the gem


Graduates step into the field to turn knowledge into practice



Graduates step into the field to turn knowledge into practice

Dr Houriiyah Tegally’s Wellcomefunded research programme brings genomics, data science, and environmental insight together to better understand how infectious diseases emerge and spread across Africa.

A €1.36 million Horizon Europe grant is enabling the South African Centre for Epidemiological Modelling and Analysis (SACEMA) to expand a long-running initiative that is reshaping how epidemic modelling is taught, applied, and sustained.

On 4 March 2026, partners from across clinical care, public health, diagnostics, government, and research gathered at the School for Data Science and Computational Thinking at Stellenbosch University for the SmartPath Roundtable: Architecting an Integrated Care Pathway for Maternal Anaemia.
the gem: Centre for Epidemic Response and Innovation (CERI) & South African Centre for Epidemiology and Modelling Analysis (SACEMA), Stellenbosch University, and KwaZulu-Natal Research, Innovation and Sequencing Platform (KRISP), UKZN, Durban director: Tulio de Oliveira editors: Tulio de Oliveira, Katrine Anker-Nilssen, Maambele Khosa, and Cheryl Baxter art director: Victor Hugo Szortyka graphic designers: Tulio de Oliveira, Katrine Anker-Nilssen, and Maambele Khosa printing: Aslam Arnolds websites: https://ceri.org.za/, https://www.sacema.org/, https://www.krisp.org.za/

As World TB Day highlights the urgency of ending one of the world’s deadliest diseases, new efforts at the National Health Laboratory Service show how training and genomic technologies are strengthening South Africa’s ability to detect, track, and respond to tuberculosis.
A new clinical study published in Cell Host & Microbe offers a long-awaited shift in how bacterial vaginosis (BV) – one of the most common infections affecting women worldwide –may be treated. Known as the VIBRANT trial, it moves beyond decades of antibiotic-only approaches to restoring the vaginal microbiome for longerterm health.

At the KwaZulu-Natal Research Innovation and Sequencing Platform (KRISP), the future of African genomics is being shaped by a new generation of scientists whose journeys are as compelling as the research they lead. Though their paths differ, Thembelihle Tombo and Nqobile Mthembu share a common purpose – to use science in service of society.


As autumn – a season that invites a moment to pause and take stock – settles over the beautiful Stellenbosch University campus, this issue of the gem focuses on something important: what happens when young scientists are given the right support, connections, and space to step into their work.
Across these pages, four graduates – Akhil Maharaj, Danilo De Silva, Thembelihle Tombo, and Nqobile Mthembu – capture that transition clearly. At CERI and KRISP, these emerging researchers are moving beyond coursework into applied science, working on real questions in genomics, data science, and infectious disease. What stands out is not just their technical ability, but their practical thinking, their contributions within teams, and the way ideas are shared and strengthened.
Alongside them, congratulations to two early-career researchers at CERI – Khomotso Mohlala and Graeme Dor – on receiving the SAMRC Biostatistics Capacity Development Initiative Award. Their work reflects a critical shift: as health data grows more complex, the ability to interpret and apply it is becoming central to public health.
That same transition is visible in two of our African STARS fellows from the first Advanced and Translational Training (ATT) cohort. With access to networks, mentorship, and industry exposure, Charleen Wormsberg and Megan Hanslow have moved into meaningful roles – applying their skills in ways that are tangible, relevant, and already making a difference. Their stories are a reminder that talent is not the barrier, but access is.
Collaboration also runs through much of this issue. The SmartPath initiative is a strong example, bringing together clinicians, researchers, policymakers, and industry to rethink how maternal anaemia is addressed in real-world settings – a practical, grounded approach to solving complex challenges.
We also highlight Prof Houriiyah Tegally’s work, which integrates genomics, data, and environment to better understand disease emergence; SACEMA’s long-term investment in modelling capacity; and the growing role of geospatial AI in shaping how we understand risk and response.
These stories point to something encouraging: a connected community of scientists who are not waiting for opportunities, but stepping into them – and, in many cases, creating them for others.
Prof Houriiyah Tegally’s Wellcome-funded research programme brings genomics, data science, and environmental insight together to better understand how infectious diseases emerge and spread across Africa.
text: Katrine Anker-Nilssen photos: MidJourney & CERI Media
Understanding why some pathogens spill over into human populations –and why some go on to spread while others do not – is one of the most complex and urgent challenges in global health. For Prof Houriiyah Tegally, Head of Data Science at the Centre for Epidemic Response and Innovation (CERI), this question sits at the centre of a new research programme supported by a prestigious Wellcome Career Development Award.
Her project focuses on pathogen emergence and expansion in Africa, a region where ecological diversity, rapid urbanisation and shifting environmental conditions are reshaping how diseases move between animals and people. By developing new analytical approaches, the work aims to better understand the conditions that allow pathogens to spill over, establish, and expand.
“This project will bring together genomics, epidemiology, and advanced data science through an integrated, multi-layered analytical framework designed to capture the full complexity of pathogen transmission in changing environments,” Prof Tegally explains.
At its core, the research integrates multiple data streams to build a more complete picture of disease dynamics. Genomic data is used to reconstruct how pathogens evolve and spread, while epidemiological data provides insight into population-level processes. Environmental and climate data, in turn, help define the ecological conditions in which transmission occurs. These datasets are brought together using phylodynamic approaches, ecological modelling, and machine learning to generate both retrospective understanding and forward-looking predictions.
“These diverse data streams will be integrated into unified models that can both explain past dynamics and generate forward-looking risk predictions,” says Prof Tegally.
The scale of this ambition is matched by the long-term support of the Wellcome award, which provides eight years of funding – a rarity in research environments often constrained by shorter funding cycles. For Prof Tegally, this creates space not only to explore complex questions in depth, but also to build sustainable research capacity.
“This award allows us to drive longer-term and more robust exploration around this topic,” she explains. “It also allows us to grow our team, support postdocs in developing their own careers, and integrate novel technologies – such as artificial intelligence – into pathogen transmission research.”
Within CERI, Prof Tegally leads a multidisciplinary
team working at the intersection of data science and infectious disease research. The group brings together modellers, genomic epidemiologists, computational biologists, ecologists, and geospatial specialists. “We focus on building analytical solutions to solve research challenges related to pathogen genomics and infectious disease risk distribution,” she explains, describing a collaborative environment where research direction is shaped collectively and training and mentorship are central.
Working with large, complex datasets presents its own set of challenges. Data must often be harmonised across formats and sources, while some inputs –such as satellite imagery used to infer environmental context – can be costly and technically demanding to process. In response, the team is exploring new approaches, including the use of artificial intelligence and strengthened data engineering capacity, to streamline workflows and unlock new insights.
A key outcome of this work is a deeper understanding not only of how outbreaks grow, but also of why they sometimes do not. “By understanding exactly the factors that help or hinder pathogens to spread in human populations, we can better detect, monitor, and respond to emerging infectious disease threats,” she explains.
Leading a project of this scale from Africa carries particular significance for Prof Tegally. “The quality of science coming out of Africa is often overlooked,” she notes. “This is a chance for us to lead high-quality, highly novel research on the continent.”
Her own journey reflects that commitment. After studying in the United States and the United Kingdom, she returned to South Africa for her PhD and has since built her career within leading African research institutions. “I have always been passionate about returning to Africa to do high-quality science here,” she says, adding that recognition at this level also holds broader meaning. “It means a lot to be a young African woman in science and to be recognised in this way.”
Alongside her research career, Prof Tegally is also navigating a new personal chapter. Becoming a mother, she says, has reshaped how she approaches both time and ambition. “It has reminded me that I can do hard things and that I am strong even on the most overwhelming days,” she reflects. “I’m thinking more long-term, being more time-efficient, and learning to balance both.”
Outside of research, she finds balance in simple, grounding activities – swimming, spending time at the beach, and being in nature.
Together, Prof Tegally’s work and trajectory point to a broader shift in global health science: one where data, context and leadership from within Africa are central to understanding and responding to the diseases of the future.
*Turn to the next page for more info on the Wellcome-funded project.
Last year, Prof Houriiyah Tegally received the Stellenbosch University Research and Innovation Excellence Award for Most Cited Open-Access Output, recognising her contributions to publications characterising SARS-CoV-2 variants as they emerged and spread globally during the pandemic. The award reflects the far-reaching impact of this work, demonstrating how research originating in South Africa informed global understanding of the virus. “It is clear the impact of this work emanating from South Africa was global,” says Prof Tegally. “It’s an honour to be recognised at the institutional level, which reminds me of the value that our work brings locally as well as internationally.”

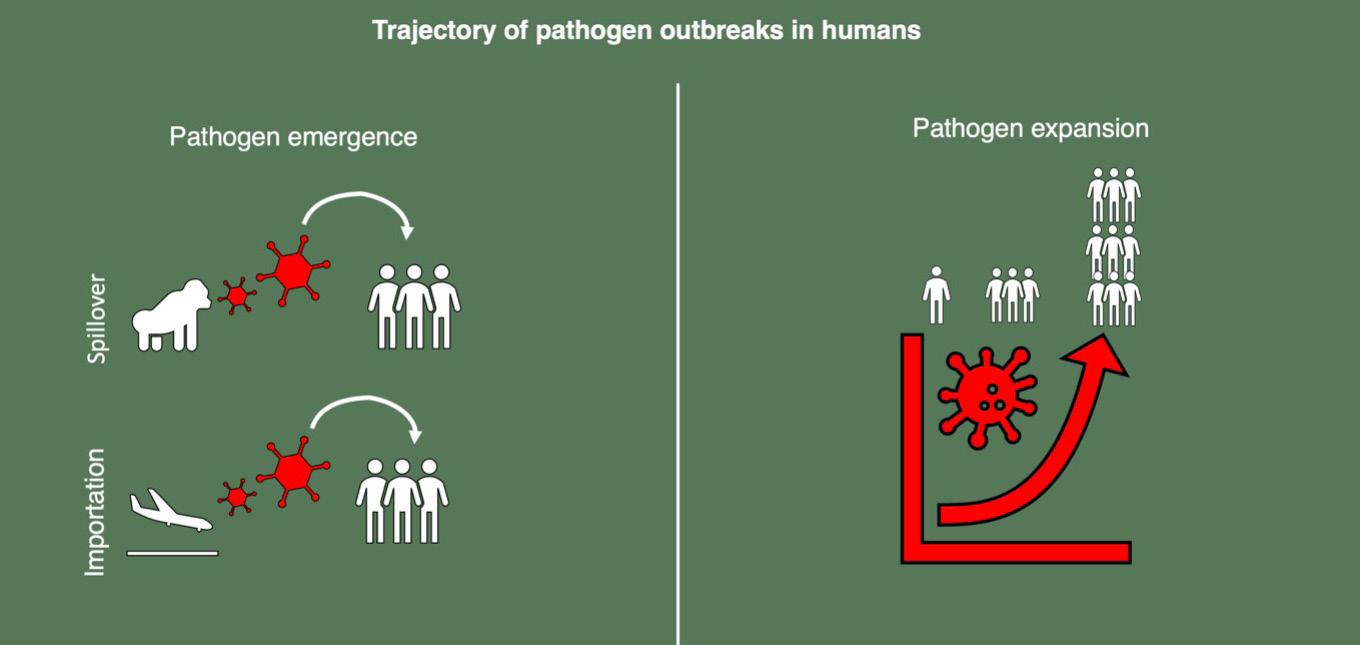
Project summary, research questions, and conceptual figure (above).
Shifting global trends such as urbanisation, land-use change, wildlife trade, and increased connectivity are amplifying pathogen spillover and expansion. Africa is central to this dynamic. The continent faces converging factors which make it both highly vulnerable to emerging pathogens, but also ideally positioned for better preparedness.
While Africa’s primary forests, savannas and wetlands, and wildlife diversity already positions the continent as an ecological hotspot for zoonotic emergence, rapid urbanisation, growing population numbers, agricultural demands, changing land-use, and climate change increase human-animal-pathogen interactions.
The goal of this project is to develop and execute integrated data analytical methods and propose a new framework to improve understanding of pathogen emergence and expansion in Africa. The main hypothesis is that the latter is determined by the dynamic interplay between reservoir ecology, pathogen evolution, human-mediated introduction pathways, and local transmission conditions.
This fellowship will focus on three categories of
pathogens, spanning distinct transmission modes, disease ecology and geographical distributions: i) Arboviruses, ii) Mpox virus, and iii) Respiratory viruses.
It will tackle disease spillover dynamics in the context of reservoir distributions, genetic evolution, contact at human-animal interface, and imported introductions, and forecast epidemic expansion potential after emergence across shifting landscapes in Africa, towards better pandemic preparedness.
RQ 1: What ecological, evolutionary, and behavioral conditions determine whether a pathogen successfully spills over from wildlife reservoirs into human populations?
RQ 2: What factors influence the risks of pathogen introduction and subsequent establishment in secondary populations?
RQ 3: Can we accurately predict pathogen epidemic expansion by integrating genomic, ecological, and mobility data using phylodynamic, epidemic forecasting and machine learning models?
Nanosene, a startup that attended SWEAT Africa 2026, is focused on translating academic research into practical tools for drug discovery and life-science research.
text: SWEAT Africa photo: Supplied
Founded on PhD research from Stellenbosch University, Nanosene was created to address a persistent challenge in biomedical science: the reliable study of membrane proteins. These proteins play a central role in human biology and are the basis of many important drug targets, yet they have long been difficult to analyse using conventional techniques.
According to CEO, Dr Gestél Kuyler, Nanosene was established to bridge the divide between promising academic innovation and technologies that can be deployed effectively in real laboratory settings. “We set out to enable stable, functional study of membrane proteins that are poorly served by existing methods,” she explains. “Our focus has been on moving beyond proof-of-concept science to deliver robust, accessible technologies that generate reliable, actionable insights.”
Nanosene’s polymer nanodisc technology enables researchers to extract and stabilise membrane proteins directly from their native environment, without relying on harsh detergents. By preserving the proteins’ natural structure and function, the technology supports more reliable data and opens up experimental approaches that are difficult to achieve with traditional methods.
Nanosene’s BzAM™ polymers are currently being evaluated by research partners across the U.K. and the U.S., including universities and applied research environments. These collaborators are using the materials in structural biology, functional assays, and drug-discovery workflows, providing independent validation across a range of research applications.
The company has also partnered with a leading biotech and contract research organisation in Germany to bring its first polymer-based products to market, marking an important step towards commercialisation.
Emerging from Stellenbosch University, Nanosene benefits from strong foundations in polymer science
and membrane biology, shaping its transition into a globally oriented biotech company.
Reflecting on SWEAT Africa 2026, Nanosene COO, Dr Elaine Barnard, notes: “SWEAT Africa didn’t just create visibility, it created traction. It accelerated meaningful conversations with partners across biotech, manufacturing and investment, and validated strong interest in our technology. Most importantly, it converted early awareness into real momentum, directly supporting our next phase of fundraising, scale-up and market entry. Pitching as part of the OST BRAIN 5.0 cohort also reaffirmed the value of what we are building, with the energy and engagement from the community translating into tangible opportunities, including new team connections.”

Access is often the difference between potential and progress. For two African STARS fellows, it led directly to employment, independence, and the ability to shape what comes next.
text: Katrine Anker-Nilssen photos: Supplied
For many early-career African scientists, the challenge is not capability – it is access. Access to networks, to industry pathways, and to opportunities that allow scientific knowledge to translate into real-world impact.
The African STARS Fellowship is designed to bridge that gap. Its value lies not only in training, but in what changes because of it: increased confidence, stronger professional voice, and the ability to move from academic knowledge into applied, meaningful work.
For two STARS, who completed their Advanced and Translational Training (ATT) fellowship in December 2025, that shift has been tangible – leading directly to employment, financial independence, and growing influence within their communities and professional networks. Both are now working at performance. io, a data-driven health and technology company that translates pharmaceutical and biotech innovation into measurable, applied outcomes.

Megan Hanslow’s path into science has always been grounded in purpose. Growing up in the Cape Flats and becoming a first-generation university graduate, her work has focused on women’s health – an area where science directly intersects with lived experience.
“My background has shaped my focus on using science to create meaningful impact in my community and on the African continent,” she explains. “Through my working experience I found that knowledge finds its true value in how it is applied.”
Despite strong academic performance, entering the workforce proved difficult for Megan. “The recurring question, ‘Do you have industry experience?’ revealed a frustrating reality: experience requires opportunity,” she says. “I treated entering the biotech workforce like a full-time job. But the lack of response reinforced just how limited access to networks can be.”
What the fellowship shifted was not only her access, but her mindset. “The African STARS Fellowship presented an opportunity to step into that next phase to connect the two, and begin shaping work that is meaningful and impactful,” she says. “The fellowship completely shifted how I think about science, innovation, and impact in Africa. Real impact doesn’t happen alone – it happens when people come together.”
Through exposure to peers across 16 African countries and direct engagement with industry, Megan began to see science differently – not as an endpoint, but as a tool.
“The fellowship programme’s conferences, site visits, and conversations with entrepreneurs and industry leaders also opened doors to new possibilities,” she explains. And that shift translated directly into
confidence and action for her. “The fellowship gave me a level of confidence I didn’t have before. It helped me realise that I could step into spaces and contribute meaningfully.”
Now entering the workforce, her understanding of impact has evolved. “Dignified and fulfilling work is work that combines purpose and impact,” she says. “It means being in an environment where my contributions are valued and where I can continue to grow.”
Importantly, that impact is already extending beyond her own role. “I have been involved in planning community engagement events for girls in science,” she says. “Inspiring girls that navigating a career in science is worthwhile and definitely possible.”
Megan’s path reflects a broader shift – from accessing opportunity to beginning to create it. “The fellowship’s programme truly empowers early-career scientists to transform their potential into real-world impact,” she says. “This experience has changed my life. I have gone from the shadows to the stars.”
Charleen Wormsberg’s experience reflects a very similar and critical barrier: the invisible gap between academic training and industry access.
“While I had strong academic training, I found it challenging to access opportunities – due to lack of structured pathways from academia into industry, and minimal networks,” she explains.
Even within an environment of innovation, that divide was clear. “I felt like there was still a definite divide between entrepreneurs in the industry versus students,” she says.
This is where the fellowship provided both access and direction for Charleen. “I wanted to gain practical, industry-relevant skills, expand my professional network, and better understand how to position myself,” she explains.
A key shift was learning how to translate science into something usable. “Learning how to translate scientific knowledge into practical, business-relevant insights was particularly impactful,” she notes. But just as important was the shift in confidence. “When I saw the level of achievement, I initially felt intimidated,” she recalls. “But there must be something in me that others believed in. And that helped me start believing in my own potential more deeply.”
That belief translated into tangible outcomes. “The fellowship helped me build the confidence to recognise my value, and to advocate for myself and negotiate a
salary that reflected my worth,” she says. It also gave Charleen the tools to access opportunities directly. “The fellowship taught me how to effectively use platforms like LinkedIn, This made me more visible to opportunities and showed that I was proactive and serious about transitioning into the industry.”
Now working at the intersection of science, data, and technology, she sees her role as part of a broader system of impact. “We translate pharmaceutical and biotechnological advancements into measurable, databacked outcomes – supporting the growth of local innovation and helping advance treatments that are more relevant to African populations,” she explains.
Beyond her own career, the ripple effect is already visible. “Everything has changed. I am now financially independent. And I’ve already been able to help friends find work,” she says.
Charleen’s ambition is explicitly outward-facing. “My goal is to create those opportunities, uplift others, and contribute to building a more equitable environment,” she says.
These stories show how the right training, networks, and opportunities enable young scientists to move into meaningful work and extend that impact beyond themselves.
From greater confidence and income to a stronger professional voice, the effects build over time. Individual opportunity begins to shape wider systems.
This is how potential becomes impact: through access, application, and action.

A €1.36 million Horizon Europe grant is enabling the South African Centre for Epidemiological Modelling and Analysis (SACEMA) to expand a long-running initiative that is reshaping how epidemic modelling is taught, applied, and sustained.
text: Katrine Anker-Nilssen photo: MidJourney
For over 16 years, the International Clinics on Infectious Disease Dynamics and Data (ICI3D) have brought together researchers from across Africa to tackle one of public health’s most complex challenges: understanding how diseases spread – and how to stop them.
Now, with new Horizon Europe funding, that work is entering a new phase. “This award reflects something we’ve been building consistently over many years,” says Dr Cari van Schalkwyk, Senior Researcher at SACEMA. “It’s about persistence – and about continuing to invest in this space over time.”
At its core, the ICI3D programme is focused on strengthening advanced epidemic modelling capacity across sub-Saharan Africa. The need is clear. As governments and health systems increasingly rely on data to guide decisions, demand for modelling expertise has grown – but opportunities for sustained, high-level training and mentorship have remained limited.
“There is increasing demand for modelling to inform policy decisions, but the capacity to do this work consistently and at scale is still developing,” says Dr van Schalkwyk. “This programme is about helping to build that capacity in a more sustained way, and ICI3D is designed to close that gap.”
Epidemiological modelling sits at the intersection of mathematics, statistics, and public health. It allows researchers to explore questions that cannot always be answered in real time: What impact could a vaccination campaign have? How might treatment
coverage change disease outcomes? Which interventions will make the greatest difference when resources are constrained?
In fast-moving health crises, these are not abstract questions – they are urgent ones. Modelling provides a way to test scenarios, anticipate outcomes, and guide decision-making when time and evidence are limited. It is increasingly central to how public health responses are designed, both during outbreaks and in long-term planning.
While training programmes in modelling do exist, they are often short-term – intensive workshops that end without ongoing support or connection. One of the most significant shifts enabled by the Horizon Europe grant is the move beyond this model.
ICI3D will not only expand its clinics and training activities, but also build a structured alumni network – creating a space where participants can continue collaborating, sharing ideas, and developing their work long after the initial training ends. “The aim is to create stronger continuity between training, applied work, and long-term scientific engagement,” Dr van Schalkwyk explains.
For early-career scientists, the impact is both practical and long-term. Participants gain advanced technical skills, but also the opportunity to work alongside experienced modellers on real public health questions. Just as importantly, they build networks that extend across countries and institutions – strengthening their ability to collaborate, secure funding, and grow their careers. The programme is intentionally rooted in

African institutions, helping to ensure that expertise is not only developed, but retained.
Infectious diseases move across borders – and understanding them requires a similarly connected approach. “It’s important that modelling work is not only applied in African contexts, but also shaped and led by researchers based in African institutions,” Dr van Schalkwyk notes.
ICI3D brings together researchers from multiple disciplines, including epidemiology, mathematics, statistics, and clinical science. This collaboration strengthens both the quality of modelling and its relevance to different contexts. It also shifts the role of African researchers within the global scientific landscape – from applying existing models to actively contributing to new methods and approaches.
Building on SACEMA’s long-standing work, the ICI3D programme represents a move toward more sustained, locally embedded modelling capacity. The ambition is not only to train individuals, but to strengthen systems – enabling African institutions to generate and use their own modelling evidence when it matters most. “Stronger local modelling capacity means being able to respond more quickly to emerging questions, and to generate evidence that is grounded in local realities,” adds Dr van Schalkwyk.
Over time, this could transform how public health decisions are made: faster responses to emerging threats, more context-specific insights, and stronger, self-sustaining research ecosystems across the continent.
“There is increasing demand for modelling to inform policy decisions. But the capacity to do this work consistently and at scale is still developing.”
– Dr Cari van Schalkwyk
Haingo Andry attended the FDL Foundations Africa Workshop, and shares his highlights and key takeouts.
text: Katrine Anker-Nilssen photo: Supplied & MidJourney
Researchers, technologists, and policymakers gathered in Stellenbosch for the FDL Foundations Africa Workshop, on March 9 and 10, to explore how emerging geospatial AI tools could be applied to challenges across the continent. Rather than a traditional lecture-style event, the workshop focused on collaborative thinking – bringing together diverse expertise to identify where these technologies could have meaningful impact.
Among the participants was Haingo Andry, a PhD candidate in Applied Mathematics at Stellenbosch University and affiliated with CERI. His experience reflects both the exploratory nature of the workshop, and the growing relevance of geospatial approaches to African research contexts.
The central aim of the workshop was to brainstorm applications of geospatial foundation models (GFMs) – large-scale AI systems trained on spatial data

– particularly in scientific and high-impact domains. Participants worked in groups to generate and refine ideas, using structured discussions and voting systems to prioritise the most promising concepts. “We developed ideas or refined existing ones, following the workshop’s approach, and used voting systems to filter out ideas,” Haingo explains.
A recurring theme across discussions was the use of these models to better understand and respond to environmental challenges. Many ideas focused on predicting or managing natural disasters such as floods and wildfires, while others explored how geospatial insights could support agriculture, for example by helping farmers improve yields and income. As Haingo puts it, “the recurring theme was to find problems that Geospatial Foundation Models (GFMs) could solve and bring impact to people’s lives.”
“The diversity of the group stood out most,” Haingo notes, highlighting how this mix helped sharpen thinking and ground ideas in real-world contexts. Participants came from a wide range of backgrounds – including computer science, astronomy, policy, management, Geographic Information Systems (GIS), and machine learning – creating a space where technical ideas could be challenged and reshaped through different perspectives.
While the workshop was not focused on teaching the technical foundations of AI, it provided a shared understanding of how GFMs operate: pre-trained on large-scale geospatial datasets, they can be adapted to specific applications such as disaster monitoring or object detection. The emphasis, however, remained firmly on application rather than theory.
For CERI, where work sits at the intersection of genomics, epidemics and data science, the relevance of these approaches is particularly compelling. Geospatial AI offers new ways to map and analyse the relationships between environmental factors and disease dynamics – an area central to epidemic modelling and response. As Haingo explains, “This is
especially relevant for work involving geospatial data and mapping relationships between data distributions,” pointing to use cases such as “estimating mosquito populations, which are closely linked to disease transmission.”
More broadly, these tools hold significant promise for African public health contexts. They could support early warning systems by monitoring environmental and climatic changes, help identify high-risk zones, and improve understanding of healthcare access across regions. They may also enable more targeted interventions by predicting vector habitats or modelling how disasters influence disease spread.
Although direct applications to genomics or microbial research were not a focus of the workshop, the intersection between geospatial data and epidemic modelling presents a clear opportunity. For CERI, this opens the door to integrating spatial intelligence with genomic surveillance – strengthening how outbreaks are detected, understood and responded to in real time.
The workshop concluded with a set of potential research directions, many of which could evolve into collaborative projects or funding proposals. “Some ideas could evolve into research collaborations or funding proposals,” Haingo says, noting how multiple concepts were refined through discussion and voting, with a focus on real-world challenges where GFMs could deliver tangible societal benefit.

In that sense, the Foundations Africa Workshop was less about delivering answers and more about shaping questions – highlighting where emerging technologies like geospatial foundation models might meaningfully contribute to solving some of Africa’s most pressing challenges, and where institutions like CERI are well positioned to lead.


On 4 March, partners from across clinical care, public health, diagnostics, government, and research gathered at the School for Data Science and Computational Thinking at Stellenbosch University for the SmartPath Roundtable: Architecting an Integrated Care Pathway for Maternal Anaemia
The six-hour session brought together a deliberately diverse group – clinicians, policymakers, academia, community health leaders, and industry partners –to confront a persistent but preventable challenge:
maternal anaemia. Despite being both detectable and treatable, too many women continue to fall through the gaps between screening, diagnosis, and care.
SmartPath, a collaborative initiative developed through the African STARS Fellowship at CERI by fellows Fezokuhle Khumalo, Joel Alukwe and Harries Molepo, seeks to address exactly this problem – not by introducing a single intervention, but by redesigning the entire pathway of care.
As Alukwe explains, the idea evolved quickly beyond its initial scope: “By the time a woman needs IV iron, the system has already failed her multiple times. That
pushed us to rethink everything – to go earlier, and build one connected pathway from awareness through to treatment.”
Structuring the Conversation: From Problem to Pathway
The roundtable was designed not as a series of presentations, but as a working session. The morning opened with framing discussions on the burden of iron deficiency and the SmartPath model, before moving into a multidisciplinary panel focused on a central question: why is the current system failing women – and what would it take to fix it?
What emerged quickly was a shared recognition that maternal anaemia is not a single-point failure, but a system-wide breakdown. “Maternal anaemia is a multilevel problem – it starts early, and frontline healthcare workers are often not trained to recognise the symptoms,” said Prof Vernon Louw, Executive Head of the Department of Medicine at the Faculty of Medicine and Health Sciences, University of Stellenbosch.
Across the panel, participants pointed to gaps at every stage – awareness, detection, monitoring, treatment, and follow-up.
“Iron deficiency develops long before pregnancy, but symptoms are often dismissed as normal,” noted Khumalo (pictured, right). “By the time women are diagnosed, the condition has often progressed.”
A recurring theme throughout the discussion was that knowledge – or lack of it – sits at the centre of the problem. “The biggest problem is awareness,” said Nomonde Tengwa, Innovation Hub Lead at mothers2mothers. “Mothers are given iron tablets but don’t understand why they are important.”
This gap extends beyond patients. “Community health workers are the first point of care, but if they don’t understand the importance, they can’t communicate it,” she added.
Even communication itself presents challenges. “There isn’t even a word for iron in some languages,” Prof Louw noted, underscoring how deeply structural some of these barriers are.
At the clinical level, the limitations of current diagnostic approaches were also clear. “Haemoglobin testing is widely used, but it misses almost half of iron deficiency cases,” said Prof Louw. This means many women are only identified once anaemia is already advanced – a point echoed across both panel and SmartPath team insights.
If the problem is fragmentation, the proposed solution is integration. SmartPath is built around a simple but powerful idea: closing the loop – connecting awareness, diagnosis, treatment, and follow-up into a continuous system of care.
“When the loop works, it starts before pregnancy,” explained Khumalo. “From adolescent screening and awareness, through antenatal care, to community monitoring and escalation when needed.”
Central to this model is the role of point-of-care diagnostics. “Point-of-care testing would make a huge difference,” said Prof Louw. “If you can diagnose and act immediately, you don’t lose the patient.”
“Pregnant women tend to fall through the cracks at several points along the care pathway: awareness, detection, monitoring, and treatment.”
– Fezokuhle
Khumalo, African STARS Fellow, SmartPath

The current system often requires women to return for results – something many cannot do. “Patients are told to come back, but they don’t – they face long queues and lose a day of work,” he added.
SmartPath instead proposes a shift from a one-time prescription model to a continuous care pathway, supported by community health workers and structured follow-up.
In the afternoon, participants moved into breakout groups to stress-test the SmartPath pilot against realworld conditions.
The focus was practical:
• What would make this work in a busy antenatal clinic?
• What evidence would policymakers need?
• How could the model be sustained beyond donor funding?
These discussions surfaced the realities of implementation.
“Primary healthcare is already stretched – integration is key,” said Megan Marais from the Western Cape Department of Health. “If we want real change, we need to build on processes that already work.”
Participants emphasised the need to avoid adding complexity. “We don’t need to reinvent the wheel – we should use the resources we already have,” added Natasha Macey, Medical Advisor from Acino.
This directly shaped SmartPath’s evolving design. “The roundtable pushed us toward a leaner approach,” Harries Molepo noted. “Focusing on only the most essential data and steps, rather than overburdening already stretched systems.”
While education and awareness were repeatedly highlighted, there was also a strong call to move beyond discussion. “We have the intellectual capacity and willingness in this room to solve this problem,” said Dr Benjamin Botha. “But education alone is not enough –we are at the interface of implementation.”
His conclusion captured a broader sentiment across the room: “Talk is cheap – implementation is what matters.”
Cost and sustainability also emerged as critical considerations. “Point-of-care testing is not yet part of the guidelines – so the question is, who pays?” noted Prof Tulio de Oliveira, Director of the Centre for Epidemic Response and Innovation (CERI).
At the same time, there was recognition that investment upfront could reduce long-term costs, particularly by preventing complications such as postpartum transfusions.
The immediate next step for SmartPath is the launch of a pilot in selected antenatal clinics, designed to generate evidence on feasibility, acceptability, and impact.
“The goal is to test this in real-world settings,” Harries Molepo explains, “and refine the model before any broader rollout.” If successful, the implications could extend far beyond maternal anaemia. “The core architecture – point-of-care diagnostics, decision support, and structured follow-up – is adaptable,” Molepo notes. “It could be applied to other conditions where care is currently fragmented.”
Ultimately, the SmartPath Roundtable made one thing clear: the challenge is not whether maternal anaemia can be addressed – but whether systems can be designed to do so consistently. Or, as Nomonde Tengwa put it: “We need to ensure the patient truly understands, before she leaves.”
“We don’t need to reinvent the wheel – we should use the resources we already have.”
– Natasha Macey, Medical Advisor, Acino

Be a junior and earlycareer researchers in Europe or Africa
Be an African Stars fellow/ alumni



Topics of interest: infectious diseases/ rare diseases or familial syndromes
Duration: Short stays up to 90 days
Destination: Africa-Europe, Africa-Africa, Europe-Europe
Deadlines: July 1st or Oct 1 st, 2026 https://tinyurl.com/ye25dxa2s



Rethinking neonatal sepsis care.
text: Katrine Anker-Nilssen photos: Supplied
Neonatal sepsis remains a leading cause of newborn mortality in sub-Saharan Africa, yet the systems designed to manage it are increasingly out of step with reality. In many clinical settings, care is shaped less by formal guidelines and more by constraints – limited diagnostics, delayed results, inconsistent antibiotic access, and the growing pressure of antimicrobial resistance.
New findings from the INSIGHTS study bring this tension into focus, revealing a fundamental misalignment between global recommendations and the conditions under which clinicians are actually working. The issue is no longer simply about access to guidelines, but whether those guidelines are still fit for purpose.
For Prof Angela Dramowski, a paediatric infectious diseases

specialist at Stellenbosch University and Tygerberg Hospital who leads neonatal infection research in the Department of Paediatrics and Child Health, the publication reflects the complexity of frontline decision-making. “Clinicians are often forced to act quickly, without the diagnostic certainty they need,” she explains. “They must decide how to treat a critically ill newborn while balancing effectiveness, safety, and the risk of further resistance – often with incomplete or delayed information.”
The study arrives at a pivotal moment. With global efforts underway to revisit neonatal sepsis diagnostics and treatment strategies, it adds important evidence from the settings most affected. “There is increasing recognition that we need better data, better diagnostics, and treatment approaches that are grounded in local realities,” Prof Dramowski says. “Without that alignment, even well-intentioned guidelines can fall short.”
A more responsive approach, she argues, starts with context. “Countries need reliable, local data on infection patterns and antimicrobial resistance, and that data must directly inform treatment guidelines and antibiotic access,” she notes. “At the same time, strengthening diagnostic systems and infection prevention is essential to reduce the burden of sepsis itself.”
For Dr Larisse Bolton, a
researcher at the South African Centre for Epidemiological Modelling and Analysis (SACEMA) and computational/data science lead for the neonatal infections research group at Tygerberg Hospital, the work is both scientific and deeply personal. Her research focuses on improving maternal and neonatal health in Africa by integrating applied mathematics, epidemiology, and data science to strengthen realworld healthcare systems.“The life expectancy of its youngest members mirrors the well-being of a society,” she reflects. “To build healthy societies, we must start with healthy neonates.”
Her involvement in the INSIGHTS study included supporting data systems and contributing to the research process, but it is the people behind the work that have had the greatest impact. “I have been deeply inspired by the relentless efforts of clinicians and researchers working to save infant lives in under-resourced settings,” she says. “My role has been to support that effort –contributing my computational expertise and helping generate the evidence needed to strengthen care where it matters most.”
Neonatal sepsis is a key driver of under-5 mortality in Africa, contributing to the about 94,100 sepsis and other infectious condition deaths among newborns in sub-Saharan Africa in 2021. These life-threatening bacterial
infections within the first 28 days of life unevenly affect low-andmiddle income countries (LMICs) where diagnostic, prevention and treatment resources are limited. Neonates with these infections deteriorate rapidly, with survival hinging on rapid detection and treatment. Sadly, survivors of neonatal sepsis are often plagued with long-term effects such as vision loss and seizures. In resource-limited settings, clinicians are often left to treat neonatal sepsis in the absence of laboratory confirmation (i.e. empiric treatment) due to limited laboratory capacity, long delays in diagnostic results and high costs of processing blood cultures –the gold-standard diagnostic test for neonatal sepsis. The WHO provides recommendations for the empiric treatment of neonatal sepsis; however, these recommendations have not been revised since 2005 and do not consider local pathogen and antimicrobial resistance (AMR) patterns, reducing their effectiveness with increasing AMR within the region. In a recent study, clinicians caring for subSaharan African neonates were surveyed to obtain a complete view of the challenges facing the management of neonatal sepsis in the region, particularly the current state of access to diagnostics and guideline use in practice. Study responses were received from 83% of the targeted countries in sub-Saharan Africa and comprised of mostly senior doctors (72%), those working in central (58%) public healthcare facilities (70%) and involved in neonatal sepsis treatment daily. 29% of respondents rarely received blood culture results in time to guide treatment, with low- and low-middle income countries (LLMICs) similarly challenged by slow diagnostic turnaround times compared to their high-income counterparts that seldom face these difficulties. More than 50% of respondents reported difficulty accessing
antibiotics, with supply issues (including antibiotic registration, formulation, access and cost) being the most frequently reported hindrance to access; more so for some antibiotics than others. The WHO guidelines are still widely used to manage neonatal sepsis in practice across the region, with more than 45% of the respondents having attempted to or been involved in the writing of local guidelines during their careers. Those who have not opted to be involved in local guideline writing mostly cited the scarcity of local AMR data as the primary obstacle. Study findings emphasise the overall dependence on empiric treatment guidelines which are misaligned with the local pathogen and AMR patterns in much of sub-Saharan Africa. This is against the backdrop of growing evidence of the rapid rise of multi-drug resistant neonatal sepsis-causing organisms. This misalignment between guidelines and AMR patterns increases the chances of ineffective initial treatment for neonatal sepsis, resulting in poor patient outcomes and increasing the spread of resistant organisms. Neonatal sepsis care in sub-Saharan Africa is thus constrained not by a lack of guidelines, but by a lack of alignment – between diagnostics, data, and the realities of AMR infections. This misalignment should be at the forefront of global health priorities, encouraging decision-makers to leverage successes from other domains to secure access to inexpensive, rapid diagnostic tests and effective antibiotics for neonatal sepsis to optimise neonatal survival and health in sub-Saharan Africa.
Stanley JL, Hettle D, Poffley R, Bolton L, Gres E, Coelho I, et al. Investigating neonatal sepsis: anti-Infectives, diagnostics and Guidelines used in Health sysTems across sub-Saharan Africa –The INSIGHTS study. BMJ Paediatrics Open. 2026;10:e004132. https://doi.org/10.1136/

Dr Larisse Bolton, a researcher at SACEMA and computational/ data science lead for the neonatal infections research group at Tygerberg Hospital, has received international recognition for her work in maternal and neonatal health.
She has been selected as a heiAIMS Incoming Young Researcher at Heidelberg University, where she will collaborate with Dr Florian Marx on the NEO-CARE project. This research will evaluate the costeffectiveness and antimicrobial resistance impact of empiric antibiotic use for early-onset neonatal sepsis in South African neonatal units. She has also been awarded a Baden-Württemberg Scholarship, supporting her two-month research visit to Heidelberg. The work is expected to generate evidencebased recommendations for neonatal sepsis treatment, while strengthening international collaboration and enabling valuable skills and knowledge exchange. Congratulations!
Larisse’s research bridges applied mathematics, epidemiology, and data science to improve maternal and neonatal health systems in Africa, with a focus on real-world impact in low- and middle-income settings.
text: Emilyn Costa Conceicao, Nabila Ismail, Sarishna Singh, Yonas Ghebrekristos, and Robin Warren
photos: Supplied
On any given day inside the National Health Laboratory Service (NHLS) laboratory in Green Point, Cape Town, laboratory staff work with samples that represent real people, real infections, and real opportunities to interrupt tuberculosis (TB) transmission with accurate diagnostics. Increasingly, these efforts are being strengthened by genomic technologies that help researchers understand how TB spreads and how drug resistance emerges.
This work reflects the spirit of World TB Day, observed each year on 24 March, which renews the global commitment to ending TB. The World TB Day 2026 campaign highlights the importance of leadership, innovation, and collaboration in addressing one of the world’s deadliest infectious diseases.
In November 2025, laboratory scientists and researchers gathered at NHLS Green Point for a two week-programme on “Theoretical and Practical Training in Whole-Genome Sequencing (WGS) for Mycobacterium tuberculosis Clinical Samples”. The initiative was delivered as part of GenPath Africa, integrating work on genomic surveillance, the Precision Action for Rifampicin-Resistant Tuberculosis (PARRTB) sub-study, and capacity development across the network.
The aim of the programme was to strengthen national capacity for TB genomic surveillance by equipping NHLS scientists with the theoretical knowledge and practical skills required to generate and interpret WGS data. These technologies are transforming TB
research and public health by enabling faster detection of drug resistance and improved understanding of transmission dynamics. It also highlights the role of academic institutions in strengthening public health systems.
The programme brought together NHLS scientists, technologists, pathologists, and early-career researchers, highlighting the multidisciplinary expertise required to implement genomic surveillance within routine laboratory practice.
By hosting the training within the NHLS laboratory environment, the initiative helps embed these skills directly within the national diagnostic infrastructure, strengthening the ability of laboratories to detect and monitor drug-resistant TB.
This partnership between researchers, laboratories, and public health institutions illustrates the importance of collaboration in strengthening health systems.
The training programme combined online theoretical learning with hands-on laboratory training, allowing participants to build both conceptual understanding and practical experience.
Participants first accessed recorded lectures covering next-generation sequencing technologies, DNA extraction and quality control, sequencing library preparation, and bioinformatics pipelines used to analyse TB genomic data.
They then attended a week-long practical training at NHLS, working directly with clinical samples and
following the full sequencing workflow – from DNA extraction and quality assessment to sequencing preparation and data management.
By the end of the course, participants had experienced the complete pipeline of TB genomic analysis, bridging laboratory techniques with the interpretation of genomic data for clinical and public health decision-making.
For Dr Emilyn Costa Conceição, one of the organisers of the training, strengthening human capacity remains central to the success of genomic surveillance.
“Training scientists within the NHLS is essential. If we want genomic technologies to support TB diagnosis, patient care, and surveillance, we must ensure that local teams have the skills and confidence to use them.”
Genomic technologies are increasingly enabling precision approaches to TB control. At the patient level, sequencing data can identify mutations associated with drug resistance, supporting precision medicine by
“Training scientists within the NHLS is essential. If we want genomic technologies to support TB diagnosis, patient care, and surveillance, we must ensure that local teams have the skills and confidence to use them.”
helping clinicians select the most effective treatment strategies. At the population level, genomic surveillance allows researchers and public health authorities to identify transmission networks and detect emerging resistance patterns, contributing to precision public health strategies.
Through the PARR-TB study and the broader GenPath Africa network, scientists are working to integrate these genomic insights into routine TB control programmes across Western Cape, South Africa.
Many of these collaborations involve partners connected to the Tuberculosis Genomic Group (TBG) and Centre for Epidemic Response and Innovation (CERI), whose interdisciplinary networks bring together researchers working across genomics, epidemiology, and public health.
The training was made possible through the efforts of a multidisciplinary team working across research, diagnostics, and public health. The course was organised by Dr Emilyn Costa Conceição, Dr Nabila
– Dr Emilyn Costa Conceição

Ismail, MSc Felicia Wells-Hunter, Prof Robin Warren, and Janré Steyn from Stellenbosch University, alongside with colleagues from NHLS: Dr Yonas Ghebrekristos, Dr Sarishna Singh, Dr Stefan Opperman, François Barton and Dr Nasima Mohamed. This training also drew on contributions from the GenPath Africa network and CERI teams, including Prof Cheryl Baxter and Prof Tulio de Oliveira.
Together, the team designed and delivered a programme that combined theoretical learning with practical laboratory experience, reflecting the collaborative spirit required to strengthen TB genomics capacity within national laboratory systems.
The training forms part of broader efforts under the GenPath Africa consortium and the PARR-TB study, supported by the Global Health EDCTP3 Joint Undertaking and its members, the Gates Foundation, and the Health Emergency Preparedness and Response (HEPR) Programme managed by the World Bank Group.
A Continuous Process
Capacity building is a continuous process. As a continuation of this initiative, the team is already preparing the next training programme for 2026, focusing on the targeted next-generation sequencing
(tNGS) Deeplex® Myc-TB method, a technology already implemented in South Africa as a diagnostic tool for detecting TB drug resistance.
The upcoming training will introduce laboratory scientists to the Deeplex workflow and its diagnostic applications, further strengthening the integration of advanced genomic tools within the NHLS laboratory network.
Through sustained investment in training, collaboration, and innovation, initiatives such as PARRTB and GenPath Africa are helping ensure that genomic technologies translate into meaningful improvements in tuberculosis diagnosis, treatment, and surveillance.
And as World TB Day reminds us each year, ending TB will depend not only on technology, but on the scientists, laboratories, and communities working together to turn innovation into action.
Disclaimer: The information and perspectives presented in this document represent the views of the authors and do not necessarily reflect the official position of the National Health Laboratory Service (NHLS).
Yes! We Can End

The launch of the Social Science Unit (SSU) at CERI marks a strategic shift toward integrating social science into epidemic response and health research in Africa.
text & photo: Ameera Crew
The SSU was officially launched at the CERI Strategic Meeting that took place on 30 January 2026, and is led by Dr Astrid Treffry-Goatley – with Dr Gill Black as supporting lead. It brings together expertise across immunology, public health, ethnography, visual arts, communication, climate change, psychology, and political science.
The SSU responds to a critical gap: the need to embed social science within health research in ways that produce contextually grounded and impactful solutions. “The objective of the unit is to advance a human-centred approach by addressing the socio-political, socio-economic, cultural and ethical dimensions of health research”, explains Dr Treffry-Goatley.
In a changing global context, this integration becomes especially important. Health challenges are increasingly complex and interconnected. Addressing them requires collaboration across multiple disciplines and stakeholders. Funding expectations are also changing as major funders are placing growing emphasis on transdisciplinary research, recognising its greater impact, scalability and sustainability.
The SSU also strengthens institutional capacity at CERI by formalising social science within research programmes and enabling deeper integration across projects. The unit will:
• Co-develop competitive grant proposals.
• Lead and support social science components in funded projects.
• Build networks of local and international collaborators.
• Provide training in participatory methods, knowledge exchange, and policy translation.
A key example is the WEMA project: The impact of extreme weather events on the mental health of vulnerable populations in Africa. This project investigates the mental health impacts of extreme weather events across Africa.
Working across Mozambique, Kenya, South Africa, and Burkina Faso, the project uses participatory approaches to understand lived experiences and generate socially grounded evidence. The SSU team leads the social science and community engagement components, including the use of digital storytelling to capture personal narratives.
Throughout 2026, in collaboration with the Public Squares and African STARS Fellowship Programme, the unit will offer training opportunities focused on transdisciplinary research – including co-production, stakeholder engagement, and creative methodologies.
By embedding social and ethical insight into health research, the SSU ensures that scientific advances are shaped by the lived realities of communities. The unit represents an important step toward more inclusive, responsive, and impactful research in Africa and positions CERI at the forefront of transdisciplinary innovation in global health.
BELOW: Core team members of the CERI SSU. Front, left to right: Dr Gill Black, Dr Chanelle Mulopo, Dr Astrid Treffry-Goatley, Maambele Khosa. Back, left to right: Nicala Zeeman, Ameera Crew, Dr Moise Majyambere.

KRISP at South Africa’s inaugural Innovation Week.
text: Shirelle Naidoo & Paul Harris photos: Shirelle Naidoo & MidJourney
In a rapidly evolving scientific and technological landscape, the need for collaboration, visibility, and real-world impact has never been greater. It was within this spirit that South Africa Innovation Week 2026 brought together a dynamic cross-section of the country’s innovation ecosystem – from researchers and entrepreneurs to policymakers, funders, and the broader public.
Hosted by the Technology Innovation Agency (TIA), the event was positioned as South Africa’s official National Innovation Platform – a space designed not only to showcase ideas, but to accelerate their translation into meaningful outcomes. For the KwaZulu-Natal Research Innovation and Sequencing Platform (KRISP), participation in this landmark event represented an opportunity to both contribute to and help shape a more connected, forward-looking innovation landscape.
Unlike traditional conferences, South Africa Innovation Week offered a vibrant, accessible environment that encouraged interaction across disciplines. With exhibition spaces, presentations, and informal
networking areas, the event created a dynamic blend of structure and spontaneity. As a TIA-supported Technology Platform, KRISP showcased its work in genomics and data-driven science, engaging with a diverse audience that included researchers, industry stakeholders, students, and the public. The response was encouraging and insightful, reflecting growing curiosity about the role of genomics in advancing health and innovation.
For many visitors, the KRISP exhibition stand became a space of discovery – an opportunity to explore how complex scientific concepts translate into real-world impact. From pathogen genomics to KRISP’s role in the COVID-19 pandemic and HIV drug resistance research, the team demonstrated how genomic technologies are shaping public health responses in South Africa and beyond.
A striking aspect of the event was the diversity of engagement. School learners, university students, and individuals from non-scientific backgrounds interacted with researchers, asking questions and exploring ideas that might otherwise feel out of reach. This created an

important opportunity not only to showcase innovation, but to make it accessible.
By translating genomics into relatable, real-world applications, KRISP helped bridge the gap between scientific research and public understanding. For some attendees – whose exposure to genomics had been limited to consumer-facing platforms – the experience broadened their perspective on the field and its potential. In doing so, the event reinforced a key message: innovation is most powerful when it is inclusive, understood, and shared.
KRISP’s presence extended beyond the exhibition floor. The organisation’s Director, Prof Tulio de Oliveira, contributed to a high-level panel discussion titled “Owning South Africa’s Technological Future.” The session brought together thought leaders to reflect on the country’s innovation trajectory, with a focus on leadership, collaboration, and building local capacity. His insights – particularly on KRISP’s role in variant discovery during the COVID-19 pandemic – underscored the tangible impact of genomics in addressing real-world challenges.
Prof de Oliveria’s contribution highlighted not only the importance of scientific excellence, but also the value of investing in local expertise and infrastructure to drive sustainable innovation.
For KRISP, participation also carried strategic significance. As an event hosted by TIA – a key funder supporting its work – it provided an opportunity to demonstrate impact while strengthening relationships within the broader innovation ecosystem. The platform enabled meaningful engagement with potential collaborators, clients, and fellow TIA-funded initiatives, opening the door to new interdisciplinary partnerships. It also offered valuable insight into the breadth of innovation taking place across the country, reinforcing the importance of creating spaces where ideas can intersect and evolve.
Reflecting on the experience, members of the KRISP team described the event as energising and inspiring – a reminder of the creativity, curiosity, and potential across South Africa’s innovation landscape. With its blend of formal presentations, informal discussions, and even festival-like elements – from outdoor sessions to shared social spaces – the event created an environment where ideas flowed freely and connections formed naturally.
For KRISP, the takeaway was clear: innovation thrives through collaboration and shared knowledge. As South Africa invests in its innovation future, platforms like South Africa Innovation Week will remain key – with KRISP continuing to contribute to science that delivers real-world impact.
A conversation at SWEAT Africa 2026 explored both the promise of AI and the profound uncertainties that come with increasingly autonomous systems.
text & photo: SWEAT Africa

Amajor highlight of SWEAT Africa
2026 was the Side Event which featured an AI panel discussion – where Ozan Somnez, Kate Hach, Jayden Finaughty, Richard Rabbat, Jennifer Miles Thomas, and others unpacked the realities of AI integration in the modern workplace.
Moving beyond the usual hype, the conversation explored both the promise of AI and the profound uncertainties that come with increasingly autonomous systems.
For Kate Hach, who works at the intersection of deep tech, entrepreneurship, and organisational transformation, the answer to the panel’s central question: AI – will we make it or will it break us?,
ultimately comes down to human choices. “In short: us,” she said. “Humans built AI to support them, but we’ve also designed it for increasing autonomy –systems that can train themselves, make decisions, and take actions based on those decisions.”
Hach compared AI development to raising children: humans define the environment, provide examples and guidance, and gradually reduce oversight as autonomy grows. But there is a crucial difference.
“With humans, autonomous decision-making is always tied to responsibility and accountability,” she explained. “Even when parents are no longer involved, institutions exist to hold people accountable for their actions. With AI, we’re building systems that can act autonomously without that same accountability.”
A recurring theme during the panel was the need for AI conversations to involve founders, scientists, investors, and policymakers alike. “We’re building the ship while sailing it,” Hach said. “And in Africa especially, we need to make sure we are part of the conversations shaping its design.”
For AI to deliver real value on the continent, collaboration across science, investment, industry, and government will be essential, along with investment in infrastructure and locally relevant data.
Without indigenous datasets and locally developed applications, Africa risks becoming merely a consumer of technologies designed elsewhere.
Despite the rapid rise of AI tools, most organisations are still in the early stages of adoption. According to Hach, many companies are currently using AI primarily to optimise existing workflows – making processes faster, cheaper and more reliable – rather than fundamentally redesigning their systems. “It’s still largely known territory,” she said. “The architecture of the organisation stays the same.”
The next phase will come when organisations allow AI to play a greater role in designing systems themselves. “For now, we’re treating AI like a clever 14-year-old.”
Embedding AI into real workflows is rarely straightforward. Large organisations often struggle because clearly defined roles, locked-in processes, and incentive structures are designed for stability, not experimentation. “Innovation becomes difficult when companies are built for structure and predictability,” Hach said.
For founders, the challenge is different. In such a fastmoving field, what they build today may quickly become obsolete as new models emerge. This creates a constant strategic question: build, wait, or buy.
Hach’s experience working with accelerator programmes in Europe and supporting innovation ecosystems in Africa has shaped her view that emerging markets may sometimes adopt disruptive technologies more rapidly.
“In places where infrastructure is lacking, AI can do more than optimise systems – it can replace them entirely,” she said. At the same time, barriers such as
compute capacity and reliable energy remain major constraints in many African contexts.
One insight from the panel highlighted the human dimension of technological change.
Panelist Jayden Finaughty noted that integrating AI into database management requires cooperation from the very professionals whose roles may be affected by automation. “The labour force relenting their own erasure is a huge factor,” Hach said. Businesses and economies are human systems, and resistance to change is inevitable when livelihoods are involved.
Despite the risks, Hach sees enormous promise in AI –particularly its democratising potential.
Language barriers, literacy limitations, and technical training requirements have historically excluded many people from participating fully in digital economies. “AI has the potential to eliminate many of those barriers,” she said.
Tools that once required specialised engineering knowledge can now be built by individuals with far less technical training, opening new opportunities for innovation.
But the technology also raises serious concerns. “We’re building systems that can define their own capabilities without accountability mechanisms in place,” she warned. Combined with growing global investment in defense technologies, the race to deploy AI quickly may outpace efforts to regulate it responsibly.
The tone of the discussion reflected the atmosphere of SWEAT Africa itself. Held in an intimate garden setting, the gathering created space for founders, scientists and investors to engage openly with difficult questions about technology’s future. “Everyone felt they were part of something new,” Hach said. “That created an environment where people were willing to experiment with ideas and learn from each other.”
If the panel made one thing clear, it is that AI’s future is still being written – and the choices humans make now will determine whether it strengthens the systems we rely on, or destabilises them.
For Hach, the conversation is only just beginning. She is excited about what lies ahead. “I’m already looking forward to SWEAT 2027,” she said.
SU has joined a leading global network shaping how emerging technologies are understood, governed and applied. Through its Policy Innovation Lab, SU is strengthening international collaboration on AI and digital policy while positioning South Africa within key global conversations.

Stellenbosch University’s Policy Innovation Lab has joined the Global Network of Internet & Society Centers (NoC), coordinated by the Berkman Klein Center for Internet and Society at Harvard University. This makes SU one of only three members from Africa, and the only South African university represented in the network, says Prof Willem Fourie, head of the Policy Innovation Lab.
This membership is significant as the NoC has launched an ambitious 2024–2026 roadmap aimed at strengthening international collaboration on the societal impacts of emerging technologies, from artificial intelligence to quantum computing.
Through the Lab’s membership of the NoC, it has also now been invited to become the South Africa expert node for the AI Policy Observatory, a global initiative run by the University of Zurich and Kyoto University.
The NoC is a collaborative initiative that brings together leading academic centres focused on interdisciplinary research into the development, social impact, policy implications, and legal dimensions of the internet and emerging technologies.
Over the past decade, the network has supported global research collaborations, policy engagement, and educational initiatives that address how digital technologies are transforming societies, explains Prof Fourie. “Building on this foundation, the new roadmap sets out shared priorities for the next phase of collaboration. These include increasing the visibility and influence of the network in global technology policy debates, bridging information gaps around emerging technologies, and strengthening interdisciplinary partnerships across regions and sectors.”
To achieve these goals, the roadmap identifies five core areas of engagement that will guide the network’s activities over the next two years: internal collaboration and engagement; external engagement; shared knowledge and education; collaborative research and events; and global dialogue.
The roadmap identifies several thematic areas where collaborative research will be particularly important. One of the most prominent is generative artificial intelligence, a rapidly evolving technology that has raised questions about governance, ethics and social inclusion. The network aims to contribute to the development of governance frameworks that reflect diverse cultural, institutional and regional perspectives. This component of the NoC’s work aligns close with
the Lab’s work on the use of AI to transform the policymaking process, and in particular with its piloting of Africa’s first AI for policy platform.
Other areas of focus include the societal implications of quantum technologies, the rise of spatial computing and immersive digital environments, and the impact of technology on labour markets and future skills.
Researchers will also examine how digital technologies affect electoral processes and democratic systems, as well as the broader relationship between technology and human wellbeing.
Safeguarding children and young people in digital environments is another priority area, reflecting growing global concern about the effects of online platforms and emerging technologies on younger users.
The roadmap encourages experimentation with new approaches to understanding and regulating technology, explains Fourie. These include tools such as regulatory sandboxes and policy prototypes that allow governments and researchers to test new regulatory ideas before implementing them more broadly.
This component of the NoC’s work also resonates strongly with the activities of the Lab. Through its partnerships with leading global universities, such as the Technical University of Munich, the Lab hosts regulatory sandbox workshops focused on the regulatory changes required to implement digital public infrastructure in South Africa.
The network also highlights the role of creative practices – including storytelling, visual media and public art – in helping society better understand the complex relationship between technology and human life.
“By combining interdisciplinary research, global collaboration and innovative public engagement, the NoC aims to strengthen its role as a leading international platform for examining the societal implications of emerging technologies,” says Fourie.
Through this roadmap, the network hopes to deepen cooperation across regions, disciplines and sectors, ensuring that research on digital technologies contributes meaningfully to policy development, public debate, and more inclusive and responsible digital futures.
text: Katrine Anker-Nilssen photos: CERI Media & Thabiso Matemotja
Two emerging researchers at CERI, Khomotso (Kitty) Mohlala and Graeme Dor, have been awarded the SAMRC Biostatistics Capacity Development Initiative Award – a recognition that speaks not only to their individual potential, but to a broader need in health research: the ability to turn complex data into meaningful, actionable insight.
At its core, biostatistics sits at the intersection of data and decisionmaking. As health datasets grow in scale and complexity – from genomic sequences to behavioural and environmental data – the ability to analyse, interpret, and apply this information is becoming increasingly critical to how public health challenges are understood and addressed.
For Kitty, currently pursuing an MSc in Medical Virology, this
journey into biostatistics began during her Honours studies in Bioinformatics and Computational Biology at Stellenbosch University. “Through biostatistics, I learned that combining statistical analyses with biological data to derive valuable insights is both impactful and essential for evidence-based research,” she explains.
That foundation now underpins her current work analysing Human Papillomavirus (HPV) and Human Leukocyte Antigen (HLA) genotyping data, alongside behavioural data, to better understand the population-level impact of HPV vaccination in an HIV-endemic setting.
For Graeme, a PhD researcher, the path into biostatistics evolved from an early interest in spatial patterns and environmental data, developed through studies in Geographic Information Systems (GIS) and remote sensing. As his

work moved into public health, that analytical foundation found new application in infectious disease research. “The kinds of questions we’re trying to answer –around how diseases spread and what drives transmission – rely heavily on statistical thinking,” he says. His work now focuses on phylodynamic approaches, using statistical models to understand how viruses evolve and spread over time and space.
The SAMRC award plays a critical role in enabling this kind of work. While it provides financial support, both researchers emphasise that its value extends far beyond funding. For Kitty, it represents “recognition of the importance of my research and its potential impact on public health,” while also allowing her to focus fully on generating meaningful insights from complex datasets. For Graeme, the award marks an important step in building a sustainable research career, providing the space to deepen his work and contribute more directly to the development and application of statistical methods.
Importantly, the initiative is designed not only to support individuals, but to strengthen national capacity. By investing in postgraduate researchers, it builds the analytical expertise needed to address complex and evolving health challenges. This is particularly relevant in fields such as genomic surveillance and infectious disease modelling,

where the challenge is no longer just generating data, but making sense of it.
Both Kitty and Graeme’s work reflects this shift. Kitty’s research aims to generate evidence that can inform HPV vaccination strategies and support cervical cancer prevention efforts in South Africa. At the same time, she is developing advanced analytical and modelling skills to translate complex health data into actionable public health insights. Graeme’s work, meanwhile, seeks to integrate genomic, environmental, and ecological data to better understand disease transmission dynamics. By building tools that link these data streams, he aims to support more targeted and informed public health responses, particularly in the context of climate change and increasing human–environment interaction.
Together, their research highlights a central reality:
effective public health responses increasingly depend on the ability to interpret complex, multi-layered data. This requires not only strong technical skills, but also interdisciplinary thinking and the capacity to translate findings into practical impact.
Initiatives like the SAMRC
Biostatistics Capacity Development Initiative Award are therefore not simply funding mechanisms – they are strategic investments in the future of public health. By enabling researchers to develop the skills, tools, and insights needed to navigate complex data landscapes, they help ensure that scientific advances can be translated into better health outcomes.
Within CERI, this kind of capacity is central to advancing genomic surveillance, strengthening research, and supporting timely, evidence-based responses to infectious disease threats. And through researchers like
Kitty and Graeme, that capacity continues to grow – shaping not only individual careers, but the broader systems that underpin public health worldwide.
“The kinds of questions we’re trying to answer –around how diseases spread and what drives transmission – rely heavily on statistical thinking”
– Graeme Dor
Groundbreaking clinical study, VIBRANT, signals a new direction for Bacterial Vaginosis (BV) treatment.
text: CERI Media photo: MidJourney
Anew clinical study published in Cell Host & Microbe is offering a long-awaited shift in how bacterial vaginosis (BV) – one of the most common infections affecting women worldwide – may be treated. Known as the VIBRANT trial, the research moves beyond decades of antibiotic-only approaches, instead focusing on restoring the vaginal microbiome to support longerterm health.
For over 40 years, BV treatment has remained largely unchanged, relying on the same two classes of antibiotics. While these can clear symptoms in the short term, recurrence remains a major challenge, with up to 50–60% of women experiencing a return of infection within months. This reflects a deeper issue: while antibiotics eliminate harmful bacteria, they do
“By monitoring inflammatory cytokines in vaginal fluid over time, we can determine if introducing beneficial bacteria promotes a shift towards a more favourable, less inflammatory state.”
– Prof Lenine Liebenberg
not reliably restore the protective bacteria needed to maintain a healthy vaginal environment.
The VIBRANT study, a Phase 1 clinical trial, tested an early-stage live biotherapeutic product candidate designed to restore beneficial bacteria after antibiotic treatment. Specifically, the intervention delivers multiple strains of Lactobacillus crispatus – bacteria commonly found in healthy vaginal microbiomes – giving protective microbes the opportunity to reestablish, take hold, and persist.
Unlike traditional treatments that focus solely on eliminating infection, this approach aims to rebuild what BV removes. The candidate formulations were developed using vaginal L. crispatus strains isolated from women in both South Africa and the United States, ensuring representation of microbial diversity across populations.
Researchers from the Centre for Epidemic Response and Innovation (CERI) at Stellenbosch University played a central role in the study. Prof Jo-Ann Passmore led earlier work isolating and characterising more than 20 South African Lactobacillus strains, while Prof Lenine Liebenberg led analyses examining how the intervention influences inflammation in the genital tract.
“By monitoring inflammatory cytokines in vaginal fluid over time, we can determine if introducing beneficial bacteria like L. crispatus promotes a shift towards a more favourable, less inflammatory state,” said Prof Liebenberg. “The early findings are encouraging.”
The trial included participants at sites in both South Africa and the United States. After receiving standard antibiotic treatment (metronidazole), participants were given either a placebo or one of four live biotherapeutic product candidate regimens for seven days.
The results showed:
• The candidates were safe and well tolerated.
• 66% of participants receiving the live biotherapeutic candidate showed colonisation by at least one beneficial strain within five weeks.
• Among those who initially colonised, 49% remained colonised 10 weeks later – despite treatment lasting only one week.
• Colonisation patterns were consistent across both study sites, suggesting broader applicability across populations.
While these findings do not yet demonstrate prevention of BV recurrence, they provide important early evidence that beneficial vaginal bacteria can successfully establish and persist after treatment.
BV is a highly prevalent yet often under-recognised condition, affecting an estimated one in two women of reproductive age globally. Beyond discomfort, it is associated with increased susceptibility to sexually transmitted infections, including HIV, as well as adverse pregnancy outcomes such as miscarriage and preterm birth.
If future studies confirm that microbiome-based therapies can reduce recurrence, this approach could represent the first major advancement in BV treatment in decades – offering longer-lasting protection, improving reproductive health outcomes, and reducing infection risk globally.
Built through Global Vollaboration – and Grounded in Africa
The VIBRANT study reflects a large-scale international collaboration involving institutions across South Africa, the United States, and Europe, including Stellenbosch University, the University of Cape Town, CAPRISA, Harvard Medical School, and others. The work builds on years of research through the Vaginal Microbiome Research Consortium and was supported by the Gates Foundation.
Importantly, the study also reflects a shift in how global health research is conducted. Rather than being tested on African populations, the science was built with African women and grounded in African microbial data – ensuring relevance, representation, and equity in innovation.
As Prof Tulio de Oliveira, Director of CERI, notes: “This study shows how scientific collaboration across continents can generate new solutions to long-standing health challenges. It also reflects the growing leadership of African scientists in microbiome research.”
With safety established and early signs of durable colonisation observed, researchers are now planning further early-phase studies to better understand how these multi-strain L. crispatus live biotherapeutic candidates may reduce BV recurrence and support long-term vaginal health.
If successful, this approach could mark a milestone in women’s health – addressing a condition that has long needed new tools, new research, and new hope.
Full publication: https://lnkd.in/dmchzh48
text & photo: CERI Media
So... What does it take to identify a disease you have never seen before? Or to build the systems that hold critical health data together across an entire continent? These are not hypothetical questions. They are the problems that Akhil Maharaj (pictured below, right) and Danilo De Silva (pictured below, left) spent their Master’s years trying to solve.
On 24 March, at the Stellenbosch University’s graduation ceremony, both walked across the stage to show what that work looks like when it comes to completion.
Akhil’s research explored the applications, complexities and opportunities of metagenomics in clinical diagnosis and viral discovery. Where traditional diagnostics search for a known pathogen, metagenomics
sequences everything in a biological sample at once. Akhil describes it as “a Pandora’s box of sequencing.” For clinicians facing patients with unknown diagnoses, it is a powerful supplementary tool to uncover hidden pathogens, inform targeted therapeutics and ultimately enhance patient care.
Akhil’s journey began at the KwaZulu-Natal Research Innovation and Sequencing Platform (KRISP), before deepening at the Centre for Epidemic Response and Innovation (CERI) at Stellenbosch University. Guided by Prof Tulio de Oliveira, Dr Monika Moir, Prof Houriiyah Tegally, and Dr Marije Hofstra, Akhil emerged not just as a scientist but as someone who knows how to think through hard problems. That same thinking is now taking him into Corporate and Investment Banking. “The skills developed in science translate well into finance:

identifying problems, testing hypotheses, and turning findings into clear, actionable insights.”
Danilo’s story began in Governador Valadares, Brazil, where an early love of computers and science set him on a path that would eventually lead him across the ocean to South Africa. After completing his undergraduate degree and working briefly in industry, he received an invitation to pursue a Master’s at CERI, developing data management systems for the INFORM Africa Research Study Group. Behind every major health study is infrastructure holding the data together. Danilo evaluated and designed data management tools to develop a broader infrastructure to support health-related research applications.
Under the supervision of Dr Marcel Dunaiski, with co-supervision from Dr Monika Moir and Dr Joicy Xavier, he contributed to academic publications and found his long-term direction. Danilo is now staying at Stellenbosch to pursue a PhD. “My experience at CERI and within SU helped me challenge these perceptions and see the transformative potential of research.”
Together, Akhil and Danilo’s stories are a reminder that world-class research is being done in and for the Global South, and the people producing it are going on to shape science, finance, technology and beyond.
We are incredibly proud of you both.
How Akhil Maharaj took his research skills from the lab into corporate and investment banking.
text: CERI Media
Not every great scientist ends up in a lab. Some take everything science taught them, the rigour, the logic, the ability to look at a problem, work through it systematically, and carry it into industries that never saw them coming.
On 24 March, at the Stellenbosch University Faculty of Science graduation ceremony, Akhil Maharaj walked across the stage to receive his MSc in Bioinformatics and Computational Biology. But the road to that moment started long before, in Durban, where a passion for science, and specifically the statistics behind it, was already taking root.
Akhil completed his undergraduate degree in genetics and microbiology, followed by honours in genetics at the University of KwaZulu-Natal. His first real encounter with high impact science came through an opportunity at KRISP, the KwaZuluNatal Research Innovation and Sequencing Platform, where he witnessed science operating at its most consequential. That experience set the direction. He went on to pursue his Master’s at the Centre for Epidemic Response and Innovation (CERI) at Stellenbosch University, where the work deepened and the stakes became even clearer.
His Master’s research focused on the applications, complexities and
opportunities of metagenomics in clinical diagnosis and viral discovery. Metagenomics is the science of sequencing everything in a biological sample at once, not just searching for one known pathogen, but casting the widest possible net.
For public health, the implications are profound. In clinical settings, where a patient’s diagnosis remains unknown, metagenomics gives clinicians a powerful supplementary tool to uncover hidden pathogens, inform targeted interventions, and enhance patient care. In a world still reckoning with the lessons of COVID-19 and the constant threat of emerging infectious diseases, that kind of science matters enormously.
His supervisors, Prof Tulio de Oliveira, Dr Monika Moir, Prof Houriiyah Tegally and Dr Marije Hofstra, played a role that went far beyond the technical. “My supervisors played a pivotal role in shaping my research journey. Beyond sharing their expertise, they consistently challenged me to think critically and push the boundaries of my work. They were always available for guidance when needed, while also instilling in me a clear understanding of what rigorous, high-impact science should look like.” Akhil reflects.
After years immersed in genomics and research, Akhil made a bold and deliberate move into Corporate and Investment Banking. Far from leaving science behind, he carried its most powerful asset with him: the
ability to think critically. “The skills developed in science translate remarkably well into the financial field. While the underlying theory may differ, the core approach remains the same; identifying problems, formulating and testing hypotheses, and most importantly, translating findings into clear, actionable insights. These shared foundations underpin many of the key qualities required in finance.”
He had long held a keen interest in finance, particularly foreign exchange (FX) trading, commodities and geopolitics. Today he is part of a graduate cohort rotating across multiple business units in Corporate and Investment Banking, gaining the practical experience and exposure that will shape his long-term career.
For young graduates sitting with a science degree and an open horizon, Akhil’s path is a powerful reminder that what you study gives you a way of thinking, and that way of thinking is currency in almost any field.
“Stay curious, and take in everything you can. You don’t need all the answers right now, what matters is your willingness to explore and grow. Every experience adds a stroke to your journey. The canvas is yours, and you hold the brush. Paint Boldly.”
– Akhil Maharaj
title:
Approaches to the Management of Virologic Failure on Dolutegravir-Based Antiretroviral Therapy in the 50 Countries With the Highest Adult HIV Prevalence
authors:
Cameron T Nutt, Richard J Lessells, Winnie R Muyindike, Lyra S Johnson, Roger D Kouyos, Tom Loosli, Loice Achieng Ombajo, Jaysingh Brijkumar, Emily P Hyle, Mahomed-Yunus S Moosa, Monica Gandhi, Marco Antonio De Avila Vitoria, Michael R Jordan, Mark J Siedner, Suzanne M McCluskey
As access to dolutegravir-based antiretroviral therapy (ART) continues to expand globally, it has transformed HIV treatment – offering a potent, durable, and widely adopted regimen for millions of people. Yet as its use scales, a critical question is emerging for clinicians and policymakers alike: how should virologic failure be managed in patients receiving this first-line therapy?
A recent global review of national HIV treatment guidelines across the 50 countries with the highest adult HIV prevalence highlights a complex and evolving policy landscape. While dolutegravir-based regimens remain highly effective, instances of virologic failure – though relatively uncommon – are prompting divergent strategies in clinical management. These differences reflect a balance between scientific evidence, resource availability, and public health priorities, underscoring the need for clearer data to guide future decisions.
Publication Summary
Expanded access to dolutegravir as part of a fixeddose combination with tenofovir disoproxil fumarate and lamivudine (TLD) has revolutionized global HIV treatment, with more than 25 million people prescribed this regimen in 2024. With a team led
by our clinical collaborators at Massachusetts General Hospital and Harvard Medical School, together with African and international partners (including WHO representatives), we reviewed national clinical guidelines for management of virologic failure on first-line TLD in the 50 countries with the highest prevalence of HIV among adults; recent guidelines were not available online for four. Among the remaining 46 country guidelines, we identified four distinct approaches: 1) empiric switch to a protease inhibitor (PI)-based regimen without genotypic resistance testing (GRT) (n=28; 61%); 2) GRT to guide antiretroviral therapy selection (n=14; 30%); 3) continuation of TLD without GRT (n=3; 7%); and 4) empiric switch to a PI-based regimen with concurrent GRT (n=1; 2%). The diversity of guidelineendorsed approaches underscores the critical need for additional data to inform policy regarding this important clinical scenario.
For countries carrying the highest burden of HIV –including many in Africa – these differences in clinical guidance are not theoretical. They directly shape how millions of people are treated when first-line therapy no longer suppresses the virus.
At the heart of the issue is a fundamental trade-
off. Switching patients too quickly to second-line regimens may introduce higher costs, greater side effects, and more complex treatment pathways. But delaying a switch – particularly without access to resistance testing – risks keeping patients on failing regimens, with implications for both individual health outcomes and the potential spread of drug-resistant virus.
The variability in national approaches also reflects deeper system realities: unequal access to diagnostic tools like genotypic resistance testing, differences in healthcare infrastructure, and the financial pressures facing HIV programmes globally. As funding constraints tighten, these decisions become even more consequential.
What this review makes clear is that the science is still catching up with policy. As new evidence emerges – from ongoing clinical trials such as our own RESOLVE study and the Ndovu study – it will be critical to align guidelines with what works best in practice. Doing so will help protect the long-term effectiveness of dolutegravir-based treatment, while ensuring that patients receive the most appropriate care at the right time.
To read the publication and access a full list of references, please visit:
https://pmc.ncbi.nlm.nih.gov/articles/ PMC12380185/
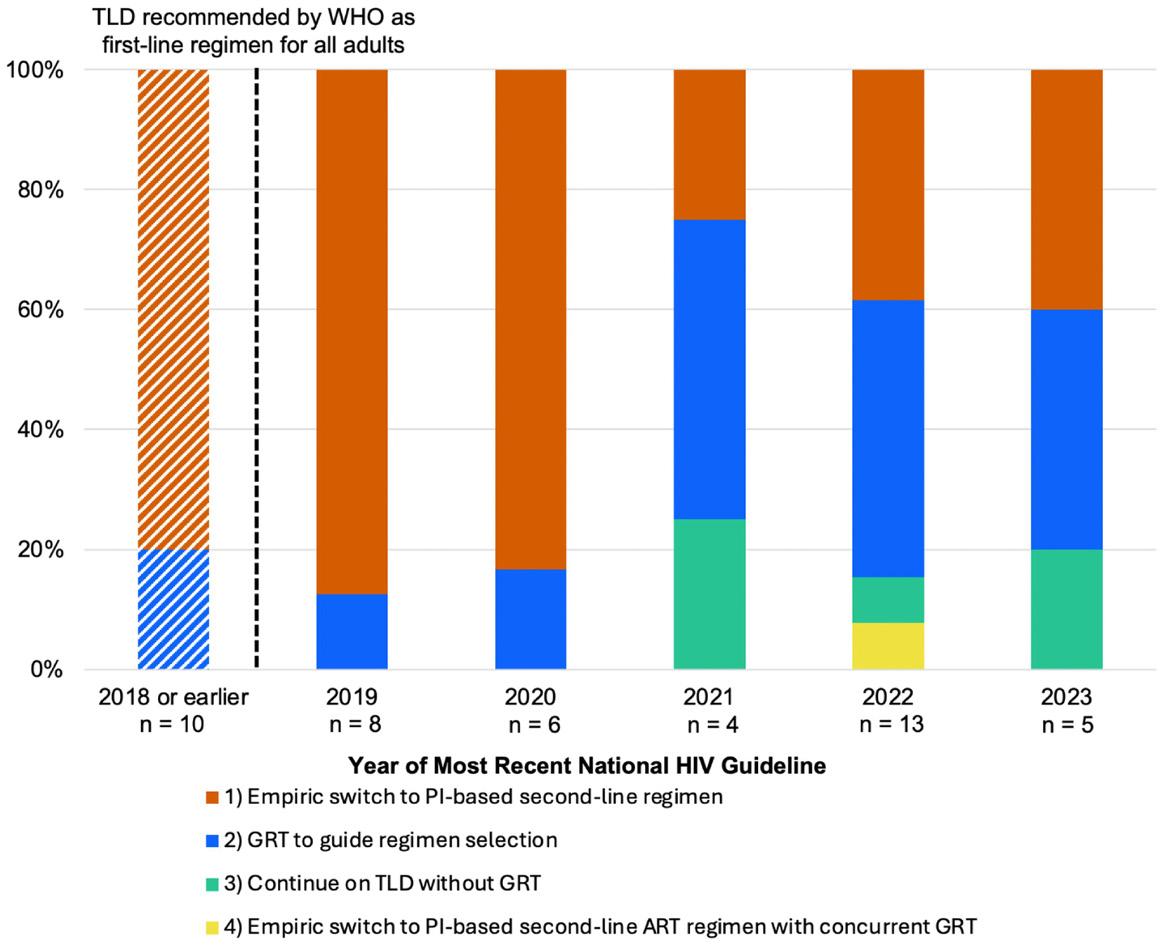
Dr Lessells is an Infectious Diseases clinician scientist at the KwaZulu-Natal Research Innovation & Sequencing Platform (KRISP), a research centre at the University of KwaZulu-Natal in Durban, South Africa; and an honorary research associate at the Centre for the AIDS Programme of Research in South Africa (CAPRISA). He did his clinical specialist training in Infectious Diseases and Internal Medicine in the U.K., and his PhD at the London School of Hygiene and Tropical Medicine. Dr Lessels has been actively involved in HIV and TB research in South Africa since 2007, initially in rural KwaZulu-Natal and then at UKZN since 2017. His current research includes an NIH-funded multi-country observational study of dolutegravir drug resistance in people with HIV (DTG Resist); an NIH-funded randomised controlled trial of strategies for the management of people with HIV who have virological failure on dolutegravir-based ART (RESOLVE); and an international multicentre randomised controlled trial of high-dose rifampicin for TB meningitis (HARVEST).
Figure 2 (top, right):
Evolution of recommended approaches to management of virologic failure on firstline TLD by year of most recent published national HIV guidelines. Abbreviations: ART, antiretroviral therapy; GRT, genotypic resistance testing; HIV, human immunodeficiency virus; PI, protease inhibitor; TLD, tenofovir disoproxil fumarate + lamivudine + dolutegravir

text & photos:
Paul Harris
t the KwaZulu-Natal Research Innovation and Sequencing Platform (KRISP), a recent weeklong Illumina Library Preparation and Sequencing Workshop brought together more than just samples, reagents, and sequencing platforms – it brought together people, ideas, and a shared vision for the future of science on the African continent.
Seventeen participants from across the region –including the University of Limpopo, University of the Free State, Sefako Makgatho Health Sciences University, and the Armauer Hansen Research Institute – gathered in Durban for an intensive, handson training experience. What unfolded over the course of the week was not only a deep dive into nextgeneration sequencing, but also a powerful reminder of how science can unite individuals across borders through a common purpose.
Laying the Foundation
The workshop opened with an introduction to Illumina

sequencing technology, grounding participants in the principles that underpin modern genomics. But it was the human element – the exchange of ideas, backgrounds, and expectations – that set the tone. Participants shared insights into their respective institutions and research focus areas, immediately establishing a sense of community rooted in collaboration.
From there, the group moved into the laboratory, where the sequencing journey began. Early sessions focused on observing the critical steps of the workflow – from tagging and amplification to post-tag clean-up – offering participants a clear view of the precision and care required at every stage.
As the week progressed, the laboratory became a space of growing confidence and curiosity. Participants engaged with increasingly complex processes, including quality control, gel electrophoresis, and library quantification. Discussions flowed easily, questions were encouraged, and knowledge was shared openly – a reflection of KRISP’s commitment to meaningful capacity building.
By the third day, the shift was tangible. No longer observers, participants stepped into the role of practitioners. Working in pairs, they carried out their own library preparation and quality control, translating theory into hands-on experience. It was here, at the bench, that learning truly came alive – through collaboration, problem-solving, and a shared sense of purpose.
As Refilwe Motswiane, a PhD student from SMU,

reflected: “The hands-on experience we got this week really showed us how capable we are to work with and perfect new protocols and workflows in our own laboratories.”
The final stages of the workshop saw participants preparing their libraries for sequencing and loading them onto the Illumina MiSeq. This moment – where individual efforts converged into a collective output –marked a significant milestone in the week’s journey. For many, the experience opened new doors. Tshepiso Khoza, an MSc student from the University of Limpopo, shared: “It was my first time being exposed to Whole Genome Sequencing and this type of data analysis, which will be a critical part of my research going forward.”
On the final day, participants assessed their results and reflected on the skills they had gained. The sense of achievement was evident, but equally important was the recognition of what lay ahead: the application of these skills within their own institutions, and the continued strengthening of genomics capacity across the continent.
While the technical training formed the backbone of the workshop, its broader impact lies in the connections forged along the way. Across institutions, disciplines, and countries, participants found common
ground in their commitment to advancing science for public health.
This spirit of collaboration was echoed by Fekadu Acemu, a Senior Health Researcher at AHRI in Ethiopia: “It is a great opportunity to be here at KRISP to see how they work and learn about all of the exciting research going on. There is a lot of common ground and areas where our two institutes can collaborate together. We are excited for the future.”
The workshop also highlighted the importance of partnership – with support from sponsors and collaborators playing a key role in making this initiative possible. Together, these efforts contribute to a growing network of skilled scientists equipped to respond to current and future health challenges.
As the group transitions into the next phase of training in bioinformatics data analysis, the journey continues – moving from the bench to the screen, and from data generation to insight.
Workshops like this serve as a powerful reminder that science does not happen in isolation. It thrives in environments where knowledge is shared, skills are nurtured, and collaboration is embraced. At KRISP, this vision remains clear: by investing in people and partnerships, Africa is not only building capacity – it is shaping the future of genomics and global health.
The Centre for Epidemic Response and Innovation (CERI) invites applications for a five-day intensive Training in Metagenomic Virus Discovery. This hands-on workshop will be led by global experts, offering participants practical skills and strategies in virus discovery—from project design to advanced analysis.
About the Training
The programme combines core lectures in metagenomics and phylogenetics with hands-on sessions focused on:
• Project planning and experimental design.
• Building metagenomic analysis pipelines.
• Downstream analysis (e.g. phylogenetics, structural analysis).
• Interpreting and presenting results (e.g. virus evolution, genomic epidemiology).
Who Should Apply
This training is designed for individuals with some experience in bioinformatics and the command line, and basic knowledge of virology. It is suitable for:
• Postgraduate students.
• Postdoctoral researchers and fellows.
• Scientists and senior scientists.
While not tailored to a specific virus application, the training includes examples from animal-infecting viruses, using a mammalian dataset for tutorials.
Funding and Support
Supported by the Rockefeller, full funding is available for selected researchers residing in Africa. This includes:
• Airfare and local ground transport .
• Accommodation.
• Registration.
Registration Fee
R13 000 (covers training meals and two networking events).
Application
To apply, please visit www.ceri.org.za/training or scan the QR code on the right.
For any questions, email Dr Erin Harvey: mvdworkshop2025@gmail. com



Are you a scientist working in virus evolution, bioinformatics, genomic surveillance, or related computational fields? Applications are now open for the Virus Evolution & Molecular Epidemiology (VEME) 2026 workshop — a leading training programme at the forefront of bioinformatics and evolutionary analysis.
VEME 2026 provides essential, in-depth training for scientists working across these cross-cutting fields.
The workshop brings together leading researchers from around the world to deliver both practical and theoretical foundations in bioinformatics. Participants will gain the skills needed to analyse complex genomic datasets, address key bioinformatics questions, and contribute meaningful insight to research communities and public health systems.
The 2026 programme consists of two specialised modules designed to provide a comprehensive overview of current methods used to analyse the vast data generated by modern DNA sequencing technologies: Phylogeny Inference & Evolutionary Hypothesis Testing.
Location: Bertha Retreat, Stellenbosch, South Africa
Dates: 6–11 September 2026
Apply Here: https://veme.climade.health/
At KRISP, two emerging scientists show how resilience, faith, and purpose are driving a new generation of genomics research.
text & photos: Paul Harris
At the KwaZulu-Natal Research Innovation and Sequencing Platform (KRISP), the future of African genomics is being shaped by a new generation of scientists whose journeys are as compelling as the research they lead. Though their paths differ, Thembelihle Tombo and Nqobile Mthembu share a common purpose – to use science in service of society. Their stories reflect resilience, faith, quiet determination, and the courage to grow, showing what is possible when talent meets opportunity.
THEMBELIHLE TOMBO
Born in Mpumalanga and raised in Richards Bay, KwaZulu-Natal, Thembelihle Tombo describes herself as naturally quiet and reserved – a trait she now recognises as a strength. “People sometimes mistake quietness for weakness,” she reflects, “but I have come to understand that it can also be a form of strength.”
Like many students, she excelled at school without full exposure to career possibilities. What she did know was that she wanted to make a difference. That purpose took shape when she began to see science not

just as an academic pursuit, but as a tool for impact. At university, representation played a pivotal role.
“Seeing women in science changed the way I viewed the field,” she says. “I had imagined science as a difficult and lonely path, but I met women who were hardworking, confident, and full of life.” Among those who inspired her was Dr Yanga Mdleleni, whose example helped her see that she belonged.
Her path into genomics and medical research brought uncertainty, self-doubt, and social anxiety. With support from her supervisors, the KRISP team, and her family, she found her footing. “What helped me was staying committed, working hard, and leaning on the people who believed in me – even when I struggled to believe in myself.”
Thembelihle’s MSc research, completed cum laude, focused on making sequencing more accessible. Titled Evaluation of a single assay for surveillance of multiple viral pathogens using Next Generation Sequencing, it explored whether lower-cost approaches could still deliver high-quality results when detecting multiple viruses. In simple terms: can we do more with less –without compromising quality?
The implications are significant. In resource-limited settings, where sequencing costs are a barrier, even small efficiencies can expand access to genomic surveillance tools. “This research is important because affordable genomic surveillance supports better monitoring of viruses and stronger public health responses,” she explains. “If we can reduce costs, more laboratories can generate the data needed to protect communities.”
Learning she had graduated cum laude came as a surprise. “Honestly, I was in disbelief,” she says. “It felt unreal because I knew how difficult the journey had been.” More than an academic milestone, it marked a turning point: “It helped me start letting go of selfdoubt. Hard work – even when it feels unseen – can lead to something meaningful.”
Now pursuing her PhD at KRISP, she is focused on deepening her research and impact. “I want to become a researcher who makes a tangible difference in people’s lives,” she says. She also hopes to inspire others who may doubt they belong in science. “I want to show that there is space in science for people with different personalities, backgrounds, and voices.”
Her story carries a message beyond the lab: “Being quiet does not mean you have nothing to offer. Sometimes strength is not loud.” Guided by humility and perseverance, she holds onto the lesson: “I try, I fail, but at least I never failed to try,” alongside the Zulu proverb Ophansi akawi.
Thembelihle’s journey shows that impact does not always begin with confidence – sometimes it begins quietly, with the courage to keep going.
Raised in Pietermaritzburg, Nqobile Mthembu ’s path into science was shaped by both academic ability and a desire to contribute to something greater than herself. Her time at UKZN was formative.
“My interest in science grew through my exposure to women in science,” she reflects. “Seeing women thrive made me believe that I, too, could contribute in a meaningful way.” Inspired by mentors such as Dr Danisile Tembe and Dr Yanga Mdleleni, she came to see science as a space where she could belong – and make a difference.
Like many young researchers, she had to step into unfamiliar spaces. “One of the biggest challenges has been stepping into new spaces,” she says. “I learned to overcome that by being patient, staying committed, and trusting the process.”
Drawn to KRISP through its role in COVID-19 genomics, she entered an environment where innovation meets real-world impact.
Her Master’s research, completed cum laude, focused on using existing resources more effectively. Titled Advancing Genomic Surveillance: A Single Streamlined Next-Generation Sequencing Approach for South Africa’s Common Respiratory Viruses, it explored how COVID-19 sequencing infrastructure could be repurposed to monitor other respiratory viruses. In simple terms: how can we extend the life and value of the tools we already have?
“This work is about affordability and accessibility,” she explains. “Many laboratories have COVID-19 sequencing infrastructure, but not all know how to use it beyond the pandemic.” The implications are significant,
especially in resource-limited settings. “If we can repurpose existing infrastructure, we can continue generating valuable public health data without needing entirely new systems.”
When she graduated cum laude, the moment was deeply emotional. “I felt incredibly happy, grateful, and relieved,” she recalls. “It reflected the early mornings, late nights, prayers, and perseverance.” It also affirmed her confidence in contributing meaningfully to science.
Looking ahead, she is guided by both ambition and purpose. “I want to be part of work that makes genomic technologies more accessible… Science should not only be innovative – it should be practical and useful.”
Nqobile draws strength from her family, friends, church community, and faith. Proverbs 16:9 – “We can make our plans, but the Lord determines our steps” – has helped her navigate uncertainty with trust. “It reminds me to work hard, but also to stay open to where life leads.”
She values balance and encourages young women entering science: “Work hard, believe in your ability, and do not be afraid to take up space… but do not lose yourself in the process.”
Her definition of success reflects this: “Growing into the fullest version of yourself, using your gifts well, and making an impact while staying true to who you are.”
Nqobile’s journey reflects a strong sense of purpose and commitment to science that serves society.


Prof Tulio de Oliveira and Dr Richard Gordon on Africa’s Startup Future: Join us on the ground at Sweat Africa 2026, Africa’s first experiential startup and venture festival taking place at the Bertha Retreat, Stellenbosch, South Africa, where founders, investors, students and ecosystem builders are working, learning and sweating together to shape the future of African innovation.
Watch the full video here: https://youtu.be/u6lGRAwe-7k
Fluorobiotech is a South African biotech startup at the forefront of innovation, and behind every great company is the next generation of talent shaping it. In this video, interns at Fluorobiotech share their firsthand experience working at the intersection of science, entrepreneurship, and social impact. These interns were placed through the Chan Soon-Shiong Family Foundation (CSSFF) Programme of the South African Medical Research Council (SAMRC) – a proud partner of SWEAT Africa.
Watch the full video here: https://youtu.be/4ubZGIuoJ5M


Which Skills Help Scientists Succeed Beyond the Lab? Writing skills! Prof Tulio de Oliveira shares his top advice for scientists: while technical and lab expertise are vital, soft skills such as writing for non-specialists can open new doors – from securing grants to creating broader impact. Learn why upskilling yourself beyond the field of science is just as important as conducting it.
Watch the full video here: https://www.youtube.com/watch?v=Tu4bl3PJPHQ
What does the future of African innovation look like? At the #SWEATAfrica Tech Event, some of the continent’s most influential voices in science, entrepreneurship, biotech, and economic leadership came together to shape that answer.
Watch the full video here: https://www.youtube.com/watch?v=NXw8xGUxiWk


This bootcamp brought together pan-African founders building science-based ventures with a cross-continental circle of experts and investors focused on market and investment readiness. It was a two-way exchange. Founders got challenged on positioning, customer reality, and go-tomarket strategy. Experts gained real market context from the cohort. The result: honest, rigorous work that DeepTech demands.
Watch the full video here: https://www.youtube.com/watch?v=6vq6cohq16Y
The difference between good ideas and real impact? The ecosystem around them. As Tebogo Machethe from University of the Witwatersrand shares, when leading institutions plug into networks like #SWEATAfrica, research moves faster, collaborations deepen, and knowledge actually translates into change.
Watch the full video here: https://youtu.be/4JPXOdpoYh8

