

Tirzepatide(Zepbound),themostpopularweightlosspeptide in2024even2025!
-ByPhcoker
Introduction
Tirzepatidepeptideisaglucose-dependentinsulinotropicpolypeptide(GIP)receptorandgluc agon-likepeptide-1(GLP-1)dualreceptoragonistThedrugiscurrentlyapprovedbytheF DAunderthebrandnameofMounjaroforthetreatmentoftype2diabetes.AccordingTirz epatideresearchresult,thecurrentclinicaltrialresultsshowthattheparticipantsintheexp erimentshowedasubstantialweightloss.Participantswhousedthehighestdoseof15mgl ostanaverageof15.7%oftheirweight,whichisamajormilestoneinthehistoryofweig htlossdrugs.
WhatIsObesity?HowToTellIfYouAreObese?
BeforetohaveadetailedreviewofTirzepatide,theimportantinformationweneedtoknow isobesity.Obesityisaseriousepidemicaffectinghealthworldwide.Factorssuchasgenetic sandenvironmentcancontributetoitsdevelopment.Butthegoodnewsis,it'streatable.S ohowdowejudgewhetherweareobese?
Generally,healthcareproviderswilluseyourBMItodiagnoseobesityBMIiscalculatedb ydividingweight(kg)byheight(m)squared[BMI=weight(kg)/height2(m)]
Overweight:ABMIbetween25.0and29.9meansyouareoverweight.Thisisahealthissu e.Beingoverweightputsastrainonyourbodyandcanleadtootherhealthproblems,suc
PeptideGMPManufacturers www.phcoker.com


Obesity:ObesitymeansthatyourweightisinanunhealthystateYourBMIbetween300 and399meansthatyouareobeseThisiswhenyouneedtocontactyourhealthcarepro viderpromptlytounderstandandtreatobesity
Severeobesity:WhenyourBMIisgreaterthan40.0,youareinthestageofsevereobesity, whichisverylikelytocauseyoutodevelopotherhealthproblems.Whenyourealizeyou areseverelyobese,youneedtotalktoyourhealthcareprovideraboutweightlossmanag ementoptions
WhatIsTirzepatidePowder?
Tirzepatideisaweightlossdruginjectedonceaweekforweightmanagement.Thedrug,developed byEliLillyandCompany,iscurrentlyapprovedbytheFDAtotreattype2diabetes.Tirzepatide powderistheactiveingredientinthepopulardiabetesdrugMounjaroandweightlossdrug ZepboundIfyouareconsideringtobuyTirzepatideforweightlossmanagement,itisimportantto findareliabletirzepatidesupplierItcanbeachallengetochoosethemostsuitabletirzepatide supplierfromthemanypeptidemanufacturersAsaprofessionalTirzepatideGMPmanufacturer, Phcokercannotonlyprovidehigh-qualityTirzepatidelyophilizedpowderinvials,butalsoraw powder.
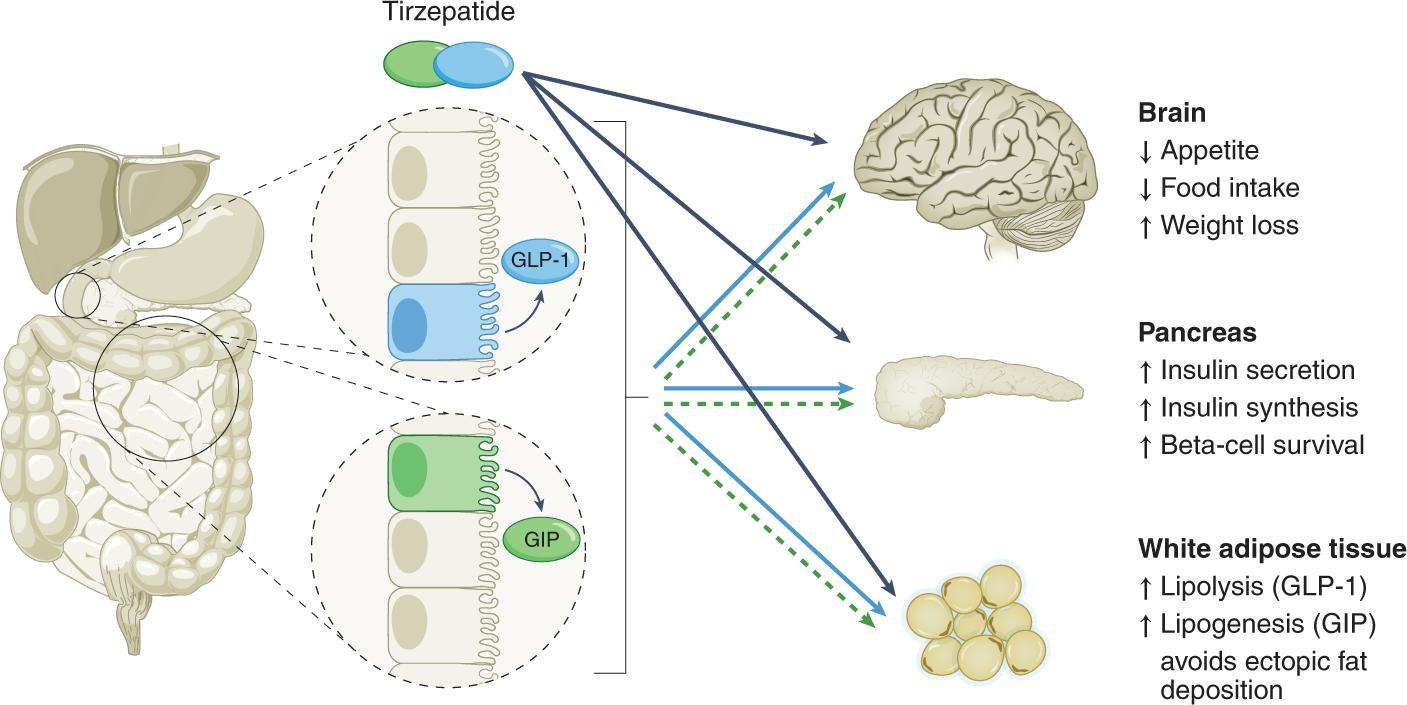
AsadualreceptoragonistofGLP-1andGIP,Tirzepatidenotonlystimulatesinsulinsecretionand release,butalsohasacertaininhibitoryeffectonglucagonThroughthedualeffectsofGLP-1and GIP,Tirzepatidecaneffectivelysuppressappetite,reducefoodintake,andprolongthefeelingof satiety,therebypromotingweightloss
ThedualeffectsofTirzepatidecannotonlyeffectivelyreduceweight,butthisinnovativemethod makesfulluseoftheadvantagesofthesetwohormonestosolvemanyproblemssuchas
PeptideGMPManufacturers www.phcoker.com hasobesity.


TheHistoryofTirzepatidePeptide

TheabovearethekeyeventsinthedevelopmentofTirzepatide.Currently,Tirzepatidepeptidehas beenproventobeasafeandeffectivedrugforweightlossandtype2diabetestreatment,andhas alsoshownmanybenefitsonmetabolism.Whenusingit,inadditiontoconsultingaprofessional healthcareproviderbasedonyourspecificsituationandneeds,itisalsoimportanttofindaregular andreliablepeptidemanufacturertopurchaseTirzepatidepowder
TirzepatideMechanismofAction
IfwanttohowisTirzepatidehelpweightmanagement,herelet’shavealookofthemechanismof actionofTirzepatide.Asasyntheticpeptide,Tirzepatideworksmainlybytargetingtworeceptor agonists,GLP-1andGIP.
ResearchstudieshaveshownthatTirzepatidehasgreateraffinityforGIPreceptorsthanforGLP-1 receptorsAndcomparedtoasingleGLP-1receptoragonist,thedualreceptoragonisteffectof
PeptideGMPManufacturers www.phcoker.com metabolism,andalsomakesitaneffectivetreatmentfortype2diabetes.apromisingtreatment option


Tirzepatidepowderhasbeenshowntoreducehyperglycemiatoagreaterextentandreducethe user'sappetitetoagreaterextent
Accordingtosignalingreports,TirzepatidewillmimictheactionofnaturalGIPontheGIPhandle; however,forGLP-1receptors,TirzepatideismorelikelytofavortheproductionofcAMP,rather thanrecruitingβ-arrestin,whichbindstosugar.,amessengerrelatedtotheregulationofglycogen andlipidmetabolism. AlthoughtheexactmechanismofactionofTirzepatidehasnotbeenfullyelucidated,current researchshowsthatTirzepatide'sdualreceptoragonisteffectonGLP-1andGIPincreasesinsulin secretionandcaneffectivelytreatobesityandtype2diabetes
WhyTirzepatideIsSoPopular?BenefitsOfTirzepatide
Asabiologicalpeptidedrug,Tirzepatideispopularinthemarketbecauseofthebenefitsitbringsto peopleItisawell-knowndrugfortreatingdiabetesandaneffectiveweightlossdrugHerearethe primarybenefitsofTirzepatide:
PeptideGMPManufacturers www.phcoker.com


PromoteweightlossStudieshaveshownthatTirzepatideactivateshumanGIPandGLP-1receptors GIPandGLP-1receptorsaretworeceptorscrucialtoregulatingappetiteinthehumanbrain Tirzepatidehasbeenshowntoreducefoodintake,suppressappetiteandregulatefatutilization
Numerousclinicalstudieshaveshownthatpatientstakingtirzepatidelost157%(344poundsor 15.6kilograms)ofbodyweight,makingitapotentialoptionforthoseseekingeffectiveweight management.
Increasessatiety.Tirzepatideslowsthepassageoffoodthroughthebody,reducesthedesiretoeat, andenhancesthebody'sfeelingoffullness,makingiteasiertosticktoahealthydietandmaintain weightloss
Controlandstabilizebloodsugarlevels.Type2diabetesisachronicprogressivediseaseandthe mostcommonformofdiabetes.Thisdiseasecausesthebody'sowninabilitytoreleaseinsulin, resultinginhighlevelsofglucoseintheblood.Tirzepatidecanregulatethereleaseanduseof insulinbyregulatingGLP-1andGIP,therebykeepingglycatedhemoglobin(HbA1c)atahealthy level
Treattype2diabetes.TirzepatidepeptidehasbeenapprovedbytheFDAforthetreatmentoftype 2diabetesandcaneffectivelyhelppatientscontrolbloodsugarRegulatesbloodsugarlevelsmainly byincreasingthesecretionofinsulin.
Treatobesity-relateddiseases.Obesitycanleadtophysicaldiseasessuchashighbloodpressure, hyperglycemia,hyperlipidemia,andevenheartdiseaseTirzepatidecaneffectivelyreducethe occurrenceanddevelopmentofthesediseaseswhileprotectingthecardiovascularsystemby promotingweightloss
Convenient.Unlikeotherdrugsthatmustbetakeneveryday,Tirzepatideonlyneedstobeinjected onceaweek,whichgreatlyhelpspatientssavealotoftimeintheirdailylivesandmakestakingthe
PeptideGMPManufacturers
www.phcoker.com

drugmoreconvenient.

Flexibledosing.TirzepatidecanbeadministeredflexiblyItcanbeusedaloneorincombination withotherdiabetesmedicationsThisflexibledosingmethodallowspatientstomodifythedosage accordingtotheirneeds,therebyachievingbettertherapeuticeffects
Treatobesewithtype1diabetes.ThelatestresearchshowsthatTirzepatidemayhelpobesetype 1diabetesalso.ResearchersreportedthatTirzepatideledtoanaverageweightlossof18.5%(>46 pounds)inpatientswithtype1diabetesoveroneyearandimprovedglycemiccontrol
Inaddition,researcherscontinuetostudythepotentialbenefitsofTirzepatidepowderPotential benefitsofTirzepatideareasfollows:
Protectheart
Reduceinflammation
Treatalcoholaddiction
ThesestudiesareongoingtoexploreTirzepatide'sbroaderbenefitsbeyondobesityandtype2 diabetesThepowerfuleffectsshownbyTirzepatidepeptidehavemadeitwidelypopularinthe marketIfyouareinterestedinTirzepatidepowder,makesuretopurchaseitfromareliablepeptide manufacturersuchasPhcoker.com.
Chronickidneydiseaseisacommoncomplicationoftype2diabetesandcanleadtokidneyfailure inseverecases.Resultsofameta-studyshowthatanydoseofTirzepatidereducesproteinurialevels inpatientswithtype2diabeteswithoutaffectingkidneyfunction.
PeptideGMPManufacturers www.phcoker.com
TirzepatideEmergesasNewStarinKidneyProtectioninType2Dia betesTreatment


Theexperimentwillconductanobservationalstudyonpatientswithtype2diabeteswhoreceived TirzepatideandacontrolgroupwhoreceivedplaceboandSemaglutideItmainlymeasuredthe urinaryalbumin-creatinineratio(UACR)andglomerularfiltrationratebetweentheTirzepatide experimentalgroupandthecontrolgroup(eGFR)forcomparisonTheresultsofthestudyshowed thatcomparedwiththecontrolgroup,theUACRintheTirzepatidegroupwasreducedby approximately27%,buthadnosignificanteffectoneGFRlevels.ThisshowsthatTirzepatidehas moreobviousbenefitsforkidneyprotection.
Allinall,Tirzepatide'sdualagonisticeffectonGIPandGLP-1cannotonlypromoteweightloss andcontrolbloodsugar,butalsoprotectthekidneys,whichisveryimportantforthetreatmentand careoftype2diabetesInordertomorefullyunderstandandconfirmtheprotectiveeffectof Tirzepatideonthekidneys,alargenumberofclinicaltrialsarestillneeded.Ifyouarearesearcher orinterestedinthis,makesuretopurchasefromaspecialtypharmaceuticalcompanyorpeptide powdermanufacturer(suchasPhcoker).
PeptideGMPManufacturers www.phcoker.com


ReferencedCitations:
[1]"Australianprescriptionmedicinedecisionsummaries:Mounjaro"TherapeuticGoods AdministrationArchivedfromtheoriginalon5February2023Retrieved28February2023
[2]"Mounjarotirzepatide15mg/05mLsolutionforinjectionpre-filledpen(379334)"Therapeutic GoodsAdministration.Archivedfromtheoriginalon3January2023.Retrieved28February2023.
[3]"PublicSummary:Mounjarotirzepatide15mg/0.5mLsolutionforinjectionpre-filledpen". TherapeuticGoodsAdministration.Retrieved28February2023
[4]"Notice:MultipleAdditionstothePrescriptionDrugList(PDL)[2023-03-08]"HealthCanada 8March2023Archivedfromtheoriginalon22March2023Retrieved21March2023
[5]"SummaryBasisofDecision-Mounjaro"HealthCanada17March2023Archivedfromthe originalon25April2023Retrieved24April2023
[6]ThomasMK,NikooienejadA,BrayR,CuiX,WilsonJ,DuffinK,etal.(January2021)."Dual GIPandGLP-1ReceptorAgonistTirzepatideImprovesBeta-cellFunctionandInsulinSensitivityin Type2Diabetes".TheJournalofClinicalEndocrinologyandMetabolism.106(2):388–396.
doi:10.1210/clinem/dgaa863.PMC7823251.PMID33236115
[7]"FDAApprovesNovel,Dual-TargetedTreatmentforType2Diabetes"USFoodandDrug Administration(FDA)(Pressrelease)13May2022Archivedfromtheoriginalon13May2022 Retrieved13May2022PublicDomainThisarticleincorporatestextfromthissource,whichisin thepublicdomain
[8]NewDrugTherapyApprovals2022(PDF).U.S.FoodandDrugAdministration(FDA)(Report). January2024.Archivedfromtheoriginalon14January2024.Retrieved14January2024.Public DomainThisarticleincorporatestextfromthissource,whichisinthepublicdomain.
[9]MinT,BainSC(January2021)"TheRoleofTirzepatide,DualGIPandGLP-1Receptor Agonist,intheManagementofType2Diabetes:TheSURPASSClinicalTrials"DiabetesTherapy 12(1):143–157doi:101007/s13300-020-00981-0PMC7843845PMID33325008
[10]KolataG(8November2023)"FDAApprovesNewObesityDrugTirzepatideThatWill CompeteWithWegovy".TheNewYorkTimes.Archivedfromtheoriginalon9November2023. Retrieved9November2023.
PeptideGMPManufacturers www.phcoker.com


[11]FriasJP,NauckMA,VanJ,BensonC,BrayR,CuiX,etal.(June2020)."Efficacyand tolerabilityoftirzepatide,adualglucose-dependentinsulinotropicpeptideandglucagon-like peptide-1receptoragonistinpatientswithtype2diabetes:A12-week,randomized,double-blind, placebo-controlledstudytoevaluatedifferentdos"Diabetes,Obesity&Metabolism22(6): 938–946doi:101111/dom13979PMC7318331PMID31984598
[12]DahlD,OnishiY,NorwoodP,HuhR,BrayR,PatelH,etal.(February2022)."Effectof SubcutaneousTirzepatidevsPlaceboAddedtoTitratedInsulinGlargineonGlycemicControlin PatientsWithType2Diabetes:TheSURPASS-5RandomizedClinicalTrial".JAMA.327(6): 534–545doi:101001/jama20220078PMC8826179PMID35133415
[13]FriasJP,NauckMA,VanJ,KutnerME,CuiX,BensonC,etal(November2018)"Efficacy andsafetyofLY3298176,anoveldualGIPandGLP-1receptoragonist,inpatientswithtype2 diabetes:arandomised,placebo-controlledandactivecomparator-controlledphase2trial"The Lancet.392(10160):2180–2193.doi:10.1016/S0140-6736(18)32260-8.PMID30293770
[14]WillardFS,DourosJD,GabeMB,ShowalterAD,WainscottDB,SuterTM,etal.(September 2020)."TirzepatideisanimbalancedandbiaseddualGIPandGLP-1receptoragonist".JCIInsight. 5(17).doi:10.1172/jci.insight.140532.PMC7526454.PMID32730231
[15]ØstergaardS,PaulssonJF,KofoedJ,ZoselF,OlsenJ,JeppesenCB,etal(October2021) "TheeffectoffattydiacidacylationofhumanPYY3-36onY2receptorpotencyandhalf-lifein minipigs"ScientificReports11(1):21179Bibcode:2021NatSR1121179O doi:101038/s41598-021-00654-3PMC8551270PMID34707178
Authorofthisarticle: Dr.JeanZenggraduatedfromking’scollegelondonFacultyofLifeSciences&Medicine.
ScientificJournalpaperAuthor: 1GiovanniPacini
IndependentResearcher,Padova,Italy
2.KazuhikoKido
PeptideGMPManufacturers
www.phcoker.com


PharmD,BCCP,BCPS,ClinicalAssociateProfessor,DepartmentofClinicalPharmacy,West VirginiaUniversitySchoolofPharmacy
3JessicaWSkelley
PharmD,BCACP,Professor,DepartmentofPharmacyPractice,McWhorterSchoolofPharmacy, SamfordUniversity,Birmingham,AL
4.QiLiu
DepartmentofCardiology,RenminHospitalofWuhanUniversity,Wuhan430060,Hubei,PRChina
5StephenJNichollsMBBS,PhD
VictorianHeartInstitute,MonashUniversity,VIC,Melbourne,Australia
PeptideGMPManufacturers www.phcoker.com








