HYBRIDANDCOMBINED PROCESSESFORAIR POLLUTIONCONTROL
HYBRIDANDCOMBINED PROCESSESFORAIR POLLUTIONCONTROL
Methodologies,Mechanisms andEffectofKeyParameters
Editedby
AYMENAMINEASSADI
UniversityofRennes,ENSCR/UMRCNRS,Alléede Beaulieu,Rennes,France
ABDELTIFAMRANE
UniversityofRennes1,InstituteofChemicalSciencesof Rennes,Rennes,France
TUANANHNGUYEN
InstituteforTropicalTechnology,VietnamAcademyof ScienceandTechnology,Hanoi,Vietnam
Elsevier
Radarweg29,POBox211,1000AEAmsterdam,Netherlands
TheBoulevard,LangfordLane,Kidlington,OxfordOX51GB,UnitedKingdom 50HampshireStreet,5thFloor,Cambridge,MA02139,UnitedStates
Copyright©2022ElsevierInc.Allrightsreserved.
Nopartofthispublicationmaybereproducedortransmittedinanyformorbyany means,electronicormechanical,includingphotocopying,recording,orany informationstorageandretrievalsystem,withoutpermissioninwritingfromthe publisher.Detailsonhowtoseekpermission,furtherinformationaboutthePublisher’s permissionspoliciesandourarrangementswithorganizationssuchastheCopyright ClearanceCenterandtheCopyrightLicensingAgency,canbefoundatourwebsite: www.elsevier.com/permissions
Thisbookandtheindividualcontributionscontainedinitareprotectedunder copyrightbythePublisher(otherthanasmaybenotedherein).
Notices
Knowledgeandbestpracticeinthisfieldareconstantlychanging.Asnewresearchand experiencebroadenourunderstanding,changesinresearchmethods,professional practices,ormedicaltreatmentmaybecomenecessary.
Practitionersandresearchersmustalwaysrelyontheirownexperienceandknowledge inevaluatingandusinganyinformation,methods,compounds,orexperiments describedherein.Inusingsuchinformationormethodstheyshouldbemindfuloftheir ownsafetyandthesafetyofothers,includingpartiesforwhomtheyhaveaprofessional responsibility.
Tothefullestextentofthelaw,neitherthePublishernortheauthors,contributors,or editors,assumeanyliabilityforanyinjuryand/ordamagetopersonsorpropertyasa matterofproductsliability,negligenceorotherwise,orfromanyuseoroperationof anymethods,products,instructions,orideascontainedinthematerialherein.
ISBN:978-0-323-88449-5
ForInformationonallElsevierpublicationsvisitourwebsiteat https://www.elsevier.com/books-and-journals
Publisher: SusanDennis
AcquisitionsEditor: AnitaKoch
EditorialProjectManager: KathrineEsten
ProductionProjectManager: ManjuThirumalaivasan
CoverDesigner: MarkRogers
TypesetbyAptara,NewDelhi,India
Contributors....................................................................xi Foreword.......................................................................xv
Chapter1Roleofnanomaterialsinsensingairpollutants1 KomalRizwan,MuhammadBilalandHafizM.N.Iqbal
1.1Introduction1
1.2Roleofnanomaterialsinsensingairpollutants3
1.3Conclusionandoutlook11 Conflictofinterests13 References13
Chapter2Anoverviewoftheadvancesinporousandhybrid materialsresearchforairpollutionmitigation17
A.Antony,H.Saini,K.Vinayakumar,S.N.Kumar,N.S.KumarandS.Sil
2.1Introduction17
2.2Carbon-basedadsorbents21
2.3Metal–organicframeworksandhybridmetal–organic frameworks24
2.4Mesoporoussilicananomaterials31
2.5Zeolites34
2.6LayeredDoubleHydroxides37
2.7CovalentOrganicFrameworks39
2.8Computationalstudyoftheporousmaterials43
2.9Conclusion46 References47
Chapter3Chemicalandbiologicalairremediationby photocatalyticbuildingmaterials63
FedericoSalvadores,SilviaMercedesZacarías,OrlandoM.Alfanoand MaríadelosMilagrosBallari
3.1Introduction63
3.2Outdoorairremediation64
3.3Indoorairremediation72
3.4Biologicalairremediation80
3.5Conclusions87 Acknowledgments87 References88
Chapter4Advancedoxidationprocessesforairpurification95 WibawaHendraSaputeraandAntoniusIndarto
4.1Nonthermalplasma95
4.2Photocatalysis101 References113
Chapter5Integratedprocessesinvolvingadsorption,photolysis, andphotocatalysis117
NarminaO.BalayevaandZaminMamiyev
5.1Introduction117
5.2Generaloverviewofadsorption,photolysis,and photocatalysis119
5.3Advancementsintheintegratedprocessinvolving adsorption–photocatalysis:nanomaterialsprospects124
5.4Isotherms,kineticsmodels,andmechanicsof adsorption–PCOhybridprocesses138
5.5Reactors144
5.6Conclusionsandfutureperspectives145 References146
Chapter6Biologicalprocessesforairpollutioncontrol153
ElhamFaroukMohamedandGamalAwad
6.1Introduction153
6.2Airpollutioncontroltechnologies154
6.3Biologicalremediationofairpollutants155
6.4Futuretrendsinbiofuelproduction162
6.5Conclusions162 References163
Chapter7Functionalizedmembranesformultipollutantsbearing airtreatment167
YutangKang,ZhaoxiangZhongandWeihongXing
7.1Introduction167
7.2Membraneforgas–solidseparation170
7.3Membranematerialsforairpurification178
7.4Functionalmembranematerialsforintegratedpurification ofairmultipollutants186
7.5Conclusionandoutlook192 Acknowledgment194 References194
Chapter8Hybridmaterialstoreducepollutioninvolving photocatalysisandparticulatematterentrapment201
AntonellaCornelio,AlessandraZanoletti,LauraEleonoraDepero andElzaBontempi
8.1Introductiontoparticulatematter201
8.2ConventionalmethodstoremoveairbornePM203
8.3Photodegradationprocess205
8.4Nanoparticlesentrapment206
8.5Photodegradationoforganicpollutants216
8.6Conclusions225 Acknowledgment225 References225
Chapter9Advancesinphotocatalytictechnologiesforair remediation229
ZhiyuZhang,YongRen,JunHeandJingWang
9.1Introduction229
9.2Classificationandenhancementofphotocatalysts230
9.3Photocatalytictechnologiesforthetreatmentof variousgases231
9.4Conclusionsandoutlook249 Acknowledgments250 References250
Chapter10Indoorairpollutionandtreatmentstrategies—Hybrid catalysisandbiologicalprocessestotreatvolatileorganic compounds257
HamzaRafeeq,MuhammadAnjumZia,AsimHussain,MuhammadBilaland HafizM.N.Iqbal
10.1Introduction257
10.2Sourcesofpollution259
10.3Eliminationofindoorairpollutants265
10.4VOCremovalbycatalyticoxidation266
10.5HybridcatalysisfortheremovalofVOCs266
10.6Catalyticoxidativedegradationmechanisms (adsorption/desorption)269
10.7Methodsofpurificationbasedonbiologicalprocesses271
10.8Conclusionandfuturestandpoints274 Acknowledgments275 Conflictofinterests276 References276
B.Karthikeyan,K.SivasankariandM.Sivanathan
11.1Introduction283
11.2Fabricationofvanadiumpentoxide/tyrosinecomposite285
12.1Introduction:indoorairpollutionanditsongoingsignificance291
AhmedAmineAzzaz,AchrafAmirAssadiandAymenAmineAssadi
14.1Filtrationprocessforairtreatment345
14.2Absorptionprocessforairtreatment348
14.3Adsorptionforairtreatment350
Contributors
OrlandoM.Alfano
InstituteofTechnologicalDevelopmentfortheChemicalIndustry,National UniversityoftheLitoralandCONICET,SantaFe,Argentina
PrashantAnand
DepartmentofArchitectureandRegionalPlanning,IITKharagpur,West Bengal,India
A.Antony
DepartmentofEnvironmentalProtection,DefenceBioengineering& ElectromedicalLaboratory(DEBEL),Bangalore,India
AchrafAmirAssadi
ResearchUnitAdvancedMaterials,AppliedMechanics,Innovative ProcessesandEnvironment,HigherInstituteofAppliedSciencesand TechnologyofGabes(ISSAT),UniversityofGabes,Tunisia
AymenAmineAssadi
UniversityofRennes,ENSCR/UMRCNRS,AlléedeBeaulieu,Rennes, France
GamalAwad
ChemistryofNaturalandMicrobialProductsDepartment,Pharmaceutical andDrugIndustriesResearchDivision,NationalResearchCentre,Dokki, Giza,Egypt
AhmedAmineAzzaz
UniversitySavoieMont-Blanc,CNRS,EDYTEM,Chambéry,France
NarminaO.Balayeva
InstituteofTechnicalChemistry,GottfriedWilhelmLeibnizUniversityof Hannover,Hannover,Germany
MaríadelosMilagrosBallari
InstituteofTechnologicalDevelopmentfortheChemicalIndustry,National UniversityoftheLitoralandCONICET,SantaFe,Argentina
MuhammadBilal
SchoolofLifeScienceandFoodEngineering,HuaiyinInstitute ofTechnology,Huai’an,China
ElzaBontempi
INSTMandChemistryforTechnologiesLaboratory,UniversityofBrescia, Brescia,Italy
AntonellaCornelio
INSTMandChemistryforTechnologiesLaboratory,UniversityofBrescia, Brescia,Italy
LauraEleonoraDepero
INSTMandChemistryforTechnologiesLaboratory,UniversityofBrescia, Brescia,Italy
AbrahamGeorge
DepartmentofArchitectureandRegionalPlanning,IITKharagpur,West Bengal,India
JunHe
DepartmentofChemicalandEnvironmentalEngineering,Universityof NottinghamNingboChina,Ningbo,China
AsimHussain
DepartmentofBiochemistry,RiphahInternationalUniversity,Faisalabad, Pakistan
AntoniusIndarto
DepartmentofChemicalEngineering,InstitutTeknologiBandung,Labtek X,Jl.Ganeshano.10Bandung,Indonesia;DepartmentofBioenergy EngineeringandChemurgy,InstitutTeknologiBandung,KampusITB Jatinangor,Indonesia
HafizM.N.Iqbal
TecnologicodeMonterrey,SchoolofEngineeringandSciences, Monterrey,Mexico
YutangKang
StateKeyLaboratoryofMaterials-OrientedChemicalEngineering, NationalEngineeringResearchCenterforSpecialSeparationMembrane, NanjingTechUniversity,Nanjing,China
B.Karthikeyan
AnnamalaiUniversity,AnnamalaiNagar,TamilNadu,India
N.S.Kumar
DepartmentofEnvironmentalProtection,DefenceBioengineering& ElectromedicalLaboratory(DEBEL),Bangalore,India
S.N.Kumar
DepartmentofEnvironmentalProtection,DefenceBioengineering& ElectromedicalLaboratory(DEBEL),Bangalore,India
ZaminMamiyev
InstituteofPhysics,ChemnitzUniversityofTechnology,Reichenhainer, Chemnitz,Germany
ElhamFaroukMohamed
AirPollutionDepartment,EnvironmentalResearchDivision,National ResearchCentre,Dokki,Giza,Egypt
HamzaRafeeq
DepartmentofBiochemistry,RiphahInternationalUniversity,Faisalabad, Pakistan
SanjuRani
DepartmentofPhysics,SRMInstituteofScienceTechnology,Ramapuram Campus,Chennai,India
YongRen
DepartmentofMechanical,MaterialsandManufacturingEngineering, UniversityofNottinghamNingboChina,Ningbo,China;ResearchGroup forFluidsandThermalEngineering,UniversityofNottinghamNingbo China,Ningbo,China
KomalRizwan
DepartmentofChemistry,UniversityofSahiwal,Sahiwal,Pakistan
SomnathC.Roy
SemiconductingOxideMaterials,NanostructuresandTailored Heterojunction(SOMNaTH)Lab,DepartmentofPhysics,IITMadras, Chennai,India
H.Saini
DepartmentofEnvironmentalProtection,DefenceBioengineering& ElectromedicalLaboratory(DEBEL),Bangalore,India
VigneshSajeev
DepartmentofArchitectureandRegionalPlanning,IITKharagpur,West Bengal,India
FedericoSalvadores
InstituteofTechnologicalDevelopmentfortheChemicalIndustry,National UniversityoftheLitoralandCONICET,SantaFe,Argentina
WibawaHendraSaputera
DepartmentofChemicalEngineering,InstitutTeknologiBandung,Labtek X,Jl.Ganeshano.10Bandung,Indonesia;CentreforCatalysisand ReactionEngineering,InstitutTeknologiBandung,Jl.Ganeshano.10 Bandung,Indonesia;ResearchCentreforNewandRenewableEnergy, InstitutTeknologiBandung,Jl.Ganeshano.10,Bandung,Indonesia
S.Sil
DepartmentofEnvironmentalProtection,DefenceBioengineering& ElectromedicalLaboratory(DEBEL),Bangalore,India
M.Sivanathan
AnnamalaiUniversity,AnnamalaiNagar,TamilNadu,India
K.Sivasankari
AnnamalaiUniversity,AnnamalaiNagar,TamilNadu,India
K.Vinayakumar
DepartmentofEnvironmentalProtection,DefenceBioengineering& ElectromedicalLaboratory(DEBEL),Bangalore,India
JingWang
DepartmentofElectricalandElectronicEngineering,Universityof NottinghamNingboChina,Ningbo,China
WeihongXing
StateKeyLaboratoryofMaterials-OrientedChemicalEngineering, NationalEngineeringResearchCenterforSpecialSeparationMembrane, NanjingTechUniversity,Nanjing,China
SilviaMercedesZacarías
InstituteofTechnologicalDevelopmentfortheChemicalIndustry,National UniversityoftheLitoralandCONICET,SantaFe,Argentina
AlessandraZanoletti
INSTMandChemistryforTechnologiesLaboratory,UniversityofBrescia, Brescia,Italy
ZhiyuZhang
DepartmentofMechanical,MaterialsandManufacturingEngineering, UniversityofNottinghamNingboChina,Ningbo,China;ResearchGroup forFluidsandThermalEngineering,UniversityofNottinghamNingbo China,Ningbo,China
ZhaoxiangZhong
StateKeyLaboratoryofMaterials-OrientedChemicalEngineering, NationalEngineeringResearchCenterforSpecialSeparationMembrane, NanjingTechUniversity,Nanjing,China
MuhammadAnjumZia
DepartmentofBiochemistry,UniversityofAgriculture,Faisalabad, Pakistan
Foreword
Airpollutionisthereleaseoftoxicfumesintotheatmosphere,which couldbethroughnaturalcausesormanmadeevents.Theformerrelatestotheeruptionofvolcanosthatcanreleaseamixtureofgases composedofmostlyCO2 ,H2 S,andSO2 ornatural-occurringfiresin driedvegetationthatreleasesCO2 .Thelatterrelatestothecombustion offuelincarengines,factories,ships,andothersimilarcombustion processes.Theartificialexpansionofcattleraisingovertallvegetation tomakeroomforgrassandwalkingspaceforfeedstockisanotherfactorthatcontributestoairpollutionsince,duringthedigestiveprocess ofruminants,thereissomereleaseofCH4 .Eventhoughtheseissues seemlocalized,airpollutionispresentinthegreatmajorityofsocieties spreadaroundtheglobe;accordingtotheWorldHealthOrganization (WHO),99%ofthepopulationbreathesairofpoorqualityduetothe highconcentrationofpollutants.Thisfactorcontributestotheemergenceofrespiratorydiseasessuchastheobstructionofthelungs,acute respiratoryinfection,lungcancer,andevenmoreseverecasesrelated toheartdiseasesandstroke.Thereareothermajorenvironmental implicationsofairpollution,whichincludeacidrainfromtherelease ofNO2 andSO2 .Also,globalwarmingcausedbytheexcessofCO2 ,CH4 , andothergasesintheatmosphereiswidelydebatedasitcanleadto climatechangeduetotheincrementofthegreenhouseeffect.Based onthat,addressingthecausesofairpollutionisamuch-desiredneed forsocietytolivehealthilyandsustainably.Severaltechnologiesare arisingtoaddressthissituation.Chemicalandbiologicalairremediationprocessesaregettingscientificandindustrialattention.Themost commononereliesontheuseofhighlyporousmaterialstoabsorb suchtoxicgasses.Eventhoughitisalow-costandconvenientalternative,theprocessingtechnologyofthesecompoundsoftenrequires aggressivechemicaltreatments.Afollow-uptypeoftechnologyforthat istheuseofmetaloxidestosynthesizehybridnanomaterialsthatare gainingvisibilityontheairpurificationmatter.Thesehybridmaterials canbefunctionalizedtoselectivelycapturespecificgases,whichcover commonlyknowntoxicsubstancesaswellaschemicalwarfaregases. Henceitisconvenientforaircleaningandtheremovaloftoxicfumes. Anothertechnologyusedtocapturetoxicgasesistheuseofbiological agents,whichcanadsorbthesegasesandconvertthemintoenergy throughabiochemicalprocess.Thistypeoftechnologyisalsovery attractivebecauseitnotonlyusesgasesresponsibleforenvironmental issuessuchasCO2 asfeedstockbutalsoconvertsitintoenergy,making
theoverallprocesshighlysustainable.Yetfurtherresearchisrequired toreachanefficientprocessthatcanbescalable.Hence,alongthose lines,severalapproachescanbeadoptedtosolvetheissueofairpollution,whichcanbedividedintoadsorption,biofixation,absorption, membranepurification,chemicallooping,hydration,andcryogenic technologiesforCO2 capture.Thuseventhoughairpollutionisamajor concernaroundtheglobe,therearefeasibletechnologiesavailableto tackleit.Thisbookcoversthefundamentalconcepts,materials,and technologiesusedtocontrolairpollution.Itprovidesadetailedinsight intothesignificantadvancementinhybridmaterialsforairpollution controlandwillbeverysuitableforscientists,students,andindustries workinginthisarea.
Thanks, RamGupta,Ph.D. AssociateProfessor DepartmentofChemistry
KansasPolymerResearchCenter
PittsburgStateUniversity
Pittsburg,Kansas,UnitedStates
1.2Roleofnanomaterialsinsensingair pollutants
1.2.1Inorganicnanomaterialsforsensingair pollutants
Gasesarebecomingextremelysignificantduetothegreatdevelopmentinelectronicdevicesthatareusedtodetectairpollutants. Materialsobtainedfrommineralsandrocksinnanosizearecalled inorganicnanomaterials.Inorganicmaterialsincludesmetal/metal oxides.Metalnanomaterialsarewidelyusedtodetectdifferentair pollutants(JohnandRubanKumar,2021).Semiconductingmetaloxidesarethemostpromisingmaterialsforthechemicalsensingof environmentalgaseouspollutants.Therearetwotypesofmetaloxidesemiconductors:p-typeandn-type.N-typesemiconductorsincludetitaniumdioxide,zincoxide,andferricoxide,whosecarriersaremostlyelectrons,whilep-typesemiconductorsincludenickel andcobaltoxides, whosecarriersareholes(Pearce,Schiffman,Nagle,andGardner,2003).Chemiresistivemetaloxide–basedsensors responseisobservedathightemperatures.Theabsorbanceofoxygenoccursonthefilmofmetaloxides,followedbytheformationofionicspecies,andthen,athightemperatures,itisdesorbed (Labidi,Gillet,Delamare,Maaref,andAguir,2006).Theresistance ofn-typesemiconductingmetaloxidesincreasesbecauseofreductioninelectronconcentrationduetothetransferofelectronsfrom theconductionbandtotheabsorbedoxygen,whileadecreasein theresistanceofp-typemetaloxidesoccursduetotheformation ofholesduetothetransferofelectronsfrom theconductionbandtotheabsorbedoxygen. Gaseouspollutantsmaybeoxidizing(NO, NO2 ,CO2 )orreducing(CO,H2 S,CH4 ,SO2 ) innature.Reductioninelectronconcentrationoccursonthemetaloxidesurfaceduringthedetectionofoxidizingairpollutants. Thereforeanincreaseintheresistanceofthe n-typemetaloxidelayeroccursandadecrease intheresistanceofthep-typemetaloxidesurfacehappens.Bylimitingtheanalyte,thesensingefficacyofmetaloxidescanalsobelimited.Thesensingmechanismofindiumoxide nanobriks(Han,Zhai,Gu,andWang,2018)for nitrogendioxidegasispresentedin Fig.1.2.
Figure1.2. Theschematic diagramofnitrogendioxide (NO2 )gassensingbyusing indiumoxidenanobricks(Han, Zhai,Gu,andWang,2018).
Figure1.3. Pd–SnO2 nanocompositeforsensing methanegas(Yaoetal.,2020).
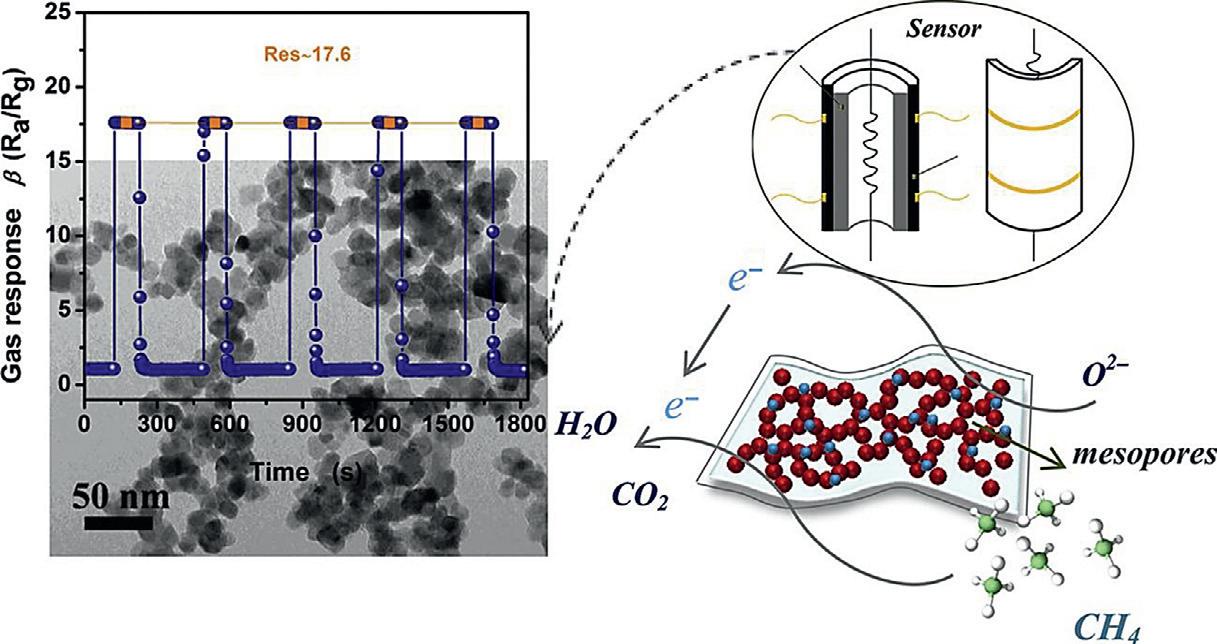
Wangandcolleaguessynthesizedcerium-dopedSnO2 nanocompositebyusingthegreenPechiniprotocol.Thesynthesizedmaterial wasusedasasensorforsensingethanolgas.Ce-dopinganSnO2 -based sensorshowedexcellentsensingofethanolwitharesponseof69.4at 50ppm.Thisresponsevalueis2.4timeshigherincomparisontothat withpristineSnO2 .Moreover,alowoperatingtemperature,greatsensitivity,selectivity,andlinearitywereobservedinsensoroperation.So cerium-dopedSnO2 nanocompositeshavegreatpracticalapplication insensinggases(Wangetal.,2022).Recently,aPd–SnO2 nanocompositewasfabricatedviathehydrothermalmethod.Tofacilitatethe fabricationofnanoporousstructures,theweakacidglucosemethodis proposed.Thefabricatednanocompositeshowedgreatsensingtoward methanegasincomparisontopristineSnO2 .Thesensor,whichis basedona2.5mol.%Pd–SnO2 nanocompositearchitecture,exhibited anultrafastresponseof17.6at3000ppmin3s.Recoverywasobtained in5satatemperatureof340°C.Thesensorshowedgreatstability andrepeatability.Theexcellentresponseofthesensormaybedue tothenovelnanoporousframeworkofthecompositeandthechemicalandelectronicsensitizationofpalladium,whichoffersapotential strategytoobtainpromisingmethanegassensing(Fig.1.3)(Yaoetal., 2020).
Gas-sensingfeaturescanbeinfluencedbyadjustingthecrystal phaseframeworkofthesensingmaterial.Inthiswaythebandgap andadsorptionpotentialofoxygenareoptimized.Thereforedifferent crystalphasestructuresoftitaniumdioxide,likerutileTiO2 nanorods (R-TiO2 NRs),brookiteTiO2 nanorods(B-TiO2 NRs),andanatase TiO2 nanoparticles(A-TiO2 NRs),werefabricatedusingthesingle-pot hydrothermalprocess.TheR-TiO2 NRs–basedsensorshowedagreat responseof12.3inthedetectionofacetonevaporsat100ppmat320°C incomparisontoA-TiO2 NRs(4.1)andB-TiO2 NRs(2.3),aswellasgreat
selectivityandrepeatability.Thegreatperformanceoftherutilesensor maybeattributedtothenarrowbandgapandgreatoxygenvacanciesof therutilephaseandthisshowsaroutetodesigningmetaloxide–based sensorsbyengineeringcrystalphases(Cao,Sui,Zhang,Zhou,Tu,and Zhang,2022).
Recently,porousZnOnanosheetsweresynthesizedbyusingthe solvothermalprotocol.Thesynthesizedmaterialwasusedforsensing NO2 .ZnOnanosheetsdetectedNO2 gaswithgreatsensitivitywitha responseof74.68at10ppm.Theobservedtemperaturewas200°C. Becauseofthehighsurfacearea,agreatresponsewasobtained.The morphologyofthematerialplaysagreatroleinsensingefficacy.Previously,ZnOnanoflowers(Kim,Porte,Ko,Kim,andMyoung,2017), ZnONRs(Jiao,Chien,VanDuy,Hao,VanHieu,Hjort,andNguyen, 2016),andZnOthinfilms(Patil,Nimbalkar,andPatil,2018)wereused tosenseNO2 gasandsignificantresultswereobtained,butporousZnO nanosheetsshowedgreatsensingperformanceintermsoflowsensing temperatureandagreatresponseatevenlowconcentrations.Recently, FanandcolleaguessynthesizedCo3 O4 nanoparticlesthroughtheelectrospraymethodtosenseacetonegas.Thesensorshowedaresponse of8.61at100ppmconcentrationat200°Cwithresponseandrecovery timesof43sand92s,respectively,towardsacetoneeveninthepresenceofbenzene,ethanol,andethylacetate(Fan,Xu,Ma,andHe,2021). Differentinorganicnanomaterialsusedforsensingairpollutantsare presentedin Table1.1.
1.2.2Organicnanomaterialsforsensingair pollutants
Organicmaterialsareobtainedfromanimalandplantsources. Organicnanomaterialsincludenanopolymericmaterialsandsmall organiccompounds.Organicpolymersareextensivelyusedacrossthe worldbecausetheyhaveadvantageslikefacileprocessing,flexibility, stability,cost-friendliness,lightweightandtailorability.Polymersare madeupofmanyrepeatingunitsarrangedinaspecificsequence,like polyethylene,inwhichethaneisthemonomericunit.Polymercharacteristicsdependonmoleculararchitecture,weight,andchemical compositionandmorphology.Anopticalsensingprinciplebasedon colorimetric,luminescence,andfluorescenceeffectsandachangein lightrefractiononthesurfaceofthepolymerinthepresenceofgases hasbeenexplainedby Adhikarietal.(2010).Organic-basedsemiconductingnanomaterialsmaybeincorporatedassensinglayersusing thesamesensingmechanism.Uponinteractionoftheconducting polymerswiththegaseousanalytes,p-typeconjugatedpolymersgive electronstogaseousspecieswhilen-typepolymersacceptelectrons fromgaseousspecies.Consequently,thepolymer’sholeconductivity
Table1.1Inorganicnanomaterialsforsensingairpollutants.
Ce-dopedSnO2 Ethanol69.450ppm23s13s265°C(Wangetal.,2022)
Pd–SnO2 nanoporous composites Methane17.63000ppm5s3s340°C(Yaoetal.,2020)
Au/SnO2 /ZnO nanosheets Ethanol36.2100ppm59s2s300°C(Li,Zhang,Han,Wang,Zhang,and He,2021)
ZnO–SnO2 Ethanol20.2100ppm5s87s250°C(Feng,Ma,Yu,Dong,Wang,and Liu,2020)
WO3 nanowireFormaldehydeSignificant5–15ppm--100°C(Bouchikhi,Chludzi ´ nski,Saidi, Smulko,Bari,Wen,andIonescu, 2020)
RutileTiO2 nanorodsAcetone12.3100ppm3s421s300°C(Caoetal.,2022)
PorousZnO nanosheets NO2 74.6810ppm--200°C(SikChoietal.,2021)
ZnOthinfilmsNO2 12.3100ppm3s37s200°C(Patiletal.,2018)
In2 O3 –SnO2 N-butanol76.550ppm--140°C(Anetal.,2021)
CuONPsTolueneSignificant240ppm194s297s160°C(ThangamaniandPasha,2021c)
(continuedonnextpage)
WO3 nanosheetsXylene24.5100ppm150s-200°C(Liangetal.,2019)
In2 O3 Ozone44200ppb70°C(Sui,Zhang,Zhou,andZhang, 2021)
CuAlO2 Ozone1.9200ppb--200°C(ThirumalairajanandMastelaro, 2016)
Co3 O4 @TiO2 CO15.99100ppm--140°C(Li,Wei,Zhu,Zhou,andGui,2021)
Co3 O4 NPsAcetone8.61,100ppm43s92s200°C(Fanetal.,2021)
h-MoO3 nanorodAmmonia0.35500ppm210s241sRT(Chua,Yaacob,Tan,andOng,2021)
MoSe2 nanoflakesH2 S18.5750ppb15s43s200°C(Jha,D’Costa,Sakhuja,andBhat, 2019)
α -Fe2 O3 LPGSignificant90ppm52s107s-(Choudhary,Annapoorni,and Malik,2021)
Au-sensitized SnO2 film LPG57780ppm60s-598K(Nakateetal.,2017)
ZnFe2 O4 NPsSO2 Significant10ppm15s15sRT(Vinhetal.,2021)
ZnOnanoflakesCO2 Significant200–1025ppm9–17s9–17s250°C(KanaparthiandSingh,2019)
WO3 nanoparticlesCl2 72.61ppm58s28s140°C(Wangetal.,2018)
CdO–ZnOnanoricesFormaldehyde34.5300ppm10s6s350°C(Umar,etal.,2021)
Sm-dopedSnO2 Ethanol138.950ppm--160°C(An,Liu,Li,Zhou,Wang,Zou,and Lian,2021)
isenhancedasitdonateselectronstogases,andsimilarly,adecrease inconductivityisobservedaftertheacceptanceofelectronsfrom gases.Thisisthemainreasonforthedecreaseoftheconductivityof conjugatedpolymers(polyaniline[PANI],polypyrrole[PPy],polythiophenes,etc.)uponexposuretodifferentnucleophilicgasesasethanol, ammonia,H2 S,methanol,andhydrazine,whileelectrophilicgaseslike oxygen,nitrogendioxide,andphosphorustrichlorideexhibittheoppositephenomenon.Theinteractionofpolymerswithpollutantsmay alsooccurthroughsomekindofweakinteractionsandsomecharge inductiontoaffecttheconductingcharacteristicsduringthesensing process.
Recently,ananosensorbasedonalignedsingle-walledcarbon nanotubes(SWNTs)wasfabricatedforsensingNO2 gasatroom temperature.Thesensorwasfoundtobehighlystableandsensitive anddisplayedashortrecoverytime,eveninthepresenceofUV light.ThealignedSWNTswerefabricatedbyemployingtheAC-DEP (Alternatingcurrent-dielectrophoresis)technique.Sensorsemploying randomnetworkedSWNTswerealsousedandtheefficaciesofthe twosensorswerecompared.Bothsensors,(randomandaligned)were usedtosenseNO2 atroomtemperatureandthealignednetworkbasedsensorshowed3.5timeshighersensitivityincomparisonto therandomlynetworkedsensor.Therecoverytimeforrandomand alignedSWNTsat0.5ppmofNO2 wasfoundtobe124sand50s, respectively(Chauhan,Kumar,Chaturvedi,andRahman,2019).A PANI-basedsensorwassynthesizedforsensingammoniagas.The filmsofPANIweredepositedonPET(polyethyleneterephthalate)basedsubstratethroughafacileinsitupolymerizationtechnique. PANIfilmswereusedtosenseammoniagasatroomtemperature. Functioningofthesensoratroomtemperaturebutitfacilitates operationatlowpowerandalsoenhancesthesensorlifetime. Thesensorshowedexcellentstabilityandreproducibilityatwide concentrationrange(5to1000ppm).Itwasmechanicallyrobust andcanbebentwithoutdamage,showinggreatstability,andits efficacywasnotaffectedevenaftervariouscyclesofbending.Conclusively,PANIfilmsmaybeusedasefficientportablesensorsforon-site detectionpurposes(Kumar,Rawal,Kaur,andAnnapoorni,2017).Yang andcolleaguesdevelopedporouspolyaniline/polyvinylidenefluoride (PANI/PVDF)compositefilmusingtheoxidativepolymerization method.Thesynthesizedfilmexhibiteda3Dporousframework, whichincreasedthesensingpotentialofthePANIsensor.Great sensitivityat25°Cwasexhibitedbythesensorforsensingammonia. Thesensorcansenseammoniaat1ppmwith27%responsealong withgreatreproducibilityandstability.ThePANI/PVDfilmexhibiteda responseof6.5%with0.2ppmammoniaat25°C.Bendingstabilitywas alsoexhibitedbyfilmandlittlevariationinresponsewasobservedat
variousbendingangles.ThePANI/PVDFfilmcanbeusedforreal-time monitoringoftheenvironment(Yangetal.,2020).Theroleofdifferent organicnanomaterialsforsensingairpollutantsispresentedin Table1.2.
1.2.3Organic-inorganicnanocompositesfor sensingofairpollutants
Fortheformationofnanohybrids,variousorganicandinorganic nanomaterialshavebeenintroduced.Innanohybrids,onecomponent ofthemixtureshouldbeinthenanodimension.Nanomaterial-based compositesincludeacombinationofpolymersormetaloxideswith carbon,metal,graphene,andmetaloxides.Nanocompositesplaya significantroleinsensingairpollutants.Huangandcolleaguessynthesizedthenanocompositeofpoly(4-styrenesulfonicacid)(PSSA)dopedPPy/tungstenoxide(WO3 )/reducedgrapheneoxide.The compositewassynthesizedbydispersinganappropriatequantityof respectivematerialsinaqueousmedium.Thesynthesizednanocompositewasspincoatedonthesurfaceofanacousticwaveresonatorto formasynthesizednitricoxidegassensor.Theas-fabricatedsensordetectednitricoxidegasatroomtemperatureataconcentrationof1–110 ppbwithasensitivityof12Hz/ppb.Therecoveryandresponsetimes werefoundtobe <2min.TheLOD(limitofdetection)valuewasfound tobe0.31ppb.Thesensorshowedquickresponse,repeatability,and greatrecoveryatroomtemperature.Thesensingefficacyofthesensor remainedstableover30dayswitha6.3%reductioninsensitivity.It wasfoundhighlyselectiveforNOgaseveninthepresenceofcarbon dioxide,ammonia,andnitrogendioxide.Thismaybeduetothesynergisticeffectofreducedgrapheneoxide,tungstenoxidenanoparticles, andPPymaterial(Hung,Chung,Chiu,Yang,Tien,andShen,2021).
Recently,Shanavasandcolleaguessynthesizedanickeloxide andreducedgrapheneoxide(NiO/rGO)nanocompositeviatheeasy single-potmicrowaveirradiationmethod.Themorphologicalcharacteristicsofthenanocompositeincludeasizeof50–60nm.Nickel oxidenanoparticleswerefoundcoveredbythesheetsofreduced grapheneoxidethoroughly,whichexhibitsexcellentsynthesisofthe binarynanoarchitecture.Thepresenceoffunctionalmoietieswas analyzedbyusingFouriertransforminfraredspectroscopy(FTIR). Thefabricatedsensorwasusedtodetectacetoneandcarbondioxidegas.TheNiO/rGO-basedsensorshowedagreatsensingresponse forcarbondioxidegaswithasensitivityof83counts/ppm.Theresponseandrecoverytimeswerefoundtobe16sand22s,respectively(Shanavas,Ahamad,Alshehri,Acevedo,andAnbarasan,2021). Khanandcolleaguessynthesizedanammoniagassensorbasedon PVP/WO3 nanocomposite.Todepositthesensorymaterialonthe