mmc2025 incorporating EMAG 2025: We’ll see you there!
Don’t judge a book by its dust
BioImagingUK Workshop on User Engagement: Sales Skills for Facility Staff
Modified stereo-image acquisition technique for Scanning Electron Microscopy (SEM)


mmc2025 incorporating EMAG 2025: We’ll see you there!
Don’t judge a book by its dust
BioImagingUK Workshop on User Engagement: Sales Skills for Facility Staff
Modified stereo-image acquisition technique for Scanning Electron Microscopy (SEM)




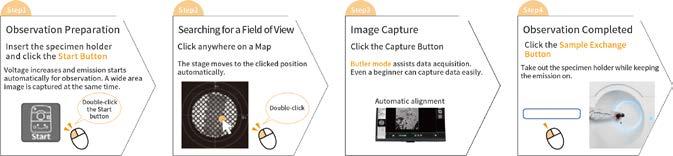
Specimen observation in 4 steps
We developed the JEM-120i with the concept of "Compact", "Easy To Use", and "Expandable". With the new external appearance, this instrument has evolved into a useful tool that anyone can use easily, from operation to maintenance.
It takes only 4 steps from loading a specimen to completing observation. The JEM-120i is equipped with an enhanced TEM control system and fully automated apertures, eliminating the need for switching magnification modes and selecting an aperture. Observation operations can be performed more smoothly than with previous models.
Annelies
infocus is the Magazine of the Royal Microscopical Society (RMS) –the only truly international microscopical society. The RMS is dedicated to advancing science, developing careers and supporting wider understanding of science and microscopy.
infocus Magazine
37/38 St Clements
Oxford, OX4 1AJ, UK
Tel. +44 (0)1865 254760
Email: infocus@rms.org.uk Website: www.infocus.org.uk
Scientific Editor
Leandro Lemgruber, University of Glasgow, UK
Editor
Owen Morton
Tel. + (0)1865 254763, Email: editor@infocus.org.uk
Editorial Board
Susan Cox, King’s College, London, UK
Rebecca Higginson, Loughborough University, UK
Myfanwy Adams, John Innes Centre, Norwich, UK
Maadhav Kothari, Zeiss Microscopy, UK
Hilary Sandig, Cancer Research, UK
Trevor Almeida, University of Glasgow, UK
Mark Rigby, Nikon UK
Advertising
Email: advertising@infocus.org.uk
ISSN: 1750-4740
© 2025 Royal Microscopical Society
infocus is published four times per year by the RMS. Designed and produced by Creative Design. Reproduction in whole or in part without permission from the RMS is forbidden. Views expressed in the Magazine are those of the individual contributors and do not necessarily reflect those of the RMS.

Dear Readers,
Welcome to our June 2025 issue of infocus Magazine!
For those in the northern hemisphere, I hope you are enjoying the summer; for our readers in the south, I hope winter has been kind so far!
As always, there’s a huge amount going on in the world of microscopy – including the final preparations for mmc2025 (incorporating EMAG 2025), the RMS’s flagship event for the international microscopy community, taking place in Manchester from 1 – 3 July.
There is always so much to see and do at mmc, and you can read all about what this year’s Congress has to offer in our special preview. If you’re heading to Manchester, then please come and chat to me or our board members about infocus Magazine, especially if you would like to contribute an article. Much like mmc2025, our June issue certainly has a diverse range of content for your reading pleasure.
On a training and professional development theme, we have a pair of articles reminding us that whatever one’s role in microscopy, a wide range of operational, technical and so-called ‘soft’ skills remain essential. From our colleagues at BioImagingUK, we have an account of a recent survey and training workshop on sales skills for core facility staff. Meanwhile, Annelies van der Laan gives a thorough account of her time job shadowing at the University of York’s Imaging and Cytometry Facility - led by RMS President Peter O’Toole.As it happens, I myself recently spent some time shadowing the team at York thanks to the Technical Specialist Job Shadowing Scheme from BioImagingUK. I can fully recommend job-shadowing as a highly effective means of upgrading expertise and operational knowledge.
For the history buffs, we have a fascinating article from Charlotte May, Luke Norman and colleagues at University of Nottingham, in which we find out how microscopy and imaging provided new perspectives on a Medieval manuscript – including the tiny creatures hidden in the binding! For the art lovers among you, we take a look some microscopy-themed tattoos (full disclosure, they belong to me!) and wonder out loud if anyone else has been similarly inspired by the microscope in their choice of body art?
Finally, from the realms of technical tips and tricks, we have an excellent article by Jim Buckman and Vladimir Krivtsov on an ingenious and user-friendly method for the acquisition of stereo-image pairs by SEM.
I hope you enjoy reading all the content in this issue, and hope to see you at mmc! Slàinte!
Leandro Lemgruber

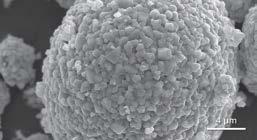
COVER IMAGE: Carnation flower.
By Shiva Pesaran, Central Laboratory, Shiraz University. Flower-like structure of manganese and protein. TESCAN SEM VEGA3. Magnification: 10KX.



mmc2025 incorporating EMAG 2025
The excitement is well and truly building as preparations are finalised for Microscience Microscopy Congress (mmc2025 incorporating EMAG 2025)!
mmc
is renowned as one of the leading international events in microscopy, imaging, and flow cytometrybringing together hundreds of people who use microscopes for work, study and pleasure. Alongside a huge, three-day conference, the event boasts a world-class exhibition, showcasing the very best in microscopy for research and industry visitors. There is also a wide range of workshops, satellite meetings, social networking opportunities and more. This year, we’re also aligning our flagship event more closely with our peer-reviewed publication - the Journal of Microscopy – by encouraging poster presenters and speakers to submit papers for a special issue!
Ahead of what promises to be one of the biggest and best editions of the congress series, we’re taking a look at some of the highlights of this year’s event – coming once again to Manchester Central, UK, from 1 – 3 July 2025. Registration is still open, so now is the time to book your place at this fantastic event for the microscopy and imaging community.
Book now: https://www.mmc-series.org.uk/ We look forward to seeing you there!
With more than 400 talk and poster abstracts submitted and a bumper scientific programme covering all aspects of microscopy and imaging, our mmc2025 (incorporating EMAG 2025) conference is shaping up to be one of the best in the history of the congress series.
Organised across six parallel streams over three days, the conference will feature multiple sessions covering:
• AFM and SPM Microscopies
• Frontiers in BioImaging
• Life and Physical Sciences
• EMAG 2025
Our ‘late-breaking’ session on Tuesday 1 July will also enable researchers to present the most exciting recent developments in the field.
Now is the time to get acquainted with the full, Scientific Programme , to make note of the sessions and talks that interest you the most, and ensure the best possible mmc2025 experience!



Peter Crozier (Arizona State University, USA)
Talk title: Seeing Atomic-Level Structural Dynamics in Materials: Managing Temporal Resolution and Electron Dose Rates

Peter A. Crozier is a Professor of Materials in the School for Engineering of Matter, Transport and Energy at Arizona State University. He has extensive experience in characterising ceramics, metal nanoparticles and catalytic materials for applications related to energy and the environment.
Read more
Uri Manor (University of California San Diego, USA)
Talk title: Novel tools for studying organelle dynamics in health and disease

Dr Uri Manor is the Faculty Director of the Goeddel Family Technology Sandbox and Assistant Professor of Cell & Developmental biology at the University of California, San Diego
School of Biological Sciences. The Manor Lab develops new methods and tools for studying cellular dynamics with nanometer precision.
Read more

Rachel Oliver (University of Cambridge, UK)
Talk title: Microscopy of Mesoporous Nitrides

Professor Rachel Oliver FREng is a materials engineer, inventor and spinout founder, and Director of the Cambridge Centre for Gallium Nitride. Her passion for understanding and engineering the small-scale structure of semiconductor materials to enable new technologies has been recognised in 2021 by her selection as an IEEE Photonics Society Distinguished Lecturer and in 2023 by the award of a Royal Academy of Engineering Chair in Emerging Technologies.
Read more

Jessica Wade (Imperial College London, UK)
Talk title: Navigating academia’s hidden curriculum

Dr Jess Wade is a Royal Society University Research Fellow and Lecturer in Functional Materials in the Department of Materials at Imperial College London. Her research considers new materials for optoelectronic devices and quantum technologies, with a focus on chiral systems and the identification of strategies to control photon and electron spin.
Read more
Assaf Zaritsky (Ben-Gurion University of the Negev, Israel)
Talk title: Extracting the invisible from live cell microscopy

Assaf Zaritsky is an Associate Professor in the Department of Software and Systems Information Engineering at Ben-Gurion University of the Negev (BGU), Israel. He performed his graduate training in computer science at Tel Aviv University, and postdoctoral training at UT Southwestern Medical Center at Dallas and joined BGU in October 2018.
Read more
As always, mmc2025 will feature a huge exhibition, with some of the leading companies in microscopy and imaging showcasing their latest products and technology. At the time of publication, almost 100% of all available space had already been sold out, with more than 90 exhibitors set to descend on Manchester Central.
Exhibition attendance is completely
FREE, and visitors will have access to a wide range of product demonstrations, commercial workshops and practical advice all under one big roof!
With three days of conference sessions on the menu, there will be ample opportunity to work your way around the stands during breaks and at other times when you’re not attending a talk.
Be sure to pay a visit to all the stands and find out what our generous sponsors have to offer!
Exhibitor list
3i - Intelligent Imaging
Innovations
AdvaScope s.r.o.
Agar Scientific, a Calibre Scientific Company
Agilent Technologies
AHF analysentechnik AG
Apex Probes Ltd
BioImagingUK
Blue Scientific Ltd
Bristol Nano Dynamics
Bruker
Cairn Research
Cephla
Chip-Nova Ltd
CN Tech
Confocal NL
ConnectomX
CoolLED Ltd
CooperSurgical Fertility and Genomics
CrestOptics spa
CSR Biotech
D-Tails
DAEIL SYSTEMS
DEBEN UK Ltd
DECTRIS
Delong Instruments
DENSsolutions
DRTEM / Single Particle
ELLIOT SCIENTIFIC LTD
EM Resolutions
EM Systems Support Ltd

EMSIS
Euclid Techlabs
Evident
Ferrovac
Gatan/EDAX
Graticules Optics Ltd
Hamamatsu Photonics UK
Limited
Henry Royce Institute
Hitachi High-Tech Europe
GmbH
HORIBA
HÜBNER Photonics
Hummingbird Scientific
IUCr Journals
IMC21
INSCOPER
ISS Group Services
JEOL
Journal of Microscopy
Kashiyama Europe GmbH
Lambda Photometrics Ltd
Laser 2000 (UK) Ltd
Leica Microsystems

Lightcore Technologies
Mel-Build
Merrow Scientific Ltd
Microbiology Society
Microscopy Society of America
MicroSupport Co.,Ltd.
Miltenyi Biotec
Molecular Machines and Industries
NanoMEGAS
Nanosurf
Nikon Healthcare UK
NINGBO YONGXIN OPTICS
CO., LTD.
Northern Light Microscopy
ONI
Oxford Instruments
Park Systems UK
Photon Lines Ltd
Prior Scientific Instruments Ltd
Protochips, Inc.
Quantum Design UK
Quantum Detectors Ltd
Quorum Technologies
Renishaw plc
RMC Boeckeler
SciMed Ltd
SenseAI
SmarAct GmbH
Spicer Consulting Limited
Technoorg Linda Co. Ltd.
Teledyne Photometrics
Telight
TESCAN
The Advanced Materials Show
Thermo Fisher Scientific
Toptica Photonics
turboTEM
VitroTem
Vmicro
XEI Scientific, Inc.
YPS
ZEISS
Journal of Microscopy
FocalPlane

Although the conference and exhibition takes place from 1 - 3 July, the preliminary action gets underway on Monday 30 June, with our pre-congress meetings and workshops.
These are as follows:
This pre-congress meeting provides an opportunity for the UK Bioimaging community to discuss priorities and strategies in national infrastructure, technology development, training, careers and ways to share knowledge across different disciplines. The session will consist of short talks from industry and academia, and interactive Q+A sessions.
For the first time, the event will conclude with an exciting Industry-Academia ‘Speed Networking’ Event. This pilot initiative aims to bridge the gap between academic researchers and industry partners in the bioimaging field.
The Early Career Symposium is an interdisciplinary event aimed at students, postdocs and early career professionals working in the field of microscopy. This will be a great opportunity for networking
ahead of the main mmc2025 conference, as well as enabling attendees to showcase their research with peers.
A highlight of the meeting will be the climax of the 2025 RMS Early Career Award, with the shortlisted entrants each delivering a flash talk about their research. The winner will be announced at the end of the meeting.
Pre-Conference workshops will take place on the afternoon of Monday 30 June.
They are a great way for attendees to boost their research by learning the most up-to-date tips and techniques. Three workshops will be taking place from 1.30 - 4.30pm on Monday 30 June, and they can be added during the booking process.
The confirmed workshops are:
• ImageJ
• EMAG - Introduction to Python for Electron Microscopists
• Quantitative Electron Backscatter Diffraction (EBSD) interpretation: what do we do with all those acronyms?
Find out more about all our pre-conference workshops


A u t o m a t e d T i s s u e P r o c e s s o r
T h e u l t i m a t e p r o d u c t i v i t y t o o l f o r
l a b s h a n d l i n g h i g h v o l u m e s o f
t i s s u e s p e c i m e n e m b e d d i n g
D i s c o v e r o u r m i c r o s c o p y s a m p l e
p r e p a r a t i o n s y s t e m s a n d w o r k f l o w
i n s t r u m e n t s a t w w w . b o e c k e l e r . c o m


The RMS International Scientific Imaging Competition is always a highlight of mmc, and especially so this year, as we have received a record number of entries – more than 230!
The competition features six different categories covering all forms of microscopy across the sciences. Once our judges have completed the difficult task of whittling down the entries, the
shortlisted micrographs in each category will be displayed in an attractive gallery throughout mmc2025.The competition always showcases some superb technical and artistic talent, and it’s well worth taking some time to see what our entrants have come up with this year.
Check out some of our previous winning entries

A major part of our flagship event since 2002, The Learning Zone is a tremendous, free, feature which is open to all visitors and conference delegates. Our team of expert volunteers will be on hand to answer questions, provide demonstrations and give advice throughout the exhibition.
The Learning Zone has its own seminar theatre hosting a daily programme of introductory talks. These talks are also free to attend and there is no need to book in advance - simply turn up and enjoy!



















As ever, mmc2025 will provide a wide range of networking opportunities – enabling delegates to catch up with friends and colleagues, and to make new connections across the international microscopy community.
As well as regular coffee and lunch breaks, our early evening poster sessions on the Tuesday (1 July) and Wednesday (2 July) present the perfect opportunity to grab some refreshments and discuss some of the latest research against the backdrop of the buzzing exhibition hall.
For those joining us on the Monday (30 June) for the pre-congress meetings and workshops, an informal BioImagingUK / Early Career Networking Reception will be held in the foyer of Manchester Central.
Conference registration also includes the option of attending one of our exclusive evening networking dinners at top Manchester restaurants. These have been organised respectively for attendees of the AFM and Scanning Probe Microscopies; BioImagingUK; and EMAG 2025 conference sessions.

The new mmc2025 App is a great tool to help you navigate your way through the Congress. You can create a personal schedule of the talks and workshops you want to catch, take notes and network with other delegates – and more.
The App is hosted by vFairs, so you will simply need to download the ‘vFairs’ App on your mobile or other device, and search for ‘mmc2025’.

If you haven’t already booked your hotel room, you need to get your skates on! Accommodation is not included with registration for mmc2025 but there are a huge number of hotels located just a short walk away from Manchester Central.
Visit our mmc2025 accommodation page for more information and a list of guide prices.There may still be time to take advantage of exclusive rates offered to conference delegates and exhibitors - though bookings will be subject to availability and prices may now vary.
Alternatively you can telephone the Reservation Highway helpdesk on +44 (0) 1423 525577 or email admin@reservation-highway.co.uk. You can also complete the downloadable booking form for mmc2025 if you prefer.
Check out the lectures and hands-on equipment in the RMS Learning Zone. These are great ways to learn more and improve your skills.
Socialise! Attend social events. Quite simply, this is the best way to network and get to know the people you ought to stay in touch with.
Check out the programme online in advance. Plan which sessions you wish to attend and familiarise yourself with their general location.
Wear comfortable shoes. You’ll probably be on your feet a fair bit, so don’t let them get sore – a sure-fire way to ruin any day!
Remember to visit the Exhibition, Workshops, Scientific Imaging Competition Display and Poster Village. Free refreshments are available in the Exhibition all day!
First time at mmc? – Here’s what to expect:
• Meet those who have shaped and changed the field of microscopy, imaging and flow cytometry, in both life and physical sciences.
• Network with mentors, professors and students – all of whom share your passion for microscopy and imaging.
• Seize the opportunity to forge relationships with potential future colleagues and collaborative partners for your research.
…And some helpful tips
Dress the part. Attire for the congress is business casual.
Attend the sessions that interest you. Sadly, there just won’t be time to see and do everything, so focus on the things you are most passionate about, rather than trying to attend every session.
Download the app. It’s a mobile and fast way to view the programme while you are attending sessions.
Find time to get out and about. Explore the amazing city of Manchester. The birthplace of the industrial revolution boasts great historical and cultural attractions, as well as shops, restaurants and nightlife.
Book your hotel – if you haven’t already!
Make booking easy and book your hotel room via the hotel booking agency on the mmc2025 website
Share your experience via Facebook, LinkedIn, and Twitter and use the event hashtag: #mmc2025

We are very pleased to continue offering a range of ‘in-person’ and virtual events this year, in order to maximise accessibility and provide opportunities to those who might not otherwise be able to attend.
The following information was correct at the time infocus went to print but could potentially be subject to change in the coming weeks. Please visit our event calendar at www.rms.org.uk for the latest updates.
If you have any questions about a booking you have already made for an event, or need any help or advice, please contact us at info@rms.org.uk
4 MidsEM 2025, Birmingham, UK (RMS exhibiting at event)
4 Expansion Microscopy User Group Meeting - Canada Hosted - June 2025, (Online)
9 GW4 Networking Day, Exeter, UK (RMS exhibiting at event)
9 – 10 Light Microscopy Summer School 2025, York, UK
10 – 11 South West Electron Microscopy 2025 (SW-EM), Plymouth, UK (RMS exhibiting at event)
11 – 12 Getting the most from your Confocal Course 2025, York, UK
30 June – 3 July mmc2025: Microscience Microscopy Congress 2025, Manchester, UK
4 Super-resolution Workshop 2025, Leeds, UK
9 – 10 The Advanced Materials Show, Birmingham, UK (RMS exhibiting at event)
27 – 31 Microscopy & Microanalysis, Salt Lake City, Utah, USA (RMS exhibiting at event)
31 August – 4 September
Microscopy Conference 2025, Karlsruhe, Germany (Affiliate event)
8 – 12 Flow Cytometry Course 2025, York, UK
18 – 20 Microscopy of Oxidation 12, Loughborough, UK
28 August – 5 September IMC21, Liverpool, UK
For further information on all these events, please visit our Event Calendar at www.rms.org.uk

THUNDER Imager Cell Spinning Disk
Get a clearer picture by combining the power of THUNDER technology with the CrestOptics CICERO spinning disk confocal.
> More convincing, high-quality data
> Fast, versatile confocal imaging of challenging 3D samples
> Streamlined workflows with intelligent automation

mmc2025: Microscience Microscopy Congress 2025
30 June – 3 July, Manchester, UK
Co-chairs of organising committee: Andy Brown, University of Leeds; Maddy Parsons, King’s College London
Registration is now officially OPEN for mmc2025 incorporating EMAG 2025!
One of the biggest events of its kind in Europe, mmc2025 incorporating EMAG 2025 will bring you the very best in microscopy, imaging and cytometry from across the globe. With six parallel conference sessions, a world-class
Super-resolution Workshop 2025
4 July, Leeds, UK
Scientific organisers: Aleks Ponjavic & Michelle Peckham (University of Leeds)
This meeting is designed to talk about the current challenges in developing and using superresolution microscopy. With short talks and lots
8 – 12 September, York, UK
Scientific organisers: Dan Payne (James Cook University Hospital); Derek Davies (Derek Davies Cytometry); Karen Hogg & Peter O’Toole (University of York)
This Flow Cytometry Course is aimed at both clinical applications and applications in cell biology, with the common fundamentals covered on days one and two. The course then splits into clinical applications and applications in cell biology streams, from practical demonstrations to lectures highlighting not just the applications, but best practise as well.
exhibition, workshops, satellite meetings, an international Imaging Competition and more, it is simply the place to be for anyone who uses a microscope for work, study or pleasure.
The Congresss also includes Frontiers in Bioimaging 2025 and AFM & Scanning Probe Microscopies 2025
Find out about our range of ticket options - including discount rates for RMS Members and students. As always attendance to the exhibition will be completely FREE throughout mmc2025.
Go to page 4 for a full preview.
of time for discussion, the workshop will discuss recent advances in super-resolution imaging from new developments in imaging to analysis of super-resolution data. We particularly want to encourage early career researchers to attend and contribute to the meeting. Please contact the organisers if you would like more information on how to contribute.
The course is constructed as a set of three modules. You can elect to attend the course from between two to five days, depending on the modules selected.
The modules consist of lectures interspersed with sessions in the laboratory. It is anticipated that instruments from two manufacturers will be available for practical work.
This course is open to all and is suitable for those who are relatively new to flow cytometry and who wish to expand their experience with applications and specific analysis.
A trade exhibition will be held at this event on Wednesday 10 September, if you are interested in exhibiting please contact Jess Cole.

With the HEM6000 CIQTek set a new standard in high-speed scanning electron microscopy. With its tailored design for volumetric imaging of serial sections, it delivers nanoscale resolution across large sample volumes with unmatched speed.
Perfect for large-area, high-resolution analysis, the HEM6000 empowers researchers worldwide to uncover the intricate details of biological tissues with clarity and con dence.

CIQTEK High-speed Scanning Electron Microscope Volume-EM solution helps scienti c researchers explore and reveal mysteries
A continuous ultramicrotome system to collect a large volume of biological thin sections in sequence.









The HEM6000 high-speed imaging system and AutoMap so ware to perform large-area stitching.
2D imaging and stitching capabilities

Apply algorithms to process and align the collected electron microscope images, forming a 3D dataset.

Utilise 3D so ware to visualise, image, and perform statistical analysis on the segmented data.
CIQTEK 3D cross-scale solution simpli es key steps in TEM imaging and analysis, providing customised solutions for your laboratory.
















How can microscopy add to our understanding of Medieval books?

Words by Dr Charlotte May (Impact and Engagement Officer, Manuscripts and Special Collections, University of Nottingham) and Dr Luke Norman (Knowledge Exchange Fellow, Nanoscale and Microscale Research Centre, University of Nottingham).
Light microscopy by Dr Richard Cousins (Senior Technician, Nanoscale and Microscale Research Centre, University of Nottingham).
Electron microscopy by Lorelei Robertson (Electron Microscopy Technician, Nanoscale and Microscale Research Centre, University of Nottingham).
Manuscripts and Special Collections at the University of Nottingham holds over 800 years of historical material that tells the stories of Nottinghamshire’s people, land, and estates. Among this, the Wollaton Library Collection houses rare medieval texts, once part of the private library of Wollaton Hall, a stately home just a short walk from the University’s main campus.
Caring for Medieval manuscripts is a real privilege and provides opportunities to find out more about the historic practices of writing, reading, and creating. This is where there is exciting potential for microscopy to reveal new insights into the material, not just informing archives’ conservation activities, but also telling us about the people, places and practices of textual production that have shaped the world we live in and provide inspiration for future generations.
An important work of the Wollaton Library Collection that has been re-examined recently is the Speculum Vitae, a text dating from the early 1400s which retains an early and perhaps original binding shared with another text, The Lay Folks’ Catechism, a shorter form of doctrinal instruction than the Speculum vitae. The Speculum is an illuminated copy of a text ascribed to William of Nassington who died in 1354. The title translates from Latin to English as ‘Mirror of Life’.




The text itself is a 16,000 line Middle English commentary on the Lord’s prayer and is referred to as ‘a monument of scholastic erudition and a breathtaking synthesis of moral philosophy’ in the Oxford Dictionary of National Biography entry for Nassington.
In the extensive study and survey of the Wollaton Library Collection undertaken by Ralph Hanna and Thorlac Turville-Petre (York Medieval Press, 2010), it is suggested that the Speculum vitae was brought

into the Wollaton Library Collection through the marriage of Elizabeth Fitzhugh to Henry Willoughby in 1491, evidenced by a dedication to her at the end of the volume by William Fletcher, possibly Elizabeth’s Chaplain. Hanna’s Reading Edition of the Speculum vitae (Oxford University Press, 2008) also notes that this is one of 45 currently existing manuscript versions of the text, many in libraries in Cambridge, Oxford (the Bodleian library houses 11) and the British Library among others. All copies



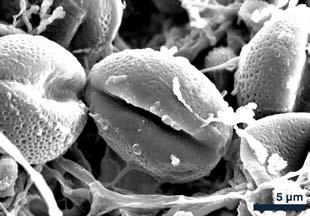
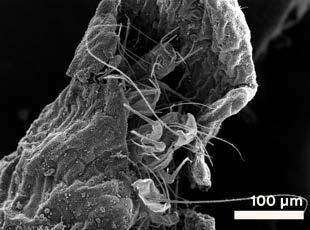
of the Speculum vitae are distinct, and the University of Nottingham’s copy has a section of deleted lines that are present elsewhere.
Scholarly work has revealed so much about the content and language of the text and its owners, readers and possible writer, but further knowledge of the material artefact can shed more light on its environment and potential uses throughout the centuries. This is where microscopy and imaging

can offer new perspectives – literally – on our understanding of this Medieval text.
During conservation work on the Speculum vitae, sweepings from the binding of the text had been kept by the professional conservator to retain as much original material as possible for future study and insight. And after a chance encounter by two Knowledge Exchange Fellows in 2022, Dr Luke Norman from the Nanoscale and Microscale Research Centre (nmRC) and Dr Charlotte May from Manuscripts and Special Collections identified

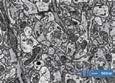
a unique collaboration that could identify a neverbefore-seen aspect of this important manuscript. The sweepings, which to the naked eye resembled little more than standard dust, were first imaged using light microscopy at the nmRC. Almost immediately, they were revealed to be plant material — a mix of shrivelled flowers and petals, dried and partially decomposed with age. Hints of green, yellow, and orange remained visible in the fragments. But what were plants doing in the binding of a 14th-century manuscript? One plausible answer lies in medieval reading habits — it was common for readers to use leaves, flowers, or other natural materials from their surroundings as bookmarks.
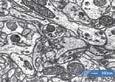
After identifying the plant material through light microscopy, scanning electron microscopy (SEM) was used for its ability to reveal finer details and structural aspects at high magnification, which would be essential for investigating the complex, microscopic features of the sweepings. SEM’s versatility allowed us to image the samples at a range of magnifications, from 60x to over 25,000x, revealing exquisite structural details. Prior to imaging, the samples were coated in gold to create a conductive surface, enabling the electron beam to interact effectively with the material.
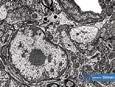
The findings could be broadly grouped into four categories: plant fibres, pollen grains, fungal spores, and — most intriguingly — creatures.
Pollen grains provided the most direct link to potential plant species. Several morphologies were observed, but the most common type was elliptical, with a net-like (reticulate) surface texture and two to three apertures. Fortunately, the Nanoscale and Microscale Research Centre (nmRC) has built an internal reference library of pollen images, enabling comparison. Based on these characteristics, the grains most closely matched those of the Ranunculus genus — specifically buttercups. Re-examining the light microscopy images of the floral material, this identification seemed consistent: the fragments showed small petals, now yellow to brown, matching the colour palette of aged buttercups. It’s easy to imagine a medieval reader sitting in a field, plucking a buttercup, and slipping it between the pages. The “creatures” discovered within the sweepings
presented a more mysterious challenge. Nestled inside a curled petal — like a tiny protective burrito — were at least three insect fragments. No complete bodies remained; all examples were partial, complicating identification. Each measured approximately 50 μm wide, slightly thinner than a human hair, with multiple segmented antennae (at least seven observed), bristled appendages, and haircovered surfaces resembling plant trichomes.
While our initial suspects included common manuscript pests like silverfish or booklice, the remains didn’t quite align. Silverfish, known for their tapered bodies, were ruled out. The fragmented head structure shared some similarities with a book louse but differed in scale and antenna count. Unfortunately, these insect fragments remain enigmatic — their precise identity lost to time, but their presence offers a tantalising hint of the book’s long and complex life.
Microscopy has offered us an extraordinary glimpse into the micro-history of this manuscript — not just the words written, but the lives and habits of those who handled it centuries ago. Trapped within its binding, these fragile traces — petals, pollen, spores, and creatures — speak of meadows, hands turning pages, and the passage of time.This project highlights how scientific imaging techniques can illuminate the invisible stories hidden in our cultural heritage, deepening our understanding of the objects that connect us to the past.
This work formed part of the nmRC’s Under the Microscope public engagement initiative powered by JEOL, designed to open up the fascinating world of microscopy to new audiences. If you’re interested in discovering more hidden worlds, we invite you to get in touch and suggest items to image — there’s so much more still waiting to be seen.
If you have a project in mind using archives and printed items, you can find out more about Manuscripts and Special Collections here: https://www.nottingham. ac.uk/manuscriptsandspecialcollections/ https://www.nottingham.ac.uk/nmrc/under-themicroscope.aspx

The Journal of Microscopy publishes top quality research articles, review articles and Hot Topic papers covering all aspects of microscopy and analysis. This includes cutting-edge technology and innovative applications in physics, chemistry, material and biological sciences.
You can read the latest Early View papers online at www.journalofmicroscopy.org
They include:
RESEARCH ARTICLE - Open access
CellPhePy: A python implementation of the CellPhe toolkit for automated cell phenotyping from microscopy time-lapse videos
Laura Wiggins, Stuart Lacy, Graeme Park, Joanne Marrison, Ben Powell, Beth Cimini, Peter O’Toole, Julie Wilson, William J. Brackenbury
We previously developed the CellPhe toolkit, an open-source R package for automated cell phenotyping from ptychography time-lapse videos. To align with the growing adoption of python-based image analysis tools and to enhance interoperability with widely used software for cell segmentation and tracking, we developed a python implementation of CellPhe, named CellPhePy. CellPhePy preserves all of the core functionality of the original toolkit, including single-cell phenotypic feature extraction, time-series analysis, feature selection and cell type classification. In addition, CellPhePy introduces significant enhancements, such as an improved method for identifying features that differentiate cell populations and extended support for multiclass classification, broadening its analytical capabilities. Notably, the CellPhePy package supports CellPose segmentation and TrackMate tracking, meaning that a
set of microscopy images are the only required input with segmentation, tracking and feature extraction fully automated for downstream analysis, without reliance on external applications. The workflow’s increased flexibility and modularity make it adaptable to different imaging modalities and fully customisable to address specific research questions. CellPhePy can be installed via PyPi or GitHub, and we also provide a CellPhePy GUI to aid user accessibility.
Subcellular localisation and identification of single atoms using quantitative scanning transmission electron microscopy
A. A. Sheader, G Vizcay-Barrena, R. A. Fleck, S. J. L. Flatters, P. D. Nellist
Heterogeneous elemental distributions drive fundamental biological processes within cells. While carbon, hydrogen, oxygen and nitrogen comprise by far the majority of living matter, concentrations and locations of more than a dozen other species must also be tightly controlled to ensure normal cell function. Oxaliplatin is a first-line and adjuvant treatment for colorectal cancer. However, pain in the body’s extremities (fingers and toes) significantly impairs clinical usage as this serious and persistent side

patient cancer care and quality of life. Annular dark-field (ADF) imaging in the scanning transmission electron microscope (STEM) provides an image with strong atom-number contrast and is sufficient to distinguish between different cell types and different organelles within the cells of the DRG. We also show that Pt may be imaged at the single atom level and be localised at very high resolution while still preserving a degree of ultrastructural context. The intrinsic image contrast generated is sufficient to identify these features without the need for heavy metal stains and other extensive processing steps which risk disturbing native platinum distributions within the tissue. We subsequently demonstrate that by considering the total elastic
scattering intensity generated by nanometre-sized Pt aggregations within the cell, the ADF STEM may be used to make a measurement of local concentration of Pt in units of atoms per cubic nanometre. We further estimate the minimum atomic number required to visualise single atoms in this setting, concluding that in similar samples it may be possible to detect species as light as sodium with atomic sensitivity.
Natalia R. Lobanova, Nadezda A. Dolzhenkova, Elena V. Boyakova, Olga A. Anastasia A. Frolova, Svetlana L. Kotova,Yuri M. Efremov, Peter S. Timashev
The morphological characterisation is crucial for analysing cell states, especially for red blood cells (RBCs), which are used in transfusions. This study compared the applicability of atomic force microscopy (AFM) and confocal optical profilometry in the accurate characterisation of the RBC morphological parameters. The imaging of RBCs thawed after cryopreservation with immediate and delayed washing steps (deglycerolisation) was performed, and the morphological data obtained with AFM and optical profilometry were compared with the clinical laboratory studies. Both techniques provided close data on the morphological parameters, but optical profilometry allowed a

faster and more convenient data acquisition. However, the membrane roughness analysis on discocytes and the submembrane cytoskeleton analysis on RBC ghosts was only possible with AFM due to its higher spatial resolution. Both techniques confirmed that delayed washing did not have negative effects on cells compared to immediate washing. Additional 3-day storage of both types of RBCs resulted in increased haemolysis. A decrease in the fraction of area occupied by pores in the submembrane cytoskeleton with the storage time was observed, possibly associated with the cytoskeleton deterioration. The studied conditions model the transportation of thawed RBCs in a cryoprotectant solution to medical facilities that have technical conditions to wash thawed RBCs and confirm its feasibility.
ORIGINAL ARTICLEOpen access
Deep learning assisted highresolution microscopy image processing for phase segmentation in functional composite materials

Ganesh Raghavendran, Bing Han, Fortune Adekogbe, Shuang Bai, Bingyu Lu,William Wu, Minghao Zhang,Ying Shirley Meng
In the domain of battery research, the processing of high-resolution microscopy images is a challenging task, as it involves dealing with complex images and requires a prior understanding of the components involved. The utilisation of deep learning methodologies for image analysis has attracted considerable interest in recent years, with multiple investigations employing such techniques for image segmentation and analysis within the realm of battery research. However, the
analysis of highresolution microscopy images for detecting phases and components in composite materials is still an underexplored area. This work proposes a novel workflow for FFT-based segmentation, periodic component detection and phase segmentation from raw high-resolution Transmission Electron Microscopy (TEM) images using a trained U-Net segmentation model. The developed model can expedite the detection of components and their phase segmentation, diminishing the temporal and cognitive demands associated with scrutinising an extensive array of TEM images, thereby mitigating the potential for human errors. This approach presents a novel and efficient image analysis approach with broad applicability beyond the battery field and holds potential for application in other related domains

characterised by phase and composition distribution, such as alloy production.
Arthur R. C. McCray, Stephanie M. Ribet, Georgios Varnavides, Colin Ophus
Electron ptychography is a powerful and versatile tool for high-resolution and dose-efficient imaging. Iterative reconstruction algorithms are powerful but also computationally expensive due to their relative complexity and the many hyperparameters that must be optimised. Gradient descent-based iterative ptychography is a popular method, but it may converge slowly when reconstructing low spatial frequencies. In this work, we present a method for accelerating a gradient descent-based iterative reconstruction algorithm by training a neural network (NN) that is applied in the reconstruction loop. The NN works in Fourier space and selectively boosts low spatial frequencies, thus enabling faster convergence in a manner similar to accelerated gradient descent algorithms.We discuss the difficulties that arise when incorporating a NN into an iterative reconstruction algorithm and show how they can be overcome with iterative training. We apply our method to simulated and experimental data of gold nanoparticles on amorphous carbon and show that we can significantly speed up ptychographic reconstruction of the nanoparticles.
Edward Felder, Jan L. Rüth, Bassam Abu-Omar, Martin Wohlwend, Paul Walther, Clarissa Read
High-pressure freezing (HPF) is an electron microscopy (EM) preparation technique with superb ultrastructural preservation. Combined with EM tomography it provides virtual EM serial sections with extraordinary spatial resolution. For HPF, cells are usually cultured on a rigid sapphire disc that provides a tight fit in the holding bracket of the HPF apparatus. Since we are using extensible elastic silicone membranes as a growth support to perform cell stretch experiments, we developed a method to clamp the stretched silicone membrane and place it instead of the sapphire disc into the HPF holding bracket. Compared to chemical fixation the HPF immobilised cells showed improved structural preservation, partly even on a molecular level. However, the outstanding quality of HPF immobilised cells on sapphire discs was not achieved. Moreover, regions with obvious freezing artefacts seemed to be more abundant in the HPF silicone membranes, probably caused by lower heat transfer rates of the silicone membrane during the HPF process.
Taken together, we have shown that HPF immobilisation can be performed on growth supports different than sapphire discs. Since even stretched membranes can be used with the new method, also other unconventional growth supports should not pose a problem.
Preparation and topographical studies of various biological specimens using alternate method to critical point drying: Scanning electron microscopy
Najeeb Ullah, Darakhshan Guhar, Saifullah Khan
Background: The major advantage of scanning electron microscope (SEM) in biological research is that one can examine the morphology and surface
features of specimens at high resolution. Specimens may differ from individual cells grown in culture to solid tissues or entire organisms measuring several centimetres in size. It literally permits an ‘in-depth’ study of such specimens with great topography due to the incredible depth of field obtainable to the operator. Current study covers practical approaches of various biological samples’ preparation and visualisation via scanning electron microscope.
Methods: Alternate method of drying was employed over standard drying method; Critical Point Drying (CPD). Natural state of the microstructures of delicate specimens could be preserved by applying recommended reagents/ fixatives. Samples were treated with 2.5% w/w glutaraldehyde and reduced 1% Osmium tetroxide as primary and secondary fixatives. Samples were then serially dehydrated by graded ethanol (EtOH) and finally treated with chemical dehydrant Hexamethyldisilazane (HMDS).
Results: Biological specimens, bacteria (Salmonella typhi and Staphylococcus aureus), bacterial crystal proteins, viruses (SARS-CoV-2), fungi (Aspergillus flavus), immune cells (monocytes) and invertebrates (Aedes aegypti), were studied and high-resolution images were captured. Detailed structural features were studied using high voltage electron beams (10–20 KV). Secondary electrons and backscattered electrons were detected to reveal detailed surface features of the specimens.
Conclusion: Chemical critical drying was found to be an economic and yet effective method with less apparent deterioration of the surface features. The advantages of using a chemical dehydrant like Hexamethyldisilazane (HMDS) include ease of use,


relative quickness, and less expense than a CPD. Same technique can be applied for different specimens with same results.

1. No submissions fees
2. No page or colour charges
3. No page limit
4. Simple online submission
5. Helpful, friendly editorial team
6. Average time from submission to first decision is less than 50 days
7. High readership figures
8. Online tracking system – authors can easily check the status of an article in production and receive emails at key stages
9. Rapid publication with Early View papers published online in advance of print, significantly shortening time from acceptance to publication
10. Free electronic offprints Journal of Microscopy App Available for iPhone and Android
Search for Journal of Microscopy on the App Store or Google play and access your personal or institutional subscription wherever you are, whenever you want.
View the Guidelines for Authors and full submission details online at:

The world’ s fastest nanoindenter Ultra High Strain Rates >10 000 s-1


The Journal of Microscopy, published by Wiley on behalf of the Royal Microscopical Society, is the world’s oldest journal dedicated to the science of microscopy. The Journal is critical to the RMS, as almost all the income it generates comes back into the society to help support our activities.
It was first published as ‘The Microscopic Journal and Structural Record’ in 1841, shortly after the society was formed in 1839. It then became ‘the Transactions of the Microscopical Society of London’, then ‘the Quarterly Journal of Microscopical Sciences’, and finally, the Journal of Microscopy in 1878.
All of these issues are available online in the backcatalogue, and are a fascinating read, including articles from some very famous microscopists such as Ernst Abbe, who published several articles in the journal, and was made an honorary fellow by the society. Interestingly, even then, part of the aim of publishing the Journal was to provide a source of
income for the society, over and above subscriptions from members, and of course, this is still true today.
The Journal of Microscopy is currently a hybrid open access journal, which means that authors can pay to have their papers published open access (or have this charge covered by a Read and Publish agreement with their institution) while other papers remain behind the paywall.
Eventually, the Journal will switch to being solely open access. This is great, scientifically, but when this happens, the amount of money that the Society will receive per article for each paper published, will drop by well over 50%. This will severely impact the income that the society receives from publishing the Journal, potentially affecting the RMS’s ability to support the wide range of activities it currently delivers.
We are not alone, in that this is a problem for all journals published by scholarly societies. The only way we can start to make up this deficit is by publishing more papers – and this is where you, our members, come in! We strongly encourage you to submit your papers to the journal, to help us increase the number of papers we publish, and we also encourage any ideas for special themed issues.
mmc2025!
The Journal of Microscopy is a great place to publish
your paper because the editors and the editorial board are expert microscopists, so your paper will be reviewed by a very knowledgeable team. The Journal should be the natural home for papers focussed on microscopy, and specifically those from members of the Society as well as microscopists generally. This was one of the drivers for starting up the Journal many years ago, to provide a place where the papers are focussed on the topic of microscopy, and it is still one of its main goals today.
RMS members can support the Journal of Microscopy in many ways including:
The Journal will have a dedicated stand at mmc2025 and the Editors would love to hear your ideas on how we can improve the Journal and any ideas you may have for special issues or review papers.
Please do consider us when you are next putting your paper together, and if you are uncertain as to whether it is a good fit or not, please do email us and ask! Please continue to support the Journal through publishing a paper with us, reading the Journal and promoting the Journal at meetings and events.
Thank you for your continuing support.
Professor Michelle Peckham
General Editor of the Journal of Microscopy
m.peckham@leeds.ac.uk



Bernardo Cesare, FRMS, FRPS, Geosciences, University of Padua, Italy.
Polarized light photomicrograph of caffeine crystals formed after drying a saturated solution on a glass slide. Crossed polarized light, width 2,7 mm. Nikon D5500 on Zeiss Axioscop 40pol.
Bernardo is Professor at the Department of Geosciences, University of Padova, Italy. He studies metamorphic rocks and the origin of granitic magmas.
He said: “I like to show the hidden beauty of small crystals under the microscope. What I like most about this image is the pattern and the unusual interference colours I succeeded in obtaining.”
SEMPREP SMART is an award-winning ion milling solution designed for highprecision SEM and EBSD sample preparation. The system can be equipped with a high-energy and a low-energy argon ion source. Ion polishing allows for the improvement and cleaning of mechanically polished SEM samples and the preparation of damage-free surfaces for EBSD analysis. Outstanding cross-sectional results and precision are achieved even in demanding and sensitive cases, such as semiconductor testing and investigation of Li-ion battery separator membranes.



High-energy ion source for multiplied throughput cross-sectioning of sample dimensions workflow control software , easy-to-use operation


Aluminium plate after Ar-ion polishing and its EBSD inverse pole figure
23rd-25th April 2025
In April 2025, the Collaborative Computational Project for Electron cryo-Microscopy (CCP-EM) held its 11th annual Spring Symposium at the East Midlands Conference Centre in Nottingham. Over 370 scientists attended in person, and 600 signed up to join the hybrid conference online via Zoom.
The symposium focused on recent developments in computational cryogenic electron microscopy (cryo-EM), new biological applications, and sample preparation techniques for cryo-EM. The first day was the Diamond Light Source, Biological CryoImaging (BCI) User Meeting for eBIC and B24. Recent updates for eBIC and B24 beamlines were shared, including new auto processing pipelines being implemented at eBIC for users and CryoSoft X-Ray Tomography highlights. The keynote speaker, Dr Stephen Muench, shared his work at the University of Leeds on the ‘Speed Blot’, which can freeze cryo-EM grids within 54 ms to preserve protein stability for single particle analysis.
One of the symposium’s highlights was the networking Ceilidh dance sponsored by Dectris. It was a novel way to meet new people and quickly say ‘Hi, I’m a PhD Student working on...’ before you were swung onto the next person in the dance and repeated the speedy introductions throughout the dancing. There was also a cryo-EM themed pub quiz organised by eBIC scientists, as part of the social programme, with questions ranging from naming the cellular organelles in an electron tomogram to guessing how many Titan Krios microscopes are on each continent. Many thanks to the sponsors, Thermo Fisher Scientific and Dectris, who supported the social programme at the symposium.

During the symposium, I presented a poster entitled “Visualising the in cellulo protein crystallisation pathway of Bacillus thuringiensis with integrated Cryogenic Soft X-Ray and Electron Microscopy” on work I’ve been doing during my PhD project. It was great to share my research, explain my ideas, and get lots of new ideas to try when I’m back in the lab. I found it helpful to also spend time with other researchers using similar techniques to discuss sample preparation approaches and data processing pipelines, especially for the work I have been doing using Cryo-Soft X-Ray Tomography at B24.
The second and third days highlighted new data processing packages and results from different cryo-EM datasets, including the benefits and drawbacks of using deep learning approaches

for protein structure determination. One of the speakers, Dr Maud Dumoux, focused on presenting new approaches for Serial FIB/SEM imaging to the community using commercially available equipment to reduce SEM imaging artefacts. Another speaker, Dr Alister Burt from Genentech, shared new developments in Warp, a cryo-electron tomography processing software and discussed how to use and implement these data processing packages in workflows at other institutions.

One of the things that struck me most at the CCPEM Spring Symposium was how each speaker and presenter really emphasised their willingness to connect, collaborate, and explore new possibilities with the whole community. It was a real testament to the supportive and welcoming environment created by the CCP-EM organisers.
Finally, I would like to thank the Royal Microscopical Society for its financial support, which enabled me to attend and present my PhD project at the CCPEM symposium.
Angharad Smith
PhD Researcher at the Rosalind Franklin Institute and the University of Leeds.
1Buckman, J., and 2, 3Krivtsov, V.
1 Institute of GeoEnergy Engineering, Centre for Environmental Scanning Electron Microscopy (CESEM), School of Energy, Geoscience, Infrastructure and Society, Heriot-Watt University, Riccarton, Edinburgh, EH14 4AS, Scotland. J.buckman@hw.ac.uk
2 Royal Botanic Garden Edinburgh, UK, EH3 5LP, Scotland,
3 812 Livingstone Tower, Department of Mathematics and Statistics, University of Strathclyde, 26 Richmond Street, Glasgow G1 1XH, Scotland.
Abstract: The acquisition of three-dimensional (3D) stereo paired images by scanning electron microscopy (SEM) typically requires the tilting of the SEM stage about the eucentric working distance, which is typically set at 10 mm working distance (WD). This is often time consuming and is only 100% applicable under the stage X and Y homed position (X=0, Y=0). Other 3D options exist that require specialised detectors (e.g., Quad backscattered electron detector), third-party software, or dynamic rocking of the beam path. Here we describe and illustrate a method for the acquisition of stereo-image pairs by SEM that works at any working distance, is easy to use, and does not require the purchase of third-party software. We illustrate the technique in reference to the examination of marine protists and freshwater testate amoebae and associated material. The technique is applicable to a large range of samples from the fields of Earth Sciences, Materials Sciences as well as other areas in the Biological Sciences.
Scanning electron microscopy (SEM) can be used for simple, non-destructive, cost and time effective stereo (3D) imaging. Such can be used to extract both qualitative and quantitative 3D surface information. A number of methods are available for SEM stereo-imaging which include:
I. The eucentric method, whereby stereo images are acquired at different angles at the eucentric working distance and used to produce coloured stereo anaglyphs (Bethal et al., 2001).
II. The use of a quad-backscattered electron (BSE) detector and specialised software to render three-dimensional images and data extraction (Slowko and Krysztov, 2013; Mignot, 2024).
III. The rocking beam method, where the beam rather than the stage is manipulated to
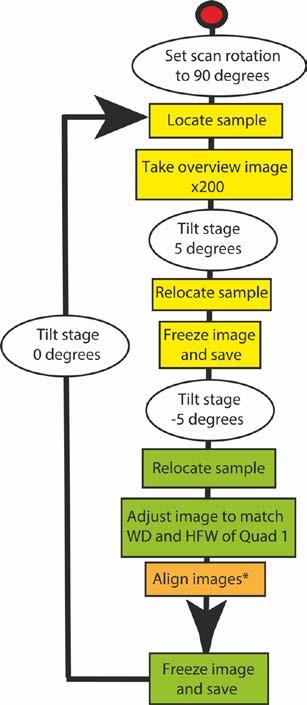
step-by-step method
acquiring SEM images for stereographic pairs, followed in the current work.Yellow boxes = quad 1 window, green = quad 2 window, and orange = quad 3 window (combination of 1 and 2). * Images aligned by overlapping of small features in the background.
produce live stereo images (e.g., standard on Tescan SEM’s, Molineux, 2015).
IV. The utilisation of software that reconstructs 3D data utilising shading interpreted from 2D SEM images (e.g., on Phenom desktop SEM).
V. 3D stereoscopic images generated from single SEM images based on depth interpretation (dark = deeper, further away, while brighter



areas are closer), which is then interpreted by the brain in effectively the same way as traditional stereoscopic anaglyphs (Sturm, 2018).
Here we introduce an additional stereoscopic method that can be utilised with a standard tilting stage but does not require imaging at the microscope eucentric position. This method therefore importantly can be performed at any working distance and is less time consuming. The method will work on any SEM model, regardless of manufacturer, and does not require the purchase of expensive third-party software.
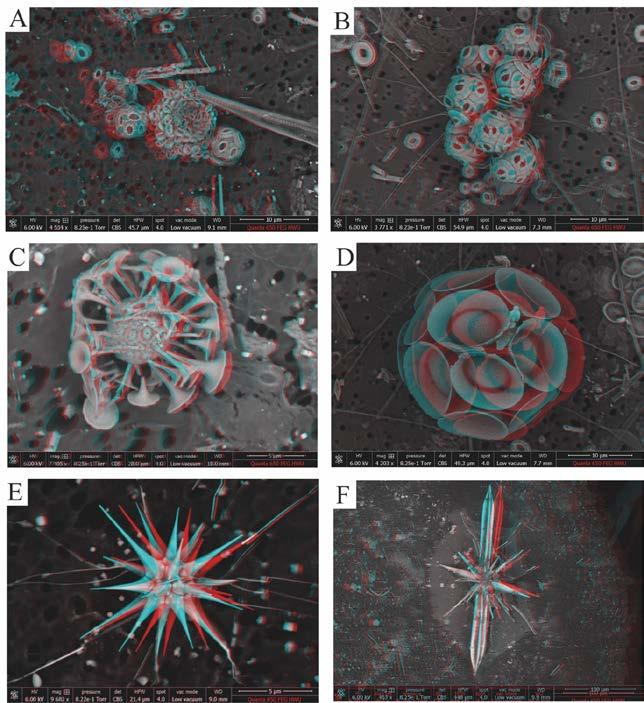
Samples were obtained from the Benguela upwelling, RRS Discovery, May 2018 as part of Controls on Oceanic Mesopelagic Carbon Storage (COMICS) project, which includes a number of protists (Coccolithophores, diatoms etc.). Other samples (testate amoebae) were collected from a number of Scottish locations, including leaf litter at HeriotWatt University (Riccarton Campus) and from an ornamental pond at the Royal Botanic Gardens Edinburgh (RBGE).
Images were taken using a Quanta 650 FEG scanning electron microscope (SEM). Chamber operated in low-vacuum mode (0.82 Torr), 5 to 10 kV with a working distance of around 8 to 10 mm. Images recorded using a backscattered (BSE) detector, with a 90˚ beam rotation.
Images were acquired utilising by stage tilting: An overview image is taken of the area that contains the subject of interest. Then the stage is tilted by 5˚. The first image is then acquired in the top
left imaging quadrant (Quad 1), saved as a jpeg, representing the left-side of the stereo pair, and the image left frozen. The stage is then tilted to -5˚ and using the overview image the subject relocated in the top right imaging quadrant (Quad 2). For the Quad 2 image the same parameters such as working distance (WD), magnification and horizontal field of view are used as those from Quad 1. The bottom left quadrant (Quad 3) was set up to display an overlay of quadrants 1 and 2 which allows the accurate alignment of both images. The image in Quad 2 is then saved as the right-side stereo image, also in jpeg format. The step-by-step method of image capture is illustrated in the form of a flowchart (Figure 1) and pictorially (Figure 2).
The freeware “Anaglyph Maker V1.08” by Sekitani (2024) was used for image processing to produce 3D anaglyphs (Figure 2), and images were saved in bmp format suitable for viewing with red-cyan stereo imaging glasses. This programme allows the saving of anaglyphs in a variety of formats, as well as adjustment of contrast and brightness of the master images, and where necessary for a degree of lateral and vertical repositioning for improved 3D image fine tuning.
Glasses for viewing 3D anaglyphs can be purchased online from a variety of shopping platforms. In addition, suitable glasses can be homemade by sourcing red- and cyan-coloured semi-transparent plastic sheets such as coloured acetate commonly used for photographic affect-lighting.
Three-dimensional anaglyphs were successfully acquired for all stereo pairs imaged (Figures 3, 4).
Using eucentric stage WD for 3D-SEM imaging can involve much trial and error and is strictly speaking only relevant at the stage central homed position (X=0, Y=0). Therefore, as the new technique does not require samples to be imaged at the eucentric
WD (typically 10 mm) it is generally simpler and more efficient to collect stereo-pairs and possible to use a range of working distances, more suited to the nature of specimens being examined. In the case of protists and other microscopic sized samples this allows for imaging at low kV closer to the polepiece, thus improving image quality due to reduced beam penetration. In addition, samples can be examined at much longer WDs with broader fields of view. In the current case a 5 mm field of view was achievable, in comparison to approximately 2 mm when working at the 10 mm eucentric WD. This opens the possibility of producing stereopairs over larger fields of view. The latter is significant with SEMs that have restricted fields of view due to the presence of physical apertures within the beam path at the polepiece.
Proper alignment of the two images acquired during collection is crucial for achieving good stereo images and anaglyphs, and consequently can be the hardest part of this new technique.Alignment is best achieved by overlapping and merging small features in the background that are out of the plane of the feature of interest. Nevertheless, the “Anaglyph Maker” software can be utilised to correct minor horizontal and vertical misalignments, to improve the quality of the produced anaglyphs. This is likely to be necessary where no suitable background features can be located to optimise image 3D quality during image acquisition.
On models of SEM where only one or two live images are available (i.e., no quad view), the protocol can be modified, to achieve similar results. The technique is also suitable for high-vacuum imaging of gold or carbon coated samples and can be used with all available detectors (i.e., backscattered electron (BSE), secondary electron (SE), and low vacuum gaseous secondary electron (GSED)).
Stereo-pairs can be used within certain constraints to produce topographic maps to quantify measurements in the X, Y and Z directions (see Pouchou et al., 2002; Andruleit et al., 2006; Mignot, 2024). Such images have also been used to construct



3D surface models of coccolithophores (Andruleit et al., 2006).
Andruleit, H., Geisen, M., and Stäger, S. (2006). Stereo-microscopy of coccolithophores – modern Interestingly, stereo-pairs also have the potential to applications for imaging and morphological analysis. be viewed using virtual reality (VR) headsets (Figure J. Nannoplankton Research, 28, 1-16. 5). The latter has great potential for educational purposes, where stereo-pairs could realistically be Bethal, E.W., Bastacky, S.J., Schwartz, K.S. |(2001). combined in a form to aid in the visualisation and Interactive stereo electron microscopy enhanced interpretation of structures at the micron and sub- with virtual reality. LBNL-48336. https://www.osti. micron scale.The use of this technique is not limited gov/servlets/purl/790407 to protists but is equally applicable to other areas
Kelly, S., El-Sobky, H., Torres-Verdin, C., and Balhoff, within the fields of Earth Science, Materials Science
M.T. (2016). Assessing the utility of FIB-SEM images as well as other areas in the Biological Sciences. for shale digital rock physics. Advances in Water
The stereo-pair capture, and imaging illustrated
Resources, 95, 302-316.
Mignot, C. (2018). Color (and 3D) for Scanning here is simpler to use than the ‘standard’ eucentric Electron Microscopy. Microscopy Today, doi:10.1017/ working distance method. In addition, it does not S1551929518000482 require the addition of costly commercial software
Mignot, C. (2024). Using a four-quadrant detector: packages or hardware and is universally applicable how to give your SEM images a 3D upgrade. to all SEM systems. Freed from the constraints Accessed 5th March 2024. https://www.digitalsurf. of WD allows images to be optimised based on com/blog/using-a-four-quadrant-detector-in-3d- the needs of the sample, giving more degrees of reconstruction/ freedom for improving image quality.
Molineux, A. (2015). The Different Imaging
Modes Offered by Tescan’s SEM. Accessed 5th Alex Poulton supplied material illustrated in figure March 2024 https://www.azom.com/article. 3 from the RRS Discovery May 2018 oceanic aspx?ArticleID=11694 project, a NERC funded consortium grant NE/ Pouchou, J-L., Boivin, D., Beauchêne, P., Besnerais, G. M020835/1 and NE/M020835/2, “Controls on Le, andVignon, F. (2002). 3D Reconstruction of Rough Oceanic Mesopelagic Carbon Storage” (COMICS). Surfaces by SEM Stereo Imaging. Mikrochimica Acta, The centre for environmental scanning electron 139, 135-144. microscopy (CESEM) is acknowledged for access to SEM facilities used for this paper.
Sekitani, T. (2024). “Anaglyph Maker” software. Downloaded February 2024. https://www.stereoeye. jp/software/index_e.html
Slowko, W., & Krysztov, M. (2013). Detector System for Three-Dimensional Imaging in the Variable Pressure/Environmental SEM. Proceedings of the IX International Conference ION 2012, Kazimierz Dolny, Poland, June 25-28, 2012. https://doi.org/10.12693/APhysPolA.123.877
Sturm, R., 2018. Stereoscopic effects from single SEM images.
Miocroscopy Today. https://doi.org/10.1017/ S1551929518000640
Jim Buckman is a Research Fellow at the Institute of GeoEnergy

Engineering, Heriot-Watt University, where he runs the scanning electron microscope facility. His research interests include the analysis of sedimentary rocks, palaeontology, trace fossils, biomineralization, testate amoebae and heliozoans.

Vladimir Krivtsov has a diverse background in natural sciences, which includes SEM analysis of microbiota and particulate materials. Also relevant to this publication are degrees in geology, and in biology with chemistry. Vladimir is currently working for Strathclyde University and is also a visiting researcher in RBGE.
You provide the text and images and we take care of the rest. It’s the ideal way to share your work with the microscopical community.
Full submission information and guidelines are available at www.infocus.org.uk.
To submit an idea or if you have any questions about the process please email the Editor (editor@infocus.org.uk)


























Join us for:
On-site demonstrations of our new high-resolution SEM with EDS and EBSD for materials science as well as SEM, volume EM, and cryo-EM for life sciences
Dedicated commercial workshops of the first advanced analytical (S)TEM for materials science and the latest developments in plasma FIB-SEM and cryo-EM for biology
Learn more at thermofisher.com/mmc


A chance to speak with our experts about how automated workflows can improve imaging scale and efficiency in materials and life sciences research
The Royal Microscopical Society would like to welcome our new members who have joined us in the last three months. We hope they enjoy a long and rewarding membership with the RMS.
Miss Anjelika Galapon
Mrs Bethan Davies-Jones
Mr Francesco De Batté
Dr Marjolaine Thomas
Miss Florence Do
Associate Professor
Ivna Kavre Piltaver
Mrs Elizabeth Ison
Darren Robinson
Dr Tushar Dandekar
Mr Muhammad Hammad
Dr Martina Kocijan
Dr Andrea Corno
Mr Claudi V. Soler
Mr Adam Agbamu
Mr Ewan Drever-Smith
If you know of anyone who might be interested in becoming a member of the Royal Microscopical Society and if you would like us to contact them, please send their details to our Membership Administrator, Debbie Hunt – debbie@rms.org.uk
Application forms are available to download at www.rms.org.uk/membership
Don't forget you can now log into the RMS website and check your membership status, renew and download receipts. If you have never logged into the RMS website, please enter the email address that is linked to your membership and then click 'forgotten password'.
If you have any queries or questions about your membership please contact Debbie Hunt debbie@rms.org.uk

DECTRIS develops and manufactures hybrid pixel detectors to spark scientific breakthroughs around the world. While CCD cameras capture and integrate electrons indirectly, DECTRIS detectors count individual electrons and X-ray photons. Our customers benefit from ultimate speed, accuracy and dynamic range when working with the most demanding TEM/STEM applications.
In addition to pushing the limits of Transmission Electron Microscopy for materials sciences, DECTRIS detectors are well suited for protein and small-molecule electron crystallography. We support researchers everywhere from our offices in Switzerland, Japan, and the United States.
Twitter: @DECTRIS_News Website: www.dectris.com/ LinkedIn: https://linkedin.com/company/ dectris-ltd-/
Name Kumbha Ravindra
Tell Us About You?
Pharmaceutical Engineering student with expertise in toxicology, drug Discovery and development.
Name Andrea Corno
Tell Us About You?
I am a Postdoctoral Research Assistant at the University of Dundee, with a background in Maths


Microscopy allows me to investigate how human cells preserve genome integrity during mitosis and assess the deleterious effects of a perturbed cell cycle. I am a strong supporter of the motto "seeing is believing"!
Why did you become a member of the RMS?
I joined the Royal Microscopy Society to connect with researchers who extensively use microscopy and image analysis to study the cell cycle. Networking within the RMS community will allow me to exchange ideas, stay updated on cutting-edge imaging techniques, and refine my microscopy skills. By interacting with specialists in the field, I aim to enhance my research and contribute to the broader microscopy community.
How do you feel being an RMS member benefits you?
Being an RMS member provides valuable opportunities for my career growth. Training courses will help me refine my microscopy skills, while conferences will keep me updated on the latest advancements in the field. Networking with experts and fellow researchers will allow me to exchange ideas, explore collaborations, and gain insights that can shape my research. At this stage of my career, these resources are invaluable for expanding my expertise and building connections within the microscopy community.
by Helena Coker, Georgina Fletcher and James Bancroft
Most microscopy facility roles require an aspect of ‘selling’, although this is often not the perception. While the stereotype of sales involves persuading customers to purchase physical products, facility staff must convince users to invest time and money in new imaging techniques. Additionally, ongoing service provision—a major aspect of sales for microscopy companies—is crucial. Therefore, training typically given to industry colleagues, such as application specialists, proves valuable for facility staff.
Top 5 tips when talking to users:
1. Credibility Statements: Practice short sentences that explain your authority over the service/microscope you’re discussing. This helps build confidence in what you’re offering.
2. Open Questions: Discover user needs by asking questions that encourage detailed explanations rather than yes/no answers.
3. Active Listening and Coaching: These skills can be developed through various courses (including online options). Even if not specifically tailored to microscopy, they provide valuable development opportunities for both work and life.
4. Find Your Sales Style: There are many sales styles which can be explored with a bit of reading. The one which might come least naturally is being a ‘Challenger’.
5. Practice Makes Perfect: You can only effectively promote your service if you believe in it and your abilities. Advocate for time to practice and develop your skills.
The majority of participants have worked in
bioimaging-aligned careers for over eight years. It was anticipated that these participants would have come across most of the workshop content during their career and have already adapted their ‘sales’ style. During the workshop, they were prompted to share their experience with more junior members of the group and play some of the stereotypical user characters in the exercises.
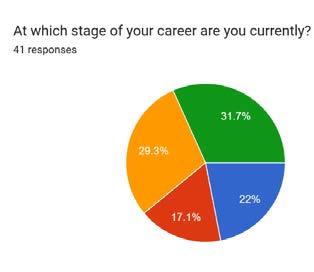

Among 41 respondents, there were 34 different job titles, which consolidated to 27 when grouping variations of ‘specialist’ or ‘facility manager’. Survey results revealed that both the perceived importance of sales skills and confidence levels increase with
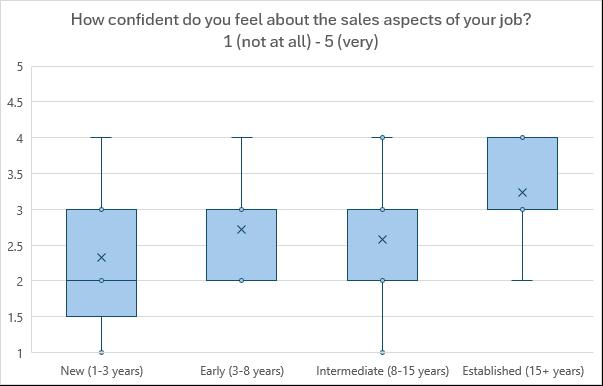
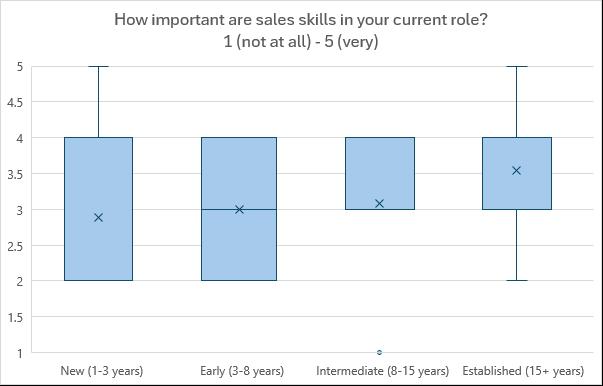
career progression (See Figures 2, 3 and 4).
Notably, no participant at any career stage reported maximum confidence (score of 5) in this aspect of their role. Common factors contributing to lack of confidence included limited experience with, or
perceived lack of authority on, specific technologies, and insufficient training. There was also feedback at this stage that sales skills aren’t needed in facility roles, as well as a lack of enjoyment and desire to use these skills impacting confidence.
Average of “On a scale of 1 to 5, how important are sales skills in your current role?”
The workshop aimed to address these concerns head-on, not only providing practical strategies for each challenge but also demonstrating why these skills are both valuable and achievable for all staff members with proper preparation and practice.
If you don’t feel fully confident about the sales aspects of your job, what factors contribute to this lack of confidence?
Responses included the following:
• Lack of confidence - including lack of confidence in a particular technology; in ability; in authority
• Lack of experience – including lack of training, being new to a role, and a miss-match between technical and biological expertise
• Not wanting to ‘overpromise’
• Being understaffed, and not having the time
• Not having many external users
• Not enjoying / wanting to do sales
The workshop combined insights from ZEISS colleagues and professional development courses. It covered six key concepts:
• Building trust through Communication
• Building confidence through upskilling and credibility statements
• Showcasing your equipment/service
• Sales styles
• Managing challenging customers/applications
• Stakeholder Engagement Beyond Users
Attendees were encouraged to implement effective communication as the cornerstone of building trust with facility users.They were introduced to coaching methodology as a key approach to help users achieve their goals through exploratory dialogue and active listening. Participants were advised that every user interaction should ideally begin by understanding objectives and collaboratively discussing potential solutions.
Trust forms the bedrock of lasting relationships in facility management and industry settings. This trust develops through conversations centered on open-ended questions that invite users to fully express their needs and concerns. Instead of limiting responses with closed questions, open questions encourage detailed explanations that reveal deeper insights. For instance, rather than asking “What magnification do you need?” a more effective approach would be “Could you describe the structures you’re hoping to observe in your images?” This invites users to share more research context and allows facility staff to provide tailored solutions.
Attendees were encouraged to explore all training and development opportunities, including those not directly linked to imaging, as well as professional registration opportunities. Similarly, managers were encouraged to support and fund such opportunities.
A key insight from confidence-building workshops introduces the concept of Credibility Statements - concise, authentic declarations of experience and expertise that naturally demonstrate professional authority. When integrated naturally into conversations, these statements help establish trust and validate recommendations. For example, statements like “I’ve successfully worked with similar samples across five years of projects” or “My experience includes optimizing imaging protocols for this specific material type” demonstrate expertise while maintaining approachability.
Attendees were informed that effective demonstrations require dedicated practice time with new techniques, as highlighted in pre-event surveys where lack of practice correlated with reduced confidence. They were advised that before presenting to an audience, they should familiarise themselves thoroughly with instruments and user samples, and prepare reliable backup examples.
Consider demonstrating as a pair with a colleague or experienced user. This approach ensures one person remains available to address questions and explain processes while the other handles technical aspects or troubleshooting.
In sustainability-focused facilities, it may feel difficult to justify spending time learning new techniques. However, your ability to attract users and promote services depends on your confidence and expertise. Advocate for adequate practice time to develop and maintain your skills.
Most attendees were surprised that ‘selling’ is a leadership style. On the Tannenbaum Schmidt continuum, selling is found where the leader relies on their influence as a manager to affect the decisions or actions of people who have less experience or expertise. You might be encouraging someone to invest in equipment you need, to use a different microscope or try a different protocol. Perhaps you want to persuade them that knowing theory is more important than just pushing buttons. Selling is therefore part of the leadership process within your facility, which has to adapt as users’ experience increases. Attendees were encouraged to often question, perhaps for each user, ‘what am I trying to achieve in this case?’.
Attendees were introduced to the five main sales styles which can be explored with some reading: Hard Worker, Challenger, Relationship Builder, Lone Wolf, and Problem Solver. They were informed that while each has its merits, the Challenger style often proves most challenging to adopt naturally.
The Challenger approach involves taking control of conversations by customising your communication to each individual. For example, when dealing with a distant or formal user, you might adopt a direct, precise tone rather than defaulting to an accommodating manner. This helps establish clear boundaries and expectations while maintaining professional relationships.
Adopting strategic flexibility in communication style can significantly improve facility utilisation and user engagement. Rather than maintaining a single approach across all interactions, tailor your style to match each user’s preferences and needs.
Attendees were informed that the Challenger approach is valuable when handling difficult users, whether they are overconfident, disengaged, or uncertain. They were taught that success
Topic-Agenda-Benefit (TAB) Model helps structure user meetings effectively:
Topic: Define the core subject and scope clearly before meeting
Agenda: Plan both discussion points and desired meeting outcomes
Benefit: Identify specific value you’ll deliver to the user, building confidence to manage expectations
requires active listening, open questions, and clear communication - and learned that facility staff may need to minimise technical language while users provide lay explanations of their work.
Key strategies include:
• Using the Topic-Agenda-Benefit model to set clear expectations (see Figure 6)
• Understanding different personality styles and their dynamics
• Recognising that tension and silence can be productive tools
Attendees were asked to consider what to do when faced with resistance like “this is how my group
does it”. How does this response change when addressing a PI rather than a user?
Attendees were informed that while facilities often focus on user engagement, effective outreach to other stakeholders is crucial. They were introduced to various stakeholder needs including:
• PIs require guidance on grant funding requirements
• Departments need clear facility value propositions
• Management expects justified budgets and expenditures



Attendees were asked to consider what to do when faced with resistance like “this is how my group does it”. They were encouraged to reflect on how this response changes when addressing a PI rather than a user.
The workshop received an average rating of 4.38/5, with participants valuing open questioning techniques, credibility statements, and broad skill applicability. While logistical improvements were suggested, the content received positive feedback.
User prospecting emerged as a key area for future development, as current focus remains on engaging existing users. Additionally, participants expressed interest in standardised procedures for user engagement, though implementation faces challenges due to varied operational models and charging structures.
Connecting pre-course experience levels with post-course feedback would enhance
future training effectiveness by better understanding impact across career stages. Future iterations could expand to allow more time for prospecting discussions and interactive elements, with flexible delivery options including online breakout rooms or in-person venues. Industry partnerships are providing insights to enhance the curriculum, though questions remain about creating content suitable for both facility and industry staff.
BioImagingUK is organic open network of UK-based Imaging Scientists who use, administer or develop imaging solutions for life science. Co-funded by the RMS, UKRI-BBSRC, UKRI-MRC and UKRIEPSRC, the network has four major aims around national strategy, careers, training and community engagement. To become a member, simply sign-up to the mailing list here mailto:georgina@rms.org. uk and to find out more check out our webpage You can also contact the network’s Project Officer, Georgina Fletcher at Georgina@rms.org.uk

Dear Readers,
I hope you are well and enjoying the year so far –wherever you are in the world.
It probably won’t surprise you to learn that my thoughts are very much turned towards the Society’s flagship event, mmc2025 incorporating EMAG 2025, which is now almost upon us!
The lead-up to mmc is always an exciting time and I’m convinced we’re in for a really great show this year. We’ve received more than 400 talk and poster abstract submissions in total, which is just amazing, and means we can expect one of the richest and most diverse conference programmes ever. Likewise, the support from our industry colleagues has been immense, with almost 100 per cent of all available exhibition space sold out as I write. I can’t wait to see that bustling exhibition hall in full flow, with some of the leading companies in microscopy showcasing their latest products and technology.
Looking at the numbers so far, it just shows how important this event is to the international microscopy community. It is one of the few meetings where life and physical sciences, electron and light microscopists, and our other related section and focus areas can all share the same space, enabling an exciting cross-fertilisation of ideas and information. For me, that is its greatest strength.

It’s not all about mmc however. In June I’ll be helping out as a teacher on the RMS Light Microscopy Summer School here in York – something I look forward to every year.
It’s always great to be in a room full of people who share a passion for learning about microscopy. This event follows a highly successful series of RMS courses to have taken place in recent months, including our Electron Microscopy Spring School, Virtual Facility Management Training Course and Virtual Flow Data
Analysis Course. We also have our well established
Dr Peter O'Toole.
‘Getting the Most From Your Confocal’ course immediately following the LM Summer School (see Events Calendar on page 22). I know how highly these courses are valued by attendees, from beginners to more experienced users, and it’s always a privilege to be involved.
One thing that isn’t always visible to our members is the work undertaken by all the RMS committees to help ensure the Society continues to evolve as an organisation and stay at the forefront of microscopy. To this end, we recently launched an online Membership Survey (still live as I write!) which we hope as many members as possible have been able to complete. We look forward to feeding back the results in the near future, and using this valuable information to make improvements across the Society’s full range of activities. From the RMS Council and Executive Committee, through to our Science Sections and Focussed Interest Groups, huge efforts are being made to maximise the support the RMS can provide for microscopists of all descriptions and at all stages of their career; it makes me very excited about the future.
One important way in which members can support their Society is by submitting papers to our flagship publication, the Journal of Microscopy (see page 38). The JoM is making great strides with a flurry of recent special issues and a growing number of articles published under ‘open access’ agreementswhich cover the publication charge, enabling authors to submit their papers for free. An mmc2025 special issue of the Journal is in the pipeline, so if you’re presenting a talk or a poster at the conference, we strongly encourage you to submit a paper!
Before signing off for this issue, I would like to say some final words of appreciation for all the RMS volunteers and staff who do so much work behind the scenes to ensure the smooth running of our activities – and the best possible experiences and outcomes for the microscopy community. None of this would be possible without your efforts, and my sincere thanks go to you all.
I’ll see you at mmc2025!
Peter O’Toole, RMS President











More than 400 abstracts have now been submitted for mmc2025 (incorporating EMAG 2025) following the closure of ‘late-breaking’ submissions.
An additional 25 abstracts were submitted for the ‘late-breaking’ conference session, due to take place on Tuesday, 1 July. The session will provide an opportunity for researchers to present the most exciting recent developments in the field.
With 388 poster and oral abstracts already having been received, it brings the total number of submissions to 413. An additional 66 invited speakers will also be presenting at what promises to be a fully loaded conference, covering all aspects
of microscopy, imaging and flow cytometry.
RMS Chief Executive Sali Davis said: “It really has been a brilliant response from the microscopy community, and we would like to thank everyone for submitting their abstracts and ensuring that mmc2025 will provide delegates with a fantastic scientific programme.
“It’s particularly pleasing to have received so many abstracts this year, as we are encouraging all our presenters to submit papers to the Journal of Microscopy - linking our flagship publication more closely with our flagship event.”
View the Scientific Programme


Second highest number of entries on record leaves difficult task for judges!
An impressive 234 entries have been submitted to the 2025 RMS Scientific Imaging Competition following a great response from the microscopy community.
The submissions were received across six different categories covering all forms of microscopy and imaging within the sciences. It’s the second highest number of submissions on record - just falling two short of the 236 received in 2017.
A team of RMS judges will now decide on a shortlist of the best entries, which will be displayed in a gallery at mmc2025 in Manchester - where the final winners will also be announced.
The RMS Imaging Competition takes place every two years, providing a great outlet for those who can capture the breath-taking and engaging beauty of the microscopical world. It now also includes a ‘short video’ category, showcasing the very best moving images captured under the microscope.
See the winners from previous competitions
We are pleased to announce the publication of our special Microscopy Techniques issue
The May 2025 issue of the Journal of Microscopy is a special issue focussing on Microscopy Techniques and it was guest edited by Stefan Linder, Universitätsklinikum HamburgEppendorf, Germany.
The cover image, from the paper by Svetllana Kallogjerovic et al, shows a vibratome section of Dorsal Root Ganglia from a tumor bearing mouse injected with Cholera Toxin Subunit B (grey) and labeled with NF200 (green), CGRP (blue) and IB4 (red).
The issue features the following papers:
Introduction to special issue on ‘Microscopy Techniques’. Stefan Linder
Characterisation and correction of polarisation effects in fluorescently labelled fibres. Nandini Aggarwal et al – OPEN ACCESS
Quantifying superimposed protein flow dynamics in live cells using spatial filtering and spatiotemporal image correlation spectroscopy
Unravelling molecular dynamics in living cells: Fluorescent protein biosensors for cell biology. Colline Sanchez et al
Rodrigo A. Migueles-Ramírez et al – OPEN ACCESS
MINFLUX nanoscopy: Visualising biological matter at the nanoscale level. Alexander Carsten et al –OPEN ACCESS
Retrograde tracing of breast cancer-associated sensory neurons. Svetllana Kallogjerovic et al – OPEN ACCESS
The issue is available to view here: https:// onlinelibrary.wiley.com/toc/13652818/2025/298/2
Society lends instrument for episode on ‘The secret genius of modern life’
The RMS is very pleased to have provided assistance for BBC science series ‘The secret genius of modern life’ in the form of a microscope which featured in a recent episode.
The instrument, which is usually loaned out to schools as part of the Society’s Microscope Activity Kit (MAK) scheme, appears during a section of the programme in which presenter Hannah Fry examines fridge installation.
The episode, which aired on 9 April and is currently available on BBC iPlayer,
takes a deep dive into the workings of modern refrigerators and also examines the history of the appliance, including the sequence of events that ultimately led to its invention.
The secret genius of modern life. Series 3: 2. Fridge



Revolutionary wide area pixelated sensor technology for EBSD
eWARP is a pioneering EBSD (Electron BackScatter Diffraction) detector powered by a Brukerengineered camera that combines direct electron detection and CMOS technologies to boost signal acquisition and processing performance, elevating the EBSD technique to the next level.
Direct Electron Detection (DED) for maximized signal efficiency
Ultrafast EBSD map acquisition at low accelerating voltages and beam currents
Patented on-chip binning enabling ForeScatter Electron (FSE) imaging at up to 350,000 points/second
For more information please visit www.bruker.com/ewarp
The RMS has signed a ‘Memorandum of Understanding’ with the Australian Microscopy and Microanalysis Society (AMMS) to seek new opportunities for collaborative working.
The agreement sets out ways in which the two societies should seek to work together towards their shared purpose of furthering the science of microscopy.
Examples include holding regular meetings to share information about future activities, helping to promote each other’s events, and opportunities to collaborate on outreach and education initiatives.
RMS Chief Executive Sali Davis said: “As an international, outward-looking society, the RMS has always sought to reach out to the global microscopy community and forge closer links with other organisations that share our aims.
“We’re really looking forward to exploring new opportunities for joint-working with our colleagues at the AMMS. This agreement is symbolic of our desire to work collaboratively, but it also helps both societies by setting out some practical steps we can take to support each other’s activities and bring our communities closer together.”
AMMS President Louise Cole said: “I, and our AMMS

Executive members, are delighted to be working more closely with the Royal Microscopical Society in the UK to cross-promote our activities.We share similar objectives and passions to support and connect our members, as well as showcase new and emerging microscope technologies and applications at our conferences and meetings.”
“We recently held our biennial meeting ACMM28 together with the Asia-Pacific Microscopy Congress (APMC13) in Brisbane, and at our AMMS booth displayed the IMC21 banner outlining the RMShosted event that will take place in Liverpool, 28 Aug - 5 Sep 2026. There was certainly interest among attendees who are now planning on attending IMC21, myself included!”

We are very pleased to welcome Steven Matthews and Stuart King as our latest additions to the RMS Diploma programme.
Steven (University of Aukland, New Zealand) and Stuart (University of Leeds, UK) join a growing number of microscopists studying for the Society’s specially designed qualification, which is intended to be of a similar standard to a Masters degree and is only available to RMS members.
The RMS Diploma is a flexible, portfolio-based course of study that is designed by the candidate with the assistance of their line-manager, and with input from existing Fellows of the Society.
The scheme has been steadily growing in recent years, with 12 candidates currently in the process of completing their projects.
Stuart and Steven’s projects are as follows:
Steven Matthews, University of Auckland, New Zealand:
Thermally sprayed carbide composite microstructural characterisation by SEM, EDS and EBSD
The wear resistance of carbide composite thermally sprayed coatings is critically dependent

upon their microstructure, which is typically analysed via scanning electron microscopy using backscatter electron imaging, energy dispersive spectroscopy and electron backscatter diffraction. However, little has been presented on the effect of imaging conditions on the depth of BSE or EDS analysis, which is particularly challenging given the dramatic variation in material properties. This work aims to optimise sample preparation for SEM and quantify the depth of BSE/EDS analysis as a function of imaging conditions across composite phase interfaces from both the surface and the crosssection using FIB-capable SEM.

Stuart King, University of Leeds, UK:
Optimisation of the preparation and analysis of Cementitious materials for Scanning Electron Microscopy
Scanning electron microscopy plays a major part in investigations of the microstructure of cementitious materials. The preparation, imaging and analysis of materials can be challenging due to the differences between the many types of cementitious materials. With the ongoing evolution in understanding these materials, the purpose of the study is to look at ways of developing the processes to optimize procedures in research.
Schools across South-East Asia are set to receive the gift of microscopy thanks to an epic fundraising effort by RMS Ambassador Michael Harrison. Michael, who is co-founder of Asian-based consulting and research organisation, The Eolais Institute, embarked on a two-week, charity bike ride across Thailand to raise awareness of microscopy and bring science – particularly marine biologycloser to children at rural schools in the region.

His journey began in Bangkok, before heading northwards through the ancient kingdom of Ayutthaya, and then on to the cities of Sing Buri and Sukhothai. Stretching further north, Michael’s gruelling ride took him via Thailand’s highest mountain, Doi Inthanon, after which he descended
into the historic city of Chiang Mai. After setting his course for the Isaan region in the northeast of the country, Michael took a coastal route back south, through the bustling town of Lopburi, before reaching his final destination, Pattaya.
Along the way, Michael visited universities and schools, helping to promote science and microscopy on behalf of the RMS. In Pattaya, he also visited an orphanage, where he made a substantial donation of essential supplies. Michael said: “My goal was simple: to inspire young minds by providing microscopes to rural schools, giving them the opportunity to peer into a world they had never seen before - and hopefully ignite an interest in science. The excitement in those classrooms reaffirmed my belief in the power of curiosity - the very force that had driven me to undertake this journey in the first place.”
With the funds raised from his ride, Michael will be purchasing microscopes to distribute to underprivileged schools across Southeast Asia. He added: “My hope is that, through these small instruments, I can open doors to a world of discovery for children who might otherwise never have had the chance.”

4D-SPED is a well-established technique in TEM, known for its ability to map material orientations & phase mapping, and strain analysis with nanometer-scale resolution. By leveraging precession, which reduces the dynamical effects of electron scattering, the quality and accuracy of these maps are significantly improved.
Two novel software, extend the applications of 4D-SPED to two cutting-edge material characterizations: mapping electric fields (EF) to investigate charge distribution in electronic, optical, and electromechanical materials, and mapping the electron pair distribution function (ePDF), a powerful tool for studying disordered, amorphous, and nanocrystalline systems. Applications include semiconductors, amorphous solids, glass, and polymers.
The uniformization of the transmitted beam intensity by the precession helps to disentangle the structure contribution from the physical properties, resulting in more accurate analysis. As a case study, the apparent EF at an interface of STO doped with 1% Ca and AlO is shown with and without the use of beam precession by employing a fully automated software through a COM algorithm




The ePDF is computed for every pixel of the electron diffraction data series using the novel ePDF Mapping data analysis software. The computed ePDFs were used to determine the Pearson correlation function, enabling a rapid assessment of similarities and differences across the scanned area. The current study of a semiconductor material, reveals that the scanned area primarily consists of two distinct layers: amorphous Si₃N₄ and SiO₂ layer
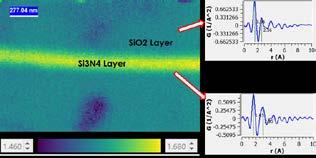


The RMS is delighted to have agreed a ‘Collaboration Framework’ with the Global BioImaging network to seek new opportunities for joint-working.
The framework sets out ways in which the two organisations aim to work more closely to advance Global Imaging Science and Community Engagement.
Examples include helping to promote each other’s events, combining communication efforts to increase the reach of mutually supported initiatives, and sharing knowledge, resources and best practices to strengthen the global imaging community.
RMS Chief Executive Sali Davis said: “We’re really looking forward to exploring new opportunities for joint-working with our colleagues at Global BioImaging.
“Wherever opportunities present themselves, we’re really keen to work more closely with partner organisations which share our commitment to advancing imaging science and building a global community of imaging professionals.”
Global BioImaging is an international network
of imaging infrastructures and communities, established in 2016. It currently brings together 13 partners representing imaging communities from 64 countries across Africa, Asia, Australia, Europe, Latin America, and North America. The network is dedicated to addressing the challenges faced by the global imaging community by promoting international cooperation and proposing solutions that advance imaging technologies and research infrastructures.
GBI Coordinator Antje Keppler said: “At Global BioImaging, we believe that collaboration is the cornerstone of scientific progress. We’re genuinely excited about this first-of-its-kind agreement with the RMS, which underscores our shared values and contributes to a more interconnected and impactful global imaging community.”
Strategic Alliances Manager Yara Reis added: “Together, we are advancing international cooperation that not only empowers today’s imaging scientists but also drives transformative breakthroughs with far-reaching benefits for all.”

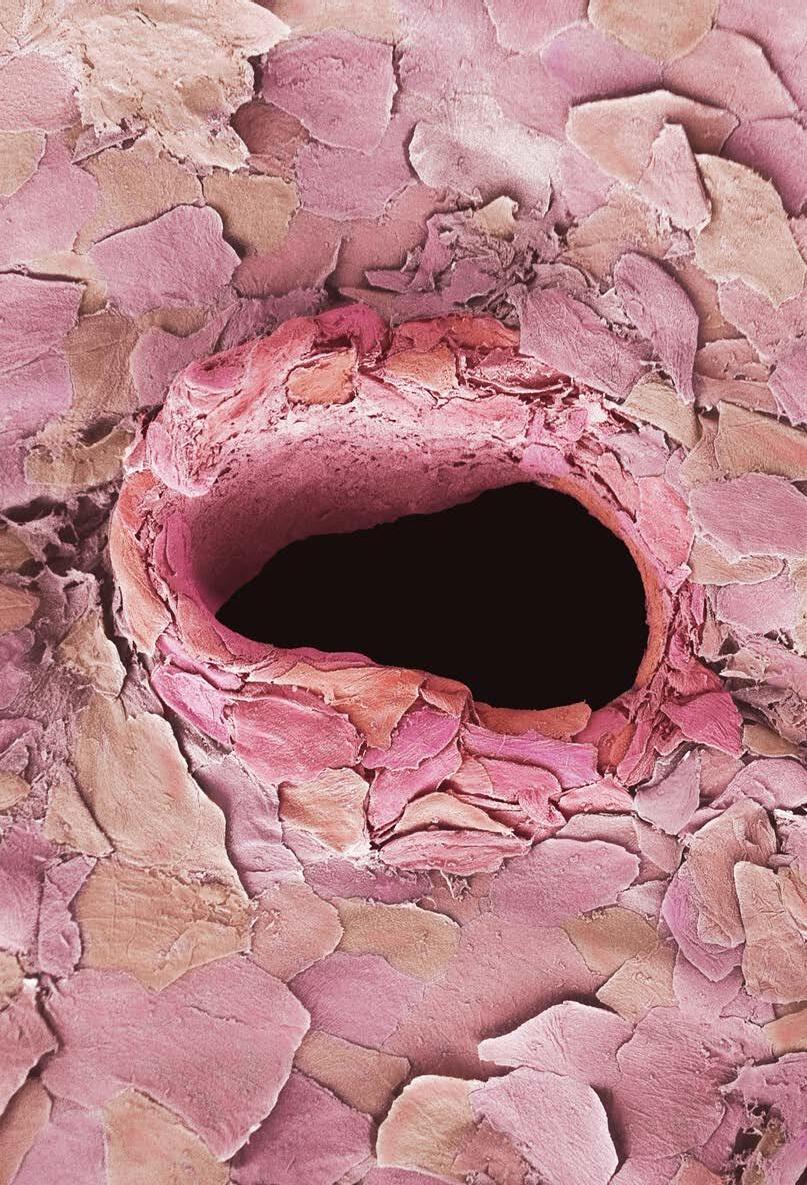
Arecent front cover image (infocus Magazine Issue 72, December 2023) got our editorial team thinking about the subject of tattoos.
Titled ‘For the Love of Art’, the image by Anne Weston (Francis Crick Institute) was also shortlisted in the 2023 RMS Scientific Imaging Competition (Electron Microscopy, Life Sciences category). It’s a wonderfully stark micrograph that shows, in no uncertain terms, what’s really going on at the business end of the tattooist’s needle!
For millennia, human beings have undoubtedly chosen to undergo this procedure for the love of art – or at least, perhaps, their desire to project cultural affiliation through symbolic expression. The love of body art remains strong in 2025, and the stories and motivations behind tattoos are presumably as many and various today as they have always been. But could the love of microscopy really be the driving force for a trip to the tattoo parlour?
As it happens, our very own Scientific Editor (and ink enthusiast) Dr Leandro Lemgruber is the proud owner of two microscopy-themed tattoos, which he was kind enough to share with infocus. (overleaf; readers will be reassured to know that neither of these images has been etched on a body part that would normally be considered ‘intimate’!)
Leandro says: “I have always loved tattoos and I have a large number of them. In fact – I’m booked in for two further tattoos later this year!
“I have used microscopes so much throughout my career that they have become a big part of my life. It just seemed natural for me to recognise that in tattoo form, and pay tribute to these amazing instruments.
“The first one, which I got done in 2018, was to reflect my interest in both light microscopy and electron microscopy. The tattoo shows the paths taken by both light and electrons in the two forms of microscopy, combining to create a unique image. I suppose it’s a little bit like the famous image of light going through a prism from the Pink Floyd album cover, Dark Side of the Moon
“The other one is of an old, antique microscope which I got done in 2020. I think the old, brass-made microscopes always look nice and fancy, and it’s a

This image by Anne Weston (Francis Crick Institute) was shortlisted in the Electron Microscopy Life Sciences category in our recent Imaging Competition. Skin tattooing has been practised for thousands of years. To make tattooing permanent, needles are used to punch through the top layer (epidermis) of the skin into the next layer (dermis). This image shows human skin cells, some damaged, from the epidermis surrounding a hole created by a tattoo needle. Magnification: x305 (Additional credit: Science Photo Library).

The microscopical pathways of both light and electrons were intertwined for artistic effect on the back

privilege to use them if you ever get the chance. it just seemed the right thing to do!”
Leandro added: “There might be some more microscopy-themed tattoos in the pipeline. I’m not sure yet, so we’ll have to wait and see.”
With the gauntlet now firmly thrown down by Leandro, we’re bound to wonder if any of our readers have been similarly inspired by microscopy in their choice of body art?
Do you know someone who has a microscopythemed tattoo they might be willing to share with us? If not, this will prove to be a one-off feature –which would surely be a shame!
Contact: editor@infocus.org.uk

Annelies van der Laan, Leiden University Medical Center (LUMC)
Microscopic images and data are often essential for answering and supporting complex research questions. In most institutes, expensive equipment, such as advanced microscope systems, are sited within a core facility, making them accessible to all researchers. To run such a core facility and maintain high standards, many factors need to be controlled: regulation of infrastructure and administration, organisation of multi-modal training and courses, clear communication with users, support for systematic data management and analysis, and equally important, effective management of facility finances. Lots can be learned if we discuss these subjects with staff from other core facilities, on both a national and international level. A highly productive way to achieve this is through job-shadowing, a practice that allows someone to visit and ‘shadow’ staff members performing similar tasks. Face-to-face knowledge exchange is highly effective, as it offers firsthand insights into operational routines. By visiting the location one can actually experience the facility’s workflow and the gained information is highly useful. In some cases, it can be directly applied to reconsider and improve procedures at one’s own facility.
Like many other microscopy core facilities worldwide, the Leiden University Medical Center (LUMC) facility provides access to advanced light and electron microscopy systems, as well as highcontent analysers and slide scanners. Approximately 400 researchers, PhD students, technicians, and students at various academic levels rely on the facility to image a wide variety of samples, from live cells to organoids, organ-on-a-chip models, and entire organisms, often related to clinical research. These images contribute to our understanding of
cell biology and its pathology, with the ultimate goal to improve healthcare. Although we strive to offer the best service, there is always room for improvement. The idea of visiting another core facility to gain new insights led us to make a jobshadowing visit a reality.
When seeking a location for a job-shadowing opportunity, I knew there were a lot of excellent microscopy facilities to choose from. The UK has many, and they are well-organised and supported by the RMS. As such, the university of York was on my short-list. Peter O’Toole, RMS President and head

of the Bioscience Technology Facility of the York University, is a well-known microscopy expert and core facility manager. Peter allowed me the chance to come and visit his excellent microscopy facility. I was very excited because the York University Imaging & Cytometry facility has a well-established track record of operational management. Last November Peter and his staff kindly hosted me for a threeday visit, during which we highlighted all desired key topics, exchanged ideas and I gained valuable knowledge. The visit exceeded my expectations, thanks to the openness and generosity of Peter and his team in sharing their experience. In this article I would like to reflect and highlight the significance of job-shadowing from my perspective.
One standout feature of the York University infrastructure is the close physical proximity of their technical facilities, which fosters seamless collaboration. The six sections - Imaging and Cytometry, Genomics, Metabolomics and
Proteomics, Molecular Interactions, Protein Production and Data Science - are housed in a single building and have regular meetings together. This encourages optimal collaboration and provides a transparent workflow for researchers in need of advanced technologies.While our LUMC facilities are structured differently, after my visit, we recognised the importance of cross-facility interaction. We are now investigating better communication strategies and will hold more frequent meetings to overcome the spatial distance. We already have implemented combined booking and charging software for some of the facilities, this uniformity is beneficial for both users and facility staff.
At the York Imaging and Cytometry facility they have a well-documented training workflow, which is another aspect we might adopt at our facility. Clear training protocols ensure consistency and allow for smoother knowledge transfer to new

users. One innovative practice we use in our own workflow, which could be applicable to other facilities, is the use of video tutorials. New users are required to watch videos about handling the microscope and software before attending the training session. This idea emerged during the COVID-19 restrictions but is still very useful today. These videos have proven to be an effective tool, helping users prepare for training sessions and grasp the basics more quickly. While recording the videos and editing the video material does take some effort and is time-consuming, they are reusable for the lifetime of the system and have saved valuable time in recurrent training sessions.
During my visit, I also gained insights into York’s facility booking rules and financial management, which was particularly useful because we are in
the process of implementing user contributions for microscope usage. Starting this year the research facilities within the LUMC need to make progress in increasing their level of self-sufficiency, something the technical facilities in York already implemented from the start. As such, there was a lot be learned from Peter and his colleague Joanne Marrison because of their long-term experience on the subject. This was incredibly helpful as we work towards implementing similar systems. I highly recommend their publication (O’Toole & Marrison (2024)), for further reading on this important topic.
By maintaining high-quality microscopes and providing the latest imaging technologies, we, as an imaging facility, make valuable contributions to research projects. While this is sometimes recognised, it is not always formally acknowledged and often underappreciated in scientific publications.
To reward the facility’s effort, which results in beautiful and valuable microscopy images and data, an acknowledgement in published research would be appreciated. Additionally, if a staff member significantly contributes to the content of an article, co-authorship should be considered. To address the visibility and recognition of high-standard microscopy facilities, we intend, together with the Dutch microscopy society (NVvM), to adopt the Imaging Facility Guidelines for Acknowledgement, as published on the RMS website (Stephen et al.).
In addition, as discussed in a publication by Marqués et al., imaging methods are highly underreported in publications. Scientific papers including microscopy images, should mention full details on how the images were obtained, supported by input from imaging facility staff to ensure the accuracy and completeness of these sections.
Another key discussion point during my visit, which could be of great value for the Dutch facility community, is the career prospects for technical staff. In many Dutch institutes there is no specific career pathway for technical staff. For instance, a research technician may be responsible for providing training on microscopy systems and managing facility administration but may no longer be involved in hands-on research.We need to create newly defined career pathways that align with the evolving responsibilities of these professionals. The Research Technical Professional Career Pathway has been well defined by Prior (2023), professor in molecular oncology at the Liverpool University. It provides a helpful framework to open up discussion and aim for better regulation of career paths for technical staff.
Reflecting on the insights I gained during my three-
day job-shadowing experience, it’s clear that jobshadowing is an incredibly effective way to review and upgrade both expertise and operational knowledge. It can elevate your core facility in terms of efficiency, and inspire new ideas for future-proofing operations. With the ongoing development in cell research technologies, it’s essential that we continue to innovate and adapt our equipment and procedures. Futuristic ideas become reality with the rapidly evolving microscope technologies that are being discovered.This visit has not only led to new insights to further optimise the facilities workflow, but also encouraged a broader national collaboration on key issues associated with running a core facility, such as acknowledgement guidelines and career pathways for technical staff.A strong, open-minded community will strengthen our position in the global research environment. I am grateful for the opportunity that allowed me to visit the Imaging & Cytometry facility directed by Peter O’Toole, and look forward to implementing these valuable insights to improve our microscopy facility and contribute to the status of technical facilities nationwide.
Marqués G, Pengo T, Sanders MA., 2020 Aug 11, Imaging methods are vastly underreported in biomedical research., Elife., 9:e55133.
O’Toole PJ, Marrison JL., 2024 Jun, A perspective into full cost recovery within a core facility/shared resource lab., J Microsc., 294(3):372-379.
Prior I, Research technical professional career pathway, 2023 Feb, https://www.liverpool. ac.uk/research/research-environment/enablingenvironment/research-technical-pathway/, 2025 Jan
Stephen N, EM-UK community, BioImagingUK focused interest group, Imaging facility publication guidelines, year of publication unknown, https:// www.rms.org.uk/community/networks-affiliates/ bioimaginguk-network/resources/im aging-fa cilitypublication-guidelines.html, 2025 Jan
Annelies van der Laan started working at the Leiden University Medical Center (LUMC) in 2001 as a research technician, studying the movement of mRNA in live Drosophila embryonic muscle fibres using photobleaching techniques such as Fluorescence Recovery After Photobleaching (FRAP) and Fluorescence Loss In Photobleaching (FLIP). In 2006, when the laboratory and its microscopes were relocated to a new building closer to the Medical Center, Annelies played a key role in establishing a core facility for light microscopy.The facility allows the use of advanced microscopy techniques for all LUMC researchers and external collaborators. As a microscope specialist, she provides training sessions, offers advice on staining and sample preparation, and manages communication and administration tasks. Today, over 400 researchers annually use the microscope systems in the core facility, and Annelies takes great pride in contributing to their important work.


Axially Swept Light Sheet Microscope for Imaging Cleared Specimens
Thin 2µm light sheet across a 10mm field of view for isotropic resolution
Custom-designed excitation objectives for a high resolution, aberration-free light sheet in 1.33 to 1.58 RI
Multiple sample chambers for specimens of all sizes up to 100 x 30 x 30mm

High Resolution Live Cell Imaging of Plant Biology
Turn key imaging system designed for study of dynamic plant biological processes in their native growth environment
Built upon the powerful Marianas inverted microscope platform supporting a wide range of imaging modalities including spinning disk confocal & super-resolution
SlideBook software enables automatic tracking and imaging of multiple growing root tips
SlideBook Microscopy Software • Widefield • Spinning Disk Confocal • LightSheet • Multiphoton • TIRF Optogenetics • Photomanipulation • Photoablation • Computer Generated Holography • Adaptive Optics
Intelligent Imaging Innovations USA | UK | DE | FR
www.intelligent-imaging.com sales@intelligent-imaging.com
It was such an honour to be awarded the RMS prize at the 2024 actin conference, particularly as the microscopy is always of such high quality. I would like to thank Harry Mellor for the work he has put in to organising the conference over the last 18 years. I was sorry to hear that he won’t be organising the event in the future, but I think it’s fair to say he’s earned a break! I hope that the community will be able to maintain an equivalent event in the future.
At the 2024 conference I presented work on an actin structure that has interested me for a long time: actin waves. Actin waves are diverse in form and function and are involved in a range of processes from the regulation of cell shape to the control of migration and macropinocytosis. This is important as the presence of actin waves has been shown to correlate with the metastatic potential of cancer cells. These structures are observed in the cortex of cells across a wide range of organisms and cell types and the patterns they generate are incredibly beautiful. It feels a bit like cheating, winning a microscopy prize when you have such exciting samples! It also helped to have access to the wonderful Wolfson Light Microscopy Facility here at Sheffield.
In (a) I present a montage taken from a widefield fluorescence movie of a mammalian cell, expressing LifeAct, responding to the addition of a drug (at t=0) that triggers the formation of actin waves. I include a kymograph (b) which shows the fluorescence overtime along the yellow line marked in (a) with the addition of the drug marked by the white
arrowhead. I also include an image (c) coloured by time. Together I hope this demonstrates the dynamic nature of these structures.


/ 96 / 384 / 1536 well formats.
/ For sophisticated microscopic applications and high content screening.
/ Highly transparent cycloolefin film bottom.
/ Adherent TC surface treatment.
Well design: 96-well plate

Well design: 384-well plate
/ 96 well format.
/ Outstanding image quality and resolution.
/ Cycloolefin-based frame with 0.17 mm glass bottom.
/ Recessed well bottom facilitating the use of lenses with low working distance and high aperture.
/ Ditch at the perimeter can be filled with liquid to minimise edge effects and evaporation.
/ Compatible with advanced automated microscopic systems.

Well design: 1536-well plate

Well design: 96-well plate

Metastasis—the spread of cancer cells—is a leading cause of cancer-related deaths. Migrastatics represent a groundbreaking class of drugs designed to inhibit cancer cell movement, effectively preventing metastasis without harming healthy cells. Unlike traditional treatments that target tumour growth, migrastatics focus on halting the invasive behaviour of cancer cells.
Pioneered by Dr. Jan Brábek, the concept of migrastatics has gained traction in the scientific community. Utilising advanced tools like Telight’s Q-Phase microscope, researchers can observe
and analyse cell migration in real-time, providing invaluable insights into the efficacy of these novel treatments.
Here is a full interview on YouTube, to better understand what migrastatics are.
This innovative approach has led to the identification of promising migrastatic candidates, such as Rebastinib, which targets specific pathways involved in cell movement.
https://telight.eu

Telight has announced a new strategic partnership with PHYSIO MCKINA, making them the exclusive distributor of Telight’s Q-Phase imaging systems in Japan. This collaboration significantly strengthens Telight’s presence in one of the world’s leading research markets. Japan’s strong focus on life sciences and demand

for label-free, high-resolution imaging tools aligns perfectly with Telight’s cutting-edge technologies. Through this partnership, Japanese researchers will benefit from localised access and expert support, driving innovation in fields such as cancer research, regenerative medicine, and drug development.
https://telight.eu
Welcome to the new edition of Microscopy and Analysis, a result of the successful merger between Wiley’s B2B magazines, Imaging & Microscopy and Microscopy and Analysis. Our dedicated editorial team is thrilled to bring you the latest trends and developments in the ever-evolving field of microscopy. The magazine’s fresh and engaging layout symbolises this exciting new beginning.
Microscopy techniques are advancing at a remarkable pace, offering diverse methods to address a wide range of scientific questions in both life and material sciences. As innovations in this field continue to unfold rapidly, our commitment is to keep the microscopy community wellinformed about all significant breakthroughs. Our magazine features popular sections such as news, event updates, and important announcements from microscopy societies and initiatives. We are proud to maintain our enduring partnerships with the European Microscopy Society and the Royal Microscopical Society, and we are
pleased to welcome Global Bioimaging as a new collaborator.
In addition, we will provide readers with insights into the latest techniques and applications in microscopy through articles from both industry and academia. Our profiles and interviews will introduce you to influential figures, companies, and research areas, offering a personal glimpse into the lives of researchers who are shaping the future of microscopy and its applications.

Our editorial team, comprising Chris Parmenter, Rebecca Pool, Martin Friedrich, and Birgit Foltas, is eager to share our combined expertise and years of experience with you. Together, we aim to keep you informed about all the important developments in microscopy. We are honored to be a vital part of the microscopy community and look forward to engaging with you on this exciting journey.
https://analyticalscience.wiley.com/ publication/microscopy-and-analysis
If you would like your Company News to appear on these pages, please contact infocus Magazine at advertising@infocus.org.uk
The announcements in this Section are compiled by the manufacturers. They in no way represent a recommendation by the Royal Microscopical Society for any particular instrument or equipment. The Royal Microscopical Society does not endorse, support, recommend or verify the information provided on these pages.
Get ready for two of the biggest highlights on this year’s microscopy calendar! If you’re attending mmc2025 in Manchester or ELMI in Heidelberg,

you’ll have the chance to see a range of our powerful and controllable LED Illumination Systems. Whether you’re a facility manager or researcher, at
CoolLED we’ve got you covered whatever your fluorescence needs:
• High-speed live cell applications – elevate your research with the pE-800, offering eight individually controllable LEDs and lightning-fast TTL triggering speeds of <7 μs.
• Versatile wavelengths for imaging facilities –the pE-4000 includes 16 LEDs spread across four channels, ensuring there’s always a perfect combination for your experiment.
• Routine to advanced fluorescence – the latest pE-400 Series has four LEDs, covering DAPI and YFP through to Cy5, with a few extra useful features to get the most information from your fluorescence samples.
Make sure you stop by the CoolLED booth at mmc2025 (#512) or ELMI (#R04) to see how we can help.
www.coolled.com
Telight has introduced a new application note showcasing how its Q-Phase microscope, combined with SophiQ software, enables precise, label-free monitoring of cell death. This technology distinguishes between apoptotic and necrotic processes by tracking realtime changes in cell mass, area, and density. Apoptosis is characterised by cell shrinkage and membrane blebbing, while necrosis involves cell

swelling and membrane rupture. By integrating Quantitative Phase Imaging (QPI) with fluorescence markers, researchers can validate morphological observations with biochemical indicators, enhancing the accuracy of cell death analysis. This application is particularly valuable for studying drug responses and disease mechanisms in cancer and neurodegenerative research.
Read the f ull application note.
www.telight.eu
Vision Engineering, a UK based manufacturer of ergonomic microscopy and digital 3D visualisation technology unveils the EVO Cam HALO, a new 4K digital microscope that redefines the standard for clarity, precision and consistency in inspection and measurement tasks.
Suitable for a wide range of inspection applications, EVO Cam HALO combines stunning 4K image quality with advanced, adaptive illumination, and new intelligent software, empowering users to capture the most challenging details with unparalleled accuracy.
HALO’s key features include 4K resolution which reveals previously obscured details with remarkable clarity, ensuring nothing escapes the user’s view. A
and more informed decision-making. Vision Engineering’s popular 360 Direct/Oblique Viewer also works with HALO, enabling users to inspect details from different angles, revealing hidden features that would otherwise be inaccessible.
HALO is suitable for a wide range of applications, including Medical Devices Manufacture and testing especially in the practices of inspecting translucent tubes, electro-polished devices, and other medical components with enhanced clarity and accuracy.
EVO Cam HALO is available for purchase from April 2025, with configurations starting at 6500GBP.
Commenting on the launch, Paul Newbatt, Group Sales and Marketing Director at Vision Engineering says “We
Light sheet microscopy image processing software – unlocking superior quality with just a few clicks
MACS iQ View – 3D Large Volume is a user-friendly light sheet microscopy image processing software tailored for managing, stitching, and processing large 3D datasets captured by the UltraMicroscope Blaze™. It empowers you to gain meaningful insights and enhance image quality with ease. Its user-friendly interface simplifies navigation and operation, making it accessible to users of all skill
levels. Its comprehensive toolkit covers various tasks, including 3D cropping, destriping, denoising, deconvolution, stitching, and contrast compression.
Learn more here:
https://www.miltenyibiotec.com/GBen/products/macs-iq-view-3d-largevolume-software.html

At the upcoming Microscience Microscopy Congress 2025 taking place from 1st to 3rd July in Manchester, UK, Thermo Fisher Scientific will present the latest advancements in integrated electron microscopy with the Thermo Scientific™ Apreo™ ChemiSEM™. This powerful system seamlessly combines scanning electron microscopy (SEM), energy-dispersive X-ray spectroscopy (EDS), and electron backscatter diffraction (EBSD) in a single, easy-to-use platform.
The Apreo ChemiSEM addresses the integration challenges often associated with incorporating EBSD into existing workflows, offering users unparalleled sensitivity, speed, and precision for complex material characterisation. To meet the demand for high resolution imaging and accurate chemical composition analysis, this system combines advanced SEM imaging with integrated EBSD and Energy Dispersive Spectroscopy (EDS) capabilities, all within a single, streamlined interface.
This eliminates the need for separate hardware and complex software, reducing the time and effort required for system alignment and analysis.
orientation map of a deformed copper alloy
Key features such as automated alignment, advanced autofocus and Smart Frame Integration (SFI) make the system user-friendly, even for less experienced operators, while improving imaging and data acquisition speed. The TruePix EBSD detector and dedicated software further simplify the process by guiding users from setup to data interpretation, ensuring high-quality results with minimal manual intervention. These design improvements make

the Apreo ChemiSEM a practical solution for laboratories looking to adopt EBSD without disrupting their workflow or significantly increasing operational complexity.
Visit Thermo Fisher Scientific at Booth 107 during mmc2025 to experience a live demo and see the Apreo ChemiSEM in action.
www.thermofisher.com
Used extensively in industrial applications and consumer product and food packaging, multilayer polymer films have become increasingly complex in terms of design and structure whilst the specifications have become more rigid. Remarkably, and as a feat of engineering, each layer serves a unique function addressing the likes of oxygen, moisture, dust, microbes, and light, integral to safety and performance.
But how do you characterise each micron-sized layer?
By employing confocal Raman microscopy, it is possible to perform cross-section analysis and confocal depth profiling of these micron-sized layers of polymer film, establishing and identifying core characteristics, identified in terms of chemical nature and thickness.
LabRAM Soleil and Layers to the rescue!
Key to performing the cross-section analysis of these important multilayer polymer films are hardware and software solutions that
are not only intuitive and robust but are also quick to perform accurate and reliable material characterisation in a manner that is easy and straightforward. In addition, HORIBA’s NEW Layers app offers one-click analysis to automatically determine the thickness and composition of polymer layers, offering peace of mind without unnecessary complications or delays to processing. Find out more about the LabRAM Soleil:

Scan or click the QR code to download our Application Note:

To find out more about the power of Layers and to see a video of it in action scan or click the QR code here: www.horiba.com
Delmic and Digital Surf are proud to bring to market new CL Workspace software powered by Mountains® technology, an innovative analysis solution for use with Delmic’s advanced cathodoluminescence (CL) imaging systems.
Delmic’s state-of-the-art imaging tools detect the cathodoluminescence emitted when materials are irradiated with a focused electron beam. This technique generates high-resolution CL maps that can be correlated with secondary-electron (SE) images, allowing researchers to gain new insights on material properties including composition, crystal structure, defectiveness and electronic band gap.
Tailored software for enhanced data analysis
The newly launched CL Workspace software is specifically engineered for Delmic’s CL imaging tools, empowering users with an intuitive interface that dramatically simplifies advanced data analysis. The unique Mountains® document-based analysis makes it easier to perform reporting, especially in the context of repetitive analyses. Furthermore, full data compatibility with TIFF and HDF5 formats ensures seamless integration with all existing Delmic microscopes. Two customised product levels are made available to users depending on their applications.
CL Workspace 2D for intensity mapping includes the following features:
• Image optimisation: de-noising, flattening and proper scaling for accurate visualisation.
• Particle analysis: quantification of defect densities and types in semiconductor materials and heterostructures.
• False-color merging: integration of color-filtered images into single RGB images to highlight subtle differences in emission.
CL Workspace 3D for multidimensional spectroscopic and time-resolved CL data offers tools for:
• Visualisation of regions of interest: enhanced identification of specific sample areas in all dimensions of a hyperspectral dataset.
• Numerical data fitting: extraction of quantitative information from spectroscopic and timeresolved datasets.
• Dynamic tools and movie export: creation of dynamic figures and movies from hyperspectral image stacks for comprehensive analysis with correlative insights.
“We’re thrilled to bring more intuitive CL data analysis to Delmic customers,” said Christophe Mignot, Digital Surf CEO. “At Digital Surf, it is our goal to make generating consistent, reportready outputs effortless, without the need for programming. This enables both researchers and industrial professionals to quickly capture, visualise and communicate deep insights on their samples, significantly accelerating their processes.”
www.digitalsurf.com
If you would like your new product information to appear on these pages, contact infocus Magazine at advertising@infocus.org.uk
The announcements in this Section are compiled by the manufacturers. They in no way represent a recommendation by the Royal Microscopical Society for any particular instrument or equipment. The Royal Microscopical Society does not endorse, support, recommend or verify the information provided on these pages.

By Francisco Rangel, National Institute of Technology, INT/MCTI, Brazil.
The image shows the transverse section of a small fragment of a turtle eggshell pick up from a nest from the Brazilian Cretaceous dating to around eighty million years ago. Horizontal Field Width 249 µm. The sample was tilted at 45 degrees to provide a 3D appearance. SEM QUANTA 450 FEI COMPANY.
Francisco said: “The image captivates me by offering an unprecedented glimpse into the past. Its microscopic details, invisible to the human eye, provide paleontologists with invaluable data to meticulously trace the evolutionary path of turtles.”
infocus is the Royal Microscopical Society’s (RMS) vibrant and striking quarterly magazine for members. It provides a common forum for scientists & technologists who use any form of microscope, including all branches of microscopy. Published four times a year, infocus is free to members of the RMS. infocus features articles on microscopy related topics, techniques and developments, an events calendar, news, event reports, book reviews, new product information, and much more. infocus welcomes submissions of:
Articles - Full articles or reviews of general interest to microscopists, of approximately 30004000 words (excluding references), with images/ figures (as many as appropriate, 4-8 as a guide). Longer articles can also be considered.
Short Articles - Short topical articles, review articles or articles providing hands-on help for microscopy methods.
Primer Articles - Short general articles that are focussed on specific techniques.
Debuts - Student articles publishing emerging results from a project. Results may still be incomplete, but areas of progress/problems should be highlighted, with the aim of provoking feedback.
Book Reviews – if you are a member of the RMS and are interested in writing book reviews for infocus, please contact Owen Morton owen@rms.org.uk.
Please see recent issues of infocus for examples of articles and reviews. To request a sample copy of infocus contact owen@rms.org.uk
If you are interested in submitting to infocus, contact: editor@infocus.org.ukj
Article Text
• Text should be in a standard font (e.g. Times New Roman or Arial) at a size of 12 pt.
• Articles should begin with a brief summary, which accurately summarises the content and is intelligible without reference to the text.
• Footnotes and appendices should not be used unless absolutely necessary.
• The hierarchy of headings within the text should be clear.
• Spelling should conform with The Concise Oxford Dictionary and SI units must be used.
• Abbreviations should be used sparingly and only if a lengthy name/expression is repeated throughout the article. When used, the abbreviated name or expression should be cited in full at first usage, followed by the abbreviation in parentheses.
• Authors should provide a photograph, brief biography as well as contact information that will be published.
References in the text should be in the form Joy (2000) or Joy & Williams (2000). For three or more authors, use the form Echlin et al. (2000). The reference list should:
• be listed in alphabetical order of first authors’ surnames.
• (where a journal is cited) - include authors’ surnames and initials, date of publication, title of paper, name of journal, volume number, and first and last page numbers.
• (where a book is cited) - include authors’ surnames and initials, title of book, year of publication, edition, followed by publisher and town, county/state (and country if necessary) of publication.
• (where a URL is cited) – include authors’ surnames and initials, year of publication, title of page, URL and date accessed.
• Figures can be one column/half page width, 65.5 mm or two column/full page width, 135 mm.
• Larger images may fill the page/spread. A full page of the magazine is 170 x 250 mm, a double page is 340 x 250 mm.
• Text in figures (labels, axis labels, legends, etc) should be Helvetica or Arial, 8pt size.
• Figure and table captions should be listed numerically at the end of the article text
• Line weights and line strokes should have a maximum value of 1 and a minimum of 0.25.
• For graphs and plots, whenever possible, please submit vectorized images.
• As much as possible, please avoid white spaces.
• All images must be high resolution – 300dpi or more.
• Submission files should be in CMYK format and can be supplied as tiff, jpeg or eps files.
• Images MUST include scale bars or field widths where relevant.
Double page of magazine, 340 x 250mm (Trim size)
• Total number of images/figures/tables should not exceed 15 including tables.
Prior to publication, authors will be sent a PDF of the article by email for approval.
Authors should ensure articles are thoroughly checked before submission – proof amendments should be limited to minor corrections only.
Five hard copies of the issue in which the article is published will be sent to the author, together with an emailed PDF of the article.
Authors are requested to assign copyright to the RMS. However, authors may make copies of their own articles without seeking permission from the RMS, provided that such copies are for free distribution only (they must not be sold) and provided that infocus is properly acknowledged (issue number, month and page number should be given). Permission to reproduce material from infocus in other publications will not be given to third parties except with the consent of the authors concerned.
Authors are responsible for obtaining permission to reproduce copyright material from other sources. Approval for reproduction/modification of any material (including figures and tables) published elsewhere should be obtained by the authors before submission of the manuscript and the source of the material should be properly acknowledged. Authors are responsible for any copyright fee involved.
One column/half page width, 65.5mm
Authors are requested to complete and submit a signed copy of our copyright sign-off form. This is available on the RMS website (www.infocus.org.uk).