https://ebookmass.com/product/processing-contaminants-in-
Instant digital products (PDF, ePub, MOBI) ready for you
Download now and discover formats that fit your needs...
Phenomenology Shaun Gallagher
https://ebookmass.com/product/phenomenology-shaun-gallagher/
ebookmass.com
Processing for Android: Create Mobile, Sensor-aware, and XR Applications Using Processing 2nd Edition Colubri
https://ebookmass.com/product/processing-for-android-create-mobilesensor-aware-and-xr-applications-using-processing-2nd-edition-colubri/
ebookmass.com
Processing for Android: Create Mobile, Sensor-aware, and XR Applications Using Processing, 2nd Edition Andrés Colubri
https://ebookmass.com/product/processing-for-android-create-mobilesensor-aware-and-xr-applications-using-processing-2nd-edition-andrescolubri/
ebookmass.com
A Dish for All Seasons Kathryn Pauline
https://ebookmass.com/product/a-dish-for-all-seasons-kathryn-pauline/
ebookmass.com
https://ebookmass.com/product/comparative-international-law-roberts/
ebookmass.com
Corruption and Fraud in Financial Markets: Malpractice, Misconduct and Manipulation Carol Alexander
https://ebookmass.com/product/corruption-and-fraud-in-financialmarkets-malpractice-misconduct-and-manipulation-carol-alexander/
ebookmass.com
A First Look at Communication Theory - 11th Edition Em Griffin
https://ebookmass.com/product/a-first-look-at-communicationtheory-11th-edition-em-griffin/
ebookmass.com
Spacecraft Systems Engineering 4th Edition – Ebook PDF Version
https://ebookmass.com/product/spacecraft-systems-engineering-4thedition-ebook-pdf-version/
ebookmass.com
Managerial Economics & Business Strategy, 10e ISE 10th/ISE Edition Michael R. Baye
https://ebookmass.com/product/managerial-economics-businessstrategy-10e-ise-10th-ise-edition-michael-r-baye/
ebookmass.com
https://ebookmass.com/product/smart-money-strategy-your-ultimateguide-to-financial-planning-luke-smith/
ebookmass.com
PROCESSING CONTAMINANTSIN EDIBLEOILS
Thispageintentionallyleftblank
PROCESSING CONTAMINANTSIN EDIBLEOILS
MCPDandGlycidylEsters
SECONDEDITION
Editedby SHAUNMACMAHON
U.S.FoodandDrugAdministration, CenterforFoodSafetyandAppliedNutrition(CFSAN), MD,UnitedStates
JESSICAK.BEEKMAN
U.S.FoodandDrugAdministration, CenterforFoodSafetyandAppliedNutrition(CFSAN), MD,UnitedStates
AcademicPressandAOCSPress
AcademicPressisanimprintofElsevier 125LondonWall,LondonEC2Y5AS,UnitedKingdom 525BStreet,Suite1650,SanDiego,CA92101,UnitedStates 50HampshireStreet,5thFloor,Cambridge,MA02139,UnitedStates TheBoulevard,LangfordLane,Kidlington,OxfordOX51GB,UnitedKingdom
Copyright © 2022AOCSPress.PublishedbyElsevierInc.Allrightsreserved.Published incooperationwithAmericanOilChemistsSociety.
PublishedincooperationwithAmericanOilChemists’ Society www.aocs.org Director, ContentDevelopment:JanetBrown
Nopartofthispublicationmaybereproducedortransmittedinanyformorbyanymeans, electronicormechanical,includingphotocopying,recording,oranyinformationstorage andretrievalsystem,withoutpermissioninwritingfromthepublisher.Detailsonhowto seekpermission,furtherinformationaboutthePublisher’spermissionspoliciesandourarrangementswithorganizationssuchastheCopyrightClearanceCenterandtheCopyright LicensingAgency,canbefoundatourwebsite: www.elsevier.com/permissions.
Thisbookandtheindividualcontributionscontainedinitareprotectedunder copyrightbythePublisher(otherthanasmaybenotedherein).
Notices
Knowledgeandbestpracticeinthis fieldareconstantlychanging.Asnewresearch andexperiencebroadenourunderstanding,changesinresearchmethods,professional practices,ormedicaltreatmentmaybecomenecessary.
Practitionersandresearchersmustalwaysrelyontheirownexperienceandknowledgein evaluatingandusinganyinformation,methods,compounds,orexperimentsdescribed herein.Inusingsuchinformationormethodstheyshouldbemindfuloftheirownsafety andthesafetyofothers,includingpartiesforwhomtheyhaveaprofessionalresponsibility.
Tothefullestextentofthelaw,neitherthePublishernortheauthors,contributors,or editors,assumeanyliabilityforanyinjuryand/ordamagetopersonsorpropertyasa matterofproductsliability,negligenceorotherwise,orfromanyuseoroperationof anymethods,products,instructions,orideascontainedinthematerialherein.
LibraryofCongressCataloging-in-PublicationData
AcatalogrecordforthisbookisavailablefromtheLibraryofCongress
BritishLibraryCataloguing-in-PublicationData
AcataloguerecordforthisbookisavailablefromtheBritishLibrary
ISBN:978-0-12-820067-4
ForinformationonallAcademicPressandAOCSPresspublications visitourwebsiteat https://www.elsevier.com/books-and-journals
Publisher: NikkiP.Levy
AcquisitionsEditor: NancyMaragioglio
EditorialProjectManager: SamW.Young
ProductionProjectManager: BharatwajVaratharajan
CoverDesigner: MarkRogers
1.Introduction1
ShaunMacMahon
Formation1
Mitigation1
Analysis2
Toxicology3
Regulations4
References5
2.Formationmechanisms7
BrianD.CraftandFrédéricDestaillats
Introduction7 MCPDesters8 Glycidylesters12
Conclusionsandperspectives20 References20
3.MitigationofMCPDandglycidylestersinedibleoils23
BertrandMatthäusandFrankPudel
Introduction23
Influenceofprecursorsonesterformation26
Mitigationin field37
Mitigationoftherawmaterial39
Mitigationinrefining42
Additives52
Removalfromfullyrefinedoils(post-refining)56 References59
4.Indirectdetectiontechniques65 KarelHrncirík
Introduction65
Mainstepsintheanalysisof2-and3-MCPDesters66 Mainapproachestowardtheanalysisofglycidylesters75
Officiallyadoptedmethods83 Methodcomparison89 Methodautomation96 Analysisofoil-basedfoodstuffs97 Challengesinfoodstuffanalysis98 Conclusions103 References104
5.Directanalyticaldetectionmethodsforthefood-borne toxicantsglycidylesters109 AliceEwertandMichaelGranvogl Introduction109 Directmethods111 References142
6.DirectdetectiontechniquesforMCPDesters145
ShaunMacMahon Background145 Directmethods146 TheInstituteofChemicalTechnology162 Conclusion191 References191
7.MethodstodetectMCPDandglycidylestersincomplexfood matrices195
ShaunMacMahon,JessicaBeekmanandMichaelGranvogl Background195 Discussion196 Conclusions217 References217
8.Toxicologicalpropertiesofglycidylesters221
GabrieleScholzandBenoîtSchilter Introduction221 Biologicalpropertiesofglycidylesters222 Biologicalpropertiesoffreeglycidol225 Conclusions231 References232
9.ToxicologicalpropertiesofMCPDfattyacidesters235
AlfonsoLampen
Abbreviations235
Introduction235
Toxicologyof3-MCPD236
Hazardpotentialof3-MCPDfattyacidesters242
Toxicologyof2-MCPDesters247
Riskcharacterization248 Conclusions249 References250
Thispageintentionallyleftblank
Contributors
JessicaBeekman
FDACenterforFoodSafetyandAppliedNutrition,CollegePark,MD,UnitedStates
BrianD.Craft
NestléResearchCenter,FoodScienceandTechnologyDepartment,Lausanne, Switzerland
FrédéricDestaillats
NestléResearchCenter,FoodScienceandTechnologyDepartment,Lausanne, Switzerland
AliceEwert
GermanResearchCenterforFoodChemistry,Freising,Germany
MichaelGranvogl
UniversityofHohenheim,InstituteofFoodChemistry,DepartmentofFoodChemistry andAnalyticalChemistry(170a),Stuttgart,Germany;TechnicalUniversityofMunich, DepartmentofChemistry,ChairforFoodChemistry,Freising,Germany
KarelHrncirík
Upfield,Rotterdam,theNetherlands
AlfonsoLampen
FederalInstituteforRiskAssessment(BfR),Berlin,Germany
ShaunMacMahon
FDACenterforFoodSafetyandAppliedNutrition,CollegePark,MD,UnitedStates
BertrandMatthäus
MaxRubner-Institut,DepartmentforSafetyandQualityofCereals,WorkingGroupfor LipidResearch,Detmold,Germany
FrankPudel
PilotPflanzenöltechnologieMagdeburge.V.,Magdeburg,Germany
BenoîtSchilter
SociétédesProduitsNestléS.A.,NestléResearch,Lausanne,Switzerland
GabrieleScholz
SociétédesProduitsNestléS.A.,NestléResearch,Lausanne,Switzerland
Thispageintentionallyleftblank
CHAPTER1
Introduction
ShaunMacMahon FDACenterforFoodSafetyandAppliedNutrition,CollegePark,MD,UnitedStates
Formation
Toimproveconsumeracceptance,edibleoilsareindustriallyprocessedto removeormodifycomponentsthatcannegativelyimpactappearance,taste, andshelfstability.However,undesirablechemicalchangescantakeplace duringtherefiningprocess.Fattyacidestersof3-chloro-1,2-propanediol (3-MCPD),2-chloro-1,3-propanediol(2-MCPD),andglycidolareheatinducedcontaminantsthatarenotpresentinvirginunrefinedoils,but theycanbeproducedbythehightemperaturesappliedduringdeodorization [1 3].Thereisevidencethat3-MCPDestersareformedfromironchloride and/ornaturalorganochlorinespresentinnativeoils[4 7].ThepredominantprecursorsandformationpathwaysforMCPDandglycidylesterswill bethoroughlyreviewedinChapter2ofthistext.
Mitigation
ThefactthatMCPDestersbeginformingat200 Cmakesmitigation difficult,asdeodorizationsaregenerallyrunattemperaturesgreaterthan 200 C[4].ManyfactorscontributetotheformationofMCPDandglycidyl esters.Thegrowingconditionsandharvestingofthepalmfruitcanhave profoundeffectsonanoil’scapacitytoformcontaminants.Theextraction, washing,andprocessingstepsthattakeplacepriortodeodorizationcan influencetheformationofthesetoxicantsduringdeodorization,ascanthe specificsofthedeodorizationscheme.ItisalsopossibletoremoveMCPD andglycidylestersusingappropriateadsorbentsorenzymes.Therehave beenanumberofrecentadvancesinmitigationofMCPDandglycidyl esters,includingindustrialscaleproductionofmitigatedpalmoilforusein infantformulaintheUnitedStates[8]. Chapter3 ofthistextdiscussesthe optimizationofallofthesestepstoreduceandeliminatethepresenceof thesecontaminantsinrefinededibleoils.
ProcessingContaminantsinEdibleOils
ISBN978-0-12-820067-4
https://doi.org/10.1016/B978-0-12-820067-4.00007-3
Copyright © 2022AOCSPress. PublishedbyElsevierInc.Allrightsreserved. Publishedincooperationwith AmericanOilChemistsSociety. 1
Analysis
Processededibleoilsarecommonlyconsumedworldwideandusedinthe productionofmanyfoodsincludinginfantformula,highlightingtheneed foraccurateanalyticalmethodologyfortheirdetection.Indirectapproaches requiringesterhydrolysisfollowedbyderivatizationandanalysisbyGC MSwerethe firstmethodsdevelopedtodetectMCPDandglycidylesters [9 11].Whiletheseearlymethodsplayedacrucialroleinbringing attentionfromindustryandregulatorstothepresenceofthesecontaminantsinrefinedoils,theearlyconditionsforbase-catalyzedhydrolyzeswere showntobepotentiallyunreliable,raisingquestionsaboutthetrustworthinessofindirectmethodology[12,13].Morerecently,however,these issueshavebeenaddressedandthequalityandreliabilityoftheseindirect methodshasimprovedgreatly.Severalindirectapproaches(includingthose involvingautomation)haverecentlycompletedsuccessfulcollaborative studies,reachingofficialcompendialmethodstatuswithorganizationssuch astheAmericanOilChemists’ Society(AOCS)andtheInternational OrganizationforStandardization(ISO).Theapplicationoftheseindirect methodstotheanalysisofMCPDandglycidylestersinoilsandhighfat matriceslikedressings/spreadswillbecoveredin Chapter4.
Partlyinresponsetothelackofdependabilityofearlyindirectmethodology,directmethodsweredevelopedforglycidylesters(GEs)and 3-MCPDesters,withcontaminantsanalyzedintactastheyoccurinprocessedoils.Althoughdirecttechniquesrequirealargenumberofstandards (someofwhicharenotcommerciallyavailable)forquantitativeanalysis,this methodologycanprovideinformationabouttheindividualMCPDand glycidylesterspresentinarefinedoil indirectmethods,ontheother hand,providequantitativeinformationregardingthetotalestercontentin anoilexpressedasboundMCPDandglycidol.Detailedinformationabout thestructureoftheindividualesterscanbeusefulinanumberofapplications.Somestudieshaveindicatedpotentialtoxicologicaldifferences betweendifferent3-MCPDesters;thisinformationislostwhenapplying indirectmethodology.Inaddition,asmethodshavebeguntobeextended tocomplexfoods,theuseofadirectapproachcanensureacceptable extractionrecoveriesofallthedifferentesters,whichdohavedistinct polaritydifferencesinsomecases.Informationaboutindividualesterscan alsobeusefulinevaluatingtheeffectivenessofmitigationapproaches.The useofdirectmethodsfortheanalysisofintactestersofglycidol(Chapter5) and3-MCPD(Chapter6)inedibleoilswillbediscussed.
Sincerefinedoilsaretheprimaryfatsourceincommercialinfant formulas,thepresenceofMCPDandglycidylestersisasourceofpotential concernduetoinfants’ lowbodyweightandconsumptionofinfant formulaasthesolesourceofnutritioninsomechildren.Asaresult,research effortshavefocusedonthedevelopmentofanalyticalmethodsforthe analysisofMCPDandglycidylestersininfantformula,aswellasinother complexfoodscommonlyconsumedbychildren. Chapter7 willreview theanalysisofMCPDestersandglycidylestersininfantformulaaswellas inothercomplexfoods.
Toxicology
Freeglycidol,3-MCPD,and2-MCPDallposeconcernsfromafoodsafety perspective.Glycidolisagenotoxiccarcinogenthatisprobablycarcinogenictohumans[14].AccordingtotheFederalInstituteforRiskAssessmentinBerlin,Germany(BfR),concentrationsshouldbekeptaslowasare reasonablyachievableinfood[15].Duetoitsgenotoxicity,theFoodSafety CommissionofJapanhassuggestedglycidolintakeshouldbebasedonthe ALARAprinciple(AsLowAsReasonablyAchievable)[16].Negative effectsonkidneysandreproductivesystemshavebeenseenfrom3-MCPD intoxicologicalstudies[17],anditwasclassifiedbytheEuropeanScientific CommitteeonFoodasanongenotoxicthresholdcarcinogen[18].Free 3-MCPDhasbeenlabeledasagroup2Bcarcinogen(possiblycarcinogenic tohumans)bytheInternationalAgencyforResearchonCancer(IARC) [19].Therearetoxicologicalconcernsshowninlimitedstudiesrelatedto 2-MCPD;oneunpublishedreportshowedthathighdosesaffectedstriated musclesandtheheart,aswellasthekidneysandtheliverinrats[20].
Relativetothefreeformsofthesecontaminants,researchonthefatty acidestersthatareformedindeodorizedoilshasonlybeeninitiatedmore recently[15,20,21].Invivotoxicologicalworkhasdemonstratedthatfree 3-MCPDisliberatedfromthediesterforminrats[22]asisglycidolfrom glycidylesters[23]inrelativelyhighyields.Initialriskassessments conductedbytheBfRhaveconcludedthatusingaworst-casescenario, infantswhoarefedonlycommercialinfantformulascouldpotentially ingestamountsofglycidoland3-MCPDexceedingtheJointFoodand AgricultureOrganization/WorldHeathOrganizationExpertCommittee onFoodAdditives(JECFA)recommendedmaximumtolerabledailyintake levels[21].Exposuresto3-MCPDestersintheUnitedStatesfrom consumptionofinfantformulas,estimatedusingdataon3-MCPDester
concentrationsinformulasamplescollectedbetween2013and2015,are higherthanthoseestimatedforEuropeaninfants.However,estimated glycidylesterexposureswerecomparableforUSandEuropeaninfantsin thattimerange[8].Thefullresultsofalltoxicologicalstudiesonthese contaminantswillbediscussedin Chapter8 (glycidylesters)and Chapter9 (MCPDesters).
Regulations
Inresponsetothedetectionoffree3-MCPDinhydrolyzedvegetable protein,soysauce,andbakedgoods,manyinternationalorganizations addressedtheissueinthosematrices.TheJECFArecommendeda maximumtolerabledailyintakefor3-MCPDof2 m[mu]g/kgbodyweight perday[24].TheEuropeanCommissionestablishedamaximumlevelof 20 m[mu]g/kg(ppb)for3-MCPDinhydrolyzedvegetableproteinandsoy sauce[25],whichwasalsoadoptedbyFoodStandardsAustraliaNew Zealand(FSANZ)[26].TheCodexAlimentariusadoptedamaximumlevel of400 m[mu]g/kg(ppb)inliquidcondimentscontainingacid-hydrolyzed vegetableprotein(excludingnaturallyfermentedsoysauce)in2008[27]. TheUSFoodandDrugAdministrationCompliancePolicyGuidestates thathydrolyzedvegetableproteinthatcontains3-MCPDatlevelsgreater than1 m[mu]g/g(ppm)isnotgenerallyrecognizedassafe(GRAS),and thereforeisanunsafefoodadditive[28].HealthCanadaalsosetamaximum contaminantconcentrationof1 m[mu]g/g(ppm)inAsian-stylesauces[29].
RegulationsregardingMCPDorglycidylesterconcentrationsin processedoilshavebeguntoappearinrecentyears.TheEuropean Commission(EC)publishedaninitialmaximumlevelforglycidylesters (expressedasglycidol)inpowderedinfantformulaof75 mg/kgthatwas reducedto50 mg/kginJuly2019;andamaximumlevelinliquidformula of10 mg/kgthatwasreducedto6 mg/kginJuly2019[30].Inaddition,the maximumlevelforboundglycidolinvegetableoilsandfatsis1000 mg/kg exceptforthoseusedtoproducebabyfoodandprocessedcereal-basedfood forinfantsandyoungchildren,wherethelimitis500 mg/kg[30].TheEC hasproposedlevelsforthesumoffree3-MCPDandbound3-MCPDfor severalproducts,althoughthesehavenotbeen finalized.Theproposed limitsforthesumofboundandfree3-MCPDare1250 mg/kgforoils/fats fromindividualoroilmixesfromcoconut,corn,canola,sunflower, soybean,andpalmkernel;2500 mg/kgforoils/fatsfromindividualormixes fromothervegetableoilsand fishoil,and2500 mg/kgformixturesofoils frombothcategories.Vegetableoilsandfatsdestinedfortheproductionof
babyfoodandprocessedcereal-basedfoodforinfantsandyoungchildren haveaproposedlimitof750 mg/kg.Infantformula,follow-onformula,and foodsforspecialmedicalpurposesintendedforinfantsandyoungchildren haveproposedlimitsof125 mg/kgforpowdersand15 mg/kgforliquids. TheEChasalsodiscussedbringing fish/marineoilsintotheexisting publishedlimitsforboundglycidol,althoughno finaldecisionshavebeen madeatpresstime.
References
[1]HrncirikK,vanDuijnG.Aninitialstudyontheformationof3-MCPDestersduring oilrefining.EurJLipidSciTechnol2011;113:374 9.
[2]MatthäusB,PudelF,FehlingP,VosmannKL,FreudensteinA.Strategiesforthe reductionof3-MCPDestersandrelatedcompoundsinvegetableoils.EurJLipidSci Technol2011;113:380 6.
[3]PudelF,BeneckeP,FehlingP,FreudensteinA,MatthäusB,SchwafA.Onthe necessityofedibleoilrefiningandpossiblesourcesof3-MCPDandglycidylesters.Eur JLipidSciTechnol2011;113:368 73.
[4]DestaillatsF,CraftBD,SandozL,NagyK.Formationmechanismsofmonochloropropanediol(MCPD)fattyaciddiestersinrefinedpalm(Elaeisguineensis)oiland relatedfractions.FoodAdditContamA2012;29:29 37.
[5]DestaillatsF,CraftBD,DuboisML,NagyK.Glycidylestersinrefinedpalm(Elaeis guineensis)oilandrelatedfractions.PartI:formationmechanism.FoodChem 2012;131:1391 8.
[6]NagyK,SandozL,CraftB,DestaillatsF.Mass-defect filteringofisotopesignaturesto revealthesourceofchlorinatedpalmoilcontaminants.FoodAdditContam 2011;28:1492 500.
[7] SmidrkalJ,TesarováM,HrádkováI,BercíkováM,AdamcíkováA,FilipV.Mechanismofformationof3-chloropropan-1,2-diol(3-MCPD)estersunderconditionsof thevegetableoilre fining.FoodChem2016:124 9.
[8]SpungenJH,MacMahonS,LeighJ,FlanneryB,KimG,ChirtelS,SmegalD. EstimatedUSinfantexposuresto3-MCPDandglycidylestersfromconsumptionof infantformula.FoodAdditContamA2018:1085 92.
[9]DivinováV,SvejkovskáB,DolezalM,VelísekJ.Determinationoffreeandbound3chloropropane-1,2-diolbygaschromatographywithmassspectrometricdetection usingdeuterated3-chloropropane-1,2-diolasinternalstandard.CzechJFoodSci 2004;22:182 9.
[10]WeibhaarR.Determinationoftotal3-chloropropane-1,2-diol(3-MCPD)inedible oilsbycleavageofMCPDesterswithsodiummethoxide.EurJLipidSciTechnol 2008;110:183 6.
[11]ZelinkováV,SvejkovskáB,DolezalM,VelísekJ.Fattyacidestersof3-chloropropane1,2-diolinedibleoils.FoodAdditContam2006;23:1290 8.
[12]HainesTD,AdlafKJ,PierceallRM,LeeI,VenkitasubramanianP,CollisonM.Direct determinationofMCPDfattyacidestersandglycidylfattyacidestersinvegetableoils byLC-TOFMS.JAmChemSoc2011;88:1 14.
[13]KazeN,SatoH,YamamotoH,WatanabeY.Bidirectionalconversionbetween3monochloro-1,2-propanediolandglycidolincourseoftheprocedureofDGFstandardmethods.JAmOilChemSoc2011;88:1143 51.
[14]IARC(InternationalAgencyforResearchonCancer).Someindustrialchemicals.In: IARCmonographsontheevaluationofcarcinogenicriskofchemicalstohumans,vol. 77.Lyon,France:InternationalAgencyforResearchonCancer;2000.p.469 86.
[15]BakhiyaN,AbrahamK,GürtlerR,AppelKE,LampenA.Toxicologicalassessmentof 3-chloropropane-1,2-diolandglycidolfattyacidestersinfood.MolNutrFoodRes 2011;55:509 21.
[16] FoodSafetyCommissionofJapan(FSCJ).Considerationsonglycidolanditsfattyacid estersinfoods.RiskAssessRep-NovelFoods&FoodAddit2015.FS/185/2015,Japan. [URL: http://www.fsc.go.jp/english/evaluationreports/others/annex_glycidol_26-52.pdf
[17]ChoWS,HanBS,NamKT,ParkK,ChoiM,KimSH,JeongJ,JangDD.Carcinogenicitystudyof3-monochloropropane-1,2-diolinsprague-dawleyrats.Food ChemToxicol2008;46:3172 7.
[18]EuropeanCommissionHealthandConsumerProtectionDirectorate.Opinionofthe scientificcommitteeonfoodon3-monochloro-propane-1,2-diol(3-MCPD).2001.
[19]IARC(InternationalAgencyforResearchonCancer).IARCmonographsonsome chemicalspresentinindustrialandconsumerproducts.Food&DrinkWkly:3Monochloro-1,2-Propanediol2016:349 74.
[20]SchilterB,ScholzG,SeefelderW.Fattyacidestersofchloropropanolsandrelated compounds:toxicologicalaspects.EurJLipidSciTechnol2011;113:309 13.
[21]BuhrkeT,WeißhaarR,LampenA.Absorptionandmetabolismofthefood contaminant3-chloro-1,2-propanediol(3-MCPD)anditsfattyacidestersbyhuman intestinalcaco-2cells.ArchToxicol2011;85:1201 8.
[22]AbrahamK,AppelKE,Berger-PreissE,ApelE,GerlingS,MielkeH, CreutzenbergO,LampenA.Relativeoralbioavailabilityof3-MCPDfrom3-MCPD fattyacidestersinrats.ArchToxicol2013;87:649 59.
[23]AppelKE,AbrahamK,Berger-PreissE,HansenT,ApelE,SchuchardtS,VogtC, BakhiyaN,CreutzenbergO,LampenA.Relativeoralbioavailabilityofglycidolfrom glycidylfattyacidestersinrats.ArchToxicol2013;87:1649 59.
[24]WHO.Safetyevaluationofcertainfoodadditivesandcontaminants,3-chloro-1,2propanediol.WHOFoodAdditSer2002;48. http://www.inchem.org/documents/ jecfa/jecmono/v48je18.htm
[25]EuropeanCommissionHealthandConsumerProtectionDirectorate.Commission regulation(EC)No1881/2006of19December2006:settingmaximumlevelsfor certaincontaminantsinfoodstuffs.2006.
[26]FSANZ.Chloropropanolsinfood,ananalysisofthepublichealthrisk;technicalreport seriesNo.15;foodstandards,Australia/NewZealand.2003. http://www.foodstandards. gov.au/publications/Documents/TechnicalReportChloropropanolReport11Sep03. doc.
[27]CodexAlimentarius.Codexgeneralstandardforcontaminantsandtoxinsinfoodand feed,codexstan193 1995;amended.2012. www.codexalimentarius.org/download/ standards/17/CXS_193e_2012.pdf
[28]U.S.FoodandDrugAdministration.Guidancelevelsfor3-MCPD(3-chloro-1,2propanediol)inacid-hydrolyzedproteinandasian-stylesauces.CompliancePolicy GuideSection500.500.March14,2008. http://www.fda.gov/ICECI/Compliance Manuals/CompliancePolicyGuidanceManual/ucm074419.htm
[29]HealthCanada.Canadianstandards(maximumlevels)forvariouschemicalcontaminantsinfoods.Modified.June28,2012. http://www.hc-sc.gc.ca/fn-an/securit/chemchim/contaminants-guidelines-directives-eng.php
[30]EuropeanCommission.Commissionregulation(EU)2018/290of26February2018 amendingregulation(EC)No1881/2006asregardsmaximumlevelsofglycidylfatty acidestersinvegetableoilsandfats,infantformula,follow-onformulaandfoodsfor specialmedicalpurposesintendedforinfantsandyoungchildren.2018.
CHAPTER2
Formationmechanisms
BrianD.CraftandFrédéricDestaillats NestléResearchCenter,FoodScienceandTechnologyDepartment,Lausanne,Switzerland
Introduction
Sincethepublicationof[1]andtheheightenedawarenessoffattyestersof monochloropropanediol(MCPD-FE)inrefinededibleoils,thecircumstancessurroundingtheirformationhavebeensubjecttolargeamountsof speculation.Forinstance,someresearchersspeculatedthatprecursorsfor MCPD-FEformation(e.g.,chlorineanddiacylglycerols)arepresentin partiallyrefinedoils[2].Otherresearcherssuspectedthattherefining processresultsintheuncontrolledintroductionofcertaincompoundsto theoils(e.g.,inorganicchloridesinthestrippingstream),soitshouldbethe firstplacetoexploremitigationstrategies[3].Further,amidanalytical developmentsinMCPD-FEquantification,anotherfamilyofcompounds wasdiscoveredinrefinededibleoils,namelythefattyestersofglycidol (G-FE).G-FEwerefoundtobepartiallyresponsibleforinflationofthe resultsofMCPD-FEquantificationsduetothegenerationofartifacts duringsamplepreparationbeforeanalysisusingindirectmethods[4].
Despitetheseearlyhurdles,somerecentbreakthroughsweremadeby Refs.[5 7]ontheformationmechanismsofbothMCPD-FEandG-FE duringpalmoilrefining.Withinthischapterwewilltakeafocusedlook atthestatusoftheliteraturetodateasitpertainstotheformationpathways ofMCPD-FEandG-FEinrefinededibleoils.Criticaltopicswillbe covered,includingthemostprevalentprecursorcompoundsanddetailed formationmechanismsresponsibleforthegenerationoftheseprocess contaminantsduringoilproductionandrefining.BecausebothMCPD-FE andG-FEhavebeenfoundinthehighestaverageabundanceinpalmoil, themajorityoftheresearchreviewedhereininvolvescrudepalmoil productionandrefining.
ProcessingContaminantsinEdibleOils
ISBN978-0-12-820067-4 https://doi.org/10.1016/B978-0-12-820067-4.00004-8
Copyright © 2022AOCSPress. PublishedbyElsevierInc.Allrightsreserved. Publishedincooperationwith AmericanOilChemistsSociety.
MCPDesters
Precursors
Chlorineisubiquitousinnature.Thus,onecanspeculateaboutawide varietyofchlorinesources,whetherorganicorinorganic,aspotential precursorcompoundstoMCPD-FEformationduringedibleoilproduction.Further,ahostoflipidtypesandcompositions(e.g.,acylglycerols, phospholipids,glycolipids)areavailableintherawmaterialsusedtoproduceedibleoils.Manyoftheselipidscouldtheoreticallyinteractwith chlorinesourcesandresultintheformationofMCPD-FEduringoil refining.Thecriticalprecursorsresponsible,however,aremostlydependent ontheoiltype,quality,and,toalesserdegree,thecircumstances ofmanufacture,aswillbedescribedbelow.Giventhatrefinedpalmoil specificallyhasbeenshowntocontainsignificantlevelsofMCPD-FE (2.7mg/kg)[8],ithasbeenexclusivelyusedasamodelmatrixinthe literature.
The firstquestionoftenraisedregardingMCPD-FEprecursorsisthe originofchlorineinvolvedintheMCPD-FEreactionduringoilrefining andwhyitispotentiallymoreabundantincrudepalmoil(CPO)in comparisontoothercrudevegetableoils[9].Recently[5],demonstrated thatmanysourcesofcovalentlyboundinorganicchlorineexistatppm (mg/kg)levelsincrudepalmoil,includingFeCl3,FeCl2,MgCl2,and CaCl2.Further,a “pool” (n ¼ 300)oforganicmonochlorinatedcompoundswasalsofoundanditappearstoundergoatransformation throughoutthestagesofoilrefiningwithcertaincompoundsbeingformed whileothersdecomposeovertime.Inordertoelucidatethecompositionof themorepredominantchlorine “donor” compounds[5],usedLC-MSn in theframeworkofmodelexperiments.Thereintheauthorsidentifieda specificfamilyofchlorinatedcompoundspresentinboththelipids extractedfromhand-pickedMalaysianpalmfruitsandcommerciallyprocuredCPOsamples. Fig.2.1,takenfromRef.[5];showstheproposed structureandchemicalformulasofthismonochlorinatedfamilyofcompounds.Theauthorssuggestthatgiventheirstructuralsimilaritiesto phytosphingosines,itisperhapsmorelikelythatthechlorinedonors identifiedareendogenousplantmetabolitesasopposedtochlorinated contaminantsintroducedtotheoilpalm’sdirectenvironmentduring growthandmaturation.Extrapolatingfromthishypothesis,onemighttend tothelogicthattherawmaterialsintendedforproductionofeachedible vegetableoilhavetheirownreactive-chlorinepoolcapableofdonating chlorineduringoilrefiningandultimatelyresultinginMCPD-FE generation.
C42H83O
4NCl700.60280
C42H85O4NCl702.61807
C42H83O5NCl716.59723
C42H85O5NCl718.61357
C42H85O6NCl734.60809
Figure2.1 Proposedstructureandchemicalformulasofanorganochlorinefamilyof compoundsfoundincrudepalmoil. (ReprintedwithpermissionfromNagyK,SandozL, CraftBD,DestaillatsF.Mass-defect filteringofisotopesignaturestorevealthesourceof chlorinatedpalmoilcontaminants.FoodAdditContam2011;28:1492 1500).
IntermsofthemostpredominantlipidprecursorsofMCPD-FEin edibleoils[1],proposedthattheremaybealinkbetweenthecontentof diacylglycerols(DAGs)inrefinededibleoilsandtheirMCPD-FElevels. ThisassertionwaslikelyduetothefactthatthehighestMCPD-FElevels wereobservedwithinthefruitpulpoilsanalyzed.Fruitpulpoils,suchas oliveandpalm,areknownforhavinghighDAGcontentscomparedto seedoils,duetothegreaterprevalenceoflipolyticreactionsduringharvest [10].Thiscorrelation,however,hasbeendisprovedinrecentliterature [11,12].AlthoughDAGscouldpotentiallyreactwithchlorinedonors duringoilrefiningandresultintheformationofMCPD-FE,theyarenot themostcriticallipidprecursorsoftheseprocesscontaminants.Further, lipidssuchasmonoacylglycerols(MAGs),phospholipids,andglycolipidsare largelyremovedduringoildegummingandarenotpresentduringthelater stagesofoilrefining[10].
BecausethebulkofMCPD-FEhavebeenshowntobegenerated duringoildeodorization[2,13],theentiretyoftheaforementionedlipid classesisnotexpectedtobegreatlyinvolvedinMCPD-FEformation reactions.Thisofcourseleavesthetriacylglycerols(TAGs)upforconsideration.TAGscanrepresentmorethan90% 95%(v/v)ofrefinedvegetableoils,whetherpressedfromnuts,seeds,orfruitpulps.TAGsare, therefore,themostlogicalcriticallipidprecursoravailableforMCPD-FE formationduringoildeodorization.TheresultsofinvitrothermalreactionexperimentscarriedoutbyRef.[7]appeartoconfirmthishypothesis [7].demonstratedincontrolledconditionsthatTAGs,notDAGs,are preferentiallyreactingwithchlorinedonorstoformMCPD-FE.
Formationpathways
Aspreviouslymentioned,MCPD-FEareformedalmostcompletelyduring thedeodorizationunitoperationofedibleoilrefining.Assuch,themajorityofscientificresearchconductedonMCPD-FEformationpathways hasbeencarriedouteitherinconditionsmimickingoildeodorizationor withinbench-top,pilotscale,orcommercialdeodorizationunits[7]. showedthroughinvitroexperimentsthat3-MCPDdiesters,whicharethe mostpredominantformofMCPD-FEinrefinedoils[14],canbegenerated attemperaturesaslowas180 200 C.Itfollowsfromthisobservationthat withineithertypeofedibleoilrefining(chemicalorphysical),thetypical deodorizationconditionsemployedstronglyfavorMCPD-FEformation. Forexample,typicalchemicalrefiningoperationsforpalmoilinvolvea deodorizationstepataround240 C,whereasphysicalrefiningoperations caninvolveadeodorizationstepatevenhighertemperatures(260 270 C) inordertoremoveexcessfreefattyacids(FFAs)[10].
InordertodeterminetheoriginofchlorineinvolvedinMCPD-FE formationduringoilrefining[2,11],examinedthecontentofchloride ionspresentinoilspre-andpost-deodorization.Theythenattemptedto correlatetheselevelswiththeultimatelevelsofMCPD-FEobservedinthe fullyrefinedoils.Unfortunately,littleornocorrelationwasobserved[3]. attemptedtodetermineifthechlorineresponsibleforMCPD-FEformation wasoriginatingfromthestrippingsteamappliedduringoildeodorization, butwithasimilarnegativeresult.Onlyrecently[5],demonstratedthatboth inorganicandorganicchlorinatedcompoundsarepresentatppm(mg/kg) levelsinpartiallyrefinededibleoils.Further,theauthorsdemonstratedthat thethermaldecompositionoforganicchloride-containingcompoundsin CPOwasfoundtocoincidestronglywiththeevolutionof3-MCPD diestersduringthethermaltreatmentofCPO(see Fig.2.2).Giventhat 3-MCPDdiestersarethepredominantclassofMCPD-FEinrefinedoils, thestudyof[5]hasprovencausalitybehindthegreatestportionof MCPD-FEformedduringthedeodorizationofedibleoils.
Agivenorganic(orinorganic)chlorinatedcompoundmaydecompose atacertaintemperature,abovewhichthereleasedreactivechlorinecan theninteractwithTAGandresultintheformationofMCPD-FE.Thefact thatthegraphicin Fig.2.2 crossesat180 C,whereastheformationtemperatureofMCPD-FEinthestandardizedinvitroexperimentsof[7]was 180 200 C,isquiteacoincidence.Inordertouncoverwhetheracertain reactivechlorineintermediatewasmostresponsibleforMCPD-FE
Sumoforganochlorines SumofMCPDs
Figure2.2 Thesimultaneousdecompositionofsomekeyorganochorines(n ¼ 8) monitoredandtheformationof3-MCPDdiestersduringthethermaltreatmentof crudepalmoil. (ReprintedwithpermissionfromNagyK,SandozL,CraftBD,DestaillatsF. Mass-defect filteringofisotopesignaturestorevealthesourceofchlorinatedpalmoil contaminants.FoodAdditContam2011;28:1492 1500).
formation[5],monitoredthedecompositionoftheorganochlorinesfound incrudepalmoilsviaLC-MSn experiments.Theauthorsreportedthat hydrogenchloride(HCl)isatypicalthermaldecompositionproductofthe organochlorinepoolmonitored.Thus,HClcouldprovetobethepredominantformofreactivechlorineresponsibleforMCPD-FEformation duringoildeodorization.
SeveralpotentialformationmechanismsofMCPD-FEhavebeen recentlyreviewedintheliterature[15,16].Giventhatmanyofthepast mechanisticstudiesofMCPD-FEformationwerecarriedoutinhydrophilicmedia,ithaslongbeensuggestedthatTAGunderwenthydrolysisto DAGasa firststepintheMCPD-FEformationreaction.DAGsthen reactedwithchlorinedonorcompoundsresultingintheformationof acyloxoniumionintermediatesandeventualnucleophilicsubstitutionof chlorideionontheglycerolbackbone[17].Morerecently,however,itwas proventhatTAGscanactdirectlyasasubstrateforMCPD-FEformation [7].Further[18],provedwithinfrared(IR)spectroscopythatheatingTAG inthepresenceofLewisacidscanleadtocyclicacyloxoniumionformation.Thus,isittheoreticallyplausibletoassumethatthetwopathways couldbefavoredinthecaseofhydrophobicsystemslikeoildeodorization.
OnepathwayinvolvesthereactionofTAGdirectlywithHClformedby thermaldegradationofchlorinedonors,nucleophilicsubstitutionofchlorideionontheglycerolbackbonetoformanMCPD-FE,and finallythe releaseofafattyacid.Thesecondmechanism firstinvolvestheformationof anacyloxoniumionintermediatecompound.ThesetwoMCPD-FEformationmechanismsweresummarizedbyRef.[7]andappearin Fig.2.3 [7]. alsoprovedthroughinvitroexperimentsthattheMCPD-FEformation reactionisregioselectiveandpreferentialtothesn-1(3)positionsonthe glycerolbackbone.
Becausethecriticalprecursorsandpredominantformationmechanisms havebeenelucidatedinthecaseofpalmoilproductionandrefining,itwas thenpossibletospeculateaboutthepotentialpredominantrootcauses responsibleforthemanifestationofMCPD-FEtherein[5].demonstrated thatchlorinatedcompounds(n ¼ 300found)canbemonitoredincrude palmoilsutilizingmass-defect filteringofisotopesignatures.Further,the authorsthendiscoveredthatthesesamecompoundscanbeutilizedto segregatecommercialpalmoilsamples(n ¼ 26)basedontheirprocessing stageusingmultivariatestatisticalanalysis(see Fig.2.4).Thegroupingof commercialsamplesfromcrudetorefined-bleachedtorefined-bleacheddeodorized,asshownin Fig.2.4,suggeststhatchlorinatedcompounds undergoatransformationthroughoutpalmoilproductionandrefining. Thistruthmightlendtothelogicthatchlorineissimilarlytransformed throughouttheagriculturalprocessinvolvedinpalmoilgrowth,maturation,andharvest. Fig.2.5 isaschematictakenfromRef.[19]andservesasa root-causeanalysisofMCPD-FEformationduringrefinedpalmoilproduction.Itsummarizespotentiallocations/sourcesfortheinfluxofchlorine fromtheenvironment,accumulationofthechlorineinthepalmplantand fruits,andtransformationofchlorineintomoreliposolubleformsduring CPOproduction,followedbytheresultantformationofMCPD-FE duringoilrefining.Asimilartypeofroot-causeanalysiscouldprove beneficialinthecaseofotherrefinedvegetableoilcropsandtheassessment oftheirpotentialforproductionofMCPD-FEafterharvestandrefining.
Glycidylesters
Precursors
GiventhehistorybehindthediscoveryofG-FEinedibleoils,researchers oftenpooledG-FEwithMCPD-FE(oftentermedMCPDestersand relatedcompounds)[3].Thisfactledtotheassumptionthatthese
Figure2.3 ProposedmechanismsfortheformationofMCPD-FEfromTAGathightemperaturesinthepresenceofHClevolvedfromthe thermaldecompositionoftraceorganochlorines.Twoputativepathwaysincludingtheformationofreactivecycloacyloxoniumionintermediate(upperpanel)oradirectnucleophilicsubstitutionreaction(lowerpanel)aredisplayed.Bothpathwaysresultintheformationof anMCPD-FEmoleculeandthereleaseofafattyacid. (ReprintedwithpermissionfromDestaillatsF,CraftBD,SandozL,NagyK.Formation mechanismofmonochloropropanediol(MCPD)fattyaciddiestersinrefinedpalm(Elaeisguineensis)oilandrelatedfractions.FoodAdditContam 2012;29:29 37).
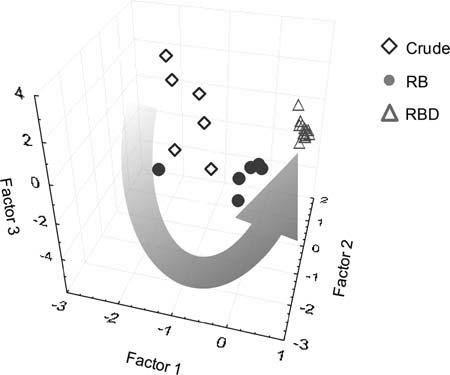
Figure2.4 Principalcomponentanalysisofchlorine-containingcompounds(n ¼ 300) presentincrude,partially,andfullyrefinedpalmoilsamples(n ¼ 26).Thegroupingof oilsamplesbasedonrefiningstage(i.e.,fromcrudetorefined-bleached[RB],to refined-bleached-deodorized[RBD])suggeststhatthesecompoundsundergoa transformationduringoilprocessing. (ReprintedwithpermissionfromNagyK,SandozL, CraftBD,DestaillatsF.Mass-defect filteringofisotopesignaturestorevealthesourceof chlorinatedpalmoilcontaminants.FoodAdditContam2011;28:1492 1500).
compoundfamilieswereverycloselyrelatedandpotentiallysubjectto interconversion.Assuch,bothMCPD-FEandG-FEwerethoughttoshare thesameprecursors.Thisassumptionhasbeendisprovedrecently[20]. reportedastrongpositivecorrelationbetweenDAGlevelsaloneandthe amountofG-FEcontainedinrefinedpalmoils.Becausefruitpulpoilslike palmoilnaturallycontainhigherlevelsofDAG( 3% 4%)[21],thismay suggestpossiblecausalityastowhytheirdeodorizedcounterpartsalso containhigherlevelsofG-FEincomparisontoseedoilcrops,as demonstratedbyRef.[4].
ThestrongcorrelationbetweenDAGcontentsandG-FEformationhas essentiallybeenvalidatedonanindustrialscale[22].recentlyreportedthat high-DAGoilsmarketedforhealthandwellnesshadtobetakenoffthe marketpurportedlydueto “highlevels” ofG-FE.Further[23],demonstratedthatcommerciallyrefinedoilsrichinDAG(87%)cancontainmore than10-foldgreaterG-FElevelsrelativetooilswithlowerDAG (3.9% 6.8%)contents.TheonlyotherprovenlipidprecursortoG-FEis MAG[6].showedthatbothMAGandDAGcanresultinformationofGFEuponthermaltreatment,althoughformationfromDAGismost
Oil Palm Growth & Maturation
Biosynthesis of hydrophilic organochlorines in palm fruits
Palm Oil Production
Formation of lipophilic organochlorines during fruit bunch sterilization
Palm Oil Refining
Reaction of liposoluble organochlorines with palm oil triacylglycerols at high temperatures
Accumulation of inorganic chloride in the oil palm plant
Influx of inorganic chloride from the environment (e.g., KCl, NH4Cl, MgCl2, FeCl3, FeCl2) (5)
MCPD diester formation
Soil, Fertilizer, & Irrigation
Figure2.5 Root-causeanalysisofthefactorsinvolvedintheformationofMCPD-FE duringrefinedpalmoilproduction,including(1)chlorineinfluxfromtheenvironment,(2)accumulationofinorganicchlorideintheplant,(3)bioconversionofinorganicchloridestoorganochlorinesinpalmfruits,(4)formationofliposoluble organochlorinesduringfruitbunchsterilization,and(5)reactionofliposolubleorganochlorineswithTAGinpalmoilduringoildeodorization. (Reprintedwithpermission fromCraftBD,NagyK,SandozL,DestaillatsF.Factorsimpactingtheformationof monochloropropanediol(MCPD)fattyaciddiestersduringpalm(Elaeisguineensis)oil production.FoodAdditContam2012;29:354 361).
favored.ThefactthatMAGlevelsinrefined-bleachedoilsareoftenquite low(mean < 0.1%)[24],however,rendersthisreactionroutelesssignificantinthecaseofediblevegetableoils.
Formationpathways
TheformationofG-FEfromDAGduringoildeodorizationwasshownto besignificantattemperaturesatorabove230 C[11].Ref.[20]confirmed thistemperatureofG-FEformationbydeodorizingrefined-bleachedpalm oilfrom180to240 Catincrementsof20 C.G-FEvalueswerereportedas thesumoftheglycidyl-palmitate(16:0G-FE),stearate(18:0G-FE),oleate (18:1G-FE),linoleate(18:2G-FE),andlinolenate(18:3G-FE)species














