25e Jaargang • Nummer 2 • November 2025


25e Jaargang • Nummer 2 • November 2025

How to build a patent portfolio around a new therapeutic product
Pag. 12
Biotech companies at Utrecht Science Park Genmab and Merus join forces
Pag. 15
The Leiden-Lund Collaboration: a model for European Success in Cell and Gene Therapy
Pag. 44
Eli Lilly chooses Leiden Bio Science Park for major expansion, boosting the Netherlands’ life sciences ecosystem
Pag. 45
Enabling successful AI transformation in Life Sciences
Pag. 60




Patient access to biopharmaceutical innovations: an industry perspective
Pag. 6
What BioBusiness Summer School taught me about transitioning to industry
Pag. 22
The Compounding Failure of Women’s Health Diagnostics in Europe
Pag. 40
Aisling Foley and the ExCulture team’s vision for changing the way heparin is produced
Pag. 62
The HH Global Investor Forum, in conjunction with Innovation for Health, gives you the opportunity to present your company and your innovation to leading investors and meet potential partners. It offers two days of pitch presentations, 1-to-1 meetings with investors, workshops and sessions.
Besides presenting your company and meeting key players in industry and finance, you will have the chance to win Startup Pitch Award, sponsored Sponsor AXON Lawyers and F.Institute.































We have room for 30 start-up in Biotech, MedTech, AI, and Digital Health to:
» Practice your pitch in front of a targeted audience before the big day.
» Present your innovation to investors and open doors to funding and strategic partnerships.
» Engage directly with investors who are eager to hear your story and explore collaboration opportunities.
» Boost your entrepreneurial skills through programme sessions designed to help you succeed.
» Connect with industry leaders and fellow innovators at the Innovation for Health conference.


The life sciences sector is defined by its ability to adapt, collaborate, and lead—and this new edition of BiotechNEWS & Life Sciences captures that spirit in full. Over the past months, our industry has seen transformative developments, bold leadership, and inspiring personal journeys. This edition of our sector magazine reflects both the momentum and the personal stories behind biotech innovation.
Our cover photo revisits this year’s TOPX Leadership Summit theme: “Leading Together in Life Sciences & Health: Building High-Performing Teams to Drive Innovation and Benefit Society.” It is a timely reminder that scientific progress is not driven by individuals alone, but by excellent teams that dare to challenge, support, and push each other toward meaningful breakthroughs.
This spirit of collective progress is also visible in two major sector developments: Genmab’s acquisition of Merus, a landmark deal between two companies that have grown side
BiotechNEWS & Life Sciences is onderdeel van: Hyphen Projects BV
Veerstraat 37, 1211 HJ Hilversum
KvK: 32110979, BTW nr : NL8184.34.491.B01 ING NL26INGB0007452062
Telefoon: 035 623 07 81 www.hyphenprojects.nl/biotechnews
Uitgever Haifen Hu, Eigenaar en Directeur
Redactie Drs. Ilse Kuiper, Hoofdredacteur
Erwin Gevers, Redacteur
Yilmaz Biter, Redactielid
Ivo Horn, Redactielid
Nilgun Gonenc, Redactielid
E-mail: biotechnews@hyphenprojects.nl
Verkoop
by side at Utrecht Science Park; and Eli Lilly’s decision to invest more than USD 3 billion in a new manufacturing facility at Leiden Bio Science Park. Both moves highlight the Netherlands as an increasingly strategic hub for global biotech.
We further spotlight emerging innovators shaping the next wave of biomedical solutions: ExCulture, led by founder Aisling Foley, and AdJane, headed by Anita Gashi. Their work exemplifies the creativity and determination driving early-stage biotech today.
For companies navigating the path from discovery to market, this edition also offers practical insights, including NLO’s guide on building a strong patent portfolio and Venn Life Sciences’ article on due diligence for drug development and start-ups.
In this edition, we also share powerful personal stories—on resilience, ambition, and navigating the realities of academic and professional life.
Haifen Hu, Sales Algemeen
Lotte Bramer, Sales Arbeidsmarkt
Ferdinand Oldemaat, Sales Strategisch Adviseur Miriam Amiguetti, Marketing
Vormgeving Reclamemakers.nl
Nathalie Wessels
Fotografie O.a. Joshua Quicken
Druk Senefelder Misset (Doetinchem) ISSN 2666-1012
Verspreiding PostNL
25e Jaargang • Nummer 2 • november 2025
INFORMATIE Biotechnologie is een sterk groeiende industrie. Nederland is wereldwijd sterk in deze sector en de vooruitzichten zijn zeer goed. BiotechNEWS & Life Sciences is een magazine dat inzicht biedt in de onderwerpen en ontwikkelingen die spelen binnen deze Nederlandse economische topsector. Nederlandstalig en laagdrempelig. Biotechnologie en life sciences is een internationale industrie. Vrijwel alle publicaties over dit onderwerp verschijnen in het Engels. BiotechNEWS vindt het essentieel om juist in het Nederlands te publiceren.
COPYRIGHT © 2025 Niets uit deze uitgave mag worden verveelvoudigd en/of openbaar gemaakt door middel van druk, fotokopie, microfilm of op welke andere wijze dan ook, hetgeen ook van toepassing is op de gehele’of gedeeltelijke bewerking, zonder voorafgaande schriftelijke toestemming van de uitgever. Alle artikelen welke in deze uitgave gepubliceerd zijn, vallen onder de verantwoordelijkheid van de desbetreffende journalist. Het verlenen van toestemming tot publicatie in BiotechNEWS houdt in dat de auteur de uitgever, met uitsluiting van ieder ander, onherroepelijk machtigt de bij of krachtens de Auteurswet door derden verschuldigde vergoeding voor kopíëren te irmen of daartoe in en buiten rechte op te treden. Wij maken u er op attent dat de verstrekte gegevens zoals, naam, adres en woonplaats zijn opgenomen in onze klantenadministratie. Wij willen het gebruiken om u te blijven informeren over onze voordeelaanbiedingen, produkten en dergelijke van zorgvuldig uitgekozen andere bedrijven. Deze administratie is aangemeld bij de registratiekamer te Rijswijk onder nummer P-0015 122 Indien u bezwaar heeft tegen dit gebruik van uw gegevens dan kunt u dit schriftelijk aan ons laten weten.
I hope this edition informs, inspires, and encourages you to keep building the future of biotech—together.
Haifen Hu Uitgever | BiotechNEWS & Life Sciences

Your news in BiotechNEWS?
BiotechNEWS & Life Sciences is the magazine that provides the Dutch & Belgium Life Sciences sector with sector news and keeps you informed of the latest developments and interesting trends.
Do you have interesting company news, product launches or personal stories to share with the Life Sciences ecosystem in the Netherlands and Belgium? Send us your press release or contact our editorial board to biotechnews@ hyphenprojects.nl.
Beschikbaar: lab- en kantoorvloeren in Gebouw Elisabeth en Rosalyn – dé plek voor innovatieve bedrijven in health & technology.
In het hart van het Amsterdam Life Sciences District ligt de AHTC Campus, een levendige en duurzame omgeving waar organisaties in Life science, Health en Technology samenkomen. Met gedeelde voorzieningen, een sterke community en nauwe samenwerking met Amsterdam UMC, biedt de campus de ideale basis voor groei en innovatie.















Ralph Bosmans (manager Biotech) en Dennis Gielissen (Scientist Biotech) van Basic Pharma
Sinds de oprichting in 2003 is Basic Pharma uitgegroeid tot een innovatief en veelzijdig farmaceutisch bedrijf met ca 275 medewerkers en een internationale klantenkring in meer dan 25 landen. Vanaf de Brightlands Chemelot Campus in Geleen worden de faciliteiten steeds
De huidige producties binnen de Biotech-afdeling van Basic Pharma zijn op dit moment nog niet geschikt voor farmaceutische toepassingen. De stappen om ook farmaceutische producten te mogen produceren zijn echter al in gang gezet.In de tussentijd richt de afdeling zich op het bedienen van klanten in diverse segmenten, waaronder onderzoeksinstituten en industriële partners wereldwijd. Basic Pharma biedt eiwitten aan uit haar bestaande portfolio, maar ontwikkelt en produceert ook klantspecifieke recombinante eiwitten. Dankzij deze flexibiliteit kan de Biotech-afdeling inspelen op uiteenlopende behoeften en innovatieve projecten ondersteunen binnen de internationale biotechsector. Please contact us via biotech@basicpharma.nl or +31 (0)88 255 40 10 www.basicpharma.nl/biotech
verder uitgebreid en is flink geïnvesteerd in productie- en laboratoriumcapaciteit en state-of-theart apparatuur. Zo kan Basic Pharma zich richten op de volle breedte van de farmaceutische industrie van productontwikkeling, registratie, productie en commercialisatie van (bio) farmaceutische producten tot en met farmaceutische dienstverlening. Recent is aan deze Full Service Package de ontwikkeling en fermentatie van recombinante eiwitten toegevoegd onder de naam Biotech Services & Partnering. Manager Biotech Ralph Bosmans en Director Business Development Peter Hollanders vertellen.
‘We zijn trots op de ontwikkelingen die Basic Pharma de afgelopen jaren heeft doorgevoerd, ‘vertellen Peter Hollanders en Ralph Bosmans, beiden sinds acht jaar werkzaam bij het bedrijf in Geleen. ‘We zijn op een mooie manier gegroeid. De directe lijnen zijn er nog steeds, en we zijn even mean-and-lean als decennia geleden toen we ooit begonnen met de fabricage van crèmes en zalven. Er kan snel geschakeld worden bij nieuwe ontwikkelingen wat de toekomstbestendigheid en de klantgerichtheid ten goede komt.’
Biotech Services: medicijnen voor de toekomst ‘Om nog beter in te spelen op de wensen van onze klanten en ze volledig te ontzorgen, besloten we ons enige tijd geleden ook te richten op de productie van recombinante eiwitten middels microbiële fermentatie van gisten en bacteriën. Deze organismes worden genetische gemodificeerd zodat ze het gewenst eiwit produceren en vervolgens gekweekt in bioreactoren, legt Ralph uit die een achtergrond heeft in de chemische biologie. Vervolgens worden deze eiwitten gezuiverd, zodat ze veilig kunnen worden toegediend aan patiënten. Onze faciliteit is ontworpen om met Genetische
Equipment
• Bioreactors: 0.5 LT – 5 LT and 50 LT total volume
• High-speed centrifuge
• Bioprocess homogenizer, up to 1200 bars for cell lysis
• Chromatography systems: R&D and pilot scale
• Ultrafiltration: Tangential flow filtration (TFF) system
• Analytical equipment, including: HPLC Electrophoresis, Absorbance plate reader
gemodificeerde organismes te werken, tot aan ML-II containment level. Omdat wij de ontwikkeling van nieuwe medicijnen onder één dak aanbieden, het zogenoemde one-stop-shopping, zijn we niet alleen in staat om snel te schakelen, maar worden de biotechnologische producten, die ook wel de medicatie van de toekomst worden genoemd, ook snel op de markt gebracht. Zodra het bioproces is ontworpen, kunnen we de klant bijstaan met opschaling en productie van de eiwitproductie. Daarbij kan de klant ook gebruik maken van onze laboratoria services en vervolgens ook van onze fill & finish mogelijkheden.’
Partnering
Samenwerking staat bij Basic Pharma hoog in het vaandel. Onlangs is een samenwerking tot stand gekomen met Bio-Connect, een toonaangevende distributeur van life science- en diagnostische reagentia in Europa. Samen met het Duitse Artes Biotechnology maakte Basic Pharma grote stappen en beleefde het een primeur, toen de eerste commerciële samples van rHSA (Recombinant Human Serum Albumin) beschikbaar kwamen geproduceerd in gisten. Inmiddels is het assortiment eiwitten uitgebreid met o.a. Histon H3 en GFP. Dit toont aan dat Basic Pharma daadwerkelijk in staat is om dit soort producten te fabriceren.
‘Groei en ontwikkeling staan bij Basic Pharma, maar ook bij onze klanten, steeds centraal,’ besluiten Ralph en Peter. ‘Dat vind je niet alleen terug in kwaliteit en kwantiteit, maar ook in de groei van ons servicepakket. Zo kunnen we door onze nieuwe Biotech Services, onze klanten nu en in de toekomst nog beter bedienen.’
By Anne Sinke, Ph.D., Medical Director, Gilead Sciences
On this we can all agree: Everyone deserves access to the best possible medical care and treatments. But the path to care is often shaped by where you live, what you can afford, and how health systems prioritize access.
Last year, a Gilead Sciences breast cancer treatment made headlines because it took nearly three years for patients in the Netherlands to gain access, despite the treatment being accessible for some time in most European countries.
The latest reports show that access to innovative medicines in the Netherlands takes on average 459 days and increases by about six weeks each year.1 In addition, year after year, the same reports show a concerning, declining trend in the proportion of EMA-approved innovations that become accessible to patients in the Netherlands: 31 out of 41 drugs (76%) were accessible between 2016-2019, versus only 24 out of 56 oncology drugs (43%) and 30 out of 66 orphan drugs (45%) between 2020-2023. 2, 3
This is resulting in troubling and emotionally charged scenarios, with patients in the Netherlands seeking alternative pathways to access treatments in neighboring countries – some paying out of pocket, others turning to crowdfunding, and some even contemplating relocation.
One of the contributing factors to this situation is the Dutch government’s “Lock” (“Sluis”) Procedure, introduced in 2015 to regulate the reimbursement of new medicines. Under this procedure, the Dutch National Health Care Institute evaluates a medicine’s efficacy, cost-effectiveness, necessity, and feasibility. If the outcome is positive, it initiates price negotiations, which can be lengthy.
A New Perspective: Balancing Accessibility and Affordability as a Polarity, Not a Problem
Groundbreaking therapies are only valuable if they reach the right patients at the right time and in the right way. But the Netherlands and many other countries are at a crossroads – between the accessibility and affordability of biopharmaceutical innovations. This dilemma is often perceived as a problem, with either a right or wrong solution, thereby causing a heated and polarized debate. Another

perspective is to look at this through the lens of the “polarity model.”
The polarity model recognizes the dilemma as a polarity – an issue that is ongoing, unsolvable, and contains seemingly opposing ideas. However, the model also sees the opposing views as two sides of an issue that are both necessary for balance. 4 Solving the polarity requires collaboration between all stakeholders, something that Gilead is strongly in favor of.
In fact, a byproduct of securing patient access to Gilead’s breast cancer treatment in the Netherlands is that it led to our company becoming a more vocal advocate for the role of collaboration and understanding in effecting positive change. We are actively engaged in a dialogue with other stakeholders
– including patient advocacy groups, professional medical and scientific societies, healthcare professionals, government health agencies, payers and industry partners – not just in the Netherlands, but worldwide.
Discovering, developing and delivering innovations for life-threatening diseases takes a long time and involves significant risk and substantial investments. Typically, only one drug emerges from a pool of 10,000 molecules and 10 to 15 years of rigorous research, testing, and regulatory approval, including preclinical studies, clinical trials, and manufacturing optimization. An analysis of the R&D input, output, and outcome of 16 leading research-based pharmaceutical companies over 20 years (2001-2020) showed an average of $6.16 billion total R&D expenditures per new drug. 6
At the heart of the issue is the value of innovation. In 1996, people living with HIV were young, not expected to live past 40, and took on average over 20 pills a day. Today, almost half of people living with HIV are over 50, have a normal life expectancy, and take a single daily pill. However, although great progress has been made in the treatment of HIV, new HIV infections are on the rise in many countries and there is still much more innovation needed to end the HIV epidemic for everyone, everywhere.
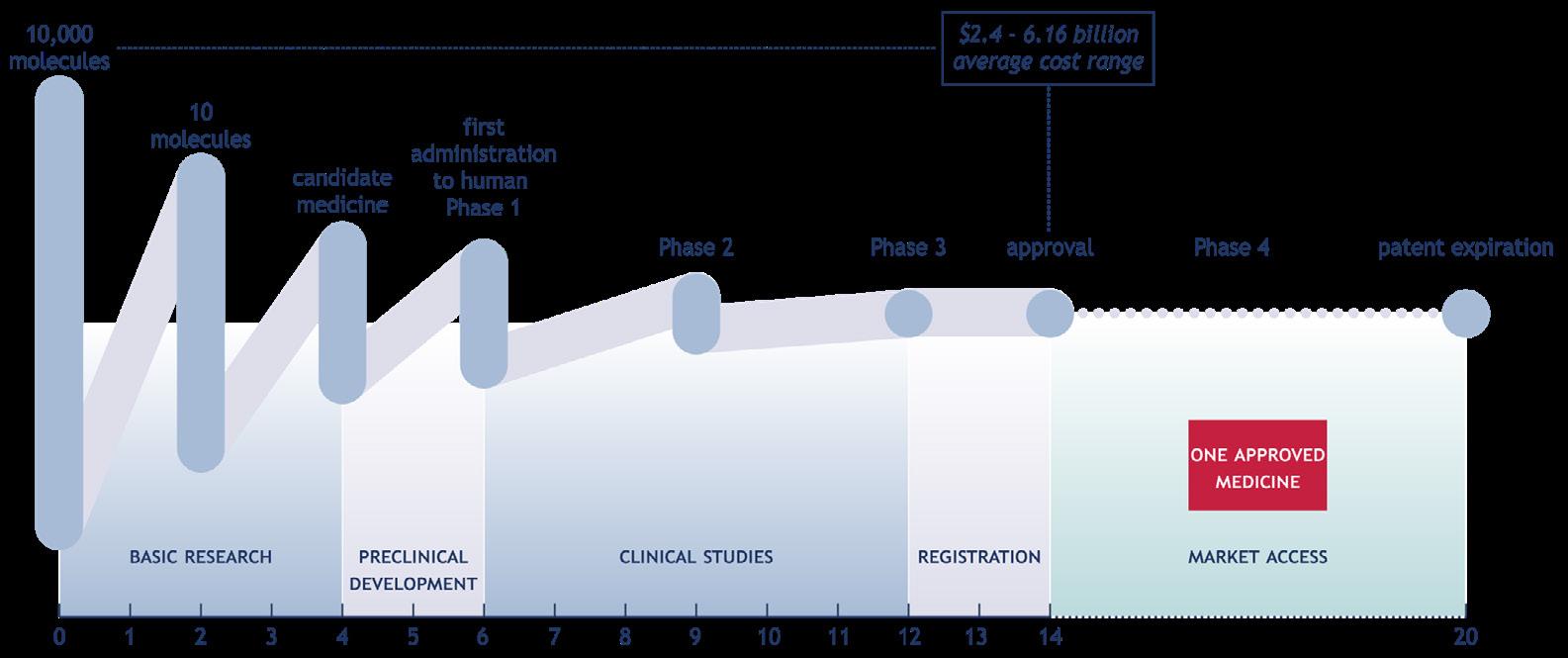
Continuous drug development is associated with significant investments. 10, 11, 12
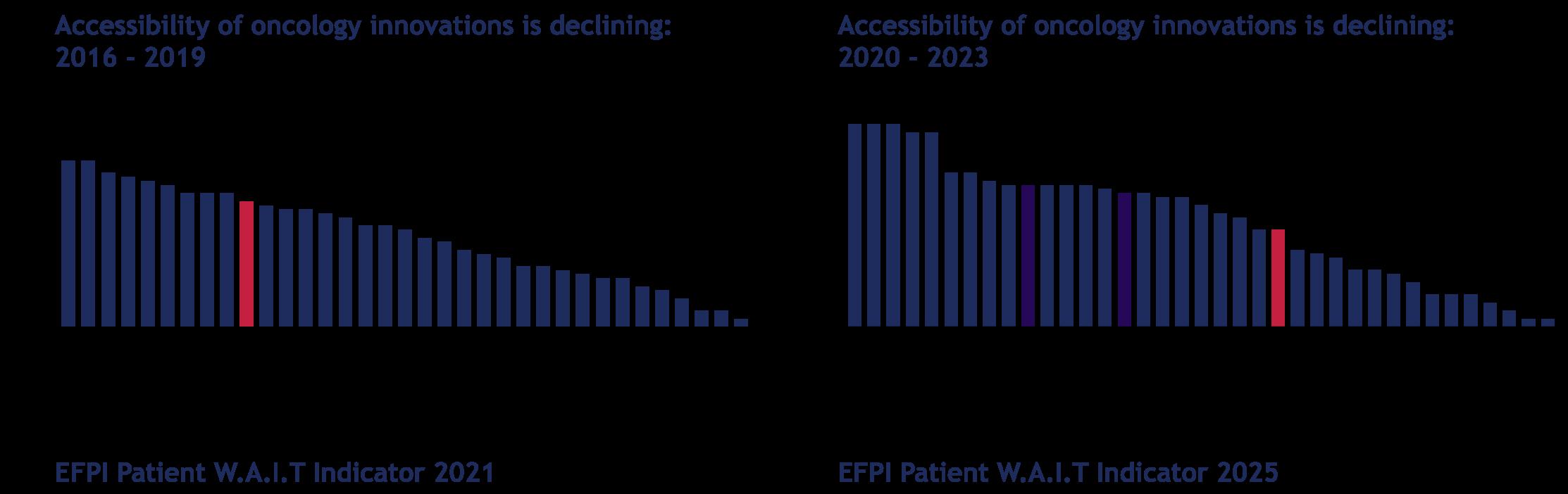
In the Netherlands in particular, oncology drug accessibility is decreasing over time.
Similarly, the Dutch Cancer Registry reported recently that the four-year survival rate for cancer patients has risen to 72%, up from 51% thirty years ago. This clearly shows that great improvements have been made, thanks to ongoing research into new treatment methods and medications. In addition, over 30 years, survival of patients with newly diagnosed metastatic cancer improved, although modestly and unevenly among the different cancer types. 7 Metastatic cancer remains a very lethal disease.
Therefore, there is still a clear need for better treatment options, better preventive measures and early detection to reduce the incidence of metastatic disease. The challenge is that only 1 out of 20 (5%) of cancer drugs in Phase 1 clinical trials and less than 1 out of 2 (44%) of cancer drugs in Phase 3 clinical trials ever see the light of day. 8
In the Netherlands, as already stated, this is further exacerbated by the declining accessibility to oncology drugs over time. As the bar graphs above from the EFPIA (European Federation of Pharmaceutical Industries and Associations) Patients W.A.I.T. (Waiting to Access Innovative Therapies) Indicator illustrate, we are moving in the wrong direction.
Currently, decision-making is driven in part by the perception that spending on innovative medicines is spiraling out of control. The graph below, however, reveals that while medical specialty care spending (blue line) in the Netherlands has steadily increased over six years, the expenditure on add-on expensive medications (red line) has remained relatively stable. 9 Unfortunately, hospitals do not always recognize this, as the financial benefit due to central agreements often does not return to them.
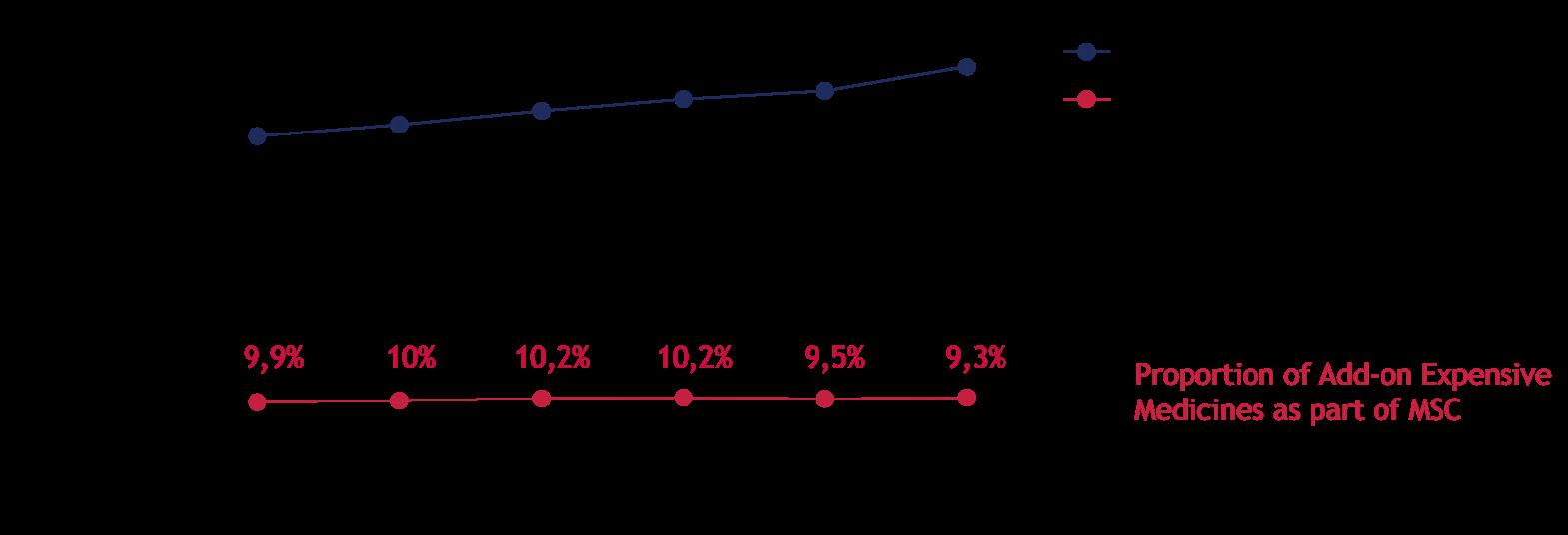
Proportion of add-on innovative medicines as part of Medical Specialty Care (MSC) budget remained stable over time.
1 Patiënten moeten in Nederland nóg langer wachten op nieuwe geneesmiddelen. Vereniging Innovatieve Geneesmiddelen. Published May 13, 2025. https://www.vereniginginnovatievegeneesmiddelen.nl/ kennisbank/patienten-moeten-nog-langer-wachten-op-nieuwe-geneesmiddelen/ Newton M, Scott K. EFPIA Patients W.A.I.T. Indicator 2021 Survey. (Troein P, ed.). EFPIA, IQVIA; 2022. Accessed June 19, 2025. https://www.efpia.eu/media/676539/efpia-patient-wait-indicator_update-july-2022_final.pdf
3 Schwaiger C. Netherlands continues negative trend, sees further decline in drug wait time rankings. Euractiv. Published May 19, 2025. Accessed June 19, 2025. https://www.euractiv.com/section/ health-consumers/news/netherlands-continues-negative-trend-sees-further-decline-in-drug-wait-timerankings/
4 Leading with Balance: Polarity Management for Effective Leadership. Systems Thinking Alliance. Published November 11, 2024. https://systemsthinkingalliance.org/leading-with-balance-polarity-man-
agement-for-effective-leadership/
5 Research & Development PhRMA. Phrma.org. Published 2025. https://www.phrma.org/policy-issues/ research-development
Schuhmacher A, Hinder M, von Stegmann und Stein A, Hartl D, Gassmann O. Analysis of pharma R&D productivity – a new perspective needed. Drug Discovery Today. 2023;28(10):103726. doi:https:// doi.org/10.1016/j.drudis.2023.103726
7 Luyendijk M, Visser O, Blommestein HM, et al. Changes in survival in de novo metastatic cancer in an era of new medicines. Journal of the National Cancer Institute. 2023;115(6):628-635. doi:https://doi. org/10.1093/jnci/djad020
8 Thomas D, Chancellor D, Micklus A, et al. Clinical Development Success Rates and Contributing Factors 2011-2020.; 2021. https://go.bio.org/rs/490-EHZ-999/images/ClinicalDevelopmentSuccessRates2011_2020.pdf
Accessibility of medicines is shaped by a complex interplay of factors: regulatory approval, government policies, pricing negotiations, willingness to pay, health technology assessments, and healthcare coverage. At the individual level, availability of comprehensive healthcare services, disease awareness, stigma, health literacy, and trust in healthcare institutions play critical roles. Achieving equitable access means aligning political will, economic capacity, and public understanding, in other words, accepting the polarity but working together toward the common goal.
As the accessibility and affordability of pharmaceutical innovations continues to be under pressure, it is of utmost importance that we have an ongoing and constructive dialogue with all relevant stakeholders. We need to understand the medicine’s value, identify a price that reflects its worth, and explore solutions that ensure optimal patient care - both in the Netherlands and beyond. Gilead is open to constructive dialogue and committed to contributing to a workable outcome, always with patients’ best interests at heart.
9 NZa Kerncijfers intramurale dure geneesmiddelen 2018 tm 2022 en voorlopige cijfers 2023 - Nederlandse Zorgautoriteit. Overheid.nl. Published 2018. Accessed June 19, 2025. https://puc.overheid.nl/ nza/doc/PUC_775444_22/ Prasad V, Mailankody S. Research and development spending to bring a single cancer drug to market and revenues after approval.JAMA Intern Med. 2017;177(11):1569–1575. doi:10.1001/jamainternmed.2017.3601
11 SiRM; L.E.K. Consulting; RAND Europe. The financial ecosystem of pharmaceutical R&D: An evidence base to inform further dialogue. Published February 2022. Accessed November 5, 2025. https:// www.rand.org/pubs/external_publications/EP68954.html
12 Schuhmacher A, Hinder M, von Stegmann Und Stein A, Hartl D, Gassmann O. Analysis of pharma R&D productivity—a new perspective needed. Drug Discov Today. 2023;28(10):103726. doi:10.1016/j. drudis.2023.103726

One of a kind. Iedere cleanroom is uniek. Sommige zijn wat meer standaard. Andere zijn uiterst complex en vereisen hoogwaardige kennis en expertise. Cleanroom Combination Group bv (CCG) is gespecialiseerd in de laatstgenoemde categorie. Daarvoor moet je flexibel zijn; denken in constructieve oplossingen en beschikken over korte lijnen met betrouwbare, vaste partners. Het zijn randvoorwaarden om de nieuwste uitdaging, een complexe interne ‘nieuwbouw’ voor een GMP-radionuclide productie-omgeving bij GE HealthCare tot een succes te maken.
In een GMP-radionuclide-productieomgeving worden radioactieve stoffen voor medisch gebruik geproduceerd. De omgeving is ontworpen om de kwaliteit en veiligheid van de radiofarmaceutica te waarborgen door middel van een uitgekiende bouw en infrastructuur, specifieke facilitaire ontwerpeisen, strikte hygiëneprotocollen en kundig personeel. GE HealthCare is gespecialiseerd in radioactieve diagnostiek om onder meer kankers aan te tonen. Hierbij wordt gebruik gemaakt van een radioactieve glucose-oplossing (FDG). PET/CT scans en SPECT zijn state-of-the-art technologieën die inzicht bieden op cellulair niveau, wat essentieel is voor een nauwkeurige diagnose en effectieve behandelplanning. Bij GE HealthCare op de TU campus in Eindhoven werken zo’n 200 professionals en wereldwijd krijgen zo’n 100.000 mensen per jaar de radioactieve diagnostische producten van GE HealthCare Eindhoven toegediend.
Uitbreiding…
Om aan de groeiende vraag naar radiofarmaceutica te voldoen, bleek uitbreiding van de cleanroomcapaciteit voor GE HealthCare noodzakelijk. Dat CCG als eerste op het netvlies verscheen, is volgens Koen Kodde, projectmanager bij GE HealthCare, geen wonder:
‘De samenwerking tussen deze 2 organisaties dateert al van 25 jaar geleden.’
Michiel van Kooten, CEO van CCG en Harald Leenders, Manager Facilities GE HealthCare vertellen over de leegstaande ruimte op de TU/e Campus die voor dit doel werd gebruikt. ‘We begonnen met de gedeeltelijke sloop van de bestaande omgeving. Daarna werden de nodige ruwbouwkundige aanpassingen doorgevoerd zoals boorpalen, staalconstructie en een nieuwe tussenvloer etc.’
Dat klinkt nu als een peulenschil, vertelt projectmanager Erwin Lindberg van CCG maar heeft heel wat hoofdbrekens gekost. ‘We moesten binnen extra heipalen plaatsen waarbij de faciliteit gewoon door moest blijven draaien. Dat kan je alleen succesvol aanpakken met ervaren partners die je door en door vertrouwt.’
Van box-in-box naar beloopbaar plafond Samen met onze partners – waaronder Kuijpers, PHF Services BV, BroekBakema Architecten en Movares , hebben we voor dit project een innovatief box-in-box systeem toegepast. Dit systeem bestaat uit een overdruk cleanroom binnen een onderdrukschil. In de cleanroom wordt meer gefilterde lucht toegevoerd dan afgevoerd, waardoor een positieve druk ontstaat. Dit voorkomt dat ongewenste deeltjes van buiten naar binnen dringen en waarborgt de GMP-kwaliteit. De omringende schil werkt precies omgekeerd: hier heerst een onderdruk, zodat er geen lucht ongecontroleerd naar buiten kan ontsnappen. Dit ontwerp waarborgt zowel productveiligheid als strikte containment-eisen.
Dat deze aanpak uitstekend werkt, is inmiddels bewezen bij andere projecten, op diverse locaties van GE HealthCare in Leiderdorp en Eindhoven.
Ook het beloopbare cleanroomplafond dat in dit project is toegepast, mag inmiddels tot een specialisatie van CCG worden gerekend. Het systeem is eenvoudig aanpasbaar en goed toegankelijk. Onderhoud aan kleppen, roosterbakken, verlichting en andere installaties wordt aanzienlijk vereenvoudigd, omdat alles van bovenaf bereikbaar is. Bovendien blijft de cleanroom tijdens onderhoud volledig operationeel.
Ook tijdens de bouw biedt dit grote voordelen: er kan gelijktijdig in de cleanroom én bovenop het plafond worden gewerkt, wat de efficiëntie
verhoogt. CCG paste hiervoor een volledig modulair plafondsysteem toe. Het systeem is relatief dun, neemt weinig ruimte in en is uitstekend belastbaar (tot 150 kg).
Daarnaast maakt CCG binnen dit project gebruik van duurzame TRESPA-wandpanelen en Colorexvloerafwerking.
De eindfase…
Nu (november 2025) werkt het team naar de eindfase van de cleanroomafbouw. Een laatste uitdaging is de plaatsing van de zogenoemde “hot cells”, afgeschermde isolator-kamers die gebruikt worden voor het werken met radioactieve materialen. Michiel: ‘Ook dit bijzondere project voor GE HealthCare is uniek en klantspecifiek en daarmee een uitdagende puzzel, die we samen met klanten en partners, oplossen om tot een optimaal resultaat te komen.’
CCG werkt en werkte aan de meest prestigieuze en duurzame projecten op het gebied van Healthcare en Lifesciences in Nederland.
1. Voor Bilthoven Biologicals is een zeer hoogwaardige polio research faciliteit gerealiseerd (850 m2) met een BSL3 en GMP compliant faciliteit. CCG werkte samen met architect KuiperCompagnons, Kropman Contamination Control en DPS Engineering.
2. In de bouwcombinatie met Kuijpers, DuPrie en EGM architecten, was CCG verantwoordelijk voor de engineering en bouwkundige realisatie van de cleanrooms bij Halix in Leiden. Het nieuwe pand is op de groei gebouwd. De eerste en tweede verdieping zijn voorzien van flexibele GMP cleanrooms d.m.v. het box-in-box principe.
3. De radiofarmaceutische apotheek van GE Healthcare in Leiderdorp, in opdracht van het Alrijne Ziekenhuis, samen met Kuijpers en DuPrie gerealiseerd.
Hitma Retour- en Recycleservice
Het is de eerste toeleverancier in Nederland en België die de capaciteiten en infrastructuur in huis heeft om daadwerkelijk iets te doen tegen vervuiling en verspilling in de farmaceutische industrie door een Retour- en Recycleservice op te zetten: Hitma.
Ongebruikte single-use plastics waarvan de houdbaarheidsdatum (bijna) is verstreken, worden ingezameld en krijgen een tweede leven bij onder andere farmaceutische startups en trainingsfaciliteiten. Materialen die niet meer bruikbaar zijn, worden zorgvuldig gerecycled tot nieuwe producten. Liesbeth Gebuijs, Coördinator Hitma Retour- en Recycleservice, Mitch van Nieuwkerk, Business Developer en Savanna Bosman, Sustainability Consultant beantwoorden prangende vragen.

Mitch: Hoe is dit initiatief tot stand gekomen en hoe werkt het?
‘Binnen Hitma onderzochten we al lange tijd de mogelijkheid tot een Retour- en Recycleservice. We keken naar de materialen die geretourneerd kunnen worden en wat we kunnen doen met materialen die niet hergebruikt kunnen worden. Jaren geleden is hierdoor een recyclestroom opgezet met een van onze partners, Biotech Training Facility in Leiden. Hier kregen ongebruikte materialen een tweede leven, maar het aanbod van materiaal was vele malen groter dan hun behoefte. Hierdoor gingen we op zoek naar een manier om deze recyclestroom breder in te zetten. (Bio)farmaceutische startups bieden


hiervoor de perfecte uitkomst. Uiteindelijk kwamen we tot het volgende model: we halen de ongebruikte producten op bij onze klanten, die hiervoor betalen.
Dat leidt tot de reductie van een aanzienlijke afvalstroom, en vermindering van CO2 uitstoot, want zeker 25% van de geleverde producten wordt door onze klanten niet gebruikt en gaat de verbrandingsoven in. De geretourneerde producten leveren we gratis aan startups, en rekenen alleen transportkosten. Ook onze assemblagepartner UltraPure International, heeft voordelen bij deze service. Bij assemblage blijven vaak producten over waar ze niets mee kunnen, die -vaak in kleinere hoeveelheden- eveneens graag afgenomen worden door startups.’
Liesbeth: Waarom is de Retour- en Recycleservice een klassiek voorbeeld van een echte win-win?
‘Ongebruikte producten waarvan de houdbaarheidsdatum is verstreken, worden ingezameld en krijgen een tweede leven bij onder andere farmaceutische startups en trainingsfaciliteiten. Daarin speelt zeer zeker een rol dat startups in de R&D fase maar ook de Biotech Training Facility in hun trainingen wel gebruik mogen maken van producten die over de houdbaarheidsdatum heen zijn. Materialen die niet meer bruikbaar zijn, worden zorgvuldig gerecycled tot nieuwe producten of herbruikbare grondstoffen. Om dit hele systeem goed te laten lopen, heb je naast een goede infrastructuur ook veel kennis en expertise nodig. Stel een startup is op zoek naar een bepaald product. Dan kijken we eerst: ligt dit in het magazijn van de Retouren Recycleservice? Of ligt er iets vergelijkbaars in het magazijn dat als alternatief kan dienen?
Zowel de klant, de startup en onze partner, Biotech Training Facility en assemblagepartner UltraPure International, profiteren van onze nieuwe service. We hebben inmiddels een zeer efficiënt systeem opgezet waarmee we snel kunnen achterhalen of we een product wel of niet tweedehands hebben. Zo geven we een enorme boost aan vrijwel alle stadia van een circulaire farmaceutische industrie.’
Savanna: Waarom is de Retour– en Recycleservice van belang voor de gehele farmaceutische industrie?
‘Het is al lang bekend dat de farmaceutische industrie zeer vervuilend is. De keten van productie, distributie en consumptie van
Over Hitma Hitma is een technische handelsmaatschappij die sinds 1926 producten en service levert aan de proces- en maakindustrie, farmacie, voedingsmiddelenbranche, energiebedrijven, hightech-sector, laboratoria, ziekenhuizen, utiliteitsbouw en installatiebranche. Het team van technische specialisten begrijpt bedrijfsprocessen en knelpunten en levert hiervoor maatoplossingen.
Zeker 25% van de geleverde producten gaat de verbrandingsoven in
geneesmiddelen veroorzaakt uitstoot van broeikasgassen. Inmiddels zijn nagenoeg alle farmaceutische bedrijven op internationaal niveau bezig om hun broeikasgasemissie te verminderen. Sommige grote bedrijven hebben de ambitie om al in 2025 volledig klimaatneutraal te zijn. Binnen Hitma staat duurzaamheid sinds een paar jaar hoog op de agenda. Als Sustainability Consultant bij Hitma ben ik verantwoordelijk voor de implementatie van de duurzaamheidsstrategie, de reductie van emissies en een zo duurzaam mogelijke supply chain. Hitma heeft een officiële duurzaamheidsstrategie samengesteld waarin we streven om in 2050 net zero/klimaatneutraal te zijn. Dat doen we door stappen te zetten in de verschillende scopes op het gebied van mensen, milieu, producten & klanten en ethiek & naleving. Daarnaast zijn we ook druk bezig met de EcoVadis rating, die het duurzaamheidsmanagement van een bedrijf weergeeft in een cijfer, en een duurzame supply chain in samenwerking met leveranciers en klanten. Onze Retour- en Recyclingservice is een unieke stap in het internationale streven om de CO2-voetafdrukin de farma te verminderen.’
‘Ook wij hebben geen totaaloplossing voor een geheel circulaire farmaceutische industrie, ‘besluiten Mitch, Liesbeth en Savanna. ‘Maar we hopen het goede voorbeeld te kunnen geven. Onze hele supply chain profiteert van een win-win situatie. We bewijzen dat er voor ondernemers met lef, doorzettingsvermogen en een goed bestuur vaak meer mogelijk is dan je denkt. En daarmee is de cirkel rond!’


























































































BCF Career Event Belgium – 27 November 2025




















BCF Career Event Netherlands – 21 May 2026

















































BCF Career is the largest Life Sciences career event in the Netherlands since 2005, offering an unparalleled opportunity to connect with thousands of highly skilled job seekers. With participants from Biology, Chemistry, Food, and Pharma, the event brings together pharma and biotech companies, CROs, research institutes, universities, recruitment agencies, and career coaching organisations - all eager to meet and recruit future talent.
Exhibiting at BCF Career Event gives companies the chance to boost their brand awareness, connect with highly qualified candidates, and fill their vacancies with top-tier talent.
Discover this year’s company line-up at BCF Career Event NL & BE!





















Contact us to discuss potential participation bcf@hyphenprojects.nl
+31 35 303 5588
























“For us, it’s not necessarily about an immediate return on investment, but rather a long-term commitment to visibility & engagement. We’re always excited to hear from applicants – now colleagues – who tell us that BCF inspired them to apply” GenDx











The chances for a successful development of a new therapeutic product have proven to be dependent on a strong patent portfolio. When building a patent portfolio around a newly developed pharmaceutical compound platform many opportunities occur to steadily extend the patent protection, using and applying the growing insights during the early stage, pre-clinical and clinical development stages of the new therapeutic.
In early stage of development, new active compounds, such as small molecule compounds or other therapeutic modalities, are identified. Such development is commonly based on a known pharmaceutical target, which is related to a medical indication.
Once the principal activity of the new compounds has been found in (in vitro) models related to the target, a first patent application (called basic patent) can be filed at the relevant patent authority for the new compounds.
A filing of the first patent application can be used to early protect the development of the new pharmaceutical compound platform. For instance, such an early filing may be desirable to reach out to possible investors to pursue investments.
The patent application beneficially contains claims to provide a broad coverage of the chemical structures of the new active ingredients. Commonly for small molecules a Markush formula approach is used to broaden the coverage including relevant variations of substituents of the chemical structures.
After filing the first basic patent application many options remain for getting additional protection for further aspects of the pharmaceutical compound and its application, for example the medical use, the pharmaceutical formulation, the dosage regime and/ or the administration form of the compound. Thinking ahead of all possible building blocks in protection is key to the development of a strong patent portfolio for the new medical compound.
An example of successful protection by a patent portfolio is Keytruda, which is approved across multiple indications. The basic compound patent expiration year is 2028, while the patent portfolio for Keytruda, which is built over time by many
patent applications, gives extensive protection for new indications, formulations, and combination therapies.
However, when not proactively thinking ahead of options along the road for building the patent portfolio and the timings of addressing these options, we see the occurrence of pitfalls in first or other early patent applications which, once overlooked, often cannot be remedied later.
For example, in early stage it is common practice to identify as many medical applications of the new pharmaceutical compound as possible, which could be targeted by the new pharmaceutical compounds. Such a long list of medical indications is then used to select a first focus area of application of the new pharmaceutical compounds and to select suitable models to assess the feasibility for addressing said focus application area.
Concurrently, we see often a tendency to mention all or at least many of these possible medical indications in a first patent application. For example, the target may be very relevant and tested for one or more cancer indications on relevant models, and at the same time no data is available which provide evidence for the use of the same compounds for other medical indications.
However, mentioning a medical indication in a patent application does not guarantee that a protection can be claimed for this medical indication based on this (e.g. first) patent application. Patent law and patent authorities apply stringent requirements to medical use claims (which cover the medical indication), which restrict the possibility for obtaining protection for such medical use claims. For example, in case no model data, which relate to the specific medical indication, is enclosed in the patent application, it is not upfront certain that a patent authority will grant protection. Besides, the requirements in such a situation also differ among the various jurisdictions or patent authorities.
At the same time, mentioning a possible medical indication in general in the first application may also restrict or hinder to obtain protection later, based on another later (secondary) patent application, which claims the same medical indication. Even if the later secondary patent
application does contain relevant data as evidence based on the pharmaceutical compounds, while the first application does not contain the data.
Thus, when building a patent portfolio, and starting from a first patent application, it makes sense to think ahead of further options for protection along the road of pre-clinical development and clinical development and make thoughtful decisions what to include at what time and what not to include in the first patent application and each of the following patent applications.
Even more, the relevancy of a strong patent portfolio for a successful development and introduction of a new therapeutic product can be illustrated by the following quote of a CEO of a successful innovator: “Our patent portfolio is our lifeline or the reason of our existence.”
For any questions for building your patent portfolio, please contact us at:
Kolja Adamczyk; European patent attorney, pharma & life sciences, at NLO
NLO – B.V. Nederlandsch Octrooibureau
Email: Adamczyk@nlo.eu
Telephone: + 31 682789121

Understanding Due Diligence
Due diligence is a thorough investigation and evaluation of an asset before finalizing an investment, partnership, or acquisition. It is one of the last and essential steps before a formal investment is committed. The process ensures that there are no hidden issues or risks that could affect the investment’s success. In the (bio-) pharmaceutical sector, due diligence encompasses a multi-dimensional assessment of scientific, regulatory, financial, and legal parameters to identify and quantify material risks and opportunities. This process is particularly critical in drug development, where the complexity and uncertainty of clinical and commercial outcomes necessitate rigorous scrutiny. For investors and acquirers, due diligence validates the scientific merit, developmental feasibility, and commercial potential of a therapeutic asset, reducing the risk of investing in suboptimal or non-viable technologies. For startups and scaleups, the due diligence process is an opportunity to demonstrate the robustness of their development strategy, the integrity of their data, and the scalability of their operations. A successful due diligence outcome can facilitate fundraising, licensing agreements, or acquisition deals.
Key Focus Areas in Drug Development Due Diligence
Due diligence in drug development typically spans several domains. Scientific and clinical due diligence evaluates robustness of non-clinical and clinical data, including non-clinical safety/efficacy observations and their translation into a clinical setting. Chemistry, Manufacturing, and Controls (CMC) assessments focus on manufacturing processes, formulation stability, and scalability. Regulatory reviews involve regulatory strategies and prior correspondence with regulatory agency (FDA/EMA).
The Role of Mock Due Diligence
Mock due diligence, or gap analysis, is a simulated third-party review that identifies deficiencies or risks before formal investor or partner evaluations
in advance. For emerging biotechs, this step is indispensable. It highlights potential red flags, such as. insufficiently developed analytical methods or inconsistent impurity profiles between GLP and clinical batches, allowing companies to address issues proactively.
Although mock due diligence does not guarantee a term sheet or investment, it significantly enhances the probability of a favorable outcome. It also provides founders with insights into how their assets will be perceived by external stakeholders. Demonstrating transparency, technical competence, and strategic foresight during this process builds trust with potential investors.
Expertise in Drug Development Due Diligence
Venn Life Sciences (“Venn”) offers specialized consultancy services in drug development due diligence, with a focus on CMC, non-clinical and early clinical development, regulatory strategy, quality assurance, as well as health technology assessment via an external partner. Over the years, Venn has conducted more than 100 due diligence and gap analysis projects across a wide spectrum of therapeutic modalities, including small molecules, biologics, antibody-drug conjugates (ADCs), advanced therapy medicinal products (ATMPs), drug-device combination products, and gene therapies. These projects have supported decision-making for investors (including several renowned life-science investors), pharmaceutical companies, and biotech startups.
Venn supported a biotech developing a combination product (ATMP + medical device) by conducting a gap analysis involving all necessary disciplines. The review uncovered a large hiccup in regulatory roadmap, which is considered critical for coming a meeting with investors. Therefore, Venn advised and helped set a regulatory strategy and associated actions to proactively address the foreseeable issues. This proactive approach enabled the biotech company to secure a term sheet, illustrating how the gap analysis prior to the due diligence can
directly influence investment and partnership success.
In drug development ‘s high-stakes environment, due diligence is more than a procedural requirement: it is a strategic imperative. Venn exemplifies the value of integrated due diligence support, combining deep domain expertise with a holistic understanding of the drug development lifecycle. Their track record across diverse modalities and development stages underscores the importance of early, informed, and strategic planning in the pursuit of therapeutic innovation.
Want to learn more? Join our upcoming webinar, featuring three experts: Erik Gout (left, Head of CMC), Arthur Noach (middle: Senior Consultant Non-Clinical Expert) and Katsuhiro Mihara (right, Head of Clinical Development). Scan the QR code to register.


Utrecht Science Park:
Ecosystem Utrecht Science Park important for the Netherlands' earnings capacity and tackling societal challenges

A fertile ecosystem for innovation
With 31,000 employees and 55,000 students, Utrecht Science Park is the biggest science park in the Netherlands. Within this close-knit ecosystem, aptly called “the golden square kilometer” by Mayor Sharon Dijksma, leading organizations collaborate closely, all within walking distance of each other.
A recent example of the network’s economic impact is the announced acquisition of the biotech company Merus, located at Utrecht Science Park, by its neighbor Genmab for €6.8 billion. This ensures that valuable knowledge remains in the Netherlands and strengthens Utrecht’s position as a “powerhouse of the biotech sector”, according to financial analyst Sebastiaan van der Schoot (in Financieele Dagblad). The ecosystem also gives rise to numerous start-ups and scale-ups, such as Gyes, NanoCell Therapeutics, GenDx, and Hub Organoids.
Strong scientific foundation
What makes Utrecht Science Park unique is its powerful combination of academic institutions, healthcare organizations, research institutes, and R&D companies. The strong scientific foundation is formed by Utrecht University (one of Europe’s leading research universities), UMC Utrecht (one of the largest public healthcare institutions in the Netherlands), and the Princess Máxima Center (Europe’s largest pediatric oncology center). These are complemented by prominent research institutes such as the Hubrecht Institute, Westerdijk Institute (both part of the KNAW), RIVM, and TNO.

With more than 55,000 students, Utrecht Science Park is an important source of talent. Each year, over 12,000 graduates enter the labor market, strengthening the regional workforce and making the area attractive for business. The Graduate School of Life Sciences, with nearly 4,000 PhD and Master’s students, is the largest in Europe. The young, highly educated population has helped make the Utrecht region the most competitive in Europe (Regional Competitiveness Index, 2022). Through internships, challenges, and educational programs, Utrecht University and HU University of Applied Sciences Utrecht foster collaboration between students, companies, and institutions. University-linked organizations such as UtrechtInc and the Centre for Entrepreneurship also support students and start-ups in transforming knowledge into entrepreneurship.
Utrecht Science Park plays a crucial role in driving employment and business growth. According to a 2024 report by Buck Consultants International, the park with 31,000 employees serves as a major job engine, spanning vocational to academic levels, for the city, region, and country. Since 2018, employment at R&D companies has grown by 76%, nearly three times faster than the Utrecht average, and the number of companies has increased by 64%. This underscores the importance of continued investment in knowledge and innovation.
Through its innovation ecosystem, Utrecht Science Park makes a significant contribution to national and European policy goals. Investments in R&D and innovation strengthen economic resilience and help maintain public services. The park is a key hub for both economic and societal impact, building on strong research and innovation centers.
It particularly distinguishes itself in biotechnology and medical technology, with expertise in areas such as oncology (immuno-, cell-, and gene therapy), regenerative medicine, organoid and stem cell research, infectious diseases, and AI applications.
It is therefore a logical step that, starting in 2026, the leading conference Innovation for Health and the Global Investor Forum will be hosted in Utrecht for multiple years. The Global Investor Forum (March 25) will take place at Utrecht Science Park, and Innovation for Health (March 26) will be held at the Royal Jaarbeurs. Several Utrecht-based organizations within the life sciences & health ecosystem, including Dutch Health Hub, Utrecht Science Park, Oncode Accelerator, ROM Utrecht Region, and the Municipality of Utrecht, are jointly shaping the program and organization. Their combined efforts convinced event organizer Hyphen Projects: Utrecht is the place to be.

Sources: Utrecht Science Park, Genmab, Financieel Dagblad
The Danish-Dutch biotech company Genmab is acquiring its Dutch counterpart Merus. Both companies have their main facilities next to each other at Utrecht Science Park. Genmab is paying $8 billion (€6.8 billion) for its neighbor, thereby acquiring a promising experimental drug for head and neck cancer and colorectal cancer.

High expectations for petosemtamab drug
Genmab is considered one of the most successful biotech companies in Europe, with a market capitalization of approximately €15.8 billion. The company is headquartered in Copenhagen and its main R&D center is located at Utrecht Science Park, where approximately 700 people work. The company is led by CEO Jan van de Winkel. Merus was founded by Ton Logtenberg, who was previously a professor of immunology at Utrecht University at the same time as Van de Winkel.
The high acquisition price for Merus is primarily based on the high expectations for the head and neck cancer drug petosemtamab. Merus’s share price had already soared earlier this year following the publication of new interim research results. Of 43 seriously ill patients, 34 were still alive twelve months after treatment with petosemtamab.
Advanced form of colon cancer
Shares in Merus rose significantly in value in May after the pharmaceutical company published positive research results for its experimental drug
against head and neck cancer. The company is also testing the efficacy of the same drug against an advanced form of colon cancer, a larger and potentially even more lucrative market.
“The proposed acquisition of Merus clearly fits our long-term strategy,”

Van de Winkel said in a joint press release from the two companies. He stated that the acquisition significantly advances his goal of making Genmab a global biotechnology leader.
Genmab is paying in cash. The company is also taking out $5.5 billion in debt financing. Shares in the Danish-Dutch biotech company have already risen by a fifth this year. Investors reacted cautiously: Genmab shares fell 2.6% on the Danish stock exchange.
‘Utrecht as a powerhouse of the biotech sector’ The knowledge and expertise of the successful

biotech company Merus will be retained in the Netherlands. According to experts, this is the most important consequence of this acquisition. “If it had been acquired by a foreign pharmaceutical company, there was a good chance that Merus and all its activities would have left the Netherlands,” says Ton Logtenberg, founder and former CEO of Merus.
“This deal will make Utrecht a true powerhouse of the biotech sector,” says Sebastiaan van der Schoot, financial analyst at investment bank Van Lanschot Kempen. Jan van de Winkel also indicates that the development of new medicines in the Netherlands will receive a tremendous boost thanks to this acquisition: “This is an exceptionally powerful conglomerate of research and drug development capacity. Genmab has built an impressive track record, and now we will be bringing our own medicines to market. I think that’s very good for the Netherlands.”

Regeneratieve Regeneratieve & Cellulaire & Cellulaire
Industrie Industrie
Herbruikbare cleanroom oplossingen voor 1 productiebatch, 1 leven. Ondersteuning van deze industrie in hun levensreddende missie.
Wij hebben de kledingoplossing die u nodig hebt!

Door te kiezen voor Elis Cleanroom investeert u niet alleen in de juiste Cleanroomkleding, maar kiest u ook voor een partner in herbruikbare wasoplossingen die zich inzet voor de toekomst met duurzame, kosteneffectieve en innovatieve diensten.

Eurofins BioPharma Product Testing, is één van de grootste, Good Manufacturing Practice (GMP) geaccrediteerde test laboratoria netwerken wereldwijd. 8200 experts werken vanuit meer dan 50 state-of-the-art laboratoria in ruim 20 landen. Wat het uniek maakt, is dat het klantcontact, ondanks de omvang van het netwerk, onveranderd is gebleven. Klanttevredenheid blijft het primaire doel. Lokaal zorgen korte communicatielijnen, efficiënte, en flexibele processen ervoor dat klanten zo optimaal mogelijk bediend kunnen worden.
Wie daar alles van weet, is Marianne Budde, Business Unit Manager Manufacturing, die sinds 16 jaar bij Eurofins BioPharma Product Testing Netherlands (Eurofins BPT NL) werkt, en daarvoor in de labs van PROXY en MicroSafe Laboratories. ‘Onze klanten van start-ups tot grote farmaceutische bedrijven, bieden we een state-of-the-art dienstenpakket zoals een breed scala aan GMP testing services, maar ook fill & finish en compounding services, allemaal volgens ons ‘one-stop-shop’ principe. Eurofins BioPharma Product Testing is een netwerk van laboratoria en experts wereldwijd, waardoor wij zowel lokaal en als netwerk een compleet dienstenpakket kunnen bieden aan onze klanten.’
Klantbetrokkenheid staat centraal
‘Onze laboratoria zijn gespecialiseerd in GMPdiensten voor farmaceutische productie en kwaliteitscontrole. Van het testen van grondstoffen en eindproducten tot small-scale compounding,
Eurofins BioPharma Product Testing Netherlands is part of the the international Eurofins BioPharma Product Testing network of harmonised product testing laboratories. We have many years of experience serving the (bio)pharmaceutical industry with laboratory, manufacturing, and consultancy services. Our laboratories offer a broad range of methodologies, operating under GMP authorisation and accredited to ISO standards. Our services are fully comprehensive and include testing of drug substances, final products, intermediates, starting materials for both small and large molecule drug products. We can support our customers with developing test methods from scratch or transfer client methods under regulatory compliant conditions. Aside from testing, we have multiple aseptic manufacturing units for small-scale compounding and fill & finish activities, including labelling and packaging services. We also support customers with QP release, ensuring their products can be brought to the market reliably and in full compliance. Leveraging our international network of Eurofins companies, we can offer our customers a comprehensive service portfolio tailored to their product, while ensuring full regulatory compliance throughout the development process.
fill & finish en zelfs QP-release: onze lokaal ingerichte processen bieden klanten efficiënte, flexibele en op maat gemaakte dienstverlening, van begin tot eind. Onze manufacturing afdeling telt een klein, deskundig team met jarenlange ervaring. De korte lijnen zorgen voor efficiënte samenwerking en vele voordelen voor onze klanten. Zo zijn we betrokken bij iedere klant of het nu een startup is die advies vraagt, of een grote farmaceut: als we testen of afvullen hebben we niet alleen kennis van het product, maar ook van het productieproces, de bijbehorende uitdagingen en het uiteindelijke doel voor de patiënt. Klantbetrokkenheid staat centraal: wijzigingen worden tijdig gecommuniceerd en klantgerichte oplossingen snel gerealiseerd, zodat vertragingen worden voorkomen, zeker omdat
strakke tijdslijnen vaak cruciaal zijn binnen onze samenwerking. Dit creëert wederzijds vertrouwen en vormt de basis voor een succesvolle samenwerking. En wanneer we een bepaalde expertise niet in huis hebben, dan is er altijd wel een laboratorium binnen ons netwerk dat wel over de juiste expertise beschikt. De bundeling van kennis en expertise biedt vele voordelen. Wereldwijd hoeven we klanten nooit ‘nee’ te verkopen. Het geeft een goed gevoel om echt iets te kunnen bijdragen.’
Alles onder 1 dak: Ruimte voor groei ‘Efficiënt is ook is dat we fysiek bij elkaar gevestigd zijn op het Leiden Bio Science Park. Eurofins PROXY Laboratories en Eurofins MicroSafe Laboratories, beide gevestigd in Leiden, liggen op minder dan vijf minuten afstand van elkaar. De aanhoudende groei van beide laboratoria maakte uitbreiding van de capaciteit noodzakelijk, met als doel de efficiëntie en kwaliteit van onze dienstverlening aan klanten te waarborgen. Gelukkig biedt het nieuwe pand, dat momenteel wordt gebouwd op het Leiden Bio Science Park, te Oegstgeest, een oplossing voor de capaciteitsuitbreiding. Naast een substantiële vergroting van onze capaciteit stelt dit ons in staat om een compleet en geïntegreerd pakket aan ‘testing services’ vanuit één locatie aan te bieden, zowel voor Biologics als voor Small Molecule Drugs. Hierdoor kunnen we onze klanten nog meer efficiëntie en kwaliteit bieden, met een dienstverlening die nog beter afgestemd is op hun specifieke behoeften.’


CP Services is de gespecialiseerde merkonafhankelijke servicedienstverlener op het gebied van validatie, kalibratie, procesonderhoud en modificaties van cleanrooms en procesinstallaties. In een sector waar betrouwbaarheid en compliance regelgeving de norm zijn, is CP Services een naam die steeds vaker valt. Ontstaan uit het gespecialiseerde Kropman Contamination Control, onderscheidt CP Services zich sinds 1 januari 2024 als zelfstandig bedrijf binnen de farmaceutische- en biotechwereld. En met reden: hun dienstverlening ademt compliance, hun aanpak is oplossingsgericht, en hun missie sluit naadloos aan op de behoeften van gereguleerde omgevingen. De diensten zijn afgestemd op de eisen binnen de farmaceutische en life sciences sector: een schot in de roos naar verdere ontwikkeling en professionalisering van het servicegerichte bedrijf. Dat bleek recent onder meer uit het starten van een nieuwe locatie op het Pivot Park te Oss. Directeur Carlos Vendrig en Engineer Anton Willems aan het woord.
De nieuwe vestiging op het PivotPark in Oss markeert een belangrijke mijlpaal in de landelijke dekking van het bedrijf, ‘stelt Carlos. ‘Door onze positie in het hart van de Nederlandse farma industrie op deze manier te versterken, wordt onze
ambitie om biotechbedrijven en farmaceutische startups nog dichter bij huis te ondersteunen in hun groei en innovatie, realiteit.’Wat deze vestiging bijzonder maakt, is de toevoeging van een Engineering-afdeling. Deze afdeling richt zich o.a. op procesanalyse, energie-efficiëntie, capaciteitsuitbreiding en compliance-advies. Denk hierbij aan het optimaliseren van het aantal luchtwisselingen, het implementeren van energiezuinige HVAC-systemen en het begeleiden bij GMP Annex 1 wijzigingen. ‘Tevens levert de afdeling support aan bestaande serviceactiviteiten. Hierdoor kunnen wij onze klanten direct ondersteunen met oplossingen die zowel praktisch uitvoerbaar als toekomstbestendig zijn.’
Duurzame oplossingen implementeren ‘Wij gaan verder dan het signaleren van een afwijking, ‘vertelt Anton. Bijvoorbeeld bij het testen van HEPA-filters kijken we naar de volledige constructie: het filter, de behuizing, de aansluiting en de luchtdichtheid van het geheel. Bij het constateren van een lekkage – of dit nu in het filter of in de constructie zit – wordt niet alleen gerapporteerd, maar ook direct een oplossing geboden. In overleg met de opdrachtgever wordt de lekkage gerepareerd en/of het filter vervangen, zodat het systeem weer voldoet aan de gestelde eisen. Deze proactieve en oplossingsgerichte

Referentie Produlab Pharma
CP Services B.V. is onderdeel van Eiffage Energiesystemen (73.500 medewerkers in 70 landen) één van de 4 businesslines binnen de Eiffage Groep. Door de stabiliteit en kracht van een grote internationale groep te combineren met de flexibiliteit en nabijheid van haar dochterondernemingen ontstaat een krachtige synergie. Het portfolio omvat een breed spectrum aan hoogwaardige diensten waaronder:
• Cleanroom kwalificatie
• Kallibratie en meetdiensten
• Procesmodificaties aan Clean Utilities (vloeistof en gasdistributie)
• Preventief en correctief onderhoud
• Engineering en procesoptimalisatie
werkwijze voorkomt vertragingen in productieprocessen en garandeert compliance met GMP-richtlijnen.
Opleiden in eigen huis
Het aantal technische vakmensen in de markt neemt af. Bij opdrachtgevers in de farma en Life Science ziet CP Services steeds vaker dat serviceactiviteiten worden uitbesteed. Het bedrijf speelt hierop in door een breed dienstenpakket aan te bieden waarmee een complete shutdown in samenspraak met de opdrachtgever volledig kan worden uitgevoerd- van planning, begeleiding en uitvoering tot en met rapportage inclusief QC-beoordeling. Een belangrijk voordeel van werken met CP Services is dat hun medewerkers multidiscipliair zijn opgeleid. Hierdoor kunnen zij meerdere taken binnen een shutdown uitvoeren, wat leidt tot efficiëncy voordeel. Minder schakelmomenten, kortere doorlooptijden en betere afstemming tussen disciplines.
Om aan geschikt personeel te komen voor meerdere disciplines, heeft CP Services een eigen opleidingscentrum opgezet in Lijnden. Dit centrum beschikt over een cleanroom voorzien van een lafplafond, een kalibratielab, instrumentenlab met apparatuur en een RVS prefabwerkplaats. Hier worden zijinstromers en nieuwkomers in de techniek opgeleid in cleanroom- en procestechniek.

Een concreet voorbeeld van deze praktijkgerichte opleiding is orbitaallassen: een gemechaniseerde TIG lastechniek die essentieel is voor het realiseren van hygiënische en lekvrije verbindingen in farmaceutische procesinstallaties. Nieuwe medewerkers leren hier niet alleen de techniek zelf, maar ook de bijbehorende kwaliteitsborging en documentatie die vereist is in GMP-omgevingen. Zo worden ze klaargestoomd voor een carrière in de farmatechniek, met een stevige basis in zowel theorie als praktijk.
‘Het is fijn werken bij CP Services, ook omdat je daadwerkelijk meerwaarde kunt bieden in oplossingen en compliance voor onze opdrachtgevers, ‘stelt Anton. Onze infrastructuur versterkt niet alleen onze eigen expertise, maar draagt tevens bij aan het kennisniveau en de compliance zekerheid van onze partners. Kwaliteit wordt daarbij geborgd door een eigen QA/QC afdeling die conform ISO 9001 en op GMP-niveau toeziet op de betrouwbaarheid van bestaande en nieuwe diensten.’
‘Als volledig merkonafhankelijke servicepartner voeren we onafhankelijke inspecties, validaties en kalibraties uit voor uiteenlopende installateurs en eindgebruikers, ‘besluiten Carlos en Anton. ‘Daarmee garanderen we objectiviteit, betrouwbaarheid en een hoge mate van compliance. Precies de waarden waar wij voor staan.’ Voor bedrijven in de Farma en biotech die willen groeien zonder concessies te doen aan kwaliteit, is CP Services de logische keuze.
Produlab Pharma: ‘CPservices is een fijne club om mee te werken’ ‘De contacten tussen CP services (voorheen Kropman) en Produlab Pharma dateren al van een jaar of 10 geleden,’ zegt Peter Zijlmans, head of technical services bij het in Raamsdonkveer gesettelde bedrijf gespecialiseerd in vloeibare en halfvaste diergeneesmiddelen.
De grote kennis en expertise, zowel algemeen als van de specifieke situatie binnen Produlab, maar vooral de korte, directe lijnen binnen CP services maken het volgens Peter een fijne club om mee te werken. Dat blijkt niet alleen uit de efficiënte werkcontacten, maar ook uit het maandelijks overleg bij Produlab waar ook de medewerkers van CP services aanwezig zijn. ‘Ook is het prettig dat CP ons werk uit handen neemt en creatief meedenkt, bijvoorbeeld door zelfstandig input aan te leveren voor ons onderhoudsbeheersysteem.’
Die kennis en expertise van CP services was vooral van groot belang toen in 2018 werd besloten de fabriek van Produlab niet alleen te upgraden maar ook uit te breiden tot een (papierloze) productiefaciliteit van ruim 22.000 m2. CP services verzorgde toen de kwalificatie van de cleanrooms en de kalibratie van de sensoren van het proces,
Vulmachine Produlab Pharma

Voor meer informatie www.cpservices.nl info@cpservices.nl
Vacatures: https://www.cpservices.nl/werken-bij
HVAC en monitoringssysteem.
Door onze state-of-the-art fabriek en productiefaciliteiten die in 2020 werden opgeleverd, kunnen we flexibel inspelen op iedere vraag naar orale en steriele vloeibare en halfvaste medicijnen. Of het nu om een batch van 1 liter gaat of 2.000 liter, of om 5 ml injectieflacons of 500 ml orale vloeistoffen (en alles wat daartussen zit) mede door de ondersteuning van CP services kunnen wij op onze beurt onze opdrachtgevers flexibel en adequaat ontzorgen.’
Onze werkzaamheden bij Produlab:
Kalibratie
-EMS Systeem
-Proces installaties
-Productie installaties
-Sterilisatie tunnel
Proces onderhoud
-PW / WFI installaties
-Formulatie installaties
-CIP / SIP installaties
Validatie
-Cleanroom validatie
-Depyrogenisatie tunnel
-UDF systemen incl RABS

At Pivot Park in Oss, we provide a go-to location for early-stage biopharmaceutical start-ups. A unique combination of state-of-the-art R&D facilities, tailored entrepreneurial support and a vibrant pharmaceutical ecosystem. The Pivot Park Incubator in Oss is the ideal launchpad for young life-science companies ready to accelerate their growth. To accelerate biotech innovation, we combine readyto-use R&D facilities with hands-on entrepreneurial support within a connected pharma ecosystem.
Fully Equipped and Ready to Use
All Incubator laboratories are now completely fitted out and operational, for emerging biopharma ventures. Each start-up receives a dedicated, flexible lab bench with a core set of calibrated equipment, while a shared-lab model grants access to specialized resources such as ML-I cell culture suites, microbiology and microscopy labs, centrifuges, fume hoods, and water baths.
Personal refrigerators and freezers, as well as shared storage for cell lines and samples, ensure secure handling of valuable materials. Essential utilities, CO2, nitrogen, vacuum, ice, and compressed air, are provided as standard. Our onestop infrastructure includes utilities and shared high-end equipment maintained to Dutch/EU standards, giving founders peace of mind on quality and safety. All lab work is supported by Sci-Notes, which underpins traceability, data integrity, and reproducibility across every bench.
New Momentum: Open Access Lab
As an additional facility, Pivot Park is currently transforming space into a brand-new Open Access Lab. This cutting-edge environment will soon offer entry to advanced analytical technologies and high-end equipment normally reserved for large pharmaceutical companies or academic centers.

The result: faster and more efficient R&D cycles and the ability to scale up more quickly.
“The Open Access Lab will be a true gamechanger,” says Donna Algra, Business Development Manager at Pivot Park. “It gives start-ups the opportunity to perform complex analyses without investing in costly instruments, lowering barriers and speeding innovation.”
Smart Support and a Strong Community
Beyond world-class laboratories, the Incubator delivers a full spectrum of business and technical support. A dedicated lab manager is on site daily to help with operations and maintenance of shared equipment. Entrepreneurs also gain access to experienced mentors, subject-matter experts, and business coaches.
More than sixty biopharmaceutical companies already call Pivot Park home, creating a dynamic network where knowledge sharing, collaboration, and direct connections to investors are part of everyday life.
Ready Resources, Rapid Start
Because speed is critical for early-stage companies, the Incubator keeps research resources and consumables in small on-hand stocks. Start-ups can begin experiments immediately, without waiting for large orders or long lead times.
Growth
The recent expansion means more room for new ventures eager to establish their R&D at the Oss campus. With fully equipped labs, access to shared high-end instruments, and comprehensive business support, Pivot Park Incubator offers one of the most attractive landing spots for biopharmaceutical innovation.
Start your journey today!
Pivot Park is where biopharmaceutical entrepreneurs find the community, space, standards and support to turn ideas into medicines. Discover how Pivot Park can accelerate your biotech venture. Connect directly with Donna Algra, Business Development Manager, to explore opportunities: donna.algra@pivotpark.com


Dale Wright and Brian Bond from Inotiv discuss the importance of taking a holistic approach to glucagon-like peptide-1 research using systems pharmacology. The therapeutic potential of targeting glucagon-like peptide-1 (GLP-1) has become increasingly evident over the past 20 years, both as notable diabetes and weight loss drugs, such as exenatide, liraglutide, and semiglutide, and as drug candidates beyond insulin regulation. Research in this field has progressed from a niche focus on diabetes to a powerful therapeutic platform with far-reaching implications for human health.
Looking beyond GLP-1 as a singular drug target and considering its multifaceted roles in metabolism may help scientists uncover treatment opportunities in cardiovascular and neurological diseases. In this Innovation Spotlight, Dale Wright, executive director of inflammation & systems biology and Brian Bond, vice president of drug discovery and translational sciences at Inotiv, discuss the importance of taking a holistic approach to GLP-1 therapeutic research using systems pharmacology.
Where did the GLP-1 research field begin and how is it evolving?
Brian Bond: Initial research discoveries in the 1980s and 1990s highlighted GLP-1’s role in glucose metabolism and appetite regulation, paving the way for the first GLP-1 receptor agonists (GLP-1 RAs) used for type 2 diabetes treatment.1 Since then, the field has evolved dramatically. The success of early GLP-1 therapies, such as exenatide and liraglutide, inspired further research into their broader metabolic and cardiovascular benefits.
Dale Wright: Today, GLP-1-based drugs are revolutionizing not only diabetes management but also obesity treatment, and show promise in addressing conditions such as metabolic dysfunction-associated steatohepatitis (MASH), cardiovascular disease, and neurodegenerative disorders. Advances in drug design, such as dual and triple agonists (e.g., GLP-1/gastric inhibitory peptide (GIP) and GLP-1/GIP/glucagon receptor agonists), are expanding the therapeutic impact of these molecules, making GLP-1 one of the most exciting areas in drug development.
What makes GLP-1 RAs particularly enticing for scientists doing drug discovery research?
Bond: GLP-1 RAs offer a unique combination of metabolic benefits, which makes them highly attractive for drug discovery. Unlike traditional
insulin therapies, GLP-1 RAs enhance insulin secretion only when blood glucose levels are elevated, reducing the risk of hypoglycemia. By acting on the central nervous system, GLP-1 RAs decrease appetite and promote weight loss, making them valuable in obesity treatment. Additionally, emerging evidence shows that GLP-1 therapies lower cardiovascular risk, reduce inflammation, and improve lipid metabolism, independent of glucose control and weight loss, opening doors to broader applications. Preclinical and clinical studies also suggest that GLP-1 RAs may slow cognitive decline and provide therapeutic benefits in neurodegenerative diseases such as Alzheimer’s and Parkinson’s diseases. These multi-faceted effects are driving intense research efforts to develop more potent and longer-lasting GLP1-based therapies as well as the discovery of mechanism related new targets.
What are systems biology and systems pharmacology? How do these approaches uniquely support therapeutic research?
Wright: A systems biology or a systems pharmacology approach is a data-driven approach that integrates multi-omics and network biology to understand disease mechanisms at a holistic level. Applying these insights can help one understand how drugs interact with biological networks and optimize drug target interactions and safety. These advanced methodologies expedite drug development, bridging the gap between preclinical research and clinical success. Inotiv is committed to partnering with researchers to unlock deeper drug discovery insights through this approach.
Bond: Inotiv’s drug discovery engine strategically runs with the critical components that are required to provide insights into the mechanism of therapeutic intervention, the safety of novel therapies, and the predicted clinical success of drug
discovery programs. By integrating expertise from early drug discovery through investigational new drug (IND) filing and beyond, Inotiv streamlines the path from discovery to clinical success.
What are the disease areas that Inotiv is interested in as the next frontier for GLP-1 therapeutic research?
Wright: One of many benefits that Inotiv brings is that the drug discovery engine is strategically built to support many different therapeutic and disease areas. While diabetes and obesity remain the core areas of GLP-1 drug development, GLP1 has been shown to affect multiple pathways branching into therapeutic and disease areas such as cardiovascular, neurodegenerative, and gastrointestinal disease. Inotiv’s sophisticated bioanalytical and system pharmacology platforms, coupled with in vitro and in vivo models across many disease areas, help bridge the gap between preclinical findings and human applications. This approach aims to de-risk drug development across numerous therapeutic programs.
What also excites researchers is the convergence of pharmacology, AI-driven drug discovery, and personalized medicine. The potential for once-weekly or even oral GLP-1 formulations, novel receptor co-agonists, and precision medicine approaches could transform treatment paradigms across multiple diseases.
A year ago, I probably would have found it hard to imagine a world where my day would be less about capturing image stacks and more about stacked calendars. Yet here I was—a freshly minted postdoc who had stepped out of academia’s familiar chaos into the vast, varied world of industry. And judging by the crowd at Hyphen Project’s BioBusiness Summer School 2025, I wasn’t alone. The room brimmed with PhDs and postdocs plotting their own moves beyond the bench, CVs ready and LinkedIn pages gleaming.
The “great migration” from academia to industry isn’t just a trend anymore—it’s practically a rite of passage. Some are drawn to startups where one person wears five hats; others prefer the stability and reach of big pharma. And many simply wonder: what happens when you take a scientist out of the lab but not the science out of the scientist?
What I’ve learned—through my own leap and from the stories around me—is that no two transitions look the same. Some use postdoc years as a springboard; others skip it altogether. Startups often value versatile academic skill sets, while larger companies increasingly appreciate analytical agility and persistence—qualities honed by research life itself.
So, whether you dream of launching a biotech startup or joining a global firm, this is my perspective as a former postdoc: a candid snapshot of that uneasy, exciting space between science and business—and how one program helped make sense of it.
The BioBusiness Summer School: Learning the Language of Industry
into what it truly takes to move from academia to industry.
The first lesson? It’s not you—it’s the market Understanding macroeconomic currents matters. When ECB interest rates rose post 2022, funding dried up, hiring slowed, and competition stiffened. You can excel technically, but if you ignore market realities, frustration follows.
Second, not all “obvious” transitions are straightforward. Writing and communication roles attract many academics but are often oversaturated. Knowing the nuances between a medical writer, technical writer, or science editor can make or break your strategy.
Preparation emerged as another recurring theme—especially for internationals facing visa and cultural barriers. One speaker’s advice stuck with me: build it before you need to bank on it. Networking, learning, and developing “power skills” (as opposed to “soft skills”)— communication, adaptability, and self awareness— pay long term dividends in any industrial role.
A memorable session by the CTO of Single Cell Discoveries focused on generational dynamics in the workplace. Managing millennials and Gen Z, he said, means balancing freedom and responsibility. His hiring philosophy—choose character and aptitude over experience—was refreshingly pragmatic, especially for startups or scale ups. As postdocs, we often underestimate the edge our curiosity and rapid learning provide. Still, bridging communication gaps across generations remains vital to effective teamwork.

started companies out of passion, others out of sheer frustration. Whatever the motivation, aligning vision with sustainability was the common thread.
The week closed with a keynote from Jaan Winkel, CEO of Genmab, whose story tied it all together. He spoke of two scientist archetypes: the curiosity driven and the application driven. The latter, he said, often thrive in biotech or pharma, where discoveries turn tangible faster. His emphasis on evolution—adapting as Genmab moved from research to commercialization—felt especially relevant for anyone reinventing themselves. Despite leading a global organization, his message stayed grounded: success comes from sharp focus and a genuine commitment to improving patient lives.
Leaving academia isn’t merely about changing jobs—it’s a mindset shift. The BioBusiness Summer School turned an intimidating unknown into a map of opportunities built on adaptability, collaboration, and purpose. It reminded me that while we may leave the bench, the habits that make us scientists—resilience, curiosity, and critical thinking—are precisely what help us thrive beyond it. Crossroads
When a former colleague recommended the BioBusiness Summer School in the Netherlands, I went in with modest expectations. I imagined it as another entrepreneurial boot camp, but it turned out to be a compact masterclass on how life sciences and business intersect. In five intensive days, we covered everything from IP and venture capital to regulatory affairs, business models, program management, and personal branding. More importantly, it offered unflinching insight
What inspired me most were the diverse transition stories. One alumnus shared how he started as a part time business developer, leveraging his academic network until he moved full time. Others pursued academic consultancy, partnering with TTOs to broker public private collaborations. These examples showed there’s no single “right” way out of academia—only creative, well timed entries. Entrepreneurship, too, wore many faces. Some

Are you ready to make the move from the lab to industry? Join the renowned BioBusiness Summer School 2026 – a five-day intensive programme designed to give you a real-world edge in the life sciences sector. Held in Amsterdam from 22–26 June 2026, this immersive experience is ideal for PhD candidates, post-docs and early-stage professionals.
» In-depth interactive lectures from industry leaders covering product development, business models, intellectual property rights, regulatory affairs and finance.
» Hands-on case studies, company visits and real-life business simulations to bridge the gap between academia and industry.
» A powerful international network of peers and experts. Collaborate, connect and build relationships that last.
Who Should Attend:
» PhD candidates and post-docs exploring a career outside academia
» Young professionals in life sciences seeking business skills
» Scientists, technologists and innovators ready to expand impact
Sponsored by Powered by


Kennis delen en partneren, samenwerken met vertrouwen. Voor Niels Höfte, algemeen directeur van Interflow is het noodzakelijk om -ook internationaal- een prominente positie in te nemen in healthcare, biotech, pharma en hightech. Stuk voor stuk markten die sterk in beweging zijn; niet alleen in Nederland maar ook internationaal. Sterke voorbeelden hiervan zijn de ontwikkeling en realisatie van circulaire cleanrooms in Oostenrijk voor de Biotech en in Eindhoven voor de Hightech-sector die door de klant wordt geleased. “Een mooi voorbeeld van wat je kunt bereiken als je samenwerkt in de gehele keten en hoe je de klanten net dat stapje extra kunt geven”, aldus Höfte.
Niels Höfte is inmiddels bijna anderhalf jaar de nieuwe algemeen directeur van Interflow in het Noord-Hollandse Wieringerwerf. Höfte is niet onbekend met Interflow. Na zijn studie Technische Bedrijfskunde aan de TU Twente en zijn carrière bij Stork kwam hij elf jaar geleden binnen bij Interflow. De laatste jaren daarvan in de functie van operationeel directeur, alvorens hij Bart-Jan Freriks opvolgde die binnen de BAM-organisatie zijn loopbaan vervolgt.
cleanrooms en operatiekamers en aanbieder van validatiediensten. Höfte: “Continu wordt er doorontwikkeld in alle disciplines, maar echt grote stappen en waar we het verschil maken is op duurzaamheid, modulariteit en industrialisatie. Op al die vlakken hebben we de laatste jaren een enorme sprong gemaakt wat geresulteerd heeft in circulaire cleanrooms met een EPD certificaat. Alle bouwstenen van de cleanroom worden als ‘legostenen’ geproduceerd en zijn voor 90 procent direct herbruikbaar. Slechts 5 procent wordt gebruikt voor restwarmte en de rest kunnen we ‘refurbishen’. Sterke argumenten om klanten te overtuigen”, vindt Niels Höfte.
Duurzaamheid is niet het enige argument; ervaring, kennis en de nieuwste technieken zijn evengoed belangrijk. Samenwerken met partners is vereist: “We delen onze kennis, zelfs met concurrenten als dat nodig is. Als je samenwerkt met partners is het verschil wat je kunt maken in product ontwikkeling en expertise voor de klant veel groter. Zeker, je moet elkaar dan wel vertrouwen en de kaarten niet voor de borst houden. Openheid en eerlijkheid zijn belangrijk en sterke waarden van Interflow.”
Omdat ze behandelingen maken voor patiënten met erfelijke ziekten willen ze zo dicht mogelijk bij hun patiënten zijn. Een ander aspect is de modulaire opzet van bijvoorbeeld farmaceutische productielijnen. En vergeet ook niet dat the time to market van bijvoorbeeld technologische ontwikkelingen of medicijnen steeds korter moet worden. Dat heeft ook gevolgen voor de doorlooptijden van de cleanrooms. Dat je binnen slechts een paar maanden een cleanroom moet kunnen aanbieden, ontwerpen, en opleveren is geen uitzondering meer. De vraag is meer: hoe snel kun je bouwen.”
De modulaire aanpak, de filosofie van de bouwstenen die Interflow voor alle disciplines heeft ontwikkeld en in haar digitale bibliotheek heeft opgeslagen, is dan in het voordeel. De voorwaarden zijn er: vorig jaar werd, bij de viering van vijftigjarig bestaan, een nieuwe volledig gerobotiseerde productiehal in gebruik genomen. Maar, benadrukt Niels Höfte, robots werken snel maar de mens blijft belangrijk: “We zijn een goed team. Ervaren mensen met kennis en motivatie. Mensen met blauw bloed, zeggen we hier: blauw is de originele bedrijfskleur. De sfeer en mentaliteit van een familiebedrijf is er nog altijd.”
"We zitten dan wel in de Noord-Hollandse polder, maar onze impact reikt veel verder. Voor onze relaties maakt afstand geen verschil: of het nu een biotechbedrijf uit Zwitserland is of een farmaceut uit Amerika, zij kiezen niet op locatie maar op kwaliteit en snelheid. En precies daarin maken wij het verschil, in heel Nederland en ver daarbuiten”.
"Ik heb meegebouwd aan waar we nu staan en ben trots op wat we samen hebben bereikt. Het delen van kennis, het versterken van partnerships en het ontwikkelen van nieuwe producten en diensten zijn essentieel om onze positie te versterken. Interflow zet hier vol op in met drie pijlers: industrialisatie, digitalisatie en duurzaamheid. Voor mij ligt er een mooie uitdaging om samen met ons team en partners verder te bouwen aan innovatieve oplossingen die de standaard in onze sector bepalen”. Interflow
In die elf jaar leerde hij Interflow door en door kennen. “En nee, ik heb niet de behoefte naar elders te vertrekken. Dit bedrijf heeft zoveel unieke elementen, zoals onze mensen, de diversiteit van de klanten en de complexe projecten waarvan het hart sneller gaat kloppen.” Een van die complexe projecten is de bouw van een pilot plant voor de productie van radionucliden in Breda. “Een eer om dat te mogen doen, je werkt mee aan de allernieuwste technologie”, aldus Hófte.
Op het gebied van cleanrooms heeft Interflow in de achterliggende vijf decennia een uitstekende reputatie opgebouwd. Begonnen met de bouw van en verkoop van Laminar Airflow Units groeide Interflow uit tot ontwikkelaar en bouwer van

Niet alleen in de hightech-sector maar ook in de life sciences in de breedste zin zal de dynamiek toenemen. Bijvoorbeeld omdat de wensen qua omvang of energiezuinigheid van de cleanrooms sterk veranderen, zegt hij: “Dat kan van klant tot klant enorm verschillen en dat moet je aankunnen. Ook in de life sciences die internationaal een groeimarkt is. Daarom zijn we steeds meer internationaal gericht, maar wel selectief. We zijn nu actief in zeven landen in Europa en volgen hierbij onze klanten over de grens”.
Sinds de laatste jaren is schaalbaarheid, snelheid, en flexibiliteit belangrijker geworden. Höfte: “We werken bijvoorbeeld voor een pharma-bedrijf dat meerdere cleanrooms door heel Europa wil.


Subjects tailored to the Life Sciences industry:
» Business Models in Life Sciences



» Partnerships
» Business Development
» Finance
» Intellectual Property


» Venture Capital
» Product Development
» Career Orientation




» Meet your Match sessions
» Case studies












Confirmed speakers (status 1 November 2025)
















Jochem Bossenbroek Executive in Life Sciences & Health | IMAGINE leader
Faced with burnout end of last year, I accepted the challenge. How can I overcome this situation as quickly as possible? Not an easy task at barely 20% of my normal energy level, a brain fog that blocked any spark of brightness and a headache as punishment for any serious activity. Still, I managed to ask myself one key question, inspired by earlier Supply Chain Management teachings:

“What process will make the desired outcome inevitable?”
This question forced me to take responsibility for my situation. It pushed me to start with a baseline analysis, to think about the desirable outcome, and to design a process that would bridge the gap. It also allowed me to trust that a well-structured process would automatically lead to the desired result. Finally, it turned out that asking this question would lead me to full recovery.
Step 1: Baseline analysis
I began with a baseline analysis, including a medical examination to rule out other health issues. Fortunately, no illness was found. A comprehensive blood test revealed no serious deficiencies—other than low Vitamin B12, which I addressed with a simple supplement. From there, I knew my symptoms were solely attributable to burnout. But what is burnout, really, on a biophysical level?
The little energy I had in those first weeks went into a deep dive into burnout and mental health literature, along with guidance from my GP and an excellent physiotherapist. Three fundamental insights from these sources proved extremely helpful:
1. Chronic stress can cause irreversible brain damage. When the brain is overloaded, it produces excessive cortisol. If this continues for too long, it can lead to permanent (brain) damage. I had always assumed stress symptoms were easy to overcome but had never realised how serious it could be. This was my wake-up call. Fortunately, my GP assured me that my condition was still reversible.
2. Over-breathing exacerbates stress. Under high mental load, we tend to breathe too much air, leading to various detrimental health effects. The good news? This can be controlled with specific breathing techniques that have an immediate
positive impact. The Oxygen Advantage by Patrick McKeown explains this very well. I started walking in nature while practising the breathing exercises, which brought instant relief.
3. The mind plays tricks on us. Left unchecked, our thinking is riddled with illusions, blind spots, and unhelpful mental loops. Mo Gawdat, former Chief Business Officer at Google X, explains this brilliantly in Solve for Happy, illustrating how joy can be engineered. I wish I had read this book when I was 24. It helped me begin unravelling and rewiring my thinking towards joy.
Step 2: Defining the desired outcome
Once I accepted and understood my situation at both a mental and biophysical level, I considered my desired outcome. My first thought was full recovery, but to what extent? How I felt last summer? In 2023? 2022? Defining those states proved difficult. A more tangible goal was simply to feel full of energy again, with a smile on my face
for no reason, ready to embark on new missions. That became my new focus. I also set a timeline: on my birthday, 1st March, I would conduct an intermediate progress check and fine-tune my approach if needed.
Step 3: Defining the process
With a clear understanding of my situation and a measurable objective, I needed to design a process to make the desired outcome inevitable. In practice, I didn’t map it all out from the start; rather, the process evolved along the way. Looking back, it followed these key steps:
1. Stepping down as CEO. I had no choice but to replace myself and communicate this to my colleagues and shareholders. This was by far the hardest step, but I believe it was the right and honest thing to do. I remain deeply grateful for the support and understanding I received, particularly from my business coach, who helped me see what was necessary.
2. Allowing my body to rest. After making the tough decision, my energy levels dropped drastically— almost to survival mode. During this phase, I bonded with our cat, watched a lot of Netflix and Disney documentaries (Limitless with Chris Hemsworth is highly recommended), practised breathing techniques, and took short walks. The patience of my wife and the love of my kids were the real treatment here.
3. Walking and reflecting. As my energy crept back to 20-25%, I took longer walks in the beautiful Dutch forests (De Veluwe). Each walk was an opportunity to practise Oxygen Advantage breathing techniques and reflect on the root cause of my burnout. Eventually, I realised the uncomfortable truth: a misalignment between my mind, heart, and gut.
4. Journaling. I made it a habit to capture insights on paper. My notebook has been with me constantly, helping me clear my mind and revisit my thoughts later.
5. Priotising sleep and nutrition. I aimed for eight hours of sleep, which usually follows sufficient physical activity during the day and mindful evening routine. Nutrition-wise I mostly follow the Mediterranean diet principles, tuned to my gluten intolerance and personal nutrition needs. No big changes here.
6. Creating a ‘happy list’. I listed activities that bring me joy. My happy list now includes 26 items such as playing with my kids, making a wood fire and learning something new. Whenever I need a happiness boost, I pick an activity from this list.
7. Systemic constellations. With rising energy levels I did something that was slightly uncomfortable first, but a huge game changer overall: I enrolled to a sequence of two individual sessions and a threeday retreat with a focus on systemic constellations, under expert guidance. This helped reveal and dissolve energy leaks, obstructive patterns, and other negative distortions in my system. It also helped (re-)surfacing my true personal mission for this life. Never have I gained so much relief and clarity in such a short time frame. After day one of the retreat I found myself smiling again – for no apparent reason.
8. Listening to music. Music can be enjoyed at any moment, at any energy level and even with closed eyes. Whenever I felt like it, I put on my noisecancelling headphones and let the music transport me.
9. Adjusting physical exercise. Here’s something I overlooked. Under the impression that physical exercise would bring me closer to my objective, I returned to resistance training too soon. I quickly discovered that high intensity workouts had a draining rather than a supportive effect. On my physiotherapist’s advice, I refrained from intense exercise for two months, focusing instead on lighter activities.
10. Studying the Yoga Sutras. For a reason I can’t recall, I ordered a book by Dr. Taimni explaining
the Yoga Sutras by Patanjali – originating from the period 200 – 500 AD. It contains 196 sutras (concise statements with a deep meaning) that reflect true wisdom on a range of worldly and spiritual topics, including mental wellbeing. Submerging myself in this ancient wisdom created space to deepen my thinking and build on the insights from the systemic constellations sessions.
11. Re-introducing my Yoga breathing routine. Inspired by the Yoga Sutras, I picked-up the breathing routine I learned years ago during courses offered by The Art of Living Foundation. Pranayama, SKY Breathing and other techniques nicely complement The Oxygen Advantage exercises. The breath, I discovered, is a remote control for the nervous system—allowing me to shift from stress to clarity within minutes. It’s now a daily habit.
12. Meditation. Though new to me, meditation has proven invaluable. I still have a long way to go, but even now, I notice its calming effect and the clarity it brings afterward.
Fortunately, the process has brought me to the desired outcome. On March 1 I already woke up with 70-80% of my target energy level and today I’m fully charged again, with much more clarity, and, most importantly, a smile on my face. Looking back, this journey has been about more than recovery. It has been a recalibration—aligning my mind, heart, and gut in ways I had never considered before. That has brought me to the wonderful position of co-initiating a novel mental performance platform, and launching the Sublime Coherence blog, where I share stories of courageous leaders working towards positive impact.
Vivalyx – enabling more donor organs, saving more lives
In the rapidly evolving field of transplantation medicine, one of the greatest bottlenecks remains the shortage and sub-optimal quality of donor organs. Over a million patients are waiting worldwide, and many organs are lost simply because current preservation methods do not maintain vitality sufficiently. Against this backdrop, the German organ vitality start-up Vivalyx emerges with a strong and worldwide mission: to raise the number and quality of transplantable organs by making organ preservation smarter, more scalable and safer.
Vivalyx combines medical-, biotech- and regulatory know-how to tackle this challenge. Their product, Omnisol, is a fully synthetic, blood-free organ preservation solution designed for all organs and all preservation methods (static cold storage, cold machine perfusion, warm machine perfusion). In preclinical trials, Omnisol has demonstrated superior performance compared to blood-based perfusates and traditional cold-storage fluids.
What makes Vivalyx’s approach particularly compelling is the combination of eliminating the dependency on donor blood - which limits scalability of warm perfusion nowadays - while offering compatibility across multiple preservation modalities with one optimized solution. This scientific and engineering foundation is paired with a clear regulatory and commercial strategy backed by strong early funding, including a € 5.4 million seed round in 2024 and up to € 8.5 million in support from the European Innovation Council (EIC), underscoring confidence in the technology.
Vivalyx is now advancing toward market readiness: the company has submitted its first technical dossier for CE-marking (expecting first approval in 2026) and is starting its fully cleared clinical trial in the Netherlands. Vivalyx is becoming a key player in the organ-transplant ecosystem, offering a transformational solution that could significantly increase the pool of viable donor organs, and ultimately improve outcomes for patients facing organ failure.

Vooruitdenken is voor producenten de sleutel tot succes
Productiefaciliteiten binnen de life sciences en farma moeten tegelijk flexibeler, energiezuiniger én toekomstbestendiger worden. “Juist die combinatie vraagt om een integrale blik als men het heeft over projecten,” vertelt Michel Croes, Business Development Manager van NIRAS in Nederland: een engineeringsbureau gespecialiseerd in process en packaging engineering en het bijbehorende project management
Al decennialang ondersteunt NIRAS toonaangevende producenten bij het optimaliseren van hun productieprocessen, capaciteitsvergroting en cGxP-naleving. De kracht schuilt in de samenwerking tussen disciplines: van procesontwerp en gebouwontwerp tot duurzaamheid en digitalisering. “We brengen technologie, regelgeving en procesontwerp samen. Dat is waar efficiënte productie en marktwaarde begint,” aldus Croes.
Kunnen voldoen aan de eisen Een belangrijk aandachtspunt is compliance. De lokale en internationale specialisten van NIRAS hebben jarenlange ervaring met cGMP-processen, cleanroomdesign, kwalificatie en validatie. Ze helpen bedrijven kwaliteits- en veiligheidseisen in hun processen te verankeren.Daarnaast zet NIRAS sterk in op duurzaamheid en circulariteit. Met adviezen rond energie- en watergebruik helpt het bedrijf producenten hun ecologische voetafdruk te verkleinen en tegelijk te voldoen aan duurzaamheidsdoelen die, ook vanuit de overheid, scherper en scherper worden.
Early-stage drug discovery companies operate in a high-stakes environment. Scientific innovation moves quickly, yet resources are limited. The ability to access, organize, and protect real-time research data is critical for productivity and longterm success. In this context, data security and scalability are strategic necessities.
Protecting intellectual property is fundamental. For small biotech and academic spinouts, the entire value often rests on the integrity and confidentiality of data. A breach, loss, or compliance failure can jeopardize partnerships, funding, and regulatory approval. Choosing a platform with robust security standards ensures results remain protected while giving teams and investors peace of mind.
Scalability is equally important. Discovery efforts may start with a handful of scientists managing modest datasets, but growth can be rapid. As collaborations expand and data complexity increases, rigid systems become bottlenecks. A flexible informatics solution adapts to more users, projects, and integrations without forcing disruptive transitions.
Choosing the right informatics partner early on makes all the difference. CDD Vault was designed with both security and scalability at its core. Its AWS-based infrastructure safeguards research data while meeting compliance needs, and its intuitive interface enables seamless expansion to support future workflows and collaborations.
For early-stage drug discovery companies, investing in a secure, scalable solution builds a foundation for tomorrow’s developments and data requirements, including AI readiness. CDD Vault offers that balance: a trusted platform to protect your science today and empower growth into the future.

Gegronde beslissingen alvorens de realisatiefase Belangrijke onderwerpen die gekoppeld zijn aan de digitale transformatie zijn traceerbaarheid, de mogelijkheid om processen real-time te analyseren en om bijvoorbeeld intra-logistieke stromen in beeld te krijgen. Digitalisering biedt nieuwe kansen. NIRAS ontwikkelt digitale plantdesigns, BIM-modellen, schrijft functionele specificaties en maakt simulaties die bedrijven ondersteunen bij het nemen van datagedreven, toekomstbestendige beslissingen. “In een wereld waarin veel verschillende ontwikkelingen elkaar in een rap tempo beïnvloeden, is overzicht het beste startpunt,” besluit Croes. “Onze rol is om deze te bieden en vanuit deze basis te kijken naar de toekomst, hoe groot of klein een project of aanpassing ook is.”
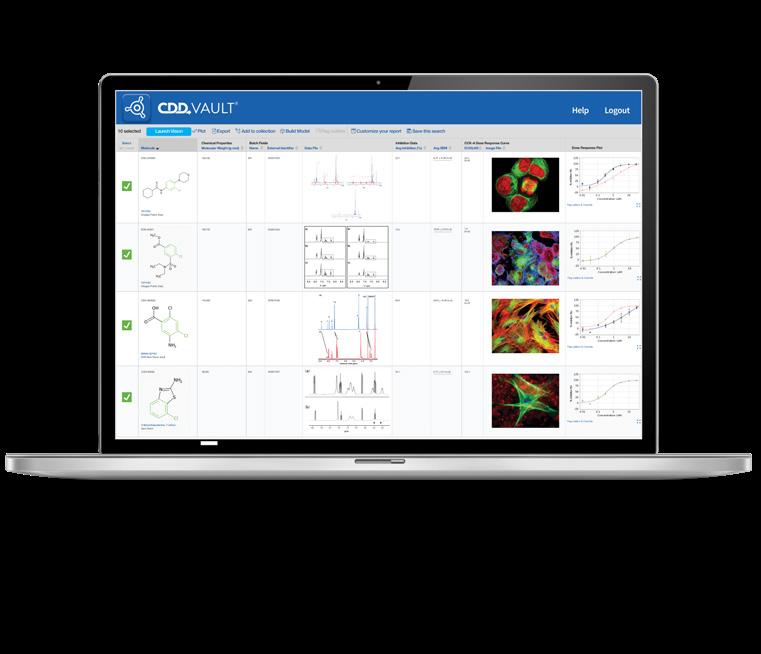

BioLizard: Vlaamse innovatiekracht met wereldwijde impact
BioLizard is een Vlaams, ondernemend en snelgroeiend biotechbedrijf dat data, AI en biologie samenbrengt om vooruitgang in de life sciences te versnellen. Met een multidisciplinair team van computationele biologen, AI-experts en data-ingenieurs ondersteunt BioLizard biotech, farma en academische centra wereldwijd in het ontdekken van nieuwe biomarkers, het versterken van translational research en het bouwen van future-proof data-architecturen.
Als Gentse scale-up bouwt BioLizard verder aan een internationale reputatie waarin wetenschappelijke excellentie en ondernemerschap hand in hand gaan. Die aanpak wordt breed erkend: het bedrijf werd de voorbije jaren geselecteerd voor onder meer Deloitte FAST 50, FAST 500 EMEA, VLAIO’s Economic Impact Award, Trends Gazellen en UNIZO KMO van het Jaar. CEO Liesbeth werd opgenomen in Inspiring Fifty Belgium 2022 en won de Vlerick Venture Award 2025. Daarnaast haalde BioLizard internationale nominaties binnen HealthTech en women-led business.
Tijdens BCF wordt BioLizard onder andere vertegenwoordigd door CEO Liesbeth, een inspirerende stem binnen de Vlaamse en Europese biotech- en technologiesector. Als vrouwelijke ondernemer met een sterke wetenschappelijke achtergrond en een duidelijke visie op AI-gedreven biologie fungeert zij als rolmodel voor vrouwen in wetenschap, technologie en business. Haar leiderschap toont hoe deep science, innovatie en inclusief ondernemerschap elkaar versterken.
In haar lezing tijdens BCF spreekt Liesbeth over hoe het wetenschappelijke landschap razendsnel verandert door de vooruitgang in data-science, AI en biotechnologie, en waarom loopbaanwendbaarheid daarbij essentieel wordt. Ze gaat in op de kerncompetenties die wetenschappers nodig hebben om te groeien in deze dynamische omgeving—zoals digitale geletterdheid, interdisciplinair samenwerken, veerkracht en levenslang leren. Ze deelt praktische inzichten om je professionele ontwikkeling actief te sturen, veranderingen als kansen te zien en een betekenisvolle, toekomstbestendige carrière uit te bouwen.

Some people get a job. Others become a Legend.
Launch your career at Legend Biotech. At Legend Biotech, your work truly matters, not just to us, but to patients around the world. Join us in our mission to save lives and shape the future of medicine.
About Legend Biotech ���� Legend Biotech is a global biotechnology company transforming cancer care through advanced cell and gene therapies. With 2,800+ employees worldwide, we are the largest standalone cell therapy company and a recognized pioneer in CAR-T innovation.
Our lead therapy, CARVYKTI® ��, offers a onetime treatment for relapsed or refractory multiple myeloma. It’s already changing lives, and we’re just getting started.
We operate as a fully integrated global organization, with manufacturing facilities in the US and Europe. Our sites in Belgium support the clinical and commercial supply of CAR-T therapies across the EMEA region.
Why join us?
• A job with impact ��
From the cleanroom to the office, every role plays a vital part in delivering life-saving therapies to patients who need them most.
• A culture of care ��
We care deeply, for patients and for each other. Respect, inclusion, and teamwork define how we work.
• Ownership that empowers ��
We trust you to take initiative, shape your role, and bring ideas to life, with the freedom to grow and the support to succeed.
• Room to grow ��
From day one, you learn by doing, supported by peers, tools, and tailored programs through our Legend Campus.
• Pioneering innovation ⚖
Driven by curiosity and powered by science, we challenge what’s possible to shape the future of medicine.
• Balance that works ⚖️
Enjoy flexibility, trust, and respect, including our 4-on-4-off schedule that keeps life in balance.
La Lorraine Bakery Group
La Lorraine Bakery Group (LLBG) is a 100% Belgian family-owned company with more than 80 years of experience in the milling and bakery sector. Every day, more than 5,300 enthusiastic employees create and sell bakery products worldwide to consumers, retailers and foodservice professionals in more than 35 countries.
At LLBG, we get up everyday to bake a better world together. We build wide access to more natural, tastier and fresher bakery products, contributing to well-balanced daily nutrition and consumer delight. We do it sustainabily while growing people to their full potential.
We are structured into 4 business units. In our ‘bakery fresh’ & ‘bakery frozen’ units, we produce over 1500 products in different categories: bread (loaves, buns, baguettes), viennoiserie, donuts, patisserie and savoury snacks. In our ‘store concepts’ unit, we’ve built up a strong franchise network of Panos & Deliway shops. Paniflower, our ‘milling’ unit, is one of the largest flour producers in the Benelux, focusing on technological innovation.
Our brands (La Lorraine, Panesco, Donut Worry Be Happy, Tarte à moi) are on the shelves of retailers across the globe. Or they are served by foodservice professionals to visitors of hotels, restaurants, cafés and to-go shops.
For over 80 years, we have proudly upheld five core values at LLBG: entrepreneurship, shared passion, respect for the three Ps, ownership and nononsense. It’s these values that allow our company and our people to keep growing. They inspire us in our work and actions, so we can make a difference together every day.
Entrepreneurship is embedded in the DNA of our family business. At La Lorraine Bakery Group, we love to roll up our sleeves and take pride in making a difference.
Ready to make a difference together?
BioLizard blijft zo bouwen aan één missie: science opschalen tot echte, mondiale impact—vanuit Vlaanderen, voor de wereld.






Recycling
De publiek-private samenwerking tussen TNO en Johnson & Johnson (J&J) vormt een belangrijke stap richting een duurzaam zorg-ecosysteem. De geneesmiddelensector speelt een sleutelrol in het verbeteren van de wereldwijde gezondheid. Tegelijkertijd groeit de aandacht voor de milieuvraagstukken. Volgens de Wereldgezondheidsorganisatie (WHO) maakt de geneesmiddelensector gebruik van grote hoeveelheden plastics, waarvan een aanzienlijk deel bestemd is voor eenmalig gebruik. Deze hoogwaardige ‘single-use plastics’ verdwijnen vaak in de verbrandingsoven, terwijl ze materialen bevatten die juist geschikt zijn voor recycling. Om de verspilling te doorbreken, bundelen TNO en J&J hun krachten.

De gezamenlijke ambitie van TNO en J&J kreeg al snel concrete vorm in het single-use bags project. Deze single-use plastic zakken worden in de geneesmiddelenproductie op grote schaal gebruikt. Uit veiligheidsoverwegingen worden ze na éénmalig gebruik verbrand; het is de vraag of dat in alle gevallen wel nodig is. Volgens
Marieke Havermans, Senior Business Developer Circular Packaging bij TNO, is het systeem aan heroverweging toe: “Het kan toch niet waar zijn dat zulke waardevolle plastics zomaar in de oven belanden?”, aldus Havermans. “We moeten het perspectief kantelen: niet langer denken in termen van afval, maar in hoogwaardige plastics die een tweede leven verdienen.” Vanuit het hernieuwde uitgangspunt gingen TNO en J&J aan de slag met het recycling vraagstuk. Om tot innovatieve antwoorden te komen, brachten ze partijen uit de hele keten samen; van grondstofleveranciers en folieproducenten tot recyclers en assemblagebedrijven.
De samenwerking leverde op labschaal veelbelovende resultaten op: gebruikte plastics konden worden omgezet in nieuwe films (dunne laag plastic) die opnieuw inzetbaar zijn binnen de geneesmiddelenketen. “Voor direct gebruik, zoals infuuszakken, laat de wetgeving dat nog niet toe, maar als omverpakking bieden de gerecyclede materialen een duurzaam alternatief”, legt Havermans uit. De volgende stap is het opschalen naar een recyclinglijn. Hiervoor is een goede businesscase nodig, wat vraagt om een andere denkwijze: “niet denken in barrières, maar in opportuniteiten.” Deze liggen onder meer in het verkrijgen van hoogwaardig recyclaat, het verlagen van afvalverwerkingskosten en het verminderen van CO2-uitstoot.
De kracht van publiek-private samenwerking Het single-use project sluit goed aan bij de bredere duurzaamheidsambities van J&J. Volgens Fenne Bouma, Sustainability Lead J&J Nederland, bewijst het project dat publiek-private samenwerking een sleutelrol speelt in het versnellen van duurzame innovatie; “We willen in 2030 onze CO2uitstoot (scope 1 & 2) met 44 procent verlagen ten opzichte van 2021,” zegt Bouma. “We liggen goed op koers: inmiddels hebben we al 26 procent reductie gerealiseerd. Dit project helpt ons die doelstelling te bereiken; het maakt onze producten duurzamer en verkleint onze CO²-voetafdruk.

” Samenwerking door de waardeketen ziet ze als een randvoorwaarde voor verder succes. “Duurzaamheid is geen soloproject. Alleen door krachten te bundelen kunnen we echt verschil maken.”
Vooruitblik
Wat begon als een kleinschalig initiatief binnen de strategische alliantie tussen J&J en TNO, is uitgegroeid tot een coalitie die de zorgsector in beweging brengt. Onder de partners bevinden zich J&J Innovative Medicine, Bristol Myers Squibb, Avantor, BioBTX, Halix, Polymer Science Park, Wittenburg, Ortessa, Renolit, Steris en Thermo Fisher. Samen werken zij aan het opschalen van circulaire oplossingen binnen de waardeketen.
De ambitie van Havermans en Bouma blijft daarbij onverminderd groot. “Over vijf jaar willen we samen bij de recyclinglijn staan,” zegt Havermans. “Maar belangrijker nog: we willen een systeemverandering op gang brengen die de sector structureel verduurzaamt.” Bouma benadrukt dat duurzame zorg voor de mens alleen mogelijk is wanneer we ook goed zorgen voor onze planeet.

Nieuwe thuisbasis voor innovatie:

Onder grote belangstelling is donderdag 2 oktober Plus Ultra Utrecht feestelijk geopend. Wederom een mijlpaal in de verdere ontwikkeling van het grootste science park van Nederland. Met zijn duurzame ontwerp, flexibele laboratoriumen kantoorruimtes en gedeelde faciliteiten biedt Plus Ultra Utrecht een inspirerende omgeving voor start-ups, scale-ups en gevestigde bedrijven in de Life Sciences & Health sector.
Inspirerende middag tijdens Utrecht Science Week
Deze middag, als onderdeel van de Utrecht Science Week, beloofde vele verhalen en ontdekkingen. Prof. Edward Nieuwenhuis (Chief Medical Officer Prinses Máxima Centrum) en Prof. Peter van der Spek (Professor Clinical Bioinformatics Erasmus MC) openden de middag met keynotes over funding, samenwerking en de impact van data binnen de life sciences, waaronder oncologie. Bezoekers kregen een exclusieve blik achter de schermen bij eerste gebruiker QVQ, een kijkje binnen de Innovation HUB (i-HUB) en het Shared Open Innovation Lab met ICAT, en werden geïnspireerd door kunstenaar Vincent Mock in de Science Gallery.
Niet alleen deze middag, maar het hele gebouw staat in het teken van verbinding: tussen wetenschap en ondernemerschap, tussen kunst en technologie, en vooral tussen mensen met een gedeelde ambitie om ontwikkelingen in de life sciences te versnellen.
Plus Ultra Utrecht
Plus Ultra Utrecht biedt de perfecte plek om als start-up of scale-up de volgende stap te zetten. Het ontwerp door architectenbureau cepezed combineert een transparante uitstraling met hoge duurzaamheidsambities. Met circa 23.000 m² aan toekomstbestendige lab- en kantoorruimte en een groots en licht atrium voor ontmoetingen en evenementen, is het gebouw ontworpen om kruisbestuiving te stimuleren. De ligging op het Utrecht Science Park naast belangrijke instellingen zoals het UMC Utrecht en Prinses Maxima Centrum maakt het een ideale plek voor ondernemers in de Life Sciences & Health.
Steijn Ribbens, CEO Kadans Science Partner: “We zijn er trots op dat we met Plus Ultra Utrecht een uiterst duurzame ruimte hebben gecreëerd die er niet alleen goed uitziet en goed aanvoelt, maar
hopelijk vooral ook goed doet. Ik kijk uit naar alle nieuwe samenwerkingen en ideeën die Plus Ultra zal inspireren.”
Innovation HUB: broedplaats voor start-ups
Een belangrijk onderdeel van Plus Ultra Utrecht is de i-HUB, een gedeelde werkomgeving speciaal ontwikkeld voor start-ups en spin-offs. In deze zone bevinden zich volledig ingerichte laboratoria en kantoren waar ondernemers direct aan de slag kunnen.
Het hart van de i-HUB is het gedeelde laboratorium van circa 600 vierkante meter in samenwerking met ICAT. In dit Shared Innovation Lab werken startende onderzoekers naast elkaar en ontstaat kennisuitwisseling vanzelf. Door de integratie met de rest van het gebouw vormt de i-HUB een natuurlijke opstap naar verdere groei binnen Plus Ultra Utrecht en het Utrecht Science Park.
Kadans Science Partner: een Europese speler Plus Ultra Utrecht is onderdeel van het uitgebreide netwerk van Kadans Science Partner, dat zich uitstrekt over Europa. Met locaties in Nederland,
België, Frankrijk, Duitsland, het Verenigd Koninkrijk en Spanje biedt Kadans bedrijven toegang tot een internationaal netwerk van innovatie.

Dit maakt het mogelijk om verbindingen te leggen tussen organisaties in verschillende landen, waardoor kennisdeling en samenwerking over grenzen heen worden gestimuleerd. Plus Ultra Utrecht is daarmee niet alleen een lokale hub, maar ook een toegangspoort tot een internationaal netwerk van partners, investeerders en kennisinstellingen.
Actieve community
Plus Ultra Utrecht is niet zomaar een locatie. Het is een community, een plek waar ideeën tot leven komen en waar samenwerking centraal staat. Door bedrijven, onderzoekers en investeerders samen te brengen, draagt het gebouw bij aan de ontwikkeling van oplossingen die de toekomst van gezondheid en wetenschap vormgeven. Kadans draagt hier actief aan bij door evenementen te organiseren en te stimuleren. Zo kijkt Kadans ook al vooruit naar 2026, waarin twee toonaangevende evenementen de innovatieve kracht van Nederland in de life sciences in de schijnwerpers zetten. Op 25 maart 2026 is Kadans met haar gebouw Accelerator Utrecht weer host sponsor van het Global Investor Forum, waar start-ups en scale-ups hun innovaties presenteren aan investeerders. Een dag later, op 26 maart 2026, volgt Innovation for Health in de Jaarbeurs Utrecht. Kadans is hierbij aanwezig als Gold Sponsor
Bekijk nu een sneak peek van Plus Ultra Utrecht of neem contact op om kennis te maken tijdens een van deze evenementen, scan de QR code:

en vertelt je er graag meer over haar Europese netwerk. Bovendien organiseert Kadans komend jaar ook zelf weer een scala aan internationale netwerkevenementen, zo ook in o.a. Glasgow en Parijs.

Biotech Booster is nu anderhalf jaar operationeel, de ontbrekende schakel in de keten van kennis naar toepassing. Tijd voor een terugblik: waar staat het Groeifondsprogramma nu? En een vooruitblik vanuit het perspectief van drie initiatiefnemers van het eerste uur: CEO Nettie Buitelaar, CBO Rein Strijker en Hollandbio-directeur Annemiek Verkamman. Ze delen een missie: versnel de valorisatie!
1. Historisch en strategisch perspectief, Annemiek Verkamman
In de praktijk draait het om verbinding

‘Stapels rapporten zijn er al over vol geschreven: we doen te weinig met onze kennis. Internationaal is Nederland een voorloper als het gaat om biotechnologische publicatie, maar verhoudingsgewijs blijft de vertaling in bedrijvigheid en toepassingen die de markt bereiken achter. En dat terwijl we staan te springen om nieuwe medicijnen, maar ook slimme oplossingen zoals waterzuivering door champignonschimmels of duurzamere landbouw door biologisch afbreekbare zaadcoatings. Om maar een paar voorbeelden te noemen uit de huidige projectenportfolio van Biotech Booster. Mijn persoonlijke missie is dat iedereen zo snel mogelijk profiteert van de nieuwste wetenschappelijke inzichten, maar de kloof tussen kennis en vermarkten is groot, complex en bureaucratisch. Het breed gevoelde idee dat het beter kan en moet was de aanleiding voor Biotech Booster. Bij het Nationaal Groeifonds diende een coalitie van koepels van Nederlandse kennisinstellingen, hollandbio, dsm-firmenich, J&J en TNO het plan in voor het programma. Anderhalf jaar na de start staat er een stevige organisatie, waar de initiatiefnemers nog altijd samen het bestuur vormen. Biotech Booster is de missende schakel tussen kennisinstellingen en het bedrijfsleven. Biotech Booster geeft projecten in de eerste fase de middelen om de technische en economische haalbaarheid te onderzoeken en projecten in de tweede fase te werken aan onderzoek en ontwikkeling. Meer dan ooit blijkt dat het in de praktijk draait om verbinding. Biotech Booster brengt de verschillende werelden bij elkaar en bevordert het onderlinge begrip.’

2. Operationeel perspectief, Nettie Buitelaar
Tweede tranche uit
Nationaal Groeifonds is erkenning van hard werk en strategische waarde

‘Het oprichtingsjaar 2024 was een rollercoaster. Er was geld voor projecten, het aantal Business Developers groeide en we creëerden een pool van ervaren ondernemers. De animo bleek enorm, de behoefte is groot. Vanuit operationeel perspectief was 2025 een ‘tussenjaar‘ waarin onder meer de 1e fase is geëvalueerd door een extern bureau. Dat is positief verlopen en heeft er onder meer toe geleid dat we de 2de tranche van de subsidie uit het Nationaal Groeifonds hebben gekregen. Deze is onderdeel van de totale subsidie van 246 miljoen euro die in 2022 werd gereserveerd. Met deze toekenning werd de voorwaardelijke financiering definitief en is de continuïteit van het programma gegarandeerd tot 2032. Een erkenning voor het harde werk van ons team en een belangrijk signaal dat de overheid de strategische waarde van biotechnologie voor Nederland onderschrijft. Het is duidelijk: de booster staat! We hebben gedegen processen ingericht voor het verwerken en beoordelen van alle aanvragen. We hebben veel tijd besteed aan onderlinge samenwerking bij de verschillende kennisinstellingen en ons team is er trots op dat er nu echt een community ontstaat. Ondertussen bouwen we verder aan de contacten met de industrie en investeerders, want alleen zo komen de goede plannen uiteindelijk in de maatschappij. Positief is dat er zowel op nationaal als Europees publiek gebied veel gebeurt, biotechnologie is zeer relevant. Dat komt ons werk ten goede.’
3. Ondernemersperspectief, Rein Strijker
De kracht om veranderingen te genereren

‘Om te weten wat ondernemers bezighoudt, is het handig als je er zelf een bent. Na decennia in de lifesciences en biotechnologie te hebben gewerkt (o.a. Pharming en het huidige VitalneXt) was ik gepassioneerd om mijn kennis en expertise in te brengen in Biotech Booster. In de afgelopen tijd zijn er zo sterke partnerships gebouwd tussen kennisinstellingen, industrie en biotechondernemingen. De cultuurkloof tussen deze werelden met hun verschillende belangen is groot. Dat maakt verbinden soms onnodig complex, want het is eigenlijk zo simpel: je brengt mensen bij elkaar die toch al bij elkaar betrokken zijn. Startups en spin-offs proberen we actief te begeleiden. Deelname aan Biotech Booster is ook een kwaliteitskeurmerk en het is spijtig als je plannen niet worden goedgekeurd. Niet getreurd: ook dan zijn er ondernemers in het Biotech Booster netwerk die je willen begeleiden. Wat ook mooi werkt binnen Biotech Booster, zijn de ‘neven effecten’ van een innovatie. Als ik terugblik dan is onze mooiste verdienste de veranderende mindset en het toegenomen begrip voor waar de andere partij mee worstelt. Zo vlak voor mijn pensioen (opvolger per 1 januari 2026 Marco de Boer) blik ik tevreden terug: Biotech Booster heeft de kracht om daadwerkelijk veranderingen te genereren.’
In mei 2024 werd het operationele startsein gegeven voor een uniek verbond van kennisinstellingen en bedrijven met als doel kennis over biotechnologie beter en sneller door te zetten naar de markt. Dat dit doel de hoogste prioriteit heeft, blijkt wel uit het riante budget van het Nationaal Groeifonds. Biotech Booster kreeg bijna € 50 miljoen direct onvoorwaardelijk toegekend voor 3 jaar. Voor de periode vanaf 2025 werd bijna € 200 miljoen voorwaardelijk toegekend. Combineer deze aangename geldpot met de kennis en expertise van het team en partners en een unieke organisatie is geboren. Biotech Booster identificeert, begeleidt en financiert kansrijke ideeën op een overzichtelijke en pragmatische manier. Kijk op www.biotechbooster.nl.
MimeCure is een innovatieve farmaceutische startup opgericht door onder meer Luke van der Koog. Luke, die summa cum laude afstudeerde in de Farmacie aan de Universiteit van Groningen, ontwikkelde tijdens zijn PhD traject een nieuwe inhalatietherapie in poedervorm tegen chronische longziekten, zoals COPD en longfibrose. De therapie is gebaseerd op het lichaamseigen eiwit mimecan. De startup rondde een Level 1 traject binnen het Biotech Booster programma af en is van plan om in 2028 met de eerste menselijke studie te starten.
Al vrij snel werd de potentie van Luke’s ontdekking duidelijk en kreeg hij contact met Jasper Dijkstra, Business Generator aan de Universiteit van Groningen en daarbij ook een business developer van Biotech Booster. Jasper – en zijn collega Herman Groen - loodste het jonge bedrijf in het Biotech Booster programma door de eerste stappen op het gebied van business, valorisatie en zelfstandig ondernemerschap, waaronder het verkrijgen van patent en het aantrekken van investeringen.
Detecting Parkinson
Luke: ‘In 2023 kwamen we via Jasper in contact met Biotech Booster, eigenlijk nog voor het officieel werd gestart. Omdat geneesmiddelenontwikkeling een duur traject is, was aanvullende financiering uit meerdere bronnen noodzakelijk. Daarom trokken we, parallel aan het Biotech Booster-traject, ook private investering aan.’
‘April 2024 was een belangrijke maand. De BV MimeCure werd opgericht en we kregen toezegging om deel te nemen aan het Level 1 Biotech Boosterprogramma. Ondertussen was duidelijk dat denken in business cases daadwerkelijk een andere mindset vereist dan werken in het onderzoek. Om te denken als een ondernemer volgde ik enerzijds een ‘mini-MBA’ en was ik anderzijds zoveel mogelijk aanwezig op de Biotech Booster dagen. Een geweldige inspirerende plek om te netwerken en je bedrijf onder de aandacht te brengen.’
Biotech Booster als kwaliteitsstempel
‘Iedere startup is beducht op de fase die we nu naderen: de Valley of Death, wanneer uitgaven hoog zijn en inkomsten laag. Het is geweldig dat Biotech
Detecting Parkinson develops the FibrilRuler Test, a bloodand spinal-fluid assay that measures amyloid fibrils to enable earlier, more accurate detection and monitoring of Parkinson’s disease. By providing objective insights into disease progression, their test supports therapy development, improves patient care, and reduces healthcare burdens.
Haplotagging
Booster door middel van een Level 2 programma daar een oplossing voor biedt. We gaan daar zeker op inschrijven. Maar meer nog werkt deelname aan Biotech Booster als een kwaliteitsstempel. Het laat zien dat je onderneming potentie heeft en serieus wordt genomen in het veld. Daarnaast biedt Biotech Booster je toegang tot een nuttig netwerk wat cruciaal blijkt voor de slagingskans van je onderneming. Aan mede-ondernemers wil ik meegeven: omarm wat Biotech Booster aanbiedt, zowel wat betreft subsidies, netwerk als begeleiding: het is echt een booster voor je onderneming.‘

Haplotagging introduces a novel DNA sequencing method that makes Smart Breeding faster, cheaper, and more accurate. By adding haplotype context through a simple bead-based reagent, it empowers breeders of all sizes to develop high-yield, climate-resilient crops while preserving genetic diversity – helping secure the future of food.
Meer informatie: Bekijk alle 70+ projecten op www.biotechbooster.nl/projects.
The life sciences sector is brimming with talent — and the TOPX Females to Follow Awards 2025 aim to shine a spotlight on some of its most promising rising stars. Organized by the TOPX Network, an initiative of Hyphen Projects, this award program identifies and highlights outstanding women across Science, Industry, and Entrepreneurship in Life Sciences & Health. The awards serve not only as a recognition of individual achievement but also as a catalyst for broader change within the industry, fostering a more diverse, inclusive, and innovative environment.
TOPX Network – Platform for Visibility, Inspiration & Empowerment
The awards’ core mission is threefold: to give visibility to remarkable female professionals, to celebrate their achievements, and ultimately to inspire the next generation to follow in their footsteps. With
30 nominees selected across three categories –Science, Industry, and Entrepreneurship – TOPX is not just honouring individual success; it is building a movement that champions gender equality and diversity in the life sciences.
This initiative is part of a larger effort by the TOPX Network, a vibrant community dedicated to fostering connections, mentorship, and leadership among women in life sciences and healthcare. The network provides a platform for knowledge exchange, professional development, and strategic partnerships, helping women advance their careers and influence the future of the industry. The Awards ceremony, held during the networks flagship event TOPX Summit, serves as a rallying point for this community, offering visibility and recognition to rising stars who are shaping the future of healthcare and scientific discovery.
“My commitment to diversity, equity, and inclusion is inseparable from my work in explainable AI for biomedical applications. We must go beyond merely recognizing our differences and embed them into the way we design transparent, trustworthy AI tools that truly work for children, women, and individuals in low-access settings. Through my research, I aim to ensure that no user/patient is treated as an afterthought, and that the technology we build reflects the diversity of the people it is meant to serve. I am also part of the DE&I team of my faculty at the University of Twente, where I aim to bring this mindset into our daily work and our classrooms, building an environment where inclusion is practiced, not just discussed.”
The Future of Life Sciences Leadership
This year’s winners exemplify what the Females to Follow Awards stand for: scientific excellence, bold innovation, and a steadfast commitment to improving human health. Their words reflect not just their work, but their core values – equity, collaboration, and patient-centered progress. They are the future leaders who will shape policies, inspire change, and drive breakthroughs in biotech and healthcare.
Together, they embody a new generation of leadership – ambitious, inclusive, and impact-driven. Their success stories serve as powerful examples for aspiring women in the industry, reinforcing the importance of perseverance, mentorship, and community support.
“As female founder and CEO of AstriVax Therapeutics, I fully embrace our company values of authenticity, entrepreneurial, caring and disciplined, because they are fully overlapping with my own values. I try to role model and encourage people to take on new assignments, small and big ones, to allow personal and professional growth. By doing so, we grow a diverse, caring team in the company, with a well-balanced gender, age and expertise spread. Ultimately, encouraging people to go out of their comfort zone, with full support, is key to get to more diverse and inclusive teams.”


Rising Star in Industry:
Anne Vogel, Senior Manager Process Engineering at Kite Pharma
“As one of the first members of the TOPX community and as a chapter lead for Women@Gilead (Kite NL), I am actively influencing the narrative around equity, diversity and inclusion. Winning the Females to Follow award is a huge milestone for me: it is a powerful acknowledgment that progress is possible when we act with intention. But for me, this is not the finish line. Instead, it is a motivator to keep working toward permanent, indisputable change that ensures equity is the standard for future generations.”

A highlight of this year’s summit was the speeches of the Inclusive-3 — a select group of three distinguished individuals who have made significant contributions to advancing equity, diversity, and inclusion within life sciences and healthcare sectors. These individuals were chosen as the 2025 Inclusive-3 and invited to take the stage to share personal insights, experiences, and key lessons on fostering a more inclusive workplace. Their stories serve as both inspiration and practical guidance for industry peers.
The 2025 TOPX Inclusive-3 winners are:
Annick Kronenburg, former Neurosurgeon & Senior Investment Manager at Oost NL
Annick Kronenburg is a neurosurgeon turned into a venture capitalist. Together with fellow doctors, Annick Kronenburg has started a campaign called “van ik naar wij”, that turned into a movement with the aim to encourage healthcare professionals to break down the (non-functional) hierarchy, to promote more equal collaboration in healthcare, and ultimately to increase job satisfaction and job retention in healthcare with the ultimate goal: from individual success to collective professional belonging.

Wiesje van der Flier, Executive Director at Alzheimer NederlandL
Wiesje van der Flier is since 1 November executive director of Alzheimer Nederland. Before that she was the scientific director of Alzheimer Center Amsterdam of Amsterdam UMC. As a scientist and executive, Wiesje has dedicated her career to find out the how we can prevent or stop Alzheimer, a devastating disease that affects many people at old age, and particularly more women than men. Not only are women at greater risk of developing Alzheimer, we are also more often caregiver for somebody with dementia. With this Inclusive-3 award TOPX would like to honor her for her relentless work and dedication and would like to encourage her in her mission to a future without dementia, for men and women.

Judith Heikoop, Co-Founder of Mitochondrial Capital
Judith Heikoop is the co-founder of Mitochondrial Capital, a women-led investment movement. With her initiative Judith aims to shift the flow of global capital and empower women as investors as well as entrepreneurs, integrating scientific breakthroughs with smart venture financing to unlock high-return investments in Life Sciences & Health, and generate health, longevity and prosperity for generations to come.

By Charisma Hehakaya
If you don’t have a laptop, studying becomes difficult. If you don’t realize that education costs more than just tuition fees, you may be caught off guard by unexpected expenses. If you don’t fully master the language, writing a motivation letter isn’t easy. In an environment where going to university is not a given, intrinsic motivation often awakens only once you finally sit in a lecture hall.
First-generation students have no one in their immediate surroundings who has attended university before. Some come from stable families whose parents encourage and support them wholeheartedly. Others grow up in less stable households, without financial backing or access to helpful social networks. Some live in areas where universities are literally far away—whether that means a remote village, rural area, city, or even another country.
These students bring with them a wealth of diversity—in ethnicity, skin color, gender, and social class. Yet they all share one experience: if you don’t know what to expect when you start university, you are bound to encounter many surprises along the way.
At university, I soon discovered many unwritten rules. Studying isn’t just about absorbing knowledge; it’s also about understanding how to study program. Each program has its own culture, norms, and expectations: how to participate in lectures, how to ask questions, how to present yourself, and even how to afford the tools needed for success, like a laptop or housing near campus. For many of my classmates, these things were normal. For me, they were not.
I grew up bilingual—Dutch at school, Malay at home. Because of that, I struggled with Dutch, and my English was almost nonexistent. Finding the right words was hard, and understanding academic texts was even harder. I took extra classes in Dutch, English, presenting, and academic writing and reading. I learned that repetition helps and that learning takes time.
Universities organize open days, but that doesn’t automatically make them open to everyone. Once you earn your high school diploma, you can apply for a selective program, such as medicine. Since the shift from a lottery-based system to selection-based admissions, student populations in such programs have become less diverse. This is striking because, especially in fields like medicine, it’s crucial that future doctors reflect the society they will serve. Yet most medical students today come from families with higher socioeconomic backgrounds. Many of them receive early encouragement, private tutoring, or expensive coaching to help them succeed in selection procedures. For students who can’t afford such support—or don’t even know it exists—the threshold is much higher.
Educational institutions may decide for themselves how to select students, as long as they use at least two different methods. But if the criteria reward knowledge that only privileged students are likely to possess—such as familiarity with the healthcare system—they unintentionally exclude others. First-generation students are rarely in a position to prepare years in advance. Research has already shown that it’s essential to consider the social and cultural background of applicants when assessing their potential.
What did being a first-generation student ultimately teach me? Not to compare myself with others. I learned to bridge the gap between people with and without academic backgrounds, and between theory and practice. I saw the same in other first-generation students: they become connectors. I discovered that language is a key to progress and that asking for help is a strength.
Equal opportunity is a social issue that universities can address. They can provide more targeted support to students from non-academic or low-income backgrounds—and make that support visible and easy to access. For many first-generation students, feeling at home at university is anything but obvious.
They often feel caught between two worlds: home and academia. Hearing that others share those feelings helped me feel less like an outsider. This is where role models matter most. And when role models are absent, it becomes even more important for teachers to make universities truly inclusive spaces. Support for “soft skills,” like finding housing or coping with setbacks, can be invaluable. Sharing experiences helps identify what students truly need. Financial support is also crucial.
When you’re young and impressionable, hearing something from an authority figure can easily shape your self-image—especially in an unfamiliar environment. But that unfamiliar world is also a source of strength. First-generation students are pioneers who, through sheer determination, bridge divides and connect social groups. They excel at translating theory into practice. They are the bridge builders between worlds—people our society urgently needs in times of growing inequality and social tension.


















At Innovation for Health 2026, we’re putting Women’s Health in the spotlight with a special pitch session to showcase your innovation and impact. Are you developing a bold, impactful solution addressing unmet needs in women’s health?
We will select various impactful initiatives to present their innovative solutions to industry leaders, investors, and healthcare professionals at Innovation for Health on 26 March 2026. These ventures will have the unique chance to gain visibility and access to IDE Group’s expertise and services to accelerate their journey.
Don’t miss your chance—apply via the website and help shape the future of women’s health.











When people talk about “inequality” in women’s health, the conversation often stops at representation: not enough women in research, on boards, or in funding rooms.
But the deeper truth is more structural. Even if tomorrow every investor and researcher were gender-balanced, the pipeline that turns ideas into diagnostics for women would still be broken. What we face isn’t simply gender inequity. It’s a market failure. One that can be measured, quantified, and, crucially, redesigned.
1. The R&D Focus Barrier: Only 1% of Pharmaceutical Research Targets Women’s Health Let’s start at the source.
Across Europe, less than 1% of pharmaceutical R&D spending outside oncology is directed toward women’s health conditions. [1]
This means 99% of early-stage scientific discovery bypasses the unique biological and clinical needs of women altogether.
EU frameworks such as Horizon Europe have funded more than €2 billion across over a thousand women’s-health projects, but this still represents a fraction of overall EU health R&D.
In other words: the scientific seedbed itself is thin. [2] When 99 out of every 100 research euros never touch women’s health, the downstream scarcity of validated diagnostics isn’t surprising - it’s inevitable.
2. The Translational Capital Barrier: Only 9–12% of Health VC in Europe Reaches FemTech Even when research exists, it rarely translates into products.In 2024, European FemTech companies raised between €339 million and €437 million, accounting for roughly 9–12% of all medtech and digital-health investment across the continent.[3,4] That may sound promising - until you realise it includes the entire spectrum: fertility tracking, pregnancy, menopause, endometriosis, mental health, and diagnostics. Within that slice, diagnostic tools remain a small minority, often considered “too niche,” “too slow,” or “too uncertain” for mainstream venture returns. The result? promising biomarkers and AI-based diagnostic models for conditions like endometriosis or PCOS rarely advance beyond pilot studies.
3. The Diagnostics Capital Barrier: Only 16% of EU Medtech Deals Involve Diagnostics Companies Diagnostics themselves — regardless of gender focus — have long struggled to attract capital.
In 2024, only 47 of 291 European medtech and digital-health deals involved diagnostic companies, representing about 16% of deal volume. [4]
While therapeutics promise billion-euro exits, diagnostics are typically seen as slow-burn investments requiring regulatory validation, clinical partnerships, and reimbursement alignment - all of which are high friction in fragmented European markets. For women’s-health diagnostics, this compounds with the upstream scarcity of genderspecific research. You can’t commercialize what hasn’t been studied, and you can’t validate what isn’t funded.

4. The Women-Founder Funding Barrier: Less Than 1% of EU VC Capital Goes to All-Female Teams Finally, even when a viable diagnostic idea exists, the leadership shaping it faces its own barrier. Across the European Union, all-female founding teams received less than 1% of total venture capital in 2024, while mixed-gender teams received about 27.8%. [5]
This founder gate matters because the majority of FemTech and women’s-health diagnostic startups are led (or co-led) by women.
If women founders systematically raise less capital, women’s-health diagnostics will remain underdeveloped by design.
5. Why This Matters
These aren’t abstract numbers. They explain why the average time to diagnose endometriosis remains 8–9 years in the UK and Europe; [6] why non-invasive diagnostic tests
for female-specific conditions remain largely unvalidated;[7] and why clinical guidelines rely on symptom exclusion rather than precision diagnostics. In economic terms, this is a failure of allocation efficiency: capital, data, and talent are misdirected away from half the population.
6. The Women’s Health Investment Opportunity
Closing the women’s health gap could add at least $1 trillion to global GDP annually by 2040 [8], driven by reduced morbidity, higher labor participation, and more healthy days lived per woman. For investors, that is a secular tailwind—bigger than a category; it is an economy-wide efficiency gain that rewards solutions which shorten diagnostic timelines and expand access.
Shortening time-to-diagnosis for conditions like endometriosis or accelerating HPV screening doesn’t only reduce suffering - it recaptures workforce participation and productivity at national scale.
Endometriosis-related sick leave alone is estimated at ≈ €30 billion per year [9]
The World Economic Forum/McKinsey analyses project ~$3 in economic growth for every $1 invested in women’s health (a 3:1 macroeconomic return). [8]
The women’s-health gap is not just a moral issue, it’s an economic inefficiency waiting to be corrected.
7. The Path Forward
Fixing this isn’t about charity or quotas, it’s about correcting a mispriced market.
Europe can act decisively by:
• Expanding targeted R&D tax credits for women’shealth conditions.
• Creating translational grant bridges between
Horizon research outputs and clinical validation programs.
• Incentivising diagnostics-specific funds with longer time horizons.
• Requiring gender equity reporting for venture portfolios.
Because until women’s health is treated as a growth market and not a “special interest”, these numbers won’t move.
This analysis was created by Dr. Derrick Khor of Medical Consulting Group in collaboration with Carmen van Vilsteren, an exited healthtech founder with deep involvement in multiple women’s health companies who’s lived this reality from both sides, building companies and navigating the barriers first-hand.


AseptiBag LT
Withstands temperatures up to -80OC
Withstands multiple freeze thaw cycles
PFAS free For cell therapy, mAbs, intermediate frozen storage

MDI AseptiBag LT single-use storage and transfer systems are designed for cold chain applications. These systems are suitable for low-temperature storage of up to -80°C, as well as multiple freeze-thaw applications involving high-value drug substances and process intermediates.

AseptiBag ULO
Protects Light sensitive drugs
Chemically defined proprietary media for mammalian cell culture

MDI AseptiBag™ ULO systems are specially designed for light sensitive media and drug products.
The MDI AseptiFlex™-T Film has a UV and light obstructing layer, co-extruded with the outer barrier layer, which minimizes transmission of light within the wavelength range of 200 nm to 780 nm.
Utrecht Science Park Bilthoven

Het Utrecht Science Park Bilthoven (USPB) biedt hoogwaardige GMP-productiefaciliteiten en groeiruimte voor biofarmaceutische en medtechbedrijven. Met Bilthoven Biologicals als anchor tenant en een terrein van 225.000 vierkante meter, is USPB dé locatie waar bedrijven soepel doorgroeien van klinische naar commerciële fase. Mede dankzij een eigen energie-infrastructuur is het park niet gevoelig voor netcongestie – dit waarborgt de betrouwbaarheid en continuïteit van productie.
Bij het vertrek van het RIVM ontstaat zo’n 50.000 m² extra ruimte voor verdere expansie en flexibiliteit. Ter voorbereiding hierop ontwikkelt USP Bilthoven het multi-tenant gebouw Impetus (10.000 m²), waarvoor recent de bouwvergunning definitief werd. Dit state-of-the-art gebouw gaat huisvesting bieden aan kantoren, labs en clean rooms voor biopharma en medtech – ingericht voor ontwikkeling, opschaling en productie volgens de hoge kwaliteitsnormen.
Jorrit van Hoorn, Director Corporate Development:
“Als onderdeel van Utrecht Science Park, groeit in Bilthoven een ecosysteem waarin wetenschap, productie en maatschappelijke impact samenvloeien.
De nieuwbouw van project Impetus markeert de volgende fase – van historische vaccinlocatie tot een internationale hotspot voor biomanufacturing en medtech. Hier werd door Jonas Salk de visie geïntroduceerd dat wetenschap in dienst moet staan van de samenleving. Bilthoven Biologicals produceert jaarlijks miljoenen vaccins voor de hele wereld. Dat is de basis waarop wij verder bouwen. Onze ‘From cell to society’-strategie staat voor een ecosysteem waarin innovaties daadwerkelijk het verschil maken in de maatschappij,” zegt Van Hoorn.
“Onderzoek naar nieuwe vaccins, kennisdeling met internationale partners en de aanwezigheid van medtech-bedrijven onderstrepen de kracht van USP Bilthoven”, aldus Van Hoorn. “Met name bij de opschaling van late klinische fases en GMP-productie is USP Bilthoven de ideale partner voor biopharma en medtech. Bedrijven die het ecosysteem versterken zijn nadrukkelijk welkom. We werken ook aan initiatieven om shared infrastructure aan te bieden met hoogwaardige GMP cleanrooms.”
Cultureel erfgoed en toekomstvisie USP Bilthoven is ontstaan uit bijzonder cultureel erfgoed. Sinds 1953, toen het terrein in gebruik werd genomen door het Rijksinstituut voor
Volksgezondheid, ligt de vaccinontwikkeling hier aan de basis. Na privatisering en overname door de Cyrus Poonawalla Group in 2012 werd Bilthoven Biologicals geboren en kwam in 2014 Utrecht Science Park Bilthoven tot stand. “De synergie tussen publieke en private samenwerkingen is door die historie vanzelfsprekend”, zegt Van Hoorn. “Die verbondenheid – met Utrecht Science Park, de academische ziekenhuizen en de brede life sciences-sector – blijft essentieel.”
Het erfgoed ondergaat de komende jaren een metamorfose: industrieel karakter blijft behouden, maar wordt geschikt gemaakt voor moderne

kantoren, labs, cleanrooms en GMP-productie.
Zo ontstaat volgens Van Hoorn een campus waar historie, innovatie en maatschappelijke impact hand in hand gaan.
Ruimte voor groei, innovatie en volledige ontzorging
Door het vertrek van het RIVM ontstaat 50.000 m² ontwikkelruimte. Met Impetus komt daar snel 10.000 m² moderne, flexibele voor labs en clean room ruimte bij. “De groene campus biedt ruimte voor samenwerking en innovatie, met centrale voorzieningen, hoogwaardige cleanrooms en productieruimtes – alles binnen een landschap dat erfgoed respecteert”, zegt Van Hoorn.
De eigen energie-infrastructuur maakt USP Bilthoven aantrekkelijk voor bedrijven die willen opschalen zonder risico op onderbrekingen, aldus Van Hoorn. Voor biomanufacturing en medtech zijn betrouwbaarheid en compliance cruciaal. USP Bilthovenbiedt GMP-faciliteiten, cleanrooms en gedeelde infrastructuur waarmee bedrijven veilig van prototype naar productie groeien, zegt Van Hoorn. “Of het nu gaat om medical devices, diagnostiek of biologicals: USP Bilthoven biedt
letterlijk de ruimte en zekerheid om productie op te schalen.”
Pandemic preparedness en maatschappelijke rol USP Bilthoven speelt een strategische rol in pandemic preparedness. Bilthoven Biologicals is wereldwijd bekend vanwege haar essentiële vaccins, nu aangevuld met Pandemic Preparedness faciliteiten voor viral vectors. Dit biedt mogelijkheden voor partners die bestaande GMP productiecapaciteit zoeken voor de opschaling en productie van vaccins, viral vectors of immunotherapie. “De unieke kennis en capaciteit vormen het fundament voor USP Bilthoven’s maatschappelijke missie, aldus Van Hoorn.
Pandemic preparedness vraagt om schaalbare, betrouwbare infrastructuur, die direct ingezet kan worden. De campus brengt dit samen – en kan snel schakelen wanneer de situatie daarom vraagt.”
Facilitaire ontzorging
USP Bilthoven onderscheidt zich met uitgebreide services: 3.000 zonnepanelen, professioneel gebouwbeheer, beveiliging, toegangscontrole en horeca. “Huurders concentreren zich zo volledig
op innovatie; USP Bilthoven regelt de rest. Die volledige ontzorging staat centraal in de visie: een duurzame campus die groei en impact ondersteunt”, aldus Van Hoorn.
Van wetenschap tot impact
Met ‘From Cell to Society’ positioneert USP Bilthoven zich als dé plek waar wetenschap, technologie en productie samenkomen – direct midden in Nederland. Hier groeien innovaties uit tot toepassingen met wereldwijde impact. Zoals Jorrit van Hoorn afsluit: “USP Bilthoven is een campus waar innovatie kan opschalen, medtech en biopharma zich thuis voelen, en wetenschap tastbare impact maakt – van cel tot maatschappij.”


What are the underlying principles that defines a real collaborative success? One true success story is the growing partnership between Leiden in the Netherlands and Lund in Sweden. It is a crossborder alliance that is accelerating advances in cell and gene therapy, showing that combined efforts and merged ecosystems can provide foundation for breakthrough therapies.
Meanwhile, LUMC had already in 2021 become one of three global sites for the Novo Nordisk Foundation’s prestigious reNEW stem cell medicine initiative.
The following year, the Netherlands inaugurated NecstGen, a state-of-the-art facility for GMP manufacturing of cell and gene therapies. On the Swedish side, Lund complemented its first-in-human trial facility with a new Pre-GMP center, opened early

From royal visit to real value
The partnership in cell and gene therapy was officially launched in October 2022 during a state visit by the King and Queen of the Netherlands to Sweden. A memorandum of understanding (MoU) was personally signed by LUMC dean, Lund University vice chancellor, and Skåne University Hospital CEO, ensuring highlevel commitment. The MoU was only possible thanks to world-class research environments on both sides, a strict deadline and aspiration for excellence.
Boosting talent and infrastructure
At the working level, the collaboration has been steered by Prof. Stefan Jovinge (Lund) and Prof. Niels Geijsen (Leiden). What began as a scientific project and an idea of a joint master’s program, rapidly expanded. Later in 2022, Lund was selected to join Sweden’s national doctoral program in advanced therapies. Now the Leiden–Lund collaboration is leading RAMP-UP, a Marie Skłodowska-Curie doctoral training network, funding 55 PhD students across Northern Europe. Most of the students will study in Leiden or Lund, ensuring access of top-tier talent in cell and gene therapy.

in 2025. Together, these infrastructures in Leiden and Lund, form a complete pipeline from early research to clinical application.
An integration of ecosystems
What sets this collaboration apart is that it goes beyond single projects. The universities frequently exchange lecturers and internships, and infrastructures connect and collaborate across ecosystems. Dutch companies have become regular participants in Sweden’s conferences on Advanced Therapy Medicinal Product (ATMP), and Swedish biotech firms are exploring partnerships in the Netherlands.
One example is Utrecht-based Scinus Cell Expansion that recently teamed up with Lund Stem Cell Center in a project funded through Eurostars. Another example is Amarna Therapeutics from Leiden and Sweden’s NorthX Biologics, that are combining efforts to support future clinical trials of next-generation gene therapy. Both Sweden and the Netherlands are small countries with world class research mutually benefiting from working together.
Regional to national level
Sweden has already built a strong platform called ATMP Sweden, which brings together hospitals, universities, and companies under one umbrella. The Netherlands is now developing a similar node, inspired by the Swedish model.
Looking forward, the partnership is set to scale. In early 2025, the Netherlands Embassy in Stockholm hosted a roundtable with key stakeholders from both countries, discussing production, training, and industry–academia collaboration. ATMP-production and development are complex endeavors and offer many opportunities. Combining the ecosystems will provide a critical mass to deliver complete therapies.
Internationalization of ATMP is crucial
Cell and gene therapies are among the most promising advances in modern medicine. Cell therapy involves restoring or modifying cells to fight disease, using the patient’s own cells or those from a donor. Gene therapy addresses inherited disorders at the DNA level, offering not just treatments but potential cures. Together with tissue-engineered products, they form a class of treatments known as ATMPs. But while science is progressing fast, the path to clinical application is complex. Manufacturing capacity, training of skilled personnel, and sustainable healthcare financing are major hurdles. To tackle these challenges, international collaboration is crucial.
Successful international collaboration is possible when all parties see clear benefits. In the LeidenLund case, hospitals and universities shared a strong incentive to speed up the development and delivery of advanced therapies to patients. Companies discover business opportunities in a larger ecosystem and for governments, cell and gene therapy offers a gamechanging opportunity to treat more patients with fewer resources. By connecting the ecosystems of northern Europe, starting with Sweden and the Netherlands, collaboration can drive innovation and strengthen leadership in life science.
By Björn Ursing, the Embassy of the Kingdom of the Netherlands in Stockholm

Eli Lilly and Company’s decision to invest more than USD 3 billion in a new manufacturing facility in Katwijk marks a significant step forward for the Dutch life-sciences landscape. The planned site, which will become part of the Leiden Bio Science Park ecosystem, is intended to strengthen the company’s global supply chain and increase its capacity to deliver next-generation medicines.
The investment comes with 500 new high-skilled positions. These will range from engineers and laboratory specialists to scientists and operational staff. Working with advanced technologies, these teams will focus on the production of complex, often biologically derived medicines. As a result, the site is expected to contribute not only to economic growth in the region but also to the development of expertise within the Netherlands’ rapidly expanding biopharmaceutical sector.
A catalyst for the regional knowledge economy Over the last decades, the Leiden Bio Science Park has grown into a leading European cluster where research institutions, start-ups, established biotech companies and industrial partners cooperate intensively. Stakeholders view Lilly’s arrival as a further reinforcement of this collaboration model.
According to the company, the combination of a skilled talent pool, reliable logistics and a proven manufacturing environment was decisive in choosing the location. David A. Ricks, Lilly’s chair and CEO, highlighted Leiden’s long track record in
pharmaceutical R&D and described the region as a stable, innovation-friendly environment suited for substantial, long-term investment.
Dutch Minister of Economic Affairs Vincent Karremans similarly welcomed the decision, noting that the selection of Katwijk and Leiden—after comparisons with various European sites—confirms the Netherlands’ attractiveness for innovative, research-driven industry. He emphasized that the project is expected to strengthen national and regional collaboration in the development of new medical treatments that directly improve patients’ lives.
Strengthening an end-to-end life-sciences ecosystem
Once operational, the new plant will become embedded in Leiden’s “end-to-end” life-sciences chain, integrating early-stage research, technology development and large-scale biopharmaceutical
production within a single region. Representatives of the Leiden Bio Science Park consider this structure essential to maintaining international competitiveness, particularly within red biotechnology and the broader life-sciences and health domain.
By keeping knowledge creation, innovation and manufacturing geographically close, the region continues to attract new companies and investment. Lilly’s choice is seen as evidence that this integrated model works—and that it offers a platform capable of supporting the development and production of modern, high-value medicines.
Additional momentum comes from recent findings by the European Patent Office, which identified Leiden University Medical Center (LUMC) as one of Europe’s leaders in technology transfer. This strong connection between academic research and market-ready innovation aligns closely with the type of activities Lilly aims to expand, reinforcing Leiden’s role as a place where scientific breakthroughs are translated into real-world health solutions.
Broad impact for the sector and the region
The combined effects of job creation, scientific collaboration, knowledge exchange and added manufacturing capacity make Lilly’s investment a milestone for the Dutch life-sciences sector. Once the Katwijk facility becomes operational, it is expected to play an important role in the global distribution of a new generation of therapeutics.
With this project, the Leiden Bio Science Park strengthens its ambition to develop into one of Europe’s most influential health and biomedical innovation hubs.

Anchoring innovation in a changing world:

In a time when biomedical innovation is accelerating, the challenge is no longer just about developing new therapies. It’s about making them matter — for professionals, patients, and society alike. At Schuttelaar & Partners, we believe that science alone isn’t enough. It needs to be embedded in a broader societal context. How do you communicate in a clear and engaging manner? How do you keep the dialogue with your stakeholders going in times of polarization and how do we build a healthcare system that is both sustainable and fair? These questions drive our work.
Over the past years we have covered diverse topics: vaccination, oncology breakthroughs, a healthy lifestyle, prevention of smoking, orphan drugs and many more. The common thread in our work? To bring stakeholders together. Through thoughtful positioning, impactful communication and interactive events – both small and large. Our team is well equipped to dive into each topic and context as it consists of professionals with roots in life sciences, public health, policy making, communication and public perception. Our mission — to contribute to a healthy and sustainable world — guides us in our day-to-day work.
Understanding the landscape and the people in it
One of our strengths lies in knowing the field. Not just the technologies or the regulations, but the people. We understand the dynamics of biomedical professionals, the expectations of policymakers, and the concerns of patients and the public. That insight allows us to help organizations position themselves — not just in terms of what they do, but why it matters.
Take the launch of Oncode Accelerator. We helped shape Oncode Accelerator not just as a brand, but as an initiative that connects scientific excellence with societal relevance. By crafting a
Mark your calendars
Start your new year with some fresh perspective. S&P organizes a Health Knowledge week from 19 to 23 January 2026. Learn more about what’s cooking in Brussels, the how and what of our LCA approach and get insights into healthy aging through a guest lecture of Prof. Eline Slagboom and Dr. Joris Deelen of the Leiden University Medical Center.You can find more information and a registration form on our website, scan the QR code below.
Not through one-off meetings, but through a structured engagement process of several years that has led to concrete steps forward, recommendations, and renewed momentum. Visit www.weesgeneesmiddelen.nl to learn more about this initiative.
Sustainability: from compliance to impact Health and sustainability are deeply connected. In the Netherlands, about 7% of the national carbon footprint comes from the healthcare sector. At the same time, climate change and nature degradation increasingly threaten global health — through extreme weather, shifting disease patterns, and the loss of ecosystems that support wellbeing. We believe the healthcare sector has both the responsibility and the opportunity to lead. Society expects more than medical safety and efficacy; it calls for visible commitment to reducing environmental impact. We help organizations respond by shaping ESG strategies and conducting environmental impact assessments.
Want to move forward?
Whether you’re developing breakthrough therapies, navigating ESG regulations, or seeking to engage stakeholders meaningfully — we’re here to help. Let’s reconnect and build a healthier, more sustainable future. Together.
Curious about Schuttelaar & Partners?
To celebrate our 30 years anniversary we have made a new brand manifesto. Scan the QR code below to have a look and join the movement.
clear narrative, designing a strong visual identity, and bringing together key stakeholders for a festive launch, we helped build a force in oncology innovation.
Building bridges that lead to action Meaningful innovation doesn’t happen in isolation. To have societal impact, innovators require trust, dialogue, and shared direction with the societies they work in. That’s why stakeholder engagement is at the heart of our work since our inception in 1995. After 30 years, we see it’s time to go back to these roots. In a polarizing society, it’s more important than ever to reconnect business and society. S&P helps its clients with making progress. Not only from our headquarters in The Netherlands, but also internationally from our office in Brussels. And these efforts deliver results.
A recent example is our work with the Association for Innovative Medicines (VIG) on orphan medicines. These treatments often face complex developmental, regulatory and reimbursement hurdles, and their societal value is not always well understood. We brought together patient representatives, policymakers, healthcare professionals, researchers, payers and industry representatives to explore shared solutions.
Take ESG reporting. While the CSRD has brought complexity and delay, we see reporting not as a burden but as a strategic tool. Done well, it informs decisions, builds trust, and connects companies to the broader societal transition. We support clients with sector-specific insight and environmental expertise. Together with Healthbridgers, we’ve built the capacity to conduct life cycle assessments of pharmaceutical products and diagnostics. These assessments quantify carbon footprints, reveal nature-related dependencies, and identify reduction hotspots — turning data into action for a more sustainable healthcare system.
A story that moves forward
What ties our work together is a commitment to clarity, connection, and collective progress. Whether we’re helping an innovative organization find its voice, facilitating dialogue on rare diseases, or calculating the environmental footprint of a new therapy — we approach each challenge with a cool head, a sharp pen, and a clear purpose. In a world full of opinions, we anchor ourselves in facts. In a sector full of complexity, we help bring clarity. And in a time of rapid change, we offer direction.

Nederland #2 wereldwijd in quantumwetenschap; investeringen quantumbedrijven verzestienvoudigd
Nederland staat nu tweede wereldwijd in de wetenschap rond quantumcomputing, -sensing en -communicatie. Tegelijkertijd groeide het private kapitaal voor Nederlandse quantumbedrijven in vijf jaar van €10 miljoen (2019) naar €160 miljoen (2025). Dat blijkt uit de data-update, De Staat van Quantum, die Quantum Delta NL in november publiceerde. De update bevat kerncijfers over wetenschappelijke output, ondernemerschap, werkgelegenheid en patenten in het Nederlandse quantumecosysteem. Volgens Quantum Delta NL vraagt het vasthouden en verzilveren van deze voorsprong om stabiele, meerjarige investeringen.
Sinds 2021 versterkt Nederland via Quantum Delta NL het nationale quantumecosysteem. Rond verschillende hubs door heel Nederland komen onderzoek, talent, ondernemerschap en maakcapaciteit samen. Met steun van het Nationaal Groeifonds positioneert Nederland zich als internationaal centrum en knooppunt voor quantumtechnologie.
De in november gedeelde data-update laat zien dat het ecosysteem in korte tijd in omvang en volwassenheid is toegenomen. In 2025 telden Nederlandse quantumstartups 735 banen, waarvan circa 400 in Delft; dat is ruim vier keer zoveel als in 2020. Het aantal start-ups groeide in vijf jaar naar 29 en de private investeringen namen in dezelfde periode met 1.600 procent toe, van €10 miljoen naar €160 miljoen. Ook de innovatiekracht versnelt: het aantal quantumgerelateerde patentaanvragen steeg van 11 in 2020 naar 60 in 2025. Recente Series A-rondes bij onder meer QuantWare, Qblox, QuiX en Orange Quantum Systems onderstrepen dat internationale investeerders Nederland weten te vinden.
De eerste toepassingen dienen zich inmiddels aan. In de zorg worden quantumsensoren ingezet voor scherper MRI-onderzoek. En bijvoorbeeld QTSense, een startup gespecialiseerd in Life Science and Health, gebruikt geavanceerde quantumsensoren om in real-time individuele cellen te analyseren. En zo zijn er nog meer voorbeelden die laten zien dat de stap van lab naar fabriek in zicht komt.
Quantum Delta NL zoekt contact met de Life Science and Health sector om te onderzoeken waar quantum impact kan maken. Bezoek onze stand en seminar op de Innovation for Health conferentie om meer te weten te komen.
– The
Anacura, a family-owned, growing center of excellence in healthcare with over 250 employees consists of 4 independent business units: Labo Nuytinck, active in medical analytics and AnaBioTec, OHMX.bio and Discovery Studio, active in drug development. The focus is on advising health delivering outcomes. The labs have their main activities in a state-of-art building in a green surrounding near Ghent, Belgium.
Labo Nuytinck delivers a high quality service in the area of laboratory medicine. Our vision is that a person’s health is served by their own strength and insight, supported by a network of trusted professionals. We envision a healthcare ecosystem where partners – practitioners, healthcare providers and the laboratory – jointly guarantee high-quality and affordable care.
AnaBioTec provides analytical services for characterization, assay development and quality assessment of novel and established medicines, ranging from synthetic molecules and biologics, to cell & gene therapies. With more than 25 years of experience we contribute to the medicines of tomorrow by helping our customers to accelerate their drug development journey. We support startups, biotech as well as large pharma companies worldwide from pre-clinical drug development, through clinical phases I, II, III all the way to commercialised treatments.
OHMX.bio specializes in multi-omics technologies, enabling a deeper understanding of biological systems through the integration of (epi)genomics, transcriptomics and proteomics data. The portfolio meets the increased demand for characterization of DNA, RNA, and proteins.
Discovery Studio is a biotech accelerator focused on translating groundbreaking concepts into therapies.



Science Talents – Think like a proton, always positive.
Heb je een passie voor wetenschap en ben je klaar voor een nieuwe uitdaging? Bij Science Talents vind je een ruime waaier aan jobs die perfect aansluiten bij jouw achtergrond en ambities. Dankzij onze specialisatie zijn we al jaren dé referentie in sectoren zoals pharma, biotech, healthcare, chemie en voeding.
Wij bieden jou verschillende carrièremogelijkheden. Als Consultant of Freelancer kom je terecht bij gerenommeerde klanten en werk je op uitdagende projecten binnen Pharma, Biotech, Healthcare, Voeding of Chemie. Een carrière bij Science Talents betekent variatie, persoonlijke ontwikkeling en een sterk netwerk van wetenschappelijke collega’s. Bovendien geniet je als consultant van aantrekkelijke voordelen, waaronder een contract van onbepaalde duur, extralegale voordelen én een bedrijfswagen.
Ook via Werving & Selectie helpen we je de volgende stap te zetten. In opdracht van onze klanten zoeken we vaste medewerkers voor complexe wetenschappelijke functies. Daarbij zorgen we voor de perfecte match tussen jouw expertise en hun noden, zodat jij meteen in dienst gaat bij onze klant. Of je nu net afgestudeerd bent en je carrière een vliegende start wil geven, al ervaring hebt en je loopbaan naar een hoger niveau wil tillen, of liever kiest voor de vrijheid van freelancen: Science Talents biedt kansen op maat.
Onze expertise strekt zich uit over verschillende domeinen. In de Life Sciences werken we rond clinical research, process validation, regulatory affairs, market access, quality assurance en meer. Binnen de Chemische Industrie ondersteunen we labo’s, R&D en productie. In de Voedingsindustrie vind je projecten binnen R&D, QA/QC en productie. Daarnaast detacheren we preventieprofielen die bedrijven helpen hun veiligheids- en welzijnsbeleid te versterken.
Samen met jou bouwen we aan de wetenschap van morgen. Ontdek jouw mogelijkheden bij Science Talents!


By Pernilla Isberg
After many great conversations at recent events, I’ve been reflecting on what really makes networking meaningful — and why it’s such a powerful catalyst for both personal and professional growth.
Success - for both you and your company - often comes down to one thing: your network. And here’s the truth: only you can build, nurture, and make use of it.
Networking isn’t about collecting business cards or polite small talk. It’s about creating potential - by provoking coincidences, thinking creatively, and daring to connect ideas, people, and possibilities.
Challenge yourself to change your approach: Instead of asking “How are you?”, try something that opens up the conversation and gives insight into what drives the person in front of you. For example:
�� “What brought you to this event?”
�� “What part of the presentation resonated most with you?”
�� “What are you currently most curious or excited about?”
People connect with your energy, not your title. Be curious. Be authentic.

�� Mindset
• Be confident, warm, and friendly - others are likely just as uneasy as you.
• Go out of your comfort zone - this is work, not hiding behind familiar faces.
• Bring your personality - curiosity, positivity, and genuine interest make you memorable.
�� Preparation
• Check the guest list (if available) - identify who you’d like to meet and possible links.
• Know your key messages - what do you want to share or learn? Be ready for the unexpected.
• Have a short, passionate “elevator pitch” (20–30 sec).
• Appearance matters - dress as you, with confi dence. When you feel comfortable, you radiate it.
☕ At the Event
• Work the room - don’t cluster with colleagues.
• Ask questions that spark real dialogue. Connect people if you see overlaps
• Focus on what you can give to others, and not primarily on what you might gain yourself.
• Listen actively - focus on quality over quantity.
• Be generous with your contacts. Introduce people to eachother - even people you just met.
• Be memorable - in a positive, authentic way.
• Exit gracefully - “Good luck, let me know how it goes!”
• And yes - drink coffee and dive on people in the coffee queue! ☕��
�� Post Event
• Follow up within 2 days - email or LinkedIn while energy is still fresh.
• Personalize your message - mention something from your conversation.
• Track and tag your contacts - think long-term, not one-off.
• Keep connections alive - small gestures go a long way.
Pernilla Isberg is a pharmacist with +30 years of experience from working within AstraZeneca with marketing, clinical trials and cross functional project management. Since 2018 Pernilla is Director at AZ BioVentureHub –A unique innovation platform located within AstraZeneca’s Gothenburg R&D site in Sweden, supporting emerging companies to grow. Access to AstraZeneca’s scientific expertise and facilities, collaboration opportunities, and a vibrant community. The focus is on non-competitive synergies and shared challenges we can solve together across Data & AI, DeepTech, SustainTech, and Pharma (new modalities and drug delivery technologies).

Networking can be trained like a muscle - with practice, purpose, and presence. By putting yourself out there, sharing what excites you, and staying curious, you increase your chances of encountering the kind of “luck” �� that transforms opportunities into impact.
Huurgarantie voor innovatief bedrijfsverzamelgebouw Beagle Marconi

Met de recente huurgarantie van het Stadshavens Ontwikkelingsfonds Innovatie en Economie komt de realisatie van Beagle Marconi een belangrijke stap dichterbij. Dit innovatieve bedrijfsverzamelgebouw in de Rotterdamse Merwe-Vierhavens (M4H) biedt straks ruimte aan jonge, snelgroeiende bedrijven die actief zijn op het snijvlak van (bio)technologie, energietransitie en maritieme innovatie.
De huurgarantie doorbreekt een klassieke kipei-situatie: ontwikkelaars en financiers willen zekerheid over huurders, terwijl huurders pas tekenen als het project van start gaat. Dankzij de steun van het fonds is deze impasse doorbroken.
Beagle Marconi wordt een belangrijke schakel in de transformatie van M4H tot toonaangevend innovatiegebied. Het gebouw stimuleert samenwerking tussen ondernemers, onderzoekers en studenten, en draagt bij aan duurzame economische groei in de Rotterdamse havenregio. De steun van het Stadshavensfonds biedt niet alleen financiële zekerheid, maar ook een krachtig signaal van vertrouwen in de toekomst van de innovatieve maakindustrie in de stad.
Beagle Marconi is een ontwikkeling van Beagle Marconi B.V., een samenwerking tussen TBIonderneming J.P. van Eesteren en Beagle Building B.V. Geïnteresseerd in samenwerking of huisvesting? Kijk op www.beaglemarconi.com.

How The Think Room Helps Organizations and Professionals Scale Authentically

In an era where artificial intelligence can replicate almost anything, the most valuable asset isn’t what you can generate, it’s what makes you uniquely you. This challenge resonates deeply across the life sciences, where professionals face pressure to leverage AI tools while maintaining the human insight that drives breakthrough discoveries.
The Think Room, led by CEO Liora Kern, addresses this modern paradox with unique industry insight. Kern brings hands-on experience from her background in medical technology at 3M, and years of collaborations with organizations like Johnson & Johnson, Mylan, Vaccines Europe and The Influenza Hub. This foundation informs her current mission: Helping organizations and professionals find what makes them unique and then scale it with AI, without losing their core identity or their humanity.
The Think Room’s approach recognizes that while AI can amplify capability, it cannot replace authentic expertise and human judgment. The Think Room works with clients to first identify what makes them irreplaceable: Their unique perspective, expertise, and value proposition, before strategically integrating AI tools to amplify these strengths.
The Think Room’s uses a bespoke methodology to unlock organizational and professional DNA and has in-depth knowledge of how to use AI tools (LLMs).
The Think Room’s process begins with organizational DNA discovery, uncovering the authentic core that makes it different. From there, they develop strategies to scale this uniqueness using AI tools, ensuring that increased efficiency never comes at the cost of authenticity or integrity.
Discover your unique organizational DNA at thethinkroom.eu or contact liora@thethinkroom. eu to learn how AI can amplify your authenticity.


Nieuwe hightech hartbewakingsafdeling geopend in Albert Schweitzer ziekenhuis
De nieuwe afdeling voor hartbewaking (CCU) van het Albert Schweitzer ziekenhuis in Dordrecht is officieel in gebruik genomen. Het ontwerp van de afdeling is gerealiseerd door KuiperCompagnons, in nauwe samenwerking met het ziekenhuis en de gebruikers.
Ruimte, licht en technologie vormen de kern van het ontwerp. De ruime suites zijn uitgerust met geavanceerde medische apparatuur en profiteren van overvloedig daglicht. Glazen puien verbinden de kamers met de gang en zorgen voor een open, transparante omgeving die zowel patiënten als zorgprofessionals ondersteunt.
De nieuwe CCU sluit naadloos aan bij de bestaande architectuur van het ziekenhuis en vormt een moderne, toekomstbestendige zorgomgeving waarin menselijkheid en hightech zorg samenkomen.
KuiperCompagnons werkte intensief samen met het bouwteam en de gebruikers om een afdeling te creëren die zowel functioneel als helend is.



Do you also have a new job or won a prize? Let our editors know via biotechnews@hyphenprojects.nl
Wiesje van der Flier
Executive Director at Alzheimer Nederland (November 2025)
Marco de Boer
CBO at Biotech Booster (November 2025)
Antoine Maupu
Vice President & Site Head of Kite’s European cell therapy manufacturing facility in Hoofddorp (October 2025)
Joost Kreijtz
CEO of Trained Therapeutix (October 2025)
Marja van Genugten
COO at Hycult Biotech (October 2025)
Kim Heylen
COO ad interim at BioLizard (October 2025)
Katie Chapman
CSO at Tessellate Bio (October 2025)
Richard Janssen
VP Portfolio Insights and External Innovation at Byondis (October 2025)
Mathias Vinther Partner at Forbion (October 2025)
Bassem Saleh
CEO Julius Clinical (September 2025)
Sanne Weijzen
Head of Cell Therapy Business and Portfolio Strategy at AstraZeneca (September 2025)
Jasper Levink CEO Khondrion (September 2025)
Roland Bürli
Chief Scientific Officer at Scenic Biotech (September 2025)
Jetty van Ginkel
Managing Director & Board Member of hDMT (August 2025)
Esther Breij
Sr VP, Head of Translational Sciences at Avidicure (August 2025)

Marloes Pennings of TU Eindhoven has won the jury prize and Thomas van Gelder of Utrecht UMC has won the public prize of the FIGON Dutch Medicines Days PhD Student Competition of 2025.
Manon den Uil of UMC Utrecht, Rosalie van Dijken of UMC Groningen and Tomás Santos Coimbra of MIMETAS won the poster awards at the FIGON Dutch Medicines Days 2025.
Professor Luke O’Neill of Trinity College Dublin is the 2025 laureate of the Ariëns Award of the NVF (Dutch Pharmacological Society).
Thijn Brummelkamp received the Spinoza Prize awarded by NWO.
Maroeska te Loo, Veroniek Harbers, Edith Klappe and Carine van der Vleuten of Radboudumc SAVE team won the first Drug Repurposing Venture Challenge.

Arlene John, Assistant Professor at University of Twente has won the TOPX Females to Follow Award – Rising Star in the category Science.
Hanne Callewaert, CEO of AstriVax Therapeutics has won the TOPX Females to Follow Award – Rising Star in the category Entrepreneurship.
Anne Vogel, Sr Manager Process Engineering at Kite Pharma has won the TOPX Females to Follow Award – Rising Star in the category Industry.
Annick Kronenburg (Oost NL), Wiesje van der Flier (Alzheimer Nederland) and Judith Heikoop (Mitochondrial Capital) have received the TOPX Inclusive-3 award at the 2025 edition of TOPX Summit.
Liesbeth Ceelen, CEO of BioLizard, won the Venture Award of Vlerick Business School
Jan van de Winkel, CEO of Genmab, won the European Lifestars – Lifetime Achievement Award





The Dutch Medicines Days bring together professionals from across the pharmaceutical sciences to share new research, exchange experiences, and strengthen collaboration both in the Netherlands and abroad.
At the conference, experts from universities, industry, and regulatory organizations meet to discuss recent developments in research, new therapeutic approaches, and future directions in drug innovation. Through inspiring talks, focused sessions, and poster presentations, participants can gain fresh insights, connect with colleagues, and build partnerships that contribute to the future of healthcare.



“Dutch Medicine Days provides unique opportunity where cutting-edge science meets frontier innovation, and ideas quickly turn into collaboration.”


us for Sponsorship & Exhibition Opportunities: E-mail: dmd@hyphenprojects.nl Tel: 035 628 66 59


Zorg en bedrijven versterken health-innovatie in Noord-Nederland:

GRONINGEN – Noord-Nederland bundelt de krachten om de zorg toekomstbestendig te maken. Zorginstellingen, kennispartners, overheden en technologiebedrijven werken voortaan structureel samen binnen het Digital Health Ecosysteem Noord-Nederland (DHENN) en de European Digital Innovation Hub Noord-Nederland (EDIH NN). De samenwerking versnelt de inzet van AI, robotisering en andere digitale innovaties om de zorg betaalbaar en werkbaar te houden.
Zorginnovaties sneller laten slagen “De zorg staat voor een enorme opgave,” zegt Martin Smit, directeur van het UMCG Innovation Center en vertegenwoordiger vanuit DHENN. “De zorgvraag groeit, maar het aantal professionals niet. Alleen door technologie slim in te zetten, kunnen we de kwaliteit en betaalbaarheid van zorg behouden. Daarvoor hebben we het bedrijfsleven nodig.”
Precies daar zet de samenwerking op in. DHENN vertegenwoordigt het ecosysteem rond digitale gezondheid, EDIH NN brengt het bedrijfsleven en technologische oplossingen samen. “We kijken niet alleen naar vraag of aanbod, maar naar de hele markt,” legt Robin Brouwer, programmamanager bij DHENN, uit. “Zo ontstaat een directe lijn tussen wat de zorg nodig heeft en wat de markt kan bieden.”
“Die koppeling is essentieel”, vult Paul Mulder, innovatiemakelaar Life Science & Health bij EDIH NN, aan. “Negen van de tien zorginnovaties stranden in de pilotfase. Door de zorgvraag en het bedrijfsaanbod beter op elkaar af te stemmen, vergroten we de kans dat innovaties slagen.”
Technologie in dienst van de zorg
Door de samenwerking krijgen zorginstellingen sneller toegang tot relevante technologie en bedrijven beter zicht op wat de zorg écht nodig heeft. Dat levert tastbare voordelen op. “De grootste uitdaging is dat technologie in de zorg ook echt wordt toegepast,” zegt Smit. “Dat vraagt om een scherp beeld van welke innovaties de zorg nodig heeft, zodat bedrijven daarop de juiste oplossingen kunnen ontwikkelen.”
Alle innovaties staan in dienst van de zorg, benadrukt Leo van der Burg, business developer bij FME, een consortiumpartner van EDIH NN.
“We richten ons op autonome toepassingen waarin AI, robotisering en digitalisering samenkomen. Zulke systemen nemen routinetaken over en ondersteunen professionals bij complexe beslissingen, terwijl de regie bij de mens blijft.”
Door gezamenlijke standaarden hoeven ITafdelingen minder te koppelen en kunnen ondernemers hun technologie makkelijker
opschalen. Tegelijkertijd worden zorgprofessionals aangemoedigd om hun behoeften actief te delen met het bedrijfsleven.
Op naar een gezonder Noord-Nederland “Innovatie gebeurt niet op afstand, maar tussen mensen die elkaar kennen en samen willen leren,” zegt Van der Burg, die vanuit FME inzet op de regionale samenwerking en zichtbaarheid. “Dat maakt dit zo’n kansrijke ontwikkeling voor NoordNederland.”
De aanpak zorgt bovendien voor focus en continuïteit, vult Mulder aan. “Kennis, zorg en bedrijfsleven werken nu samen in één ontwikkelproces. Daardoor stijgt de kans dat innovaties echt slagen, en daar profiteert iedereen van: van zorginstelling tot ondernemer, en de patiënten, voor wie we het doen.”
Zo raakt innovatie direct aan het belangrijkste doel: een gezonder Noord-Nederland. “We voelen ons niet alleen verantwoordelijk voor de patiënten in ons ziekenhuis,” besluit Smit. “In heel NoordNederland willen we mensen gezond houden.”
Agomab announced positive topline phase 2a data for Ontunisertib in Fibrostenosing Crohn’s Disease.
myTomorrows raised $29 Million to Scale and Transform Access to Innovative Therapies for Patients Worldwide.
Citryll presented positive data from phase I study of CIT-013 in Rheumatoid Arthritis at ACR convergence 2025.
Sensius secured €15 million to advance breakthrough cancer thermotherapy.
AstraZeneca announced a $70 million investment to expand its manufacturing facility in Nijmegen, the Netherlands.
MRM Health raised €55 million Series B financing to develop first-in-class therapeutics for patients with inflammatory bowel diseases, auto-immune diseases and cancer by harnessing the full power of the gut microbiome.
Sam Possemiers, CEO of MRM Health: “This funding marks a pivotal moment for MRM Health. With the support of our experienced international investor syndicate, we are now well positioned to deliver transformative therapies for patients with chronic inflammatory diseases, where current treatments often fall short. Our CORAL® platform gives us a unique edge in designing potent, resilient and scalable microbiome-based therapeutics. This new funding will allow us to further build both our platform and pipeline.”

Rapidemic has received a $2.7M grant from the Gates Foundation to accelerate an ultrarapid, power-free molecular diagnostic that delivers results for Chlamydia trachomatis and Neisseria gonorrhoeae in under 15 minutes.
Aiosyn has raised €2.4 million to accelerate the development and rollout of its AI-driven diagnostic solutions, aimed at improving cancer care.
VarmX announced that their lead asset VMX-C001 has been granted Fast Track Designation by the US FDA.
Citryll has successfully dosed the first patient in its Phase IIa clinical trial evaluating CIT-013 in rheumatoid arthritis.
Amplio Pharma received EMA approval for Phase 1b trial in patients with rheumatoid arthritis.
Marguerite Mensonides, CEO of Amplio Pharma: ”Our close collaboration with Amsterdam UMC, Leiden UMC, CHDR and OctoRelease have made it possible to reach this first milestone on our clinical journey with NovoBioJect. We look forward to reaching the next ones in the coming months.”
Confo Therapeutics awarded VLAIO grant for development of ultra-long-acting antibody-based GPCR therapeutics in endocrine and metabolic conditions.
VarmX partners with CSL in a strategic collaboration & option agreement to develop novel investigational coagulation treatment.
SkylineDx has been named “Diagnostics Company of the Year” at the 2025 BioTech Breakthrough award.
Leyden Labs announced a new equity investment of €30M to advance ongoing development of novel antibody-based nasal sprays to protect against respiratory viruses, such as influenza.
Koenraad Wiedhaup, co-founder and CEO of Leyden Labs: “With PanFlu, we are taking a bold step to stop influenza viruses at the gate. We are proud to welcome these two esteemed European funds, each with the goal of accelerating innovations in Europe that deliver public benefit. Their support for our strategy to develop novel products to protect against respiratory viruses is a wonderful endorsement of our relentless efforts.”


AM-Pharma completed enrollment in Phase 2 Study of Ilofotase Alfa for prevention of cardiac surgery-associated renal damage.
Juliane Bernholz, CEO of AM-Pharma: “Reaching this enrollment milestone is a key inflection point in the clinical development of ilofotase alfa, bringing us closer to addressing a significant unmet need in hospital care. We are grateful to our dedicated clinical partners and the patients for their vital support and trust. With recruitment now complete, we are focused on the data that will inform our next steps to realize the potential for ilofotase alfa to become the first approved therapy to prevent CSA-RD.”

Name Start-up Company: AdJane
Name (co-)Founder: Family Dynamic Innovations (FDI)
Leadership: Anita Gashi, Director
Mission Statement: Amplifying the vaccines of tomorrow

antigenic composition and intrinsic adjuvanticity of OMVs, delivering a versatile and clinically validated platform for safer, more effective, and longerlasting vaccination. The company’s lead candidate targets gonorrhea, a WHO-prioritized pathogen with no preventive solution.
reproductive complications such as infertility, untreated infections increase susceptibility to HIV and can cause serious neonatal outcomes, including infection-related blindness. Despite decades of research, no licensed vaccine exists, underscoring the urgent need for a preventive solution.
Our Solution:
OMVs naturally mimic pathogens, presenting antigens in their native conformation and triggering strong immune responses without the need for external adjuvants. AdJane enhances this innate biology through precise engineering of OMV composition - optimizing its content to strengthen intrinsic adjuvanticity and achieve balanced, durable immune activation. Positioned at the forefront of OMV innovation, this technology combines built-in immunostimulation, safety, and scalable manufacturing.
Building on their platform, AdJane is developing a prophylactic vaccine to combat gonorrhea, a persistent and increasingly drug-resistant infection. The OMV-based vaccine expresses gonococcal antigens while overcoming immune evasion, a key limitation of previous approaches. The program represents a crucial step toward protecting reproductive health worldwide and mitigating one of the fastest-rising antimicrobial threats.
USP:
AdJane’s OMV platform eliminates the dependency on added adjuvants, uniquely balancing intrinsic immunostimulatory properties and precise antigen presentation to achieve potent immune activation with an excellent safety profile, as demonstrated in a Phase 1 study. Backed by decades of research, it represents a next-generation approach to bacterial and viral vaccine design.
AdJane is a Netherlands-based biotech developing next-generation vaccines using its proprietary Outer Membrane Vesicle (OMV) platform. Spun out of Intravacc by FDI, AdJane deploys advanced genetic engineering to precisely tailor both the
Problem Aiming to Solve Antimicrobial resistance (AMR) is one of the most urgent threats to global health. Gonorrhea, a leading AMR pathogen of concern with over 80 million new cases each year, has developed resistance to nearly all antibiotic classes and is now often treated with last-line therapies. Beyond
As a female-led biotech, AdJane brings a distinctive perspective to innovation and global health leadership, championing inclusive, purpose-driven progress across the life sciences.
Seeking:
AdJane welcomes discussions with collaborators and partners interested in our vaccine platform or specific pipeline assets.
PHC Europe, renowned for its evergreen product lines of refrigerators, freezers, and cell culture incubators, is expanding its innovation portfolio with a novel product line supporting research and development in the Cell & Gene Therapy (CGT) field and Cell-based R&D.

The flagship product of PHCbi’s CGT line is the Live Cell Metabolic Analyzer (LiCellMo), an innovation that enables long-term, continuous, manual sampling-free measurement of glucose and lactate in natural cell culture environments. LiCellMo visualizes real-time changes in glycolysis-based cellular metabolism, providing precise information about in vitro metabolism shifts and cellular statuses.
At the heart of LiCellMo is PHCbi’s proprietary sensor technology, developed in-house by PHC’s engineering team in Japan. This electrochemical sensor, where glucose dehydrogenase and lactate dehydrogenase are conjugated, detects and records mild currents released during dehydrogenation reactions in the cell culture medium. To date, eight patents covering the sensing technology, its manufacturing, and the LiCellMo system have been issued in various countries, highlighting its uniqueness and originality.
LiCellMo’s real-time measurement of glucose and lactate can sustain up to 10 days without

manual sampling, creating a labor-free working environment for researchers and scientists, thereby increasing R&D efficiency. Measurements are taken once per minute, with data smoothed and output every 15 minutes. Additionally, the metabolic rate based on glucose and lactate is calculated automatically. Furthermore, the LiCellMo system is housed in a standard CO2 incubator, ensuring cells remain in their natural growth environment and can be used for further experiments, evaluations, or cell expansion after metabolic data collection.
LiCellMo is a versatile tool for monitoring:
1. Shifts in metabolism pathways (glycolysis VS mitochondrial respiration)
2. Long-term changes in cellular statuses
3. Cell differentiation and activation
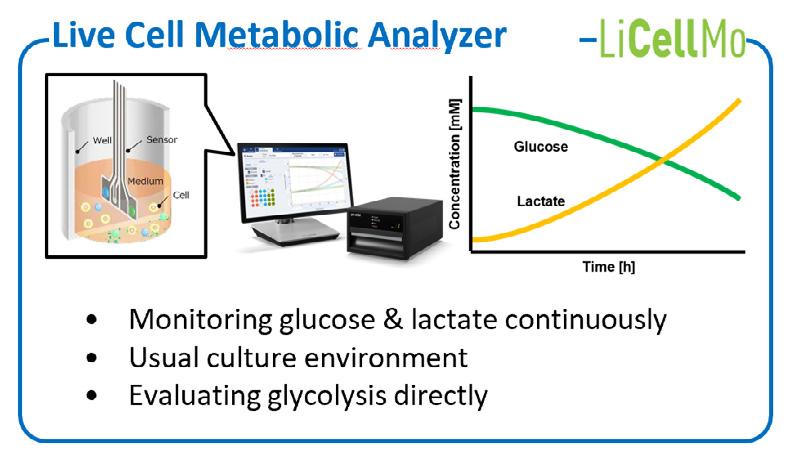
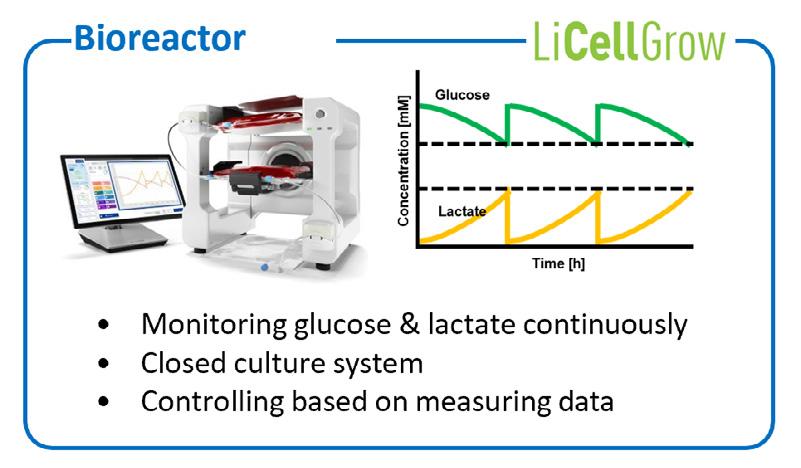
Its applications span various fields, including but not limited to metabolism-related research (e.g., diabetes, liver, cardiovascular), oncology and tumor microenvironment studies, immunology, primary cell and stem cell cultivation and differentiation, cell culture medium optimization and testing, process development (e.g., upscaling to bioreactors,
pharmaceutical component screening, and mode of action determination), and sports medicine.
Building on this, PHCbi’s engineering team has developed LiCellGrow, an automatic bioreactor controlled via the real-time glucose and lactate measurements mediated by the same sensing technology as LiCellMo. LiCellGrow offers various approaches to control the bioreactor system, such as via the threshold value or the range value of the glucose and lactate measurement, and when necessary replacing the old cell culture medium with the new one. It also allows users to monitor cell conditions based on four critical elements: glucose, lactate, pH, and oxygen concentration.
PHCbi’s new CGT product line, grounded in proprietary technology, is poised to provide biomedical scientists with a paradigm-shifting solution for monitoring cell statuses and controlling cell culture growth. While LiCellMo is currently available, LiCellGrow is scheduled for launch in October 2025. We look forward to collaborating with academic and industry partners dedicated to biomedical research and development, with LiCellMo and LiCellGrow efficiently supporting your efforts to advance innovation.
Connect with us for an in-depth discussion and a demo on location:
Ashley Shih, Application Specialist, CGT department (ashley.shih@eu.phchd.com)
https://www.phchd.com/eu/biomedical/ cell-analysis/live-cell-metabolic-analyzer/ licellmo


Join TOPX Network — a platform for ambitious women in the life sciences and health sectors who are ready to lead, innovate and connect. Our mission is to support your personal growth and empower you to take on leadership, management or entrepreneurial roles.
Grow. Connect. Shine.
» Expand your network with peers and senior leaders.
» Engage in trusted peer groups and events.
» Elevate your visibility through speaking opportunities and features.
What You’ll Experience
» TOPX Meetings & Expert Sessions — both online and in-person — designed to inspire, inform, and connect.
» Annual TOPX Summit — our marquee event. Two full days of keynotes, panels, workshops, coaching, networking, speeddates, and more.
» Exclusive Opportunities — member-only benefits including exclusive discounts on Hyphen Projects events, and coaching programmes.
Become a Member Today and visit www.hyphenprojects.nl/topx
Scan the QR code & start your journey.


Empowering ambitious women to rise to the top in Life Sciences!
Be Part of a Circle of Trust
What you get:
» Networking & collaboration: Join a trusted group with shared goals.

TOPX Inner Circles (TIC) — a tailored initiative where a small group of like-minded women from the life sciences sector meet regularly in a trusted peer-group environment.
» Support & encouragement: Connect with peers who understand your unique challenges.
» Learning & development: Share insights, uncover blind spots, and grow together.


Deadline for registration: 5 December 2025
TIC participation is free for Full Members of TOPX Network. Groups start annuallynext round kicking o in January 2026.
Apply to join a TIC today and visit www.hyphenprojects.nl/topx/topx-inner-circles
Make Your Career Tick with a TIC!
















Cantoni Therapeutics secures financing to advance novel non-incretin obesity drug into IND enabling phase
Cantoni Therapeutics B.V. (‘Cantoni’), a biotech company spun out from Leiden University, developing best-in-class small molecule inhibitors of nicotinamide N-methyltransferase (NNMT), a promising target for cardiometabolic diseases, announced the closing of additional financing to advance its lead asset into IND enabling phase for obesity and its comorbidities. In preclinical studies, Cantoni’s NNMT inhibitors have demonstrated an ability to reduce fat mass, without negatively impacting lean mass, and improve metabolic health through increased energy expenditure.
Furthermore, Cantoni was announced as winner of a Novo Nordisk validatioNN grant supporting translational research work on the mode of action of NNMT inhibition.
CEO Annegret Van der Aa said:
“This combined equity and loan financing, and the research grant from Novo Nordisk will help significantly as we progress towards our

ultimate goal to validate NNMT as a novel target for obesity and its comorbidities in the clinic. This financial support is a strong validation of our approach, enabling us to start IND enabling studies.”
Cantoni Therapeutics B.V. is pioneering the discovery and development of small molecule bisubstrate inhibitors of NNMT, a metabolic enzyme highly expressed in metabolic tissues, including the liver, adipose tissue and muscle. NNMT is an attractive drug target for cardiometabolic disease where elevated NNMT levels are correlated with obesity, type 2 diabetes and cardiovascular diseases. In diet-induced obese mice, Cantoni’s NNMT inhibitors induce a reduction in fat mass, without negatively impacting lean mass, and improved metabolic health through increased energy expenditure rather than reduced energy intake. Furthermore, targeting the metabolic enzyme also brings opportunities in other indications; a role for NNMT has been described in the development of chronic kidney disease as well as in cancer progression.
The Invest International activities span five sectors of which healthcare is one. Life Sciences in the sense of medicine development and specialized activities within that segment are excluded from our activities. Within healthcare the range of activities is broad with a bias towards reducing pressure on the healthcare system through prevention and shortening time in the care facilities. With the increased value creation through data insights, we also see more focus on data availability and processing, especially in low to medium income countries.
The mandate of Invest International is closely aligned with the Dutch ministry of Foreign Trade and Development and for healthcare by extension with the international health strategy as defined by that ministry in cooperation with the Dutch ministry of health. Health system strengthening and the interaction between public health and climate change feature frequently as a result. Examples of companies supported can be found on the disclosure section of the Invest International website.
Invest International opened its doors in 2021 when existing mandates from FMO, the Dutch DFI, and the Netherlands Enterprise Agency were combined to offer Dutch companies a single offering for their international funding needs. The organization therefore acts as the Dutch National Promotional Institute, helping Dutch companies move or expand abroad. The Dutch Ministry of Finance is 51% shareholder with FMO holding the remaining. The financing activities of Invest International are additional to the market, addressing a market failure, and are provided within the state aid framework. We generally start at TRL 7, frequently following up from where the regional development corporations and Invest NL joined in an earlier stage.
For more info visit: www.investinternational.nl

HAN BioCentre bestaat 20 jaar. Het Centre of Expertise viert dit jubileum met een symposium op dinsdag 27 januari 2026 in Nijmegen.
Al 2 decennia werkt HAN BioCentre aan een betere toekomst, waarbij slim gebruik wordt gemaakt van de levende natuur. Dat doet ze samen met studenten, andere kennisinstellingen en met het bedrijfsleven. In die 20 jaar zijn veel mooie en bijzondere resultaten behaald. Een verkoopbaar en heerlijk product maken van afgekeurde tomaten bijvoorbeeld. Of samen met BIOMA Lab biobased leer maken van het restproduct van kombucha. En ook op het gebied van drug discovery timmert het biocentre aan de weg. Zo is samen met partner TropIQ succesvol getest op nieuwe antimuggenstoffen.
In 20 jaar is het onderzoekscentrum gegroeid. Qua aantallen medewerkers, maar zeker ook qua werkterrein. De duurzame wereld stond altijd voorop en dat is niet veranderd. Maar er zijn wel onderzoeksthema’s bijgekomen. HAN BioCentre werkt nu aan biobased research, data science|bio-informatie en drug discovery. Vanuit diverse locaties middenin de praktijk. In Arnhem is er een lab in HAN@Connectr, op het Cleantech Park, waar zich het centrum bevindt van de biobased materialen. Oss is de belangrijkste plek voor medicijnontwikkeling, waar HAN BioCentre een laboratorium heeft tussen de start-ups en gevestigde farmaceutische bedrijven.
De drie onderzoeksthema’s staan centraal in het symposium eind januari. Zij krijgen een plek tijdens diverse parallelsessies en paneldiscussies. Meteoroloog en ondernemer Gerrit Hiemstra vormt als keynote speaker de link ertussen. Het symposium is voor iedereen die interesse heeft in hoe HAN BioCentre door middel van de inzet van lab-expertises bijdraagt aan een gezonde en duurzame samenleving.
20-jarig jubileum HAN BioCentre Dinsdag 27 januari van 12.30 – 18 uur Laan van Scheut 2 in Nijmegen
Aanmelden kan via de site https://www.han.nl/evenementen/2026/01/ symposium-biocentre-20-jaar/


Name event: TOPX Summit
Took place on: 13-14 November 2025
Theme: Leading Together in Life Sciences & Health

• Dare to Ask (Hello, Dopamine!)
• Dare to Challenge (Serotonin!)
• Dare to Standout (Oxytocin!)

I left feeling energized, strategically focused, and armed with fresh perspectives on leadership and career growth, especially for empowering women to rise to the top in the Life Sciences & Health sector. That was the clear pulse of the day!
The energy was phenomenal, and the lessons were GOLD, perfectly aligning with TOPX's mission to support and encourage women's development in leadership roles.
Key Learnings for Career & Leadership:
• Marjoleine van der Zwan’s Journey: a powerful reminder to "Keep feeding your soul - just keep going forward!" and to always "Stay true to your talents." Essential advice for women navigating competitive leadership tracks—if a path isn't leveraging your core strengths, have the courage to shift! Also, loving the "Every setback has a hidden gift" mindset for resilience.
• The Power of Daring: Sangbreeta Moitra nailed it with the call to action for high-impact growth, particularly relevant to women stepping into the spotlight:
• Future-Proofing & Self-Advocacy: Loved the focus on embracing AI as a superpower (Marcelina Dutkiewicz ) and the crucial message from Barbara Rogoski on self-motivation and self-care. It’s vital for women to watch those subtle self-put-downs—it's not bragging to speak about your achievements—it's necessary!
• Career Navigation & Development: The panel discussions, including the fantastic session moderated by Anne Vogel, and the closing plenary moderated by Laura Duran, offered invaluable blueprints for navigating career growth.
• Marketing Your Career: Practical strategies from Karen Rensma on taking charge of your professional narrative—a critical skill for visibility and promotion.
Connections & Community
The best part? Making incredible new connections and having a thrilling reunion with a former colleague and friend!
Graded (on a scale of 1 to 10): 8/10
Thank you to the TOPX team for a brilliantly organized and genuinely inspiring event, focused squarely on empowering ambitious women to rise to the top!
Already looking forward to 2026!
Generative AI (GenAI) superusers notice 40% higher quality of their work, while 91% of super users say they are more happy and motivated at work. AI broadly has the power to reshape how science and operations are run.
If we were to consider the AI transformation through data, the picture could be as follows:
It starts with excitement:
● 85% of top pharma execs call AI an “immediate priority” and are scaling their budgets accordingly
Includes worries:
● 60% of leaders worry their organization’s leadership lacks a plan and vision to implement AI; while 45% of white collar workers are worried about being replaced by AI
And currently is a challenge:
● 95% of AI pilots fail; and only 14% of employees become superusers when GenAI licences are deployed (without job-specific training)
Why? Answering that requires understanding the story of AI in many Life Sciences companies, which followed a path similar to this:
Over the last decades, some organizations invested in AI/ machine learning - but mostly in niche applications.
In 2022, OpenAI lowered barriers to AI by releasing ChatGPT 3.5 to the public. For those who learned to use this GenAI effectively, it democratised knowledge, experience and it made them much more efficient.
It changed how we work forever. Fast adopters were thrilled. Yet many senior leaders, some treating GenAI as a hype, some as “interesting opportunity” - didn’t personally upskill. Compelling visions about the future of work in organisations were often not shared.

Due to overwhelming media coverage of AI but little leadership, employees began to fear for the future of their jobs.
GenAI adoption and change management (if any) in most companies were led by enthusiasts and tech functions. Adoption barriers were underestimated, Generative AI (like ChatGPT/Copilot…) was released to employees as a “yet another” useful tool. Without proper change management, communication, leadership and job-specific / tailored training.
Resulting in too few super users to spark real momentum.
In 2025, larger implementations often began as grassroots pilots driven by employee excitement and not the strategic vision, combined with the challenges above - ended up as pilots failing or failing to scale.*
At the same time there are pharma and biotech companies that are already harvesting return on investment from their AI initiatives. How to do it well then?
EfficiencyTactics is a Danish advisory and upskilling company focused on people centric AI adoption, partnering with top pharma and biotech companies to enable successful AI transformation. Its CEO and Founder Marcelina Dutkiewicz, advises stepping away from thinking about AI as an ‘implementation’ and treating it as a major transformation.
Marcelina recommends considering AI as a deep and profound change in the ways of working for individuals and ways of operating for companies, highlights the importance of senior leadership upskilling to enable them to lead by example and reimagine the strategy with AI capabilities in mind. BIO: Marcelina holds a MSc in Mathematical Modelling, over a decade of experience from R&D, commercial and innovation roles from both large pharma - Novo Nordisk, Leo Pharma - and biotech - Genmab - where she served as Director of AI and Digital R&D Strategic Initiatives 2023-2025. Now she shares her knowledge and experience supporting the life sciences sector to strategically implement AI.
*Adapted from book: “It starts with vision - A guide to navigating AI transformation across people, leadership, and life sciences companies” by Marcelina Dutkiewicz, published Nov2025; sales via: www.efficiencytactics.com
For more information: info@efficiencytactics.com
Connect with Marcelina:


Define Ventures “Inside the C-Suite - Pharma Leaders Share Their Vision for AI.” Jul, 2025
Data source - BCG, Jan 2025: “GenAI in The Nordics”
Source: MIT “State of AI in Business 2025 Report”; Microsoft “Work Trend Index”, 2024

On a Monday morning at a mid-sized biotech, the weekly program review felt like a ritual of exhaustion. Slide decks were heavy, calendars were full, and the team left meetings with more to do but less clarity about what mattered. Of twenty-two initiatives on the board, seven hadn’t moved in months. They were busy, but not forward. That pattern—activity without momentum—is what I call the Progress Trap. If you add too many activities, the result will be a diminishing progress overall
The surprising truth is that progress often comes from subtraction, not addition. Two simple, repeatable routines changed the dynamic for that team: Offload and Talent Mapping & Activating. Both are low-friction, high-impact habits that free capacity and focus without asking people to work harder.
1) OFFLOAD, to start stopping faster. Three key areas where you can free-up capacity and reduce significant waste of scarce human resources.
a. The number of running projects & initiatives in the organizations. First step is to check which initiatives are hardly making any progress. Often a clear sign that it isn’t perceived as a mission critical initiative or team members lost trust that it will be finished anyway. A recent finding in a biotech company was that 7 out of 22 initiatives were identified as unfinished business, without any drive to move it forward. By applying the two-way-door decision (meaning you can reverse the decision overnight) they were stopped.
b Ineffective collaboration. The usual suspects are there for sure, like meetings with no clear objective, too many participants and open endings, to name a few. And many communication channels, often with personal preferences by individuals, leading to a lot of miscommunication and waste of time to search for information, when a clear agreement is missing on what channel should be used for what type of communication. But what I see the most in Pharma and biotech companies is the excessive amount of data and information shared in cross functional meetings. And if there hasn’t been agreed on ‘how do we collaborate and what’s the scope (clarity needed) ‘, the result of these dozens of slides packed with information, create a freezone, where different people of different functions just pick their own preferences. Resulting in a non-stop ask for priority setting and reprioritizing. The following
principles will create an engaging and productive collaboration.
→ Shared measurable goals
→ Clear roles, responsibilities and decision making
→ How do we work together (meetings/messages/ filing etc.)
→ Encourage safety and trust by asking feedback and sharing what went wrong and key learning for the team.
→ Share success (steps)
c. Needless complexity. Examples of needless complexity are people trying to create an approach for something or a solution for a challenge without first checking if there are colleagues working in another country if they already dealt with it. Simply first asking yourself, who could have faced a similar situation and can help me setting up the same approach instead of coming up with a different approach (leading to different approaches for the same kind of challenge.
But most needless complexity has a very simple reason, if you put enough smart people in the room, anything you discuss will become more (and often not needed) complex. People by nature have the tendency to add things much more than to subtract.
So, start asking with any solution or approach, first what can we subtract instead of what should be added!
2) Talent mapping & matching
Teams are often unaware of the full range of skills, knowledge, and abilities within their own ranks. As a result, valuable talent goes untapped—often at the very moment it’s most needed, I discovered a simple, three-step exercise that include:
• Self-Reflection: Team members list their strengths, acknowledging talents they might take for granted.
• Team Input: Colleagues provide additional perspectives on each other’s strengths, fostering trust and appreciation.
• Matching Strengths to Challenges: Assigning tasks and challenging based on talents and strengths rather than job titles, dynamically redefining roles.
To unlock your team’s hidden potential, schedule a dedicated meeting to surface the full capabilities
on your team. Give each person 10 minutes to reflect and write down their talents and skills. Then, ask everyone to share their list aloud. As they do, encourage colleagues to add strengths they’ve observed that weren’t mentioned.
This straightforward exercise is consistently eye-opening. It can reveal hidden strengths, help This exercise builds trust within the team and fuels collaboration. One remarkable, unexpected outcome I discovered was:
The Quietest Voices (the introverted and modest team members) often receive the most additions from their colleagues, which boosts their appreciation and increase their self-confidence they didn’t recognize in themselves (or simply took for granted).
Q1 2026 Published


From farm to fermentation:
By Yilmaz Biter

For almost a century, heparin has been a trusted protector against blood clots in hospitals and dialysis centres around the world. However, this life-saving drug is not without its issues. Its production relies on the intestinal mucosa of pigs, creating a highly vulnerable supply chain. When African swine fever swept through China’s pig farms, hospitals worldwide began to feel the effect of the heparin shortages. The paradox became impossible to ignore: one of our most essential drugs was still tied to a twentiethcentury extraction process and was therefore vulnerable to any disease that could affect pigs.
Instead of accepting the traditional extraction approach, Aisling Foley, together with her cofounder Ilaria Poledri and the ExCulture team have progressed a microbial fermentation platform that uses mixed bacterial cultures that originates from the group of Mark van Loosdrecht and Yuemei Lin at TU Delft, to produce sulfated glycosaminoglycans,

the macro-class that anticoagulant heparin comes from.
Aisling grew up on a small farm in south-eastern Ireland, where she learned early on about the intersection of agriculture and science which also sparked her interest in biology and sustainability. ‘’I grew up on a small farm in the southeast of Ireland. My parents work outside of the farm too, but it’s something my dad has always worked on in his spare time. We mainly have cattle for beef, but we also have a vegetable garden for the house, and I think that’s where my interest in science started. I loved growing vegetables and flowers and helping my dad and sister look after the animals.’’
She isn’t the first person in her family to start her own company. She says it could be that it’s in her blood to be an entrepreneur. ‘’My dad came across an archive of interviews from the Irish national radio
service, one of which was with my grandfather in 1966. He was a carpenter and wheelwright, and in the interview, my grandfather talked a bit about working for himself, how when you must build things up bit by bit from the foundation it’s difficult, you don’t have any spare time when you’re your own boss.’’ Her uncle on her mother’s side founded his own company in the computer programming space in the 1990s and even further back one of her maternal granduncles opened a motor garage and filling station in the 1930s.
A personal motivation
Her original ambition was to work in biomedical sciences, driven by a desire to combat the illnesses that had affected her loved ones. ‘’There’s a history of various diseases and conditions in my family, including the autoimmune disease lupus. Unfortunately, two of my aunts had that. Another aunt had cancer, which she overcame. However, she later died from sepsis. That’s what initially sparked
my interest in setting up ExCulture. Like many people who want to study science, I went to university thinking I would focus on oncology. But it turns out that I’m not a natural organic chemist. I just can’t intuitively follow those reactions. The Environmental and Sustainable Chemistry specialisation was relatively new in Dublin at the time, and I found that very compelling.’’
Driven by a desire for international experience, she pursued a semester-long international exchange at the University of Virginia. ‘’The chemistry studies were extremely tough. I really enjoyed it and found the environment very motivating, but they use a bell curve grading system, which made me feel under a lot of pressure to work really hard. I did a couple of projects there, but the main one was working in an organometallic chemistry group. We were developing ligands for palladium catalysts.’’
Upon her return, despite her growing certainty that her future lay outside the pharmaceutical industry, she adopted a ‘try before you decide’ approach. She took an internship at Merck & Sharpe and Dohme. ‘’Even though I was quite confident at the time that I didn’t want to be in the pharmaceutical industry in the future I always think: don’t make a decision without trying something first. The most interesting thing for me was learning how to scale up a lab reaction for manufacturing, as well as learning just how many different people from engineers to finance and quality personnel must give their input before the process could move to the manufacturing site.’’
The heparin crisis
TU Delft’s technology transfer office (Delft Enterprises) was seeking a founder to commercialise a patent-pending bioprocess that had emerged from decades of wastewater treatment research. This technology uses mixed cultures of microorganisms to naturally produce sulfated biopolymers that are remarkably similar to heparin. ‘’I had also learned about the licensing and ventures group while I was in Virginia. I think I was kind of disillusioned at times during my studies, because while a lot of the
innovations happening at the university are super cool and innovative, they don’t necessarily make it into the real world. I was connected via Female Ventures with someone working at Delft Enterprises. I started following them on LinkedIn and saw that they were looking for a founder to spin out a company based on a patent-pending bioprocess for environmental wastewater treatment.’’
This project had been originally worked on by Winnifred Noorlander, who, after coming across the patent application of Professor’s Mark van Loosdrecht and Yuemei Lin et al, had developed a proof of concept for an antiseptic. Unfortunately, Noorlander had to stop working on the project due to a severe injury, but she is an on-going and inspirational supporter of Aisling and Ilaria, founders of ExCulture. ‘’From our experience of doing a marketing project together during our business studies courses we knew we could work well together, and I felt that Ilaria’s molecular genetics and biotechnology background was relevant to the microbial bioprocess and complementary to my chemistry background so I approached her one month after starting the project myself and asked if she would be my co-founder. She said yes on the spot and we’ve been working together for over three years now.’’
‘’Our aim is to produce a safer heparin via a more sustainable production method, and although we still have quite a lot of work ahead of us, we’re optimistic that we can achieve this,’’ she continues. ‘’Ideally, we would like to have onsite or onshore production to ensure regional sovereignty of supply. If we think back to the pandemic time, that was a major challenge from a medicine perspective. We feel that setting up a bioreactor in the Netherlands, Ireland or the USA, for example, should not be a problem, and that this would be more localised to the major markets.’’
Lessons from a pioneer
As a Termeer Fellow, Aisling draws inspiration from the legendary founder of Genzyme – who transformed the treatment of rare diseases – the late Henri Termeer. ‘’When you hear about someone
like Henri Termeer, it’s mainly about Genzyme, the multi-billion-dollar company. But when you read this book (Conscience and Courage: How Visionary CEO Henri Termeer Built a Biotech Giant and Pioneered the Rare Disease Industry), you get a true insight into the journey they and Henri Termeer followed from the early days.’’
What does the Termeer Fellowship mean to you? ‘’Everyone involved in the Henri Termeer programme has a personal story or anecdote about how he helped, inspired or mentored them. The people who formed and support the Termeer Institute really have this pay-it-forward mentality and are always willing to help. There’s just this ethos of helping each other. It’s not about knocking people down. When you think about business, you sometimes think that everybody’s in competition with each other, but they’re really not. I can’t speak for any other industry, but I really like this aspect of the biotech industry. We’re very lucky to have such a collaborative, supportive ecosystem in the Netherlands.’’
Aisling Foley


















Join us at the Leading Conference for Key Players in Life Sciences & Health.


Innovation for Health (I4H) is the Netherlands’ premier platform for health and life sciences innovation. On March 25-26, 2026, I4H and the Global Investor Forum will bring together over 700 high-level participants, 70+ expert speakers, and 50+ exhibitors.

Join these key leaders from the ecosystem to bridge the gap from idea to innovation—accelerating the development, funding, and adoption of impactful healthcare solutions.
There is an opportunity for everyone:
» Sponsors – Elevate your brand among industry leaders.
» Exhibitors – Showcase solutions and innovations to decision-makers.
» Start-ups – Connect, pitch, and grow with investors and partners.
» Investors – Discover high-potential ventures and breakthrough technologies.
» Delegations – Showcase your region’s expertise and ambitions .

» Attendees – Learn, network, and explore cutting-edge innovations.

Meet the right people and achieve your goals with guaranteed one-to-one meetings, easily arranged via PartneringONE®.






