Listofcontributors
LucaAnsaloni SINTEFIndustry,Oslo,Norway
PietroArgurio DepartmentofEnvironmentalEngineering,Universityof Calabria,Rende,Cosenza,Italy
AngeloBasile Hydrogenia,Genova,Italy;UnitofChemical-Physics FundamentalsinChemicalEngineering,DepartmentofEngineering,University CampusBio-MedicalofRome,Rome,Italy
SlametBudijanto DepartmentofFoodScienceandTechnology, IPBUniversity,KampusIPBDarmaga,Bogor,Indonesia
LoredanaDeBartolo CNR-ITM,NationalResearch CouncilofItaly,Institute onMembraneTechnology,Rende,Cosenza,Italy
KiwintaDiaussie DepartmentofFoodScienceandTechnology, IPBUniversity,KampusIPBDarmaga,Bogor,Indonesia
AlbertoFigoli InstituteonMembraneTechnology(CNR-ITM),Rende, Cosenza,Italy
FrancescoGaliano InstituteonMembraneTechnology(CNR-ITM),Rende, Cosenza,Italy
FaustoGallucci SustainableProcessEngineering,ChemicalEngineeringand Chemistry,EindhovenUniversityofTechnology,Eindhoven, TheNetherlands
EllenGapp InstituteforMicroProcessEngineering(IMVT)atKarlsruhe InstituteofTechnology(KIT),Eggenstein-Leopoldshafen,Germany
HuaHuang DepartmentofMaterialsScienceandEngineering,Clemson University,Clemson,SC,UnitedStates
CristinaLavorato DepartmentofEnvironmental Engineering,Universityof Calabria,Rende,Cosenza,Italy
MarcoMartino DepartmentofIndustrialEngineering,UniversityofSalerno, Fisciano,Italy
EugenioMeloni DepartmentofIndustrialEngineering,UniversityofSalerno, Fisciano,Italy
PierreMillet Paris-SaclayUniversity,ICMMO(UMR8182),Orsay,France
RaffaeleMolinari DepartmentofEnvironmentalEngineering,Universityof Calabria,Rende,Cosenza,Italy
SabrinaMorelli CNR-ITM,NationalResearchCouncilofItaly,Instituteon MembraneTechnology,Rende,Cosenza,Italy
VincenzoPalma DepartmentofIndustrialEngineering,UniversityofSalerno, Fisciano,Italy
ThijsA.Peters SINTEFIndustry,Oslo,Norway
PeterPfeifer InstituteforMicroProcessEngineering(IMVT)atKarlsruhe InstituteofTechnology(KIT),Egge nstein-Leopoldshafen,Germany
AntonellaPiscioneri CNR-ITM,NationalResearch CouncilofItaly,Institute onMembraneTechnology,Rende,Cosenza,Italy
CarmellaRosabel DepartmentofFoodScienceandTechnology, IPBUniversity,KampusIPBDarmaga,Bogor,Indonesia
SimonaSalerno CNR-ITM,NationalResearchCouncilofItaly,Instituteon MembraneTechnology,Rende,Cosenza,Italy
SergioSantoro DepartmentofEnvironmental Engineering,Universityof Calabria,Rende,Cosenza,Italy
AzisBoingSitanggang DepartmentofFoodScienceandTechnology, IPBUniversity,KampusIPBDarmaga,Bogor,Indonesia
JianhuaTong DepartmentofMaterialsScienceandEngineering,Clemson University,Clemson,SC,UnitedStates
HildeJ.Venvik DepartmentofChemicalEngineering,NorwegianUniversity ofScienceandTechnology(N TNU),Trondheim,Norway
J.Vital DepartmentofChemistry,NOVASc hoolofScienceandTechnology, LAQV-Requimte,UniversidadeNo vadeLisboa,Caparica,Portugal
ZeyuZhao DepartmentofMaterialsScienceandEngineering,Clemson University,Clemson,SC,UnitedStates
MindaZou DepartmentofMaterialsScienceandEngineering,Clemson University,Clemson,SC,UnitedStates
Listofcontributorsxiii
Prefacexvii
1.Introductiontomembraneandmembranereactors1
VINCENZOPALMA,MARCOMARTINO,EUGENIOMELONIANDANGELOBASILE
1.1Introductionandprinciples1
1.2Membranes6
1.3Membranebioreactors8
1.4Combinationofmembranesandcatalyticreactions10
1.4.1Interfacialcontactormode15
1.4.2Flow-throughcontactormode15
1.5Conclusionsandfuturetrends15
Nomenclature16
Acronyms16
Symbols16 References17
2.Protonicelectrocatalyticmembranereactors21
ZEYUZHAO,MINDAZOU,HUAHUANGANDJIANHUATONG
2.1Introduction21
2.2Ammoniasynthesis22
2.2.1Thecommondesignofprotonicelectrocatalytic membranereactorsfortheammoniasynthesis23
2.2.2Electrocatalyticnitrogenreductionreactionmechanism24
2.2.3Electrolytematerials26
2.2.4Cathodematerials27
2.2.5Anodehydrogenfeedstocks28
2.3CO2 reduction30
2.3.1ThecommondesignofProtonicelectrocatalytic membranereactorsfortheCO2 reduction31
2.3.2MechanismsoftheCO2 electrocatalyticreduction31
2.3.3Electrolytematerials32
2.3.4Cathodicmaterialsandcatalysts34
2.3.5Anodicmaterials35
2.4Hydrocarbondehydrogenation36
2.4.1Methaneupgrading36
2.4.2Conversionofalkanestoalkenes42
2.5Otherreactions44
2.6Conclusionandfuturetrends45
Nomenclature46
Acronyms46 References47
3.Packedbedmembranereactors59 FAUSTOGALLUCCI
3.1Introduction59
3.2Latestdevelopmentsinpackedbedmembranereactors62
3.3Conclusionsandfuturetrends73
Nomenclature73
Acronyms73 References74
4.Fluidizedbedmembranereactors77
FAUSTOGALLUCCI
4.1Introduction77
4.2Latestdevelopmentsinfluidizedbedmembranereactors79
4.3Conclusionsandfuturetrends90 Nomenclature91 Acronyms91 References91
5.Microstructuredmembranereactorsfor processintensification95
ELLENGAPP,LUCAANSALONI,HILDEJ.VENVIK, THIJSA.PETERSANDPETERPFEIFER
5.1Introduction95
5.2Designandfabrication96
5.3Examplesofmicrostructuredmembranereactors105
5.3.1Polymeric105
5.3.2Metallicmembranes110
5.3.3Zeolitemembranes113
5.3.4Ceramicoxygenandprotonconductingmembranes115
5.4Conclusionandfuturetrends117 Nomenclature118 Acronyms118 Symbols118 References118
6.Pervaporationmembranereactor127
SERGIOSANTORO,ALBERTOFIGOLIANDFRANCESCOGALIANO
6.1Introduction127
6.2Pervaporationmembranereactors130
6.3Fieldsofapplication134
6.3.1Esterificationreactions135
6.3.2Etherificationreactions139
6.3.3Acetalizationreactions140
6.3.4Condensationreactions141
6.3.5Bio-alcoholproduction(pervaporationbioreactors)141
6.4Conclusionsandfuturetrends142 Nomenclature144
Acronyms144 References145
7.Polymericmembranereactors151
J.VITAL
7.1Introduction151
7.2Polymericmembranes152
7.2.1Structureofpolymericmembranes152
7.3Classificationofmembranereactors167
7.3.1Extractor-typemembranereactors168
7.3.2Contactor-typemembranereactors171
7.3.3Distributor-typemembranereactors176
7.4Polymericmembranemicroreactors177
7.5Conclusionsandfuturetrends179
7.6Acronyms179 References181
8.Currenttrendsinenzymaticmembranereactor195
AZISBOINGSITANGGANG,KIWINTADIAUSSIE, CARMELLAROSABELANDSLAMETBUDIJANTO
8.1Introduction195
8.2Designsofenzymaticmembranereactor196
8.3Membranecharacteristics197
8.4Enzymeimmobilizationinenzymaticmembranereactor203
8.5Enzymaticmembranereactorversusotherreactor configurations209
8.6Applicationsofenzymaticmembranereactor210
8.7Conclusionandoutlook217
Nomenclature217 Acronyms217 References218
9.Membranereactorsinbioartificialorgans227
SABRINAMORELLI,SIMONASALERNO, ANTONELLAPISCIONERIANDLOREDANADEBARTOLO
9.1Introduction227
9.2Bioartificialorgans designissues228
9.3Transportphenomena229
9.4Membranebioreactorasbioartificialliver231
9.4.1Membranebioartificialliversinflatconfiguration232
9.4.2Membranebioartificialliversinhollowfiber configuration233
9.5Membranebioreactorsforbioartificialkidney236
9.5.1MembranesforBAK236
9.5.2BAKdevicesinanimalstudiesandclinicaltrials237
9.6Membranebioreactorasabiomimeticmodelfor nervoustissueanalogue239
9.7Conclusionsandfutureperspectives243 Nomenclature243 References244
10.Photocatalyticmembranereactors251 RAFFAELEMOLINARI,CRISTINALAVORATOANDPIETROARGURIO
10.1Introduction251
10.2Basicprinciplesofphotocatalysis253
10.3Basicofphotocatalyticmembranereactors257
10.3.1Typesofphotocatalysts257
10.3.2Typesofmembranes260
10.3.3Membranemodulesandsystemconfigurations263
10.4Applicationsofphotocatalyticmembranereactors268
10.4.1Photocatalyticmembranereactorsin photodegradationofpharmaceuticalsinwater268
10.4.2Photocatalyticmembranereactorsinthe conversionofCO2 insolarfuels273
10.5Advantagesandlimitationsofphotocatalyticmembrane reactors274
10.6Conclusionandfuturetrends275
Listofsymbols277
Listofacronyms277
Acknowledgments278 References278
11.Electrochemicalmembranereactors285
PIERREMILLET
11.1Introduction285
11.2Electrochemicalreactors286
11.2.1Generalprinciples286
11.2.2Endergonictransformers289
11.2.3Exergonictransformers290
11.2.4Cellseparators290
11.3Diaphragmsforliquidelectrolytes292
11.3.1Asbestos293
11.3.2Thermoplasticdiaphragms293
11.4Polymermembranematerials295
11.4.1Protonconductingionomers295
11.4.2Hydroxyl-ionconductingionomers298
11.5Ceramicmembranematerials300
11.5.1Nonorganicprotonconductors300
11.5.2Oxide-ionconductors302
11.6Selectedendergonicapplications304
11.6.1Waterelectrolysis304
11.6.2Mainwaterelectrolysistechnologies305
11.6.3Brineelectrolysis305
11.7Conclusionsandfuturetrends310 Nomenclature310 References311 Furtherreading313
12.Modelingofmembranereactors315
FAUSTOGALLUCCI
12.1Introduction315
12.2Packedbedmembranereactors316
12.2.11Dpseudo-homogeneousmodel316
12.2.22Dpseudo-homogeneousmodel322
12.2.3Modelingoffluidizedbedmembranereactors325
12.3Conclusionsandfuturetrends331 Nomenclature332 References334
13.Techno-economicanalysisofmembranereactors337 FAUSTOGALLUCCI
13.1Introduction337
13.2Latestdevelopmentsintechno-economicanalysisfor membranereactors340
13.3Conclusionsandfuturetrends351 Nomenclature353 References353 Index355
Preface
Membranereactors(MRs)aremultiphase,multifunctionalsystemsinwhichreactionsand separation(throughamembrane)areintegratedwithinasinglevessel.
Membranescanbeusedtoeitherextractoneormorecomponentsfromthereactionsystemortofeedareactantintothereactionsystem.Inanycase,theintegrationofmembrane separationandreactionisusedtoenhancetheperformanceofthereactorintermsofconversion,selectivities,and/orenergyefficiency.
MRsareespeciallyinterestingforreactionsystemsthatarelimitedbythermodynamic equilibriumorwherethereactionsystemisacombinationofreactionsinseriesinwhichthe productcanbefurtherconvertedtoby-products.Inbothcases,byaddingamembrane,the systemcanenhancetheyieldoftheproducts.
MostoftheMRsintheliteratureareusedforequilibrium-limitedreactions.Shortly,ifat leastoneoftheproductsorthereactionisremovedfromthereactionsystem,theequilibriumisshiftedtowardtheproducts,thushigherconversionscanbeachievedunderthe sameoperatingconditions,orsimilarconversionsareachievedundermilderconditions. OtherMRsareusedtofeedareactantinthesystem,suchasinpartialoxidationreactionsor inbioreactorswhereairisfedthroughporousmembranes.
Inthisbook,atfirstdifferenttypesofMRsareshown.Indeed,dependingonthecatalyst system,therearepackedbedMRs(PBMRs),fluidizedbedMRs(FBMRs),microstructured MRs,orcatalyticMRs.
DifferenttypesofmembranesarealsousedinMRs:frommetallictoceramic,topolymericones.Forexample,mostbioreactorsusepolymeric(hollowfibers)orceramicmembranes.Inthesecases,someMRsusealsoparticlestoremoveordecreasethefoulingofthe membranes(tobeunderlinedthatthisisverydifficulttocompletelypreventinmembrane bioreactors).Forgas-phasereactionsinMRs,generallymetallicorceramicmembranereactorsareused,asthesereactionsoccurathightoveryhightemperatures.
Goingabitintodetails,thebookstartswithageneralintroductiontobothmembranes andinorganicMRs,giveninChapter1(Palma,Martino,Meloni,andBasile).Inthiswork, hydrogenisconsideredthemostpromisingenergycarrierforsustainableenergysystemsof thefuture.Thisisbecausetheproductionofhighpurityhydrogenisessentialforseveral applications,includingfeedingofprotonexchangemembranefuelcellstacks,whichisthe mostpromisingalternativetotheinternalcombustionenginesforseveraltransportation applications.However,theauthorsalsostressthathigh-gradehydrogenisdifficulttostore andtransportsuggestingthattheseissuesmaybesolvedthroughthegenerationofhydrogen utilizingMRssystems,whichhavegainedgreateffortsfromthescientificcommunity.Infact, inrecentyears,aconsistentstreamofstudiesaddressedinvestigatingthecombinationof hydrogenproductionandseparation.Tosummarize,inthischapter,briefdescriptionsof
boththegeneralprinciplesofmembraneseparationprocessesandthepossiblewaystocoupleacatalystandamembraneincatalyticMRsareprovided.Moreover,abriefoverviewof membranebioreactorsisalsogiven.Finally,futuretrendsandcurrentchallengesaboutMRs arediscussed.
Thenextchapter,Chapter2(Zhao,Zou,Huang,andTong),concentratesonaparticular typeofMRcalledprotonicelectrocatalyticmembranereactors(PEMFCs).Followingthe authors,theincreasingenergyandpowergenerationdemandmainlydependsontheconsumptionoffossilfuels,whichcontributestoclimatechange.Inthiscontext,PECMRsoffer promisingpotentialsforsustainableenergyconversionandstoragewithlowenergyconsumptionandlowemissionduetotheirhighprocessandenergyefficiencies.Manypractical applicationshavebeendemonstratedbasedonPECMRs,suchasammoniasynthesis,the reductionofCO2,thedehydrogenationofhydrocarbons,andotherenvironmentalapplications.Inthisinterestingscenario,thechaptersummarizesthemostrecentdevelopmentof PECMRsbysortingapplicationtypes,includingtheintroductionoftheoreticalprinciples,the progressofthematerialdevelopment,andpresentingchallengesandperspectives.
InChapter3(Gallucci),aparticularkindofMR,namedPBMR,isconsideredtheeasiest configurationstudiedinlaboratorysettingsfortheproof-of-conceptofMRs.Theauthor reportsafewexamplesofthesesystemsasappearinginthespecializedliteratureinthelast 5years(2018 22).Thechapteralsoillustratedafewofthelatestexamplesofthiskindof reactor.
ThesameauthorcontinuesthediscussioninthenextChapter4(Gallucci),withthe FBMRs,consideredanextensionoranimprovementofPBMRs.Infact,thefluidizedbed configurationallowsmoreuniformtemperatureduetothemovementofparticlesevenfor veryexothermicreactionsanddecreasedconcentrationpolarization.Also,inthiscase,variousexamplesofMRsappearinginliteratureinthelast5years(2018 22)arepresented.
Duringthelasttwodecades,anewkindofduringthelasttwodecadeshasattractedwide interestandundergonerapiddevelopment.Forthisreason,Chapter5(Gapp,Ansaloni, Venvik,Peters,andPfeifer)illustratestheimportanceofmicrostructuredMRsforprocess intensification.TheauthorsfocusonmicrostructuredMRsforprocessintensification employingmembraneswithacombinedgas-selectiveand/orcatalysisfunction.Firstly,the designandfabricationstrategiesofsuchdevicesareintroduced,thenfollowedbyvarious examplesofmicrostructuredMRsemployingpolymeric,andceramic.Andaswellmetallic membranesarealsogivenbyintroducingpossibleapplicationsofmicrostructuredMRs whichinvolvegas liquidandgas gasreactions.Thechapterendsbydiscussingthepotentialoutlookforthetechnology.
AnotherkindofMR,theso-calledpervaporationMRs(theacronymisPVMRs),isdiscussedindetailinChapter6(Santoro,Figoli,andGaliano).Shortly,alsoPVMRsrepresent anintegratedseparationsystemwhereachemical(orbiochemical)reactioniscoupledwith amembrane-basedseparationin,inthiscase,apervaporationunit.Thishybridprocess offersaseriesofadvantagesintermsofbothinvestmentcostsreductionandhigheroperationalperformance.TobeaddedthatPVMRsaretodayconsideredaconsolidatedtechnologyinesterificationreactions,butencompassalsocondensationreactions,acetylation
reactions,etherificationreactions,andbiochemicalreactions,whereaseriesofproductsare generatedfromamicrobialconversion.
Chapter7(Vital)introducesanddeeplyillustratesthepolymericMRs.Polymericmembranes,applicablein-lowtemperatureprocesses(below150 C),duetotheirhighversatility andvarietyintypesandproperties,areverysuitableandadvantageousforapplicationsin thefinechemistryfield,whencomparedwiththeirinorganiccounterparts.Inthischapter, anoverviewofpolymericMRsispresented,andmembranestructures,suchasdense, porous,symmetric,asymmetric,integral,composite,andmixedmatrices,untilthemembraneroleinthereactorisgiven.Techniquesformembranepreparation,suchasphase inversionorthemethodsusedtopreparemetalnanoparticlesloadedmixedmatrices,are reviewedindetail.MRs’ classificationaccordingtothemembranematerial'snature,therole ofthemembraneinthecatalyticprocess,thetransportfunctionofthemembrane,orthe reactorconfigurationiswidelyrevisited.Moreover,recentprogressonnewmembranetypes orMRs,suchasionicliquidmembranes,polymericmicroporousmembranes,orpolymeric membranemicroreactorsisreferredto.
Thecurrenttrendsinenzymaticmembranereactor(EMR)aredeeplydiscussedin Chapter8(Sitanggang,Diaussie,Rosabel,andBudijanto).Followingtheauthors,theterm EMR,usedforbothsingleormonophasicreactorsandmultiphasicreactors,isgenerally acceptedforanybiochemicalreactioncatalyzedbyacertainenzyme(s)orenzymeproducingcellsandcoupledwithmembraneseparation.EMRhasshowntheabilityto improvetheefficiencyofenzyme-catalyzedbioconversion,increaseproductyield,andiseasilyscaledupforindustrialpurposescomparedtoconventionalreactors.Especiallyinfood andpharmaceuticalapplications,EMRisusedmostlyfortheenzyme-catalyzedhydrolytic reactionstoimprovetheproduct’snutritionalandfunctionalproperties,thusincreasingtheir economicvalues.Inparticular,thischapterfocusesonthedesigns,membranecharacteristics,andapplicationsofEMRinassortedfieldstoproduceaproductinsingleandmultiphasesystems.
TheaspectsofMRsinbioartificialorgansaredeeplydiscussedinChapter9(Morelli, Salerno,Piscioneri,andDeBartolo),whereanoverviewoftheapplicationofmembranebioreactortechnologytoengineerbioartificialorgansthatcanbeusedasextracorporealdevices providingtemporarysupportforpatientswithorganfailurewaitingfortransplantationoras implantablesystemsisprovided.Thesedevicescanalsoofferaninvitroplatformfordrug toxicitytestingandstudies.Indetail,afterabriefintroductiononthecriticalissuesinthe designofamembranebioreactortobeusedasabioartificialorgan,asummaryofthetransportphenomenawithinthebioreactorbyusingcomputationalmodelingisreported,since theyhavetobeanalyzedtooptimizetheoveralloperationalconditions.Then,specialattentionisgiventothemembranebioreactordevicesusedasbioartificialliver,bioartificialpancreas,andbiomimeticmodelofthenervoussystem.Thecurrentstatusoftheirdevelopment ininvitroandinvivostudies,aswellasinclinicaltrialsperformedwithinthelastdecadesis alsodiscussed.
Chapter10(Molinari,Lavorato,andArgurio)regardsthephotocatalyticMRs. Heterogeneousphotocatalysisislargelystudiedinthefieldofenvironmentrecoverybythe
totaldegradationoforganicandinorganicpollutantsandforthesynthesisofchemicals.In thiscontext,thechapterdiscussesthebasicprinciplesofphotocatalysistogetherwithboth theadvantagesanddisadvantagesrelatedtoitscouplingwithamembraneseparationin photocatalyticMRs.Thetypesofmembranesusedandtheircriteriaofselectionarebriefly examined.Onthebaseoftheirconfigurationandmembraneoperation,photocatalyticMRs areusedinreactionsofphotodegradationofpollutantsandreactionsofsynthesis,evidencingastheappropriatechoiceoftheconfigurationisakeystepgivenlargescaleimplementation.Somecasestudiesinwatertreatment(i.e.pharmaceuticalsremoval)andthereaction ofsynthesis(CO2 reduction)arediscussed,evidencingpotentialities,drawbacks,andfuture trends.
InChapter11(Millet),anoverviewofsomeelectrochemicalMRsofgreatpracticalinterestforthechemicalindustryandtheenergytransitionisprovided.First,thedifferenttypes ofelectrochemicalreactorsarecategorizedfromgeneralthermochemicalconsiderations.In detail:thegeneralfeaturesofelectrochemicalmembranereactorsaresummarized,followed bythediaphragmsusedwithliquidelectrolytesandthenanion-conductingpolymerelectrolyteusedeitherdirectlyorsoakedinelectrolytesisdescribed.Moreover,solidoxidesare usedaselectrolytesandcellseparators;andafewselectedendergonicelectrochemicalprocessesofindustrialinterest(waterelectrolysis,brineelectrolysis,andelectrodialysis)are described.Variousprocessesaredescribedtoo,includinglimitationsofcurrentmaterials togetherwithsomeprospectiveissues.
AveryimportantaspectofMRsistheirmodeling,whichisdiscussedinChapter12 (Gallucci),whereafewgeneralmodelsthatcanbeusedformembranereactorsarereported andvariousexamplesofmodelresultsfromtheliteraturearealsogiven.Althoughthelistof modelsisnotexhaustive,mostoftheMRsreportedinthisbookcanbemodeledwithoneof themodelsproposedinthischapter.Ofcourse,closureequationsforkineticsandmembranefluxarepresentedfortheexamplesreportedinthischapterandshouldbechanged andverifiedbytheusersfortheirspecificproblems.
ThebooksendwithChapter13(Gallucci)withatechno-economicanalysisofMRs.As said,MRsareoftenusedastheadvancementofexistingtechnologiesbecause,byintegrating reactionandseparationinasinglevessel,theefficiencyofthesystems/processesincreases. However,therealindicatorthatshowsiftheMRisoutperformingaconventionalsystemis thefinalcostoftheproduct,whichcanbecalculatedbyusingatechno-economicanalysis. Inthischapter,afewexamplesoftechno-economicstudiesofmembranereactorsappearing inliteratureinthelast5yearsareshown.
Theeditorswouldliketotakethisopportunitytoexpresstheirsinceregratitudetoallthe contributorstothisbook,whoseexcellentsupportresultedinitssuccessfulcompletion.We aregratefultothemforthecommitmentandsinceritytheyhaveshowntowardstheircontributions.Withouttheirenthusiasmandsupport,thecompilationofthisbookwouldnothave beenpossible.Lastbutnotleast,wewouldalsoliketothankthepublisher,inparticularspecialthankstotheresponsibleatElsevier,IvyDawnC.Torre,SantosVeronica,AnitaKoch, NarmathaMohan,andKostasMarinakis,fortheirgreathelp.
Introductiontomembraneand membranereactors
VincenzoPalma1,MarcoMartino1,EugenioMeloni1,AngeloBasile2,3
1 DEPARTMENTOFINDUSTRIALENGINEERING, UNIVERSITYOFSALERNO,FISCIANO,ITALY
2 HYDROGENIA,GENOVA,ITALY 3 UNITOFCHEMICAL-PHYSICSFUNDAMENTALSINCHEMICAL ENGINEERING,DEPARTMENTOFENGINEERING,UNIVERSITYCAMPUSBIO-MEDICALOF ROME,ROME,ITALY
1.1Introductionandprinciples
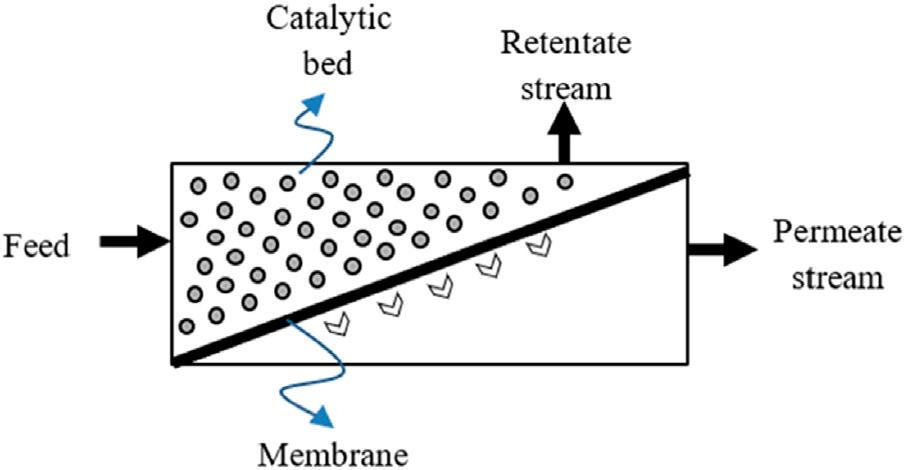
AccordingtotheInternationalUnionofPureandAppliedChemistryrecommendations,a membraneisa “structure,havinglateraldimensionsmuchgreaterthanitsthickness, throughwhichmasstransfermayoccurunderavarietyofdrivingforces” [1].Amembrane allowscontrollingthemasstransferbetweentwoadjacentfluidphasesbyactingasasieveto separatedifferentspeciesandcontrollingtherelativeratesoftransportacrossthemembrane [2].Throughthemembrane(Fig.1 1),afluidstream(retentatestream),isdepletedofsome ofitsoriginalcomponents,toformanotherfluidstream(permeatestream),whichisconcentratedinthesecomponents.Thedrivingforcethatregulatesthetransportprocessacrossthe membranetypicallydependsonagradientofconcentration,pressure,temperature,electric potential,etc. [3].
Themembranecanbeusedtocontinuouslyremovetheproductsandincreasetheconversion,thusovercomingtheequilibriumlimitations(LeChatelierprinciple),ortoincrease theselectivitybydistributivefeedingareactant [5].
Themembranescanbeclassifiedaccordingtotheir nature(biologicalorsynthetic),geometry, andseparationregime,aswellasorganic,inorganic,ororganic/inorganichybrids [5].
Biologicalmembranescanbeeasilyfabricated;however,theyhavemanylimitations, includinglowtemperaturesofuse,tolerancetoalimitedpHrange,andsusceptibilityto microbialattack [5].Syntheticmembranescanbeorganicorinorganicinnature;theorganic membranesarepolymericmaterials [6],suchaspolyamideorpolystyreneandinsomecases canbeusedupto300 C,whiletheinorganicmembranescanbeceramic,suchaszeolitesor oxides,ormetallic,suchaspalladiumormetalalloys,andshowhighstabilityinawiderange oftemperatures(upto1000 C,insomecases)andtolerancetoabroadpHrange [7]. Inorganicmembranescanalsobeclassifiedbasedonporediameter(dp)sizes,microporous (dp , 2nm),mesoporous(2nm , dp . 50nm),andmacroporous(dp . 50nm),and
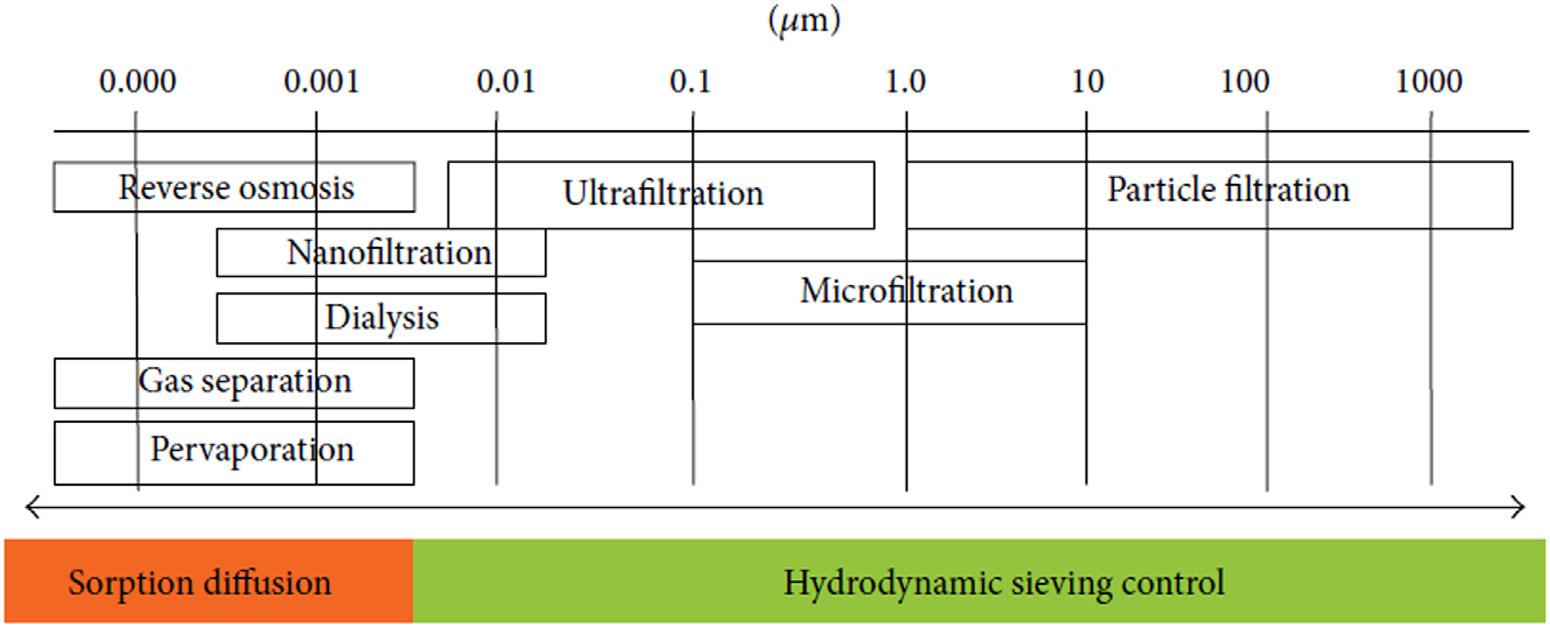
currentmembraneprocessesincludemicrofiltration,ultrafiltration,nanofiltration,gasand vaporseparation,andpervaporation(Fig.1 2).
Thelattermentionedprocessinthelasttwodecadesisfindinganincreasingapplication inliquidhydrocarbonsseparations(petrochemicalapplication,alcohol/etherseparations), removalofvolatileorganiccompoundsfromwater,removalofwaterfromglycerin,and dehydrationtointensifyesterificationreaction [8].Moreover,themetallicmembranescanbe classifiedassupportedandunsupported [5].Thesupporteddensemembranes [9],obtained bydepositionofmetalliclayersonaporoussupport,suchasalumina,silica,carbon,orzeolite,areparticularlyinterestinginthefieldofhydrogenproduction.
Themechanismsthatregulatethemasstransportthroughporousanddensemembranes areverydifferent;inporousmembranes,themechanismdependsontheporosity,whilein densemetallicmembranesasolution-diffusionmechanismiseffective [5].
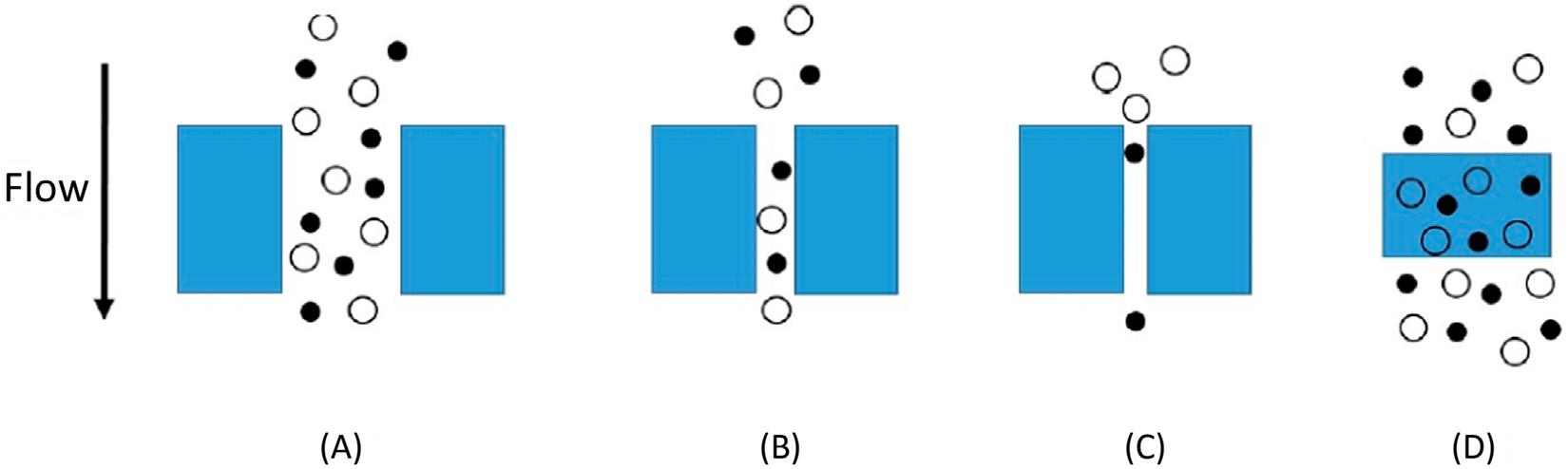
Thegastransportmechanismsthroughaporousmembraneincludemolecularsieving, Knudsendiffusion,capillarycondensation,andlaminarflow(e.g.,Poiseuilleflow),dependingonthemembraneporesizeanddiameterofgasmolecules [10].Differentmechanismsof gastransportthroughmembranesareshownschematicallyin Fig.1 3.
FIGURE1–2 Thechoiceofmembranewithrespecttothesizeofparticles [8].
FIGURE1–1 Conceptualschemeofamembranereactorsystem [4]
–3 Diffusionmechanisms:(A)bulkflowthroughpores;(B)Knudsendiffusionthroughpores;(C)molecular sieving;(D)solutiondiffusionthroughdensemembranes [10]
Thepermeability(P)isacharacteristicpropertyofthemembrane,andinthecaseofdense membranes,itisproportionaltosolubility(S)anddiffusivity(D),accordingtothe Eq.(1.1)[11].
Thesolubilityisrelatedtotheaffinitybetweenthegasmoleculesandthemembranes materials,thediffusivitytothefreevolume,andthesizeofgases.Theperm-selectivity(α) dependsontheoperativeconditions,includingtemperatureandpressure,andhasbeen definedastheratiobetweenthepermeabilityoftwogases [12],accordingtothe Eq.(1.2)
Inthecaseofdensepolymermembranes,theperm-selectivitycanbealsoinfluencedbythe plasticizationphenomenon [13],duetothephysicaldissolutionofthepenetrantgasinthepolymermatrix,whichinducesanincreaseinthesegmentalmobilityofthepolymerchains [14].
Thegaspermeance(Pe)dependsonthegaspermeabilityandthemembranethickness (δ)accordingtothe Eq.(1.3)[11].
Theefficiencyoftheseparationprocessisdefinedastheseparationfactor(SF),whichis relatedtothemolarfractionsofthecomponentsinthepermeate(Xi,p, Xj,p)andfeedstream (Xi,f, Xj,f),accordingtothe Eq.(1.4)[11]
Asmentionedaboveinthecaseofporousmembranes,themechanismsdependonthe sizeofthepores.Whenthemeanporediameterislargerthanthemeanfreepathofthefluid
FIGURE1
molecules(macroporousmaterials)thePoiseuilleflow(viscousflow)isoperating(Fig.1 4), thecollisionbetweenthemoleculesismoreprobablethanthecollisionbetweenthemoleculesandtheporewalls [15].
Inthecaseofmesoporousmaterials,themoleculestendtocollidemorewiththepore wallsthanamongthemselves [16] (Fig.1 4).Whenthemeanporediameterandthemean freepathofthefluidmoleculesaresimilar,theKnudsenmechanismoccurs,andtheflow throughthemembraneiscalculatedaccordingto Eq.(1.5)[17],where G isthegeometrical factorrelatedtothemembraneporosityandporetortuosity.
–4 Masstransportmechanisminsideporousmaterialsandtheirperm-selectivity [4]
FIGURE1
InthecaseofKnudsendiffusion,unlikethePoiseuilleflow,theflowdoesnotdependon absolutepressure.Inthiscase,thehighestseparationfactorobtainableforabinarymixture, whenthevacuumisimposedonthepermeateside,isequaltothesquarerootoftheratio betweenthemolecularweightsofthetwodifferentmolecules,thusthesmallermolecules aretransferredmoreintensivelyacrossthemembrane [15].Whenthemoleculesarephysisorbedorchemisorbontheporewalls,surfacediffusionoccurs [17],andselectivetransport takesplace,however,theadsorbedmoleculesreducetheporesize,hinderingthefurther transferring [15].Capillarycondensationtakesplacewhenoneofthecomponentscondenses withinthepores,asaresultofcapillaryforces,thecondensatefillstheporesandthenevaporatesatthepermeatesidewherelowpressureiskept [15].Multi-layerdiffusionisconsidered anintermediateflowregimebetweensurfacediffusionandcapillarycondensation [5,18].In thecaseofmicroporousmaterials,themechanismiscomparabletoamolecularsieve,only smallmoleculescanpermeate,makingitpossibletoachieveveryhighselectivity [4].Ithas beendemonstratedthatthepermeatingflowthroughthemicroporousmaterialsincreases withthetemperature [19],accordingto Eq.(1.6).
where Eact istheapparentactivationenergy,rangingfrom2to40kJ/mol,dependingon microporesizeandgasmoleculesize.Moreover,DeLangeetal. [20] describedthegastransportandseparationinmicroporousmembranematerials,thustheactivatedtransportmay beexpressedaccordingtothe Eq.(1.7)
where Do (m2/s)isthemeanintrinsicdiffusioncoefficientformicroporediffusion,and ko is theintrinsicHenryconstant,themembraneporosity, l themembranethickness, ρ thebulk density, qst theisostericheatadsorption, Ei theactivationenergyforgasspecies, R theuniversalgasconstant,and T thetemperature [4].
Pervaporationisacombinationofpermeationandevaporation,whichconsistoftheseparationofliquidmixtures(feedstream)bypartialvaporizationthroughadensemembrane, thereforeitisbasedonaliquid-vaporphasechange [16],andthepermeatestreamisrecoveredasvapor.Theprocessconsistsofseveralphases,theliquidfeedisheateduptothe operatingtemperature,thensenttotheactivesideofthemembrane,wheretheseparation occurs,andfinally,thepermeatevaporiscontinuouslyremovedfromtheothersideofthe membrane.Thecontinuousremovalofthepermeatevaporgeneratesaconcentrationgradientacrossthemembranewhichactsasthedrivingforceoftheprocess [21].Themasstransferofaspecieacrossthemembranecanbeexplainedthroughasolution-desorptionmodel [16],whichconsistsofthreemainsteps:sorptionofthepermeatingspeciesatthefeedside, transportacrossthemembraneaccordingtotheFick’slaw,anddesorptionatthepermeate sideundervacuum [21] orasweepgas [16] (Fig.1 5A).
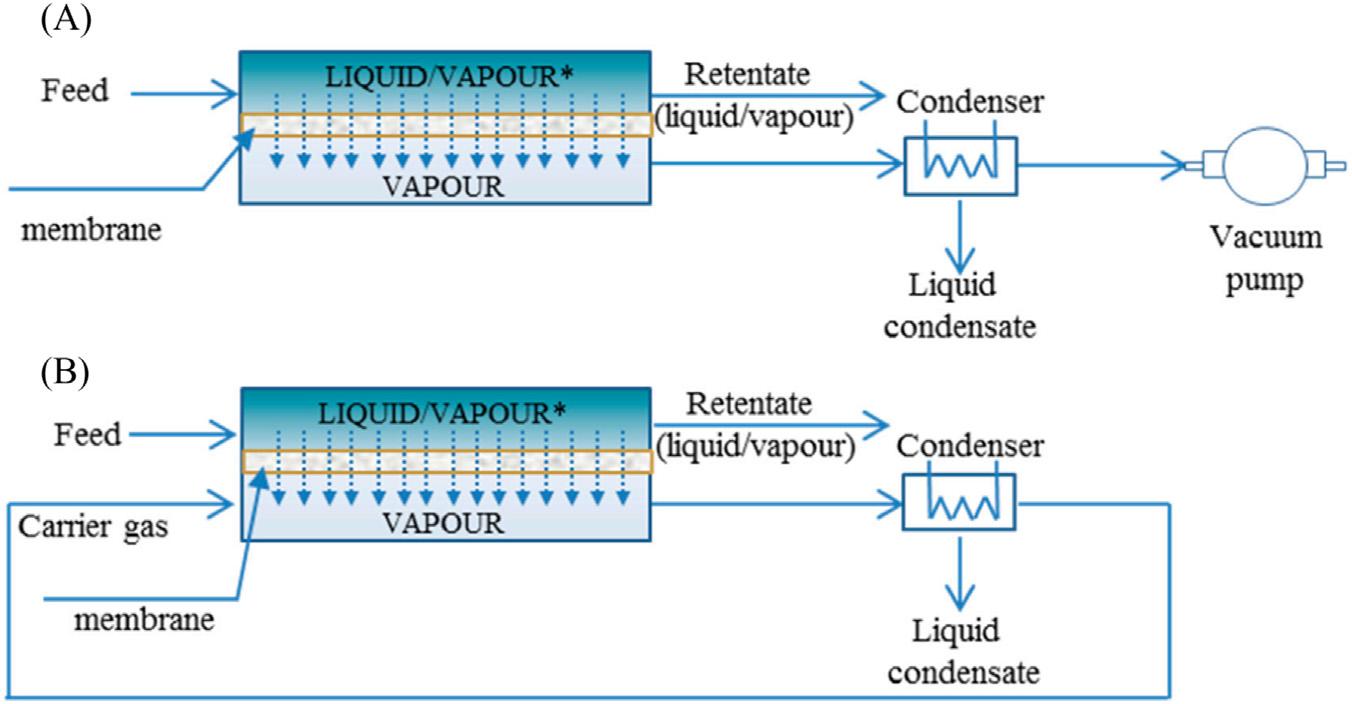
FIGURE1–5 Schematicrepresentationofpervaporationorvaporpermeationprocess:(A)byvacuum;(B)bya carriergas. Forpervaporation,thefeedisliquid;forvaporpermeation,thefeedisvapor [16]
Inadditiontomasstransfer,thechangeinthephysicalstateofthepermeatecomponent impliesalsoheattransfer;thedecreasingofthetemperaturebringstothedecreaseofthe partialpressureandconsequentlytothedrivingforceofthemasstransfer,sothatsometimes,atindustrialscale,upstreamheatexchangeisusedtocompensate [16].
Unlikegases,theadsorptionfromtheliquidfeedisalmostindependentofthepressure, thusthedrivingforcefortheadsorptiondependsonthefugacityofthespecies(Eq.1.8) [22].
where xi isthefeedmolefraction, ɣi istheactivitycoefficientand pi isthesaturatedvapor pressure.Thefugacityincreaseswiththetemperature,moreover,inmixtures,theadsorption selectivityseemstoincreasewiththeincreaseofthefeedfugacityratio [22].
Contrarytopervaporation,invaporpermeationthefeedisamixtureofvaporandgases,thereforetherehappensnophasechangeandconsequently,anon-heatsupplyisnecessary.Inthis case,thedrivingforceisthechemicalpotentialgradientduetothedifferenceinthepartialpressure throughthemembranes,thustheseparationisregulatedbyasolution diffusionmechanism [16].
Polymericandzeolitemembranesareusedinpervaporationandvaporpermeationprocesses;whilethemostcommonapplicationsarewaterremovalfromorganicsandviceversa,separationoforganicmixtures,andconcentrationofaqueoussolutions.Themain advantageoftheseprocessesresidesinthepossibilitytoeasilyseparateazeotropicmixtures ormixturesforwhichahighnumberoftheoreticalstagesisrequired [21].
1.2Membranes
Aspreviouslymentioned,membranescanbeclassifiedaccordingtothematerials,andthe choiceofthetypeofmembranedependsonthetypeofprocessinwhichtheyaretobeused.
Polymericmembranesareparticularlyinterestingduetotheirlowcost,however,canbe usedonlyatlowtemperatures,upto150 C [6].Theseparationprocessdependsonseveral parameters;however,thecharacteristicsofthepolymerplayacrucialrole.Thepolymers usedasmembranescanberubberorglassy,theformerisusuallyhighpermeableforgases butlowselective,onthecontrary,thelatterisselectivebutlowerpermeable.Themainphysicochemicalfactorsinfluencingthegaspermeabilityandpermselectivityofthepolymeric membranesarethefreevolumeofthepolymer,thechainsmobility,andthesolubilityofthe gasinthepolymer [6].Thechainrigidityfavorsthepermselectivitybutisdisadvantageous forthepermeability,asinthecaseofglassypolymers,however,toimprovethepermeability anincreaseinthefreevolumecanbebeneficial.Ontheotherhand,thehighflexibilityof thepolymerchainintherubberypolymersallowsthehighpermeabilityandhighselectivity. Glassypolymermembranesprovidehighmechanicalresistanceandgoodreproducibility; however,theysufferfrompoortemperatureresistance,surfacecorrosion,andswellingeffect duetoplasticizationphenomena.Themostusedglassypolymersarepolysulfone,polyethersulfone,polyetherimide,andpolyimide [11].Rubberypolymersgenerallyshowahighaffinityfor CO2 gasmolecules;themostusedpolymersarepoly(ethyleneoxide)-basedpolymers,poly (amide-6-b-ethyleneoxide),poly(dimethylsiloxane),andpolyvinylamine [11].Reallyattractive arepolymersfromnaturalsources,theso-calledbio-polymers,whicharemostlysynthesizedby livingorganisms,andarebiodegradable,compostable,andenvironmentallysustainable [11]. Themostinterestingexamplesare:
• Celluloseacetate,aglassypolymercommercializedinthe1980s,isusedforCO2 separation.
• Thermoplasticstarch,obtainedfromplants,includingpotatoes,corn,etc.,isusedfor packagingapplications.
• Cross-linkedchitosan,obtainedbydeacetylationofchitin,isusedinwatertreatment processes.
• Polylacticacid,isusedinpackagingapplicationsandtissueengineering.
• Polyhydroxyalkanoates,obtainedbymicrobialfermentation,arecharacterizedby hydrophobicity,opticalpurity,andhighprocessability.
• Polyvinylalchol,ischaracterizedbyhighhydrophilicityandgoodbarrierproperties.
• Polyurethane,isobtainablefromplantoil,suchascastororsoybeanoil.PUspossess goodphysicalandtensilestrength,chemicalresistance,andmechanicalproperties.
Inorganicmembranesconsistofmetals,oxidescarbon,orelementarycarbon,theyare highlyselectiveandpermeableandcanoperateinsevereoperativeconditions [23]. Althoughmoreexpensive,inorganicmembranespresentseveraladvantagescomparedto polymericones:awell-definedstableporestructure,highmechanicalstability,andthermal andsolventresistance [5].Themaincategoriesofinorganicmembranesare:
• Metalmembranesarecategorizedasdenseorporous;mostarecharacterizedbya gradientcompositestructureofthemetal,metaloxide,ormetalalloy,themostused metalsarePd,Ag,theiralloys,andsteel.Theunsupportedmembranesaremadewith
puremetal,whilethesupportedmembranesaremadebycoatingwithametalormetal oxideontheprimarystructurewhichisaporousmetal [24].Metalmembranesareused forgasseparation,andfood,drug,andbeverageapplications.
• Ceramicmembranesarebasedonalumina,silica,ortitaniumoxide;theyareinertand stableathightemperaturesandpossesshighpermeabilityandmoderateselectivity.They areparticularlysuitableforfood,biotechnology,andpharmaceuticalapplications [5], silicaandsilica,andfunctionalizedceramicmembranesareusedforhydrogen productionandseparation [25].Amongthenegativeaspectsrelatedtotheiruse,thereare membranesealingproblemsinhigh-temperaturemodules,crackingproblemsrelatedto temperaturegradients,andlowchemicalstabilityofperovskite-typematerials [5].
• Zeolitemembranesarebasedonmicroporouscrystallinealuminasilicate,usually obtainedbydirectgrowthonaporousceramicormetalsupport.Thesynthesisisusually carriedoutbyhydrothermaltreatmentsinthepresenceofanorganicstructure-directing agent,whichallowsforcontrollingtheshapeandsizeofthepores [26].Themolecular sievingaction,thelargesurfacearea,andthecontrollableinteractionhost-sorbateare consideredthemainadvantagesofusingzeolitemembranes,moreover,thepossibilityto combinecatalyticactivityandseparationcapabilityisanattractivefeature.Zeolite membranesareusedascatalystsandsensorsfortheseparationofgasand/orliquids [27] Themaindrawbacksofusingzeolitemembranesarethelowgasfluxcomparedtothe otherinorganicmembranesandthelowthermalstabilityofthezeolitelayer,whichcan expandwiththetemperature [5].
• Carbonmembranesarecomposedofmicroporous,amorphoushigh-carbonmaterials, canbeproducedbythermaltreatmentofawidevarietyofpolymerprecursors,andcan besupported(flatortube)andunsupported(flat,capillary,orhollowfiber).Thehollow fiberpresentahighseparationperformance,highpackingdensity,andlowcost, however,thebrittlenessmakesitdifficulttohandle,sosupportedcarbonmembranesare preferable [28].Thesupportedmembranesarefabricatedbythecarbonizationofa polymericprecursorlayeronresistantsupport.Carbonmembranescanbeusedingas separationforCO2,N2,andH2 removal,however,theselectivitystronglydependsonthe precursorusedforthefabrication.Althoughtheuseofcarbonmembranesisvery promising,itstillappearstobeimmature;theproblemsoffragilityandtheoptimization ofpreparationmethodsconstitutealimittotheiruse.
1.3Membranebioreactors
Amembranebioreactor(MBR)canbedefinedasaspaceinwhichabiochemicaltransformationandamembraneseparationprocessoccur [29].InMBR,themembranecanbe usedfordifferentpurposes,suchasaddingareactantorforselectivelyremovingoneofthe reactionproducts [30].Moreover,membranescanbeutilizedtoretainthebiocatalystoract asthesupportforthebiocatalyst,ortheseparationofenzymesbysizeexclusion [31].MBR processesarecharacterizedbyseveraladvantages,includingsmallfootprint,lowersludge