AdvancesinExperimentalMedicine andBiology
AdvancesinMicrobiology,InfectiousDiseases andPublicHealth
Volume971
EditorialBoard
IrunR.Cohen,TheWeizmannInstituteofScience,Rehovot,Israel N.S.AbelLajtha,KlineInstituteforPsychiatricResearch,Orangeburg,NY,USA
JohnD.Lambris,UniversityofPennsylvania,Philadelphia,PA,USA RodolfoPaoletti,UniversityofMilan,Milan,Italy
SubseriesEditor
GianfrancoDonelli,MicrobialBiofilmLaboratory,FondazioneSantaLucia IRCCS,Rome,Italy
TheAdvancesinMicrobiology,InfectiousDiseasesandPublicHealthSeries willprovidemicrobiologists,hygienists,epidemiologistsandinfectious diseasesspecialistswithwell-choosencontributedvolumescontaining updatedinformationintheareasofbasicandappliedmicrobiologyinvolving relevantissuesforpublichealth,includingbacterial,fungalandparasitic infections,zoonosesandanthropozoonoses,environmentalandfoodmicrobiology.Theincreasingthreatofthemultidrug-resistantmicroorganismsand therelatedhostimmuneresponse,thenewstrategiesforthetreatmentof biofilm-based,acuteandchronicmicrobialinfections,aswellasthedevelopmentofnewvaccinesandmoreefficaciousantimicrobialdrugstoprevent andtreathumanandanimalinfectionswillbealsoreviewedinthisseriesin thelightofthemostrecentachievementsinthesefields.Specialattentionwill bedevotedtothefastdiffusionworldwideofthenewfindingsofthemost advancedtranslationalresearchescarriedoutinthedifferentfieldsof microbiologicalsciences,withtheaimtopromoteapromptvalidationand transferatclinicallevelofthemostpromisingexperimentalresults.Thebook seriespublishesreviewandoriginalresearchcontributions,short(data) reportsaswellasguesteditedthematicbookvolumes.Allcontributions willbepublishedonlinefirstandcollectedin(thematic)bookvolumes.There arenopublicationcosts.ThisseriesisasubseriesofAdvancesinExperimentalMedicineandBiology2015ImpactFactor:1.953AdvancesinExperimentalMedicineandBiologyhasbeenpublishingexceptionalworksinthe fieldforover30yearsandisindexedinMedline,Scopus,EMBASE, BIOSIS,BiologicalAbstracts,CSA,BiologicalSciencesandLiving Resources(ASFA-1),andBiologicalSciences.
Moreinformationaboutthisseriesathttp://www.springer.com/series/13513
LorenzoDrago Editor
Editor LorenzoDrago DepartmentofBiomedicalSciencesHealth UniversityofMilan
Milan,Italy
ISSN0065-2598ISSN2214-8019(electronic)
AdvancesinExperimentalMedicineandBiology
ISBN978-3-319-52273-9ISBN978-3-319-52274-6(eBook) DOI10.1007/978-3-319-52274-6
LibraryofCongressControlNumber:2016935504
# SpringerInternationalPublishingAG2017
Thisworkissubjecttocopyright.AllrightsarereservedbythePublisher,whetherthewholeor partofthematerialisconcerned,specificallytherightsoftranslation,reprinting,reuseof illustrations,recitation,broadcasting,reproductiononmicrofilmsorinanyotherphysicalway, andtransmissionorinformationstorageandretrieval,electronicadaptation,computersoftware, orbysimilarordissimilarmethodologynowknownorhereafterdeveloped. Theuseofgeneraldescriptivenames,registerednames,trademarks,servicemarks,etc.inthis publicationdoesnotimply,evenintheabsenceofaspecificstatement,thatsuchnamesare exemptfromtherelevantprotectivelawsandregulationsandthereforefreeforgeneraluse.
Thepublisher,theauthorsandtheeditorsaresafetoassumethattheadviceandinformationin thisbookarebelievedtobetrueandaccurateatthedateofpublication.Neitherthepublishernor theauthorsortheeditorsgiveawarranty,expressorimplied,withrespecttothematerial containedhereinorforanyerrorsoromissionsthatmayhavebeenmade.Thepublisher remainsneutralwithregardtojurisdictionalclaimsinpublishedmapsandinstitutional affiliations.
Printedonacid-freepaper
ThisSpringerimprintispublishedbySpringerNature TheregisteredcompanyisSpringerInternationalPublishingAG Theregisteredcompanyaddressis:Gewerbestrasse11,6330Cham,Switzerland
Contents
TheConceptofBiofilm-RelatedImplantMalfunction and“Low-GradeInfection” .............................1 CarloLucaRomano ` ,DeliaRomano ` ,IlariaMorelli, andLorenzoDrago
MechanismsofBacterialColonizationofImplants andHostResponse ....................................15 ChristofWagnerandG.MariaHansch
AnimalModelsofImplant-RelatedLow-Grade Infections.ATwenty-YearReview ........................29 AriannaBarbaraLovati,MartaBottagisio,ElenadeVecchi, EnricoGallazzi,andLorenzoDrago
MicrobiologicalDiagnosisofImplant-Related Infections:ScientificEvidenceandCost/Benefit AnalysisofRoutineAntibiofilmProcessing ..................51 LorenzoDragoandElenaDeVecchi
TheRoleofBiomarkersfortheDiagnosisofImplant-Related InfectionsinOrthopaedicsandTrauma ....................69 AbtinAlvand,MaryamRezapoor,andJavadParvizi AntibacterialBioactiveGlass,S53P4,forChronicBone Infections–AMultinationalStudy ........................81 NinaLindfors,JanGeurts,LorenzoDrago,J.J.Arts, VesaJuutilainen,PekkaHyvonen,ArnoldJ.Suda, AlojDomenico,StefanoArtiaco,ChingizAlizadeh, AdrianBrychcy,JertzyBialecki,andCarloL.Romano ` ProstheticJointInfectionsandCostAnalysis? ...............93 F.S.Haddad,A.Ngu,andJ.J.Negus
AlgorithmtoDiagnoseDelayedandLatePJI:Role ofJointAspiration ....................................101 OlivierBorens,PabloS.Corona,LarsFrommelt,StergiosLazarinis, MichaelRichardReed,andCarloLucaRomano
Erratum:MicrobiologicalDiagnosisofImplant-Related Infections:ScientificEvidenceandCost/BenefitAnalysis ofRoutineAntibiofilmProcessing .........................113 LorenzoDragoandElenaDeVecchi
Erratum:AntibacterialBioactiveGlass,S53P4, forChronicBoneInfections–AMultinationalStudy ..........115 NinaLindfors,JanGeurts,LorenzoDrago,J.J.Arts, VesaJuutilainen,PekkaHyv€ onen,ArnoldJ.Suda, AlojDomenico,StefanoArtiaco,ChingizAlizadeh, AdrianBrychcy,JertzyBialecki,andCarloL.Romano `
Index
AdvExpMedBiol-AdvancesinMicrobiology,InfectiousDiseasesandPublicHealth(2017)5:1–13
DOI10.1007/5584_2016_158
# SpringerInternationalPublishingSwitzerland2016
Publishedonline:19October2016
TheConceptofBiofilm-RelatedImplant Malfunctionand“Low-GradeInfection”
CarloLucaRomano
` ,DeliaRomano ` ,IlariaMorelli, andLorenzoDrago
Abstract
Biofilmshaveatremendousimpactonindustrialmachinesworkingin moistenvironments,whileinbiologicalsystemstheireffectisfurther complicatedbythehost’sresponse.
Implant-relatedinfectionsareacomplexprocess,startingwithbacterialadhesionandbiofilmformation,followedbythevariableinteraction betweenhost,implant,microorganismsandtheirby-products.Depending onthebalanceofthesefactors,differentclinicalpresentationsare observed,whichmayeventually,attimes,shiftfromoneintotheother.
–“Implantmalfunction”displaysonlymildclinicalsigns/symptoms–light painand/orslightsofttissuecontractureorfunctionalimpairment–with negativeinfection/inflammatorymarkers;itrequiresprolongedcultures, antibiofilmandeventuallygenomicinvestigationsforpathogendetection; –“Low-gradeinfection”featuresrecurrentorpersistentpainand/orsoft tissuecontracturewithvariousfunctionalimpairmentandmixedpositive/ negativemarkersofinfection/inflammation;pathogenidentification requiresprolongedculturesandantibiofilmtechniques;
–“High-gradeinfection”displaysclassicalsigns/symptomsofinfection/ inflammationwithpositivetests;pathogenidentificationisoftenpossible withtraditionalmicrobiologicaltechniques,butisbetterachievedwith prolongedculturesandantibiofilmprocessing.
C.L.Romano ` (*)
CenterforReconstructiveSurgeryofOsteoarticular Infections(CRIO)andMilanoBiofilmCenter(MBM), I.R.C.C.S.,GaleazziOrthopaedicInstitute,Milan,Italy
e-mail: carlo.romano@grupposandonato.it
D.Romano ` andI.Morelli
CenterforReconstructiveSurgeryofOsteoarticular Infections(CRIO),I.R.C.C.S.,GaleazziOrthopaedic Institute,Milan,Italy
e-mail: delia-romano@libero.it; ilaria.morelli@gmail.com
L.Drago
LaboratoryofClinicalChemistryandMicrobiologyand MilanoBiofilmCenter(MBM),I.R.C.C.S.,Galeazzi OrthopaedicInstitute,Milan,Italy
e-mail: lorenzo.drago@unimi.it
Understandingbiofilms-relatedclinicalpresentationsiscrucialfor physicians,toimplementthebestdiagnosticandtherapeuticmeasures, andforregulatorybodies,todefinetheevaluationprocessoftechnologies aimedatreducingimplants’malfunctionsandinfections,likeantiadhesiveandantibiofilmcoatings,thatshouldberegulatedas(partof) medicaldevices,requiringasuitablepost-marketingsurveillance.
Onlyaneffectiveantibiofilm-targetedapproachfromallplayerswill hopefullyallowthemedicalcommunitytomitigatethecurrentunacceptablesocialandeconomicalburdenofimplant-relatedinfectionsand malfunctions.
Keywords
Biofilm•Implant•Malfunction•Infection•Low-gradeinfection
1Introduction
Biofilmsaredefinedascommunitiesofmicrobial cellsandintercellularmatrix,attachedto surfacesinmoistenvironments,whilebiofouling orbiologicalfoulingistheaccumulationof biofilmsonwettedsurfaces.
Leeuwenhoek(1684),usinghisprimitive lightmicroscope,foundmicrobesattachedto toothsurfaces,formingsessilecommunities, whichcouldbeconsideredasthefirstobservationofmicrobialbiofilms.
Biofilmsareprobablytheprevalentmodeof lifeformicroorganismsinnature,butitwas notuntilthe1920sthattheconceptofbacterial biofilmswasformulated.Angst(1923)observed thatthenumberofmarinebacteriaonthesurface ofshipshullswashigherthanthesurrounding floatingcells,andproposedthatbacterial biofilmsledtoseriouscorrosionsofshipshulls. Bythe1980s,bacteriawereobservedonthesolid surfacesofmanyecologicalenvironments includingwastewatertreatmentsystems,industrialwatersystems,equipmentusedtomanufacturevinegar,etc.
Nowweknowthatthegeneralprinciplesof biofilmformationandfactorsleadingtosettlementonhardsurfacesaresimilarinmedical, marineandindustrialapplications(Bixler etal. 2014).
Intheindustrialandmarinesettingbiofouling hasawellknownimpactonperformance,
causingbiodegradation,malfunctionandfinally energyloss(Fig. 1)andhundredsbilliondollar costseveryyear.
Intheindustrialenvironment,biofoulingis oftenassociatedwithmicrobiologically influencedcorrosion(MIC)orbiocorrosionand inorganicfouling.Biocorrosionisthedegradationofmaterials,usuallymetals,duetotheactivityofmicroorganisms(Table 1),whileinorganic foulingistheaccumulationofnonbiological particles,thatmayforminadditiontoor indipendentlyfrombiofouling.
Ontheotherhand,inthemedicalsetting,and particularlyinimplant-relatedinfections,the effectsofbacterialadhesionandbiofilmformationontissuesandimplantedbiomaterialsare complicatedbythevariablehost’sresponse, whileconsequencesareequallydevastating.
Infact,itisestimatedthat20%offatalities worldwideareduetoinfectiousdiseases,of whichapproximately80%arebiofilm-related (Harrisonetal. 2005;Prenticeetal. 2004), includingthegreatmajorityofchronicbacterial andfungalinfectionsandvirtuallyall biomaterials-associatedinfections(Gristina etal. 1990;Romano ` etal. 2014;Stoodley etal. 2011;Nanaetal. 2016).
Thepresenceofbiofilmsmakesboththediagnosisandtreatmentparticularlychallenging (Dragoetal. 2013;Romano ` etal. 2013a, b), giventheprotectionofferedbytheextra-cellular matrixtothemicroorganisms.Infact,bacterial
Fig.1 Effectofbiofoulingonworkingmachinesand systems.Maindetrimentaleffectsonperformanceconcernimplantsdegradationandmalfunctionandenergy
loss.Theeconomicalimpactintheindustrialfieldranges from10to >30%oftheoperatingcosts,dependingon differentsettingsandreports(seetextformoredetails)
Table1 Mechanisms(CoetserandCloete 2005)andprocesses(Leeetal. 1995)underlyingMicrobiological InfluencedCorrosion(MIC)intheindustrialenvironment
Mechanisms
Utilizationofoxygenbyaerobicorganismsresultinginanodicareas.Localizeddifferencesinconcentrationshiftthe potentialofmetalsurfacesresultinginthecreationoflocalizedcorrosioncells.
Utilizationofhydrogenbymicroorganismsviaacathodicreactiondepolarizesthecathodewhichincreasestherateof metallossattheanode
Microbialdegradationofprotectivecoatingsonmetalsurfaces
Microbialdegradationofcorrosion-inhibitingchemicalsaddedtoprotectmetalsinindustrialwatersystems–corrosionorscalinginhibitors
Microbialproductionofmetaboliteswhicharecorrosiveorganicandinorganicacidsareoftenend-productsofthe metabolismofmicroorganisms
Metabolicby-productssuchasH2Scanprecipitatemetalions,suchasirontoformFeS,whichiscorrosiveitself.
Processes
Transportandaccumulationofmaterialsfromthebulkliquidtothemetalsurface.Thesematerialscanbesoluble (microbialnutrientsandcorrosivechemicals)orparticulate(viablemicroorganismsorinorganicparticles)
Microbialandelectrochemicaltransformationprocesseswithinthebiofilmandthemetalsurface.Microorganisms excreteextra-cellularpolymers,whichcontributetothebiofilmdepositandpromoteadherenceofcorrosionproducts. Microbialtransformationprocessesinfluencethecorrosivityofthemicroenvironmentatthebiofilm-metalinterface. Abioticprocessesinfluencetherate,extent,anddistributionofcolonizingmicrobialspecies,aswellasthechemical compositionandphysicalpropertiesoftheresultingbiofilm.
Erosionanddetachmentfromthesurfaceofthefilm.Theseprocesseslimittheoverallextentoffoulingdeposit accumulation.
slimenotonlyreducestheimmunesystemability tofightinfections,butmayincreaseantibiotic resistancebymorethan1000times;inlinewith thisobservation,introducingantibiofilm strategiesshouldprobablyberegardedasabetter responsethaninvestinginnewantibioticsin ordertoovercomethealarmingincreasingantibioticresistanceworldwide(WHOReport, 2014).
Bothintheindustrialandinthemedical settings,theprocessfrombacterialadhesionto
theprogressivelossofperformanceisinfluenced bymanyvariablesandtheimpacttotheoverall performanceoftheaffecteddeviceorsystem mayrangefromadifficult-to-detectlightmalfunctiontoaseverefunctionalimpairment.
Here,afterabriefreviewoftheimpactof biofoulingonindustrialsystems,wefocuson implantedbiomaterials,introducingtheconcept ofbiofilm-relatedimplantmalfunction,low-and high-gradeinfection,withitspossiblepractical implications.
2BiofoulinginIndustry andWorkingMachines
Industrialbiofoulingandbiocorrosionis estimatedtocosttogovernmentsandindustries overtwohundredsbilliondollarsperyear (Colauttietal. 2006;Schmitt 2009;Schultz etal. 2011).
Microbialbiofilmscontaminateandclog waterandwaterfiltrationunits(affectingdrinkingwater,wastewater,desalinizationandindustrialcoolingwater)(ChmielewskiandFrank 2003),corrodeandblockpipelinesandinterfere withoilandgasextractionprocesses,affecting severalindustrialsystemsandmanufactures (Table 2).
Industrialprocesswaterorpotablewateris notsterile,sothereisbiofilminallsystemsthat isinherentlypresentwithoutcausingproblems. Problemsoccurwhenthebiofilmbuildsup,creatingdeadbiomassandthereforeanutrient sourcethatleadstore-growthoforganismsin thewater.Biofilmstructuresvaryaccordingto flowconditionsinawatersystem,forexample,a turbulentflowproduceshomogeneousandslimy biofilms,whicharehardertoinactivatethanbiofilmproducedbylaminarflows.Alsotheeffectivenessofadisinfectantorbiocidedependson theageofthebiofilmaswellasitsparticular physicalandchemicalstructure.Thepresent trendinindustrialwatersystemsistominimize bothwaterconsumptionandwaterdischargeby recirculation.Thisresultsintheconcentrationof dissolvedandsuspendedsubstancespromoting
Table2 Listofsomeofthemainindustrialand manufacturingsectorsinwhichbiofoulinghasamajor impactonperformanceandefficiency
Waterproductionandpipelines
Foodandbeverageindustry
Petrochemicalindustry
Pharmaceuticalandcosmeticmanufacturing
Shippingindustry
Heatexchangerandcoolingsystems
Paperproduction
Automotiveindustry
Steelproduction
Nuclearandhydro-electricplants
growthofwaterbornemicrobes,andshiftingthe microbialcommunitytoamorecopiotrophic state.
Theoilindustryhascitedmanyproblems resultingfrombiofilmformationbysulphatereducingbacteria.Examplesincludepipeand rigcorrosion,blockageoffiltrationequipment andoilspoilage(Voordouwetal. 1996).
Thepresenceofbiofilmsiscommoninfood industry.Biofilmscanexistonalltypesof surfacesinfoodplantsrangingfromplastic, glass,metal,wood,tofoodproducts (ChmielewskiandFrank 2003),causingserious engineeringproblemssuchasimpedingtheflow ofheatacrossasurface,increasesinfluidfrictionalresistanceofsurfacesandincreasesinthe corrosionrateofsurfacesleadingtoenergyand productionlosses(VerranandJones 2000).Pathogenicmicrofloragrownonfoodsurfacesandin processingenvironmentscancross-contaminate andcausepost-processingcontamination (GaneshandAnand 1998).Ifthemicroorganisms fromfood-contactsurfacesarenotcompletely removed,theycanleadtomaturebiofilmformationandsoincreasethebiotransferpotential. Examplesofthefoodsectorsthatpayparticular attentiontothepossibilityofcrosscontaminationarethemilkindustry(Chye etal. 2004)andtheslaughterindustry(Petrak etal. 1999).Agriculturalcropsarealsonegativelyimpactedbycertainpathogenicmicrobial biofilms,whichcause“blights”andotheragriculturaldiseasethatcanruincrops.
Themostcommonfoodbornebiofilm producersbelongtothegenera Pseudomonas spp., Listeria, Salmonellaspp.,Escherichia coli,Enterobacter, Flavobacterium,Alcaligenes, Staphylococcus,Bacillusspp.,etc. (Chmielewski andFrank 2003;ShiandZhu 2009).
Inthemarineshippingindustry,biofilmsthat formonshiphullsleadtocorrosionandcause “drag,”whichresultsinmuchhigherconsumptionoffuelduringtransportaswellashighership hullmaintenancecosts(Flemming 2011; Kamino 2013).Itisinfactestimatedthat 25–50 μmbiofilmsonashiphullincreasehydrodynamicdragby8–22%respectively,withan increaseinfuelconsumptionthatmayraiseupto
40%andadditionalgreenhousegasproduction (estimatedtobe384milliontonnesperannum) (Townsin 2003;Schultzetal. 2011).
Theinfluenceofbiofoulingoncoastaland oceanographicmeasuringinstruments,which areroutinelyusedinmarineandcoastalresearch andmonitoringprograms,isverystrongandthe earlieststagesofbiofouling,withinafewdaysof immersion,significantlyaffectdataqualityand instrumentperformance.Thereisaneedtoprotecttheinstrumentsfrombiofoulingsothatthey areabletogatherbetterqualitydataandrequire lessmaintenance.Currentlytherearenoeffectivecoatingstocontrolthisproblem,theonly solutioninvolvesexpensivemanualcleaningby divers.
Biofoulingofintakestructures,screens,seawaterpipingsystemsandheat-exchangertubes indesalinationandpowerplantscausesanoveralldeclineinplantefficiencyatgreateconomic cost.Forexamplethepresenceofabiofilmon transfersurfacesofheatexchangerscooledby seawaterreducestheheattransferrateby 20–50%andincursaglobalexpenditureof over$15billionsperannumtocontroltheproblem.Themajorityofcurrentmeasurestocontrol biofoulinginvolvetheuseofbiocides.
Intheareaofmembranetechnology, microfiltrationandultrafiltrationmembranesare usedfordrinkingwaterproductionandwastewatertreatment.Theprimarylimitationtothemore widespreadadoptionofmembranefiltrationis foulingwithmicroorganismsandorganic moleculeswhichleadstoasignificantdecline ofthepermeateflux,higherenergyconsumption, andeventually,failuretomeettheregulatory standards(FlemmingandSchaule 1988).Frequentcleaningofthemembranesiscostlyand maydamagethemembranematerials/barrier layers(Flemming 2009).
3BiofilmsandBiofouling inImplantedBiomaterials
Thepresenceofaforeignbody,suchasanorthopedicimplant,hasbeenshowntosignificantly increasesusceptibilitytoinfection.Whilethisis
atleastpartiallyduetoalocallyacquiredgranulocytedefect,biofilmformationisofmajor importance(Zimmerlietal. 1982;Costerton etal. 1995).
Adherenceofmicro-organismstothesurface oftheimplantinvolvesrapidattachmentbyspecific(e.g.adhesins)ornon-specificfactors (e.g.surfacetension,hydrophobicity,andelectrostaticforces).Thisinitialphaseisfollowedby anaccumulativephaseduringwhichbacterial cellsadheretoeachotherandformabiofilm. Depletionofnutrientsand/orwasteproductaccumulationinbiofilmscausesmicro-organismsto enteraslowornon-growing(stationary)state makingthemupto1000timesmoreresistantto mostantimicrobialagentsthantheirplanktonic (freeliving)counterpartsandallowthemto eventuallypersistformonthsoryears(Donlan 2002).
Orthopedicsisamongtheleadingspecialties forimplantedbiomaterials.Inspiteoftheaverageexcellentclinicalresults,implant-related infectionsisthefirstreasonsforfailureinthe first2yearsafterimplant.Evenifcurrentperioperativeinfectionpreventionmethods,like antibioticprophylaxis,havesignificantly reducedtheincidenceofsurgicalsiteinfections, upto2.5%ofprimaryhipandkneejoint replacementandto10%ofrevision arthroplastiescanstillbecomplicatedby periprostheticjointinfection(PJI)(Lentino 2003)(Fig. 2).
Moreover,accordingtorecentanalysis,these figurescouldevenbeunderestimatedandareon therise(Daleetal. 2009).
Thepresenceofbiofilmsandofsessilebacteriaonjointprosthesismakespathogen (s)detectionmoredifficultandoftenleadsto treatmentfailures.Infact,theoccurrenceofPJI isconsideredadevastatingcomplication,often requiringimplantremoval,prolongedhospitalizationandlong-lastingmedicaltreatment,with highmorbidityandpossiblelong-terminfection recurrence(Costertonetal. 1999;Scarponi etal. 2013;Romano ` etal. 2014);PJIhasbeen showntobeassociatedwithmortalityraise (Zmistowskietal. 2013)andelevatedeconomicalandsocialcosts(Kurtzetal. 2012).

Asimilarworryingimpactisassociatedwith biofilm-relatedinfectionsafterosteosynthesisfor fracturefixation(GomezandPatel 2011), pacemakers,cathetersandcardio-vascularprosthesis(Baddouretal. 2010),maxillo-facialsurgery(Prakasametal. 2016),breastimplants (Pittetetal. 2005)andvirtuallyallsurgeries involvingimplantedbiomaterials,withanaverageriskofbiofilm-relatedinfectionrangingfrom 0.5tomorethan20%,depending,amongother variables,onthetypeofimplant,thelengthofthe operation,thedegreeofsurgicalfieldcontaminationandhost’sco-morbiditiesandriskfactors (Table 3).
4Biofilm-RelatedMalfunction, Low-andHigh-GradeInfection andThresholdsforClinical Interference
Intheindustrialsetting,biofoulingworksasan operationaldefinition,referringtothatamountof biofilmsdevelopmentthatinterfereswithtechnical,aestheticoreconomicalrequirements.
Forexample,virtuallyallnonsteriletechnical watersystemsbearbiofilms,butnotallofthem sufferfrombiofouling.Thetermbiofoulingisin factrelatedtotheinterferenceofbiofilmswith theefficiencyandtheperformanceofagiven
machineorasystemandathresholdlevelexists abovewhichbiofoulingbegins.
Inindustry,this“levelofinterference”, illustratedbythecurveproposedmorethantwo decadesagobyFlemmingetal.(1994)(rewritten inFig. 3),isdefinedmostlybyeconomical considerations,connectedtotheextenttowhich biofilmeffectscanbetoleratedwithoutunacceptablelossesinprocessperformanceorproductqualityandquantity.Beyondthispoint, whichcanbequitedifferentinvariousindustries, biofoulingbegins.
Thisthresholdofinterferenceisafeltlimit, whichreflectsthefoulingtoleranceofanoperator.Flemming(2009)speculatedthat,althoughit maybefeltdifferentlyindifferenttechnical fields,itmaybeassumedthata30%lossof productivity,productqualitylossorprocessefficacywillalertanyoperator,whowilltryto identifyandeliminatethereason.
Weherehypothesizethat,intheclinical setting,asimilarthresholdofinterferencecan betracedforbiofilmformationonimplanted biomaterials.Atvariancewithitsindustrial counterpart,thethresholdof“clinical”interferenceismostlydefinedbytheclinicalperformanceoftheimplanteddeviceandbyour abilitytodetectandinterpretsignsand symptomsofimplantfailure;infact,hereare inflammatorysymptomsorinfectionmarkers
Fig.2 Visiblebiofilm remnantsonthetitanium surfaceofafailedhip
TheConceptofBiofilm-RelatedImplantMalfunctionand“Low-GradeInfection”7
Table3 Listofmostcommonlyusedbiomaterialsbyapplication
Cardiovascular
Stents–Pacemaker–Implantablecardiacdefibrillators–Heartvalves–Catheters–Guidewires–Vasculargrafts–Sensors–Others(ventricularassistdevice(VAD),sternumclosuredevices,andintroducersheaths)
Orthopedic
Jointreplacement(Knee-,Hip-,Shoulder-,Ankle-,Elbow-,Wrist-,Finger-arthroplasty)
Spine(spinalfusion,motionpreservation/dynamicstabilization,interspinousspacers,discarthroplasty)
Bioresorbable(Sutureanchors,Interferencescrews,Meniscalrepairtacks,mesh)
Orthobiologics(Allografts,bonesubstitutes,autografts)
Dental
Dentalimplants
Dentalbonegrafts&substitutes
Dentalmembranes
Plasticsurgery
Acellulardermalmatrices
Craniofacialsurgery
Bioengineeredskins
Breastimplants
Trauma
Fracturefixationdevice(boneplates,screws,pins,rods,wires
Tissueengineering
Scaffoldsforregenerativemedicine
Ophthalmology
Contactlens
Intraocularlens
Functionalreplacementsofoculartissues
Syntheticcorneas
Others
Neurologicaldisorders/CentralNervousSystems
Shuntingsystems
Corticalneuralprostheticsandimplantableneurostimulators
Otherapplications
Drugdeliverysystems
Urinarycathetersandprosthesis
Fig.3 Schematicbiofilm developmentbelowand abovethearbitrary “thresholdofinterference” (dottedline)(rewritten afterFlemming etal. 1994). Δ represents theeffectofbiofilms development(e.g.: thickness,friction resistance,etc.)
thatmaytriggeran“alert”totheinvolved “operators”,thepatientandhis/herphysician, raisingthesuspectofabiofilm-relatedimpaired performanceoftheimplantedmedicaldevice; thisinturnwilleventuallyelicitfurther investigationsandanappropriateresponseor treatment.
Inanygivenpatient,thethresholdof“clinical”interferencedependsonthenetbalanceof differentvariables,includingthetypeof
microorganism(s)andoftheimplant,the antibacterialprophylaxis/treatmentand,most importantly,theextentofthehost’simmune andinflammatoryresponse.
Accordingtothelevelsetasanalerttodefine thepresenceofapathologicalcondition,thefollowingclinicalscenarios(Fig. 4),withrespective thresholdsofinterference,canbedistinguished (Fig. 5).
Fig.4 Schematicrepresentationofpossibleclinical scenarios,inthepresenceofamedicalimplantandadheringbiofilm-producingmicroorganisms.(a)Subclinical presentation.Bacteriaandbiofilmsdonotinterferein anydetectablewaywithimplantfunction.Symptoms andmarkersofinfectionareabsentandthedeviceisfelt asnormallyfunctioning.(b)Implantmalfunction.Bacteriaandbiofilmsinduceonlyminorclinicalsignsand
symptoms,butmarkersofinfectionremainnegative.(c) Low-gradeinfection.Bacteriaandbiofilminteraction induceamildhost’sreaction,withmoderateclinical signsandsymptomsandslightchangesininflammatory markers.(d)High-gradeinfection.Aconditioninwhich theclassicalsignsandsymptomsofinfectionandinflammationarepresent,withpositivemarkers
Fig.5 Schematicbiofilmdevelopmentbelowandabove arbitrary“thresholdsofclinicalinterference”forimplant malfunction,low-andhigh-gradeinfection.Subclinical presenceofbacteriaandbiofilmsistheoreticallypossible andisrepresentedbythe dottedgreycurve. Δ represents theeffectofbiofilmsdevelopment(clinicalsignsand
4.1SubclinicalContamination andImplantMalfunction
Whilesubclinicalcontaminationisdefinedasthe presenceofbacteriaandbiofilmsthatdonot interferewiththenormalfunctionofthe implantedmedicaldevice,implantmalfunction isassociatedwithmildclinicalsigns/symptoms, thatmaybereportedasmildbutpersistentor recurrentpainatthesiteofsurgeryand/ormild softtissuecontractureorfunctionalimpairment, withnegativelaboratoryandimagingmarkersof infection/inflammation;theidentificationofthe slow-growingmicroorganisms,thatgenerally causethiscondition,requiresprolonged microbiologicalcultures,antibiofilmandeventuallygenomicormoleculartechniques(Drago etal. 2013;Palmeretal. 2011).
4.2Low-GradeInfection
Low-gradeinfectionisaconditioninwhichthe patientcomplainsaboutrecurrentorpersistent painand/orsofttissuecontracturewithvarious functionalimpairment,withmixedpositive/negativemarkersofinfection/inflammation;
symptoms,positivemarkersofinfection/inflammation) andistheresultoftheinteractionbetweenthe microorganismsandtheirby-products,thehostresponse, thetypeofimplantandtheantibacterialprophylaxis/ treatment
pathogenidentificationrequiresprolonged culturesandantibiofilmtechniques.
4.3High-GradeInfection
High-gradeinfectionsdisplaytheclassicalsigns/ symptomsofinfection/inflammation,including variousdegreesofredness,swelling,painand localwarmthand/ordelayedwoundhealingor drainingsinus,withpositivelaboratoryand imaginginvestigations;inthesecases,pathogen identificationisoftenpossiblewithtraditional microbiologicaltechniques,butisbetter achievedwithprolongedculturesandantibiofilm processing,especiallyifpatientsunderwent empiricalantibiotictreatmentpriortocultural examination.
Ontheaverage,theestimatedincidenceof high-gradeinfectionlaysinarangebetween approximately0.5–2.5%aftercleansurgery;on theotherhand,low-gradeinfectionsandbiofilmrelatedmalfunctionsprobablyoccurinapproximately8–12%ofthepatientsreceivingan implantedbiomaterial,accountingforunexplainedpain,softtissuecontractures,jointstiffness,delayedbonehealingornon-unionafter
fracturefixation,etc.,thatmayeventuallybeleft untreatedofmanagedonlybymedicationsor rehabilitationmeasuresandneverleadtoimplant removal.
Examplesofimplantmalfunctionor low-gradeinfectionscanbeobservedinvarious surgicalfields.
Inplasticsurgery,Pajkosetal.(2003) reportedastatisticallysignificantpositive,direct relationshipbetween Staph.epidermidis sonicationcolturesinremovedbreastimplantsandthe degreeofcapsularcontracture,intheabsenceof clinicalsignsofinfection.
Beswicketal.(2012),inarecentsystematic reviewofprospectivestudiesinpatients undergoingtotalhiporkneereplacementforosteoarthritis,foundaproportionofpeoplewithlongtermpainofunknownoriginrangingfromabout 7–23%afterhipandfrom10to34%afterknee replacement,whileotherstudieshaveshownthat “between4%and13%ofpatientswithpreoperativediagnosisofasepticlooseningwere infected”,whenretrievedimplantswereanalyzed withgenomicidentificationmethods(Moojen etal. 2010).Furthermore, P.acnes hasbeen identifiedinrecentyearsasanoccultcausative agentofpainaftershoulderprosthesis(Millett etal. 2011).
Infracturefixation,ananimalmodelshowing theimpactoflow-gradeinfectionoftherateof non-uniondueto Staph.epidermidis hasbeen recentlypublished(Lovatietal. 2016),while “aseptic”tibialnon-unionin23patientshad beenrecentlyreportedbyGilleetal.(2012)to beassociatedin2cases(8.7%)withpathogens thatcouldonlybedetectedbyinvestigatingbacterialrRNAwithpolymerasechainreaction (PCR).
Eachoneoftheabovementionedclinical conditionsmayeventually,attimes,shiftfrom onetotheother,dependingontreatments,host’s immunesystem,bacteriallifecycle,etc.(Fig. 6).
Forexample,itisacommonobservationin theclinicalsettingthata wellconductedantibiotictreatmentmaysometimessuppressinflammatorysignsassociate dwithaperi-prosthetic jointinfection,thuschangingahigh-grade infectiontoalow-gradeoneoreventoamild
Fig.6 Theclinicalpresentationofbiofilm-related infectionsofmedicaldevicesandbiomaterialsis influencedbymanyfactorsandshiftsfromoneclinical conditiontoanothermayoccasionallyhappen
implantmalfunction,withonlyminorclinical signsandnegativeserummarkersofinflammation.Ontheotherside,abreachintheimmune systemcompetence,dueforexampletoanconcurrentchemotherapy,mayeventuallyshifta previouslow-gradeinfectiontoahigh-grade, acutesepsis.
5Conclusions
Biofilmsandbiofoulinghaveawellknowndetrimentalimpactonmostindustrialand manufacturingprocesses.
Thresholdofinterferenceinindustrydepends mostlyoneconomicalconsiderationsandis believedtobereachedinanycasewhena30% lossofproductivity,productqualitylossorprocessefficacyoccurs.
Implantedbiomaterialsalsoaregreatly affectedbypossiblebacterialcontaminationand biofilmformation,thatmayultimatelyinterfere withimplantfunction,durabilityandperformanceorpatient’swellbeing.
Similartothethresholdofinterferencein industry,herethresholdsof“clinical”interferencecanbeimagined,whichidentifyvarious possibleclinicalconditions,rangingfroman implantmalfunctiontoahigh-gradeinfection. Theoccurrenceofeachoftheseclinical conditionsdependsontherelativebalance betweenthetypeofimplant,theantibacterial
prophylaxis,thebehaviorofthecolonizing microorganismsandthehost’sresponse.
Itacommonobservationthat,eventhemedicalfieldtheperceivedthresholdofclinicalinterferenceissetataratherhighlevelandusually onlythehigh-gradeinfectionstriggersomeadequateresponseintheoperators.However,abetterunderstandingofbiofilms-relatedclinical presentationsandacknowledgingthefactthat biofilmsonanimplantcanbeassociatedwith onlyminorornosignsofinfection,iscrucial forphysicians,inordertoimplementthebest diagnosticandtherapeuticmeasuresforall patientswithlow-performingimplant;apractical exampleistheintroductioninthesurgical
Fig.7 Removedimplant,abonecementspacer,issent formicrobiologicalexaminationwithchemical antibiofilmprocessingbymeansofdithiothreitol(DTT), apureantibiofilmagentthatdoesnotinterferewithbacterialgrowth.Thedisposablekit(microDTTect,4iSrl, Monza,Italy)allowstocollectsamplesatsurgeryandto transportandprocesstheminacompletelyclosedsystem, avoidingcontamination,andincreasingsensitivityofculturalexamination
routineofsystemsthatmayallowculturalexaminationwithantibiofilmprocessingofallfailed implants(Fig. 7).
Ontheotherhand,recognizingbacteriaand biofilmsasapossiblereasonofimplantmalfunction,shouldpromptregulatorybodiestoconsider anti-adhesiveandantibiofilmimplantcoating technologiesasa(partof)medicaldevice aimedatreducingimplantmalfunction,thus adoptingtherelativeevaluationprocessandcertificationprocedure,alsoinconsiderationofthe promisingclinicaloutcomesreportedindifferent clinicaltrials(Tsuchiyaetal. 2012;Romano ` etal. 2015, 2016).
Aneffectiveantibiofilm-targetedapproach fromallplayersistheonlywaythemedical communitymayhavetomitigatethecurrent unacceptablesocialandeconomicalburdenof implant-relatedinfectionsandmalfunctions.
References
AngstEC(1923)Thefoulingofshipbottomsbybacteria. Report,BureauConstructionandRepair,United StatesNavyDepartment,Washington,DC BaddourLM,EpsteinAE,EricksonCC,etal.,American HeartAssociationRheumaticFever,Endocarditis,and KawasakiDiseaseCommittee,CouncilonCardiovascularDiseaseinYoung,CouncilonCardiovascular SurgeryandAnesthesia,CouncilonCardiovascular Nursing,CouncilonClinicalCardiology,InterdisciplinaryCouncilonQualityofCare,AmericanHeart Association(2010)Updateoncardiovascularimplantableelectronicdeviceinfectionsandtheirmanagement:ascientificstatementfromtheAmericanHeart Association.Circulation121:458–477
BauerTW,GrossoMJ(2013)Thebasicscienceofbiofilm anditsrelevancetothetreatmentofperiprosthetic jointinfection.OrthopKnowlOnlineJ11(9):12–20 BeswickAD,WyldeV,Gooberman-HillRetal(2012) Whatproportionofpatientsreportlong-termpain aftertotalhiporkneereplacementforosteoarthritis? Asystematicreviewofprospectivestudiesinunselectedpatients.BMJOpen2:e000435.doi:10.1136/ bmjopen-2011-000435
Bixleretal(2014)JcolloidInterfaceSci419:114–133 ChmielewskiRAN,FrankJF(2003)Biofilmformation andcontrolinfoodprocessingfacilities.ComprRev FoodSciFoodSaf2:22–32.doi:10.1111/j.1541-4337. 2003.tb00012.x
ChyeFYF,AbdullahA,AyobMK(2004)Bacteriological qualityandsafetyofrawmilkinMalaysia.Food Microbiol21:535–541
CoetserSE,CloeteTE(2005)Biofoulingand biocorrosioninindustrialwatersystems.CritRev Microbiol31:213–232
ColauttiRI,BaileySA,VanOverdijkCD,AmundsenK, MacisaacHJ(2006)Characterisedandprojectedcosts ofnonindigenousspeciesinCanada.BiolInvasions 8:45–59.doi:10.1007/s10530-005-0236-y
CostertonJW,LewandowskiZ,CaldwellDE,KorberDR, Lappin-ScottHM(1995)Microbialbiofilms.Ann RevMicro49:711–745
CostertonJW,StewartPS,GreenbergEP(1999)Bacterial biofilms:acommoncauseofpersistentinfections. Science284:1318–1322
DaleH,HallanG,HallanG,EspehaugB,HavelinLI, EngesaeterLB(2009)Increasingriskofrevisiondue todeepinfectionafterhiparthroplasty.ActaOrthop 80:639–645
DonlanRM(2002)Biofilms:microbiallifeonsurfaces. EmergInfectDis8(9):881–890
DragoL,SignoriV,DeVecchiE,VassenaC,PalazziE, CappellettiL,Romano ` D,Romano’CL(2013)Useof dithiothreitoltoimprovethediagnosisofprosthetic jointinfections.JOrthopRes31:1694.doi:10.1002/ jor.22423
FlemmingHC(2009)Microbialbiofouling:unsolved problems,insufficientapproaches,andpossible solutions.In:FlemmingHCetal.(eds)Biofilm highlights.SpringerSeriesonBiofilms5.SpringerVerlag,Berlin/Heidelberg.doi:10.1007/978-3-64219940-0_5,2011
FlemmingHC(2011)Microbialbiofouling:unsolved problems,insufficientapproaches,andpossible solutions.In:FlemmingH-C,WingenderJ,Szewzyk U(eds)Biofilmhighlights.Springer,Essen,pp 81–109
FlemmingHC,SchauleG(1988)Biofoulingon membranes–amicrobiologicalapproach.Desalination70:95–119
FlemmingH-C,SchauleG,McDonoghR,RidgwayHF (1994)Mechanismandextentofmembranebiofouling.In:GeeseyGG,LewandowskiZ,FlemmingH-C (eds)Biofoulingandbiocorrosioninindustrialwater systems.Lewis,Chelsea,pp63–89
GaneshCK,AnandSK(1998)Significanceofmicrobial biofilmsinfoodindustryareview.IntJFood Microbiol42:9–27
GilleJ,WallstabeS,SchulzAPetal(2012)Isnon-union oftibialshaftfracturesduetononculturablebacterial pathogens?AclinicalinvestigationusingPCRand culturetechniques.JOrthopSurgRes7:20–25
GomezE,PatelR(2011)Laboratorydiagnosisofprostheticjointinfection,partI.ClinMicrobiolNewsl33 (8):55–60
GristinaAG,NaylorPT,WebbLX(1990)Molecular mechanismsinmusculoskeletalsepsis:theracefor thesurface.InstrCourseLect39:471–482
HarrisonJJ,TurnerRJ,MarquesLLR,CeriH(2005) Biofilms.Anewunderstandingofthesemicrobial
communitiesisdrivingarevolutionthatmaytransformthescienceofmicrobiology.AmSci93:508–515
KaminoK(2013)Mini-review:barnacleadhesivesand adhesion.Biofouling29:735–749
KurtzSM,LauE,WatsonH,SchmierJK,ParviziJ(2012) Economicburdenofperiprostheticjointinfectionin theUnitedStates.JArthroplast27:61–65
LeeW,LewandowskiZ,NielsenPH,HamiltonWA (1995)Roleofsulfate-reducingbacteriaincorrosion ofmildsteel:areview.Biofouling8:165–194
LentinoJR(2003)Prostheticjointinfections:baneof orthopedists,challengeforinfectiousdisease specialists.ClinInfectDis36:1157–1161
LovatiAB,Romano ` CL,BottagisioM,MontiL,De VecchiE,PrevidiSetal(2016)Modeling Staphylococcusepidermidis-inducednon-unions:subclinical andclinicalevidenceinrats.PLoSOne11(1): e0147447.doi:10.1371/journal.pone.0147447
MillettPJ,YenYM,PriceCS,HoranMP,vander MeijdenOA,ElserF(2011)Propionibacterium acnesinfectionasanoccultcauseofpostoperative shoulderpain:acaseseries.ClinOrthopRelatRes 469(10):2824–2830
MoojenDJF,vanHellemondtD,VogelyHCetal(2010) Incidenceoflow-gradeinfectioninasepticloosening oftotalhiparthroplasty:aprospectivemulticenter studyusingextensiveroutineandbroad-range16S PCRwithreverselineblotdiagnostics.ActaOrthop 81(6):667–673
NanaA,NelsonSB,McLarenA,ChenAF(2016)What’s newinMusculoskeletalinfection:updateonbiofilms. JBoneJointSurgAm98:1226–1234
PajkosA,DevaAK,VickeryKetal(2003)Detectionof subclinicalinfectioninsignificanbreastimplant capsules.PlastRecontrSurg111:1605–1610
PalmerM,CostertonW,SeweckeJ,AltmanD(2011) Moleculartechniquestodetectbiofilmbacteriainlong bonenonunion:acasereport.ClinOrthopRelatRes 469:3037–3042
PetrakT,KaloderaZ,NovakovicPetal(1999)Bacteriologicalcomparisonofparallelandcounterflowwater chillingofpoultrymeat.MeatSci53:269–271
PittetB,MontandonD,PittetD(2005)Infectioninbreast implants.LancetInfectDis5:94–106
PrakasamS,SteinK,LeeMK,RampaS,NalliahR, AllareddyV,AllareddyV(2016)Prevalenceand predictorsofcomplicationsfollowingfacialreconstructionprocedures.IntJOralMaxillofacSurg45 (6):735–742
PrenticeT,BeagleholeR,IrwinA(2004)TheWorld HealthReport,2004:changinghistory.WorldHealth Organization,Geneva
Romano ` CL,DeVecchiE,VassenaC,ManziG,DragoL (2013a)Acaseofalateandatypicalkneeprosthetic infectionbyno-biofilmproducerPasteurella multocidastrainidentifiedbypyrosequencing.PolJ Microbiol62(4):435–438
Romano ` CL,ToscanoM,Romano ` D,DragoL(2013b) Antibiofilmagentsandimplant-relatedinfectionsin
orthopaedics:wherearewe?JChemother25 (2):67–80
Romano ` CL,LogolusoN,DragoL,PeccatiA,Romano ` D (2014)RoleforirrigationandDebridementin Periprostheticinfections.JKneeSurg27:267
Romano ` CL,ScarponiS,GallazziE,Romano ` D,DragoL (2015)Antibacterialcoatingofimplantsinorthopaedicsandtrauma:aclassificationproposalinan evolvingpanorama.JOrthopSurgRes10:157–163
Romano ` CL,MalizosK,CapuanoN,MezzopreteR, D’ArienzoM,VanDerStraetenC,ScarponiS, DragoL(2016)Doesanantibiotic-loadedhydrogel coatingreduceearlypost-surgicalinfectionafterjoint arthroplasty?BoneJointInfect1:34–41
ScarponiS,DragoL,Romano ` D,LogolusoN,PeccatiA, MeaniE,Romano ` CL(2013)Cementlessmodular intramedullarynailwithoutbone-on-bonefusionasa salvageprocedureinchronicallyinfectedtotalknee prosthesis:long-termresults.IntOrthop35:413
SchmittG(2009)Globalneedsforknowledgedissemination,research,anddevelopmentinmaterialsdeteriorationandcorrosioncontrol.WorldCorrosion Organization,NewYork
SchultzMP,BendickJA,HolmER,HertelWM(2011) Economicimpactofbiofoulingonanavalsurface ship.Biofouling27:87–98.doi:10.1080/08927014. 2010.542809
ShiX,ZhuX(2009)Biofilmformationandfoodsafetyin foodindustries.TrendsFoodSciTechnol20:407–413
StewartPS,CostertonJW(2001)Antibioticresistanceof bacteriainbiofilms.Lancet358(9276):135–138
StoodleyP,EhrlichGD,SedghizadehPP,Hall-StoodleyL,BaratzME,AltmanDT,SotereanosNG,Costerton
JW,DemeoP(2011)Orthopaedicbiofilminfections. CurrOrthopPract22(6):558–563
ThurlowLR,HankeML,FritzT,AngleA,AldrichA, WilliamsSH,EngebretsenIL,BaylesKW,Horswill AR,KielianT(2011)Staphylococcusaureusbiofilms preventmacrophagephagocytosisandattenuate inflammationinvivo.JImmunol186(11):6585–6596
TownsinRL(2003)Theshiphullfoulingpenalty.Biofouling19(Suppl):9–15
TsuchiyaH,ShiraiT,NishidaH,MurakamiH,KabataT, YamamotoNetal(2012)Innovativeantimicrobial coatingoftitaniumimplantswithiodine.JOrthop Sci17(5):595–604
VerranJ,JonesM(2000)Industrialbiofouling.Wiley, NewYork
VoordouwG,ArmstrongSM,ReimerMFetal(1996) Characterizationof16SrRNAgenesfromoilfield microbialcommunitiesindicatesthepresenceofa varietyofsulfate-reducing,fermentative,andsulfideoxidizingbacteria.ApplEnvironMicrobiol 62:1623–1629
WorldHealthOrganization(2014)Antimicrobialresistance:globalreportonsurveillance2014. http://www. who.int/drugresistance/documents/surveillancereport/ en/
ZimmerliW,WaldvogelFA,VaudauxP,NydeggerUE (1982)Pathogenesisofforeignbodyinfection: descriptionandcharacteristicsofananimalmodel.J InfectDis146(4):487–497
ZmistowskiB,KaramJA,DurinkaJB,CasperDS,Parvizi J(2013)Periprostheticjointinfectionincreasesthe riskofone-yearmortality.JBoneJointSurgAm 95:2177–2184
AdvExpMedBiol-AdvancesinMicrobiology,InfectiousDiseasesandPublicHealth(2017)5:15–27
DOI10.1007/5584_2016_173
# SpringerInternationalPublishingSwitzerland2016
Publishedonline:5November2016
3ColonizationofImplants:The FirstStepofImplantInfection
Afterbreakageofthephysiologicalbarriers invadingbacteriarapidlyseekadequatesurfaces tosettledownandformbiofilms,alifestyle,in which–incontrasttotheirplanktonic counterparts–theyareprotectedagainsthost defensemechanisms.Accordingtothephrase “racetothesurface”introducedbyGristina (Gristina 1987)microbialadhesioncompete withtissuecellintegrationforcolonizationof thebiomaterial’ssurface.Gristina’sconceptis stillvalid,andisnowsupportedbyanabundance ofdata,particularlyregardingthesuitable surfaces,meansofbacterialattachmenttothe saidsurfaces,andsignalscontrollingbiofilmformation.Itisnowapparent,thatbiofilmsformationisarathercomplex,geneticallydriven process,mediatedbynumberofbacteria-derived signalingmolecules,alsoknownas“quorum sensing(QS)molecules”orautoinducers.The basicstepsofbiofilmformationarequitesimilar amongthebacteriaspecies:bacteriaattachtoa surfacebymeansofspecializedadhesion molecules,thensignalingmoleculesarereleased, whichinturndrivethebiofilmformationby inducingtheproductionoftheextracellular matrix(extracellularpolymersubstances,EPS), thename-giving,insomeinstancesvisible,film orslime,andalsobyalteringbacteria-inherent featuresandproperties,forexamplethelossof flagella.Thebacteriaarethenembeddedinthe extracellularmatrix,themostconspicuousfeatureofthebiofilm,yieldingawell-organized bacterialcommunity.Ofnote,althoughthe basicmechanismsappeartobesimilar,theadhesionmoleculesaswellasthesignalingquorumsensingmoleculesdiffergreatlyamongthespecies,asdoesthequalityandcompositionofthe extracellularmatrix(Heilmann 2011;Vander MeiandBusscher 2012;Fosteretal 2014, reviewedinDickschat 2010;Gargetal 2014; Büttneretal 2015).Thisisimportanttorealize whenattemptingtointerferewithattachmentor biofilmformationasapreventiveregimen (Dragoetal 2013,reviewedinArciola 2009;
Shunmugaperumal 2010;Beloinetal 2014; Wilkinsetal 2014).
Biofilmsareconsideredasaninteractivesymbiotic“cityofmicrobes”.Biofilmsareanefficientandprotectivesurvivalstrategyina potentiallyaggressiveenvironment(forin-depth understandingofthemolecularmechanismsof biofilmformationandresistanceresp.tolerance pleaseseee.g.thefollowingreferencesand reviews:Costertonetal 1999;O’Tooleetal. 2000;DonlanandCosterton 2002;Greenberg 2003;Wuertzetal 2004;Costertonetal 2005; Williamsetal 2007;KaratanandWatnick 2009; Agarwaletal 2010;Ha ¨ nsch 2012a;Wolcottetal 2013;Olsen 2015)(Fig. 1).
Colonizationoftheimplantisthedecisive stepinimplant-relatedinfections(reviewedin Hall-Stoodleyetal 2004),anddependsonthe abilityofbacteriatoadheretoagivensurface. Adhesionisinfluencedbyagreatvarietyof components,includingbacteriaspecies,properties oftheimplantsurface,suchasmicroarchitecture, roughness,orelectricalcharge,asareenvironmentalparameters,includingflowconditions,rheology,ortemperature.Adhesioninvolvesthe classicalphysicochemicalforces(VanderWaals attraction,electrostaticcharges,gravitational forcesand/orhydrophobicinteractions),and specializedadhesionmoleculesonthebacteria (reviewede.g.inPavithraandDoble 2008; Harmsenetal 2010;Otto 2014;Persatetal 2015). Invitro,colonizationappearswithina fewhours,slimeproductionwithinseveraldays, dependingonexperimentalconditions,particularlye.g.species,initialnumberofbacteria,or flowconditions.However,verylittleisknown aboutthe invivo situation,andinsightsprovided bystudiesinanimalmodelsarealsolimited.
Biofilmformationisadynamicprocess.Singlebacteriacanleaveormaybearereleasedfrom thebiofilm,andalsotheorganizationofbacteria withinthebiofilmandthequalityoftheextracellularmatrixissubjecttomodifications,reconstructionorself-inhibition(NagarandSchwarz 2015).Thecomponentsoftheextracellular matrixvaryamongspecies(reviewedin Sutherland 2001;FlemmingandWingender
bacterial adhesion molecules
surface structures promoting adhesion
surface
binding of bacteria to implant
formation of microcolonies production of extracellular polymeric substances (EPS) („slime“) in which bacteria then are embedded maturation of biofilm to complex threedimensional structure
Fig.1 Biofilmformationonimplantsurface. (a)Sequenceofbiofilmformation:From right to left: Bindingofbacteriatoimplant(adhesion,colonization), formationofbacterialmicrocoloniesandproductionof extracellularpolymericsubstances(EPS,slime)inwhich thebacteriaareembedded,finallybiofilmmaturationtoa complexthree-dimensionalstructure. Top:schematic
2010),whichisalsoimportanttotakeinto accountwhenattemptingtosanitizeortodisrupt biofilms.Onimplants,bacterialbiofilmsmaybe
cartoon; below:illustrationofstep-by-stepformationof biofilms(white:bacteria)usinglaserscanmicroscopy (b+c).Detectionofbiofilmformationonimplantby scanningelectronmicroscopy(SEM)(b)(Courtesyof Prof.UrsulaObst,KarlsruherInstitutfürTechnologie) (c)andbystainingwithmira-ton(c)(Figureadapted fromWagnerandHansch 2015)
single-species,ormaycomprisemultiplespecies andalsofungae,suchas Candidaalbicans, can formbiofilms(RenduelesandGhigo 2012;
Sherryetal 2014,reviewedinLynchand Robertson 2008).
Artificialsurfacesareexcellentsubstratafor biofilmformation;however,itremainsstillelusivewhythey,comparedtohosttissue,arepreferentiallycolonizedbybacteria.Alikely explanationisthatartificialsurfacesareinert andhencelackdefensemechanismsasthereare foundontissuecellsthatpreventorfendoff colonization(Chunetal 2004;Hastings 2004). Moreover,afterimplantation,biomaterialsare readilycoveredbybloodandserumproteins (e.g.fibrinogen,fibronectin,vitronectin) resultingintheformationofaso-calledconditioningfilmorlayer,whichinturnpromotes adhesionofbacteriabyprovidingexclusive receptorsites(Rochfordetal 2012).Consistently,mimickingthe invivo microenvironment bycoatingnon-biologicalsurfaceswithhuman serumorplasma,itcouldbeshownthatbacterial adherenceandbiofilmformationisincreased (Wagneretal 2011).Takingintoaccountthat immersioninbloodoccursimmediatelyafter placingtheimplant insitu,strategiesmodifying theimplantsurface,e.g.bypotentially antibacterialsubstances(e.g.silverorcopper)–althoughshowingpromisingresultsinthe invitro experimentsorinanimalmodels (reviewedinSchmidmaieretal 2006;Goodman etal. 2013;Gbejuadeetal 2015;Francolinietal
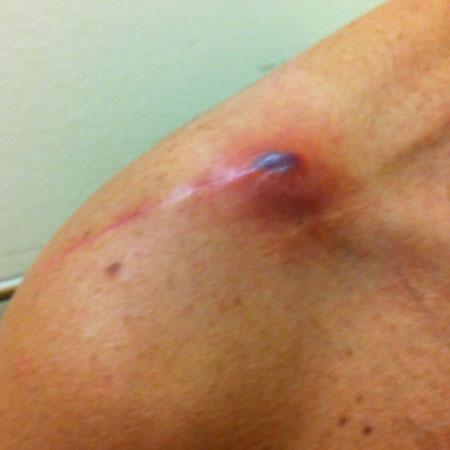
Fig.2 Characteristic clinicalsignsoflocal inflammationfollowing osteosynthesisoftheright lateralclavicula
2015;Romanoetal 2015)–arenotreally promisinginalong-termsetting.
4Inflammation:AnEssential andTellingResponse toInfection
Entryofbacteriaintotissuesignals“danger”to thehost.Viamessengermolecules,systemicand localmeansofhostdefenseareactivated,and alsothetissueattheinfectedsitesisaltered,a processcollectivelyaddressedas“inflammation”.Majoralterationsaretheincreasedblood flow,theenhancedpermeabilityofbloodvessels andtheexpressionontheendothelialcellsof molecules,attractingandbindingphagocytic cells,whichconsequentlysqueezethroughthe vesselwallandmigrateactivelytowardsthe infectedsite.Additionally,alsoserumand bloodseepthroughtheopenings.Cytokinescontrolthemotilityandthedirectedmigrationofthe cells,aswellasthesurfacemoleculesonthe phagocyticcellsandontheendothelium,which areessentialforadhesionandorientation.The enhancedpermeabilityofbloodvesselsisusually restrictedtotheinfectedareaandaccountsfor thetraditionalsymptomsofinflammationknown as“rubor,calor,dolor,tumor,andfunction laesa”(Fig. 2).Thegenerationoftheso-called
“pro-inflammatoryenvironment”isanessential, indispensableresponseofthehosttoinjuryanda crucialprerequisiteforanefficienthostdefense.
Forclinicians,inflammationisanindicatorfor infection,anddetermininginflammationassociatedalterationssuchasincreasednumber ofleukocytesintheperipheralbloodorenhanced serumconcentrationsoftheC-reactiveprotein havealongtradition.Infact,bacterialinfections areprimarilyrecognizedbytheinflammatory responsetheyhavecreated,ratherthanbydetectionandidentificationofthebacteriathemselves. However,symptomsofinflammationoralack thereof,donotnecessarilyproveordisprove bacterialinfections,becausealsosterile irritationsoftissuescauseinflammation.Moreover,localizedbacterialinfectiondonotinevitablyinduceanotablesystemicinflammatory response.
5TheHostResponsetoBacterial Infection
Asdescribedabove,whenbacteriacrossthe defendingbarriers,forexamplefollowingdamagetotheskinorthemucousmembranes, respectively,thelocalcellssignal“danger”and alerttheimmuneresponse.Theexactpathway thatlinksthelocaldangersignaltoasystemic responseisstillunderinvestigation.Cytokines havebeenidentifiedthatinducetheincreaseof thebloodC-reactiveproteinconcentration,and thatmobilizegranulocytesfromthebonemarrow,resultinginleukocytosis,animportantindicatorofinfection.
Thefirstcellstorespondarephagocyticcells, especiallygranulocytes(polymorphonuclear neutrophils,PMN)as“firstlinedefense”, which,toefficientlycombatbacteria,infiltrate theinfectedsiteinacomplexandwell-regulated controlledmanner(thesequenceofthe neutrophil-mediatedinflammatoryhostresponse tobacteriaisillustratedinFig. 3).Briefly,dueto chemokines(e.g.complementC5),generatedat thesiteofinfectionanddiffusedintothetissues, thenearbyendothelialcellsbecome“sticky”by up-regulationofspecializedadhesionproteins,
thatcapturePMNfromtheperipheralblood. Afterbeingactivatedandattachedfirmly,PMN thensqueezebetweentheendothelialcells (so-calleddiapedesis)andmigrateactively towardsthebacteriaviachemotaxis.AsphagocyticcellsPMNbindandtakeupplanktonic bacteria,whichthen,afterbeingengulfedintoa plasmamembrane-derivedvacuole,the phagosome,arekilledintracellularly,predominantlybycytotoxicoxygenradicals,generated byasequentialreductionofoxygen.Inaddition tooxidativekilling,granulocytescarryalarge arsenalofbactericidalentities,amongthose e.g.lysozyme,defensins,collagenaseandelastase(anoverviewisshowninTable 1)(reviewed inFaurschouandBorregaard 2003),whichare storedpreformedinthecells,andarereleasedin responsetobacteria-derivedagentsorto cytokineseitherintothecellorintotheenvironment.Successfulphagocytosisinitiatesapoptosis oftheneutrophils,whichtheninturnarecleared byinvadingmacrophages;therebyspillingof PMN’scytotoxicandproteolyticcontentis prevented.Insummary,ideally,phagocytosis resultsintheclearanceoftheoffendingbacteria, theterminationoftheinflammatoryresponse, andtherestorationofthetissue,thewound healing.Inthat,thehostresponseislimitedina time-,andinmanyinstancesalsoinaspacedependentmanner(reviewedine.g.Savill 1997;Kobayashietal 2003;Leeetal 2003; WagnerandHansch 2005).
Invadingbacteriaalsoalerttheadaptive immuneresponse(Wagneretal 2008;Karauzum andDatta 2016).B-andT-lymphocytesare activated,andthegenerationofantibodiesis induced.Basically,anincreasedbloodantibody concentrationisanindicatorofanongoingadaptiveimmuneresponse,andcanbealsousedasa diagnostictool.However,becausethemajority ofbacteriafoundinimplantinfectionsarethe samethatarepermanentlycolonizingtheskinor themucousmembranesasopportunists, antibodiesarepresentatanytime,andtherefore arenotusefulasdiagnostictoolfordeviceassociatedinfections.
Becauseweareconstantlyexposedtobacteria,theimmunesystemispermanentlyinaction,
granulocytes
2.adhesion
1. release of mediators
3.diapedesis
endothelial cells ba
vascular leak
4.chemotaxis
bacteria
5a.phagocytosis
5.phagocytosis
5b.“frustrated phagocytosis“ release of bactericidal and cytotoxic entities bacterial biofilm
Fig.3 Theroleofgranulocytesinhostresponsetobacteria:(1)Mediatorsgeneratedandreleasedattheinfected site,actontheclose-byendothelium.(2)Byup-regulation ofadhesionproteins,theendothelialcellsbecome“sticky” andcapturecirculatinggranulocytesfromtheperipheral blood.(3+4)Afterbindinggranulocytestransmigratein betweentheendothelialcells(diapedesis)towardsthesite ofinfectioninadirectmanner(chemotaxis).(5)Having reachedthesitetheytakeupbacteria(phagocytosis)which
thenarekilledintracellularly.(5a)Dependingonmaturationstatebacterialbiofilmscanalsobephagocytosedby granulocytes(successfulphagocytosis);thesiteofinfectionwillbecleared.(5b)Incasethatthebiofilmresistthe attackofthegranulocytes,PMN,notabletotakeup bacteria(“frustratedphagocytosis”),arefurtheractivated andconsequentlyreleasetheirproteolyticandcytotoxic entitiesintothesurroundingscausingprogressivetissue destruction
Table1 Bactericidalandproteolyticcontentofneutrophilgranules(selection)
AzurophilgranulesSpecificgranulesGelatinasegranules α1-antitrypsinCollagenaseLeukolysin α-mannosidaseGelatinaseGelatinase
thoughmainlyunperceived.Thelatterbecomes obviouswhendealingwithpatientsonimmunosuppressivetherapy,orwithcongenitalor acquiredimmunodeficiencysyndromes(e.g. reviewedinArmengaud 1976;Doriaetal 2008; ShadyabandCrum-Cianflone 2012).
Theextentandtheefficiencyofthelocalhost responsedependsonvariousfactors,particularly thenumberofbacteria,thebacteriaspecies,the abilityofthebacteriatoinvadethetissue,and theirvirulence,thelatterdefinedasthepropensityofthebacteriatodamagehostcells,e.g.by producingcytotoxicsubstancesortoxinsthat interferewiththecellularsignalingorthecell metabolism(e.g.pertussistoxin).Ontheother hand,alsotheextentandqualityoftheimmune responsevarieswidelyamongindividuals.The geneticallydeterminedrepertoireofimmune cellsdeterminestherecognitionofforeignand potentiallydangerousmaterials(suchasbacteria);moreover,thealsogeneticallyimprinted capacitytoproducemessengermolecules(such asinterleukins)thatregulate,supportorcontrol theindividualimmuneresponsevariesamong individuals,asdoesthedensityofcytokine receptors,orthenumberofmoleculesthatsense “danger”.Consequently,theefficiencyof eliminatingagivenbacteriavariesamong individuals,asdoestheaccompanyinginflammatoryresponse.Thus,thehostdefensecanoccur “virtuallyunnoticedbythehost”,orleadto moderatelocalsymptomslikeswellingand redness,oreventoextremesystemicreactions, thesepsis.
6HostDefenseAgainstBacterial Biofilms
Howthehostreactstobiofilmsisnotreally known(reviewedinZimmerliandSendi 2011; Hansch 2012a).Inpatientswithimplantassociatedinfectionswesee–mostlikely–only anextremesituationwithfulminantinflammationandmoreorlessextensivetissuedamage. Ontheotherhand,onroutinelyremoved implants,bacterialbiofilmsarefoundwithout signsofinflammationoradversetissuereactions
(Neutetal 2003;Trampuzetal 2007;Obstetal 2012;Yanoetal 2014;Dapuntetal 2014b). Toreconcilethesetwoextremes,thefollowing scenariosarefeasible:
1.Thebacteriaarerecognizedbytheimmune response,andareeliminated,probablyeven beforeamaturebiofilmisformed.Because quorum-sensingmoleculesarerecognizedby cellsofthehostresponse,e.g.byphagocytes, itisadistinctpossibility(Vikstrometal 2005; Zimmermannetal. 2006;Maureretal 2015, reviewedinHansch 2012b).Suchanearlyand efficienthostresponsewouldgounnoticedby thehost
2.Alternatively,thebacteriaformabiofilmthat escapesrecognitionbytheimmuneresponse. Thebiofilmthenpersists,butwithouteliciting animmuneresponseoraninflammatory response.Thisisadistinctpossibility, becausethereistheclaimthatbacteriain biofilmshavealimitedmetabolismanddo nonotdivideoratleastnotthatasrapidlyas theirplanktoniccounterparts.Moreover,the surroundingextracellularmatrixmightnotbe recognizedasdangerous.
3.Bacterialbiofilmsarerecognizedbythe immunesystem,phagocyticcellsinfiltrate, butareunabletoeliminatethebiofilm.This wouldhavetwoimportantconsequences: (a)thebiofilmcontinuouslyactivatesthe hostresponse.Morephagocyticcellsand eventuallyalsoT-lymphocytesinfiltratethe sites(Wagneretal 2003;Wagneretal 2006),pro-inflammatorymediatorsareproducedwhichinturncausemorecellinfiltration,buteventuallyalsoactivationoflocal cells,forexampleosteoblasts.Theinflammatoryimmuneresponseisthusnotself-limited, butratherprogressesandexpands.(b)When phagocyticcellsarefurtheractivatedbutare unabletotakeupbacteria,theyreleasetheir cytotoxicandbactericidalentitiesintothe surroundings.However,eveninthiscase, thereisstillachancefortheneutrophilsto attackordestroyabiofilm,because invitro datashowclearlythatbiofilmsarenot inherentlyprotectedagainsttheattackby
MechanismsofBacterialColonizationofImplantsandHostResponse23
phagocyticcells(Wagneretal 2004;Günther etal 2009;Meyleetal 2010;Hansch 2012a). Incasethatthebiofilmresiststheattackbythe phagocyticcells(“frustratedphagocytosis”or “attemptwithoutsuccess”)(Fig. 3),thehost reactionatthelocalsiteproceedsandeven progresses,resultinginanongoingreleaseof cytotoxicandproteolyticentitieswith subsequentprogressivetissuedestruction (DallegriandOttonello 1997;Wardand
Lentsch 1999)(Fig. 4)asdirecollateraldamagebythelocalhostdefence(Wagneretal 2005).Bygenerationofaproinflammatory microenvironmentwithincreasedcytokine levels(e.g.tumornecrosisfactoralpha, TNFalpha;interleukin8,IL-8,MRP-14)the differentiationofboneresorbingosteoclasts (osteoclastogenesis)frommyeloidprecursor cellsisinduced(Fig. 5),perpetuatingthe self-inflictedtissuedamage,eventually
Fig.4 Progressivetissuedestructioninimplant-associatedinfectionfollowingplateosteosynthesis
Fig.5 Linkbetweeninflammationandosteolysis. Left: Schematicillustrationofdifferentiationandfusionof monocytesbypro-inflammatorymediatorswithformationofgiantcellsandfinallymulti-nucleatedbone-
resorbingosteoclasts. Right:bonebiopsyshowing osteoclasts(red kathepsinK, blue multiplenuclei)and infiltratedphagocyticcells(blue)inthesurrounding (FigureadaptedfromWagnerandHansch 2015)
Another random document with no related content on Scribd:
99
100
Fig. 101
Figs. 96 to 101.—Arrangement and Details of Collapsible Monoplane
At the other end of the fuselage is attached the hooks for the elastic, and the wire forming them is also utilised as a protector. The whole construction is made from a 10½-in. length of No. 18 �.�.�. piano wire, reinforced with a strip of thin bamboo, bent to shape over a blue flame and bound with silk tape. The part is shown unbound in
Fig.
Fig.
Fig. 101, and Fig. 102 shows an alternative protector; but it is not so effective.
The main plane is made of bamboo and piano wire of No. 18 �.�.�., and measures 2 ft. 10 in. by 6¼ in. The leading edge is of bamboo 3 ft. 2 in. long, ³/₁₆ in. wide, tapering to ⅛ in. at the ends, with a uniform thickness of ⅛ in. When planed down the length should be heated over a blue flame, and bent to the shape shown in Fig. 96, the outside of the bamboo being kept on the outside of the curve. The trailing edge should now be attached to the ends of the leading edge, a length of ½ in. being bent up at each end of the wire, and securely bound to the bamboo. The ribs should now be cut from the same gauge wire. The ends should be bent out at an angle so that they may be bound to the leading and trailing edges, as shown in Fig. 103; the projecting ends should be about ⅜ in.
Fig. 102
Fig. 103
Fig. 104
Fig. 105
Fig. 106
Fig. 107
Fig. 108
Fig. 109
Figs. 102 to 109. Details of Collapsible Monoplane
The two centre ribs are shaped as shown in Fig. 104. It will be seen that the leading edge is raised ¼ in. above the fuselage, and the rear edge is level with it. The ends of the ribs are fitted into small holes drilled in the top of the fuselage, and kept in position by means of small metal clips, as shown in Figs. 105 and 106. Four of the clips are required, and they may be easily made from thin tinplate and soldered. The ribs should be soldered to the trailing edge so as to make them secure. The framework should be covered with proofed silk, and neatly glued on all edges.
Fig. 110.—Rear Skid
Fig. 111 Fig. 112
Figs. 111 and 112.—Details of Screw
The chassis is shown by Fig. 107, No. 18 gauge piano wire being used for the framing, and an ordinary cycle spoke for the axle. Figs. 108 and 109 show the flexible joints of the chassis, which folds up flat when the hooks at C (Fig. 107) are withdrawn.
The wheels are 2 in. in diameter, and are rubber tyred, the ends of the spoke being burred over to keep them in position.
The rear skid is shown in Fig. 110, and is made of No. 18 gauge piano wire. A single length is used, being bent to shape, passed through the end of the fuselage, and held to its work by the projecting end D, which fits in a hole in the under-side of one of the pieces of wood. When not in use the skid may be folded flat.
The two propellers are of the simple bentwood type, 10 in. in diameter and 1 ft. 8 in. pitch. They are made of ¹/₁₆-in. birch in the usual way. The shape of the blades is shown in Fig. 111, and the angle at which the blades are bent is shown in Fig. 112. Six strands of strip rubber should be attached to each propeller, ordinary soft soap being used as a lubricant.




















































