https://ebookmass.com/product/medical-device-regulations-a-
Instant digital products (PDF, ePub, MOBI) ready for you
Download now and discover formats that fit your needs...
Advances in Nanosensors for Biological and Environmental Analysis Aakash Deep
https://ebookmass.com/product/advances-in-nanosensors-for-biologicaland-environmental-analysis-aakash-deep/
ebookmass.com
Diagnosis: A Guide for Medical Trainees Graham Kennedy
https://ebookmass.com/product/diagnosis-a-guide-for-medical-traineesgraham-kennedy/
ebookmass.com
Breastfeeding: A Guide for the Medical Profession Ruth A. Lawrence
https://ebookmass.com/product/breastfeeding-a-guide-for-the-medicalprofession-ruth-a-lawrence/
ebookmass.com
All the Little Hopes Leah Weiss
https://ebookmass.com/product/all-the-little-hopes-leah-weiss-2/
ebookmass.com
Unforeseen https://ebookmass.com/product/unforeseen-love-love-in-lockdownchronicles-book-1-l-s-pullen/
ebookmass.com
Atomically Precise Nanochemistry 1st Edition Rongchao Jin
https://ebookmass.com/product/atomically-precise-nanochemistry-1stedition-rongchao-jin/
ebookmass.com
Interwar London after Dark in British Popular Culture Mara Arts
https://ebookmass.com/product/interwar-london-after-dark-in-britishpopular-culture-mara-arts/
ebookmass.com
Content Inc., Second Edition : Start a Content-First Business, Build a Massive Audience and Become Radically Successful (With Little to No Money) Joe Pulizzi
https://ebookmass.com/product/content-inc-second-edition-start-acontent-first-business-build-a-massive-audience-and-become-radicallysuccessful-with-little-to-no-money-joe-pulizzi/
ebookmass.com
Dermatology Secrets 6th Edition Whitney A. High
https://ebookmass.com/product/dermatology-secrets-6th-edition-whitneya-high/
ebookmass.com
Doing Social Research and Publishing Results: A Guide to Non-native English Speakers Candauda Arachchige Saliya
https://ebookmass.com/product/doing-social-research-and-publishingresults-a-guide-to-non-native-english-speakers-candauda-arachchigesaliya/
ebookmass.com
MedicalDeviceRegulations AAKASHDEEP DepartmentofPharmaceuticalSciences,ChaudharyBansiLal University,Bhiwani,Haryana,India
AcademicPressisanimprintofElsevier
125LondonWall,LondonEC2Y5AS,UnitedKingdom
525BStreet,Suite1650,SanDiego,CA92101,UnitedStates
50HampshireStreet,5thFloor,Cambridge,MA02139,UnitedStates
TheBoulevard,LangfordLane,Kidlington,OxfordOX51GB,UnitedKingdom
Copyright©2022ElsevierInc.Allrightsreserved.
Nopartofthispublicationmaybereproducedortransmittedinanyformorbyanymeans, electronicormechanical,includingphotocopying,recording,oranyinformationstorageand retrievalsystem,withoutpermissioninwritingfromthepublisher.Detailsonhowtoseek permission,furtherinformationaboutthePublisher’spermissionspoliciesandourarrangements withorganizationssuchastheCopyrightClearanceCenterandtheCopyrightLicensingAgency, canbefoundatourwebsite: www.elsevier.com/permissions .
Thisbookandtheindividualcontributionscontainedinitareprotectedundercopyrightbythe Publisher(otherthanasmaybenotedherein).
Notices
Knowledgeandbestpracticeinthisfieldareconstantlychanging.Asnewresearchand experiencebroadenourunderstanding,changesinresearchmethods,professionalpractices,or medicaltreatmentmaybecomenecessary.
Practitionersandresearchersmustalwaysrelyontheirownexperienceandknowledgein evaluatingandusinganyinformation,methods,compounds,orexperimentsdescribedherein.In usingsuchinformationormethodstheyshouldbemindfuloftheirownsafetyandthesafetyof others,includingpartiesforwhomtheyhaveaprofessionalresponsibility.
Tothefullestextentofthelaw,neitherthePublishernortheauthors,contributors,oreditors, assumeanyliabilityforanyinjuryand/ordamagetopersonsorpropertyasamatterofproducts liability,negligenceorotherwise,orfromanyuseoroperationofanymethods,products, instructions,orideascontainedinthematerialherein.
BritishLibraryCataloguing-in-PublicationData
AcataloguerecordforthisbookisavailablefromtheBritishLibrary
LibraryofCongressCataloging-in-PublicationData
AcatalogrecordforthisbookisavailablefromtheLibraryofCongress
ISBN:978-0-323-91126-9
ForInformationonallAcademicPresspublications visitourwebsiteat https://www.elsevier.com/books-and-journals
Publisher: MaraConner
AcquisitionsEditor:CarrieBolger
EditorialProjectManager:RafaelG.Trombaco
ProductionProjectManager:SojanP.Pazhayattil
CoverDesigner: ChristianJ.Bilbow
TypesetbyMPSLimited,Chennai,India
Preface xiii
1.Introductiontomedicaldevices1
1.1 Introduction 1
1.2 Classificationsofmedicaldevicesonthebasisofrisk1
1.2.1 Medicaldevicesafetyandperformance1
1.3 Thebasicdesignandmanufacturingprincipleslistedinthissectionapplyto IVDandmedicaldevices2
1.3.1 Commonprinciples2
1.3.2 Clinicalinvestigation3
1.3.3 Physicochemicalandbiologicalproperties3
1.4 Historyofmedicaldeviceregulationsglobally3
1.5 Productlifecycleofmedicaldevice5
1.6 Thefivestagesofthemedicinalproductlifecycle5
1.6.1 Investigation5
1.6.2 Design5
1.6.3 Validation6
1.6.4 Launch6
1.6.5 Postmarketreview6
1.7 InternationalMedicalDeviceRegulatorsForum7
1.8 IMDRFManagementCommittee7
1.8.1 PurposeofIMDRF7
1.8.2 OrganizationalconvergenceIMDRF7
1.9 GlobalHarmonizationTaskForce(GHTF)8
1.9.1 Safetymanagement8
1.10 SummaryTechnicalDocumentation8
1.11 Globalmedicaldevicenomenclature10
1.12 Conclusion 10 References 10
2.Ethicsofclinicaltrialsofmedicaldevices13
2.1 Introduction13
2.1.1 Clinicalinvestigationofmedicaldevices13
2.2 Clinicalinvestigationalplanformedicaldevices13
2.3 Clinicalinvestigationconduct13
2.3.1 General13
2.3.2 Investigationsiteinitiation14
2.3.3 Investigationsitemonitoring14
2.3.4 Adverseeventsanddevicedeficiencies14
2.3.5 Clinicalinvestigationdocumentsanddocumentation14
2.3.6 Additionalmembersoftheinvestigationsiteteam14
2.3.7 Subjectprivacyandconfidentialityofdata14
2.3.8 Documentanddatacontrol15
2.3.9 Investigationaldeviceaccountability15
2.3.10 Accountingforsubjects15
2.3.11 Auditing15
2.4 ISO14155:201115
2.5 InternationalCouncilonHarmonizationofGoodClinicalPractice16
2.6 InternationalCouncilonHarmonizationofGoodClinicalPracticeprinciples16
2.7 ISO13485:2016:qualitymanagementsystemofmedicaldevicesrequirements forregulatorypurposes17
2.8 Generalrequirementsofqualitymanagementsystem18
2.9 ISO14971:2019medicaldeviceriskmanagementapplications18
2.10 Riskmanagementapplicationthroughoutthelifecycleofthedevice19
2.10.1 Planning19
2.10.2 Documentation19
2.10.3 Riskanalysis/evaluation19
2.10.4 Riskcontrol20
2.10.5 Productionandpostproductioninformation20
2.10.6 Riskmanagementprocess20
2.11 Conclusion 21 References 21
3.RegulationsformedicaldevicesintheUnitedStates23
3.1 Introduction23
3.2 USFood&DrugAdministration23
3.3 Classificationofmedicaldevices(I,II,andIII)23
3.3.1 ClassIdevices23
3.3.2 ClassIIdevices24
3.3.3 ClassIIIdevices24
3.4 Regulatoryapprovalprocessformedicaldevice24
3.5 Premarketnotification510(k) 21CFRPart807E25
3.6 Premarketapproval25
3.7 ApprovalprocessofmedicaldevicesintheUSA:itfollowssomesteps27
3.8 Investigationaldeviceexemption28
3.9 Qualitysystemrequirements21CFRpart82028
3.10 Labelingrequirements21CFRPart80129
3.11 Postmarketingsurveillanceofmedicaldevice30
3.12 Uniquedeviceidentificationofmedicaldevice30
3.13 Conclusion 31 References 31
4.RegulationsofmedicaldevicesinEurope33
4.1 Introduction33
4.2 Classificationofmedicaldevices33
4.3 Medicaldevices’ lawsinEurope34
4.4 Thenewapproachforregulatingproducts keyfeatures35
4.5 Regulatoryapprovalprocessofmedicaldevice35
4.6 NotifiedbodiesinEurope37
4.7 CEMarkinginEuropeformedicaldevices37
4.8 Procedure 37
4.9 Documentsrequiredformedicaldevicemarketingapproval37
4.10 Labelingrequirements38
4.11 Medicaldevicelabeling:EURegulationMDR2017/74538
4.12 ProductlabelingandQMS EUMDR38
4.13 Conclusion 39 References 39
5.RegulationsofmedicaldevicesinASEANcountries41
5.1 Introduction 41
5.2 MembersoftheAssociationofSoutheastAsianNations41
5.3 ASEANCommonSubmissionDossierTemplate42
5.4 ProductregistrationonthebasisofCSDTASEAN42
5.5 Generalcriteriatoregisteramedicaldevice42
5.6 ASEANCSDTformat43
5.6.1 Summary43
5.6.2 Elementsofthecommonsubmissiondossiertemplate43
5.7 RegistrationprocessinASEANcountries44
5.7.1 Thailand44
5.7.2 Singapore45
5.7.3 Malaysia45
5.7.4 Indonesia47
5.7.5 Vietnam48
5.7.6 BruneiDarussalam49
5.7.7 Laos 50
5.7.8 Cambodia50
5.7.9 Myanmar51
5.7.10 Philippines53
5.8 Conclusion 53 References 54
6.RegulationsofmedicaldevicesinJapanandChina57
6.1 IntroductiononmedicaldeviceinJapan57
6.2 Regulatoryauthorities57
6.2.1 Japan’sagencyforpharmaceuticalproductsandmedicaldevices57
6.2.2 Japan’sMinistryofHealth,Labor,andWelfare58
6.3 Medicaldeviceclassification58
6.4 RegulatoryapprovalprocessinJapan58
6.4.1 MarketingauthorizingholderinJapan58
6.4.2 Reviewprocedure59
6.4.3 Approvalprocess:processflowchartshowingtheapprovalprocess59
6.5 IntroductiontomedicaldeviceinChina62
6.6 Regulatoryauthorities62
6.7 Classification62
6.8 Registrationprocedure63
6.9 Labelingrequirements63
6.10 Challenges 64
6.11 Conclusion 64 References 65
7.RegulationsofmedicaldevicesinCanada67
7.1 Introduction67
7.2 Definitionofmedicaldevice67
7.3 ActionplanformedicaldevicesfromtheCanadianHealthMinistry67
7.4 Classificationofmedicaldevices68
7.5 MedicaldevicepremarketrequirementsinCanada69
7.6 CanadianMedicalDevicesConformityAssessmentSystem69
7.7 Applicationformforregistrationofmedicaldevice69
7.8 Registrationrequirements74
7.9 Note 74
7.10 Registrationprocedure75
7.11 MedicalDeviceLicense76
7.12 LabelingrequirementsofmedicaldeviceinCanada(Section21)77
7.13 Timeframe:theapprovalprocessvariesbydeviceclass78
7.14 Specialrequirements79
7.15 Localfees 79
7.16 Licensevalidity79
7.17 Licensetransfer79
7.18 Authorizedrepresentative/licenseholder79
7.19 Conclusion 79 References 80
8.RegulationsformedicaldevicesinIndia83
8.1 Introduction83
8.2 ClassificationofmedicaldeviceinIndia84
8.3 Proposedclassificationsystemformedicaldevices84
8.4 RegulationsinIndia85
8.5 Marketofmedicaldevices85
8.6 CentralDrugsStandardControlOrganization(CDSCO)85
8.7 MedicaldevicedefinitionasperCDSCO86
8.8 Medicaldeviceregistrationprocess87
8.9 Thedocumentsneededforregistration87
8.10 ApprovalprocessofmedicaldeviceinIndia88
8.11 Manufactureofmedicaldevicesforsaleorfordistribution88
8.11.1 ClassAandB88
8.11.2 ClassCandD89
8.12 Importofmedicaldevices90
8.13 Inspectionofforeignmanufacturingfacilities90
8.14 AproposaltoregulatemedicaldevicesinIndia90
8.15 Newproposedregulationsforclinicaltrials92
8.16 ReportingofmedicaldevicesinIndia93
8.17 Labelingofmedicaldevices94
8.18 Recallofmedicaldevices94 8.19
9.RegulationsofmedicaldevicesinAustralia97 9.1 Introduction97
9.2 AnoverviewofAustralia’smedicaldevicesregulatoryauthority97
9.3 DefinitionofmedicaldeviceasperTGA97
9.4 Classificationofmedicaldevice98
9.5 Lifecycleapproachtoregulation98
9.6 Postmarketingregulations99
9.7 MedicaldeviceregistrationinAustralia99
9.8 MedicaldeviceregulatorysysteminAustralia100
9.9 EssentialprincipleofmedicaldevicesinAustralia100
9.10 DeclarationofConformity101
9.11 OverviewofmedicaldeviceregulationsinAustralia101
9.12 RegulatoryframeworkofmedicaldeviceinAustralia101
9.13 ConformityassessmentandARTGinclusion103
9.14 TemplatesDeclarationofConformity(medicaldevices)103
9.15 Medicaldeviceinclusionprocess106
9.16 Postmarketsurveillance107
9.17 FeesofmedicaldevicesinAustralia107
9.18 Conclusion111 References 112
10.RegulationsofmedicaldevicesinGulfCooperationCouncilcountries113
10.1 Introduction:GulfCooperationCouncilcountries113
10.2 SaudiArabia113
10.3 SaudiFoodandDrugAuthority114
10.4 MedicaldevicedefinitionasperSFDA115
10.5 ClassificationofmedicaldevicesinSaudiArabia115
10.6 MedicaldeviceregulationsinSaudiArabia116
10.7 Requireddocuments116
10.8 Medicaldevicetechnicaldocumentation117
10.9 Essentialprinciplesofmedicaldevices119
10.10 SupplyingmedicaldevicestotheKSAmarket119
10.11 Registrationandlistingrequirements120
10.12 Medicaldevicesmarketingauthorization121
10.13 TheapprovalprocessformedicaldevicesinSaudiArabia122
10.14 Timeframe123
10.15 Specialrequirements123
10.16 Localfees123
10.17 Licensevalidity123
10.18 Licensetransfer123
10.19 Authorizedrepresentative/licenseholder124
10.20 Labelingrequirementsofmedicaldevices124
10.21 Postmarketingsurveillanceofmedicaldevice124
10.22 Kuwait 124
10.23 Classificationofmedicaldevice126
10.24 Documentsrequiredforregistration126
10.25 RegistrationandregulationsofmedicaldevicesinKuwait126
10.26 Timeframe127
10.27 Specialrequirements127
10.28 Localfees127
10.29 Licensevalidity127
10.30 Licensetransfer127
10.31 Authorizedrepresentative/licenseholder127
10.32 Bahrain 128
10.33 Classificationofmedicaldevice128
10.34 Authorizedrepresentativeregistration128
10.35 Registrationofmedicaldevices129
10.36 Validity 130
Classificationofmedicaldevices131
Documentsrequiredforregistrationofmedicaldevice131
MedicaldeviceregulationsinOman133
Timeperiod133
Licensevalidity133
Authorizedrepresentative133 10.48 UnitedArabEmirates133
Documentsrequiredfortheregistration:tworegistrationsrequired135
Authorizedrepresentative138
11.1 Introduction141
11.2 ScopeandresponsibilitiesoftheNMRAfortheapprovalofmedicaldevices141
11.3 MedicaldevicedefinitionasperNMRA142
11.4 MedicalDeviceEvaluationCommittee142
11.5 Medicaldeviceclassificationsystem142
11.6 Registrationprocessofmedicaldevice142
11.6.1 MAHorapplicantresponsibilities143
11.6.2 Samplelicenserequest143
11.6.3 Applicationsubmission143
11.6.4 Multipleapplications147
11.6.5 Filesubmissionprocedure148
11.6.6 Specialneeds148
11.6.7 Technicaldocuments149
11.6.8 Evaluationprocessing149
11.6.9 Extensionofthevalidityperiodofregistrationcertificateandlicense150
11.6.10 Applicationforimportlicense150
11.7 Conclusion151 References 151
12.RegulationsofmedicaldevicesinRussia153
12.1 Introduction153
12.2 Medicaldevicedefinition154
12.3 Classificationofmedicaldevices154
12.4 Documentsrequired155
12.5 Russiamedicaldeviceregistrationrequirements156
12.6 Labelingrequirements156
12.7 Documentsrequiredforforeignmanufacturer157
12.8 ApprovalofmedicaldevicesinRussia158
12.9 RegulationsofmedicaldevicesinRussia159
12.10 RenewalofcertainnationalstandardsrelatedtomedicaldevicesinRussia161
12.11 Stateregistrationofmedicaldevices161
12.12 Conclusion162
Preface TheideaofwritingthisbookwasconceivedwhenmyownstudentsfoundgreatdifficultyingettingtherecentliteratureregardingtheRegulationofMedicalDevices. Amajoraimofthisbookwastoproviderecentliteratureregardingthecurrent RegulationofMedicalDevices.Thecurrentbookisacompilationofabriefreview ofvariousregulatorybodiesofmajordevelopedanddevelopingcountriesaroundthe worldandtheRegistrationProcedureofMedicalDeviceofsuchPharmaceutical RegulatoryOrganizationsindeliveryofsafeandeffectivehealthcareproducts.
IthanktheworthyViceChancellorofChaudharyBansiLalUniversity,Bhiwani, Prof.RajKumarMittal,forhisconstantmotivationandsupport.IthankDr.Jitender KumarBhardwaj,Registrar,ChaudharyBansiLalUniversity,Bhiwani,forthemotivationandguidance.
ItismyproudprivilegetoexpressmysinceregratitudetoProf.NarasimhanB., DepartmentofPharmaceuticalSciences,MaharshiDayanandUniversity,Rohtak,for hisconstantunceasingencouragementtocombinemymentalimageswithemotionof desiretoacceleratetheirrealization.
IgratefullyacknowledgemyChairmanProf.NitinBansal,Departmentof PharmaceutcialSciences,ChaudharyBansiLalUniversity,Bhiwani,Dr.Prabodh ChanderSharma,DelhiPharmaceuticalSciencesandResearchUniversity,New Delhi,andProf.D.N.Mishrafortheirblessings.
Ifeeltothanktomyparents,Sh.RajKaranandSmt.KamlaDeviandmybetter halfPriyanka,sistersSharda,Sangeeta,andMeenakshi,andmykidsHargunandSeerat whohavehelpedmeinbringingoutthisbook.
Ihumblyandgenerouslycalluponallmycolleaguestogivedueconsiderationsto thispieceofart.ThanksarealsoduetomyenergeticanddynamicpublisherElsevier PublishingCompanytobringoutthisbookwellintime. Dr.AakashDeep
CHAPTER1 Introductiontomedicaldevices 1.1Introduction Theterm “medicaldevice” includeseverythingfromhighlycomplexcomputerized medicalequipmenttosimplewoodentongueinhibitors.Unlikedrugs,thefundamental wayofworkingthemedicaldeviceonthephysicalbodyisnotmetabolic,immunological,ormedicinal.
“Medicaldevice” meansanydevice,implant,reagent,ortitratorinthelaboratory, anyprogram,tool,oranyothersimilarorrelatedmaterialintendedbythemanufacturerforusealoneortogetherwithhumanswithoneormoreofthetargetssetfor
• diagnosis,control,prevention,treatment,orrelieffromdisease;
• diagnosis,monitoring,treatment,relief,orcompensationforinjury;
• investigate,replace,modify,orsupportanatomy;
• physiologicalprocess;
• supportorpreservelife;
• designcontrol;
• disinfectionofmedicaldevices;and
• providingmedicalinformationthroughalaboratoryexaminationofthephysical bodysamplesthatdonotaccomplishtheintendedprimaryactiononoronthe physicalbodybymedicinal,immunological,ormetabolicmeans,butcanbe accompaniedinitsfunctionbythesemeans [1].
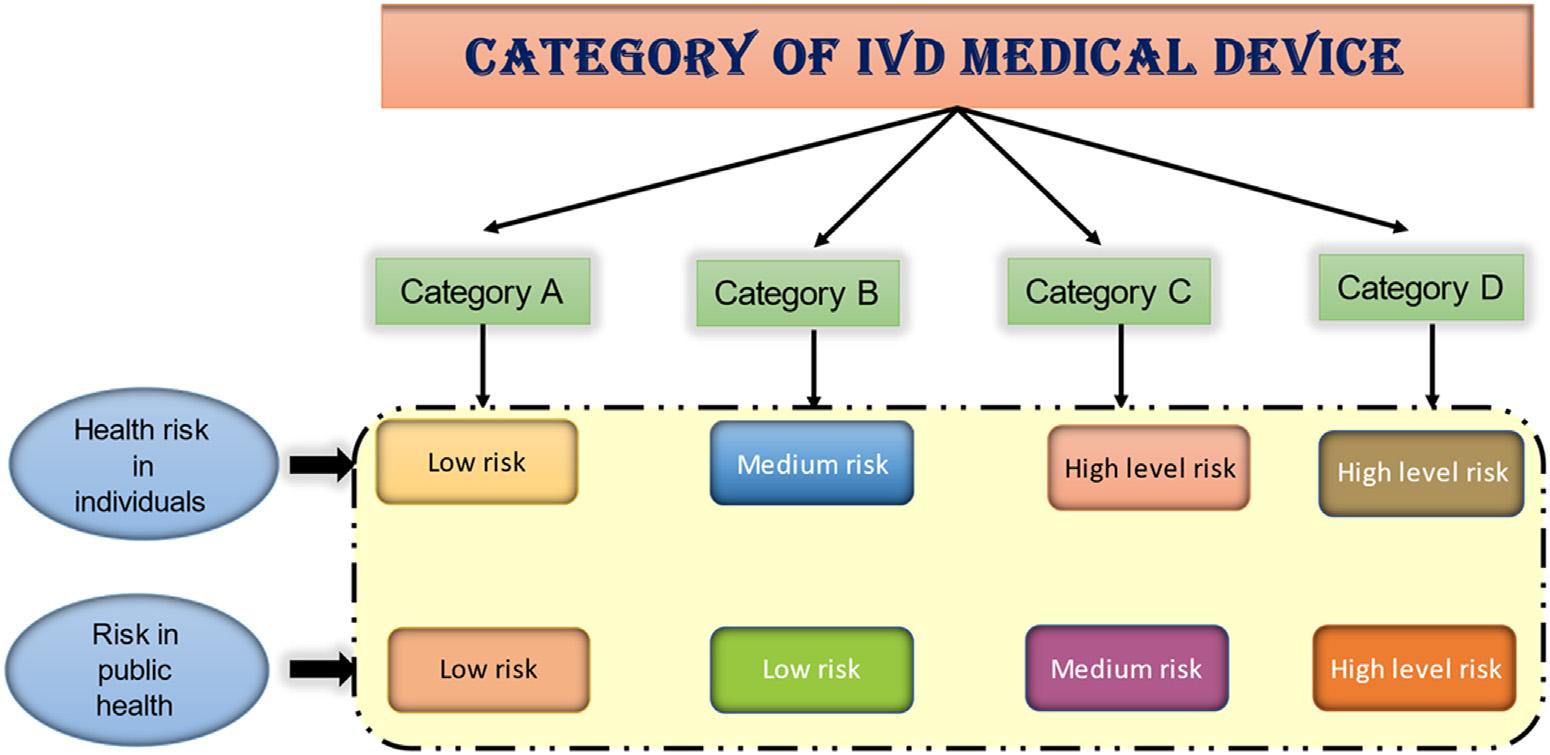
1.2Classificationsofmedicaldevicesonthebasisofrisk TheInternationalMedicalDeviceRegulatorsForum(IMDRF)classifiesmedicaldevices basedonrisktoassessthelevelofpremarketregulatorycontrolthatisnecessaryforusing medicaldevices,withthegoalofensuringthatthesecontrolsareappropriateforeach classtoprotectthehealthandsafetyofpatients,users,andotherpeople.Asshownin Fig.1.1,theoutcomeofthesystemistogroupIVDs(invitrodiagnosticdevice)into oneoffourriskclasses(A D) [2]
1.2.1Medicaldevicesafetyandperformance AmedicaldeviceandIVDmanufacturershoulddesignandmanufactureaproduct thatisbothsafeandeffectivethroughoutitslifecycle.Thisdocumentoutlinesthe
basicdesignandmanufacturingstandards,referredtoasthe “EssentialPrinciplesof SafetyandPerformance,” thatmustbemettoachievethisresult [3].
1.3Thebasicdesignandmanufacturingprincipleslistedinthis sectionapplytoIVDandmedicaldevices
1.3.1Commonprinciples IVDandmedicaldevicesshouldmeetthemanufacturer's [4,5] performanceexpectations,andtheyshouldbedesignedandmanufacturedinsuchawaythattheyare suitablefortheirintendeduseundertheconditionsofintendeduse.Theyshouldbe safeandoperateasintendedandpresentacceptableriskscomparedtothebenefitsfor thepatient,andtheymustnotharmtheclinicalconditionorpatientsafety,orthe safetyandhealthofusers,orothers,ifany.
Toensurethequality,safety,andcontinuousperformanceofthemedicaldevice andIVD,manufacturersshoulddevelop,implement,document,andmaintainarisk managementsystem.Riskmanagementshouldbeviewedasanongoingiterativeprocessthatrequiresregularperiodicupdatesthroughoutthelifeofthemedicaldevice andIVD.Toimplementriskmanagement,manufacturersmust:
• makeariskmanagementplanforeachIVDanddocumentit;
• providesafetyinformation(precautions/warnings/contra-indications)andappropriatetrainingtousers;and
• ascertaintheknownandexpectedrisksassociatedwithandmedicaldevice.
Figure1.1 ClassificationofIVDmedicaldevices.
1.3.2Clinicalinvestigation Aclinicalevaluationmayberequiredinsomecases,dependingonjurisdictional requirements.Aclinicalevaluationshouldevaluateclinicaldatatodetermineifthe medicaldeviceandIVDhaveafavorablebenefit riskdeterminationintheformof oneormoreofthefollowing:
• clinicalresearchreports(forclinicalperformanceappraisalreports);
• publications/scientificjournalspublished;and
• clinicalexperience.
Clinicalresearchshouldbecarriedoutinaccordancewiththeethicalprinciples outlinedintheDeclarationofHelsinki.Theseprinciplesprotecthumanrights,security,andwelfare,andtheyarethemostimportantconsiderationsinscienceandsociety,andtheywillprevail.Ateverystageofclinicalinvestigation,theseprinciplesmust beunderstood,observed,andapplied.Inaddition,somecountriesmayhavespecific regulatoryrequirementsforprestudyprotocolreview,informedconsent,andtheuse ofexcessIVDsamples [6].
1.3.3Physicochemicalandbiologicalproperties Withregardtothechemical,physicalandbiologicalpropertiesofamedicaldevice andanIVD,specialattentionshouldbegiventothefollowing [7]:
• Thechoiceofmaterialsused,especiallywithregardto:
• biocompatibility,
• toxicity,and
• flammability;
• Theeffectofoperationsonthepropertiesofmaterials;
• Wherepossible,previouslyvalidatedbiophysicalormodelingresearchresults;
• Themechanicalpropertiesofthematerialsused,which,whenapplicable,reflectissues likestrength,ductility,breakstrength,wearresistance,andfatigueresistance;and
• Surfaceproperties.
1.4Historyofmedicaldeviceregulationsglobally
1. JanetE.Trunzo,SeniorAdvisortothePresidentandSeniorExecutiveVice President,TechnologyandRegulatory Affairs,fortheAdvancedMedical TechnologyAssociation(AdvaMed)wholeadsateamofregulatoryexperts,presentedasummaryofeffortstoharmonizeregulatoryapproachesformedical devices.Thecoordinationdecisioncamefromvariousstakeholders,including governments,industry,andthereforethepublic.Coordinationprovidesaconsistentapplicationoforganizationalpri nciplesandapproachesandimprovesthe
effectivenessandefficiencyoftheorganizationalsystem.Thereisadiscounton repeatedorganizationalactivities,savingtimeandcost.Newproductsandtechnologiescometomarketinacumulativeandimprovedway,andtheprocessis moretransparent [8]
2. Manyregulatoryprogramsuseinternationalstandardsandguidelinesasabasisfor theirnationaltechnicalregulations.TrunzonotedthatalargenumberofFoodand DrugAdministration(FDA)staffparticipatedinvariousregulatorystandardscommittees.Itisalsoimportantthatregulatorysystemsseektheviewsofstakeholders aspartofthecoordinationprocess.
3. GlobalHarmonizationTaskForce(GHTF)wasavoluntarygroupcreatedin1992 asalinktoregulatorsandthereforetoanorganizedindustry.ThefoundingmembersweretheUnitedStates,theEuropeanUnion,Canada,Australia,andJapan. Therewerelinkswithotherorganizationsaroundtheworld,includingtheAsian CoordinationWorkingGroup;theGHTFhadaMemorandumofUnderstanding (MOUs)withISOand,accordingly,withtheIndependentEthicsCommittee, workingdirectlywiththeWorldHealthOrganization,andthuswiththePan AmericanHealthOrganization.GHTFhasbeenpermanentlyreplacedby InternationalMedicalDeviceRegulatorsForum(IMDRF)in2011 [9].
4. TheobjectivesoftheGlobalForumtoPromoteGrowthweretopromotetheconvergenceofglobalregulatorypracticesandthecommercializationoftechnologyinnovation andinternationaltradethroughcoordinated regulatoryprocesses.Theworkinggroup wasalsodesignedtoactasaforumfortheexchangeofinformation(GHTFdidnot evaluatetheeffectivenessofregulatorysystemsaroundtheworld) [10].
5. GHTFStructure:TheGHTFForumwaschairedbyapanelof4regulatoryand4 industryrepresentativesfromeachofthethreegeographicregions:NorthAmerica, Europe,andAsiaPacific(totalof24members).Thedirectionofthecommittee changedevery3years.Inadditiontothecommittee,whichguidedtheworkand definedtheorganization'sstrategicplan,therewerefivestudygroupsand unplannedworkinggroups,asneeddictated.
6. Thepremarketresearchgroup(StudyGroup1)producedanumberofpapersthat helpedtoshapetheconceptofaharmonizedregulatorymodel.StudyGroup2, whichfocusedonpostmarketingissues,waschargedwithdevelopingprocedures tomonitorandreportnegativeevents.Qualitysystems,theprimaryobjectiveof StudyGroup3,benefitfromtheInternationalStandardISO13485Quality ManagementSystems.
7. Themainthemeoftheactivityistheprinciplesofclassification;inparticular, thecreationofstandardvocabulary.Othercoretopicsincludetechnical requirements,theshapeandc ontentofsalesapplication s,evaluationandreview practices,postmarketingactivities,qu alitymanagementsystem,andbusiness functionrequirements [11]
1.5Productlifecycleofmedicaldevice TheMedicalProductLifeCycleisassociatedwithregulatoryprocessesforindustry leadersintheUnitedStates,theEuropeanUnion,andothercountriesthatmimic theirpolicies.However,therelationshipbetweentheLegislativeCouncilandregulationisnotalwayscleartomany.Managingproductsthroughouttheirlifecycleis essentialforendusers,andthusforthecompaniesthatproduceandmarketthem. Understandingtheinseparablelinkbetweenregulationandmarketsandtherefore themedicaldeviceindustryisessentialfortheassemblyofsafeandeffectivedevices, sustainableclinicalimprovementsfortheindustry,andthusforlong-termmedical ethicsdatingbackto “donothurt” [12]
1.6Thefivestagesofthemedicinalproductlifecycle Medicaldevicesaremostcommonlyusedtodiagnoseortreatpatients,butsomeare alsofrequentlyusedtoeducatepeopleabouttheirhealth.Regardlessoftheirintended use,thesedeviceswouldgothroughthesamefivesteps:research,production,testing, launch,andpostmarketassessment.
1.6.1Investigation Theconceptmaybeanideaforaneworimprovedsystembasedontheuseofan existingdevice.Inanycase,thoroughresearchisessentialtoensureaviabledefinition. Manyproductswillnotprogressbeyondthisstagebecausetheirdevelopershavenot thoroughlyresearchedtheiridea.Theycan,ideally,respondtothefollowing questions:
• Whoisthetargetaudienceforthisproduct?
• Whatarethethreatsassociatedwithmechanicalandmanufacturingprocessed?
• Istheconceptuniqueinrelationtoalloranyotherdevicesdesignedtoperforma similarfunction?
• Itisbesttodiscussaconceptwithanexperiencedengineerwhohaspreviously workedonsimilarmedicaldevices.Tohelpanswerthosequestions,theywillconductacriticalassessmentofacommodity.
1.6.2Design Themachinehasbeendeveloped,reviewed,updated,andredesignedusingagile productengineering.Computermodelsandprototypesareusedtotestthedesignand assessitsmarketability.Herearesometoolstohelpyouthinkpositively:
• 3Dprinting:3Dprintingmaderapidprototypesavailabletothegeneralpublicby allowingmanufacturerstocreatemodelsquickerandgainabetterunderstanding
ofthem.Thisgivesthedesigneraroughideathatheorshecanusetodevelopthe product.
• PowderLayerMergers ahigh-densitylaserthatfusespowderedmetalmaterials into3Dmodelsandshapes.Powderbedcombinationsenabledesignerstobuild strongerdesigns,makingthemacommonoptionformedicaldeviceprototypes.
• Computernumericalcontrol(CNC)machining:thisapproachisparticularlyuseful whenworkingwithrawmaterialstocreateadesign.Sincethemachinecontrols manufacturingequipmentand3Dprinters,acomplex,detaileddesign,andhighqualityprototypesareproduced.
1.6.3Validation TheFDAhasdevelopedguidelinesforcertainmedicaldevices.Thecontrolsandparametersvariedbydevicetype,withdevicesbeingclassifiedonascaleofonetothreein termsoftheirfunction,invasiveness,andrisklevel.Whenitcomestocheckingthe resultandensuringapositiveoutcome,thedevice'sclasswilldecidewhatcontrols arerequired.Clinicaltrialsshouldbeperformedandsubmittedatthisstage,iftheyare needed,sothatthemakercanrequestpremarketapproval.
1.6.4Launch
• ThecontentcreatorwillbeginpromotingandsellingitsgoodsafterobtainingpresaleapprovalfromtheFDA,ifnecessary.
• PremarketapprovalmaybeaanFDAmessagetocontentcreatorsindicatingthatit issecureforthegeneralpublic.
• Ifthedevicerequiressuchapproval,itwillnotbepossibletostartmarketingor sellingbeforereceivingit.Onthepositiveside,onlyabout5%ofallmedical devicesrequirethisstrictandcostlyprocess.Thisisgreatnewsformanyyoung creators,whomaynothavethe$94millionneededtoprovidethegeneralpublic withatoolthatrequirespremarketapproval.
• Toensurethatthematerialscomplywithlegislationandthatthemarketingstrategiesareacceptable,marketingmustworkcloselywithalegalteam.Marketing messagesandmethodsmustbecarefullyplannedthroughthismatterandarelikely tobebettermanagedbycompanieswithspecificmedicaldeviceexpertise.
1.6.5Postmarketreview Theprojectdoesnotstopuntilthedeviceisreleasedtothepublic.Aftertheproduct isreleased,thecreatormustmonitorforharmfuleffects,completenecessarychecks, reportadverseevents,andlikelycompleterecallsanddeviceremovals.Postmarket monitoringisanimportantaspectofthis,asdevelopersmustkeeptrackoftheeffects oftheirgoodsandkeepmeticulousrecordsofthem.Itisalsoimportanttokeeptrack
ofmedicalhistoryandkeeptrackofpatientregistries.Thisstagemayalsoinclude launchingthedeviceinsecondarymarkets [13].
1.7InternationalMedicalDeviceRegulatorsForum TheInternationalMedicalDeviceRegulatorsForum(IMDRF)wasdesignedin February2011asaforumtodiscussfuturedirectionsforcoordinatingtheregulation ofmedicaldevices.Itisavoluntarygroupofmedicaldeviceorganizersfromallover theworldwhohavebeenabletobuildonthestrongfoundationalworkoftheGlobal MedicalDevicesCoordinationGroup(GHTF)andacceleratethefulfillmentofaims totheinternationaldevicecoordinationandconvergenceofphysicians [14]
1.8IMDRFManagementCommittee TheIMDRFManagementCommittee,madeupofregulators,advisesonforumstrategies,procedures,directives,membership,andevents.Themanagementcommittee alsomanagesworkinggroupsthatdrawontheexpertiseofnumerousstakeholder groups,includingindustry,academia,healthpractitioners,andcustomersandbusiness groups.Thecurrentmembersare [7]:
• Brazil
• Canada
• Australia
• Singapore
• SouthKorea
• UnitedStates
• China
• Europe
• Japan
• Russia.
1.8.1PurposeofIMDRF ThemissionoftheIMDRFistostrategicallyacceleratetheregulatoryconvergenceof internationalmedicaldevicestocommercializeaneffectiveandefficientmedical deviceregulatorymodelthatrespondstonewchallengesinthissectorwhileprotectingpublichealthandmaximizingsecurity.
1.8.2OrganizationalconvergenceIMDRF Organizationalconvergence(hereinafterreferredtoas “ convergence ”)isavoluntary mechanisminwhichregulatorycriteriaandapproachesincountriesandregions becomemoresimilarorharmonizedovertimeasaresultoftheadoptionof
comparabletechnicaldocuments,norms,andscientificprinciples.Coordinationand similarorganizationalpracticesandproceduresareencouraged.Theprocessofconvergenceisacriticalformoforganizationalcooperationthatpermitsadditionaland improvedtypesofcooperationandsynergybetweenregulatoryauthorities [15]
1.9GlobalHarmonizationTaskForce(GHTF) 1. GlobalHarmonizationTaskForcewasestablishedtocoordinate(GHTF)in1993by governmentsandindustryrepresentativesfromAustralia,Canada,Japan,andthe EuropeanUnion,andtheUnitedStates.GHTFaimedtopromotetheconvergenceof standardsandregulatorypracticesassociatedwiththesafety,performance,andqualityof medicaldevices.TheGlobalTechnicalForum alsopromotedtechnologicalinnovation andfacilitatedinternationaltrade.Themainwaytoachieveitsgoalsweretopublish andpublishharmonizedguidancedocumentsforcoreorganizationalpractices.
2. Medicaldevicesaresubjecttoinadequatenationalpoliciesandlegislationinthemajorityofdevelopingcountries.WHOcooperationwiththeWorldHealthForumcan facilitatedevelopingcountries(importersandindustrialists)accessto [16]:
• informationonthemajormedicaldeviceregulatoryframeworks;
• approvalsformedicalequipmentandreviewsofhealthtechnologyfromhighly regulatedmarkets;
• adoptionofasinglenomenclatureformedicaldevices;
• innovativetechnologyadvances;and
• postmarketmonitoringandvigilancenetworks.
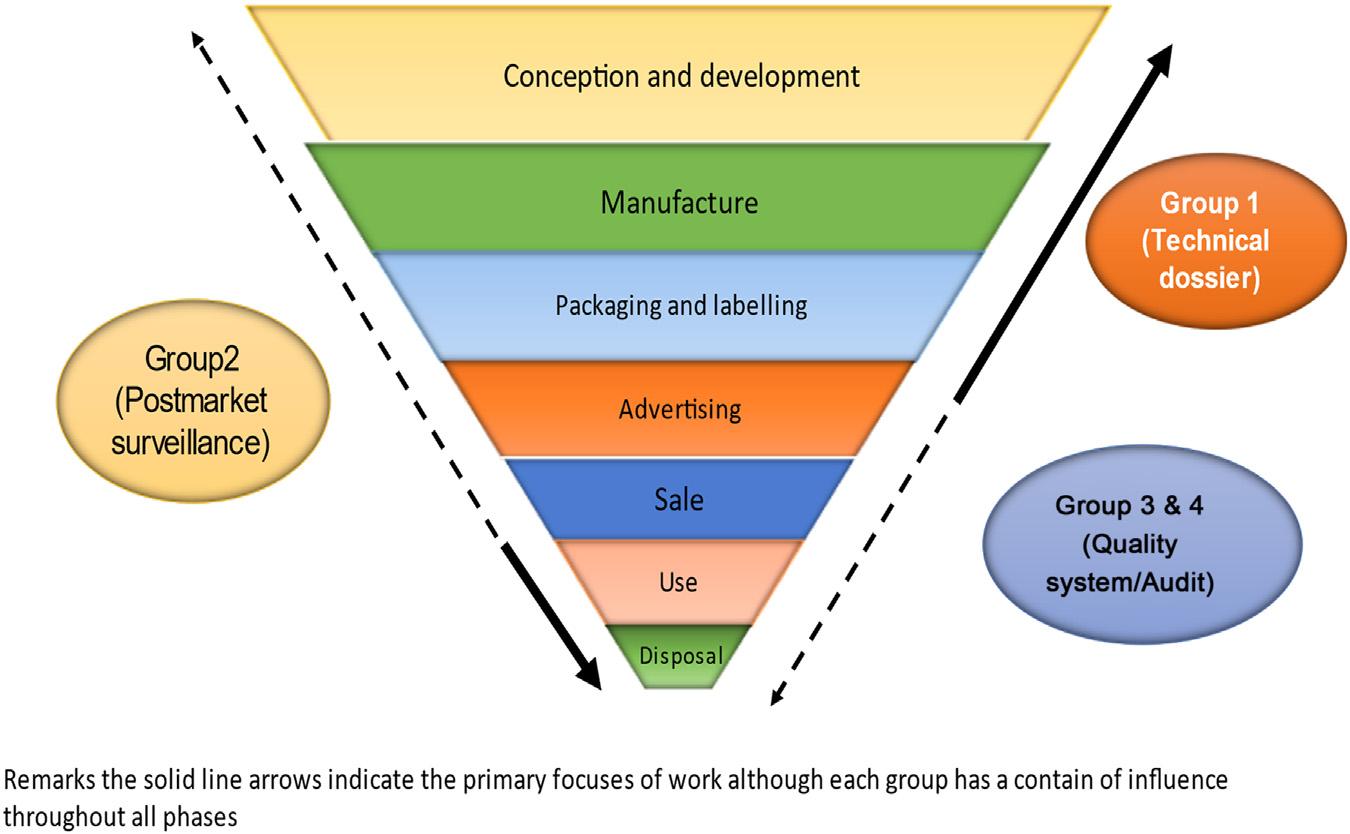
1.9.1Safetymanagement Thesafetyandperformanceofmedicaldevicesdependontwomaincriticalelements (premarketandpostmarketing surveillance).Premarket(group1)reviewcontributesto productcontrol,andpostmarketingsurveillance(group2)ensuresthatmedicaldevicesin usecontinuetobesafeandeffective.Thereisanimportantthirdelement,whichisthe representationoftheproducttotheuser [17].Allthreeelementsaredepictedin Fig.1.2.
1.10SummaryTechnicalDocumentation 1. TheSummaryTechnicalDocumentation(STED)hasbeendevelopedtoencourage furtherstandardizationofregulatorycardsformedicaldevicesinallmarkets [18,19].
2. STEDisrecognizedbyAmerican,European,Canadian,Australian,andJapanese regulators,aswellasinothermarkets.
3. TheIMDRFhasplannedSTEDformatupdates,butsofarthereislittleinthe scopefordetermination.
4. ManufacturersofallcategoriesofIVDmustdemonstratecomplianceofIVDwith thebasicprinciplesofsafetyandperformanceofmedicaldevices5bypreparing andstoringtechnicaldocumentsexplaininghowtodevelopthemedicaldevice, designandmanufactureeachIVDwithdescriptionsandexplanationsnecessaryto understandthemanufacturer'sdecisionregardingthiscompliance.Thesetechnical documentshavebeenrevisedtoreflectthecurrentstateoftheIVDviathemanufacturer'sregularQualityManagementSystemapplication.
5. Forconformityassessmentpurposes,themanufacturercollectsSTEDfromexisting technicaldocumentstosupplyapprovedrepresentative(AR)/conformityassessmentbody(CAB)provingthattherelevantIVDconformstobasicprinciples. STEDreflectstheconditionoftheIVDataspecificpointintime(e.g.,atthe timeofpremarketrenderingorwhenorderingARforpostmarketingpurposes) andisreadytorespondtoregulatoryrequirements.
6. Beforeandaftermarketuse,however,theconditionsforuseofSTEDaredifferent.WhenSTEDissenttotheAR/CAB,itmustbewritteninalanguage acceptabletotheexaminationauthority.Thedepthanddetailofknowledgefound inSTEDismainlybasedonclassificationtopicofIVD.
7. Sendfeedback.
8. History.
9. Saved.
10. Community.
Figure1.2 Elementsofsafetymanagement.
1.11Globalmedicaldevicenomenclature 1. GHTF(nowIMDRF)proposesGlobalMedicalDeviceNomenclature(GMDN) foruniquedeviceidentifier(UDI) [20].
2. EthicsCommittee(EC)proposesGMDNfortheEUDAMED(marketsurveillance database).
3. EUCOMEDsupportstheutilizationofGMDNinmeetingtheneedsofEuropean manufacturers.
4. EChastranslatedtheGMDNinto20languages.
5. WHOandMedecinsSansFrontieres(MSF)useGMDNinguidancefordevelopingcountriesCollaborationAgreementwithInternationalHealthTerminology StandardsDevelopmentOrganisation(IHTSDO)(SnomedCT).
6. USFDAisusingGMDNwithinthefirstnationalimplementationofUDI.
1.12Conclusion Thepresentstudyprovidesdesirableinformationofmedicaldevicesformedicalpurposesandprovidesallinformationregardingtheiruse,marketedsurveillance,andall safetyprotocolstoreducerisksthatarouseduringtheiruseandinformationonthe mainregulatorysystemsformedicaldevices.Medicaldevicesareregulatedbyvarious regulatoryauthoritieswhoarerecruitedbymanagementcommitteestosmoothlyrun thedevicesandreduceallrisks.
References [1]WHO,MedicalDeviceRegulations.GlobalOverviewandGuidingPrinciples[Internet].[citedApril 29,2020]. https://www.who.int/medical_devices/publications/en/MD_Regulations.pdf ,2020.
[2]PrinciplesofInVitroDiagnostic(IVD)MedicalDevicesClassification.AvailableFrom: https://www.imdrf. org/sites/default/files/docs/imdrf/final/technical/imdrf-tech-wng64.pdf.2021,2021(cited08-08-22).
[3]EMERGO,India:DraftEssentialPrinciplesforMedicalDeviceSafetyandPerformanceOutfor Comment.[citedApril29,2020]. https://www.emergobyul.com/blog/2017/07/india-draft-essential-principles-medical-device-safety-and-performance-out-comment ,2020.
[4]RAPS,IMDRFGuidanceAddressesEssentialPrinciplesforMedicalDevices,IVDs.[cited29April 2020]. https://www.raps.org/news-and-articles/news-articles/2018/1/imdrf-guidanceaddressesessential-principles-for,2020.
[5]GovernmentofIndia,EssentialPrinciplesforSafetyandPerformanceofMedicalDeviceGuideline. [citedFebruary2,2021]. https://cdsco.gov.in/opencms/export/sites/CDSCO_WEB/Pdf-documents/medical-device/Essentialprinciples.pdf ,2021.
[6] S.S.Altayyar,Theessentialprinciplesofsafetyandeffectivenessformedicaldevicesandtheroleof standards,Med.DevicesEvid.Res.13(2020)49 55.
[7]IMDRF,EssentialPrinciplesofSafetyandPerformanceofMedicalDevicesandIVDMedical Devices.[citedApril30,2020]. http://www.imdrf.org/docs/imdrf/final/technical/imdrf-tech181031-grrp-essential-principles-n47.pdf ,2020.
[8]AdvancedMedicalTechnolgyAssociation,JantE.Tronzo.SeniorAdvisortothePresidentand SeniorExecutiveVicePresident,Technology&RegulatoryAffairs.[cited17February2021]. https://www.advamed.org/about/leadership/janet-trunzo ,2021.
[9]IMDRF,AboutIMDRF.[cited30April2020]. http://www.imdrf.org/about/about.asp ,2020.
[10]Lexology,ABriefOverviewofRegulatoryFrameworkforMedicalDevicesinIndia.[cited30April 2020]. https://www.lexology.com/library/detail.aspx?g 5 e39ba922-f7c6-4568-a7e0-9b753769ada6,2020.
[11]T.Wizemann,TheGlobalFrameworkforRegulationofMedicalDevices.[citedApril30,2020]. https://www.ncbi.nlm.nih.gov/books/NBK209785/ ,2020.
[12]B.Fiedler,Y.David,Reframingproductlifecycleformedicaldevices,in:ManagingMedical DevicesWithinaRegulatoryFramework,2017,pp.3 16.[cited30April2020]. https://www. sciencedirect.com/science/article/pii/B9780128041796000010 .
[13]S.Daugherty,UnderstandingtheMedicalDeviceProductLifeCycle.[citedApril30,2020]. https://www.pacific-research.com/understanding-the-medical-device-product-life-cycle-prl/ ,2020.
[14]InternationalMedicalDeviceRegulatorsForum,[citedApril30,2020]. http://www.imdrf.org/,2020.
[15]MassMEDIC,InternationalMedicalDevicesRegulatorsForum(IMDRF).[citedApril30,2020]. https://www.massmedic.com/wp-content/uploads/2014/12/IMDRF-Presentation-Dec2014.pdf,2020.
[16]WHO,GlobalHarmonizationTaskForce(GHTF).[citedApril30,2020]. https://www.who.int/ medical_devices/collaborations/force/en/ ,2020.
[17] S.Ramakrishna,L.Tian,C.Wang,S.Liao,W.Teo,Globalharmonizationofmedicaldevices, Med.Devices(2015)207 213.
[18]EMERGO,WhatIstheSTEDFormatforMedicalDeviceTechnicalDocumentation?[citedApril 30,2020]. https://www.emergobyul.com/blog/2016/10/what-sted-format-medical-device-technical-documentation ,2020.
[19]IMDRF,SummaryTechnicalDocumentation(STED)forDemonstratingConformitytothe EssentialPrinciplesofSafetyandPerformanceofInVitroDiagnosticMedicalDevices.[citedApril 30,2020]. http://www.imdrf.org/docs/ghtf/archived/sg1/technical-docs/ghtf-sg1-n063-2011-summary-technical-documentation-ivd-safety-conformity-110317.pdf ,2020.
[20]WHO,GlobalMedicalDeviceNomenclature(GMDN).[citedApril30,2020]. https://www.who. int/medical_devices/global_forum/Workshop_10_GMDN.pdf ,2020.
CHAPTER2 Ethicsofclinicaltrialsofmedicaldevices 2.1Introduction
2.1.1Clinicalinvestigationofmedicaldevices
Theregulationsforconductingmedicaldevice(MD)clinicaltrialsworldwidehavevaried widely [1].Consequently,complicationsarise whenatrialisconductedinonecountry havingprotocolstobefollowedwhichisdifferentfromtheclinicaltrialprotocolsdefined inanothercountrywherethedeviceistobeusedsothattheuseofatooltoplugthe gapsinGCP,whichispossibleinonecountry,mightnotbeallowedinthestricter country [2].Besides,dataproducedunderonesetofrulescouldalsobeconsideredquestionablegivendifferentrequirementsindifferentcountries.Reciprocalacceptanceof GoodClinicalPractices(GCPs)wouldfacilitatemultinationalstudiesandpromotethe utilizationofclinicaldatatosupportregulatorysubmissionsinmultiplecountries [3]
2.2Clinicalinvestigationalplanformedicaldevices 1. General:Allpartiesinvolvedinconductingclinicalresearchmustbeeligible througheducation,training,orexperiencetoperformtheirtasks,andthismust bedocumentedcorrectly;
2. Riskassessment;
3. Therationaleforplanningclinicalresearch;
4. ClinicalResearchPlan(PIC);
5. IBInvestigatorBooklet;
6. CaseReportingForms;
7. Monitoringscheme;
8. Searchsitechoice;
9. Agreement(agreements);
10. Signaturestobeappended;and
11. DataMonitoringCommittee [4].
2.3Clinicalinvestigationconduct 2.3.1General
TheclinicalinvestigationmustfollowtheprotocollaidoutintheCIP.Theclinical investigationwillnotbeginuntiltheEthicsCommittee(EC)and,ifnecessary,the
DOI: https://doi.org/10.1016/B978-0-323-91126-9.00006-7
appropriateregulatoryauthorityofthecountrieswheretheclinicalinvestigationistakingplacehasgiventheirwrittenapproval/positiveopinion [5].
2.3.2Investigationsiteinitiation
Theestablishmentofatrialsiteensuresthatallnecessarytrialauthorizationsanddocumentsareinplaceandthattheprotocolandtrialproceduresarediscussedandagreed withtheprincipalinvestigatorandtheprincipalinvestigator'strialpersonnelincompliancewiththeprotocols,standardoperatingprocedures(SOPs),GCP,andrelevant regulatoryrequirements.
2.3.3Investigationsitemonitoring Monitoringisanimportantpartofaclinicaltrial'squalitymanagementproceduresand isusedtoensurethestudy'senduringquality.Onceit'sdecidedwhichSOPtoadhere to,itwillbefollowedwithalltheclinicaltrialsfundedorcosponsoredbyoneor moreofthepartnerorganizations.Fromtimetotime,trialssponsoredbyorganizations otherthanthepartnerorganizationsmaybetrackedinaccordancewiththeSOP.
2.3.4Adverseeventsanddevicedeficiencies
Anyunfavorablemedicalcase,accidentaldiseaseorinjury,orunfavorableclinicalsigns, suchasanabnormallaboratoryfinding,insubjects,consumers,orotherstakeholders, whetherornotconnectedtothedeviceunderinvestigation,maycomeundertrialin theformofaclinicalinvestigation.
2.3.5Clinicalinvestigationdocumentsanddocumentation
Allrelevantformsandrecordsmustbeamended,ifneed,andupdatedinamanner appropriatetoGCP,withastatementexplainingthechange,ifany.Itisimportantto keeptrackofthesubjectswhohavedecidedtoparticipateintheclinicaltrial.TheEC mustapproveanysignificantchangestotheinvestigationplan.
2.3.6Additionalmembersoftheinvestigationsiteteam
ApartfromthePrincipalInvestigatorwhoconductsclinicalinvestigation-relatedproceduresand/ormakesessentialclinicalinvestigation-relatedandmedicalcaredecisions, individualmembersoftheinvestigationsiteteamataninvestigationsiteareappointed toworkunderthesupervisionofthePrincipalInvestigator.
2.3.7Subjectprivacyandconfidentialityofdata TheInstitutionalReviewBoard(IRBs)mustdecideifsufficientcareistakentoprotecttheconfidentialityofdatacollected.
2.3.8Documentanddatacontrol
Allrelateddocumentsofexternalorigin,suchasnationalstandardsorclientcontracts anddrawings,mustbetrackedandregulated.Theteammustmaintainrecordcontrol procedures,controlnotices,andothertemporarydatasources,suchashand-written information,withadataarchivalsystemforbackingupyourmachine.
2.3.9Investigationaldeviceaccountability Deviceaccountabilitylogsofallunopenedstudydevicesmustberecordedupon receipt(keepshippinglogs),disposition(persubjectusage,includingtheamountused, amountremaining,etc.),transfer(ifapplicable),andreturntothesitemustbe maintained.
2.3.10Accountingforsubjects Thecostsincurredduringthestudyfromthemomentapatienttakespartinaclinical trialofadevicetoitswindingupwillbeaccountedfor.DespiteCROshandlingthe analysis,itneedssomeonewithexpertiseintheaccountingsystemtocarryoutthe costingofthestudy.Contingencies,likethedeathofapatientduringthestudy,must beaccountedforascostsbytheaccountingmodel.
2.3.11Auditing Usuallydoneonlyonceortwiceduringthedurationofaclinicaltrial,auditingcovers awiderangeoftopics.Theauditorsanalyzeaselectionofdatafromacross-sectionof researchsites.Theyalsolookoverregulatorydocuments,includingtheresearchprotocol,IRBcorrespondenceandapprovals,informedconsentdocumentation,andinvestigatorbiographies.
2.4ISO14155:2011 ThedataintegrityrequirementisensuredusinginternationalstandardssuchasISO 14155:2011
1. ISO14155:2011examinesGCPforplanning,conducting,recording,andreportinghuman-managedclinicalresearchforregulatorypurposestoassessthesafetyor performanceofMDs.
2. TheprinciplesoutlinedinISO14155:2011refertoalloranyotherclinical researchandwillbeappliedtothegreatestextentpossible,takingintoaccountthe scopeofclinicalresearchand,therefore,nationalregulatoryrequirements.
3. ISO14155:2011specifiesthegeneralcriteriaforprotectingtherightsofhuman subjects,andtheirsafetyandwell-being,ensuring,clinicalscientificresearchisconductedwiththegreatestattentiontonationalandinternationalconcernsand,