

Preser vative -free Phosphate -free Hypotonic Osmoprotec tive



Preser vative -free Phosphate -free Hypotonic Osmoprotec tive




Indications: Treatment of mild non-infectious allergic or inflammator y conjunctival diseases. Posology: The recommended dosage is 2 drops 2 to maximum of 14 days Gradual tapering off up to one administration ever y other day may be recommended in order to avoid a relapse In case active substance or to any of the excipients; Known glucocorticosteroid-induced ocular hypertension and other forms of ocular hypertension; ulceration (except when combined with specific chemiotherapeutic agents for herpes virus), conjunctivitis with ulcerative keratitis even at the initial conjunctivitis and purulent blepharitis, stye and herpes infection that may be masked or aggravated by anti-inflammator y drugs Pregnancy: administered glucocorticoids are excreted in breast milk and may cause suppression of growth or of endogenous corticosteroid production or may newborns/infants cannot be excluded Undesirable effects: Hydrocortisone: Eye disorders: Not
Burning, stinging Corticoid class effects: topical corticosteroids Eye disorders: Not known: Allergic and hypersensitivity
corticosteroid-induced uveitis, changes in corneal
corneas.
ZENFLORE ® CALM with Bifidobacterium longum 1714™
For mental wellbeing and emotional balance.
SAFFRON EXTRACT* SUPPORTS EMOTIONAL BALANCE AND RELAXATION 1
VITAMIN B6** SUPPORTS NORMAL PSYCHOLOGICAL FUNCTION
ZENFLORE ® TEEN with Bifidobacterium longum 1714™
Targeted support for teenagers looking for a calm mind and emotional balance.
SUPPORT MENTAL PERFORMANCE***

SUPPORT NORMAL PSYCHOLOGICAL FUNCTION***




Page 5: New Leadership Team at Medicines for Ireland
Page 8: IPHA Annual Conference hears of exceptional economic contribution
Page 10: United Drug supporting Pharmacy Growth
Page 12: How healthcare education is expanding in Ireland
Page 18: Pharmacy to provide enhanced medicines optimisation
Page 26: The role of pharmacy in driving Bowel Screening uptake
Page 40: Supporting patients beyond the dispensary
Page 47: OTC & Retail Pharmacy Product Award winners 2026
PUBLISHER:
IPN Communications Ltd.
77 Camden Street Lower, Dublin X02 XE80 Office: +353 (01) 2339121
MANAGING DIRECTOR
Natalie Maginnis natalie@ipn.ie
EDITOR
Kelly Jo Eastwood: 0044 787654 8989
kelly-jo@ipn.ie
SALES DIRECTOR
Debbie Graham
debbiegraham@ipn.ie +353 8727 99317


This issue comes at a time of significant progress and transformation across the pharmacy sector, with developments that continue to expand both the role and impact of community pharmacy in Ireland.
A major milestone is the rollout of the new Common Conditions Service, supported by a national training programme developed by the Irish Institute of Pharmacy in partnership with RCSI’s School of Pharmacy and Biomolecular Sciences. With more than 3,000 community pharmacists now trained, the profession is stepping confidently into an enhanced clinical role—one that allows pharmacists to assess and treat patients for a range of common conditions, including, for the first time, the prescribing of certain prescription-only medicines. This represents a meaningful shift in patient care, improving access to treatment while reinforcing the position of pharmacists as accessible, highly skilled healthcare providers within the community.
Alongside these clinical advancements, the wider pharmaceutical sector continues to demonstrate its critical importance to Ireland’s economic strength and global standing. As highlighted in new analysis published by the Irish Pharmaceutical Healthcare Association (IPHA), the industry has reached record levels across employment, output, exports and tax contribution. Presented at the IPHA Annual Conference 2026, the findings come at a time of increasing global uncertainty, underscoring the sector’s resilience and strategic value—not only to Ireland, but to Europe as a whole.

This issue also celebrates excellence through the OTC & Retail Pharmacy Product Awards, where this year’s winners highlight the continued innovation shaping the sector. From trusted household brands to emerging product developments, the awards showcase those delivering real value in terms of efficacy, patient outcomes and consumer engagement. The winning products reflect a strong focus on accessibility, evidence-based formulation and clear communication with both pharmacists and patients—key factors in today’s increasingly competitive and informed marketplace.
MARKETING & EDITORIAL
EXECUTIVE
Anna Hadfield Office: +353 (01) 2339121 Mobile: +353 89 947 7171 anna@ipn.ie
CONTRIBUTORS
Donna Cosgrove
Dr Iseult Sheehan
Dr Martina Ní Chúlain
Victoria Spillane
Ian Clarke
Máirín Ryan

Professor Christine Loscher
Mairead McCarthy
DESIGN DIRECTOR
Ian Stoddart Design


of this magazine may be reproduced, stored in a retrieval system or transmitted in any form without written permission. IPN Communications Ltd. has taken every care in compiling the magazine to ensure that it is correct at the time of going to press, however the publishers assume no responsibility for any effects from omissions or errors.
Now firmly established, these awards remain an important benchmark for quality within the industry. Since their launch in 2008, they have consistently recognised the brands and products that not only respond to evolving consumer needs, but also support pharmacists in delivering informed, effective recommendations at the counter.
Together, these developments reflect a sector that is not standing still. From expanded clinical services and strengthened economic impact to continued innovation in product development, pharmacy in Ireland is evolving at pace. As always, we remain committed to bringing you the insights, developments and achievements that matter most to your practice and your patients.
FEATURE: HAYFEVER SEASON P14
FEATURE: SHINGLES VACCINATION P32
TEAM TRAINING: SLEEP DISORDERS P38
CPD: MANAGEMENT OF PAIN P41
FEATURE: INFANT FEEDING P84
CLINICAL PR: P96
A new national training programme developed by the Irish Institute of Pharmacy and RCSI’s School of Pharmacy and Biomolecular Sciences has equipped more than 3,000 community pharmacists to deliver Ireland’s new Common Conditions Service.
The RCSI designed online course, created in partnership with the Pharmaceutical Society of Ireland (PSI) and the Department of Health, provides the mandatory training required for pharmacists to assess and treat patients for eight common conditions, including the prescribing of certain prescription only medicines for the first time. Developed in response to recommendations of the Expert Taskforce to Support the Expansion of the Role of Pharmacy, the training ensures that pharmacists nationwide are equipped to provide safe, standardised clinical care as the service goes live. 94% of community pharmacies have already registered to provide the Common Conditions Service.
The RCSI-led programme includes a regulatory module and eight condition specific modules, built by a multidisciplinary team of
pharmacists, doctors, educators and learning technologists.
Designed using adult learning theory, the course incorporates case-based learning, interactive clinical scenarios, decision-making exercises and reflective practice. Patient and public involvement and universal design for learning principles were integrated throughout to ensure accessibility and patient-centredness.
Delivered on-demand through the Irish Institute of Pharmacy (IIOP) platform, the course supports completion by practising pharmacists without disrupting routine services.
Professor Helena Kelly, Head of the RCSI School of Pharmacy and Biomolecular Sciences, said, “This national training programme is a significant milestone for pharmacy practice in Ireland. RCSI is proud to have worked
with the IIOP and PSI to develop the educational foundation that enables pharmacists to deliver the Common Conditions Service safely, consistently and confidently. By preparing more than 4,000 pharmacists to take on this expanded clinical role, the training supports better patient access to care in line with Sláintecare’s ambition for responsive, community based services.”
Catriona Bradley, Executive Director of the Irish Institute of Pharmacy, adds, “Ensuring pharmacists have access to high quality, evidence based education is central to supporting the safe and effective rollout of the Common Conditions Service. The strong engagement from pharmacists across the country reflects their commitment to expanding their clinical role and improving access to care for patients.”
Over three quarters of a million dosage units of falsified and illegal medicines were detained by the Health Products Regulatory Authority (HPRA) in 2025. Announcing its annual enforcement figures this month, the HPRA confirms that it detained a total of 763,027 dosage units which included just under 14,000 individual packages each linked to a separate purchase by a member of the public of illegal or falsified medicines. This represents a threefold (180%) increase of individual consignments since 2024. A significant proportion of these were presented as GLP 1 products for personal use.

Jennifer McCartan, Compliance Manager at the HPRA
Announcing the figures, the HPRA reiterated the serious health risks associated with sourcing prescription medicines online or through any unauthorised channels.
In the 12 months of 2025, the most significant categories of illegal products detained included sedatives (27%), erectile dysfunction medicines (14%), anabolic steroids (12%), diabetes/ slimming (9%) and analgesics (5%). The breakdown is as follows:
• Sedative medicines – 205,270 units detained
• Erectile dysfunction – 103,185 units detained
• Anabolic steroids – 94,647 units detained
• Diabetes/Slimming – 65,148 units detained
• Analgesic medicines – 40,669 units detained
The HPRA highlights the substantial increase in the volume of GLP-1 type medicines detained, rising from 1,582 in 2024 to 48,752. Authorised GLP-1 prescription-only medicines are intended for specific medical purposes such as diabetes or weight management under certain conditions. The majority of the detained products were promoted as containing semaglutide or tirzepatide and were presented primarily as GLP 1 drops (27,329 items) or microneedle patches (17,170 items).
Pregabalin is another product that saw a significant increase in detentions in 2025, rising from 23,442 in 2024 to 59,905.
Minister for Health, Jennifer Carroll MacNeill TD has launched a call for expressions of interest to serve as voluntary members on the new Clinical Trials Advisory Council (CTAC). This marks a significant milestone in the Government’s ongoing work to strengthen Ireland’s clinical trials ecosystem and to position the country as a global leader in clinical research and innovation.
The establishment of the CTAC follows on from the successful work of the National Clinical Trials Oversight Group (NCTOG) and fulfils a recommendation in its final report, published by the Minister last November.
The CTAC will oversee the implementation of transformative recommendations through creating more agile and responsive systems, enhancing public and patient engagement, promoting value and incentivising activity, ensuring strategic workforce development, and promoting the use of innovative technology.
The work of CTAC, particularly as Ireland takes up the Presidency of the EU in July 2026, will help ensure Ireland is well positioned to benefit from these developments and to support the growth of the biotechnology sector. The CTAC will also play a key role in supporting the forthcoming National Life Sciences Strategy, currently being developed by the Department of Enterprise, Tourism and Employment (DETE).
Minister Carroll MacNeill said, “Clinical trials are essential and have significant impact in achieving better health outcomes for patients. Earlier access to innovative treatments, improving clinical outcomes and attracting investment are vital for building Ireland’s research capacity and supporting the wider economy.
“By working together, we can accelerate the development of novel treatments from the laboratory to real-world use and provide earlier access to treatments and preventative solutions that improve health outcomes for our patients.”
The closing date for receipt of applications is 15:00 Monday 20 April 2026. Full details, including the Information Booklet and how to apply are available on the Department website.
The Irish Pharmacy Union (IPU) has welcomed the announcement that tax relief for health expenses will now apply to pharmacist consultations and medicines prescribed through the Common Conditions Service (CCS).
The CCS allows patients the chance to access timely care for a range of everyday health issues directly from their local pharmacist. This new measure empowers individuals to seek treatment conveniently in their community, without unnecessary delays.
The CCS enables pharmacists to prescribe certain prescription only medicines where this is the most appropriate option. Conditions covered include allergic rhinitis, cold sores, conjunctivitis, impetigo, oral thrush, shingles, uncomplicated urinary tract infections (UTIs) and vulvovaginal thrush.
Welcoming the announcement the IPU said, “Extending tax relief to the Common Conditions Service is a very positive step for patients. It ensures that people who choose to access care from their local pharmacist will receive the same tax relief as they would for a GP visit. This supports patient choice and recognises the important role pharmacists are playing in delivering timely care in the community.
“Pharmacists are medicines experts and have long sought the opportunity to use their expertise in new ways to help patients. The CCS is already making a real difference by improving access to treatment, reducing demand on GPs and ensuring patients can receive safe protocol based care close to home.”
The Common Conditions Service is now accessible in pharmacies across Ireland, offering patients convenient and private consultations tailored to their needs. The service marks a significant step forward in community healthcare, ensuring patients receive expert support close to home.
Medicines for Ireland (MFI), the representative body for the suppliers of the majority of medicines in Ireland, has announced its new senior leadership team, with Mr Donagh O’Leary appointed as Chairperson and Mr Joe Keane as Vice-Chairperson.
Since its foundation in 2016, Medicines for Ireland has been promoting the benefits of the increased use of generic, biosimilar, and value-added medicines in Ireland. The organisation’s core objective is to improve the way Ireland procures and supplies medicines, in order to expand patient access to affordable treatment.
As Managing Director of Clonmel Healthcare Ltd, Mr Donagh O’Leary has been elected Chairperson of the trade association, having previously served in the role of ViceChairperson. He brings over 25 years of experience in the pharmaceutical sector across Ireland and the UK. During his tenure at Clonmel Healthcare Ltd, Mr O’Leary has expanded the business to become Ireland’s largest supplier of consumer healthcare products and generic medicines.
Mr O’Leary and Mr Keane’s appointments follow the conclusion of the new Framework Agreement on the Pricing and Supply of Medicines, a landmark agreement that marks a significant step forward in strengthening Ireland’s pharmaceutical sector. Developed in partnership with
the Department of Health, the Department of Public Expenditure and the HSE, the Agreement sets out a comprehensive package of measures to improve security of supply, enhance affordability, and support a more predictable and competitive market environment through to 31 December 2029.
Taken together, the measures have the potential to generate up to ¤1.4 billion in value, which can be reinvested to support innovation and frontline patient services.
Mr O’Leary comments, “With the new framework agreement now in place, we are entering a fresh phase of growth that will strengthen our impact and sharpen our strategic direction. I look forward to harnessing this opportunity to build momentum, deepen collaboration across the sector, and deliver meaningful progress for our members.”
Joe Keane, who has been elected Vice-Chairperson, is Head of Ireland Operations at Sandoz, trading as Rowex in Ireland. Mr Keane has led the Bantry operation for almost two decades and brings extensive experience in the pharmaceutical industry.

Also speaking at the recent MFI EGM, Mr Keane expressed his gratitude to members for their support, adding that he “looked forward to working closely with members to address both the opportunities and challenges facing the sector.” He highlighted the importance of tackling emerging issues, including the EU’s Urban Wastewater Treatment Directive, noting that a proportionate transposition is imperative to protecting the sustainability of medicine supply in Ireland.
Vaccination remains one of the most effective public health interventions for preventing infectious diseases and reducing morbidity and mortality. Yet vaccination coverage, particularly among adults, remains below recommended targets in many countries.
Existing literature shows that recommendations from healthcare providers are one of the strongest drivers of vaccine acceptance and uptake. To support pharmacists in this role, FIP has launched a new toolkit and accompanying infographic on vaccination communication today. The toolkit focuses on three key areas of communication: pharmacist–patient, pharmacist–public, and pharmacists–other healthcare professionals communication.
“Pharmacists are among the most accessible healthcare professionals and are one of the first points of contact for people seeking advice about vaccines,” said Ms Nisa Masyitah, FIP Data and Intelligence Manager.
“Effective communication skills are therefore essential for improving vaccine uptake, and this toolkit provides practical guidance to support pharmacists in having those conversations with confidence.”
Effective communication is therefore central to pharmacists’ capacity to support vaccination uptake. Conversations about vaccines may involve addressing concerns, correcting misinformation, providing clear and evidence-based recommendations, and collaborating with other healthcare professionals.
This toolkit has been developed to support pharmacists in their communication role within
vaccination programmes. Building on evidence that healthcare provider recommendation is a key driver of vaccination uptake, it focuses on strengthening pharmacists’ capacity to communicate effectively about vaccines with different audiences, including patients, the public, and other healthcare professionals with whom they collaborate.
The toolkit also aligns with FIP Development Goal 16 on Communicable Diseases, which emphasises expanding the role of the pharmacy workforce in the prevention, surveillance, and management of communicable diseases.
McCabes Pharmacy has announced price cuts on 200 essential health and wellbeing products, alongside promotions on more than 1,000 additional items, across its 110 pharmacies nationwide and online.

The reductions make everyday health and wellness essentials affordable with savings across vitamins, first aid essentials, skincare, dental care, feminine hygiene and baby products, helping Irish families manage rising household costs without compromising on their health and wellbeing.
This initiative forms part of McCabes Pharmacy’s ongoing
commitment to “Better Health for Every Body,” ensuring customers have access to trusted healthcare products at better value and helping to support local communities during the ongoing cost-of-living crisis.
The new value offering is designed to make everyday essentials more accessible, helping customers get more of what they rely on without stretching their budgets.
The Pharmaceutical Managers’ Institute is hosting a Stakeholder Briefing on April 23rd, 2026 at The Address Citywest in Dublin.
Brian O’Keeffe, Managing Director of McCabes Pharmacy, said, “At McCabes Pharmacy, we know that households across Ireland are feeling the pressure of rising costs, and everyday health products should never feel out of reach.
“By cutting prices across 200 essential items and offering promotions on over 1,000 products, we want to make it easier for people to look after themselves and their families without stretching their budgets.”
The price cuts span the categories customers rely on most, helping customers continue to prioritise their everyday health and wellness routines.
Alongside competitive pricing, McCabes Pharmacy continues to support customers with convenient services designed to make healthcare easier and more manageable, especially in challenging times.
Through DigiScript, patients can order prescriptions online or via the McCabes Pharmacy app, while the Free Text Service keeps them informed when repeat prescriptions or refills are ready. Reminders for GP renewals, along with notifications about available vaccines and health checks, help patients stay organised, in control of their health, and confident that essential care is always within reach.
“Empowering pharmacists for healthier futures” will be the theme for World Pharmacists Day 2026, celebrated annually on 25 September.
The theme highlights the essential role pharmacists play in improving health outcomes across communities and health systems. As healthcare evolves due to demographic shifts, noncommunicable diseases, digital innovation and climate pressures, empowering pharmacists with the right skills, recognition and systems support is critical. It will also emphasise the shared responsibility of governments, policymakers, regulators, health insurers and the public to create enabling environments in which pharmacists can fully realise their impact.
World Pharmacy Week (19–25 September) will continue to recognise the contributions of the wider pharmacy workforce, including pharmaceutical scientists, pharmacy technicians and collaborative partners.
Community pharmacy is undergoing significant transformation, with the pharmacist’s role expanding across several key areas. The recently launched Common Conditions Service marks a policy shift towards an enhanced role for community pharmacy in supporting improved access to care.

This Stakeholder Briefing will explore developments across community pharmacy including:
Vaccination rollout in pharmacy
Shared care records
ePrescriptions
CPA 25 developments
Introduction of Common Conditions services
There will also be an opportunity for questions from the floor.
Speaking at the event will be Susan O’Dwyer, Head of Professional Services – Irish Pharmacy Union. Visit www.thepmi.com for further information.
“Empowering pharmacists means equipping them with the competencies, recognition and working environments necessary to meet evolving health needs,” said FIP president Mr Paul Sinclair, AM. “Pharmacists are uniquely positioned at the intersection of medicines supply, scientific innovations and patient care delivery. When they are enabled to practise to the full extent of their expertise, health systems are more accessible, more responsive and more sustainable.”
FIP emphasises that empowering the profession is not optional, but essential to building stronger, future-ready systems.
World Pharmacists Day (25 September) marks the anniversary of the inception of FIP in 1912 and was adopted by the FIP Council in 2009. As such, the World Pharmacists Day campaign is led by FIP every year, with the theme chosen by the FIP Board.
In 2020, FIP also created World Pharmacy Week, extending the celebrations of the entire profession and overtly encompassing all sectors of the pharmacy profession. Any reference to “World Pharmacists Day” or “World Pharmacy Week” should, therefore, include FIP.
Wegovy® delivers quality weight loss1,2,5 and provides cardiovascular risk reduction1,3ɬ

Safety and tolerability profile comparable to the GLP-1 RA class in general1
tThis product is subject to additional monitoring. ESC = European Society of Cardiology. CCS = Chronic Coronary Syndrome. GLP-1 RA = Glucagon Like Peptide 1 Receptor Agonist.
Wegovy®t(semaglutide) Please refer to the full Summary of Product Characteristics (SmPC) before prescribing. Wegovy® 0.25 mg FlexTouch® solution for injection in pre-filled pen. Wegovy® 0.5 mg FlexTouch® solution for injection in pre-filled pen. Wegovy® 1 mg FlexTouch® solution for injection in pre-filled pen. Wegovy® 1.7 mg FlexTouch® solution for injection in pre-filled pen. Wegovy® 2.4 mg FlexTouch® solution for injection in pre-filled pen. Indication(s): Adults: Wegovy® is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management, including weight loss and weight maintenance, in adults with an initial Body Mass Index (BMI) of ≥30 kg/m2 (Obesity) or ≥27 kg/m2 to <30 kg/m2 (overweight) in the presence of at least one weight-related comorbidity e.g. dysglycaemia (prediabetes or type 2 diabetes mellitus), hypertension, dyslipidaemia, obstructive sleep apnoea or cardiovascular disease. For trial results with respect to cardiovascular risk reduction, obesity-related heart failure, and populations studied, see section 5.1. of the Wegovy® SmPC. Adolescents: Wegovy® is indicated as an adjunct to a reduced-calorie diet and increased physical activity for weight management in adolescents ages 12 years and above with obesity* and body weight above 60 kg. Treatment with Wegovy® should be discontinued and re-evaluated if adolescent patients have not reduced their BMI by at least 5% after 12 weeks on the 2.4 mg or maximum tolerated dose. *See table 1 in the Wegovy® SmPC for BMI cut-off points for obesity by sex and age. Posology and administration: Administered once weekly at any time of the day, with or without meals. Injected subcutaneously in the abdomen, in the thigh or in the upper arm. The injection site can be changed. It should not be administered intravenously or intramuscularly. For the 7.2 mg dose, inject three doses of 2.4 mg one after each other. The injections can be administered in the same body area but should be at least 5 cm apart. Injection sites should always be rotated to reduce the risk of injection site amyloid deposits. The day of weekly administration can be changed if necessary, as long as the time between doses is at least 3 days (>72 hours). After selecting a new dosing day, once-weekly dosing should be continued. Adults: The maintenance dose of semaglutide 2.4 mg once-weekly is reached by starting with a dose of 0.25 mg. To reduce the likelihood of gastrointestinal symptoms, the dose should be escalated over a 16-week period to the maintenance dose. If needed, the dose can be increased to 7.2 mg once weekly after a minimum of 4 weeks on the 2.4 mg dose in adults with BMI ≥ 30 kg/m2 at treatment initiation. If no additional clinical improvement in body weight is observed with 7.2 mg, lower the dose to 2.4 mg once weekly. In case of significant gastrointestinal symptoms, consider delaying dose escalation or lowering to the previous dose until symptoms have improved. Adolescents: For adolescents ages 12 years and above, the same dose escalation schedule as for adults should be applied. The dose should be increased until 2.4 mg (maintenance dose) or maximum tolerated dose has been reached. Weekly doses higher than 2.4 mg are not recommended in the adolescent population. Patients with type 2 diabetes: When initiating Wegovy®, consider reducing the dose of concomitantly administered insulin or insulin secretagogues (such as sulfonylureas) to reduce the risk of hypoglycaemia. Missed dose: If a dose is missed, it should be administered as soon as possible and within 5 days after the missed dose. If more than 5 days have passed, the missed dose should be skipped, and the next dose should be administered on the regularly scheduled day. If more doses are missed, reducing the starting dose for re-initiation should be considered. Elderly: No dose adjustment is required based on age. Renal impairment: No dose adjustment is required for patients with mild or moderate renal impairment. Experience in patients with severe renal impairment is limited. Semaglutide is not recommended for use in patients with severe renal impairment (eGFR <30 mL/min/1.73m2) including patients with end-stage renal disease. Hepatic impairment: No dose adjustment is required for patients with mild or moderate hepatic impairment. Experience in patients with severe hepatic impairment is limited. Semaglutide is not recommended for use in patients with severe hepatic impairment and should be used cautiously in patients with mild or moderate hepatic impairment. Paediatrics: The safety and efficacy of semaglutide in children below 12 years of age have not been established. Contraindications: Hypersensitivity to the active substance or to any of the excipients. Special warnings and precautions for use: Cases of pulmonary aspiration have been reported in patients receiving GLP-1 receptor agonists undergoing general anaesthesia or deep sedation. Therefore, the increased risk of residual gastric content due to delayed gastric emptying should be considered prior to performing procedures with general anaesthesia or deep sedation. Use of GLP-1 receptor agonists may be associated with gastrointestinal adverse reactions. This should be considered when treating patients with impaired renal function, as nausea, vomiting, and diarrhoea may cause dehydration, which in rare cases can lead to a deterioration of renal function. Patients treated with semaglutide should be advised of the potential risk of dehydration in relation to gastrointestinal side effects and take precautions to avoid fluid depletion. Acute pancreatitis has been observed with the use
of GLP-1 receptor agonists. Patients should be informed of the characteristic symptoms of acute pancreatitis. If pancreatitis is suspected, Wegovy® should be discontinued; if confirmed, Wegovy® should not be restarted. Caution should be exercised in patients with a history of pancreatitis. In the absence of other signs and symptoms of acute pancreatitis, elevations in pancreatic enzymes alone are not predictive of acute pancreatitis. Data from epidemiological studies indicates an increased risk for nonarteritic anterior ischaemic optic neuropathy (NAION) during treatment with semaglutide. There is no identified time interval for when NAION may develop following treatment start. A sudden loss of vision should lead to ophthalmological examination and treatment with semaglutide should be discontinued if NAION is confirmed. Wegovy® should not be used as a substitute for insulin in patients with type 2 diabetes. Wegovy® should not be used in combination with other GLP-1 receptor agonist products. Patients treated with Wegovy® in combination with a sulfonylurea or insulin may have an increased risk of hypoglycaemia. The risk of hypoglycaemia can be lowered by reducing the dose of sulfonylurea or insulin when initiating treatment with a GLP-1 receptor agonist. In patients with diabetic retinopathy treated with semaglutide, an increased risk of developing diabetic retinopathy complications has been observed. Patients with diabetic retinopathy using semaglutide should be monitored closely and treated according to clinical guidelines. There is no experience with Wegovy® in patients with type 2 diabetes with uncontrolled or potentially unstable diabetic retinopathy. In these patients, treatment with Wegovy® is not recommended. Semaglutide treated patients with gastroparesis may experience more serious or severe gastrointestinal adverse events. Semaglutide should be used with caution in these patients, and semaglutide is not recommended if gastroparesis is severe. The safety and efficacy of Wegovy® has not been investigated in patients treated with other products for weight management, with type 1 diabetes, with severe renal or hepatic impairment or with congestive heart failure New York Heart Association (NYHA) class IV. Use in these patients is not recommended. There is limited experience with Wegovy® in patients aged 85 years or more, with mild or moderate hepatic impairment, with inflammatory bowel disease. Use with caution in these patients. If semaglutide is used in combination with a sulfonylurea or insulin, patients should be advised to take precautions to avoid hypoglycaemia while driving and using machines. Fertility, pregnancy and lactation: Women of childbearing potential are recommended to use contraception when treated with semaglutide. There are limited data from the use of semaglutide in pregnant women. Therefore, semaglutide should not be used during pregnancy. If a patient wishes to become pregnant, or pregnancy occurs, semaglutide should be discontinued. Semaglutide should be discontinued at least 2 months before a planned pregnancy due to the long half-life. In lactating rats, semaglutide was excreted in milk. A risk to a breast-fed child cannot be excluded. Semaglutide should not be used during breast-feeding. Effect on fertility unknown. Undesirable effects: Very common (≥1/10): Headache, vomiting, diarrhoea, constipation, nausea, abdominal pain, fatigue. Common (≥1/100 to <1/10): Hypoglycaemia in patients with type 2 diabetes, dizziness, dysgeusia, dysaesthesia, diabetic retinopathy in patients with type 2 diabetes, gastritis, gastrooesophageal reflux disease, dyspepsia, eructation, flatulence, abdominal distension, cholelithiasis, hair loss, injection site reactions. Uncommon (≥1/1,000 to <1/100): Hypotension, orthostatic hypotension, increased heart rate, acute pancreatitis, delayed gastric emptying, increased amylase, increased lipase. Rare (≥1/10,000 to <1/1,000): Anaphylactic reaction, angioedema. Very rare (<1/10 000): Non-arteritic anterior ischaemic optic neuropathy (NAION). Not known (cannot be estimated from the available data): Intestinal obstruction. The SmPC should be consulted for a full list of side effects. MA number(s): Wegovy® 0.25 mg FlexTouch® EU/1/21/1608/006. Wegovy® 0.5 mg FlexTouch® (1.5 ml cartridge) EU/1/21/1608/007. Wegovy® 0.5 mg FlexTouch® (3 ml cartridge) EU/1/21/1608/012. Wegovy® 1 mg FlexTouch® EU/1/21/1608/008. Wegovy® 1.7 mg FlexTouch® EU/1/21/1608/009. Wegovy® 2.4 mg FlexTouch® EU/1/21/1608/010. Legal category: Product subject to prescription which may not be renewed. For complete prescribing information please refer to the SmPC which is available on www.medicines.ie or by email from infoireland@novonordisk.com or from the Clinical, Medical and Regulatory Department, Novo Nordisk Limited, 1st Floor, Block A, The Crescent Building, Northwood Business Park, Santry, Dublin 9, Ireland. Date last revised: February 2026. IE26SEMO00055.
tThis medicinal product is subject to additional monitoring. This will allow quick identification of new safety information. Adverse events should be reported to the Health Products Regulatory Authority. Information about adverse event reporting is available at www.hpra.ie. Adverse events should also be reported to Novo Nordisk on Tel: 01 8629700 or complaintireland@novonordisk.com.
*From baseline to week 72. Data presented here from the STEP UP trial are based on the trial product estimand, which describes the treatment effect if all people adhered to treatment, whereas the primary treatment policy estimand describes the treatment effect regardless of treatment adherence. When applying the treatment policy estimand, people treated with Wegovy® 7.2 mg achieved a superior weight loss of 18.7% vs placebo of 3.9%. The proportion of patients with a body weight reduction of ≥25% was greater with Wegovy® 7.2 mg (31.2%), vs placebo (0%).1
ɬ People living with overweight or obesity and established cardiovascular disease without diabetes.
Ŧ The co-primary endpoints were percentage change in body weight and the proportion of patients with a body weight reduction of 5% or greater for Wegovy® 7.2 mg vs placebo.1
Applying the trial product estimand, the proportion of patients with a body weight reduction of ≥5% was greater with Wegovy® 7.2 mg (93.2%), vs placebo (35.7%).1
¥Confirmatory secondary endpoint.
References: 1. Wegovy® Summary of Product Characteristics www.medicines.ie 2. Wharton S, Freitas P, Hjelmesæth J, et al. Once-weekly semaglutide 7.2 mg in adults with obesity (STEP UP): a randomised, controlled, phase 3b trial. Lancet Diabetes Endocrinol. 2025; S2213-8587(25)00226-8. 3. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and cardiovascular outcomes in obesity without diabetes. N Engl J Med. 2023;389(24):2221-2232 4. Vrints C, Andreotti F, Koskinas KC, et al. 2024 ESC Guidelines for the management of chronic coronary syndromes. Eur Heart J. 2024;45(36):3415-3537. 5. Hjelmesæth J, Bhat S, Garvey WT, et al. Effect of semaglutide on body composition and proximal muscle strength: the STEP UP trial. Presented at: The 61st European Association for the Study of Diabetes (EASD) Annual Meeting; September 15-19, 2025; Vienna, Austria.
The Irish Pharmaceutical Healthcare Association (IPHA) has published new analysis demonstrating the exceptional economic and strategic contribution of the pharmaceutical sector to Ireland, as industry leaders gathered for the IPHA Annual Conference 2026 – “Steering Through Disruption” at the Dublin Royal Convention Centre.

Against a backdrop of geopolitical uncertainty, shifting global trade dynamics and preparations for Ireland’s upcoming Presidency of the Council of the EU, the Goodbody Economic Report for IPHA shows that the pharmaceutical sector has reached record levels of employment, output, exports and tax contribution, underscoring its central role in Ireland’s and Europe’s economic resilience.
Crucially, the recent conference highlighted how this economic strength can now be matched by a system that delivers faster access to innovative medicines for Irish patients, supported by the newly concluded IPHA Framework Agreement.
The report confirms that the pharmaceutical industry is a cornerstone of Ireland’s economy,
revealing that the sector continues to outperform all historic benchmarks:
• ¤139 billion in pharma exports in 2025, representing 53% of all goods exports – the highest on record.
• Pharma exports accounted for 41% of Ireland’s GNI* in 2025, with exports to the US alone representing 25% of GNI*.
• 75,000 workers are employed directly and indirectly in pharma and related activities, with sectoral employment growing three times faster than the wider labour market. *
• The industry paid an estimated ¤6 billion in total taxes in 2023, including an estimated 18% of all corporation tax receipts. ¤4.1bn in corporation tax was paid in 2024.
Ireland is now the EU’s second largest pharmaceutical exporter and hosts the highest number of FDA registered drug manufacturing sites per capita in Europe, reflecting its global leadership in high value biologics, advanced therapeutics and complex manufacturing.
As Europe seeks to compete globally in advanced therapies, biologics and next generation medicines, the report shows that Ireland is one of the continent’s strongest and most competitive life sciences hubs, with deep expertise in high value biologics, sterile injectables and advanced therapeutics manufacturing.
A central theme of the conference was turning this economic strength into patient impact, faster, fairer, and more predictable access to innovative medicines.
The newly concluded IPHA Framework Agreement is designed to deliver precisely that:
• creating structural reforms in pricing and reimbursement so that Ireland complies with the 180 day timing for HSE decisions on reimbursement;
• enabling sustainable investment in innovation;
• supporting Ireland’s competitiveness as a location for manufacturing and R&D; and
• so that patients in Ireland can access the newest treatments at the same pace as other leading European countries.
The Agreement also aligns with Ireland’s priorities for its forthcoming EU Presidency, where regulatory efficiency, innovation pathways and Europe
wide patient access will be central policy themes.
Industry leaders emphasise that the Agreement is a directional change — shifting Ireland toward a system that values innovation, supports long term investment, and strengthens Ireland’s role as a global manufacturing and R&D hub.
Shane Ryan, IPHA President, said, “Ireland’s pharmaceutical sector is one of the most competitive anywhere in the world – powered by exceptional talent, deep expertise and a strong track record of delivery. We are committed to improving the health and wellbeing of people across Ireland, while also making a major contribution to jobs, growth and the economy. Today’s Goodbody report shows just how important our sector has become, not just for Ireland, but for Europe, and for global healthcare.
“The challenge now is to ensure patients can access innovation as quickly as we deliver it. The new Framework Agreement is a critical step forward in supporting patients in Ireland gain faster access to innovative and life-changing medicines, whilst empowering clinicians to provide the best care available. The Agreement is the result of a shared ambition and significant collaboration between Government and our sector –creating the basis for a lasting strategic partnership that backs innovation, strengthens supply chains, and, above all, improves outcomes for patients.”
IPHA Chief Executive Oliver O’Connor, added, “This report confirms that the pharmaceutical industry is one of Ireland’s greatest strategic strengths. It is an engine of stability and growth — delivering record exports, high value employment and major tax contributions — even as the global environment becomes more uncertain.
“This economic strength should clearly translate into patient benefit. The new IPHA Framework Agreement marks a decisive shift towards a system that values innovation and ensures patients in Ireland can access breakthrough medicines faster. With Ireland soon taking up the EU Presidency, we have a major opportunity to lead Europe toward a more competitive, innovation friendly, and patient focused future.”

Alan Finn, Head of Consumer Sales & Marketing at United Drug, outlines how the company is helping Irish pharmacies navigate a changing retail landscape — and what's coming up for the year ahead.


Having joined United Drug in June 2025, Alan Finn brings over 20 years of experience in the Irish healthcare sector to his role as Head of Consumer Sales & Marketing. Leading a team of 18, Alan is well versed in the evolving demands of the pharmacy market, having held a range of senior sales and marketing management roles throughout his career.
"I have always enjoyed working in the Irish healthcare industry and found pharmacy customers very welcoming and supportive," he says. It is an industry he knows intimately — and one that has undergone considerable transformation over the past two decades.
Deregulation and reductions in the medicine reimbursement scheme
have fundamentally altered how pharmacies operate, compelling many to rethink and refocus their business models. The growth of OTC, front-of-shop and vitamin categories has been central to this shift, helping stores drive footfall and boost revenue in a more competitive environment.
United Drug Consumer is well positioned to support pharmacies in sustaining this momentum. Over the years, the team has carefully cultivated a comprehensive yet focused portfolio of trusted brands — household names that Irish consumers know and reach for, including Batiste, Sterimar, Anusol, Mitchum, John Frieda, Bondi Sands, NUK, Swedish Nutra, Baylis & Harding, Sanctuary, Tom Ford and Estée Lauder.
With Irish consumers continuing to feel the pressure of rising inflation, Alan is clear about what pharmacy retailers need from their suppliers right now. "We need to provide our pharmacy customers with strong product promotions and the supporting materials to ensure they maximise sales during 2026," he explains.
Plans are already in place to expand the United Drug Consumer range further — deepening and broadening its reach across the pharmacy
channel. "We want United Drug Consumer to be recognised as an innovative company that is continuously bringing new brands to the Irish pharmacy market and supporting our customers to be more informed about our range," Alan adds.
Central to that ambition is a recognition of the time pressures faced by pharmacy staff. "Pharmacy staffs' time is limited in-store, so it's up to United Drug Consumer to make their jobs easier — by providing product information, staff training, supporting materials and promotions to drive sales and customer retention for pharmacy retailers."
In an evolved healthcare marketplace, where consumers increasingly seek expert advice, United Drug Consumer delivers value-added service through its experienced sales and product trainer team.
Meet the Team at the Cosmetics Show, May 2026
United Drug Consumer will be exhibiting at the Cosmetics Association Trade Show in May 2026, and the team are looking forward to connecting with pharmacy customers. The stand, located at E13 - E18 will showcase the Christmas catalogue alongside a full display of products from across the portfolio — giving retailers a first look at seasonal ranges and the opportunity to plan ahead for the key trading period.
The team will be on hand throughout the show for meetings, to discuss ranging options, and to place orders. If you would like to arrange a dedicated meeting time, please get in touch with your United Drug Consumer representative in advance.
For more information or to arrange a meeting at the Cosmetics Show, contact the United Drug Consumer team.

VISIT US AT THE CHRISTMAS COSMETIC & GIFT TRADE FAIR IN THE MAIN HALL, RDS, BALLSBRIDGE, DUBLIN - SUNDAY 17 TH MAY TO TUESDAY 19 TH MAY










University of Galway is strengthening its position as a key provider of healthcare education with the launch of a new MPharm programme and significant Government investment in a new School of Medicine building. Dr Martina Ní Chúlain outlines how these developments will support workforce expansion, innovation, and future models of care.


University of Galway is set to play an increasingly important role in shaping the future of healthcare education in Ireland, following the recent announcement of Government funding under the Healthcare Education Activation Programme (HEAP). The investment, part of a ¤75 million capital programme under Project Ireland 2040, will support the development of a new School of Medicine building and the continued expansion of healthcare training across the University.
A key milestone in this expansion has already been achieved, with the launch of the University’s new Master of Pharmacy (MPharm) programme. The first intake of students commenced in September 2025, marking a significant step forward for both the University and the wider region.
According to Dr Martina Ní Chúlain, Director of Strategic Development and Process Improvement, the programme represents a major achievement, not least because it was the first pharmacy programme to be delivered through the Government’s Healthcare Activation Programme.
The introduction of the MPharm programme forms part of a broader strategic response to workforce shortages across healthcare professions.
Dr Martina
Ní
Chúlain, Director of Strategic Development and Process Improvement
Dr Ní Chúlain explains that the programme aligns closely with the University’s College FutureCare Strategy (2022–2025), which was reshaped in the aftermath of the Covid-19 pandemic to address growing workforce challenges.
“Our approach has been to expand capacity where we can and to develop new programmes where there is a clear regional need and strong alignment with our strengths,” she notes.
Pharmacy was identified as a natural fit within the University’s existing education and research portfolio. The programme has been developed in close collaboration with the regional health authority, with strong input from local pharmacy teams to ensure it reflects both current practice and future workforce requirements.
A defining feature of the new MPharm programme is its focus on producing graduates who are equipped for evolving clinical roles.
Dr Ní Chúlain highlights that prescribing competencies are embedded throughout the curriculum from the outset, reflecting the growing role of pharmacists as clinical practitioners within integrated healthcare systems.
“This investment reflects the increasing recognition of pharmacists as essential members of the healthcare team,” she says. “Our programme is designed to ensure graduates are ready to contribute to patient-centred care from the very beginning of their careers.”
Students will also benefit from Ireland’s established national placement system through the APPEL model. This interinstitutional framework provides structured, high-quality clinical placements across a range of settings, including community pharmacy, hospital practice, industry, and research.
This exposure will support students in developing the practical skills and experience needed to pursue diverse and flexible career pathways within the profession.
The development of a new School of Medicine building will further strengthen the University’s capacity to deliver high-quality healthcare education.
The facility will provide dedicated teaching spaces, including rooms designed specifically for small-group learning—an essential component of modern healthcare education. It will also enable greater opportunities for interdisciplinary and interprofessional training, reflecting the collaborative nature of realworld healthcare delivery.
“Students will be trained in ways that mirror how healthcare teams operate in practice,” Dr Ní Chúlain explains, highlighting the importance of preparing graduates for integrated care environments.
Beyond teaching, the new building will support the expansion of research and innovation activities. In particular, it will enhance the University’s capabilities in areas such as digital health and connected health—fields that are expected to play an increasingly important role in the future of healthcare.
Alongside the expansion of pharmacy education, the University is also progressing initiatives aimed at addressing broader workforce challenges, particularly in rural and remote areas.
The introduction of a Rural and Remote Graduate Entry Medicine Stream is expected to play a significant role in improving access to healthcare in underserved regions.
Drawing on international best practice, including models used in Scotland and other jurisdictions, the programme is designed to train doctors in rural settings with the aim of encouraging them to build long-term careers in those communities.
Dr Ní Chúlain notes that this approach has been shown to improve retention and support more sustainable workforce planning in rural healthcare systems.
The investment under HEAP is also enabling the University to accelerate innovation in how healthcare professionals are educated.
As healthcare continues to evolve, there is a growing need for graduates who are not only clinically competent, but also adaptable, digitally literate, and comfortable working within new models of care.
“This is an opportunity to develop graduates who are digitally ready and prepared for the future of healthcare,” Dr Ní Chúlain says.
By integrating digital health, innovation, and interdisciplinary learning into its programmes, the University aims to ensure that graduates are equipped to meet the changing demands of healthcare delivery.
The combination of new programme development and infrastructure investment represents a significant step forward for University of Galway and for healthcare education in Ireland more broadly.
For pharmacy, in particular, the introduction of the MPharm programme signals a continued shift towards expanded clinical roles, greater integration within healthcare teams, and a stronger focus on patient-centred care.
As these developments take shape, they are expected to contribute not only to addressing workforce shortages, but also to enhancing the quality, accessibility, and sustainability of healthcare services across the country.























































FOR DRY EYES
Expertly developed eye care solutions with DIFFERENT THERAPEUTIC STRENGTHS of SODIUM HYALURONATE available as PRESERVATIVE-FREE & PRESERVED lubricating eye drops to cover the spectrum of DRY EYE severities and patient needs





A SPECIALIST RANGE OF EYE CARE TREATMENTS FOR DRY EYES & LIFESTYLE EYE CONDITIONS

FOR LIFESTYLE
EYE CONDITIONS
PRESERVATIVE-FREE LIFESTYLE positioned eye care solutions created to attract new customers to the category

FOR PRICE ADVANTAGE FOR YOU AND YOUR PATIENTS







In recent years it has become increasingly apparent in clinical practice that the hay fever season is no longer confined to the traditional late spring and summer months. Patients are presenting earlier in the year, often in late winter or early spring, with symptoms that may initially be mistaken for recurrent viral upper respiratory infections. Early recognition of seasonal allergic rhinitis is therefore important in order to initiate timely and effective management.

The timing and intensity of pollen seasons are strongly influenced by prevailing weather conditions. Following the exceptionally wet winter experienced across Ireland this year, airborne pollen levels have remained relatively suppressed during the colder months. However, this does not necessarily indicate that the upcoming season will be mild. As weather conditions improve and become milder and drier, pollen release may increase more rapidly.
Early-season tree pollens are typically the first allergens to appear, including species such as birch, which is highly allergenic and increasingly prevalent in Ireland. Grass pollen remains the dominant allergen and is responsible for the majority of moderate-to-severe allergic rhinitis symptoms. In Ireland, the grass pollen season typically peaks between May and July. This period often coincides with the examination season for many secondary school and university students, during which allergic symptoms may significantly impair sleep quality, concentration and academic performance.
There is also growing clinical recognition that the pollen season may be lengthening, with earlier onset and more prolonged periods of symptom burden. While the underlying drivers are multifactorial, including environmental and climatic influences, the practical implication
Written by Dr Iseult Sheehan, Clinical Director, Allergy Ireland, MSc (Allergy), AFHEA, MICGP, FRACGP, MB, BCh, BAO, LRCP&SI, BA (Hons) Biochemistry & Immunology
is that patients may present earlier in the year and may require treatment for longer durations than previously observed.
Clinical Burden and Associated Conditions
Although hay fever is sometimes perceived as a relatively minor condition, the clinical burden can be considerable. Patients frequently present with the classical symptoms of sneezing, rhinorrhoea, nasal congestion and ocular irritation. However, the broader impact of allergic rhinitis is often underestimated. Disturbed sleep, daytime fatigue, impaired concentration and reduced productivity at work or school are common and may significantly affect quality of life.
An important patient group to identify is those with co-existing asthma. Approximately one in ten individuals in Ireland have asthma and an estimated 80-90% of these patients also experience concomitant allergic rhinitis. Allergic rhinitis and asthma represent a continuum of airway inflammation with inadequate control of upper airway disease being associated with poor asthma control and increased risk of exacerbations. Patients presenting with cough, wheeze or breathlessness during the pollen season should therefore prompt consideration for medical review.
Another common scenario encountered in community practice is the undertreated patient. Many individuals rely solely on oral antihistamines each year despite persistent symptoms. While antihistamines can be effective for sneezing, itching and ocular symptoms, they provide limited
benefit for nasal congestion and sinus symptoms.
Optimising Pharmacological Management
Intranasal corticosteroids remain the most effective first-line pharmacological therapy for moderate-to-severe allergic rhinitis. These agents act directly on the underlying inflammatory process and are effective in improving the full spectrum of nasal symptoms, including congestion. Despite their well-established efficacy, intranasal corticosteroids are frequently initiated too late in the season, used inconsistently or discontinued prematurely.
Pharmacists therefore have an important role in advising patients that these treatments are preventative anti-inflammatory therapies rather than immediate symptom relievers. When commenced early and used regularly throughout the pollen season, they can significantly improve symptom control.
Correct administration technique is also essential for optimal efficacy and tolerability. Spraying directly onto the nasal septum may increase the likelihood of irritation or epistaxis and reduce therapeutic effectiveness. Patients should be advised to angle the spray slightly outward, away from the nasal septum.
Combination intranasal sprays containing both an antihistamine and corticosteroid may provide additional benefit in patients with persistent or more severe symptoms and are increasingly utilised in specialist practice.
Prevention and Adjunctive Measures
Early intervention remains an important component of effective hay fever management. Patients with a known history of seasonal allergic rhinitis should ideally commence intranasal corticosteroid therapy one to two weeks prior to the expected onset of symptoms and often in combination with an oral antihistamine. In Ireland, this frequently corresponds to early spring, particularly for individuals sensitised to tree pollens.
Adjunctive measures may also contribute to improved symptom control. Saline nasal irrigation can assist in reducing allergen load within the nasal passages. Additionally, practical measures such as keeping windows closed during high pollen periods, wearing wrap-around sunglasses, showering after outdoor exposure and limiting outdoor activities during peak pollen times may also be beneficial.
Topical nasal decongestants may be used for short periods during acute exacerbations or when concurrent infection is present, however, patients should be advised to limit use to a maximum of five to seven days to avoid the development of rhinitis medicamentosa. Smoking cessation should also be encouraged where relevant, as tobacco smoke may exacerbate airway inflammation and worsen respiratory symptoms. Patients should also be advised that vaping is not a benign alternative, as exposure to aerosolised chemicals can similarly irritate the respiratory mucosa and may aggravate allergic and airway symptoms.
While the majority of patients can be effectively managed within the community setting, referral for further assessment should be considered where symptoms remain poorly controlled despite appropriate therapy, where there is significant impairment of daily functioning and where co-existing asthma is present.
Allergen immunotherapy represents an established treatment option for selected patients with confirmed allergic rhinitis. In Ireland, sublingual immunotherapy is available for grass pollen, tree pollen and house dust mite allergy. Grass pollen preparations are currently included under the Drugs Payment Scheme. Treatment courses typically continue for three to five years, although clinically meaningful improvement is often observed within the first 12-18 months. Importantly, allergen immunotherapy is diseasemodifying and may provide sustained long-term benefit beyond symptomatic relief.

Always read the leaflet.
Abbreviated prescribing information

As summer comes to a close, there is always a lot to be done to get kids ready to go back to school. Returning to
schools being fully open after the Covid-19 lockdown is going to be hard, and harder still to try and predict what it will look like.

When children do return it will be to a very different experience than before and will carry greater complexities. However what will remain the same is the health and care of school children as they re-integrate with their peers and continue with their lifelong learning.
This special feature takes a closer look at some of the most common presenting issues, ranging from headlice to hayfever and from injuries to skincare.
Parents can help their kids have a great school year by making sure they are healthy and ready to learn – because healthy students are better learners.
However, getting kids ready to start the school year involves more than finding the perfect backpack, buying school supplies, and shopping for new clothes. It means safeguarding their health so they’ll be physically ready for the challenges.
For community pharmacists, the event leads to opportunities for effective front of shop category management. Now is the time for retail pharmacies to get ready for
the onslaught of pharmacy needs that crop up among their younger patients as they return to school.
Children, ‘tweens, and teens cannot be underestimated, and need to be treated with importance. Pharmacists have a key role to play in advising the younger generation on how to take medications and how to stay away from toxic medications.
Back to school is an opportune time to enhance that discussion.
Providing customers with easy to navigate back-to-school sections in community pharmacy, with relevant point of sale and prominent window displays during August and September, will help remind parents and carers that preparation is key when it comes to the management of back-toschool self-treatable conditions.
In addition, many parents worry about whether they should keep their children at home or send them to school when they are ill. Pharmacy teams should be prepared to ask appropriate questions and offer advice to help them to make that decision. For
example, does the child seem well enough to carry out the usual day-to-day activities they would at school, or is their condition serious or contagious enough to be passed on to classmates or teachers? Helping people to make such decisions will add to their self care skills and knowledge for managing coughs and colds.
Highlighting the availability of products, giving advice for back to school ailments and communicating the principles of self care to parents is an important service that community pharmacy offers families as the first port of call.
From head lice and rotavirus, to stomach upsets and cold/ flu symptoms, there are many self-treatable conditions that can hit families hard when September comes. Consider these commonly presented concerns when thinking about your front of shop and category management within the community pharmacy.
Managing Allergies
The sun may still be out, and September historically sees some
of our sunniest weather, so children will be out playing sports during school. This increases the risk of not only sports-related injuries but also exposure to allergens.
Nasal allergy in children, if unrecognised or badly managed, can have a significant toll on emotional as well as physical well-being.
Kids allergies can leave sufferers incredibly troubled when it comes to getting through the school day.
Itchy eczematous skin, snuffly and irritable noses and wheezy chests, carry a significant burden of ill health. Dr Paul Carson of the Slievemore Clinic Ireland says, “If the nasal allergy is especially troublesome the child may get intermittent hearing loss. One day he’s bright and alert in class, inter-reacting and co-operating. Next day he seems distant and detached, ignoring questions or not fully grasping what’s going on. The teachers are at a loss to explain these variations in attentiveness and the boy’s parents can’t quite understand the situation either. It’s not uncommon for these children to be labelled ‘difficult’.
“Nasal allergy also provokes intense fatigue. If an affected child is not treated he misses out on ordinary children’s activities and can be isolated and ignored.
“The term Allergic Irritability Syndrome (ARS) has been coined to explain the many unpleasant symptoms and features children with untreated nasal allergy show.” Children with un-recognised ARS have:
• A significantly impaired quality of life.
• Significant learning difficulties.
• A lower ability to achieve different types of knowledge (factual, conceptual and knowledge application) compared with healthy children.
• May suffer sleep apnoea, snoring and disturbed sleep pattern. This in turn leads to daytime drowsiness, grumpy mood and poor school performance.









Fitzgerald Power is the leading financial advisor to the Irish community pharmacy sector and the leading pharmacy sales brokerage firm in Ireland.
Get in touch today with our specialist advisor y team and we will help you really understand what’s financially driving your business.
We can help you with:
Financial Health Checks
Pharmacy Valuation
Pharmacy Sale Brokerage
Due Diligence & Fundraising
Tax & Compliance
www.fitzgeraldpower.ie
Management Accounts Payroll
Year End Accounts & Audit
Book Keeping
Company Secretarial Ser vices

• In severe cases it may cause or at least contribute to attention deficit hyper-activity disorder (ADHD).
• May cause impaired hearing if fluid collects in the inner ear (medical term: serous otitis media).
• May suffer repeated ‘head colds’ that go down to the chest (which is really an untreated nose and sinus allergy triggering early asthma).
• Nasal blockage and irritation (sneezing, rubbing at the nose to relieve itch); dark circles around the eyes with puffiness of the lower lids.
• Poor concentration; disruptive behaviour and unexplained mood swings.
Below are the common effects of persisting nasal allergy in children.
Nasal Congestion
Sometimes a child’s nose is congested (obstructed) to the point that he or she breathes through the mouth, especially while sleeping.
If the congestion is left untreated this forces air currents through the mouth. The strength of the air changes the way the soft bones of the face grow. The features may become abnormally elongated in a pattern called ‘adenoidal face’. This causes the teeth to come in at an improper angle as well as creating an overbite. Braces or other dental treatments may be necessary to correct these problems.
Nasal allergy and ear infections
Nasal allergy can lead to inflammation in the ear and may cause fluid accumulation which in turn can trigger ear infections and decreased hearing. If this happens when the child is learning to talk, poor speech development may result. Hayfever can also cause earaches and ear itching, popping and fullness (‘stuffed up ears’).
Nasal allergy while at school Nasal allergy can last throughout the school year. For some kids this means absences due to allergy flares. Here are some of the problems to look out for so that the condition can be properly diagnosed and treated, as well as several suggestions for helping the allergic child.
• Dust irritation: reducing dust in the home will be helpful to most allergic family members. At school, children with allergy problems should sit away from the blackboards to avoid irritation from chalk dust.
• School pets: furry animals in school may cause problems for allergic children. If your child has more problems while at school, it could be the class pet.
• Asthma and physical education: sports are a big part of the school day. Having asthma does not mean eliminating these activities. Often medication administered by using an inhaler is prescribed before exercise to control their symptoms. Children with asthma
and other allergic diseases should be able to participate in any sport the child chooses –provided the doctor’s advice is followed.
• Dry air: with the onset of cold weather using a humidifier to accompany forced air heating systems may be helpful in some regions of the country. Adding a small amount of moisture to dry air makes breathing easier for most people. However, care should be taken not to allow the humidity above 40%, which promotes the growth of dust mites and mould.
• Change in behaviour: children cannot always vocalise their
annoying or painful symptoms. Their discomfort may manifest as behaviour problems. Be on the alert for possible allergies if your child has bouts of irritability, temper tantrums or decreased ability to concentrate in school. These may be signs of ‘allergic irritability syndrome’ caused by nose and ear symptoms in allergic children. Sometimes allergic children are badly behaved and have short attention spans. Needless to say their schoolwork suffers. When a child’s allergies are properly treated, his symptoms, behaviour and school performance can improve dramatically.

The Irish Pharmacy Union (IPU) has welcomed a new agreement that will enable community pharmacies across Ireland to provide enhanced medicines optimisation supports to those with the greatest clinical need. The agreement focuses on delivering practical, clinically appropriate supports, including access to medicines optimisation supports, which may include blister packs, where required.
Welcoming the agreement, IPU
President Tom Murray said,
“Pharmacists use their expertise every day to support those managing complex medicines, living with cognitive or physical impairments, or facing significant social challenges. This agreement strengthens our ability to protect vulnerable patients while supporting the safe, effective and evidence-based use of medicines.
“This marks a significant development for patients and for the wider health service. It puts in place a robust, structured support
that will improve how medicines are managed in the community while building on the ongoing expansion of the role of pharmacists.”
Patients who are not eligible under this new service for these supports may still choose to avail of medicines optimisation supports including blister packing services through their pharmacy, where this is offered, for an agreed fee, as a private service.
The IPU has said they look forward to continued engagement with the Department of Health and
the HSE on the implementation of this agreement.
Under the agreement, medicines optimisation supports may be provided under the GMS Scheme where appropriate, to the following:
• Patients who are on high-risk medicines and are at risk of misuse and/or abuse of those medicines.
• Patients with physical impairment affecting the ability to use conventional packaging (who do not have carers that could support them to use original packs).
• Patients diagnosed with cognitive impairment or dementia who have carers to support them (including home care) by prompting them to take their medications at the appropriate intervals.
• Patients with an intellectual disability.
• Patients who, for social inclusion reasons, including homeless or marginalised people, are unable to appropriately manage their medication.




The trusted name behind Ireland’s leading ostomy and urology care service has a new look — and a renewed commitment to the patients and pharmacy professionals who depend on it every day.
United Drug Wholesale is pleased to announce that its Ostomy and Urology Care business unit has been rebranded as UroStomaCare. The new name and identity reflect a sharper focus on the specialist care that patients with stoma and continence care needs require — while preserving every element of the exceptional service that pharmacy partners across Ireland have come to rely on.
Same great service, powerful new identity.
Stoma and continence care patients face deeply personal healthcare challenges. The rebrand to UroStomaCare signals a dedicated, specialist offering — one that goes beyond product supply to encompass clinical support, patient education, and genuine quality of life improvements. For community pharmacists, this means a more intuitive, clearly positioned service to refer patients to, and a stronger partnership to lean on.
UroStomaCare is Ireland’s number one provider of stoma and continence care products and services. The breadth and depth of what is on offer is unmatched nationally:
• With over 3,000 products sourced from 35 leading manufacturers, ensuring pharmacists can meet the needs of every patient.
• State-of-the-art cutting technology with two dedicated cutting machines, enabling complimentary custom bag cutting at an average of 10,000 bags per week.
• 48-hour turnaround on emergency orders, giving pharmacy teams and patients the confidence that critical supplies will always arrive when needed.
• A dedicated Customer Care team providing responsive, expert support to pharmacy professionals.
• Access to Stoma Nurses across Ireland, offering patients clinical guidance and reassurance beyond the pharmacy counter.
• Supplier partnerships that unlock in-store training for pharmacy staff, online educational resources, and one-to-one Quality of Life consultations with patients — supporting both clinical outcomes and business development for pharmacy partners.
Introducing the UroStomaCare Hub on UDW.ie
Alongside the rebrand, a new UroStomaCare Hub is now live on UDW.ie. Designed with pharmacy professionals in mind, the hub brings together in one place:
• Comprehensive product catalogues
• Application guides for stoma and continence care products
• Educational resources for pharmacy staff
• Clear referral pathways to help pharmacists connect patients with the right support
The hub is designed to reduce the burden on busy pharmacy teams — making it quicker and easier to find the right product, access training, and refer patients appropriately.
The Same Commitment, A Stronger Platform
The launch of UroStomaCare represents more than a name change. It is a reaffirmation of the commitment to delivering the best in customer care, product range, service, and training to the pharmacy community across Ireland. For patients living with a stoma or continence care needs, the rebrand means continued access to Ireland’s most trusted, most comprehensive care service — now under a name that better reflects its specialist focus. Pharmacy professionals interested in learning more, accessing the new UroStomaCare Hub, or exploring training opportunities for their team are encouraged to visit UDW.ie today, or contact the customer care team on 01 463 2300.
Researchers from RCSI University of Medicine and Health Sciences recently came together for RCSI Research Day 2026, showcasing the breadth of research underway across the University. The event provides an opportunity for researchers at all career stages to share new findings and connect with colleagues across the RCSI community.

Through presentations, posters and the Research and Innovation Showcase session, the programme highlights outstanding contributions from RCSI researchers and the worldclass research that is driving improvements in human health. Selected presentations from undergraduate, early career and postgraduate researchers explored a range of topics across three themed sessions including physiotherapy, nursing, immunology, drug delivery, neurosciences, cancer and biomaterials.
This year’s distinguished speaker for the John J. Ryan Lecture was Professor Margaret A. PericakVance, Director of the John P.
Hussman Institute for Human Genomics and Executive Vice Chair of the Dr John T. Macdonald Foundation Department of Human Genetics at the University of Miami Miller School of Medicine. She will deliver a lecture titled 'The Global Genetics of Alzheimer’s Disease: Pathway to Prevention and Treatment'.
Professor Pericak-Vance is a world-renowned human geneticist and author of more than 750 peer-reviewed publications, cited over 115,000 times. Her work has made a major contribution to understanding genetic risk factors for Alzheimer’s disease, as well as conditions including autism, age-related macular degeneration, glaucoma and multiple sclerosis.
Introducing UroStomaCare Ireland’s No. 1 Provider of Stoma & Continence Care Products
Cutting an average of 10,000 bags per week Over 3000 products
Dedicated customer care team
Sourcing from over 35 leading manufacturers
48 hour emergency UroStomaCare Hub on UDW.ie

The bacteria in a child’s gut plays an important role in keeping them healthy, both mentally and physically
Gut health has become a hot topic in recent years and the discussion is usually about gut microbiome which are the trillions of bacteria that live inside our gut. These bacteria play a key role in keeping children mentally and physically healthy and also reduce the risk of illness and disease.
Symptoms of a poorly functioning gut include uncomfortable digestion, stomach aches, poor nutrient absorption, poor immunity and sleep, mood fluctuations and general fatigue. All can have a detrimental impact on children. The gut microbiome is established in the early years of life. It’s particularly important to support a child’s gut microbiome to thrive while they’re growing. In fact, it is believed that before the age of four or five children’s microbiome remains flexible. This is a great time to build a strong and healthy gut health in children. Beyond this age the microbiome is harder to change as it becomes well established. Everyone’s microbiome is unique and, interestingly, diet can be responsible for up to 75% of this variation!
Elimination of Waste
The gut removes waste from the body. This is important to help support liver function. So, to keep a child’s bowel movements regular (and prevent uncomfortable constipation), encourage a diet with plenty of fibre. For instance, include wholegrains, veggies and fruit in their diet. This will keep the bowels regular and also feed the healthy gut bacteria. Water is also important to help keep bowel movements soft and regular.
The gut is home to 70% of the body’s immune system. It is also the main entry point for bugs to get into the body. Therefore, if a child’s gut is not a strong barrier they will be more likely to pick up nasty bugs. This can lead to illness. Research suggests that a poor balance of bacteria in the gut during early childhood can increase risk of gut-related allergies like asthma and eczema. Advise parents that they can help build their child’s immunity by fuelling the good bacteria in their gut. This will, in turn, keep children feeling energetic and healthy.
The Gut-Brain Axis
The Gut-Brain Axis is the physical and chemical connection between the gut and brain. To clarify, it is a network of millions of nerves sending messages both ways. Chemicals called neurotransmitters, such as serotonin and GABA (which controls feelings of fear and anxiety), are sent to and from the brain. Many of these are produced in the gut. A thriving, healthy gut microbiome contributes to production of chemicals and signalling messages for good mental and physical health in children.
Probiotics are the “healthy” bacteria that make up the gut microbiome. Probiotics work to maintain health and immunity. They also fight inflammation and disease. Parents can boost the good bacteria in a child’s gut by feeding them probiotic rich foods. Good bacteria can help to form a strong, healthy microbiome. A probiotic supplement is also an option.
Researchers are still learning about all the benefits of probiotics and how they work. Probiotics may help with digestion, preventing infectious diseases and even playing a role in maintaining a healthy weight.
Some studies say probiotics help children with digestive issues such as:
• Constipation
• Inflammatory bowel disease
• Irritable bowel syndrome
• Infectious diarrhea
Though it is unclear if probiotics benefit children, it's clear they typically won't cause harm in healthy children. However, pharmacy teams should recommend parents wait until a child is over 1 year of age before giving probiotics.

Children who are seriously ill or who have a compromised immune system should consult their GP before taking probiotics. Some studies suggest that children with a central line or port should also avoid probiotics. In children with these medical devices, there have been reported cases of sepsis.
Fibre
A child’s diet should contain fibre as an essential component. It is required to achieve good gut health in children and different fibres help the gut in their own unique ways.
While we often think of fibre as a singular nutrient there are actually many different types, for example:
• Insoluble fibre: Wholegrain breads and cereals and the skin of fruit and vegetables help to soften bowel content, promoting regular (and comfortable) bowel movements.
• Resistant starch: This is a type of fibre which is not digested as normal by the body. Resistant starch is found in wholegrain cereals, legumes and starchy vegetables like potato. It travels to the large intestine where it feeds good bacteria in the gut. These good bacteria produce what you need for a healthy digestive system and protection against disease.
• Prebiotics: These are another type of fibre which also feed the friendly bacteria in the gut. This provides another nourishing food source to the healthy bacteria in your gut. The great news is prebiotics are found in foods that are very child-friendly and delicious. Think bananas, apples and oats plus foods that are easy to sneak into meals like barley, onion or flaxseed.
• Soluble fibre: These help to slow the emptying process in the stomach, which can help children to feel fuller for longer. Soluble fibre is found in fruits, vegetables, oats, barley and legumes.
E45 cream is used for treating dry skin conditions such as dry/flaky skin, eczema, dry psoriasis, dermatitis and sunburn. Suitable for the whole family. Apply 2-3 times daily for best results*.


I was thirteen when I first heard the words Crohn’s Disease. At an age when most of my friends were worrying about school exams and general adolescent issues, I was learning how to navigate scopes, steroids, and the fear that my body had suddenly turned unpredictable. What I didn’t realise then was that Crohn’s would shape not just my health, but my entire life - from the work I do today to the way I understand resilience, community, and compassion.

Over two decades later, I live with a permanent ileostomy after years of exhausting every possible treatment. My stoma, lovingly nicknamed Guiney, gave me back the life that Crohn’s had stolen. But as anyone with IBD or a stoma will tell you, the medical procedures and medications are just one part of the story. The practical, emotional, and day-to-day realities shape your world just as powerfully.
And throughout every chapter - the flares, the surgeries, the setbacks, the wins - one person outside my immediate family has remained absolutely constant: my community pharmacist.
My pharmacist Sheila has known me since childhood. She has watched me grow from a scared teen picking up liquid diets and steroids, to an adult navigating complex long-term immunosuppressants, to now, a woman living confidently with a stoma. Even after moving location, I never changed pharmacy. That continuity mattered. She mattered.
At a time when so much in my health felt unstable, she offered a rare sense of stability - someone who didn’t need the shorthand, who always remembered the small
details, and who saw me as more than a diagnosis.
One of the most meaningful examples happened only recently. A change had been made to the type of stoma bags dispensed for me. Instead of simply processing the prescription and moving on, she picked up the phone to check whether I actually knew about the change. She wanted to make sure it wasn’t an error, that I wouldn’t be caught off guard, and most importantly, that I felt comfortable and supported.
That phone call may seem small, but to someone living with a stoma, it is huge. The wrong product, or an unexpected change, can mean leaks, skin damage, or hours of distress. Her attention to detail wasn’t just professional; it was deeply human.
Living With a Stoma: The Realities Pharmacists Help Ease
People often think that once you have a stoma, the surgery is the end of the story. But in truth, it’s the beginning of a new way of living. You learn what products work, how your skin behaves, how your output changes with stress or diet, and how important reliable, accessible, knowledgeable support becomes.
Community pharmacists play a quiet but powerful role in this.
By Victoria Spillane, interim Chief Operations Officer, Crohn’s and Colitis Ireland
dietitians, mental health supports - they all matter deeply.
But pharmacists are often the unsung members of that village. They see us far more frequently than specialists. They see us on the days we’re exhausted, anxious, sore, and stretched thin. And they are in the perfect position to offer practical advice, early guidance, and simple kindness - the things you can’t always schedule into a clinic appointment.
If I could share one message with pharmacists and pharmacy teams, it’s this:
“Your role is profound. Please never underestimate it!”
They are often the first to spot patterns - repeated requests for barrier sprays, more frequent prescriptions for pain relief, or questions that hint someone may be struggling emotionally as much as physically.
For me, my pharmacist has been that safety net. She has:
• Explained medication adjustments in plain, practical language
• Flagged interactions and helped me avoid unnecessary flares
• Reassured me through difficult treatment transitions
• Helped navigate supply challenges - a real issue for stoma products
• Treated every question, no matter how sensitive, with dignity
Those moments accumulate. They build trust. They make living with a chronic illness not just manageable, but less lonely.
Community Pharmacy as Part of the Village
In my advocacy work, I often talk about the idea that “It Takes a Village” to support someone living with IBD or a stoma. Surgeons, gastroenterologists, IBD nurses, stoma nurses,
People with IBD or stomas may look “well” on the outside. We may collect our prescriptions quickly, or seem quiet, or appear rushed. But many of us are carrying years, sometimes decades, of pain, fear, fatigue, and resilience.
The reassurance you give, the questions you ask, the care you take with products and prescriptions, and the moments when you truly see us… they make an enormous difference.
Community pharmacy is often the first place we turn when something feels off, and the last place we leave when we need to feel understood.
My IBD journey has been long and winding, filled with challenges I never expected and strengths I never knew I had. But I’ve never walked it alone. And my pharmacist, the same one who supported me as a frightened teenager, has been one of the most constant, compassionate figures throughout it all.
She embodies what community pharmacy can and should be: knowledgeable, reliable, proactive, and deeply human.
It truly takes a village. And I am profoundly grateful that she has always been part of mine.






Vocalzone Throat Pastilles are a unique blend of powerful ingredients formulated to provide effective relief from throat irritation caused by singing or talking. They may also help soothe sore throats during a common cold. Available in 3 flavours: Orginal, Honey & Lemon and Blackcurrant. Gluten free. Lactose free. Suitable for Vegetarians and Vegans. Contact: ask@pamex.ie



As BowelScreen expands and demand on services grows, pharmacists are uniquely positioned to influence participation, address misconceptions, and support both prevention and early detection—particularly through the programme’s ability to identify and remove pre-cancerous abnormalities before they develop into cancer—but challenges remain.
Bowel cancer is one of the most common cancers in Ireland, but screening plays a powerful role in both preventing cancer through the removal of pre-cancerous abnormalities and detecting it at an early stage when outcomes are significantly improved.
BowelScreen, our national bowel screening programme has made significant progress in reducing mortality by identifying cancers at an earlier stage and preventing disease through the removal of abnormal growths. Yet despite its proven clinical and economic benefits, participation remains stubbornly below target.
This gap between availability and uptake is now one of the most pressing challenges facing the programme. With planned expansion to wider age groups and increasing demand across the health system, attention is turning to how participation can be improved—and critically, who is best placed to influence it.
Community pharmacy, with its accessibility, trust, and frequency of patient interaction, is increasingly being recognised as a key part of the solution.
The Current Landscape: Progress and Pressure
BowelScreen currently offers free screening every two years to men

and women aged 57 to 71 as of April 1st, 2026. The programme is designed primarily to prevent bowel cancer developing through the identification and removal of polyps which, if left untreated, can progress to cancer over time. It can also detect cancer at an earlier stage, when treatment is more effective.
Each year, around 3,000 people undergo removal of pre-cancerous growths following screening— representing thousands of cancers that may never develop. When cancers are detected through the programme, they are more likely to be identified at an earlier stage, significantly improving treatment outcomes.
However, participation remains a major concern. As of 2022-2023 (latest available data), uptake stood at 46.4%, well below the programme’s achievable target of 60%. This represents a substantial missed opportunity for early intervention. Increasing uptake is essential if screening is to deliver its full benefit in reducing cancer burden and improving outcomes.
At the same time, expansion of the programme is expected to significantly increase demand.
HIQA analysis indicates that extending screening to younger age groups could result in up to 219,000 additional invitations annually. This will have implications not only for primary care, but also for downstream services such as endoscopy, histopathology, and radiology.
Improving screening uptake is a complex challenge, and international evidence offers valuable insights into what can influence participation.
HIQA recently examined international evidence relating to screening uptake in the context of a Health Technology Assessment (HTA) examining potential further age expansion
Máirín Ryan, Deputy Chief Executive and Director of Health Technology Assessment at HIQA
“Improving participation is essential if we are to maximise the benefits of early detection and prevention that screening programmes can offer.”
of the BowelScreen programme. Specifically, HIQA were requested by the National Screening Advisory Committee to undertake a HTA of extending BowelScreen to include those aged 50 to 54 years. This HTA was delivered to the National Screening Advisory Committee in December 2025.
HIQA analysis indicates that extending screening to those aged 50 to 54 could result in up to 219,000 additional invitations annually. This will have implications not only for primary care, but also for downstream services such as endoscopy, histopathology, and radiology.
According to HIQA, a range of interventions have been shown to improve engagement. These include enhanced communication strategies, behavioural scienceinformed messaging, and alternative delivery models.
Digital engagement is also playing an increasing role. In Ireland, BowelScreen uses a combination of traditional, digital and social media campaigns to raise awareness and encourage participation, with further national campaigns planned. Alongside this, targeted messaging— particularly for younger eligible populations—has shown potential in supporting awareness and engagement, including among individuals aged 50 to 54.
In Ireland, the BowelScreen programme has trialled the FIT Direct approach, whereby test kits are sent directly to eligible individuals without requiring them to opt in. This significantly increased participation rates,
particularly among those who had not responded to previous invitations.
However, this approach also highlighted operational challenges. While uptake improved, there was a corresponding increase in unsatisfactory samples, demonstrating the importance of balancing accessibility with quality and usability.
Behavioural science is now being incorporated more systematically into programme design. The “Better Letters” initiative, developed by the National Screening Service, aims to improve uptake by redesigning invitation materials to better align with how people respond to information.
Marketing campaigns have also demonstrated impact. A 2023 BowelScreen campaign linked to the lowering of the screening age resulted in a 279% increase in registrations during the campaign period, highlighting the importance of sustained public engagement.
Despite these efforts, participation continues to be shaped by a complex interplay of emotional, psychological, and practical factors. HIQA’s assessment noted that many barriers are rooted in patient perception rather than system design. Feelings of embarrassment or discomfort associated with stool-based testing remain common, while fear—both of a cancer diagnosis and of follow-up procedures—can deter participation.
A super supplement with 10 active ingredients designed to support your skin, hair and nails from within.
Agree their skin is glowing and more radiant 93%
Agree their hair feels stronger 82% Would recommend to others 96%
*Based off a 2026 survey of 45 participants after taking the product for 90 days.


Misunderstanding is another key issue. Many individuals incorrectly believe that screening is only relevant if they are experiencing symptoms or have a family history of cancer. This misunderstanding undermines the fundamental purpose of screening, which is to detect disease before symptoms appear. Broader social determinants further influence participation. Lower uptake is associated with socioeconomic deprivation, rural residence, and certain ethnic backgrounds, reflecting wider inequalities in access to healthcare services.
Vulnerable populations—including individuals with intellectual disabilities, mental health conditions, or unstable housing— face additional barriers. These may include difficulties receiving invitations, understanding screening information, or attending appointments.
Gender differences are also evident, with men consistently less likely to participate in screening programmes than women.
Addressing these barriers requires a multifaceted approach, combining system-level interventions with personalised engagement.
Pharmacy: A Unique Point of Influence
Within this context, community pharmacy offers a unique opportunity to influence behaviour.
HIQA’s findings highlight the scale of engagement within the pharmacy setting. A 2024 study found that 88% of the population had visited a pharmacy in the previous month, and more than half within the past week. This frequency of contact is unmatched by most other healthcare settings.

HIQA noted that this accessibility positions community pharmacy as a valuable platform for opportunistic health promotion. Evidence supports this potential. A small Irish pilot study found that pharmacist-assisted enrolment increased uptake to 73.9%, compared with 38% in the general population. While further research is needed to confirm these findings at scale, they highlight the potential impact of pharmacybased interventions.
The Community Pharmacy Agreement 2025 reflects this growing recognition. The agreement includes provisions for pharmacist involvement in promoting BowelScreen enrolment, with financial support for this activity.
The HSE has confirmed that implementation will begin in 2026, starting with training and health promotion initiatives. Pharmacists will play a role in encouraging participation and supporting registration, while the HSE will retain responsibility for issuing and processing FIT kits.
Frontline Reality: Opportunity Meets Constraint
While the strategic case for pharmacy involvement is clear, successful implementation will depend on practical considerations and effective system design.
Ian Clarke, Superintendent Pharmacist at Clarke’s Pharmacy in Galway, welcomes the recognition of pharmacy’s role and the opportunity this presents for the profession.
“There is no other profession that is as accessible to the public as community pharmacy,” he says.
“It is very encouraging to see this recognised at policy level, and that there is a move towards appropriately supporting and resourcing these services.”
He notes that with the right structures in place, pharmacy is well positioned to play a meaningful role in supporting screening initiatives.
“As with any new service, ensuring that processes are efficient and well-designed will be key. If implemented effectively, this has the potential to deliver real benefits for patients while making best use of the accessibility and expertise within community pharmacy.”
“The uptake rate represents the largest gap in the screening service at present—we need to focus on the quick wins that will drive participation.” - Ian Clarke
This reflects a broader tension between policy ambition and operational capacity. For pharmacy involvement to be sustainable, processes will need to be streamlined and aligned with existing workflows.
Improving uptake will require both broad awareness campaigns and targeted, local engagement. National campaigns have demonstrated their effectiveness, but sustained visibility is key. Clarke suggests that partnerships with organisations such as the GAA could help maintain public awareness and reach key demographics, particularly men.
At a local level, pharmacy teams can integrate screening into everyday interactions. Linking bowel screening to other preventative services—such as flu vaccination or blood pressure monitoring—can help normalise the conversation and reduce stigma.
Correcting Misconceptions and Supporting Patients
Pharmacists are well placed to address common misconceptions.
“A lot of patients think screening is only for people with symptoms or a family history,” Ian explains. “There can also be fear of an invasive procedure, when in reality it’s a simple at-home test.”
Máirín Ryan, Deputy Chief Executive and Director of Health Technology Assessment at HIQA, emphasises that clear communication is essential. Screening is designed for individuals without symptoms, and a positive result does not mean a cancer diagnosis—it simply indicates that further investigation is required.
While screening targets asymptomatic individuals, pharmacists must remain vigilant for red flag symptoms.
These include persistent changes in bowel habits, blood in the stool, unexplained weight loss, fatigue, and abdominal discomfort.
Appropriate referral remains essential, ensuring that symptomatic patients are directed to further investigation without delay.
Preparing for the Future
Looking ahead, bowel screening is likely to evolve.
Emerging research is exploring new approaches, including riskbased screening models and less invasive testing methods such as blood-based biomarkers. While these innovations are not yet part of national programmes, they may shape future strategies.
At the same time, increased participation will place additional demands on healthcare services, highlighting the need for coordinated planning across the system.
Improving bowel screening uptake in Ireland will require a coordinated effort across policy, practice, and public engagement.
Community pharmacy has a critical role to play, offering accessibility, trust, and frequent patient contact. However, realising this potential will depend on ensuring that pharmacists are supported and integrated effectively into the programme.
As BowelScreen expands, the challenge will be not just to extend access, but to translate that access into meaningful participation.
Ian Clarke, Superintendent Pharmacist, Clarke’s Pharmacy, Galway
Pharmacists can also reinforce the importance of repeat participation, explaining that intermittent bleeding means regular screening increases the likelihood of detection.
In that effort, pharmacy may prove to be one of the most powerful tools available—helping to close the gap between invitation and action, and ultimately saving lives.
“BowelScreen is the free national bowel screening programme for men and women aged 58 to 70. The primary goal of BowelScreen is to reduce mortality from bowel cancer. We aim to prevent cancers from developing by finding and removing small growths (called polyps), which can turn into cancer if left untreated. Every year around 3,000 people have pre-cancerous growths removed because they did our BowelScreen test. These are all potential cancers prevented. In addition, if cancer is diagnosed through BowelScreen testing it is more likely to be found at an early stage when treatment may lead to a better outcome.
“Over time, the HSE will expand the screening programme to people aged 55 to 74.
“People can expect their first invitation for bowel screening between their 58th and 60th birthday. Taking part in BowelScreen is now even easier as people can request their kit 24/7 through an online register. You can use the new online system to check if you are registered with BowelScreen, provide your contact details, check when your next test is due and request a home test kit.
Pharmacist Involvement
“The HSE is engaging with pharmacies and other relevant stakeholders and planning is under way to implement the measures outlined in the Community Pharmacy Agreement 2025, in relation to pharmacist involvement in the BowelScreen programme. We have met the Irish Pharmacy Union, and look forward to working with community pharmacies to implement this agreement and work together to support more people to choose screening.
“The agreement sets out the key actions to ensure collaboration and integration between the HSE and pharmacists. In relation to the respective responsibilities in the agreement, it sets out that the HSE will “maintain responsibility for issuing FIT kits and processing returned tests”.
“As per the timelines identified in the agreement, it will be rolled out in a phased basis starting in 2026, starting with the training and health promotion activity.”
Bowel cancer is one of the most common cancers in Ireland, but it is also one of the most treatable when detected early. The national BowelScreen programme plays a vital role in identifying early signs of disease and preventing cancer from developing altogether.
For community pharmacists, understanding how the programme works—and how to communicate its benefits effectively—can make a meaningful difference in improving uptake and patient outcomes. What is BowelScreen?
BowelScreen is Ireland’s free national screening programme for colorectal cancer. It is currently offered every two years to men and women aged 58 to 70, with plans to expand eligibility to those aged 55 to 74.
The programme uses a Faecal Immunochemical Test (FIT), which detects tiny amounts of blood in stool that may not be visible. Participants complete the test at home and return it by post for analysis.
If blood is detected, further investigation—usually a colonoscopy—is offered to determine the cause.
Screening is designed for people who feel well and have no
symptoms. Its purpose is to detect cancer early, when treatment is more effective, or to identify and remove pre-cancerous polyps before they develop into cancer.
Each year, thousands of people in Ireland have these pre-cancerous growths removed through BowelScreen—preventing cancer from developing in the first place.
It is important to reinforce to patients that:
• A positive FIT result does not mean cancer
• A normal result does not rule out cancer completely
• Ongoing participation every two years is essential
Pharmacists frequently encounter misunderstandings about bowel screening. Common misconceptions include:
• “I don’t need screening because I have no symptoms” Screening is specifically for people without symptoms.
• “It’s only for people with a family history”
Most cases of bowel cancer occur in people with no family history.
• “The test is invasive or complicated”
The FIT test is simple, quick, and completed at home.
Addressing these misconceptions is key to improving participation.
Recognising Red Flags
While screening targets asymptomatic individuals, pharmacists should remain alert to symptoms that require referral.
These include:
• Persistent changes in bowel habits
• Blood in the stool
• Unexplained weight loss
• Ongoing fatigue
• Abdominal pain or discomfort
Patients presenting with these symptoms should be advised to seek medical evaluation, regardless of screening history.
Encouraging participation in screening often requires more than simply providing information. Pharmacists can support behaviour change by:
• Using clear, simple language
• Reassuring patients about the process
• Emphasising the benefits of early detection
• Addressing concerns around follow-up procedures
Even brief interventions can have a significant impact.
As the BowelScreen programme expands, pharmacists are likely to play an increasingly important role in supporting awareness and engagement.
By combining clinical knowledge with trusted patient relationships, pharmacy teams can help bridge the gap between invitation and participation—ultimately contributing to earlier detection and better outcomes for patients.
Proanthocyanidins are a potent antioxidant with wide-ranging health benefits, yet they’re becoming difficult to obtain from diet alone. This has important implications for modern patients.

For decades, healthcare conversations have focused on vitamins A, B, C, D, E and K. However, growing scientific interest continues to highlight a group of plant compounds once referred to as Vitamin P: proanthocyanidins.
Although not classified as essential vitamins, these powerful polyphenols play an important role in vascular integrity, connective tissue support and protection against oxidative stress. And in today’s environment, where inflammatory burden is steadily rising, their importance has never been greater.
The Burden of Modern Living Oxidative stress occurs when free radicals overwhelm the body’s antioxidant defences. While this is a natural biological process, modern environmental and lifestyle pressures have significantly increased that burden. Contributing factors include air pollution, ultraviolet radiation, processed and nutrient depleted diets, psychological stress, sedentary lifestyles and chronic inflammatory conditions.
Over time, excess oxidative stress contributes to connective tissue breakdown, endothelial dysfunction and low-grade systemic inflammation, presentations that are increasingly encountered in pharmacy practice.
Patients may not describe “oxidative stress”, but they often present with its downstream effects including joint stiffness, reduced mobility, poor circulation, slower recovery and visible skin ageing.
Historically, proanthocyanidins were abundant in diets rich in berries, grape skins and seeds, apples and other deeply coloured plant foods. Many of these compounds are concentrated specifically in the skins and seeds of fruit, the very parts increasingly absent from modern consumption patterns.
In pursuit of convenience, longer shelf life and sweeter taste profiles, much of today’s produce has been selectively bred for palatability. Seedless grapes, seedless citrus and thinner-skinned fruits now dominate supermarket shelves. While appealing to consumers, these adaptations often reduce exposure to phytonutrients naturally concentrated in seeds and skins, including proanthocyanidins.
At the same time, agricultural practices prioritise yield, size and uniformity rather than phytonutrient density, while extended transport and storage may influence polyphenol levels. In addition, ultra-processed foods continue to displace whole, fibre-rich plant foods in everyday diets. The paradox of modern abundance is that while food is plentiful, exposure to diverse plant compounds may be diminishing.
This widening gap between oxidative burden and phytonutrient intake helps explain the renewed clinical interest in standardised supplementation.
Proanthocyanidins: Mechanism and Clinical Relevance
Proanthocyanidins are among the most potent naturally occurring antioxidants, with particular affinity for collagen-rich tissues and vascular structures.
Their mechanisms of action include:
• Neutralising excess free radicals
• Supporting collagen stability and connective tissue integrity
• Promoting healthy microcirculation and endothelial function
• Helping modulate inflammatory pathways
For pharmacists, this positions proanthocyanidins as a valuable adjunct in categories such as:
• Joint and mobility support
• Vascular and circulation health
• Skin integrity and healthy ageing
• Recovery and active lifestyles
• General antioxidant support within chronic inflammatory contexts
Importantly, they work synergistically with Vitamin C and Vitamin E, both of which are involved in antioxidant recycling and collagen support.
Introducing Procydin®: Standardised Grape Seed Extract
Procydin® is a scientifically formulated supplement containing a high-quality grape seed extract (Vitis vinifera L.) delivering 70 mg of proanthocyanidins per capsule.
The Irish formulation is presented in a vegetarian capsule (hydroxypropyl methylcellulose), making it suitable for vegan consumers.
Each capsule also contains:
• Orange fruit extract (bioflavonoids)
• Calcium L-ascorbate (Vitamin C)
• D-alpha-tocopherol acetate (Vitamin E)
This synergistic blend is designed to support antioxidant defence, connective tissue integrity and healthy circulation as part of consistent, long-term use.
Real-World Consumer Feedback
Procydin® has been available in South Africa for over two decades, where it has developed a strong base of repeat users and healthcare professional familiarity.
Post-market consumer feedback indicates consistent longterm use, often integrated into broader preventative wellness strategies. Reported themes align with the known mechanisms of proanthocyanidins, including perceived support for mobility, circulation and overall vitality when taken consistently over time.
High levels of repeat purchase and sustained daily use suggest that
consumers experience tangible value in incorporating grape seed–derived proanthocyanidins into their routines.
By targeting excess free radical activity and supporting connective tissue integrity, Procydin® addresses oxidative stress, a recognised contributor to chronic inflammatory processes, at a foundational physiological level.
As pharmacy continues to shift toward preventative health and long-term wellness strategies, the role of polyphenols such as proanthocyanidins warrants greater attention.
With growing awareness of chronic inflammation, oxidative stress and vascular health, patients are increasingly seeking evidenceinformed, plant-based solutions that integrate into daily routines.
By bridging the gap between dietary insufficiency and modern inflammatory load, proanthocyanidins may represent one of the most overlooked tools in the preventative health toolkit.
Procydin® offers pharmacists a standardised, synergistic and vegan-friendly option to support patients seeking everyday antioxidant defence.





Shingles is a viral infection caused by the reactivation of the varicella-zoster virus—the same virus responsible for the common childhood illness, chickenpox. Following initial infection, the virus does not leave the body but instead remains dormant in nerve cells, often for decades.

As individuals age or experience a weakening of the immune system, the virus can reactivate. When this occurs, it travels along nerve pathways to the skin, producing the characteristic blistering rash typically seen on one side of the body or face. This process is often accompanied by intense pain, which can persist even after the visible rash has resolved. In some cases, this leads to postherpetic neuralgia, a debilitating condition that can significantly impact quality of life.
A vaccine for shingles has been available for approximately 20 years, with a newer, more effective version introduced in 2017. This updated vaccine offers around 90% protection against infection and represents a major advancement in the prevention of shingles and its complications. While the primary aim of vaccination is to prevent shingles itself, emerging research suggests that its benefits may extend beyond this. A recent large-scale study in Wales, which compared vaccinated and unvaccinated older adults, found that those who received the shingles vaccine had a 20% lower risk
of developing dementia. This may be linked to the role of the varicella-zoster virus in triggering chronic inflammation in the brain, which has been associated with neurodegenerative processes.
In addition, research from Korea has indicated a 26% reduction in cardiovascular events among individuals who had been vaccinated against shingles. This may be explained by the fact that shingles infection can contribute to blood vessel damage, inflammation, and clot formation— factors known to increase the risk of heart disease. By preventing infection, vaccination may therefore help reduce these downstream risks.
Community pharmacy has played an increasingly important role in vaccine delivery in Ireland over the past decade. Pharmacists first became involved in national vaccination efforts in 2011 with the rollout of the influenza vaccine. Their role expanded significantly during the Covid-19 pandemic, when pharmacy teams were instrumental in delivering vaccines at scale across the country.
Written by Professor Christine Loscher, Head of School and Full Professor of Immunology, School of Biotechnology, Dublin City University
Pharmacies are deeply embedded within communities and have established, trusted relationships with patients—particularly older adults, who are most at risk from shingles. This accessibility and trust are key reasons why pharmacyled vaccination programmes have proven so effective.
Given this track record, community pharmacy is well positioned to support the delivery of a national shingles vaccination programme, should one be introduced. Pharmacists have the clinical expertise, infrastructure, and patient engagement necessary to improve access and uptake.
Despite the availability of an effective vaccine, access remains a significant challenge in Ireland.
The National Immunisation Advisory Committee (NIAC) has approved the shingles vaccine for use in Ireland. However, it has not yet been prioritised for inclusion in a publicly funded national programme. As a result, the vaccine is currently only available on a private basis, at a cost of approximately ¤500 for the required two-dose course.
For many patients—particularly older adults—this cost represents a substantial barrier. The consequence is that a highly effective preventative intervention remains out of reach for a large proportion of the population.
This situation is in stark contrast to other countries. The shingles vaccine is available free of charge in several healthcare systems, including those in the UK, the United States, Canada, Australia, and a number of European countries. These programmes recognise not only the direct benefits of preventing shingles, but also the wider health and economic benefits associated with reducing complications and hospitalisations.
The growing body of evidence linking shingles vaccination to reduced risks of dementia and cardiovascular disease adds further weight to the case for wider access. These findings suggest that the benefits of vaccination extend beyond infection control and into broader areas of public health.
Preventative medicine offers significant long-term value, both in improving patient outcomes and reducing pressure on healthcare systems. However, it is not always prioritised in funding decisions. Ireland has a strong and successful childhood vaccination programme, but adult vaccination remains more limited. Vaccines such as influenza and shingles are often not publicly funded, leaving patients to bear the cost themselves. This has a direct impact on uptake, particularly among those who may benefit most.
Lower uptake not only increases the burden of disease but also contributes to seasonal pressures on healthcare services, as seen each winter with influenza.
The current situation presents a clear opportunity for policy reconsideration. The availability of an effective shingles vaccine, combined with emerging evidence of wider health benefits, strengthens the argument for its inclusion in a national vaccination programme.
Community pharmacy stands ready to support such an initiative. With its accessibility, trusted relationships, and proven track record in vaccine delivery, pharmacy could play a central role in improving uptake and ensuring equitable access.
Ultimately, the question is not whether the shingles vaccine works—it does—but whether it should be made more widely available to those who need it most.
As the focus of healthcare increasingly shifts towards prevention, ensuring access to effective vaccines such as this will be critical in improving long-term health outcomes for the population.











Shingrix is indicated for those aged 50 years of age or older and those 18 years of age or older at increased risk of HZ.1
Established safety profile. Most vaccination reactions were mild to moderate, with a median duration of 2-3 days3,4
79.7% efficacy against shingles during ZOE-Long Term Follow Up study (Y6 to Y11 since vaccination)5
87.7 % overall efficacy against shingles up to year 11 after initial vaccination5
Shingrix powder and suspension for injection in vials [Herpes Zoster vaccine (recombinant, adjuvanted)].
(Please refer to SmPC before prescribing)
Composition: After reconstitution, one dose (0.5 mL) contains: Varicella Zoster Virus glycoprotein E antigen1,2 50 micrograms. (1 adjuvanted with AS01B containing: plant extract Quillaja saponaria Molina, fraction 21 (QS-21) 50 micrograms, 3-O-desacyl-4’-monophosphoryl lipid A (MPL) from Salmonella minnesota 50 micrograms,2 glycoprotein E (gE) produced in Chinese Hamster Ovary (CHO) cells by recombinant DNA technology). Therapeutic indications: Prevention of herpes zoster (HZ) and post-herpetic neuralgia (PHN) in adults 50 years of age or older and in adults 18 years of age or older at increased risk of HZ. The use of this vaccine should be in accordance with official recommendations. Posology and method of administration: For intramuscular injection only, preferably in the deltoid muscle. Primary
Vaccination: Initial dose of 0.5 ml followed by a second 0.5 ml dose 2 months later. For flexibility the 2nd dose can be administered between 2 and 6 months after the first dose. For subjects who are or might become immunodeficient or immunosuppressed and whom would benefit from a shorter vaccination schedule, the 2nd dose can be given 1 to 2 months after the initial dose. Booster doses: need not established. Contraindications: Hypersensitivity to the active substances or any of the excipients. Special warnings and precautions for use: The name and the batch number of the administered product should be clearly recorded. Appropriate medical treatment and supervision should be readily available in case of an anaphylactic event. Administration of Shingrix should be postponed in subjects suffering from an acute severe febrile illness. However, the presence of a minor infection, such as cold, should not result in deferral. A protective immune response may not be elicited in all vaccinees. Never administer intravascularly or intradermally; subcutaneous administration not recommended as it may lead to an increase in transient local reactions.
Caution in individuals with thrombocytopenia or any coagulation disorder since bleeding may occur following intramuscular administration. Syncope can occur following, or before any vaccination as a psychogenic response. This can be accompanied by several neurological signs such as transient visual disturbance, paraesthesia and tonic-clonic limb movements during recovery. It is important that procedures are in place to avoid injury from
faints. There are no data to support replacing a dose of Shingrix with another HZ vaccine. Contains 0.08 mg of polysorbate 80 per dose. Interactions: Shingrix can be given concomitantly with unadjuvanted inactivated seasonal influenza vaccine, 23-valent pneumococcal polysaccharide vaccine (PPV23), 13-valent pneumococcal conjugate vaccine (PCV13), reduced antigen diphtheria-tetanusacellular pertussis vaccine (dTpa) or coronavirus disease 2019 (COVID-19) messenger ribonucleic acid (mRNA) vaccine or respiratory syncytial virus (RSV) vaccine (recombinant, adjuvanted). The vaccines should be administered at different injection sites. In adults aged 50 years and above, systemic adverse reactions that are very commonly and uncommonly reported, following administration of Shingrix alone were reported with increased frequency when Shingrix was co-administered with a COVID-19 mRNA vaccine. Fertility, pregnancy and lactation: There were no effects on male or female fertility in animal studies. It is preferable to avoid the use of Shingrix during pregnancy. The effect on breast-fed infants of administration of Shingrix to their mothers has not been studied. It is unknown whether Shingrix is excreted in human milk. Effects on ability to drive and use machines: Shingrix may have a minor influence on the ability to drive and use machines in the 2-3 days following vaccination. Undesirable effects: Very common (≥1/10): Headache, GIT symptoms, myalgia, injection site reactions, fatigue, chills, fever. Common (≥1/100 to <1/10): injection site pruritus, malaise. Uncommon (≥1/1000 to <1/100): lymphadenopathy, arthralgia, injection site induration. Rare (≥1/1000 to <1/100): Hypersensitivity reactions. Very rare: Guillain-Barré syndrome. Legal Category: POM A. Marketing Authorisation Number: EU/1/18/1272/001 Marketing Authorisation Holder: GlaxoSmithKline Biologicals S.A., Rue de l’institut 89, 1330 Rixensart, Belgium. Further information is available from GlaxoSmithKline (Ireland) Ltd. 12 Riverwalk, Citywest Business Campus, Dublin 24. Telephone: 01-4955000. Code: PI-7757. Date of preparation: February 2026.
Adverse events should be reported directly to the Health Products Regulatory Authority (HPRA) on their website: www.hpra.ie. Adverse events should also be reported to GlaxoSmithKline on 1800 244 255 or via online form https://gsk.public.reportum.com
References : 1. Shingrix, Summary of Product Characteristics (SPC), available on https://www.medicines.ie/medicines/shingrix-powder-and-suspension-for-suspension-for-injection- herpes-zoster-vaccine-recombinant-adjuvanted--35192/spc. Last Accessed: October 2025. 2. Kawai, K et al. BMJ Open. 2014 Jun 10;4(6):e004833. doi: 10.1136/bmjopen-2014-004833. 3. Lal H, Cunningham AL, Godeaux O, Chlibek R, Diez-Domingo J, Hwang S-J, et al. Efficacy of an adjuvanted herpes zoster subunit vaccine in older adults. N Engl J Med. 2015 May;372(22):2087-96. 4. Cunningham AL, Lal H, Kovac M, Chlibek R, Hwang S-J, Diez-Domingo J, et al. Efficacy of the herpes zoster subunit vaccine in adults 70 years of age or older. N Engl J Med. 2016 Sep;375(11):1019-32. 5. Strezova A et al. Final analysis of the ZOE-LTFU trial to 11 years postvaccination: efficacy of the adjuvanted recombinant zoster vaccine against herpes zoster and related complications. eClinicalMedicine 2025;83: † SHINGRIX does not protect 100% of individuals vaccinated. Secondary objective (pre-planned and descriptive analysis): VE against first or only shingles episode from 1-month post-dose 2 in the ZOE-50/70 studies.
Trade marks are owned by or licensed to the GSK group of companies ©2025 GSK group of companies or its licensor. PM-IE-SGX-JRNA-240002 | Date of Preparation: February 2026.
If the skin’s water content drops below 10%, the enzymes responsible for corneocyte desquamation no longer function properly. The corneocytes shrink reducing levels of NMF and the lipid lamellae appears broken (analogous to crumbling mortar in an old brick wall). These will create breaches in the skin’s barrier, allowing for greater water loss.
Clinically, xerotic skin appears rough and flaky, with white patches (which represent adherent corneocytes due to reduced desquamation) that is both uncomfortable and pruritic. Dry skin is commonly seen on the lower legs, the trunk, forearms, hands and face. The feet are normally less prone to dryness, being protected by socks and tights, but dryness can be problem for those who walk barefoot or wear sandals.
Left untreated, xerotic skin becomes red, dull and rough, and ultimately starts to crack. These cracks deteriorate over time and can bleed, providing an entry portal for irritants and bacteria that can lead to infections.
To prevent dry skin, customers should be advised to maintain skin hydration through adequate fluid intake, avoid hygiene products that dry out the skin, reduce fullbody bathing to every other day, and avoid long, hot showers. To treat dry skin, there are several suggestions pharmacists can offer. Patients can apply oil-based emollients (eg, petrolatum and mineral oil) after bathing, or they may use colloidal oatmeal or bath oil while bathing to enhance skin hydration. Humectants, such as glycerin, draw water into the skin. More serious cases of dry skin may require a urea- or lactic acid containing product.
The words eczema (which comes from the Greek word meaning ‘to boil’) and dermatitis (meaning inflammation of the skin) are often used interchangeably to describe several conditions that cause the skin to become dry, itchy, inflamed or irritated.
There are a number of different types of eczema but the term is most often used to refer to atopic eczema (also called atopic dermatitis).
The word atopic refers to hypersensitivity reactions to something in the environment.
Atopic eczema is a very common, non-contagious, chronic (long-term) inflammatory skin condition. While the exact cause is not known, certain factors are thought to be important in its development, these include an inherited (genetic) predisposition to have a weakened skin barrier, as well as altered inflammatory and allergy responses. Atopic eczema most frequently begins in infancy but may also develop for the first time in adulthood. It affects approximately 1 in 5 children and 1 in 12 adults in Ireland.
For most children affected, the disease is mild. However, often those who have seemingly outgrown the condition will continue to have life-long sensitive skin and may have recurrences of eczema following long symptomfree spells.
Atopic eczema can run in families and frequently occurs alongside other atopic conditions, including hay fever and asthma.
Symptoms include red, dry, itchy skin, which can sometimes weep, become blistered, crusted and thickened. However, the appearance of eczema and the locations of the body affected can vary greatly depending on the age of the person affected.
Atopic eczema has been described as the ‘itch that rashes’. Itch is an unpleasant sensation which activates the desire to scratch. The desire to scratch can be so intense that people can sometimes scratch until the skin becomes painful and bleeds. Occasionally, this can lead to cuts, abrasions and skin infections.
Scratching is a behaviour used to relieve itching, but sometimes it
can also be prompted by emotion, or it can become habitual. Some people scratch when they are stressed or anxious. Some people scratch through subconscious repeated behaviour, and can get stuck in the itch scratch cycle. When they feel itchy they scratch – when they scratch chemicals are released that causes more itch.
Alongside conventional management and treatments for atopic eczema some simple techniques may help to relieve the itch.
Habit reversal is a behaviour modification technique that can be beneficial in helping to replace an old habit with a new one.
To stop scratching, the person first needs to become aware of the desire to scratch and the action of scratching. However, people living with eczema can get fed up of family saying “stop scratching!”, so it may help by developing a code word to provide encouragement instead. Maybe gently ‘pinch’ or ‘tap’ or count the number of times scratching is observed to create awareness of the action. Some people choose to use a tally counter.
The main aim is to improve symptoms and achieve longterm control. In atopic eczema, the skin’s protective barrier is weakened which allows moisture to be lost, and irritants and allergens to pass through the skin more easily. Some common everyday substances e.g. soap, contribute to the weakening of the skin’s barrier and should be avoided.
Emollient (moisturiser) therapy is often used to manage a number of
skin conditions, such as eczema and psoriasis. However, with such a wide variety of emollients available on the market today, it is important you and your team are aware about which products are the best fit for people who need to use emollients regularly.
Emollients are moisturisers that come in the form of lotions, creams and ointments. They repair and protect the skin barrier. They hydrate and trap moisture in the skin. Emollient therapy improves symptoms resulting in decreased dryness, flaking, cracking, scaling, resulting in reduced itch and inflammation.
Emollients are used in two ways: Leave on emollient or wash emollient. A leave-on emollient is applied directly to the skin to moisturise and trap moisture. Wash emollient comes in the form of a soap substitute as an alternative to soap, emollient body wash in place of shower gel, or emollient bath additive or bath oil instead of bubble bath. Soaps, shower gels and bubble-bath should be avoided as they contain detergents, which strip away the natural oils and dries out the skin.
Finding the right emollient is often a matter of trial and error, but the best emollients are ones customers prefer to use and will continue to use every day.
Practical tips for emollient therapy
Establish a good daily skin care routine and try to stick to it.
Don’t stop moisturising when the skin is clear.
Apply emollients in a smooth, downward motion, in the direction of the hair growth.
Never stick fingers into a tub of emollient – Remember to use a CLEAN spatula, spoon or a pump dispenser to apply so it does not become contaminated.
A child with inflamed skin often requires a minimum of 250g of ‘leave-on’ emollient per week, while an adult may need approximately 500g –1000g per week.
Bathwater should be a lukewarm temperature. 5 minutes with an emollient bath additive is sufficient.
Remember – avoid soap, bubble bath and shower gel
After bathing or showering, gently pat skin dry and apply emollient all over when the skin is still slightly damp.










Following on from the March issue Continuing Professional Development on the Pharmacy Role in Sleep Disorders, this 5-Minute Learning Module is designed to enhance the community pharmacy team understanding and ask further questions as to how you can support and advise patients.

Sleep disorders are one of the most common concerns presented in community pharmacy, with patients frequently seeking advice before consulting a GP. From short-term insomnia linked to stress, to longer-term sleep disruption associated with lifestyle, menopause, or mental health, pharmacy teams are often the first point of contact.
Sleep problems are rarely caused by a single factor. In practice, they are often the result of a combination of lifestyle habits, psychological stress, underlying health conditions, and, in some cases, medicines. Patients may describe difficulty falling asleep, frequent waking during the night, or waking too early and feeling unrefreshed. Understanding how long symptoms have been present, and how they impact daily functioning, is key to distinguishing between short-term and chronic insomnia.
A brief but structured conversation can provide valuable insight. Asking about sleep patterns, caffeine and alcohol intake, screen use, and current medicines can quickly highlight potential contributing factors. It is also important to explore whether the patient has already tried any remedies, including over-thecounter or herbal products, as this can indicate both expectations and potential misuse.
Pharmacy teams should also be alert to underlying causes. Medicines such as antidepressants, corticosteroids, stimulants,
Consider:
and some decongestants can interfere with sleep, while hormonal changes—particularly during perimenopause—are an increasingly common driver of sleep disturbance. Mental health is another key consideration, as anxiety and depression are closely linked to poor sleep, often creating a cycle that can be difficult to break.
While many cases of insomnia can be managed within pharmacy, there are clear situations where referral is required. Persistent symptoms lasting more than four weeks, significant daytime impairment, or sleep disturbance linked to worsening mental health should prompt GP referral. Symptoms such as loud snoring, choking during sleep, or excessive daytime sleepiness may indicate obstructive sleep apnoea and require further investigation. Similarly, patients who appear to be relying on sleep aids over a prolonged period should be reviewed and, where appropriate, referred.
For the majority of patients, non-pharmacological strategies remain the cornerstone of effective sleep management. Practical, tailored advice is far more effective than generic recommendations. Encouraging a consistent sleep routine, reducing screen use in the evening, limiting caffeine intake, and creating a comfortable sleep environment can all have a meaningful impact. Establishing a wind-down routine before bed and avoiding irregular sleep patterns are also key messages.
Am I/my team confident in carrying out a brief but structured sleep assessment (duration, pattern, lifestyle, medicines, underlying causes)?
Are we identifying when sleep issues may be linked to medicines, mental health, or hormonal changes (e.g. perimenopause)?
Do we recognise red flags that require referral, such as chronic insomnia, suspected sleep apnoea, or deterioration in mental health?
Are we monitoring repeat requests for OTC sleep aids and recognising potential overuse or dependency?
Importantly, pharmacists can help patients understand that sleep is not always perfect, and that occasional disruption is normal. Reassurance can be particularly valuable for patients who have developed anxiety around sleep itself, which can perpetuate the problem.
Sedating antihistamines remain one of the most widely used and clinically appropriate OTC options for the short-term management of transient insomnia. Products such as diphenhydramine-based sleep aids can provide effective relief where sleep disturbance is temporary, for example during periods of stress, travel, or shortterm lifestyle disruption.
Clear counselling is essential. Patients should be advised that these products are for short-term use only, typically no longer than one to two weeks. They should also be made aware of potential next-day drowsiness, the need to avoid alcohol, and the importance of not combining them with other sedative medicines.
In practice, these products can play a useful role when used appropriately, particularly when combined with sleep hygiene advice and clear expectations around short-term use.
Particular caution is required in older adults, who are more susceptible to side effects such as confusion, dizziness, and falls. In this group, nonpharmacological approaches
Key Points:
Sleep disorders are often multifactorial, involving lifestyle, psychological, hormonal, and medicationrelated causes
Most sleep problems can be effectively improved with behavioural changes, with medicines playing a short-term supportive role
Pharmacists and pharmacy teams are in a strong position to identify patterns, intervene early, and guide appropriate care
Patients may feel anxious or embarrassed discussing sleep—sensitivity and reassurance are essential
OTC sleep aids (e.g. sedating antihistamines) are suitable for shortterm, situational insomnia only, and require clear counselling
should always be prioritised. Melatonin, while frequently discussed by patients, remains prescription-only in Ireland, and pharmacy teams should guide patients accordingly. Similarly, patients enquiring about long-term or stronger sleep medications should be referred for medical assessment.
An important part of the pharmacist’s role is recognising patterns of behaviour. Repeat purchases of sleep aids can indicate underlying chronic insomnia or inappropriate reliance on medication. These situations present an opportunity for intervention, allowing pharmacists to support safer, more sustainable approaches to sleep management. Sleep health is also evolving. Increasingly, patients are presenting with data from wearable devices or sleep tracking apps. While these can provide useful insights, they can also contribute to anxiety around sleep performance. Pharmacists should focus on clinical symptoms and overall wellbeing, rather than numerical sleep metrics alone.
Ultimately, community pharmacy is uniquely positioned to support patients with sleep disorders. With accessibility, trusted relationships, and frequent patient contact, pharmacy teams can provide early intervention, practical advice, and appropriate referral when needed.
By taking a structured, patientcentred approach, pharmacists can help improve sleep outcomes, reduce inappropriate medicine use, and support overall health and wellbeing—often through simple but effective interventions delivered at the pharmacy counter.
Ensure all pharmacy team members understand:
How to ask simple, structured questions to assess sleep problems effectively
The common causes of insomnia, including lifestyle habits, medicines, mental health, and menopause
The appropriate role, benefits, and limitations of OTC sleep aids, including short duration of use
How to identify red flags and confidently refer patients to a GP when needed



*Based on IQVIA sales data MAT 12/2025. Nytol One-A-Night 50 mg Tablets contains diphenhydramine hydrochloride. A symptomatic aid to the relief of temporary sleep disturbance in adults. Adults: One tablet to be taken 20 minutes before going to bed, or as directed by a physician. Do not exceed the maximum dose of one tablet in 24 hours. Elderly patients or patients with liver or kidney problems should consult their doctor before taking this medicine. Children under 18 years: Not recommended. The product should not be taken for more than 7 days without consulting a doctor. Contraindications: hypersensitivity to the active substance or to any of the excipients, stenosing peptic ulcer, pyloroduodenal obstruction, phaeochromocytoma, known acquired or congenital QT interval prolongation, known risk factors for QT interval prolongation. Special warnings and precautions: pregnancy/lactation, renal and hepatic impairment, myasthenia gravis, epilepsy or seizure disorders, narrow-angle glaucoma, prostatic hypertrophy, urinary retention, asthma, bronchitis, COPD. Patients should be advised to promptly report any cardiac symptoms. Tolerance and / or dependence may develop with continuous use. Do not take for more than 7 consecutive nights without consulting a doctor. Should not be used in patients currently receiving MAO inhibitors (MAOI) or patients who have received treatment with MAOIs within the last two weeks. Use in the elderly should be avoided. Avoid concomitant use of alcohol or other antihistamine-containing preparations. Do not drive or operate machines. Cases of abuse and dependence were reported in adolescents or young adults for recreational use and/or in patients with psychiatric dis-orders and/or history of abuse disorders. Contains lactose. May suppress the cutaneous histamine response to allergen extracts and should be stopped several days before skin testing. Interactions: Alcohol, CNS depressants, MAO inhibitors, anticholinergic drugs (e.g. atropine, tricyclic antidepressants), metoprolol and venlafaxine, CYP2D6 inhibitors, Class Ia and Class III anti-arrhythmics. Side effects: dry mouth, fatigue, sedation, drowsiness, disturbance in attention, unsteadiness, dizziness, thrombocytopenia, hypersensitivity reactions, confusion, paradoxical excitation, convulsions, headache, paraesthesia, dyskinesias, blurred vision, tachycardia, palpitations, thickening of bronchial secretions, gastrointestinal disturbance, muscle twitching, urinary difficulty, urinary retention. Product not subject to medical prescription. PA1186/016/001. MAH: Chefaro Ireland DAC. The Sharp Building. Hogan Place. Dublin 2. Ireland. Date of preparation: Nov. 2023. SPC: https://www.medicines.ie/medicines/nytol-one-a-night-50-mg-tablets-34889/spc MAT-10247
As workplace pressures continue to rise, more patients are presenting in community pharmacy with symptoms linked not to acute illness, but to the demands of modern working life. Stress, poor sleep, musculoskeletal pain and burnout are increasingly common, yet often go unrecognised or unmanaged until they begin to impact day-to-day functioning.
For pharmacists, this presents both a challenge and an opportunity. As one of the most accessible healthcare professionals, pharmacy teams are often the first point of contact for individuals struggling with workrelated health concerns—whether through requests for pain relief, sleep aids, or general advice. These interactions offer valuable moments to identify underlying issues, provide practical support, and guide patients towards healthier coping strategies.
Marking National Workplace Wellbeing Day, Mairead McCarthy, Director of Wellbeing and Founder of WellPlace Ireland, outlines the key workplace-related health challenges facing employees today and shares practical, accessible advice that pharmacists can use to support patients in managing
stress, improving wellbeing, and maintaining balance in both their professional and personal lives.
At WellPlace, our focus is on helping companies look after their employees by providing an extensive range of workplace wellbeing services.
We work with organisations of all sizes and see a number of common workplace related conditions including stress and anxiety due to workload, lack of support, or conflict and uncertainty; musculoskeletal disorders (such as back pain, neck and shoulder pain, tendonitis, and carpal tunnel syndrome); and repetitive strain injuries affecting hands, wrists, elbows or shoulders.
We all experience stress from time to time due to the demands of

daily life, but not all stress is bad. Stress can actually help motivate us to get things done when necessary in the short term, and once we have a rest period to recover, there are often no lasting ill-effects.
However, ongoing stress and anxiety can lead to sleep disruption, and possibly burnout if not identified and managed properly.
Burnout typically results from ongoing workplace stress that hasn't been successfully managed. If people are presenting with exhaustion, feeling drained and lethargic, overwhelmed, often unable to cope with even small day to day tasks, then they are more likely to be suffering from burnout rather than normal workrelated stress.
If patients are struggling with work-related stress, here is some practical advice that can be offered to them:
Be kind to yourself - we often beat ourselves up about not doing enough and not looking after ourselves well enough. But being kind to yourself is really important, particularly when experiencing stress.
Try to include some self-care in your daily routine. We all need to put on our own life jackets first and take time to prioritise our physical and mental health in order to stay well. Getting out for a walk can really help and people should aim for 30 minutes of daily activity. Eating balanced meals as much as possible - it's okay to eat sweet things but in moderation and it is really important to include plenty of protein and healthy fats and we often don't get enough of these.
Do what you can to improve your sleep routine by incorporating some time to wind down before bed. Limiting caffeine and alcohol also can help improve sleep and reduce stress hormones.
Take it one step at a time - it can feel overwhelming all the things we need to do in life and even in looking after ourselves. So taking it one step at a time is a good start to manage this.
Mairead McCarthy, Director of Wellbeing and Founder of WellPlace Ireland
Introduce breathing and mindfulness techniques to help reduce stress during the dayand this can also help people to wind down before a bed and help improve sleep.
Connection with others is really important for everybody. Whether it's a family member or a friend, taking time to connect with people can help us feel less alone when we're struggling and help reduce our stress levels.
Setting boundaries with work but also with your personal life, knowing that it's ok to say no. It's important to prioritise breaks during the day to manage workload.
Pharmacists should note if there is frequent use of sedatives, painkillers, or laxatives as possible stress signs, and suggest nondrug coping methods first. Use products that promote stress relief including bath salts, roll on oils and supplements.
Leadership and culture in an organisation make a real difference to a healthy work environment. Commitment from leaders to wellbeing and allowing people flexibility where possible and giving them control over how they do their work can help to boost engagement and reduce stress.
It's so important that people feel safe, supported, and valued in their workplace. Open communication is also key, where people feel it is safe to speak up about concerns, and to push back if they feel their workload is becoming overwhelming.
Having positive relationships in work can make a real difference to how people feel they belong in the organisation, and whether they feel respected, all of which are associated with better overall wellbeing.
Most important of all when dealing with patients experiencing work related stress or health issues is to take the time to listen and to give practical advice that isn't overwhelming and that the patient can implement quite soon. Whether it's trying a natural technique, a supplement or medication, let them know that there is support available to them and that they're not alone.
AUTHOR: Donna Cosgrove PhD MPSI
Donna graduated with a BSc in Pharmacy from the Royal College of Surgeons in Ireland. She then returned to university to complete a MSc in Neuropharmacology. This led to a PhD investigating the genetics of schizophrenia, followed by a postdoctoral research position in the same area. Currently Donna works as a pharmacist in Galway, and as a clinical writer.
Pain management represents a major component of community pharmacy practice, with widespread use of OTC analgesics.
Effective management requires distinguishing acute from chronic pain and recognising the biological, psychological, and social factors that influence symptom presentation. Acute pain arises from tissue injury and inflammatory processes, whereas chronic pain reflects persistent nociceptive, neuropathic, or nociplastic mechanisms and is associated with significant functional and psychosocial burden.
Evidence indicates that ibuprofen/paracetamol combinations and fastacting NSAIDs offer the most effective OTC relief for acute pain, while topical NSAIDs provide comparable efficacy with lower systemic risk. Chronic pain management increasingly emphasises non-opioid pharmacological options and nonpharmacological interventions, including exercise therapy, psychological support, TENS, and acupuncture.
Prescription data show rising analgesic use in Ireland that may be mitigated by increased availability to services that offer non-pharmacological management of pain. Multimodal approaches integrating medication with targeted physical and psychological therapies remain essential for safe, effective pain management.

1. REFLECT - Before reading this module, consider the following: Will this clinical area be relevant to my practice?
2. IDENTIFY - If the answer is no, I may still be interested in the area but the article may not contribute towards my continuing professional development (CPD). If the answer is yes, I should identify any knowledge gaps in the clinical area.
3. PLAN - If I have identified a
Introduction
Approximately 47% of EU consumers report using an over-the-counter (OTC) analgesic at least once weekly within the last month,1 positioning pain management as one of the main reasons for consultation within community pharmacy practice.2 An effective OTC analgesic should demonstrate satisfactory analgesic efficacy in a substantial proportion of patients, exhibit a rapid onset of action, and provide an adequate duration of effect. Self-care - pain management undertaken without prescription - remains appropriate for numerous minor, self-limiting conditions such as bruising, viral upper respiratory tract infections, minor wounds, ear pain, fever, headache, dysmenorrhoea, mild musculoskeletal pain, sinusitis or cold/flu symptoms, sprains and strains, and teething or dental discomfort. However, the extensive range of OTC analgesic options frequently complicates patient decision-making.3 Consequently, pharmacy professionals must be equipped to deliver evidencebased recommendations and identify red flags warranting referral for clinical evaluation or escalation to prescription-only analgesia, particularly when pain persists or deteriorates beyond the expected clinical trajectory.
Acute pain is defined as pain lasting less than 12 weeks and

knowledge gap - will this article satisfy those needs - or will more reading be required?
4. EVALUATE - Did this article meet my learning needs - and how has my practise changed as a result? Have I identified further learning needs?
5. WHAT NEXT - At this time you may like to record your learning for future use or assessment. Follow the

4 previous steps, log and record your findings. Published by IPN. Copies can be downloaded from www.irishpharmacytraining.ie
Disclaimer: All material published is copyright, no part of this can be used in any other publication without permission of the publishers and author. Panadol has no editorial oversight of the CPD programmes included in these modules.
may arise secondary to tissue injury, nerve insult, or surgical intervention. It is commonly associated with inflammatory responses, including swelling and reduced function, and may substantially impair activities of daily living and health-related quality of life.4 A wide range of analgesic interventions are available, including paracetamol, non-steroidal anti-inflammatory drugs (NSAIDs), opioids, complementary therapies, and herbal preparations. Within each class, multiple formulations exist, e.g. rapid-release tablets, effervescent preparations, liquid formulations, and fixeddose combinations, which may influence onset and extent of analgesic effect.
Chronic pain persists beyond the expected period of tissue healing, typically exceeding 3–6 months, and is associated with considerable functional limitation and psychosocial burden. Established non-modifiable risk factors include female sex, age greater than 65 years, highintensity acute pain, and lower socioeconomic status. Modifiable psychological risk factors (e.g. elevated pain catastrophising, heightened pain-related fear, and increased symptoms of anxiety, depression, or post-traumatic stress disorder) also contribute to chronic pain development and persistence.5 Chronic pain prevalence across Europe is high, with reported rates ranging from 12% to 48%, and an estimated




Donna Cosgrove
of pain (acute versus chronic) and the underlying nociceptive mechanism, including degenerative pathology (e.g. osteoarthritis), traumatic injury, or muscle spasm (4).
Table 1. Management considerations based on pain type: acute vs. chronic6
Table 1. Management considerations based on pain type: acute vs. chronic (6).
Characteristics Acute Pain
Duration < 3-6 months
Function Physiological (protective)
Chronic Pain
> 6 months
Pathologic (non-protective)
Cause Acute illness, injury, trauma, surgery Injury, chronic illness, cancer, may have no definable pathology
Treatment options
Goals of Treatment
NSAIDs, paracetamol, opioids, muscle relaxants, immobilisation, heat/cold, elevation, exercise, physical therapy
● Resolution of pain and underlying cause
● Facilitate recovery
● Minimise side effects
● Prevent chronic pain
Nociception and Types of Pain
prevalence of 43.5% in the UK. Selection of appropriate analgesic strategies requires consideration of both the temporal profile of pain (acute versus chronic) and the underlying nociceptive mechanism, including degenerative pathology (e.g. osteoarthritis), traumatic injury, or muscle spasm.4
Nociception and Types of Pain
Non-opioid analgesics (opioids necessary in some cases), physical therapy, CBT, exercise, yoga, relaxation, tai chi, massage, acupuncture
● Restore physical, emotional, social function
● Decrease pain
● Treat underlying cause
● Correct secondary consequences, e.g. maladaptive behaviour
associated with, or resembling that associated with, actual or potential tissue damage.”
of an inflammatory response and gather at the site of injury via the bloodstream, followed by the release of other chemical mediators. Inflammation can induce mast cell degranulation, releasing platelet activating factor, resulting in the subsequent release of 5-HT from the circulating platelets. This culminates in a hot inflamed site due to increase in blood flow towards the region, redness, swelling due to vascular permeability, pain caused by the activation and sensitisation of primary afferent neurons, and loss of function.
The localised inflammatory response induces the release of free arachidonic acid from phospholipids. Arachidonic acid (which is converted via cyclooxygenase (COX) pathways into prostaglandins (PGs)), along with mediators such as 5-HT, bradykinin, histamine, nerve growth factor (NGF), adenosine triphosphate (ATP), glutamate, leukotrienes, nitric oxide (NO), noradrenaline (NE), and protons, activates and sensitises nociceptors.
Nociception refers to the neural processes encoding actual or potential tissue damage. It arises from the activation of primary afferent nociceptors (unmyelinated C-fibres and thinly myelinated Aδ-fibres) which remain quiescent under normal physiological conditions and fire in response to noxious mechanical, thermal, or chemical stimuli. Once activated, signals are transmitted through peripheral afferents to the dorsal horn of the spinal cord and relayed via ascending pathways to supraspinal centres. Motor and autonomic responses are mediated through descending pathways projecting back to the spinal cord.
Nociception refers to the neural processes encoding actual or potential tissue damage. It arises from the activation of primary afferent nociceptors (unmyelinated C-fibres and thinly myelinated Aδ-fibres) which remain quiescent under normal physiological conditions and fire in response to noxious mechanical, thermal, or chemical stimuli. Once activated, signals are transmitted through peripheral afferents to the dorsal horn of the spinal cord and relayed via ascending pathways to supraspinal centres. Motor and autonomic responses are mediated through descending pathways projecting back to the spinal cord.
Pain is inherently subjective and influenced by biological, psychological, and social factors. Notably, pain intensity does not reliably correlate with the extent of nociceptive input or injury severity; psychosocial determinants often account for a greater proportion of variance in pain reports. Early identification of such factors is therefore essential to prevent progression to persistent pain states.
Pain, in contrast, represents the conscious perception of this activity and incorporates sensory, emotional, and cognitive dimensions. The International Association for the Study of Pain (IASP) defines pain as “an unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage.” Pain is inherently subjective and influenced by biological, psychological, and
Pain, in contrast, represents the conscious perception of this activity and incorporates sensory, emotional, and cognitive dimensions. The International Association for the Study of Pain (IASP) defines pain as “an unpleasant sensory and emotional experience

Aδ- and C-fibres terminate in specialised free nerve endings distributed across the skin, musculature, joint capsules, bone, and some visceral organs. Superficial structures are typically innervated by both fibre types, whereas deep somatic tissues such as muscle and joints are predominantly supplied by C-fibres. Acute inflammatory pain is generally sharp, short-lived, and mediated largely by Aδ-fibres. Chronic inflammatory pain, which persists beyond the expected healing period, is primarily C-fibre mediated. Additionally, “silent” nociceptors—abundant in visceral tissues—do not respond to mechanical stimuli under normal conditions but become sensitised during inflammation by locally released chemical mediators.

Neuropathic pain arises from lesions or diseases affecting the somatosensory nervous system and is frequently associated with allodynia (where pain is caused by a stimulus that does not normally provoke it) and spontaneous pain. Common aetiologies include metabolic disorders (e.g. diabetes), trauma, neurotoxins, malignancy, infectious processes (e.g. herpes zoster), and degenerative neurological disease. Structural injury such as compression or stretch alters axonal neurochemistry and promotes hyperexcitability of affected neurons. During neuropathic pain, nociceptors demonstrate a dynamic expression of ion channels, e.g. sodium channels, which are the major channels involved in neuronal excitability. Neurogenic inflammation occurs when inflammatory mediators, (e.g. substance P, calcitonin gene-related peptide, neurokinin) are released locally by the afferent neurons, which further amplifies peripheral and central sensitisation.
Inflammatory pain is a natural biological response to harmful stimuli that helps eradicate the necrotic cells and initiate the tissue repairing process. Neutrophils are usually the first respondents
Hyperalgesia refers to an increased pain response to a stimulus that is normally painful. Primary hyperalgesia occurs at the site of injury and is predominantly due to peripheral mechanisms, whereas secondary hyperalgesia and allodynia arise from central sensitisation within the CNS. Prostaglandin E2 (PGE2), generated through COX activation, is a major mediator of these processes. Repeated noxious stimulation leads to reduced activation thresholds and increased firing of primary afferents. The collective release of inflammatory mediators at the injury site, often described as the “inflammatory soup” (ATP, bradykinin, 5-HT, NE, PGE2, NGF, substance P), drives sustained sensitisation through intracellular signalling cascades (e.g., cAMP/PKA, PKC/DAG). The net effect is heightened nociceptor responsiveness, producing amplified pain perception even when stimulus intensity remains unchanged.
Most Effective OTC Analgesia
A Cochrane review examining the efficacy of OTC analgesics in acute pain4 concluded that simple, inexpensive, and widely


accessible analgesics provide clinically meaningful pain relief for many patients presenting with conditions such as dental pain, sprains, and strains. This is frequently described by the Number Needed to Treat (NNT), which is the average number of patients who need to be treated with a particular therapy for one additional person to achieve a defined beneficial outcome compared with a control (usually placebo). A lower NNT indicates a more effective treatment, e.g. an NNT of 2 means that for every two people treated, one will experience meaningful benefit who would not have benefited with placebo. Among OTC products available in Ireland, the most effective options for acute pain were combination ibuprofen/paracetamol formulations (400 mg/1,000 mg and 200 mg/500 mg), which relieved pain in approximately 70% of patients and demonstrated an NNT of <2. Fast-acting ibuprofen formulations were also highly effective, achieving pain relief in over 50% of patients with an NNT of 2–3. Paracetamol was less effective comparatively, benefiting around 40% of patients with an NNT between 3 and 5. Notably, the review found no evidence supporting the efficacy of lowdose codeine-containing products for acute pain.
The common advisory to take ibuprofen or other NSAIDs with food was also challenged in this review. NSAIDs are absorbed more rapidly on an empty stomach, which may enhance their analgesic effect. This has been reflected in updates to some product information leaflets: for example, the PIL for Nurofen Express Maximum Strength Tablets now advises administration with water only, without reference to food.9
Topical NSAIDs were also shown to provide substantial pain relief in acute conditions such as sprains, strains, and overuse injuries, with efficacy likely comparable to oral NSAIDs.10 A 2020 guideline developed by the American College of Physicians and the American Academy of Family Physicians11 similarly identified topical NSAIDs as among the most effective therapeutic options for acute, non–low back musculoskeletal pain in adults. Importantly, topical formulations did not demonstrate a significant increase in adverse effects. Diclofenac, ibuprofen, and ketoprofen gels achieved
significantly higher rates of ≥50% pain reduction than placebo, with diclofenac Emulgel showing the most favourable NNT (1.8). Diclofenac medicated plasters also performed well (NNT 3.2), while ibuprofen gel demonstrated an NNT of 3.9 based on marked improvement or complete symptom remission.10 Oral NSAIDs and paracetamol both offered pain reduction; however, the guidelines noted that oral NSAIDs were associated with increased gastrointestinal risk.11
Mechanisms of Action of Analgesics
A range of pharmacological classes are used in the management of acute and chronic pain, each acting through distinct molecular pathways that modulate nociceptive processing and inflammatory responses.
Paracetamol reduces prostaglandin (PG) synthesis from arachidonic acid through COX-1 and COX-2 inhibition and is thought to exert its primary analgesic effects centrally. Owing to its favourable tolerability and modest efficacy, it is widely used alone or in combination with NSAIDs.12
NSAIDs remain foundational in the treatment of many painful conditions, including osteoarthritis and chronic low back pain. They inhibit the COX-1 and COX-2 enzymes, which convert arachidonic acid into prostaglandins and thromboxanes. Their use is limited by gastrointestinal, renal, and cardiovascular risks, necessitating appropriate patient counselling and monitoring.
COX-2 selective inhibitors (e.g. celecoxib, etoricoxib) provide analgesia comparable to traditional NSAIDs with fewer gastrointestinal adverse effects but may increase cardiovascular risk with longterm use. COX-2 is expressed in inflammatory cells, damaged tissue, synovium, vascular endothelium, and the CNS.
Topical NSAIDs (e.g. diclofenac) are effective for localised chronic or acute musculoskeletal pain and are preferred when oral formulations are contraindicated because systemic exposure is significantly lower.
Opioids act as agonists at μ, δ, and κ receptors in the brain, spinal cord, and peripheral tissues. Key adverse effects include respiratory depression, sedation, nausea,

vomiting, and constipation. They are no longer recommended as first-line therapy for chronic primary pain; short- to mediumterm use may be appropriate in certain patients.13 Among patients with chronic pain receiving opioids, estimated rates of misuse range from 21–29%, and addiction from 12–18%.
For neuropathic pain, preferred therapies include certain antidepressants, antiepileptics, topical lidocaine, and topical capsaicin. In nociplastic pain (e.g. fibromyalgia), non-pharmacological approaches are prioritised, though TCAs, SNRIs, and gabapentinoids may provide benefit.4
Anticonvulsants (e.g. gabapentin, pregabalin, carbamazepine) are used for neuropathic pain conditions including diabetic neuropathy, post-herpetic neuralgia, and spinal cord injury pain.12 Gabapentinoids bind to the α2δ subunit of voltage-gated calcium channels, reducing calcium influx in hyperexcitable neurons. Carbamazepine is thought to act by binding to and stabilising inactive sodium ion channels, and modulating the release of glutamate and GABA.
Tricyclic antidepressants (e.g. amitriptyline, nortriptyline) exhibit analgesic effects independent of antidepressant activity, believed to stem from presynaptic inhibition of serotonin and noradrenaline reuptake.
SNRIs (e.g. duloxetine) are effective in diabetic neuropathy, fibromyalgia, osteoarthritis, and low back pain. SSRIs may be considered in fibromyalgia but generally lack evidence of efficacy in neuropathic pain.
Musculoskeletal agents such as baclofen and tizanidine are commonly used adjuncts. Baclofen, a GABA-B agonist, likely exerts its effects through inhibition of spinal reflexes, though the mechanism is not fully defined. Tizanidine (and clonidine) act as central α2adrenergic agonists producing muscle relaxation via presynaptic inhibition. This reduces dorsal horn neuronal activity and inhibits substance P release, producing analgesia and sedation.
Anxiolytics, particularly benzodiazepines, may help manage anxiety associated with acute pain flares but should be used only short-term;
longer-term anxiety management should shift to safer agents such as antidepressants.
Topical lidocaine patches (applied 12 hours on/12 hours off) are useful for localised nociceptive pain, neuropathic pain, and postherpetic neuralgia.
Ketamine, an NMDA receptor antagonist, provides analgesia at sub-anaesthetic doses by reducing central sensitisation and attenuating excitatory pain signalling.14 Evidence for its use in chronic or neuropathic pain remains limited. An RCT in cancer-related neuropathic pain found ketamine to be comparable with placebo, though subgroups—particularly those with features of central sensitisation—may benefit.15
Glucocorticoids (e.g. prednisolone) are potent anti-inflammatory agents that suppress gene transcription of pro-inflammatory mediators while promoting transcription of anti-inflammatory proteins. These effects are mediated by glucocorticoid receptor binding and nuclear translocation.16
Analgesia
A cross-sectional study from England and Ireland examined trends in prescription analgesic use between 2014 and 2022.17 Prescribing data for England were obtained from NHS sources, while Irish data were derived from the Primary Care Reimbursement Service (PCRS) for items dispensed under the General Medical Services (GMS) scheme. The analysis included opioids, systemic NSAIDs, paracetamol, topical analgesics, antimigraine medicines, and agents used for neuropathic pain.
In Ireland, analgesic dispensings per 1,000 GMS population increased from 3,388.6 in 2014 to 4,316 in 2022. Most individual opioids showed increased dispensing (with the exception of hydromorphone, dihydrocodeine, and tramadol). Use of topical analgesics, lowdose amitriptyline, gabapentin, pregabalin, and paracetamol also rose. In contrast, NHS data demonstrated a reduction in analgesic dispensings per 1,000 population over the same period. Despite these divergent trends, the proportional distribution of analgesic classes remained relatively stable across both settings. Codeine was the most commonly dispensed opioid in




England (48% of opioid items) and Ireland (45%), primarily for analgesic rather than antitussive use.
The World Health Organization defines the Defined Daily Dose (DDD) as the assumed average maintenance adult dose for a drug’s principal indication. Using this metric, Irish DDDs for all analgesics increased from 116 to 147.6 per 1,000 GMS population per day between 2014 and 2022; opioid DDDs alone increased from 33 to 40.3. The largest relative growth was observed for tapentadol (389%) and low-dose amitriptyline (194%). In England, DDDs declined from 97 to 80.6 per 1,000 population per day over the same timeframe. Subgroup analysis of English data showed substantially higher analgesic dispensing in areas of greater socioeconomic deprivation.
Some of the observed differences between countries reflect differences in the underlying populations sampled. Irish GMS data disproportionately represent older adults and individuals with socioeconomic disadvantage, i.e. two demographic groups known to experience higher rates of pain, whereas NHS data encompass a broader demographic. Evidence-based NICE guidance for chronic pain recommends first-line non-pharmacological interventions such as structured exercise, psychological therapies, acupuncture, and TENS. While these services are available within the NHS, access for GMS patients

in Ireland is often more limited, and waiting times can be prolonged, potentially contributing to higher reliance on prescribed analgesics.
The Organisation for Economic Co-operation and Development (OECD) incorporates opioid and NSAID prescribing within its 11 quality indicators for prescribing in primary care.18 Despite increasing opioid prescribing in Ireland, a recent study examining prescription drug–related harms from 2010 to 2020 found that opioid-related harm remained relatively stable over time.
OTC sales of low-dose codeine preparations are also substantial: an international comparison ranked Ireland and England second and fourth highest, respectively, among 31 countries.19
NICE guidelines for chronic pain management20 include the following recommendations:
A person-centred assessment — including biological, psychological and social contributors.
Non-pharmacological first-line therapies for chronic primary pain, such as supervised exercise programmes, psychological therapy (e.g. CBT), and, in some cases, acupuncture.
Cautious use of medications — with careful consideration of benefits vs harms, especially for opioids or long-term pharmacotherapy.

Importance of a shared care plan, ongoing re-evaluation, and involving patients in decision-making.
Multimodal analgesia, i.e. using more than one analgesic class in combination with nonpharmacological strategies, can enhance pain control while limiting reliance on any single agent and reducing the adverse-effect burden associated with higher doses of individual drugs.5 This synergistic approach is particularly valuable when addressing complex or persistent pain.
Musculoskeletal pain is frequently driven by muscle spasm. Thermal therapies can reduce muscle shortening associated with trauma or underlying neuromuscular or skeletal conditions.8 However, evidence supporting these modalities remains limited due to a lack of large, high-quality clinical trials.
Cryotherapy decreases bleeding, vasodilation, inflammation, oedema formation, and pain perception. The PRICE protocol (protection, rest, ice, compression, elevation) is commonly applied for acute sports injuries and some chronic presentations.
Heat therapy increases collagen extensibility, blood flow, metabolic rate, and facilitates resolution of subacute or chronic inflammation. Heat combined with stretching can reduce muscle contraction, joint
stiffness, and chronic inflammatory symptoms, improving mobility and function. Heat elevates the pain threshold and reduces muscle excitability. Optimal protocols for both heat and cryotherapy, including ideal duration and frequency, remain uncertain.21
Transcutaneous electrical nerve stimulation (TENS) operates via the “gate control theory,” whereby activation of Aβ fibres inhibits nociceptive transmission. Evidence supports its use in osteoarthritis and neuropathic pain, and it has been applied in postoperative pain, complex regional pain syndrome, phantom limb pain, peripheral nerve injury, and during pregnancy and labour. TENS is also a useful adjunct in acute orthopaedic injury and postoperative pain within a multimodal regimen.
Acupuncture, an invasive technique involving insertion of fine needles at defined points, must be performed by trained practitioners. While evidence does not show superiority over NSAIDs for certain pain conditions, acupuncture may benefit individuals with back or neck pain, knee pain associated with osteoarthritis, postoperative pain, and arthralgia related to aromatase inhibitor therapy.
Therapeutic exercise, including passive mobilisations, active or assisted movements, stretching, and relaxation techniques, can complement other pain management strategies. Early management of musculoskeletal pain prioritises immobilisation, compression, and cryotherapy. As pain subsides, gradual mobilisation prevents chronic muscle shortening and secondary pain generation.
Psychological factors significantly influence pain perception. Interventions such as cognitive behavioural therapy (CBT), education, reassurance, stressreduction training, and counselling can improve coping and reduce symptom burden. CBT, including digital/online formats, has demonstrated benefit in pain relief. Meta-analyses show that music therapy reduces anxiety and improves sleep in chronic illness, with additional evidence of pain reduction and decreased opioid requirements.5 Similarly, systematic reviews of aromatherapy report anxiolytic and analgesic effects.
References available on request







Scan to learn more





A standout event in the Irish pharmacy calendar, the Irish Pharmacy News OTC & Retail Pharmacy
Awards remain the only industry accolades dedicated to recognising and rewarding excellence within the over-the-counter (OTC) market.
Now firmly established, these Awards continue to provide a valuable platform to celebrate innovation, showcase best-in-class products, and highlight the brands that are delivering real value to both pharmacists and patients. Since their launch in 2008, they have consistently reflected the evolving needs of the pharmacy sector—recognising advancements in product development, consumer engagement, and education.
Community pharmacy continues to play a central role in the delivery of consumer health. As the most trusted and accessible healthcare setting, pharmacies offer far more than product supply—they provide expert advice, personalised care, and a vital link between patients and safe, effective treatment options. In an increasingly complex health landscape, pharmacists remain a key source of guidance, helping patients navigate a growing range of OTC medicines, supplements, and wellness products with confidence.
The OTC market in Ireland continues to evolve, shaped by changing consumer behaviours, digital influence, and growing demand for self-care solutions. While patients are more informed than ever, they are also increasingly self-managing conditions—making the role of the pharmacist in guiding safe and appropriate product use more important than ever.
According to IQVIA data, the Irish OTC market remains a significant and competitive sector, with strong contributions from key categories including pain relief, cough, cold and respiratory products, vitamins and minerals, digestive health, and habit treatments. Seasonal demand, shifting consumer priorities, and ongoing public health challenges continue to influence performance across these categories.
Innovation remains critical. As external retail channels and online platforms expand their reach, pharmacy continues to differentiate itself through expertise, trust, and clinical support. The products recognised in this year’s Awards demonstrate how manufacturers are responding to these challenges—through improved formulations, targeted solutions, and enhanced consumer education.
Over the following pages, we showcase the winners of the 2026 OTC & Retail Pharmacy Product Awards—those products that stood out to our judging panel across a wide range of categories, from Best Beauty Product and Best Baby Care Product to Best Sleep & Stress Solution, Best Digestive Health Product, and Best Marketing & Training Campaign.
These Awards highlight not only product innovation, but also the importance of effective
education, strong brand support, and meaningful engagement with pharmacy teams. In a market where choice is vast, these elements are key in supporting pharmacists to make informed recommendations and deliver value to their customers.
While the OTC landscape continues to shift, one constant remains: the critical role of pharmacy. As patients increasingly turn to self-care and seek solutions outside of traditional healthcare settings, pharmacists are uniquely positioned to ensure that this care is safe, appropriate, and effective.
The OTC Awards serve as a reminder of the strong partnership between manufacturers and pharmacy, and the shared goal of improving patient outcomes through accessible, high-quality healthcare solutions.





Best Baby Skincare Product was won by Child’s Farm Baby OatDerma Moisturiser, who were also the winners of this category last year.
Childs Farm’s OatDerma™ three-step routine— OatDerma Body Wash (250ml), Bubble Bath (250ml), and Body Moisturiser (200ml)—is designed to gently cleanse, soothe, and hydrate dry, itchy, and sensitive skin. Each fragrance-free formula is enriched with colloidal oatmeal, sustainably sourced from Finland, known for its soothing, moisturizing, and barriersupporting properties.
The quick-rinse Body Wash and gentle Bubble Bath cleanse effectively without stripping, while the Body Moisturiser provides intensive hydration to relieve dryness and maintain skin comfort. Mild enough to be comparable to water, the range is suitable for newborns and up, clinically proven to support the skin barrier, and approved by dermatologists and paediatricians.
“A well-trusted brand in the baby wash space, no complaints. The re-branding looks well and oats are definitely having a moment so it’s a clever pivot”
“Great product with lovely packaging. Always well supported within pharmacy for activations and promotions. great knowledge and training behind the brand. good advertising on TV” Judges Comments









Medicare Gel Arch Wrap won the 2026 title of Best Footcare Product.
The new Medicare Gel Arch Wrap is a discreet, comfortable footcare solution designed to support individuals experiencing arch pain and plantar fasciitis. Containing medical-grade, shockabsorbing Biogel®, the wrap cushions the arch, redistributes pressure, and helps alleviate discomfort caused by prolonged standing, walking, or repetitive strain.
Designed for everyday wear, the slim and flexible wrap fits comfortably inside footwear, offering targeted support without bulk. The gel pad provides consistent cushioning directly where it is needed most, while the soft fabric wrap ensures a secure and comfortable fit throughout the day.
“The Medicare Gel Arch Wrap provides targeted cushioning and support for customers experiencing arch discomfort or foot fatigue. It is soft, flexible, and easy to use in everyday footwear. It complements a pharmacy footcare and pain relief range well. The packaging is professional and easy to read”
“Excellent Irish owned product with a brand that is known and trusted in pharmacies over the decades. Comfortable product. Plantar fasciitis is something I am asked about more often in the pharmacies, so good niche in the market. Suitable for all day use and well marketed on blogs and social media. Excellent product” Judges comments

Alleviates metatarsal pain, corns, calluses and more.






Alleviates pain and applies support for plantar fasciitis.

Reduces pressure on the toes and metatarsals.







Corrects curled toes, bunions and supports toe fractures.




Our Medicare® Gel Arch Wrap is designed to provide targeted relief for those suffering from foot discomfort, including plantar fasciitis, flat arches, and cavus foot. The shock-absorbing Biogel® reduces pressure and alleviates pain by supporting the arch.




Kinvara 24HR Rosehip Face Serum won the 2026 Best Natural Product Award.
The Kinvara 24HR Rosehip Face Serum is a mega calming hydrator designed to support healthy, radiant and comfortable skin throughout the day and night. Made in the west of Ireland, it blends carefully selected plant oils and botanical extracts to help hydrate, nourish and protect the skin while remaining gentle enough for everyday use.
At the heart of the formula is rosehip oil, naturally rich in vitamins and essential fatty acids that help improve skin tone, smoothness and elasticity. This is combined with sea buckthorn to support skin vitality and antioxidant protection, and aloe vera to soothe and calm, making the serum suitable for all skin types, including sensitive and easily irritated skin.
“This is a beautiful product; it has really eye catching, feminine packaging and fantastic key ingredients suitable for most skin types and ages. It’s smooth and makes skin feel like silk once applied. Also enjoys great longevity”
“A huge fan of this brand. I think the training, ethos and brand message is beautiful and fits the Irish Skincare market perfectly. So much in-store support is given by the brand. We have a lot of returning customers, including our members of staff hooked to the whole line of Kinvara”





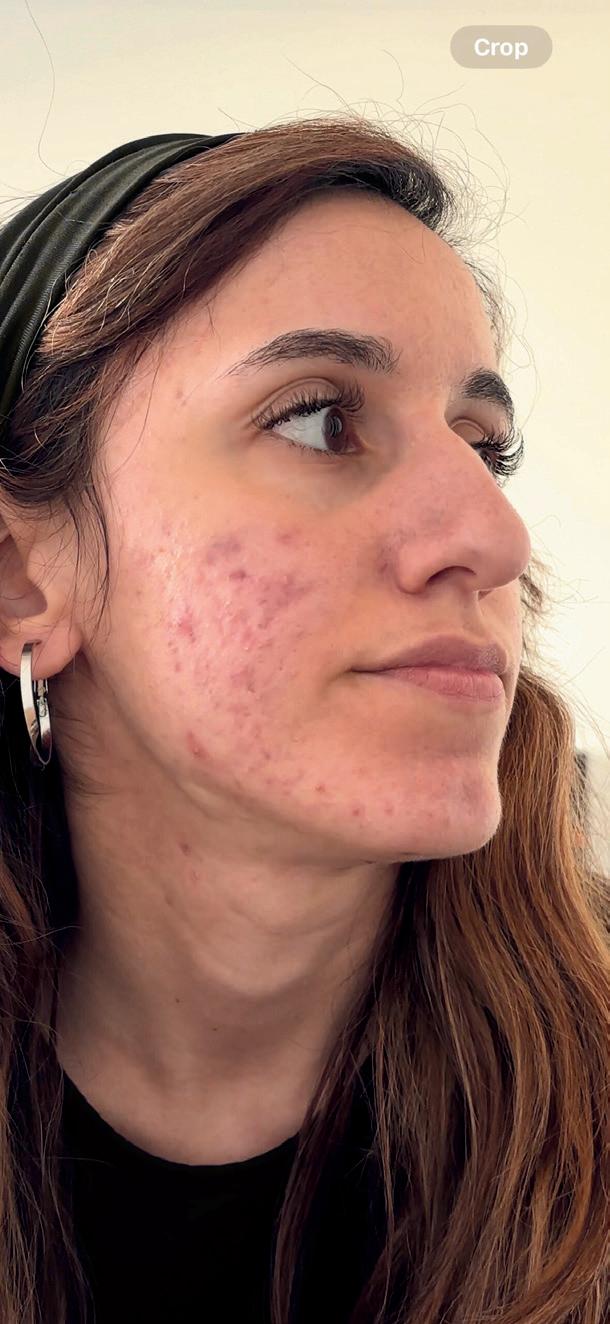





of customers would recommend Kinvara to a friend*

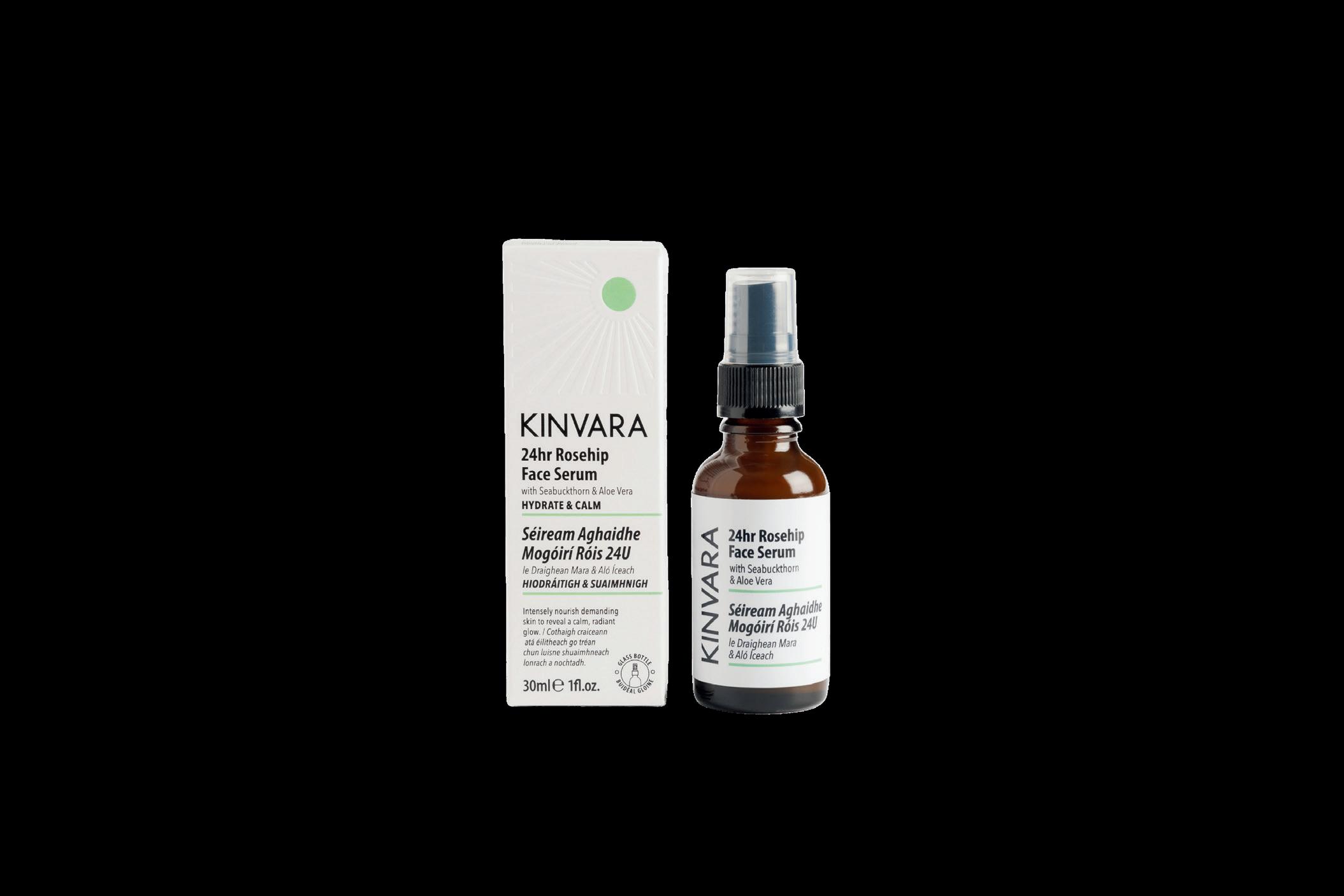
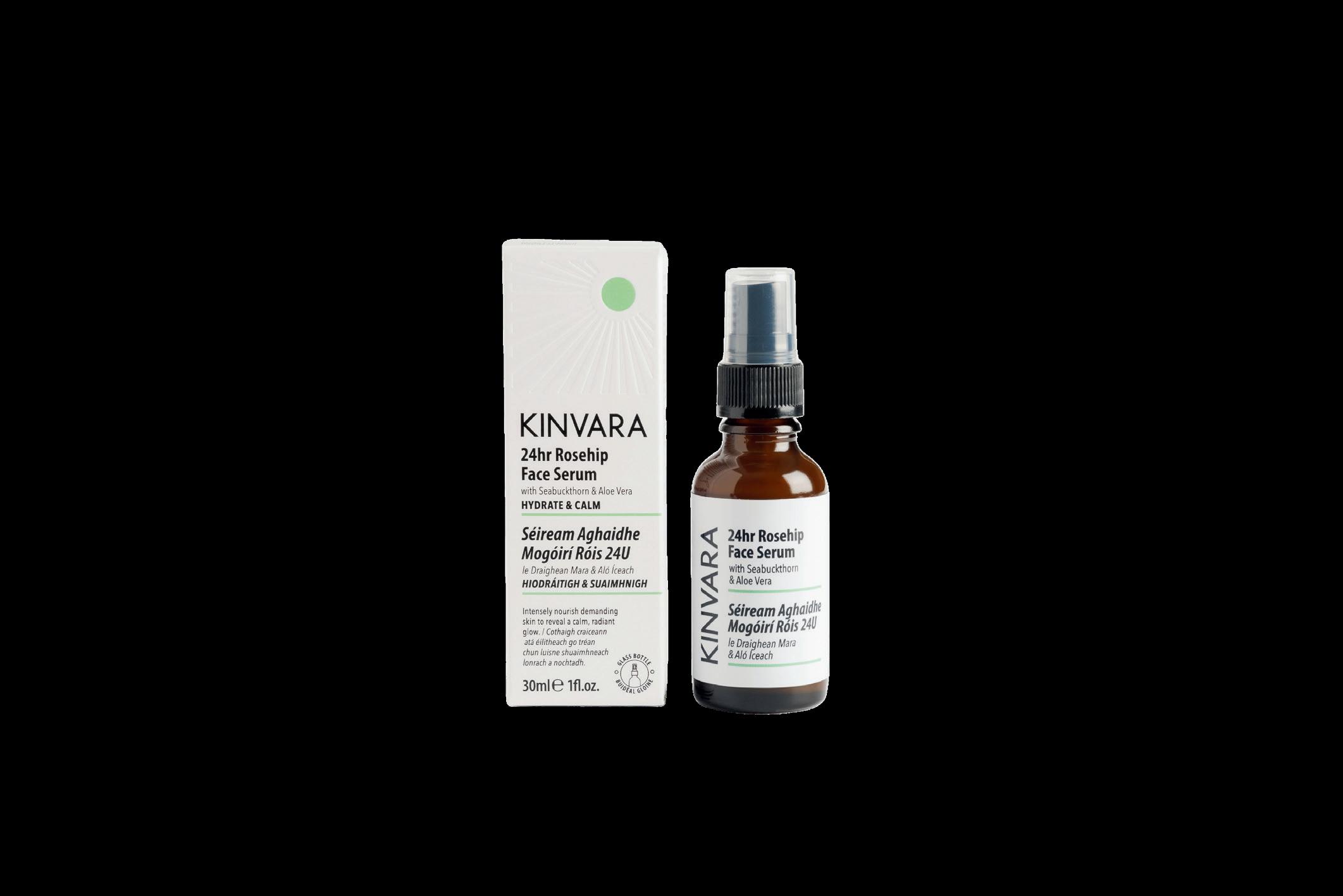






Optibac Babies & Children takes the 2026 Award for Best Children and Baby Supplement.
Optibac Babies & Children is a premium probiotic for digestive health and wellbeing, from the brand of friendly bacteria supplements most trusted by parents†. Expertly formulated for babies, toddlers and children aged 0-12, this supplement contains 3 strains of friendly bacteria including the world’s most scientifically studied strain, L. rhamnosus GG. It also contains added Vitamin D3 and prebiotic FOS fibres, offering little ones all-round gut and immune support.
†Optibac is the brand of friendly bacteria supplements most trusted by parents in Ireland. See website T&Cs for survey details, 500 people, August 2025.
“Optibac Babies & Children is a well-formulated probiotic designed specifically for young children, which supports confident pharmacy recommendation. Overall, it is a trusted, pharmacy-suitable product that I would be happy to stock and recommend to customers”
“Great Brand and trusted well-known brand. Consumer offers work well for this brand in pharmacy” Judges Comments











CALPOL® Vapour Plug won the Award for Best Children and Baby Product.This was the second consecutive title for Calpol Vapour Plug in this category.
CALPOL® Vapour Plug & Nightlight releases soothing lavender and camomile vapours that comfort your child through the night and help with clear & easy breathing. Its soft nightlight provides a gently reassuring glow, as well as guiding you in the room so you can avoid disturbing your child while they rest.
“Very useful and innovative product to help with restfulness at night. Excellent soothing aroma, a well branded and marketed product. Good packing information and material. And being suitable from a very young age is highly beneficial”
“This is a fantastic product with good and clear packaging . It works great and customers have reported its ease of use. Provides great relief to children. This is a trusted brand which parents love. Good marketing of this product with the range”
Judges Comments




The 2026 Award for Best Marketing/ Launch Initiative went to Nuromol Dual Action Pain Relief.
Nuromol Dual Action Pain Relief, from the makers of Nurofen, is redefining OTC pain relief in Ireland. Launched in August 2025, its patented technology delivers faster, longer-lasting, and more effective relief than standard painkillers alone. Unmissable in-store and backed by bold outdoor campaigns, Nuromol exemplifies Nurofen’s commitment to driving pharmacy footfall through innovation and impactful media. A true game-changer in 2025.

Nuromol Dual Action Film-Coated Tablets contain paracetamol 500 mg and ibuprofen 200 mg for short-term symptomatic treatment of mild to moderate pain. This product is especially suitable for pain which has not been relieved by ibuprofen or paracetamol alone. For use in adults aged 18 and older.

“The Reckitt brand and Nurofen is widely well known and loyalty is still very high despite other brands available. The launch of Nuromol has been very effective with lots of support from the manufacturer and very effective advertising. The demand has increased for the product and repeated sales have been seen. It is unique and we see it doing well compared to another brand on the market”
“Highly recognised global brand and trusted by consumers. Significant investment in promoting the launch of Nuromol - both across all media channels and with prominent instore activations and training has maximised awareness of this new innovative product” Judges comments











MACU-SAVE Junior+ won the 2026 Award for Most Innovative Product.
MACU-SAVE Junior+ is a next-generation Irish food supplement created to support children’s eye health in today’s digital world, while delivering benefits far beyond vision alone. Expertly formulated with Omega-3 Fish Oil, Lutein, Zeaxanthin, and Vitamin D, it provides vision support, with added benefits for normal brain function, and the normal functioning of the immune system, as well as the maintenance of healthy bones and teeth. What truly sets
MACU-SAVE Junior+ apart is its use of patented Concordix® technology, designed for the body to enhance nutrient bioavailability, delivering 44.9% greater nutrient absorption compared. This helps children receive maximum benefit from every dose, supporting both short and long-term health outcomes.
“Brilliant product that includes benefits for eyes, their brain function, immunity (Bones & Teeth). It includes it all without having to take more than one product”
“Very new product; from an innovation perspective it is a good combination product which could benefit children, strong packaging”
Judges Comments




Once again, Elave took the win for the Best Skincare Product category with Elave Intensive Cream.
Elave Intensive Cream is a certified medical device treatment formulated to restore and protect the skin’s natural barrier, providing lasting relief for dry, itchy, ultra-sensitive and eczema- or dermatitisprone skin. Developed by Gardiner Family Apothecary, an Irish, family-owned pharmaceutical skincare company with over 90 years of dermatological expertise, the cream reflects rigorous scientific formulation and a deep understanding of sensitive-skin needs. Suitable for all ages—from newborns to older adults—it delivers safe, effective care for even the most reactive skin.

“A well-rounded product suitable for all ages. Excellent emollient and good value. A brand well recognised in pharmacy as being a reliable one and and a good overall maintenance product”
“Elave is very well known to customers; it's unique packaging makes it stand out on the shelf and is easily identifiable. The various sizes the product comes in gives great variety and choice to customers. The formulation is nongreasy and easily absorbed. Customer loyalty to the brand is exceptional and it is a product we are confident to recommend due to repeat sales and positive feedback from those who've tried it”













Pregnacare Max scooped the Award for Best Pregnancy/ Fertility Product 2026.
Pregnacare Max is an advanced prenatal supplement designed for mums-to-be who want maximum nutritional support throughout pregnancy. This expertly formulated dual-pack provides key vitamins, minerals, and essential Omega-3 DHA to help support maternal health and normal foetal development. Each daily serving includes 400µg of folic acid, incorporating the highly bioavailable form L-Methylfolate, along with 10µg of vitamin D, 500mg of calcium, and 300mg of Omega-3 DHA to contribute to normal brain and eye development in the foetus.
“The Vitabiotics range and Pregnacare range are well known and trusted. This is a more pocket friendly option and the Omega 3 makes this one their top option. There’s no effort to sell this product. Virtually sells itself” “Strong market leader - clear packaging and good investment in the target market”
Comments




Pregnacare® Conception with 21 nutrients including folic acid as recommended for all women trying to conceive.
Pregnacare® Max with over 19 nutrients including folic acid, calcium and omega 3.
2 Most recommended by midwives 2,3
Pregnacare® New Mum supporting nutritional requirements of new mothers including vitamin B12 and iron.

Helping to keep mums and babies healthy for over 30 years, Pregnacare® provides the recommended level of folic acid4 and vitamin D which is advised by the Department of Health.5 It also provides a range of micronutrients, and is supported by unique clinical research with mums-to-be.6,7 DURING AFTER





Valeo Healthcare, Merrywell Industrial Estate, Ballymount, Dublin 12. For general queries: 01 405 1500. Email: valeohealthcare@valeofoods.ie. 1. Journal of the American College of Nutrition, Vol.18, No.5, 487-489 (1999). 2. For more information on this research, please visit www.pregnacare.com/mostrecommended. 3. Based on a survey of 1000 midwives. 4. Folic acid contributes to maternal tissue growth during pregnancy. Pregnacare® has always contained 400mcg folic acid, the level recommended for all women from the start of trying to conceive until the 12th week of pregnancy. 5. The Department of Health recommends that all adults, including pregnant and breastfeeding women, should consider taking a daily supplement containing 10μg of vitamin D between September and March. (Source: www.NHS.uk). 6. Agrawal, R. et al. Prospective randomised trial of multiple micronutrients in women undergoing ovulation induction, Reproductive BioMedicine Online December 2011. 7. L Brough et al. Effect of multiple-micronutrient supplementation on maternal nutrient status, infant birth weight and gestational age at birth in a low-income, multi-ethnic population. British Journal of Nutrition (2010), 104, 437-445. For more information on the Vitabiotics range, contact your Valeo Healthcare Territory Manger Lisa Kelly 086 206 1449 Lisa.Kelly@valeofoods.ie Mike Peters 086 856 2077 Mike.Peters@valeofoods.ie Gary Considine 087 933 4227 Gary.Considine@valeofoods.ie Chris Ferncombe 086 033 8162 Chris.Ferncombe@valeofoods.ie






Alflorex® scooped the title of Best Digestive Product for the third year in a row
Alflorex® is supported by a robust clinical evidence base, including randomised controlled trials and large real-world studies demonstrating meaningful improvements in symptom severity and quality of life. Costing approximately ¤1 per day, Alflorex® is an accessible, evidence-based option for ongoing IBS management and is the best-selling probiotic in Ireland (source: IQVIA, 2025).Alflorex® is Ireland’s leading probiotic for irritable bowel syndrome (IBS), a chronic and relapsing condition affecting up to 1 in 7 people and frequently managed in community pharmacy. Taken once daily, Alflorex® helps manage IBS symptoms and reduce their recurrence, supporting sustained symptom control.
“Well trusted, well researched product. Great to have clinical evidence to back up the product when recommending to customers. Good price point for a unique product”
“Well established Irish product with consumers returning to repurchase frequently, Packaging clean and clear. Customer feedback is positive on its effectiveness” Judges comments



Also awarded
Best Gastrointestinal Product (2016 & 2017) and Best Natural Product (2018 & 2020) Scan to explore the clinical research behind each strain.




So proud to be recognised as Best Digestive Product 2026 — thank you for your continued trust and support.

Backed by over 20 years of microbiome research and the clinically studied Bifidobacterium longum 35624™ strain, Alflorex® delivers targeted digestive support where it’s needed most.

Talk to your Pamex representative to learn more
Email: info@pamex.ie | Phone: 094 9024000





Best Women’s Product was voted for by our esteemed judging panel and the winner for 2026 is Utipro Plus AF.
Utipro Plus AF is an antibioticfree option designed to help relieve cystitis symptoms and prevent recurrent infections. Its unique dual mode of action works in both the bowel and the bladder, offering a comprehensive approach to cystitis management without over-reliance on antibiotics. Clinical studies have shown that Utipro Plus AF can help reduce the need for antibiotics, ease cystitis symptoms, and lower the risk of recurrent infections. The formulation of ingredients is also recognised by the European Association of Urology (EAU) for its effectiveness.
“The marketing around this product has been effective, including the sponsorship of the ghosted podcast which focused on the right customer group. The fact that it doesn’t need to be held behind the counter is a big plus also”
“Great unique product for cystitis. Only real option available in pharmacy without antibiotics.”
Judges comments











It was another win for fabÜ SHROOMS MENO & PERI as they took the title of Best Menopause Product 2026, their second year running in winning this title.
fabÜ SHROOMS MENO & PERI is designed to help you reconnect with your vibrant, balanced self as you navigate perimenopause and menopause.
Powered by Lion’s Mane mushroom alongside essential vitamins and minerals, this multi-award-winning blend supports hormonal balance, helps ease fatigue and tiredness, and boosts mental clarity and focus -helping to reignite your mojo.
Formulated with seven carefully selected active ingredients, it works in harmony with your body’s natural rhythms to support both mind and body during this transition. By promoting energy, balance, and resilience, fabÜ SHROOMS MENO & PERI makes this transformative stage of life easier to embrace - helping you feel like you again.
“Very effective and impactful support behind the brand with social media campaigns, and pharmacy colleague training. Huge loyalty and trust in the brand from patients and pharmacists”
“The clear winner for me in this category, with many and varied options. Meno&Peri has become the one I come back to. The brand owner has delivered strong and distinctive marketing that truly engages, reaches, and educates customers about the product” Judges Comments




The beauty industry is fast-moving and highly saturated. Understanding changing consumer opinions and the behavioural indicators behind global trends is integral to securing the success of any company or brand within the space.
The Award was won by fabÜ SKIN-HAIR-NAILS GLOW.

fabÜ SKIN-HAIR-NAILS GLOW is a carefully crafted, multiaward-winning blend designed to nourish your hair, skin, and nails from within. Powered by functional Reishi mushroom and 12 active ingredients-including vitamins, amino acids, and zinc this formula supports collagen formation, protects cells from oxidative stress, and enhances your natural glow. With consistent use, it helps promote healthier, more radiant skin, thicker, fuller hair, and stronger nails. Tailored to support beauty from the inside out, fabÜ SKIN-HAIR-NAILS GLOW empowers you to look and feel your best, unlocking a luminous, confident version of you.
“The product features strong, impactful packaging that fits seamlessly within the Fabu range, maintaining brand consistency. Clear call-outs on the pack help communicate key benefits effectively, ensuring it stands out and is easy for shoppers to understand”
“A strong media presence, particularly on social platforms, along with other marketing and PR activity has seen the Fabu brand grow in recent years. Loyalty to this pharmacist-led brand continues to grow. Providing complimentary products to pharmacy staff completing the training is a great initiative as it gives teams the opportunity to see and feel the benefits they have learnt through training - giving even greater confidence when recommending the product to customers”



Judges comments








Scoring a hat-trick for the brand, fabÜ won the 2026 Award for Best Irish Pharmacy Brand.
fabÜ is a pharmacist-founded wellness brand creating science-led, plantpowered formulas that deliver real, measurable results. Developed by award-winning pharmacist Laura Dowling, each product is expertly formulated using carefully selected ingredients with clinical evidence to support customers through every stage of life.
The brand’s 10 nutritional supplement blends combine functional mushrooms, vitamins, minerals, and botanicals to support energy, balance, immunity, cognitive function, and overall wellbeing. In addition, fabÜ has expanded into intimate wellness with the launch of hello joy, a water-based lubricant, and hello healing, vaginal ovules designed to support comfort, confidence, and intimate health.
“FabU is still growing every year and is driven by strong marketing support as well as comprehensive training available. The product range has brought Mushroom therapies to the Pharmacy masses, and customers are now more confident in purchasing from the growing range that has been launched, with Core lines such as Meno & Peri being the backbone of sales volume”
“A fabulous range with very strong brand loyalty. Lovely packaging - contains all of the necessary information. Fantastic brand support to pharmacies. Laura is doing great work for female health in Ireland, it's a pleasure to watch her success”







5 stars
Based on 2112 reviews
blends Lion’s mane, vitamins, and minerals to support balance, energy, and clarity through perimenopause and menopause.
Helping you feel like yourself























Revive Active won the Best Immunity Product for 2026.
Revive Active, a super supplement, formulated with 26 active ingredients in one convenient daily sachet. It supports immune function, energy levels, heart health, and helps reduce fatigue.
Formulated using only high-quality, bioavailable ingredients, carefully measured to work in synergy so consumers can truly feel the benefits across multiple areas of health. Glutenfree, vegan & halal certified, it carries the Guaranteed Irish & Love Irish Food marks, and is both formulated and manufactured in Ireland at Revive Active’s dedicated facility in Mullingar.
Sold in over 1,500 pharmacies, many of which have instore advocates who use & recommend the product with confidence to their customers, a clear testament to the quality that many pharmacies are proud to stand behind.
“Revive Active has a really good blend of immune boosting ingredients. It is good as a pickme-up during and following flu like symptoms, recovery after surgery and illness due to the amino acids & as a boost for those suffering with fatigue.
It is my go to customer recommendation.”
“Price point is very affordable. Alot of repeat buyers for these products as they work really well. Packaging is very big which is great for catching the eye”
Judges Comments





Panadol Extra Soluble Tablets are the winners of the Best Pain Relief Product 2026.

Panadol Extra Soluble Tablets are clinically proven to fight tough pain and are consistently a best-selling pain relief product. Formulated with paracetamol (500 mg) and caffeine (65 mg), this powerful combination delivers enhanced pain relief compared to standard paracetamol alone. The soluble format ensures rapid absorption, providing fast and effective relief when you need it most. Whether it’s a severe headache, toothache, muscle ache, or fever, Panadol Extra Soluble Tablets help you get back to living life without pain holding you back.
Made in Ireland, Panadol Extra is produced at Haleon’s world-class facility in Dungarvan, Co. Waterford, the global home of Panadol, the world’s #1 pain relief brand (excl. US. Based on sales data).

“Who doesn't have Panadol in their house, work, car, handbag etc. Panadol Extra is just a faster and much enhanced pain relief product. It's a very trusted and reliable brand and Panadol Extra doesn't disappoint. The caffeine also helps and gives a burst of energy”
“Alot of customer loyalty for the brand and is hugely trusted”
Judges comments







The Best Sports Product was only introduced in 2025. Taking home the title for this year was AYA Electrolytes Plus.
AYA Electrolytes Plus is a premium hydration solution designed for athletes and active lifestyles. Formulated with optimal levels of electrolytes, magnesium, and essential minerals, it supports rapid rehydration, muscle function, and endurance during training and recovery.
The clean, low-sugar formula delivers effective hydration without unnecessary additives, making it suitable for daily use and high-performance sport. Trusted for quality, taste, and efficacy, AYA Electrolytes Plus aligns with pharmacy-led wellbeing and evidence-based nutrition. Its convenient format and Irish-focused innovation make it a standout choice for supporting performance, recovery, and overall hydration for professionals, patients, and athletes seeking reliable, pharmacy-approved sports nutrition solutions.
“Great tasting product. Low in sugar which is great. Supports hydration and recovery, ideal for athletes and the general population. easy use packaging. Stand out profile of electrolytes and minerals. This product is Irishowned with an excellent marketing strategy”
“It is refreshing to see an electrolyte that combines so many additional vitamins and magnesium. Low sugar is a good selling point, tastings in pharmacies would be useful. Excited to see the AYA name becoming a household brand”
Judges Comments
















BENYLIN® DAY & NIGHT came out on top within the 2026 Best Cough, Cold and Flu Product category.
BENYLIN® DAY & NIGHT tablets are the only cold/flu remedy to offer 24 hour relief with tablets specifically designed for Day and Night.
Each white tablet contains 500mg Paracetamol and 60mg Pseudoephedrine hydrochloride.
Each blue tablet contains 500mg Paracetamol and 25mg Diphenhydramine hydrochloride.
During the day the white tablet relieves fever and decongests the nose.
During the night the blue tablet helps relieve cold symptoms to aid a better sleep.
Each pack contains 12 ‘’day’’ tablets and 4 ‘’night’’ tablets.
“This is a gold star cold remedy for me, as it combines a day and night solution in one pack. I recommend products based on evidence of effectiveness and all the products in this are proven to provide symptomatic relief for colds”
“It has been a challenging Cold and Flu season, with many new and unusual symptom sets presenting. Benylin Night & Day Tablets have been a very strong seller this season due to their great combination of active ingredients as well as a recognised quality and great Brand Identity”





BENYLIN DAY & NIGHT TABLETS: Composition: Each white (day) tablet contains Paracetamol 500 mg, Pseudoephedrine Hydrochloride 60 mg. Each blue (night) tablet contains Paracetamol 500 mg, Diphenhydramine Hydrochloride 25 mg. Pharmaceutical Form: Day Tablet: Tablet; Night Tablet: Film coated tablet. Indications: For the short-term symptomatic treatment of nasal and sinus congestion associated with daytime symptoms of cold and flu such as pain, headache and/or fever when in combination with bedtime symptoms which are causing difficulty in getting to sleep. Benylin Day & Night is indicated in adult and adolescents aged 15 to 17 years. Dosage: Adults and Adolescents over 15 years: Four tablets should be taken daily. One white tablet (paracetamol and pseudoephedrine) to be taken every 4 to 6 hours during the day (one tablet in the morning, at midday and in the afternoon). Do not take more than 3 white day-time tablets in 24 hours. One blue tablet (paracetamol and diphenhydramine) to be taken at night. Do not take the night-time tablets during the day. Patients should consult their doctor or pharmacist if symptoms persist for more than 3 days or worsen. Not recommended for children and adolescents under 15 years. Elderly: Experience has indicated that normal adult dosage is usually appropriate. However, in frail, immobile, elderly subjects or in elderly patients with renal or hepatic impairment, a reduction in the amount or frequency of dosing may be appropriate. The maximum daily dose should not exceed 60mg/kg/day (up to a maximum of 2g paracetamol per day) in the following situations, unless directed by a physician: Weight less than 50kg, Chronic alcoholism, Dehydration, Chronic malnutrition. Hepatic impairment: In patients with hepatic impairment or Gilbert’s Syndrome, the dose should be reduced or the dosing interval prolonged. The daily dose should not exceed 2g paracetamol/day unless directed by a physician. Renal impairment: Caution should be exercised when administering Benylin Day & Night to patients with mild to moderate renal impairment. It is recommended, when giving paracetamol to patients with renal impairment, to reduce the dose and to increase the minimum interval between each administration to at least 6 hours unless directed otherwise by a physician. See SPC for full details: Contraindications: BENYLIN Day & Night is contraindicated in patients with: hypersensitivity to paracetamol, pseudoephedrine, diphenhydramine or any of the excipients listed in section 6.1. Concomitant use of other sympathomimetic decongestants, beta-blockers or monoamine oxidase inhibitors (MAOIs), or within 14 days of stopping MAOI treatment (see section 4.5). The concomitant use of MAOIs may cause a rise in blood pressure and/or hypertensive crisis. Cardiovascular disease including hypertension, Diabetes mellitus, Phaeocromocytoma, Hyperthyroidism, Closed angle glaucoma, Severe acute or chronic kidney disease /renal failure, Urinary retention in patients at risk of developing respiratory failure. Special warnings and precautions for use: Paracetamol should be administered with caution under the following circumstances: Hepatic impairment, Chronic alcoholism, Renal impairment (GFR ≤ 50ml/min), Gilberts syndrome (familial non-haemolytic jaundice), Concomitant treatment with medicinal products affecting hepatic function, Glucose-6-phosphate dehydrogenase deficiency, Haemolytic anaemia, Glutathione deficiency, Dehydration, Chronic malnutrition, Weight less than 50kg, Elderly patients. Hepatotoxicity at therapeutic dose of paracetamol. Cases of paracetamol induced hepatotoxicity, including fatal cases, have been reported in patients taking paracetamol at doses within the therapeutic range. These cases were reported in patients with one or more risk factors for hepatotoxicity including low body weight (<50 Kg), renal and hepatic impairment, chronic alcoholism, concomitant intake of hepatotoxic drugs and in acute and chronic malnutrition (low reserves of hepatic glutathione). Paracetamol should be administered with caution to patients with these risk factors. Caution is also advised in patients on concomitant treatment with drugs that induce hepatic enzymes and in conditions which may predispose to glutathione deficiency (see sections 4.2 and 4.9). Doses of paracetamol should be reviewed at clinically appropriate intervals and patients should be monitored for emergence of new risk factors for hepatotoxicity which may warrant dosage adjustment. Prolonged or frequent use is discouraged. Paracetamol overdose warning: Taking more than the recommended dose (overdose) may result in severe liver damage. In case of overdose, medical help should be sought immediately. Urgent medical attention is critical for adults as well as for children even if you do not notice any signs or symptoms. Patients should be advised not to take other paracetamol-containing products concurrently. Taking multiple daily doses in one administration can severely damage the liver. In such cases, medical assistance should be sought immediately. Caution should be exercised when using the product in the presence of severe hepatic impairment or moderate to severe renal impairment (particularly if accompanied by cardiovascular disease). The hazards of overdose are greater in those with non-cirrhotic alcoholic liver disease. Patients with hepatic disease should consult a doctor before use. In general, medicinal products contains paracetamol should be taken for only a few days without the advice of a physician or dentist and not at high doses. If high fever or signs of secondary infection occur or if symptoms persist for longer than 3 days, a physician should be consulted. Hypersensitivity reactions, including skin rashes, angioedema and anaphylaxis have been reported very rarely with paracetamol. Serious skin reactions such as acute generalised exanthematous pustulosis (AGEP), Stevens-Johnson syndrome (SJS) and Toxic epidermal necrolysis (TEN) and drug reaction with eosinophilia and systemic symptoms (DRESS), have been reported very rarely in patients receiving paracetamol. Patients should be informed about the signs of serious skin reactions and use of the drug should be discontinued at the first appearance of skin rash or any other sign of hypersensitivity. Ischaemic colitis: Some cases of ischaemic colitis have been reported with pseudoephedrine. Pseudoephedrine should be discontinued and medical advice sought if sudden abdominal pain, rectal bleeding or other symptoms of ischaemic colitis develop. Severe skin reactions such as acute generalized exanthematous pustulosis (AGEP) may occur with pseudoephedrine-containing products. This acute pustular eruption may occur within the first 2 days of treatment, with fever, and numerous, small, mostly non-follicular pustules arising on a widespread oedematous erythema and mainly localized on the skin folds, trunk, and upper extremities. Patients should be carefully monitored. If signs and symptoms such as pyrexia, erythema, or many small pustules are observed, administration of this medicine should be discontinued and appropriate measures taken if needed. Ischaemic optic neuropathy Cases of ischaemic optic neuropathy have been reported with pseudoephedrine. Pseudoephedrine should be discontinued if sudden loss of vision or decreased visual acuity such as scotoma occurs. Although pseudoephedrine has virtually no pressor effects in normotensive patients, this medicine should be used with caution in patients taking antihypertensive agents, tricyclic antidepressants or other sympathomimetic agents (such as appetite suppressants and amphetamine-like psychostimulants). The effects of a single dose on the blood pressure of these patients should be observed before recommending repeated or unsupervised treatment. Posterior reversible encephalopathy syndrome (PRES) and reversible cerebral vasoconstriction syndrome (RCVS). Cases of PRES and RCVS have been reported with the use of pseudoephedrine-containing products (see section 4.8). The risk is increased in patients with severe or uncontrolled hypertension, or with severe acute or chronic kidney disease/renal failure (see section 4.3). Pseudoephedrine should be discontinued and immediate medical assistance sought if the following symptoms occur: sudden severe headache or thunderclap headache, nausea, vomiting, confusion, seizures and/or visual disturbances. Most reported cases of PRES and RCVS resolved following discontinuation and appropriate treatment. This product is contraindicated for use in patients with pre-existing cardiovascular disease, particularly those with coronary heart disease and hypertension (see section 4.3). This product is contraindicated for use in patients with thyroid disease, diabetes, glaucoma and, severe renal impairment (see Section 4.3). Patients with difficulty in urination and/or enlargement of the prostate, should be advised to consult a physician before using pseudoephedrine. This product may act as a cerebral stimulant giving rise to hyperpyrexia, tremor and epileptiform convulsions. Care should be taken when used in epileptic patients. If any of the following occur, this product should be stopped: Hallucinations, Restlessness, Sleep disturbances. Use with caution in occlusive vascular disease. Pseudoephedrine may induce positive results in certain anti-doping tests. Diphenhydramine may enhance the sedative effects of central nervous system depressants. While taking this product, avoid alcoholic beverages and consult a healthcare professional prior to taking with central nervous system depressants. Patients with the following conditions should be advised to consult a physician before using diphenhydramine: a respiratory condition such as emphysema, chronic bronchitis, or acute or chronic bronchial asthma; prostate hyperplasia with urinary retention. Concomitant use of other products containing paracetamol or decongestants with Benylin Day & Night could lead to overdosage and should, therefore, be avoided. Night time tablets only: May cause drowsiness. Patients should be advised not to drive or operate machinery if affected. Alcoholic drink should be avoided. Night time tablets only: Patients should be advised not to use with any other product containing Diphenhydramine. The stated dose should not be exceeded. Use in patients with congenital long QT-syndrome should be avoided. Cases of high anion gap metabolic acidosis (HAGMA) due to pyroglutamic acidosis have been reported in patients with severe illness such as severe renal impairment and, sepsis, or in patients with malnutrition or other sources of glutathione deficiency (e.g. chronic alcoholism) who were treated with paracetamol at therapeutic dose for a prolonged period or combination of paracetamol and flucloxacillin. If HAGMA due to pyroglutamic acidosis is suspected, prompt discontinuation of paracetamol and close monitoring is recommended. The measurement of urinary 5-oxoproline may be useful to identify pyroglutamic acidosis as underlying cause of HAGMA in patients with multiple risk factors. Close monitoring, including measurement of urinary 5-oxoproline, is recommended. The concurrent use of medicinal products, which also prolong the QT interval or result in hypokalemia should be avoided. Immediate medical advice should be sought in an event of an overdose, even if you feel well. Please read the enclosed leaflet carefully (label). Immediate medical advice should be sought in an event of an overdose, because of the risk of irreversible liver damage (leaflet). Do not take with any other paracetamol containing products. Do not use with any other product containing diphenhydramine, even one used on skin. If symptoms persist or get worse, or if new symptoms occur, patients should stop use and consult a physician. Use when only clearly necessary. This medicine contains less than 1 mmol sodium (23 mg) per dose, that is to say essentially ‘sodium-free’. Undesirable effects: Immune system disorders: Not known Anaphylactic reaction, hypersensitivity. Investigations: Not known Blood pressure increased; transaminases increased. Psychiatric disorders: Not known Anxiety, confusional state, euphoric mood, hallucination, irritability. Nervous system disorders: Not known: Agitation, coordination abnormal, convulsion, headache, paraesthesia, psychomotor hyperactivity, sedation, sleep disturbances, tremor, somnolence cerebrovascular accident, Posterior Reversible Encephalopathy Syndrome, Reversible Cerebral Vasoconstriction Syndrome.. Eye disorders: Not known Blurred vision, Ischaemic optic neuropathy. Ear and labyrinth disorders: Not known Tinnitus. Cardiac disorders: Not known Palpitations, tachycardia, arrhythmia. Vascular disorders: Not known Hypotension. Respiratory, thoracic and mediastinal disorders: Not known Chest discomfort, dry throat, nasal dryness, dyspnoea. Gastrointestinal disorders: Not known Abdominal pain, constipation, diarrhoea, dyspepsia, vomiting Ischaemic colitis. Skin and subcutaneous tissue disorders: Not known Rash, pruritus, rash pruritic, urticaria, Angiodema, Serios skin reactions, including acute geralised exanthematous pustulosis (AGEP). Renal and urinary disorders: Not known Urinary retention, dysuria. Blood and the lymphatic system disorders: Not known Blood disorders, blood dyscrasias such as thrombocytopenia and agranulocytosis have been reported following paracetamol use, but were not necessarily causally related to the drug. Hepato-biliary disorders: Not known Liver dysfunction. Metabolism and nutrition disorders: Not known - High anion gap metabolic acidosis. MAH: JNTL Consumer Health I (Ireland) Ltd. Block 5, Hight Street, Tallaght, Dublin 24, Ireland. MA Number: PA23490/006/001. Date of revision of text: March 2025. Product not subject to medical prescription. Supply through pharmacies only. Full prescribing information available upon request. IE-BYL-2026-308038






The title of Best Sleep/Stress Product goes to Nytol Once-A-Night.
Nytol remains the #1 sleep aid brand in Ireland*, available exclusively through pharmacies to help people achieve a restful night’s sleep. Quality sleep is a cornerstone of overall health, playing a vital role in immune function and well-being.
Nytol One-A-Night Tablets are specifically formulated to address temporary sleep difficulties in adults, supporting the entire sleep cycle: helping you fall asleep smoothly, improving sleep quality, and promoting a refreshed wake-up.
Nytol One-A-Night Tablets contain 50 mg of diphenhydramine hydrochloride as the active ingredient. Diphenhydramine is an antihistamine with sedative properties that works by blocking histamine receptors in the brain, helping to reduce alertness and promote drowsiness. This makes it effective for short-term relief of temporary sleep disturbances in adults.
“Nytol has performed really well since it launched; it was launched very well with outdoor campaigns and activations in store to drive customer awareness”
“On-Counter investments and product placement is really pivotal. The brand is well recognised and its effectiveness is widely known” Judges Comments





*Based on IQVIA sales data MAT 12/2025. Nytol One-A-Night 50 mg Tablets contains diphenhydramine hydrochloride. A symptomatic aid to the relief of temporary sleep disturbance in adults. Adults: One tablet to be taken 20 minutes before going to bed, or as directed by a physician. Do not exceed the maximum dose of one tablet in 24 hours. Elderly patients or patients with liver or kidney problems should consult their doctor before taking this medicine. Children under 18 years: Not recommended. The product should not be taken for more than 7 days without consulting a doctor. Contraindications: hypersensitivity to the active substance or to any of the excipients, stenosing peptic ulcer, pyloroduodenal obstruction, phaeochromocytoma, known acquired or congenital QT interval prolongation, known risk factors for QT interval prolongation. Special warnings and precautions: pregnancy/lactation, renal and hepatic impairment, myasthenia gravis, epilepsy or seizure disorders, narrow-angle glaucoma, prostatic hypertrophy, urinary retention, asthma, bronchitis, COPD. Patients should be advised to promptly report any cardiac symptoms. Tolerance and / or dependence may develop with continuous use. Do not take for more than 7 consecutive nights without consulting a doctor. Should not be used in patients currently receiving MAO inhibitors (MAOI) or patients who have received treatment with MAOIs within the last two weeks. Use in the elderly should be avoided. Avoid concomitant use of alcohol or other antihistamine-containing preparations. Do not drive or operate machines. Cases of abuse and dependence were reported in adolescents or young adults for recreational use and/or in patients with psychiatric dis-orders and/or history of abuse disorders. Contains lactose. May suppress the cutaneous histamine response to allergen extracts and should be stopped several days before skin testing. Interactions: Alcohol, CNS depressants, MAO inhibitors, anticholinergic drugs (e.g. atropine, tricyclic antidepressants), metoprolol and venlafaxine, CYP2D6 inhibitors, Class Ia and Class III anti-arrhythmics. Side effects: dry mouth, fatigue, sedation, drowsiness, disturbance in attention, unsteadiness, dizziness, thrombocytopenia, hypersensitivity reactions, confusion, paradoxical excitation, convulsions, headache, paraesthesia, dyskinesias, blurred vision, tachycardia, palpitations, thickening of bronchial secretions, gastrointestinal disturbance, muscle twitching, urinary difficulty, urinary retention. Product not subject to medical prescription. PA1186/016/001. MAH: Chefaro Ireland DAC. The Sharp Building. Hogan Place. Dublin 2. Ireland. Date of preparation: Nov. 2023. SPC: https://www.medicines.ie/medicines/nytol-one-a-night-50-mg-tablets-34889/spc MAT-10247

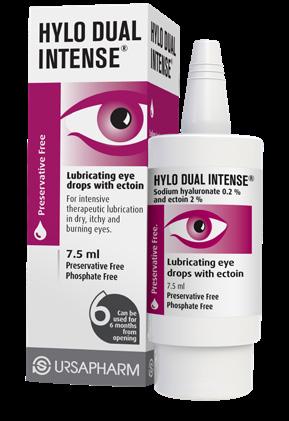




Scope Healthcare’s HYLO DUAL INTENSE® won the Best Eye Care Product for 2026.
HYLO DUAL INTENSE® is the next generation eye drop for the management of symptoms of dry eye disease, providing patients with instant, long-lasting relief.
HYLO DUAL INTENSE® contains two dual-acting ingredients that work together to break the vicious cycle of dry eye. Working in combination with 2% Ectoin® osmoprotectant, it also contains 0.2% High Molecular Weight Hyaluronic Acid, providing patients with a preservative-free & phosphate-free formulation that gives superior hydrating relief.
HYLO DUAL INTENSE® is conveniently presented in the patented COMOD bottle which offers patients precise dosing with a guaranteed 225 drops and 6 months sterility from first opening.
“Really like how this product can be kept open for six months. Very good value for money considering that. The type of container that it is stored in really lends itself to easy administration of the drops”
“Hylo Dual Intense Preservative-free Eye Drops are truly exceptional! This product stands out as a top-tier solution for dry, irritated eyes, providing long-lasting relief and comfort. The preservative-free formula is gentle on the eyes, making it an ideal choice for those with sensitive eyes or for those who wear contact lenses. It’s one of the few eye drops I’ve used that provides immediate hydration and relief without any stinging or discomfort.”
Judges Comments


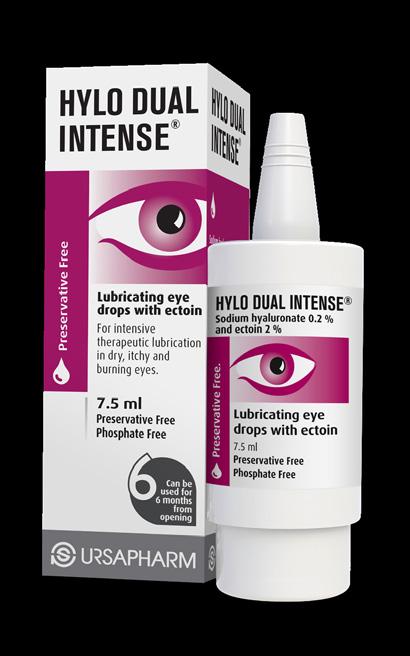









TePe Interdental Brushes scooped the Award for Best Oral Care Product.
TePe Interdental Brushes are designed to do what a regular toothbrush cannot: clean effectively between the teeth, where plaque builds up and gum disease often begins. TePe Interdental Brushes combine clinical effectiveness, comfort, and high-quality design to support optimal daily oral hygiene.
Available in seven sizes, the range ensures a precise fit for every interdental space. Correct sizing is essential for effective plaque removal, and TePe’s colour-coded system makes it easy to choose the right brush. The flexible yet stable handle provides excellent grip and control, allowing gentle, controlled movements for safe and thorough cleaning.
Beyond performance, TePe is strongly committed to sustainability. Fossil-based raw materials have been replaced with fossil-free pine oil, reducing the carbon footprint of TePe Interdental Brushes by up to 80%—without compromising product quality, efficiency, or design.
“Trusted by dental professionals and customers around the world. Well recognised brand. Development to make the range more sustainable isvery positive. Packaging easy to differentiate and added benefit of customers being able to choose from a range of 7 products to ensure they have the right tool to maintain their oral health”
“TP Interdental Brushes (sizes 0–5) offer a comprehensive, clinically trusted solution for effective interdental cleaning, supporting improved gum health across a wide range of patient needs. The clear size differentiation with colour and professional packaging make selection straightforward for both pharmacy staff and consumers”





1965
Developed in collaboration with dental experts, TePe is the UK’s No. 1 selling Interdental Brush (IDB) and most recommended by dental hygienists.* IDB’s are highly effective in removing dental plaque between teeth and TePe offers a wide range in multiple sizes and handle lengths to suit everybody.
We’re using a range of initiatives from raw materials to energy supply that reduces our IDBs carbon footprint by 80% – without
Get the details at tepe.com
*Source: A survey of 201 dental hygienists in the UK, Ipsos (2019)



Mark Saunders, Managing Director


Best VMS Product 2026 was awarded to O.R.S Hydration Tablets.
O.R.S Hydration tablets scientifically proven formula follows the World Health Organisations hydration guidelines. It contains the perfect blend of electrolytes, minerals and small level of glucose. Proven to hydrate you faster and keep you hydrated for longer than water alone. O.R.S Hydration is the fastest proven way to restore a healthy hydration level.
“This is a very popular rehydration product. They have adapted to the market by making a pleasant tasting rehydration product at a reasonable cost. Their packaging is simple and easy to follow.
“Nice to see a sport rehydration pack available for pharmacy sales. Pharmacys are the centrepoint for all things health focused, I would like to see pharmacies supporting more sporting supplements and gym supplements. Pharmacists are the ideal candidate to give advice on vitamins and proteins.













Formulated by Pharmacists, to follow World Health Organisation oral rehydration solution guidelines O.R.S Hydration Tablets contain a scientific blend of electrolytes glucose, salts and minerals for fast and effective hydration that tastes great.

Infant feeding is one of the most common and often sensitive topics raised in community pharmacy. For new and expectant parents, decisions around feeding can be influenced by a combination of medical advice, personal preference, practical challenges, and, increasingly, external pressures such as product availability.
Pharmacists are uniquely positioned to support parents during this time. As highly accessible healthcare professionals, pharmacy teams are often the first point of contact for reassurance, guidance, and product advice—particularly in the early weeks and months of a baby’s life.
In recent months, challenges around product availability have further highlighted the importance of clear, evidencebased advice. Parents may present feeling anxious, overwhelmed, or unsure about how to safely manage feeding changes. In these moments, the role of the pharmacist extends beyond product supply to providing calm, practical support.
Infant feeding decisions are highly individual. While breastfeeding is recommended as the optimal source of nutrition for infants, many families will use formula feeding either exclusively or in combination. Pharmacists should approach all conversations with sensitivity and without judgement, recognising that parents may already feel under pressure. A supportive, open approach helps build trust and encourages parents to seek advice when needed.
Understanding the different types of infant formula available is important in guiding appropriate recommendations. Standard infant formulas are suitable for most
babies, while specialist formulas may be indicated for specific conditions such as reflux, colic, lactose intolerance, or cow’s milk protein allergy.
However, it is important to note that not all feeding issues require a change in formula. In many cases, reassurance and practical advice can resolve concerns without the need for unnecessary product switching.
Pharmacy teams frequently encounter a range of feedingrelated concerns.
Colic and unsettled behaviour are among the most common reasons parents seek advice. While distressing for parents, these symptoms are often self-limiting. Pharmacists can provide reassurance, suggest feeding techniques, and advise on appropriate products where necessary.
Reflux is another frequent concern. Parents may report frequent regurgitation, irritability, or feeding difficulties. In mild cases, advice on feeding position, smaller and more frequent feeds, and winding techniques may be sufficient. More persistent or severe symptoms should be referred for medical assessment.
Constipation and changes in bowel habits can also prompt consultation. It is important to distinguish between normal variation and true constipation,

particularly in formula-fed infants. Pharmacists can advise on hydration, feeding practices, and when further investigation is required.
In some cases, symptoms may suggest cow’s milk protein allergy (CMPA), particularly where there are additional signs such as eczema, diarrhoea, or poor weight gain. These cases should always be referred for appropriate diagnosis and management.
Recent supply challenges have meant that some parents may need to switch between formula brands or types. This can be a source of significant anxiety, particularly for first-time parents.
Pharmacists play a key role in reassuring parents that, in most cases, switching between standard infant formulas is safe. These products are tightly regulated to ensure they meet the nutritional needs of infants.
When advising on switching, pharmacists should encourage a gradual transition where possible and monitor for any changes in tolerance. Clear guidance can help reduce unnecessary worry and prevent inappropriate or frequent switching between products. It is also important to address misinformation, particularly from online sources, which can sometimes lead to confusion or unsafe practices.
Often, the most valuable support pharmacists can offer is simple, practical guidance.
Advice on feeding techniques— such as correct bottle preparation, appropriate feeding volumes, and winding—can make a significant difference. Supporting parents in establishing feeding routines, where appropriate, can also help reduce anxiety.
Equally important is reassurance. Many parents worry about feeding patterns, particularly in the early weeks. Helping them understand what is normal can prevent unnecessary concern and build confidence.
Pharmacy teams should also be mindful of parental wellbeing. Feeding challenges can be stressful and exhausting, and taking the time to listen and offer reassurance can have a meaningful impact.
While many feeding issues can be managed within pharmacy, there are clear situations where referral is required.
These include:
• Poor weight gain or feeding refusal
• Persistent vomiting or severe reflux
• Blood in stools or ongoing diarrhoea
• Suspected allergy
• Parental concern that is not resolving with advice
Timely referral ensures that underlying conditions are identified and managed appropriately.
The
The role of community pharmacy in infant nutrition continues to evolve. Beyond product supply, pharmacists are increasingly recognised as a trusted source of advice and support for parents navigating early childhood.
This is particularly important in times of uncertainty, where clear, consistent messaging can help reduce anxiety and support informed decision-making.
By combining clinical knowledge with a compassionate, patientcentred approach, pharmacy teams can play a vital role in supporting families during one of the most important stages of life.
Infant feeding is rarely straightforward, and parents often require reassurance as much as guidance. Community pharmacy offers a valuable and accessible point of support, helping parents navigate challenges with confidence.
As demand for advice continues to grow, pharmacists have an opportunity to strengthen their role in this area—providing practical, evidence-based support that benefits both infants and their families.











We
leading the way in quality standards, and have introduced additional safeguards
We all know how important these products are to the families we serve. That’s why we’re mobilising all available resources across our global network to ensure continuity of supply.
We introduced extra safeguards and raw material verification steps to further strengthen our processes so that parents can continue to feel fully confident in the products they choose for their babies.
of breast milk. Parents should consider the social and financial implications of the use of infant formula. As babies grow at different rates, healthcare professionals should advise on the appropriate time for a baby to begin eating complementary foods. Infant formula and complementary foods should always be prepared, used and stored as instructed on the label in order to avoid risks to a baby’s health. The product should be used only on the advice of independent persons having qualifications in medicine, nutrition, pharmacy, or other professionals responsible for maternal and child care. SMA LF® is a lactose-free milk based formula for babies who are intolerant to lactose or sucrose, or who are experiencing symptoms such as diarrhoea, tummy ache or wind caused by temporary lactose intolerance. It is suitable as the sole source of nutrition up to 6 months of age, and in conjunction with solid food up to 12 months of age. The following products must be used under medical supervision. SMA® Comfort is a special formula intended for the
With the recent and ongoing events in Iran and across the wider Middle East, market volatility can feel uncomfortable in the moment. In over 20 years advising members of the pharmacy community we know that periods of decline, uncertainty, and sharp swings are not interruptions to the investment journey, they are the journey. The challenge for investors is not avoiding volatility, but understanding it and crucially responding appropriately. In most cases, the correct response is remarkably simple, do nothing.

The accompanying chart of the S&P 500, which is the 500 largest companies listed on U.S stock exchanges, makes this point exceptionally well. It shows that market declines occur with striking regularity. In almost every calendar year, markets experience a meaningful intra-year drop, often in the region of 10%–20%, and occasionally much more severe. These declines are not rare events they are a routine part of how markets function.
What is particularly informative about the chart is the contrast between intra-year declines and the final outcome for that same year. While markets frequently fall sharply during the year, sometimes by as much as 30%, the calendar-year return is often positive by year-end. For example, the chart shows major drawdowns during events such as the dot-com crash, the global financial crisis, and the COVID-19 pandemic. In some cases, intrayear declines reached as much as -34%, -49%, or -34%, respectively. Yet, despite these sharp falls, many years still finished with positive returns. Even in years with negative returns, recovery often followed quickly in subsequent periods.
The most compelling statistic in the chart is however the long-term trajectory. Despite these temporary setbacks, the S&P 500 rose from 1,148 in January 2002 to 6,878 by February 2026. This is not a smooth upward line, it is a jagged, volatile path marked by fear, uncertainty and frequent declines. If you are to take anything from this article, it should be that volatility is the price of admission for long-term returns and not to be feared.
Investors often mistake volatility for risk. In reality, volatility is simply
Written by Colm Moore CFP™, Moore Wealth Management
Colm Moore CFP™ and his firm, Moore Wealth Management, will be at the IPU National Pharmacy Conference in Mount Wolseley on the 9th May and can be contacted at colm@mwm.ie or via www.mwm.ie
movement, markets adjusting to new information, changing expectations and shifting economic conditions. True risk, particularly for long-term investors, is not shortterm declines but the possibility of missing out on long-term growth. Risk is most often realised when investors react to volatility rather than endure it.
This is where behaviour becomes critical. When markets fall, the instinctive reaction is to “do something”, to move to cash, reduce exposure, or wait for things to “settle.” But history shows that this is precisely the wrong approach. Markets do not send invitations when they are about to recover. The strongest days often occur very close to the worst days, and missing even a small number of these recovery periods can significantly damage long-term returns.
The chart reinforces this behavioural challenge. If an investor were to exit the market during each intra-year decline, say at -10%, -15%, or -20%, they would likely miss the rebound that often occurs before the year ends.
Doing nothing, therefore, is not passive, it is a deliberate and disciplined decision. It requires an understanding that short-term movements are largely noise in the context of a long-term plan. It requires confidence in the underlying resilience of markets and the global economy and requires acceptance that discomfort is part of the process.
There is also a deeper structural reason why doing nothing works. Markets are forward-looking. Prices adjust rapidly to new information, meaning that by the time an investor reacts, much of the movement has already occurred, often in microseconds. Attempting to time exits and re-entry is not just difficult, it is statistically improbable to do consistently well. The evidence overwhelmingly supports staying invested rather than trying to outmanoeuvre the market.
Another key idea here is the concept of average declines. The
data shows that the average intrayear drop is around 16%. This means that a double-digit decline is not an outlier, it is the norm. When investors experience a 10% or even 15% drop, it should not be viewed as a signal that something is fundamentally wrong. Instead, it is entirely consistent with historical patterns.
Framing is everything. If investors expect markets to rise smoothly year after year, volatility will always feel like a problem. But if you understand that declines are a regular and predictable feature, those same movements become far less concerning.
Successful investing is less about finding the perfect strategy and more about sticking with a good one. The temptation to act during periods of volatility is strong, but the evidence is clear: the best course of action is usually no action at all.
The investors who succeed are not those who avoid volatility, but those who understand it, accept it, and resist the urge to react to it. In a world where action is often equated with control, the real discipline lies in doing nothing.
For healthcare professionals in Ireland only. Abbreviated Prescribing Information can be found below.
Nintedanib Teva is indicated in adults for the treatment of idiopathic pulmonary fibrosis (IPF).
Nintedanib Teva is also indicated in adults for the treatment of other chronic fibrosing interstitial lung diseases (ILDs) with a progressive phenotype.
Nintedanib Teva is indicated in adults for the treatment of systemic sclerosis associated interstitial lung disease (SSc-ILD).
Nintedanib Teva Soft Capsules Abbreviated Prescribing Information. Presentation: Each soft capsule contains nintedanib esilate equivalent to 100 mg and 150 mg nintedanib. Indications: Nintedanib Teva is indicated in adults for the treatment of idiopathic pulmonary fibrosis (IPF); for the treatment of other chronic fibrosing interstitial lung diseases (ILDs) with a progressive phenotype; the treatment of systemic sclerosis associated interstitial lung disease (SSc-ILD). Dosage and administration: For oral use. Treatment should be initiated by physicians experienced in the management of diseases for which Nintedanib Teva is approved. Adults: The recommended dose is 150 mg nintedanib twice daily administered approximately 12 hours apart. The 100 mg twice daily dose is only recommended to be used in patients who do not tolerate the 150 mg twice daily dose. The recommended maximum daily dose of 300 mg should not be exceeded. Children: Nintedanib should not be used in children. Elderly: No overall differences in safety and efficacy were observed for elderly patients. No a-priori dose adjustment is required in elderly patients. Patients ≥75 years may be more likely to require dose reduction to manage adverse effects. Renal impairment: Adjustment of the starting dose in patients with mild to moderate renal impairment is not required. The safety, efficacy, and pharmacokinetics of nintedanib have not been studied in patients with severe renal impairment (<30 ml/min creatinine clearance). Hepatic impairment: In adult patients with mild hepatic impairment (Child Pugh A), the recommended dose of Nintedanib Teva is 100 mg twice daily approximately 12 hours apart. In patients with mild hepatic impairment (Child Pugh A), treatment interruption or discontinuation for management of adverse reactions should be considered. The safety and efficacy of nintedanib have not been investigated in patients with hepatic impairment classified as Child Pugh B and C. Treatment of patients with moderate (Child Pugh B) and severe (Child Pugh C) hepatic impairment with Nintedanib Teva is not recommended. Contraindications: Hypersensitivity to the active substance or to any of the excipients. Precautions and warnings: Serious cases of diarrhoea leading to dehydration and electrolyte disturbances have been reported in the post-marketing. Patients should be treated at first signs with adequate hydration and antidiarrhoeal medicinal products, e.g. loperamide, and may require dose reduction or treatment interruption. Nintedanib Teva treatment may be resumed at a reduced dose or at the full dose. In case of persisting severe diarrhoea despite symptomatic treatment, therapy with Nintedanib Teva should be discontinued. Nausea and vomiting are frequently reported gastrointestinal adverse reactions, with most patients experiencing mild to moderate intensity. If symptoms persist despite appropriate supportive care (including antiemetic therapy), dose reduction or treatment interruption may be required. The treatment may be resumed at a reduced dose or at the full. In case of persisting severe symptoms therapy with Nintedanib Teva should be discontinued. Cases of drug-induced liver injury have been observed with nintedanib treatment, including severe liver injury with fatal outcome. The majority of hepatic events occur within the first three months of treatment. Therefore, hepatic transaminase and bilirubin levels should be investigated before treatment initiation and during the first month of treatment with Nintedanib Teva. Patients should then be monitored at regular intervals during the subsequent two months of treatment and periodically thereafter, e.g. at each patient visit or as clinically indicated. Elevations of liver enzymes and bilirubin were reversible upon dose reduction or interruption in the majority of cases. If transaminase elevations > 3x ULN are measured, dose reduction or interruption of the therapy with Nintedanib Teva is recommended and the patient should be monitored closely. Once transaminases have returned to baseline values, treatment with Nintedanib Teva may be resumed at the full dose or reintroduced at a reduced dose which subsequently may be increased to the full dose. If any liver test elevations are associated with clinical signs or symptoms of liver injury, e.g. jaundice, treatment with Nintedanib Teva should be permanently discontinued. Alternative causes of the liver enzyme elevations should be investigated. Adult patients with low body weight (<65 kg), Asian and female patients have a higher risk of elevations of liver enzymes. Nintedanib exposure increased linearly with patient age, which may also result in a higher risk of developing liver enzyme elevations. Close monitoring is recommended in patients with these risk factors. Cases of renal impairment/failure, in some cases with fatal outcome, have been reported with nintedanib use. Patients should be monitored during nintedanib therapy, with particular attention to those patients exhibiting risk factors for renal impairment/failure. In case of renal impairment/failure, therapy adjustment should be considered. Vascular endothelial growth factor receptor (VEGFR) inhibition might be associated with an increased risk of bleeding. Patients at known risk for bleeding including patients with inherited predisposition to bleeding or patients receiving a full dose of anticoagulative treatment were not included in the clinical trials. Nonserious and serious bleeding events, some of which were fatal, have been reported in the postmarketing period (including patients with or without anticoagulant therapy or other medicinal products that could cause bleeding). Therefore, these patients should only be treated with Nintedanib Teva if the anticipated benefit outweighs the potential risk. Patients with a recent history of myocardial infarction or stroke were excluded from the clinical trials. Caution should be used when treating patients at higher cardiovascular risk including known coronary artery disease. Treatment interruption should be considered in patients who develop signs or symptoms of acute myocardial ischemia. The use of VEGF pathway inhibitors in patients with or without hypertension may promote the formation of aneurysms and/or artery dissections. Before initiating Nintedanib Teva, this risk should be carefully considered in patients with risk factors such as hypertension or history of aneurysm. Due to the mechanism of action of nintedanib patients might have an increased risk of thromboembolic events. Due to the
Teva Pharmaceuticals Ireland, Digital Office Centre Swords, Suite 101 - 103, Balheary Demesne, Balheary Road, Swords, Co Dublin, K67E5AO, Ireland.
Freephone: 1800 - 201 700 | Email: info@teva.ie
Product subject to prescription which may not be renewed (A)
mechanism of action of nintedanib, patients might have an increased risk of gastrointestinal perforations. Cases of gastrointestinal perforations and cases of ischaemic colitis, some of which were fatal, have been reported in the postmarketing period. Particular caution should be exercised when treating patients with previous abdominal surgery, previous history of peptic ulceration, diverticular disease or receiving concomitant corticosteroids or NSAIDs. Nintedanib Teva should only be initiated at least 4 weeks after abdominal surgery. Therapy with Nintedanib Teva should be permanently discontinued in patients who develop gastrointestinal perforation or ischaemic colitis. Very few cases of nephrotic range proteinuria with or without renal function impairment have been reported postmarketing. Histological findings in individual cases were consistent with glomerular microangiopathy with or without renal thrombi. Reversal of the symptoms has been observed after nintedanib was discontinued, with residual proteinuria in some cases. Treatment interruption should be considered in patients who develop signs or symptoms of nephrotic syndrome. Some cases of posterior reversible encephalopathy syndrome (PRES) have been reported postmarketing. PRES is a neurological disorder (confirmed with magnetic resonance imaging) which can present with headache, hypertension, visual disturbances, seizure, lethargy, confusion and other visual and neurologic disturbances, and can be fatal. PRES has been reported with other VEGF inhibitors. If PRES is suspected, nintedanib treatment must be discontinued. Administration of Nintedanib Teva may increase blood pressure. Systemic blood pressure should be measured periodically and as clinically indicated. Nintedanib Teva should not be used in patients with severe pulmonary hypertension. Close monitoring is recommended in patients with mild to moderate pulmonary hypertension. No increased frequency of impaired wound healing was observed in the clinical trials. Based on the mechanism of action nintedanib may impair wound healing. No evidence of QT prolongation was observed for nintedanib in the clinical trial programme. As some other tyrosine kinase inhibitors are known to exert an effect on QT, caution should be exercised when nintedanib is administered in patients who may develop QTc prolongation. Interactions: Nintedanib is a substrate of P-gp. If coadministered with Nintedanib Teva, potent P-gp inhibitors (e.g. ketoconazole, erythromycin or cyclosporine) may increase exposure to nintedanib. In such cases, patients should be monitored closely for tolerability of nintedanib. Management of adverse reactions may require interruption, dose reduction, or discontinuation of therapy with nintedanib. Potent P-gp inducers (e.g. rifampicin, carbamazepine, phenytoin, and St. John’s Wort) may decrease exposure to nintedanib. Selection of an alternate concomitant medicinal product with no or minimal P-gp induction potential should be considered. Only a minor extent of the biotransformation of nintedanib consists of CYP pathways, therefore, the likelihood of drug-drug interactions with nintedanib based on CYP metabolism is considered to be low. Co-administration of nintedanib with oral hormonal contraceptives did not alter the pharmacokinetics of oral hormonal contraceptives to a relevant extent. Co-administration of nintedanib with bosentan did not alter the pharmacokinetics of nintedanib. Pregnancy and lactation: Nintedanib may cause foetal harm in humans. Women of childbearing potential should be advised to avoid becoming pregnant while receiving treatment with Nintedanib Teva and to use highly effective contraceptive methods at initiation of, during and at least 3 months after the last dose of Nintedanib Teva. The efficacy of oral hormonal contraceptives may be compromised by vomiting and/or diarrhoea or other conditions where the absorption may be affected. Women taking oral hormonal contraceptives experiencing these conditions should be advised to use an alternative highly effective contraceptive measure. As nintedanib may cause foetal harm also in humans, it must not be used during pregnancy and pregnancy testing must be conducted prior to treatment with Nintedanib Teva and during treatment as appropriate. Female patients should be advised to notify their doctor or pharmacist if they become pregnant during therapy with Nintedanib Teva. If the patient becomes pregnant while receiving Nintedanib Teva, treatment must be discontinued and she should be apprised of the potential hazard to the foetus. Breast-feeding should be discontinued during treatment with this medicinal product. Effects on ability to drive and use machines: Nintedanib Teva has minor influence on the ability to drive and use machines. Patients should be advised to be cautious when driving or using machines during treatment with nintedanib. Adverse reactions: Thrombocytopenia, myocardial infarction, bleeding, pancreatitis, colitis, hyperbilirubinaemia, renal failure, PRES. Very Common: Diarrhoea, nausea, abdominal pain, vomiting, hepatic enzyme increased. Common: Weight decreased, decreased appetite, hypertension, alanine aminotransferase (ALT) increased, aspartate aminotransferase (AST) increased, gamma glutamyl transferase (GGT) increased, blood alkaline phosphatase (ALKP) increased, rash, headache. Consult the Summary of Product Characteristics in relation to other side effects. Overdose: There is no specific antidote or treatment for nintedanib overdose. Two patients in the oncology programme had an overdose of maximum 600 mg twice daily up to eight days. Observed adverse reactions were consistent with the known safety profile of nintedanib, i.e. increased liver enzymes and gastrointestinal symptoms. Both patients recovered from these adverse reactions. In the INPULSIS trials, one patient was inadvertently exposed to a dose of 600 mg daily for a total of 21 days. A non-serious adverse event (nasopharyngitis) occurred and resolved during the period of incorrect dosing, with no onset of other reported events. In case of overdose, treatment should be interrupted and general supportive measures initiated as appropriate. Legal category: POM. Marketing Authorisation
Number: PA22579/007/001-002. Marketing Authorisation Holder: Teva GmbH, Graf-Arco-Str. 3, 89079 Ulm, Germany. Job Code: MED-IE-00106. Date of Preparation: February 2026.
Adverse events should be reported. Reporting forms and information can be found at www.hpra.ie.
Adverse events should also be reported to Teva UK Limited on +44 (0) 207 540 7117 or medinfo@tevauk.com
Date of Preparation: March 2026 | Job Code: GEN-IE-00179
Further information is available on request or in the SmPC. Product Information also available on the HPRA website.
The OTC Counter Assistant of the Year Award recognises excellence in knowledge and service to retail customers. Nominations for the category can be made through self-nomination, by colleagues or by pharmacy business owners.
Judges will be looking to reward those who bring something extra to the consumers experience of the pharmacy setting within a community.

1. Clear demonstration of how the nominee/ counter assistant has successfully placed customer care at the forefront of their pharmacy.
2. The details and rationale for any specific initiatives developed by the nominee for customer care.
3. Levels of excellence displayed above and beyond that expected from a counter assistant role.
4. Clear evidence of team working and excellent communication between the individual, their peers/colleagues and their patients and local community.
* Open to all pharmacies with more than 3 stores

Applications are sought from those demonstrating clear enthusiasm and commitment to the enhancement of community pharmacy in Ireland, exceptional quality above and beyond what is expected and an ability to overcome challenges in pursuit of goals.
1. Activities that may involve pioneering new models or systems that improve pharmacists’ impact as members of the healthcare team; patient safety and outcomes; patient care in general and other professional development.
2. Development of a system or tool for pharmacy that will directly or immediately impact patient care or the profession and/or serve as an example or template for other pharmacy professionals to follow.
3. Measurable benefits of your initiative. Please use financial data wherever possible (percentages, rations, graphic images etc.), as well as other statistics to show how your project has brought added value, profits, customer satisfaction improvements, productivity increases or any other benefit relevant to this award category.
Applications Close - Friday, April 10th, 2026 Send your entries to - kelly-jo@ipn.ie
Download application forms from: https://irishpharmacyawards.ie/
* Open to all pharmacies with less than 3 stores

Judges will look for a business initiative that stands out in terms of its scale, scope or approach. This Award will credit the company that has demonstrated excellent levels of customer service b acked by clear standards and adequate monitoring performance.
Awards Criteria:

* Open to all pharmacies with more than 3 stores
Entries must showcase a strong commitment to patient-centred care, innovation in service delivery, and positive customer feedback.
Mystery Shopper Assessment:
1. Clear demonstration of an identified need and personal rationale along with details of the process of implementation from concept to design, planning and results.
2. Evidence will be displayed for plans for further research and development.
3. Evidence of a sound business plan, sales and marketing strategies.
4. Strong leadership skills with the ability to drive the business forward through innovation and ambition.
As part of the judging process, a mystery shopper element will be incorporated. Mystery shoppers will visit shortlisted pharmacies to assess the quality of customer service, including friendliness, professionalism, attentiveness, and problemsolving skills. The results of this assessment will contribute to the final judging decision.
Awards Criteria:
1. Outstanding Patient Care and Compassion: Evidence of personalised, empathetic, and proactive customer service that enhances the patient experience.
2. Innovation in Service Delivery: Creative approaches, new initiatives, to improve customer interactions and accessibility.
3. Proven Impact and Customer Feedback: Measurable improvements in customer satisfaction, supported by testimonials, reviews, or survey results.
4. Commitment to Continuous Improvement: Ongoing staff training, service enhancements, and efforts to maintain high standards of patient care.
Applications are sought from those demonstrating clear enthusiasm and commitment to the Independent community pharmacy business in Ireland, exceptional quality above and beyond what is expected and an ability to overcome challenges in pursuit of goals.

The Award will be presented to the team or individual who can demonstrate a significant positive impact on the experience of those who use Pharmacy services. This may be through campaigns, promotions or initiatives which have identified a need within the community to address certain health issues.
1. Measurable benefits your pharmacy has brought to the community; with evidence to show how you have brought added services or value to your customers; productivity increases or other benefits relevant to this Award.
2. Evidence of strong team working, clear goals and achievements.
3. Demonstration of how your independent pharmacy has had an impact on the community its serves.
4. Evidence of how your way of working, within an independent, rivals the larger multiple stores to bring the same benefits to patients.




















1. Evidence of long-term, consistent dedication and outstanding achievements that have led to the advancement of the profession of pharmacy and self-care.
2. Evidence of how you/your team have researched and identified a need within the self-care market and how you have met this need for the enhancement of patient care.
3. Examples of entries may include evidence of the impact your self-care offering has had on the community you serve/improved your links with allied healthcare professionals or organisations and/or tackled a health inequality through self-care initiatives and promotions.

Judges will be looking for pharmacies that can showcase a commitment to cultivating exceptional skills and that can demonstrate a positive impact on business goals and a return on investment.
Awards Criteria:

1. Evidence of training, education and/ or support initiatives undertaken by the pharmacist/pharmacy team.
2. Demonstration of how your training and development initiative(s) developed, utilised and/or motivated your pharmacy team(s).
3. An ability to show what impact your training and development initiative(s) had on patients.
4. Examples of how this training and development initiative has impacted your pharmacy/ies.


The judges will be looking for those who can demonstrate promotion of the role of the Pharmacy Technician and those who continue to champion excellence through forward thinking and innovation.
Awards Criteria:
1. Evidence of long-term, consistent dedication and outstanding achievements that have led to the advancement of the profession of pharmacy and public health.
2. Evidence of a large variety of skills, attributes and accomplishments.
3. Evidence of an individual strong in character, cumulative professional accomplishments and the ability to properly represent and model what pharmacy technicians as a profession encompasses.
4. Evidence of an understanding the goals of pharmacy, and significantly contributing to how these goals may be achieved.

Judges will be looking for applications from those that are focused on establishing a framework for achievement of a high quality, safe and consistent service for the benefits of the patient, as well as facilitating the development of the professional role of the pharmacist.


This Award is open to pharmacists aged up to 30 - at the date of entry submission - who are working within any pharmacy, multiple or independent, where their involvement has been greater than six months.
It is the individual qualities that will be evaluated, rather than those of any of the projects worked on.
Applications are invited from independent and multiple Superintendent Pharmacists who can demonstrate one or more of the following:
1. Understanding patient needs when delivering healthcare in the community.
2. Examples of great patient experience and care, innovation and ambition.
3. Best practice in delivering professional services and patient reviews.
4. Encouragement of staff education in patient health and wellbeing advice.
5. Strong leadership skills with the ability to drive the business forward.
1. Judges will want to see effective communication skills with both staff and customers.
2. Demonstration of a commitment to mentoring or other leadership activities.
3. Operation within their own pharmacy liaising with key staff members and management and developing key communication skills.
4. A dedication and commitment to furthering the profession into the future.
Applications Close - Friday, April 10th, 2026 Send your entries to - kelly-jo@ipn.ie
Download application forms from: https://irishpharmacyawards.ie/

How to Enter:
To be eligible, a nominee must have been practicing for at least 4 years. Those working under the age of 30 should apply for the Young Community Pharmacist of the Year category.
Awards Criteria:


The key to any successful pharmacy is teamwork and this Award recognises the power and potential of a focused and unified approach to healthcare initiatives. Teams can be based within one organisation or spread over multiple organisations; but they must comprise individuals working towards the same objective or goal.
1. Evidence of long-term, consistent dedication and outstanding achievements that have led to the advancement of the profession of pharmacy and public health.
2. Evidence of a large variety of skills, attributes and accomplishments.
3. Evidence of an individual strong in character, cumulative professional accomplishments and the ability to properly represent and model what pharmacy as a profession encompasses.
4. Evidence of an understanding the goals of pharmacy, and significantly contributing to how these goals may be achieved.
Awards Criteria:
1. How the team has demonstrated their ability to deliver clear benefits to patients; and/ or staff members through working together efficiently and effectively.
2. How the team has worked together to achieve its objectives over the past twelve months.
3. Projects that the team has successfully managed which demonstrate excellence in quality, innovation, productivity and prevention.
4. A clear display of the principles underpinning their success as a team.
Applications Close - Friday, April 10th, 2026 Send your entries to - kelly-jo@ipn.ie Download application forms from: https://irishpharmacyawards.ie/
The judges will be looking for a hospital pharmacist who can clearly demonstrate sustained clinical impact, leadership and innovation, alongside a strong commitment to advancing the profession. Submissions should highlight measurable improvements in patient care, service delivery or professional practice, and illustrate how the entrant models excellence within hospital pharmacy.
Awards Criteria:

1. Evidence of sustained leadership and outstanding achievements that have contributed to advancing hospital pharmacy practice and improving patient care.
2. Examples of initiatives, service enhancements or models of care that have strengthened the role of pharmacy within the multidisciplinary healthcare team.
3. Demonstration of strong character, professionalism and the ability to represent and exemplify the core values of the pharmacy profession.
4. Evidence of understanding the evolving goals of hospital pharmacy and actively contributing to their achievement through strategic thinking, mentorship or advocacy.
The judges will be looking for a Hospital Pharmacy Technician who can clearly demonstrate their impact on patient care, their contribution to service development, and their commitment to advancing the technician role within hospital pharmacy. Evidence of leadership, innovation and professionalism will be key, alongside examples of how the entrant has championed excellence and inspired colleagues through their work.
Awards Criteria:
1. How their work has delivered measurable benefits to patients and/or colleagues, contributing to safer, more efficient or improved pharmacy services.
2. Evidence of promoting and advancing the role of the Hospital Pharmacy Technician within their organisation and the wider profession.
3. Examples of projects, initiatives or service developments that demonstrate excellence in quality, innovation, productivity or prevention.
4. A clear display of the values, teamwork and professional principles underpinning their success, including mentoring, training or supporting colleagues.
Applications Close - Friday, April 10th, 2026 Send your entries to - kelly-jo@ipn.ie Download application forms from: https://irishpharmacyawards.ie/
McLernons are proud to continue their sponsorship of the Irish Chemists' Golfing Society annual InterProvincial Cup, held on Thursday 12th March at Seapoint Golf Links, Termonfeckin, Co Louth.
Despite the weather warnings and inclement conditions almost 30 intrepid golfers from across the island of Ireland took to the fairways and enjoyed a great day’s golf as they fought to win the McLernons Claret jug and the ICGS trophy for the Best Individual.
McLernons are delighted to support an event that brings together pharmacists from every Province in Ireland for an outing which has seen great friendly rivalry throughout the years.
Jim McElwee, President of the ICGS, said, “Even though the wind and rain seemed determined to cause havoc we all persevered and

had a wonderful day at Seapoint. This annual InterProvincial event happened to be the first of our outings this year and I would strongly encourage my fellow pharmacists to pick up their clubs and come along and join us for the rest of the year.”
Captain Peter Finnegan added, “Munster were worthy winners of the McLernons interprovincial Cup dethroning Leinster who were last year’s winners.”
Congratulations to all participants and especially to our winners across the three categories:
Overall Winner –Gerry Rooney (12) – 43 pts
Category 1
1st Paul O’Keeffe (12) – 39 pts
2nd Bernard Crombie (15) - 37 pts
3rd Pat Downey (12) – 36 pts
Category 2
1st Ming Ng (19) – 38 pts
2nd Pearse Adams (21) – 38 pts
3rd Paddy Digan (20) – 37pts
Category 3
1st Jerry O’Connor (24) – 41 pts
2nd Tim Doody (26) – 40 pts
3rd Matt Murphy (35) – 39 pts



Thank you to all who attended and helped make the day a success! Provisional dates for 2026 outings are: 19th& 20th April: Slieve Russell, PGA National, Ballyconnell, Co Cavan 13th &14th May: Rosslare Golf Links, Co Wexford 17th June: Ballyneety Golf Club, Co Limerick 18th June: Limerick Golf Club, Ballybough, Co Limerick 13th July: Carlow Golf Club, Deerpark, Co Carlow 23rd & 24th August: The Heritage Golf Resort, Killenard, Co Laois 13th & 14th September: Carne Golf Links, Belmullet, Co Mayo


BEONE’S TISLELIZUMAB NOW AVAILABLE IN IRELAND, OFFERING A NEW FIRST LINE IMMUNOTHERAPY OPTION FOR ADVANCED OESOPHAGEAL CANCER
BeOne Medicines (Nasdaq: ONC; HKEX: 06160; SSE: 688235), a global oncology company, revently announced that the Health Service Executive (HSE) in the Republic of Ireland has approved reimbursement of tislelizumab, in combination with platinumbased chemotherapy, for the first-line treatment of eligible adult patients with unresectable, locally advanced recurrent or metastatic oesophageal squamous cell carcinoma (OSCC) whose tumours express PD-L1.
From 10 March 2026, tislelizumab will be available to prescribe to eligible patients across Ireland through HSE-funded hospital oncology services, in line with reimbursement approval.
Oesophageal cancer has a relatively low five-year survival and, according to the National Cancer Registry Ireland, approximately 500 patients are diagnosed each year. OSCC is the most common subtype, accounting for nearly 90% of cases. Squamous cell carcinoma represents one of the primary histological subtypes of oesophageal cancer globally.
The HSE reimbursement decision is based on results from the Phase 3 RATIONALE-306 trial, a randomised, double-blind, placebo-controlled study evaluating tislelizumab in combination with chemotherapy as a first-line treatment in 649 patients with unresectable, locally advanced recurrent or metastatic oesophageal squamous cell carcinoma.
The study met its primary endpoint and demonstrated a statistically significant improvement in overall survival in the intention-to-treat population. Median overall survival was 17.2 months for patients receiving tislelizumab in combination with chemotherapy compared with 10.6 months for those receiving chemotherapy alone (hazard ratio 0.66; 95% CI 0.54–0.80). In patients with PD-L1 expression ≥5%, median overall survival was 19.1 months versus 10.0 months, respectively (HR 0.62; 95% CI 0.49–0.79).
The safety profile of tislelizumab in combination with chemotherapy was consistent with the known profile of the individual agents. The most frequent serious adverse reactions (≥2%) were pneumonia, dysphagia, diarrhoea, fatigue and oesophageal stenosis. The most common adverse reactions were anaemia, fatigue, decreased appetite, nausea, constipation,
decreased weight, diarrhoea, peripheral sensory neuropathy, vomiting and stomatitis.
James Harris, Interim General Manager, BeOne Medicines UK & Ireland, said: “This approval marks our first launch in solid tumours in Ireland and reflects our commitment to supporting patients with advanced oesophageal squamous cell carcinoma. We are pleased that eligible patients will now have access to tislelizumab as a treatment option, and we remain focused on working with the oncology community to support patient care in Ireland.”
Tislelizumab first received approval by the European Commission on 15 September 2023 for the second-line treatment of OSCC. It is approved in multiple advanced cancer indications across Europe, including in gastric and gastro-oesophageal junction adenocarcinoma, non-small cell lung cancer (NSCLC), extensivestage small cell lung cancer (SCLC) and nasopharyngeal carcinoma.
PATIENTS WILL PAY THE PRICE FOR THE LACK OF URGENCY AROUND NATIONAL CANCER STRATEGY, WARNS IRISH CANCER SOCIETY
The Irish Cancer Society is calling on Government to provide clarity on its plans to develop a new and adequately funded National Cancer Strategy. The charity’s call comes ahead of a Dáil debate this evening following a Sinn Féin motion on Cancer Services.
The Society has consistently highlighted the lack of ringfenced funding for the current National Cancer Strategy, which has caused staffing shortages, outdated cancer care equipment and a lack of progress reporting on the Strategy's defined targets. The result is that patients continue to experience chronic delays for diagnostics, treatment and access to medicines.
Steve Dempsey, Director of Advocacy & Communications at the Irish Cancer Society, said:
“Previous national cancer strategies have worked. In the 90s just 44% of Irish people were alive five years after a cancer diagnosis. By 2018, after our first two National Cancer Strategies, this had increased to 65%. Planning and centralisation have worked. Patients have benefited from multidisciplinary services in one location, treating cancer in its complexity, including surgery, radiation therapy and medical oncology.
“With cancer effecting one in two of us, and incidences projected to rise in the coming years, it’s vital that the Government builds on the good work of the first two cancer strategies and doesn’t take progress for granted. This means vision, long-term planning and modelling the investment needed to minimise delays, improve outcomes, and save lives. On behalf of cancer patients and their families, we’re calling on Government to urgently provide clarity and timelines on how it will develop and resource a new National Cancer Strategy.”
Minister for Education and Youth Hildegarde Naughton and Minister for Health Jennifer Carroll MacNeill have announced details of education services for children and young people at the new National Children’s Hospital Ireland (NCHI).
The new hospital school being established at the NCHI will bring together and build on the strengths of the four existing hospital schools currently located at the Children’s Health Ireland hospitals at Temple Street, Crumlin, and Tallaght and at Beaumont Hospital. All paediatric services under CHI will transition from their current locations to the NCHI, bringing these services together under one state of the art facility.
The new hospital school will provide enhanced educational support for children and young people during their care. The school’s patron will be the City of Dublin Education and Training Board (City of Dublin ETB).
Minister
On a visit to NCHI, Minister Naughton said:
“I want to express my deep appreciation for the outstanding support, dedication and guidance shown across all of the hospital schools involved in this exciting development. I am acutely aware of the unique environment in which hospital schools operate and I greatly admire the vocation, compassion, and professionalism demonstrated by every member of staff. I would also like to sincerely thank each of the patrons and their representatives for their constructive and collaborative engagement throughout this process - engagement that has brought us to today’s important announcement.
“The National Children’s Hospital Ireland represents the most significant capital investment in our healthcare system in the history of the State. It will unite existing services within a modern, purpose built, digital hospital on the St. James’s campus, providing the very best care and treatment for Ireland’s sickest children and young people. Education will be at the heart of this new facility, and the new hospital school will play a vital role in supporting the holistic care of every child and young person who attends.
“I want to record my sincere gratitude to Archbishop Farrell and Archbishop Jackson for their invaluable stewardship as patrons of the Temple Street Children’s Hospital School and Tallaght University Hospital School, and indeed for their wider contributions as patrons of so many schools across the country. Their leadership has supported generations of children at the intersection of health and education.
“I also want to thank the City of Dublin ETB for its dedicated work as patron of the hospital schools at Beaumont and Crumlin, and I wish the City of Dublin ETB every success as it assumes patronage

of the new hospital school at the NCHI.
“My department will continue to fully support all patron bodies as they progress the patronage transfer and amalgamation processes. We will work closely together to ensure that all necessary arrangements are in place in the weeks ahead so that the new hospital school is ready to open in line with the opening of the NCHI.
“My department will also continue our strong partnership with the HSE, and I am grateful to Linn Dara School for extending its specialist provision into the NCHI.”
A new national quality improvement framework aimed at ensuring that every older person receives safe, timely and respectful emergency care, regardless of when or where they attend, has been published by the National Clinical Programme for Emergency Medicine in the Royal College of Surgeons of Ireland (RCSI).
The framework was developed in response to the steady and sustained increase in Emergency Department and Injury Unit attendances by older adults. In 2025, more than 560 people aged 75 years and over present to EDs nationally each day, reflecting the evolving patterns of emergency care demand.
The framework is informed by national data and earlier work led by the National Clinical Programme for Emergency Medicine, which highlighted that many patients aged 75 and over are triaged to lower acuity categories despite complex clinical needs, often resulting in prolonged Emergency Department stays and delayed care.
The National Quality Improvement Framework (QIF) for the Care of Older Adults in Emergency Departments and Injury Units sets out a structured, evidenceinformed approach, translating national policy and international standards into practical actions that can be implemented at site level, even in the context of ongoing system pressures. By strengthening post-triage assessment and standardising age-attuned practices, the framework aims to improve patient experience and outcomes while supporting more effective use of existing resources.
Dr Rosa McNamara, Clinical Lead, National Clinical Programme for Emergency Medicine said that “providing emergency care to older adults is a core responsibility of
Emergency Departments and Injury Units. As we evolve to meet the changing needs of the populations we serve, this document offers clear guidance and direction on minimum standards, best practices, and aspirational care.”
Professor Deborah McNamara, President, RCSI, welcomed the publication of the framework: “Ireland’s ageing population is reshaping the demand for emergency care, and our health services must evolve accordingly.
This Quality Improvement Framework provides practical, evidence-informed guidance to support Emergency Departments and Injury Units in delivering safe, timely and respectful care for older adults. RCSI is proud to support this important step towards more age-attuned emergency services nationwide.”
The framework builds on the Emergency Medicine Programme Model of Care (HSE, 2025) and aligns with Delivering an Age-Friendly Health System (Department of Health and HSE, 2025), as well as international best practice outlined by the International Federation for Emergency Medicine (IFEM). The framework focuses on three priority areas:
• Minimum Care Standards to ensure that all older adults have access to essentials such as hydration, appropriate seating, timely clinical assessment and clearly documented discharge and follow-up plans.
• Post-Triage Assessment and Bundles of Care, including frailty and delirium screening, to support early identification of risk, prompt intervention and avoidance of deterioration during Emergency Department stays.
• Education and Workforce Development, promoting Geriatric Emergency Medicinefocused training for all Emergency Department staff, supported by dedicated clinical leads and simulationbased learning.
The framework provides clarity on core requirements while allowing flexibility for local adaptation. Accompanying tools and implementation resources have been developed to support sites in embedding improvements in a sustainable way.
Examples of effective practice already operating within Irish Emergency Departments and Injury Units demonstrate that meaningful improvements are achievable, even within constrained environments. Some of these case studies are featured in the document.
Under the National Clinical Programme for Emergency Medicine in RCSI, this Quality Improvement Framework and accompanying resources have been developed by frontline staff in Emergency Departments and Injury Units and provide a clear, practical pathway to consistently highquality emergency care for older adults across Ireland, supporting frontline teams to meet changing patient needs while remaining mindful of the significant pressures faced by emergency services.
Mater Private Network has unveiled the latest offering in its Cherrywood Day Hospital, a new, cutting-edge CT scanner which offers both general and cardiac CT imaging. The new scanner offers
quick turnaround and convenient access, improving the patient experience, while significantly enhancing diagnostic capabilities and patient experience.
Mater Private Network is continually investing in new services and technologies to improve patient experience and care. In addition to the standard CT offering, the new scanner offers enhanced cardiac diagnostic capabilities including highresolution images of the heart to identify calcium buildup, coronary artery disease, blockages, and structural issues.
With the hospital group’s longstanding reputation for diagnosing and treating Ireland’s most complex cardiovascular cases, this state-of-the-art CT scanner significantly expands cardiac diagnostic capacity in South County Dublin by initially offering 160 extra scans a month, and scaling up to 450 as required.
A new DEXA scanner in the Cherrywood Day Hospital also improves diagnostic access for patients with conditions such as osteoporosis, rheumatoid arthritis and fracture risk.
Commenting on the new offering, David Slevin, Deputy CEO of Mater Private Network said, “At Mater Private Network we are continually looking at how we can improve our patient services. This new CT scanner will offer enhanced diagnostic capabilities, with quick turnaround times to give our patients peace of mind. This marks the latest in a series of recent investments for Cherrywood – in the past year we have opened
David Slevin, Deputy CEO of Mater Private Network at the openin

new clinics in geriatric medicine, rheumatology, urogynaecology, neurology, pain medicine and an orthopaedic foot & ankle clinic. Work is also currently underway on a new oncology suite and endoscopy suite in our Northern Cross Day Hospital, due to come on stream from September 2026.
“Our day hospitals mean that patients can access specialised care from same day clinic appointments to diagnostics and treatment plans from leading experts, without lengthy waiting times. Patients also have the reassurance that as part of our wider care network, they will have access to our full suite of hospitals and specialist teams, should the need for more acute treatment arise.”
Additional radiology services available in Cherrywood include MRI, X-ray, Ultrasound and CT. Advanced cardiac testing is also available, including cardiac MRI and cardiac CT, as well as ECG, Echo and stress testing. Treatment options include ultrasound-guided and fluoroscopic-guided joint injections for orthopaedic, rheumatology and spine pain management, while a physiotherapy clinic offers full musculoskeletal (MSK) assessments and reviews.
In 2025, Cherrywood Day Hospital saw over 15,000 patients across clinic appointments, diagnostic scans, and treatment.
Researchers at St John of God University Hospital’s Research & Innovation Hub, UCD Centre for Psychosis Research and University Medical Centre Groningen in the Netherlands have developed the first international clinical practice guideline specifically focused on the choice of first antipsychotic medicine for females experiencing a first episode of psychosis. The recommendations were developed by an international multidisciplinary panel of clinicians, researchers and experts-by-experience from Europe, North America and Australia. These guidelines are developed as part of the PROGRESS research group that has been funded by the Health Research Board.
Published in this month’s Schizophrenia Bulletin, the guideline responds to longstanding gaps in prescribing guidance, where sex-specific risks and side-effect profiles have not been adequately reflected in clinical recommendations. The recommendations are designed to support clinicians and patients
to make safer, more personalised treatment choices at the earliest stage of care.
Psychosis most commonly begins in early adulthood, with the median age of onset around 20 years. Symptom onset is often later in females, with a second peak after age 50. Early intervention at first onset is considered the gold standard of care, with antipsychotic medicines forming a key component of treatment alongside psychological, social, occupational and educational supports.
Caroline Hynes-Ryan, lead author and pharmacist at St John of God University Hospital, said “Firstepisode prescribing is a crucial window to get treatment right. By prioritising outcomes that matter for females - particularly hormonal and cardiometabolic effects - this guideline helps clinicians choose options with a lower long-term burden and supports females to stay on treatment that is both effective and acceptable.”
Professor Brian O’Donoghue, Director of the UCD Centre of Psychosis Research at the School of Medicine in UCD and lead of the PROGRESS group, said “Response rates to antipsychotic medications are high and therefore the choice of first antipsychotic medication needs to be based on the expected side-effects and tolerability. There is a very real differences in side-effect vulnerability for females and therefore, these sex specific guidelines should result in females being prescribed lower risk medications from the outset and
will have longer term benefits for physical health. This guideline brings together international evidence and expert consensus to support safer first-choice antipsychotic medicine, better shared decision-making, and more equitable care from day one.”
Addressing sex-specific risk from the start
The guideline highlights that females can experience distinct vulnerabilities to certain antipsychotic side-effectsparticularly hormonal disruption (including prolactin elevation) and cardiometabolic risk - with potential long-term implications for bone health, fertility and overall physical health.
The guideline was developed using the GRADE-ADOLOPMENT methodology and AGREE II framework, involving an international multidisciplinary panel including clinicians, researchers and experts-by-experience.
The aim of the guideline is to improve treatment acceptability and adherence, support better long-term physical health outcomes, and advance equity in psychosis care.
This work is part of the Research & Innovation Hub’s mission to translate evidence into improved patient care across the mental health sector.
The Network for Collaborative Oncology Development & Advancement (NCODA) recently
brought together international leaders across oncology, haematology, pharmacy, diagnostics and healthcare innovation for day two of its inaugural Global Oncology & Haematology Congress, held at The Shelbourne Hotel, Dublin.
The two-day Congress convened a carefully curated group of experts from Europe, North America and beyond for high-level dialogue, collaboration and clinically relevant education focused on improving patient outcomes worldwide. Discussions explored key issues shaping the future of cancer care, including precision oncology, AI-driven healthcare, treatment sequencing, sustainable oncology and multidisciplinary, patientcentred care models.
The Global Oncology & Haematology Congress reflects NCODA’s ongoing commitment to advancing quality, safety and access in cancer care through collaboration, education and realworld innovation, strengthening partnerships across disciplines and geographies.
Pictured at the Congress are Michael Reff, RPh, MBA, Founder and Executive Director of NCODA, alongside Dave Shanahan, a member of the Global Steering Committee and CEO of Athena Pharmaceuticals, Orla Dolan, CEO of Breakthrough Cancer Research, and Amy Nolan, Director of Clinical Affairs at the Irish Cancer Society.





COSMETIC & GIFT TRADE FAIR

SUNDAY 17TH MAY 10am - 6pm
MONDAY 18TH MAY 9am - 8pm
TUESDAY 19TH MAY 9am - 6pm
The Main Hall, RDS, Ballsbridge, Dublin