
Catalyst
A University of Utah Department of Chemistry Publication I 2025

Elements of Discovery

Welcome to the 2025 edition of Catalyst, our annual celebration of the people, partnerships, and discoveries that define the Department of Chemistry at the University of Utah.
This year’s issue emphasizes what makes our department truly exceptional: collaboration. Throughout research groups, disciplines, and generations, our faculty, students, and alumni are working together to tackle urgent challenges—from sustainable materials and energy innovation to molecular medicine and environmental stewardship. These efforts not only advance science but also help create a more equitable and resilient future; they are highly collaborative, crossing disciplinary boundaries and generations of mentorship.
Across politics, technology, and global markets, critical materials have become a focal point of public discussion. Understanding why requires looking beyond the headlines to the chemistry that makes these materials indispensable. In this issue’s feature article, we explore the chemistry behind the elements and compounds that power our technology—and the vulnerabilities they present. From rare earth metals essential to clean energy and electronics, to molecules like phosphate that support global agriculture, critical materials are both crucial and vulnerable. Faculty across the Department of Chemistry lead efforts to understand, recover, and rethink these resources through advanced research in spectroscopy, catalysis, molecular design, and sustainable recovery. Their work not only deepens our scientific knowledge but also paves the way toward a more resilient and circular materials economy. These efforts are the Elements of Discovery that each of us contributes to, in diverse ways, to address scientific grand challenges and promote sustainability (Page 4).
Advances in science are powered not only by discovery, but by the generosity and engagement of our donors and alumni. Whether you mentor students, fund scholarships and fellowships, or join us at events, your support strengthens the vibrant, welcoming community that makes our department a hub for innovation and learning.
As you read through these pages, I hope you’ll feel the energy and purpose that motivate our work. From undergraduate researchers making their first discoveries, to faculty pushing the boundaries of knowledge, each story reflects our shared commitment to curiosity, excellence, and impact.
Thank you for being part of our community. Let’s keep discovering. Join us in advancing science that matters.
Warmly,
Aurora Clark Professor of Chemistry and Department Chair Department of Chemistry, University of Utah
Cover
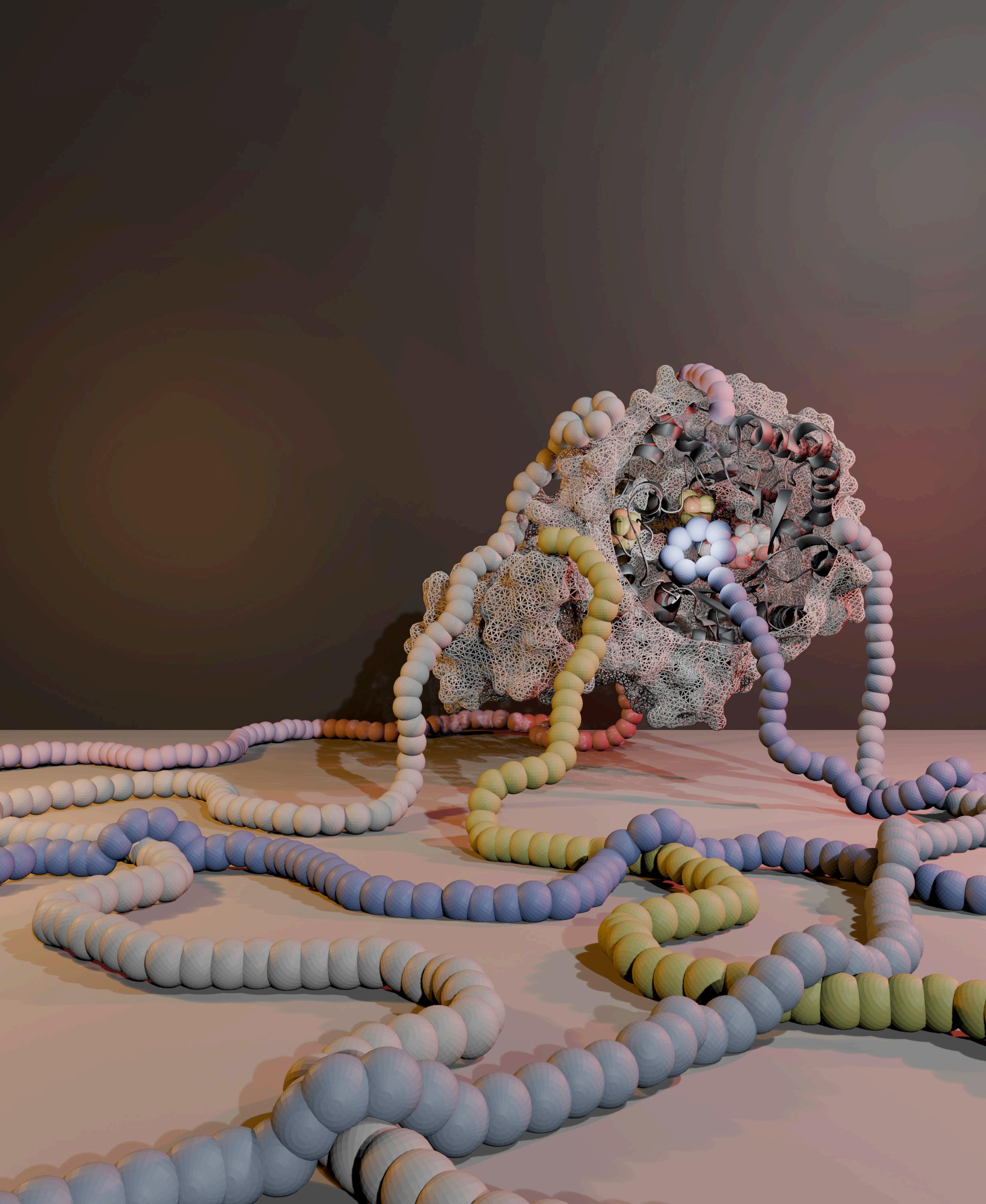
Macrocyclic and polymacrocyclic peptides are key players in biology because they provide structural constraints that increase affinity, efficacy, and stability. The recent explosive rise of peptides in treating obesity and diabetes highlight the prominent role that peptide therapeutics are likely to continue to play in the future. The Bandarian Lab studies how enzymes can be used to produce complex peptide-based architectures. The image shows a system that the lab studied and demonstrated its use to controllably macrocyclize peptides. The method was applied to analogs of the three incretin mimics, Ozempic, Tirzepatide, and Retratrutide, to illustrate the utility of the method in drug discovery applications. The paper describing the work is in press in ACS Bio & MedChem Au (https://pubs.acs.org/doi/10.1021/acsbiomedchemau.5c00152). The cover art was generated by Bandarian Lab graduate Jake Pedigo, who is first author on the manuscript.
Department of Chemistry Awards and Recognitions
Research:
Matthew Sigman - Spanish Royal Society of Chemistry GEQO Fellow
Peter Armentrout - IBBI Bowers Award
Qilei Zhu - Ralph E. Powe Junior Faculty Enhancement Award
Gabe Nagy - 2025 Rising Star in Measurement Science Award & Pacific NW American Vacuum Society Emerging Leader Award
Cynthia Burrows - ACS Organic Division Reaction Mechanisms Conference Lectureship Honoree & ETH-Zurich Albert Eschenmoser Prize
Long Luo - Pittsburgh Conference Achievement Award
Joel Harris - American Chemical Society Roland F. Hirsch Distinguished Service Award
Bethany Buck - U Faculty Fellow Award
Vahe Bandarian - U Founders of the Year
Teaching:
Janis Louie - Utah Science Teaching Association Outstanding Higher Education Educator
Rodrigo Noriega - Robert W. Parry Teaching Award
Jennifer Shumaker-Parry - U Distinguished Teaching Award
Matthew Sigman - U Distinguished Mentor Award
Ryan Vanderlinden - W.W. Epstein Outstanding Educator Award
Service:
U Environmental Health & Safety Partner Awards
Ryan Looper
James Muller
Special Thanks: Students & Faculty of the Department of Chemistry
Check out our website and social media accounts:

Credits:
Writers / Editors / Design / Photography:
Todd Anderson, Aurora Clark, Nelly Divricean, Stella Madsen & David Pace
Contact: Nelly Divricean, Director, Strategic Engagement nellyd@chem.utah.edu
What is Critical About Critical Materials

Materials are deemed critical when they are vital to modern technologies yet vulnerable to supply disruptions caused by scarcity, geopolitical concentration, or the absence of suitable alternatives. These include elements such as the lanthanides, actinides, and transition metals, whose complex electronic structures give rise to exceptional magnetic, catalytic, and optical properties—making them indispensable for electric vehicles, wind turbines, fuel cells, chemical production, electronics, and nuclear energy. Critical materials also encompass molecules like phosphate, which are essential for global agriculture and food security. Whether elemental or molecular, researchers in the Chemistry Department are advancing a fundamental scientific understanding of these materials that is crucial to enabling their efficient reuse and recycling, and for designing sustainable replacements that can reproduce their remarkable behaviors.
On the Behavior of Electrons and Chemical Bonding:
The partially filled d- and f-subshells of transition metals, lanthanides, and actinides underpin their remarkable electronic, magnetic, and catalytic properties—and drive global demand that reaches hundreds of thousands of tons annually for clean energy, high-tech manufacturing, and defense applications. The electrons in these subshells experience strong nuclear attraction and move at relativistic speeds, leading to pronounced relativistic effects and strong electron correlation. These complexities make their behavior exceptionally difficult to describe accurately using either theoretical models or experimental measurements.
One of the most fundamental properties of critical metals is the strength of the bonds they make with other elements, which derives from the specific electron occupation of atomic orbitals (which determines the electronic states of the element) and the overlap those orbitals have during bond formation. Research in the Armentrout and Morse groups is aimed at understanding both of these. Because of the complex distribution of electrons, the number of different possible arrangements of the electrons is huge, which makes traditional spectroscopic study too complex to analyze. Instead, the Morse group uses pulsed laser ablation to get the metal atoms into the gas phase, where they are picked up in a stream of helium and react with a reaction partner to make the molecule of interest.

They are then carried downstream into a time-of-flight mass spectrometer, where one or two lasers are used to probe their spectroscopy. The key is to use a combination of photons to ionize the molecule, so that the ions can be detected in a mass-specific manner, allowing a spectrum to be recorded for the specific molecule of interest. These same lasers can be used to add enough photon energy to cause these molecules to fall apart, where the amount of energy input is precisely measured until dissociation occurs – thus measuring the Bond Dissociation Energy (BDE).

A complementary method for measuring the reactivity of critical actinides, lanthanides and transition metals is guided ion beam tandem mass spectrometry, an instrument and methodology invented by Professor Peter Armentrout. There, the absolute reaction probability of metal reactions with different molecules is measured as a function of the kinetic energy available. The precision of the BDE measurements and reaction threshold energies made in the Armentrout and Morse groups is unprecedented – having many uses to the broader scientific community.
First, the measured values can be used as a benchmark to new theoretical models of chemical bonding for critical metals.
Computational chemistry is often used to predict the behavior of critical metals, however the significant relativistic effects and electron correlation require new theoretical methods to be developed and it is unclear if these methods are correct without comparison to highly accurate experimental measurements. Second, the trends in bond energies as a function of different elements reveal new periodic trends in bonding that can be leveraged for the development of new materials and to predict and understand variations in chemical reactivity. For example, metal sulfides and metal selenides are inorganic chalcogenides, where the anion (S²⁻ or Se²⁻) plays a key role in determining structural, electronic, and optical properties. The subtle differences in electronic properties of S vs. Se translate into different technological strengths.
Sulfides dominate in energy storage, catalysis, and environmental applications due to their abundance, stability, and robustness. Selenides excel in semiconductors, photovoltaics, thermoelectrics, and optoelectronics, where tunable band structures and strong light absorption are key. The Morse group has examined the correlation in BDEs found in comparing the metal sulfides and selenides, demonstrating clearly that the metal sulfides are bound 15.9% more strongly than the corresponding metal selenide, across a wide variety of different metals.
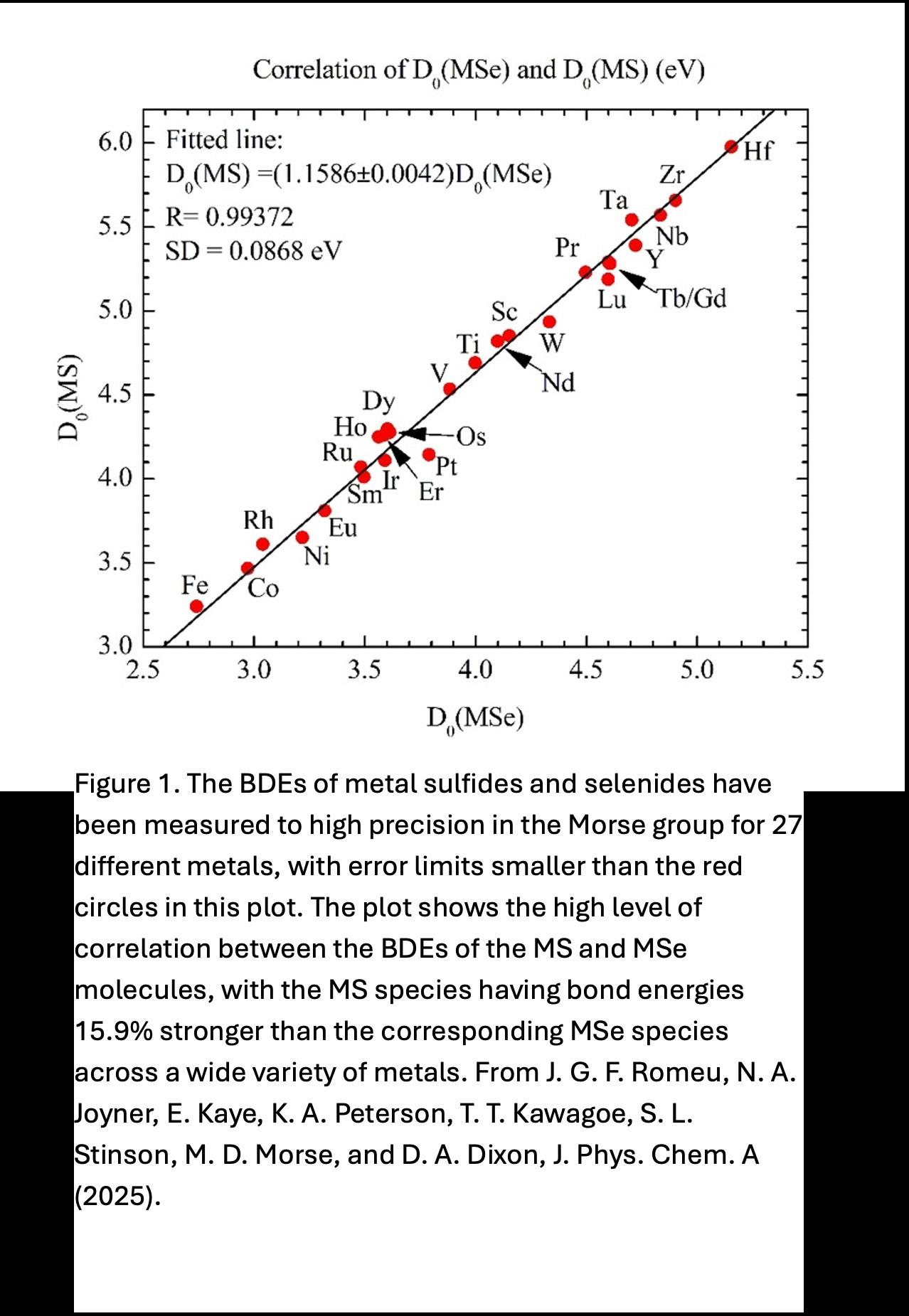

In combination, the research in the Armentrout and Morse groups reveal how electronic configuration—particularly the occupancy and availability of d- and f-orbitals—governs molecular stability, bonding trends, and reactivity across the periodic table. Beyond their intrinsic scientific importance, such studies also provide the data necessary to test and refine modern computational chemistry methods, enabling predictive modeling of complex systems involving the lanthanides and actinides that are vital for catalysis, energy applications, and materials science.

From Waste to Resource: Innovative Chemistry for Critical Element Recovery
The lifecycle of many critical materials remains largely linear— raw materials are extracted or synthesized from limited sources, used in industrial and technological applications, and ultimately discarded as waste—depleting finite reserves and leaving lasting environmental impacts.
Phosphate and gadolinium exemplify this unsustainable pattern. Phosphate, essential for global agriculture, is mined from a few geopolitical regions such as Western Sahara, with reserves expected to last only about a century. Most is used in fertilizers, but runoff into waterways causes eutrophication and dead zones, and current treatment methods remove rather than recover it. Gadolinium, a rare earth element vital for MRI diagnostics, faces a similar fate: hundreds of tons are released annually into wastewater after medical use, with only a small fraction removed by conventional treatment, leading to measurable accumulation in surface waters. These examples highlight the urgent need for chemical innovations that close the materials loop—developing selective methods to recover, recycle, and repurpose critical materials so that waste streams become renewable resources. Two complementary research strategies—molecular recognition and electrochemical recovery —illustrate how chemistry can meet this need.
The Pierre group has pioneered the development of stimuli-responsive molecular receptors for phosphate capture and release. Traditional host–guest separation systems rely on static binding affinities, forcing a compromise between strength of binding and ease of release. This tradeoff limits their effectiveness at the low phosphate concentrations found in wastewater. By contrast, the Pierre group’s approach moves beyond this paradigm, introducing electrostatic allosteric control that allows binding affinity to be switched on or off with external stimuli such as light, redox potential, or pressure. These tunable receptors, integrated into “phosphate-selective sponges,” can selectively capture phosphate at parts-per-trillion levels—even in the presence of competing ions—and release it on demand for recovery as struvite, a valuable fertilizer. This approach not only prevents environmental eutrophication but also transforms waste into a renewable resource, demonstrating a circular strategy for phosphorus management.
In parallel, the Luo group is developing electrochemical filtration systems for recovering gadolinium directly from patient urine following MRI procedures. Their system uses water electrolysis to create steep pH gradients and electric fields within a membrane filter. These gradients promote demetalation of gadolinium-based contrast agents, enabling electrophoretic migration and precipitation of Gd³⁺ as insoluble GdPO₄ and Gd(OH)₃. Using gadopentetate dimeglumine (GdDTPA) as a model, the group achieved ~70% trapping efficiency from both artificial and real urine samples. The gadolinium can then be recovered from the filter by thermal treatment, enabling direct recycling of this high-value element and mitigating its environmental footprint.

In combination, the work of Pierre and Luo aim to transform waste streams into sources of critical materials, closing the loop between use and reuse. As global demand for essential resources like phosphate and gadolinium continues to rise, such advances in chemical separation science will be central to securing the materials foundation of future technologies while protecting the environment.

From Critical to Sustainable: Redefining Material Design
Developing new strategies to use critical materials more efficiently—or replace them entirely with earth-abundant alternatives—is an urgent scientific priority that several of our faculty are addressing head-on. In creating new catalysts for chemical transformations and materials that harvest, store, or transfer energy, we emphasize three essential attributes: functionality, scalability, and sustainability.
Catalysts drive nearly one-third of the global economy, enabling essential processes in energy, chemical synthesis, and pollution mitigation. Yet, many of the most active and selective catalytic systems rely on critical precious metals—Pt, Ir, Pd, Ru, Rh, and Au. One promising approach is to drastically improve metal-use efficiency by reducing particle size so that all atoms are catalytically active. The Anderson group pushes this strategy to the extreme, creating sub-nanometer clusters composed of just 1–20 atoms. At this scale, every atom resides at the surface, fully accessible to reactants, and the clusters’ under-coordinated nature enhances their intrinsic reactivity.
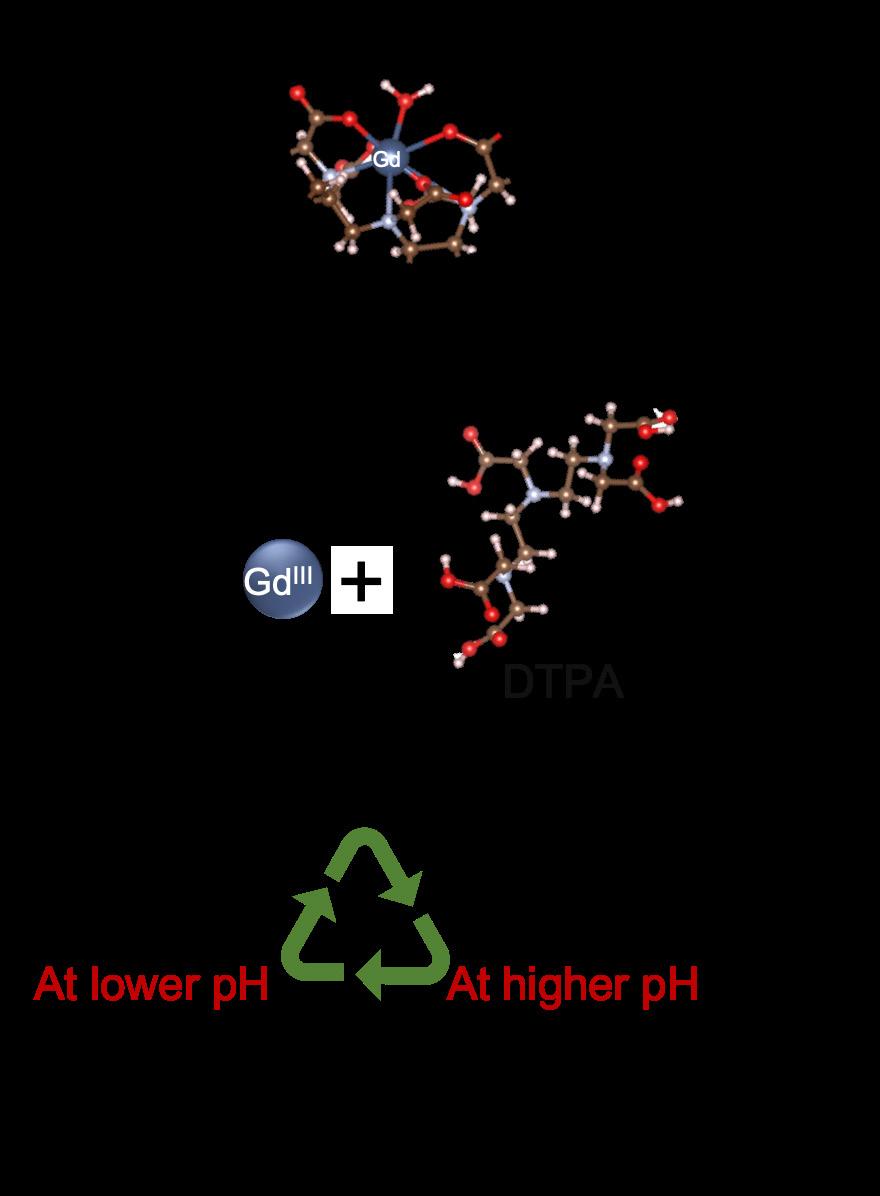
Figure 2. Electrochemical filtration method for recovering gadolinium from MRI patient urine by creating a steep pH and strong electric field to separate Gd3+ and its ligand of the Gd contrast agent, and the precipitate Gd3+ as insoluble GdPO4 and Gd(OH)3
The Anderson group also develops methods to stabilize and electronically tune these clusters through interactions with the underlying support. In electrochemical systems, for example, the group modifies electrode surfaces— either by high-energy deposition that creates defect binding sites or by implanting nitrogen or reactive metal atoms that strongly anchor the clusters. These strategies not only prevent agglomeration but can enhance catalytic activity by up to an order of magnitude. By ensuring that every precious-metal atom is utilized at maximum efficiency, this research reduces material cost and supply limitations, providing a scalable path toward sustainable catalysis.
While catalysis research focuses on minimizing reliance on rare metals, other efforts seek to replace critical inorganic materials entirely. The Bischak group investigates organic semiconductors—molecular materials composed of abundant elements such as carbon, sulfur, oxygen, and nitrogen—that can perform electronic and optoelectronic functions traditionally dominated by semiconductors made of critical elements. These organic materials offer distinct advantages: they are lightweight, flexible, and can be processed from solution using scalable, low-cost manufacturing methods such as roll-to-roll coating or inkjet printing.

The Bischak group optimizes conductivity, optical response, and stability using high-throughput robotic fabrication platforms integrated with machine learning to rapidly explore vast processing parameter spaces. This data-driven approach identifies the conditions that optimize performance and reveals the underlying structure–property relationships governing function in soft electronic materials. By connecting molecular organization to device behavior, this work accelerates discovery and lays the foundation for next-generation organic electronics built entirely from earth-abundant elements.

In a similar vein, the Stolley group, an undergraduate-focused research lab, contributes to the development of advanced conducting polymers that can achieve functionalities traditionally reliant on rare or magnetic materials. Working in collaboration with physicists and materials chemists,
Polyacetylene, the first organic electrical conductor, revolutionized the understanding of electron transport in molecular systems and earned the 2000 Nobel Prize in Chemistry. By introducing chirality into this archetypal material, the Stolley group and collaborators are exploring how structural asymmetry couples to optoelectronic and spintronic behavior—a frontier known as chiral-induced spin selectivity (CISS). Their breakthrough was the development of a new chiral polymerization catalyst that uses common, inexpensive chiral ligands to synthesize chiral polyacetylene under simple and scalable conditions, overcoming the impracticality of earlier synthetic routes. This discovery opens access to a new class of functional materials that could enable spintronic and optoelectronic devices based entirely on organic, earth-abundant components.
Together, these research efforts represent a multifaceted strategy for achieving sustainability in advanced materials. By combining molecular-level precision, data-driven discovery, and cross-disciplinary innovation, this work moves us closer to a circular materials economy that meets technological demands without depleting the planet’s critical resources.
Photos taken in the U Chemistry Laboratories of professors: Scott Anderson, Peter Armentrout, Connor Bischak, Long Luo, Michael Morse, Valerie Pierre and Ryan Stolley
U Chemistry 2025 Distinguished Alumni

by DAVID
The Department of Chemistry at the University of Utah established the Distinguished Chemistry Alumni Awards in 2011 to recognize and honor our most prominent and successful alumni. We celebrated three of our outstanding alumni on September 29, 2025 in the Department of Chemistry: Dr. Hyung Kyu Shin, Dr. Sally Hunnicutt and Dr. Michael Hunnicutt.
The Big Reveal: Honoring Mike Hunnicutt
Mike Hunnicutt’s journey from a first-generation college student to a renowned chemist and educator demonstrates the importance of mentorship, perseverance, and the transformative power of science. Recently honored with the Distinguished Alumni Award by the University of Utah’s Department of Chemistry, Hunnicutt reflects on a career influenced by key relationships and a passion for discovery. Born to a U.S. Army Master Sergeant and a Japanese mother, Hunnicutt grew up in Florida and found his calling in chemistry thanks to a high school teacher. At Duke University, he immersed himself in lab work and met influential mentors like Dr. Charles Lochmüller and Dr. Joel Harris, who guided him into graduate studies and post-doctoral research. His work with Harris on luminescent probe molecules and surface chemistry laid the foundation for a distinguished career in industry and academia.

After earning his Ph.D. in 1984, Hunnicutt joined Procter & Gamble and later Wyeth Pharmaceuticals, rising to senior leadership roles in analytical sciences and regulatory compliance. In 2011, he transitioned to teaching at Virginia Commonwealth University, where he focused on lab instruction and student mentorship —his way of “paying it forward.” CATALYST I Department of Chemistry
Now retired, Hunnicutt continues to serve Duke University and local nonprofits, carrying forward the legacy of those who believed in him. His story comes full circle with a poignant memory: learning on his first day at Duke that his father—who had grown up in Durham—had quietly arranged financial support for his education. That moment, Hunnicutt says, was “the big reveal.”

Read the full stories about Dr. Hyung Kyu Shin, Dr. Sally Hunnicutt and Dr. Michael Hunnicutt here:
Photo: Dr. Michael Hunnicutt and Professor Joel Harris
PACE


Honored with multiple teaching awards and the naming of the Sally Hunnicutt Science Hub at VCU, she also served as Associate Chair and helped design VCU’s new STEM building. Now professor emerita, she continues to champion science education through Richmond’s Blue Sky Fund. Her story is a testament to the lifelong impact of curiosity, resilience, and student-centered teaching.
A Life in Chemistry: The Enduring Passion of Hyung Kyu Shin
At 92, Dr. Hyung Kyu Shin still arrives at his University of Nevada, Reno office by 7 a.m.—a routine unchanged for decades. “My wife complains,” he says with a smile, but for Shin, chemistry is more than a career; it’s a lifelong passion. Born in South Korea, Shin’s journey to becoming a pioneering theoretical chemist began with his sister’s acceptance to the University of Utah, when he decided to follow along. There, he studied under working with J. Calvin Giddings and the legendary Henry Eyring, whose mentorship shaped Shin’s approach to research: hands-on, curious, and deeply engaged. Shin earned both his bachelor’s and Ph.D. at Utah by 1961, focusing on vibrational energy transfer in molecular collisions—a field with wide-reaching implications from pharmaceuticals to petrochemicals.
“Energy transfer is essential for the chemical reaction,” he explains.

After postdoctoral work at the National Bureau of Standards and Cornell, Shin joined UNR in 1965, serving twice as department chair and publishing over 210 papers. In 1973, he was named the university’s first Outstanding Researcher of the Year.
When he retired in 2000, Shin insisted his position be filled by a younger scholar. “Department has to grow,” he said. He continued his research independently, driven not by titles but by purpose. His wife, Young Ai, a former WHO nurse, notes that unlike other retired professors, Shin still comes home smiling, energized by his work. Even in the AI era, he remains undaunted. “We had to dig out all the answers ourselves… But these days you don’t have to do that. Just ChatGPT,” he quips. “You have to have the purpose in life,” Shin reflects. For this humble pioneer, that purpose is still the molecular dance of chemistry.
Photo: Dr. Hyung Kyu Shin and his wife, Young Ai Shin
Photo: Sally Hunnicutt and Former Chemistry Professor Chuck White
From Curiosity to Chemistry: Yexalen’s Journey of Discovery and Representation
by STELLA MADSEN

In a quiet lab tucked inside the University of Utah’s Chemistry Department, Yexalen Barrera is chasing molecules and rewriting narratives. Yexalen is a third-year graduate student, studying physical chemistry in the Morse Lab. She measures the bond dissociation energies of diatomic and triatomic lanthanide metals bonded to a p-block element utilizing resonant two-photon ionization spectroscopy.
A first-generation Ph.D. student, Yexalen is transforming serendipity into science and reshaping the narrative for her community.
Born and raised in Salt Lake City, she’s a local with deep roots in Mexico, a second-generation immigrant whose academic path is as bold and intentional as the question she’s always asked: “Why?”

“My internal passion is worth the fight.” —Yexalen, on navigating grad school
“I was labeled a traviesa (troublemaker) growing up,” she laughs, recalling her childhood curiosity. “I couldn’t stop asking why. Why is it done this way? Can it be done differently?” That instinct, once a source of mischief, has become Yexalen's greatest asset as a Ph.D. researcher.
Her fascination with chemistry began in fifth grade, sparked by the blurry line between physical and chemical changes. But it was in high school that her passion was truly ignited. “I read every chapter in the textbook twice,” she says. “I just had to know everything.” That hunger for understanding led her to the University of Utah, where she initially planned to pursue medicine—until research changed everything. Now, Yexalen is immersed in the intricate world of bond dissociation energies (BDEs), studying lanthanide-containing molecules using resonant two-photon ionization spectroscopy. Her work provides critical benchmarks for computational chemists and reveals periodic trends that deepen our understanding of molecular stability. And sometimes, science surprises her. While studying the PrN molecule, an unexpected peak on the mass spectrum led her to discover PrNC— an accidental breakthrough born from sandpaper and serendipity.
“Science is a community effort.” — Yexalen, on mentorship and collaboration
But Yexalen’s impact reaches far beyond the lab bench. As the first in her family to graduate high school, college, and enter a Ph.D. program, she carries the weight and pride of representation. “My mother sacrificed her freedoms, pride, and future to give her daughters better opportunities,” she shares. “Experiences like hers often remain silent in academic circles. I want to change that.”
Caroline Sauoma was among the first faculty with whom she shared her background, establishing a meaningful connection early on. Ryan Steele’s computational class stood out for its dynamic and engaging approach, sparking enthusiasm for complex material. Aurora Clark likewise brought vibrancy to her teaching and has provided steady support throughout her PhD journey, reinforcing the department’s commitment to mentorship and student success. Tom Richmond, though never her professor, has been a tireless advocate for her.

And Michael Morse, as well as alum Dakota M. Merriles, has become a guiding light in her research journey. Now, Yexalen is paying it forward. As a mentor herself, she’s helping younger students navigate the world of research, just as others once did for her. “Science is a community effort,” she says. “Mentorship has made me a better researcher.” Outside the lab, Yexalen finds joy in crafting—jewelry, cross-stitch, clay magnets—and catching up with friends over matcha or chai.
“I genuinely love the research I do, making the task of going to work not feel like work.”, she says. “But I believe in taking breaks to balance the stress.” Her advice to future grad students? “No one can teach you passion. It’s what carries you through the inevitable feelings of inadequacy. My internal passion is worth the fight.” As she reflects on her journey from curious fifth grader to groundbreaking researcher, Yexalen is clear-eyed about her purpose. “I’m here to challenge norms, spark new ideas, and change the narrative for my community. Chemistry lets me do all of that.”

Summer of Discovery: Undergraduates Dive into Research at the U
The Chemistry Department’s REU program offers students a transformative experience where mentorship, hands-on science, and community come together.
Every summer, the University of Utah’s Chemistry Department opens its doors to a new cohort of undergraduate researchers through the prestigious Research Experience for Undergraduates (REU) program. Designed to immerse students in full-time research, the REU offers more than just lab work—it’s a launchpad for future scientists.
Participants, drawn from both the U and universities across the country, are placed directly into faculty-led research groups tackling real-world challenges in energy, health, environment, and education. From day one, students are treated not just as learners, but as contributors.
“It was really cool to have people to look up to and answer my questions,” said Samantha Stinson, one of this year’s participants. “They helped me navigate graduation and research.”
“It was an opportunity to become my own scientist and not just a student.” — Samantha Stinson, REU participant

Images courtesy of Yexalen Barrera and Professor Michael Morse

Samantha, a senior undergraduate chemistry student and president of the University of Utah American Chemical Society student chapter, joined Professor Michael Morse’s lab. There, she worked with advanced instrumentation to measure bond dissociation energies of transition metal cyanides and isocyanides. She described the experience as a turning point. “It was an opportunity to become my own scientist and not just a student.”
“The
REU
gave me the space to fully focus
on research without distractions.” — On the difference between summer research and the academic year
Beyond the lab, REU students attended weekly breakfast seminars where they presented their research to peers, boosting their confidence in scientific communication. They also participated in campus-wide social events like game nights, ice cream socials, and more, encouraging friendships and collaboration across disciplines. Graduate students and faculty mentors play a crucial role in the program’s success. Their guidance helps students develop essential skills in data analysis, modeling, and scientific writing—skills that will benefit them in graduate school and beyond.
WHAT IS REU
Program Name: Research Experience for Undergraduates (REU) Duration: 10 weeks, full-time summer research Who Can Apply: Undergraduates from any U.S. institution Focus Areas: Chemistry research in energy, health, environment, education Highlights: Mentorship, seminars, social events, hands-on lab experience
“Having that professional setting and the opportunity to do research from nine to five every day was a game-changer,” Samantha reflected. “During the school year, I’d squeeze in lab time between classes. But the REU gave me the space to fully focus.” As another summer concludes, the Chemistry Department celebrates the growth, resilience, and curiosity of its REU scholars. Applications for next year’s program will open soon, and instructions will be posted on our departmental website. Professor Rodrigo Noriega stated, “Firsthand lab experience for undergraduate students has become a focus in the College of Science, as it naturally connects our research and education missions. With generous and ongoing support from the National Science Foundation and the Chemistry Department, our REU program has a strong tradition of enabling talented, motivated undergraduates to participate in discovery and expand scientific knowledge—the next generation of researchers is already making their mark.”
by STELLA MADSEN
Founders of the Year: Professor Vahe Bandarian and alum Karsten Eastman

Vahe Bandarian and Karsten Eastman, co-founders of the University of Utah startup Sethera Therapeutics, have developed their PolyMacrocyclic Peptide (pMCP) Discovery Platform, revolutionizing peptide-based drug treatment This kind of drug treatment utilizes a chain of amino acids that bind to cell surface receptors in the body and regulate biological processes and treat diseases from there. Sethera Therapeutics’ pMCP Discovery Platform is designed to engage multiple targets at once, and it allows company partners to modulate intricate biological pathways, install complex functionalities and engineer peptides according to their goals and needs. Bandarian and Eastman’s enzymatic

cross-linking technology places their company in a unique position in drug discovery. They have both leveraged their many years of biochemical research on enzymatic transformations at the University of Utah to commercialize and translate their findings, ranking them among the most notable researchers of the year.
Photo: (left to right): Olivia Lucas, Tristen Garner, Ashley Winterbauer, Owen Bascomb, Kitana Ramos, Calvin Crawford, Erin King, Sofia Perez, Spencer Stevenson, Samantha Stinson, Ricardo Avendaño, Derek Salinas-Escobar

Arrival 1976
Joel Harris – a Legacy of Analytical Chemistry at the U
Now in his 50th year on the faculty of the Department of Chemistry, Joel Harris will retire from the U on January 1, 2026. Joel arrived at the University of Utah on August 12, 1976, only 4 days after defending his dissertation at Purdue University. He was the first analytical chemist appointed to the faculty and was charged with launching a graduate program in analytical chemistry and developing undergraduate and graduate course offerings in this area. Since that time, analytical chemistry has grown to be a thriving part of the department’s graduate program, with 10 faculty and 21 graduate students who currently affiliate with this area.
Thousands of undergraduate students have now taken analytical chemistry courses that Joel first organized around the principle of integrating fundamental concepts taught in the classroom with practical experiences in lab. This principle was manifested in Joel’s inquiry-based ‘special project’ that was a major part of his semester-long undergraduate course in Quantitative Analysis. This project gave students an opportunity to explore questions of their own asking, using tools of chemical analysis that they learned in the first 8 weeks of the lab. It was a capstone experience of independent learning that was made successful by the individual attention that Joel provided each student as they encountered problems in lab. Joel offered a similar approach to his graduate-level teaching, where students learned the theory of modern data analysis methods in the classroom and applied these concepts in consultation with Joel to data from their dissertation research.
Joel launched a research program in analytical laser spectroscopy at the U with a modest start-up budget of $25,000. He and his first students pioneered photothermal spectroscopy, a sensitive method for detecting molecules that absorb laser radiation by measuring the increase in the sample temperature. Time-resolved photothermal measurements reported non-radiative decay rates of electronically excited states, while pulsed-laser Raman spectroscopy was employed to determine the structure of photochemical intermediates.
Another major research goal in Joel’s lab was to determine the composition and structure of molecules at liquid/solid interfaces, where a small population of molecules residing at an interface must be resolved from the host of molecules in the surrounding solution. To address this challenge, Joel and his students developed surface-selective Raman, infrared, and fluorescence spectroscopy methods to investigate chemistry at liquid/solid interfaces. The sensitivity of their fluorescence methods increased to the point where they could quantify molecular populations by counting molecules and record movies of individual molecules undergoing reversible chemical reactions at surfaces.
Joel has won numerous national, international, and University honors for his research, teaching, and professional service. The true legacy of his career, however, is found in the success and outstanding contributions that his undergraduates, graduate students, and postdocs have made in their lives and careers, following their time in his classroom and research lab.

Giving to U Chemistry
Supporting the Future of Science
Chemistry Greatest Needs
The Chemistry Greatest Needs program provides vital, flexible support for student scholarships, awards and departmental initiatives within the University of Utah’s Department of Chemistry. Contributions to this fund help ensure that students have access to transformative, educational opportunities, research experiences, and academic resources. By addressing the most immediate and impactful needs of the department, this program empowers our faculty, staff, and students to thrive—and allows donors to make a meaningful difference where it matters most.
Sigman-Marks Fellowship
The Sigman–Marks Endowed Fellowship supports outstanding graduate students pursuing research in physical organic chemistry—a field that bridges molecular structure, reactivity, and mechanism to reveal the fundamental principles governing chemical transformations. Inspired by a shared commitment to advancing student success and scientific discovery, this fellowship recognizes the profound impact of physical organic chemistry in shaping modern catalysis, materials design, and sustainable chemical innovation
Photo: Sigman Group

chemistry.utah.edu/give

Special Thank You

The Allen Foundation Establishes Presidential Endowed Graduate Fellowship in Chemistry in Honor of the Casarotto Family
Founded in 1991, The Allen Foundation aims to continue the legacy of generosity and community involvement of their founder, Marion Mathis Allen. Marion was known throughout her life for her philanthropic commitment and active participation in numerous charities and organizations. The Foundation is now a nationwide philanthropic organization, with Marion’s grandson, David Casarotto, serving as Chairman of The Allen Foundation’s Board. The foundation continues to keep Marion’s memory alive and honoring her by helping multiple charities and causes each year. The Allen Foundation donation to an Endowed Fellowship will have a lasting impact on the quality of the chemistry program at Utah, as a percentage of the endowment is reinvested to ensure that the Fellowship remains financially strong for the future.

Emma Hale, Phd Student

Meghan Rainier, PhD Student
