https://ebookmass.com/product/bioactive-food-componentsactivity-in-mechanistic-approach-1st-edition-cinthia-bau-
Instant digital products (PDF, ePub, MOBI) ready for you
Download now and discover formats that fit your needs...
Nanoencapsulation of Food Bioactive Ingredients: Principles and Applications 1st Edition Jafari
https://ebookmass.com/product/nanoencapsulation-of-food-bioactiveingredients-principles-and-applications-1st-edition-jafari/
ebookmass.com
Pericyclic reactions : a mechanistic and problem solving approach 1st Edition Kumar
https://ebookmass.com/product/pericyclic-reactions-a-mechanistic-andproblem-solving-approach-1st-edition-kumar/
ebookmass.com
Bioactive seaweeds for food applications : natural ingredients for healthy diets 1st Edition Yimin Qin
https://ebookmass.com/product/bioactive-seaweeds-for-foodapplications-natural-ingredients-for-healthy-diets-1st-edition-yiminqin/ ebookmass.com
Orban’s Oral Histology and Embryology 14/E Edition G. S. Kumar
https://ebookmass.com/product/orbans-oral-histology-andembryology-14-e-edition-g-s-kumar/
ebookmass.com
Leopard's Hunt Christine Feehan https://ebookmass.com/product/leopards-hunt-christine-feehan/
ebookmass.com
The Burnout Fix: Overcome Overwhelm, Beat Busy, and Sustain Success in the New World of Work Jacinta M. Jiménez
https://ebookmass.com/product/the-burnout-fix-overcome-overwhelm-beatbusy-and-sustain-success-in-the-new-world-of-work-jacinta-m-jimenez-3/
ebookmass.com
The epidemiology and pathogenesis of coronavirus disease (COVID-19) outbreak Hussin A. Rothan
https://ebookmass.com/product/the-epidemiology-and-pathogenesis-ofcoronavirus-disease-covid-19-outbreak-hussin-a-rothan/
ebookmass.com
Environmental Pollution in China: What Everyone Needs to Know® Daniel K. Gardner
https://ebookmass.com/product/environmental-pollution-in-china-whateveryone-needs-to-know-daniel-k-gardner/
ebookmass.com
Modern Data Architecture on Azure: Design Data-centric Solutions on Microsoft Azure 1st Edition Sagar Lad
https://ebookmass.com/product/modern-data-architecture-on-azuredesign-data-centric-solutions-on-microsoft-azure-1st-edition-sagarlad/
ebookmass.com
Let It Go: A Why Choose Story (Brain Storm Series Book 1)
https://ebookmass.com/product/let-it-go-a-why-choose-story-brainstorm-series-book-1-jonas/
ebookmass.com
BioactiveFoodComponentsActivityin MechanisticApproach Thispageintentionallyleftblank
BioactiveFoodComponents ActivityinMechanistic Approach Editedby
CinthiaBau´BetimCazarin
FoodScienceandNutritionDepartment,SchoolofFoodEngineering,UniversityofCampinas, Campinas,Brazil
JulianoLemosBicas
FoodScienceandNutritionDepartment,SchoolofFoodEngineering,UniversityofCampinas, Campinas,Brazil
GlauciaMariaPastore
FoodScienceandNutritionDepartment,SchoolofFoodEngineering,UniversityofCampinas, Campinas,Brazil
MarioRobertoMarosticaJunior
FoodScienceandNutritionDepartment,SchoolofFoodEngineering,UniversityofCampinas, Campinas,Brazil
AcademicPressisanimprintofElsevier 125LondonWall,LondonEC2Y5AS,UnitedKingdom 525BStreet,Suite1650,SanDiego,CA92101,UnitedStates 50HampshireStreet,5thFloor,Cambridge,MA02139,UnitedStates TheBoulevard,LangfordLane,Kidlington,OxfordOX51GB,UnitedKingdom
Copyright©2022ElsevierInc.Allrightsreserved.
Nopartofthispublicationmaybereproducedortransmittedinanyformorbyanymeans,electronicormechanical,including photocopying,recording,oranyinformationstorageandretrievalsystem,withoutpermissioninwritingfromthepublisher. Detailsonhowtoseekpermission,furtherinformationaboutthePublisher’spermissionspoliciesandourarrangementswith organizationssuchastheCopyrightClearanceCenterandtheCopyrightLicensingAgency,canbefoundatourwebsite: www. elsevier.com/permissions .
ThisbookandtheindividualcontributionscontainedinitareprotectedundercopyrightbythePublisher(otherthanasmaybe notedherein).
Notices
Knowledgeandbestpracticeinthisfieldareconstantlychanging.Asnewresearchandexperiencebroadenourunderstanding, changesinresearchmethods,professionalpractices,ormedicaltreatmentmaybecomenecessary.
Practitionersandresearchersmustalwaysrelyontheirownexperienceandknowledgeinevaluatingandusinganyinformation, methods,compounds,orexperimentsdescribedherein.Inusingsuchinformationormethodstheyshouldbemindfuloftheir ownsafetyandthesafetyofothers,includingpartiesforwhomtheyhaveaprofessionalresponsibility.
Tothefullestextentofthelaw,neitherthePublishernortheauthors,contributors,oreditors,assumeanyliabilityforanyinjury and/ordamagetopersonsorpropertyasamatterofproductsliability,negligenceorotherwise,orfromanyuseoroperationof anymethods,products,instructions,orideascontainedinthematerialherein.
BritishLibraryCataloguing-in-PublicationData
AcataloguerecordforthisbookisavailablefromtheBritishLibrary LibraryofCongressCataloging-in-PublicationData
AcatalogrecordforthisbookisavailablefromtheLibraryofCongress
ISBN:978-0-12-823569-0
ForInformationonallAcademicPresspublications visitourwebsiteat https://www.elsevier.com/books-and-journals
Publisher: NikkyP.Levy
AcquisitionsEditor: MeganR.Ball
EditorialProjectManager: HilaryCarr
ProductionProjectManager: KumarAnbazhagan
CoverDesigner: MatthewLimbert
TypesetbyMPSLimited,Chennai,India
Contents Listofcontributorsix 1.Introduction1
CinthiaBau ´ BetimCazarin,JulianoLemosBicas, GlauciaMariaPastore andMarioRobertoMarosticaJunior
1.1Introduction1
1.2Chronicdiseasesintheworldandthe causalroleoftheenvironmentalfactors1
1.2.1Foodsasatoolforpreventing chronicdiseasesanddisabilities2 References3
2.Mainmolecularmechanismsfor noncommunicablediseases5
MarinaVilarGeraldi,PauloSe ´ rgioLoubetFilho, AndressaMaraBaseggio,CinthiaBau ´ BetimCazarin andMarioRobertoMarosticaJunior
2.1Introduction5
2.2Mainmolecularmechanismsfor noncommunicablediseases6
2.2.1Inflammatoryresponseand oxidativestress6
2.2.2Obesityandmetabolicdysfunctions9
2.2.3Cancer14
2.2.4Inflammationand cancerdevelopment14
2.2.5Cognitionandneurodegenerative diseases17
2.3Conclusion19 Conflictofintereststatement19 Acknowledgments19 References19
3.Phenoliccompounds27
Marı´a delCarmenVillegas-Aguilar, A ´ lvaroFerna ´ ndez-Ochoa, FranciscoJavierLeyva-Jime ´ nez, A ´ ngelaMiranda-Segura,Marı´adelaLuzCa ´ diz-Gurrea andAntonioSegura-Carretero
3.1Introduction27
3.2Invitroevidenceoffoodphenolic compoundbioactivityandtheir mechanismsofaction33
3.3Antioxidantactivity33
3.4Antiinflammatoryactivity34
3.5Activitiesagainstchronicdiseases35
3.6Invivoevidencesoffoodphenolic compoundbioactivities36
3.7Neuroprotectionpotential36
3.8Antidiabeticpotential38
3.9Cardioprotectivepotential38
3.10Antiobesitypotential39
3.11Anticancerpotential39
3.12Clinicaltrials40
3.13Bioavailabilityandmetabolismstudies40
3.14Longitudinaldietaryassays42
3.15Conclusion45 References45
4.Prebioticsandprobiotics55
HenriqueSilvanoArruda,MarinaVilarGeraldi, MarinaFelixCedran,JulianoLemosBicas, MarioRobertoMarosticaJunior andGlauciaMariaPastore
4.1Introduction55 4.2Prebiotics56
4.2.1Mainconceptsanddefinitions ofprebiotics56
4.2.2Mainprebioticclasses57
4.2.3Otherprebioticcandidates81
4.2.4Concludingremarks83
4.3Probiotics84
4.3.1Mainconceptsanddefinitionsof probiotics84
4.3.2Maintraditionalstrains85
4.3.3Otherprobioticsandpotentialnew strains87
4.3.4Concludingremarks91
4.4Clinicalevidenceeproposedmechanisms ofactionofprebioticsandprobiotics91
4.4.1Gutmicrobiotainteraction92
4.4.2Immunesystemmodulation99 4.4.3Intestinalbarrierfunction100
4.4.4Productionoforganicacids101
4.4.5Othermechanisms101
4.5Conclusion102
Acknowledgments102 Conflictofintereststatement102 References102
5.Carotenoids119
AdrieleHacke,DanieleBobrowskiRodrigues, CinthiaBau ´ BetimCazarin andLilianReginaBarrosMariutti
5.1Introduction119
5.2Animalstudies120
5.3Humanstudies123
5.4Mechanisticstudies129
5.5Conclusion132 References133
6.Tocopherolsandtocotrienols: theessentialvitaminE139
PritiJain,InderbirSingh,SanjayJ.Surana andAtulA.Shirkhedkar
6.1Introduction139
6.2Chemistryoftocopherolsandtocotrienols139
6.3Sourcesoftocopherolandtocotrienol140
6.3.1Edibleoils140
6.3.2Underutilizededibleoilsasa sourceoftocols141
6.4Roleoftocopherolandtocotrienolin plantsandinhumanhealth142
6.4.1Roleinplants142
6.4.2Roleinhumanhealth143
6.5Conclusion151
Acknowledgment151 References151
7.Bioactivepeptidesagainstinflammatory intestinaldisordersandobesity155
BlancaHernandez-Ledesma, SamuelFernandez-Tome andLourdesAmigo
7.1Definingtheconcept“bioactivepeptides”155
7.2Bioactivepeptidesagainstinflammatory intestinaldiseases155
7.2.1Invitroantiinflammatoryeffectsof food-derivedpeptides156
7.2.2Invivoeffectsoffood-derived peptides162
7.3Bioactivepeptidesagainstobesity163
7.3.1Invitroeffectsofantiobesity foodpeptides163
7.3.2Invivoevidenceofantiobesity food-derivedpeptides170 7.4Concludingremarks170 References177
8.Monounsaturatedandpolyunsaturated fattyacids:structure,foodsources, biologicalfunctions,andtheir preventiveroleagainst noncommunicablediseases185
MiguelA ´ ngelRinco´n-Cervera, RobertoBravo-Sagua, RosanaAparecidaMano´lioSoaresFreitas, SandraLo´pez-AranaandAdrianoCostade Camargo
8.1Generaltopicsonmonounsaturatedand polyunsaturatedfattyacids185
8.1.1Chemistryandclassificationof fattyacids185
8.1.2Dietarysourcesandsupplements containingmonounsaturatedand polyunstaturatedfattyacids186
8.1.3Absorptionofdietaryfattyacids187
8.1.4Polyunsaturatedfattyacidfromthe n-3andn-6familiesasprecursors oflipidmediatorstomodulate inflammationprocesses188
8.1.5Biosynthesisofessential unsaturatedfattyacids190
8.1.6Monounsaturatedandpolyunsaturated fattyaciddegradation192
8.2Prospectingthepotentialhealthbenefits monounsaturatedandpolyunsaturated fattyacidsincellmodelsystems192
8.2.1Oxidativestress193
8.3Monounsaturatedandpolyunsaturated fattyacidseffectsinanimalmodels196
8.3.1Polyunsaturatedfattyacidand oxidativestressinanimalstudies196
8.3.2Inflammation197
8.3.3Fattyacidsandcardiovascular diseases198
8.4Roleoffattyacidsfortheprimaryand secondarypreventionofcardiovascular disease199
8.4.1Cardiovasculardisease199
8.4.2Relationshipbetweendietaryfatty acidsandcardiovasculardisease199
8.4.3Saturatedfatsandcardiovascular disease199
8.4.4Monounsaturatedfattyacidand cardiovasculardisease200
8.4.5 ω-3polyunsaturatedfattyacidand cardiovasculardisease200
8.4.6 ω-6polyunsaturatedfattyacidand cardiovasculardisease201
8.4.7Polyunsaturatedfattyacidandother healtheffects202
8.5Finalconsiderationsandfuturedirections202 Acknowledgments203 References203
9.Sulfurcompounds211 MohammedSaniJaafaru andAhmadFaizalAbdullRazis
9.1Introduction211
9.2Chemicaldefinitionofglucosinolates andtheirhydrolyticproducts211
9.3Sourcesofglucosinolates212
9.4Roleofisothiocyanatesin neuroprotection212
9.5Invitroandinvivoneuroprotective activitiesofisothiocyanateandtheir mechanisticpathway(s)213
9.5.1Sulforaphane213
9.5.2Phenethyl-isothiocyanate214
9.5.3Erucin214
9.5.46-(Methyl-sulfinyl)hexylisothiocyanate215
9.5.5Glucomoringin-isothiocyanate215
9.6Majorpathwaysinvolvedin neuroprotection/neurotoxicity215
9.6.1Nrf2/AREsignalingpathway215
9.6.2IκB/NF-κBsignalingpathway216
9.6.3Intrinsicmitochondrialsignaling pathway217
9.7Isothiocyanatesasantioxidantand antiinflammatoryagent217
9.8Isothiocyanatesasanticancerand chemopreventiveagent218
9.9Isothiocyanatesasantidiabeticagent219
9.10Conclusion219 References219
10.Phytosterols:physiologicalfunctions andtherapeuticapplications223
Suryamani,RakeshSindhuandInderbirSingh 10.1Introduction223 10.1.1Structure223 10.1.2Sourcesofphytosterols224 10.1.3Physiologicalfunctionsof phytosterols225
10.1.4Phytosterolcontentsinfoods227 10.1.5Absorptionofphytosterols228 10.1.6Distributionandexcretionof phytosterols228
10.2Majormodeofactionofphytosterols229 10.2.1Alterationofmembrane composition229
10.2.2Alterationofmembraneintegrity andmembraneboundenzymes229 10.2.3Effectonsignaltransduction pathway229
10.2.4Effectofonimmunesystem230 10.2.5Effectoncellcycle231 10.2.6Therapeuticactions231 10.2.7Phytosterolsandcholesterol231 10.2.8Phytosterolsasimmunomodulators andantiinflammatoryagents232
10.2.9Anticancerrolesofphytosterols232 10.3Modelsofcoloncancer233 10.4Modelsofbreastcancer233 10.5Modelsofprostatecancer234 10.5.1Antidiabeticrolesofphytosterols234 10.5.2Mechanismsofhypocholesterolemia activityandotheractivities234 10.5.3Safety235
10.5.4Phytosterolformulation235 10.6Conclusion236 References236 Index239
Thispageintentionallyleftblank
Listofcontributors LourdesAmigo DepartmentofBioactivityandFood Analysis,InstituteofFoodScienceResearch(CIAL, CSIC-UAM,CEIUAM+CSIC),Madrid,Spain
HenriqueSilvanoArruda FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
AndressaMaraBaseggio FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
JulianoLemosBicas FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
RobertoBravo-Sagua InstituteofNutritionandFood Technology,UniversityofChile,Santiago,Chile; AdvancedCenterforChronicDiseases(ACCDis), FacultyofChemicalandPharmaceuticalSciences, UniversityofChile,Santiago,Chile;AgeingResearch Network,ChileStateUniversityConsortium, Santiago,Chile
Marı´adelaLuzCa ´ diz-Gurrea Departmentof AnalyticalChemistry,UniversityofGranada,Spain
CinthiaBau ´ BetimCazarin FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
MarinaFelixCedran FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
AdrianoCostadeCamargo InstituteofNutritionand FoodTechnology,UniversityofChile,Santiago,Chile
A ´ lvaroFerna ´ ndez-Ochoa DepartmentofAnalytical Chemistry,UniversityofGranada,Spain;Berlin InstituteofHealthMetabolomicsPlatform,Berlin, Germany;MaxDelbru ¨ ckCenterforMolecular MedicineintheHelmholtzAssociation,Berlin, Germany
SamuelFerna ´ ndez-Tome ´ HealthResearchInstituteofthe UniversityHospitalLaPrincesa(IIS-IP),CIBERof HepaticandDigestiveDiseases(CIBEREHD), AutonomousUniversityofMadrid(UAM),Madrid,Spain
MarinaVilarGeraldi FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
AdrieleHacke FoodScienceandNutritionDepartment, SchoolofFoodEngineering,UniversityofCampinas, Campinas,Brazil
BlancaHerna ´ ndez-Ledesma DepartmentofBioactivity andFoodAnalysis,InstituteofFoodScienceResearch (CIAL,CSIC-UAM,CEIUAM+CSIC),Madrid,Spain
MohammedSaniJaafaru DepartmentofBiochemistry, KadunaStateUniversity,Kaduna,Nigeria
PritiJain SchoolofPharmaceuticalSciences, DepartmentofPharmaceuticalChemistry,Delhi PharmaceuticalSciencesandResearchUniversity (DPSRU),NewDelhi,India
FranciscoJavierLeyva-Jime ´ nez Departmentof AnalyticalChemistry,UniversityofGranada,Spain; ResearchandDevelopmentofFunctionalFoodCentre (CIDAF),Granada,Spain
SandraLo ´ pez-Arana DepartmentofNutrition,Faculty ofMedicine,UniversityofChile,Santiago,Chile
PauloSe ´ rgioLoubetFilho FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
RosanaAparecidaMano ´ lioSoaresFreitas Department ofNutrition,SchoolofPublicHealth,Universityof SaoPaulo,SaoPaulo,Brazil
LilianReginaBarrosMariutti FoodScienceand NutritionDepartment,SchoolofFoodEngineering, UniversityofCampinas,Campinas,Brazil
MarioRobertoMarosticaJunior FoodScienceand NutritionDepartment,SchoolofFoodEngineering, UniversityofCampinas,Campinas,Brazil
A ´ ngelaMiranda-Segura ResearchandDevelopmentof FunctionalFoodCentre(CIDAF),Granada,Spain
GlauciaMariaPastore FoodScienceandNutrition Department,SchoolofFoodEngineering,University ofCampinas,Campinas,Brazil
AhmadFaizalAbdullRazis NaturalMedicinesand ProductsResearchLaboratory, InstituteofBioscience, UniversitiPutraMalaysia, Serdang,Selangor,Malaysia
MiguelA ´ ngelRinco ´ n-Cervera InstituteofNutritionand FoodTechnology,UniversityofChile,Santiago,Chile
DanieleBobrowskiRodrigues UniversityofBrası´lia,Brazil
AntonioSegura-Carretero DepartmentofAnalytical Chemistry,UniversityofGranada,Spain;Research andDevelopmentofFunctionalFoodCentre (CIDAF),Granada,Spain
AtulA.Shirkhedkar R.C.PatelInstituteofPharmaceutical EducationandResearch,Shirpur,India
RakeshSindhu ChitkaraCollegeofPharmacy,Chitkara University,Rajpura,India
InderbirSingh ChitkaraCollegeofPharmacy,Chitkara University,Rajpura,India
SanjayJ.Surana R.C.PatelInstituteofPharmaceutical EducationandResearch,Shirpur,India
Suryamani ChitkaraCollegeofPharmacy,Chitkara University,Rajpura,India
Marı´adelCarmenVillegas-Aguilar Departmentof AnalyticalChemistry,UniversityofGranada, Spain
Chapter1 Introduction CinthiaBau´BetimCazarin,JulianoLemosBicas,GlauciaMariaPastoreandMarioRobertoMarosticaJunior FoodScienceandNutritionDepartment,SchoolofFoodEngineering,UniversityofCampinas,Campinas,Brazil
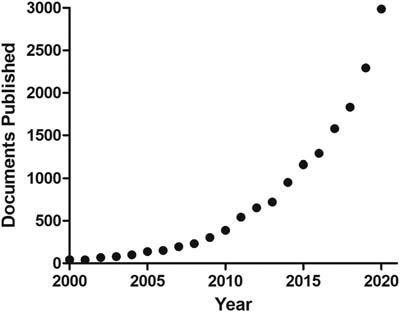
1.1Introduction Theinterestinbioactivecompoundshasbeenincreasingoverthelastfewyears.AbriefsearchinthePubmedindicates atotalof15,711publicationsusing“bioactivecompounds”asadescriptorinthelast20years,increasingfrom41(in 2001)to2986documents(in2020)atanaverageannualgrowthrateof24%.Bioactivecompoundscanbedefinedas nutrientsandnonnutrientspresentinthefoodmatrix(vegetalandanimalsources)thatcanproducephysiologicaleffects beyondtheirclassicalnutritionalproperties.Additionally,thesecompoundscanbefoundinalgae,fungal,different marinesources,etc.Nowadays,thereisalargenumberofpublicationsreportingqualitativeandquantitativedataon bioactivecompoundsinfoods,aswellasdemonstrationsoftheirprotectiveeffectsinvitroandinvivoagainstdifferent typesofdiseases.However,studiesontheirmechanismofactionarenotascomprehensiveasthisdescriptiveinformation.Thus,thisbookintendstogathertheavailableinformationonthemechanismofactionofthemostrelevantbioactivecomponentscommonlyfoundinfoodstuff(Fig.1.1).
1.2Chronicdiseasesintheworldandthecausalroleoftheenvironmentalfactors
Noncommunicablediseasesareresponsiblefor41milliondeathsperyearglobally,ofwhich85%correspondtoprematuredeath(WorldHealthOrganization,2018).In2020,theimpactofthesediseasesinpublichealthwasmoreevidencedwiththecoronaviruspandemic(COVID-19),highlightingthefragilityoftheindividualsthathavechronic diseases,whicharemorepronetotheeffectsofotherdiseases,likeSARS-CoV-2coronavirus(Gaoetal.,2020; Mauvais-Jarvis,2020;Vasetal.,2020;Zhouetal.,2020)andotherhealthproblems.Anunhealthydietisoneofthe modifiablefactorsthatcontributetodevelopingnoncommunicablediseasesanddecreasingtheimmunesystemthatprotectsourbodyagainstharmfulsubstancesandinvaderslikegermsorpathogenicmicroorganisms.
Thedemographicandnutritionaltransitionsobservedinsomenationshavecontributedtoincreasesinnoncommunicablediseaseratessincethistypeofdiseaseisrelatedtohabitsandpracticescarriedoutthroughoutlife.Theextremes malnutritionandobesitycoexistinsomeregions,andbothcompromisehumanhealthinmanyways.Foodsecurityis
FIGURE1.1 Thenumberofdocumentsreferringto“bioactivecompounds”published inPubmedfrom2000to2020.
alsoanimportanttopicassociatedwithmorethanonesustainabledevelopmentgoalinthe2030Agenda(Dı´az-Bonilla &Hepburn,2016;UnitedNations,2020).
Dataobtainedfromfoodsurveyscarriedoutindifferentnationsareusedtoformulateguidelinesandepidemiologicalsurveysaboutindividuals’qualityoffoodconsumption.However,eachnation’sculturalandenvironmentaldifferencesmustbetakenintoaccountwhenestablishingconducttoguaranteeahealthydiet.Accesstolocal,regional,or seasonalfoodscanfavortheconsumptionofhealthyfoods,withagreatersupplyofnutrientsandsustainability,contributingtothepromotionofhealthandthelocaleconomy.
1.2.1Foodsasatoolforpreventingchronicdiseasesanddisabilities Since1935,Dr.PedroEscudero,regardedasthenutritionfatherinLatinAmerica,describedthatahealthydietshould focusonquality,quantity,harmony,andadequacy.Theseprinciplesareusuallyassociatedwiththeso-calledLawsof CorrectNutrition(Cha ´ vez-Bosquez&Pozos-Parra,2016).Providingagooddietduringalllifecyclestagesisthebasis toachievegoodhealthstatusanddevelopment.Forinstance,thelackofdietqualityduringthefirstyearsoflifeand infancycanfavorstuntingthatwillimpactallthechildren’sdevelopmentandcompromisetheircapabilityandpotential intheadultstage(Hurleyetal.,2016).
Theconsumptionofcaloricfoodsrichinsugar,fat,andsalthasbeenincreasingworldwideaswellastheincidence ofoverweightandobesityinallages(Brancaetal.,2019).Ontheotherhand,ingeneral,thepopulationfromseveral countriesdonotintaketheWorldHealthOrganizationrecommendedamountsoffruits,legumes,andwholegrains, whicharetheprimarysourceoffibersinthediet,besidessupplyingminerals,vitamins,andbioactivecompounds (Franketal.,2019).Similarly, Brancaetal.(2019) attributedtheintakeofanunbalanced—poorinwholegrains,fruits, nuts,seeds,andvegetables,andrichinsodiumandsugar—totheglobalburdenofnoncommunicablediseasesobserved in2017.
MinistryofHealthofBrazil(2015) andthe Wallaceetal.(2020) areexamplesofcountrieswherethedietaryguidelinesrecommendthatfruitsandvegetablesconstituteone-halfofdietconsumptiontoimprovecitizens’mealsand health.Similarly,therecognizedMediterraneandietshowsasignificantcontributionoffruits,vegetables,nuts, legumes,andwholecerealsinitscomposition(Trichopoulouetal.,2014).Basedontheliteraturedata,thebenefitsof consumingfruitsandvegetablesareunequivocallyassociatedwithnoncommunicablediseaseprevention(KossMikołajczyketal.,2019;Milleretal.,2017).TropicalcountrieslikeBrazilhavetheadvantageofhighbiodiversityand availabilityofdifferentfruits,vegetables,andnutsrichinbioactivecompounds.However,thesetropicalregionsalso concentrateonthelow-ormiddle-low-incomecountries(TheWorldBank,2020),andsocioeconomicaspectsofthese nationsareassociatedwithinsufficientfruitandvegetableconsumptionwhencomparedtomoreadvantagedgroups (Allenetal.,2017).
Despitetheirhealth-promotingproperties,theprocessingoffruitsandvegetablesgenerateslargeamountsof by-products,whicharealsorichinphytochemicals(Kumaretal.,2021).Fromasustainableperspective,theseunderutilizedby-productscouldprovidebioactivecomponentstothefoodindustryandbetternutritionalstatusforthepopulation.Inthissense,sustainabledietsareahighlighttopicincludedinthedebateoverthelastyears,mainlybecauseof theimpactourfoodintakehasontheEarthandhownutritiousthispatternis.TheuseofaCircularEconomy,for instance,isapracticethatcanimproveourwayofexploringthesourcesprovidedbytheEarthandthedietqualityof humanity(Ghosh&Agamuthu,2018).Theutilizationofallpartsoffoodsortheirby-productscanminimizefood wasteandmaximizetheuseofthebioactivecompoundspresentinthesefoodmatrices.
Therefore,webelievethatunderstandingthemechanismsinvolvedinthehealtheffectofbioactivefoodcomponents mayaccredittheiruseforpreventinghealthissues,assuringabetter-nourishedpopulation.Consequently,theconventional(e.g.,fruitsandvegetables)andunconventional(e.g.,agro-industrialby-products)sourcesofsuchsubstances shouldbescientificallyinvestigatedandcommerciallyexploredwell.
Inthiscontext,thisbookcompilestheavailableinformationonthemainbioactivecomponentsfoundinfoods, focusingontheirmechanismofaction.Itisorganizedinninechapters:thefirstdealingwithsomemechanismsfrom noncommunicablediseases,whileeachofthefollowingeightchapterswilldetailthemaingroupsofbioactivecompounds(phenoliccompounds,preandprobiotics,carotenoids,tocopherols,bioactivepeptides,polyunsaturatedand monounsaturatedfattyacids,sulfurcompounds,andphytosterol),includingtheirfoodsources,andspecificmechanisms involvedintheprotectivehealtheffectassociatedwiththeirconsumption.
References Allen,L.,Williams,J.,Townsend,N.,Mikkelsen,B.,Roberts,N.,Foster,C.,&Wickramasinghe,K.(2017).Socioeconomicstatusandnoncommunicablediseasebehaviouralriskfactorsinlow-incomeandlower-middle-incomecountries:Asystematicreview. TheLancetGlobal Health, 5(3),e277 e289.Availablefrom https://doi.org/10.1016/S2214-109X(17)30058-X
Branca,F.,Lartey,A.,Oenema,S.,Aguayo,V.,Stordalen,G.A.,Richardson,R.,Arvelo,M.,&Afshin,A.(2019).Transformingthefoodsystemto fightnon-communicablediseases. BMJ(ClinicalResearched.), 364,l296.Availablefrom https://doi.org/10.1136/bmj.l296
Chavez-Bosquez,O.,&Pozos-Parra,P.(2016).TheLatinAmericanlawsofcorrectnutrition:Review,unifiedinterpretation,modelandtools. ComputersinBiologyandMedicine, 70,67 79.Availablefrom https://doi.org/10.1016/j.compbiomed.2015.12.019 Dıaz-Bonilla,E.,&Hepburn,J.(2016). Trade,foodsecurity,andthe2030agenda.InternationalCentreforTradeandSustainableDevelopment (ICTSD).Retrieved28thDecemberfrom https://ictsd.iisd.org/sites/default/files/research/sdgs_food_security.pdf Frank,S.M.,Webster,J.,McKenzie,B.,Geldsetzer,P.,Manne-Goehler,J.,Andall-Brereton,G.,Houehanou,C.,Houinato,D.,Gurung,M.S., Bicaba,B.W.,McClure,R.W.,Supiyev,A.,Zhumadilov,Z.,Stokes,A.,Labadarios,D.,Sibai,A.M.,Norov,B.,Aryal,K.K.,Karki,K.B., Jaacks,L.M.(2019).Consumptionoffruitsandvegetablesamongindividuals15yearsandolderin28low-andmiddle-incomecountries. The JournalofNutrition, 149(7),1252 1259.Availablefrom https://doi.org/10.1093/jn/nxz040
Gao,F.,Zheng,K.I.,Wang,X.-B.,Sun,Q.-F.,Pan,K.-H.,Wang,T.-Y.,Chen,Y.-P.,Targher,G.,Byrne,C.D.,George,J.,&Zheng,M.-H.(2020). ObesityisariskfactorforgreaterCOVID-19severity. DiabetesCare,dc200682.Availablefrom https://doi.org/10.2337/dc20-0682
Ghosh,S.K.,&Agamuthu,P.(2018).Circulareconomy:Thewayforward. WasteManagement&Research, 36(6),481 482.Availablefrom https:// doi.org/10.1177/0734242x18778444
Hurley,K.M.,Yousafzai,A.K.,&Lopez-Boo,F.(2016).).Earlychilddevelopmentandnutrition:Areviewofthebenefitsandchallengesofimplementingintegratedinterventions. Advancesinnutrition(Bethesda,Md.), 7(2),357 363.Availablefrom https://doi.org/10.3945/an.115.010363
Koss-Mikołajczyk,I.,Baranowska,M.,Todorovic,V.,Albini,A.,Sansone,C.,Andreoletti,P.,Cherkaoui-Malki,M.,Lizard,G.,Noonan,D.,Sobajic, S.,&Bartoszek,A.(2019).ProphylaxisofNon-communicableDiseases:Whyfruitsandvegetablesmaybebetterchemopreventiveagentsthan dietarysupplementsbasedonisolatedphytochemicals? CurrentPharmaceuticalDesign, 25(16),1847 1860.Availablefrom https://doi.org/ 10.2174/1381612825666190702093301
Kumar,K.,Srivastav,S.,&Sharanagat,V.S.(2021).Ultrasoundassistedextraction(UAE)ofbioactivecompoundsfromfruitand vegetableprocessingby-products:Areview. UltrasonicsSonochemistry, 70,105325.Availablefrom https://doi.org/10.1016/j. ultsonch.2020.105325
Mauvais-Jarvis,F.(2020).Aging,malesex,obesity,andmetabolicinflammationcreatetheperfectstormforCOVID-19. Diabetes, 69(9),1857. Availablefrom https://doi.org/10.2337/dbi19-0023
Miller,V.,Mente,A.,Dehghan,M.,Rangarajan,S.,Zhang,X.,Swaminathan,S.,Dagenais,G.,Gupta,R.,Mohan,V.,Lear,S.,Bangdiwala,S.I., Schutte,A.E.,Wentzel-Viljoen,E.,Avezum,A.,Altuntas,Y.,Yusoff,K.,Ismail,N.,Peer,N.,Chifamba,J., Mapanga,R.(2017).Fruit,vegetable,andlegumeintake,andcardiovasculardiseaseanddeathsin18countries(PURE):aprospectivecohortstudy. TheLancet, 390(10107), 2037 2049.Availablefrom https://doi.org/10.1016/s0140-6736(17)32253-5
MinistryofHealthofBrazil.(2015). DietaryguidelinesfortheBrazilianpopulation.MinistryofHealthofBrazi.Retrieved27thDecemberfrom http://bvsms.saude.gov.br/bvs/publicacoes/dietary_guidelines_brazilian_population.pdf
TheWorldBank.(2020). Theworldbyincome,FY2017.Retrieved28thDecemberfrom https://datatopics.worldbank.org/sdgatlas/archive/2017/theworld-by-income.html
Trichopoulou,A.,Martınez-Gonzalez,M.A.,Tong,T.Y.N.,Forouhi,N.G.,Khandelwal,S.,Prabhakaran,D.,Mozaffarian,D.,&deLorgeril,M. (2014).DefinitionsandpotentialhealthbenefitsoftheMediterraneandiet:viewsfromexpertsaroundtheworld. BMCMedicine, 12(1),112. Availablefrom https://doi.org/10.1186/1741-7015-12-112
UnitedNations.(2020). The17goals.Retrieved28thDecemberfrom https://sdgs.un.org/goals
Vas,P.,Hopkins,D.,Feher,M.,Rubino,F.,&Whyte,M.B.(2020).Diabetes,obesityandCOVID-19:Acomplexinterplay. Diabetes,Obesityand Metabolism, 22(10),1892 1896.Availablefrom https://doi.org/10.1111/dom.14134
Wallace,T.C.,Bailey,R.L.,Blumberg,J.B.,Burton-Freeman,B.,Chen,C.yO.,Crowe-White,K.M.,Drewnowski,A.,Hooshmand,S.,Johnson, E.,Lewis,R.,Murray,R.,Shapses,S.A.,&Wang,D.D.(2020).Fruits,vegetables,andhealth:Acomprehensivenarrative,umbrellareviewof thescienceandrecommendationsforenhancedpublicpolicytoimproveintake. CriticalReviewsinFoodScienceandNutrition, 60(13), 2174 2211.Availablefrom https://doi.org/10.1080/10408398.2019.1632258
WorldHealthOrganization.(2018). Noncommunicablediseases.Retrieved10thDecemberfromhttps://www.who.int/news-room/fact-sheets/detail/noncommunicable-diseases
Zhou,Y.,Chi,J.,Lv,W.,&Wang,Y.(2020).Obesityanddiabetesashigh-riskfactorsforseverecoronavirusdisease2019(Covid-19). Diabetes/ MetabolismResearchandReviews,e3377.Availablefrom https://doi.org/10.1002/dmrr.3377,n/a(n/a).
Thispageintentionallyleftblank
Mainmolecularmechanismsfor noncommunicablediseases MarinaVilarGeraldi,PauloSe´rgioLoubetFilho,AndressaMaraBaseggio,CinthiaBau´BetimCazarinand MarioRobertoMarosticaJunior
FoodScienceandNutritionDepartment,SchoolofFoodEngineering,UniversityofCampinas,Campinas,Brazil
2.1Introduction Noncommunicablediseases(NCDs),orchronicdiseases,areregardedasnoninfectioushealthconditionsoflongdurationorlonglatencyandinvolvesthecombinationofgenetic,physiological,behaviors,andenvironmentalfactors. NCDsareagroupofconditionsthatincludecardiovasculardisease(CVD),chronicrespiratorydiseases,cancers,diabetes,andmentalillness(WHO,2018a).Theburdenofthesediseasesisrising;thenumberofdeathswas41millionpeoplein2016,representing70%ofallglobalmortality.AccordingtotheNCDsglobalstatusreportbyWorldHealth Organization(WHO),in2016theCVDsarethefirstcauseofglobaldeaths(31%),followedbycancers(16%),chronic respiratorydiseases(7%),anddiabetesinthefourthplacewith3%ofdeaths(WHO,2018b).
Thesediseasesusuallyemergeinmiddleageafterlongexposuretoanunhealthylifestyleinvolvingmodifiablerisk factors,whichincludestobaccouse,harmfuluseofalcohol,physicalinactivity,unhealthydiet,overweight/obesity, raisedbloodpressure,raisedbloodsugar,andraisedcholesterollevels;allincreasetheriskofNCDs(Forouzanfar etal.,2016;WHO,2018a).Theobesityprevalencehasbeenincreasingglobally,andtheriskfactorhasbeenassociated withhighmorbidityandmortalityrates.Also,obesityisachronicsystemicinflammationthatcontributestodiabetes, somecancers,CVDs,neurodegenerativediseases,amongotherdiseasesonset(Hamjaneetal.,2020).
AccordingtotheProspectiveUrbanRuralEpidemiology,acohortstudywith135,335individualsin18countries showedthatelevatedfruit,vegetable,andlegumeintakeisinverselyassociatedwithmajorityCVDs,myocardialinfarction,cardiovascularmortality,noncardiovascularmortality,andtotalmortality.Theresultsindicatedthattheconsumptionofthreetofourservingsoffruitsandvegetablesperday(equivalentto375 500g/day)improvesboth noncardiovascularandtotalmortalityrates(Milleretal.,2017).Manybeneficialeffectswereattributedtohighamounts ofdietarybioactivecompounds,suchasvitamins,polyphenols,andcarotenoids,whichhaveantioxidantandantiinflammatoryproperties.Fruitsandvegetablesmightimprovebloodvesselendothelialfunctions(Asplund,2002; Hooperetal.,2008)andlowerbloodpressure(Appeletal.,1997;Johnetal.,2002).Moreover,fruits,vegetables,and legumesaregoodsourcesofdietaryfiberandphytochemicals;theirconsumptioncanreducetheinsulinresponsetocarbohydrates(AmericanDiabetesAssociation,2008),decreasetotaland LDL-cholesterol(low-densitylipoprotein)and triglycerides(Aguiaretal.,2018;Anderson&Major,2002;Andersonetal.,1995).Meta-analysesandcohortstudies showedaninverseassociationbetweentotalflavonoidintakeandincidenceofCVD,coronaryheartdisease,andmortalityinthesediseases(Iveyetal.,2015;Jiangetal.,2015;Wangetal.,2014).
Whilesafelymodulatinginflammationusingtargetedtherapeuticsremainsachallenge,theresultsfromrecentprospectivestudiesdemonstratethatreducinginflammationmayofferanovelapproachtoreducingriskforNCDs (Alfaddaghetal.,2020;Franceschi&Campisi,2014;Schwingshackl&Hoffmann,2014).Ahealthydietmayhave favorableeffectsoninflammationthroughweightloss-dependentandindependentpathways(Sayo ´ n-Oreaetal.,2019). Obese,diabetics,orindividualswithmetabolicsyndromemaybenefitfromchangingsomemodifiableriskfactorslike dietarypatterns,practicephysicalactivity,andlosingweight(Spahnetal.,2010;Verheijdenetal.,2004).
***ThereforetheanalysisofthemainmolecularmechanismsinvolvingNCDsiscrucialinimprovingthequalityof lifeandlongevity.Inparticular,theassociationbetweeninflammationandoxidativestresshasbeenstudiedinNCDs.
BioactiveFoodComponentsActivityinMechanisticApproach.DOI: https://doi.org/10.1016/B978-0-12-823569-0.00002-3 © 2022ElsevierInc.Allrightsreserved.
TheseprocessesturnchronicduringNCDsprogressionandcontributetotheevolutionandworseoutcomes(Prasad etal.,2012).Moreover,thischapterfocusesonthemechanismsforNCDs,mainlytheroleoftheinflammatory responseandoxidativestressinobesity-associatedmetabolicabnormalities(insulinresistance,diabetes,andmetabolic syndrome),CVD,cancer,andneurodegenerativediseases.
2.2Mainmolecularmechanismsfornoncommunicablediseases Hereareaddressedsomeofthemostcommoncellularmechanismsintheliteraturethatmaybeinvolvedintheetiology andcontributetosomepathophysiologicalevolution.
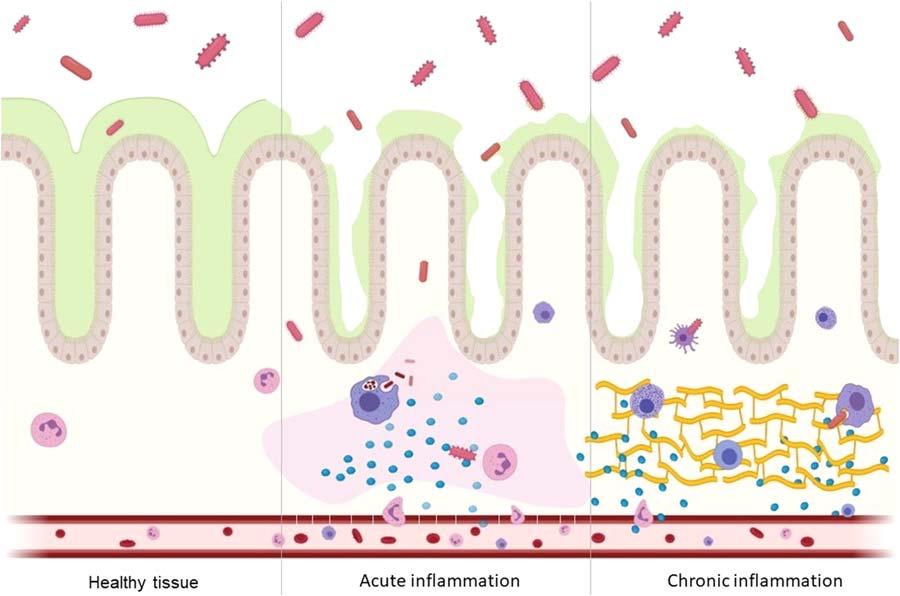
2.2.1Inflammatoryresponseandoxidativestress Inflammationisaprotectiveimmune-mediatedprocess;itsmanifestationistheresponsetoaspecificstimulusaiming toeliminatethecausalfactor,therestorationandthephysiologicalrecoveryoftheinjuredtissue(Fig.2.1);later,the twomainformsofinflammationwillbediscussed:namely,acuteandchronic.
Ingeneral,inflammationcanbeinitiatedbyinfectionsofmicroorganisms(bacteria,viruses,fungi,andparasites), varioustraumas(cuts,thermallesion,chemicaltoxicity),tissuenecrosisofanycauses,foreignbodies(splinters,dust, pollen),andbyhypersensitivityreactions(Kumaretal.,2020).Themolecularmechanismsbywhichsuchaggressive factorsarerecognizedcanbeclassifiedinto:
Cellularmicroorganismreceptors:ThereceptorsclassifiedherebelongtotheToll-likereceptors(TLR)family, identifyingextraandintracellularpathogen-associatedmolecularpatterns;afterrecognitionofthepathogen,these receptorsstimulateviatheIκBkinaseenzymetheincreaseofexpressionandproductionofproteinsthatrecognizeand induceinflammation,inadditiontorecruitingcellsoftheimmunesystem(Kumaretal.,2017)(Fig.2.1).
FIGURE2.1 Gutinflammationactivatedbymicroorganisms.Thehealthytissuepresentsathickmucuslayerblockingmicroorganisms’accessto enterocytes,andfewleukocytespresentinthetissue.Theacuteinflammationpresentsadamagedmucuslayerenablingtheaccessofmicroorganisms tothetissue;vesselandleukocytereactionsareobservedaswellastheminimumtissueinjury.Thechronicinflammationpresentsanirregularmucus layer,blockingandenablingmicroorganismstoreachthetissue,theimmuneresponse,andscarringtissue. Adaptedfrom:(Kumaretal.,2020).Own authorshipcreatedbyBiorender.com
Celldamagesensors:Thesecytosolicreceptorscalledinflammasomesidentifydamage-associatedmolecularpatternsthataremolecularproductsofthedegradationofmitochondrial,nuclear/plasmamembranes,andgeneticmaterial; amongtheinflammasomes,theNLR(nucleotide-bindingdomainandleucine-richrepeat-containing)inducetheproductionofinflammatorycytokinesviathecaspaseenzyme,stimulatingoxidativecellstressandalsotherecruitmentof cellsfromtheimmunesystem(Place&Kanneganti,2018).
Circulatingplasmaproteins:Theyareconstituentsofthecomplementsystem(mannose-bindinglectin,collectin, antibodies)withthefunctionofcoating/opsonizingtheattackingtargetandthroughproteolyticactivation,resultingin theinflammatoryresponsebyleukocytechemotaxisandphagocytosisfollowingthedestructionand/orattackthepathogenmembrane(Kumaretal.,2017).
Acuteinflammation(AI)istheorganism’sfirstresponseaftermolecularsignalingofaspecificinjuryagent;so,itis calledduetoitsrapiddevelopmentandresolutionoftheoffensivefactor,characterizedmainlybyvascularandcellular reactions(Fig.2.1).
Vascularreactionsresultfromtheinitialinteractionbetweentheinnateimmunesystem’scellsandthetissue’spathogen.Fromthehistaminereleasedbythisinteraction,vasodilationoccurs,whichincreasesbloodflowtotheinflammationsite;afterward,thereisanincreaseincapillarypermeability,facilitatingtheextravasationofplasmafluidrichin proteinsinextravasculartissue;then,thebloodflowisreducedfavoringthecontactoftheleukocyteswiththeendothelialcells,initiatingthecellularreactions(Neteaetal.,2017).
Cellularreactionsaremarkedbytheadhesionandmigrationofcirculatingleukocytestotheinflamedtissue;target recognitionoccurswhenpresentinthetissue.Macrophagesandneutrophilsareactivated,thusinitiatingtheirpathogen eliminationmechanismsthroughphagocytosisanddegradationviareactiveoxygenspecies(ROS),nitricoxide(NO) andlysosomes,andextracellulartraps(releasedpro-inflammatorycytokinesandROS).However,cellularproductsgeneratedinresponsetoAIcandestroysurroundedhealthytissue(Neteaetal.,2017).
ThusAIcanendinthreeways:resolutionoccurswhentheinflammatoryresponseeliminatestheoffendingtarget causinglittletissuedamage,thusmakingitpossibletorestoreparenchymalcellsbynormalizingtissuefunction;healing occurswhenthereisconsiderabletissuedamageduetotheinflammatoryresponseintissuesthatarenotcapableof regenerating,beingthenreplacedbyfibroustissue;or,theinflammatoryprocesscanprogressandbecomechronic (Kumaretal.,2020)(Table2.1).
Chronicinflammation(CI)maybeduetothepersistenceofthestimulusrecognizedinAI,oritcanbereproduced insidiously,assumingthecharacterofalatentprocesswithoutpresentingthecharacteristicsofAI;CIisconcomitantly characterizedbyinflammation,injury,andattemptedtissuerepair(Fig.2.1).
Thechronicinflammatoryresponsetoinjuredtissueisbasedonmononuclearleukocytes’reaction,mainlymacrophages,andlymphocytes.Macrophagescanbeactivatedinaclassical(M1)oralternative(M2)manner,detailedin Section2.2.2.M1arerelatedtotheeliminationofthecausalfactorofCIthroughtheirmediators(Table2.1),potentiatingtheinflammatoryresponsethroughleukocytechemotaxisandcausingtissuedamage;whereas,M2promotesthe processoftissuerepairthroughanti-inflammatorycytokinesandgrowth(Table2.1),stimulatingtheformationofnew bloodvesselsandcollagensynthesis(scar).Atthesametime,lymphocytesareactivatedbymacrophagesatthesiteof inflammation,producingantibodies,andcytokines,thusstimulatingtheCIcyclethroughactivationbetween macrophages-lymphocytes(Neteaetal.,2017).
TABLE2.1 Mainphysiopathologicalmarksintheinflammatoryprocess.
AcuteinflammationChronicinflammation
DurationShort-term(h)Long-term(years)
Immune response
Inflammatory mediators
Noncellular(mucuslayer,complementproteins);cellular (neutrophils,basophils,naturalkillers,gut,andbloodbrainbarriers)
IL-1β,-6;TNF-α;INF-γ;reactiveoxygenspecies;nitric oxide;lysosomalenzymes
Noncellular(mucuslayer,complementproteins); cellular(macrophages,lymphocytes,mastcells, microglia,astrocytes)
IL-5,-10,-17;INF-γ;TNF-α;reactiveoxygenspecies; nitricoxide;lysosomalenzymes;TGF-β;VEGF
OutcomesResolution;healing;chronicinflammationAngiogenesis,andfibrosis
IL:interleukins; TNF:tumornecrosisfactor; INF:interferon; TFG:transforminggrowthfactor; VEGF:vascularendothelialgrowthfactor.Adaptedfrom: (Furmanetal.,2019; Kumaretal.,2017).
Tissuerepairoccurswithinsuchinflammatoryevents;similartoAI,thetissuerepairofCIdependsonthedegreeof thelesionfromtheinflammatoryresponseandthelocalcapacityforcellregeneration.However,thecontinuityandproductionofinflammatorymediatorsofCIinducethetissuerepairfocusedonfibrosis,mainlystimulatedbythetransforminggrowthfactor(TGF)-β,leadingtolossoftissuefunctiondependingontheextentofthescar.Thereforeitis relatedtotheetiologyofnumerousNCDs(Kumaretal.,2017)(Fig.2.2).
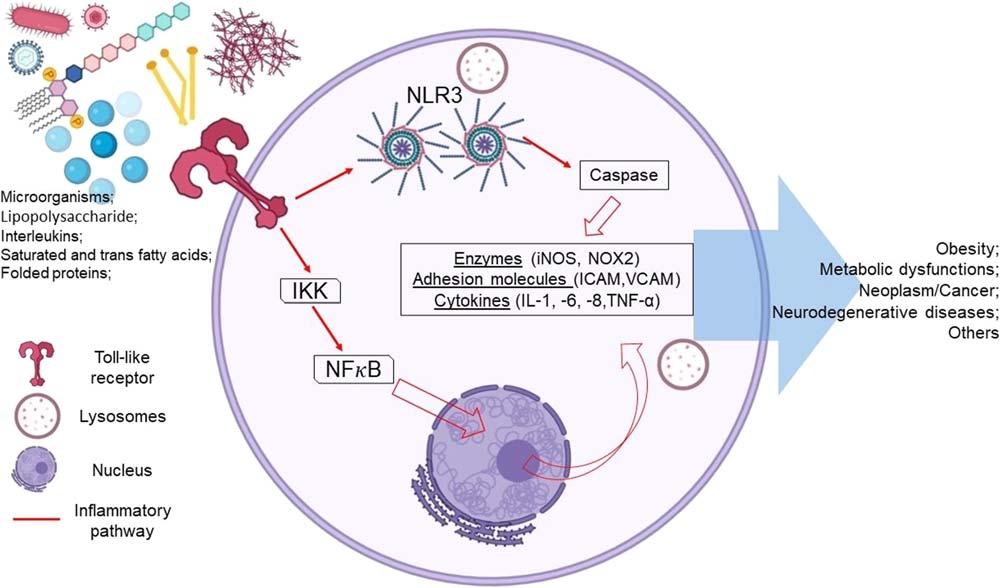
Themaininflammatorymolecularpathwaysreported,andseenasamainstayinsomeNCDs,arethenuclearfactor kappaB(NF-κB)andtheNLR3inflammasome;thesepathwaysarethemaintargetsofexperimentalandclinicaltrials interventionsfromfunctionalfoodsandnutrients,phytochemicals,-biotics,andothers(Furmanetal.,2019).
Thecytokinesandchemokinespresentintheinflammatoryprocessderivefromasignalingcascadeinitiatedbyrecognizingtheinjuryfactorandinflammatorycytokinesbythetransmembranereceptorsofthecells;afterbindingthe receptortothestimulus(canonicalpathway).TheenzymecomplexIKK(IκBkinase)phosphorylatestheIκBmolecule, whichwillproceedtodegradation,whiletheNF-κBdimers(p50/p65)disconnectedfromtheIκBmanagetotranslocate uptothenucleus,wheretheyactivatethetranscriptionofpro-inflammatorycytokines.Inthenoncanonicalpathway, afterrecognizingthestimulus,IKKα isphosphorylatedandinteractswiththep100dimer,phosphorylatinganddegradingittothep52molecule,whichbindstoRelBandtranslocatestothenucleus,activatingthenoncanonicaltranscriptionofpro-inflammatorycytokinesviaNF-κB(Khanetal.,2020)(Fig.2.2).
Theactivecaspase-1enzymeisresponsibleforcleavinginflammatorypro-interleukinstranscribedviaNF-κB,thus potentiatingtheinflammatoryresponseinthepresenceoftheinjuryfactor.Theactivecaspaseoriginatesfromthe NLR3inflammasomecomplex,calledanapoptosis-associatedspeck-likeproteincontainingCARD(ASC)speckwhen formedbytheNLRassociatedwiththeadaptermoleculeASCandinactivecaspase-1;aftersuchastructureisformed, thecaspasedemonstratesautocatalyticcleavagecapacity,thenreleasingitsenzymeintheactiveform(Place& Kanneganti,2018)(Fig.2.2).
FIGURE2.2 Primarycellresponsetoexternaldisturbances(microorganisms,lipopolysaccharide,transandsaturatedfattyacids,foldedproteins, cytokines).Theexcessiveand/orprolongedinflammatoryresponseleadstothedevelopmentofnoncommunicablediseasesviaNF-κBandinflammasome(NLR3)pathways. Adaptedfrom:Fischer,R.,&Maier,O.(2015).Interrelationofoxidativestressandinflammationinneurodegenerativedisease:RoleofTNF. OxidativeMedicineandCellularLongevity,2015. https://doi.org/10.1155/2015/610813;Place,D.E.,&Kanneganti,T.D.(2018). Recentadvancesininflammasomebiology. CurrentOpinioninImmunology,50,32 38. https://doi.org/10.1016/j.coi.2017.10.011.Ownauthorship createdbyBiorender.com
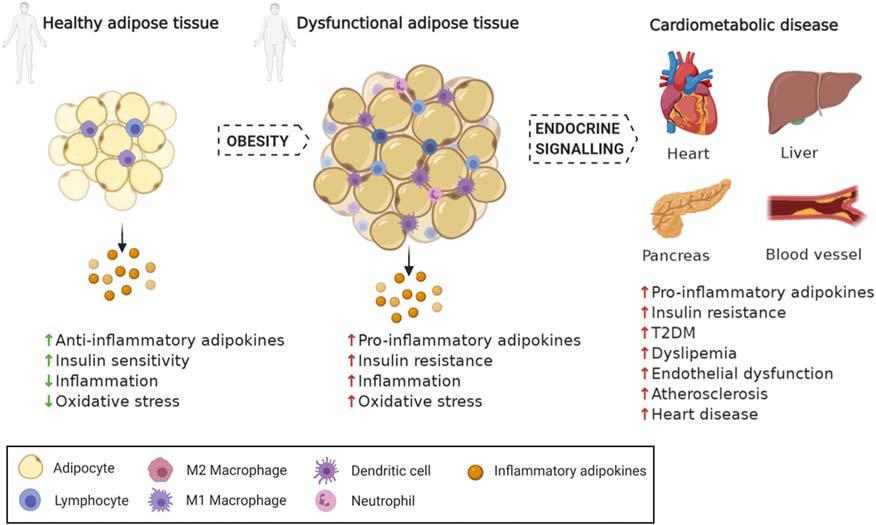
2.2.2Obesityandmetabolicdysfunctions Thecardiometabolicdiseaseincludesthepresenceofobesity,impairedcarbohydratemetabolism(insulinresistance, impairedglucosetolerance,ortype-2diabetes—T2DM),dyslipidemia,andhypertension.Inadequateeatinghabits,sedentarylifestyle,andweightgainstronglyinfluenceobesityandthedevelopmentofotherpathologiesrelatedtocardiometabolicdysfunctions(Mechanicketal.,2020).
Specificmechanisticpathwaysderivefromprimarydriversfromgenetics,environment,andbehavior,withsubsequentadiposity,insulinresistance, β-celldysfunction,andmetabolicsyndromeleadingtoCVD(Mechanicketal., 2020).Geneticsareimportanttodeterminediseasedrivernetworkstorevealpotentiallygenesandmolecularmechanismsresponsibleforthepathophysiologicalandhereditabilityofthesediseases.However,context-dependentriskvariantshavemoreeffectontheexpressionofphenotypictraits(Bjo ¨ rkegrenetal.,2015).Geneticstoidentifymolecular driversisuseful,buttheinteractionofgenes,environment,andbehaviordeterminesriskanddiseaseprocesses (Bjorkegrenetal.,2015).Theenvironmentprovidescontextfortheexpressionofgeneticcardiometabolicriskfactors, andindividualbehaviorscanmodulateandprovideinterventionopportunities(Mechanicketal.,2020).
Obesityisamajorhealthproblemthatincreasestheriskofmanyrelatedcomplications,suchasmetabolicsyndrome,CVDs,respiratorydisorders,diabeticcomplications,andcancer(Nigroetal.,2014).Adiposetissueshavebeen recognizedasmorethanastorageorgan;itisanendocrineorganthatproducesbiologicallyactivesubstancesdefined as“adipokines,”proteinhormoneswithpleiotropicfunctionsintheregulationofenergymetabolismaswellasinappetite,insulinsensitivity,inflammation,atherosclerosis,andproliferation(Rega-Kaunetal.,2013).
Here,wedescribethecurrentscientificevidenceonmolecularmechanismsofobesity-associatedinflammationonset ofinsulinresistance,T2DM,hypertension,metabolicsyndrome,andCVD.
2.2.2.1Adiposity Adiposity-basedchronicdiseaseisanewdiagnostictermforobesitythatidentifiesasachronicdiseasescientifically andlessstigmatizeddiagnostictermformedicaldiagnosisofthepathophysiologicprocess.Adiposity-basedchronic diseasereferstoadipocytesandadiposetissueandconnectstheimpactonhealth-relatedtoquantity,distribution,and/ orfunctionofadiposetissuecharacterizedbycardiometabolic,biomechanical,orpsychologicalcomplications (Mechanicketal.,2017).
Abnormaladipositydistributionreflectsanimbalanceofcaloricintakeandenergyexpenditureandleadstoan increaseinthenumberofadipocytes(adipocytehyperplasia)andsize(adipocytehypertrophy)duetotheincreasedstorageoftriglycerides(Nakamuraetal.,2014).Theadiposetissuebecomesdysfunctional,thehypertrophiedadipocytes undergonecroticand/orapoptoticcelldeath,contributingtotheinsulinresistance,pro-inflammatorycytokinesproduction,infiltration,andactivationofimmunecells(Cintietal.,2005;Strisseletal.,2007).Thelow-gradeinflammation observedintheadiposetissueresultsfromtheincreasedproductionofpro-inflammatorycytokinesthatmodulateadipocytefunction,differentiation,andadipokinessecretioninthebloodstream.Thissituationaffectsthesystemicmetabolic homeostasisandinflammationandthedevelopmentofobesity-associatedcardiometabolicdiseasessuchasinsulinresistance,T2DM,andCVDs(Fig.2.3)(Feijo ´ o-Bandı´netal.,2020).
Obesityleadstomacrophagerecruitmentandaccumulationinadiposetissue,whichisassociatedwithsystemic inflammation,andinsulinresistance(Weisbergetal.,2003).Besides,obesityalterstheexpressionoftheM1/M2state phenotype.MacrophagesinobesityexpressgenesassociatedwithM1-likeor“classicallyactivated”phenotype,whereas leanfatexpressmarkersofanM2or“alternativelyactivated”state(Lumengetal.,2007).Thelper1(Th1)-type responsesmediatedbyinterferon-γ,microbialstimuli,interleukin-4(IL-4),andIL-13promoteM1macrophagesphenotype.M1macrophagesproducepro-inflammatorycytokines,suchastumornecrosisfactor(TNF)-α,expressinducible nitricoxidesynthase(iNOS),andproduceROSandnitrogenintermediates,promotinginflammation,tissuedestruction, andinsulinresistance(Gordon,2003).M2-likephenotypebyTh2-typecytokinesexpressesanti-inflammatorycytokines,suchasinterleukin(IL)-10andtheenzymearginase-1,whichinhibitsiNOSactivity.Thismacrophagesclasspromotesangiogenesis,theresolutionofinflammation,andprotectsagainstobesity-inducedinsulinresistance(Gordon, 2003;Odegaard&Chawla,2011).
ObesityalsoinfluencesTcells’subsetsthatregulatemacrophagesphenotypeinadiposetissue,therebygenerating eitherapathogenicoraprotectiveenvironment.CD4 1 regulatoryTcellsandTh2-polarizedcellsarepresentinthe adiposetissueofleanmice,andtheycontributetothemaintenanceofadiposetissuefunctionandinsulinsensitivity,in partthroughalternativeanti-inflammatoryactivationofmacrophages.ObesityleadstoactivationofCD8 1 Tcells, whereastheCD4 1 helperandregulatoryTcellswerediminished.TheCD8 1 Tcellsaccumulationprecedesmacrophageinfiltrationinthistissue,propagatingthepro-inflammatorycascade(Nishimuraetal.,2009).Metabolically
FIGURE2.3 Obesity,inflammation,andcardiometabolicdiseases.Abnormaladiposityaccumulationleadstoderegulationofitsphysiologicalfunction,relatedtoadipocytesquantity,distribution,andfunction.Thissituationaffectsthesystemicmetabolichomeostasisandinflammationand the developmentofobesity-associatedcardiometabolicdiseasessuchasinsulinresistance,T2DM,andCVDs. Greenarrows:beneficialeffect; Red arrows:detrimentaleffect. Adaptedfrom:Feijoo-Bandın,S.,Aragon-Herrera,A.,Morana-Fernandez,S.,Anido-Varela,L.,Tarazon,E.,Rosello-Lletı, E.,Portole´s,M.,Moscoso,I.,Gualillo,O.,Gonza´lez-Juanatey,J.R.,&Lago,F.(2020).Adipokinesandinflammation:Focusoncardiovasculardiseases. InternationalJournalofMolecularSciences,21(20),1 34. https://doi.org/10.3390/ijms21207711.OwnauthorshipcreatedbyBiorender.com
dysfunctionaladiposetissuealsopresentscrown-likehistologicalstructuresrepresentingactivatedM1-likemacrophages surroundedbynecroticadipocyteandCD8 1 Tcells(Apovianetal.,2008).Thenumberofcrown-likestructuresis correlatedwithinflammationandinsulinresistance(Cintietal.,2005).
2.2.2.1.1Theactionofthemaininflammatorymoleculesinvolvedintheinflammationassociatedwith obesityanditsmetabolicabnormalities
Clinicalandepidemiologicalstudiesindicatethatobesity-relatedmetabolicdysfunctioninvolvesdevelopingasystemic, low-gradeinflammatorystateduetochangesintheexpressionofcytokinesbyadiposetissue(Nakamuraetal.,2014). Theproductionofadipokinesisupregulatedintheobesestateandleadstothesubsequentsecretionoftheseproinflammatoryproteinsinthecirculation(Berg&Scherer,2005).Obesity-relatedinflammationcanalsobeobservedin thepancreas,liver,andbrain(Cildiretal.,2013).Atthemolecularlevel,adiposityleadstotheactivationofmetabolic signalingpathways,particularlytheN-terminalkinasec-Jun(JNK),theNF-κB,andtheproteinkinaseR(Nakamura etal.,2010).Activationofthesepathwaysresultsinthereleaseofinflammatoryfactors,includingadipokineslikeleptin,adiponectin,andresistin,aswellascytokines(Gregor&Hotamisligil,2011).
Theinfiltrationofimmunecellsintoadiposetissueinducedbyobesityleadstoanincreaseinthesynthesisand secretionofinflammatoryfactors.Thesemediatorshaveeitherpro-inflammatoryandanti-inflammatoryactivities,and theirbalanceiscriticalinmaintainingsystemichomeostasis.Underphysiologicalconditions,adipocytespredominantly secreteanti-inflammatoryadipokinessuchasadiponectin,TGF-β,IL-10,secretedfrizzled-relatedprotein5,andNO, whichpromotesinsulinsensitivityandpromotescardioprotectiveandantiatherogeniceffects.However,inobesity,dysfunctionalhypertrophicadipocytesproduceandreleasepro-inflammatoryadipokinessuchasleptin,TNF-α,IL-6,IL18,angiopoietin-likeprotein2,resistin,andlipocalin2,whichhaveatherogeniceffects(Koliakietal.,2019).These dysfunctionaladipokinesproductionhasbothlocalandsystemiceffectsoninflammatorycells(Nakamuraetal.,2014). Manyoftheknowncirculatingpro-inflammatorymoleculesthatdirectlyparticipateinthedevelopmentofcardiometabolicdiseasesarereleasedfromadipocytes,linkingthedevelopmentofmanycomplicationsassociatedwithobesity, suchasmetabolicsyndrome,respiratorydisorders,CVDs,T2DM,andcancer.Alistofpro-andanti-inflammatory mediatorsandtheircardiovascularactionscanbefoundin Table2.2
TABLE2.2 Actionofprincipalsinflammatorymediatorsinvolvedintheobesity-associatedinflammation.
Inflammatory molecules InflammationRolesandeffectsReferences
LeptinProinflammatory
ResistinProinflammatory
VisfatinProinflammatory
OmentinProinflammatory
AdiponectinAntiinflammatory
Appetitecontrolthroughthecentralnervoussystem.Playsa keyroleinbody-weightregulation.HighlevelinducesIR, Ms,andseveraltypesofcancer
Pro-atherogenic,asubclinicalmarkerofatherosclerosis. PromotesIRandinflammationthroughIL-6andTNF secretionfrommacrophages
Adipokineisexpressedbymacrophages.Itcontributesto thedevelopmentofMsandhassignificanteffectson glucosemetabolismandatherosclerosis.
Pro-angiogenic,inhibitionofvascularinflammation,a subclinicalmarkerofatherosclerosisInsulin-sensitizing effects
Anti-proliferative,antidiabetic,anti-inflammatory, antiatherogenic,andcardioprotectiveeffects.Ithasa negativerelationshipwithBMI
GhadgeandKhaire(2019), RamosMunizetal.(2018)
Burnettetal.(2005), Park etal.(2017)
Chenetal.(2015), Coimbra etal.(2014)
Maruyamaetal.(2012)
Ghadgeetal.(2018)
AdipolinAntiinflammatory Improvesglucosemetabolismandinsulinsensitivit Sargolzaeietal.(2018)
RBP4Proinflammatory
Lipocalin2Proinflammatory
ANGPTL2Proinflammatory
TNFα Proinflammatory
HighlycorrelatedwithIR,otherMsfactors,andCVD Norseenetal.(2012)
RegulatedbyobesityandpromotesIR Zhangetal.(2008)
Secretedbyadiposetissueandthatitscirculatinglevelwas closelyrelatedtoadiposity,systemicIR,andinflammation
InducesIR,T2DM,andMs.Reducestheoxidationoffatty acidsandincreasestheplasmalevelsoffreefattyacids.Itis involvedinallstagesoftumorigenesis
IL-1β Proinflammatory Promotes β cellsdysfunctionanddeath,inducingIR,and T2DM.Promotestheproliferationandinvasionofcancer cellsincludingbreast,liver,andpancreaticcancers
IL-6Proinflammatory
MCP-1Proinflammatory
SFRP5Antiinflammatory
CRPProinflammatory
ItstimulatesthelivertosynthesizeandsecretetheCreactiveprotein.InvolvedinthepathophysiologyofIRand T2DM
Inducesmacrophagecelldivisioninadiposetissue,playsa crucialroleinthedevelopmentofobesity,diabetes,and CVD
Anti-inflammatoryadipokinerelatedtoglucoseintolerance andhepaticsteatosis.Suppressthepro-inflammatory WNT5asignaling,whichpromotesinflammatoryresponses involvingmacrophagesanditisupregulatedintheadipose tissue
InvolvedinT2DMandCVD.StimulatesIL-6andTNF-α production.Promotedamageonthevascularwall, producingendothelialdysfunctionandreducingthe bioavailabilityofNO.Itactivatesmonocytes,VSMC,and endothelialcells,andtriggerstheonsetofthrombotic complications
Tabataetal.(2009)
Burlakaetal.(2018), Clark (2007), Weigertetal.(2010)
Dysonetal.(2014), Jager etal.(2007), Kolbetal. (2016), Lietal.(2012)
Elluluetal.(2017)
Cranfordetal.(2016), Panee (2012)
Ouchietal.(2010)
Acquahetal.(2016), Ramos Munizetal.(2018)
BMI,bodymassindex; BP,bloodpressure; CRP,Creactiveprotein; CVD,cardiovasculardiseases; IR,insulinresistance; MCP-1,monocytechemoattractant protein1; Ms,metabolicsyndrome; NO,nitricoxide; TNF-α,tumornecrosisfactor; T2DM,type2diabetesmellitus; VSMC,vascularsmoothmusclecells.
Hyperleptnemiaresultingfromanexcessofadiposetissueproductionandhypothalamicleptinresistancehasbeen associatedwithaninflammatoryreaction,oxidativestress,endothelialdysfunction,andinsulinresistance(Katsikietal., 2018).Adiponectinisconsideredanantiatherogenicadipokine(Katsikietal.,2017).Thedysfunctionofadiposetissue islinkedtotheadiponectin/leptinratio(Fru ¨ hbecketal.,2018).Obesityandmetabolicsyndromearecharacterizedby anincreaseinleptinlevels,paralleltoadecreaseinadiponectincirculatingconcentrations.Theadiponectin/leptinratio isnegativelycorrelatedwithmarkersofinflammation,suchasCreactiveprotein(CRP)(Fruhbecketal.,2018),and correlateswithinsulinresistancebetterthanadiponectinorleptinalone(Satohetal.,2004).Inepidemiologicalstudies, anincreaseinthisratiohasbeenassociatedwithhigherinsulinsensitivity,reducedatherosclerosisrisk,anddecreased riskofsometypesofcancer(Fruhbecketal.,2018;Vega&Grundy,2013).
2.2.2.2Insulinresistance Insulinresistanceisapathologicalconditioninwhichcellsfailtorespondtoinsulin,anditisattheintersectionof abnormaladiposityanddysglycemia.Obesityisoneofthemajorfactorscontributingtothelow-gradeinflammation state,whichprovokesinsulinresistance(Rehman&Akash,2016).Thehighlevelsofglucoseand/orlipidsintheblood duringobesityleadtotheactivationofmolecularandmetabolicpathways.Theseinflammatoryresponsesaremediated bypro-inflammatoryand/oroxidativestressmediators,mainlypro-inflammatorycytokinessuchasTNF-α,IL-6, interleukin-1beta(IL-1β),CRP,chemokines,andadipocytokines(Fragosoetal.,2015;Hamjaneetal.,2020).
Thechronicconditionofnutrientsstimulation(metabolicstress)triggerscytokinesignalingproteinsthatblockthe activationofinsulinsignalingreceptorsinpancreatic β-cells.Besidesthat,cytokinesactivateJNKandNF-κB,inhibitinginsulinsignalinginperipheraltissues(Lackey&Olefsky,2016).Inflammasomesandtheinnateimmunesystem’s TLRsareactivatedandincreasetheinflammatoryresponse(Schroderetal.,2010;Stienstraetal.,2012).
GrowingevidencepointstoacrucialroleforIL-1β inmediatingthedevelopmentofinsulinresistance.Production ofIL-1β ismainlyregulatedbyelevatedglucoselevelsandfreefattyacidsintheblood,whichinducetheexpression andreleaseofIL-1β fromthe β-cellsofpancreaticislets.Onceactivatedandproduced,IL-1β regulatestheexpression ofmanyotherpro-inflammatorycytokines,adipokines,andchemokines(Jageretal.,2007).TNF-α pro-inflammatory cytokinehasbeenrecognizedasakeyfactorlinkinginflammationandobesity-associatedinsulinresistance.TNF-α promotesinsulinsignalinginhibitionbyabnormalphosphorylationofinsulinreceptorsubstrateinthemuscleandadiposetissuesanddecreasedsignaltransductionforglucosetransportertype4translocation,thuscontributingtoinsulin resistance(Lorenzoetal.,2008).AcohortstudydemonstratedthatTNF-α levelsinthebloodwereassociatedwith insulinresistance(Hivertetal.,2008).
Thisinflammatoryconditioninducesapoptosisandamyloidosis,fibrosis,recruitmentofmacrophages,andimpaired insulinsecretion(Jageretal.,2007).Theinflammatoryresponsedrivingthedevelopmentofinsulinresistanceprobably comprisesacombinationofpro-inflammatorycytokinesthatjointlyeffectuateT2DMprogression.
2.2.2.3Type2diabetes T2DMisamultifactorialdiseaseinvolvinggeneticandenvironmentalfactors.Obesity,physicalinactivity,aging,tissue lipidaccumulation,oxidativestress,endoplasmicreticulumstressin β-cells,tissueinflammation,autoimmuneinflammation,and β-celldysfunctionarethemainknownfactorsrelatedtoinsulinresistance,whichprogressT2DM(Akash etal.,2013).Hyperglycemiaanddyslipidemiapromoteoxidativestressandpro-inflammatorymediators’production. Chronichyperglycemiaalsoproducesadvancedglycationend-products,whichaccumulateinthebodyandcontributes totheoccurrenceanddeteriorationofdiabeticvascularcomplications(Rhee&Kim,2018).Insulinresistanceisdevelopedinperipheraltissues,whichincreasesthedemandforinsulinsecretionfrompancreatic β-cellstomaintainglucose homeostasis.TheonsetofT2DMoccurswhenthepancreatic β-cellfailstoadapttotheincreasedinsulindemand causedbyinsulinresistanceandleadstocelldeath(DeFronzoetal.,2015).
Pro-inflammatorycytokinesareamongthemultifactorialpathophysiology’s,themostimportantdriverforT2DM propagation(Akashetal.,2013).Asdescribedpreviously,IL-1β,TNF-α,CRP,andIL-6arepredictiveinthedevelopmentofT2DM(Pradhanetal.,2001),whereasweightlossleadstoadecreaseincirculationlevels(Espositoetal., 2003).
TheadipokineresistinwasproposedtolinkobesityandT2DM(Rosen&Spiegelman,2014).Resistinupregulates pro-inflammatorycytokinesexpression,suchasTNF-α,IL-6,IL-12,monocytechemoattractantprotein-1(MCP-1),and macrophagesviatheNF-κBpathway.Epidemiologicalandgeneticstudiesindicatethatincreasedresistinlevelsare associatedwiththedevelopmentofinsulinresistance,diabetes,andCVD(Parketal.,2017).
2.2.2.4Hypertension Agrowingbodyofevidencehasshowntheassociationbetweenobesityandhypertension.Thisassociationisattributed totheincreaseoffreefattyacids,oxidativestress,andinflammatorystateassociatedwithobesityinducingendothelial dysfunction(Jiangetal.,2016;Roush,2019).
Hyperactivityoftherenin-angiotensin-aldosteronesystemplaysanimportantroleinvascularfunctionandvascular injurythroughitsactionsonendothelialdysfunction,vascularremodeling,andvascularinflammation(Leongetal., 2015).Theoxidativestressofthearterialwallandpro-inflammatoryeffectsofhumoralfactors(suchasAngiotensinII) inducesNF-κBactivation(Sanz-Rosaetal.,2005).AngiotensinIIstimulatesROSgenerationthroughactivationof NADPHoxidase(NOX)andreleasingofROS(suchassuperoxideanion)anddecreasingNOgenerationthatimpairs endothelium-dependentvasodilatation(Donghuietal.,2019;McGarretal.,2019;Montezanoetal.,2014).Through increasedNox-derivedROSgenerationandactivationofredox-sensitivetranscriptionfactors,AngiotensinIIsystem leadstotheexpressionofcytokinesIL-1β,IL-6,TNF-α,celladhesionmolecules(ICAM-1,VCAM-1),chemokines (MCP-1),andgrowthfactors.Thesemolecularandcellularprocessesfacilitateincreasedvascularpermeability,leukocyterecruitment,calcification,andvascularfibrosisleadingtovascularinjury(Montezanoetal.,2014).
Hypertensionisassociatedwithdysregulatedplasmalevelsofadipokineadiponectinandleptin(Ghantousetal., 2018).Adiponectinisacardioprotectivehormonewithanti-atherosclerotic,anti-inflammatory,andanti-hypertrophic actionsduetoNOproductioninendothelialcellsphosphatidylinositol3kinase-dependentandAMP-activatedprotein kinasepathways(Ghantousetal.,2018).Physiologically,adiponectindecreasessmoothmusclecellproliferationand TNF-α expressioninmacrophages,andtheNOproductionrelaxesvesselsandexertsanti-inflammationandantithromboticeffectsonthevascularwall(Nigroetal.,2014).Epidemiologicstudieshavereportedthatlowadiponectinlevels areanindependentriskfactorforhypertension(Iwashimaetal.,2004;Jungetal.,2014).
Besides,leptinalsohasanimportantroleindevelopinghypertensioninobesity(Bell&Rahmouni,2016).High levelsofleptininduceatherosclerosisandvascularsmoothmusclecell(VSMC)hypertrophy.LeptinactsontheendotheliumbystimulatingthesynthesisofNOviaPI3kindependentactivationofAktandJanuskinase2orbyincreasing theNOproductioninVSMC(Becerriletal.,2019).
2.2.2.5Metabolicsyndrome Metabolicsyndromeisconsideredamultisystempathophysiologicalprocessandnotasinglediseaseentity,causedprimarilybyacombinationofadiposetissuedysfunctionandinsulinresistance(Reyesetal.,2020).
Severaldefinitionsofmetabolicsyndromehavebeenproposedbymanyorganizationsthathavesetcriteriaforthe diagnosis,whichincludeWHO,NationalCholesterolEducationProgram-AdultTreatmentPanelIII,andInternational DiabetesFederation(Hamjaneetal.,2020).Althoughthereareslightdifferencesbetweendefinitions,thecriteriaforall thesedefinitionsincludeabdominalobesitydeterminedbyincreasedwaistcircumference,elevatedbloodpressure, hyperglycemia,raisedtriglycerides,andloweredhigh-densitylipoprotein(HDL-cholesterol)(vanNamenetal.,2019).
Chroniclow-gradeinflammationhasbeenimplicatedamongthemajorfactorsindevelopingmetabolicsyndrome (Nigroetal.,2014).TheinflammatorystateisrepresentedbyelevatedconcentrationsofavarietyofinflammatoryregulatorssuchasC-reactiveprotein,TNF-α,resistin,IL-6,IL-8,visfatin,adiponectin,andleptin(Esseretal.,2014;Maury &Brichard,2010;Wisse,2004).Adiponectinmayhaveacentralroleindeterminingtheriskofmetabolicsyndrome (Caltonetal.,2013).Adiponectinisinverselyassociatedwithinflammatorystateandadiposity,whereasleptinispositivelyrelated(Coimbraetal.,2014;Maury&Brichard,2010).
2.2.2.6Cardiovasculardisease
CVDsarethenumberonecauseofdeathallovertheworld.Riskfactorsincluderaisedbloodpressure,hyperglycemia, dyslipidemia,overweight,andobesity(Forouzanfaretal.,2016;WHO,2018a).Obesity-associatedinflammationplays acriticalroleinthegenesis,progression,andCVDmanifestation(Alfaddaghetal.,2020).
Asdescribedbefore,obesity-associatedinflammationplaysamajorroleindevelopinginsulinresistance,which itselfconstitutesariskforCVDandstronglyassociateswithotherCVDrisks(dyslipidemia,hypertension)throughseveralpathophysiologicmechanisms(Duongetal.,2019).Macrophagesproducepro-inflammatorycytokines,suchas TNF-α,IL-6,andIL-1β,intheendothelium,promotingtheatherogenicprocess(Rashadetal.,2018).Thusthesecytokinesinducecellularadhesionmolecules’expressionintheendotheliumfacilitatingtheentrapmentofleukocytesand monocytesandinitiatingtheatherogenicprocess(Hamjaneetal.,2020).
Bothinsulinresistanceandimpairmentininsulinsecretionresponsestronglycorrelatewithcoronaryendothelial dysfunction,eveninpatientswithoutdiabetes(Lali ´ cetal.,2018).Theincreasesincirculatingfreefattyacidsobserved
ininsulinresistanceinducehepaticoverproductionofvery-low-densitylipoproteinsrichintriglycerides,whichhave becomeLDLduetotheremovaloffreefattyacidsbylipoproteinlipasepresentintheadiposetissueandothertissues. LDLismorepronetooxidation,enteringtheendothelium’sintima,leadingtotheformationoffoamcellscontributing toatherosclerosisdevelopment(Diffenderfer&Schaefer,2014).Furthermore,LDLparticlesarehighlycorrelatedwith CVD(Toth,2014)andendothelialdysfunction(Fordetal.,2009).
Besidesthat,insulinresistanceischaracterizedbyaclottingdiathesisduetoendothelialdysfunctionanddecreased NOproduction,increasingplateletadhesiveness,andincreasedcirculatingfibrinogenandplasminogenactivatorinhibitor 1(PAI-1)productionbyadiposetissue(Suslovaetal.,2015).Theeffectsofinsulinonthevasculaturearedeterminedby differentcellularsignalingpathwaysactivatedbystimulationoftheinsulinreceptor.Throughphosphatidylinositol 3-kinase(PI3K/Akt),insulinstimulatesNOproductionandcausesvasodilatation.However,thispathwayisinhibitedin insulinresistanceconditions,andthemitogen-activatedproteinkinase(MAPK)pathwayishyperactiveduetohyperinsulinemia.MAPKpathwaystimulatesmitogenicinsulinaction,includingexpressionofendothelin1,PAI-1,andotherinflammatorycytokinespromotingendothelialdysfunction(Manriqueetal.,2014).Themitogenicpathwaypromotes vasoconstriction,oxidativestress,cellgrowth,mitogenesisinVSMC,expressionofVCAM-1,ande-selectin(Kimetal., 2006;Shimizuetal.,2016).Also,insulinresistanceisassociatedwithincreasedsympatheticnervoussystemactivityand therenin-angiotensin-aldosteronesystem(Leongetal.,2015;Zhouetal.,2012).
2.2.3Cancer Cancer,definedasalargegroupofdiseasesthatcanaffectvarioustissuesofthebodywiththecommonfeatureofthe rapidproliferationofabnormalcellsthatcanspreadtootherorgansortissues,isamajorpublichealthchallenge (WHO,2020).Thisstatementcouldbeexplainedbythecontinuousincreaseincancerincidenceandmortality:Itis estimatedthatmalignanttumorsareresponsibleforoneinsixdeathsglobally,beingthesecondleadingcauseofdeath globally(WHO,2020).
Incontrasttootherchronicdiseases,wheretherearelackorreductionsincellfunction,cancersariseduetoan uncontrolledcelldivisionandfailureincellcyclecheckpoints.Carcinogenesisisacomplexmultistepandmultimechanismprocessconsistingofstagesofinitiation,promotion,andprogression.Theinitiationfrequentlyinvolvescarcinogenicbindingagents(asradiation,biological,pharmaceutical,andchemicalagents)totheDNA,generatingadducts ormutations(Fishbeinetal.,2020).Thesemutationsareresponsiblefortransformingthephenotypeofnormalinto cancerouscells,ensuringtheextensiveabilitytobeself-sufficientingrowthsignalsandcelldeathevasion.Theacquisitionofthistransformedphenotyperequirestheactivationofoncogenesand/orinactivationoftumorsuppressorgenes, triggeringintracellularsignalsforanincrementincellcycleproteinssynthesis,metabolicchanges,andapoptosisinhibition(Parsonsetal.,2016).
Thepromotion,thesecondstageofcancerdevelopment,occursinresponsetostimuli,inacertainfrequency,by promotingagentsininitiatedcells,leadingtoproliferation.Ingeneral,theseagentsarenotmutagenicbutcancreatean adequateenvironmentforcelldivisionandfurthermutationsofcancerouscells.Finally,thelaststageofcancerdevelopment,termedprogression,evolvesthemalignanttransformation,anirreversiblechangethatforwardsthecancerevolutionuntilmetastasis,themostharmfulconsequenceoftumordevelopment(Newkirketal.,2017).
Theincreasingknowledgeofcancerbiologyhasdemonstratedthatsomemechanismspredisposecancerinitiation,promotion,andprogression.CIandoxidativestresshavebeenlinkedtoallstepsofcarcinogenesis,actingtofuelavicious cycleinvolvingtheDNAandtissuedamage,intheactivationofoncogenesandepigeneticalterations,andalsothrough thestimulusofsomekeymediatorsofsignalingpathwaysinvolvedincancerdevelopment,ashypoxia-induciblefactor1α (HIF-1α),signaltransducerandactivatoroftranscription3,activatorprotein-1(AP-1),andnuclearfactor-erythroid2 p45-relatedfactor-2,beingthusconsideredfundamentalhallmarksofcancer(Hayesetal.,2020;Reuteretal.,2010).
2.2.4Inflammationandcancerdevelopment If,ontheonehand,theinfectiousdiseasesandchronicinflammationarerelatedtoapproximately25%ofcancercausingfactors(Murata,2018),theactivityofpro-inflammatoryfactorsinthetumormicroenvironment,ascytokines andinfiltratingimmunecells,influencestumorigenictransformationsandneoplasticprogression,asnewbloodvessels formation(angiogenesis),epithelialtomesenchymaltransition,metabolicchanges,andmetastasis(Fishbeinetal., 2020),turningtheinflammationanimportantmechanismintumorigenesis.
Thusthemaincellularmechanismsstudyinvolvedintheinterplaybetweeninflammationandcancerhasattractedthe scientificcommunity’sattentiontofurtherdevelopinganticancertherapies.Studiesreportthattumorssupplantedthe
pathwaysevolvedtomediateimmunitytoinfectionandregenerationtowardtheirbenefit(Greten&Grivennikov,2019).
TheincrementinthetranscriptionNF-κBactivityincancerouscellsplaysacriticalroleinthisresponsethroughthe downstreamofmultiplepro-inflammatorycytokinesrelease,prostaglandinsynthesisenzymes,NOS,andotheractivation signalstogrowth(Fishbeinetal.,2020).Thetumorsuppressorproteinp53,whosegeneisthemostcommonlymutatedin initiatedcells,isanantagonistofNF-κB,whichsignifiesthatthelossoffunctionalp53resultsinanincreasedexpression ofNF-κBandcontributestotheirhighestactivityincancerouscells(Greten&Grivennikov,2019).
Theactivationofoncogenes,agreatdegreeofplasticityinthetumormicroenvironment,andtheconstantcellinsultsin cancercauseastrongperturbationoftissuehomeostasis,increasingcytokineproductionandchemokinesbytheowncancerouscells.Thesealterationsstimulatetherecruitmentofsomeimmunecellstothetumormicroenvironment(asmonocytes, macrophages,neutrophils,andinnatelymphoidcells),andpersistentactivationoflocalinflammatorysignals,togetherwith thefailureofpro-resolvingmechanisms,feedachronicinflammation-inducedsignalingandimmunecellsrecruitment,ina processreferredas“cancer-promotinginflammation”(Fishbeinetal.,2020;Greten&Grivennikov,2019).
Thispro-inflammatoryphenotypeobservedincancerinvolvestheinnateimmunesystem,adaptiveimmunecells, andstromalcellsasfibroblasts,endothelialcells,andmesenchymalcellsthatactsynergically.Themacrophagesare amongthemostabundantimmunecellsinthetumormicroenvironmentandcanexhibitM1andM2phenotypesindifferenttumorstages(Galdieroetal.,2013).Atinitiationandearlypromotion,thesecellsappeartoundergoclassical activation(M1polarization)throughstimuluspromotedbyIFN-γ,TNF-α,andlipopolysaccharidesthatisrecognized byreceptorsasTLR-2and4,triggeringasignalingpathwaythatculminateswiththesecretionofclassicalproinflammatorycytokines,chemokines,andeffectormoleculesincludingIL-1β,IL-6,TNF-α,IL-23,andiNOS.These factorspromotechemoattractionofcellsfromadaptiveimmunity,asThelperlymphocytes(CD4 1 )amplifytheinitial inflammatoryresponsebyIL-4secretion,animportantfactortoM2phenotypeswitch,frequentlyobservedwhen tumorsaccumulatelacticacidandinhypoxicregions(Shalapour&Karin,2015).OncemacrophagesM2produce VEGF,afundamentalfactorinangiogenesissignalization,aswellasTGF-β andarginase-1,whichareimmunosuppressiveagents,itisproposedthatthesecellsaremostimportantinthelaterstagesoftumorgrowth(Shalapour&Karin, 2015).Interestingly,theimmunosuppressiveeffectofTGF-β cytokineisfrequentlylostincancercellsduetomutations intheTGFBRIIgene,andtheTGF-β axisplaysanextensiveroleinepithelial-mesenchymaltransition,contributingto cancerinvasiveness(Piotrowskietal.,2020).
TABLE2.3 Specificfeaturesandinflammatoryprofileofmostprevalenttumor-typesworldwide.
Cancer type
Mostcommoncharacteristicsbytumor type
Lung cancer -MutationsintheEGFRreceptorandALKgene; reductioninalveolarbarrieragainstdamage.
Breast cancer -Incrementonsteroidhormones,mainly oestradiol;increaseinactivityofestrogen receptors(asERα and β).
Colorectal cancer -HyperactivationofWNTpathway;damageto theintestinalbarrier;bacterialtranslocationand dysbiosis.
Prostate cancer -Sexsteroidhormonalimbalance;incrementin androgenreceptoractivity,andandrogendependentprofile,increaseinestrogen receptorsactivity(ERα and β).
InflammatoryprofileReferences
m cytokines:IL-6,IL-10,IL-8; k phagocytic abilityofalveolarmacrophages; m macrophages M2and k macrophagesM1; k iNOSexpression.
OrozcoMorales etal.(2016)
m CRP; m TLRsexpression; m IKKε expression,a centralkinaseinNF-κBpathway. Bhatelia etal.(2014)
m tumor-infiltratingmicrobesincreasethe chemotaxisofcellsfrominnateimmunity; m cytokines:IL-6,TNF-α,IL-1β,IL-17; k AIM-2and NLRP6expression,oneofthefactorsrelatedto inflammasomeassembly.
m cytokines:IL-17,IL-6,IL-1β,IL-10,IL-8,and TNF; m CD4 1 Tcells,mainlytheTh17 phenotype; m COX-2activity; m inflammatory cellinfiltratesintheregionofprostaticatrophy, referredtoas“proliferativeinflammatory atrophy”.
Lasryetal. (2016)
Sfanosand deMarzo (2012)
IM2,Interferon-inducibleprotein;ALK,anaplasticlymphomakinase;COX-2,Ciclo-oxygenase-2;CRP,Creactiveprotein;EGFR,epidermalgrowthfactor receptor;IKKε,IκB-Kinase-epsilon; iNOS,induciblenitricoxidesynthase;NF-κB,factornuclearkappaB;NO,nitricoxide;TLRs,Toll-likereceptors;TNF-α, tumornecrosisfactor.