TranslationalBiotechnology:AJourneyfrom LaboratorytoClinics.1stEditionHasija
https://ebookmass.com/product/translational-biotechnology-ajourney-from-laboratory-to-clinics-1st-edition-hasija/
Blockchain for Teens: With Case Studies and Examples of Blockchain Across Various Industries 1st Edition Brian Wu
https://ebookmass.com/product/blockchain-for-teens-with-case-studiesand-examples-of-blockchain-across-various-industries-1st-editionbrian-wu/
ebookmass.com
Integrated Advertising, Promotion, and Marketing Communications (8th Edition)
https://ebookmass.com/product/integrated-advertising-promotion-andmarketing-communications-8th-edition/
ebookmass.com
Russia, China and the West in the Post-Cold War Era: The Limits of Liberal Universalism Suzanne Loftus
https://ebookmass.com/product/russia-china-and-the-west-in-the-postcold-war-era-the-limits-of-liberal-universalism-suzanne-loftus/
ebookmass.com
Synergy for Clinical Excellence: The AACN Model Patient Care 2nd
https://ebookmass.com/product/synergy-for-clinical-excellence-theaacn-model-patient-care-2nd/
ebookmass.com
A Rebel's Honor: A BBW & Military Romance (Heartland Heroes: Rebel Autos Book 1) Lana Love
https://ebookmass.com/product/a-rebels-honor-a-bbw-military-romanceheartland-heroes-rebel-autos-book-1-lana-love/
ebookmass.com
https://ebookmass.com/product/tulsians-business-law-for-cafoundation-3rd-edition-p-c-tulsian/
ebookmass.com
TRANSLATIONALBIOTECHNOLOGY
Thispageintentionallyleftblank
AcademicPressisanimprintofElsevier
125LondonWall,LondonEC2Y5AS,UnitedKingdom
525BStreet,Suite1650,SanDiego,CA92101,UnitedStates
50HampshireStreet,5thFloor,Cambridge,MA02139,UnitedStates
TheBoulevard,LangfordLane,Kidlington,OxfordOX51GB,UnitedKingdom
Copyright©2021ElsevierInc.Allrightsreserved.
Nopartofthispublicationmaybereproducedortransmittedinanyformorbyanymeans,electronicormechanical, includingphotocopying,recording,oranyinformationstorageandretrievalsystem,withoutpermissioninwritingfromthe publisher.Detailsonhowtoseekpermission,furtherinformationaboutthePublisher’spermissionspoliciesandour arrangementswithorganizationssuchastheCopyrightClearanceCenterandtheCopyrightLicensingAgency,canbefound atourwebsite: www.elsevier.com/permissions
ThisbookandtheindividualcontributionscontainedinitareprotectedundercopyrightbythePublisher(otherthanasmay benotedherein).
Notices
Knowledgeandbestpracticeinthisfieldareconstantlychanging.Asnewresearchandexperiencebroadenour understanding,changesinresearchmethods,professionalpractices,ormedicaltreatmentmaybecomenecessary.
Practitionersandresearchersmustalwaysrelyontheirownexperienceandknowledgeinevaluatingandusingany information,methods,compounds,orexperimentsdescribedherein.Inusingsuchinformationormethodstheyshouldbe mindfuloftheirownsafetyandthesafetyofothers,includingpartiesforwhomtheyhaveaprofessionalresponsibility.
Tothefullestextentofthelaw,neitherthePublishernortheauthors,contributors,oreditors,assumeanyliabilityforany injuryand/ordamagetopersonsorpropertyasamatterofproductsliability,negligenceorotherwise,orfromanyuseor operationofanymethods,products,instructions,orideascontainedinthematerialherein.
BritishLibraryCataloguing-in-PublicationData
AcataloguerecordforthisbookisavailablefromtheBritishLibrary LibraryofCongressCataloging-in-PublicationData
AcatalogrecordforthisbookisavailablefromtheLibraryofCongress
ISBN:978-0-12-821972-0
ForInformationonallAcademicPresspublications visitourwebsiteat https://www.elsevier.com/books-and-journals
Publisher: StacyMasucci
Senior AcquisitionsEditor: RafaelE.Teixeira
EditorialProjectManager: MonaZahir
ProductionProjectManager: NiranjanBhaskaran
CoverDesigner: VictoriaPearson
TypesetbyMPSLimited,Chennai,India
Contents
Listofcontributorsxi
Prefacexiii 1
Introductiontotranslational biotechnology
1Translationalbiotechnology: Atransitionfrombasic biologytoevidence-based research3
DebleenaGuin,SaritaThakran,PoojaSingh,S.Ramachandran, YashaHasijaandRitushreeKukreti
1.1Introduction4
1.1.1Backgroundandemergenceofthe field4
1.2Thephasesoftranslationalresearch5
1.3Challengestosolutions6
1.4Applications9
1.4.1Drugdevelopment12
1.4.2Nanomedicine16
1.4.3Genetherapy17
1.4.4Precisionmedicineandbiomarker development19
1.4.5Microbialengineeringfor bio-therapeutics19
1.4.6Applicationofbigdataand translationalbioinformatics19
1.5Conclusionandfuturedirections21
1.6Highlights21 Acknowledgment22 Conflictofinterest22 References22
2
Biotherapeutics
2Biotechnology-basedtherapeutics27
RavichandranVijayaAbinayaandPragasamViswanathan
2.1Introduction28
2.2Humangenetherapy29
2.2.1Somaticcellgenetherapy30
2.2.2Germlinegenetherapy30
2.2.3Genetransfersystem30
2.2.4Gene-editingtechnology33
2.2.5Ethicalissue34
2.3Stemcelltherapy34
2.3.1Sourcesofstemcells35
2.3.2Benefitsofstemcelltherapyinvarious disorder36
2.3.3Challengesandproblems37
2.4Nanomedicine37
2.4.1Nanotherapeuticapplications37
2.4.2Tissueengineering39
2.4.3Nanoimaging40
2.5Drugdesigninganddelivery40
2.5.1Rationaldrugdesign41
2.5.2Computer-aideddrugdesign41
2.5.3Drugdelivery44
2.6Recombinanttherapeuticproteinsand vaccines44
2.6.1Recombinantprotein44
2.6.2Expressionsystem44
2.6.3Recombinantproteinasatreatment46
2.6.4Recombinantvaccine47
2.7Conclusionandfutureapplications48 Acknowledgments48 Conflictsofinterest48
Author’scontribution48 References49
3Advancedbiotechnology-based therapeutics53
SrividhyaRavichandranandGauravVerma
3.1Introduction54
3.2Technologiesthatleadtothediscoveryof therapy55
3.2.1Genomeeditingtechnologies55
3.2.2Roleofnanomedicineindrug discoveryapproaches56
3.2.3Antibody drugconjugates58
3.3Moleculardiagnostics60
3.3.1Translationalbioinformatics62
3.3.2Organoids—toolsfordisease models63
3.4Cell-basedtherapy65
3.5Nanotechnologyanditsusesin biomedicine67
3.6Genome-scalemetabolicmodeling68
3.7Criticalprocessesintheflowfrombasic sciencetopracticalapplicationintheclinic viaclinicaltrialsandtranslationalstudies69
3.8Majorpitfallsintranslationalresearch70
3.9Advancementindevices,biologics,andvaccines asanintroductiontobiotechnologyproducts thatarebeingusedintherapy72
3.10Conclusionandsummary74 References74
3
Pathwayandtargetdiscovery
4Humaninvitrodiseasemodelstoaid pathwayandtargetdiscoveryfor neurologicaldisorders81
BhavanaMuralidharan
4.1Introduction82
4.2Generationofhumandiseasemodelsusing iPSCs/patientfibroblasts83
4.2.1Directeddifferentiationintoneural cells84
4.2.2Directdifferentiationintoneurons/ glia86
4.2.3Directlineagereprogramming/ transdifferentiationintoneurons88
4.3Modelingneurodevelopmentaldisorders88 3.1Rettsyndrome88
4.3.2FragileXsyndrome89
4.3.3Autismspectrumdisorders89
4.3.4Schizophrenia90
4.4Modelingneurodegenerativediseases91 4.4.1Amyotrophiclateralsclerosis91
4.4.2Alzheimer’sdisease92
4.4.3Parkinson’sdisease93
4.5Cerebralorganoidsandthefutureofhuman invitrodiseasemodeling93
4.6Frombenchtobedside—identificationof pathwaysanddrugtargetsfordesigning therapies95
4.7Futureperspectives97
Keyworddefinitions97
Acknowledgments98
References98
5Importanceoftargetedtherapiesin acutemyeloidleukemia107
AjitKumarRaiandNeerajKumarSatija
5.1Introduction107
5.1.1Conventionaltherapyforacutemyeloid leukemia108
5.1.2Significanceoftargetdiscovery108
5.2Approachesintargetdiscovery109
5.2.1Systemsapproach110
5.2.2Molecularapproach111
5.3Acutemyeloidleukemia targetedtherapiesin clinics117
5.3.1BCL-2inhibitors117
5.3.2Isocitratedehydrogenaseinhibitors117
5.3.3PML-RARα targetedtherapy118
5.3.4TargetingFLT3-mutatedacutemyeloid leukemia:frombenchtobedside(acase study)119
5.4Hurdlesandemergingtargetedtherapies120
5.5Conclusion125
Acknowledgments125 References126
4 Noveltherapeuticmodalities
6Biologicaltherapeuticmodalities137 MunishChhabra
6.1Introductiontobiologicaltherapeutic modalities137
6.2Historyofclassicalmodalities139
6.3Newmodalities140
6.3.1Smallmolecules140
6.3.2Nucleicacidtherapeutics142
6.3.3Therapeuticproteins143
6.3.4Antibodies145
6.3.5Cell-basedimmunotherapies148
6.3.6Stemcells150
6.3.7Phagetherapies151
6.3.8Microbiome-basedtherapeutics153
6.4Futureofbiologicaltherapeutics154
6.5Casestudy—bio-therapeuticmodalitiesin COVID-19treatment155
6.6Conclusion156 References160
7ThejourneyofnoncodingRNAfrom benchtoclinic165
RavindreshChhabra
7.1Introduction165
7.1.1NoncodingRNAsandtheir classification165
7.1.2InsiliconcRNApredictiontools166
7.1.3Screeningandcharacterizationof ncRNAs167
7.1.4SmallnoncodingRNAs (miRNAsandsiRNAs)167
7.1.5LongnoncodingRNAs181
7.2PatentlandscapeofnoncodingRNA187
7.3Bottlenecksintheuseofnoncoding RNAsasbiomarkers/therapeutics189
7.4Conclusionsandfutureperspectives191 References192
8Peptide-basedhydrogelsforbiomedical applications203
DebikaDattaandNitinChaudhary
8.1Introduction203
8.2Peptide-basedhydrogelators204
8.2.1 β-Sheetformingpeptides204
8.2.2 α-Helicalpeptides214
8.3Biomedicalapplications215
8.3.1Therapeuticdelivery216
8.3.2Scaffoldforregenerativemedicine218
8.3.3Wounddressing219
8.3.4Antimicrobialagents220
8.4Conclusion,limitations,andfuture directions221 References223
9Bispecificantibodies:Apromising entrantincancerimmunotherapy233
SamvednaSainiandYatenderKumar
9.1Introduction234
9.2Evolutionofbispecificantibodies234
9.2.1Differentformatsofbispecific antibodies236
9.2.2Mechanismofaction238
9.3Productionofbispecificantibodies243
9.3.1Hybridhybridoma(quadroma technology)243
9.3.2Knob-into-holeapproach243
9.3.3CrossMabapproach244
9.3.4Chemicalconjugation244
9.4Biomarkersinimmunotherapyataglance246
9.4.1Biomarkersforbreastcancer246
9.4.2Biomarkersforprostatecancer247
9.4.3Biomarkersforcheckpointblockade immunotherapy248
9.5Engineeringoftherapeuticprotein248
9.5.1Bindingaffinityenhancement249
9.5.2Immunogenicityminimization249
9.5.3Stabilityenhancementand half-lifeextension250
9.6Marketanalysis:past,presentandfuture250
9.7Futurechallengesandopportunities254
9.8Conclusion255
References255
10Emergingtherapeuticmodalitiesagainst malaria267
SureshKumarChalapareddy,AndaleebSajid,MritunjaySaxena, KritiArora,RajanGuhaandGunjanArora
10.1Introduction267
10.2Heme-detoxificationdrugs268
10.3DrugstargetingDNAorprotein synthesis270
10.4Drugstargetingmembranetransporters271
10.5Naturalproducts272
10.6Protein-basedmalariavaccines273
10.7Nucleicacidvaccinesforthenewera273
10.7.1DNA-basedvaccines274
13.3.3Roleofsyntheticbiologyin personalizedmedicine340
13.3.4Regulationandethicalconsiderations ofsyntheticbiology340
13.4Conclusion341 References342 7
Drugdiscoveryandpersonalized medicine
14Translationalresearchin drugdiscovery:Tinystepsbefore thegiantleap347
SindhuriUpadrastaandVikasYadav*
14.1Introduction348
14.2Toolsinvolvedintranslationdrug discovery349
14.3Recentsuccessfuladvancesintranslationdrug discovery351
14.3.1Cancer352
14.3.2Diabetes355
14.3.3Acquiredimmunodeficiency syndrome355
14.3.4Autoimmunedisorders356
14.3.5Neurologicaldisorder357
14.3.6Cardiovasculardisease(CVD)357
14.4Opportunitiesintranslationdrug discovery358
14.5Challengesintranslationdrugdiscovery359
14.6Approachestoboosttranslationaldrug discovery360
14.7Conclusion364
14.8Futureperspective364 References365
15FLAGSHIP:Anoveldrugdiscovery platformoriginatingfromthe “darkmatterofthegenome”371
NeerajVerma,SiddharthManvatiandPawanDhar
15.1Introduction371
15.2Designingnoveltherapeuticpeptidesfrom darkmatterofthegenome373
15.2.1Antimicrobialpeptides373
15.2.2Antimalarialpeptides374
15.2.3Anti-Alzheimerpeptides374
15.2.4Drawbacksofpeptides therapeutics375
15.2.5Futureapplications375 15.3Pseudogenes:apotentialbiotherapeutic target376
15.3.1Pseudogene-directedgene regulation377 References377
Socio-economicimpactof translationalbiotechnology
16Roleofsharedresearchfacilities/core facilitiesintranslationalresearch383
VidhuSharma
16.1Introduction:socioeconomicimpactof translationalresearch384
16.1.1Challengesfacedintranslational research385
16.2Corefacility:sharedresearch shared cost386
16.2.1Corefacilitiesofprimesignificancein translationalresearch388
16.3Researchanddevelopmentsupporting mechanism:environmentalscan(theUnited StatesandCanada)389
16.3.1Supportingtranslationalresearch throughcorefacilitiesintheUnited States—frompasttopresent390
16.3.2Canada’secosystemoftranslational researchandfunding mechanism392
16.3.3Highlightsaroundtheworld394
16.3.4Glimpsesofglobalresearchand developmentexpenditure396
16.4Efficienciesandleanpracticesinresearch management399
16.4.1Corefacilitiesbusinessmodel399
16.4.2Governancemodelforcore facility402
16.4.3Corefacilitiesandresearch outcome402
x Contents
16.5Finalnotes:learningsforfuture403
16.5.1Integrationofcorefacilitieswithin theinstitutionalstrategicplan403
16.5.2Comprehensiveavailabilityof infrastructureinventory403
16.5.3Impactmeasurement404
Acknowledgments404 References404
17AnewTOPSIS-basedapproachto evaluatetheeconomicindicatorsinthe healthcaresystemandtheimpactof biotechnology407
PriyankaMajumderandApuKumarSaha
17.1Introduction408
17.2Techniquefororderofpreferencebysimilarity toidealsolutionapproach410
17.2.1Metricspace410
17.2.2Newtechniquefororderofpreference bysimilaritytoidealsolution approach411
17.3Methodology412
17.3.1Selectionofcriteria413
17.3.2Selectionofindicators414
17.3.3Applicationofnewtechniquefor orderofpreferencebysimilarityto idealsolutionapproach414
17.3.4Analysisofsensitivity416
17.4Resultanddiscussion416
17.4.1Resultfromtechniquefororderof preferencebysimilaritytoideal solution1416
17.4.2Resultfromtechniquefororderof preferencebysimilaritytoideal solution417
17.4.3Resultfromsensitivityanalysis418
17.5Conclusion418 References419
Glossary421 Index425
Listofcontributors
RavichandranVijayaAbinaya RenalResearch Lab,SchoolofBiosciencesandTechnology, CentreforBiomedicalResearch,Vellore InstituteofTechnology,Vellore,India
VidhuAeri DepartmentofPharmacognosy& Phytochemistry,SPER,JamiaHamdard,New Delhi,India
GunjanArora YaleUniversity,NewHaven, CT,UnitedStates
KritiArora ProteusDigitalHealth,Inc., RedwoodCity,CA,UnitedStates
RajkumarChakraborty Departmentof Biotechnology,DelhiTechnologicalUniversity, Delhi,India
SureshKumarChalapareddy National InstitutesofHealth.,Bethesda,MD,United States
NitinChaudhary DepartmentofBiosciences andBioengineering,IndianInstituteof TechnologyGuwahati,Guwahati,India
MunishChhabra MolecularAssemblies,San Diego,CA,UnitedStates
RavindreshChhabra Departmentof Biochemistry,CentralUniversityofPunjab, Bathinda,Punjab,India
DebikaDatta DepartmentofBiosciencesand Bioengineering,IndianInstituteof TechnologyGuwahati,Guwahati,India
PawanDhar SchoolofBiotechnology, JawaharlalNehruUniversity,NewDelhi India
NituDogra ProteomicsandTranslational ResearchLab,CentreforMedicalBiotechnology,AmityInstituteofBiotechnology,Amity University,Noida,India
RajanGuha NationalInstitutesofHealth., Bethesda,MD,UnitedStates
DebleenaGuin GenomicsandMolecular MedicineUnit,InstituteofGenomicsand IntegrativeBiology(IGIB),Councilof ScientificandIndustrialResearch(CSIR), Delhi,India;GNRamachandranKnowledge Centre,CouncilofScientificandIndustrial Research(CSIR)—InstituteofGenomicsand IntegrativeBiology(IGIB),Delhi,India
YashaHasija DepartmentofBiotechnology, DelhiTechnologicalUniversity,Delhi,India; DepartmentofBioinformatics,Delhi TechnologicalUniversity,Shahbad Daulatpur,MainBawanaRoad,Delhi,India
DeepshikhaPandeKatare Proteomicsand TranslationalResearchLab,Centrefor MedicalBiotechnology,AmityInstituteof Biotechnology,AmityUniversity,Noida, India
RitushreeKukreti GenomicsandMolecular MedicineUnit,InstituteofGenomicsand IntegrativeBiology(IGIB),CouncilofScientific andIndustrialResearch(CSIR),Delhi,India; AcademyofScientificandInnovative Research(AcSIR),Ghaziabad,India
YatenderKumar NetajiSubhasUniversityof Technology,Delhi,India
PriyankaMajumder DepartmentofBasic ScienceandHumanities(Mathematics), TechnoCollegeofEngineeringAgartala, Maheshkhola,Agartala,Tripura,India
RuchiJakhmolaMani Proteomicsand TranslationalResearchLab,Centrefor MedicalBiotechnology,AmityInstituteof Biotechnology,AmityUniversity,Noida, India
SiddharthManvati SchoolofBiotechnology, JawaharlalNehruUniversity,NewDelhi India
Preface
Theadvancementsinthefieldofbiotechnologyhavebeenmonumentalandthe pacethereof,exponential.However,the sameisnotparalleledintheclinicalsetting. Thelackofadequatetranslationofbasic biomedicalresearchintoclinicalapplicationshasbeenamatterofhugeconcernfor theentireclinicalscientificcommunity. Therefore,thereisaneedtoachievecongruenceinboth,sothattheworkdonein researchlaboratoriesgetspercolatedinto thereal-timemedicalpractice.
Thisvoidhasledtotheemergenceofan excitingfieldof“benchtobedside”translationalresearch,sothatthelastmaninthe queue,thatis,thepatient,getsbenefitted. Thisbook“TranslationalBiotechnology:A journeyfromlaboratorytotheclinics”isa sincereefforttowardunderstandingthe stepsinvolvedintranslatingpath-breaking innovativeresearchandemergingscientific insightstoreachingthepatients,andinthe process,creatingnewtherapies,preventing, diagnosingandtreatingdiseases,and improvinghealthandliving.
Thebook,interalia,isaimedattransferringfundamentalbiologicaldiscoveries andtechnologiesfromtheresearchlaboratoriesintopatientcare,inthequestfor effectivehealthcare.Itattemptstotraverse thelongjourneyfrombenchworkto healthcarereformsandalsotriestoaddress theobstacles,lowsuccessrates,failures, andchallengesinthecomplexvoyage.
Inthisbook,westringtogethercontributionsfrominternationallyacclaimed authorsfromvariousdomainsofbiotechnologytoofferuniqueinsightsintheir
respectivefieldsofexpertise.Itwilltake thereadersthroughseveralfacetsoftranslationalresearchinbiotechnologywith illustrativeexamples.Theintroductorysectionsofthebookintroduceadvancedbiotechnologyprinciplesandprocessesin diseasestudies.Thissectionemphasizes technologiesthatleadtoorassistinthediscoveryofbetterclinicaloutcomes.Itis hopedthatitwillshapetheunderstanding ofcriticalprocessesintheflowfrombasic sciencestopracticalapplicationsintheclinicalsetting,viatranslationalstudiesand clinicaltrials.Thebookalsodiscussesthe advancementsindevices,biologics,vaccines,andseveralbiologicalmodalities,as anintroductiontobiotechnologyproducts thatarebeingusedintherapy.
Thesubsequentsectionsdealwithtranslationalapproachesinnewerdisciplinesof biotechnology,likebioinformatics,systems biology,andsyntheticbiology.Itdiscusses practicalapproachesinthedevelopmentof personalizedmedicine,clinicalsystems, andtranslationalmedicine.Italsooutlines futureresearchprospectsofthebenchto bedsideapproach.
Theconclusionsectionisoneofitskind thatgivesthereadersabirds-eyeviewof thesocioeconomicaspectsassociatedwith translationalbiotechnology.Thegoalisto makethereadersawareofthefeasibilityof carryingouttranslationalresearch,its availabilitytothepublic,andtheimpact causedbydiscoveriesmadeinthelaboratory.Itdiscussesthetechnologicaland monetarychallengesfacedindeveloping andunderdevelopedcountriesincarrying
outtranslationalresearch,andwaysto overcomethem.Italsodealswiththelegal andethicalaspectsoftranslational biotechnology.
Thegoalofthebookistoprovideina lucidform,theresearchinthefieldofbiotechnologythatistranslationalinnature,is cost-effective,andreadilyavailableforuse. Isincerelyhopethatthereaderswillbenefitfromthiscomprehensivebook,which willfurtherinspireandencouragethemto adoptsuchpracticesintheirresearchwork, orientedtowardclinicalapplications.
Everybookisanembodimentofcollectiveeffort.Iexpressmygratitudetoallthe contributorsfordeliveringsuchinsightful compilationsoftheirrespectiveareasof research,andthevaluableinputprovided bythereviewers.Iamindebtedtothe entireteamatElsevierforbeinginclose collaborationatvariousstagesforbringing outthisbookandensuringasmoothsailingpublicationprocess.
YashaHasija
Thispageintentionallyleftblank
DebleenaGuin1,2,SaritaThakran1,3,PoojaSingh1,3, S.Ramachandran2,3,YashaHasija4 andRitushreeKukreti1,3
OUTLINE
1.1Introduction4
1.1.1Backgroundandemergenceofthefield4
1.2Thephasesoftranslationalresearch5
1.3Challengestosolutions6
1.4Applications9
1.4.1Drugdevelopment12
1.4.2Nanomedicine16
1.4.3Genetherapy17
1.4.4Precisionmedicineandbiomarker development19
1.4.5Microbialengineeringfor bio-therapeutics19
1.4.6Applicationofbigdataand translationalbioinformatics19
1.5Conclusionandfuturedirections21 1.6Highlights21 Acknowledgment22 ConflictofInterest22 References22
1 GenomicsandMolecularMedicineUnit,InstituteofGenomicsandIntegrativeBiology(IGIB),Councilof ScientificandIndustrialResearch(CSIR),Delhi,India
2 GNRamachandranKnowledgeCentre,CouncilofScientificandIndustrialResearch(CSIR)—Instituteof GenomicsandIntegrativeBiology(IGIB),Delhi,India
3 AcademyofScientificandInnovativeResearch(AcSIR),Ghaziabad,India
4 DepartmentofBioinformatics,DelhiTechnologicalUniversity,ShahbadDaulatpur,MainBawanaRoad, Delhi,India
4 1.Translationalbiotechnology:Atransitionfrombasicbiologytoevidence-basedresearch
1.1.1Backgroundandemergenceofthefield
Biotechnologyisanadvancedfieldofbiologythatexploitstechnologytomakeand disseminatebiologicaldiscoveriesforhumanbenefit.Biotechnologyhasabroadspectrumofapplications,spanningfromindustry toagriculture,manufacturingoffood,chemicals,probiotics,pharmaceuticals,andthelistgoeson.Oneoftheprimaryfocusesof biotechnologyhasbeenimprovinghealt hcare,andwehavecomealongwayinthat endeavor.Like,forinstance,frommicrobia lbioconversionsfortherapeuticusetovaccinedevelopment,drugdiscoveryanddevelopment,clinicaltrialdesign,community medicine,personalizedtherapy,clinicalinfo rmatics,etc.Throughthecourseoftimeand withtheaccumulationofsubstantialbiologicalinformation,biotechnologyhasprogressedtowardspecificapplication-basedresearch,whichistheneedofthedecade.It hasmadeagatewayforanewapproachtoresearchcalled“translationalresearch.”The word“translation”asdefinedbytheNatio nalInstituteofHealthfundedNational CenterforAdvancingTranslationalSciences(NCATS),is“theprocessofturningobservationsinthelaboratory,clinic,andcommunityintointerventionsthatimprovethe healthofindividualsandthepublic—fromdia gnosticsandtherapeuticstomedicalproceduresandbehavioralchanges”(USDepartmentofHealth&HumanService,2020 ). Moreover,theemergingfieldof“translationalscience”isfocusedoninvestigatingthe scientificandoperationalprinciplesunderlyingeachstepofthetranslationalprocess. Throughthebasicknowledgeofhumanphysiologyindiseaseandhowtheintervention (say,adrugoratherapy)isaffectingthedisea seswillexpeditethetranslationalprocess towardafocusedscience-driven,predictive, andeffectivedrugdevelopmentforthepreventionandtreatmentofalldiseases.
Withtheavailabilityofunlimitedhumanbiologicaldata,thisnewsubsidiaryfieldof “translationalbiotechnology”isfocused,distinct,anduniqueinoperation,application, andimplementation.Whileuniquechallengesindifferentterritorieshover,thisfieldalso providesuswithunprecedentedopportunitiestowidenthespectrumofbiomedicalenterprise.Comprisingmanydisciplinesofscienceandoperations,includingbiology,chemistry,informatics,pharmaceutical,engineering,medicine,andpublichealthmanagement, translationalbiotechnologydefinesthescientificandoperationalrelationshipsamong thesefields,buildsbridges,andcreatesatransdisciplinarynetworkforactivedevelopment anddeploymentofinterventionsthatbenefitpublichealth(Westfall,Mold,&Lyle,2007).
Inthischapter,weintroducethekeythreadsoftranslationalresearch.We,first,provide aconceptualoverviewoftheunderstandinginthisfield,thedifferentstagesinthebroad spectrumofthetranslationalresearchpipeline,followedbythenumerousscientificand regulatoryroadblocksinthisfieldandtheirpotentialsolutiontospeeduptheprocess. Finally,summingupthevariedapplicationsoftranslationalbiotechnologyinclinical sciencesandpublichealth.Fromthetranslationalapplicationindrugdiscoveryanddevelopment,precisionmedicineandbiomarkerdiscovery,genetherapy,bio-therapeutic,and applicationofartificialintelligence(AI)indiseasediagnosisandotherclinicalresearch. Withaneffectiveintegratednetworkofrobustmultidisciplinaryeffortbetweenpatients, researchers,andhealthcareproviderswithinasystem,canyieldwell-roundedand
competentteamworkwhocanultimatelycommittoimprovedcommunityhealthand healthcarecosts.
1.2Thephasesoftranslationalresearch
Thebroadspectrumoftranslationalresea rchhasbeencategorizedintodifferent phasesforaconvenienttransitionfrombasicresearchfindingtoitsapplicationmode forclinicalimplementation.Thisintegrat iveresearchefforthasbeendesignatedinto foursteps:T 1 ,T 2 ,T 3 ,andT 4 ( Lorenzi,2011 ).Thestepsinoutcome-basedclinical researchfollow:frombasicscientificlaboratoryworkbasedonunderstandingthe humanphysiologyandapplicationofmedicinal chemistrytopreclinicalstudiestovalidatetheclinicalfindinginvitro/invivomo delsystemsfollowedbyclinicaltrialstudiesforimplicationforpracticeanditsoveral leffect.Ultimately,theresearchfindings areusedforimplicationsincommunity-basedh ealthbenefitsinclinicaluse.Thefinal crucialstepinclinicalresearchisthedeliv eryofrecommendedcaretotherightpatient attherighttime,resultinginimprovedpatienthealth.Itmaybewithimprovedprognosis,diagnostictests,therapyortherapy adherence,andtreatmentoutcomeforusein clinicalpractice.Ateachofthetransitionsbetweeneachstage,thereisastepfortranslationaloutput.Thespectrumoftranslatio nalresearchisnotlinearorunidirectional; eachstagebuildsuponeachotherandm ovesupwardinthepyramidwithalarger impactandgreaterpopulationcoverage.Atallstagesofthespectrum,accordingto NCATS,newapproachesaredeveloped,demon stratingtheirusefulnessanddisseminatingthefindings.
Everystageofthetranslationalresearchsp ectrumisbasedonresearchfindingsthat derivefrombasicsciences.AtT 1 ,insightsgainedfromthefundamentalscientificdiscoveryareaccumulatedthatgivesa“proofofconcept”studywithcontrolledexperimentalconditionsand,therefore,theiroutcome.T 2 providesclinicalinsightsfrom studyfindings.Fromthepreviousstage,whereresearchersdevelopmodelstotest interventionstounderstandthepathophysi ologyofthediseaseand,ultimately,its treatment.Suchtesting/dataisusedtoelabor ateclinical/physiologicalinsights.Such testingisperformedusinginvitromodels(animaltissue-specificcelllines)orinvivo modelorganismslikemouse,rat,drosophila,zebrafish,etc.andinsilico-basedcomputer-assistedsimulationmodelsofdrug,dev ice,ordiagnosticinteractionswithinlivingsystems.Thisstepincludespreclinica lvalidationstudies,earlyphaseI,andII trials.Subsequently,practice-basedresearchincludestestinginterventionsforhuman safetyandeffectivenessinpatientswithorw ithoutthedisease,behavioral,andobservationalstudiesandoutcomesandhealthservicesresearchattheT 3 phase.Theconfirmedevidencefromhumansubjectsisintroducedanddisseminatedintoapatient caresettingforimplementationanddeploymentofclinicaldiscovery.Thisstepis calledT 4 ,anditisthefinalachievablegoal,whe rethedevelopedinterventionisused forimprovingpublichealth( Choi,Tubbs,&Oskouian,2018 ).Inthisstageoftranslation,researchersvalidatetheclinicalo utcomefindingsinanentirepopulationto determinetheburdenofthediseasesandclinicaleffortstopreventit,diagnose,and treatthem( USDepartmentofHealth&HumanService,2020 ).Thesestepshavebeen
TABLE1.1 Thetranslationalresearchcontinuum:takinganexampleofprecisionmedicineanddrug responsebiomarker—fromdiscoverytoapplicationindruglabelingbytheUSFDA.
PhasesHypothesisFinding
Basicscientific discovery
Whatcausestheinterindividual variabilityindrugresponse?
T1Translationofbasicscientificfindingfor humanhealth
Clinicalfindings (proposedhuman application)
Whichgeneticvariantisassociatedwith drugresponse,andhow?
GeneticvariantsinvolvedinPK/PDcan causeavariedresponsetodrugsinpatients
Theconceptofpharmacogenomics
Thereisavastinterindividualvariabilityin warfarindoserequiredtoachievethetarget therapeuticeffect.Thisvariabilityismostly causedbygeneticvariantsin VKORC1, CYP2C9, CYP4F2 genes(Dean,2012)
T2TranslationtoclinicalimplicationPGxbiomarkersforaltereddrugresponseto warfarindoses.Forexample,patientswith CYP2C9*2andCYP2C9*3variants,require lowerdosesofwarfarin,andtakealonger timetoreachtargetINRonstarting warfarintherapyandareatahigherriskof bleedingcomplications(Johnson,Caudle, Gong,&Whirl-Carrillo,2017)
Theimplicationfor practice(replicationin largerpopulation)
Isthegeneticvariantassociatedwith poormetabolismofwarfarininall populations(Pavani,Naushad,& Rupasree,2012)?
Theeffectofgeneticassociationof VKORC1 and CYP2C9 inAfricanandAsianare concordantwiththoseinEuropeanancestry, butthefrequencydistributionofallelic variantsvarybetweenmajorpopulations (Fungetal.,2012).Thirtypercentage variabilityinwarfarindoseisdueto CYP2C9 and VKORC1 riskvariantsamongEuropean Americansand10%amongAfrican Americans(Limdietal.,2008)
T3TranslationtopracticeDefiningwarfarindosingalgorithmfor patientscarryingspecificgeneticvariant allele/genotype(Pirmohamedetal.,2013)
Clinicalpractice guidelines
Whatdose/drugtobeadministeredto whichpatients?Alternatively,patients areatriskofadverseeffects.
Genotype-baseddrugprescription algorithmsalsobetterpredictwarfarindose thantheUSFDA-approvedwarfarinlabel table(Finkelman,Gage,Johnson, Brensinger,&Kimmel,2011)
T4Evidence-basedresearchPopulation-specificclinicalutility (Pirmohamedetal.,2013)
Clinicalutilityfor publichealthbenefit
Marketdisseminationandpracticeof PGx-basedmarkerfordrug administration
FDAapprovalfordruglabelingbasedon pharmacogenomicbiomarkers(USFDA, 2018)
CYP2C9,CytochromeP450familytwosubfamilyCmember9; CYP4F2,cytochromeP450familyfoursubfamilyFmember2; INR,internationalnormalizedratio; PGx,pharmacogenomics;PK/PD,pharmacokinetic/pharmacodynamic; USFDA,theUnited StatesFoodandDrugAdministration; VKORC1,vitaminKepoxidereductasecomplexsubunit1.
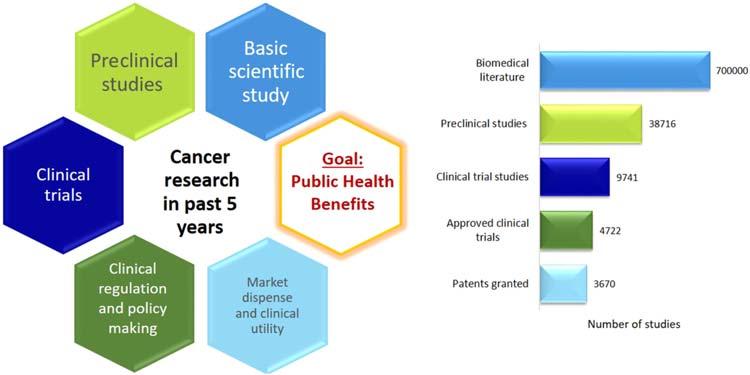
Aglanceattranslationalresearchoncancerinthepast5years.
tounderstandtheissuesintranslationalresearchbetterandtolookforpotentialsolutions (HarvardCatalystClinicalResearchCenter,2020)(Fig.1.2).
Thevastmajorityofstudiesthatgiveexcitingandimpressiveresultsinpreclinicalstudies usuallyfailatitsnascentstageofrigoroustargetvalidationanddonotreachuptoclinical development.Issuesandchallengesthatmightaccountfordifferencesbetweenmarkedsuccessindecipheringthemechanism,pathogenesis,andtreatmentofdiseasesinpreclinical studies,andlimitingsuccessratefortranslatingmostofthesediscoveriesfrombenchto bedsideincludelackofpropervalidationofpublishedfindings,reproducibilityproblem, andlackofpredictiveefficacyandsafety(Yoichi,2019).Themaincausesoftheseissues includefewerstudiesconductedtosupportanyresearchfindingforapplicationandlackof replicabilityofdatabetweendifferentstudiesduetodifferentstudydesigns.Otherfactors likesamplingcriteria,geneticallydiversesubjectsincludedinstudies,controlledexperimentalconditions,andrecruitmentofsampleswithspecificphenotypiccharacteristics,pitfallsin animalexperimentation,differentstatisticalmethodsused,andoverinterpretationofdata (Collins&Tabak,2014).Generally,academicclinicaltrialunitsconductinnumerableclinical studiesonhumansubjectsbutreportonlythemostpromisingresultsfromstudiesthat havefavorableoutcomes,andthisleadstoalackofreproducibilityofresults.Toenhance reproducibilityandtransparencyofstudiesconducted,thereisaneedtodiscusssuchchallengesplaguingpreclinicalresearchacrosstheglobeindifferentpopulationstoprovideconstructiveguidancefortherapyandrecommendreportingstandardclinicaldesign.Potential solutionstotacklethisproblemincludecallsforrandomassignmentofanimals,blindingof preclinicaltreatmentgroups,morerigoroussampleandeffectsizecalculations,andformal rulesforthehandlingofdatainvolvingoutliers,prespecifiedprimaryandsecondaryendpoints,andreplicationofkeyexperimentalfindings,developmentofnovelmethodstopredictsafety,conductionofPhase0,andinvestigationalexploratorytrialstoconfirmthemode ofaction,validatebiomarkers(Landisetal.,2012).
FIGURE1.2
Themajornon-scientificchallengesthatneedtobeaddressedaretominimizethegaps betweenscientificdiscoveryandtheirapplicationintheclinicalsetting,whichshouldbe prioritizedinadvancebyfunders/researchorganizations.Othernon-scientificfactors includeculturaldifferencesbetweencliniciansandbasicscientistsasscientistsinvolvedin basicscienceresearchhavelessexposuretotheclinicalenvironmentandthelackoflaboratoryresearchexperienceinclinicians.Additionally,communicationgap,thedifference inworkattitudeandrewardsystemofbothgroups,likeacademicresearchersinvolvedin translationalresearchgenerallydonothaveproperincentivestochannelizethemovement ofscience,astheircareertrajectoryisdependentonhigh-impactpublications,funding, andpatents,alsoaddtotheproblem.Thisgapneedstobefilledbycouplingofhospitals andresearch-orientedscientificinstitutes,todefineaseparatefieldoftranslational researchwithamultidisciplinaryviewpoint,andinstitutionsshouldensureandmake newguidelinestoproperlyevaluateandrecognizethecontributionsmadebyscientistsin translationalresearch(Homer-Vanniasinkam&Tsui,2012).
Thefinancinggapfortranslationalresearchisalsowidening.Traditionalinvestors involvedintranslationalresearcharebecomingincreasinglyrisk-averseinthefaceofescalatingchallengesintheearlystagesofthedrugdevelopmentprocess.Tocounteractthis trend,themedicalresearchfieldneedstoincreasethefieldofpromisingresearchventures thatalsoattractinvestmentopportunitiesbymodifyingboththeresearchmanagement processaswellascurrentfinancingmethods(MILReport,2012).Besidesthislackof resourcesandscatteredinfrastructure,lackofskilledinvestigatorswithspecificexpertise andwell-awareparticipantsforthestudy,timetaking,andcostlyphasesoftranslational research,lackofcollaborationsandpartnershipsbetweenclinics,researchers,andindustries,conflictofinterest,ethical,andvariousregulatoryissues,righttoprivacyandincompatibledatabasesaddstothechallengeswhichslowdownthepaceofbenchtobedside research.Potentialsolutionsforthesechallengesincludebuildingnational-levelclinical andtranslationalresearchcapability,collaborativeefforts,likeacoherentpartnership betweenacademicandindustrialresearch,whereacademiadeliversskilledandtrained researchersandindustryexploitsthosehumanresourcefortheupliftmentoftranslational activitiesformankind.Costreductioninclinicalinvestigations,properscrutinyofethical andsocialissuesshouldbelookedintopriortothestudyaspertheclinicalset-up,and studydesign.Anoverviewofobstaclesandtheirpotentialsolutionsrelevanttotranslationalresearcharepresentedin Table1.2.
1.4Applications
Regardlessofmanygapsintranslatingeveryresearchavenuetoclinicalimportance, therehavebeenseveralsuccessfulattempts.Biotechnologyhasasignificanthandintranslatingclinicalresearch.Initially,biotechnologywasthoughtofbeinglimitedtothedevelopmentofrecombinantDNA(rDNA)technologyandtheproductionofrecombinant proteinssuchasinsulin.Withadvancementsintechnology,thisfieldhasevolvedfromthe synthesisofproteinstobiopharmaceuticaldiscovery.Now,biotechnologyhascontributed tothedevelopmentofgenetherapy,immunotherapy,andpersonalizedmedicine.It includessynthesisofmonoclonalantibodies(mAb),antisensestrands,enzymes,cancer
TABLE1.2 (Continued)
IssuesPerceivedobstaclesPotentialsolutions
5Career progressionand promotion
Inthepresentscenario,currentprogression includeshigh-impactpublications,bigger grants,andscientistsinvolvedin translationalresearchhavecomparatively fewerpublicationsasitistimetaking (Homer-Vanniasinkam&Tsui,2012).
6TimeandcostTranslationoftenrequiresalargelengthof timeandiscostlyasitrequiresmany phasesofresearchearlierinthelabthen inclinics.
Theplethoraoftoolsandtechniques requiredfortranslationalbiotechnology enhancesthecost,andalsothehuman trialphaseaddstoit( Morris,Steven,& Jonathan,2011 ).
Institutionsshouldensureandmakenew guidelinestoproperlyevaluateand recognizethecontributionsmadeby scientistsinTranslationalresearch(HomerVanniasinkam&Tsui,2012).
Moreemphasisshouldbegiventoprovide bettertrainingtotranslationalscientiststo improvetheircareers.
Actionablepolicyinterventionsshouldbe madethatcouldspeedupthetranslation process,whereappropriate,andthus increasethereturnonresearchinvestment.
7Lackof resources
Lackofinterdisciplinarytraining,integrated infrastructure,skill,andtechnologyto executeandinterpretinvitroandinvivo preclinicalstudiesrequiredtodemonstrate efficacyfully.
8FundingLackoffinancialsupportforcarryingout translationalresearchasitisalongprocess andcostlyatthesametime,andthereisno guaranteeofsuccess,especiallyatthe clinicalphase(Editorial,2017).
Translationalresearchprocesseshavinga higherchanceofsuccessand commercializationtendtohavemorefund allocationsthanthosewithahigherimpact onpublicandcommunityhealthbutdifficult toconductandhaslesschanceofsuccess. Fundingoftengetsreducedduringthecostly humantrialphase.
NCATSwasestablishedbyNIHin2012to executethetranslationalscienceprocess (Mandal,Ponnambath,&Parija,2017).
Globallyotherinfrastructureswerealso developedtopromotetranslationalresearch likeTHSTIinIndia. Buildnationalclinicalandtranslational researchcapability.
Tofillthisfinancialgapintranslational research,thereisaneedforresearchthat willalsoattractinvestmentopportunitiesby modifyingboththeresearchmanagement processaswellascurrentfinancing methods.
Theurgentneedfortargetedapproachesfor fast-trackdrugdevelopmentpipelinethat canbebetteratmanagingrisk,lowercapital investment,andimproveresearch effectivenessinlessertimeandaccessnew capitalsources(MILReport,2012).
9Collaborations and partnerships
Lackofcollaborationsandpartnerships betweenclinicsandresearchers.
Thereisaneedtotakeinitiativesthat encouragepartnershipsandenhance consortium-widecollaborations. Forexample,NCATSisdevelopedto encouragecollaborativepartnerships betweencliniciansandscientistsaswellas pharmaceuticalindustries.
10EthicalissuesManyethicalissuesareinvolvedinhuman researchsuchasinformedconsent, irreversibleconsequencesinhumansubjects ontheapplication,socialinjusticeandrisk analysis,ethicsregardinganimalcrueltyfor Clear,informedconsentisverycrucialin translationalresearchtoavoidtherapeutic misconception.Drawnoutcomeof applicationsandsubjects’consentregarding thesame.
(Continued)