


Rectal spacers are biodegradable materials used to temporarily create space between the prostate and rectum. They can be made of polyethylene glycol (PEG)-based material or hyaluronic acid.
They are injected into the space between the Denonvilliers’ fascia (tissue layer covering the posterior prostate) and the rectum under transrectal ultrasound guidance and remain in place for approximately 3–6 months.
It is then naturally broken down and cleared by the body.
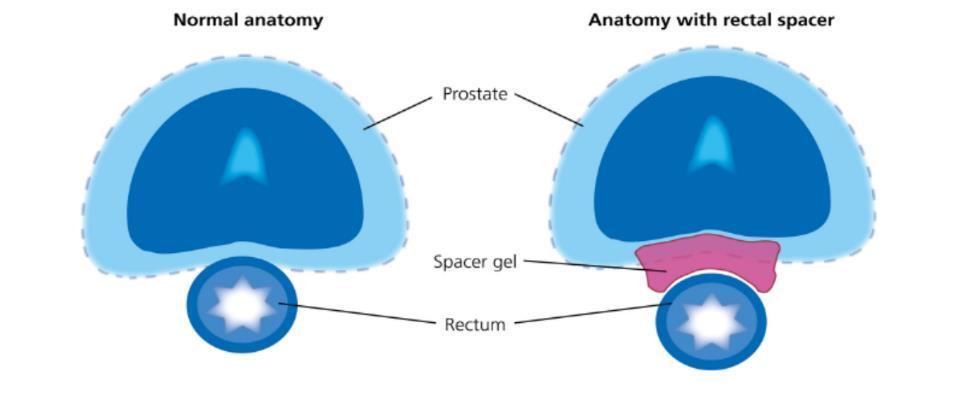
During prostate radiotherapy, high-dose radiation can unintentionally affect nearby organs, especially the rectum, leading to:
• Chronic rectal pain
• Rectal bleeding
• Changes in bowel habits
• Faecal urgency or incontinence
Rectal spacers reduce these risks by:
• Increasing the space between the prostate and rectum
• Decreasing rectal radiation exposure
• Reducing the risk of rectal toxicities
It is performed in the operating theatre under anaesthesia.
A transrectal ultrasound probe is inserted to visualise the prostate, and a needle is placed through the skin of the perineum for injection of the rectal spacer.
Do note that there is a possibility that the rectal spacer insertion may not be able to proceed at the time of surgery due to difficulty encountered during the procedure, which may be due to the extent of prostate cancer.
Post-procedure imaging (CT or MRI) is usually scheduled thereafter to confirm gel placement before the initiation of radiotherapy.
• Mild perineal discomfort or pressure sensation (typically selflimiting)
• Minimal bleeding or spotting from the perineum
• Rarely, temporary urinary symptoms (e.g., hesitancy, frequency)
The procedure is generally well-tolerated. Potential risks include:
Common (1–5%)
• Mild perineal discomfort or bruising
• Transient urinary frequency or urgency
• Minimal bleeding from needle puncture site
• Urinary retention
Uncommon (0.1–1%)
• Rectal wall penetration or hydrogel injection into rectal submucosa
• May cause pain, rectal inflammation, and rarely require early spacer removal
• Allergic or inflammatory reaction to the space material
Rare but serious (<0.1%)
• Peri-rectal or prostatic abscess formation
• Recto-urethral fistula
• Needle tract infection or sepsis
• Incorrect gel placement (e.g. in urethral wall or lateral to prostate) → may reduce effectiveness of radiotherapy protection
• Embolic events due to misplacement of spacer material
You may be asked to stop blood-thinners or anticoagulants (e.g., aspirin, warfarin, clopidogrel) prior to procedure and after the procedure
• Most patients can go home the same day
• Resume normal activities after 24–48 hours
• The gel will be absorbed naturally after a few months
Please go to the Emergency Department or contact your care team if you experience:
• Fever (≥38°C), chills
• Severe perineal pain or swelling
• Rectal bleeding
• Difficulty urinating or urinary retention
• Foul-smelling discharge from rectum or perineum
References:
1. Mariados N, Sylvester J, Shah D, et al. Hydrogel spacer prospective multicenter randomized controlled pivotal trial: Dosimetric and clinical effects of perirectal spacer application in men undergoing prostate image guided intensity modulated radiation therapy. Int J Radiat Oncol Biol Phys. 2015 Aug 1;92(5):971–7.
2. Hamstra DA, Mariados N, Sylvester J, et al. Continued benefit to rectal separation for prostate radiation therapy: Final results of a phase III trial. Int J Radiat Oncol Biol Phys. 2017 Apr 1;97(5):976–85.
