippines, detailed the progressive proliferation of occlusive retinal vasculitis. The patient had reported a progressive blurring in both eyes ever since his diagnosis of SLE in 2008. This led him to wear prescription glasses in 2015 which improved his vision. It was only in 2019 that he consulted with Dr. Ladores for dimming vision in his right eye. After having detected ischemic areas on FA and cystoid macular edema on OCT, the patient was advised to undergo PRP for the right eye and also intravitreal anti-VEGF treatment.
Unfortunately, the patient experienced a seizure and was admitted to the hospital. He also had an SLE flare, potential TB lymphadenitis, and was in an immunocompromised state.
Further investigations led Dr. Ladores to question if the patient had a more severe form of SLE — one associated with antiphospholipid antibody which is thrombogenic and is linked with CNS and ocular vaso-occlusive events (Hughes’ retinopathy).
A 1999 study by Montehermoso and colleagues4 mentioned that 77% of patients with SLE and retinal involvement had positive antiphospholipid antibody titers, whereas only 29% of SLE patients without retinal disease had positive titers. Subsequently, this patient tested positive for antiphospholipid antibody. “The occurrence of retinopathy in SLE due to antiphospholipid-induced retinal vascular thrombosis constitutes Hughes’ retinopathy,” said the study authors.
| June/July 2022 28 AOS VIRTUAL 2022 CONFERENCE HIGHLIGHTS
As a general practice, lupus retinopathy is treated by control of systemic SLE with systemic corticosteroids and immunosuppression. However, even with improved appearance of retinopathy after systemic treatment of SLE, good visual recovery is seldom achieved which may be attributed to the presence of retinal ischemia.
Indirect laser was the best treatment at that time for the patient due to the seizure in order to prevent neovascularization and risk of hemorrhage, Dr. Ladores said.
In conclusion, occlusive retinal vasculitis rarely occurs in SLE retinopathy. “When it develops, it is a sign of active SLE. Visual loss or even death may occur if left unevaluated or untreated. Systemic SLE control is essential. However, visual recovery is infrequent. Thus, the target of therapy remains the prevention of further vascular events,” concluded Dr. Ladores.
Laser accidents: Points to ponder
In another interesting case, Dr. Panisa Singhanetr from Mettapracharak Hospital, Thailand, encountered a 28-year-old Thai female patient who presented with acute vision loss after a Y10G mode laser was accidentally fired into her right eye for 1 second. She was cleaning the probe after usage and was not using any protective devices.
Two days after the incident, she only had perception of light (PL) in her right eye while her left eye was 20/20. A fundus examination of her eye showed a whitish burn lesion at the foveal center of the right eye.
The patient was diagnosed with laser-induced maculopathy and was prescribed a few oral steroids and timolol eye drops. After 5 days, her right eye improved to 20/80 but blurred vision persisted. An OCT of the patient’s macula showed there was focal subfoveal hyperreflectivity. There are also outer IS/OS disruptions subfoveally in the right eye with the RPE intact, while no abnormalities were seen in the OCTA.
On fundus autofluorescence (FAF), a faint hyper-autofluorescent is seen at the
foveal center correlating with the yellow lesion on the earlier fundus examination. Her medication was then discontinued.
After 6 weeks, a color fundus photo of her right eye showed the yellow focal burn at the center could barely be seen while her best corrected visual acuity improved to 20/20. On OCT, the hyperreflective band is gone and the slight yellow hyperfluorescence on FAF also faded.
The laser used in this case was the dual yellow multi-wavelength copper bromide laser used in cosmetic clinics. Dr. Singhanetr said the increasing popularity of cosmetic laser procedures of the face have also seen a rise in the number of reported ocular injuries. “Most of the reported injuries were from laser hair removal of the periorbital areas, in particular with 755 nm alexandrite and diode (800-983nm) lasers,” said Dr. Singhanetr.
Closing eyes is not protective enough
A majority of the ocular injuries are associated with cosmetic lasers involving the anterior segment, highlighted Dr. Singhanetr.
The eye contains pigment-rich chromophores (RPE and iris) that absorb laser energy. Eyelids are too thin to sufficiently protect the eye from any injuries. “Therefore, having your eyes closed during laser treatment is not enough,” she explained.
The injuries sustained are immediately apparent. About 62% of the reported cases did not have proper eye protection. In addition, the retina — and in particular the macula — is most susceptible to laser injuries. The extent of injuries depends on the laser’s wavelength, duration of exposure, and method of administration.
Laser injuries from laser pointers are common among adolescents; cosmetic injuries occur often among an older age group, with scotomas, a sudden decrease in vision and foveal pigmentary change, macular holes, as well as subhyaloid hemorrhage observed on fundus. It was also noted that the Nd:YAG laser causes photodisruption
and she said that many cases from this laser were linked with vitreous hemorrhages.
Most laser induced ocular injuries will improve spontaneously without treatment,5 but permanent vision loss and scotoma may last. Generally, 55% of eyes recovered to a visual acuity of 20/25 or better within a few months and 36% recovered to a VA of 20/100 or 20/30.6
Prevention and awareness are most important for laser injuries as there aren’t any evidence-based treatments so far. “Wavelength-specific goggles or spectacles should be worn during procedures and should not be taken off until the end of the procedure,” she said.
Eye protection should be essential, even for observers and operating personnel. There should also be sufficient cooling of treated areas to prevent overheating of metal corneal shields and treating physicians operating lasers should be properly trained, Dr. Singhanetr concluded.
References:
1. Tsang SH, Sharma T. Stargardt Disease. Adv Exp Med Biol. 2018;1085:139-151.
2. National Eye Institute, Stargardt disease. National Eye Institute. Available from: https://www.nei.nih. gov/learn-about-eye-health/eye-conditions-anddiseases/stargardt-disease Accessed 24 April 2022.
3. Khan KN, Kasilian M, Mahroo OAR, et al. Early Patterns of Macular Degeneration in ABCA4Associated Retinopathy. Ophthalmology. 2018;125(5):735-746.
4. Montehermoso A, Cervera R, Font J, et al. Association of antiphospholipid antibodies with retinal vascular disease in systemic lupus erythematosus. Semin Arthritis Rheum. 1999;28(5):326-332.
5. Wong EWN, Lai ACH, Lam RF, Lai FHP. Laserinduced ocular injury: A narrative review. Hong Kong J Ophthalmol. 2020;24(2):51-59.
6. Barkana Y, Belkin M. Laser eye injuries. Surv Ophthalmol. 2000;44(6):459-478.
Editor’s Note:
The 5th AOS Congress was held virtually on March 26-27, 2022. Reporting for this story took place during the event. A version of this article was first published on piemagazine.org.
| June/July 2022 29
The 8th AIOS-OPL 2022

Teamwork and Camaraderie Trumped Competition
The Ophthalmic Premiere League (OPL) — the All India Ophthalmological Society’s team video competition on managing ophthalmic complications — is in its 8th year and was held on Day 3 of the 80th Annual Conference of the All India Ophthalmological Society (AIOS) in Mumbai, Maharashtra (a.k.a. DREAMCON AIOC 2022).
OPL is the ophthalmic version of the Indian Premiere League games and this year, four teams competed:
Team 1: Delhi Divide and Conqueror (Dr. Mahipal Sachdev, Dr. Kumar Doctor, Dr. Chitra Ramamurthy and Dr. Ajoy Paul)

Team 2: Mumbai Multifocals
(Dr. T. P. Lahane, Dr. Rohit Om Parkash, Dr. Gaurav Luthra and Dr. Sonu Goel)
Team 3: Kolkata Knightriders
(Dr. Partha Biswas, Dr. Namrata Sharma, Dr. Priya Narang and Dr. Samar Basak)
Team 4: Chennai Capsules
(Dr. Mohan Rajan, Dr. Rajini Parekh, Dr. Rajesh Sinha and Dr. Arun Kshetrapal)
It would have been just another series of surgical video presentations to share clinical pearls to both peers and students … but when presented creatively in Bollywood or K-pop MTV-
by Gloria D. Gamat
style, the audience witnessed the bantering and teasing among India’s prime ophthalmic surgeons. It was a display of entertaining competition among colleagues who, in real life, are obviously close friends too. The captive audience was hanging on to each video presentation like their favorite series on Netflix. Talk about binging on eye surgery for two hours! And that’s what matters most: The sharing of clinical pearls that young ophthalmologists can get from their teachers and mentors was the main objective.
Moderated by Dr. Amar Agarwal, each team member had four minutes each to show a video and narrate the story of how they managed one complicated ophthalmic case after another: 1) a special case of bilateral corneal transplant on a pregnant woman; 2) fibrosed and calcified cortical matter firmly adhered to the anterior and posterior capsule; and 3) a bad case of white cataract during COVID-19 lockdown; among others.
Each team was given 18 minutes in total to present their most complicated surgical case. The audience and the panel of judges were given time to discuss each presentation, and by the end of the exercise, both audience and panel were expected to vote for the “Best Entertainer,” “Best Video” and “Best Team.”
But this year is special. After all, the 80th AIOC is the first physical conference in Asia, post-pandemic. We all know India had its share of dark periods related to COVID-19.
And being among the delegates here in Mumbai is a celebration of life in itself. Like surviving a war, the atmosphere is hopeful that ophthalmology (as in any other industry) is getting back on its feet and is feeling positive that everything will get better in the world again. So it wasn’t surprising when the competing OPL teams, instead of nitpicking each other’s work, turned into a comedy show (almost!).
Team Chennai Capsules was quite the entertainer. A little bit of “healthy bashing” went on here and there … but it was all delivered in ophthalmic language, of course. The room was full of eye doctors, so obviously, they all got the joke. The captain of Chennai Capsules, Dr. Mohan Rajan, was voted Best Entertainer.
The Best Video award went to Dr. Namrata Sharma, the captain of Team Kolkata Knightriders. The video, entitled Two Eyes Two Lives, is a case of a 28week pregnant patient who underwent bilateral sclerokeratoplasty using the same donor. The patient was initially not cleared for surgery due to the high chance of preterm labor. But it was a team effort that prevailed to see the patient through. In the end, not only two eyes were saved, but it was also the beginning of a new life. The mother gained her vision back and her baby survived the ordeal.
Finally, the judges and audience couldn’t decide on a single team to win the Best Team award. In the end, everyone got the Best Team award, and shared the trophy.
Editor’s Note:
The DREAMCON AIOC 2022 was held as a physical show on 2-5 June at the Jio World Centre in Mumbai, India. Reporting for this story took place during the conference. Media MICE is the Official Media Partner of AIOC 2022. A version of this article was first published on cakemagazine.org.
| June/July 2022 30 AIOC 2022 CONFERENCE HIGHLIGHTS













2.0 AN ASIAN-BASED, GLOBALLY-MINDED FUNKY OPHTHALMOLOGY AND OPTOMETRY EYE CONFERENCE A unique hybrid show with hundreds of in person guests, international speakers and exhibitors as well as engaging and entertaining coverage available online.
POINTS BY SHERATON DA NANG, VIETNAM 20TH & 21ST 2022 AUGUST Scan here or visit www.mediamice.com/expo for more information. FREE FOR DOCTORS TO ATTEND Email expo@mediamice.com to register.
FOUR
Complex Vitreoretinal Surgery Cases

Highlights from Retinawesome 14 , Part 2
by April Ingram
Following the presentation of keynote speaker Dr. Subhara Jalali’s elegantly performed surgical cases, attendees at Retinawesome 14 could not help but be inspired to become pediatric retina surgeons! Panelist Dr. Vaibhav Sethi, asked Dr. Jalali what approach adult surgeons should take to make the shift. She shared that all her trainees assist in all the cases that she does, as well as preoperatively under anesthesia, examine everything, make the drawings and write down their findings, and then compare with what the faculty is drawing. She feels that the more drawing they do, the better they understand, even now with Optos [retinal imaging systems by Optos], they
can study the images in great detail. She does not recommend starting the foray into pediatric surgery on one-eyed patients, but does start with the bad cases, where the amount of damage can be limited and slowly progress. “If I can do it, anybody can do it, anybody who is doing other retinal surgeries can do it, it's only a question of focus,” said Dr. Jalali.
On retinal cases with anterior segment complications
Dr. Andre Juca presented his case of an implantable collamer lens (ICL) and
pucker removal in high myopia. The patient was 32-years-old, had a 20D ICL, and 15 days later, a retinal detachment. Dr. Juca had to remove the ICL, the lens, the cataract and the pucker. The patient did not want the ICL removed, she had just spent so much money for it! Dr. Juca shared the surgical video, and accompanying OCT. He used trypan to stain the anterior capsule. The ICL was not difficult to remove, it was cut to the middle and turned, and cut again, like a pizza, but getting a good refractive result was going to be the challenge. Then Dr. Juca performed a lensectomy, highlighting how deep the anterior chamber was in this patient with 20D of myopia. He inserted an Alcon

| June/July 2022 32 RETINAWESOME 14 CONFERENCE HIGHLIGHTS
lens, and noted that a multifocal lens would not be good for this patient. The video proceeds, trypan blue was used to visualize the internal limiting membrane and then the epiretinal membrane is removed, as well as the vitreous attachments. Laser was applied to the periphery to prevent micro holes. Followup at 15 and 45 days showed a good result: The membrane was gone and the refractive result was also good for long distance vision. Dr. Juca highlighted the take-home messages from this case: Use the Barrett II formula; the ICL lens does not interfere with the calculation; and vitreoschisis is quite often present in highly myopic eyes; and they use triamcinolone in removal.

Dr. Wei Ching Lam agreed: Operating on high myopia is always a challenge and the vitreoschisis over the macular area can be hard to grasp. He often uses the Tano diamond-dusted membrane scraper to go over the macular area and remove vitreous. Dr. Juca noted that for this particular case, they used the probe to reach the back of the eye, but in some cases if the surgeon does not have special forceps, he recommends removing the trochanter and going a bit back to reach the posterior pole in long eyes. Surgeons discussed how to best visualize membrane peeling, and most prefer to use a contact lens; but it isn’t possible to reach the far periphery with a contact lens, so the BIOM (an ophthalmomicroscope for vitreoretinal surgery by Oculus) or NGENUITY (a 3D visualization system by Alcon) provide good stereopsis. Additionally, it was noted that for epiretinal membranes, brilliant blue is not appropriate and trypan blue is preferred.
Dr. Emmanouil Mavrikakis presented his case of a 40-yearold male with reduced vision (hand movements) for two days, fellow eye 20/30. Patient is a high myope (-8.00) with no other pertinent medical or ocular history. Dr. Mavrikakis performed a vitrectomy, clearing the vitreous hemorrhage and used the cutter to remove the anterior part
of the break in order to minimize any PVR or postoperative issues and relieve any traction. He marked the edge of the giant tear with endodiathermy, then used scleral depression to remove any remaining vitreous or redundant retinal tissue. He shared that he does not like PFO so he will reattach using air-fluid exchange. “Stay as anterior as possible and be very patient to remove the fluid under the retina so the giant tear is completely unfolded. Be patient and keep draining the fluid, and it will happen.” He then lasered the posterior part of the break, continuing to the anterior, then used cryo on the horns of the tear. Immediately postoperatively the patient was put on his left side for two hours to drain as much fluid temporally as possible, then face down. Five months post-op, the visual acuity is 20/60 and the macula is flat on OCT.
Dr. Jalali agrees, she transitioned from air to PFO and now back to air, because if one small bubble goes behind, “it's like the mosquito who just keeps on troubling you, it's a pain to remove and you don't want to leave it behind. PFO can flatten the retina, but there is vitreous left in the periphery or areas of lattice and it comes back under the oil to punish you. Air tells you the truth.”
The next case was presented by Dr. Marcelo Murillo Sasamoto. An 18-yearold male, high myopia, pseudophakic, who underwent a vitrectomy. There was fibrosis of the posterior capsule and inferior retinal detachment. Dr. Sasamoto uses triamcinolone to recognize the vitreous. He showed aspiration of the subretinal fluid, but highlighted how they attached the retina, using laser to recognize the detachment and the holes that are located inferiorly, followed by air-fluid exchange. Then they removed the trocars and put on a scleral buckle, followed by silicone oil. Dr. Sasamoto explained that there are a lot of procedures and techniques in these patients, but the scleral buckle is useful in this type of patient. Dr. S. Natarajan added that this was a very nice clean case, but added that before the oil is injected, it is important to ensure that all the vitreous is removed.
A case for inverted vitrectomy
Dr. Hudson Nakamura wowed the audience with the video of his surgical Inverted Vitrectomy, discussing a double stain with brilliant blue for taut macular pucker. The eye is phakic with a clear lens, and
| June/July 2022 33
“It's like the mosquito who just keeps on troubling you, it's a pain to remove and you don't want to leave it behind. PFO can flatten the retina, but there is vitreous left in the periphery or areas of lattice and it comes back under the oil to punish you. Air tells you the truth.”
— Dr. Subhara Jalali
he performed a core and peripheral vitrectomy using a wide-angle vitrectomy contact lens for visualization. The hyaloid is detached to free the macula from vitreous adhesions. Being careful of the patient’s phakic lens, the eye is rotated with the curve and illuminating pipe toward the far periphery. Using a macular lens, there is a clear view from the fundus for a complete airfluid exchange, allowing for enough brilliant blue to be injected toward the macular surface for a good staining. Dr. Nakamura noted that there is no need to pass the arcades for staining. The adherent and taut epiretinal tissues were removed after grabbing an edge that was carefully lifted from temporal to nasal, the pucker is peeled off, freeing up the macula. He highlighted the vascular traction and tortuosity at vessel sites, visible on the video. Again, brilliant blue staining is carried out, this time without an air-fluid exchange. Here, the remains of the adherent tissues and astrocyte proliferations were shaved, using careful controlled force, cleaning the macular surface. This is not an easy task! Endolaser was applied to protect the
peripheral retina at the sclerotomy sites. Dr. Nakamura shared the postoperative FA imaging and OCT: The macula looks good, but the eye has some chronic traction and intraretinal cysts, which can be treated with anti-angiogenics.
Dr. Jalali noted that when the patient is young there is a thick membrane, so it is important to always check the periphery because there can be angiomas or PVR causing this problem. She added that some of the edema may be surgically induced, so she usually waits two months, maybe putting them on topical dorzolamide during that time. If they do not respond during that time, she will move forward with injecting a steroid. The panel agrees that steroids are beneficial, but caution that those eyes with glaucoma may need careful monitoring for steroid response.
Dr. Nakamura also weighed the pros and cons of using gas or silicone oil in an eye like this, noting that there may be a benefit from the mechanical action, but he does not wish to leave

this patient with silicone oil, and gas would require positioning. There is also the complication of cataract if the gas touches the lens. Dr. Anita added that when she does a membrane peel, she does not remove the ILM, unless she encounters a multi-layered epiretinal membrane. She does not use stain for epiretinal membrane, only a bent 25-gauge needle or MVR blade, and she is able to see the edge come up. She advocates for a second stain to see the remaining ILM (only 20-25 seconds), but intraoperative OCT is the best way to ensure everything has been removed. Dr. Anita also keeps patients on topical pred forte for 6 to 8 weeks, which is very beneficial in ischemic diabetic eyes to calm ischemic inflammation.
Dr. Sethi asked the panel to comment on cases where they would prefer observation over surgery. Dr. Lam suggested doing an FFA: If there is petaloid leakage rather than tractional changes, then that case will benefit from steroid first and see how much the intraretinal cystic changes improve. He adds that the epiretinal membrane can cause cystic changes, but they are mechanical and should not leak. Dr. Jalali shared that if the eye is post-uveitic and some inflammation is present, the eye would benefit from Ozurdex (dexamethasone intravitreal implant, Allergan, an AbbVie company) before surgery. Also, if the vision is still good, discuss options with the patient.
Another exciting compilation of cases from Retinawesome 14!
Editor’s Note:
Mark your calendars because you don’t want to miss upcoming Retinawesome events, the last Saturday of every month, or catch them anytime by subscribing to the Retinawesome Retina & Vitreous International channel on YouTube.

| June/July 2022 34
RETINAWESOME 14 CONFERENCE HIGHLIGHTS




| June/July 2022 35 Unique , Smart and A.I. solutions for Eye Care Specialists! FAST Get all the measurements in 3” FRIENDLY Easy-to-use and patients friendly ACCURATE Suitable for adults and kids PORTABLE Take it everywhere www.adaptica.com contact@adaptica.com Adaptica srl: Via San Marco 9/H, 35129 Padova (PD), Italy MA D E IN I TA LY MULTIFUNCTIONAL Widest range of Application SAFE Safety Working distance > 1 m



6001 Beach Road, #19-06 Golden Mile Tower, Singapore 199589 CONTENT MARKETING + ADVERTISING + MEDICAL WRITING Phone: +65 8186 7677 | U.S.: +1 302 261 5379 E-mail: enquiry@mediamice.com Web: www.mediamice.com Request our 2022 Agency Kit Now! Write enquiry@mediamice.com for a copy









































 by Tan Sher Lynn
by Tan Sher Lynn
























 by Joanna Lee
by Joanna Lee


















