https://ebookmass.com/product/total-synthesis-of-bioactive-
Instant digital products (PDF, ePub, MOBI) ready for you
Download now and discover formats that fit your needs...
Studies in natural products chemistry. Volume 58. Bioactive natural products Rahman
https://ebookmass.com/product/studies-in-natural-products-chemistryvolume-58-bioactive-natural-products-rahman/ ebookmass.com
Discovery and Development of Antidiabetic Agents from Natural Products: Natural Product Drug Discovery Goutam Brahmachari
https://ebookmass.com/product/discovery-and-development-ofantidiabetic-agents-from-natural-products-natural-product-drugdiscovery-goutam-brahmachari/ ebookmass.com
Discovery and Development of Anti-Breast Cancer Agents from Natural Products Goutam Brahmachari
https://ebookmass.com/product/discovery-and-development-of-antibreast-cancer-agents-from-natural-products-goutam-brahmachari/ ebookmass.com
WOCN Core Curriculum: Ostomy Management (Wound, Ostomy and Continence Nurses Societyu00ae Core Curriculum) 1st Edition, (Ebook PDF)
https://ebookmass.com/product/wocn-core-curriculum-ostomy-managementwound-ostomy-and-continence-nurses-society-core-curriculum-1stedition-ebook-pdf/ ebookmass.com
Understanding the Prefrontal Cortex: Selective Advantage, Connectivity, and Neural Operations Richard Passingham
https://ebookmass.com/product/understanding-the-prefrontal-cortexselective-advantage-connectivity-and-neural-operations-richardpassingham/ ebookmass.com
Digital Transformations in the Challenge of Activity and Work: Understanding and Supporting Technological Changes
Marc-Eric Bobillier Chaumon
https://ebookmass.com/product/digital-transformations-in-thechallenge-of-activity-and-work-understanding-and-supportingtechnological-changes-marc-eric-bobillier-chaumon/ ebookmass.com
Culture from the Slums Jeff Hayton
https://ebookmass.com/product/culture-from-the-slums-jeff-hayton/
ebookmass.com
Warrior Kings: Planet of Kings Books 1-3 Savino
https://ebookmass.com/product/warrior-kings-planet-of-kingsbooks-1-3-savino-2/
ebookmass.com
To Die in Tuscany David P. Wagner
https://ebookmass.com/product/to-die-in-tuscany-david-p-wagner-2/
ebookmass.com
Industry 4.0 and Circular Economy: Towards a Wasteless Future or a Wasteful Planet? Antonis Mavropoulos
https://ebookmass.com/product/industry-4-0-and-circular-economytowards-a-wasteless-future-or-a-wasteful-planet-antonis-mavropoulos/
ebookmass.com
TOTALSYNTHESISOF BIOACTIVENATURAL PRODUCTS TOTALSYNTHESISOF BIOACTIVENATURAL PRODUCTS GOUTAMBRAHMACHARI DepartmentofChemistry,Visva-Bharati (ACentralUniversity),Santiniketan,India
Elsevier
Radarweg29,POBox211,1000AEAmsterdam,Netherlands TheBoulevard,LangfordLane,Kidlington,OxfordOX51GB,UnitedKingdom 50HampshireStreet,5thFloor,Cambridge,MA02139,UnitedStates
© 2019ElsevierLtd.Allrightsreserved.
Nopartofthispublicationmaybereproducedortransmittedinanyformorbyanymeans, electronicormechanical,includingphotocopying,recording,oranyinformationstorage andretrievalsystem,withoutpermissioninwritingfromthepublisher.Detailsonhowto seekpermission,furtherinformationaboutthePublisher’spermissionspoliciesandour arrangementswithorganizationssuchastheCopyrightClearanceCenterandtheCopyright LicensingAgency,canbefoundatourwebsite: www.elsevier.com/permissions.
Thisbookandtheindividualcontributionscontainedinitareprotectedundercopyright bythePublisher(otherthanasmaybenotedherein).
Notices Knowledgeandbestpracticeinthisfieldareconstantlychanging.Asnewresearchand experiencebroadenourunderstanding,changesinresearchmethods,professionalpractices, ormedicaltreatmentmaybecomenecessary.
Practitionersandresearchersmustalwaysrelyontheirownexperienceandknowledgein evaluatingandusinganyinformation,methods,compounds,orexperimentsdescribed herein.Inusingsuchinformationormethodstheyshouldbemindfuloftheirownsafetyand thesafetyofothers,includingpartiesforwhomtheyhaveaprofessionalresponsibility.
Tothefullestextentofthelaw,neitherthePublishernortheauthors,contributors,oreditors, assumeanyliabilityforanyinjuryand/ordamagetopersonsorpropertyasamatterof productsliability,negligenceorotherwise,orfromanyuseoroperationofanymethods, products,instructions,orideascontainedinthematerialherein.
LibraryofCongressCataloging-in-PublicationData
AcatalogrecordforthisbookisavailablefromtheLibraryofCongress BritishLibraryCataloguing-in-PublicationData
AcataloguerecordforthisbookisavailablefromtheBritishLibrary
ISBN:978-0-08-102822-3
ForinformationonallElsevierpublications visitourwebsiteat https://www.elsevier.com/books-and-journals
Publisher: SusanDennis
AcquisitionEditor: EmilyMcclosket
EditorialProjectManager: CarlyDemetre
ProductionProjectManager: VigneshTamil
CoverDesigner: GregHarris
TypesetbySPiGlobal,India
Dedication InMemoryofSantoshK.Brahmachari—MyBelovedFather Foreword S.Chandrasekaran
DepartmentofOrganicChemistry,IndianInstituteofScience,Bangalore,India
Naturalproductscanbeconsideredasa“gift”ofnaturetochemists,andthe diversityofstructuresandcomplexitiesthatoneencountersinthemistruly amazing.Itisawell-knownfactthatavastmajorityofdrugsapprovedfor clinicaluseareeitherderivedfromnaturalproductsorinspiredbyresearch onnaturalproducts.Ingeneral,organicchemistsareenamoredbythechallengesthatthetotalsynthesisofbioactivenaturalproductsoffers.Sincemany ofthenaturalproductsareisolatedonlyinverysmallquantitiesfromnatural sources,thereisanadditionalimpetusandnecessityforsyntheticchemiststo designandexecuteanelegant,efficient,andeconomicallyviableprotocol forprocuringthesemoleculesonareasonablescaleinthelaboratory.The strategiesandtacticsinachievingthegoalofsynthesisofbioactivenatural products,ifpresentedinanappropriateandappealingmanner,wouldbe veryusefultoyoungstudentsandactivepractitionersoforganicsynthesis.
ItisinthiscontextthatIfind TotalSynthesisofBioactiveNaturalProducts, authoredbyProfessorGoutamBrahmachari,tobeextremelyvaluable andusefultothescientificcommunity.Theauthorhascarefullychosen 60bioactivenaturalproductsandsummarizedtheirtotalsyntheses.Itis tobeappreciatedthatexcellentdescriptionsofsyntheticschemesforeach moleculeareavailableinoneplace.Thechaptersinthisbookarearranged logicallybasedonthenamesofthebioactivemoleculesinalphabeticalorder.
Thewealthofimportantinformationthatisavailableinthisbookistruly exceptionalandIwouldliketoextendmyheartycongratulationstothe authorforhisdedicatedeffortandcontribution.
Preface MotherNatureisaninexhaustiblesourceofcomplexmoleculesdisplaying anincrediblerangeofstructuraldiversitywithintriguingbiologicalproperties.Infact,themajorityoftoday’sapproveddrugsareeithernaturalproduct derivedorhavebeendevelopedbasedonnaturalproductleadstructures.On theothersideofthecoin,syntheticchemistryhasalsocontributedagreat deal.Fromourbasicneedsforfood,clothing,shelter,andmedicineto energy,defense,andevenart—andmostthingsinbetween—theadvances insyntheticstrategytomakemoleculeshasenabledusnotonlytoreplicate, ofteninquantity,whatisfoundinnaturebutimportantlytogobeyond naturebyproducingnewchemicalentitiesthatfulfillourfurtherneeds. Thehugerangesofthesemolecules—bothnaturalandnonnatural—have enormousimpactsonourmoderncivilizationaswellasonourexistence. Thatiswhychemicalsynthesisisoneofthemostvibrantareasofmodern scientificresearch,andwillcontinuetosupplythedrugs,materials,and commoditiesasperthedemandoftheday.
Inspirationfromnaturalproductshasindeedbroughtnewperspectivesto organicsynthesis.Thelast60yearsorsohavewitnessedrevolutionaryprogressinnaturalproductisolation,structuralelucidation,totalsyntheses,and biologicalstudies.Bioactivenaturalproductsareoftenfoundonlyinminisculequantities,andtohaveaparticularbioactivenaturalproductinample amountisthusoneamongmanyobjectivesofnaturalproductssyntheses. Totalsynthesis,athoroughanddidacticpresentationofthechemical synthesisofacomplexmolecule,oftenanaturalproduct,fromsimple,commerciallyavailableprecursors,isregardedasastateofthechemicalartatits highestdegreeofelegance,perception,andaestheticinnovation!
Thebirthoftotalsynthesistookplaceinthe19thcenturywhenW€ ohler accomplishedthesynthesisofurea denovo outsidethelivingsystemin1828 [1],simplyuponheatinganinorganicsubstance,ammoniumcyanate.The synthesisofaceticacidfromelementalcarbonbyKolbein1845 [2] isthe secondmajorachievementinthehistoryoftotalsynthesis.Thetotal synthesesofalizarinbyGraebeandLiebermann(1869) [3],indigobyBaeyer (1878) [4],and D-(+)-glucosebyFischer(1890) [5] representlandmark accomplishmentsinthehistoryoftotalsynthesisofthe19thcentury.Since then,chemistssynthesizedinnumerablebioactivenaturalproductsofsignificanceandpracticalutility.Thetotalsynthesisofcomplexnaturalproducts
stillremainsamongthemostexcitinganddynamicareasofresearch—total synthesisofsuchcomplexmoleculescoupledwithadvancementinsynthetic strategiesplayacrucialroleinmoderncivilization.
Fromafundamentalperspective,totalsynthesisisanauthenticplatform forprovingnewmethodologiesandnewstrategiesorwaysofthinking. Totalsynthesisalsocultivatesanunderstandingofthebasicprinciplesof chemistry:howandwhyreactionsoccur,therelationshipsbetweenmolecularshapeandfunction,stereochemicalaspects,andrelatedissues.Anability tosynthesizemoleculesremainsanessentialtrainingforthenextgeneration ofchemists.Woodwardonceargued,“Theuniquechallengewhichchemicalsynthesisprovidesforthecreativeimaginationandtheskilledhands ensuresthatitwillendureaslongasmenwritebooks,paintpictures,and fashionthingswhicharebeautiful,orpractical,orboth” [6]
Inthiscontext,anoutstandingreviewarticle,“Theartandscienceof totalsynthesisatthedawnofthetwenty-firstcentury”byProf.K.C.Nicalaouandhisgroup,mayberecommendedwheretheauthorsdemonstrated theelegancyandbeautyofthisremarkablechemicalsciencecoupledwithits necessityandaccomplishments [7].Itisalsofeltpertinenttoquotefromthe introductorywordsofProfessorA.Fredga,amemberoftheNobelPrize CommitteeforChemistryoftheRoyalSwedishAcademyofSciences,used tointroduceProf.R.B.WoodwardattheNobelceremoniesin1965,the yearinwhichWoodwardreceivedtheprizefortheartoforganicsynthesis, “Inourdays,thechemistryofnaturalproductsattractsaverylivelyinterest. Inthecourseoftheinvestigationofacomplicatedsubstance,the investigatorissoonerorlaterconfrontedbytheproblemofsynthesis,of thepreparationofthesubstancebychemicalmethods.Hecanhavevarious motives.Perhapshewantstocheckthecorrectnessofthestructurehehas found.Perhapshewantstoimproveourknowledgeofthereactionsandthe chemicalpropertiesofthemolecule.Ifthesubstanceisofpracticalimportance,hemayhopethatthesyntheticcompoundwillbelessexpensiveor moreeasilyaccessiblethanthenaturalproduct.Itcanalsobedesirableto modifysomedetailsinthemolecularstructure.Anantibioticsubstanceof medicalimportanceisoftenfirstisolatedfromamicroorganism,perhapsa mouldoragerm.Thereoughttoexistanumberofrelatedcompoundswith similareffects;theymaybemoreorlesspotent,somemayperhapshave undesirablesecondaryeffects.Itisbynomeans,orevenprobable,that thecompoundproducedbythemicroorganism – mostlikelyasaweapon inthestruggleforexistence – istheverybestfromthemedicinalpointof view.Ifitispossibletosynthesizethecompound,itwillalsobepossible
tomodifythedetailsofthestructureandtofindthemosteffectiveremedies. Thesynthesisofacomplicatedmoleculeis,however,averydifficulttask; everygroup,everyatommustbeplacedinitsproperpositionandthisshould betakeninitsmostliteralsense.Itissometimessaidthatorganicsynthesisis atthesametimeanexactscienceandafineart.… ” [8]
Syntheticorganicchemistryisnowassociatedwithbiology,medicinal chemistry,andmaterialsciences,andbeingacompositefieldofsuchsubdisciplines,organicchemistrywouldthusberequiredtoattainanappropriatecombinationofthesesubdisciplinestohaveapronouncedoverallsuccess ofanysyntheticendeavor.Thisfieldofchemicalsciencesisfacing uninterruptedchallengebynewchemicalscaffoldsisolatedfromnature’s seeminglyunlimitedlibraryofmoleculararchitectures.Atthesametime, thepracticeoftotalsynthesisisalsobeingenrichedprogressivelybynewly developedsyntheticstrategies,reagents,catalysts,aswellasadvancementin analyticalinstrumentationsfortherapidpurificationandcharacterizationof compounds.
Thustheoriginalgoaloftotalsynthesistoconfirmthestructureofan isolatednaturalproducthasbeenreplacedgraduallywithprimeobjectives relatedmoretotheexplorationanddiscoveryofnewchemistryalongthe pathwaytothetargetmolecule.Presently,biologicalissuesaredeeplyassociatedwiththetotalsynthesisprograms.Theevolutionofdrugdiscovery anddevelopmentprocessiscloselyrelatedtototalsynthesis—academic researchfocusingonorganicandnaturalproductsynthesisoffershighlyrelevantbasicknowledgeandtrainingtoyoungresearchersinthisdomain.The pharmaceuticalindustryappliestheknowledgegainedtodiscoveringand manufacturingnewdrugsforthebenefitsofsociety.Thatmedicinaland combinatorialchemistshavesomanytoolsattheirdisposaltodayintheir questsforhugenumbersofnovelanddiversesmallmolecules(natural productsand/ornaturalproduct-likestructures)isprimarilytheresultof thecontributionsoftotalsynthesisandoforganicsynthesisincombination. Thecommunityshouldconsidertotalsynthesisasavibrantandworthwhile pursuitaslongasitenablesaccesstofunctionallyusefulmoleculesand/or powerfulnewmethodsandstrategiesarebeinginvented.
Underthepurviewofthisenormouslypotentfieldoftotalsynthesis, TotalSynthesisofBioactiveNaturalProducts hasbeendesignedtocoverthe recentcutting-edgedevelopmentandstateoftheartoftotalsynthesisof potentiallyimportantandusefulnaturalsmallmolecules.Thebookoffers totalsyntheticapproachesfor60well-judgedbioactivenaturalproducts underdistinctentrieswithvividstep-by-stepdescriptionswiththehelp
ofgraphicsandschemessothatreaderscanfollowthechemistryinvolvedin eachstepofthetotalprocesstomakethemfamiliarwiththetacticsfortheir thoroughunderstandingandfollow-up.Theselectionshereinareonlythe tipoftheicebergandarerepresentativeoftheexcitingdevelopmentsthat continuetobereported.Thistimelyvolumewouldnotonlybehelpful toresearchers,professionals,andexpertsinvolvedwiththisremarkablefield butalsotoadvancedstudentsandyoungchemistsinmotivatingthemtothe dynamicfieldofthetotalsynthesisofusefulorganicmolecules.
Iwouldliketoexpressmysincerethanksanddeepsenseofgratitudeto ProfessorSrinivasanChandrasekaran,OrganicChemistryDivision,Indian InstituteofScience,Bangalore,forhiskeeninterestinthemanuscript andforwritingtheforewordtothebook.
IwouldalsoliketoexpressmydeepsenseofappreciationtoalltheeditorialandpublishingstaffmembersassociatedwithElsevierfortheirkeen interestinpublishingthisworkaswellastheirall-roundhelptoensurethat thehigheststandardsofpublicationhavebeenmaintainedinbringingout thisbook.Myeffortwillbesuccessfulonlywhenitisfoundhelpfulto readersatlarge.Everystephasbeentakentomakethemanuscripterrorfree; inspiteofthat,someerrorsmighthavecreptin.Anyremainingerrorsare,of course,myown.Constructivecommentsandapproachofthebookfrom readerswillbehighlyappreciated.
Finally,Iwouldtothankmywife(Piyasi)andmyson(Asanjan)fortheir understandingandallowingmeenoughtimethroughouttheentireperiod ofwriting;withouttheirsupport,thisworkwouldnothavebeensuccessful.
GOUTAM BRAHMACHARI ChemistryDepartment,Visva-BharatiUniversity,Santiniketan,India
References [1] F.W € ohler, Ann.Phys.Chem. 12(1828)253.
[2] H.Kolbe, Ann.Chem.Pharm.54(1845)145.
[3]a)C.Graebe,C.Liebermann, Ber.Dtsch.Chem.Ges.2(1869)332;b)firstcommercial synthesis:C.Graebe,C.Liebermann,H.Caro, Ber.Dtsch.Chem.Ges.3(1870)359.
[4]A.Baeyer, Ber.Dtsch.Chem.Ges.11(1878)1296–1297;(b)firstcommercialproduction: K.Heumann, Ber.Dtsch.Chem.Ges.23(1890)3431.
[5] E.Fischer, Ber.Dtsch.Chem.Ges. 23(1890)799–805.
[6] P.Ball, Nature 528(2015)327–329.
[7]K.C.Nicalaou,D.Vourloumis,N.Winssinger,P.S.Baran, Angew.Chem.Int.Ed.39 (2000)44–122.
[8]NobelLectures:Chemistry1963–1970,Elsevier,NewYork,1972,pp.96–123.
CHAPTERONE Aeruginosins298-AandB Abbreviations
Alloc allyloxycarbonyl
Bn benzyl
Boc tert-butoxycarbonyl
BOP (benzotriazol-1-yloxy)tris(dimethylamino)phosphoniumhexafluorophosphate
Bz benzoyl
Cbz benzyloxycarbonyl
CH2Cl2 dichloromethane
DEPBT 3-(diethoxyphosphoryloxy)-1,2,3-benzotriazin-4(3 H )-one
DEPC diethylcyanophosphinate
(2)-DIP-Cl ( )-B-chlorodiisopinocampheylborane
Et3N triethylamine
IBCF isobutylchloroformate
LS-Selectride lithiumtrisiamylborohydridesolution
NMM N-methylmorpholine
TBS tert-butyldimethylsilyl
TFA trifluoroaceticacid
THF tetrahydrofuran
TIPS triisopropylsilyl
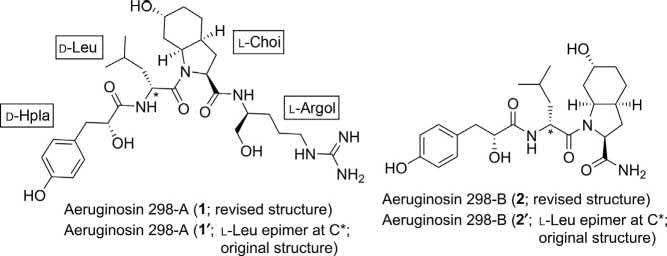
Systematicnames: (2S,3aS,6R,7aS)-N-((S)-5-Guanidino-1hydroxypentan-2-yl)-6-hydroxy-1-((R)-2-((R)-2-hydroxy-3-(4hydroxyphenyl)propanamido)-4-methylpentanoyl)octahydro-1H-indole2-carboxamide(1;aeruginosin298-A);(2S,3aS,6R,7aS)-6-hydroxy-1((R)-2-((R)-2-hydroxy-3-(4-hydroxyphenyl)propanamido)-4methylpentanoyl)octahydro-1H-indole-2-carboxamide(2;aeruginosin 298-B)
Compoundclass: Peptides
TotalSynthesisofBioactiveNaturalProducts © 2019ElsevierLtd. https://doi.org/10.1016/B978-0-08-102822-3.00001-8
Structure:
Naturalsource: Microcystisaeruginosa NIES-298(cyanobacterium) [1]
Pharmaceuticalpotential: Thrombinandtrypsininhibitors [1]
Syntheticroutes:
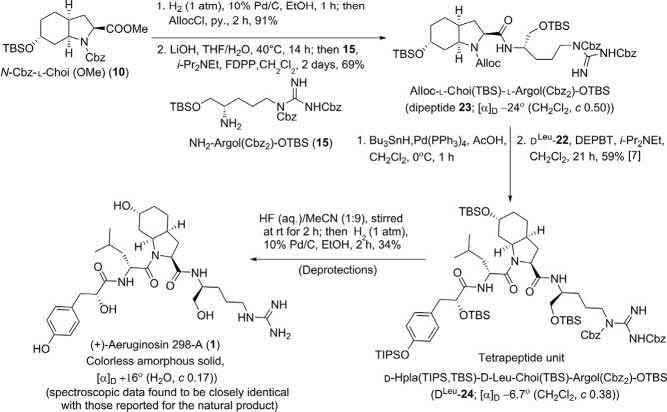
Thefirsttotalsynthesisoftheaquaticpeptideaeruginosin298-A(1)was reportedbyWipfandMethot(Schemes1–4) [2].Thepeptidemoleculeis composedoffourunitssuchas D-leucine(D-Leu;aminoacid),2-carboxy-6hydroxyoctahydroindole(L-Choi;anunusualbicyclic α-aminoacidcore), α-hydroxy-p-hydroxyphenyllacticacid(D-Hpla),and L-argininol(L-Argol).
The L-Choi(10), L-Argol(15),and D-Hpla-D-Leu(22)segmentsweresynthesizedseparately,andthenassembledalltogethertoframethestructure 1 asdepictedin Schemes1–4.
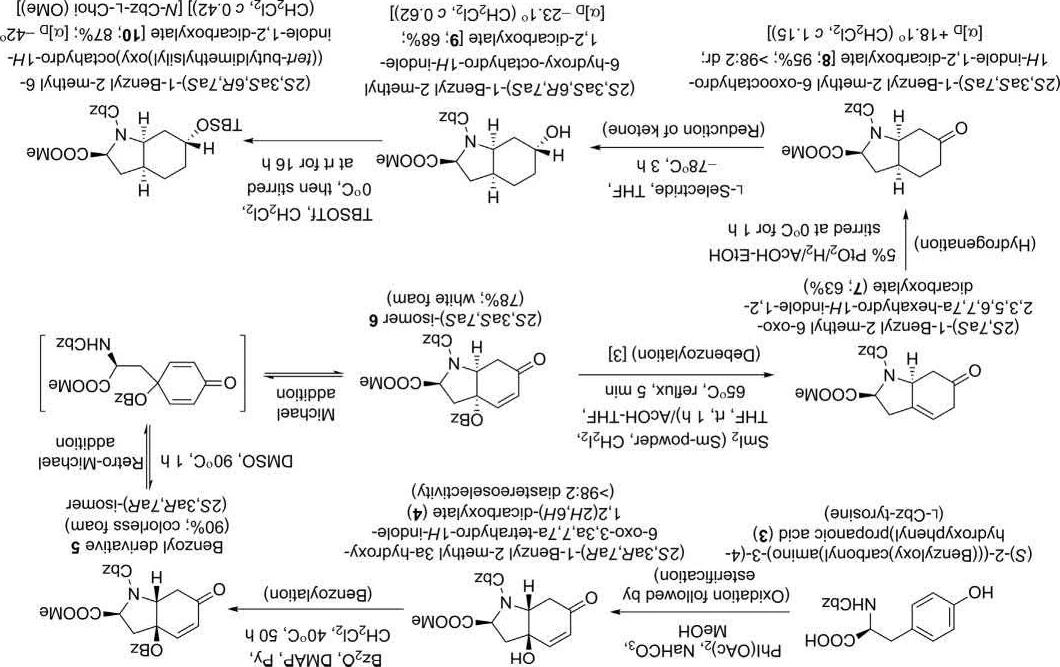
Synthesisof L-Choisegment(10):
Synthesisofthissegmentwasinitiatedwith L-Cbz-tyrosine(3)(Scheme 1).OnoxidationwithPhI(OAc)2 followedbyesterificationwithmethanol, compound 3 afforded(2S,3aR,7aR)-1-benzyl2-methyl3a-hydroxy-6oxo-3,3a,7,7a-tetrahydro-1H-indole-1,2(2H,6H)-dicarboxylate(4)ina diastereoselectivityof >98:2.Its syn-diastereomer 6 wasachievedthrough athermodynamicequilibrationofthebenzoyl-protectedalcohol 5 viaa retro-Michael/Michaeladditionsequencereaction.Compound 7 was obtainedondebenzoylationof 6 ingoodyieldusingSmI2 [3].Hydrogenationof 7 followedby L-SelectridereductionandTBSprotectionafforded the L-Choicore(10).
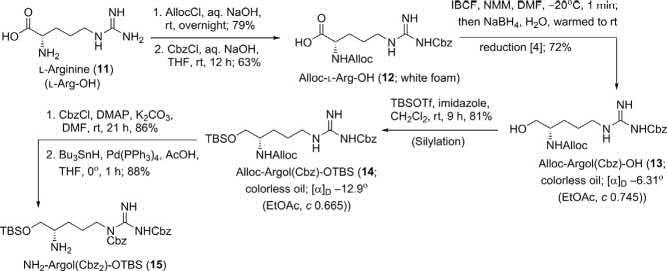
Synthesisof L-Argolsegment(15):
The L-Argolsegment(15)wassynthesizedinsixstepsfrom L-arginine (11)(Scheme2).SelectiveAllocandCbzprotectionfollowedbyinsitu sodiumborohydridereduction [4] ofthemixedanhydrideformedwith
Scheme1 Synthesisof L-Choisegment(10) [2].
Synthesisof L-Argolsegment(15) [2].
isobutylchloroformate(IBCF)yieldedtheprimaryalcoholAlloc-Argol (Cbz)-OH(13).Standardprotectionanddeprotectiontechniques implementedinthenextthreestepsaffordedthe L-Argolsegment 15 in goodyield.
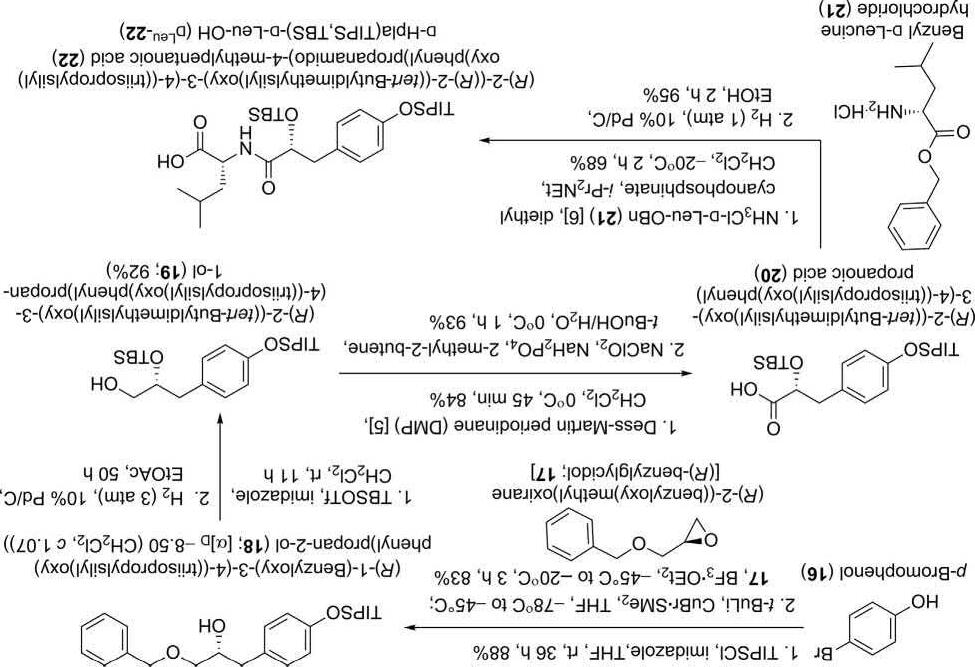
Synthesisof D-Hpla-D-Leusegment(22):
TheinvestigatorsstartedthesynthesisofthissegmentusingaBF3 OEt2catalyzedorganocuprateadditionof(R)-benzylglycidol 17 withtheprotected4-bromophenoltogeneratethe(R)-1-(benzyloxy)-3-(4((triisopropylsilyl)oxy)phenyl)propan-2-ol(18)in83%yield(Scheme3). StandardprotectivegroupmanipulationsfollowedbyDess-Martinperiodinane [5] andsodiumperchlorateoxidationsresultedin D-Hpla 20. Finally,diethylcyanophosphinate(DEPC)-mediatedcouplingto D-LeuOBn(21)andhydrogenolysisprovidedthedesired D-Hpla-D-Leusegment (DLeu-22).
Assemblyof L-Choi(10), L-Argol(15),and D-Hpla-D-Leu(22) segments:totalsynthesisofaeruginosin298-A(1):
Atthisstage,theinvestigatorsassembledtherespectivesegmentstoarrive atthetetrapeptideframeworkofthetargetmolecule 1 (Scheme4).TheCbz groupof N-Cbz-L-Choi(OMe)(10)wasexchangedwithanAllocgroup, andwasthensubjectedtosubsequentsaponificationandpentafluorophenyl ester-mediatedcouplingto L-Argol 15[NH2-Argol(Cbz2)-OTBS]toafford thedipeptideAlloc-L-Choi(TBS)-L-Argol(Cbz2)-OTBS(23).Alloc deprotectionfollowedby3-(diethoxyphosphoryloxy)-1,2,3-benzotriazin4(3H)-one(DEPBT)-mediatedcoupling [7] to D Leu -22 resultedintheformationofthetetrapeptide D Leu -24 in59%yield.Thetetrapeptidewas treatedwithHF(H2O-MeCN1:9)for2h,neutralizedwithaq.NaOH, andextractedintoCH2Cl2/EtOAc.Finally,hydrogenolysiscleavedthe Cbz-protectivegroups(34%;twosteps)tofurnish(+)-aeruginosin298-A
Scheme2
Scheme3 Synthesisof D-Hpla-D-Leusegment(22) [2].
Scheme4 Totalsynthesisof(+)-aeruginosin298-A(1) [2].
(1)aswhiteamorphoussolid;spectroscopicdataofthissyntheticcompound completelymatchedthosereportedforthenaturalproduct [1]
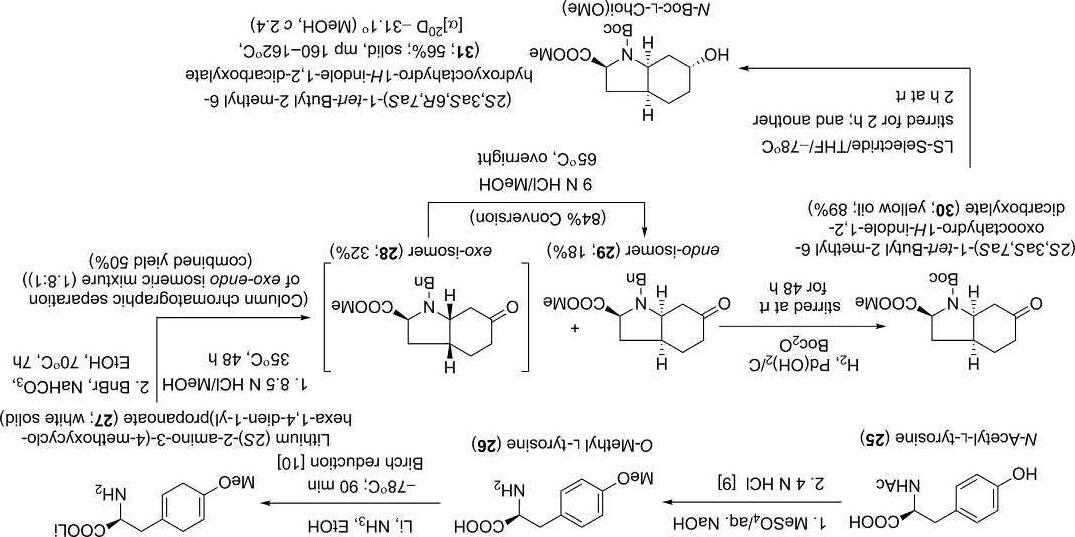
Almostinaparallelreporting,Bonjochandhisgroup [8] outlined anotherrouteforthetotalsynthesesforbothaeruginosin298-A(1)and 298-B(2)(Schemes5–7).Theinvestigatorsfirstprepared L-Choifragment 31 and D-Hplafragment 36 asdepictedbelow.
Synthesisof N-Boc-L-Choi(OMe)(31):
(2S,3aS,6R,7aS)-1-tert-Butyl2-methyl6-hydroxyoctahydro-1Hindole-1,2-dicarboxylate[N-Boc-L-Choi(OMe)(31)]waspreparedstarting from N-acetyl-L-tyrosine(25):
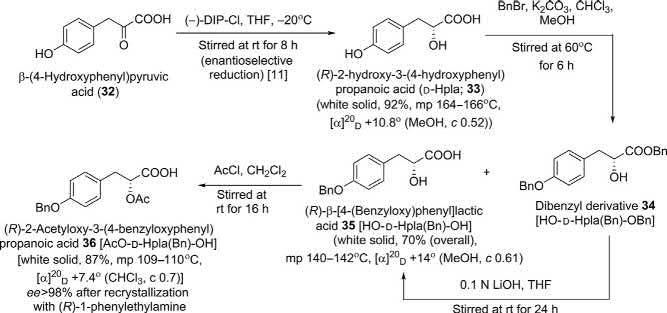
Synthesisof D-Hplafragment(36):
Theprotectedderivativeof D-Hpla 36 wassynthesizedstartingfrom β(4-hydroxyphenyl)pyruvicacid(32)(Scheme6);onenantioselectivereductionfollowingWang’sprocedure [11],compound 32 furnished(R)-2hydroxy-3-(4-hydroxyphenyl)propanoicacid(D-Hpla; 33)in92%yield with86% ee.Thenaftersequentialprotectionofbothhydroxylgroups andrecrystallizationwith(R)-1-phenylethylamine,theinvestigators obtainedthedesired 36 in >98% ee
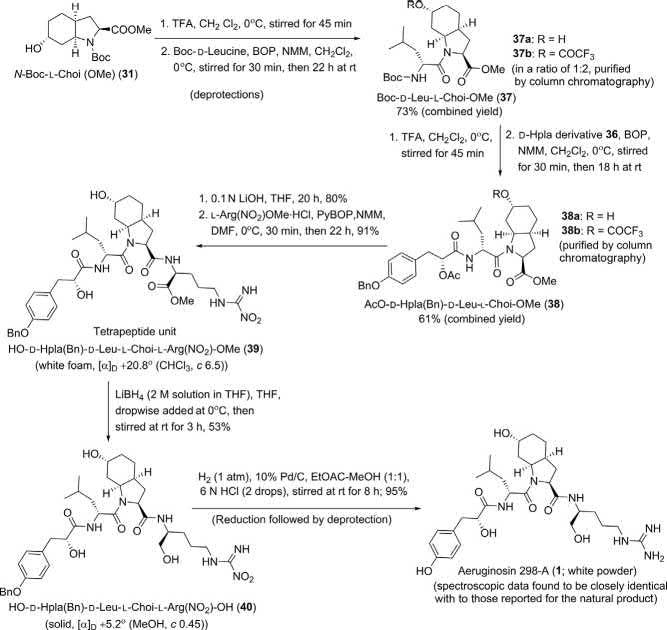
Assemblyofthesegments:totalsynthesisofaeruginosin298-A (1)andaeruginosin298-B(2):
Theinvestigators [8] thenassembledthecorrespondingfragmentsto achievethetargetmoleculesaeruginosin298-A(1)andaeruginosin298B(2)asdescribedinthefollowing Schemes7and8
Synthesisof N-Boc-L-Choi(OMe)(31) [8].
Scheme5
Synthesisofprotectedderivativeof D-Hpla(36) [8].
Scheme7 Totalsynthesisofaeruginosin298-A(1) [8].
Scheme6
Scheme8 Totalsynthesisofaeruginosin298-B(2) [8].
Lateron,Shibasakiandhisgroupalsodevelopedaversatileand enantioselectivesyntheticprocessforaeruginosin298-A(1)anditsseveral attractiveanalogsusingacatalyticasymmetricphase-transferreactionand epoxidation,andevaluatedtheirbiologicalpotentialsaswell [12,13]
References [1] (a)M.Murakami,Y.Okita,H.Matsuda,T.Okino,K.Yamaguchi,TetrahedronLett. 35(1994)3129;(b)M.Murakami,K.Ishida,T.Okino,H.Matsuda,K.Yamaguchi, TetrahedronLett.36(1995)2785;(c)K.Ishida,H.Okita,H.Matsuda,T.Okino,M. Murakami,Tetrahedron55(1999)10971.
[2] P.Wipf,J.-L.Methot,Org.Lett.2(2000)4213.
[3] G.A.Molander,Org.React.46(1994)211.
[4] M.Rodriguez,M.Llinares,S.Doulut,A.Heitz,J.Martinez,TetrahedronLett. 7(1991)923.
[5] (a)D.B.Dess,J.C.Martin,J.Am.Chem.Soc.113(1991)7277;(b)R.E.Ireland,L. Liu,J.Org.Chem.58(1993)2899.
[6] T.Wakamiya,M.Kamata,S.Kusumoto,H.Kobayashi,Y.Sai,Bull.Soc.Chem.Jpn. 71(1998)699.
[7] H.Li,X.Jiang,Y.Ye,C.Fan,T.Romoff,M.Goodman,Org.Lett.1(1999)91.
[8] N.Valls,M.Lόpez-Canet,M.Vallribera,J.Bonjoch,Chem.Eur.J.7(2001)3446.
[9] (a)B.W € unsch,M.Zott,LiebigsAnn.Chem.(1992)39;(b)W.Siedel,K.Sturm,R. Geiger,Chem.Ber.96(1963)1636.
[10] B.C.Laguzza,B.Ganem,TetrahedronLett.22(1981)1483.
[11] Z.Wang,B.La,J.M.Fortunak,X.-J.Meng,G.W.Kabalka,TetrahedronLett. 39(1998)5501.
[12] T.Ohshima,V.Gnanadesikan,T.Shibuguchi,Y.Fukuta,T.Nemoto,M.Shibasak,J. Am.Chem.Soc.125(2003)11206.
[13] Y.Fukuta,T.Ohshima,V.Gnanadesikan,T.Shibuguchi,T.Nemoto,T.Kisugi,T. Okino,M.Shibasaki,Proc.Natl.Acad.Sci.USA101(2004)5433.
AgeladineA Abbreviations
Biphenyl-PCy2 2-biphenyldicyclohexylphosphine
BOC tert-butoxycarbonyl
BOM benzyloxymethyl
DMF N,N-dimethylformamide
HPLC highperformanceliquidchromatography
MMPs matrixmetalloproteinases
n-BuLi n-butyllithium
Sc(OTf)3 scandiumtriflate
THF tetrahydrofuran
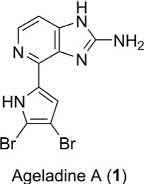
Systematicname: 4-(4,5-Dibromo-1H-pyrrol-2-yl)-1H-imidazo[4,5-c] pyridin-2-amine
Compoundclass: Alkaloid(pyrrole-imidazolealkaloid)
Structure:
Naturalsource: Agelasnakamurai Hoshino(marinespongecollectednear Kuchinoerabu-jimaIslandinsouthernJapan;family:Agelasidae) [1]
Pharmaceuticalpotential: MMPsinhibitor(antiangiogenicandanticancer) [1–10];pH-sensitivemembranepermeabledye [11,12]
Syntheticroutes:
AgeladineA(1),ananticancermarinepyrrole-imidazolealkaloid,was isolatedfromthemarinesponge Agelasnakamurai byFusetaniandcoworkers [1];thebrominatedalkaloidmetaboliteisthefirstexampleofan imidazolopyridinemarinenaturalproduct.ItspromisingMMPs’inhibitory
TotalSynthesisofBioactiveNaturalProducts
© 2019ElsevierLtd.
https://doi.org/10.1016/B978-0-08-102822-3.00002-X Allrightsreserved.
activityandunusualcompactstructuremotivatedsyntheticchemiststoward itstotalsynthesis;asaresultatleastfivesyntheticstrategiesforthetotal synthesisofageladineAanditsanalogshavebeenreportedsofar [2–10]. In2006,MeketaandWeinreb [2] reportedthefirsttotalsynthesisof 1 using a6π-1-azaelectrocyclizationandSuzuki-Miyauracouplingof N-Bocpyrrole-2-boronicacidandachloropyridinederivativeasakeystep.Soon afterinthesameyear,ShenguleandKaruso [3] reportedtheconcise synthesisof 1 basedonbiomimeticprinciplesinvolvingapivotalPictetSpengler-typecondensationbetween2-aminohistamineand N-Boc-4,5dibromo-2-formylpyrrole.Weinrebandhisgroupfurtherreportedthe thirdtotalsynthesisusingavariationofhisoriginalmethodemployinga 6π-2-azatrieneelectrocyclizationfortheformationoftheimidazolopyridine moiety [5–7].Lateron,Andoetal.reportedafourthsynthesisofageladine AanditsanalogsbasedonthePictet-Spenglercyclizationbetweenthe N-Boc-2-aminohistamineand N-protected2-formylpyrroles [8,9].Most recently,Karusoandhisgroupreportedtheone-potsynthesisof 1 anda rangeofanalogsfrom2-aminohistamineandvariousheterocyclicaldehydes [10].Hereinthefirsttwototalsyntheticstrategiesarediscussed.
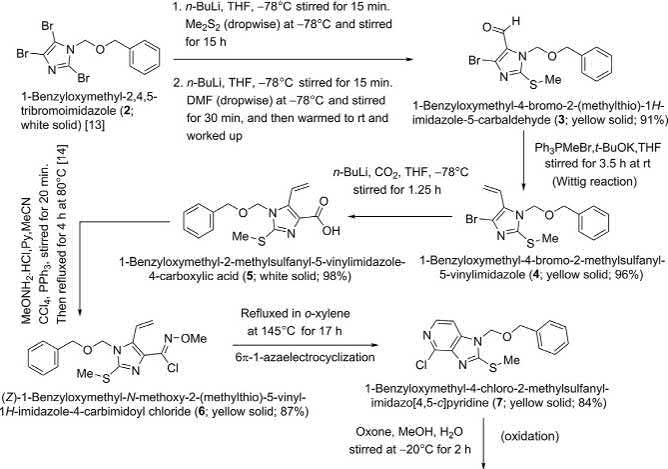
MeketaandWeinrebaccomplishedthetotalsynthesisofthetricyclic heteroaromaticmarinemetaboliteageladineA(1 )forthefirsttimein 12consecutivestepsusinga6π-1-azaelectrocyclizationandSuzukiMiyauracouplingtechniqueasthekeyreactions( Scheme1) [2] .Thereadilyavailablebenzyloxymethyl(BOM)-protectedtribromoimidazole( 2) [13] wasfirstmetalatedwith n -BuLiatC-2,andathiomethylgroup wasintroducedthereonusingdimethyldisulfide.Asecondequivalent of n-BuLiwasthenaddedtothereactionmixturetoeffectmetalation atC-5,followedbytreatmentwithDMF,leadingtotheformationof 3-benzyloxymethyl-5-bromo-2-methylsulfanylimidazole-4carboxaldehyde( 3)in91%overallyieldfortheone-potoperation.The bromovinylimidazole 4 ,obtainedfromtheimidazolealdehyde 3 viaWittigreaction,wasthenconvertedtoBO M-protectedvinylimidazolecarboxylicacid 5 bymeansoflithiationof 4 atC-4followedbytreatment withcarbondioxidein98%yield.ApplyingthemethodologyofKikugawa etal. [14] ,theinvestigatorstransformedthisBOM-protectedcompound 5 directlyinto N -methoxyimidoylchloride 6 (87%yield),whichthen underwent6π-1-azaelectrocyclizationonrefluxingin o-xyl ene at 150 °C affordingchloropyridine 7 in84%isolatedyield.
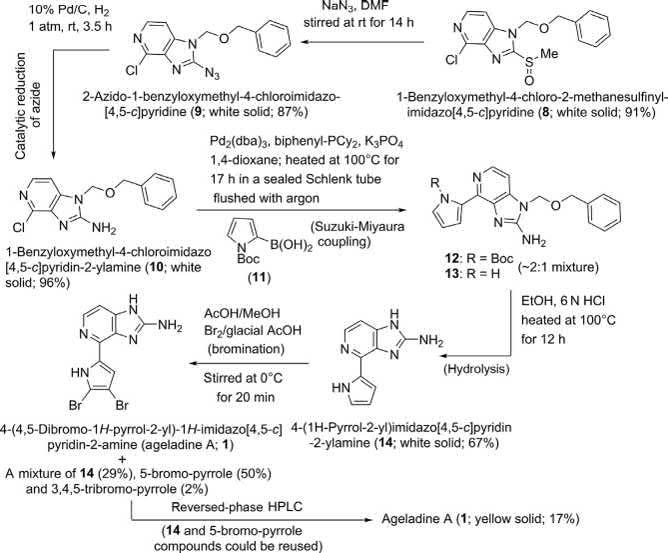
TheinvestigatorsobservedthatSuzuki-MiyauracouplingofBOMprotectedchloropyridine 7 with N-Boc-boronicacid 11 couldnotbeeffected, andthustheymodifiedtheirstrategy.Thesulfidemoietyin 7 wasfirstoxidized
Scheme1 Weinreb’stotalsynthesisofageladineA(1) [2]
withoxonetothesulfoxide 8,followedbyitsdisplacementwithsodiumazide atroomtemperatureaffordingtherebytheazide 9 in87%yield.Catalytic hydrogenationofthisazidethencleanlyproduced2-aminoimidazolopyridine 10,whichthenunderwentSuzuki-Miyauracouplingwith 11 inthepresence ofBuchwald’s2-biphenyldicyclohexylphosphineligand [15] tofurnisha 2:1mixtureofBoc-protectedtricyclicpyrrole 12 andcompound 13 (withouttheBocgroup).Thiscrudemixturewasthenhydrolyzedwith6N HClinethanoltoafforddeprotectedtricycle 14 in67%overallyieldfrom chloropyridine 10.Finally,brominationofpyrrole 14 waseffectedupon treatmentwithbromineincoldaceticacid/methanolmedium,which producedageladineA(1)(17%),alongwithrecoveredstartingmaterial (14;29%),the5-bromo-pyrrole(50%),andasmallamountof3,4,5tribromopyrrole(2%).Thesecompoundswereseparablebyreverse-phase HPLC.Boththemonobromopyrroleandthestartingmaterial 14 could berecycledtothenaturalproduct.SyntheticageladineA(C10H8N5Br2, yellowsolid)wasfoundtohaveidenticalphysicalandspectralproperties tothosereportedforthenaturalmaterial [1]
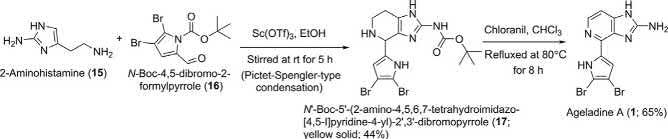
SoonafterthefirstreportofMeketaandWeinreb [2],aconcisetotalsynthesisofageladineAinjusttwostepswaspublishedbyShenguleandKaruso inthesameyear(Scheme2) [3].Theyachievedthisshorterrouteforthe synthesisof 1 byexploitingaPictet-Spengler-typecondensationbetween 2-aminohistamine(15)and N-Boc-4,5-dibromo-2-formylpyrrole(16)as thekeystep.Thestartingmaterial 15 iscommerciallyavailableorcanbepreparedfromBoc-guanidineand β-alanineasperreportedmethods [16,17] Similarly,4,5-dibromo-2-formylpyrrole(16)isalsocommerciallyavailable, andcanbeeffectedinnearquantitativeyieldfrom2-formylpyrroleintwo steps [18].Pictet-Spengler-typecondensationbetween2-aminohistamine (15)and N-Boc-4,5-dibromo-2-formylpyrrole(16)wascarriedoutbystirring thereactantsinethanolatroomtemperaturefor5hinthepresenceof theLewisacidcatalystSc(OTf)3 tofurnish N0 -Boc-50 -(2-amino-4,5,6,7tetrahydroimidazo[4,5-c]pyridine-4-yl)-20 ,30 -dibromopyrrole(17;yellow solid)in44%yield.Dehydrogenationanddeprotectionof 17 werecleanly
Scheme2 Karuso’stotalsynthesisofageladineA(1) [3].
effectedbyrefluxingitwithchloranilinchloroformfor8htoyieldageladine A(1)asafluorescentyellowsolid,identicalinallrespectstothenatural product [1].
References [1] M.Fujita,Y.Nakao,S.Matsunaga,M.Seiki,Y.Itoh,J.Yamashita,R.W.M.van Soest,N.Fusetani,J.Am.Chem.Soc.125(2003)15700.
[2] M.L.Meketa,S.M.Weinreb,Org.Lett.8(2006)1443.
[3] S.R.Shengule,P.Karuso,Org.Lett.8(2006)4083.
[4] Y.Nakao,N.Fusetani,J.Nat.Prod.70(2007)689.
[5] M.L.Meketa,S.M.Weinreb,Y.Nakao,N.Fusetani,J.Org.Chem.72(2007)4892.
[6] M.L.Meketa,S.M.Weinreb,Tetrahedron63(2007)9112.
[7] M.L.Meketa,S.M.Weinreb,Org.Lett.9(2007)853.
[8] N.Ando,S.Terashima,Bioorg.Med.Chem.Lett.17(2007)4495.
[9] N.Ando,S.Terashima,Bioorg.Med.Chem.Lett.19(2009)5461.
[10] S.R.Shengule,W.L.Loa-KumCheung,C.R.Parish,M.Blairvacq,L.Meijer, Y.Nakao,P.Karuso,J.Med.Chem.54(2011)2492.
[11] U.Bickmeyer,A.Grube,K.W.Klings,M.Kock,Biochem Biophys.Res.Commun. 373(2008)419(Erratumin: Biochem.Biophys.Res.Commun.,2009, 383,519).
[12] U.Bickmeyer,M.Heine,I.Podbielski,D.Mund,M.Kock,P.Karuso,Biochem. Biophys.Res.Commun.402(2010)489.
[13] R.W.Schumacher,B.S.Davidson,Tetrahedron55(1999)935.
[14] Y.Kikugawa,L.H.Fu,T.Sakamoto,Synth.Commun.23(1993)1061.
[15] T.E.Barder,S.D.Walker,J.R.Martinelli,S.L.Buchwald,J.Am.Chem.Soc. 127(2005)4685.
[16] R.G.Jones,E.C.Kornfeld,K.C.McLaughlin,J.Am.Chem.Soc.72(1950)4526.
[17] T.L.Little,S.E.Webber,J.Org.Chem.59(1994)7299.
[18] S.T.Handy,J.J.Sabatini,Y.Zhang,I.Vulfova,TetrahedronLett.45(2004)5057.
CHAPTERTHREE (+)-AinsliadimerA Abbreviations
Ar argon
BINOL 1,10 -bi-2-naphthol
CH2Cl2 dichloromethane
DABCO 1,4-diazobicyclo[2.2.2]octane
DBU 1,8-diazabicyclo[5.4.0]undec-7-ene
EtOAc ethylacetate
H2O2 hydrogenperoxide
HMPA hexamethylphosphoramide
iPr isopropyl
LDA lithiumdiisoproylamide
m-CPBA meta-chloroperbenzoicacid
MsCl methanesulfonylchloride
NaBH4 sodiumborohydride
NO nitricoxide
PhSe-SePh diphenyldiselenide
Py pyridine
SOCl2 thionylchloride
THF tetrahydrofuran
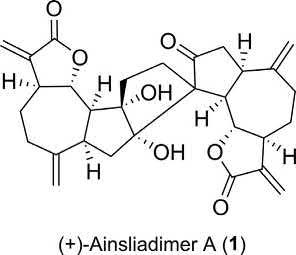
Systematicname: (3aS,3a0 S,6aR,6a0 R,7a0 S,9aR,9bS,10a0 S,10b0 S,10c0 S)7a0 ,10a0 -dihydroxy-3,30 ,6,60 -tetramethylenehexadecahydro-2H-spiro[azuleno [4,5-b]furan-9,80 -cyclopenta[2,3]azuleno[4,5-b]furan]-2,20 ,8(3H,9aH,9bH, 90 H,10b0 H,10c0 H)-trione
Compoundclass: Guaianolidesesquiterpenedimer
TotalSynthesisofBioactiveNaturalProducts © 2019ElsevierLtd. https://doi.org/10.1016/B978-0-08-102822-3.00003-1
Structure:
Naturalsource: Ainsliaeamacrocephala (wholeplants;family: Compositae) [1]
Pharmaceuticalpotential: InhibitortoNOproductionin RAW264.7stimulatedbylipopolysaccharide(IC50 ¼ 2.41 μg/mL) [1]
Syntheticroute:
(+)-AinsliadimerA(1),aguaianolidesesquiterpenedimerisolatedfrom theChinesemedicinalplant A.macrocephala,bearsauniqueheptacyclicring systemhaving11contiguousstereogeniccentersandahighlyfunctionalized cyclopentaneringwiththreequaternarycarbonsconnectingthetwomonomericsesquiterpenelactoneunits.Theseuniquearchitecturalfeaturescombinedwithitspotentinhibitoryeffectagainstnitricoxide(NO)production madethismoleculeasanidealtargetfortotalsynthesis.Leiandhisgroup [2] reportedthefirsttotalsynthesisof(+)-ainsliadimerAstartingfrom α-stantoninbasedonaconciseandbiomimeticapproachinvolving14 steps—ahydrogenbonding-promoted[4+2]-hetero-Diels-Alderdimerizationisthekeystepofthesyntheticstrategy(Scheme1).Thesynthetic approachdemonstratedthefeasibilityofusingnonenzymaticconditions toachievetheproposedbiosynthesispathofthenaturalproduct 1 [1].
TheinvestigatorsfirstsynthesizeddehydrozaluzaninC(12)fromcommerciallyavailable α-santonin(2).Onphotoirradiationwithahigh-pressureHg lamp(500W)inaceticacid, α-santonin(2)afforded O-acetylisophotosantonic lactone(3)asacolorlesssolid [3–5],whichwassubsequentlyconvertedto compound 4 inquantitativeyieldandexcellentdiastereoselectivityonhydrogenationofthedoublebondwithPd-C/H2.Selectivereductionofketone 4, followedbymesylationofthecorrespondinghydroxylgroupandinsitu antielimination,thereafterfurnishedalkene 6 (52%overtwosteps) [6]
Compound 6 underwentsmoothsaponificationreactionwith5%aq.