THE INDEPENDENT VOICE OF PHARMACY



Starts to decongest in 2 minutes 10hrs Lasts up to 10 hours
Help your patients breathe their best.
Product Information: Please refer to the Summary of Product Characteristics for full product information.
Otrivine Sinusitis Relief 0.1% w/v Nasal Spray, Solution (xylometazoline hydrochloride). Indication: As a nasal decongestant for relief of the symptoms of acute rhinitis in allergic or upper respiratory tract infections, including the common cold or influenza, and sinusitis. Dosage and administration: Adults (including the elderly) and adolescents over 12 years of age: 1 spray into each nostril, up to 3 times daily as needed. Do not exceed 3 applications daily into each nostril. Contraindications: Hypersensitivity to xylometazoline or to any of the excipients. Trans-sphenoidal hypophysectomy or surgery exposing the dura mater. Acute coronary disease, hyperthyroidism or narrow angle glaucoma. Treatment with monoamine oxidase inhibitors, or within 14 days of stopping such treatments. Rhinitis sicca and Atrophic rhinitis. Children aged less than 12 years old. Precautions: Use with caution in patients with hypertension, cardiovascular disease, diabetes mellitus, phaeochromocytoma, prostatic hypertrophy and in patients on tri- and tetracyclic antidepressants. Not to be used for more than seven consecutive days: prolonged or excessive use may cause rebound congestion and/or atrophy of the nasal mucosa. Caution in patients with a strong reaction to adrenergic substances. Patients with long QT syndrome, due to increased risk of serious ventricular arrhythmias. Contains benzalkonium chloride which may cause nasal irritation or swelling inside the nose. Do not exceed the recommended dose, especially in children and the elderly. It is recommended that each product package is used by one person only. Side effects: For full details see SPC. Common: headache, nasal dryness and discomfort, nausea, application site burning. Uncommon: Epistaxis. Legal category: Pharmacy only. Licence number: PA 0678/120/003. Licence holder: Haleon Ireland Limited, 12 Riverwalk, Citywest Business Campus, Dublin 24, Ireland.
Date of preparation: November 2023. Additional information available on request. Contains Xylometazoline hydrochloride. Always read the label/leaflet. Ask your pharmacist for advice. Trade marks are owned by or licensed to the Haleon group of companies. PM-IE-OTRN-24-00004
Pharmacists issue a Call for Action Page 4 EDUCATION: ePortfolio Review for 2024/25 Page 8
CYBER SECURITY: Protecting your Pharmacy Business Page 18
FEATURE: Managing Cold and Flu Page 26
CPD: Upper Respiratory Tract Infections Page 39
INTERVIEW: Addressing Disparities in Chronic Pain Research Page 44
TEAM TRAINING: Wound Care Page 54
FINANCE: Expectations of a Financial Advisor Page 56









Page 5: McCabes Pharmacy completes rebrand
Page 6: Proposed rollout of electronic product information
Page 8: An overview of the ePortfolio Process
Page 10: New Masters of Pharmacy Programme at ATU
Page 14: Celebrating creativity and eye health awareness
Page 18: Cyber attacks on pharmacy – are you prepared?
Page 20: New research on the internal body clock
Page 34: Biosimilars in the treatment of Cancer
PUBLISHER:
IPN Communications
Ireland Ltd.
Clifton House, Fitzwilliam Street Lower, Dublin 2 00353 (01) 6690562
MANAGING DIRECTOR
Natalie Maginnis n-maginnis@btconnect.com
EDITOR
Kelly Jo Eastwood: 00353 (87)737 6308 kelly-jo@ipn.ie
SALES MANAGER
Amy Evans | amy@ipn.ie 0872799317
EDITORIAL/ EVENTS & MARKETING EXECUTIVE
Aoife Hunter: aoife@ipn.ie
CONTRIBUTORS
Mr David Galvin | Nathan Scanlon
Dr S Griffiths | Colm Moore
Dr D Breen | Lorna Nellany
Dr Deirdre McDermott
Dr Michele Roche
Dr Ishapreet Kaur
Fionnuala Ní Áinle
Siobhán Ní Bhriain
Michael O’Connor
DESIGN DIRECTOR
Ian Stoddart Design



5

In one of your lead news stories this month, The Pharmaceutical Group of the European Union (PGEU) is calling for comprehensive measures to address the growing shortage of pharmacists in Europe, urging policymakers, healthcare providers and educational institutions to act swiftly to ensure the long-term sustainability of the pharmacy profession. In its new Position Paper on Pharmacists Workforce Shortages, PGEU outlines key recommendations to strengthen the pharmacy sector and improve patient care.
You can read more about this on page 4.
On page 8 of this issue, Áine Barrett, ePortfolio Review Project Lead with the Irish Institute of Pharmacy, gives IPN readers an overview of the 2024/25 ePortfolio process and answers some commonly posed questions.

14

10 Irish Pharmacy News is circulated to all independent, multiple Pharmacists and academics in Ireland. All rights reserved by Irish Pharmacy News. All material published in Irish Pharmacy News is copyright and no part of this magazine may be reproduced, stored in a retrieval system or transmitted in any form without written permission. IPN Communications Ltd. has taken every care in compiling the magazine to ensure that it is correct at the time of going to press, however the publishers assume no responsibility for any effects from omissions or errors.
She says, “In addition to the email communications that selected pharmacists have received from the IIOP, at this point selected pharmacists will also notice a Pending sign on their ePortfolio. This sign will remain in place until the submission period is closed (26 January) and will not change when you have submitted cycles. When the first submission period closes this will change from Pending to Active. This means that the review process is happening and is not specific to a particular pharmacist. The Active sign will remain in place until the ePortfolio Review process is complete in early May.”
Last October, Chronic Pain Ireland and the University of Galway hosted a pivotal event marking the ‘Day of the Girl.’ The hybrid event, entitled Exploring Sex and Gender Disparities in Chronic Pain, brought together researchers, clinicians, and patients to shed light on the profound differences in how chronic pain is experienced and managed across sexes and genders.
Among the event’s key voices was Dr Michelle Roche, Associate Professor in Physiology, Co Director of the Centre for Pain Research at the University of Galway, and President of the Irish Pain Society. Dr Roche’s keynote address, Developing Guidelines and an Agenda for Research on Sex and Gender as Variables in Pain, highlighted the urgent need to address these disparities.
In a follow-up interview, she elaborated on her work and vision for the future of pain research. Turn to page 44 for the full story.
I hope you enjoy the issue.
FEATURE: MANAGING COLD AND FLU P26
FEATURE: SMOKING CESSATION P28
CPD: UPPER RESPIRATORY TRACT INFECTIONS P39
As healthcare systems face mounting pressure, community pharmacists’ role is more vital than ever. However, many European countries are facing difficulties in recruiting and retaining community pharmacists, exacerbated by burnout, early retirements, and a lack of interest among students.
The Pharmaceutical Group of the European Union (PGEU) is calling for comprehensive measures to address the growing shortage of pharmacists in Europe, urging policymakers, healthcare providers and educational institutions to act swiftly to ensure the long-term sustainability of the pharmacy profession. In its new Position Paper on Pharmacists Workforce Shortages, PGEU outlines key recommendations to strengthen the pharmacy sector and improve patient care.
PGEU asks for strategic workforce planning for pharmacists at a national level and for governments’ investments to ensure that the pharmacy workforce can meet the growing demands of an ageing
population and increasingly complex therapies.
“To continue to provide highquality care, we must boost education, leveraging on innovative teaching methods, and ensure that pharmacy curricula adapt to the evolving healthcare landscape.
Fostering interprofessional education early in students’ academic careers and using effective skill-mix strategies will be instrumental in creating a more unified healthcare workforce.
It is also imperative that we not only showcase the value of community pharmacy but also work to integrate the profession more deeply into the healthcare system,” said PGEU President Aris Prins.
“We should focus on promoting continuing professional development and on retention policies that address burnout and dissatisfaction within the profession. This includes offering manageable workloads, competitive salaries, reducing bureaucracy, improving career progression pathways, and promoting work-life balance.
Technological solutions can streamline administrative tasks in pharmacy, allowing community pharmacists to spend more time on patient counselling. Embracing digitalization can improve efficiency without compromising the critical patient interaction that is the core of quality healthcare”, Prins concluded.
Chemist Warehouse recently recognised the achievements of their team celebrating all the nominees and winners of their annual Awards of Excellence. The 2024 winners included Chemist Warehouse Artane for the Store of the Year; Kayla and Maria from the Blanchardstown and Henry Street stores for the full-time Pharmacist Assistant of the Year; Fiona from the Cornmarket store for part-time Pharmacy Assistant of the Year; Drogheda’s Sinead Brady who won Pharmacist of the Year and Ballincollig’s Kevin for the Intern of the Year.

Meanwhile, the pharmacy chain announced the opening of their 12th Irish store, located on Talbot Street in the heart of Dublin.
The 5,000-square-foot space offers a wide range of beauty, wellness, and baby products.
The new store has created over 20 new jobs, including managerial and pharmacist positions, contributing to the local community.
Chemist Warehouse recently opened their 12th store in Dublin

The Department of Health is making influenza vaccination available free of charge to everyone aged 50 to 64 years of age.
This extension is in addition to groups already eligible for the flu vaccination. Those aged 50 to 64 will shortly be able to receive a free vaccination. Arrangements are being completed at pace to make the vaccination available through Community Pharmacies, Trust vaccination clinics and GP surgeries. There will be a further update in the coming days.
Health Minister Mike Nesbitt said, “This year, flu season has had an early and sharp start, with the number of people who have influenza increasing rapidly since the beginning of December. This is contributing to the pressures currently being seen in our hospitals and expect that pressure to continue or grow in the coming weeks.
“The decision to extend the programme to everyone aged 50 to 64 years of age is a pragmatic approach based on the stock of vaccines that we have available and the pressures we are facing. It will benefit individuals, the health service and society more generally.
“To date, over 526,000 influenza vaccinations have been administered to those eligible for the annual flu vaccination programme across Northern Ireland. I would urge all those who are eligible for the flu vaccine, including those in the 50 to 64 age group, to take up the offer as soon as possible.
“For all those who are eligible, flu vaccination is free and it can be accessed easily from your local pharmacy or Trust vaccination clinic. The best thing you can do right now to protect yourself and help us to protect our services for those who are sickest is to get the flu jab.”
The PSI is inviting registered medical practitioners to express their interest in being appointed as Medical Assessors to its Health Committee.
About the role
The role of the Medical Assessor is to advise the Committee with regard to medical evidence arising when considering a complaint against a registered pharmacist.
The Medical Assessor must draw upon their professional experience and knowledge to help the Committee understand medical/psychiatric issues arising.
In Ireland, it is estimated that two million visits are made to pharmacies each month. Most people have high levels of trust and confidence in their pharmacist and pharmacy, but errors do happen. Medicines are complex, and complaints can arise about the care or treatment that is received from a pharmacist or pharmacy. When this happens, the PSI is responsible for receiving and handling “fitness to practise” complaints and managing those complaints pursuant to the statutory process as set out in Part 6 of the Act. Complaints are heard before statutory Disciplinary Committees.
Applicants are asked to describe how they meet the competencies required.
These include the following:
• A clear understanding of, and commitment to, public protection.
• A proven ability to work collaboratively and constructively with others.
• Understanding of, or interest in, regulatory/legislative environments or frameworks.
• Ability to analyse information and use effective judgement to provide fair and reasoned medical advice.
• Ability to communicate effectively with good interpersonal skills.
• Understands and values diversity and fair treatment.
If you wish to apply, please complete this application form and return by e-mail to eoiexpertpharmacists@psi.ie no later than 5pm on Friday, 31 January 2025.
McCabes Pharmacy now have 110 stores nationwide. Earlier this year, they embarked on an ambitious journey with LloydsPharmacy Ireland colleagues to rebrand 100% of the LloydsPharmacy Ireland store estate by the end of the year and are proud to say that the rebrand is now complete.
Their plans were ambitious with 80 stores to rebrand in 3 months, and with a huge team effort and some amazing support from our suppliers and agencies, they have achieved our goal.
McCabes CEO Sharon McCabes said, “We celebrated the launch of these new-look stores around the country with events in our local communities, and had an opportunity to meet some of the wonderful people behind it all.
“We welcomed some special guests and our loyal patients to join us in celebrating the merging of both brands, and McCabes Pharmacy becoming Ireland’s largest, patient centric pharmacy brand.
“We’re excited about the next chapter in our journey as McCabes Pharmacy, and look forward to serving our local communities together, as 110 pharmacies nationwide.”
Jan Pieter Hallema, LloydsPharmacy Managing Director, said of the journey, “Whilst our patients will still see the same familiar faces from the teams they know and trust, this new more resilient pharmacy network combines the strengths of both brands allowing us to enhance our services and continue to meet the evolving needs of our customers.”

On August 29th, we celebrated the rebranding of LloydsPharmacy Ashleaf and LloydsPharmacy Churchtown – the first of many transformations as part of the national rollout across the country
The first rebrand stores LloydsPharmacy Ashleaf and LloydsPharmacy Churchtown

The MPharm Degree is the national pharmacy qualification that must be successfully completed to be eligible to register and practice as a pharmacist in Ireland. The MPharm is a five-year integrated programme, and the programme must be accredited by the PSI. We currently accredit programmes delivered by Trinity College Dublin, the Royal College of Surgeons in Ireland and University College Cork.
New MPharm Degrees
In 2022, in response to national skills needs, the Higher Education Authority (HEA) published a call for increased national capacity in a number of health professions, including pharmacy. Subsequently, in October 2024, the Minister for Further and Higher Education, Innovation, Research and Science announced an increase in healthcare places in Ireland through the creation of six new programmes, including three
new pharmacy programmes at the Atlantic Technological University (ATU), the South East Technological University (SETU), and the University of Galway.
What does this mean for prospective students for the new programmes?
ATU, SETU and the University of Galway must undergo an accreditation process that is managed by us. We are engaging with each of these universities
at present in relation to the accreditation process. Once the accreditation processes are complete, we will publish the accreditation reports on our website. Information updates on the progress of accreditation visits are captured in our reporting to the PSI Council in the minutes and associated Registrar’s reports, available on this website.
Prospective students may also wish to communicate with the relevant universities.
In a significant stride towards further modernising patient care, regulatory efficiency, and environmental sustainability, Pharmaceutical Industry Associations (AESGP, EFPIA, and Medicines for Europe) have launched a new series of position papers advocating for the implementation of electronic Product Information (ePI) and improvement of the patient leaflet content.

By transitioning to ePI, patients, healthcare professionals (HCPs), and civil society will benefit from the most up-to-date, accessible medicinal information, ensuring safer use of medicinal products.
Key Highlights
1. ePI Phasing-In, Paper PhasingOut: The gradual phasing in of ePI is proposed to be fully operational within 4 years after entry into force of the revised General Pharma Legislation and will precede the phasing out of paper leaflets. This will ensure patients have continuous access to critical medicinal information via secure, harmonized digital platforms. Existing ePI platforms such as National Competent Authority and Industry websites and compendia could be used as solutions to initiate the
transition before ePI becomes fully available on the EMA/HMA portal. Phasing out paper in self-administered products will be more gradual than for HCPadministered products due to individual needs, administrative capabilities and product specific requirements.
2. Improving PIL: Patient information leaflets would greatly benefit from layout and readability improvements. There are several proposals to benefit correct safe use of medicinal products, by delivering clear information to level up health literacy.
3. Patient Safety and Digital Access: With 90% of EU citizens regularly accessing the internet, ePI will allow for availability of up-to-date
leaflets, interactive elements, personalized content, and more accessible formats such as large print or multimedia. However, alternatives for those without internet access will be retained to guarantee inclusivity.
4. Safeguarding Availability in Small Markets: Multi-country packs, which are simplified by the use of ePI, language exemption and harmonised labelling requirements, will improve the availability of medicines across Europe particularly in smaller markets, reducing logistical burdens and fostering greater supply chain agility.
5. Enhancing Regulatory Efficiency: The ePI platform is designed to streamline regulatory processes, reducing administrative burdens for both pharmaceutical companies and health authorities. The centralized EMA portal will serve as a single source of trustworthy information, fostering transparency and regulatory efficiency across the EU.
Pharmaceutical Industry is urging regulatory bodies across Europe to adopt a harmonized implementation of ePI. This transition is critical not only for advancing patient care but also for enhancing regulatory operations and addressing environmental challenges.

The International Pharmaceutical Federation (FIP) world congress 2025, themed “Pharmacy Forward: Performance, Collaboration, and Health Transformation,” aims to explore the evolving role of the pharmacy profession in the context of modern healthcare challenges. The congress will explore how pharmacists can drive forward the performance of healthcare systems, foster meaningful collaboration across sectors, and catalyse transformative changes in health outcomes globally. The congress is structured around three subthemes, each addressing key facets of this overarching theme.
The subtheme “Making it sustainable” focuses on enhancing global health through sustainable resource use, innovative health solutions, and robust policy frameworks. It addresses key challenges like supply chain efficiency and antimicrobial resistance while emphasising preventive care and digital health. It also highlights the importance of professional development, aiming to integrate sustainability into pharmacy education and practice, thereby reinforcing the profession’s global health impact.
“Making the system work” emphasises pharmacists’ roles in enhancing healthcare efficiency through integrated care, patient safety and innovations like telepharmacy and AI. It highlights pharmacoeconomics, policy development, and collaboration among healthcare providers. This congress track advocates for ongoing training, health literacy, adaptive regulatory frameworks and technological advancements to create an effective, safe and patient-centred healthcare system.
Equity in access to healthcare, reimbursement for pharmacists' services, and challenges in medicine supply are among the key factors shaping global pharmacy trends in self-care, according to a new insight board report reissued by the International Pharmaceutical Federation (FIP).
The report, based on an insight board meeting held in Cape Town, South Africa, in September, explores post-COVID-19 expansions of pharmacists' roles, the rise of digitalisation, and strategies to address healthcare workforce shortages. It also includes key recommendations for enhancing self-care.
At the 82nd FIP World Congress of Pharmacy and Pharmaceutical Sciences in Cape Town, South Africa, which took place in September 2024, pharmacists from around the world joined an insight board hosted by FIP for a discussion on global pharmacy trends and their impact on public health and self-care across different countries.
Despite their potential to enhance self-care, pharmacists face several barriers that can limit their effectiveness. Key factors such as regulatory constraints, pharmacy education and training, and public perceptions of pharmacists' roles need to be addressed to achieve optimal patient outcomes and pharmacists’ potential. The COVID-19 pandemic has also highlighted gaps in health literacy among communities which impact equity and access. It is essential to tackle these challenges to maximise the benefits of evolving pharmacy practices and strengthen the role of pharmacists in promoting self-care.
FIP has established a dedicated programme focused on selfcare for the profession globally. Linked to FIP Development Goal 18 (Access to medicines, devices and services), FIP dedicates an exclusive priority programme to self-care. The programme aims to leverage pharmacists' expertise, accessibility and trusted relations in the community to provide quality, evidence-based information and advice on self-care products and strategies to the public, to promote health literacy and to empower patients towards better health and well-being.






In October 2024, the Irish Institute of Pharmacy (IIOP) sent an email to approximately 1400 pharmacists who have been selected for the 2024/25 ePortfolio Review. The IIOP also sent a reminder email to pharmacists in early December. In January 2025, these selected pharmacists will be required to submit evidence of their Continuing Professional Development (CPD) to the IIOP via their ePortfolio.
Here, Áine Barrett, ePortfolio Review Project Lead, gives an overview of the process. We talk about the key dates, the process and where you can get more support.
So Áine, how will pharmacists know whether they are selected for the 2024/25 ePortfolio Review?
In addition to the email communications that selected pharmacists have received from the IIOP, at this point selected pharmacists will also notice a Pending sign on their ePortfolio. This sign will remain in place until the submission period is closed (26 January) and will not change when you have submitted cycles. When the first submission period closes this will change from Pending to Active. This means that the review process is happening and is not specific to a particular pharmacist. The Active sign will remain in place until the ePortfolio Review process is complete in early May.
If I’ve been selected, when will I have to submit my ePortfolio extract?
On Monday 6 January 2025 an email will be sent from the ePortfolio System (info@iiop.ie) to your IIOP registered email address inviting you to submit CPD cycles from your ePortfolio for review.
You will have until Sunday 26 January to submit your extract. You may submit cycles at any stage during the three-week submission window, and you may make more than one submission.
Pharmacists must have their current, active email address registered on their IIOP profile in order to ensure they receive communications from the IIOP about the 2024/25 ePortfolio Review. Should you need help in accessing the website, retrieving your password or editing your details contact the IIOP at info@iiop.ie.
How does the ePortfolio Review process work?
The ePortfolio Review process incorporates two elements:
• A System Based ReviewEach extract (i.e. the cycles you choose to submit) will be automatically reviewed against the System Based Standards which are pre-set within the IIOP ePortfolio system.
• A proportion of the ePortfolio extracts submitted will be reviewed against the Review Standards. This will include:
o All ePortfolio extracts submitted in the first submission period that do not meet the System Based Standards
o A random sample of ePortfolio extracts submitted in the first submission period that do meet the System Based Standards
o All ePortfolio extracts submitted in the second submission period
How will I know which cycles I should submit as part of my ePortfolio Review?
To help selected pharmacists ensure the cycles they submit as part of their ePortfolio extract meet
the System Based Standards, the IIOP developed the Ready Reckoner tool. The Ready Reckoner will be available to pharmacists selected for ePortfolio Review when the submission period opens in January.
Pharmacists can use the Ready Reckoner in two ways;
• Before submissionPharmacists can use the ‘Check readiness’ function before submission to identify cycles to submit which collectively meet all of the System Based Standards.
• After Submission - The Ready Reckoner automatically appears when one or more cycles are submitted for review and uses green and red lights to show progress against the System Based Standards.
In the event that one of the standards has not been met, a red light and a warning icon are shown, and pharmacists can click on the warning icon for an overview of the action they need to take to meet this standard.
I can’t find the “Ready Reckoner” on the IIOP website, where is it?
The Ready Reckoner will only become available to pharmacists included in the 2024/25 ePortfolio Review when the submission period opens on Monday 6 January. It can be found within the Completed Cycles section of the ePortfolio.
I have undertaken CPD but not yet had the opportunity to record it in my IIOP ePortfolio. Can I backdate these records to reflect when the work was completed?
Yes, you can backdate cycles to reflect the date the CPD was undertaken. Simply choose the relevant date when entering the ‘Cycle Start Date’. You can find further information on recording your CPD retrospectively in the How to meet the cycle from previous four years standard support resource which can be found on the 2024/25 ePortfolio Review Support page on the IIOP website.
Since I began using the IIOP ePortfolio, I have been recording entries in my ePortfolio but I
haven’t completed all of my CPD cycles. I have been selected for the 2024/25 ePortfolio Review, if I completed a cycle now will it count as CPD from the year I created it?
Yes, it will count from the date it was created e.g. if a cycle is created in 2023 and completed in 2024, the system will count it as a cycle for 2023. We understand that people work in different ways, and the IIOP ePortfolio has been built to reflect this. While some people may like to complete a cycle and finish it right away other people may like to work on them over time.
I understand that the Core Competency Self-Assessment Tool (CCSAT) has been retired. How do I meet the standard for self-assessment against the PSI Core Competency Framework (CCF) in the current year?
Yes, the CCSAT was retired in 2023. The 2024/25 ePortfolio Review standards require that at least one cycle is created and submitted following selfassessment against the Core Competency Framework for Pharmacists in the current year. For the purposes of the 2024/25 ePortfolio Review, the current year is considered to be 2024 and up to 26 January 2025 i.e. the end of the submission period. Having self-assessed against the CCF, you can demonstrate evidence of this by selecting the tick box under the Self-Appraisal stage of the relevant cycle 'Completing self-assessment against the Core Competency Framework'.
I completed the CCSAT in January 2023 and completed a CPD cycle based on the outcome in July 2023. Can I submit this as a cycle which originated from my selfassessment against the PSI Core Competency Framework for assessment in the 2024/25 ePortfolio Review?
The 2024/25 ePortfolio Review Standards require that you submit one cycle created following selfassessment against the PSI’s Core Competency Framework in the current year, therefore the cycle submitted to meet this standard must have a creation date in 2024 to the end of the submission period in January 2025. This is in line with the legislative requirement to regularly self-assess against the Core Competency Framework. A cycle created in 2023 will not meet this standard, however, you may wish to submit this cycle as evidence of one of the cycles that you created in the previous four years.
I have completed some of the CPD training programmes available through Irish Pharmacy News (IPN) and I have also completed IIOP online training programmes, are there other activities that I could record in my ePortfolio?
Many pharmacists are surprised to realise the breadth of what counts as CPD. Traditionally, many pharmacists focus their CPD on their clinical expertise. The PSI Core Competency Framework indicates that pharmacists must be competent across a range of domains. It is important to aim to keep upskilled across all domains – not just those domains relating to medicines. This requires engaging in a breadth of learning.
Consequently, there are many different types of activities that can be recorded in your ePortfolio as CPD. Completing the IPN CPD modules, for example, is a very valuable means of undertaking CPD. Other examples of activities that can be recorded in your ePortfolio as CPD include:
• Attending a live learning course
• Reading an article
• Having a discussion with a colleague
• Attending a conference
• Researching a new drug that has been prescribed for a patient It is important to remember that recording any learning activity may be appropriate as long as you can demonstrate how it has contributed to your professional development. It may be helpful to refer to the 2024/25 ePortfolio Review Standards (available on the ePortfolio Review Support Resources page on the IIOP website) to see types of criteria within the scope of the Review.
How much information should I record in my CPD cycles?
There is no right or wrong answer to this. Each pharmacist will have their own style; some choose to use bullet point information, while others prefer to be more detailed. What is important, however, is that you sufficiently outline what you have learned and how that learning has contributed to your understanding, benefited your practice, improved patient outcomes or advanced your continuing professional development. You will find some sample CPD cycles on the IIOP website, in IIOP newsletters or by attending ePortfolio Review information events. You should write in a way that works for you and allows you to reflect on what you have done.
What happens if my ePortfolio extract does not meet the 2024/25 ePortfolio Review Standards?
The ePortfolio Review enables pharmacists to demonstrate evidence of appropriate and ongoing engagement with CPD, in the interest of outcomes-focused professional development. All pharmacists’ ePortfolio extracts are reviewed against the specific standards, mapped to the legislative requirements, which have been set for that year’s ePortfolio Review.
All pharmacists who submit their cycles within the submission period in January 2025 will be provided with feedback on their ePortfolio extract, should one or more of the standards not be met in the first instance. They will then have an opportunity to resubmit cycles based on this feedback to meet the standards.
In the event that a pharmacist does not meet the standards at the end of their first ePortfolio Review process, they are automatically entered into the following year’s ePortfolio Review. The PSI is not informed at this stage. However, if at the end of the second ePortfolio Review the pharmacist does not meet the standard or does not reengage with this process in year 2, the IIOP is obliged under the terms of the ePortfolio Review Policy to refer the pharmacist to the PSI. Will the PSI be aware of how I perform in the ePortfolio Review process?
The IIOP undertakes the ePortfolio Review process to enable pharmacists to demonstrate evidence of CPD, in line with the legislation. Whilst the PSI is responsible for selecting pharmacists for ePortfolio Review, the IIOP undertakes the review process itself, at arm’s length from the PSI. The IIOP will not share any aspect of a pharmacist’s ePortfolio with the PSI.
In the event that the IIOP cannot ascertain that a pharmacist is meeting his or her CPD obligations (i.e. if the standard is not met at the end of two years ePortfolio Review, or if the pharmacist fails to submit an ePortfolio extract for review within the timeframe provided) then the IIOP has a statutory obligation to refer the pharmacist to the PSI. No information from your cycles is communicated to the PSI.
When I was selected for ePortfolio Review, I didn’t apply for an exemption due to extenuating circumstances, but
my circumstances have now changed and I will be unable to submit my ePortfolio. Is it too late to apply for an exemption?
The PSI manages the selection process for ePortfolio Review including all applications for exemptions from ePortfolio Review under its Extenuating Circumstances process. If circumstances apply to you which would have an impact on your ability to submit an extract from your ePortfolio, you should contact the PSI. The PSI's Extenuating Circumstances Policy and the relevant application form are available on the PSI website. All applications submitted will be managed by the PSI on a confidential case by case basis.
Where can I get more information?
The ePortfolio Review Support Page can be accessed via the IIOP homepage and is the main source of information relating to the 2024/25 ePortfolio Review including information on communication from the IIOP, timelines and ePortfolio Review Information Events. The ePortfolio Review support resources are also hosted on this page.
You may also wish to attend an Information Event. The IIOP hosted a series of Information Events from September 2024 until January 2025 to support pharmacists selected for the 2024/25 ePortfolio Review. All events are facilitated by a Peer Support Pharmacist and specifically focus on the key requirements of the 2024/25 ePortfolio Review so that pharmacists will know exactly what to expect at each point in the process. Pharmacists who took part in previous ePortfolio Review processes consistently highlight the IIOP Information Events as a key source of support in their preparation. They felt more confident in participating in ePortfolio Review and had a clear understanding of what the standards were and how to access support if needed, after attending an event. There are two information events (webinars) scheduled on 8 and 21 January 2025. You can book via the IIOP website, under the ‘Courses & Events’ tab. There is also a recorded version of the webinar available to view in your own time on the ePortfolio Review support resources page on the IIOP website.
The IIOP team are always happy to hear from you with any queries you may have and you can contact the team on info@iiop.ie
Minister for Further and Higher Education, Research, Innovation and Science, Patrick O’Donovan TD, recently announced the launch of Atlantic Technological University’s (ATU) new Master of Pharmacy programme.

“A new Master of Pharmacy programme will help to address the significant shortage of pharmacists in our health services and will offer students a new opportunity to study pharmacy in the Northwest.”
This significant development expands ATU’s educational offerings in the pharmaceutical and healthcare sectors.
Pending accreditation by the Pharmaceutical Society of Ireland (PSI), the new Master of Pharmacy programme will begin in September 2025, initially offering 36 places in the five-year, full-time course. The programme will provide graduates with the qualifications required to register as practising pharmacists in Ireland.
The announcement by the Minister in Limerick, is part of a major increase in healthcare places in Ireland through the creation of six new programmes. Funding of ¤130 million unlocked from the National Training Fund will support the growth of key healthcare disciplines, addressing critical workforce shortages and enhancing healthcare education across the country.
As part of this initiative, following recommendations from the Higher Education Authority (HEA), several universities will establish new programmes in areas of Medicine, Pharmacy, and Dentistry. At an event to mark the announcement in UL today, Minister O’Donovan said: “I am excited to see the impact these new projects will have on the health sector and
those who avail of their services. They truly have the potential to revolutionise our higher education landscape and provide more opportunities for students to follow their passions in healthcare and medicine. University of Limerick will introduce a direct entry medicine programme, the Royal College of Surgeons will receive support for their Dental Surgery programme, University of Galway will introduce a Rural and Remote Graduate Entry Medicine stream, and three new pharmacy programmes will be introduced in Atlantic Technological University, South East Technological University, and University of Galway. This will double the number of pharmacy programmes in Ireland and make pharmacy education available in new regions. And I’m pleased to say we are not finished: this is only the first round of development. My officials are working with the HEA and the higher education sector to progress further expansion to meet the needs of the public sector and the nation.”
HEA CEO Dr Alan Wall said: “Today’s announcement demonstrates the ability of the higher education and research system to respond swiftly and effectively to evolving national priorities and skills needs. The
Dr Marita Kinsella, Head of Department of Pharmacy, Atlantic Technological University (ATU), Patrick O’Donovan, Minister for Further and Higher Education, Dr Orla Flynn, President of ATU, Dr Thomas Smyth, Head of Faculty of Science, ATU, Sligo Campus
New research on the impact of obesity medications on alcohol intake has found that these medications significantly reduce alcohol use – including by up to two thirds among heavy drinkers.
Research carried out by Medication Weight Loss Clinic in Dublin quantified changes in alcohol use when patients with the disease of obesity were treated with the obesity medications liraglutide and semaglutide.
new programmes announced today reflect the commitment of institutions to meeting society’s needs and to enhancing the range of programmes on offer to prospective students.”
The new Master of Pharmacy programme is designed to be future-focused, integrating clinical pharmacy practice with cutting-edge healthcare and pharmaceutical sciences research. It will develop graduates with the expertise required for contemporary pharmacy roles and to take on the expanded responsibilities that pharmacists now have in patient care. The programme will also include 12 months of structured work placements, offering students practical experience across community, hospital, and industrial pharmacy settings.
Speaking on the impact the new programme will have, Marita Kinsella, Head of ATU’s new Department of Pharmacy, highlighted its potential to address the growing shortage of pharmacists in Ireland, “In a time of significant advancement for the pharmacy profession, as pharmacists take on expanded roles to enhance health services and patient care, ATU is keen to play its part. A new Master of Pharmacy programme will help to address the significant shortage of pharmacists in our health services and will offer students a new opportunity to study pharmacy in the Northwest.”
Marita also emphasised the importance of collaboration within the pharmacy sector, noting that ATU will work closely with the pharmacy profession, industry leaders, healthcare professionals, and other academic institutions to ensure the programme meets the highest standards of education and practice. With the introduction of this new course, ATU is set to play a vital role in shaping the next generation of pharmacists in Ireland.
Both medications are taken by injection either daily or weekly and are already proven highly effective for treating obesity and also type 2 diabetes. On average alcohol intake among those being treated reduced from 12 to 4 units per week. No patient taking the medications experienced an increase in alcohol intake.
Intake among heavier drinkers in the research decreased from 23 units to 8 units per week, a two thirds reduction which is an equivalent change from 12 pints to 4 pints. Patients also lost 8kg of weight in the first four months with a positive association between alcohol reduction and weight loss.
The research and evaluation was carried out by the Medication Weight Loss Clinic Lead and GP Dr Maurice O’Farrell between January 2023 to March 2024 involving 262 adults living with obesity. The findings are published this month in the Diabetes Obesity and Metabolism Journal
Responding to the research Dr Farrell said: “The significant reduction in alcohol intake among patients treated with obesity medications has important clinical implications for public health in Ireland. The ability of these medications to reduce both alcohol intake and body weight suggests a potential role for these medications in patients with cooccurring obesity and alcohol use disorder.”
More than 1500 people in Ireland die per year because of alcohol. Treatments for alcohol use disorder include behavioural interventions, motivational interventions, and medications, but relapse rate is 70% within the first year of treatment.




























































In the ever-evolving world of pharmacy and healthcare, the individuals who drive innovation, demonstrate leadership, and inspire progress deserve to be celebrated. We are delighted to celebrate the 19 PHX Ireland colleagues who have been recognised in this year’s Dynamic 100. These nominations highlight those who are shaping the future of the pharmacy industry, and we are thrilled to see so many of our own teammates among this distinguished group.

PHX Ireland Group Limited comprises United Drug, Ireland’s leading pharmaceutical wholesaler and distributor; McCabes Pharmacy, Ireland’s largest, most patient centric retail pharmacy brand with 110 pharmacies across Ireland (including +80 previously LloydsPharmacy stores); and TCP Homecare, innovators in clinical home nursing services and direct to patient delivery solutions.
At PHX Ireland, we are committed to delivering excellence across every part of our organisation, and these individuals exemplify the values, expertise, and dedication that drive us forward. From leadership and operations to retail and pharmacy care, their contributions are pivotal to our success and the health of the communities we serve.
Recognising Our Dynamic 100 Nominees
We celebrate the following colleagues for their exceptional achievements and contributions:
• Paul Reilly, CEO, PHX Ireland –A leader driving innovation and growth across our organisation.
• Elaine Condon, MD, TCP Homecare – Transforming patient care through specialised homecare solutions.
• Sharon McCabe, CEO, McCabes Pharmacy – Leading one of Ireland’s most trusted pharmacy brands with unwavering dedication.
• Hillary Collins, People & Culture Director, PHX Ireland – Championing a culture of excellence and employee wellbeing.
• Laura Reynolds, Pharmacist, McCabes Pharmacy Rathmines – Delivering outstanding patient care at the community level.
• Anna Cotter, Pharmacist, McCabes Newcastle –Exemplifying clinical expertise and compassionate care.
• Rajesh Purcell, Head of Operations, United Drug Wholesale – Ensuring operational efficiency and excellence.
• Michael Taylor, Head of Sales and Retail Solutions, United Drug Wholesale – Driving retail innovation and building relationships to grow customer business.
• Sonya Lynch, Key Account Manager, United Drug Wholesale – Building strong partnerships with clients and stakeholders.
• Orla Doyle, Area Manager, United Drug Wholesale –Supporting customers in growing their business.
• Orlagh Dunne, Sales Manager, United Drug Wholesale –Delivering exceptional results through strategic sales leadership.
• Adrian O’Sullivan, Buying Groups Manager, United Drug Wholesale – Developing the buying groups’ proposition for customers.
• Alan Franklin, ARC Claims Manager, United Drug Wholesale – Streamlining claims processes with precision and care.
• Reggie Ismailov, Solutions Manager, United Drug Wholesale – Providing solutions to meet evolving customer needs.
• Karen Doherty, General Manager, McCabes Pharmacy – Steering retail operations with excellence and insight.
• Marina Timotijevic, Marketing Manager, McCabes Pharmacy – Elevating brand presence and customer engagement.
• Aine Martin, Head of Operations, McCabes Pharmacy (formerly LloydsPharmacy) – Leading operational transformation and growth.
• Ann Marie Broderick, Retail Operations Lead, McCabes Pharmacy (formerly LloydsPharmacy) – Optimising retail operations with expertise.
• Sarah Kenny, Retail Operations Scheduling Manager, McCabes Pharmacy (formerly LloydsPharmacy) – Ensuring seamless scheduling and operational efficiency.
A Collective Achievement
This recognition is not just an individual accomplishment but a reflection of the collaborative spirit that defines PHX Ireland. It underscores the strength of our team, the breadth of our expertise, and our unwavering commitment to delivering health to the communities across Ireland. We are incredibly proud of each nominee and grateful for their dedication to excellence. Their recognition in the Dynamic 100 is a testament to their hard work and commitment.
Congratulations to Ruth Maher, 3rd year pharmacy student at the School of Pharmacy, University College Cork, who presented her research poster at the College of Medicine and Health Research Conference "Futures 2024: Transforming Tomorrow's Health" on 12th December.

Dr Kieran Dalton, Ruth Maher and Dr Suzanne McCarthy, UCC School of Pharmacy
Her poster, titled "Community Pharmacists' Views on Extending Prescription Duration in Ireland: A Survey Study", explores the recent change in practice allowing pharmacists to extend prescriptions from 6 to 12 months. The national survey, which received responses from over 750 pharmacists in #Ireland, provides
key insights into the perspectives of pharmacists on this development, which should guide the planning and implementation of expanded pharmacist roles to address the rising demand for healthcare in Ireland.
Ruth's research was funded by a Summer Undergraduate Research Experience (SURE) Award and was supervised by Dr Kieran Dalton and Dr Suzanne McCarthy from the School of Pharmacy.
Delivering best-in-class products, service and business support for optimum patient care for your customers
Choice Based Buying Group.
Range of quality products including a mix of OTC, branded, generic and cold chain.
Safe and secure sourcing of unlicensed medicines. Bespoke medicines tailored to patients needs.
Ireland’s No1 provider of Ostomy products and services.
Best in class advice, service and support, enabling pharmacists to give patients confidence and peace of mind.
Compliance Based Buying Group
Rewarding you with competitive pricing.
Insights & Education.
Providing meaningful insights to help drive business forward and deliver better patient care.
Call: 01 463 2300
Email: wholesale@united-drug.com
Visit: www.united-drug.com
Login: www.udw.ie
Théa Pharma Ireland is proud to announce the winners of the second annual "A Feast for Your Eyes" art competition, held at their Country’s HQ in Castlebar. This inspiring event brought together more than 30 talented Transition Year students from across County Mayo, who showcased their creativity and artistic flair while raising awareness about the vital importance of eye health.
The competition, now in its second successful year, encouraged students to express their artistic talents and highlight the beauty of vision through their work. The top eight artists were celebrated at a special event, where the winners were officially announced:
1st Prize - ¤1000 for Molly Moran from Balla Secondary School
2nd Prize - ¤500 for Valentina Bezares from Balla Secondary School
3rd Prize - ¤250 for Grace Killeen from Gortnor Abbey, Crossmolina
The day was filled with creativity, excitement, and celebration as students, teachers, and families gathered to honour the hard work and passion behind each piece of art. Speaking at the event, Théa Pharma Ireland’s Country Manager, Diarmuid Gavin, expressed his admiration for the participants: "The talent displayed today has been nothing short of extraordinary. These young artists not only demonstrated their remarkable creativity but also helped shine a light on the importance of eye health in such a unique and meaningful way. At Théa Pharma, we believe that fostering awareness about vision care among young people is key, and this competition perfectly merges education with art. Shining a light on Art and celebrating excellence in its creation, gives recognition to students who may often escape the limelight. I would like to congratulate all the participants, especially our winners, for their outstanding work. We look forward to seeing this initiative grow even further in the years to come.
Tina Heraty, competition Judge, was amazed by the talent of display and noted the extraordinary inspiration and uniqueness behind each individual piece. The "A Feast for Your Eyes" art competition serves as part of Théa Pharma Ireland’s ongoing commitment to raising awareness about eye health and fostering creativity in local communities.
Théa Pharma is Ireland’s number one choice for preservative-free eye care. Dedicated to enhancing vision health, Théa Pharma continues to innovate and engage with local communities through meaningful initiatives like "A Feast for Your Eyes".

The competition, now in its second successful year, encouraged students to express their artistic talents and highlight the beauty of vision through their work. The top eight artists were celebrated at a special event, where the winners were officially announced
Competition Judge Tina Heraty and Diamuid Gavin, Théa Pharma Ireland Country Manager, pictured with Molly Moran – 1st prize winner


Written by Dr Deirdre McDermott, Respiratory Registrar, Galway University Hospital and Dr Sinead Walsh, Consultant Respiratory Physician, Galway University Hospital & Galway City Integrated Care Hub
Asthma is one of the most common chronic respiratory diseases in Ireland, with approximately 1 in 10 people affected by this condition. Globally, asthma impacts millions of people, making it a leading cause of morbidity and healthcare utilisation worldwide. Managing asthma effectively requires a comprehensive, multi-faceted approach that engages not only healthcare providers but also patients, their families, and the wider community. In Ireland, asthma management has evolved significantly in recent years, with community-based care playing a pivotal role in improving health outcomes for individuals with asthma.
The Global Initiative for Asthma (GINA) provides guidelines that emphasise the importance of self-management education for asthma patients. According to GINA, asthma patients need comprehensive education on inhaler use, medication adherence, symptom monitoring, and the development of asthma action plans. Effective asthma management relies on empowering patients to make informed decisions about their care, including when to use medications and how to recognise when their asthma is under control or worsening.
Sláintecare is a national reform program aimed at improving healthcare access and equity in Ireland. It focuses on providing care closer to home and ensuring that healthcare services are more accessible to all citizens, particularly those in rural or underserved areas. Under this reform, new Regional Health Areas (RHA) have been established to offer more equitable access to healthcare services across the country. Within these RHAs, Community Healthcare Networks (CHN) have been developed. (Sláintecare - the Strategy for Improving Ireland’s Healthcare System - About the HSE, n.d.).
A key component of Sláintecare is the Integrated Care Programme for the Prevention and Management of Chronic Disease (ICPCD). This initiative aims to improve the care and management of patients living with chronic conditions,
including asthma. Other chronic conditions included in the programme are COPD, diabetes and cardiovascular disease.
The ICPCD model focuses on integrating healthcare services across primary, secondary and community care, providing a patient-centred and co-ordinated approach. The model prioritises prevention, early detection, and self-management, enabling patients to receive appropriate care closer to home, reducing hospital admissions, and improving overall health outcomes. For asthma patients, this involves utilizing local services for ongoing monitoring, education, and preventative care, which greatly reduces the need for more intensive, hospitalbased interventions. (Federman et al., 2019).
One of the cornerstones of the ICPCD is the End to End Model of Care (MOC) for adults with asthma. This model of care focuses on providing comprehensive asthma management through integrated community solutions. The MOC spans a wide range of services, from primary prevention to more specialised care. The goal is to ensure that asthma patients receive continuous, appropriate care at the most suitable level, preventing unnecessary hospitalisations and improving long-term disease control. The MOC encourages patient empowerment, ensuring that individuals with asthma are involved in decision-making processes regarding their health and treatment. This model encourages patients to manage their condition independently whenever possible, reducing the need for specialist interventions and fostering greater control over their own health.
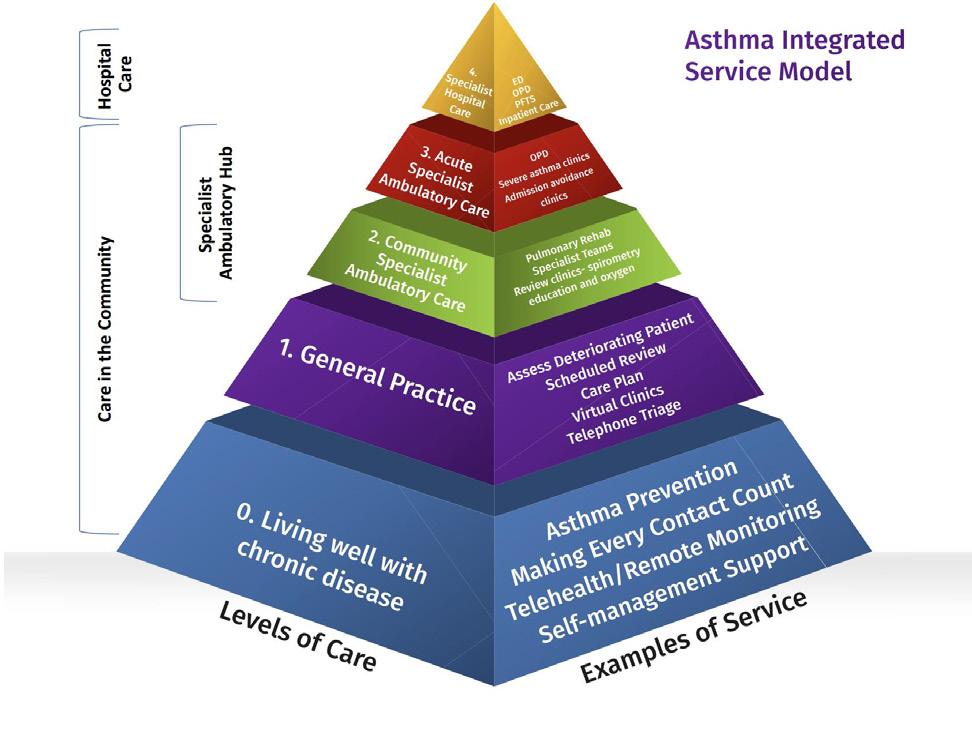
The MOC for asthma care in Ireland follows a pyramid structure, with the bulk of asthma management occurring at lower levels of care in the community setting. At Level 0, the majority of asthma patients live well with their condition and require minimal intervention. These patients are typically able to manage their symptoms with the support of general practitioners (GPs) and community healthcare services. Level 1 care involves management by GPs, who play a vital role in
monitoring asthma symptoms, adjusting medications, and providing ongoing support. At Level 2, patients may be seen in Community Specialist Ambulatory Hubs, where they can access more specialised care and diagnostic services without the need to visit a hospital. Level 3 care involves acute specialty ambulatory services, where patients who require more intensive care are treated, and Level 4 care is reserved for patients who need specialty hospital care.(Model of Care - HSE.ie, n.d.).
The pyramid approach ensures that most asthma patients are managed in community settings, with specialist interventions provided only when necessary. This structure emphasises the need for community-based care, which can prevent exacerbations and ensure that patients receive timely, appropriate care that is closer to home. It also highlights the importance of multi-disciplinary teams (MDTs), which involve a range of healthcare professionals working together to provide comprehensive care. These teams may include GP’s, respiratory physiologists, respiratory specialists, physiotherapists, and nurses, who collaborate to deliver personalised asthma management plans, including self-management strategies and action plans. Specialist Integrated Care Hubs have been

developed within the CHN’s, which offer a range of services for chronic disease management. In terms of asthma, diagnostic spirometry & FeNO testing is carried out. Consultations with asthma specialists, including the respiratory consultant and specialist respiratory nursing staff are available. After confirming the diagnosis, patients can receive educational support on their asthma self-management, including the use of peak flow meters, inhaler techniques, and the development of a personalised asthma action plan. The integrated care hubs aim to provide a “onestop-shop” for patients with asthma, where they can access both specialist care and education in a convenient, community-based setting. By bringing respiratory care out of hospital settings and into local communities, these hubs improve access to care and reduce the need for hospital visits. (Respiratory - HSE.ie, n.d.) The importance of communitybased care in managing asthma cannot be overstated. Most asthma patients in Ireland receive their care within community settings, making it essential to ensure that these services are robust, accessible, and effectively integrated into the healthcare system. Asthma management involves ongoing monitoring, education, and personalised care, all of which can be effectively addressed within a community-
based framework. By offering regular check-ups, educational resources, and support systems, community care can significantly reduce emergency interventions and hospitalisations while improving patients’ quality of life.
A key component of communitybased asthma management is the GP Chronic Disease Management Programme. For adult patients with asthma who have a medical card or GP visit card, they can attend their GP practice to obtain a structured review of their asthma with the GP or practice nurse. The chronic disease management programme allows patients to avail of a personalised care plan, and a review of existing care plans and inhalers. If a referral to a specialist is required, the integrated care hubs are there in the community, close to where the patient lives, to provide this specialist service.
Globally, the importance of community healthcare workers in asthma management has been widely recognised. The Global Strategy for Asthma Management and Prevention underscores the role of community healthcare workers in providing education and support to asthma patients. Asthma self-management education, delivered by trained community health workers, has been shown to improve patient outcomes and reduce healthcare
utilization. In many cases, community healthcare workers can deliver educational programs, monitor symptoms, and help patients develop asthma action plans, ensuring that individuals with asthma receive the support they need to manage their condition effectively.(2024 GINA Main Report - Global Initiative for Asthma - GINA, n.d.). The success of these community-driven initiatives demonstrates the critical role of local healthcare workers in ensuring asthma patients receive the education, tools, and support necessary for managing their condition independently. This approach can reduce reliance on acute healthcare services, such as emergency department visits or hospitalisations, by promoting proactive care and selfmanagement. By teaching patients how to recognise early warning signs of asthma exacerbations, adjust their medications, and manage their symptoms effectively, community healthcare workers play a crucial role in improving asthma control.
One of the key aspects of asthma self-management is the ability to monitor asthma symptoms and peak expiratory flow (PEF). PEF monitoring allows patients to track their lung function and recognise early signs of asthma exacerbations. This empowers individuals to seek timely
Asthma Integrated Service Model
medical attention and adjust their medications, helping to prevent more severe asthma episodes and reducing the need for hospital interventions. Moreover, asthma self-management education helps patients understand the importance of medication adherence, the correct use of inhalers, and avoiding triggers that can worsen asthma symptoms. Through education and ongoing support, patients can feel more confident in managing their condition independently, which leads to better asthma control and better patient wellbeing.
In both Ireland and internationally, the integration of asthma management into community care systems has been shown to improve outcomes and reduce healthcare costs. Community care not only improves access to care but also empowers patients, reduces hospitalisations, and enhances the overall quality of life for individuals with asthma.
In Ireland, initiatives such as the End to End Model of Care and Specialist Integrated Care Hubs reflect the global recognition of the importance of community-based asthma care. These initiatives align with global strategies, such as those proposed by GINA, that emphasise the role of education, self-management, and communitybased healthcare workers in improving asthma control.
In conclusion, a communityfocused approach is central to the effective management of asthma, both in Ireland and internationally. As asthma rates continue to rise worldwide, fostering communitybased solutions will be critical to ensuring that individuals with asthma receive the support they need to manage their condition effectively. Community care is essential not only in reducing the burden on hospital systems but also in providing timely, personalised care that meets the unique needs of each patient. As the burden of asthma continues to grow, fostering community-based solutions will be essential in improving asthma care, enhancing patient outcomes, and creating a more sustainable healthcare system.
References:
2024 GINA Main Report - Global Initiative for Asthma - GINA. (n.d.). Retrieved December 13, 2024, from https://ginasthma.org/2024report/
Federman, A. D., O’Conor, R., Mindlis, I., Hoy-Rosas, J., Hauser, D., Lurio, J., Shroff, N., Lopez, R., Erblich, J., Wolf, M. S., & Wisnivesky, J. P. (2019). Effect of a Self-management Support Intervention on Asthma Outcomes in Older Adults: The SAMBA Study Randomized Clinical Trial. JAMA Internal Medicine, 179(8), 1113–1121. https://doi.org/10.1001/ JAMAINTERNMED.2019.1201
ICP for Prevention and Management of Chronic DiseaseHSE.ie. (n.d.). Retrieved December 13, 2024, from https://www.hse.ie/ eng/about/who/cspd/icp/chronicdisease/
Model of Care - HSE.ie. (n.d.). Retrieved December 13, 2024, from https://www.hse.ie/eng/ about/who/cspd/ncps/ncpr/ asthma/moc/
Respiratory - HSE.ie. (n.d.). Retrieved December 13, 2024, from https://www.hse.ie/eng/ about/who/cspd/ncps/ncpr/ Sláintecare - the strategy for improving Ireland’s healthcare system - About the HSE. (n.d.). Retrieved December 13, 2024, from https://about.hse.ie/our-work/ slaintecare-our-strategy-forimproving-irelands-healthcaresystem/
To maximise treatment outcome remind your customers to:
Apply LAMISIL ONCE to both feet
Spread evenly on toes, sole and sides of feet
Do not massage or rub
Do not wash feet for 24 hours


Unique, patented film forming technology:
2.5 x
Enhances Terbinafine penetration by 2.5 times*
Days 13
Creates a reservoir that prolongs the fungicidal e ect of Terbinafine for up to 13 days**

Find out how McLernons can help you protect your business from malicious software by improving your cyber security…
What is Cyber Security?
Cyber security is the protection of devices, services and networks and the information on them from theft or damage. It’s essential for providing effective care, protecting your patients and maintaining their trust in the pharmaceutical services you provide.
Richard Horne, Head of GCHQ’s National Cyber Security Centre (NCSC) issued the NCSC Annual Review in December 2024 and said, “What has struck me more
forcefully than anything else is the clearly widening gap between the exposure and threat we face, and the defences that are in place to protect us. And what is equally clear to me is that we all need to increase the pace we are working at to keep ahead of our adversaries.”
“There is no room for complacency about the severity of state-led threats or the volume of the threat
posed by cyber criminals. The defence and resilience of critical infrastructure, supply chains, the public sector and our wider economy must improve.
“In the past year, we have seen crippling attacks ag ainst institutions that have brought home the true price tag of cyber incidents.
“The attack against Synnovis showed us how dependent we are on technology for accessing our health services.”
The NHS in England suffered months of disruption in 2024 after Synnovis, a London-based provider of pathology services, was hit by a ransomware attack in June. The attack, claimed by the Qilin ransomware group, left patients unable to get blood tests from their doctors for more than three months, and led to the cancellation of thousands of outpatient appointments and more than 1,700 surgical procedures.
Meanwhile in the US, the number of cyber attacks on the healthcare sector have doubled in the last two years.
Anne Neuberger, the US deputy national security advisor for cyber and emerging technology, highlighted the growing problem of ransomware in the healthcare industry and pointed out how healthcare data is increasingly being traded on the dark web, "with the opportunity to blackmail individuals."
The proposals follow several high-profile data breaches in the sector, exposing the data of hundreds of millions of Americans and disrupting treatment. In February 2024, a ransomware attack on UnitedHealth subsidiary Change Healthcare reportedly exposed the personal data of more than 100 million people and paused pharmacy services and billing.
The company’s handling of the hack — which is believed to have been caused by a breach of a basic user account which did not use multi-factorial authentication (MFA) was criticized by Americans who couldn’t get their medications filled or hospital stays approved, affected healthcare providers who were going broke as a result of the cyberattack,
In May 2024, healthcare provider Ascension was hit with a cyberattack that brought down the IT systems at many of its hospitals, in some cases forcing doctors to use pen-and-paper records.
YOU are the First Line of Defence
Pharmacists are particularly vulnerable to cyber-attack, and the devastating consequences that it could have on your business and your patients. Your IT systems are at the very heart of the way that you work, holding confidential patient details and dispensing histories and essential business information.
An attack could halt your business operations and severely damage your reputation, not to mention incurring potential fines from the Information Commissioner. While McLernons can advise and provide information the responsibility for cyber security ultimately rests with you. We can provide cloud backups of your pharmacy data so that it can be restored, but we are not responsible for putting in place the measures needed to protect it from cyber-attack. There are a number of measures which we recommend but it is up to the individual contractor to decide on these and implement them.
Failing to secure your systems can result in GDPR fines of up to £20 million or 4% of your global turnover. Setting aside any GDPR consequences, the loss of reputation will have huge consequences for your business. And should you be the target of a cyberattack you may be asked to pay a ransom to have your data returned to you.
Used to provide overall network security. Allows you to have a number of separate networks, so that systems such as your dispensing system can be isolated from all other networked devices, including card payment units, CCTV systems, personal staff devices etc. Firewalls will also be configured to restrict access to potentially malicious websites or specific website ‘categories’.
This gives you the various Office applications and a secure business email account. Using Hotmail, Google and other generic email addresses vastly increases your exposure to a possible cyber security attack. This can be augmented by the use of an additional email gateway defence system, such as Proofpoint Essentials, which can isolate and quarantine suspicious emails, or block emails from previously identified suspicious addresses.
When we are upgrading old systems, we strongly recommend that our customers choose a new, robust password. Restrict access according to staff roles to protect patient data and comply with GDPR.


McLernons have implemented what we believe to be the most comprehensive back up and recovery solution. Cove Data Protection offers hourly MPS and MPS Retail back ups - giving you 24/7 peace of mind in the event of system failure, ransomware attack or natural disaster – and it also backs up all of your personal files and folders to a secure cloud repository.
We have rolled out what we believe is the best available EPDR (Endpoint Protection, Detection and Response) , which uses Artificial Intelligence to scan and classify potential threats. This contrasts with legacy AntiVirus which only recognises existing known threats. This service is provided by WatchGuard
In the month of December, our EPDR programme monitored over 4.5 million interactions and detected and blocked 6573 intrusion attempts on almost 6000 McLernons’ customers’ systems that it protects.
We manage your Windows updates, providing patches for all supported devices. Windows 7 has been ‘end of life’ since 2020 and Windows 10 ‘end of life’ is October 2025. We are working hard on your behalf to upgrade your hardware and protect your data, your patients and your businesses.
As your Pharmacy IT systems partner we are here to support you. We can help, advise and
recommend steps that you should take to minimise the risk but ultimately the responsibility vests in you.
There are many layers of security that you overlay on your IT systems but these won’t prevent them from being hacked down the line if software is not updated, and the basics of cyber security are ignored.
As a bare minimum, we recommend that all pharmacies have a robust firewall, a proven back-up system and have upgraded their hardware from obsolete and out-of-date systems, such as Windows 7 machines. If you have any queries, please do not hesitate to contact us on ITSecurity@mclernons.ie
Common Security Vulnerabilities in Pharmacies
Are you guilty of any of the following...
• Using a ‘free’ insecure email account
• Allowing unfiltered web access on your pharmacy systems
• Using Default System Passwords
• Using Unsupported Windows Operating Systems
• Using Legacy Hardware
• Inadequate Anti-Virus Software
• Lacking a hardware firewall
• Using open or insecure WiFi
• Lacking staff cyber security training
IF YOU HAVE ANSWERED YES TO ANY OF THESE, THEN YOUR IT SYSTEMS ARE AT RISK
New research from RCSI University of Medicine and Health Sciences has explained how the body’s internal clock influences the inflammatory process of the immune system. The findings describe how immune cells, called macrophages, work differently at various times of the day and could pave the way for time-targeted treatments for inflammatory diseases such as arthritis.

The researchers explored the link between the immune system and the body’s circadian rhythms often referred to as the body clock. Macrophages, immune cells that detect and respond to harmful substances, are able to trigger inflammation as a defence mechanism by assembling large complexes known as
inflammasomes. Inflammasomes could be compared to ‘smoke detectors’ that will then alert the immune system of danger.
Activation of an inflammasome called NLRP3 was not found to be constant throughout the day but was regulated by the body’s 24-hour circadian clock.
Professor Annie Curtis
This daily rhythm determines when macrophages are most efficient at detecting threats and when their energy levels peak to mount a response. The research also highlights a key role for mitochondria, the cell’s energy producers, in driving these daily changes in immune activity.
“When macrophages ‘think’ it’s morning, their inflammasome activation is quicker and more robust,” explained Professor Annie Curtis, principal investigator for the
study at RCSI School of Pharmacy and Biomolecular Sciences. “This means the immune response is heightened during the early part of the day, a time when we are awake and more likely to encounter environmental challenges, such as injuries or infections.”
The study has significant implications for understanding and treating inflammatory diseases, such as arthritis, where overactive inflammasomes play a key role. Symptoms of such diseases often worsen in the morning, something this research may help explain.
“With these findings, there’s potential to refine treatments for inflammatory conditions,” said Dr James O’Siorain, lead author of the study. “For instance, new therapies targeting inflammasomes could be more effective if administered at specific times of the day when macrophage activity peaks.”
'Time of day control of mitochondria regulates NLRP3 inflammasome activation in macrophages' is published in The FASEB Journal. The research was supported by funding from Taighde Éireann – Research Ireland.
Almost a quarter of people who used the Irish Heart Foundation’s Mobile Health Unit to have a heart health check were advised to see their GP for further assessment, a new report reveals.
A blood pressure reading was so high in one case that after being examined by their doctor, one patient ended up in A&E, according to an evaluation of the Irish Heart Foundation’s Mobile Health Unit.
The charity’s nurse-led vehicle, supported since 2022 by Pfizer Healthcare Ireland, travels the country offering free heart health checks, including blood pressure and pulse checks as well as a lifestyle assessment. In 2024, 6,000 free heart health checks were given to communities and groups across 26 counties.
Researchers from South East Technological University (SETU) evaluated its effectiveness and found that dozens of patients needed further check-ups when
the health check nurse identified signs of high blood pressure or hypertension.
The ‘silent killer’ is a leading cause of cardiovascular disease, the leading cause of death globally.
High blood pressure is often present without symptoms and the World Health Organisation estimates that 46% of adults have it but are unaware of it.
A sample of 300 men and women who used the Mobile Health Unit across Ireland showed, more than a fifth (22.1%) were found to have high blood pressure, while almost double that number (42.9%) were deemed to be higher than normal.
A worryingly high 39.5% reported having no prior awareness of their blood pressure levels and almost
a quarter (23.2%) were signposted to their GP based on the Mobile Health Unit readings.
Six weeks later, 182 of participants completed a followup questionnaire over the phone, which also revealed that of those who were signposted to their doctor, 28.2% underwent additional testing recommended by their GP, such as using an ambulatory blood pressure monitor.
A total of 10.3% received a diagnosis of high blood pressure and began taking medication, while 2.6% went to the emergency department due to dangerously high blood pressure levels.
The Mobile Health Unit checks also inspired reported lifestyle changes among almost half, with 20.9% making dietary improvements,
almost 20% becoming more physically active, a further 1.1% quit smoking and the same proportion reduced or abstained from alcohol.
Janis Morrissey, Director of Health Promotion, Information and Training with the Irish Heart Foundation said the findings underline the importance of the charity’s work in identifying people at high risk of heart disease and stroke.
“The findings from the SETU report are crucial, as 80% of premature heart disease and stroke is preventable,” she said.
“The Mobile Health Unit service plays a vital role in identifying people at risk of heart disease and stroke.

Parents and caregivers are being encouraged to familiarise themselves with the symptoms of respiratory syncytial virus (RSV) as the highest numbers of infections are usually reported in December and January every year.

RSV is a common virus that causes coughs and colds and may cause infection in the lower part of the respiratory system. These are known as lower respiratory tract infections (LRTIs).
A recent online survey was funded and conducted by Sanofi among 500 adults who are either currently pregnant or who have children under the age of four in Ireland to understand their level of awareness and knowledge of RSV. The survey results revealed that just over half (51%) of Irish parents had heard of RSV and understand the risk it poses to infants and babies, but almost 1 in 5 (19%) have not heard of RSV.
Approximately 90% of children are infected with RSV by two years of age, with RSV being the leading cause of LRTIs in babies. RSV is a seasonal virus and is spread by coughs and sneezes or close physical contact.
In Ireland, the RSV season is generally from early October, with peak outbreaks typically occurring in the winter months.
Dr Máire Finn, GP at Ennis Centric Health said, “Most RSV infections in babies are mild and will clear up within a few weeks. However, some cases can be more serious and may require hospitalisation. RSV can initially present with symptoms that might easily be mistaken for a common cold, such as fever, sniffles, wheezing, reduced appetite, or even ear infections. If symptoms progress to include changes in feeding habits, prolonged periods without wet nappies, rapid breathing, persistent high fever despite medication, or uncharacteristic fussiness, it's best to consult your GP. And if your child shows noticeable breathing difficulties or significant pauses in breathing, it's imperative to seek urgent medical attention, including dialling 999.”
Dr Máire Finn, a GP at Ennis Centric Health
According to the Health Service Executive (HSE), symptoms of RSV can often resemble a cold at the beginning, including:
• cough
• wheezing
• runny nose
• difficulty feeding or decreased appetite
• fever (temperature of 38 degrees Celsius or higher)
• sore throat
Precautionary steps can be taken to help protect infants. These include:
• keeping a child home from school, childcare and other activities if they are not well enough to attend
• washing their hands properly and regularly
• always carrying tissues and cover their mouth and nose with a tissue when they cough or sneeze
• cough or sneeze into a bent elbow if you do not have a tissue and wash your hands after
• put used tissues in the bin and wash hands after
The HSE also recommends that parents or caregivers contact a GP if they are worried about their baby’s symptoms or if:
• they’re worried about their child
The National Office of Clinical Audit has launched the Irish Heart Attack Audit National Report 2022 and 2023.
• The baby has had less than 3 to 4 wet nappies in a 24 hour period
• The baby has gone 12 hours without having a wet nappy
• The baby has no energy to breastfeed, or if they are refusing to feed. Or if baby is too breathless to attach to the breast
• They are bottle feeding and notice baby is taking less than half of what they would normally take
• The child has a temperature of 38 degrees Celsius or higher that does not settle or improve
• The child seems short of breath
• The child has no energy or is irritable
Furthermore, the HSE recommends immediately calling 999 or 112 for an ambulance or go to the nearest emergency department if a parent or a caregiver notices that their child:
• is having visible difficulty breathing - their nostrils are getting wider as they breathe, or their ribs are sucking in
• has gone blue on their lips or tongue
• will not stay awake or wake up
• has long gaps in breathing (more than 10 seconds at a time or regular short gaps in breathing of 5 to 10 seconds)
• is breathing a lot faster than usual (more than 60 breaths per minute)
• is pale and sweaty
• has had a dry nappy for 12 hours or more
The report reveals the key quality indicators in the care provided to the patients who experienced a major heart attack known as an ST elevation myocardial infarction (STEMI). A STEMI happens when the supply of blood to the heart is suddenly cut off, usually by a blood clot (thrombosis). Key statistics regarding the lifestyle factors and treatment of those patients was also audited.
Receiving timely treatment is important for patients, and the report revealed that of the 3,102 patients treated in one of the ten primary percutaneous coronary intervention (PCI) centres in Ireland, 55% were brought directly by ambulance to a PCI centre. 32% of patients were transferred from a non-specialist hospital to a PCI centre.
The audit continues to show that the likelihood of achieving timely treatment is highly dependent on how a patient accesses a primary PCI centre. Patients who arrive directly at a PCI centre, usually by ambulance and after having the diagnosis confirmed by a pre-hospital ECG with the ambulance service, receive timely treatment in 79% of cases compared to 45% of those who are transferred from another hospital.
Pauline O’Shea, Patient and Public Interest Representative on the IHAA Governance Committee and Advocacy Campaign Manager with the Irish Heart Foundation, commented, “The audit clearly shows, year on year, the impact of the initial transport choice for many patients having a heart attack is significant in terms of quality and quantity of life after a heart attack.”































It is well known that many consumers struggle to ensure they are eating a healthy, balanced diet and the result can be low immunity and a general lack of energy which will leave them prone to picking up viruses and infections.
Millions take vitamins and dietary supplements hoping to achieve good health, ease illnesses or defy ageing. Recent years have seen a massive boom in supplement use as products that were once the preserve of specialist health food stores have become more widely available, especially in pharmacy. However, the huge range of dietary supplements now available makes the area something of a minefield for consumers. It should be explained that vitamins and minerals are two completely different things, and that minerals help the vitamins work.
Customers will take supplements for all kinds of reasons, usually relating to their health. They hope these will boost vitality, limit the signs of ageing, extend life, cut the risk of chronic disease such as cancer and treat specific ailments such as arthritis. Pharmacy staff should understand the evidence regarding the efficacy of supplements and be able to explain this to patients. They
must also be up-to-date on any new products and formulations and be familiar with the benefits of each item. It is essential that customers feel that they are being recommended the correct VMS for them, not just whichever product is on special offer that particular day.
Buyer Categories
Supplements are products that contain vitamins, minerals, herbs or plant material and come in pill, capsule, tablet or liquid form.
Women are more likely to purchase VMS products than men, as will older people with poor health. Over 50% of children are given vitamins regularly and/or a mineral supplement.
The Department of Health & Children recommend specific vitamins for specific groups of people, and the EU has daily recommended allowances for vitamins and minerals these are:
Certain vitamins are important for certain people, these are:
conceive and who are in the first 12 weeks of pregnancy – 400mcg daily is recommended for women at low risk of conceiving a child with neural tube defects and 5mg daily for those at high risk. Some pregnant women might require additional calcium or iron.
Vitamin C
Vitamin C is the most popular single vitamin supplement. Other supplements that people often turn to when they have a cold include zinc and echinacea.
All of these have been found to have antiviral properties in animal or laboratory studies. Researchers have been interested in using vitamin C to prevent and treat colds for more than 60 years. Some animal studies have found that vitamin C strengthens resistance to infections. One theory for this is that the vitamin protects cells against oxidative stress.
excessive build-up of iron in their body, such as haemochromatosis. Large doses may cause nausea, diarrhoea and stomach cramps. Refer to the pharmacist anyone with any medically significant conditions.
Zinc
Zinc is a trace element that is essential for normal growth. The mineral has been promoted for many years as being able to boost the immune system. Customers purchasing zinc must keep to the recommended dosages. The correct dose is important and there may be side effects including nausea and an unpleasant taste in the mouth, or more serious events such as vomiting, diarrhoea and abdominal pain.
Vitamin D
Vitamin D is recommended for all pregnant and breastfeeding women. Older people also require additional vitamin D and it is recommended for children aged from six months to five years, together with vitamins A and C.
Vitamin B9
More commonly known as folic acid, vitamin B9 is recommended for women who are trying to
Vitamin C is not suitable for everyone. It can cause dangerous rises in the blood sugar levels of people with diabetes. It can also be dangerous for people with genetic conditions that cause an
Combination products are also growing in popularity, such as combined fish oils and garlic for heart health. In fact, along with multivitamin formulations, products containing fish oils are amongst the highest top-selling supplements in recent years in Ireland.



Written by Amy Oates, MPSI
Every year during the cold and flu season, most patients consider their community pharmacy as the first port of call to help manage symptoms. Consumer research shows that an average of 80% of the population are likely to visit a pharmacist with cold and flu symptoms.

The majority of patients will look to treat symptoms of cold and flu within three days of onset, and many will expect an all-in-one product to relieve all symptoms. Every interaction with a patient is an opportunity to provide the most appropriate advice and treatment recommendations. However, engaging in a consultation with patients about their cold or flu symptoms can sometimes be difficult. Many just simply ask for a specific product and are not looking for additional advice from the pharmacy team. This is where our pharmacy teams can focus on ensuring that all patients are getting the best possible care, and most appropriate treatment.
Consultation pathway
There are several stages in the consultation pathway, and the whole pharmacy team should use the same system to promote consistent care. The first stage during any consultation begins when the patient presents to the counter and ends with the patient satisfied with their recommended treatment:
• Initiating the consultation: Greet the patient, identify what brings them to the pharmacy and build a rapport;
• Gathering information: Use your usual questioning technique such as WWHAM to gather information related to the patient’s problem. Example questions, Stage 2:
1. Can you describe your symptoms and how long they have been present?
2. Is there anything about these symptoms that is worrying you?
3. Have you tried anything to relieve your symptoms?
4. Do you have any medical conditions or allergies?
• Shared decision-making: Put the patient at the centre of decisions about their own care. This is an important stage where the pharmacy team has the opportunity to provide detailed information on symptom management, appropriateness of antibiotics, the product
options available and other patient-specific factors to allow them to make a fully informed decision about their own care. As self-care is essential for the future management of cold and flu, any decisions should be made in collaboration with the patient;
• Patient education: This important stage ensures the patient knows how to use the medication effectively and safely, and to store it appropriately. During the consultation, patients should be educated on the correct dosage and administration, what to expect in terms of symptom relief and improvement, and any red flags to watch out for. Patients should be told what to do if there is no improvement or if an adverse reaction is experienced, and also when to look for advice or need to return to the pharmacy. Pharmacy teams can also provide patients with details regarding the next steps for their management, including nonpharmacological treatments and self-care measures; and
• Close the consultation: Check that the patient is happy with the agreed management and treatment plan. It is also important to check the patients understanding of all the information provided and give them the opportunity to ask questions.
Patients wishing to purchase specific products can be less willing to engage in a consultation. This is where our pharmacy teams need to focus to ensure that the product is suitable
Patients who present wishing to purchase specific products can be less willing to engage in a consultation. This is where our pharmacy teams need to focus to ensure that the product is suitable. Rather than going straight to select the product for the patient we can direct the conversation back through the consultation pathway. Start by asking if the product is for them or someone else. By engaging in a conversation with the patient it will initiate the consultation journey and work through the stages outlined. If a patient has preselected a cold and flu product to purchase, discuss their symptoms alongside the appropriateness of the product selected. Alternative management options should be presented with reasons why these options may be suitable.
Many patients can find it difficult to find the appropriate treatment, with so many options available. Patients often choose incorrect treatments to relieve their symptoms. Patients are at risk of using too many products to treat their symptoms and can often be over-medicating and overusing some ingredients. This is where our pharmacy teams can ensure the most effective treatment for cold and flu symptoms. Pharmacological management consists primarily of symptomatic relief for the presenting symptoms. Following the consultation pathway, the choice of product should be based on patient preference, presenting symptoms, and clinical considerations. Pharmacists and their team need to ensure that patients are aware of the active ingredients in combination products
and that the correct product is selected to manage symptoms. Special consideration should be given to certain groups where their management may require additional care, intervention and attention. For instance, older people, those who are pregnant or immunocompromised.
Identifying need for referral
A community pharmacist can easily diagnose a common cold. If a more serious infection is suspected, or involvement of the lower airways, then patients should be referred to a medical practitioner for further assessment. When assessing patients’ presenting symptoms, pharmacists can identify red-flag symptoms which warrant direct referral to a GP:
• Fever ≥39°C in adults;
• Fever for five days or more, or fever returning after a fever-free period in adults;
• Fever ≥38°C in infants; or
• Rising fever or fever lasting more than two days in children of any age.
Some red-flag signs and symptoms accompanying cough / sore throat can include but is not limited to:
• Cough that lasts for more than three weeks;
• Unexplained cough that recurs regularly;
• Discoloured or purulent sputum present with cough;
• Coughing up blood or blood present in sputum;
• Chest pain;
• Shortness of breath;
• Wheezing (may be suggestive of other conditions, such as asthma);
• Persistent nocturnal cough in children;
• Stridor;
• Sore throat that lasts for more two weeks;
• Difficulty in swallowing;
• Sore throat with mouth ulceration or blistering; or
• Suspected adverse drug reaction or medicine-induced agranulocytosis:
o Examples of medicines known to contribute to agranulocytosis include captopril, carbimazole, cytotoxic medicines, clozapine, sulfasalazine and sulphur-containing antibiotics. Further information can be found in the International Pharmaceutical Federation (FIP)’s handbook for pharmacists, Cold, Flu and sinusitis: Managing symptoms and supporting self-care (available at fip.org/file/5089).
Role of the pharmacist
Pharmacists have a unique opportunity to make a differential diagnosis, educate patients on the management of these common conditions, and provide
appropriate therapy and advice. Ensuring pharmacy teams engage in effective consultations when patients present with cold and flu symptoms will ensure the highest standard of care for all patients. There is also an important role in antimicrobial stewardship and supporting self-care.
Antimicrobial stewardship
Pharmacists have a huge role in antimicrobial stewardship (AMS). A lot of patients can expect an antibiotic for treatment of their cold and flu symptoms. AMS not only looks at challenging inappropriate prescribing of antimicrobials but also plays a role in the prevention of infection and promoting self-care for cold and flu, right from the initial contact with the patient. Pharmacists are ideally placed to educate patients about the indications for use of antibiotics and the importance of avoiding their misuse and overuse. Patients should be educated on the difference between bacterial and viral infections as well as the reasons why antibiotics are not effective against viruses, which are the primary cause of the common cold and flu. Pharmacy teams can help reshape the patients perception of managing cold and flu symptoms. Pharmacists must encourage their teams to reinforce public health messaging around prevention of infection and self-care for cold and flu, and they must strongly discourage patients from automatically seeking antibiotics. This message needs to begin at the start of the patient journey — at first
presentation, when initiating the consultation. This can be achieved through an effective consultation, with appropriate treatment recommendations resulting in successful symptom relief.
Supporting self-care
According to the World Health Organisation, self-care is defined as; “the ability of individuals, families and communities to promote health, prevent disease, maintain health, and to cope with illness and disability with or without the support of a healthcare provider”. Self-care is a broad area that looks at many factors such as a patient lifestyle, their nutrition, general and personal hygiene and self-medication. Pharmacists can empower patients to help them make healthy lifestyle choices, recommend appropriate non-prescription medicines, and educate them on when they should visit a GP.
Flu vaccination
Flu vaccination is one of the most effective methods of preventing flu illness. Vaccination has been associated with a reduced risk of flu infection and transmission, hospitalisation, and risk of flurelated death in at-risk patients. As pharmacists, we can help to ensure our patients are offered a flu vaccine each year, especially those in at-risk groups. By providing a flu vaccination service in our pharmacies we are reinforcing our roles as firstline, easily accessible primary healthcare professionals.
Results of the 2024 National Inpatient Experience Survey have been published, highlighting the experiences of patients in public acute hospitals across Ireland. This year, 12,367 patients took part in the survey, sharing feedback on their experience of care in Irish hospitals.
The survey revealed that the majority of respondents (85%) rated their hospital care experience positively, with 58% rating their care as “very good” and 27% as “good”.
Compared to 2022 survey findings, the 2024 results show that patients’ rating of their overall care experiences have improved, particularly regarding being discharged from hospital, which had consistently received the lowest scores in previous surveys.
In 2024, patients indicated positive interactions with healthcare staff, with:
• 83.8% reporting they always felt treated with respect and dignity
• 79.1% always had confidence and trust in hospital staff hospital staff
• 78.7% reporting adequate pain management
• 79% feeling fully confident in the safety of their treatment and care.
Despite overall positive feedback, patients identified a number of
key areas to be improved, and highlighted long waiting times in the emergency department:
• 19.7% said they were unable to find staff to discuss their worries
• 15.3% reported not receiving enough information on managing their condition postdischarge
• 13.4% said their family did not have enough opportunities to talk to a doctor
• 72.6% reported waiting longer than six hours before being admitted to a ward, with 1,078
people (13.4%) waiting more than 24 hours.
The survey found that patients from disadvantaged areas were more likely to report being treated with respect and dignity while in hospital, as well as receive more frequent introductions from hospital staff, and have higher satisfaction with hospital food in comparison to those from affluent areas. In contrast, patients from affluent areas were less likely to feel they received adequate information on managing their condition post-discharge.
Written by Dr S Griffiths, Dr D Breen, Department of Respiratory Medicine, Galway University Hospital
Abstract: Smoking remains the single leading cause of preventable disease, disability and death worldwide. Currently 18% of Irish adults are active smokers, and recent years have seen an upward trend of young people developing a nicotine addiction, in part due to the rise in popularity of electronic nicotine delivery systems (ENDS). Smoking cessation services are an essential component of tobacco control, and play a key role in meeting the goal of a Tobacco-Free Ireland.
Introduction:
In this paper we will analyse current prevalence and trends in smoking habits in Ireland, and discuss the impact of ENDS in the market, particularly the impact on Irish teenagers and potential legislation to tackle this novel challenge. We aim to identify successful smoking cessation frameworks worldwide and discuss the benefits to both the patient and the health system. A local audit conducted in Galway University Hospital provides insight into current smoking cessation practise and we have identified potential methods to improve local systems. We will review the National Stop Smoking Guideline 2022, in particular it’s application to the hospital setting and discuss the need for increased training among all healthcare staff in smoking cessation provision.
Meeting the goal of being tobacco-free by 2025 is dependent on accelerating progress with smoking cessation in Ireland, and this paper aims to identify current challenges to Irish smoking habits and identify an effective approach to improving our smoking cessation services.
Tobacco use is the leading cause of preventable death, disease and disability worldwide. (1) The Organisation (WHO) describes current tobacco usage worldwide as a global epidemic. The Healthy Report 2022 (HI) reports that 18% of the population of Ireland are current smokers. There has significant shift in demographics in Ireland with the Central Statistics Office publishing data for 2022, showing the largest 12 -month increase in the population of Ireland since 2008, in part accounted 15-year high in immigrant arrivals (2). These demographic shifts will have a major impact on smoking prevalence; for example the rapid increase in Ukrainian nationals to Ireland will ultimately resul in overall smoking numbers; it was estimated that in 2022, 27.4% of the Ukrainian adult population (3).
2022, 27.4% of the Ukrainian adult population smoked.3
Current prevalence and trends in smoking habits
Tobacco use is the leading cause of preventable death, disease and disability worldwide.1 The World Health Organisation (WHO) describes current tobacco usage worldwide as a global epidemic. The Healthy Ireland Report 2022 (HI) reports that 18% of the population of Ireland are current smokers. There has been a significant shift in demographics in Ireland with the Central Statistics Office publishing data for the year ending 2022, showing the largest 12-month increase in the population of Ireland since 2008, in part accounted for by a 15year high in immigrant arrivals.2 These demographic shifts will have a major impact on smoking prevalence; for example the rapid increase in Ukrainian nationals to Ireland will ultimately result in an increase in overall smoking numbers; it was estimated that in
The HI survey also reflects WHO data on long term health effects of tobacco use, reporting that 32% of smokers and 39% of ex-smokers suffer from longterm illness and chronic health problems, compared with 27% of never smokers.4 Year on year, the HI Survey identifies 25–34-yearolds as the age group most likely to smoke, and the proportion of smokers in this group has experienced a 4-point increase to 24%, significantly higher than the national average.4 Smoking prevalence by age and gender is illustrated in Figure 1 and of note highlights the persistent high smoking rates within young adults, particularly males, and the decrease in smoking prevalence among older adults.
Current prevalence and trends in smoking habits:
The use of electronic nicotine delivery systems (ENDS), often referred to as e-cigarettes or ‘vapes’ is increasingly prevalent, with a major expansion of this market in Ireland over the past two decades. Overall, 6% of the population reported using ENDS regularly, with the highest use reported in those under the age of 25 (11%).4 ENDS are highly variable, without a strict regulatory framework, and at present there is no definitive research on the long-term impacts of ENDS use, or second-hand exposure. As the popularity of ENDS grows, there is concern about their use in younger people. The Public Health (Tobacco Products and Nicotine Inhaling Products) Bill 2023 is currently before Dáil Éireann, and proposes to introduce a regulatory framework for the retail sale of ENDS, termed ‘nicotine inhaling products’. These measures focus on reducing access
to ENDS for children by restricting the sale of these products, and plans to prohibit advertisement of ENDS around schools and on public transport
The European Schools Project on Alcohol and other Drugs (ESPAD) Ireland 2019 Report5 provides information about substance use among Irish teenagers attending secondary school, and reported that the decline in smoking has halted in Irish teenagers for the first time in 25 years. This correlated with a higher prevalence of e-cigarette use among Irish teenagers than smoking tobacco, with 39% of 15- to 16-year-olds reporting having used ENDS, compared with 32% smoking tobacco. There are concerns about the potential role of ENDS
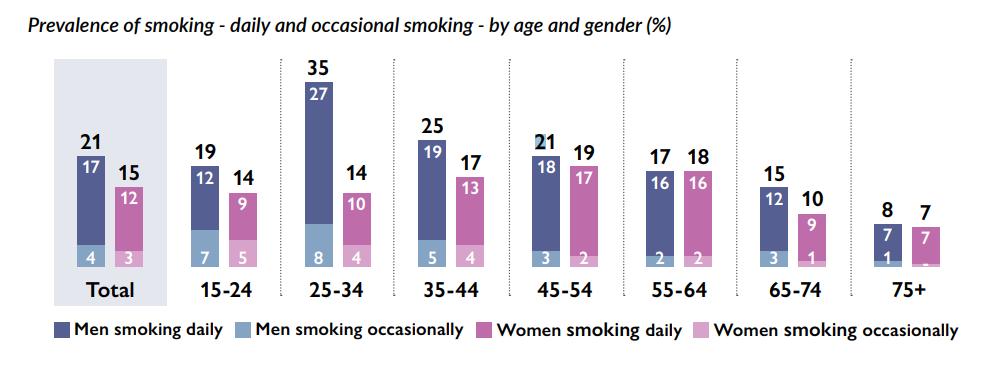
The HI survey also reflects WHO data on long term health effects of tobacco use, reporting that and 39% of ex-smokers suffer from long-term illness and chronic health problems, compared with smokers. (4 ) Year on year, the HI Survey identifies 25 –34-year-olds as the age group most likely the proportion of smokers in this group has experienced a 4 -point increase to 24%, significantly national average. (4 ) Smoking prevalence by age and gender is illustrated in Figure 1 and of note persistent high smoking rates within young adults, particularly males, and the decrease in smoking among older adults. Figure 1: Data from the Healthy Ireland Report 2022 on prevalence of smoking by age and gender


*based on 2 x 1mg spray
Nicorette QuickMist 1 mg/spray, oromucosal spray, solution. One spray delivers 1 mg nicotine in 0.07 ml solution. 1 ml solution contains 13.6 mg nicotine. Excipient with known effect: Ethanol (less than 100 mg of ethanol/spray). Propylene glycol, Butylated hydroxytoluene. Pharmaceutical form: Oromucosal spray, solution. A clear to weakly opalescent, colourless to yellow solution. Indications: For the treatment of tobacco dependence in adults by relief of nicotine withdrawal symptoms, including cravings, during a quit attempt (or to cut down smoking before stopping completely. Permanent cessation of tobacco use is the eventual objective. Nicorette QuickMist should preferably be used in conjunction with a behavioral support program. Dosage: Subjects should stop smoking completely during the course of treatment with Nicorette QuickMist. Adults and Elderly: The following chart lists the recommended usage schedule for the oromucosal spray during full treatment (Step I) and during tapering (Step II and Step III). Up to 4 sprays per hour may be used. Do not exceed 2 sprays per dosing episode and do not exceed 64 sprays (4 sprays per hour, over 16 hours) in any 24-hour period. Step I: Weeks 1-6: Use 1 or 2 sprays when cigarettes normally would have been smoked or if cravings emerge. If after a single spray cravings are not controlled within a few minutes, a second spray should be used. If 2 sprays are required, future doses may be delivered as 2 consecutive sprays. Most smokers will require 1-2 sprays every 30 minutes to 1 hour. Step II: Weeks 7-9: Start reducing the number of sprays per day. By the end of week 9 subjects should be using HALF the average number of sprays per day that was used in Step I. Step III: Weeks 10-12: Continue reducing the number of sprays per day so that subjects are not using more than 4 sprays per day during week 12. When subjects have reduced to 2-4 sprays per day, oromucosal spray use should be discontinued. To help stay smoke free after Step III, subjects may continue to use the oromucosal spray in situations when they are strongly tempted to smoke. One spray may be used in situations where there is an urge to smoke, with a second spray if one spray does not help within a few minutes. No more than four sprays per day should be used during this period. Regular use of the oromucosal spray beyond 6 months is generally not recommended. Some ex-smokers may need treatment with the oromucosal spray longer to avoid returning to smoking. Any remaining oromucosal spray should be retained to be used in the event of sudden cravings. Gradual cessation through progressive reduction in smoking: For smokers who are not willing or ready to quit abruptly. The oromucosal spray is used between periods of smoking in order to prolong the smoke-free intervals and with the intention to reduce smoking as much as possible. The patient should be aware that an incorrect use of the spray may enhance adverse effects. A cigarette is replaced with one dose (1-2 sprays) and a quit attempt should be made as soon as the smoker feels ready and no later than 12 weeks after start of treatment. If a reduction in cigarette consumption has not been achieved after 6 weeks of treatment, a healthcare professional should be consulted. After quitting smoking, gradually reduce the number of sprays per day. When subjects have reduced to 2-4 sprays per day, oromucosal spray should be discontinued. Regular use of the oromucosal spray beyond 6 months is not recommended. Some ex-smokers may need treatment with the oromucosal spray longer to avoid returning to smoking. Any remaining oromucosal spray should be retained to be used in the event of sudden cravings. Paediatric population: Do not administer this medicine to persons under 18 years of age. There is no experience of treating adolescents under the age of 18 with this medicine. Method of administration: After priming, point the spray nozzle as close to the open mouth as possible. Press firmly the top of the dispenser and release one spray into the mouth, avoiding the lips. Subjects should not inhale while spraying to avoid getting spray into the respiratory tract. For best results, do not swallow for a few seconds after spraying. Subjects should not eat or drink when administering the oromucosal spray. Behavioural therapy advice and support will normally improve the success rate. Contraindications: Hypersensitivity to nicotine or to any of the excipients. Children under the age of 18 years. Those who have never smoked. Special warnings and precautions for use: This medicine should not be used by non-smokers. The benefits of quitting smoking outweigh any risks associated with correctly administered nicotine replacement therapy (NRT). A risk-benefit assessment should be made by an appropriate healthcare professional for patients with the following conditions: Cardiovascular disease: Dependent smokers with a recent myocardial infarction, unstable or worsening angina including Prinzmetal’s angina, severe cardiac arrhythmias, recent cerebrovascular accident and/or who suffer with uncontrolled hypertension should be encouraged to stop smoking with non-pharmacological interventions (such as counselling). If this fails, the oromucosal spray may be considered but as data on safety in this patient group are limited, initiation should only be under close medical supervision. Diabetes Mellitus. Patients with diabetes mellitus should be advised to monitor their blood sugar levels more closely than usual when smoking is stopped and NRT is initiated as reduction in nicotine induced catecholamine release can affect carbohydrate metabolism. Allergic reactions: Susceptibility to angioedema and urticaria. Renal and hepatic impairment: Use with caution in patients with moderate to severe hepatic impairment and/or severe renal impairment as the clearance of nicotine or its metabolites may be decreased with the potential for increased adverse effects. Phaeochromocytoma and uncontrolled hyperthyroidism: Use with caution in patients with uncontrolled hyperthyroidism or phaeochromocytoma as nicotine causes release of catecholamines. Gastrointestinal Disease: Nicotine may exacerbate symptoms in patients suffering from oesophagitis,
gastric or peptic ulcers and NRT preparations should be used with caution in these conditions. Seizures: Use with caution in subjects taking anti-convulsant therapy or with a history of epilepsy as cases of convulsions have been reported in association with nicotine. Paediatric population: Danger in children: Doses of nicotine tolerated by smokers can produce severe toxicity in children that may be fatal. Products containing nicotine should not be left where they may be handled or ingested by children. Transferred dependence: Transferred dependence can occur but is both less harmful and easier to break than smoking dependence. Stopping smoking: Polycyclic aromatic hydrocarbons in tobacco smoke induce the metabolism of drugs metabolised by CYP 1A2 (and possibly by CYP 1A1). When a smoker stops smoking, this may result in slower metabolism and a consequent rise in blood levels of such drugs. This is of potential clinical importance for products with a narrow therapeutic window, e.g. theophylline, tacrine, clozapine and ropinirole. The plasma concentration of other medicinal products metabolised in part by CYP1A2 e.g. imipramine, olanzapine, clomipramine and fluvoxamine may also increase on cessation of smoking, although data to support this are lacking and the possible clinical significance of this effect for these drugs is unknown. Limited data indicate that the metabolism of flecainide and pentazocine may also be induced by smoking. Excipients: This medicine contains about 7 mg of alcohol (ethanol) in each spray which is equivalent to 97 mg/ml. The amount in one spray of this medicine is equivalent to less than 2 ml beer or 1 ml wine. The small amount of alcohol in this medicine will not have any noticeable effects. This medicinal product contains less than 1 mmol sodium (23 mg) per spray, i.e. essentially ‘sodium- free’. This medicine contains 12 mg propylene glycol in each spray which is equivalent to 157 mg/mL. Due to the presence of butylated hydroxytoluene, Nicorette QuickMist may cause local skin reactions (e.g. contact dermatitis), or irritation to the eyes and mucous membranes. Care should be taken not to spray the eyes whilst administering the oromucosal spray. Undesirable effects: Effects of smoking cessation: Regardless of the means used, a variety of symptoms are known to be associated with quitting habitual tobacco use. These include emotional or cognitive effects such as dysphoria or depressed mood; insomnia; irritability, frustration or anger; anxiety; difficulty concentrating, and restlessness or impatience. There may also be physical effects such as decreased heart rate; increased appetite or weight gain, dizziness or presyncopal symptoms, cough, constipation, gingival bleeding or apthous ulceration, or nasopharyngitis. In addition, and of clinical significance, nicotine cravings may result in profound urges to smoke. This medicine may cause adverse reactions similar to those associated with nicotine given by other means and these are mainly dose-dependent. Allergic reactions such as angioedema, urticaria or anaphylaxis may occur in susceptible individuals. Local adverse effects of administration are similar to those seen with other orally delivered forms. During the first few days of treatment irritation in the mouth and throat may be experienced, and hiccups are particularly common. Tolerance is normal with continued use. Daily collection of data from trial subjects demonstrated that very commonly occurring adverse events were reported with onset in the first 2-3 weeks of use of the oromucosal spray, and declined thereafter. Adverse reactions with oromucosal nicotine formulations identified from clinical trials and during post-marketing experience are presented below. The frequency category has been estimated from clinical trials for the adverse reactions identified during post-marketing experience. Very common (≥1/10); common (≥1/100 to <1/10); uncommon (≥1/1 000 to <1/100); rare (≥1/10 000 to <1/1 000); very rare (<1/10 000); not known (cannot be estimated from the available data). Immune system disorders Common Hypersensitivity Not known Allergic reactions including angioedema and anaphylaxis Psychiatric disorders Uncommon Abnormal dream Nervous system disorders Very common Headache Common Dysgeusia, paraesthesia Not known – Seizures (Cases of seizures have been reported in subjects taking anti-convulsant therapy or with a history of epilepsy). Eye disorders Not known Blurred vision, lacrimation increased Cardiac disorders Uncommon Palpitations, tachycardia Not known Atrial fibrillation Vascular disorders Uncommon Flushing, hypertension Respiratory, thoracic and mediastinal disorders Very common: Hiccups, throat rritation Uncommon Bronchospasm, rhinorrea, dysphonia, dyspnoea, nasal congestion, oropharyngeal pain, sneezing, throat tightness Common: cough Gastrointestinal disorders Very common Nausea Common Abdominal pain, dry mouth, diarrhoea, dyspepsia, flatulence, salivary hypersecretion, stomatitis, vomiting Uncommon Eructation, gingival bleeding, glossitis, oral mucosal blistering and exfoliation, paraesthesia oral Rare Dysphagia, hypoaesthesia oral, retching Not known Dry throat, gastrointestinal discomfort, lip pain Skin and subcutaneous tissue disorders Uncommon Hyperhidrosis, pruritus, rash, urticaria Not known Erythema General disorders and administration site conditions Common Burning sensation, fatigue Uncommon Asthenia, chest discomfort and pain, malaise. MAH: JNTL Consumer Health I (Ireland) Ltd. Block 5, High Street, Tallaght, Dublin 24, Ireland, Ireland. PA Number: PA23490/019/013 Date of revision of text: December 2023. Product not subject to medical prescription. Supply through pharmacy and non-pharmacy outlets. Full prescribing information available upon request.
providing new routes into nicotine addiction, with 68% of adolescents in a 2019 study reporting they had never used tobacco prior to first use of e-cigarettes.6
In the adolescent population, e-cigarettes are rarely cited as a smoking cessation technique, with only 3% of adolescents reporting quitting smoking as the reason for first using e-cigarettes.6 This contrasts with the high ENDS use as a smoking cessation tool in adults; the HSE National Stop Smoking Guideline reports 38% of people choosing e-cigarettes as a quit smoking aid in 2019.7
Tackling tobacco use
in the early 1990s. The OMSC is a validated, evidence-based process to embed comprehensive smoking cessation treatments and support as an integral aspect of routine patient care. Since its inception the OMSC has continued to expand. In 2020 the OMSC had been implemented in over 500 outpatient, inpatient and primary care sites across Canada. A 2015 study reported that 35% of patients who received the OMSC interventions were smoke-free at 6 months, and had a 40% reduction in risk of death over 2 years.11
This project was supported by encouragement of nursing staff during the daily safety-pause, and a poster campaign on the pilot ward. During the pilot programme, smoking documentation rate increased to 85.7%, and 95% of tobacco users were referred to local smoking cessation services, a significant improvement from the reported pre-intervention results.
Tackling tobacco use has become a priority for government policy makers worldwide. In 2008 the WHO implemented the MPOWER policy as a worldwide tobacco control strategy, aiming to assist in the country-level implementation of effective interventions to reduce the demand for tobacco.
smoking cessation services significantly improved practice. This project was supported by encouragement of nursing staff during the daily safety-pause, and a poster campaign on the pilot ward. During programme, smoking documentation rate increased to 85.7%, and 95% of tobacco users were referred smoking cessation services, a significant improvement from the reported pre-intervention results.
In Ireland, the HSE implemented the National Stop Smoking Guideline (NSSG) in 20227 aimed at improving the identification and treatment of tobacco addiction across all healthcare settings. Healthy Ireland reports that 29% of smokers are either trying to quit or are actively planning a stop date.4 However, only 18% of smokers who saw their GP over a 12-month period discussed methods of smoking cessation, a decline of 50% compared to data from 2019.4
of health behaviour change that should be utilised by healthcare professionals in their day-to-day practice. Sometimes referred to as a ‘teachable moment’, these conversations aim to motivate individuals to adopt risk reducing health behaviours. The 5 A’s model (figure 2), recommended by the Centre for Disease Control (CDC) and WHO, summarises the activities that a healthcare provider can do within 3-5 minutes during a consultation. Multiple studies have shown the benefits of a brief intervention program, delivered by staff who are appropriately trained, with a statistically significant difference in smoking cessation and continued abstinence.14
The British Thoracic Society (BTS) and the National Institute for Health and Care Excellence (NICE) recommend that all frontline healthcare staff should receive training to identify smoking status and to offer very brief advice, as well as the local referral process for behavioural support.15,16
HSE implemented the National Stop Smoking Guideline (NSSG) in 2022 (7) aimed at improving identification and treatment of tobacco addiction across all healthcare settings. Healthy Ireland reports smokers are either trying to quit or are actively planning a stop date (4) However, only 18% of saw their GP over a 12-month period discussed methods of smoking cessation, a decline of 50% data from 2019. (4)
In 2004, Ireland became the first country to pass a smoke-free law,8 and furthermore in 2021 was recognised by the WHO as a global leader in tobacco control. The Department of Health has set the goal of being tobacco-free by 2025, defined as a smoking prevalence of less than or equal to 5%. However, as the HI Survey shows, ongoing improvements in smoking cessation policy and practise are required to maintain progress towards this goal. As demonstrated in the HI Survey 2022, a large proportion of smokers suffer from chronic illness4 which in turn often results in hospital admissions and prolonged inpatient stays. This was demonstrated in a 2016 study which attributed 309,117 beddays to smoking related illnesses. The HSE recommends that each patient encounter in the hospital setting is used as a valuable opportunity for a brief intervention to discuss smoking cessation.
In 2017, the OMSC was adapted in the Greater Manchester Area in the UK as the CURE project; a comprehensive secondary care treatment program for tobacco addiction. The CURE project has been shown to be cost-effective, with a highly significant return on investment.12 The CURE project estimates a 50% reduction in readmissions at 30-days (3,273 admissions), and a reduction in re-admissions at 1 year from 38.4% to 26.7%. This project is estimated to save 30,880 bed days per year in the Greater Manchester Area. Furthermore, it has been proven to deliver high-quality care for patients by providing access to highly effective interventions and providing individualised smoking cessation treatment.13 The success of the CURE project has led to a commitment from the NHS that by 2023/24 all active smokers admitted to hospital will be offered NHS-funded tobacco treatment services, based on the successful and favourable results from the OMSC and CURE models.
Local data
recommends that all healthcare professionals ask and document an individual’s smoking behaviours, advise all active smokers about the harms of smoking and the benefits of cessation Healthcare should discuss the individual treatment needs and preferences, and advise that making an quit attempt is less effective than using recommended supports. This is supported by WHO data demonstrates that brief advice from health professionals can increase quitting success by up to 30%, advice increases the chance of quitting by 84%.
The NSSG recommends that all healthcare professionals ask and document an individual’s smoking behaviours, and, in turn advise all active smokers about the harms of smoking and the benefits of cessation. Healthcare professionals should discuss the individual treatment needs and preferences, and advise that making an unsupported quit attempt is less effective than using recommended supports. This is supported by WHO data which demonstrates that brief advice from health professionals can increase quitting success by up to 30%, while intensive advice increases the chance of quitting by 84%.
The NSSG identifies asking about smoking behaviour and offering advice to quit as a key element
identifies asking about smoking behaviour and offering advice to quit as a health behaviour change that should be utilised by healthcare in their day-to -day practice. Sometimes referred to as a ‘teachable these conversations aim to motivate individuals to adopt risk reducing behaviours. The 5 A’s model (figure 2), recommended by the Centre for ontrol (CDC) and WHO, summarises the activities that a healthcare do within 3-5 minutes during a consultation. Multiple studies have benefits of a brief intervention program, delivered by staff who are trained, with a statistically significant difference in smoking continued abstinence. (14)
In our service, this is particularly applicable in the Rapid Access Lung Clinic, a service for diagnosis and treatment of patients with suspected lung cancer. A recent analysis highlights the benefit to overall survival of stopping smoking at any time for a patient with lung cancer.9, 10
Effective smoking cessation framework
Thoracic Society (BTS) and the National Institute for Health and Care (NICE) recommend that all frontline healthcare staff should receive identify smoking status and to offer very brief advice, as well as the process for behavioural support. (15,16)
There is precedent for the implementation of smoking cessation frameworks within healthcare settings, as evidenced by the success of the Ottawa Model for Smoking Cessation (OMSC) which was first developed
In Galway University Hospital we conducted a review of current smoking prevalence and smoking cessation services. An initial chart review revealed that 19.7% of audited patients were active smokers; overall in line with the national data. However, initially, only 64.4% of patients had smoking status documented, and only 13% received brief advice, with the same percentage referred to smoking cessation services. A standardised smoking cessation proforma was implemented on a pilot ward as part of the admission bundle, aiming to document smoking status on admission to hospital and encourage brief advice, and, in addition, all identified smokers were referred to smoking cessation services with their consent. These simple interventions including the introduction of a standardised approach to identification of smoking status and an optout referral of active smokers to smoking cessation services significantly improved practice.

options for behavioural support include individual or group telephone or text-message support Currently, behavioural support specialist smoking cessation officers, both in hospital and based facilities, who can also provide prescription for NRT if Novel methods of delivering counselling for smoking cessation have been developed including the counselling service ‘Florence’, an artificial intelligence bot developed by the WHO. Florence is a 24/7 worker, capable of providing brief conversations by voice or text, and to signpost patients to

Additional options for behavioural support include individual or group counselling, telephone or text-message support. Currently, behavioural support is available specialist smoking cessation officers, both in hospital and community-based facilities, who can also provide prescription for NRT if required. Novel methods of delivering counselling for smoking cessation have been developed including the digital counselling service ‘Florence’, an artificial intelligence bot developed by the WHO. Florence is a 24/7 virtual health worker, capable of providing brief conversations by voice or text, and to signpost patients to other digital cessation programmes in their country, and can found at https://www.who.int/campaigns/ Florence. This technology was developed in 2021, primarily aimed at overcoming the barriers arising from the COVID-19 pandemic such as easy access to smoking cessation services. The HSE has also developed a digital cessation service, and offers a 28-day plan including personalised daily support via email and text message through the QUIT.ie website, with a personalised web page to track progress.
For patients who wish to use pharmacological therapy in combination with behavioural supports, the NSSG recommends that combination NRT treatment or NRT monotherapy should be prescribed. NRT is available in multiple forms and prescription can be tailored to the patient’s preference (figure 3). A recent Cochrane review confirmed the effectiveness of NRT in smoking cessation.17
Varenicline (alone or in combination with nicotine replacement therapy (NRT)) is recommended by the HSE as first-line treatment for smoking cessation.1
Available on private prescription only.
Varenilcine 0.5mg and 1mg Film-Coated Tablets Abbreviated Prescribing Information Presentation: Each film-coated tablet contains varenicline citrate equivalent to 0.5mg and 1mg varenicline. Indications: Varenicline is indicated for smoking cessation in adults. Dosage and administration: Oral use. Adults: The recommended dose is 1mg Varenicline twice daily following a 1-week titration (see SmPC for details). Children: Not recommended for use. Elderly: No dosage adjustment is necessary. Elderly patients are more likely to have decreased renal function, prescribers should consider the renal status of an elderly patient. Renal impairment: No dosage adjustment is necessary for patients with mild (estimated creatinine clearance >50ml/min and ≤80ml/min) to moderate (estimated creatinine clearance ≥30ml/min and ≤50ml/min) renal impairment. For patients with severe renal impairment (estimated creatinine clearance <30ml/min), the recommended dose of Varenicline is 1mg once daily. Hepatic impairment: No dosage adjustment is necessary. Contraindications: Hypersensitivity to the active substance or to any of the excipients. Precautions and warnings: Physiological changes resulting from smoking cessation, with or without treatment with Varenicline, may alter the pharmacokinetics or pharmacodynamics of some medicinal products, for which dosage adjustment may be necessary (examples include theophylline, warfarin and insulin). As smoking induces CYP1A2, smoking cessation may result in an increase of plasma levels of CYP1A2 substrates. Changes in behaviour or thinking, anxiety, psychosis, mood swings, aggressive behaviour, depression, suicidal ideation and behaviour and suicide attempts have been reported in patients attempting to quit smoking with Varenicline. Depressed mood, rarely including suicidal ideation and suicide attempt, may be a symptom of nicotine withdrawal. Clinicians should be aware of the possible emergence of serious neuropsychiatric symptoms in patients attempting to quit smoking with or without treatment. If serious neuropsychiatric symptoms occur whilst
Teva Pharmaceuticals Ireland, Digital Office Centre Swords, Suite 101 - 103, Balheary Demesne, Balheary Road, Swords, Co Dublin, K67E5AO, Ireland.
Freephone: 1800 - 201 700 | Email: info@teva.ie
Prescription Only Medicine.
on Varenicline treatment, patients should discontinue Varenicline immediately and contact a healthcare professional for re-evaluation of treatment. Smoking cessation, with or without pharmacotherapy, has been associated with exacerbation of underlying psychiatric illness (e.g. depression). In clinical trials and post-marketing experience there have been reports of seizures in patients with or without a history of seizures, treated with Varenicline. Varenicline should be used cautiously in patients with a history of seizures or other conditions that potentially lower the seizure threshold. At the end of treatment, discontinuation of Varenicline was associated with an increase in irritability, urge to smoke, depression, and/or insomnia in up to 3% of patients. In such instances, tapering should be considered. Patients taking Varenicline should seek immediate medical attention if they experience signs and symptoms of myocardial infarction or stroke. Interactions: Varenicline has no clinically meaningful drug interactions (see SmPC for further details). No dosage adjustment of Varenicline or co-administered medicinal products listed below is recommended. In vitro studies indicate that Varenicline is unlikely to alter the pharmacokinetics of compounds that are primarily metabolised by cytochrome P450 enzymes. Furthermore, since metabolism of Varenicline represents less than 10% of its clearance, active substances known to affect the cytochrome P450 system are unlikely to alter the pharmacokinetics of Varenicline, therefore a dose adjustment of Varenicline would not be required. Varenilcine is not known to affect the pharmacokinetics of metformin, digoxin, bupropion and warfarin. Coadministration of cimetidine, with Varenicline increased the systemic exposure of varenicline by due to a reduction in varenicline renal clearance. In patients with severe renal impairment, the concomitant use of cimetidine and Varenicline should be avoided. Pregnancy and lactation: As a precautionary measure, it is preferable to avoid the use of varenicline during pregnancy. A decision on whether to continue/discontinue breast-
feeding or to continue/discontinue therapy with varenicline should be made taking into account the benefit of breast-feeding to the child and the benefit of varenicline therapy to the woman. Effects on ability to drive and use machines: Varenicline may have minor or moderate influence on the ability to drive and use machines. Varenicline may cause dizziness, somnolence and transient loss of consciousness, and therefore may influence the ability to drive and use machines. Adverse reactions: Diabetes mellitus, suicidal ideation, depression, hallucinations, psychosis, seizure, cerebrovascular accident, transient loss of consciousness, myocardial infarction, angina pectoris, tachycardia, atrial fibrillation, electrocardiogram ST segment depression, gastritis, haematemesis, severe cutaneous reactions including Stevens Johnson Syndrome and Erythema Multiforme, angioedema. Very Common: Nasopharyngitis, abnormal dreams, insomnia, headache, nausea. Common: Bronchitis, sinusitis, weight increased, decreased appetite, increased appetite, somnolence, dizziness, dysgeusia, dyspnoea, cough, gastrooesophageal reflux disease, vomiting, constipation, diarrhoea, abdominal distension, abdominal pain, toothache, dyspepsia, flatulence, dry mouth, rash, pruritus, arthralgia, myalgia, back pain, chest pain, fatigue, liver function test abnormal. Consult the Summary of Product Characteristics in relation to other side effects. Overdose: In case of overdose, standard supportive measures should be instituted as required. Legal category: POM. Marketing Authorisation Number: 0.5mg PA1986/129/001, 1mg PA1986/129/002. Marketing Authorisation Holder: Teva B.V., Swensweg 5, 2031GA Haarlem, Netherlands. Job Code: MED-IE-00084. Date of Preparation: July 2024.
References: 1. Stop Smoking. National Clinical Guideline No. 28, 2022.


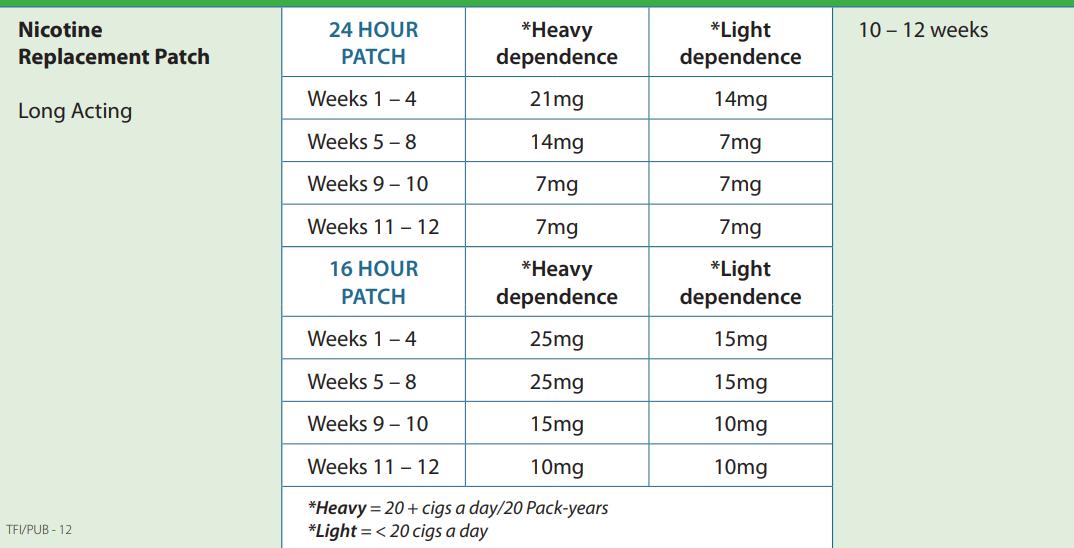
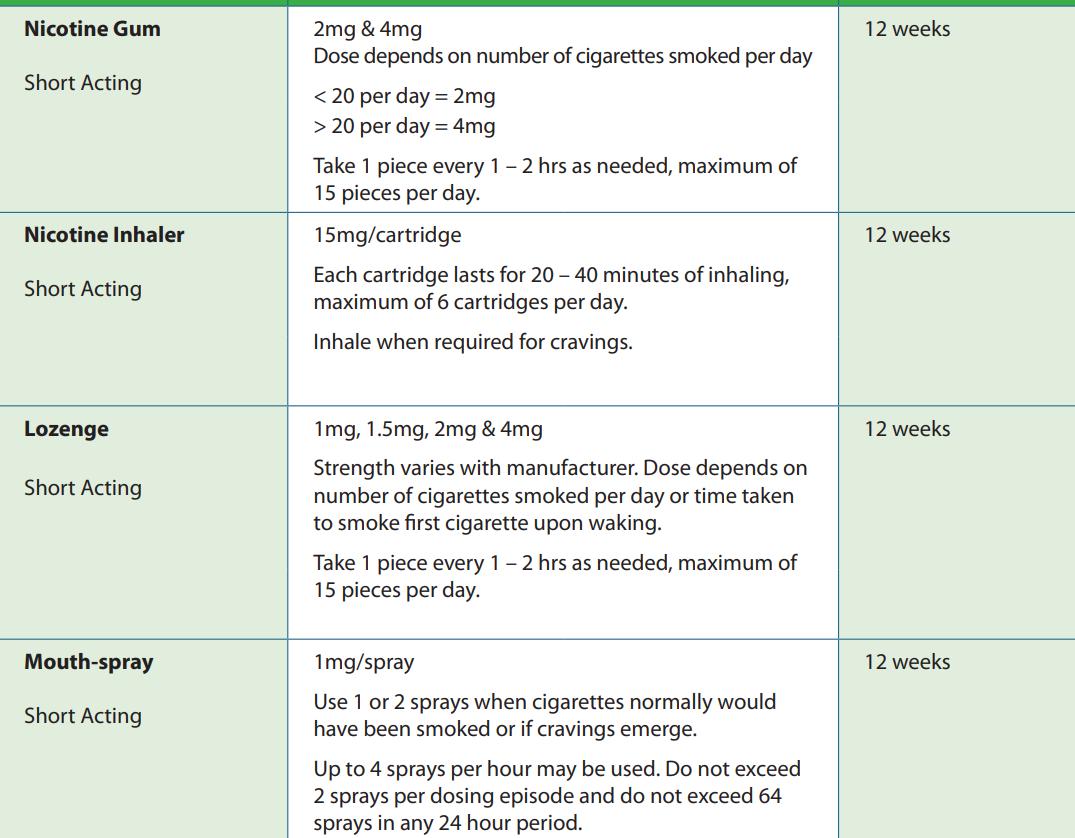
dependent on adequate access to behavioural and pharmacological supports for all patients, irrespective of chronic illness or socioeconomic status.
Conclusion
The development of an effective smoking cessation framework in the hospital setting has been proven by the Ottawa Model (Canada) and the CURE project (UK) to be a cost-effective intervention, which benefits patients at any stage of a disease course, and in addition can equal the benefit of costly pharmacological or surgical interventions. Recent data
indicates a slowdown in the momentum towards the target of a tobacco-free Ireland by 2025. Meeting this goal is dependent on accelerating progress with smoking cessation programs across all healthcare settings. Additional challenges are also appearing, for example, the smoking habits of Irish people are evolving with the increasing popularity of e-cigarettes and this will require the adaptation of current smoking cessation practise and
legislation to tackle these changes particularly amongst younger people. Providing standardised clinical care to patients in healthcare is challenging, due to the diversity in environments of care and patient presentations. However, there is an opportunity during all patient encounters to provide a brief intervention and in turn direct patients to appropriate smoking cessation treatments. Improvements in smoking cessation services in Ireland are
At present, we risk reversing the gains which have been made to date in Ireland towards the goal of a tobacco-free nation. The impact of the COVID-19 pandemic, novel nicotine delivery systems, and the rise of tobacco addiction among young people cannot be underestimated, and additional funding and training of healthcare professionals is required for smoking cessation services to maintain momentum in tackling tobacco addiction in Ireland. References available on request
Call for papers: make your contribution to Irish Pharmacy News
Articles
Research Papers
Reviews
Programme Descriptions
Reports
Case Reports
Letters to editor
In-depth review articles critique fundamental concepts, issues, and problems that define a field of research or practice and support advanced practitioners as well as aspiring early-year pharmacists.
Practice reports share innovations on any area of practice, including delivering clinical services, pharmacy administration, or new approaches to inform and engage with patients with the aim to improving pharmacy practice.
Perspective articles focus on a specific field or discipline and discuss current advances or future directions, and may include original data as well as expert insight and opinions.












Nursing services at the Royal Victoria Eye and Ear Hospital (RVEEH), Dublin, are improving access to integrated eye care for patients, protecting people’s eyesight while reducing waiting times for ophthalmology services by managing stable chronic patients in a controlled environment.
The HSE is highlighting key figures from the RVEEH nursing services, who managed 13,762 ophthalmology patient visits independently and 908 patient visits as part of a shared care clinic in 2023, an increase of 217% from 2019.
The eye conditions managed include some of the main causes of blindness in Ireland, such as age-related macular degeneration, glaucoma, and cataracts with nursing also supporting ocular emergency, cornea and oncology patients.
Key 2023 data shows:
• 9,965 of all injections to patients for the management of their eye condition were administered by nurses
• Only 12% of patients attending the nurse glaucoma clinics required follow-up with a consultant
• Patients have indicated almost 100% satisfaction with the nurse-led service.
The service optimises access to care, reducing waiting times and ensuring continuity of care, as patients often see the same staff member at each visit. Using innovation, digitalisation and an integrated team approach, it is a key example of healthcare reform as it affords medical colleagues the opportunity to see more complex patients.
Cathriona Ennis, Director of Nursing, Royal Victoria Eye and Ear Hospital said: “The nurse-led service in RVEEH improves access to care and timely diagnosis for patients. Nurses are supported in a culture that empowers them to practise at the top of their licence and are instrumental in reducing the risk of disease progression or preventable blindness. The multidisciplinary
approach optimises medical capacity, enhances the patient experience and designs a service that is built on robust governance, competency-based training, evidence-based outcomes best practice and patient and staff feedback.”
Integrated Eye Care Teams in the community are being rolled out to deliver specialty services including assessment, prioritisation, diagnoses and pre-op or postop care. In line with Sláintecare, the RVEEH clinic is also referring patients to community-based services. For example, the community health teams in Tallaght, Wicklow and Churchtown looked after 7,899 new and review patient episodes closer to home in 2023.
Shared Care Initiative
A shared care refers to a collaborative and well-monitored
care plan designed to promote coordinated care for individuals with chronic diseases by facilitating communication between healthcare teams and patients as well as across different healthcare providers and over time.
Linda Fabbian, a Clinical Nurse Specialist (CNS) in glaucoma ophthalmology, has been supported by the Advanced Nurse Practitioner and lead consultants on a pilot basis to expand her role to meet the needs of the community population. “My role involves monitoring and treating patients with glaucoma and assessing patients after cataract surgery. The community care pathway ensures the right patient gets the right care at the right time, improving access to care for patients. This removes the need for some patients to be seen on the hospital site.”



References:1)*Countries: Europe (Austria, Belgium, Bulgaria, Croatia, Czech Republic, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Netherlands, Norway, Poland, Portugal, Romania, Serbia, Slovakia, Slovenia, Spain, Sweden, Switzerland, UK) Source: Based on internal analysis by Laboratoires Théa using quarterly data from the following source: IQVIA Consumer Health Customized Insights for the period 2023, volume and value sales in Euros MNF, limited to CHC 07A3, reflecting estimates of real-world activity in these countries: Austria, Belgium, Bulgaria, Croatia, Czech Republic, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Netherlands, Norway, Poland, Portugal, Romania, Russia, Serbia, Slovakia, Slovenia, Spain, Sweden, Switzerland, UK. Copyright IQVIA. All rights reserved. 2) Pinto-Bonilla JC et al. Ther. Clin Risk Manag 2015:11;595-603 3) Schmidl D et al. Cornea 2015;34(4):421-6 TP24032
& Protects
Suitable for all dry eye sufferers including contact lens wearers A single drop Lasts for up to 4 hours3 Clinically Proven2 to treat symptoms of dry eyes
www.theapharma.ie
Deirdre Bannon, a 78 year old widow from Tallaght, is delighted to be able to attend the RVEEH nursing services, based in Kilnamanagh/Tymon Primary Care Centre. Speaking about her positive experience, Deirdre explained how she was diagnosed with glaucoma several years ago after she was screened for pressure in her eye Following an initial examination, Deirdre was referred to the Royal Eye and Ear Hospital for further treatment.
“While attending my regular appointment in the Eye and Ear Hospital in January 2024, I was offered an outpatient appointment at Kilnamanagh/Tymon Primary Care Centre, which is a local clinic near my home in Tallaght. The
nurse asked me if I would like to go along there for my appointments. I jumped at the chance as it was so close to where I live. The care is amazing. They keep a good eye on me and it’s such an added bonus to have my care locally in Tallaght,” Deirdre added.
An estimated 10% to 30% of the population older than 40 years suffers from some degree of dry eye disease (DED). The condition tends to affect people above 60, and it is more common in women than men.

Around one in 13 people who are in their fifties experience dry eye syndrome, and the condition becomes more common with age. Up to a third of people age 65 or older may have dry eye syndrome.
DED, which is also sometimes referred to as dry eye syndrome or keratoconjunctivitis sicca, is considered the most prevalent ophthalmic disorder that affects the anterior eye and is most often associated with the aging process, especially in postmenopausal women.
DED is a multifactorial disease of the tears and ocular surface that results in discomfort, tear film instability, and visual disturbance, with potential for damage to the ocular surface. DED can be classified as chronic or temporary. DED can be also attributed to Bell palsy, collagen disorders such as rheumatoid arthritis, corneal or eye lid defects, Sjögren syndrome, and thyroid-related eye disease.
Other medical conditions associated with DED include diabetes, lupus, and scleroderma.
Dry eyes can be caused by ordinary things that increase tear evaporation, such as looking at a computer screen too long; being outside in windy, dry conditions; or just being tired. Cigarette smoke may also cause dry eyes. Other common causes of dry eye include:
• Aging. Tear production tends to decline with age. Dry eyes are common in individuals older than 50 years.
• Gender. A deficiency of tears is more common in women, especially with hormonal changes caused by pregnancy, the use of birth control pills, or menopause.
• The use of cold or allergy medicines, antidepressants, and drugs for high
blood pressure; acne; birth control; and Parkinson’s disease.
• Wearing contact lenses.
• An eye injury or other problem with your eyes or eyelids.
• Diabetes.
• Thyroid disorders.
• Vitamin A deficiency. Symptom Checker
The symptoms of dry eye syndrome usually affect both eyes and may include:
• feelings of dryness, grittiness or soreness, which get worse throughout the day
• redness of the eyes
• watering eyes, particularly when exposed to wind
• eyelids that stick together when waking up These symptoms may get worse in smoky or hot environments.
When a patient presents with symptoms of a dry eye condition, such as irritation, grittiness, burning, soreness, watery eyes and visual disturbances generally affecting both eyes, a detailed history should be recorded by the pharmacist because it may elicit information about contributing factors.
Briefly, this should include details of the signs and symptoms, duration of symptoms and exacerbating factors, such as the environment, changes in humidity or computer use.
It should also record details of topical and systemic medicines taken by the patient,
whether the patient wears contact lenses and if the patient has any dermatological, inflammatory or other systemic diseases.
A differential diagnosis for other eye conditions (such as conjunctivitis, allergy and acute red eye) should be established because initial presentation may be similar.
Once dry eye syndrome develops, some people have recurring episodes for the rest of their lives. There is no cure for dry eye syndrome, but a range of treatments can control the symptoms. In rare cases, more severe cases of dry eye syndrome may require surgery.
The ultimate goal of dry eye treatment focuses on symptomatic relief, usually using tear supplements. Despite this, the underlying mechanism of symptomatic improvement with tear supplementation is still poorly understood. It is thought that increased tear volume, improved tear stabilisation, reduced tear osmolarity or a dilution of inflammatory biomarkers or a combination of these factors play a vital role.
Topical ocular lubricants are the mainstay of dry eye treatment, with the choice of tear substitute depending on the severity of the condition. Pharmacological interventions in all forms of dry eye conditions range in formulation, such as drops, sprays, gels and ointments.
For occasional or mild dry eye symptoms, OTC eyedrops (artificial tears) used regularly may provide relief. Preservative-free artificial tears are preferred, as they cause less irritation. Wearing glasses or sunglasses that fit close to the face (wraparound shades) or that have side shields can help slow tear evaporation from the eye surfaces. An indoor air cleaner to filter dust and other particles can help prevent dry eyes, as can a humidifier by adding moisture to the air, avoiding dry conditions, and allowing the eyes to rest when performing activities that require someone to use their eyes for long periods of time.
Pharmacologic agents that have anticholinergic properties—including antihistamines, decongestants, and antihypertensives such as antidepressants, beta-blockers, and diuretics— are common causes of DED.
Additionally, DED may be caused or exacerbated by allergens and environmental conditions such as dry climates, failure to blink regularly when staring for long periods at electronic devices, smoke, and wind. Laser eye surgery may also cause temporary dry eye.








pH balanced – helps prevent stinging
isotonic – won’t dehydrate eyes
contact lens friendly
















HYLO® - FORTE For more severe and persistent dry eye symptoms
HYLO® - FRESH For milder dry eye symptoms such as tired, red eyes caused by screen use
HYLO® -TEAR For more moderate dry eye symptoms and discomfort caused by contact lens
HYLO® - NIGHT For severe dry eye symptoms that persist throughout the night
HYLO - CARE® Contains Dexpanthenol which supports healing post-surgery or after

HYLO® - DUAL Contains Ectoin which creates a shield against allergens and tear evaporation
HYLO DUAL INTENSE® For intense inflammatory dry eye symptoms such as stinging and burning

When the SARS-CoV-2 pandemic hit, infectious respiratory diseases were brought into an unprecedented, generalized focus, with COVID-19 dominating news, politics and our daily lives. MURTIs are complex viral diseases of the upper airways caused by a diversity of viruses, including multiple strains of rhinovirus, coronavirus, influenza virus, parainfluenza virus, respiratory syncytial virus, adenovirus, enterovirus and other viruses.
In summary, MURTIs are amongst the most widespread infectious diseases. On average, adults have two to four MURTIs, whilst children have six to eight MURTIs per year, accounting for millions of lost working and school days and relevant direct costs per episode.
Treatment of MURTIs is purely symptomatic and can speed up recovery and mitigate symptoms. A variety of OTC solutions, home remedies, food supplements or plant-derived substances, which can be taken orally, topically or via inhalation, are available and proven to soothe symptoms by influencing different functional triggers within the molecular network of symptom development.
Nasal decongestants, mainly sympathomimetics, constrict nasal blood vessels; antitussives centrally or topically reduce the frequency and/or intensity of coughs and include opium alkaloids and derivatives; expectorants increase the production of fluid in the lungs to reduce the viscosity of mucus, whilst mucolytics break down disulfide bonds in the mucus.
First-generation antihistamines block both histaminic and muscarinic receptors and, in contrast to second-generation antihistamines, pass the blood–brain barrier, exerting their effects on the cough centre located in the brainstem. With their anticholinergic properties, they disrupt nerve signals that trigger mucus secretion and sneezing. Analgesics can provide relief from pain-associated common cold symptoms and may be antipyretic.
AUTHOR: Andrew Smith1, Peter Kardos2, Oliver Pfaar3, Winfried Randerath4,5, Guillermo Estrada Riolobos6, Fulvio Braido7,8, Laura Sadofsky9
1School of Psychology, Cardiff University, Cardiff, UK
2Centre of Allergy, Respiratory and Sleep Medicine, Maingau Clinic of the Red Cross, Frankfurt am Main, Germany
3Department of Otorhinolaryngology, University Hospital Marburg, Philipps University Marburg, Marburg, Germany
4Institute of Pneumology, University of Cologne, Cologne, Germany
5Bethanien Hospital, Clinic of Pneumology and Allergology, Centre for Sleep Medicine and Respiratory Care, Solingen, Germany
6Spanish Society Clinical Family and Community Pharmacy (SEFAC), Paseo de las Delicias, Madrid, Spain
7Istituti di Ricovero e Cura a Carattere Scientifico, Ospedale Policlinico San Martino, Genova, Italy
8Università di Genova, (DIMI), Genova, Italy
9Hull York Medical School, University of Hull, Hull, UK
1. REFLECT - Before reading this module, consider the following: Will this clinical area be relevant to my practice?
2. IDENTIFY - If the answer is no, I may still be interested in the area but the article may not contribute towards my continuing professional development (CPD). If the answer is yes, I should identify any knowledge gaps in the clinical area.
3. PLAN - If I have identified a
knowledge gap - will this article satisfy those needs - or will more reading be required?
4. EVALUATE - Did this article meet my learning needs - and how has my practise changed as a result? Have I identified further learning needs?
5. WHAT NEXT - At this time you may like to record your learning for future use or assessment. Follow the
4 previous steps, log and record your findings.
Published by IPN. Copies can be downloaded from www.irishpharmacytraining.ie
Disclaimer: All material published is copyright, no part of this can be used in any other publication without permission of the publishers and author. P&G has no editorial oversight of the CPD programmes included in these modules
This position paper addresses the treatment of mild upper respiratory tract infections (MURTIs) and provides recommendations for best practice. When the SARSCoV-2 pandemic hit, infectious respiratory diseases were brought into an unprecedented, generalized focus, with COVID-19 dominating news, politics and our daily lives. COVID-19 is a potentially life-threatening disease, affecting many organ systems and requiring intensive care in many patients.1,2 However, patients may also experience milder forms of COVID-19 with flu-like symptoms1,3,4 and the evolution of the virus as well as increased immunity of general populations have led to a decline in hospitalizations and deaths during the year 2022.2 Today, SARS-CoV-2 variants often lead to MURTIs, suggesting that a review of our knowledge of MURTIs is desirable and a reassessment of treatment options is required. Unmet needs and appropriate healthcare provision will also be evaluated. Background information on the cause and outcomes of MURTIs provides the context for discussion of treatment.
MURTIs are complex viral diseases of the upper airways caused by
a diversity of viruses, including multiple strains of rhinovirus, coronavirus, influenza virus, parainfluenza virus, respiratory syncytial virus, adenovirus, enterovirus and other viruses (Table 1). In total, it is estimated that >200 viral strains are causative for MURTIs.1,5 Viral incubation typically lasts 2–4 days.6 Depending on the viral trigger, MURTIs can manifest as a common cold – a self-limiting disease in immunocompetent patients, usually lasting about 2 weeks. The multiple symptoms related to MURTIs (Table 2) are not directly caused by the viruses but by an immune-mediated response to them,7,8 and include typical nasal congestion, cough, sore throat and malaise, which causes a strong negative effect on general well-being.9
The large number of viral strains involved in MURTIs means that the development of effective vaccines is complex as respiratory viruses frequently mutate to escape immunity8 but important public health messages can help prevent or reduce infections.20,21 In most cases, antibiotics are inappropriate, as they do not influence the natural history of viral MURTIs but contribute to antibiotic
resistance22–27 and possible side effects such as gastrointestinal symptoms and allergies. Drugs for symptomatic treatment of different manifestations of MURTIs are available, including analgesics, decongestants, antihistamines, herbal remedies or essential oils for inhalation, oral and transdermal application.28,29 Many of these over-the-counter (OTC) drugs have proven efficacy in improving symptoms and QoL in both randomized controlled trials (RCTs)30 and clinical use.31
In summary, MURTIs are amongst the most widespread infectious diseases.5,32,33 On average, adults have two to four MURTIs, whilst children have six to eight MURTIs per year,5 accounting for millions of lost working and school days5 and relevant direct costs per episode.34
Pathophysiology and current management of MURTIs
Clinical symptoms of the common cold, flu and other MURTIs are caused by an immune response to the viruses7,8,35,36 (Figure 1). Typically, the onset of symptoms is staggered.37 Symptoms of MURTIs include sore throat, nasal congestion, nasal discharge, sneezing, headache, cough

Table 1
Common cold-causing viruses.
Virus The estimated annual proportion of cases
Rhinoviruses 30–50%
Coronavirusesa 15–30%
Influenza viruses 5–15%
Respiratory syncytial virus 5%
Parainfluenza viruses 5%
Adenoviruses <5%
Enteroviruses <5%
Metapneumovirus Unknown Unknown 20–30%
aThere are four human coronaviruses known to cause symptoms of the common cold: 229E, NL63, HKU1 and OC43; this does not include SARS, MERS and SARS-CoV-2.119
Based on data from Heikinnen and Järvinen.5
Table 2.
Table 2
and general malaise (Table 2).38 Symptom triggers include bradykinin release into the pharyngeal area as a cause for sore throat;39 cytokine release causing breakdown of muscle tissue to facilitate protein release to support responsiveness leading to general aches, headaches and pain as well as fever;40–44 bradykinin and histamine release cause nasal congestion due to vasal dilation;39,45,46 and excess mucus production and inflammatory mediators47,48 sensitize and stimulate sensory nerve endings, inducing cough, which starts around 48 hours after the onset of symptoms and usually persists well beyond all other symptoms.37 Because the viruses do not directly cause the symptoms, it is difficult to distinguish the common cold, influenza and mild COVID-19 based on clinical presentation only, though some differences are observed.1,3,4,49
Common symptoms of common cold, influenza and COVID-19.
Common symptoms of common cold, influenza and COVID-19.
Common cold Influenza COVID-19a
Gradual onset of symptoms Abrupt onset of symptoms Symptoms range from mild to severe Abrupt onset of symptoms
Length of symptoms <14 days 7–14 days 7–25 days
Sneezing
Runny or stuffy nose
Shortness of breath
Wheezing
Treatment of MURTIs is purely symptomatic and can speed up recovery52–54 and mitigate symptoms. A variety of OTC solutions, home remedies, food supplements or plant-derived substances, which can be taken orally, topically or via inhalation, are available28,29,55 and proven to soothe symptoms by influencing different functional triggers within the molecular network of symptom development (Figure 1). Intense research over the last decades has not only led to a better understanding of these triggers but also of the molecular processes of a diversity of pharmacologically active substances, thus allowing for more holistic approaches with adapted and well-targeted therapies.
Nasal decongestants, mainly sympathomimetics, constrict nasal blood vessels;56 antitussives centrally or topically reduce the frequency and/or intensity of coughs and include opium alkaloids and derivatives;57 expectorants increase the production of fluid in the lungs to reduce the viscosity of mucus, whilst mucolytics break down disulfide bonds in the mucus.55 Firstgeneration antihistamines block both histaminic and muscarinic receptors and, in contrast to second-generation antihistamines, pass the blood–brain barrier, exerting their effects on the cough centre located in the brainstem. With their anticholinergic properties, they disrupt nerve signals that trigger mucus secretion and sneezing.58 Analgesics can provide relief from pain-associated common cold symptoms and may be antipyretic.31
In addition, pharmacologically active plantbased molecules are also treatment options, including systemic use, topical application or inhalation of aromatics such as menthol, eucalyptus, camphor, thyme, ivy, primrose, African geranium, gentian root, elderflower and vervain herb, which have long been wellestablished treatments for respiratory tract illnesses. As an example, so-called inhaled therapeutic vapours are widely used to improve breath, reduce cough, improve alertness and improve sleep during a common cold.36
a Information is still evolving.1,3,4,49
a Information is still evolving.1,3,4,49
Note: Som e symptoms of the common cold and COVID-19 overlap, and testing may be required for a correct diagnosis. Symptoms for seasonal allergies without asthma.
Note: Some symptoms of the common cold and COVID-19 overlap, and testing may be required for a correct diagnosis. Symptoms for seasonal allergies without asthma.
The large number of viral strains involved in MURTIs means that the development of effective vaccines is complex as respiratory viruses frequently mutate to escape immunity8 but important public health messages can help prevent or reduce infections.20,21 In most cases, antibiotics are inappropriate, as they do not influence the natural history of viral MURTIs but contribute to antibiotic resistance22–27 and possible side effects such as gastrointestinal symptoms and allergies. Drugs for symptomatic treatment of different manifestations of MURTIs are available, including analgesics, decongestants, antihistamines, herbal remedies or essential oils for inhalation, oral and transdermal application.28,29 Many of these over-the-counter (OTC) drugs have proven efficacy in improving symptoms and QoL in both randomized controlled trials (RCTs)30 and clinical use.31
In this respect, a recent systematic review59 provided an overview of pharmacological studies showing that aromatic compounds
Professional Development supported by

derived from natural plant extracts, such as menthol, eucalyptus or camphor, have multifaceted modulating effects on transient receptor potential channel (TRP) ‘cough’ receptors and/or the nasal TRP channels, such as TRPM8 and TRPA1,59,60 and therefore have the potential to target multiple symptoms during the course of MURTIs.38,59,61 Clinically, the modulation of TRP receptors results in a significant reduction of coughing, as shown in three single-blind crossover studies (total n=104 with induced cough),62 where a combination of menthol, camphor, eucalyptus, turpentine oil, cedar leaf oil, myristica oil and thymol significantly reduced the frequency of coughing more effectively than single components. Further, the application of this combination significantly reduced the time taken for the feeling of nasal cooling and, importantly, the time to nasal decongestion compared with a petrolatum control (reviewed in detail by Smith and Matthews36). Nasal airflow also influences TRP channels,38,61 and the absence of nasal airflow is associated with the feeling of stuffiness. Interestingly, as recently described in a systematic review by Stinson et al.,59
The clinical efficacy of a combination of cowslip, gentian root, black elder, sorrel and common vervain was tested in patients with acute viral rhinosinusitis (n=386)65 in a placebo-controlled, double-blind, randomized trial and led to a significant and clinically relevant improvement of the investigator-assessed symptom score on day 15.65 Furthermore, a combination of gentian root, primula flower, sorrel, elderflower and common vervain was shown to be safe and efficacious in patients with acute rhinosinusitis in phase IIb/III and phase III placebo-controlled clinical trials (total n=589).66
Wagner et al.67 and Kardos68 provided summaries of the efficacy of plant-based cough remedies. A preparation of Andrographis paniculata (creat or green chiretta) was tested against placebo in five RCTs and versus echinacea and bromhexine in a sixth study (total n=807),67 and was shown to significantly improve cough-related symptoms in all but one small pilot study.67,69 Four RCTs using a preparation based on ivy, primrose and thyme (total n=1428)67 showed strong evidence for the beneficial effect of this combination, which not only reduced the frequency and severity of cough but also facilitated secretolysis.67,70 Furthermore, oral treatment with a syrup containing thyme and ivy was shown to significantly reduce the Bronchitis Severity Score and cough severity and to improve health-related QoL in an observational, prospective, uncontrolled study (n=730).71
A meta-analysis of randomized, placebocontrolled trials assessing the effects of a Pelargonium sidoides extract (South African geranium), including 11 trials and 2195 patients,72 showed a reduced burden, earlier remission of cough and an increase in disease-
Sensitization of airway receptors
Viral infection of nasal epithelial cells
Sneezing Sore throat
Inflammatory mediators
associated QoL. Furthermore, patients treated with the geranium extract felt able to resume normal daily routines sooner than those treated with pacebo.72 Several studies on the effect of a mixture of eucalyptus oil, sweet orange oil, myrtle oil, and lemon oil on coughing have been published. For example, Gillissen et al. reported significantly superior effects to placebo in coughing-related endpoints and sleep disturbances (n=413).53 Fürst et al. reviewed clinical trials conducted with this mixture in China and confirmed its efficacy in the treatment of respiratory tract diseases in a Chinese patient population.73
Not all studies successfully demonstrated the efficacy of essential oils in objective endpoints or lasting superiority over placebo controls, but they still reported efficacy in subjective endpoints, including the nasal sensation of airflow and quality of sleep.38,75–77 Assessment of patient-centric outcomes is part of evidencebased medical research and should thus not be disregarded; symptomatic relief perceived by the individual patient is considered beneficial for well-being, stress reduction, and sleep quality9,78 and is considered the most important endpoint in a self-limiting disease. Even in RCTs of asthma, interstitial lung disease and chronic obstructive pulmonary disease, the FDA and EMA require patient-related outcomes (i.e. QoL Questionnaires) as coprimary or secondary endpoints. Given the importance of sleep for overall survival and individual well-being, treatment of sleep disorders and increasing quality of sleep are essential to improving immunity15,78; thus, further studies and focus are required on holistic health approaches in the therapy of MURTIs.
In conclusion, relief of nasal congestion and cough is key to breathing appropriately, which
is not only important for sleep quality but also relevant for well-being. This may further lead to reduced susceptibility to infection and faster recovery, highlighting the importance of these therapeutic targets and holistic approaches, and acknowledging the significance of patientcentric efficacy outcomes.
Unmet needs in public health messages
Despite the high number of patients affected by and costs associated with MURTIs, many have regarded these diseases as nothing more than a slight nuisance, adding to the somewhat neglected status of MURTIs. With the pandemic, a new public and medical focus towards MURTIs has developed. As previously stated, clinical symptoms may overlap between the common cold, influenza and mild COVID-19, making identification of the infecting agent difficult (Table 2). Diagnostic testing may be required depending on local regulations or at the health professional’s recommendation based on risk factors.
Whilst MURTIs are usually not severe or lifethreatening diseases in immunocompetent patients, immunocompromised patients are at risk of developing lower respiratory tract infections, including potentially fatal conditions like bronchiolitis obliterans syndrome in transplant patients15 and pneumonia.79 Viral MURTIs may also cause exacerbation of pre-existing respiratory conditions such as asthma,80 chronic obstructive pulmonary disease,81 allergic rhinitis,80 chronic rhinosinusitis,82 interstitial lung diseases83 and breathing disturbances during sleep.84
In the paediatric patient population, the use of OTC cough and cold medications often lacks support from well-designed, contemporary research and proof of efficacy.85–88 However,

OTC products for the treatment of MURTIs are widely used, increasing the trend of abuse and potential toxicity,85,86,88 which may even cause fatalities if used in very young children and/or in overdose.89
Finally, yet importantly, a holistic medical approach allows for centring the attention of medical professionals on the individual patient’s needs, preferences and well-being, thus triggering compliance, increasing security, and reducing psychosocial stress and chronic disease.92 Stress, as described, can negatively influence immunity and could contribute to the worsening or prolongation of the symptoms of MURTIs.93
The pandemic has also put a new focus on the importance of pharmacists as first-line health consultants. MURTIs are the main cause of consultation in both primary care and community pharmacy. With long opening hours and short anticipated waiting times, pharmacies provide low-threshold access to medication advice and play an important role as the primary point of contact with a healthcare professional as patients consult the pharmacist for health advice, health literacy and social support.99,100 During the COVID-19 pandemic, a wide range of pharmaceutical interventions was provided, and expanded powers were granted to pharmacies, enhancing their role in a multi-professional healthcare setting.101 Furthermore, approximately 20% of consultations with doctors could be dealt with by other professionals like pharmacists, which would save 1 out of every 5 hours of medical doctor time that could be devoted to other activities or patients.102
Enhancing the role of pharmacists as firstline consultants within applicable local legal boundaries could further reduce the use of antibiotics in patients with the common cold. Pharmacists are highly educated, giving advice regarding symptomatic treatment for viral upper and lower respiratory tract infections. Patients may erroneously believe that antibiotics could aid in the fast relief of symptoms. However, antibiotics were shown to be ineffective in changing the natural history of common colds and are not indicated in uncomplicated MURTIs, as shown in a number of RCTs and observational studies.22–27,103 Still, 41% of all antibiotic prescriptions are for respiratory conditions,104 which is one of the most important contributions to antibiotic resistance, whilst several scientific societies advise against antibiotic use in MURTIs.105–107
Symptomatic therapies versus subjective endpoints?
Despite accumulating clinical evidence and a better understanding of the mode of action of available pharmacotherapies, the availability of data, especially regarding the efficacy of plant-based substances, may often be considered limited due to study designs or endpoints used in clinical trials.
With patient-centred research becoming more important in medical care and research, patient-reported outcomes should not be disregarded because, beyond contagiousness, this is the cause of absenteeism from work or school. With various OTC products available, including plant-based medicines, patients can benefit from tailored treatment approaches when experiencing MURTIs. The scheme summarizes chemical and plant-based actives as classified by World Health Organization Anatomical Therapeutic Chemical/Defined Daily Dose (WHO ATC/DDD) Index Codes108 and by the latest research findings described earlier, and presents available treatment options for the therapy of MURTI symptoms over the illness time course. Treatment options include throat preparations to treat sore throat at the beginning of the illness (day 1); analgesics, non-steroidal anti-inflammatory drugs and antipyretics to treat pains and fever as well as nasal decongestants to treat nasal congestion (days 2–5); and cough suppressants and expectorants to treat cough (days 6–7). Plant-based medicines are often provided as either single substances or as combinations of various essential oils (e.g. inhaled therapeutic vapours), potentially covering all phases of MURTIs with one pharmaceutical product. Therefore, (co)treatment with plant-based combinations is considered suitable to treat multiple symptoms throughout the course of the illness, as shown by in vitro data and/or clinical trials.37,59
Overall, plant-based medicines and honey may provide a safer alternative to synthetic OTC products in a paediatric patient population,87,109,110 with similar or improved efficacy.111,112 Safety and tolerability endpoints for plant-derived substances are reported in the literature66,67,75,113 but are also limited in children,114 with reported adverse events being of a mild irritant nature.115
An estimated one-third of patients in the United States prefer and use ‘natural alternatives’ to chemical drugs in the treatment of the common cold and influenza, including plant-derived essences. Consideration of integrative therapies could thus increase patient compliance and positive patient care experiences.116
The first pillar on which our best practice recommendations to manage MURTIs are based is health literacy. Educated patients can understand and crosslink the interdependency and general importance of a healthy lifestyle, good sleep quality, and their immune system. In the case of MURTIs, healthcare professionals should educate and advise patients about treatment options, facilitating patients to make an educated decision to comply with the selected therapy. Most importantly, patients and caregivers should become aware that antibiotics are inappropriate and ineffective in the treatment of MURTIs,22–27 and vaccines are not available, except for COVID-19, influenza and, most likely soon, respiratory syncytial virus.
The second pillar consists of healthcare professionals creating and improving a complementary multi-professional healthcare setting. Pharmacists play a fundamental role not only in dealing with MURTI symptoms but also in health promotion and education. They can effectively reach patients and disburden medical doctors.102 Ideally, the patient’s initial consultation is with the pharmacist, who advises the patient to closely monitor a potential worsening of symptoms and may recommend suitable OTC or plant-based drugs.
If symptoms worsen, a physician is to be consulted. Home assessment of oxygen saturation could increase patient safety and shorten response times in complicated cases of URTIs. Pulse oximeters have been successfully used for self-monitoring in patients with mild COVID-19;117 they are inexpensive and easy to use118 and could be included in common medicine cabinets like fever thermometers in the future.
Furthermore, home tests for influenza A and B, SARS-CoV-2, and respiratory syncytial virus are available. Thus, acute testing should be advised to monitor for possible post-COVID-19 cases.
The proposed guide for the treatment of patients with MURTIs and/or mild fever includes the following steps:
• Help increase self-awareness for symptoms and symptom development: consider signs of infection and illness, for example, by home assessment of fever and monitoring oxygen saturation using a pulse oximeter as appropriate.
• Focus on optimization of symptomatic treatment: use adapted continuous (co) treatment(s) targeting multiple symptoms of the natural history of MURTIs, considering the patient’s preferences between plantderived remedies and chemical drugs, and avoiding initial antibiotic treatment.
• If relevant, use appropriate rapid tests (e.g. influenza, SARS-CoV-2): this is important for the identification of viruses during endemics and pandemics where targeted causal therapies are available and to monitor contagiousness.
• Advice to seek professional medical attention immediately if symptoms worsen or last longer than 3 weeks.
The COVID-19 pandemic highlighted unmet needs in the management of MURTIs. Antibiotics are still inappropriate for MURTIs, and vaccines are available only for a few viruses, mainly mitigating the symptoms rather than preventing the diseases. Relevant prophylactic measures may include testing and reduced crowd densities in shops and events as well as personal measures such as wearing masks and improved hand hygiene. A strong advancement of patient-centred approaches is required, including the improved appraisal of subjective treatment outcomes within adapted holistic therapies.
References available on request

In October, Chronic Pain Ireland and the University of Galway hosted a pivotal event marking the ‘Day of the Girl.’ The hybrid event, entitled Exploring Sex and Gender Disparities in Chronic Pain, brought together researchers, clinicians, and patients to shed light on the profound differences in how chronic pain is experienced and managed across sexes and genders. The program examined groundbreaking findings, including the influence of the latest PAINDIFF research, and their implications for diagnosis, treatment outcomes, and health policies.

Among the event’s key voices was Dr Michelle Roche, Associate Professor in Physiology, CoDirector of the Centre for Pain Research at the University of Galway, and President of the Irish Pain Society. Dr. Roche’s keynote address, Developing Guidelines and an Agenda for Research on Sex and Gender as Variables in Pain, highlighted the urgent need to address these disparities. In a follow-up interview, she elaborated on her work and vision for the future of pain research.
Can you tell us about your background and what inspired your interest in sex and gender as variables in pain research?
Dr Roche’s career has focused on the neurobiology underlying mood disorders, chronic pain, and their interactions. She noted
a glaring disparity in previous research, much of which focused predominantly on males, despite the higher prevalence of these conditions among females.
She says, “I am an Associate Professor in Physiology with a research focus on understanding the neurobiology underlying mood disorders, such as depression and anxiety; pain and their interactions. Both mood disorders and chronic pain are more prevalent in females compared with males, however up until relatively recently the majority of research conducted in these fields was conducted in males only. This disparity could account, at least in part, for the failure of several clinical trials and lack of new treatment strategies for these conditions, thereby reduce quality of life for individuals experiencing such conditions.
“As a consequence, research in our lab examines the biological mechanisms that underlie sex differences in the comorbidity between mood-disorders and chronic pain. Ultimately, we hope to identify new sexspecific therapeutic targets and management strategies for these conditions, bringing us one step closer to personalised medicine. While research examining the influence of sex on pain has expanded over the past decade, the influence of gender on pain is less well understood, with research in this area is only in its infancy.”
How would you describe the current state of research on sex and gender in pain research? What gaps do you see?
Increased awareness and funding requirements have catalysed a surge in studies examining sex and gender differences across biomedical sciences, including pain. However, Dr Roche identified critical gaps.
She continued, “Increased awareness of the importance of sex and gender as key contributors to health and disease, together with requirements of funding agencies, has resulted in profound increases in the number of studies investigating sex and/ or gender differences across the biomedical sciences including pain. This welcome development has resulted in mounting evidence for genetic, molecular, cellular, systems-level, social and psychological differences in the pathophysiology and experience of pain between and across sexes and genders.
“However, many gaps still remain which we hope will be addressed into the future. For example, there are a lack of studies examining the interaction between biopsychosocial factors and as mentioned, the study of gender as a variable in pain has largely been overlooked. Furthermore, inconsistency in research findings
across and between research groups is widely recognised, and as such there is a pressing need for guidelines on how best to incorporate sex and gender as variables into pain research.”
What are some common misconceptions about sex and gender differences in pain and how can we prevent these misconceptions?
A common misconception in pain research is the conflation of sex and gender, often treated interchangeably and limited to binary categories (male/ female, man/woman). Dr Roche stressed the importance of recognizing sex and gender as distinct, fluid, and dynamic variables that extend beyond binaries. Misunderstandings also persist about the causes of sex differences in pain, often attributed solely to hormonal changes.
“One common misconception is that sex is all that matters, and that gender does not impact on pain experience or management,” she highlighted.
“As such until recently the terms sex and gender were used interchangeably and only on a binary level (male: female and/or, man:women) in both research and clinical settings which has lead to confusion and inconsistencies. It is important that researchers, clinicians and allied health professions are aware of the definitions of sex and gender, acknowledge that this extends beyond the binary, and is fluid and may change over the life course of an individual. As such it is critical that information on both sex and gender is collected where possible to enable full understanding of the influence of these variables on pain.
“Other misconceptions include that increased pain responding and pain experience reported in females is primarily due to changes in hormone levels, despite multiple
“My hope would be that by considering sex and gender as variables in pain research would allow for more personalised treatments and care to be provided to individuals with chronic pain. For example, current data suggests that cox inhibitors and glucocorticoids elicit greater analgesia with reduced side effects in males, while CGRP based treatments and cognitive based therapies may be more effective in females”
lines of evidence demonstrating a wealth of biological, psychological and social factors that underlie the effects of sex on pain. Recognition of the interactions between biopsychosocial factors that underlie the effects of sex and gender is important in dispelling such misconceptions.”
What are your primary objectives for developing guidelines on sex and gender as variables in pain research?
At this time of rapid acceleration in the number and scope of studies examining sex and gender as variables in pain, it is essential that the researchers adopt consistent research approaches in order to maximize progress in this critically important area. With this in mind, with the support of ERA-NET NEURON and the Irish Health Research Board (HRB), we established an international group of 32 interdisciplinary experts, including leading senior scientists, early career researchers and patient advocates, from 22 different institutes across 8 countries, termed the PAINDIFF Network. The aim of this network are to (1) identify areas of methodological consensus and best practice, (2) identify gaps in the knowledge or inconsistencies in the application and (3) develop a set of consensus recommendations on how best to study sex and gender as variables in pain research To achieve this we have conducted an extensive review of the literature, completed a comprehensive
will reduce variability and lack of reproducibility between laboratories, and improve the translatability of research findings, ultimately leading to better outcomes for people living with chronic pain. Furthermore, much of the content and recommendations will have general relevance beyond pain research specifically, to many different areas of neuroscience research where sex and gender are studied as variables,” she added.
How can researchers be encouraged to incorporate sex and gender considerations into their studies?
and glucocorticoids may work better in males, while CGRPbased therapies and cognitive interventions could be more effective for females. By deepening our understanding of sex and gender in pain, researchers can tailor treatments to individual needs.
Emerging technologies such as machine learning and AI also offer exciting possibilities for integrating sex and gender considerations into research design.
international survey of the pain research community on current methodological practices and opinions, and following extensive discussion and consensus, have developed a set of guidelines and recommendations for the best practice study in this area.
Can you outline the main components that you think should be included in these guidelines?
Dr Roche envisions these guidelines being widely adopted in clinical, research, and hospital settings. Beyond pain research, they hold relevance for neuroscience and other disciplines studying sex and gender variables. Implementation could lead to better reproducibility, personalized treatments, and improved outcomes for diverse patient populations.
“The PAINDIFF guidelines and recommendations will focus on experimental design, methodological approaches, data analysis, presentation and reporting when conducting pain research with sex and/or gender as variables. Furthermore, specific recommendations pertaining to key stakeholders including researchers, ethics boards, journal editors and funding bodies will be highlighted.”
How do you envision these guidelines being implemented in clinical and research settings as well as in hospital settings?
“Adoption and implementation of these recommendations
“Due to the increased awareness of the importance of including sex and gender into research, and requirements from funding agencies, there has been a steady increase in the number of studies including all sexes and/or genders in their design. However, data from our survey and published data demonstrate that the majority of studies choose not to analyse or report on sex specific effects, and rarely examine effects of gender.
“We propose that by analysing and reporting on the effects of sex and/or gender in their studies, researchers will yield additional rich source of data, understanding and insight, into sex and gender specific pain management and treatment options. In studies were sex and gender are not the primary outcome variable, analysis and reporting will enable future researchers to use and combine data across studies to provide greater understanding of pain perception and management across a diverse sex and gender population.
“However, there also needs to be widespread recognition and acknowledgement by funding agencies, journal editors, journal and grant reviewers, regulatory authorities, institutional ethics committees, facilities management and researchers themselves, that greater resources are required to study sex and gender as variables in pain research, including but not limited to financial resources, infrastructural resources, time, and human resources.”
Looking ahead, what are your hopes for the future of pain research as it relates to sex a nd gender?
Dr Roche’s hopes for the future of pain research are rooted in the promise of personalised medicine. For example, certain treatments like COX inhibitors
“My hope would be that by considering sex and gender as variables in pain research would allow for more personalised treatments and care to be provided to individuals with chronic pain. For example, current data suggests that cox inhibitors and glucocorticoids elicit greater analgesia with reduced side effects in males, while CGRPbased treatments and cognitive based therapies may be more effective in females. Thus, expanding our understanding of the influence of sex and gender in pain will ultimately result in better pain treatment and management for all.”
Are there any emerging trends or technologies that you believe could enhance our understanding of sex and gender in pain?
Dr Roche adds, “There are many trends and technologies that will enable and facilitate the incorporation of sex and gender into research practice, including machine learning and AI. Multi-and inter-disciplinary research from basic science through to clinical practice and back, is critical to further our understanding in this field. However at the outset, researchers need to consider these variables in their research design and analysis and the PAINDIFF guidelines will provide them with a framework on how to undertake such studies.”
She concluded, “Integrating sex and gender specific approaches into pain research and clinical practice will improve development of novel therapeutic strategies (pharmacological and non-pharmacological) for all. However, collaboration between researchers, healthcare professionals, funders and policymakers will be crucial in addressing sex and gender disparities in pain care and management.”
Written by Dr Ishapreet Kaur1 and Mr David Galvin1


Bladder cancer affects around 540 people in Ireland every year. It accounts for 15 percent of cancer cases worldwide.1 Most cases (90%) are transitional cell carcinomas (TCC), arising from bladder lining cells, while squamous cell carcinoma (SCC), often linked to chronic infection, accounts for most of the rest.1
Major risk factors include smoking, occupational exposure to harmful chemicals, obesity, and the male sex. Inflammation of bladder tissue is the primary SCC risk, especially in regions with schistosomiasis.2-3
Bladder cancer commonly presents with painless haematuria (85%), up to 20% of patients present with urinary urgency or suprapubic pain. A small cohort of patients (3%) will present with nonvisible haematuria.4
Diagnosis involves cystoscopy and upper tract imaging which includes either CT Urogram or a renal ultrasound. Some hospitals within the HSE have developed protocols with the triage of patients with Visible Haematuria including the introduction of Haematuria Clinics which echo principles of the triple-assessment breast clinics. Patients triaged into these clinics will have their urine cytology taken, flexible cystoscopy completed and upper tract imaging filmed on the day of their visit.
With their diagnosis, patients with frank haematuria will undergo CT Thorax Abdomen and pelvis with a Urographic phase to assess local spread and metastasis. The TNM staging system assesses tumour invasion, nodal involvement, and metastasis, while grading determines tumour cell differentiation.5
Over the past decade the use of immunotherapy can be considered in patients that are unfit for cisplatin/carboplatin treatment.8
Additionally, the use of immune check-point inhibitors led to longer survival in patients with metastatic bladder cancer
Treatment and management of bladder cancer depends on the invasive nature of the cancer. The bladder wall consists of three layers, the innermost mucosa, muscularis propria (or detrusor
muscle), and the outermost serosa and peri-vesical fat. Patients are then classified to have non-muscle invasive bladder cancer (NMIBC), muscle invasive bladder cancer (MIBC), and metastatic disease.
Non Muscle Invasive Bladder Cancer
80% of patients have NMIBC.1 They will undergo a transurethral resection of bladder tumour (TURBT) with the intention of making a correct histological diagnosis and to remove all macroscopic lesions. This is sometimes followed by intravesical chemotherapy (Mitomycin C or Epirubicin) which has an ablative effect on residual tumour cells. It has shown to reduce recurrence soon after the TURBT, and is generally offered to high grade lesions.6
Intravesical BCG (bacillus Calmette-Guérin) decreases the risk of recurrence in intermediate and high risk tumours. It is given at least two weeks after TURBT and the initial induction course involves six, weekly treatments. Following this, maintenance therapy as per the Lamm regime can be commenced.7 These involve once-weekly instillations for three weeks at three months, six months, and 12 months posttumour resection. Three onceweekly courses every six months

Available over the counter. No prescription required. Always read the leaflet. Available in a 4 or 8 pack.
ABBREVIATED PRESCRIBING INFORMATION
Product Name: Sidena 50 mg Tablets.
Composition: Each tablet contains, 50 mg sildena l (as citrate) .
Description: Light blue, round, slightly dotted tablets. Cross breaking notch on one side and marked ‘50’ on the other side. Can be divided into equal quarters. (Only two quarters of the 50 mg is covered by posology).
Indication(s): Treatment of men with erectile dysfunction, which is the inability to achieve or maintain a penile erection su cient for satisfactory sexual performance.
Dosage: Adults and elderly: 50 mg taken as needed approximately one hour before sexual activity. Dose may be decreased to 25 mg. Max dose: 50mg once daily. Impaired renal and hepatic function: Sildena l clearance is reduced in hepatic and severe renal impairment. Consider a dose of 25 mg. Dose may be increased step-wise to 50 mg if tolerated. Children and adolescents below 18 years of age: Contraindicated. Use in patients using other medicines: Starting dose of 25 mg with CYP3A4 inhibitors (not advised to use with ritonavir). To minimise postural hypotension in patients receiving and alpha-blocker, stabilise patient rst on the alpha blocker and use a starting dose of 25 mg sildena l. Contraindications: Hypersensitivity to sildena l or any of the excipients. Concomitant with ritonavir, nitric oxide donors or nitrates in any form, guanylate cyclase stimulators e.g. riociguat. In patients that sexual activity is inadvisable (e.g. severe cardiovascular disorders such as a recent (6 months) acute myocardial infarction (AMI) or stroke, unstable angina or severe cardiac failure). Refer these patients to a doctor. Patients with loss of vision in one eye due to NAION. Known hereditary degenerative retinal disorders. Severe hepatic impairment. Hypotension. Anatomical deformation of the penis. Not intended if no erectile dysfunction. Women. Warnings and Precautions for Use: First diagnose erectile dysfunction and determine potential underlying causes (e.g. hypertension, diabetes mellitus, hypercholesterolaemia or cardiovascular disease), before considering pharmacological treatment. Consider the cardiovascular status of patients, since there is a degree of cardiac risk associated with sexual activity. Serious cardiovascular events, including myocardial infarction, unstable angina, sudden cardiac death, ventricular arrhythmia, cerebrovascular haemorrhage, transient ischaemic attack, hypertension and hypotension have been reported post-marketing in temporal association with the use of sildena l. Most, but not all, of these patients had pre-existing cardiovascular risk factors. Sildena l has vasodilator properties, resulting in mild and transient decreases in blood pressure. Caution: Patients with anatomical deformation of the penis (such as angulation, cavernosal brosis or Peyronie’s disease), or in patients who have conditions which may predispose them to priapism (such as sickle cell anaemia, multiple myeloma or leukaemia). Advise patients that in case of priapism, prolonged erections (longer than 4 hours) or sudden visual defect, they should stop taking sildena l and consult a physician immediately. Administer to patients with bleeding disorders or active peptic ulceration only after careful bene t-risk assessment, as there is no safety information available. Interactions: See SPC for detailed information. Inhibitors of the cytochrome P450 (CYP) isoforms 3A4 (major route) and 2C9 (minor route) isoenzymes such as CYP3A4 inhibitors: Itraconazole, ketoconazole, erythromycin, cimetidine, HIV protease inhibitor saquinavir: May reduce sildena l clearance and increase sildena l plasma levels. Consider a starting dose of 25 mg. Strong CYP3A4 inducers e.g. rifampicin may increase sildena l clearance and decrease sildena l plasma concentrations. Grapefruit juice: May give rise to modest increases in plasma levels of sildena l. Nicorandil (Hybrid of potassium channel activator and nitrate): Due to the nitrate component it has the potential to have serious interaction with sildena l. Sildena l potentiates the hypotensive e ect of nitrates. Alpha blocker: Concomitant administration of sildena l may lead to symptomatic hypotension in a few susceptible individuals. Patients should be hemodynamically stable on alpha-blocker therapy prior to initiating sildena l treatment. Sildena l potentiates the antiaggregatory e ect of sodium nitroprusside in vitro. Not recommended in patients with a history of bleeding disorders or active peptic ulceration. Not recommended to use with other pulmonary arterial hypertension treatment containing sildena l. Caution when sildena l is initiated in patients treated with sacubitril/valsartan. May result in a increase of bosentan availability. Ability to Drive and Use Machinery: Minor in uence, dizziness and altered vision were reported. Patients should be aware of how they react to sildena l before driving or using machinery. Undesirable E ects: Very common: Headache. Common: Dizziness, visual disorders, visual colour distortion, vision blurred, ushing, hot ush, nasal congestion, nausea, dyspepsia. See SPC for more adverse e ects.
Marketing Authorisation Holder: Rowex Ltd, Bantry, Co. Cork. Marketing Authorisation Number: PA 0711/170/002. Further information and SPC are available from: Rowex Ltd., Bantry, Co. Cork. Freephone: 1800 304 400 Fax: 027 50417
E-mail: rowex@rowa-pharma.ie
Legal Category: Not subject to medical prescription. Date of Preparation: Jan 2024
of preparation: (10-24) CCF: 26643
are recommended thereafter up to three years post-resection.
Common side-effects include dysuria, frequency, and malaise with a mild fever for up to 24 hours post-instillation. If patients develop a symptomatic urinary-tract infection, BCG should not be administered until they have recovered. There is a small risk (0.4-2%) of developing BCG sepsis, which may present as persistent fever, arthralgia, headaches, rash, and increased malaise. This requires hospitalisation, blood cultures, resuscitation, and commencement of anti-tuberculous treatment.7
Follow up for patients with NMIBC according to current European Association of Urology (EAU) guidelines recommend regular follow-up with flexible cystoscopy accompanied by annual imaging for high-risk tumours.6
Muscle Invasive Bladder Cancer
Approximately 20% of patients are diagnosed with MIBC.1 It is treated with radical cystectomy, radiotherapy, chemotherapy, or immunotherapy, depending on the patient’s health status and tumour stage.
Radical cystectomy and radiotherapy are the standard for localised cancers, often combined with chemotherapy. Evidence shows that neo-adjuvant cisplatin containing chemotherapy has been shown to improve overall five-year survival rates by 8% compared to cystectomy alone.8
There is no difference shown between open and robotic cystectomies with regards to oncologic outcomes or major complications. Robot-assisted cystectomy has been shown to decrease length of stay and blood loss and is now the standard of care in our hospital.
Radiotherapy is delivered as part of a tri-modal treatment (TMT) option. It combines TURBT, chemotherapy and radiotherapy with the aim to achieve local tumour control in the bladder to preserve quality of life. Both cystectomy and radiotherapy have similar oncological outcomes over 5 years.
Patients with metastatic disease can be categorised based on their fitness to undergo cisplatin-based chemotherapy, carboplatin-based chemotherapy, and finally platinum based therapy which has been the standard of care for decades.
Over the past decade the use of immunotherapy can be considered
in patients that are unfit for cisplatin/carboplatin treatment.8 Additionally, the use of immune check-point inhibitors led to longer survival in patients with metastatic bladder cancer.9
Immunotherapy is also being considered in neoadjuvant settings. Agents currently showing significant pathologic response includes Pembrolizumab and atezolizumab. These have yet to be included in routine practice.10
Looking forward
Emerging trials include CheckMate-901 that provided compelling results supporting the use of upfront chemotherapy and immunotherapy (Nivolumab + gemcitabine-cisplatin) in patients with previously untreated unresectable metastatic bladder cancer, or recurrence post-chemotherapy.11
An exciting second trial, called EV-302 (Ertofumab-Vedotin), compared the effectiveness of enfortumab plus pembrolizumab versus platinum-based chemotherapy as initial treatments for advanced bladder cancer. Tumours shrank or stopped growing in 67% of the enfortumabplus-pembrolizumab group versus 44% in the chemotherapy group. Pathological complete response was seen in 30% of the enfortumab-plus-pembrolizumab group, compared to 12% in the chemotherapy group. Most importantly, a median follow-up period of 18 months showed patients in the ertofumab-pluspembrolizumab group lived nearly twice as long as those in the chemotherapy group: a median of 31 months versus 16 months.12 It has recently been approved (September 2024) by the European Medicines Agency for its use for upfront chemotherapy for inoperable or metastatic bladder cancers that have not been treated before.13
The phase 3 NIAGARA is the first to challenge the standard neo-adjuvant approach for MIBC. Here, durvalumab was added to neoadjuvant chemotherapy before radical cystectomy and durvalumab monotherapy after surgery produced significant and clinically meaningful improvements in event-free survival and overall survival compared with NAC alone. Importantly, the addition of neoadjuvant durvalumab did not compromise the ability to complete radical cystectomy.14
Currently, HSE approved immunotherapies include:
1. Avelumab: First line maintenance for patients’ progression free after platinum-based therapy
2. Atezolizumab: Approved for patients post platinum or cisplatin- ineligible
3. Pembrolizumab: for prior platinum-treated or cisplatinineligible patients
4. Ertofumab-Vedotin: Approved for inoperable or metastatic bladder cancer in patients that have not been treated previously Bladder cancer is increasing in incidence with an ageing population, and it varies in severity. With the progress currently made in bladder cancer treatment, there will be increased emphasis on tailored therapies. Ongoing studies looking at circulating tumour DNA is increasingly being investigated to assess which patients need perioperative systemic therapy after radical surgery.15 Bladder sparring treatment approaches are also being considered and evaluated to improve the quality of life of patients with MIBC.
References
1. Irish Cancer Society Bladder cancer. [Online]. Irish Cancer Society. Available at: www.cancer. ie/cancer-information-and-support/ cancer-types/bladder-cancer [Accessed July 11, 2024]
2. Brennan P, Bogillot O, Cordier S et al. Cigarette smoking and bladder cancer in men: a pooled analysis of 11 case-control studies. International Journal of Cancer 2000; 86(2):28994
3. Rezaei F, Tabatabaee H, Rahmanian V, Mirahmadizadeh A, Hassanipour S. The correlation between bladder cancer and obesity, overweight, physical inactivity, and tobacco use: an ecological study in Asian Countries. Annal Global Health 2019; 85(1):102
4. National Cancer Registry Ireland. (2021). Incidence Statistics. NCRI. Available at: www.ncri.ie/data/ incidence-statistic [Accessed 12 July 2024
5. Lin W, Pan X, Zhang C, Ye B, Song J. Impact of age at diagnosis of bladder cancer on survival: a surveillance, epidemiology, and end results-based study 2004. Cancer Control 2023. 30:1-14. doi: 10.1177/10732748231152322
6. EAU Guidelines. Edn. presented at the EAU Annual Congress Paris 2024. ISBN 978-94-92671-23-3.
7. Improving Patient Outcomes: Optimal BCG Treatment Regimen to Prevent Progression in Superficial Bladder Cancer, Lamm, Donald, European Urology Supplements, Volume 5, Issue 10, 654 – 659
8. Gunlusoy B, Arslan M, Vardar E et al. The efficacy and toxicity
of gemcitabine and cisplatin chemotherapy in advanced/ metastatic bladder urothelial carcinoma. Actas Urológicas Españolas 2012; 36(9):515-20
9. Maiorano B, Giorgi U, Ciardiello D et al. Immunecheckpoint inhibitors in advanced bladder cancer: seize the day. Biomedicines 2019; 10(2):411
10. Powles T, Csőszi T, Özgüroğlu M, Matsubara N, Géczi L, Cheng SY, Fradet Y, Oudard S, Vulsteke C, Morales Barrera R, Fléchon A, Gunduz S, Loriot Y, RodriguezVida A, Mamtani R, Yu EY, Nam K, Imai K, Homet Moreno B, Alva A; KEYNOTE-361 Investigators. Pembrolizumab alone or combined with chemotherapy versus chemotherapy as first-line therapy for advanced urothelial carcinoma (KEYNOTE-361): a randomised, open-label, phase 3 trial. Lancet Oncol. 2021 Jul;22(7):931-945. doi: 10.1016/S1470-2045(21)001522. Epub 2021 May 26. PMID: 34051178.
11. LBA7 Nivolumab plus gemcitabine-cisplatin versus gemcitabine-cisplatin alone for previously untreated unresectable or metastatic urothelial carcinoma: Results from the phase III CheckMate 901 trial van der Heijden, M.S. et al. Annals of Oncology, Volume 34, S1341
12. Powles, T. et al. (2024) ‘Enfortumab vedotin and pembrolizumab in untreated advanced urothelial cancer’, New England Journal of Medicine, 390(10), pp. 875–888. doi:10.1056/ nejmoa2312117.
13. Authorisation details: EMA product number EMEA/ H/C/005392 Additional monitoring Padcev, European Medicines Agency (EMA). Available at: https://www.ema.europa.eu/en/ medicines/human/EPAR/padcev s
14. Powles TB, et al. A randomized phase 3 trial of neoadjuvant durvalumab plus chemotherapy followed by radical cystectomy and adjuvant durvalumab in muscle-invasive bladder cancer (NIAGARA). ESMO Congress 2024, LBA5
15. Ascione C, Napolitano F, Esposito D et al. Role of FGFR3 in bladder cancer: Treatment landscape and future challenges. Cancer Treat Rev 2023; 115(1):1
16. Staging and grading of bladder cancer, Macmillan Cancer Support. Available at: https:// www.macmillan.org.uk/cancerinformation-and-support/bladdercancer/staging-and-grading-ofbladder-cancer.
The National Clinical Programme in Surgery (NCPS) has launched a new HSE eLearning Module which provides education on the management of neck haematomas when they occur on the ward after thyroid surgery.
Neck haematomas are rare but life-threatening and can occur in approximately 1-3% of patients who have thyroid surgery. The haematomas can enlarge rapidly and compromise the patient’s airway requiring urgent intervention. There are a number of factors that can increase the risk including age, hypertension, Grave’s disease or history of previous thyroid surgery.
The eLearning module aims to provide education on recognition, escalation, and the management of this condition and is recommended for all doctors and nurses involved in the post-operative care of all patients who have undergone thyroid surgery.
The module was funded by the HSE and its development was led by the National Clinical Advisor for Otorhinolaryngology-Head and Neck Surgery, Professor Michael Walsh. It was developed by Aurion Learning and was supported by an expert working group that included a range of ENT consultants, nurses and anaesthesiologists.
Professor Walsh, National Clinical Advisor for Otorhinolaryngology
– Head and Neck Surgery, NCPS, said, “The development of this eModule was a collaborative effort
Pictured (l-r): Ciara Hughes, Programme Manager, NCPS; Prof. Michael Walsh, National Clinical Advisor for OtorhinolaryngologyHead and Neck Surgery; Prof. Deborah Mc Namara, President RCSI; and Sharon Casey, Administration Officer, NCPS
involving multiple stakeholders and the NCPS is grateful to all those who contributed to this work, particularly to Mr Greg Sadler Consultant Endocrine Surgeon, John Radcliffe Hospital, Oxford, UK and Mr Shad Khan Consultant Endocrine and General Surgeon, Oxford University Hospitals for the use of their video.
“This module is easily accessible through HSELanD for all relevant healthcare staff and should give staff the knowledge and confidence to act quickly and safely in this emergency scenario which will ultimately save lives.”
The National Clinical Programme in Surgery is a strategic initiative between the HSE and RCSI. The programme aims to design and implement change initiatives to improve and standardise the quality of care and access for all patients in a cost-effective manner.

The programme works closely with other National Clinical and Care Programmes under the governance of the HSE CCO to ensure integration across multiple areas of care.
Dr Siobhán Ni Bhriain, HSE National Clinical Director Integrated Care, welcomed the launch, noting “the integrated approach taken in the development of the
protocol, ensuring that it meets the highest standards internationally. It demonstrates the importance of continued collaboration between the HSE and RCSI in the development of these guidelines to support the delivery of high quality, standardised clinical care. Congratulations to Prof. Walsh and team and the NCPS for bringing this to completion.”
The Health Research Board recently hosted an event – A Decade of Impact: Celebrating our Journey which acknowledged ten years of HRB investment in trials methodology research and capacity building in Ireland, through the HRB-TMRN.
One of the speakers at this event was Dr Conor Judge, Consultant Nephrologist and Senior Lecturer in Applied Clinical Data Analytics who spoke on - Testing Artificial Intelligence Interventions in Pragmatic Trials. Here, he gives Hospital Professional News an overview.
“Artificial intelligence (AI) holds immense promise in transforming healthcare, particularly in areas like clinical decision support systems (CDSS). My talk at the HRB Trials Methodology Research Network event - ‘A Decade of Impact: Celebrating Our Journey’ explores the intersection of AI and pragmatic randomised controlled trials (RCTs), focusing on ensuring these technologies are safe, effective, and scalable.
“The core of my research is developing an AI-CDSS for
hypertension management. This project is funbded by the Health Research Board. The system integrates data from electronic health records (EHRs) and employs advanced machine learning techniques to provide clinicians with evidencebased recommendations for antihypertensive therapy. However, introducing AI into clinical care raises questions about trust, acceptability, and effectiveness, which can only be answered through robust testing frameworks like RCTs.
“Pragmatic trials are critical for evaluating AI interventions because they reflect real-world conditions. Unlike traditional RCTs, which often operate under idealised scenarios, pragmatic trials account for the variability and complexity of everyday clinical settings. For example, a
Dr Conor Judge, Consultant Nephrologist and Senior Lecturer in Applied Clinical Data Analytics
recent multicenter trial tested an AI-driven decision-making tool in stress echocardiograms. While the AI showed potential, it also highlighted the challenges of achieving non-inferiority compared to standard care, emphasising the need for careful evaluation.
“Building infrastructure for pragmatic trials is another focus of my work. In Ireland, we face gaps like the lack of fully integrated EHR systems, though we do have strong data linkage capabilities, such as connecting clinical data to mortality and audit registries. Collaborations with entities like the Health Service Executive (HSE) are vital for leveraging existing datasets to support such trials.

“In conclusion, testing AI interventions requires blending technical innovation with rigorous research methodology. Through pragmatic trials, we can address unmet clinical needs while ensuring AI's safe integration into healthcare systems.”
Written by Fionnuala Ní Áinle, Siobhán Ní Bhriain, Michael O’Connor
(1) HSE National Clinical Lead, National Clinical Programme in VTE (2) HSE National Clinical Director Integrated Care, Office Chief Clinical Officer (3) HSE National Clinical Advisor and Group Lead, Access and Integration
Introduction
Venous thromboembolism (VTE, comprising deep vein thrombosis and pulmonary embolism) represents a major healthcare risk throughout the world. In the community, it is a common disease affecting 1 in 12 people overall during their lives and symptoms and signs can be overlooked. VTE is a leading cause of hospital-associated morbidity and mortality and is the leading cause of maternal death overall in the UK and Ireland. Of all survivors, at least half will have long-term sequelae including post-thrombotic syndrome of the lower limbs, post-pulmonary embolism dyspnoea, chronic thromboembolic pulmonary hypertension, anxiety and posttraumatic stress disorder.
Despite the incidence, mortality and long-term health impact of VTE, awareness of this condition in the general population is very poor. A US survey reported that 75% of respondents had never heard of DVT and fewer than 1 in 10 had an awareness of DVT symptoms. This is a major clinical and societal risk that must be urgently addressed, given the missed opportunities to prevent death and long-term disability.
VTE is a major burden for health care systems worldwide. For example, VTE costs EU health systems ¤1.5-2.2 billion per year in direct costs. However, the effects of chronic disability and loss of productive life years due to thromboembolic diseases can cause indirect costs, that in Europe have been estimated to be as high as 13.2 billion per year (source: International Society on Thrombosis and Haemostasis).
Despite the fact that VTE is so prevalent and avoidable, it often lacks the prioritisation it deserves in many countries. In a transformative step towards raising VTE awareness and driving systemic improvements in VTE prevention and care, the HSE launched the National Clinical Programme in VTE (NCP-VTE) in October 2022. The programme
aims to establish national standards for the prevention, diagnosis and management of VTE and for a post-VTE recovery programme, integrated in the community. Under the mandate of Dr Colm Henry, Chief Clinical Officer, HSE, and with the strong advocacy of Melanie SheehanCleary, who tragically lost her beloved daughter, Eve Cleary, due to a pulmonary embolism (PE), the NCP-VTE has been commissioned to form a taskforce to write “Eve’s Protocol”— a first national guideline intended to guide multidisciplinary care providers in the community and in hospitals in managing VTE across the patient journey and to raise VTE awareness among the public. Establishing the National VTE Programme: A Milestone in VTE Care
The role of the NCP-VTE is to develop guidance and advice for the healthcare system on the prevention, diagnosis, management, and long-term care of VTE through an integrated care approach across the community (including primary and prehospital care) and hospitals. Since its inception, the NCP has built a robust governance structure at both national and regional levels, aiming to establish a streamlined, standardised approach to the implementation of the recommendations for patient safety in VTE. This governance structure includes a VTE Clinical Advisory Group with extensive multidisciplinary clinical representation from all healthcare regions. Moreover, the NCP-VTE has been extremely fortunate to include amongst its working team a range of multidisciplinary leadership from around the country, including nursing, advance nurse practitioner, psychology, physiotherapy and clinical colleague which has led to the formation of very active VTE working groups focussing on the highest priority issues facing VTE.
The NCP-VTE team, working with active and committed colleagues throughout the country, has
made significant advances to date since its inception in 2022. A key performance indicator (KPI) estimating suspected hospitalacquired thrombosis rates is collected and reviewed, with feedback provided to support local assessment and to drive quality improvement. The programme’s strategic alignment with the 2024 National Service Plan and the anticipated resource approvals from the Department of Health demonstrate the prioritisation of VTE care in Ireland. Resources have been dedicated particularly in the context of unscheduled emergency care and women’s healthcare—two areas where VTE risk and management needs are especially high.
Eve’s Protocol: A National Guideline for a Safer Future At the centre of the NCP-VTE mission is the development of a National Guideline for the prevention, diagnosis and management of VTE and supporting evidence-based strategies supporting VTE recovery. The Guideline taskforce is honoured to name this “Eve’s Protocol”, in memory of Eve Cleary. Eve’s Protocol is intended as a living legacy to the lessons learned from Eve’s passing and her mother Melanie SheehanCleary’s determined advocacy. This protocol will commemorate all those who died from VTE. In July 2024, the National Clinical Director for Integrated Care commissioned the development of the protocol, highlighting the protocol’s importance to improving patient safety and quality of care in Ireland. The commissioning is a clear mandate from the highest levels of the HSE, with support from HSE CEO.
The Protocol will be built on evidence and an understanding of the patient experience and patient and family representatives will serve on the guideline task force along. This patient i involvement is deeply appreciated and serves as an important reminder of the need for a patient-centered approach to healthcare policy and guideline
creation. This will ensure that the voices of those directly affected by VTE remain central to the NCP-VTE mission. This mission is enshrined in one of the most important of the draft protocol’s recommendations highlighting the need to improve awareness of VTE signs and symptoms in the community and for patients who have been hospitalised. A truly collaborative approach to improving VTE awareness was exemplified by the development of a “Blood Clot Alert Card” by the HSE Quality and Patient Safety directorate in 2018 working closely with the patient organisation Thrombosis Ireland (Figure 1). The Protocol will include the messaging included in this valuable resource and will also promote alternative strategies aimed at a wider audience, including videos suitable for dissemination on social media and a wide range of patient information leaflets. The blood alert card will undergo further review to update it.
Collaboration: A Foundation of the National VTE Programme
The day to day work of the NCP-VTE is characterised by a strong multidisciplinary approach, including perspectives from many varied professionals, which will ensure that recommendations are reflective of different healthcare settings. The development othe Protocol will further benefit from this strong professional collaboration. This collaborative approach is particularly important, given the broad spectrum of VTE’s impact across patient demographics and clinical scenarios.
A key pillar of the NCP-VTE’s emerging strategy has been its focus on data and performance metrics. The programme already advises in the HSE’s measurement of the rate of suspected hospitalassociated VTE and assists in data interpretation. Through this work, opportunities are identified for
42 Billion Live Bacteria, 8 ‘friendly’ bacterial strains + Vitamin C


*Vitamin C contributes to the normal function of the immune system Nutritionist and Author

This is the formation of a clot inside a blood vessel, usually in the leg, which may break off and go to the lungs. This can be fatal.
Blood clots can be very serious - but there are effective treatments to deal with them and help prevent them 60%of clots happen in HOSPITAL or in the 90 DAYS following admission.
Ask for your risk of blood clots to be assessed, especially if you are in one of the higher risk groups listed opposite
➢Walk and move as much as possible
➢Drink plenty of fluids
You have a HIGHER CHANCE of getting a clot in HOSPITAL than on an AEROPLANE!
➢If directed to use stockings or medication to prevent or treat a clot follow instructions exactly
Remember, a blood clot in the veins is more likely up to 90 days after being in hospital
If you have any signs or symptoms of a clot, take immediate action to seek medical help
quality improvement and valuable feedback is provided to key stakeholders with responsibility for quality improvement. This data-driven approach fosters accountability and transparency, with the goal of reducing the incidence of preventable VTE and enhancing the quality of VTE care nationwide. The NCP-VTE’s vision is that similarly, the implementation of the Protocol recommendations will also be driven by formal Quality Improvement strategies.
The NCP-VTE started its work in 2023 and is now progressing
with the Protocol which is scheduled to complete in early 2025. The programme plans to make recommendations on the standardisation of VTE prevention, diagnosis, and management through implementation of this protocol. As the NCP-VTE continues to evolve, its objectives will continue to reflect the core values of safety, quality, and patient-centered care.
As the NCP-VTE’s work moves forward, the collaborative efforts of multidisciplinary colleagues and patient advocates will always stay vital to its success. Each healthcare provider has a
Swelling or pain in one leg or arm
Warmth or redness in the leg or arm
➢Short of breath or rapid breathing
Chest pain
(particularly when breathing deeply)
Coughing or coughing up blood
If you have one or more of these, you may have a clot and need urgent treatment

YOU MAY BE AT HIGHER RISK IF YOU: are admitted to hospital and for 90 days after you go home
have active cancer or receiving cancer treatment are pregnant or have had a baby less than 6 weeks ago become immobile (more than 3 days in bed / travel non-stop more than 6 hours / in a leg cast)
RISK MAY INCREASE FURTHER IF:
you or a close relative had a blood clot you had surgery in the last 90 days you have thrombophilia (tendency to clot) you are on the oral contraceptive pill or HRT you have heart, lung or inflammatory disease you are over 60 years of age or are overweight you have varicose veins that become red and sore
role to play in preventing VTE and safeguarding patients from its potentially life-threatening complications. With the Protocol as a guide, the NCP-VTE will advise and guide in the creation of a safer healthcare environment for all Irish patients and to establish Ireland as a leader in comprehensive VTE care.
Conclusion
The establishment of the National VTE Programme and the drafting of Eve’s Protocol mark a major step forward in Ireland’s approach to VTE. Through leadership, multidisciplinary collaboration, and a strong commitment to
Thrombosis Ireland/NCPVTE Blood
patient-centred care, the NCPVTE is committed to advise on the implementation of strategies that will potentially improve VTE outcomes and reduce preventable VTE incidents. We wish to see a safer future. For all involved in the implementation journey— clinicians, patient advocates, and healthcare professionals across Ireland—this initiative will honour the memory of Eve and others who died from VTE by driving lasting changes in VTE care.











A community pharmacy environment that fosters teamwork ensures high levels of consumer satisfaction. This series of articles is designed for you to use as a guide to assist your team in focusing on meeting ongoing CPD targets and to identify any training needs in order to keep the knowledge and skills of you and your team up to date.

The below information, considerations and checklist provide support to enable you to run a team training session and identify opportunities for learning within the topic of Wound Care. It is estimated that 1.5% of the population are affected by a wound at any one point in time.
Patients will regularly present to the pharmacy to treat minor wounds rather than attending a GP clinic. Most wounds can be treated cheaply and effectively with over the counter products. Wound care management in the pharmacy could potentially be a service that pharmacists offer in the consultation room.
This is a practice that other jurisdictions offer such as Australia and US and has been successful.
Consider:
In these situations it is utilising the skills of community pharmacists to provide enhanced first aid and wound management services and freeing up other highly skilled health professionals to perform health interventions requiring specialised skills. The Pharmacy Guild of Australia report that the introduction of this service gives greater recognition of the role of community pharmacists as a member of the primary health care team and allows community pharmacy the opportunity to develop a viable business involving service provision as an adjunct to product supply.
Wound care is valued at ¤4.3 million in Ireland. Many pharmacy counter assistants and pharmacists are trained in first aid and are familiar with the appropriate equipment
The different types of wounds, causes and symptoms
Know how to advise customers on preventative measures to avoid wounds
Be able to pair the right treatment to the relevant issue
When to refer customers to the pharmacist.
and advise for certain ailments and this can encourage loyal and repeat customers.
Skin Wounds are a common health concern, which can vary from abrasions, lacerations, rupture injuries, punctures, to penetrating wounds. Treating pressure sores in bedridden or elderly patients is a large part of wound care.
Addressing wounds appropriately and timely with dressings and wound closures will minimise cosmetic scaring and infection while making the patient more comfortable.
When offering patients advice on wound care it is essential to ask the patient about their lifestyle and general health so the dressing and healing process is appropriate for the patient. The pharmacist should ask the patient about the duration of the wound, if a patient who has had a wound for longer than one week should be referred to their doctor.
Acute wounds will generally proceed through an orderly and timely process to produce a healed wound which has anatomic and functional integrity.
Chronic wounds are defined as those whose healing is impaired. The inflammatory phase is dysfunctional due to intrinsic and extrinsic factors that impact on the person, the wound or the healing environment (Swanson et al., 2015). Chronic wounds include a variety of aetiologies, yet they share a number of characteristics such as a persistent state of inflammation due to high levels of
Key Points:
Check your pharmacy team are aware and understand the following key points:
The pharmacy team knows how to manage wounds and select appropriate OTC treatments and dressings with confidence
The team knows how to assess a wound and the stages of wound healing
My pharmacy assistants can meet the points in this training checklist.
pro-inflammatory cytokines and unregulated proteolytic activity as a result of the high levels of active proteases and defective neutrophils, with an imbalanced expression of tissue inhibitors i.e. metalloproteinases.
In the treatment of wounds endeavour to facilitate the following objectives of wound healing:
1. The wound should be allowed to heal in a moist wound environment unless the clinical goal is to maintain a dry wound bed, e.g. ischaemic foot
2. To address the issues observed in the assessment process
3. To promote wound healing
Wound Pain
Wound pain can be categorised as:
• Background pain - continuous or intermittent pain that is felt even at rest
• Incident pain - pain that occurs during day-to-day activities such as mobilisation or coughing
• Procedural pain - pain that results from routine procedures such as dressing changes or wound cleansing
• Operative pain - pain associated with significant wound intervention, e.g. debridement or wound biopsy.
Actions:
Ensure efficient sign posting to discreet consultation areas within the pharmacy for further help and advice
Spend time raising awareness within the team about how the body heals following an injury, exploring some of the internal and external factors preventing or delaying this
Check your pharmacy team are able to recognise a infected wound
Ensure staff are educated about wound care terminology
Train the team to meet all the above considerations

Discover an innovative wound management range that covers all phases of wound healing.
Developed especially for the treatment of acute and chronic wounds, our range offers effective, patient friendly solutions for conditions such as: Acute Wounds, Chronic Wounds, Ulcers and Burns.








As our business marks 20 years advising members of the pharmacy community we would like to thank all our clients in the sector for their trust in us as advisers. Below we look back on our processes and methodologies that have evolved with time and technology to allow us to continue serving your sector for so long.

The New Year always brings a focus on financial planning with people wondering if they are getting the best advice and how to determine this. Financial Advice is a broad term with many definitions and interpretations. Depending on the provider you choose what you receive can vary greatly along with the outcomes. The need for a definitive process to guarantee the best solutions for consumers was the primary driver behind the introduction of the CERTIFIED FINANCIAL PLANNER™ designation in Ireland back in 2010.
There are less independent advisers operating over the last 10 years as the market place shrinks and consolidates. The pillar banks, Allied Irish and Bank of Ireland have bought up Goodbody and Davy Stock Brokers respectively. The Canadian owned Irish Life (whose pensions and investments you are sold if you take your advice from the AIB bank channel) are buying multiple medium to large size brokerages to develop their assets under management.
The CERTIFIED FINANCIAL PLANNER™ designation is the benchmark in the market place for those who want all options evaluated for them. With an estimated 400 acting independently in Ireland they are highly qualified professionals
Written by Colm Moore, a Certified Financial Planner™ with Moore Wealth Management and has been advising the
pharmacy community for 20 years. www.mwm.ie
who ensure you are getting the best advice available from all the options in a process that’s accountable and transparent. They like you are small business owners who understand your hopes, fears and financial issues as they deal with these themselves on a daily basis.
A CERTIFIED FINANCIAL PLANNER™ operates with a clearly defined process of developing strategies to help people manage their financial affairs to meet life goals defined as
1. Establishing and define the client-planner relationship.
2. Gathering client data, including goals.
3. Analysis and evaluation of your financial status.
4. Developing and presenting financial planning recommendations and/or alternatives.
5. Implementing the financial planning recommendations.
6. Monitoring the financial planning recommendations.
So when you decide you want independent advice what does this mean to you. When carrying out a review for any new client we look at the following and this is what you should be looking for.
What do you need for family protection. There are methods to quantify this and one of the most useful is to map out the impact of the death of either spouse on future cashflows for a defined period which can be to the point for example where any children are through college and independent. Once this figure is established we look at any existing policies to see if they fulfil this need. In some cases we find that clients were over insured and they ended up saving money by
cancelling policies. In other cases we find clients are overpaying for cover because due to intense competition in the market place insurance companies are reducing premiums to drive business and increase market share. For over 5 years now one of the main insurers in Ireland has had a discount of 15% on life cover premiums. However this discount has to be passed on by the broker which does not happen as much as it should as the commission paid is a function of the premium and discounts impact broker remuneration.
Takeaway #1 If you have taken out life cover in the last 5 years make sure you have the best premium and the right amount of cover for your dependants.
Business protection has a similar methodology in that you
quantify the impact of the death of the business owner and insure accordingly. But what you need to understand is that if the company pays the premium in the event of claim the policy proceeds are paid into the business and not directly to your family. This type of cover is suitable for shareholder protection where a business has more than one owner and the proceeds are used to buy back shares under agreement. It is not suitable where the intention is for the proceeds to go directly to your family as they will have to extract the funds from the company and pay the same taxes as drawing salary. This is a very common problem.
Takeaway # 2 If you were told to have your business pay for life cover to be more tax efficient are you aware the policy proceeds are going to the company and subject to normal taxation on extraction.

There should be a clear distinction between life cover(death protection) and living benefits which are income protection are serious illness. Without doubt income protection is the most important cover you can have. Your ability to earn is your most valuable unrecognised asset and a culmination of years of hard work, study and determination on your part. These policies are designed to pay out if you cannot work in your own occupation and protect you and those who depend on your income. Serious illness is different to income protection in that it pays out a lump sum on diagnosis, typically heart attack, cancer and stroke and works well in tandem with income protection if you are going to be out for a long time or unable to work again.
Takeaway #3 Medical advances mean you are more likely to survive major illness than die, but what will you do if you are unable to return to work and earn a living.
This section has to have a performance and cost review accompanied by full cashflow modelling that shows the future projections of your pension and the drawdown scenarios when you
reach retirement age. Your pension is a supremely tax efficient tool for wealth accumulation and cash extraction. It is one asset in your retirement armoury and needs to be overlayed with your other assets and income streams to give you a model of retirement and how best to draw cash. It can make perfect sense to use resources such as the proceeds of business sale first and allow your pension assets to continue to grow in a tax free bubble until needed.
Takeaway #4 Make sure your pension is projected out to end of life using full cashflow modelling mapping various drawdown scenarios.
This should look at how you are investing personally held funds and determining if you are optimising the most tax efficient environment for them. Most personally held investments in Ireland suffer from a 1% entry levy, 1-2% annual management charge and 41% tax on gains. This is a tough regime under which to make returns and guidance is crucial. Hope however is on the horizon with a recent government review clearly stating the tax rate needs to reduce to 33%.
This is also the section of the report that ties into inheritance
tax planning and it should be quantified if you should be accumulating assets in your children’s names to take advantage of the small gift allowance which allows any one person to gift another ¤3,000 per annum with no tax implications. This has no impact on the current lifetime parent/child threshold of ¤400,000.
Takeaway #5 Determine what the best way to accumulate personally held assets is and identify the appropriate tax efficient structure for holding them
An often overlooked but key section. This is not on most people’s radar but when you realise ¤400,000 per child is the inheritance tax threshold the problem comes into focus. Only on calculating the combined value of your home, business, pension and investment assets do you realise that everything above ¤800,000 (two children), ¤1,200,000 (three children) etc is going to be taxed at 33% that the problem become stark and quantifiable.
There are innovative solutions in the market place for this including one of the more interesting policies ever launched in Ireland. This policy insures both spouses for the inheritance tax liability. On death
the cover amount is received by the estate tax free for settlement of the bill thus leaving all the assets in the children’s names. The key part of this policy is that after 16 years if you have not used the policy you are guaranteed that 70% of the premiums paid are returned to you. This is not conditional on anything other than you deciding you want to end the policy. This life cover can protect your family from a substantial tax bill in the medium term while giving you time to plan for alternative ways to reduce the liability. The time provided will give you an idea for example if business assets will be passed to children as they grow older which is very tax efficient.
Takeaway #6 A comprehensive review will identify if this is an issue for you. Seek out a CERTIFIED FINANCIAL PLANNER™ for guidance.
The advice market is changing and this is natural. The larger players and banks are further developing one size fits all strategies and campaigns. You do not have to fit into one of these templates. The independent financial adviser section of the market place is maintaining its independence and delivering world class solutions for clients.

Written by Lorna Nellany, Respiratory Advanced Nurse Practitioner, Nathan Scanlon, COPD Outreach Clinical Nurse Specialist, Sligo University Hospital


Introduction
Chronic obstructive pulmonary disease (COPD) is a progressive lung disease characterised by persistent airflow obstruction that is partially reversible. Clinically, it manifests as worsening dyspnoea and, in advanced stages, frequent exacerbations requiring medical intervention in primary care, emergency settings, or hospital admissions. COPD represents a major public health challenge in Ireland, significantly contributing to respiratory mortality. Despite global improvements, Ireland continues to report one of the highest agestandardised mortality rates for COPD among European nations.
The prevalence of COPD in Ireland is estimated at approximately 380,000 people. Despite this, only about 110,000 individuals have been formally diagnosed, leaving a significant proportion undiagnosed and untreated. It is a leading cause of morbidity and mortality in Ireland, with at least 1,500 deaths annually and over 15,000 hospital admissions attributed to the condition.
Sligo University Hospital (SUH) is part of the HSE West North West region, and is a public model three acute hospital with a catchment area extending to Sligo, Leitrim, South Donegal, West Cavan, as well as parts of Mayo and Roscommon. The hospital has 288 inpatient beds and provides 24/7 Emergency Medicine care, as well as various Medical and Surgical specialities
Over the years, the hospital's respiratory service has expanded from a team of one Respiratory Physician, one Clinical Nurse Specialist (CNS), and one Respiratory Physiologist to a multidisciplinary team including three Respiratory Physicians, two acute CNS, one CNS in Non-Invasive Ventilation, one CNS in COPD Outreach and one Advanced Nurse Practitioner (ANP) 50:50 Hospital and Integrated Care. There is also a Respiratory Integrated Care Team in the community compromising of two Respiratory CNS, and two Clinical Specialist Physiotherapists, two Respiratory Physiologists and a Pulmonary Rehabilitation Team.
The inpatient Respiratory CNS service plays an integral role in the care of patients with COPD. Key areas of focus include inpatient reviews, education, management of non-invasive ventilation (NIV), long term oxygen ordering and referrals to Respiratory Physiology and Pulmonary Rehabilitation. One of the most recent significant developments was the CNS team’s integration into the Emergency Department (ED) which allows the team to identify COPD patients earlier in their hospital stay, enabling timely interventions and better care coordination ensuring these patients are transferred to the Respiratory ward.
Inpatient Reviews
The respiratory CNS inpatient review service is a key component
of COPD care, offering guidance to multidisciplinary teams across various specialties. Patients are typically referred with an established diagnosis of COPD, but those who present with symptoms suggestive of COPD are also assessed then referred to the Respiratory ANP for further investigations if appropriate. The team also optimise inhaler regimens, ensuring appropriate medications, while providing education on correct inhaler techniques to improve adherence. More complex patients are discussed with the respiratory physician on consults. Through this comprehensive approach, the CNS service ensures COPD patients receive tailored, evidence-based care during their hospital stay.
Education is a cornerstone of effective COPD management, empowering patients to take control of their condition and improve their quality of life. A primary focus is on inhaler technique and the importance of medication adherence, as incorrect use of inhalers or inconsistent medication routines can significantly worsen symptoms and lead to exacerbations. Patients are also educated on the importance of winter vaccines in reducing exacerbations and maintaining lung function. Smoking cessation remains a critical area of education, as it is the most effective intervention for slowing disease progression. Additionally, patients benefit from learning to recognise the early signs of exacerbations, enabling them to seek timely intervention and avoid hospital admissions.
The CNS team plays a pivotal role in initiating and managing non-invasive ventilation (NIV) for patients with acute respiratory failure due to COPD exacerbations. We assess the patient’s clinical condition and recommend appropriate ventilator settings, including pressures and FiO2 levels, to optimise oxygenation and carbon dioxide clearance. Once NIV is initiated ongoing support and troubleshooting is provided to address challenges such as mask fit, pressure leaks, or discomfort. These patients are transferred to
the Respiratory Unit for ongoing specialist care. Ensuring effective use of NIV and addressing complications promptly significantly improves patient outcomes.
Managing patients on home oxygen therapy is another vital aspect of our role, ensuring optimal care and safety for those requiring long-term oxygen support. We work closely with our respiratory consultants to titrate oxygen, tailoring therapy to meet the patient’s needs while preventing complications such as hypercapnia. Our responsibilities also include providing phone support to patients, addressing concerns, monitoring their progress, and coordinating care. Additionally, we are often called to assess patients to determine their eligibility for home oxygen. For those who are not candidates, we evaluate alternative management plans to ensure that oxygen therapy is used effectively to enhance patient outcomes and quality of life.
The COPD Outreach service at Sligo University Hospital was established in May 2023 to address the growing burden of COPD on acute healthcare services. COPD exacerbations are a leading cause of hospital admissions and bed occupancy, yet they are recognised as an ambulatory care sensitive condition—meaning timely and appropriate community-based care can significantly reduce hospitalisations. With funding from the Enhanced Community Care (ECC) programme, the service was launched as a proactive, multidisciplinary initiative designed to improve patient outcomes, reduce healthcare utilisation and empower individuals to manage their condition more effectively.
The team consists of one Respiratory CNS and one Clinical Specialist Physiotherapist with governance from the Respiratory Consultant. National COPD Outreach guidelines provided the foundation for the programme, which were customised to meet the unique needs the catchment area.

Initially, the service began with a targeted approach, where respiratory consultants handpicked patients for inclusion. By June 2023, it expanded to include all medical patients admitted with an acute exacerbation of COPD who met the inclusion criteria. This criteria focused on patients with confirmed COPD who could engage with a self-management program and lived within a 35km radius of the hospital. Exclusion criteria included conditions such as pneumonia, pneumothorax, or social circumstances that made home care unfeasible.
Service Implementation
The COPD Outreach service has two major streams.
1. Supported Discharge
Patients admitted with an acute exacerbation are followed up postdischarge for two weeks during this time they remain under the care of the respiratory consultant. They typically receive two to three home visits, depending on their clinical needs. Each visit includes:
• Monitoring of vital signs and symptoms.
• Clinical assessments, including chest auscultation and symptom questionnaires.
• Education on inhaler technique, early exacerbation recognition, and medication adherence.
• Discussions about goals of care and referrals to pulmonary rehabilitation or other services.
Patients who avoid admission during the two-week follow-up are reviewed again at six weeks, with a discharge summary sent to their GP.
2. Admission Avoidance
Admission avoidance targets patients who had previously
engaged with COPD Outreach. Patients experiencing exacerbations at home contact the team directly for an initial phone triage. If deemed appropriate, a same or next day home visit is arranged. Nurse prescribed treatments are initiated during these visits, including oral steroids and antibiotics, often avoiding the need for hospital admission. These interventions are supported with advice from the respiratory consultant.
Both arms of the service emphasise patient empowerment, equipping individuals with the knowledge and tools to manage their disease proactively. In July 2024 we undertook an audit to measure the impact of the COPD outreach service on hospital admissions and bed days. There were 68 accepted patients enrolled in the programme between May 2023 and March 2024, see table 1 for patient
demographics of the audit carried out on 38 patient’s meeting predefined inclusion criteria.
Results and Evaluation
In this time the COPD Outreach service demonstrated significant benefits with hospital admissions: Reduced by 78%, from 1.68 to 0.37 admissions per patient and hospital bed days reduced by 70%, from 10.39 to 3.08 bed days per patient. See Figure 1.
The COPD Outreach service has demonstrated the significant impact of a tailored, communitybased approach to managing COPD. By reducing hospital admissions and bed days through proactive supported discharge and admission avoidance pathways, the programme has not only improved patient outcomes but also alleviated the strain on hospital resources.
The introduction of a Respiratory ANP post in 2022 led to a focus on capturing people who attended the hospital with chronic respiratory conditions but were not known to respiratory services. Currently one of the aims of the service is to reduce COPD exacerbations and ED/ hospital visits, through early diagnosis, assessment and management. This development is aligned with the Integrated Model of Care for the Prevention and Management of Chronic Disease, ensuring timely diagnosis, streamlined patient pathways, and comprehensive follow-up care.
Service implementation
The ANP-led service identifies and manages patients via two pathways
1. Emergency Department (ED) Pathway:
Patients presenting to the ED with respiratory conditions, including suspected or exacerbated COPD, are reviewed by the Respiratory CNS or ANP, a full assessment is carried out and treatment is optimised based on symptoms. If appropriate these patients are followed-up in the Respiratory ANP clinic in the Benbulbin Chronic Disease Management (CDM) Hub for further assessment, investigations and management. For patients who present Out of Hours a referral form is sent to the Respiratory ANP who contacts the patient to triage review and arrange follow-up in the Benbulbin CDM Hub. Patients are typically reviewed within 1–4 weeks of their initial presentation, gaining
access to diagnostics, tailored management plans, and selfmanagement education.
2. Inpatient Pathway:
Patients admitted to nonrespiratory specialties with COPD symptoms are flagged for review. The respiratory CNS optimises treatment during their admission, with follow-up review by the ANP
Past medical history
Health history
Risk factors
Smoking
Occupation
Medications
Family history
Referral for:
Pulmonary function test
FENO
Pulmonary rehab RIC
Smoking cessation
Respiratory Consultant
in the Benbulbin CDM hub this ensures ongoing follow-up care in the community. See figure 2 for ANP referral pathway.
The ANP-led clinic operates twice weekly at the Benbulbin Chronic Disease Management (CDM) Hub, offering an efficient, “one-stop shop” approach to patient care. The patient undergoes pulmonary function testing followed by a
consultation with the ANP. A comprehensive health assessment is conducted. The clinic offers ABG analysis, referral for chest x-ray, bloods and sputum monitoring. Education on COPD management is provided, ensuring patients are informed about their condition. Treatment plans are reviewed and optimised in line with national and international
Comprehensive health assessment including physical assessment
Review of available results
Reconciliation of medication
Revised treatment plan
Health promotion
Self management plan
Discharge to GP, RIC vs follow-up review with ANP. Refer to consultant clinic if complex needs – CT thorax, bronchoscopy
COPD guidelines to ensure best practices. See Figure 3 for an overview of the clinical pathway. Most patients are discharged back to their GP with follow-up in the Chronic Disease Management Programme in primary care. This integrated approach supports early intervention, enhances patient education, and facilitates continuity of care.
Assessment of symptoms: SOB, cough, wheeze
Sputum
Respiratory triggers
mMRC
CAT assessment ACT
Vital signs
Sputum C&S
Bloods – IgE, RAST, Alpha 1 Antitrypsin
Allergy testing
ABG & oxygen assessment
Referral for chest x-ray
Service outcomes
Between January 2023 and June 2024, 268 patients were reviewed in the ANP clinic.
COPD specific outcomes
• 93 patients with COPD were seen, of which 81 were new referrals.
• 72% of new referrals had sameday PFTs.
• 37 new diagnoses of COPD were made see figure 4
• 20 patients were referred to pulmonary rehabilitation.
• 52 patients were discharged from the ANP clinic, while 7 were referred to a respiratory consultant.
The ANP-led COPD service is closely aligned with the HSE COPD Model of Care and the Respiratory National Clinical Programme. By bridging emergency, inpatient, and community services, this model ensures that patients receive diagnostic services, treatment plans, and access to multidisciplinary teams—all within a streamlined, patient-centred framework. This seamless integration of acute and community care improves health outcomes and supports the sustainability of respiratory services at Sligo University Hospital.
Future plans
There are many exciting projects happening in terms of COPD care
but nationally and internationally. We would like to focus on the use of remote patient monitoring for real-time assessment of COPD exacerbations and expansion of tele-monitoring to detect exacerbations early and reduce hospital admissions.
in quality of life biologics have made to people with asthma and there has been exciting research on expanding the role of biologics for people who have severe COPD with asthma overlap.
to better manage symptoms such as breathlessness.
We are concentrating more on treatable traits and identifying characteristics and phenotypes which respond to specific targeted treatments which means we can provide personalised precision medicine. We see the improvement
We also know that COPD rarely exists in isolation. We are beginning to appreciate that many diseases co-exist with COPD and they also interact. This is obviously the case for cardiovascular disease. There is a potential to hold joint Respiratory and Cardiology clinics in the CDM hub
COPD care by the Respiratory Nursing Team at Sligo University Hospital exemplifies a patientcentred, multidisciplinary approach, integrating acute and community services. From innovative inpatient reviews to the establishment of COPD Outreach and the ANP-led clinic, these services have demonstrated significant improvements in patient outcomes, resource utilisation, and quality of life.
The ANP-led COPD service is closely aligned with the HSE COPD Model of Care and the Respiratory National Clinical Programme. By bridging emergency, inpatient, and community services, this model ensures that patients receive diagnostic services, treatment plans, and access to multidisciplinary teams all within a streamlined, patient-centred framework. This seamless integration of acute and community care improves health outcomes and supports the sustainability of respiratory services Sligo University Hospital.
In a recent survey* 7 in 10 people did not know that when a person living with HIV is on effective treatment, they cannot pass it on to sexual partners
There are many exciting projects happening in terms of COPD care but nationally and internationally
The HSE in partnership with people living with HIV, has launched ‘You, Me and HIV,’ a new nationwide campaign to address misconceptions around HIV and help reduce the stigma experienced by people living with HIV in Ireland. The campaign features people living with HIV and their loved ones.
We would like to focus on the use of remote patient monitoring for real-time assessment of COPD exacerbations and expansion of tele-monitoring to detect exacerbations early and reduce hospital admissions.
Research has found that late diagnosis is often related to fear of diagnosis and stigma, highlighting the need to reduce stigma around HIV and testing. The latest figures from the Health Protection Surveillance Centre (HPSC) show a decrease in the rate of first-time HIV diagnoses in Ireland in 2023. However, nearly two in five people (39%) were diagnosed late. HIV Fact Check:
• HIV, regardless of whether or not a person is on treatment, is not passed on from kissing, or from using the same cups, plates, forks or toilet seats.
• People on effective treatment cannot pass HIV to sexual partners. When a person living with HIV is on effective treatment, the viral load is so low that it is not detected in their blood. This is often referred to as 'undetectable' equals 'untransmittable' (U=U).
• People living with HIV on effective treatment can have healthy pregnancies and go on to deliver healthy babies without passing on the virus. Effective treatment in pregnancy prevents infection in babies.
• HIV and AIDS are not the same thing. HIV is a virus that attacks the human immune system weakens its ability to fight infection and disease. AIDS describes the group of illnesses that you can get in the late stage of HIV infection. Most people with HIV will not develop AIDS-related illnesses because of the advancements in treatments.
We are concentrating more on treatable traits and identifying characteristics and phenotypes which respond to specific targeted treatments which means we can provide personalised precision medicine. We see the improvement in quality of life biologics have made to people with asthma and there has been exciting research on expanding the role of biologics for people who have severe COPD with asthma overlap.
Professor Fiona Lyons, National Clinical Lead, HSE Sexual Health Programme said, “Thanks to the generosity, bravery and passion of Veda, Robbie, Aoife, Pat, Ainara and Peter, we are confident that ‘You, Me and HIV’ will make a real and lasting impact on the HIV-related stigma that persists in Ireland today.
“Through sharing their real life experiences, advice and reassurance it helps us address misconceptions, and helps ensure the wider public has the most up to date facts and information about HIV. Additionally, we hope the campaign will alleviate fears about getting tested for HIV or seeking treatment. Please do not delay, we are here to help you. You deserve to live a healthy and happy life like everyone else.”
MAKE IT YOUR NEW YEAR’S RESOLUTION TO GET ANY WORRYING SIGNS OR SYMPTOMS OF CANCER CHECKED OUT: IRISH CANCER SOCIETY
The Irish Cancer Society’s Your Health Matters roadshow visited 26 locations across the country last year, engaging with over 27,300 people about the signs and symptoms of cancer.
The charity is urging the public to continue to get any signs or symptoms of concern checked out as we head into 2025.
The Your Health Matters roadshow initiative offers free health checks from Cancer Nurses in communities across Ireland.
Over 1,950 health consultations took place at the roadshows, and the Irish Cancer Society’s Cancer Nurses and volunteers are looking forward to getting out on the road again in 2025 and engaging with even more members of the public
Kevin O’Hagan, the Irish Cancer Society’s Cancer Prevention Manager, said the roadshows play an important role in the early detection of cancer, and he urged the public to seek medical advice if they notice any worrying changes in their body.
“We were delighted to be able to bring our Your Health Matters roadshow to 26 locations nationwide in 2024 with over 27,300 people engaging with our nurses, and we are looking forward to continuing to bring the roadshow to communities across the country in 2025. The roadshows are an important tool for helping with the early detection of cancer.
“People attending the roadshows had a wide range of concerns ranging from risks of cancer, diagnosis, treatment and support after treatment. Feedback from the nurses at the roadshows indicated that 70% of people who attended were concerned about signs and symptoms of cancer, and 14% had a family member or loved one with cancer.
“Many people reported that they had not been to their GP for some time. For 16%, it was more than a year since they last saw their GP, 40% up to a year, and 3% did not have a GP, so they were very grateful for the opportunity to speak with a Cancer Nurse.
“We are urging people with any worrying symptoms to go to their GP and get them checked out. If you’ve noticed any unexplained weight loss, a new lump or a persistent cough, make it your New Year’s resolution to get it addressed and find out what’s causing it.
“We know that the early detection of cancer is vital and saves lives, and healthcare services are there for you and will help you to uncover what is causing your symptoms.
“Screening programmes like BreastCheck, CervicalCheck, and BowelScreen are another valuable tool that help to pick up cancer early in people who may be showing no other symptoms, so it is vital that those who are eligible attend for screening when they are invited.”
Anyone with a concern or query about cancer can contact the Irish Cancer Society’s Freephone Support Line at 1800 200 700 or email supportline@irishcancer.ie.
TEVA
Teva Pharmaceuticals is pleased to announce the launch of Varenicline Teva, a generic prescription medication indicated for smoking cessation.
Varenicline Teva works by targeting nicotine receptors in the brain, reducing cravings and withdrawal symptoms associated with quitting smoking.
For any queries or further information, please contact your local Teva representative or contact Teva on 1 800 201 700.
Product information is available upon request or from the SmPC available at HPRA.ie.
Adverse events should be reported. Reporting forms and information can be found at www. hpra.ie. Adverse events should also be reported to Teva UK Limited on +44 (0) 207 540 7117 or medinfo@tevauk.com.
Date of preparation: December 2024
Job code: Vcli-IE-00013


BOOTS NIGHT WALK RAISES RECORD ¤189,977 FOR IRISH CANCER SOCIETY NIGHT NURSES, FUNDING
Boots Ireland’s Night Walk for Night Nurses has achieved a remarkable fundraising milestone, raising a record ¤189,977 for the Irish Cancer Society Night Nursing Service. This incredible generosity will fund approximately 370 nights of end-of-life care, allowing patients to spend their final days at home surrounded by loved ones and providing essential respite for their families.
Since 2012, Boots customers and colleagues have raised over ¤3.5 million - funding approximately 9,500 nights of care. Now in its 13th year, the Night Walk has inspired over 6,000* participants to collectively walk an incredible 300,000 kilometers* in solidarity with patients and families impacted by cancer.
Stephen Watkins, Managing Director of Boots Ireland, expressed his gratitude: “We are immensely proud to partner with the Irish Cancer Society and deeply grateful for the outpouring of support for this year’s Night Walk. This recordbreaking achievement will bring comfort and support to so many families facing incredibly difficult times. Thank you to everyone who participated and donated.”
Pictured at the cheque presentation above is: (l-R) Georgina Roache, Miriam Callaghan, Joe Scallan, Director of Stores, Boots Ireland, Laura Clarke, Averil Power, CEO, Irish Cancer Society, Cherise Hand, Stephen Watkins, MD, Boots Ireland, Agata Wirkus, Martha Ryan, Director of HR & ESG Lead, Caoimhe O’Connor, ICS, Claire Bowman, ICS and Louise O’Brien, Boots Ireland.
Picture Andres Poveda
The Cardiothoracic team at University Hospital Galway have won a HSE Excellence Award for their project ‘Minimally Invasive Vessel Harvesting for Coronary Artery Bypass Grafts’ in the Innovation in Service Delivery Category on 28th November 2024.
The awards are an opportunity to showcase and celebrate examples of the great work that happens every day across our health service. They aim to encourage and inspire health service staff to develop and improve care and services for patients.
Describing the project Dr Sadiq Siddiqui, Associate Specialist in Cardiothoracic Surgery at University Hospital Galway said, “Part of our job is to perform coronary artery bypass surgery which requires harvesting veins and radial artery to perform this surgery.
“Traditionally those arteries and veins were harvested via a long incision which are associated with pain, wound infection, debilitating scars and delayed mobility
“For the last eight years we have been harvesting the radial artery and long saphenous vein via a 2cm incision and this technique has shown that the wound infection rate has reduced from 7% to 0%, while also maintaining excellent clinical outcomes and improving patient satisfaction from the cosmesis of
Cardiothoracic 1: From left, Sharon Umeh, Staff Nurse; Sharon Collins, Clinical Nurse Manager 1; Emer Lambe, Staff Nurse; Dr Sadiq Siddiqui, Associate Specialist in Cardiothoracic Surgery; Patient Bernadette Benagh; Annie Flaherty, Health Care Assistant and Niamh McDermott, Clinical Nurse Manager 2
their wound and the avoidance of post-operative complications.”
Speaking about her experience, Patient Bernadette Benagh said, “I am so honoured and privileged to be able to tell you about my experience. It was a wonderful, wonderful experience from the day I started until the day I finished and the way I am now is because of these people who saved my life.”
Welcoming the news, Hospital Manager Chris Kane said, “This technique is another enhancement in cardiac surgery resulting in better patient outcomes.
“We are enormously proud of the team and the recognition they have received by winning an HSE Excellence Award; it is well-deserved.”
Bernard Gloster, HSE CEO, commending the winners, said, “These awards create the opportunity to show innovation and creativity, which ultimately improves the services that we provide. Well done and thank you to all those short-listed as well as the winners. It is your work that helps pave the way to a brighter future for Ireland’s health service.”
Anne Marie Hoey, HSE Chief People Officer, highlighting the positive outcomes achieved, said, “As well as providing an opportunity to recognise the great work of staff and teams across our wide range of services, the Excellence Awards is an important channel to share learnings with our colleagues across the health service.
“Through this pooling of knowledge and expertise, the projects inspire a sense of staff pride, teamwork and collaboration. This will positively impact the overall environment for our staff whilst improving outcomes for the people who rely on our care and support.”

Children’s Health Ireland (CHI), in partnership with St. James’s Hospital have successfully opened Ireland’s first early phase clinical trial, enrolling eligible children with Duchenne muscular dystrophy (DMD). The global trial, called DELIVER, which started in August 2022 as the first-in-human (phase 1/2) trial of a therapeutic called DYNE-251 at sites around the world targets male children with a specific type of DMD (children amenable to exon 51 skipping).
The trial is being delivered by the HRB CHI Clinical Research Centre and the Wellcome HRB Clinical Research Facility at St James’s Hospital and involves delivery of a next generation molecular therapy developed by Dyne Therapeutics, a Massachusetts based biotechnology company, specialising in the development of therapies for rare muscle diseases. The study is progressing through at sites around the world but is the first time such an early phase trial has been conducted in children in Ireland.
Duchene muscular dystrophy (DMD) is a rare neuromuscular disease that results in progressive deterioration in muscle strength and premature death. Symptoms are often first detected as early as age two, but diagnosis often occurs between four and five years of age. The disease is almost exclusively seen in boys. It is caused by gene mutations that affect a protein called dystrophin, which plays a critical role in muscle cell structure and function. As they grow, children with DMD become gradually weaker, losing the ability to walk and ultimately being confined to a wheelchair. Weakness of the muscles involved in swallowing, breathing and heart function are particularly serious and ultimately life threatening. Currently, very few treatments are available for this devastating disease, and children and their families are anxiously awaiting treatments that can target the basic defect in DMD, aimed at slowing or stopping the inevitable decline in muscle function.
Clinical trials can be extremely complex to undertake and are governed by very strict and detailed safety codes. They involve years of careful planning by a large team of experts within the hospitals, regulators and industry. Global healthcare regulatory agencies comprehensively review data on investigational medicines before
the clinical trial can start in humans.
Dr Declan O’Rourke, Consultant Paediatric Neurologist and principal investigator for the study at CHI, said: “This is a watershed moment for CHI. We have been working for many years to increase the number of trials we offer to our patients with progressive muscle diseases and have had some great successes already. DMD is a condition with very few treatment options currently, and with this trial, it is great to be at the very forefront of potentially bringing new, transformative therapies to children.”
The development of clinical trials in Neurology in CHI has been supported by a research grant from Children’s Health Foundation, CHI’s fundraising partner.
CHI’s Director of Research & Innovation, Paul McNally welcomed CHI’s participation in the ongoing study saying:
“This is an exciting day for everyone involved in clinical trials in CHI. These early phase trials are an enormous undertaking but a vitally important thing for us to be able to do if we really want to become a leading children’s hospital and offer our patients the very latest in cutting edge treatments in the world. Our partnership with St James’s Hospital has been critical here and is an example of the type of benefits that we can expect when working together on the same campus when we move to the new children’s Hospital.”
DON’T LET STIGMA BE A BARRIER TO SEEKING HELP FOR MENTAL HEALTH DIFFICULTIES
Some 20% of adults in Ireland would tell no one if they were experiencing suicidal thoughts, St Patrick’s Mental Health Services’ 2024 annual Attitudes to Mental Health and Stigma Survey has revealed. The survey also found a significant 49% of people who have experienced mental health difficulties have not sought treatment due to stigma or embarrassment. In light of these findings, St Patrick’s Mental Health Services is urging everyone to check in on their own and their loved ones’ mental health and wellbeing, particularly during the Christmas period when the pressures of the season can often take over, and to seek support if needed.
For over 10 years, St Patrick’s Mental Health Services has
commissioned this annual survey to track changing attitudes towards mental health and stigma. While there have been improvements in attitudes, the 2024 findings highlighted that stigma is still a significant barrier to seeking help, with shame or embarrassment often impacting willingness to disclose mental health difficulties and to engage in treatment. For example, this year’s survey, which polled a nationally representative of over 1,000 adults in the Republic of Ireland, found:
- 53% of respondents have experienced a mental health difficulty, yet more than one-third (34%) did not seek treatment, with the main reason for not doing so cited as shame.
- 23% of people would consider it a sign of weakness if they sought help for a mental health difficulty. At the same time, 11% would consider it a sign of weakness if a friend or loved one sought help, indicating the prevalence of self-stigma.
Speaking about the findings, Paul Gilligan, CEO of St Patrick’s Mental Health Services said: “While attitudes towards mental health are improving in many ways, as reflected year-on-year in our annual survey findings, one concerning area that has seen little change is in the disclosure of suicidal thoughts and the idea that it’s a sign of weakness to seek help for your mental health.
Speaking up about your mental health takes courage. Oftentimes, a person might feel like a burden to others or that mental health difficulties may reflect a personal weakness. This is absolutely not the case, and we need to continue to challenge these deeply ingrained attitudes; to continually demonstrate the strength in speaking out and seeking help and reinforce the possibility of recovery from mental health difficulties with the right supports.
In the busyness of the holiday season, it’s important to remember that the most important thing we can do with our time is to connect with our emotional selves and to check in on how we, and others, are really feeling. If we ourselves, or someone we love, is struggling, don’t let stigma stand in the way of seeking support.”
Despite some stagnated attitudes towards stigma, findings from the 2024 survey have also shown areas where attitudes to mental health are notably improving, for example:
• Less people are afraid of experiencing mental health difficulties themselves in the future than they were five years ago, indicating increased understanding of mental health difficulties and awareness that recovery from mental health difficulties is possible.
• Since 2020, there has been a 16% decrease in the number of people who believe that being treated for a mental health difficulty is still seen by Irish society as a sign of personal failure. In 2020, 64% of respondents agreed with this statement while this figure stood at 48% in 2024.
• 66% of people who experienced mental health difficulties in 2024 sought treatment – a 10% increase since 2023.
• 78% of this year’s survey respondents also believe that people with mental health difficulties experience less stigma and discrimination than 10 years ago.
• 52% have had a positive experience of disclosing mental health difficulties at work, at home or in the local community.
MSD IRELAND WELCOMES LAUNCH OF “ACTION PLAN TO ELIMINATE HPV-RELATED CERVICAL CANCER” IN IRELAND BY 2040
MSD Ireland welcomes the news that cervical cancer rates in Ireland continue to drop as Ireland launches an ambitious Cervical Cancer Elimination Action Plan, a “significant milestone in the global drive to eliminate cervical cancer by 2040.” The newly launched action plan aligns with MSD’s own commitment to harnessing the power of leading-edge science to address some of the world’s biggest healthcare challenges, including the elimination of HPV related cervical cancer.
Cervical cancer remains one of the most common cancers affecting women globally, with an estimated 265 cases diagnosed annually in Ireland alone, and 85 lives lost each year to this diseaseiii. Yet, with a combination of increased vaccination, enhanced screening, and more comprehensive treatment, we can work towards a future where no woman must go through the distress of a cervical cancer diagnosis in Ireland.
This Action Plan is launched on the fourth anniversary of the World Health Organisation (WHO) global initiative to eliminate
cervical cancer and one year on from the Minister for Health's announcement of Ireland's commitment to this goal, setting the target date for cervical cancer elimination by 2040. Ireland joins countries like Sweden and Australia, which are already on track to eliminate the disease.
Samantha Humphreys, Managing Director of Human Health at MSD Ireland, commented: “We welcome the introduction of the Cervical Cancer Elimination Action Plan in Ireland as a significant milestone in the global drive to eliminate cervical cancer. Ireland has an important role to play and we’re immensely proud of the role we play in supporting the goal of HPV elimination, starting with cervical cancer elimination.”
She continued: “The introduction of this action plan marks a crucial step forward in reducing the burden of the disease and improving long-term health outcomes for women. The action plan represents a vital step in Ireland's effort to eliminate the impact of the disease, through a focus on screening and vaccination programmes, and ultimately saving lives by preventing future cases of cervical cancer. To achieve this ambitious goal, stakeholders must work together on this action plan, and the specific goals outlined within, which includes: ‘increasing HPV vaccination rates for girls by age 15 to 90% by 2030 (currently at 84%); maintaining cervical screening coverage above 70% (currently at 73%); and maintaining the number of women receiving treatment above 90% (currently at 97%). We particularly welcome and will support efforts to increase awareness and education about HPV and related cancers in the community and among vulnerable and marginalised groups.”
In anticipation of the full implementation of the six new health regions in early 2025, the NCRI annual report on national cancer statistics has, for the first time, examined cancer incidence and survival3-6 across these six geographies for the four most common cancers in Ireland: lung, bowel (colorectal), breast and prostate cancer. These data are presented alongside national statistics providing an annual update on the status of cancer in Ireland.
In Ireland as a whole, an average of 44,000 tumours were diagnosed each year during 2020-2022. Prof.
Deirdre Murray, Director of NCRI, and Professor of Epidemiology at University College Cork, noted that the majority of these (24,200) were potentially life-changing invasive cancers requiring extensive treatment. On average 9,800 people died each year from cancer during 2020-2022. One in five deaths were due to lung cancer.
Considering the six new health regions, the majority (60%) of all invasive cancers diagnosed during 2020-2022 occurred in the three eastern geographical areas (Dublin and North East, Dublin and Midlands, and Dublin and South East). Compared to the national average, the lung cancer incidence rate was higher in Dublin and Midlands, and lower in Dublin and South East. By contrast, the rate of prostate cancer was higher in Dublin and South East, but lower in Dublin and North East.
The proportion of people diagnosed during 2009-2018 and surviving at 5-years following diagnosis was lower than the national average for those living in the Mid West for colorectal, lung, and female breast cancer. Fiveyear survival for prostate cancer was also lower than the national average for people resident in the Dublin and North East region at the time of their diagnosis.
The report also found that, in Ireland as a whole, the number of people living with and beyond cancer continues to grow with an estimated 220,700 people who had been diagnosed with cancer still alive at the end of 2022 (about 1 in 23 of the Irish population).
Prof. Murray, said: “One of the core functions of the NCRI is to provide data for cancer service planning, evaluation and policymaking. While geographic variation must always be interpreted with care these data can point to important opportunities for improvement across cancer care from prevention, early diagnosis, and screening, to access to services and treatment.”
Chair of the NCRI Board, Dr Robert O’Connor added: “This report provides a comprehensive overview of cancer statistics as our country began to recover from the COVID-19 pandemic. The data reveal critical insights into regional health outcome differences and emerging trends in cancer survival rates. The findings underscore the continued need for targeted, strategic investments in cancer research, prevention, treatment, and post-treatment care to equitably maximise patient outcomes and community health.”
THE HEALTH SERVICE EXECUTIVE (HSE) APPROVES FINTEPLA®(FENFLURAMINE) FOR PATIENTS IN IRELAND WITH DRAVET SYNDROME AND LENNOX GASTAUT SYNDROME (LGS)
UCB is pleased to announce that the Health Service Executive (HSE) has approved FINTEPLA® (fenfluramine) for the treatment of Dravet syndrome (DS) and Lennox-Gastaut syndrome (LGS) in patients from two years of age.
This reimbursement has allowed eligible patients to access fenfluramine much sooner than expected under the routine approval processes.
Nadeem Aurangzeb, Head of Rare & Epilepsy, UCB UK & Ireland, said: “We are immensely pleased that fenfluramine has been approved by the HSE. We know that Irish patients and their families have been looking forward to accessing this treatment, due to the burden that Dravet syndrome and Lennox Gastaut syndrome places on the lives of those living with these complex, rare conditions. We’re also pleased that Irish patients will be able to access fenfluramine over a year ahead of the expected approval date. This approval means fenfluramine is available to Irish patients ahead of those in the rest of the UK, which is almost unheard of.”
LGS is a rare, severe, earlyonset developmental epileptic encephalopathy, which is characterised by multiple types of seizures, intellectual impairment and electroencephalography (EEG) abnormalities. It affects around 0.1 to 0.28 people per 100,000 and it accounts for 1-10% of childhood epilepsies and 1-2% of all epilepsies.
The approval of fenfluramine for the treatment of LGS-associated seizures is based on phase 3 data and a long-term open-label extension study.4,5 In the phase 3 trial, 263 patients ages two to 35 years were randomised to receive either fenfluramine or placebo.
Patients assigned to fenfluramine experienced a significantly greater reduction in drop seizures, compared with the placebo group. The most frequent adverse events included decreased appetite, somnolence, and fatigue, and there were no cases of valvular heart disease (VHD) or pulmonary arterial hypertension (PAH). Furthermore, in the open-label extension study, patients taking fenfluramine continued to
experience sustained (39.4% 51.8%, 28.6% overall) reductions in drop seizure frequency.
The treatment was generally well-tolerated and long-term VHD and PAH was not observed.5 DS, another developmental epileptic encephalopathy, is also a rare and life-long form of epilepsy that begins in infancy and is marked by frequent seizures.
People living with DS experience significant developmental and motor impairments that persist into adulthood and quality of life is severely impacted for patients, their families, and caregivers due to its high physical, emotional, and financial burden.
Data from a randomised, double-blind, placebo-controlled phase 3 trial supports the use of fenfluramine in DS, where 119 patients received either fenfluramine or placebo.10 Fenfluramine provided significantly greater reduction in convulsive seizure frequency, compared with the placebo group. It was also generally well-tolerated, with no observed cases of VHD or PAH.
People living with both LGS and DS have an increased risk of sudden death, known as Sudden Unexpected Death (SUDEP), and it is estimated that there are over 130 epilepsy-related deaths in Ireland per year.
Nicola Kehoe of Dravet Syndrome Ireland said: “We are delighted to have fenfluramine available as a funded treatment for people living with Dravet syndrome and Lennox Gastaut Syndrome in Ireland. We know from the international community that fenfluramine is very effective in reducing seizures and we welcome the potential improvements in health and quality of life for our community of families living with these very complex epilepsies.”
Fenfluramine is currently only available to people living with LGS in Ireland, however, UCB is working collaboratively to secure reimbursement for patients living in England, Wales and Scotland. Marketing authorisation was previously obtained from the European Medicines Agency (EMA) in 2020, and approval was obtained from the European Commission (EC) in 2023.
The Medicines and Healthcare products Regulatory Agency (MHRA) authorised fenfluramine for the treatment of seizures associated with DS and LGS in July 2023.

Pictured at the Novartis and Deciphex AI event are (from left): Caitriona Walsh, Country President, Novartis Ireland, Dr. Donal O’Shea, CEO of Deciphex and Laura Kavanagh, Research & Advocacy Manager, the Irish Platform for Patients’ Organisations (IPPOSI). Pictured taking part in the panel discussion are (from left); Moderator Elaine Burke; Minister of State for Digital Dara Calleary TD; Laura Kavanagh and Neasa McNabola, Senior Scientific Adviser, IDA BioPharma Team
NOVARTIS AND DECIPHEX HOST ROUNDTABLE ABOUT IRELAND’S AI SECTOR TO MARK COLLABORATION
Novartis Ireland has marked its Collaboration Agreement with Deciphex, a Dublin-based digital pathology company, at a specially convened roundtable on Artificial Intelligence (AI).
The collaboration is a global agreement between Novartis and Deciphex. Harnessing cuttingedge technology, it seeks to develop a suite of AI tools that will improve the efficiency and accuracy of preclinical studies and accelerate the painstaking work of drug discovery and development. Ultimately, this collaboration has the potential to positively impact patient pathways.
In Ireland, Novartis and Deciphex marked the agreement at a panel discussion titled ‘How Ireland can be the Centre of Artificial Intelligence (AI) for Europe’. Taking place at Novartis Ireland and officiated by Minister of State for Digital Dara Calleary TD, the event featured contributions from Novartis and Deciphex leadership and representatives from IDA Ireland and patient-led platform, IPPOSI.
The event heard from panelists that government policies and
State support, ongoing investment and a strong pipeline of tech talent from universities are key factors in creating a fertile ecosystem where the AI sector in Ireland is flourishing.
Against this backdrop, partnerships between indigenous firms and multinationals such as the Novartis and Deciphex collaboration, can deliver positive economic, clinical and societal outcomes in the coming years.
The roundtable also heard that with AI set to transform sectors such as healthcare over the coming years, the public and patients are being engaged to ascertain the degree in which safeguards and regulation can provide comfort that with AI advances, a human will still be in the loop. The Irish public are being invited to share their insights for policymakers through important initiatives such as the IPPOSI ‘Citizens’ Jury on AI in Healthcare’.
Minister of State for Digital Dara Calleary TD said: “I am delighted to officiate at today’s panel discussion. Ireland’s AI sector is flourishing, with a partnership approach between State bodies, enterprise and the public key to delivering transformative societal benefits in the coming years. The Novartis-Deciphex collaboration is a great example of how this approach works in practice, with an indigenous medtech collaborating with a global leader in pharma to harness cutting-edge technology and deliver better outcomes for patients.”
Michael Lohan, CEO, IDA Ireland said, “Artificial Intelligence is a cornerstone of Ireland’s strategy to drive innovation and fuel economic growth. By fostering a robust AI ecosystem, we are not only attracting leading global tech companies but also empowering local enterprises to compete on the world stage. The integration of
AI across various sectors is pivotal in enhancing productivity, creating high-value jobs, and ensuring Ireland remains at the forefront of technological advancements”
Dr. Donal O’Shea, CEO of Deciphex said: “We are thrilled to collaborate with Novartis in leveraging AI technology to drive innovation in preclinical research. This partnership underscores our commitment to advancing healthcare through the development of cutting-edge solutions that address critical challenges in drug development lifecycles. Novartis is committed to innovating AI-based approaches with potential to accelerate drug discovery and development and bring life-changing medicines to patients faster.” Statement approved by stakeholder
Caitriona Walsh, Country President, Novartis Ireland said: “I’m excited that Novartis has entered into a collaboration agreement with Deciphex to develop Artificial Intelligencebased approaches aimed at improving the efficiency and accuracy of preclinical studies. We believe AI has transformative potential to accelerate the painstaking work of drug discovery and development. I look forward to seeing how our collaborative efforts can help us improve and streamline existing processes to deliver impactful medicines to patients more quickly.”
HSE APPROVES FINTEPLA®
UCB is pleased to announce that the Health Service Executive (HSE) has approved FINTEPLA® (fenfluramine) for the treatment of Dravet syndrome (DS) and Lennox-Gastaut syndrome (LGS) in patients from two years of age.
This reimbursement has allowed eligible patients to access fenfluramine much sooner than expected under the routine approval processes.
Nadeem Aurangzeb, Head of Rare & Epilepsy, UCB UK & Ireland, said: “We are immensely pleased that fenfluramine has been approved by the HSE. We know that Irish patients and their families have been looking forward to accessing this treatment, due to the burden that Dravet syndrome and Lennox Gastaut syndrome places on the lives of those living with these complex, rare conditions. We’re also pleased that Irish patients will be able to access fenfluramine over a year ahead of the expected approval date. This approval means fenfluramine is available to Irish patients ahead of
those in the rest of the UK, which is almost unheard of.”
LGS is a rare, severe, earlyonset developmental epileptic encephalopathy, which is characterised by multiple types of seizures, intellectual impairment and electroencephalography (EEG) abnormalities. It affects around 0.1 to 0.28 people per 100,000 and it accounts for 1-10% of childhood epilepsies and 1-2% of all epilepsies.
The approval of fenfluramine for the treatment of LGS-associated seizures is based on phase 3 data and a long-term open-label extension study. In the phase 3 trial, 263 patients ages two to 35 years were randomised to receive either fenfluramine or placebo. Patients assigned to fenfluramine experienced a significantly greater reduction in drop seizures, compared with the placebo group. The most frequent adverse events included decreased appetite, somnolence, and fatigue, and there were no cases of valvular heart disease (VHD) or pulmonary arterial hypertension (PAH).
Furthermore, in the open-label extension study, patients taking fenfluramine continued to experience sustained (39.4% 51.8%, 28.6% overall) reductions in drop seizure frequency. The treatment was generally welltolerated and long-term VHD and PAH was not observed.5 DS, another developmental epileptic encephalopathy, is also a rare and life-long form of epilepsy that begins in infancy and is marked by frequent seizures.
People living with DS experience significant developmental and motor impairments that persist into adulthood and quality of life is severely impacted for patients, their families, and caregivers due to its high physical, emotional, and financial burden.
Data from a randomised, double-blind, placebo-controlled phase 3 trial supports the use of fenfluramine in DS, where 119 patients received either fenfluramine or placebo. Fenfluramine provided significantly greater reduction in convulsive seizure frequency, compared with the placebo group. It was also generally well-tolerated, with no observed cases of VHD or PAH.
People living with both LGS and DS have an increased risk of sudden death, known as Sudden Unexpected Death (SUDEP), and it is estimated that there are over 130 epilepsy-related deaths in Ireland per year.
Nicola Kehoe of Dravet Syndrome
Ireland said: “We are delighted to have fenfluramine available as a funded treatment for people living with Dravet syndrome and Lennox Gastaut Syndrome in Ireland. We know from the international community that fenfluramine is very effective in reducing seizures and we welcome the potential improvements in health and quality of life for our community of families living with these very complex epilepsies.”
Fenfluramine is currently only available to people living with LGS in Ireland, however, UCB is working collaboratively to secure reimbursement for patients living in England, Wales and Scotland. Marketing authorisation was previously obtained from the European Medicines Agency (EMA) in 2020, and approval was obtained from the European Commission (EC) in 2023. The Medicines and Healthcare products Regulatory Agency (MHRA) authorised fenfluramine for the treatment of seizures associated with DS and LGS in July 2023.
Sligo University Hospital is delighted to announce the opening of the Medical Offsite Ward, a new 26-bed ward comprising of six four-bed rooms and two single ensuite rooms, located at St John’s Community Hospital, Sligo.
This new development is an extension of Sligo University
The new 26-bed Medical Offsite Ward which is an extension of Sligo University Hospital will provide additional bed capacity and improved accommodation for patients
Hospital and will provide additional bed capacity, improved accommodation for patients and will ease patient flow from the Emergency Department and throughout the hospital, while meeting the required national standards and delivering enhanced quality care to patients.
The additional beds was part of the capital funding from the HSE 2024 Capital Plan, costing in excess of ¤2.5m for the design, build and equipping of the ward.
The medical offsite ward which is a specialised facility will provide structured medical support to patients who meet the criteria for the ward and will continue to benefit from medical supervision and therapeutic support, provided by a full team of health and social care professionals in a calm environment. It is suitable for patients who would benefit from an additional few days under the care of hospital staff.
Grainne McCann, Hospital Manager, stated, "This new development is a welcome addition to the hospital, allowing us to expand our inpatient care in a new contemporary ward designed to provide personalised, compassionate care to support our patients' medical needs and recovery during their inpatient stay."
Patient, Elizabeth Fallon, said, “When I came to the new medical ward, I was flabbergasted with the space and facilities. Everybody here is fantastic and the atmosphere is great. Everyone was very welcoming and cheerful on my arrival to the ward. The physios are fantastic. Last night was the first night I got a good sleep. My son who came into see me was also amazed with the new environment.”



References:1)*Countries: Europe (Austria, Belgium, Bulgaria, Croatia, Czech Republic, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Netherlands, Norway, Poland, Portugal, Romania, Serbia, Slovakia, Slovenia, Spain, Sweden, Switzerland, UK) Source: Based on internal analysis by Laboratoires Théa using quarterly data from the following source: IQVIA Consumer Health Customized Insights for the period 2023, volume and value sales in Euros MNF, limited to CHC 07A3, reflecting estimates of real-world activity in these countries: Austria, Belgium, Bulgaria, Croatia, Czech Republic, Estonia, Finland, France, Germany, Greece, Hungary, Ireland, Italy, Latvia, Lithuania, Netherlands, Norway, Poland, Portugal, Romania, Russia, Serbia, Slovakia, Slovenia, Spain, Sweden, Switzerland, UK. Copyright IQVIA. All rights reserved. 2) Pinto-Bonilla JC et al. Ther. Clin Risk Manag 2015:11;595-603 3) Schmidl D et al. Cornea 2015;34(4):421-6 TP24032
& Protects
Suitable for all dry eye sufferers including contact lens wearers A single drop Lasts for up to 4 hours3 Clinically Proven2 to treat symptoms of dry eyes
www.theapharma.ie