
OriginalArticles
EcologicalIndicators
journalhomepage: www.elsevier.com/locate/ecolind
Benthicforaminiferaasbioindicatorsofpotentiallytoxicelement(PTE) pollution:Torrecillaslagoon(SanJuanBayEstuary),PuertoRico
MichaelMartínez-Colón a,⁎,PamelaHallockb,CarlosR.Green-Ruízc,JosephM.Smoakd
a SchooloftheEnvironment,FloridaA&MUniversity,FSHScienceResearchCenter(RM306B),1515SouthMLKBlvd.,Tallahassee,FL,32307,UnitedStates
b CollegeofMarineScience,UniversityofSouthFlorida,8301stStreetSouth,St.Petersburg,FL,33701,UnitedStates
c InstitutodeCienciasdelMarLimnología,UniversidadAutónomadeMéxico,Ave.JoelMontesCamarenas/n,82000,Mazatlán,Sinaloa,Mexico
d EnvironmentalScience,UniversityofSouthFlorida,1407thAveSouth,St.Petersburg,FL,33701,UnitedStates
ARTICLEINFO
Keywords:
Heavymetals
Ecology
Caribbean Bioavailability
Hypoxia


ABSTRACT
TorrecillasLagoon,onthenorthcoastofPuertoRico,hasexperiencedextensiveanthropogenicinfluenceover thepast200years.ElevatedconcentrationsofPotentiallyToxicElements(PTEs)inbulksediment(Cu,Zn,Pb, Ni,Cr,Li,V,Fe,As,Se,andMn)havebeenreportedinsurficialsedimentsandhaverelativelyuniformspatial distributions.Areaswithhigherconcentrationsareassociatedwithahigherpercentageoftotalorganiccarbon (TOC)andpercentmud(mud),aswellasanoxicconditions. Ammoniabeccarii, Quinqueloculinarhodiensis,and Triloculinaoblonga arethedominantforaminifersinthelagoonandarecharacteristicofstressedcoastalenvironments.BulkconcentrationsofCu-Zn-Fearenegativelycorrelatedwithnumerousforaminiferaltaxa,absoluteabundances,anddiversityindices,thoughveryfewcorrelationswiththebioavailablecounterparts (F2Tess-bioavailable)areobserved.Similarly,relativeabundancesof Quinqueloculina and Triloculina positively correlatewithbulkCu-Zn-FebutnotwithF2Tess-bioavailable.ThewatersinTorrecillaslagoonshowstrong stratification,withhypoxic/anoxic(dissolvedoxygen<3mg/L)andcorrosive(pH<7.4)conditionsbelow4m depth.Thepresenceofsuchstronggradientsinveryshallowwaterrepresentsadynamicchemicalenvironment, withchangesoccurringonday-nightcycles,tidalcycles,andespeciallywithstormactivitythatinducesmixing ofotherwisehighlystratified,verylocalizedwaters.RecognizingthepotentialforsequesteredPTEstoberemobilizedisanessentialinsightforcoastalmanagementagenciesthatmustassesstherisksofexistingPTEs duringcoastalengineeringactivities(e.g.,dredgeand fillactivities)andmajorstormevents.Exchangeableand oxidizablefractionsarelikelymorebioavailablethanacid-solublefractionsininfluencingtheecologyofforaminifersundermostcircumstances.
1.Introduction
Estuariesareexperiencingadversechangesthatareinfluencingthe naturalvariabilityoftheseuniqueecosystems.Coastallagoonscommonlyserveassinksforawiderangeofpollutants(Zaaboubetal., 2015 ).Rapidindustrialization,overpopulation,surface-wateralteration,andotherfactorsarechangingestuarineenvironmentsandbiotic communities(e.g., Green-Ruízetal.,2005;HunterandArbona,1995). Estuariesreceiveapproximately80–90%ofallwastereleasedto marineenvironments( Gross,1978)andthereforeareparticularly vulnerabletopollutionbypotentiallytoxicelements(PTEs).Such PTEs(see Martínez-Colónetal.,2009 fordefi nitionandfurtherdiscussion)canhavedetrimentale ffectsonwaterquality,aquaticecosystems(i.e.,reducingabundanceanddiversity,etc.),andhuman
health(e.g., Laboy-Nieves,2009 ).
InnorthernPuertoRico,TorrecillasLagoon(TL)hasbeenextensivelyalteredbyanthropogenicactivities(Figs.1and2).FromPreColombiantimestopresent,humansettlements,agriculture,deforestation,landfills,dredgingandurbandevelopmenthaveaffectedthis extensivelagoonsystem(Ellis,1976;Seguinot-Barbosa,1983).During thelate1800s,sugarcanewasgrownaroundTL(Wilson,1899).At present,TLisinfluencedbyawiderangeofpointandnonpointsources ofpollution(Table1),coupledwithlocalizedanoxicconditionswithin dredgednavigationalchannels.
Previousstudieshavereportedthatorganicpollutants(PCBs, Dieldrin,DDT),aswellaselevatedconcentrationsofPTEs(As,Se,Cu, Hg,CdNi,Zn,andPb)insedimentsofTLareanthropogenic (Martínez-ColónandHallock,2010;SanJuanBayEstuary,2000;
Abbreviations: PTE,Potentiallytoxicelement;F2Tess-bioavailable,CarbonatebioavailablefractionfromTessieretal.(1979)
⁎ Correspondingauthor.
E-mailaddress: michael.martinez@famu.edu (M.Martínez-Colón).
http://dx.doi.org/10.1016/j.ecolind.2017.10.045 Received4June2017;Receivedinrevisedform21August2017;Accepted20October2017
1470-160X/©2017ElsevierLtd.Allrightsreserved.
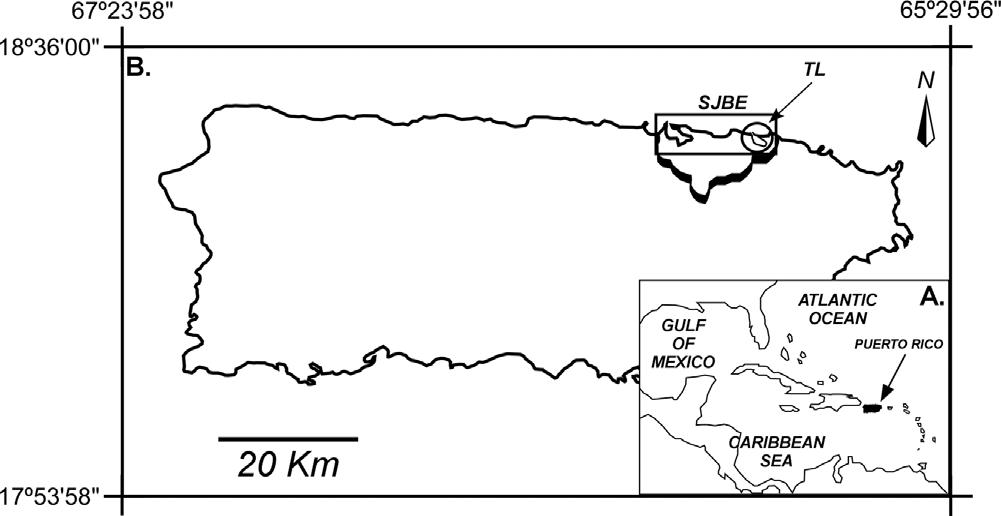
Fig.1. A.LocationofPuertoRico.B.MapofPuertoRico.Rectangle:SanJuanBay Estuary(SJBE).Circle:TorrecillasLagoon(TL).Raisedline:SJBEwatershed.
WebbandGómez-Gómez,1998 ).
Avarietyofproxieshasbeenimplementedtoidentifypollutantsin coastalwaters,includingmacro-andmicro-invertebrates,aquatic plants,and fish.Benthicforaminifers(shelledprotists)wereextensively usedasbioindicatorsofpollutionincoastalenvironmentsofPuerto Ricoby Seiglie(1968,1971,1975c) andmorerecentlyby,forexample, Oliveretal.(2014).Worldwide,foraminifersareknowntorespondto pollutionthroughchangesinassemblagemakeupandabundance(e.g., Alve,1995;Emrichetal.,2017;Yanko-Hombachetal.,2017),andto displayavarietyofmorphologicalabnormalitiesthatareconsidereda biologicalresponsetoenvironmentalstresses(e.g., SamirandEl-Din, 2001;Yankoetal.,1998).Benthicforaminifersarelikelysensitiveto exposuretoPTEsgiventhehighsurface-to-volumeratiosofthese
protistsandtheirinterrelationshipwithsedimenttypeandphysicochemicalparametersinbottomandporewaters.Benthicforaminifers alsoreacttochangesinsedimentsupply,dissolvedoxygen(DO),food (organicmatter),pH,andotherexternal(abiotic)andinternal(biotic) factors(Alve,1995;Alveetal.,2016;Schönfeldetal.,2012),andare oftenamongthelasteukaryoticorganismstodisappearcompletely frompollutedsites,makingthemexceptionalbioindicators(Alve,1991; Schafer,2000).
Thisresearchaddressestheimplementationofbenthicforaminifers inTLasbioindicatorsofPTEpollution.TheobjectivesweretodeterminethedistributionandbioavailabilityofPTEsandtheirpossible influenceonthespatialdistributionandmorphologicaldeformitiesof benthicforaminifers.
2.Studyarea
PuertoRicoistheeasternmostislandoftheCaribbeanGreater Antilles(Fig.1A).TheSanJuanBayEstuary(SJBE)system,thelargest estuary(Fig.1B)inPuertoRico,wasthe firsttropicalislandestuaryin theNationalEstuarineProgram,establishedin1993,andcomprises 240km2 ofland(drainagebasin),ofwhich25km2 areinundated (WebbandGómez-Gómez,1998).Meanannualrunoff isestimatedto be185×106 m3 withsuspended-sedimentyieldsexceeding 15,000mg/km2-yr(WebbandGómez-Gómez,1998).TheSJBEsystem consistsofsemi-enclosedbays,lagoons(e.g.,Torrecillas),andnatural anddredgedchannels(e.g.,CanalSuárez).Withintheeasternsubtidal portionoftheSJBEsystem(Fig.1B),TL(2.5km2)hasanaveragedepth of2.4mandisprobablythemostcomplexsystemwithintheSJBEdue toinflow-outflowsourcesofsalt,fresh,andbrackishwaters(Gómez-
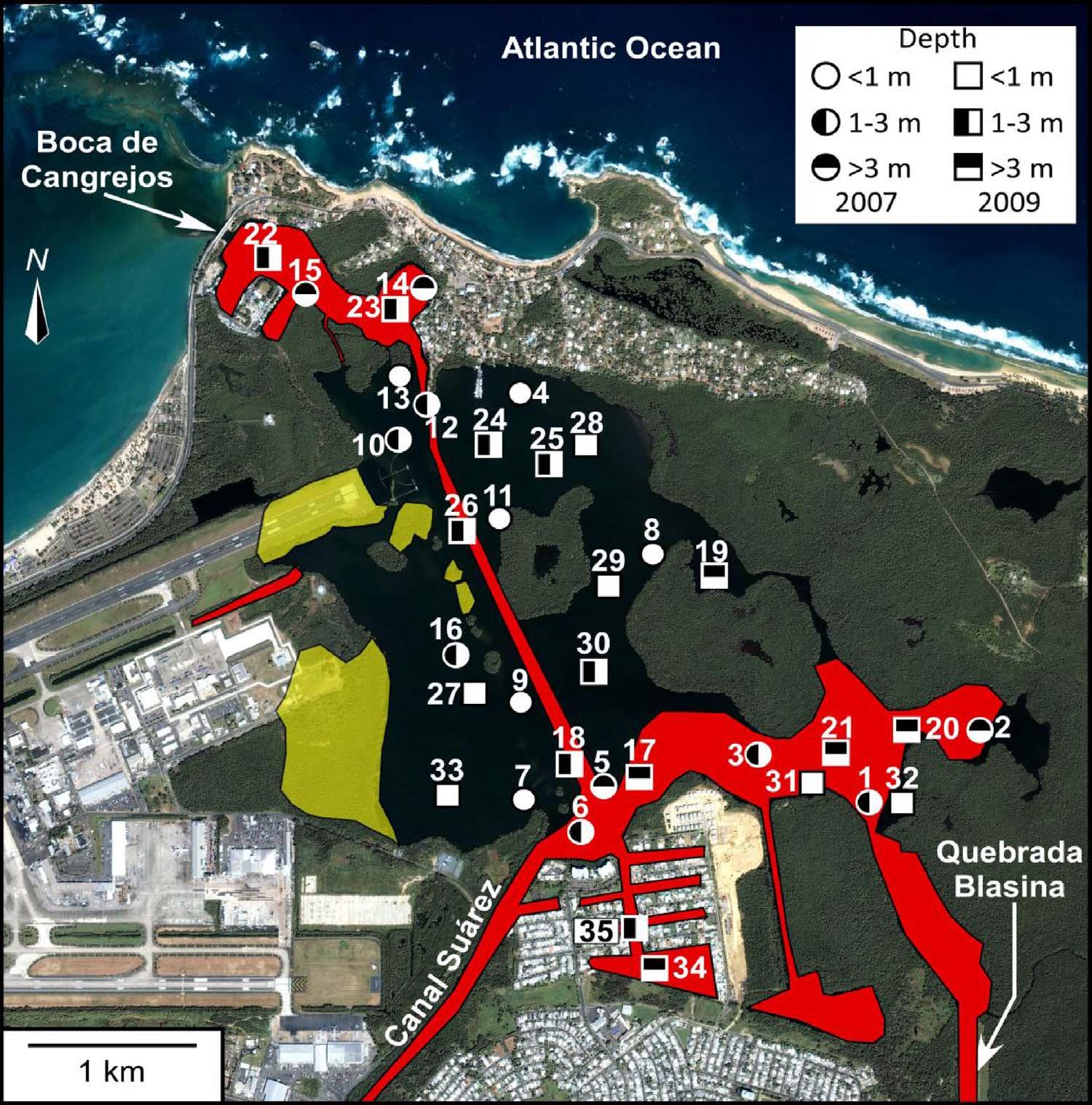
Fig.2. SamplingsitesinTorrecillasLagoon:circles=2007;squares=2009.Areasinfluencedby dredge(red)and fill(fadedyellow)areindicated (from Ellis,1976).(Forinterpretationofthereferencestocolourinthis figurelegend,thereaderis referredtothewebversionofthisarticle.)
Table1 SourcesofPollution.
PointSourceTypeLocationReference
SewagetreatmentplantRawsewage;PTEsQuebradaBlasina SanJuanBayEstuary(2000) CollectionsystemoverflowRawsewageSouthofTL
Combinedseweroutflows Rawsewage;stormrunoff SouthofTL
MarinasanitarydischargesRawsewageNorth/SouthofTL Dredgingand fillingSedimentAllaroundTL Ellis(1976)
NonpointSourceTypeLocationReference
IllegalsanitarydischargesRawsewageAllaroundTL SanJuanBayEstuary(2000)
On-siteseptictanksRawsewageWest-southwestmarginofTL Stormrunoff PTEs,PCBs,etc.AllaroundTL
Gómezetal.,1983).
BocadeCangrejoisadredgedoutletconnectingTLwiththeAtlantic Ocean(Fig.2).Thelagoonssemidiurnaltidalrangeisapproximately 0.60m(Bunchetal.,2000),withlongerresidencetimesandincreased volumeof110%attributedtodredginginthelagoon(Ellis,1976).Net outflowintotheAtlanticOceanisapproximately230,000m3/day (WebbandGómez-Gómez,1998).Below2mwaterdepth,anoxic conditionshavebeenfoundandareattributedtodisruptionofcirculationbydredging.Althoughdenserseawaterinjectedby floodtides canbringDOtotherelativelydeeperpartsoftheestuary,because neitherwindnortidalactionisstrongenoughtomixthelagoonwaters, theyaretypicallystratified,impedingdiffusionofoxygenandallowing slightlydeeperareastoserveasnutrienttraps(Ellis,1976).
3.Methods
3.1.Fieldsampling
Themethodsimplementedwerepriorto Schönfeldetal.(2012) FOBIMOprotocols.Thirty-fivesampleswerecollectedduringtwo samplingtripsin2007and2009(Fig.2).Aponargrabsamplerwas usedtocollectmostsamplesandtheupper2cmofsedimentwere collectedusingaTeflonspoon.Insomecases,sedimentsampleswere collectedviaSCUBAdiving.Eachsamplewasplacedintoanacid-washed(10%HClsolution)plasticNalgene© containerandfrozenfor subsequentchemicalandecologicalanalyses.AYSI-probewasusedto determineinsituwatercolumnmeasurementsatfourstationsfor temperature(°C),pH,salinity,andDOduring fieldsampling.
3.2.Laboratorysamplepreparation
3.2.1.Sedimentsamples
Sedimentsampleswerefreezedriedandsub-sampledforthefollowinganalyses:grainsize,totalorganiccarbon(TOC),percentcarbonate(CO3),PTEconcentrationinbulksediment,andPTEF2Tessbioavailableconcentrationinmudfraction.Forpurposesofthisstudy, whatwereportedasF2Tess-Cu,Zn,andFewastheF2Tess-CO3 second most “bioavailable” fractionasdefinedby Tessieretal.(1979) andas explainedinSection 5.3
Forgrain-sizeanalysis,sub-sampleswerewetsievedovera63 μm sieveandsubsequentlyovendried(50°C)todeterminethemud(silt andclay)fraction(<63 μm)byweightdifference.Thesandfractions (>63 μm)werethendrysieved.Eachsizefractionwasweighed,and theyweresummedandconvertedintopercentages;themediansize fractionforeachsamplewasexpressedinphi(Φ)units.Whenusedin conjunctionwithmud(%), Φ indicatessedimenttexturewithoutlisting allsizefractions.
TheTOCandCO3 analysesfollowedtheUICCarbonCoulometer manufacturerprotocolsfortitrationandanalysis.Thisprocedurerequirestheconversionofinorganicandtotalcarbonphasestocarbon dioxide(CO2).ForTOCandCO3,0.5gand0.015–0.065gofcrushed sub-samples(100mesh-size)wererespectivelycombustedat970°Cfor 6min(%TotalCarbon)andacidifiedusing5mLof2NPerchloricacid for5min(%TotalInorganicCarbon).
ForbulkPTEanalyses,eachdrysub-sample(0.5–1.2g)wascrushed intoa finepowder(100mesh-size)andsenttoACTLABSLaboratories Inc.(www.actlabs.com),forpreparationandgeochemicalanalysisof elementsofconcern(Cu,Zn,Pb,Ni,Cr,Li,V,Fe,As,Se,andMn)using InductivelyCoupledPlasmaMassSpectrometry(ICP-MS).TheanalyticalmethodimplementedwasCodeUltratrace4(ICP-MS).This methodmeasuresasuiteofelementsvia “total” digestionwithhydrofluoric,nitricandperchloricmixture,andhydrochloricacidsat260°C. Theacid-soluble(F2Tess:carbonatefraction)concentrationsof copper(Cu),zinc(Zn),andiron(Fe)weredeterminedtoestimate bioavailabilityviaAtomicAbsorptionSpectrometry(AAS)inthe GeomicrobiologyandSedimentologyLaboratoryatUniversidad NacionalAutónomadeMéxico.Sedimentsub-samplesweredrysieved (<63 μm)usingapolyethylenesieveandmesh.Sub-samplepreparationof0.25gofsedimentfollowedtheprotocolsof LuomaandBryan (1981),modifiedby Szeferetal.(1995) and Perez-Cidetal.(1998)
3.2.2.Foraminiferalsamples
Forforaminiferalassemblages,sedimentsub-samples(2.8–23.4g) werestainedinasolutionofRoseBengal(1gofdyein1Ldistilled water)for12h.Specimenswhoseinteriorexhibitedanintenseredstain werecountedasaliveatthetimeofcollection,althoughnecroticcytoplasmmaybestained.Someauthorshavenotedshortcomingsofthis technique(Bernhard,2000),butthisisastandardapproachwidely usedinbenthicstudies.Sub-samples(onepersamplingstation)were wetsievedthrougha63 μmmeshtoremoveanymudparticlesandthen dried(50°C).Wellpreserved(stained/unstained)foraminiferswere pickedfromthe>63 μmunderadissectingmicroscopeuntil,when possible,200–300individualswerecounted.Specimenswereidentified usingthegenerictaxonomyestablishedby LoeblichandTappan(1987) andtospecieslevelwhenpossible(duetosize<63 μm)following Poag (1981) and Seiglie(1971,1975a,b,c).Relativeabundancesof Ammonia (ARA), Quinqueloculina (QRA),and Triloculina (TRA)arecalculatedas thenumberofindividualsofthegenusdividedbythetotalnumberof foraminifersinthesample.Absoluteabundancesrefertonumberof individualsofaspeciesinasample.
3.3.Dataanalysis
Theselectionofdiversityindexesandstatisticaltoolsarebased uponapplicabilitytothe proposedstudy,andgeneraluseinforaminiferalandecological research(e.g., HayekandBuzas,1997).Theparametersusedinthis studyaredescribedasfollows:
(1)Speciesrichness:S=numberofspeciespersample.
(2)ForaminiferalDensity:FD=numberoftests/gram(totalmass)in eachsample.
(3)ShannonIndex:H(S)= Σ pi ×ln(pi)(Shannon,1948).
(4)EquitabilityIndex:E=eH(S)/n(Magurran,1988).
Hierarchicalclusteranalysiswasperformedonallforaminiferal species,afteradjustmentsfordistributionandtransformationstodetermineecologicalassemblages.The firstadjustmentwastheremoval ofanyspeciesthatwasnotpresentinatleast5%ofsamples.Thendata werestandardizedbycalculatingtheratioofeachtaxontodrymass(g) ineachsample(2.8–23.4g).Thesedatawerefourth-roottransformed (ParkerandArnold,1999)usingPRIMER© statisticalsoftware,thus creatingaresemblancematrixthatgeneratedagroup-averagecluster dendrogrambasedonBray-Curtissimilarity.
Pearsoncorrelationwasperformedtodetermineifanysignificant trendswerefoundbasedonlog-transformeddata(ParkerandArnold, 1999).Pearsoncorrelationanalysesincludedmud,TOC,andCO3,bulk PTEconcentration,F2Tess-bioavailable(acid-soluble)PTEconcentration,S,FD,H(S),andE.ThiswasdoneusingPRIMER© (v.6)statistical software(ClarkeandGorley,2006).
Contourmapsofmud,TOC,CO3,bulkandF2T-bioavailablePTEs, andforaminiferaldata,wereplottedusingArcGIS© (v.10)software (WorldGeodeticSystem84datum)toillustratepotentialhotspotsof pollution,distributionofPTEs,andkeyforaminiferalgenera.
4.Results
4.1.Sedimenttextureandcomposition
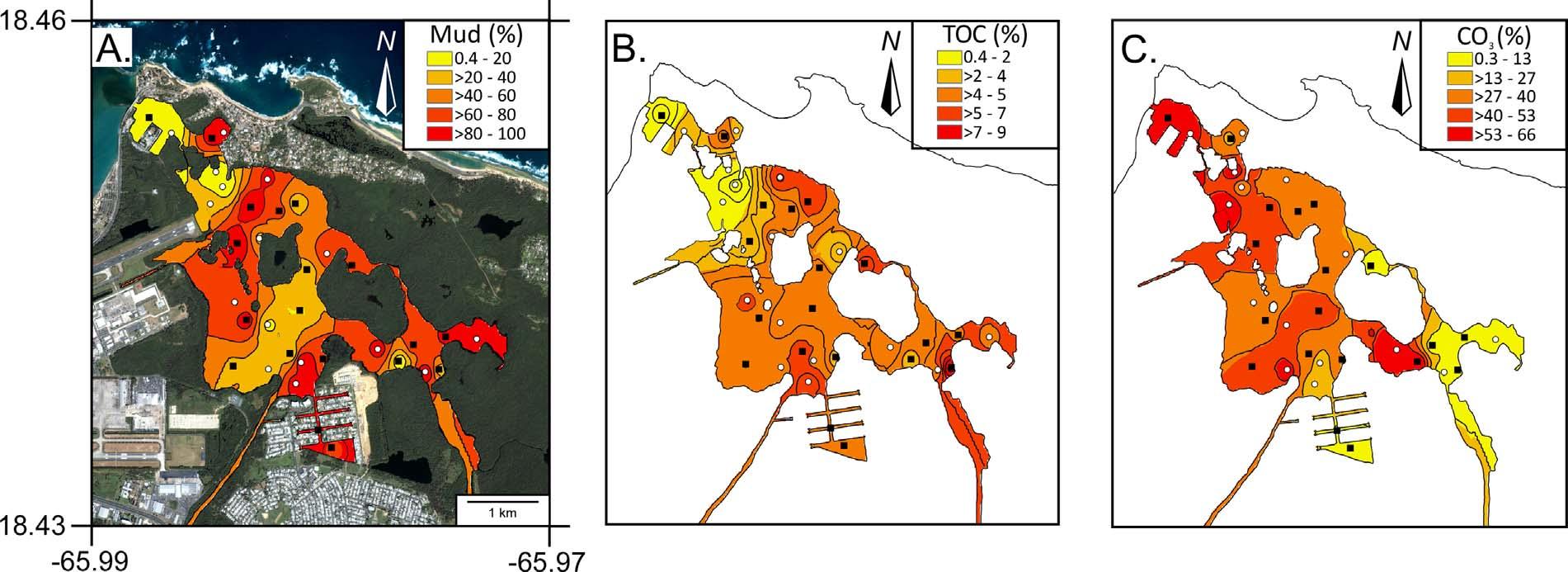
Thedominantmediangrainsizeforallsampleswasmud(Φ >4; 51%ofsamples)followedbymediumsand(Φ =2;17%ofsamples) andgravel(Φ ≤−1;17%ofsamples)(AppendixA,Supplemental material).Percentmud(mud)decreased200-fold(99.4–0.45%)from QuebradaBlasinaontheSEtowardstheNWendofTL(connectionto AtlanticOcean)(Fig.3a).Totalorganiccarbonvaluesrangedfrom 0.42–6%.Similartomud,TOCdecreased20–foldfromSEtoNW (Fig.3b).Incontrast,CO3 (0.32–66%)(AppendixA,Supplemental material)showedtheoppositetrend,increasingalmost200-foldfrom
SEtoNW(Fig.3c).
4.2.Potentiallytoxicelementdistribution
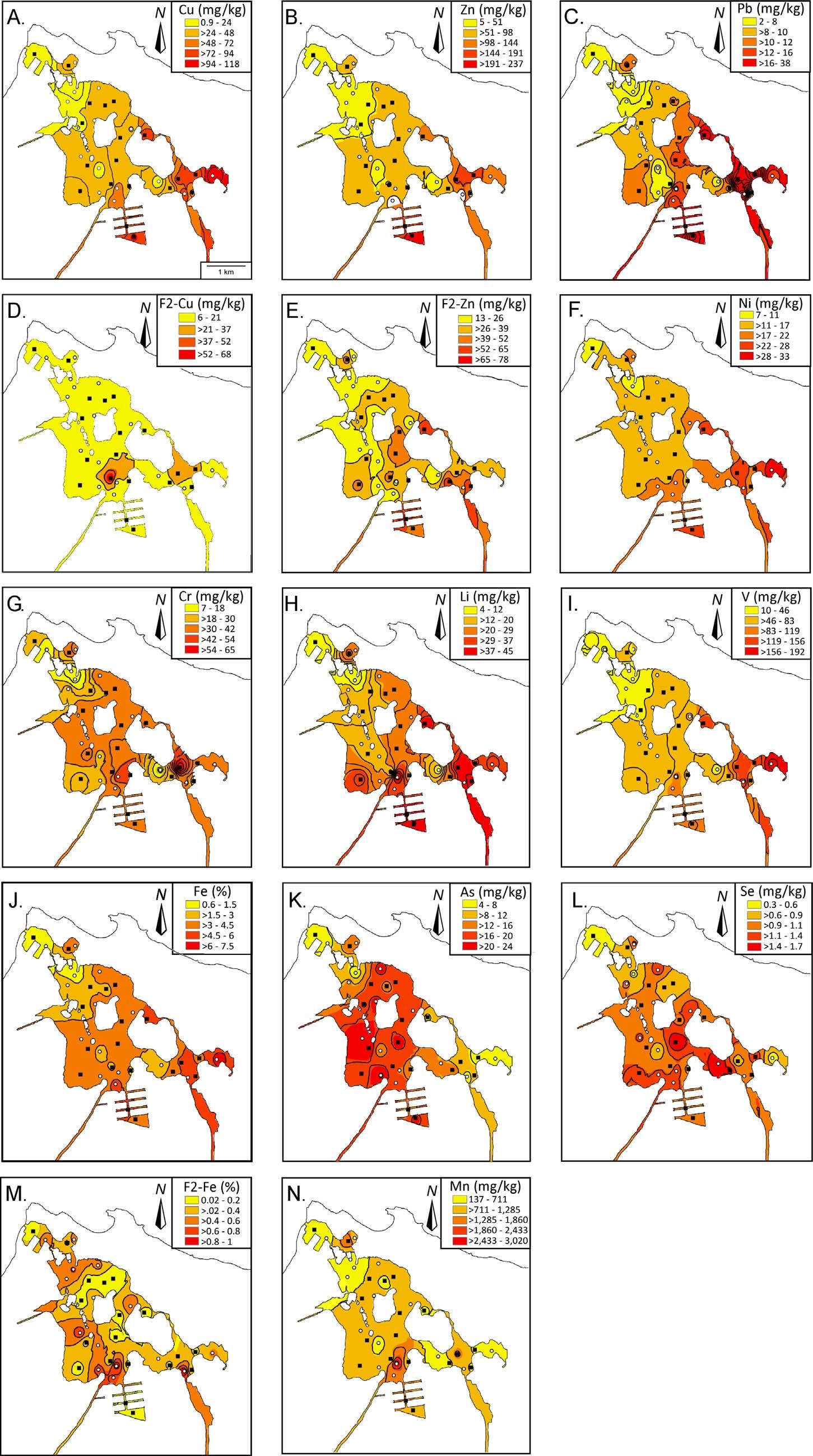
ForbulkPTEconcentrations,11PTEswereconsidered.Spatial distributionsareshownin Fig.4A–4N.Copper(0.9–118mg/kg),Zn (4.8–237mg/kg),Pb(2–38mg/kg),Ni(6.9–33mg/kg),Cr (6.64–65.39mg/kg),Li(4.2–44.9mg/kg),V(10–192mg/kg)andFe (11–134mg/kg)generallyincreasedfromNWtoSE.Concentrationsof As(4.46–24.47mg/kg),Se(0.3–1.7mg/kg)andMn(137–3020mg/kg) werehighertowardsthemiddleofTL.MostPTEsvariedoveroneorder ofmagnitudeexceptforCuandZn.
Forbioavailability,onlythreePTEs(Cu,Zn,andFe)wereconsidered.CopperandzincarewellknownPTEsthathavebeencorrelatedtoforaminiferalassemblagechangesin fieldandculturestudies. Iron,wasselectedduetoitssignificanceasaredoxindicator.F2Tessbioavailablecopper(F2Tess-Cu)(5.87–67.73mg/kg)hadanalmost uniformdistributionexceptfortwo “hotspots” closetotheCanal Suarez(east)andQuebradaBlasina(west);whileF2Tess-bioavailable zinc(F2Tess-Zn)(13.02–72.63mg/kg)showedanincreasingNW–SE trend(Fig.4D–E).TheF2Tess-bioavailableiron(F2Tess-Fe)showedno discerniblepattern(Fig.4M).Afactorof11-foldinF2Tess-Cuwasobservedandfactorof50-foldwasobservedforF2Tess-Fe.TheF2Tess-Zn varied6–foldinconcentration.ThesitesatCanalSuarez,LagoManagua,andQuebradaBlasina,showedoverallrelativelyhighconcentrationsofbothbulkandthebioavailablePTEcounterparts(except F2Tess-Cu).
APearsoncorrelationmatrix(AppendixB,Supplementalmaterial) wascalculatedforthe11elementsofconcern,aswellasforbioavailablePTEs(F2Tess-Cu,F2Tess-Zn,F2Tess-Fe),andTOC,CO3 andmud.All PTEspositivelycorrelatedwithmud,thoughforF2Tess-Cu,F2Tess-Zn, F2Tess-Fe,As,andSe,thecorrelationwasnotsignificantatthe95% confidencelevel.Similarly,allexceptF2Tess-Cu,F2Tess-Zn,andF2Tess-Fe significantlycorrelatedwithTOC.Thus,mostPTEpairswerepositively correlated(>0.34).However,AsonlycorrelatedwithSeandMn.The F2Tess-CuandF2Tess-Znpositivelycorrelatedwitheachother,aswellas withCu,Zn,Pb,Cr,andLi,andnegativelywithF2Tess-Fe.Noneofthe bioavailablePTEssignificantlycorrelatedwithTOCormud.Percent carbonatenegativelycorrelatedwithmostPTEsexceptforSewhile correlationswithF2Tess-CuandF2Tess-Fewerenotsignificant.Arsenic wastheonlyPTEthatcorrelatedpositivelywithCO3.
4.3.Watercolumndissolvedoxygen,pH,temperatureandsalinity
Dissolvedoxygenconcentrationsinthewatercolumngenerally decreasewithdepth.InallprofilesexceptforSamplingStation#17, hypoxicconditionsprevailedbetween1.5–3.5m,andanoxicconditions
werefoundat>3.5mdepth(Fig.5A).
ApH<8,whichisbelownormaltropicalseawater,wasseenat 2–3mdepthinthreeoftheprofiles(Fig.5B).Watersbelow5mwere highlycorrosivetoCaCO3 (pH<7.4).Incontrast,Site#17showed
Fig.4. PTEsurfacedistribution.A:copper(Cu);B:zinc (Zn);C:lead(Pb);D:F2Tess-Cu(acid-solublecopper);E: F2Tess-Zn(acid-solublezinc);F:nickel(Ni);G:chromium (Cr);H:lithium(Li),I:vanadium(V).Allconcentrations areinmg/kg(=ppm).Whitecircles:2007sampling;and Blackrectangles:2009sampling.J:iron(Fe);K:arsenic (As);L:selenium(Se);M:F2Tess-Fe(acid-solubleiron);N: manganese(Mn).Allconcentrationsareinmg/kg,except Fe(%).Whitecircles:2007sampling;andBlackrectangles:2009sampling.
minimalvariationinpHbetweensurfaceandbottomwaters(8.1). Awell-definedthermoclinewasobservedbetween0and6mdepth. Temperatureremainedconstantat26.3°Catdepths>6m(Fig.5C).A shallowersite(#34)hadathermoclinebetween0and4m,with
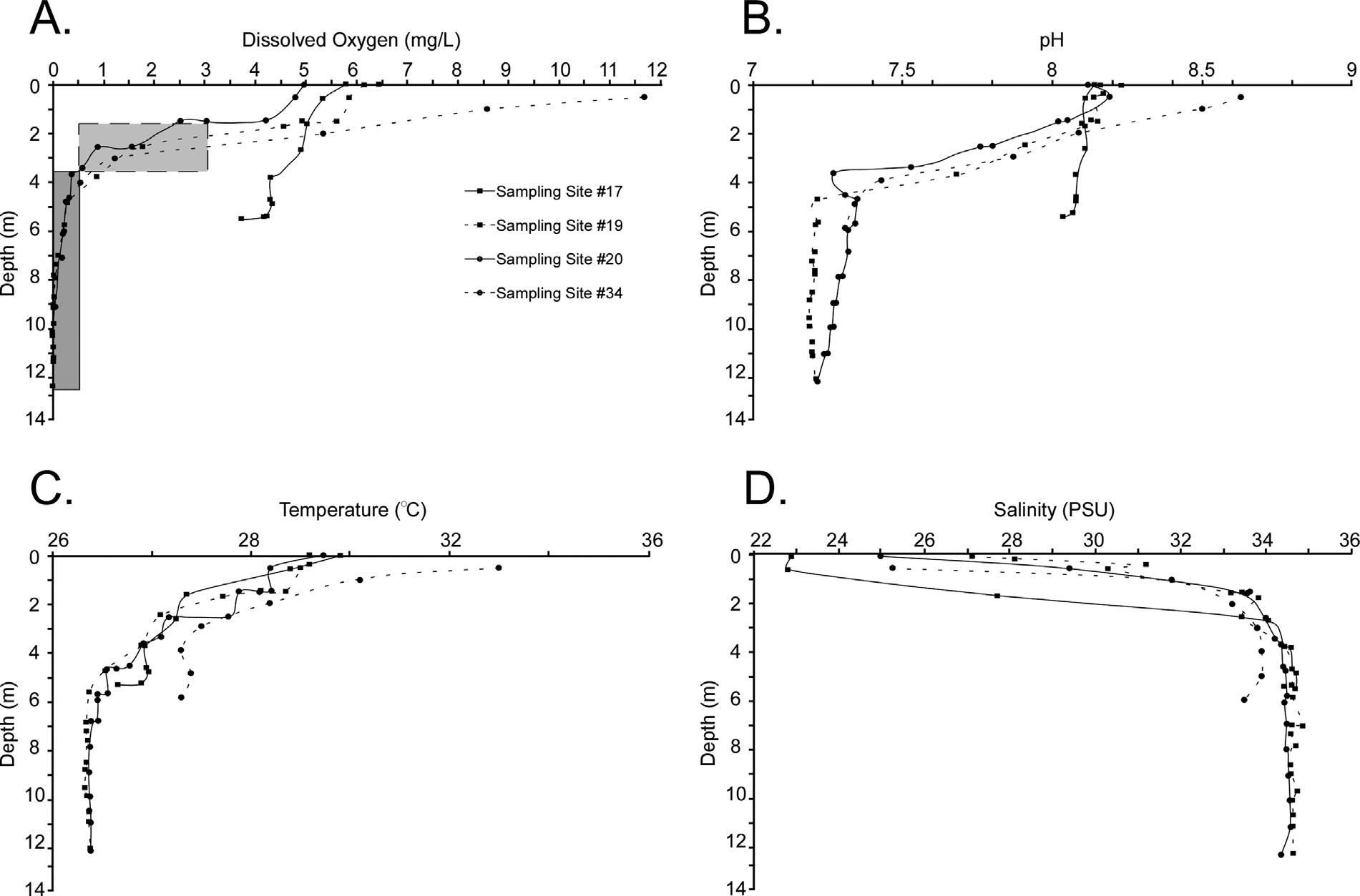
Fig.5. Watercolumnprofiles.A:Dissolvedoxygen.Lightshadedareadepictshypoxicconditions(<3mg/L)anddarkshadedareadepictsanoxia(<0.5mg/L).B:pH.C:Temperature.D: Salinity.
temperaturedroppingfrom30°to27°C.Ahaloclinewasobserved between1and2mwaterdepthwithasalinityrangebetween25and 35(Fig.5D).Site#19wasslightlylesssalineatthesurface,witha valueof23.
4.4.Foraminiferalassemblages
From35sedimentsurfacesamplescollectedinTL,6221benthic foraminiferswerepicked,with34generarepresentedamongwhich50 specieswereidentified(AppendixC,Supplementalmaterial)(Fig.6). Stainedforaminiferswereseldomencountered;therefore,allcounts
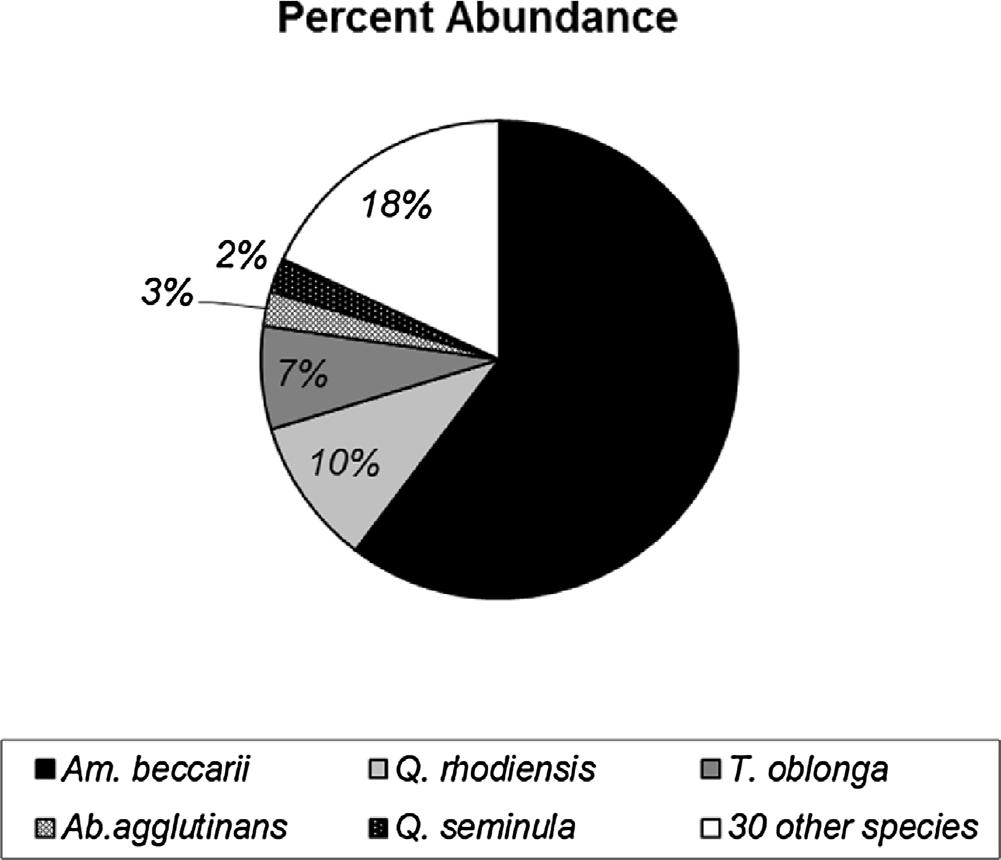
representtotalabundances.Ofthe35speciespresentinatleast5%of thesamples, Ammoniabeccarii (3752individuals),Quinqueloculina rhodiensis (621individuals)and Triloculinaoblonga (434individuals) werethemostabundantacrossallsamples. Ammobaculitesagglutinans (145individuals)and Quinqueloculinaseminula (144individuals)were thefourthand fifthmostabundantspecies.Noneoftheotherspecies accountedformorethan100individualsacrossallsamples.
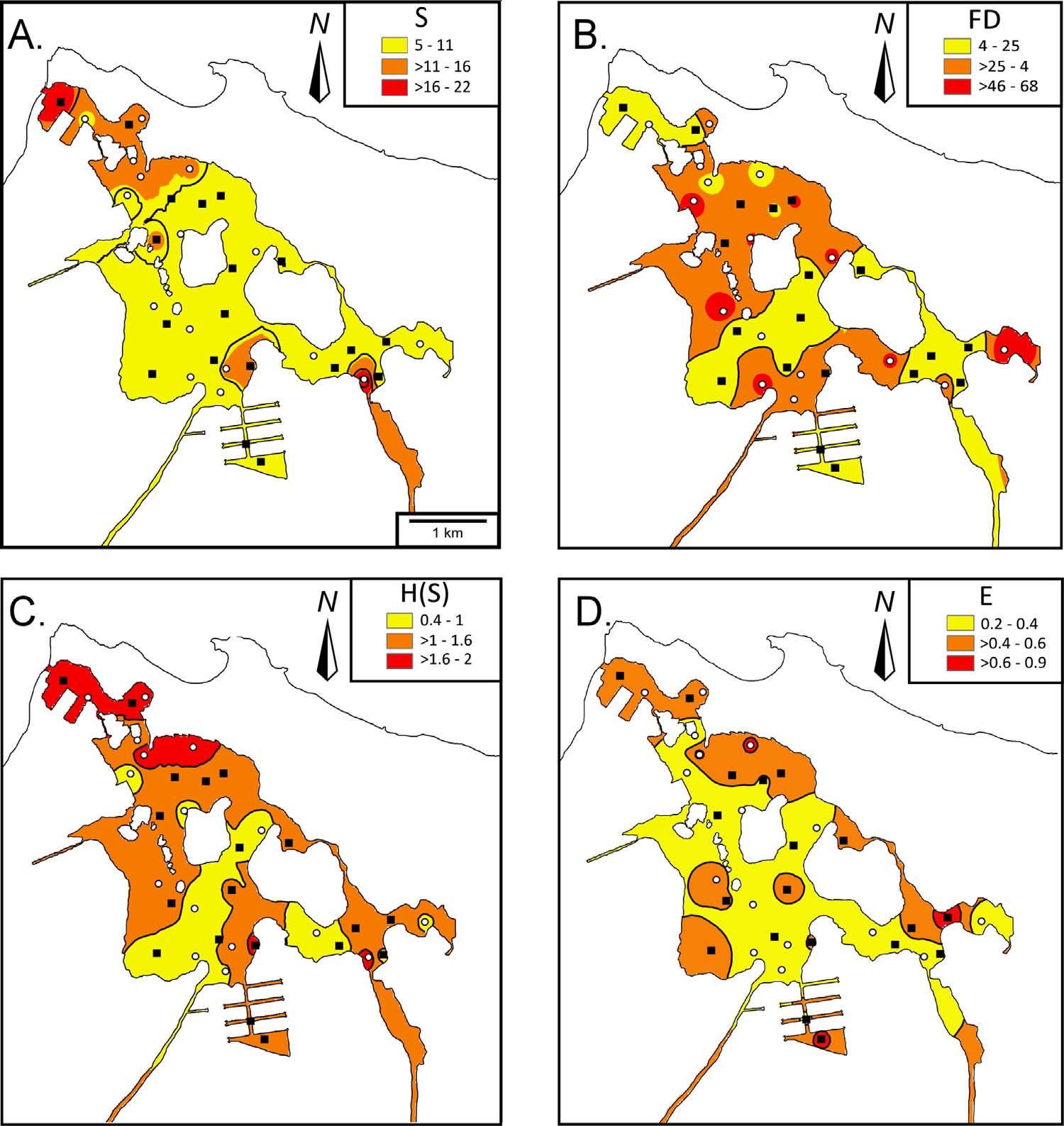
Speciesrichness(S)rangedfrom5to22species(Fig.7A).The majorityofthesamples(71%)hadrelativelowspeciesrichness(5–11). ThetwosampleswithhighSvalueswherefoundonoppositesidesof thelagoon.
Foraminiferaldensities(Fig.7B)werealsoverylowacrossTL. Variabilityrangedfrom4to68individuals/gand54%ofthesamples hadrelativelowdensities(4–25).SimilartoS,noapparenttrendwas observedexceptforareasoflowdensityontheNW,centralandSE sectionsofTL.
TheShannonIndex[H(S)](Fig.7C)wasusedtoevaluatetheheterogeneity(i.e.,diversity)ofsampleswithrespecttoforaminiferaltaxa. Indexvaluesrangedfrom0.42–2.22.Only40%ofthesampleshad intermediate(1.03–1.62)H(S)values.MostsampleswithhigherdiversitywerefoundtowardstheNWportionofTL.
TheEquitabilityIndex(E)measurestheevennessoftheforaminiferaldistributionwithintheassemblage.TheEvaluesranged from0.20–0.87(Fig.7D).Morethanhalfofthesamples(51%)hadlow equitability(0.20–0.42)values(i.e.,highdominance),while11%ofthe sampleshadhigher(0.65–0.87)equitabilityvalues(i.e.,lowdominance).
Deformitiesofforaminiferaltestswerefoundin54%ofthesamples. Alldeformitieswereobservedinmiliolidsexceptforonesamplein whichdeformed A.beccarii testswerefound.Percentdeformities(FD) rangedfrom2to18%.
4.4.1.Foraminiferalclusteranalysisandrelativeabundances
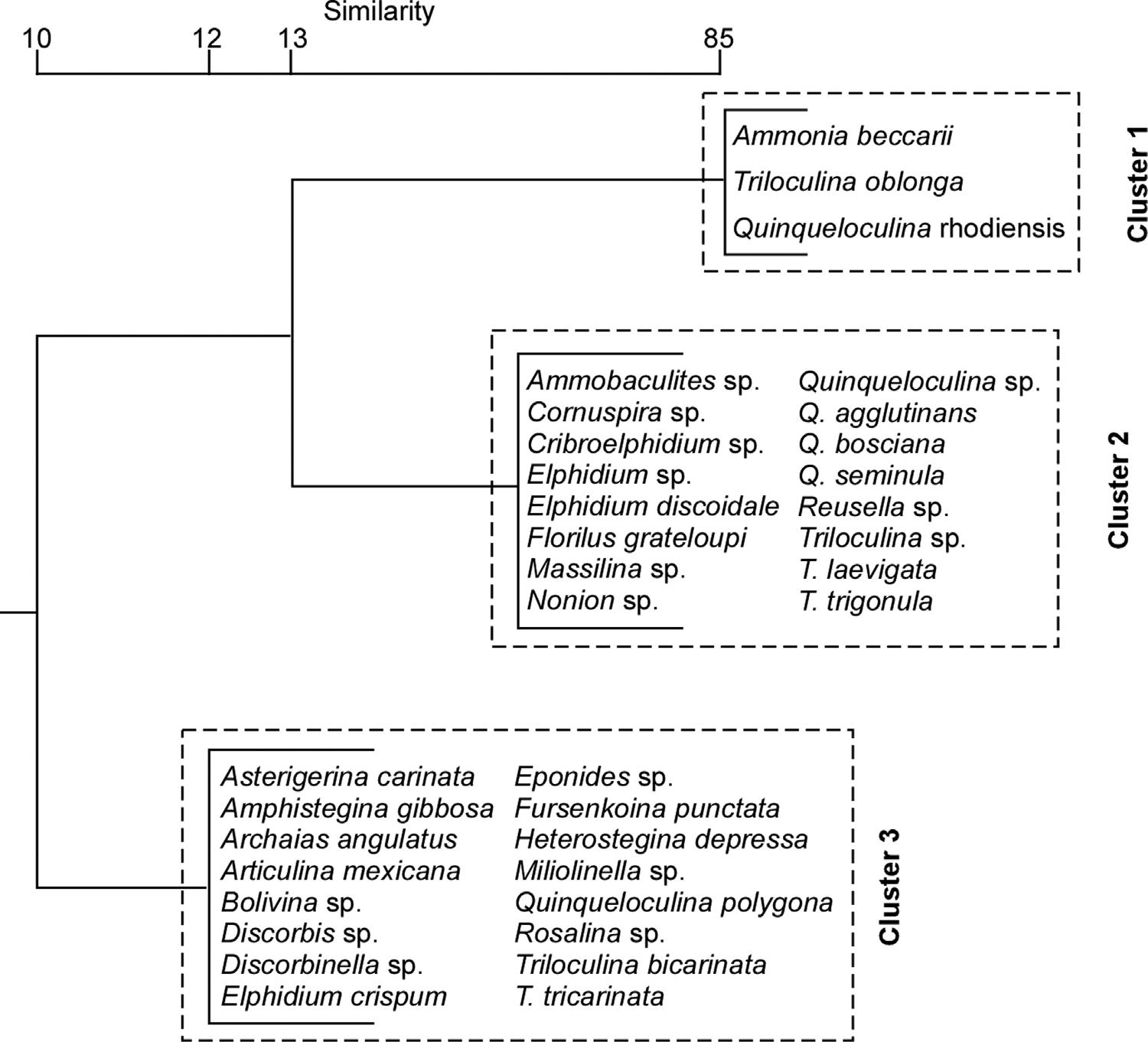
Threeclusterswereidentified(Fig.8).Cluster1wascomposedof A. beccarii (overwhelminglydominantforaminiferinTL), Q.rhodiensis, and T.oblonga.Cluster2includedothercommonnearshoreandestuarinetaxa.Cluster3includedtaxamorecharacteristicofopen-marine shelfenvironments.
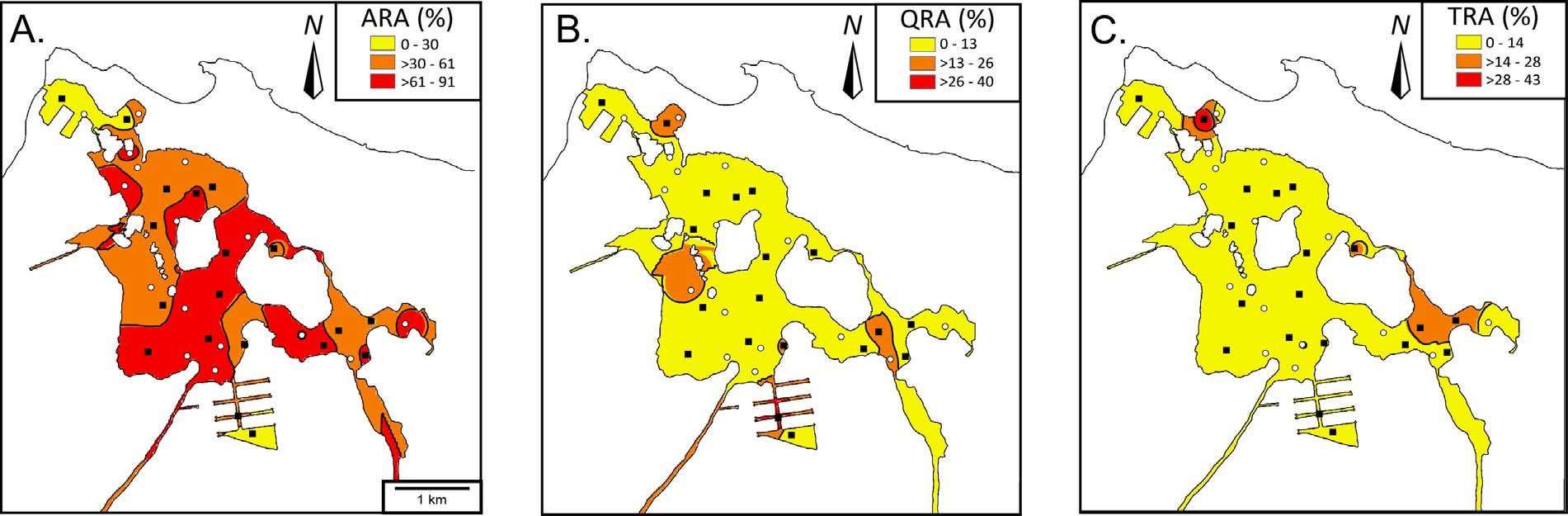
Therelativeabundances(RA)of A.becarrii, Quinqueloculina sp.and Triloculina sp.wereplotted(Fig.9). Ammonia RA(ARA)rangedfrom 0.7–91%.FivesampleswereclassifiedashavinglowARA(<30%), while13and17sampleshadmedium(30–61%)orhighabundances (>61%)respectively.SampleswithhighARAvalues(53%)were mostlyfoundtowardsthecentral–SWportionoftheestuary.Avery similardistributiontothepatternoflowspeciesrichnesswasseenin Fig.7A. Quinqueloculina RA(QRA)valuesrangedfrom0to40% (Fig.9B).Nearlyallsamples(80%)hadalowQRA(<10%).Similarly, Triloculina RA(TRA)valuesrangedbetween0and43%(Fig.9C),with 85ofthesampleshavinglowTRAvalues.
APearsoncorrelationmatrix(AppendixD,Supplementalmaterial) wascalculatedforthe11PTEsofconcern,includingbioavailableelements(F2Tess-Cu,F2Tess-ZnF2Tess-Fe),aswellasTOC,CO3,mud,foraminiferaltaxaabsoluteabundances,ecologicalindices,relative abundancesandpercentdeformities.Numerousgeneranegatively correlatedwithbulkPTEs,bioavailablePTEs,TOCandmud.Fromthe dominantassemblage, A.beccarii absoluteabundancesnegativelycorrelatedwithF2Tess-Cu,F2Tess-ZnandpositivelywithF2Tess-Fewhile Quinqueloculinarhodiensis positivelycorrelatedwithCu,Cr,Fe,Mn, TOCandmud. Triloculinaoblonga absoluteabundancescorrelated
Fig.7. Distributionofforaminiferalcharacteristics. A:Speciesrichness(S);B:Foraminiferaldensity(FD); C:ShannonIndex[H(S)];andD:Equitability(E).
positivelywithCu,Cr,Fe,V,andmud.Percentdeformitiescorrelated positivelywithF2Tess-CuandCrandnegativelywithF2Tess-Fe. ForaminiferaldensitycorrelatednegativelywithF2Tess-CuandF2TessZnandpositivelywithF2Tess-Fe.Speciesrichnesscorrelatednegatively withCu,Zn,Ni,Li,Se,Fe,VandTOC.TheH(S)showednocorrelation exceptnegativelywithSe;CO3 correlatednegativelywithE.Relative abundancesof A.beccarii showednocorrelations,whileQRAandTRA bothpositivelycorrelatedwithCu,Zn,Cr,Li,Fe,Mn,V,mud,andno correlationwithbioavailablePTEs(F2Tess-Cu,F2Tess-Zn,F2Tess-Fe). LeadcorrelatedwithTRAandTOCwithQRA.
5.Discussion
5.1.Potentiallytoxicelementsinsediments
ThedistributionsofPTEsinTLareintimatelyrelatedtosediment texture,TOCandwaterdepth.Thelagoonisafairlyrestricted,low energyenvironmentwithsurfacesedimentsmostlydominatedbymud (Fig.3A).CoarseningofsedimentstowardstheNWisrelatedtohigher energyenvironments;thisalsoexplainslowerTOCvalues(Fig.3B). HigherTOCvaluestowardstheSEarelikelyassociatedwithlocalinput, lowwaveenergy,waterstratificationandwaterdepth.Waterdepths varyfrom<1mto16m(Fig.2,AppendixA,Supplementalmaterial), withsomeareaswellmixed(e.g.,Station#17)andothersstrongly stratifiedwithrespecttooxygen,pH,temperatureandsalinity(Fig.5). Dredgingoperationsuntilthemid-1970s(Fig.2)createdunconnected deeper “holes” (>3.5m),where “oxiclines” develop(Fig.5A)and
allowforapreservationoforganicmatter.Inaddition,strongpH stratification,withvalues<7.4atdepths>5m(Fig.5B),isattributed toapparentoxygenutilization(lowDO)intheSEsectionduetohigh mudandTOC(Fig.3A–B).
Outflowsfromsewagetreatmentfacilities,residentialseptictanks, and “clandestine” solidwastedumps(e.g.,LagunaPiñones)aresources ofdissolvednutrientsandorganicmatterthatcontributetotheTOC, whichcancomplexPTEs.Inaddition,PTEsarealsoadsorbedbymudsizesediments(e.g.,terrigenousinput)fromQuebradaBlasinaand CanalSuarez.Underoxicconditions,PTEs(e.g.,Cu,Pb,Zn)aresequesteredbyFe/Mnoxidesand,conversely,underanoxicenvironments,bysulfides(Daviesetal.,2005;Lariosetal.,2012;Zaaboub etal.,2015).Copper,Zn,Ni,Pb,andsomeotherPTEspositivelycorrelatedwithFe/Mn(AppendixB,Supplementalmaterial),indicativeof oxic/anoxicboundaryconditions.Moreover,framboidalpyritewas
Fig.8. Summaryclusterdiagram(BrayCurtis)for foraminiferalassemblages.Similaritynottoscale.
commonlyseeninthetestsofforaminifers(Martínez-Colónand Hallock,2010;Seiglie,1973)andcanbeconsideredasastressresponse toPTEpollution(Buzas-StephensandBuzas,2005)orpost-mortem exposuretoanoxia.Ingeneral,thehighestPTEconcentrationswere foundontheSEsection(Cu,Zn,F2Tess-Zn,Pb,Ni,Cr,Li,V,Feand F2Tess-Fe),nearesttoanthropogenicsources,orrevealednodiscernable pattern(F2Tess-Cu,As,SeandMn)(Fig.4).
5.2.Foraminiferalspatialdistributions
ForaminiferalassemblagesinTLaretaxonomicallycharacteristicof estuarineenvironments.Clusteranalysis(Fig.8)clearlyseparatedthe threedominantspecies(Cluster1),fromcommonnearshoreandestuarinetaxathatweremuchlessprevalent.Cluster3includedtaxasuch as Amphisteginagibbosa and Heterosteginadepressa thatarecharacteristic
sp.relativeabundance (TRA).
ofopen-marineshelfenvironmentsandwerefoundprimarilyinsamplesdirectlyinfluencedbyconnectiontoAtlanticwaters.
Ammoniabeccarii iswellknowntobestress-tolerantunderanthropogenicaswellasnaturalstressors.Ingeneral, fluctuationsinsalinity, foodsupply,temperature,oxygen,andpollutants,includingPTEs(de Nooijeretal.,2007;Jorissen,1999;amongothers),aresomeofthe stressorsthataffectthedistributionofthistaxon.Inothercoastalareas inPuertoRico,suchasJobosBayandGuanyanillaLagoon, A.beccarii dominatetheassemblages,alongwith Q.rhodiensis insubstratesimpactedbyorganicpollution(Seiglie,1968,1975a,1975b,1975c). Cribroelphidiumexcavatum andsome Elphidium spp.arealsoopportunistic,stress-toleranttaxathatcansurvivechangesinoxygenconcentrations(e.g., DabbousandScott,2012;Debenayetal.,2009).Althoughfoundinthecurrentstudy,thesetaxawerenotcommon. However,afewotherspeciesrecordedinTL,notably Q.seminula,are wellknownstress-toleranttaxa(e.g., Fosteretal.,2012),especiallyin warmwaterenvironments.
Numerousstudiesinvolvingtheuseofforaminifersasbioindicators ofpollutionhavereportedtheimpactofPTEsandotherstressorson ecologicalindices(e.g., Alveetal.,2016;Bouchetetal.,2012;Schwing etal.,2016;Oliveretal.,2014).Theseindices,ascalculatedfrom foraminiferalassemblagesinTL,arecharacteristicofanimpactedestuarineenvironment.Speciesrichness,FD,H(S)andEareverylow comparedtosimilarestuarineenvironmentssuchasBiscayneBay, Florida(e.g., Carnahanetal.,2009);PointJoinville,France(Debenay etal.,2001);andCagliari,Sardinia(Schintuetal.,2016).Moreover, veryfewforaminiferswererecordedasliveinthesamplescollected fromTL.
ThehighestSvalueswerefoundtowardstheNWandSEsectionsof thelagoon(Fig.7A).Thedatashownocorrelationwitheithermudor CO3.However,thesignificantnegativecorrelationwithTOCwaslikely relatedtoredoxchanges(suboxictooxicconditions)(Fig.5A). Ellisand Gómez-Gómez(1976) previouslyrecordedanaerobicconditionsbelow 2mwaterdepthandDOlevelsbetween1and3.4mg/L,consistent withthecurrentconditionsofthelagoon(Fig.5A).Anoxiaassociated withabundantorganicmatterandlackofmixingishighlydetrimental tomostforaminiferalspecies(Jorissen,1999;Schönfeldetal.,2012). SpeciesrichnessalsonegativelycorrelatedwithseveralPTEbulkconcentrationsinTL,whichisinagreementwithsimilarstudieselsewhere (Alve,1991;ArmynotduChâteletetal.,2004;Martinsetal.,2016; Salvietal.,2015).
TheFDhaslowervaluestowardstheNWandSEofthelagoon (Fig.7B).Interestingly,FDshowsnocorrelationwithanyparameter exceptnegativelywithF2Tess-CuandF2Tess-Znandpositivelywith F2Tess-Fe. Romanoetal.(2008) and FrontaliniandCoccioni(2008) foundsimilarlowdensitiesinanthropogenicallyimpactedenvironments.Insomecoastalenvironments,lowFDhasbeenreportedto correlatetolowTOC(<2%)(ArmynotduChâteletetal.,2009).AlthoughnocorrelationwasfoundwithTOC,pHandanoxiaaremore likelycontrollingmechanisms. DebenayandFernandez(2009) and Martinsetal.(2011,2013) alsofoundthatbioavailablePTEsnegatively correlatedwithforaminiferaldensity.Inaddition,dispersalbytidal currents(e.g., Arslanetal.,2016b)intothehighlycorrosiveconditions indeeperzonesofTLarelikelytodissolvetheforaminiferaltests, whichwouldcontributetolowFDvalues,andtochangesindistributionanddominance.Dormancyisanotherfactortoconsidersincereportshaveshownthatpropagulesremaininstasisuntilenvironmental conditionsaremorefavorable(AlveandGoldstein,2003;Rossand Hallock,2016)whichcouldalsobethecaseinthedeeperportionsof TL.
SimilartoSandFD,diversityindicesareknowntobelowinareasof PTEpollution(e.g., Berginetal.,2006;Fosteretal.,2012;Kfourietal., 2005;Martínez-ColónandHallock,2010;Schintuetal.,2016).BothH (S)andEshowasimilarspatialpattern,withlowervaluestowardsthe centerofthelagoon(Fig.7C–D).Thelowdiversitiesandabundancesof foraminiferaltestsareindicativeofstressedenvironments,butPTEsdo
notappeartobetheprimaryenvironmentalfactorstressingtheassemblagesinthelagoongivenfewsignificantcorrelationswithPTEs. Thesameconclusionwasreached,baseduponashort-sedimentcore fromTLinwhichthetemporalvariabilityofH(S),EandFDreflected thecarbonatepreservationpotential(pH)andorganicmattercontentof thelagoon(Martínez-ColónandHallock,2010).
Therelativeabundancesof Q.rhodiensis andTRAweregenerally quitelow,whileARAwasgenerallymoredominanttowardsthecenter ofthelagoon(Fig.9),whichisdissectedbydredgedchannels(Fig.2). ThefactthatARAshowsnocorrelationsfurtherdemonstratesthatthis speciesisnearlyubiquitousinthelagoon.Moreover,thehigh-Mgcalcitemiliolidtestsaremorepronetodissolutionindeeper,corrosive areasofthelagoon.DissolutioncouldalsohelpexplainthelowforaminiferaldensitiesandindicesoverallinTL;thedominantspecies showingdissolutionscarssupportthishypothesis(seePlateIin Martínez-Colónetal.,2009). Diasetal.(2010) notedlowernumberof foraminifersinassemblagesdominatedbyagglutinatedformsat pH<7.6.InTL,profilesshowpHaslowas7.2atdepths>3.5m (Fig.5B),althoughagglutinatedspecimenswererarelyfound.TheQRA andTRApositivelycorrelatedwithseveralbulkPTEsandnotwiththe bioavailablecounterparts(F2Tess-Cu,F2Tess-Zn,F2Tess-Fe).Thisobservationisnotconsistentwithstudiesthathaveconcludedthatbulk PTEconcentrationsareforaminiferalstressors(e.g., Fontanieretal., 2012;Schintuetal.,2016).Thesignificanceofthis findingisthat conclusionsshouldnotbebasedonbulkPTEconcentrationsbutinstead mustconsiderchemicalfractionationsaswellasenrichment(seeSection 5.3 fordiscussion).Inaddition,thepositivecorrelationbetween absoluteabundancesof Q.rhodiensis andQRAwithTOCandmud,and T.oblonga andTRAwithmud,suggeststhatsedimenttextureandorganicmatter(asreflectedbyanoxicconditions)couldbethemain controllingfactorsandPTEsaresecondary.
Numerousstudieshavedebatedtheimportanceoftestdeformities asaproxyofpollution(seeTable4in Martínez-Colónetal.,2009). Deformedforaminiferswerefoundin54%ofthesamplesfromTL,with 18%deformedforaminifersrecordedinonesample.Theforaminifers exhibitingdeformedtestsweremostlymiliolids,especially Q.rhodiensis, aspreviouslyobservedby Martínez-ColónandHallock(2010) AlthoughdeformitiescorrelatedpositivelywithF2Tess-CuandCr,and negativelywithF2Tess-Fe, Seiglie(1975c) notedthatabnormal morphologiesof Q.rhodiensis couldbeinfluencedbyeutrophication.
5.3.Bulkvsbioavailablepotentiallytoxicelements
Inenvironmentalmicropaleontologicalstudies,thestandardmethodologyhasbeentoassesstotalPTEconcentrationsinbulksediments (e.g., ArmynotduChâteletetal.,2004;Arslanetal.,2016a;Eichler etal.,2012;Salvietal.,2015),whilefarfewerstudieshaveconsidered thebioavailabilityofPTEs(Martinsetal.,2011,2013,2015;Schintu etal.,2016). Tessieretal.(1979) notedthatPTEscouldbefoundin varioussedimentarycomponents(“fractions”).Fractionationcanbe influencedbysedimenttype,organicmattercontent,pH,alkalinity,Eh, salinity,andotherenvironmentalfactors.ThemobilityofPTEsduring sequentialextractionprocedureshasbeenrelatedtothedegreeofoperationalbioavailability(BaconandDavidson,2008;Zimmermanand Weindorf,2010).Theseinterpretationsinfractionationresultedin five differentsedimentchemicalfractionsfollowing Tessieretal.(1979) nomenclature:exchangeable(PTEsadsorbedtoclays:F1Tess aremost bioavailableandarereadilyreleasedbycomplexationandminorpH changes),acid-soluble(PTEsboundtocarbonates:F2Tess-CO3 canbe releasedduringdissolution),reducible(PTEsboundtooxidizedminerals:F3Tess-Fe/Mncanbereleasedinreducingconditions),oxidizable (PTEsboundtoorganicmatter:F4Tess-organicmatter),andresidual (PTEsfoundinthecrystallinestructureofsilicateminerals:F5Tesslithicsaretheleastbioavailableandwillnotbereleasedtotheenvironment).
Thephysico-chemicalconditionsofTLserveasagoodexampleof
Fig.10. Simplifiedmodelofthefateandtrasportof PTEs.A:lowoxygenandpHconditions.B:high oxygenandpHconditions.Positive/negativesigns: pH/oxygenincreaseordecrease.Modifiedfrom Martínez-Colónetal.(2009) M.Martínez-Colónetal.
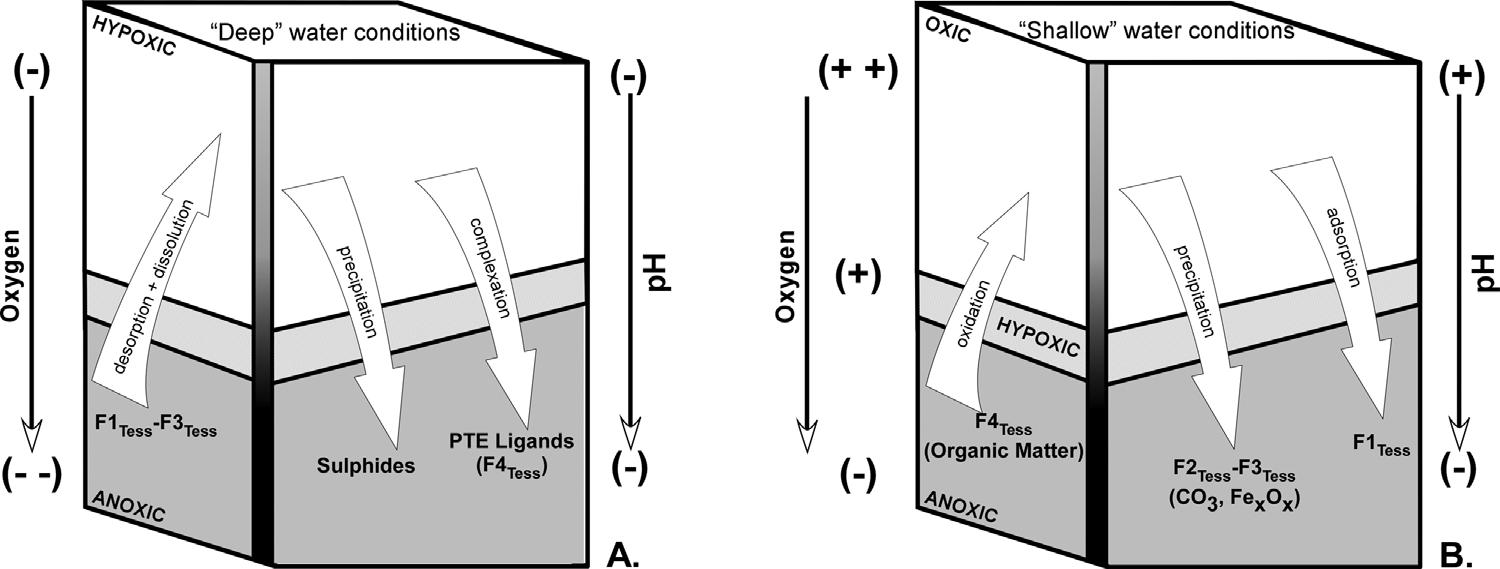
thebehavioranddistributionofPTEsamongthedifferentsediment chemicalfractions. Fig.10 illustratesthefateandtransportofPTEs betweensedimentchemicalfractions(e.g.,F1Tess–F4Tess)inthelagoon basedonpHandDOcharacteristicswithdepth.Inthedeeperareasof thelagoon(>3.5m)lowpHandanoxicconditionsprevail(Fig.5A–B). AcidicconditionsallowforPTEstodesorbanddissolvefromclays (F1Tess)andcarbonates(F2Tess)respectively,allowingthepollutantsto existinsolution(freeions)(Fig.10A).PTEsinthereduciblefraction (F3Tess)arereleasedbydissolutionofoxidemineralsunderhypoxicand anoxicconditions.ThefatesofthesePTEsinsolutionarecontrolledby lowoxygenconditionsandlowEhthatpromotetheirsequestrationby sulfideprecipitation(e.g.,pyrite-FeS2)orbybecomingcomplexedwith organicmatterandotherligands(e.g.,Cl ,SO4 2)(e.g., MartínezColónetal.,2009).PTEsinorganicmatter(F4Tess)canbereleased underhighlyoxygenatedconditionsandtheycanbeboundbysulfide precipitation,complexationoradsorptionbyclays(Fig.10B).ConditionsinTLpromotingoxidationoforganicmattercanberelatedto strongmixingevents(e.g.,tropicalstorms)ortoshallowandwelloxygenatedareas.Underoxygenatedandnormal-marinepHconditions (Fig.10B),PTEremobilizationislimitedtoadsorption(F1Tess),CaCO3 precipitation(F2Tess)andoxidemineralformation(F3Tess).Basedon theseobservations,PTEsinsolution(e.g.,free-ions)orboundtoorganic mattershouldbemorereadilybioavailableduringcalcificationand feeding(e.g.,detritus-feedingprotistssuchasbenthicforaminifera).
Forexample,absoluteabundancesof A.beccarii and Triloculina coupledwithFDhavenocorrelationwithbulkCu,ZnandFe,butare negativelycorrelatedwithF2Tess-Cu,F2Tess-Zn,andpositivelywith F2Tess-Fe(except Triloculina) (AppendixD,Supplementalmaterial). However,thesePTEsmayhavelimitedbioavailabilitytotheforaminifers.FractionF2Tess-Cu,aswellforZnandFe,translatestoCuions lockedinthecrystallinestructureofcarbonateminerals.Theonly scenarioinwhichPTEsfoundintheF2Tess fractionwouldbecome bioavailableiswhendissolutionofthecarbonatemineralsoccursand thePTEsareremobilizedinsolution(e.g.,inlowpHconditions).
NumerousauthorshavefoundcorrelationsbetweenbulkCuandkey foraminiferaltaxa(e.g., Alve,1991;DebenayandFernandez,2009; Martinsetal.,2011).InTLforexample,QRA,andTRAarepositively correlatedtobulkCu,Zn,andFe(andthesewithTOCandmud)butnot toF2Tess-Cu,F2Tess-Zn,andF2Tess-Fe(AppendixD-Supplementalmaterial).Similarly,absoluteabundancesof Q.rhodiensis positivelycorrelatedwithbulkCuandFebutnotwiththeirbioavailablecounterparts.Thesecorrelationsindicatethatthecumulativenatureofbulk concentrationsmayexplainsomestressresponses,butnotall,andthose inconsistenciesininterpretationscanoccurwhenchemicalspeciation andfractionationarenotconsidered.OurstudysuggeststhattheF2Tess acid-solublefractionisnotasatisfactoryassessmentchoiceduetoits verylimitedimpactonforaminifers,astheydonothavedigestive systems,andenvironmentsthataresufficientlyacidictodissolveCaCO3 arenotamenabletocalcareousforaminifers.
Martinsetal.(2011,2013) and Schintuetal.(2016) conductedPTE
fractionationfollowingtheEuropeanCommunityBureauofReference protocols(BCR).Forexample,theF1BCR fractioncombinestheexchangeable(F1Tess)andacid-soluble(F2Tess)fractionsof Tessieretal. (1979) andtheF2BCR andF3BCR arethereducibleandoxidizablefractionsrespectively.TheyfoundsignificantcorrelationsbetweenF1BCR andF2BCR withassemblages(Ammoniatepida and Quinqueloculina seminulum)anddeformities.ThisobservationisalsoinconclusivebecauseitischallengingtodeterminewhichPTEsintheF1BCR fraction (exchangeableoracid-soluble)areaffectingtheforaminifers.Inaddition,thesestudieshavefoundrelativehighaccumulationofPTEsin F2BCR (Fe/Mnoxides)andhavedescribedthisfractionashighlymobile andbioavailable.
Cautionalsomustbeexercisedwheninterpretingthestatistical correlationbetweenecologicalparametersandFe/Mnoxidefraction. Forexample,inTLthepositivecorrelationofCuwithFe/Mnsuggests thatthisPTEcouldbeintheF3Tess fraction(AppendixB,Supplemental material).Furthermore,thepositivecorrelationofQRAandTRAwith Fe/Mn(AppendixD,Supplementalmaterial)couldbeinterpretedasthe F3Tess beingabioavailablestressortoforaminifers.However,PTEs potentiallyfoundinthisF3Tess fractionarenotdirectlybioavailable becausethesecontaminantswillbefoundwithinthecrystallinestructureofFexOx/MnxOx minerals,butcanonlybereleasedunderreducing conditions. Lariosetal.(2012) foundthatAs+5 boundtoferrichydroxideswaslaterreleasedasAs+3 underreducingconditions.
WhenexaminingtheinfluenceofPTEsonforaminiferalassemblages,fractionationofeachPTEofinterest,includingthechemical conditionsunderwhichtheymightbecomebioavailable,shouldbe considered.Sequentialextractionsarerequiredtoelucidatewhichpotentialbioavailablefractionwillhaveanimpact.Ofthe fivefractions, theF1Tess exchangeablephaseisconsideredthemostbioavailableand theF5Tess residualphasetobetheleastbioavailable(e.g., Tessieretal., 1979).Thequestionis:Whatchemicalfractionisconsideredbioavailabletotheforaminifers?Benthicforaminifersaredetritivores,PTEs foundinF4Tess fractionshouldbeconsideredhighlybioavailablesince thisisalikelypathwayofPTEbioaccumulation(Martínez-Colónetal., 2009).However,studieshaveconcludedthatPTEsintheF3BCR fraction tobelesstoxicandbioavailablethanthosefoundintheF2BCR fraction incoastalenvironmentsinPortugal(Martinsetal.,2013)andSardinia (Schintuetal.,2016).FractionF1Tess couldalsobeconsideredbioavailable,sincePTEscouldbereadilydesorbedintofreeionsbyminor changesinpH,Ehandsalinity.Thisscenariocouldhappenatthesedimentwaterinterfaceorduringcalcificationinwhichforaminifers reducetheirinternalpHduringvacuolization.Thisisconsistentwith the findingsof Zaaboubetal.(2015),inwhichtheyconcludedthat PTEsintheexchangeableportionoftheF1Tess fractionisthemost mobileandthatremobilizationiscontrolledlargelybypHandredox conditions.
Moreover,theenvironmentalconditionsinthestudiedareasmust alsobeconsidered.Forexample,inTL,thepresenceofwelloxygenated, hypoxicandanoxicconditions,aswellasnormalmarinepHtolowpH
(7.2)gradients,withinafewmetersofeachotherinveryshallow water,representsapotentiallyverydynamicchemicalenvironment, withsomechangesinchemicalenvironmentoccurringonday-night cycles,tidalcycles,andespeciallywithstormactivitythatinduces mixingofotherwisehighlystratified,verylocalizedwaters.Thus,the bioavailabilityofindividualelementscanchangespatiallyonscales rangingfromcentimeterstokilometers,andtemporallyonscalesrangingfromhourstoseasonorpossiblyyears(e.g.,majorstormevents).
6.Conclusion
SeveralareasinTorrecillasLagoonshowstrongstratificationwith hypoxic/anoxic(DO<3mg/L)andcorrosive(pH<7.4)conditions below3.5mdepth.ThesesettingslikelyimpactbothPTEfractionation andforaminiferalassemblagedistributions.Foraminiferalassemblages dominatedbylowdensitiesof A.beccarii, Q.rhodiensis,and T.oblonga, andcoupledwithlowecologicalindexvalues[H(S),E,FD]anddeformedforaminifers(2–18%),areindicativeofstressedconditions.The spatialvariabilityofPTEsinbulkconcentrations(concentrationgradient:Zn>V>Cu>Cr>Li>Ni>As>Pb>Fe>Mn>Se) suggeststheinfluenceofnumerouspointandnon-pointsourcesof pollution.BulkPTEconcentrationsaremostlynegativelycorrelated withforaminiferaltaxaandseveralecologicalindices,butveryminimal correlationwiththeacid-soluble “bioavailable” PTEfraction(F2Tess) (concentrationgradient:F2Tess-Cu>F2Tess-Zn>F2Tess-Fe)wasfound.
Interpretationsbasedonbulkconcentrationsarenotrecommended sincethereisnodirectwayofdeterminingwhichchemicalfractionis bioavailableandishavinganimpactonforaminifers.Inretrospect,the assessmentofF2Tess asabioavailablefractionimpactingforaminifersis alsonotrecommended.Inthisstudy,PTEscorrelatedpositivelywith mudandTOC,andareinferredtobeadsorbedorcomplexedinthe F1Tess (exchangeable)andF4Tess (oxidizable)fractions.Thesetwo fractionsarelikelyofgreaterimportanceandbioavailabilitythanthe F2Tess (acid-soluble),F3Tess (reducible),andF5Tess (residual)fractions.
ThediscrepanciesobservedbetweenbulkandF2Tess-bioavailable PTEswithforaminifersstronglysupporttheideaofimplementingsequentialextractionprotocolsinsteadofthetotaldigestiontobetter elucidatetheactualimpactsonforaminifers.Cautionisrecommended whennotconsideringthebioavailabilityofPTEsinpollutionstudies duetothepotentialforoverestimation.Moreover,therecognitionof environmentalconditions,thedynamicnatureoftheenvironment,and thepotentialforsequesteredandcomplexedPTEstobemobilizedcan provideessentialinsightforcoastalmanagementagenciesthatmust assesstherisksofexistingPTEsandfuturepollutionsources,especially duringcoastalengineeringactivities(e.g.,dredgeand fillactivities)and majorstormevents.
Acknowledgments
TheauthorswouldliketothankDr.JorgeBauzá,Mrs.AdelísCabán andGladysRiverafromtheSanJuanBayEstuarysystemfortheir field support.ICONOSimagesfromTorrecillasLagoonwereprovidedbythe GeologicalandEnvironmentalRemoteSensingLaboratoryfromthe UniversityofPuertoRico-MayaguezCampus.Specialthanksto HumbertoBojórquez-LeyvafromUniversidadAutónomadeMéxico’s InstituteofMarineScienceandLimnologyforhisAtomicAdsorption SpectroscopytrainingandtoMrs.MollyR.McLaughlinfromtheUSGS CoastalandMarineScienceCenterinSt.Petersburg-FLfor field equipmentandfreezedrying.Wewillliketoacknowledgethehelpful commentsofDr.WarnerIthier-GuzmánandDr.CandiceBrownonthe improvementofthismanuscriptaswellastothreeanonymousreviewers.ThisworkwassupportedbyPuertoRicoSeaGrant(grant numberR-21-1-08)andtheU.S.GeologicalSurveyCooperative Agreement(grantnumber99HQAG0004).
AppendixA.Supplementarydata
Supplementarydataassociatedwiththisarticlecanbefound,inthe onlineversion,at https://doi.org/10.1016/j.ecolind.2017.10.045
References
Alve,E.,Goldstein,S.T.,2003.Propaguletransportasakeymethodofdispersalin benthicforaminifera(Protista).Limnol.Oceanogr.48,2163–2170
Alve,E.,Korsun,S.,Schönfeld,J.,Dijkstra,N.,Golikova,E.,Hess,S.,Husum,K.,Panieri, G.,2016.Foram-AMBIAsensitivityindexbasedonbenthicforaminiferalfaunasfrom North-EastAtlanticandArcticfjords,continentalshelvesandslopes.Mar. Micropaleontol.122,1–12
Alve,E.,1991.Benthicforaminiferainsedimentcoresreflectingheavymetalpollutionin Sørfjord,westernNorway.J.Foraminifer.Res.21,1–19
Alve,E.,1995.Benthicforaminiferalresponsestoestuarinepollution:areview.J. Foraminifer.Res.25,190–203
ArmynotduChâtelet,E.,Debenay,J.-P.,Soulard,R.,2004.Foraminiferalproxiesfor pollutionmonitoringinmoderatedpollutedharbors.Environ.Pollut.127,27–40
ArmynotduChâtelet,E.,Bout-Roumazeilles,V.,Riboulleau,A.,Trentesaux,A.,2009. Sediment(grainsizeandclaymineralogy)andorganicmatterqualitycontrolon livingbenthicforaminifera.Rev.Micropaleontol.52,75–84.
Arslan,M.,Kaminski,M.A.,Tawabini,B.S.,Ilyas,M.,Babalola,L.O.,Frontalini,F.,2016a. Seasonalvariations,environmentalparametersandstandingcropassessmentof benthicforaminiferaineasternBahrainl.Arab.GulfGeol.Quat.60,34–35
Arslan,M.,Kaminski,M.A.,Tawabini,B.S.,Ilyas,M.,Frontalini,F.,2016b.Benthicforaminiferainsandy(siliciclastic)coastalsedimentsoftheArabianGulf(SaudiArabia): atechnicalreport.Arab.J.Geosci.9,1–7
Bacon,J.R.,Davidson,C.M.,2008.Isthereafutureforsequentialchemicalextraction? Analyst133,25–46
Bergin,F.,Kucuksezgin,F.,Uluturhan,E.,Barut,I.,Meric,E.,Avsar,N.,Nazik,A.,2006. TheresponseofbenthicforaminiferaandostracodatoheavymetalpollutioninGulf ofIzmir(EasternAegeanSea).Estuar.CoastShelfSci.66,368–386 Bernhard,J.M.,2000.Distinguishinglivefromdeadforaminifera:methodsreviewand properapplications.Micropaleontology46,37–46
Bouchet,V.M.P.,Alve,E.,Rygg,B.,Telford,R.J.,2012.Benthicforaminiferaprovidea promisingtoolforecologicalqualityassessmentofmarinewaters.Ecol.Indic.23, 66–75
Bunch,B.W.,Cerco,C.F.,Dortch,M.S.,Johnson,B.H.,Kim,K.W.,2000.Hydrodynamic andwaterqualitymodelstudyofSanJuanBayEstuary.In:TechnicalReportERDCTR-00-1,U.S.ArmyEngineerWaterwaysExperimentStation.Vicksburg,MississippiUSA Buzas-Stephens,P.,Buzas,M.A.,2005.PopulationdynamicsanddissolutionofforaminiferainNuecesBay,Texas.J.Foraminifer.Res.35,248–258 Carnahan,E.A.,Hoare,A.M.,Hallock,P.,Lidz,B.H.,Reich,C.D.,2009.Foraminiferal assemblagesinBiscayneBay,Florida,USA:Responsestourbanandagricultural influenceinasubtropicalestuary.Mar.Pollut.Bull.59,221–233 Clarke,K.R.,Gorley,R.N.,2006.PRIMERv6:UserManual/TutorialPRIMER-E,Plymouth Dabbous,S.A.,Scott,D.B.,2012.Short-termmonitoringofHalifaxHarbour(NovaScotia, Canada)pollutionremediationusingbenthonicforaminiferaasproxies.J. Foraminifer.Res.42,187–205
Davies,B.E.,Bowman,C.,Davies,T.C.,Selinus,O.,2005.Medicalgeology:perspectives andprospects.In:Selinus,O.,Alloway,B.,Centeno,J.A.,Finkelman,R.B.,Fuge,R., Lindh,U.,Smedley,P.(Eds.),EssentialsofMedicalGeology.ElsevierAcademicPress, NewYork,pp.1–41
Debenay,J.-P.,Fernandez,J.-P.,2009.Benthicforaminiferarecordsofcomplexanthropogenic environmentalchangescombinedwithgeochemicaldatainatropicalbayof NewCaledonia(SWPacific).Mar.Pollut.Bull.59,311–322
Debenay,J.-P.,Tsakiridis,E.,Soulard,R.,Grossel,H.,2001.Factorsdeterminingthe distributionofforaminiferalassemblagesinPortJoinvilleHarbor(Iled’Yeu,France): theinfluenceofpollution.Mar.Micropaleontol.43,75–118
Debenay,J.-P.,DellaPatrona,L.,Herbland,A.,Goguenheim,H.,2009.Theimpactof easilyoxidizedmaterial(EOM)onthemeiobenthos:foraminiferaabnormalitiesin shrimppondsofNewCaledonia;implicationsforenvironmentandpaleoenvironment survey.Mar.Pollut.Bull.59,323–335
deNooijer,L.J.,Reichart,G.J.,Duenas-Bohorquez,A.,Wolthers,M.,Ernst,S.R.,Mason, P.R.D.,vanderZwaan,G.J.,2007.Copperincorporationinforaminiferalcalcite: resultsfromculturingexperiments.Biogeosci4,493–504
Dias,B.B.,Hart,M.B.,Smart,C.W.,Hall-Spencer,J.M.,2010.Modernseawateracidification:theresponseofforaminiferatohigh-CO2 conditionsintheMediterranean Sea.J.Geol.Soc.167,843–846
Eichler,P.P.B.,Eichler,B.B.,SenGupta,B.,RöschRodrigues,A.,2012.Foraminiferaas indicatorsofmarinepollutantcontaminationontheinnercontinentalshelfof southernBrazil.Mar.Pollut.Bull.64,22–30
Ellis,S.R.,Gómez-Gómez,F.,1976.HydrologiccharacteristicsoflagoonsatSanJuan, PuertoRico,duringaJanuary1974tidalcycle.U.S.Geol.Surv.Water-Resour. Investig.38,52
Ellis,S.R.,1976.Historyofdredgingand fillingoflagoonsintheSanJuanarea,Puerto Rico.U.S.G.SWater-ResourcesInvestigationsReport,pp.38–76
Emrich,K.,Martínez-Colón,M.,Alegría,H.,2017.Isuntreatedsewageimpactingcoral reefsofCayeCaulker,Belize?J.Foraminifer.Res.47,20–33
Fontanier,C.,Fabri,M.-C.,Buscail,R.,Biscara,L.,Koho,K.,Reichart,G.J.,Cossa,D., Galaup,S.,Chabaud,G.,Pigot,L.,2012.Deep-seaforaminiferafromtheCassidaigne Canyon(NWMediterranean):Assessingtheenvironmentalimpactofbauxiteredmud
disposal.Mar.Pollut.Bull.64,1895–1910
Foster,W.J.,ArmynotduChâtelet,E.,Rogerson,M.,2012.Testingbenthicforaminiferal distributionsasacontemporaryquantitativeapproachtobiomonitoringestuarine heavymetalpollution.Mar.Pollut.Bull.64,1039–1048
Frontalini,F.,Coccioni,R.,2008.Benthicforaminiferaforheavymetalpollutionmonitoring:acasestudyfromthecentralAdriaticSeacoastofItaly.Estuar.CoastShelf Sci.74,404–417
Gómez-Gómez,F.,Quiñonez,F.,Ellis,S.R.,1983.Hydrologiccharacteristicsoflagoonsat SanJuan,PuertoRico,duringanOctober1974tidalcycle.U.S.GeologicalSurvey Water-ResourcesInvestigationsOpen-FileReport#82-34941p
Green-Ruíz,C.,Ruelas-Inzunza,J.,Páez-Osuna,F.,2005.Mercuryinsurfacesediments andbenthicorganismfromGuaymasBay:eastcoastoftheGulfCalifornia.Environ. Geochem.Health27,321–329
Gross,M.G.,1978.EffectsofwastedisposaloperationsinestuariesandtheCoastalocean. Annu.Rev.EarthPlanet.Sci.6,127–143
Hayek,L.-A.C.,Buzas,M.A.,1997.SurveyingNaturalPopulations.ColumbiaUniversity Press,NewYork
Hunter,J.M.,Arbona,S.I.,1995.ParadiselostAnintroductiontothegeographyofwater pollutioninPuertoRico.Soc.Sci.Med.40,1331–1355
Jorissen,F.J.,1999.Benthicforaminiferalmicrohabitatsbelowthesediment-waterinterface.In:SenGupta,B.K.(Ed.),ModernForaminifera.KluwerAcademicPublishers, Boston,pp.179–191
Kfouri,P.B.P.,Figueira,R.C.L.,Figueiredo,A.M.G.,Souza,S.H.M.,Eichler,B.B.,2005. MetallevelsandforaminiferaoccurrenceinsedimentcoresfromGuanabaraBay,Rio deJaneiro,Brazil.J.Radioanal.Nucl.Chem.265,459–466
Laboy-Nieves,E.,2009.Environmentalprofileandmanagementissuesinanestuarine ecosystem:acasestudyfromJobosBay,PuertoRico.In:Laboy-NievesEddie,FredC., Schaffner,AhmedAbdelhadi,Matheus,F.A.Goosen(Eds.),Environmental Management,SustainableDevelopmentandHumanHealth.CRCPress,Leiden-The Netherlands,pp.361–398
Larios,R.,Fernández-Martínez,R.,Rucandio,I.,2012.Comparisonofthreesequential extractionproceduresforfractionationofarsenicfromhighlypollutedminingsediments.Anal.Bioanal.Chem.402,2909–2921
Loeblich,A.R.,Tappan,H.,1987.ForaminiferalGeneraandTheirClassification.Van NostrandReinholdCompany,NewYork
Luoma,S.N.,Bryan,G.W.,1981.Astatisticalassessmentoftheformofthetracemetalsin oxidizedestuarinesedimentsemployingchemicalextractants.Sci.Total.Env.17, 165–196
Magurran,A.E.,1988.EcologicalDiversityandItsMeasurements.PrincetonUniversity Press,NewJersey-USA
Martínez-Colón,M.,Hallock,P.,2010.Preliminarysurveyonforaminiferalresponsesto pollutantsintorrecillaslagoonPuertoRico.Caribb.J.Sci.46,1–6
Martínez-Colón,M.,Hallock,P.,Green-Ruíz,C.,2009.Strategiesforusingshallow-water benthicforaminifersasbioindicatorsofpotentiallytoxicelements:areview.J. Foraminifer.Res.39,278–299
Martins,M.V.A.,Yamashita,C.,Sousa,S.H.M.,Martins,P.,Laut,L.L.M.,Figueira,R.C.L., Mahiques,M.M.,FerreiradaSilva,E.,AlveirinhoDias,J.M.,Rocha,F.,2011.The responseofbenthicforaminiferatopollutionandenvironmentalstressinRiade Aveiro(NPortugal)Larespuestadelosforaminíferosbentónicosalacontaminación yelestrésambientalenlaRíadeAveiro(NdePortugal).J.Iber.Geol.37,231–246
Martins,M.V.A.,Frontalini,F.,Tramonte,K.M.,Figueira,R.C.,Miranda,P.,Sequeira,C., Fernandez-Fernandez,S.,Dias,J.A.,Yamashita,C.,Reno,R.,Laut,L.L.,Silva,F.S., Rodrigues, M.A.,Bernardes,C.,Nagai,R.,Sousa,S.H.,Mahiques,M.,Rubio,B., Bernabeu,A.,Rey,D.,Rocha,F.,2013.AssessmentofthehealthqualityofRiade Aveiro(Portugal):heavymetalsandbenthicforaminifera.Mar.Pollut.Bull.70, 18–33
Martins,M.V.A.,Silva,F.,Laut,L.L.M.,Frontalini,F.,Clemente,I.M.M.M.,Miranda,P., Figueira,R.,Sousa,S.H.M.,Dias,J.M.A.,2015.Responseofbenthicforaminiferato organicmatterquantityandqualityandbioavailableconcentrationsofmetalsin aveirolagoon(Portugal).PLoSOne10. http://dx.doi.org/10.1371/journal.pone. 0118077
Martins,M.V.A.,Pinto,A.F.S.,Frontalini,F.,MachadodaFonseca,M.C.,Terroso,D.L., Laut,L.L.M.,Zaaboub,N.,daConceicao-Rodrigues,M.A.,Rocha,F.,2016.Can benthicforaminiferabeusedasbio-indicatorsofpollutioninareaswithawiderange ofphysiochemicalvariability?Estuar.CoastShelfSci.182,211–225
Oliver,L.M.,Fisher,W.S.,Dittmar,J.,Hallock,P.,Campbell,J.,Quarles,R.L.,Harris,P., LoBue,C.,2014.ContrastingresponsesofcoralreeffaunaandforaminiferalassemblagestohumaninfluenceinLaParguera,PuertoRico.Mar.Environ.Res.99, 95–105. http://dx.doi.org/10.1016/j.marenvres.2014.04.005
Parker,W.C.,Arnold,A.J.,1999.Quantitativemethodsofdataanalysisinforaminiferal ecology.In:SenGupta,B.K.(Ed.),ModernForaminifera.KluwerAcademic Publishers,Boston,pp.71–89.
Perez-Cid,B.,Lavilla,I.,Bendico,C.,1998.Speedingupofathree-stagesequentialextractionmethodformetalspeciationusingfocusedultrasound.Anal.Chim.Acta360, 35–41
Poag,W.,1981.EcologicAtlasofBenthicForaminiferaoftheGulfofMexico.Hutchinson RossPublishingCompany Romano,E.,Bergamin,L.,Finoia,M.G.,Carboni,M.G.,Ausili,A.,Gabellini,M.,2008. IndustrialpollutionatBagnoli(Naples,Italy):Benthicforaminiferaasatoolinintegratedprogramsofenvironmentalcharacterization.Mar.Pollut.Bull.56,439–457 Ross,B.,Hallock,M.,2016.Dormancyintheforaminifera:areview.J.Foraminifer.Res. 46,358–368
Salvi,G.,Buosi,C.,Arbulla,D.,Cherchi,A.,DeGiudici,G.,Ibba,A.,DeMuro,S.,2015. Ostracodaandforaminiferaresponsetoacontaminatedenvironment:thecaseofthe Ex-MilitaryArsenaloftheLaMaddalenaHarbour(Sardinia,Italy). Micropaleontology61,115–133
Samir,A.M.,El-Din,A.B.,2001.Benthicforaminiferalassemblagesandmorphological abnormalitiesaspollutionproxiesintwoEgyptianbays.Mar.Micropaleontol.41, 193–227
SanJuanBayEstuary,2000.ComprehensiveandConservationandManagementPlan (CCMP)fortheSanJuanBayEstuary. http://estuario.org/index.php/ccmp-english (Accessed05.05.16).
Schönfeld,J.,Alve,E.,Geslin,E.,Jorissen,F.,Korsun,K.,Spezzaferri,S.,Abramovich,S., Almogi-Labin,A.,ArmynotduChatelet,E.,Barras,C.,Bergamin,L.,Bicchi,E., Bouchet,V.,Cearreta,A.,DiBella,L.,Dijkstra,N.,Disaro,S.T.,Ferraro,L.,Frontalini, F.,Gennari,G.,Golikova,E.,Haynert,K.,Hess,S.,Husum,K.,Martins,V.,McGann, M.,Oron,S.,Romano,E.,Sousa,S.M.,Tsujimoto,A.,2012.TheFOBIMO (FOraminiferalBIo-MOnitoring)initiative–Towardsastandardizedprotocolforsoftbottombenthicforaminiferalmonitoringstudies.Mar.Micropaleontol.94,1–13. Schafer,C.T.,2000.Monitoringnearshoremarineenvironmentsusingbenthicforaminifera:someprotocolsandpitfalls.Micropaleontology46,161–169 Schintu,M.,Marrucci,A.,Marras,B.,Galgani,F.,Buosi,C.,Ibba,A.,Cherchi,A.,2016. HeavymetalaccumulationinsurfacesedimentsattheportofCagliari(Sardinia, westernMediterranean):environmentalassessmentusingsequentialextractionand benthicforaminifera.Mar.Pollut.Bull.111,45–56
Schwing,P.T.,O’Malley,B.J.,Romero,I.C.,Martínez-Colón,M.,Hastings,D.W.,Glabach, M.A.,Hladky,E.M.A.,Greco,A.,Hollander,D.J.,2016.Characterizingthevariability ofbenthicforaminiferainthenortheasternGulfofMexicofollowingtheDeepwater Horizonevent.Env.Sci.Poll.Res.2010–2012. http://dx.doi.org/10.1007/s11356016-7996-z
Seguinot-Barbosa, J.,1983.CoastalModificationsandLandTransformationintheSan JuanBayArea:PuertoRico.UnpublishedPhDDissertationLouisianaState University,BatonRougeLouisiana
Seiglie,G.A.,1968.Foraminiferalassemblagesasindicatorsofhighorganiccarbon contentinsedimentsandpollutedwaters.Am.Assoc.Petrol.Geol.Bull.52, 2231–2241
Seiglie,G.A.,1971.Apreliminarynoteontherelationshipsbetweenforaminifersand pollutionintwoPuertoRicanBays.Caribb.J.Sci.11,93–98
Seiglie,G.A.,1973.Pyritizationinlivingforaminifers.J.Foraminifer.Res.3,1–6
Seiglie,G.A.,1975a.ModificationsaL’Holocenesuperieurdeforaminiferesdelabaiede jobosetsesenvirons,PortoRico.Rev.Micropaleontol.19,101–107
Seiglie,G.A.,1975b.LateHoloceneChangesontheForaminiferalAssemblagesofJobos BayandSurroundings,PuertoRico.AguirrePowerProject,EnvironmentalStudies JobosBayPuertoRicoFinalReport.PuertoRicoNuclearCenter#196
Seiglie,G.A.,1975c.ForaminifersofGuayanillaBayandtheiruseasenvironmentalindicators.Rev.Esp.Micropaleontol.7,453–487
Shannon,C.E.,1948.Amathematicaltheoryofcommunication.BellSyst.Tech.J.27, 379–423
Szefer,P.,Glasby,G.P.,Pempkowiak,J.,Kaliszan,1995.ExtractionstudiesofheavymetalpollutantsinsurficialsedimentsfromthesouthernBalticSeaoff Poland.Chem. Geol.120,111–126
Tessier,A.,Campbell,P.G.C.,Bisson,M.,1979.Sequentialextractionprocedureforthe speciationofparticulatetracemetals.Anal.Chem.51,844–851
Webb,R.M.T.,Gómez-Gómez,F.,1998.SynopticSurveyofWaterQualityandBottom Sediments,SanJuanBayEstuarySystem,PuertoRico,December1994–July1995. U.S.G.S.WaterResourcesInvestigationsReport#97-4144
Wilson,H.M.,1899.WaterResourcesofPuertoRico.U.S.GeologicalSurveyWater SupplyandIrrigationPapers#32
Yanko,V.,Ahmad,A.J.,Kaminski,M.,1998.Morphologicaldeformitiesofbenthicforaminiferainresponsetopollutionbyheavymetals:implicationsforpollutionmonitoring.J.Foraminifer.Res.28,177–200
Yanko-Hombach,V.,Kondariuk,T.,Motnenko,I.,2017.BenthicForaminiferaindicate environmentalstressfromriverdischargetomarineecosystems:examplefromthe BlackSea.J.Foraminifer.Res.47,70–92
Zaaboub,N.,Martins,M.V.A.,Dhib,A.,Bejaoui,B.,Galgani,F.,ElBour,M.,Aleya,L., 2015.AccumulationoftracemetalsinsedimentsinaMediterraneanLagoon:usefulnessofmetalsedimentfractionationandelutriatetoxicityassessment.Env.Pollut. 207,226–237
Zimmerman,A.J.,Weindorf,D.C.,2010.Heavymetalandtracemetalanalysisinsoilby sequentialextraction:areviewofprocedures.Int.J.Anal.Chem.2010,1–7
