Influence of Meningioma Molecular Subtyping and Tumour Localization on Treatment Response: A Retrospective Longitudinal
Study
Senior Research Project (TFG)
Author: Angela Mei Seiler Torres 6th Year of Medical School. University of Navarra.
Director: Dr. Bartolomé Bejarano Herruzo. Head of Neurosurgery Department, Clínica Universidad de Navarra (CUN)

INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE
Abstract

Meningioma, the most common primary intracranial tumor, varies widely in clinical outcomes based on histological subtype and location. This retrospective longitudinal study at Clinica Universidad de Navarra (CUN) from 2008 to 2022 examines the relationship between meningioma subtypes, tumor localization, and treatment responses. We evaluated medical records of 122 patients who underwent surgery for meningioma, focusing on WHO subtypes, tumor characteristics, and post-surgical outcomes, including recurrence and subsequent treatments. The cases reveal a female-to-male ratio of approximately 2:1. The predominant tumor subtype was Transitional (mixed) with the majority (90.16%) being Grade 1 tumors. Age distribution showed a median age of 60 years. In terms of proliferation and receptor status, 74.12% of tumors assessed had a positive Ki-67 index, indicating higher proliferative activity. Most tumors also exhibited moderate to high progesterone receptor immunoreactivity. Cross-tabulation analyses highlighted distinct patterns in subtype distribution across different anatomical sites. Notably, transitional (mixed) meningiomas predominantly adhered to the 2:1 female-to-male ratio, while meningothelial and atypical subtypes demonstrated gender distributions nearing a 1:1 ratio and a reversal to 1:2, respectively. Fibrous (fibroblastic) meningiomas showed a stronger female prevalence with a 4:1 ratio. Tumor location analysis indicated a significant representation of fibrous (fibroblastic) meningiomas in the frontal or anterior fossa and midline structures, while meningothelial meningiomas were more common in parietal and middle fossa areas and the skull base. A high percentage of transitional and meningothelial subtypes showed moderate to high progesterone receptor immunoreactivity, contrasting with the more varied levels observed in fibrous subtypes, and were frequently associated to hyperostosis or dural attachments Atypical and anaplastic meningiomas, indicating more aggressive behaviors, exhibit shifts in gender distribution, higher Ki-67% index levels, and higher recurrence and surgical counts than the rest
Key Words: Meningioma, Intracranial tumor, Histological subtype, Tumor localization, WHO subtypes, Recurrence, Ki-67% proliferation index, Progesterone receptor immunoreactivity, Transitional (mixed) meningioma, Meningothelial meningioma, Fibrous (fibroblastic) meningioma, Angiomatous meningioma, Atypical meningioma, Anaplastic meningioma
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE
Introduction

Meningioma is the most common primary intracranial tumor found on incidental neuroimaging (1) and histopathology analysis (2). While meningiomas are prevalent tumors within the central nervous system, and experiencing a slow rise in incidence (2,3), comprehensive information regarding their epidemiology, clinical attributes, and therapeutic strategies lags behind the extensive knowledge available for other tumors such as gliomas. This can be attributed to the prevailing benign nature of incidental meningiomas, as per the WHO grading system (4), where the steady rise in incidence is possibly a combination of factors such as a progressively aging demographic, advancements in healthcare accessibility and elevated rate of histological verification, rather than representing a substantive threat to the general population's health.
Nonetheless, the reality is medical practitioners have an obligation to address incidentalomas. In the context of meningiomas, the central question revolves around the decision of whether to proceed with removal or not. Typically, when dealing with a suspected benign meningioma that exhibits no clinical consequences, no significant growth or threat to neurovascular structures, a conservative management approach is adopted, involving regular follow-up assessments. Conversely, in cases requiring intervention, management plans exhibit considerable diversity in terms of treatment modalities and combinations. These options encompass surgical interventions with varying degrees of resection, as well as therapies such as stereotactic radiosurgery (SRS), fractionated external beam radiotherapy (RT), and experimental pharmacotherapy (5). Meningioma management plans depend on a multitude of factors, including those regarding patient suitability (patient age and degree of frailty), those affecting tumor resectability (tumor size, shape, anatomical location/accessibility), those influencing growth patterns or rates of recurrence (histopathological grading, mass irregularity, genetics, intrinsic, extrinsic risk factors, etc.) (6–11), and those involving practicality (local medical resources, individual expertise of the neurosurgeon and patient consent). Together these factors aim for providing patients with health-related quality of life (HRQOL) (12–14)
Anticipating the proliferative potential of meningiomas and treatment outcome is the hardest of the four, given that the most reliable predictor for their growth rate and recurrence remains the WHO classification (4,15), involving the grading and subtyping of these tumors. This approach necessitates histopathological assessment, which itself demands surgical intervention, rendering it less practical for decision-making prior to considering surgical removal. Furthermore, other potential determinants of growth pattern, namely tumor irregularity or the infinite combinations of intrinsic-extrinsic risk factors, are challenging to employ directly on quantitatively determining the growth rate of meningiomas. Whilst many have proposed single center models of quantification, there are still overwhelming variations in radiological surveillance frequency and/or diversity in the array of measurements for assessing tumor growth and frequency of incomplete follow-up (16), which in turn present a challenge in forming robust, aggregate, multicenter analyses and refining the overarching, simplified decision-making framework (5) for meningioma management.
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

Nevertheless, less is known about anatomical predilections and tendency to exhibit certain symptoms by histological subtype (17,18). One source broadly noted atypical (WHO Grade II) and malignant (WHO Grade III) meningiomas more notably prevalent in regions such as the jugular foramen, tentorium, sphenoid ridge, cerebellopontine angle, petroclival, orbital groove, posterior parafalcine, tuberculum sellae, and intraventricular regions (18), however, it did not consider treatment modalities and other contributing factors when measuring mortality rates. Additionally, the study failed to propose mechanisms explaining the observed findings. Nonetheless, we contend that this approach could contribute to a more nuanced characterization of meningiomas. The prospect regarding predictability of likely histological type preoperatively, based on the site of origin among its factors, has the potential to reshape the future management of meningiomas. Rating the success rate of treatment and its clinical evolution as a function of recurrence, may involve considerations such as extending resection boundaries and implementing or modifying adjuvant treatment strategies.
The primary objective of this retrospective cohort study is to analyze the relationship between anatomical characteristics, histological grading/subtyping and treatment response or patient outcomes in surgically intervened meningioma cases. Specifically, to (a) classify meningioma cases into three grades and fifteen subtypes based on WHO 2021 classification, (b) evaluate any association between meningioma histological subtypes and tumor properties/location within the central nervous system (CNS), and (c) assess any patterns in treatment response and evolution of surgically treated patients based on histological subtyping and tumor properties/location.
Materials and Methods
Materials
We conducted a retrospective, single-center, observational study on patient populations selected from medical records at Clínica Universidad de Navarra (CUN).
To evaluate any association between meningioma histological subtypes and tumor properties/location, patients were selected directly based on histopathological classification thereby extrapolating from the overall database surgically intervened, single, intracranial meningiomas between the years 2008–2020. Additionally, patients diagnosed with genetically confirmed hereditary meningioma (neurofibromatosis 2, mutations in TRAF7, AKT1, KLF4, SMO, PIK3CA, etc.), extracranial meningiomas and cases having incomplete treatment and follow-up data at CUN we excluded from the study.
Meningioma gradings, made with classifications prior to the 2021 WHO version, were recategorized based on their histopathological subtyping reports. In the present analysis, both Proliferation Index Ki67 and Progesterone Receptor immunoreactivity percentages are reclassified into ordinal categorical values to afford a more nuanced statistical assessment. The Ki-67 index, indicative of cellular proliferative activity, is bifurcated into 'High' and 'Low'
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

expressed groups, operationalized as (Positive Ki-67 Index) and Ki-67<5% (Negative Ki-67 Index) (19), respectively, denoting a clearer distinction between tumors with potentially different growth rates and aggressive behavior (20). Progesterone Receptor immunoreactivity is stratified into discrete categories ranging from complete absence to high expression. This classification encompasses Negative immunoreactivity (no detectable expression), Very Low immunoreactivity (less than 15% of cells exhibiting expression), Moderately Low immunoreactivity (between 16–50% of cells), Moderately High immunoreactivity (51–79% of cells), and High immunoreactivity (80% or more of cells) (21) Markers for Epithelial membrane antigen (EMA), were categorized as negative, patchy and intense immunoreactivity as reported on clinical history records. Tumor dimensions are quantified in millimeters, delineating 'length' as the longitudinal axis surpassing others in measurement, and 'width' as the axis the next longest in measurement, at the time closest to surgery.
Together, all the data summarized in Table 1 is grouped into divisions highlighting different important areas relevant to good clinical outcome, including patient demographics, clinical features (on presentation and follow-up) and relevant surgical details, histopathological tumor characteristics, neuroradiological/intraoperative and tumor characteristics.
Supplementary Table 1 further summarizes the clinical and demographic variables and dropdown options extracted from the cohort, in addition to the possible overall management plans. If Simpson grading was provided by surgical notes, Simpson grades I-III are simplified as gross total resections (GTR) whereas grades IV-V are considered subtotal resections (STR). This simplification reflects the contemporary evolution of neurosurgery, where Simpson grading has shifted from being a prognostic tool to a means for documenting the treatment received by the patient (22). Additionally, one can appreciate tumor locations have been classified into 27 possible categories (Supplementary Table 1).
For sub analyses on long term follow-up and recurrence rates, a minimum of two-year followup evaluations and most recent out-patient follow-up no prior to January 1, 2021 were necessary, thus patients only recently operated were excluded from those specific analyses. Incomplete treatment and follow-up data was defined as patients with missing scheduled follow-up consultations or patients continuing follow-up at other institutions, limiting a comprehensive tumor evolution understanding. Those with sufficient follow-up records are tagged as having received adjuvant radiotherapy (SRT/SRS), identified for instances of recurrence, and ascribed an intervention tally, which is specified as the aggregate quantity of surgical procedures necessitated throughout the entirety of the case up to the current date. Varying combinations of radiotherapy, and degree of resection define the overall management plan as: Surgery (GTR) monotherapy; Surgery (GTR) + adyuvant SRT/SRS; Surgery (no GTR) + adyuvant SRT/SRS; Surgery (no GTR) + adyuvant SRT/SRS + HT.

Table 1. Data Collection Overview Category Dependent Variables
Patient Demographics and History
Clinical and Surgical Details
Histopathological Analysis
Biological Sex, Age at time of surgery
Incidental, Indication of surgery, Relevant Clinical Manifestations
Extent of Resection, Simpson Grade (if known), Nº Incidences, Nº Transfusions, Relevant Surgery Descriptions, CUN Follow Up Availability
Pathology Diagnosis WHO Grading, Pathology Diagnosis WHO Subtype Proliferation Index Ki67%, Progesterone Receptor immunoreactivity %, Markers of tumor itself (e.g., Proliferation Index Ki67%, Progesterone receptor (PR), Epithelial membrane antigen (EMA), Estrogen Receptor (ER))
Tumor characteristics
Follow-Up and Recurrence
Tumor Location, Size Length (mm), Size Width (mm), Left/Right, Irregularity, Red flags signals
4 total follow ups to summarize overall management plan: Adjuvant SRS/RT, Recurrence, Total Known Surgeries Required
Table 1. Overview of collected variables from the CUN database by relevant domain: Patient Demographics and History, Clinical and Surgical Details, Histopathological Analysis, Tumor characteristics, Follow-Up and Recurrence. Clinical histories were anonymized and recoded at all times.
Methods
A cross-tabulation (contingency table) approach was utilized to examine the relationship between WHO meningioma subtypes (independent variable) and various dependent variables, including demographic and pathological characteristics. The dependent variables, delineated in rows, were cross-tabulated against the meningioma subtypes, listed in columns, to assess frequency distributions within each subtype.
For each cell in the table, the count of cases falling into the intersecting category was recorded. Subsequently, proportions were calculated to express the number of cases for each dependent variable category as a percentage of the total cases within each subtype. This method facilitates direct comparison between the frequency of each category within subtypes versus the general frequency across all cases.
The analysis aims to identify significant patterns or deviations in the distribution of dependent variables across different meningioma subtypes, providing insights into the characteristics that may be predominantly associated with each subtype. Statistical significance is assessed using Chi-square tests, where appropriate, to determine if distributions differed significantly from what would be expected by chance, taking proportions across all meningioma subtypes as reference and considering a 95% confidence interval.

Results
Descriptive Description of Demographics
The data from the present study provides detailed insights into the demographic distribution and pathology characteristics of tumor cases, totaling 122 patients receiving surgical treatment in our center as a result of a single intracranial meningioma from January 1, 2008 to December 31, 2020 (Table 2). Starting with the biological sex of the patients, females account for 78 cases (63.93%), while males are 44 cases (36.07%). Statistical analysis of age distribution among meningioma patients reveals the following: the mean age is 59.33 years, the median age is 60 years, and overall ranges from 56 years, from the youngest operated at 34 years to the oldest at 90 years. In terms of Pathology Diagnosis WHO Grading, Grade 1 tumors are the most prevalent with 110 cases (90.16%), followed by Grade 2 with 10 cases (8.20%), and Grade 3 with 2 cases (1.64%).
Figure 1. CUN 2008-2020 surgically operated meningioma distributions. (a) (b)
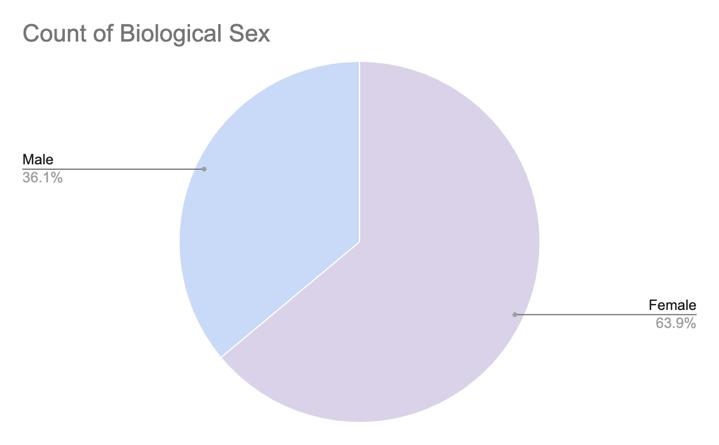
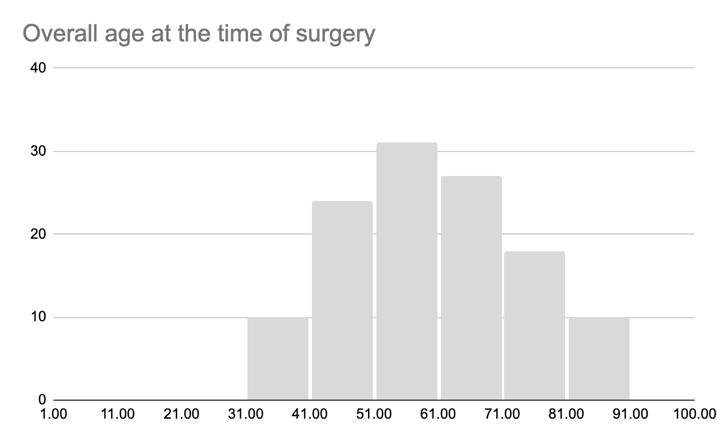
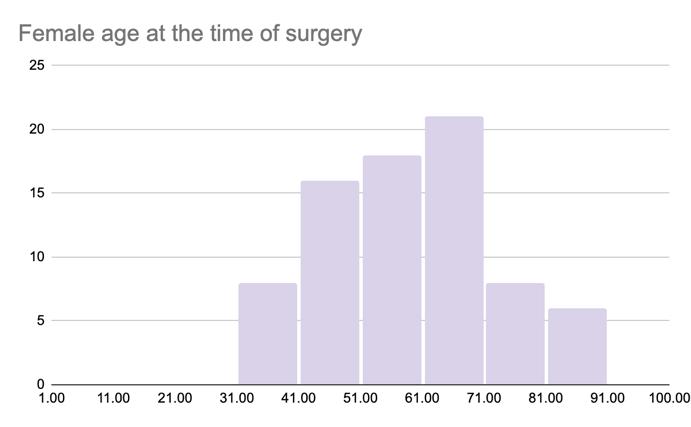
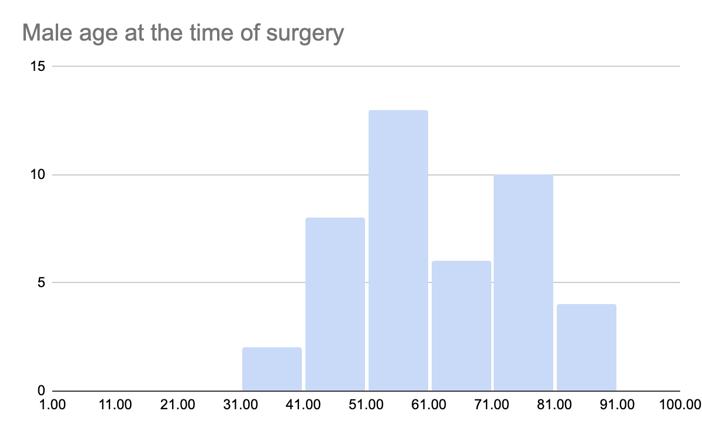
Figure 1. Demographic distribution of patients who underwent meningioma surgery between 2008 and 2020, showing (a) female-to-male prevalence, (b) overall age distribution peaks and separate age distributions for (c) females and (d) males.
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

117 meningioma cases of the 122 total were classified by WHO subtypes, the Transitional (mixed) subtype was the most prevalent, with 48 cases accounting for 41.03% of the total.
This was followed by the Meningothelial subtype with 34 cases (29.06%), and the Fibrous (fibroblastic) subtype with 15 cases (12.82%). Less common subtypes included Angiomatous and Atypical, each represented by 3 cases and constituting 2.56% of the total, respectively.
The Anaplastic subtype was found in 2 cases, making up 1.71% of the cohort. Transitional (mixed)-Psammomatous, and Fibrous (fibroblastic)-Psammomatous subtypes, as well as Meningothelial-Angiomatous subtypes, were each found in 2 cases, representing 1.71% of the cases respectively. Cases categorized under the "Other" subtype numbered 6, constituting 5.13% of the total.
For the markers of tumor proliferation and hormone receptor status, among the 85 meningioma cases assessed for Ki67% marker, 63 cases (74.12%) are positive Ki-67% Index, with only 22 cases (25.88%) being negative Ki-67% Index. Similarly, among the 81 cases assessed for progesterone receptor (PR) status, most tumors 34 cases (81.40%) and 26 cases (32.10%) —exhibit high to moderately-high immunoreactivity, respectively. Lesser extents include moderately-low and low immunoreactivity presentation in 9 cases (11.11%) and 6 cases (7.41%), respectively, and finally negative immunoreactivity in another 6 cases (7.41%).
Regarding treatment planning, among the 122 cases in the study, 28 cases (22.95%) had received or were indicated for Stereotactic Radiosurgery or Radiotherapy (SRT/SRS) at some point during their condition, while 94 cases (77.05%) were reported to not have needed SRT/SRS. Alongside rates of gross total resection/radical (GTR) and subtotal resection (no GTR) 92 cases (75.41%) and 30 cases (24.59%), respectively overall management plans are classified as requiring surgery only (94 cases, 77.05%), subtotal resection and SRS/SRT (20 cases, 16.39%), total resection and SRS/SRT (7 cases, 5.74%), or total resection and SRS/SRT with hormone therapy 1 case, 0.82%).
Cross - tabulations & Contingency tables
All contingency tables, which detail the cross-tabulations of the data, are presented in Table 3.
Crosstabulation of biological sex and meningioma subtypes in our dataset revealed notable deviations from the widely reported 2:1 female-to-male predilection for meningiomas. Our analysis was predicated on a sample distribution closely mirroring the recognized gender incidence in meningioma cases, with 63.25% females and 36.75% males among the subtyped cases. For Transitional (mixed) meningiomas, the distribution of 33 cases in females (68.75%) versus 12 in males (31.25%) aligned closely with the expected 2:1 ratio (23).
However, disparities emerged in other subtypes. Meningothelial meningiomas presented a near-equivalent distribution between females and males, with 16 cases (47.06%) in females and 18 cases (52.94%) in males, suggesting a deviation towards a 1:1 ratio. Furthermore, atypical meningiomas exhibited a reversal of the typical gender ratio, showing a female-to-
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

male ratio of 1:2. This inversion, while intriguing, may be attributable to the small sample size of this subtype, which could potentially skew these results towards statistical anomalies. In contrast, Fibrous (fibroblastic) meningiomas demonstrated a substantially greater skew towards female prevalence, with cases in females (80.00%) and males (20.00%) resulting in a 4:1 female-to-male ratio.
Tumor locations, divided into 27 different groups, produce very specific crosstabulations, which is insightful when viewing the raw data, but difficult to view patterns and draw conclusions. These are best further re-clustered into broader categories based on lobe location (frontal, parietal, temporal, occipital), superficiality (convexity, falcine/parasagittal, skull base), as well as midline and dural projections. This nuanced subgrouping strategy enables the identification of varied patterns, providing insights without confinement to preexisting categorical frameworks (Table 4).
Fibrous (fibroblastic) meningiomas demonstrated a pronounced propensity for the frontal or anterior fossa regions, with a substantial representation of 60.00% (9 cases), as opposed to the overall distribution of 48.72%.This same subtype also showed increased occurrence in the occipital and posterior fossa areas with 26.67% (versus an overall average of 15.38% in occipital and posterior fossa areas across meningioma subtypes), and was notably prevalent in midline and dural projections at 50.00% (compared to an average of 26.50%). In fact, by case count, fibrous meningiomas are also most likely to be located anteriorly (9 cases) and related to midline structures (8 cases). On the contrary, fibrous meningiomas we significantly lower in prevalence in convexity meningioma contexts at 20 00% (where normally 34.17% would present as convexity meningiomas).
Meningothelial meningiomas, however, were less represented in the frontal regions at 41.18% but significantly more common in the parietal and middle fossa regions at 56.52% (versus 36.75% overall), and overwhelmingly present at the skull base with 41.67% (versus 35.04% overall), each representing a case count of 17. Finally, transitional (mixed) meningiomas tend to follow the average, in part likely due to their highest representation as most common meningioma subtype in this study. Similar to meningiomas overall, they are mostly found anteriorly (54.17%), while convexity and skull base (35.42% and 33.33%, respectively) localizations surpass midline presentations.
The subtypes Transitional (mixed), Meningothelial, and Fibrous (fibroblastic) each exhibited a nearly 4:1 ratio of negative to positive Ki-67% proliferation indices, closely mirroring the overall results of 73.81% negative and 31.14% positive. This alignment is likely influenced by their status as the most prevalent WHO subtypes and their general association with WHO Grade 1 categorization. Despite the challenges in discerning patterns due to the extremely low number of Ki-67% determinations for the less common subgroups, all remaining subtypes with the exception of Anaplastic meningioma (Grade 3) consistently demonstrated negative Ki-67% results when more than one determination was available. This includes all variations that combine transitional (mixed), meningothelial, fibrous (fibroblastic), and angiomatous subtypes, further corroborating the consistent predominance of Ki-67%negative results observed in each of these subtypes when found alone.
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

In assessing Progesterone Receptor (PR) Marker immunoreactivity among various meningioma subtypes, distinct patterns emerge. Transitional (mixed) and meningothelial meningiomas consistently show high to moderately high PR immunoreactivity, with respective totals of 85.30% and 82.61%. In contrast, fibrous (fibroblastic) meningiomas display a broader spectrum of PR immunoreactivity levels, with 41.67% high, another 41.67% moderately high, and 8.33% each in moderately low to low, and "patchy" categories. This more varied distribution starkly contrasts with that of Angiomatous meningiomas, which, although limited in number, predominantly exhibit very low to negative PR marker immunoreactivity at a 1:2 ratio, with no instances of moderate or higher immunoreactivity. Patterns within other subtypes remain difficult to establish due to their small sample sizes.
Assessing recurrence rates in meningioma cases proves challenging due to inconsistencies in follow-up practices. Among 122 cases, only 37 either presented with recurrence either preoperatively or postoperatively or had sufficiently thorough follow-up defined as a minimum of two years of evaluations and the most recent outpatient follow-up not prior to January 1, 2021 to reliably determine that further recurrence is highly unlikely. Additionally, this assessment does not take into account the variability in management plans, which may include different surgical resections and adjuvant therapies. Despite these complexities, the data counting surgical interventions is clearer: 98 of 117 cases (83.76%) required only a single surgical procedure to date. The ratios of single interventions to reinterventions were 11:1, 7:1, and 4:1 for Transitional (mixed), Meningothelial, and Fibrous (fibroblastic) meningiomas, respectively. Conversely, higher reintervention rates, defined as necessitating two or more surgeries overall, were observed in Angiomatous, Atypical, and Anaplastic subtypes, with ratios of 0:3, 1:2, and 0:2, respectively. In other words, 100% of the Angiomatous and Anaplastic cases required more than one surgery over the course of treatment, a particularly surprising finding for Angiomatous meningiomas, which are classically categorized as Grade 1 and benign.
Chi - squared tests
Chi-squared tests, as presented in Table 5, were utilized to identify significant patterns or deviations in the distribution of meningioma WHO subtypes across various dependent variables. These tests require a minimum sample size for each subtype and expected values to ensure statistical validity. In fact, entire subtypes have been re-clustered for the purpose of these tests, leaving transitional (mixed), meningothelial, fibrous (fibroblastic), angiomatous, atypical, anaplastic, chordoid meningiomas as the groups. Additionally, due to a failure to reach a minimum of 5 expected cases in many instances, Chi-Square p-values were compared with a Fisher’s Exact Test p-values to corroborate findings. Nevertheless, Chi-Square tests were retained within the study methodology to allow for adaptability in future research with potentially larger sample sizes.
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

As detailed in Table 5, the analysis of biological sex as a dependent variable revealed Chisquared values of 60.4299 (p-value <0.00001), and 0.0249 Fisher’s Exact Test p-value, suggesting a potential association between meningioma subtype, particularly highlighting meningothelial, fibrous, and atypical subtypes and biological sex.
For anatomic location, with a considerably higher degree of freedom (df = 130), the Chisquared value being 6088.600 (p-value <0.00001), indicating very strong statistical significance and suggesting a notable association between these subtypes and their specific anatomic locations. Fisher's exact test p-value (95% CI) for 'Anatomic Location' was not assessed as this test is not well suited for contingency tables far greater than 2x2. P-values in this category were ≤ 0.05, underscoring statistical significance; nevertheless, when reclustered into different anatomical location groupings, Chi square p-values (18.6501; p = 0.0449) statistical significance decreases, barely reaching 0.05 threshold (Table 4).
The analysis involving the Ki-67% index showed 68.18003459, with corresponding p-value of <0.00001, but 0.01722 p-value with the Fisher's exact test As such, these results suggest no significant association between Ki-67% index levels and the meningioma WHO subtypes. The lower rates of statistical significance may be attributed to reduced statistical power, as not all cases may have received immunoreactivity analysis for Ki-67% index of proliferation or PR marker and leading to reduced sample sizes. However, results would make sense in the context of nearly 4:1 ratio of negative-to-positive Ki-67% proliferation indices across most groups.
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE


INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

Table 2 provides a comprehensive summary of the demographic and clinical characteristics of patients diagnosed with meningioma who underwent surgical treatment at the Clinica Universidad de Navarra (CUN) between 2008 and 2020. The table categorizes the data based on age, gender, tumor location, and size, as well as surgical outcomes and follow-up duration. Counts are provided and use of percentages (%) next to raw counts helps in understanding the distribution and proportions of the cases overall (right) and with respect to specific subcategories (left), providing a detailed statistical breakdown of the meningioma cases. *‘Petroclivus’ includes cases reported as spheno-petro-clivus.
SENIOR RESEARCH PROJECT (TFG):

INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON

SENIOR RESEARCH PROJECT (TFG): INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

Table 3 presents a contingency table that categorizes findings based on the World Health Organization (WHO) subtypes of a particular disease or condition. It systematically arranges data to show the distribution of various clinical or pathological findings across different WHO classifications: Transitional (T.), Meningothelial (M.), Fibrous (F.), Angiomatous (Ang.), Atypical (Atyp.), Anaplastic (Ana.), Transitional-Psammomatous (T.Ps.), Fibrous- Psammomatous (F.Ps.), Meningothelial -Angiomatous (M.Ang.), Meningothelial - Atypical (M.Atyp.), Chordoid (Chord.), Meningothelial - Psammomatous (M.Ps.), Microcystic (Micro.), Psammomatous (Ps.), Secretoy (Secr.). *‘No recurrence’ is subject to additional exclusion criteria defined in Materials and Methods regarding recurrence subanalysis.
Table 4 offers an in-depth contingency analysis of findings categorized by World Health Organization (WHO) subtypes and clustered anatomical locations for meningiomas. The table displays counts of cases across six distinct location categories and respective proportions out of tumor subtype total right below. A Chi-square analysis was conducted with 10 degrees of freedom was able to be conducted when only including the three most common subtypes in this study: Transitional, Meningothelial, and Fibrous meningiomas. Exact tumor localizations as per the re-clustering can be appreciated clearly in Supplementary Table 2.
SENIOR RESEARCH PROJECT (TFG): INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION

Table 5. Chi-squared analysis for Transitional (mixed), Meningothelial, Fibrous (fibroblastic), Angiomatous, Atypical, Anaplastic, Chordoid meningiomas (hidden) to determine relation to biological sex, anatomic location, Ki-67% Index, PR marker, recurrence count, total surgeries count. Due to low sample sizes and expected counts for reliable Chi-squared values and p-values, these are all compared with a Fisher’s Exact Test. * Fisher’s exact test p-value (95% CI) for ‘Anatomic Location’ and ‘Recurrence Count’ have been omitted as this test is not well suited for contingency tables as large as these.
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

Similarly, the Chi-squared values regarding progesterone receptor (PR) immunoreactivity was 653.3617108 (p-value <0.00001), without calculating for Fisher's exact test p-value for the same reasons as with 'Anatomic Location'. This may reflect tendency of fibrous (fibroblastic) meningiomas’ broader spectrum of PR immunoreactivity levels.
In the recurrence count analysis, Chi-squared value of 51.73019343 (p-value <0.00001), but non-significant for Fisher's exact test p-value of 0.9139 reflects the inconsistencies in followup practice previously highlighted. Additionally, ‘no recurrence’ counts are greatly underestimated due to added exclusion criteria for sub-analyses regarding follow-up and recurrence outlined in Materials and Methods.
Lastly, the total surgeries over the course of the entirety of the case revealed Chi-squared values of 53, with p-value of 0.0028, indicating significant association between the number of surgeries and these specific, meningioma WHO subtypes. However, a higher sample size > 5 cases is advisable for Angiomatous, Atypical, and Anaplastic subtypes to strengthen the validity of these results, given that previously they were seen to have higher rates of reinterventions.
Discussion
Meningioma is the most common primary intracranial tumor (1), comprising a family of neoplasms most likely originating from the meningothelial cells of the arachnoid mater. Currently, the World Health Organization (WHO) grading system classifies meningiomas into Grades 1, 2, and 3. This classification remains the gold standard for predicting tumor aggressiveness and includes further subdivision into 15 histopathological subtypes (4).
The overall 2.3:1 female-to-male ratio demographic distribution of meningiomas (23), aligns closely with the findings of our study. However, the literature is sparse regarding the distribution across these varying histopathological subtypes (18,23), with most studies focusing on sex distributions within the general context of meningioma presentations (11,23,24)
The incidence of meningioma increases with age, peaking at a median age of 67 years according to data from CBTRUS (23). In contrast, data from CUN reports a slightly lower median age of diagnosis at 60 years, potentially influenced by Berkson's bias, where CUN may attract a demographic with earlier presentations. The variance in meningioma subtypes may be influenced by genetic, hormonal, or environmental factors, which could correspond to these demographic variations. Furthermore, certain subtypes of meningioma may present unique clinical characteristics.
In the present study, fibrous (fibroblastic) meningiomas showed a more pronounced female predominance and were more commonly located in the frontal/anterior fossa regions and occipital/posterior fossa, along the midline (contrasting with their lower prevalence in the convexity) It is possible frontal and occipital, midline structures (such as tentorium and falx
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

cerebri) that contain very dense connective tissue with numerous longitudinal bundles of collagen fibers (25,26)normally in the sagittal direction, hold relation to the abundance of spindle cells or interlacing bundles in collagen-rich matrices characteristic of fibrous meningiomas (4,27). Additionally, fibrous (fibroblastic) meningiomas exhibited a wider range of progesterone receptor (PR) immunoreactivity levels, suggesting greater variety hormonal sensitivity may play less of a consistent role in their pathophysiology, and rather underlying characteristic, molecular features on microscopy being the central driving factor for the evolution into fibrous subtype
Meningothelial meningiomas presented a nearly equal distribution between females and males, more commonly occurring in the parietal and middle fossa regions, and were notably prevalent at the skull base and convexity. These tumors consistently showed high to moderately high PR immunoreactivity, similar to transitional (mixed) meningiomas. One study found PR immunoreactivity expression tended to be lower in the elderly, with no significant sex differences, including between premenopausal and postmenopausal women (21). Additionally, higher expression was noted in medial skull base and spinal locations (21), exactly aligning with the patterns observed in this study for sex and tumor location distribution for meningothelial meningiomas.
Transitional meningiomas typically exhibit features of both meningothelial and fibrous patterns (4), including frequent whorl formations and psammoma bodies round, microscopic calcifications. Nevertheless, among the patients with red flags including identification of calcifications intraoperatively or on imaging, there does not seem a greater proportion of transitional meningiomas, as calcifications are not exclusive to transitional subtypes. Results are not unexpected, as the majority of patterns observed are either visibly similar to fibrous and meningothelial subtypes, or a midpoint in distribution between fibrous findings and meningothelial findings. In contrast, they were significantly more prevalent in patients with findings of hyperostosis and cerebral edema (50% transitional meningiomas among this subgroup, versus 41% overall).
Angiomatous meningiomas, typically non-calcifying and favoring convexity locations (28)(as opposed to specifically frontal areas in this study), predominantly exhibited very low to negative PR immunoreactivity and negative Ki-67% index (29). Although low PR levels generally suggest a low recurrence probability reinforcing the use of PR as a routine pathological marker for meningioma prognosis by many studies the higher reintervention rate observed for angiomatous meningiomas was unexpected. It is relevant to note that the extent of peritumoral brain edema was found to be related to micro-vessel density (MVD) of tumors, rather than the expression of matrix metalloproteinase 9 (MMP9) or Vascular endothelial growth factor (VEGF) (29). This subtype’s characteristic hypervascularity, responsible for considerable cerebral edema (28), may influence the likelihood of recurrence. Peritumoral edema cases increased permeability in dural tail-positive tumors (30), which is associated with pial irrigation from intracerebral blood vessels (31), which itself is correlated with cortical invasion, regardless of tumor size or location. This is the case in this study, where a greater number of cases with angiomatous meningiomas exhibited broad-based dural attachment or a dural-tail, which along with peritumoral edema, VEGF, could collectively contribute to their recurrence profile, despite most often being Grade 1 meningiomas
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

Atypical meningiomas, typically classified as WHO Grade 2, revealed a reversal of the typical gender ratio are not entirely unexpected. Previous research has observed a decrease in the incidence of malignant meningiomas and an increase in non-malignant meningiomas among middle-aged females (32,33). Additionally, there is a general consensus suggesting that atypical meningiomas occur more commonly in men (34). This shift in gender distribution highlights potential differences in the biological behavior of meningioma subtypes, specifically those more aggressive in pattern, and underscores the need for further investigation into the factors influencing these patterns.
Anaplastic meningioma (Grade 3) consistently demonstrated positive Ki-67%, a known predictor of clinical outcome in patients with atypical or anaplastic meningioma (tumor recurrence and overall survival) (20,35). One study corroborates the higher recurrence rate of anaplastic meningiomas (34,36), which may also be related to difficulty of surgical resection based on anatomical location. The present study cannot definitively identify patterns due to small sample size, however, others have noted a predilection of anaplastic meningiomas to locations operatively difficult to resect such as falx cerebri and interventricular locations (compared to brain convexity) (34), which may make total gross resection a greater challenge
Regarding overall recurrence rates, this study initially suggests a recurrence rate just below 50% (specifically 45.95%), which appears unrealistically high when compared to general findings in the literature. However, it is crucial to note that non-recurring cases are significantly underrepresented. Many were excluded because they did not meet the stringent follow-up criteria necessary to definitively rule out recurrence. Importantly, most benign cases, which are unlikely to recur, also tend not to return for follow-up due to effective clinical management. Additionally, while there is significant statistical evidence suggesting relationships in recurrence rates across different meningioma subtypes, these findings should be interpreted with caution.
Limitations
A greater sample size would have been ideal for this study to enhance the robustness and generalizability of our findings. The limited sample size constrained our ability to perform comprehensive statistical analyses, leading to the omission of several potentially insightful analyses that could further elucidate the nuances of meningioma subtypes and their demographic distributions. Moreover, the demographic representation within our study, as a single-center study, may not fully capture the variability observed in broader populations.
To address these limitations, we propose a call for multicenter collaboration. Collaborating with multiple centers would not only increase the sample size but also diversify the demographic representation, thereby mitigating the biases associated with single-center studies. Additionally, strengthening our knowledge base for less common subtypes of meningiomas, which are often underrepresented in studies Such collaborations would
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

enhance the reliability of follow-up data, enabling more sustained and detailed tracking of clinical outcomes over time. Ideally, a comprehensive study such as this would include 500600 cases of meningiomas meeting inclusion criteria.
Future Research
This research avenue offers the potential to develop models assessing the impacts of various surgical and advanced imaging techniques on meningioma management, under the right circumstances. The first step would involve compiling a comprehensive, detailed, and representative database of retrospective data. Utilizing Cytoscape©, a software platform that is open-source and designed to visualize networks of complex molecular interactions and biological pathways, could facilitate the visualization of these relationships at a glance. This approach would set the groundwork for more specific and direct statistical analyses. The results could then inform the design or refinement of artificial intelligence algorithms, paving the way for potential prospective trials. These trials would seek to validate the algorithms as effective tools for managing meningioma, particularly in low-income, middle-income, and rural settings.
Conclusion
This retrospective cohort study has provided critical insights into the relationships between anatomical characteristics, histological subtyping, and treatment responses in surgically intervened meningioma cases. Our findings confirm that the demographic distribution of meningiomas reflects a greater prevalence in females, aligning with the general literature.
There seems to be a pronounced female predominance, especially in fibrous (fibroblastic) meningiomas, which were commonly found in the frontal/anterior and occipital/posterior fossa regions, and variability progesterone receptor (PR) expression. In contrast, meningothelial meningiomas showed a more equal gender distribution and were predominantly located at the medial skull base, frequently exhibiting high PR immunoreactivity. Transitional meningiomas, the most prevalent subtype in this study, significantly more prevalent in patients with findings of hyperostosis or broad based dural attachement or dural tail than overall.
Atypical meningiomas displayed a shift in gender distribution towards males, a finding that calls for further investigation into the biological behaviors of more aggressive subtypes. Similarly, anaplastic meningiomas demonstrated high Ki-67% index levels, correlating with their aggressive nature, in addition to challenging resection outcomes as typically locating themselves in particularly, anatomically complex locations.
SENIOR RESEARCH PROJECT (TFG):
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

Many of these differences in case findings by meningioma subtype suggest an intricate network of relationships likely influenced by genetic, hormonal, and environmental factors. Both meningioma subtype and meningioma anatomic predispositions may be considered as independent factors influencing clinical presentations, extent of surgical resection, overall survival, recurrence rate, appropriate management plan, etc. However, strong correlations between meningioma subtype and other features, including anatomical location, demographic distribution, aggressiveness/growth rate raises the idea of a possible molecular connection between all these features, which can together influence the determination of meningioma subtype.

Bibliography
1. Vernooij MW, Arfan Ikram M, Tanghe HL, JPE Vincent A, Hofman A, Krestin GP, et al. Incidental Findings on Brain MRI in the General Population From the Departments of Epidemiology A bs t r ac t [Internet]. Vol. 357, N Engl J Med. 2007. Available from: www.nejm.org
2. Ostrom QT, Price M, Neff C, Cioffi G, Waite KA, Kruchko C, et al. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2015–2019. Neuro Oncol. 2022 Oct 1;24(5 S):V1–95.
3. Ostrom QT, Cioffi G, Gittleman H, Patil N, Waite K, Kruchko C, et al. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2012-2016. Vol. 21, Neuro-Oncology. Oxford University Press; 2019. p. V1–100.
4. WHO Classification of Tumours Editorial Board. Central Nervous System Tumours WHO Classification of Tumours. 5th ed. Vol. 6. Lyon: International Agency for Research on Cancer; 2021. 283–297 p.
5. Goldbrunner R, Stavrinou P, Jenkinson MD, Sahm F, Mawrin C, Weber DC, et al. EANO guideline on the diagnosis and management of meningiomas. Neuro Oncol. 2021 Nov 1;23(11):1821–34.
6. Fountain DM, Soon WC, Matys T, Guilfoyle MR, Kirollos R, Santarius T. Volumetric growth rates of meningioma and its correlation with histological diagnosis and clinical outcome: a systematic review. Vol. 159, Acta Neurochirurgica. Springer-Verlag Wien; 2017. p. 435–45.
7. Thomann P, Häni L, Vulcu S, Schütz A, Frosch M, Jesse CM, et al. Natural history of meningiomas: a serial volumetric analysis of 240 tumors. J Neurosurg. 2022 Dec 1;137(6):1639–49.
8. Da Broi M, Borrelli P, Meling TR. Predictors of survival in atypical meningiomas. Cancers (Basel). 2021 Apr 2;13(8).
9. Dresser L, Yuen CA, Wilmington A, Walker M, Vogel TJ, Merrell RT, et al. Estrogen hormone replacement therapy in incidental intracranial meningioma: a growth-rate analysis. Sci Rep. 2020 Dec 1;10(1).
10. Johnson MD. Transforming Growth Factor Beta Family in the Pathogenesis of Meningiomas. Vol. 104, World Neurosurgery. Elsevier Inc.; 2017. p. 113–9.
11. Baldi I, Engelhardt J, Bonnet C, Bauchet L, Berteaud E, Grüber A, et al. Epidemiology of meningiomas. Vol. 64, Neurochirurgie. Elsevier Masson SAS; 2018. p. 5–14.
12. Królikowska A, Filipska-Blejder K, Jabłońska R, Haor B, Antczak-Komoterska A, Biercewicz M, et al. Quality of Life after Surgical Treatment of Brain Tumors. J Clin Med. 2022 Jul 1;11(13).
13. Benz LS, Wrensch MR, Schildkraut JM, Bondy ML, Warren JL, Wiemels JL, et al. Quality of life after surgery for intracranial meningioma. Cancer. 2018 Jan 1;124(1):161–6.
14. Haider S, Taphoorn MJB, Drummond KJ, Walbert T. Health-related quality of life in meningioma. Neurooncol Adv. 2021 Jan 1;3(1).

15. Bender L, Somme F, Lhermitte B, Ahle G, Boone M, Blonski M, et al. High risk of recurrence for grade II meningioma: a 10-year multicenter analysis of prognosis factors. Chin Clin Oncol. 2021 Jun;10(3):26–26.
16. Islim AI, Mohan M, Moon RDC, Srikandarajah N, Mills SJ, Brodbelt AR, et al. Incidental intracranial meningiomas: a systematic review and meta-analysis of prognostic factors and outcomes. Vol. 142, Journal of Neuro-Oncology. Springer New York LLC; 2019. p. 211–21.
17. Shrestha R, Bishokarma S, Shrestha S, Gurung S, Rani Thapa E, Rayamajhi S, et al. Spatial distribution of histological subtypes of Meningioma: A 14-Year longitudinal study in a developing country. Interdisciplinary Neurosurgery. 2024 Jun;36:101921.
18. Bhat A, Wani M, Kirmani A, Ramzan A. Histological-subtypes and anatomical location correlated in meningeal brain tumors (meningiomas). J Neurosci Rural Pract. 2014;5(3):244.
19. Khanna O, Fathi Kazerooni A, Arif S, Mahtabfar A, Momin AA, Andrews CE, et al. Radiomic signatures of meningiomas using the Ki-67 proliferation index as a prognostic marker of clinical outcomes. Neurosurg Focus. 2023 Jun;54(6):E17.
20. Moon CM, Lee YY, Kim DY, Yoon W, Baek BH, Park JH, et al. Preoperative prediction of Ki-67 and p53 status in meningioma using a multiparametric MRI-based clinicalradiomic model. Front Oncol. 2023 May 23;13.
21. Maiuri F, Mariniello G, de Divitiis O, Esposito F, Guadagno E, Teodonno G, et al. Progesterone Receptor Expression in Meningiomas: Pathological and Prognostic Implications. Front Oncol. 2021 Jul 15;11.
22. Rapoport BI, McDermott MW, Schwartz TH. Letter to the Editor. Time to move beyond the Simpson scale in meningioma surgery. J Neurosurg. 2021 Feb;1–2.
23. Ostrom QT, Price M, Neff C, Cioffi G, Waite KA, Kruchko C, et al. CBTRUS Statistical Report: Primary Brain and Other Central Nervous System Tumors Diagnosed in the United States in 2016 2020. Neuro Oncol. 2023 Oct 4;25(Supplement_4):iv1–99.
24. Cea-Soriano L, Wallander MA, García Rodríguez LA. Epidemiology of Meningioma in the United Kingdom. Neuroepidemiology. 2012;39(1):27–34.
25. Tatarlı N, Ceylan D, Canaz H, Tokmak M, Bay HH, Şeker A, et al. Falcine venous plexus within the falx cerebri: anatomical and scanning electron microscopic findings and clinical significance. Acta Neurochir (Wien). 2013 Nov 14;155(11):2183–9.
26. Walsh DR, Zhou Z, Li X, Kearns J, Newport DT, Mulvihill JJE. Mechanical Properties of the Cranial Meninges: A Systematic Review. J Neurotrauma. 2021 Jul 1;38(13):1748–61.
27. Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, et al. The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol. 2021 Aug 2;23(8):1231–51.
28. Liu Z, Wang C, Wang H, Wang Y, Li JY, Liu Y. Clinical characteristics and treatment of angiomatous meningiomas: a report of 27 cases. Int J Clin Exp Pathol. 2013;6(4):695–702.
SENIOR
(TFG): INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

29. Yang L, Ren G, Tang J. Intracranial Angiomatous Meningioma: A Clinicopathological Study of 23 Cases. Int J Gen Med. 2020 Dec;Volume 13:1653–9.
30. Kim BW, Kim MS, Kim SW, Chang CH, Kim OL. Peritumoral Brain Edema in Meningiomas : Correlation of Radiologic and Pathologic Features. J Korean Neurosurg Soc. 2011;49(1):26.
31. Yoon JT, Lee KM, Na K. Dural tail sign positive tumors: Points to make a differential diagnosis. Radiol Case Rep. 2024 Feb;19(2):773–9.
32. Santos RRP dos, Rampazzo ACM, Zicarelli CAM, Camargo LV de. Peculiarities of Atypical Meningiomas: Literature Review. Arquivos Brasileiros de Neurocirurgia: Brazilian Neurosurgery. 2022 Jun 3;41(02):e137–44.
33. Cao J, Yan W, Hong X, Yan H. Epidemiology and survival of non-malignant and malignant meningiomas in middle-aged females, 2004-2018. Front Oncol. 2023 Apr 26;13.
34. Maier H, Öfner D, Hittmair A, Kitz K, Budka H. Classic, atypical, and anaplastic meningioma: three histopathological subtypes of clinical relevance. J Neurosurg. 1992 Oct;77(4):616–23.
35. Bruna J, Brell M, Ferrer I, Gimenez-Bonafe P, Tortosa A. Ki-67 proliferative index predicts clinical outcome in patients with atypical or anaplastic meningioma. Neuropathology. 2007 Apr 13;27(2):114–20.
36. Brodbelt AR, Barclay ME, Greenberg D, Williams M, Jenkinson MD, Karabatsou K. The outcome of patients with surgically treated meningioma in England: 1999–2013. A cancer registry data analysis. Br J Neurosurg. 2019 Nov 2;33(6):641–7.
SENIOR
RESEARCH PROJECT
(TFG):
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE
Supplementary Materials
Table 1. Clinical and Demographic Variables

Demographic details
Biological Sex
Male
Female
Histopathology
WHO grading
1 2 3
Subtyping meningothelial Fibrous (fibroblastic)
Transitional (mixed)
Psammomatous
Angiomatous Microcystic Secretory
Lymphoplasmacyte-rich Metaplastic Chordoid
Clear Cell
Atypical Papillary Rhabdoid
Anaplastic Ki-67 % index
≥ 5% (Positive)
< 5% (Negative)
Progesterone Receptor (PR) Immunoreactivity
0% (Negative)
1-15% (Very Low)
16-50% (Moderately Low)
51-80% (Moderately High)
≥81% (Very High)
Epithelial membrane antigen (EMA)
Immunoreactivity
Negative
Patchy
Intense
Any Preoperative Symptoms
Asymptomatic
Raised ICP
Seizures
Headaches
Reduced Consciousness
Focal Neurological Deficits
Surgical details
Incidental Yes No
Age at surgery
Indication
Recurrence
Clinical Manifestations
Other
Extent of Resection
Gross Total (GTR)
Sub Total (no GTR)
Incidences
Transfusions
Tumour properties
Location3
Convexity
Frontal cerebral convexity
Fronto-parietal cerebral convexity
Parietal cerebral convexity
Frontal-temporal cerebral convexity
Occipital cerebral convexity
Entire
Entire temporal skull base
Entire anterior skull base
Falcine/Parasagittal
Frontal falcine
Occipital falcine
Frontal parasagittal
Parietal parasagittal
Occipital parasagittal
Sellar
Suprasellar (region)
Intrasellar
Cavernous Sinus/Parasellar
Other Specific Types
Cerebellopontine Angle
Foramen Magnum
Intraosseous
Intraventricular
Olfactory Groove
Petroclivus
Tentorium
Other
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE
Red Flags
Intratumoral Calcifications
Hyperostosis
Heterogenous enhancement
Broad-based dural attachement/Dural Tail
Cerebral Edema
Central/Peripheral Tumor Cysts
Neurovascular Compression
Neurovascular Invasion
None
Size (Length cm)
Size (Width cm)
Follow-up data available
Yes
No
Category Dependent Variables
Frontal or Anterior Fossa Tendencies
Parietal and Middle Fossa Tendencies
Occipital and Posterior Fossa Tendencies
Convexity Meningiomas
Skull Base Meningiomas
Midline and Dural Projections Meningiomas

Postoperative Symptoms in relation to progression
Asymptomatic
Raised ICP
Seizures
Headaches
Reduced Consciousness
Focal Neurological Deficits
Overall Management plan
Surgery (GTR) monotherapy
Surgery (GTR) + adyuvant SRT/SRS
Surgery (no GTR) + adyuvant SRT/SRS
Surgery (no GTR) + adyuvant SRT/SRS + HT
This category included the Entire Anterior Skull Base, Frontal Cerebral Convexity, Frontal Falcine, Frontal Parasagittal, Frontal-Temporal Cerebral Convexity, Fronto-parietal cerebral convexity, Fronto-Parietal Parasagittal, and Olfactory Groove Meningiomas (n = 57 cases).
Grouping included Cavernous Sinus/Parasellar, Entire Temporal Skull Base, FrontalTemporal Cerebral Convexity, Fronto-Parietal Cerebral Convexity, Fronto-Parietal Parasagittal, Intrasellar, Parietal Cerebral Convexity, Parietal Falcine, Parietal Parasagittal, Sphenoid, Suprasellar, and Temporal Cerebral Convexity Meningiomas (n = 43 cases).
Comprised Cerebellopontine Angle, Foramen Magnum, Occipital Cerebral Convexity, Occipital Falcine, Occipital Parasagittal, Petroclivus, and Tentorium Meningiomas (n = 18 cases).
Included Frontal, Frontal-Temporal, Fronto-Parietal, Occipital, Parietal, and Temporal Cerebral Convexity Meningiomas (n = 40 cases).
Composed of Cavernous Sinus/Parasellar, Cerebellopontine Angle, Entire Anterior Skull Base, Entire Temporal Skull Base, Foramen Magnum, Intrasellar, Olfactory Groove, Petroclivus, Sphenoid, and Suprasellar Meningiomas (n = 41 cases).
Included Cavernous Sinus/Parasellar, Frontal Falcine, Frontal Parasagittal, Fronto-Parietal Parasagittal, Occipital Falcine, Occipital Parasagittal, Parietal Falcine, Parietal Parasagittal, and Tentorium Meningiomas (n = 31 cases).
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE
Evaluation and Self-Reflection

The research process for my dissertation commenced late due to a third and final change in the thesis project. The initial project proposed in my fourth year of medical school was abandoned because of resource constraints. A second project, planned for my fifth year, was also terminated due to unresolved issues with a simulator that was essential for the trial, as the completion remained unconfirmed. Facing impending deadlines, it was advised by my clinical tutor to pivot to another project that was solely reliant on my efforts. This retrospective observational study did not require multicenter collaboration, which significantly simplified the process, relying instead on a sufficiently large sample size drawn from CUN clinical histories given the high prevalence of the targeted CNS condition.
The data collection phase proved to be particularly challenging and labor-intensive. The project involved an in-depth analysis of 122 cases, each requiring extensive review and frequent consultation across multiple departments, including shifts from neurosurgery to neurology and oncological radiotherapy. A significant hurdle was the variability in clinical reporting styles among physicians, which necessitated the time-consuming task of standardizing and temporarily rewriting sections into structured dropdown options for later use. Initially, the average time spent per case was approximately 30 minutes, but gradually improved to as little as 15 minutes per case as the process continued. Technical difficulties with the CUN2018 platform further complicated access to necessary data, with consistent access across devices proving nearly impossible (often costing me between 1 to 2 hours in achieving system access, each time), restricting data collection activities to specific times and conditions. Overall, inconsistencies with my access codes across different devices led to successful use of the platform on only a few occasions: once with the library's CUN2018 system and twice using devices in the neurosurgery department. Additionally, the somewhat most reliable device within the department (by comparison) was limited, often forcing me to conduct data collection exclusively late into the evening, from 6 pm to as late as 11 pm. The need to recode the cases for patient privacy added further complexity to the data handling process.
Upon data assembly, overall patterns were analyzed using Excel filters and advanced capabilities, which facilitated multitier comparisons between variables and with existing literature. The study primarily aimed to explore the impact of meningioma subtypes on factors such as anatomical location, recurrence rates, and their correlation with demographic and clinical presentation patterns. Meningioma subtypes were thus considered as the independent variable in all analyses, with data meticulously categorized into very specific items to allow flexibility in future research, including potential reclassification of anatomic locations.
This approach not only met but exceeded the initial research objectives by allowing more extensive comparisons than originally anticipated. For instance, decisions regarding the cutoff for what would be considered an adequate follow-up can be adjustable based on the structure of the dataset in timeline form. The use of Excel filters provided enhanced capabilities for data overview, particularly for specific patient subgroups reporting conditions
INFLUENCE OF MENINGIOMA MOLECULAR SUBTYPING AND TUMOUR LOCALIZATION ON TREATMENT RESPONSE

such as "calcifications". For instance, specifically among those patients identified with calcifications in neuroimaging or intraoperatively presented with seizures.
Despite the relatively high frequency of meningiomas compared to other CNS tumors, the sample size remained modest, particularly for less common but clinically significant subtypes (such as atypical or anaplastic), impacting the robustness of statistical analyses. This led to the use of Fisher’s exact test to complement Chi-square statistics, especially when expected values in contingency tables were low (<5) Even so, Fisher's exact test is not ideally suited for analyzing contingency tables significantly larger than 2x2. While I employed coding in other areas such as Javascript to systematically modify values within Excel, manual computation of Chi-square values was preferred to monitor and correct errors effectively, especially when recalculating for adjusted groups was necessary when case counts were low. For example, due to low case counts in some categories, I had to frequently recluster groups within various variables, and ensure that all additions accurately corresponded to the new group counts and adjusted degrees of freedom. Nonetheless, towards the conclusion of each test I utilized an online calculator to determine the corresponding p-values.
Despite facing significant personal challenges this year, which added to the difficulty of the project, I am ultimately somewhat satisfied with its completion. Admittedly, I find it less motivating to engage in projects that do not lead directly to publication. Therefore, the final design of this study was deliberately structured to enable potential multicenter expansion or follow-up studies, aligning with broader research objectives that could enhance its academic impact. This strategic approach enabled me to delve deeply into various facets of scientific research, including topic research, literature review, protocol design, data collection and processing, statistical analysis, and the synthesis and comparison of results with existing literature.
Furthermore, several colleagues from other institutions have expressed interest in contributing to the dataset, which suggests promising potential for expanding the study. This potential for collaboration and enlargement of the study's scope is an exciting prospect that I may pursue further.
On another note, I wish I had gained more practical experience with the Institutional Review Board (IRB) application process. Understanding the intricacies of financing plans and grant applications remains a relatively new and unexplored area for me. However, I am gradually acquiring experience in grant writing and the IRB process through other projects. Still, a deeper understanding of how these processes function within the context of CUN would significantly enhance my ability to advance several of my projects that are currently on hold due to these gaps in understanding the operational nuances at CUN.
