Ahuman iPSC platform for studying pathogenic protein aggregation in neurodegeneration
Eve Corrie1, Anna-Lena Zepernick1, Matthieu Trigano1, and Emma V. Jones1
1Medicines Discovery Catapult, Block 35, Alderley Park, Cheshire, SK10 4ZF, UK
Introduction
• Protein misfolding, phosphorylation, and aggregation are central features of many neurodegenerative diseases
• These pathogenic proteins can seed further misfolding and propagate pathology across anatomically connected brain regions
• These processes contribute to cellular dysfunction and death, and are increasingly being targeted by novel therapeutics in the drug discovery landscape
• MDC has developed human in vitro systems to study the pathology of α-synuclein and tau in iPSC-derived cortical neurons in 96- and 384-well format using high content imaging assays and analysis pipelines
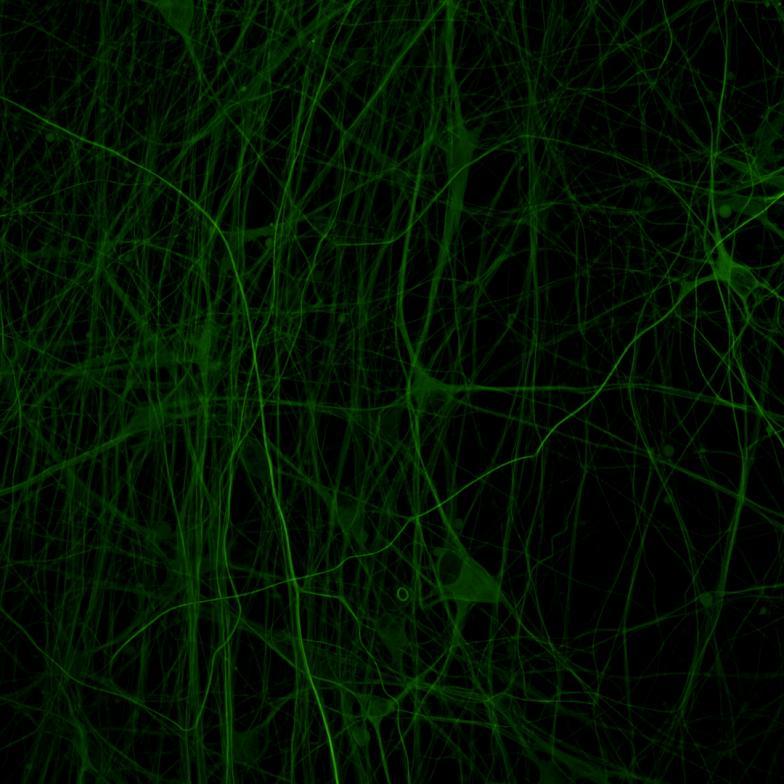
Live imaging of tau aggregation using lentiviral systems
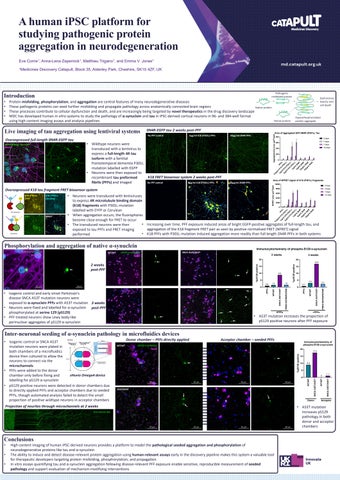
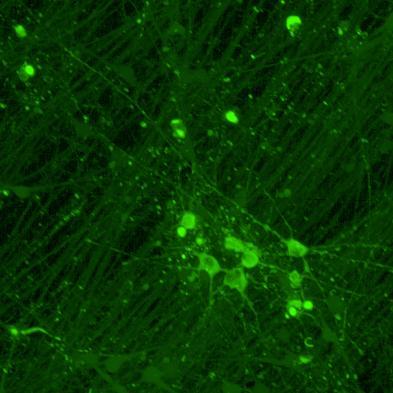
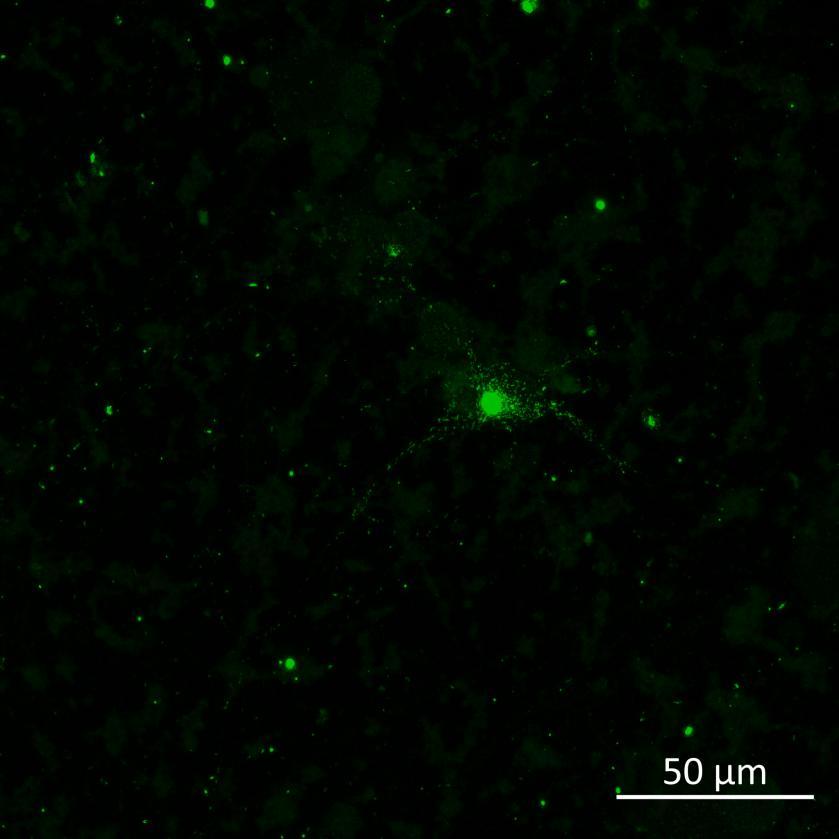
Overexpressed full-length 0N4R-EGFP tau
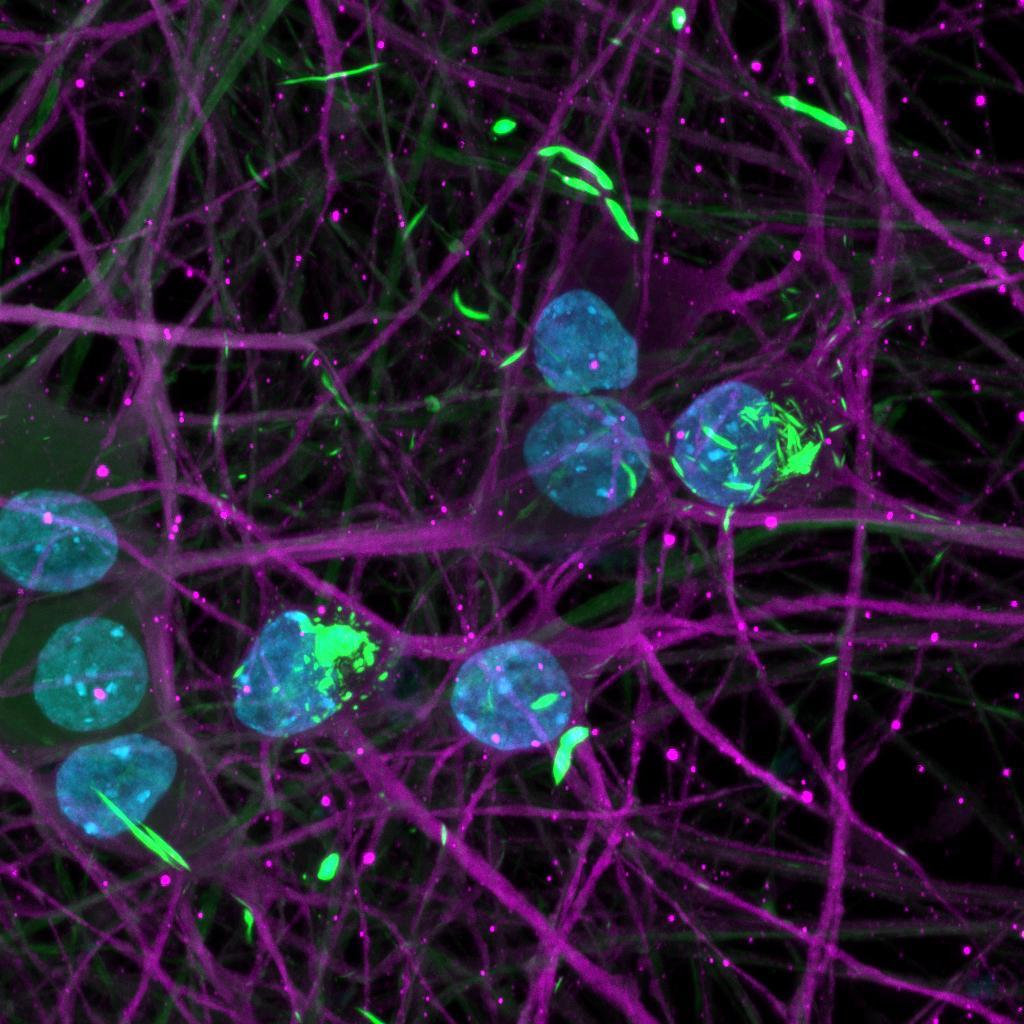
• Wildtype neurons were transduced with a lentivirus to express a full-length 4R tau isoform with a familial frontotemporal dementia P301L mutation labelled with EGFP
• Neurons were then exposed to recombinant tau preformed fibrils (PFFs) and imaged
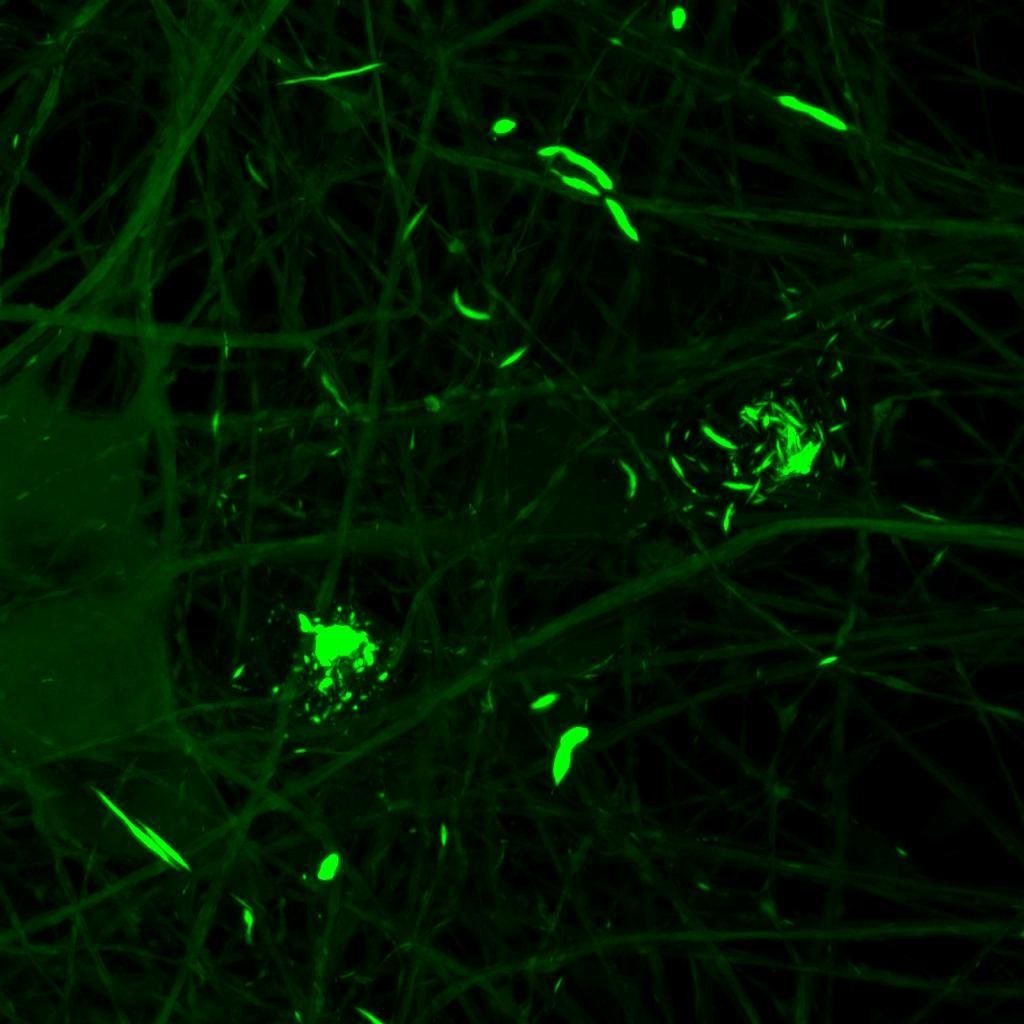
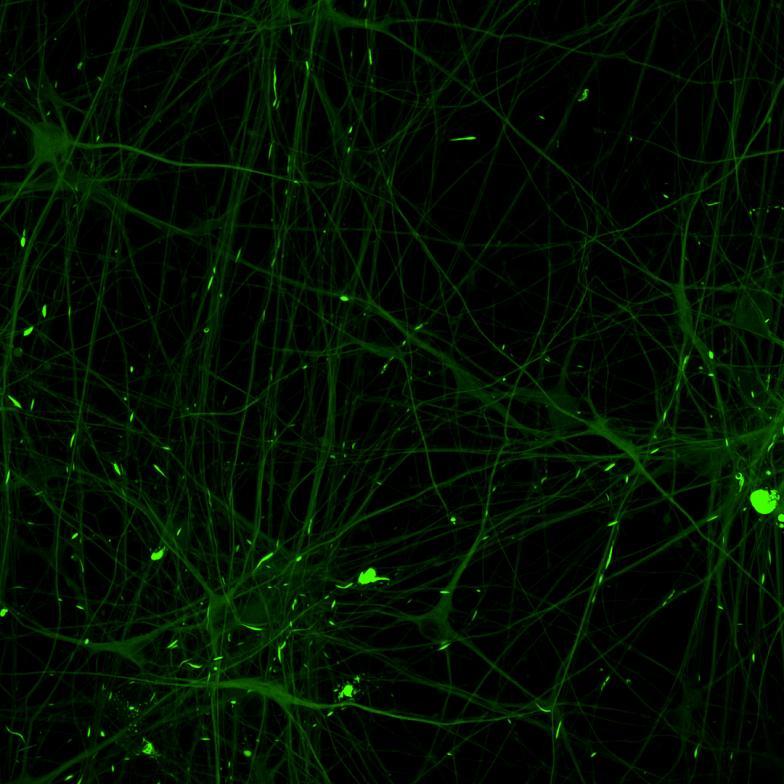
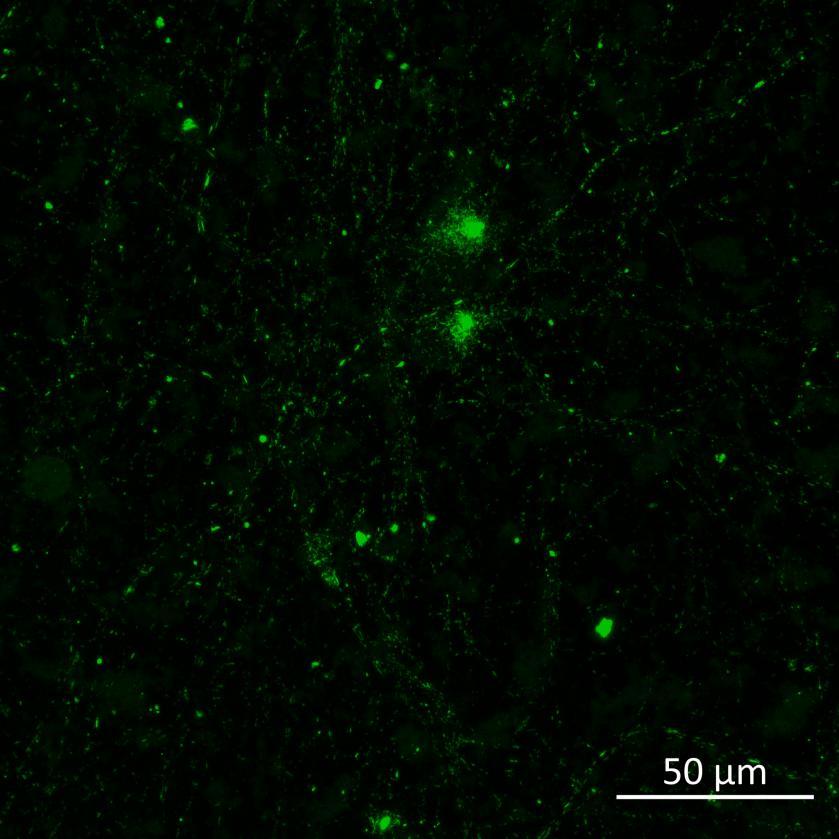
Overexpressed K18 tau fragment FRET biosensor system
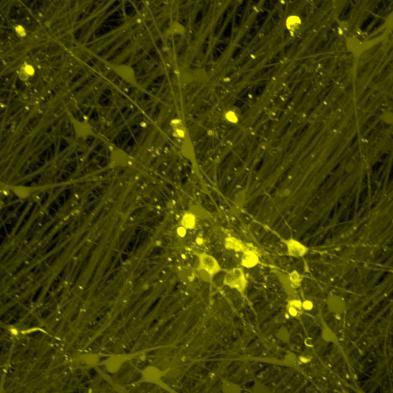
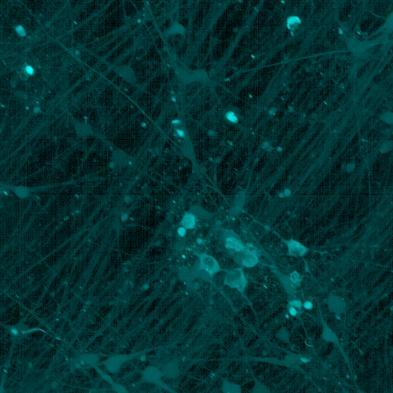
• Neurons were transduced with lentiviruses to express 4R microtubule binding domain (K18) fragments with P301L mutation labelled with EYFP or Cerulean
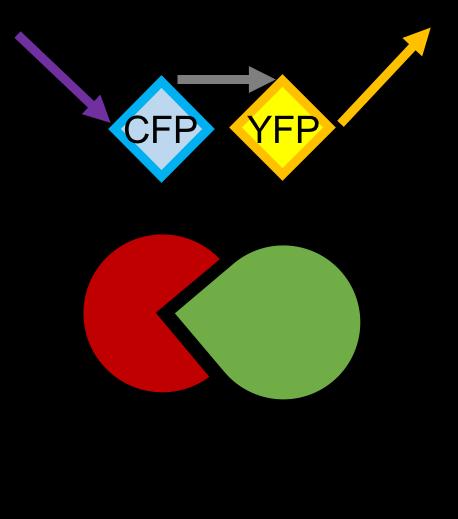
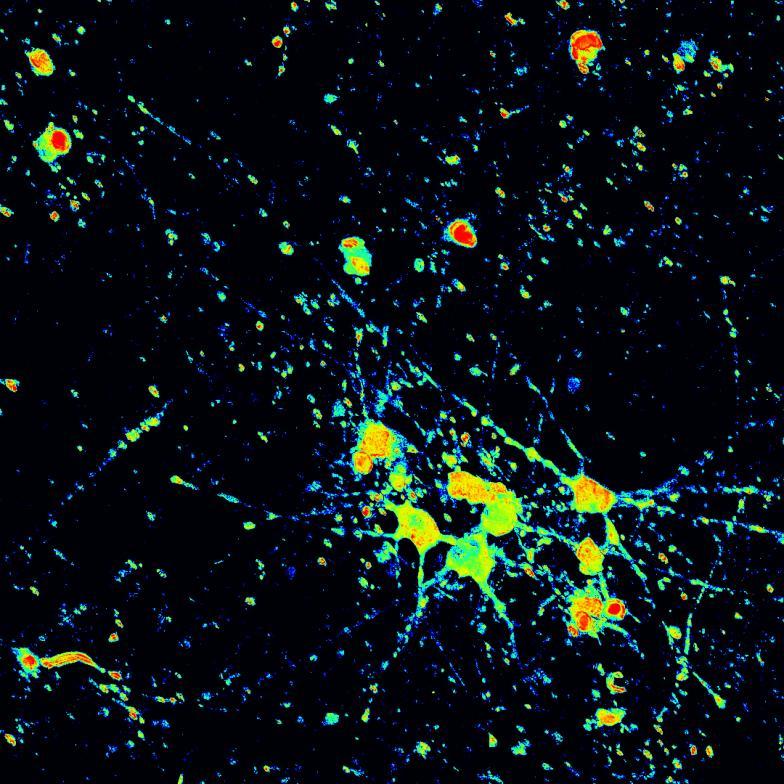
• When aggregation occurs, the fluorophores become close enough for FRET to occur
• The transduced neurons were then exposed to tau PFFs and FRET imaging performed
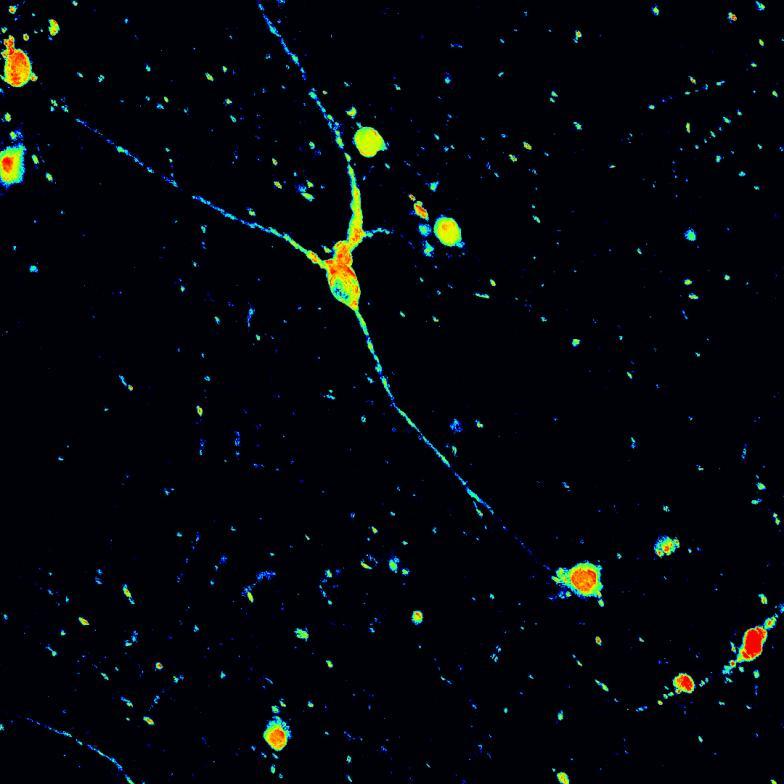
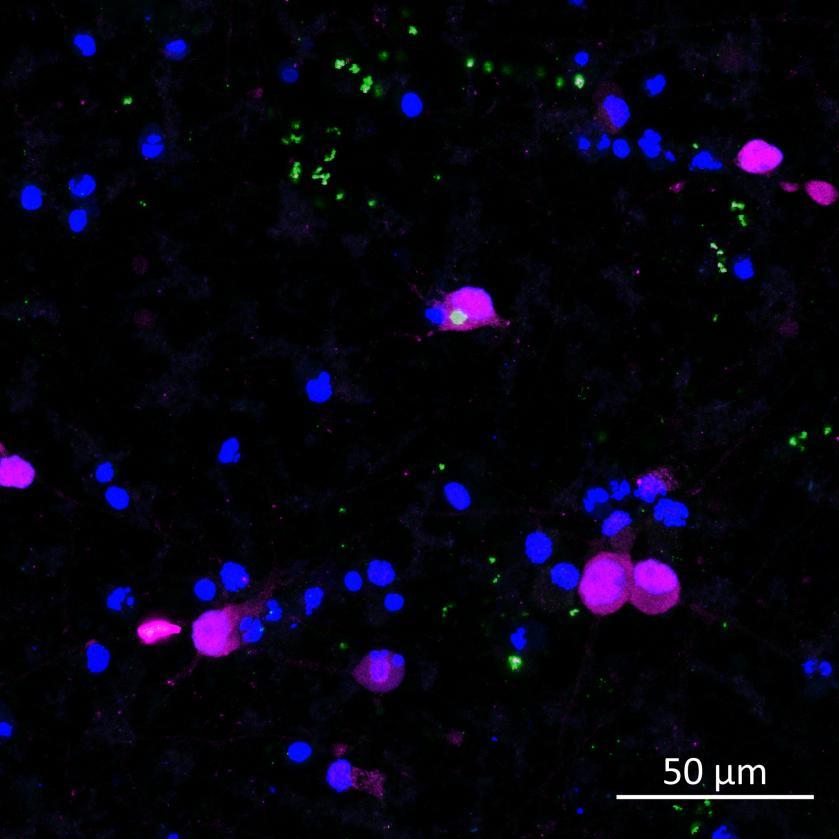
Phosphorylation and aggregation of native α-synuclein
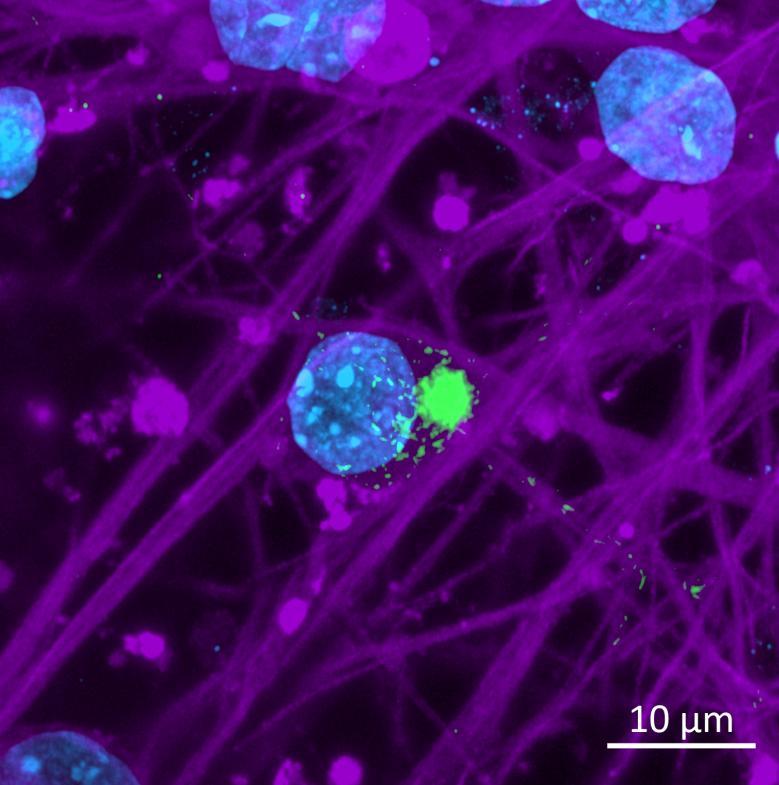
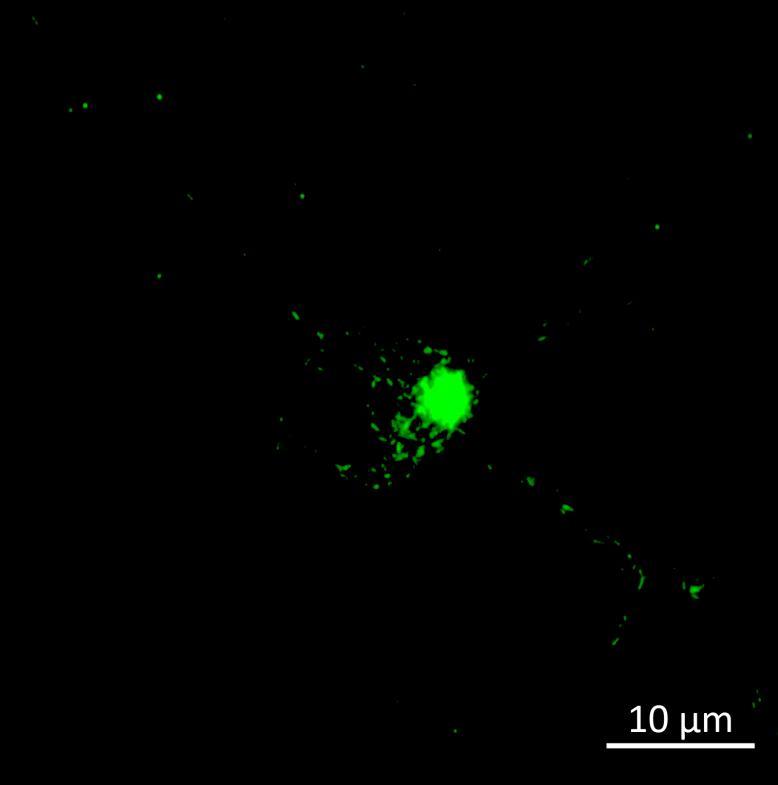
• Isogenic control and early onset Parkinson’s disease SNCA A53T mutation neurons were exposed to α-synuclein PFFs with A53T mutation
• Neurons were fixed and labelled for α-synuclein phosphorylated at serine 129 (pS129)
• PFF treated neurons show Lewy body-like perinuclear aggregates of pS129 α-synuclein
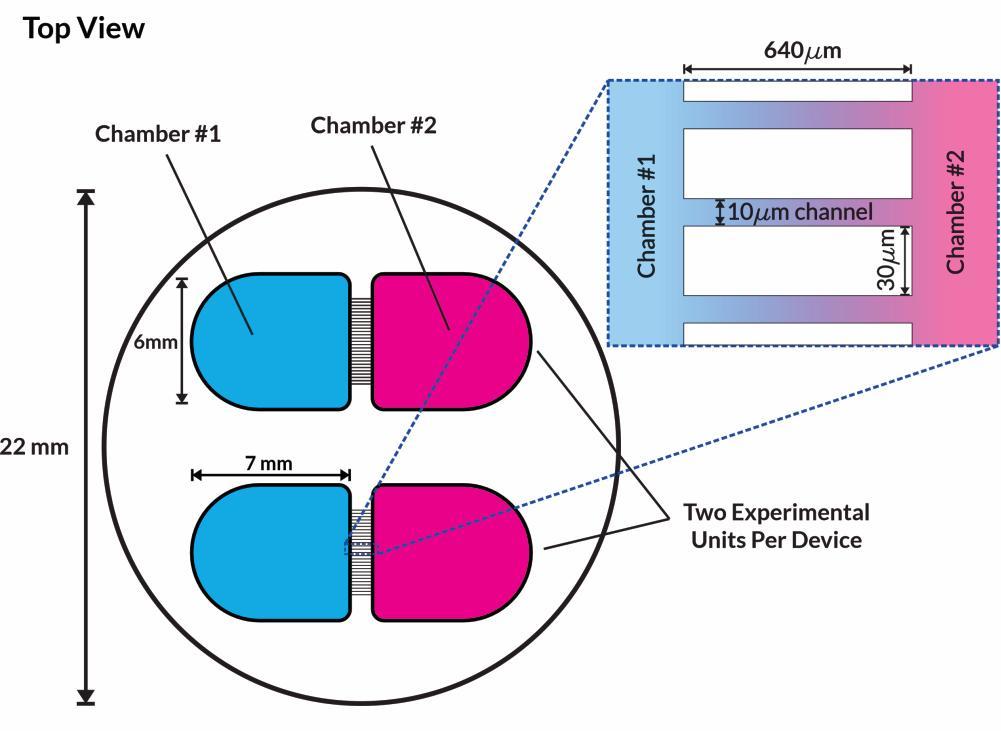
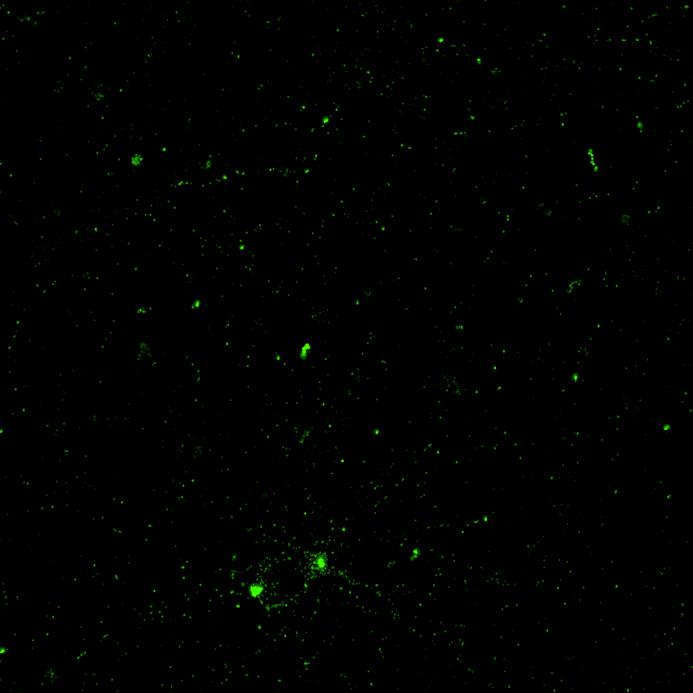
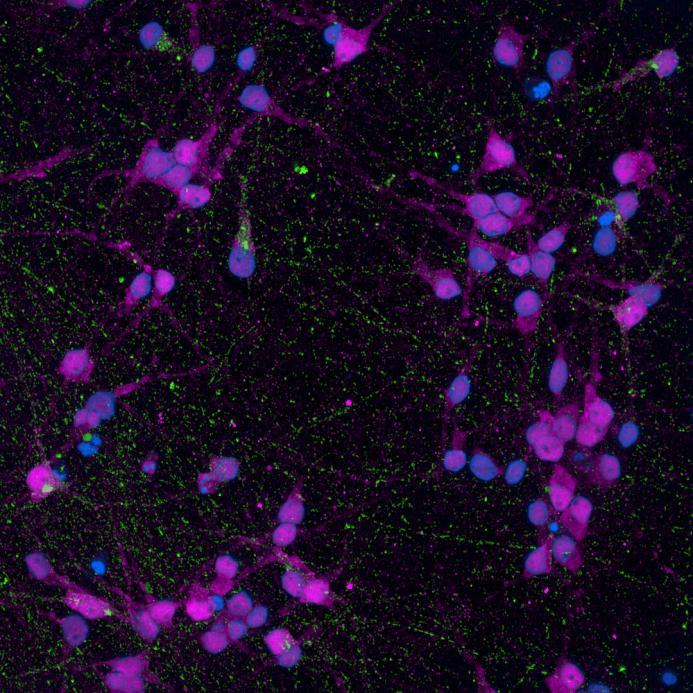
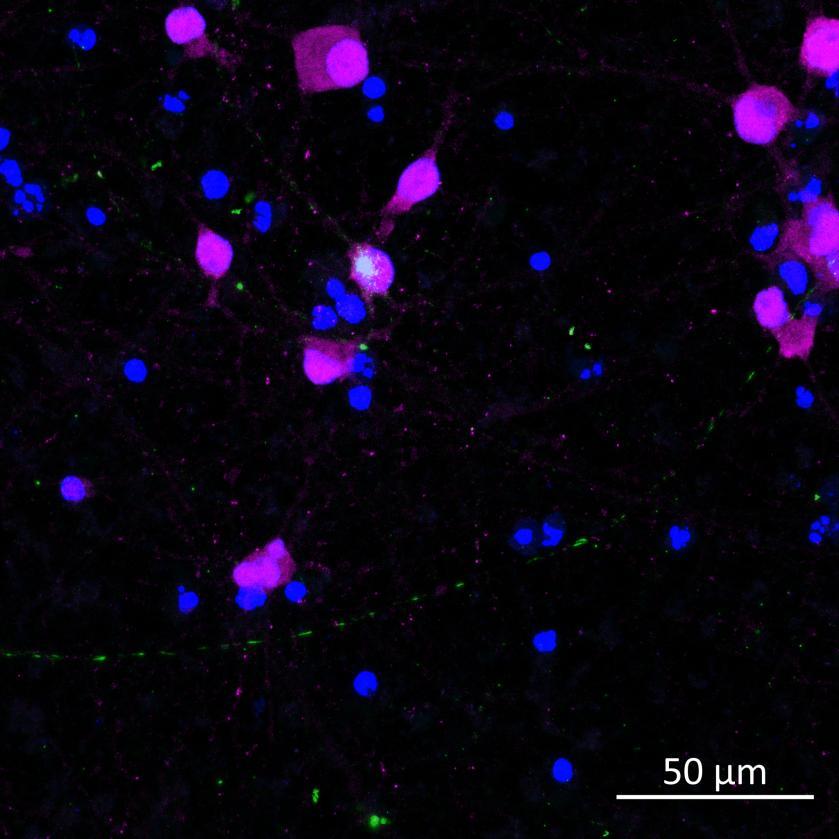
• Isogenic control or SNCA A53T mutation neurons were plated in both chambers of a microfluidics device then cultured to allow the neurons to connect via the microchannels
• PFFs were added to the donor chamber only before fixing and labelling for pS129 α-synuclein
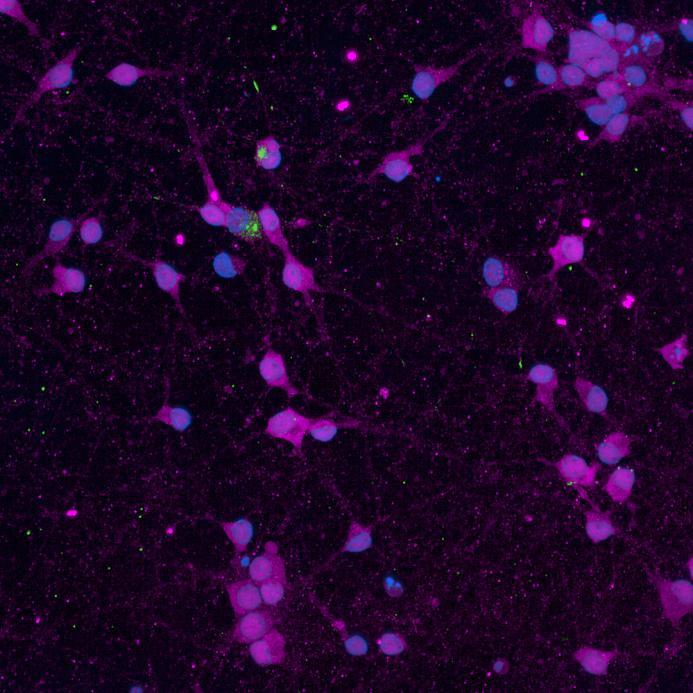
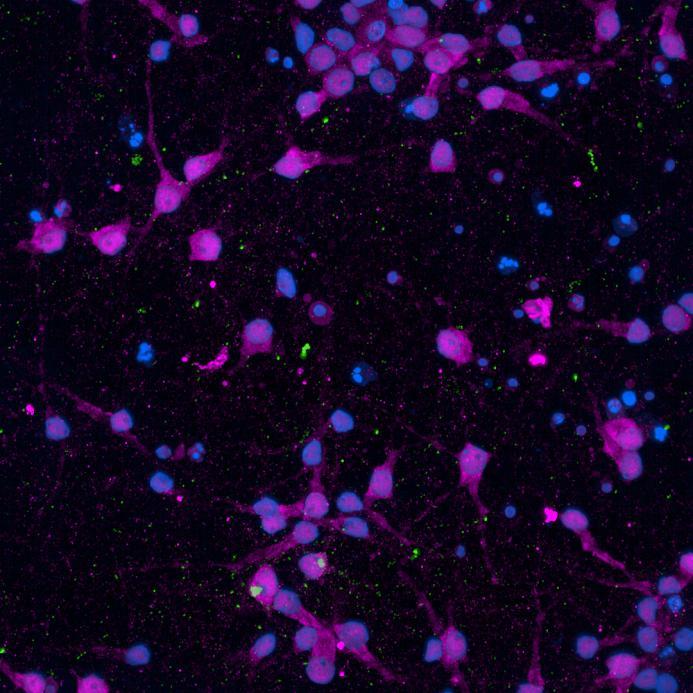
• pS129 positive neurons were detected in donor chambers due to directly applied PFFs and acceptor chambers due to seeded PFFs, though automated analysis failed to detect the small proportion of positive wildtype neurons in acceptor chambers
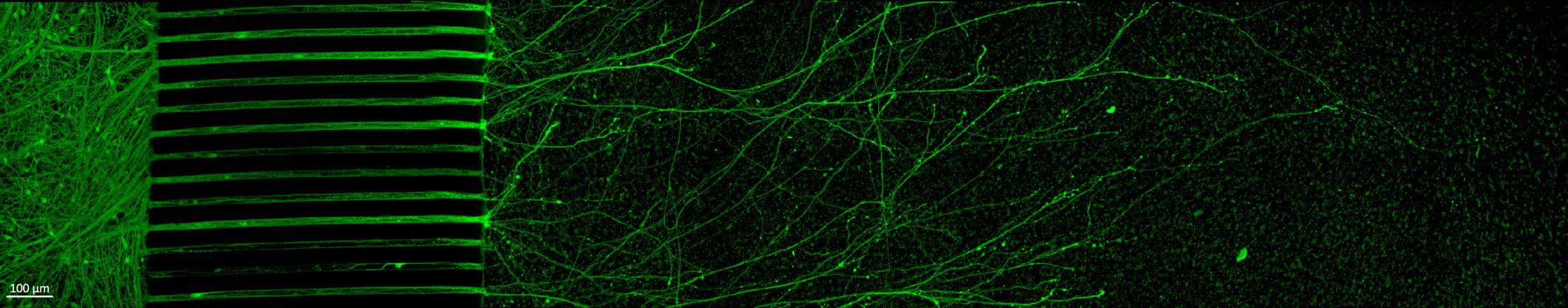
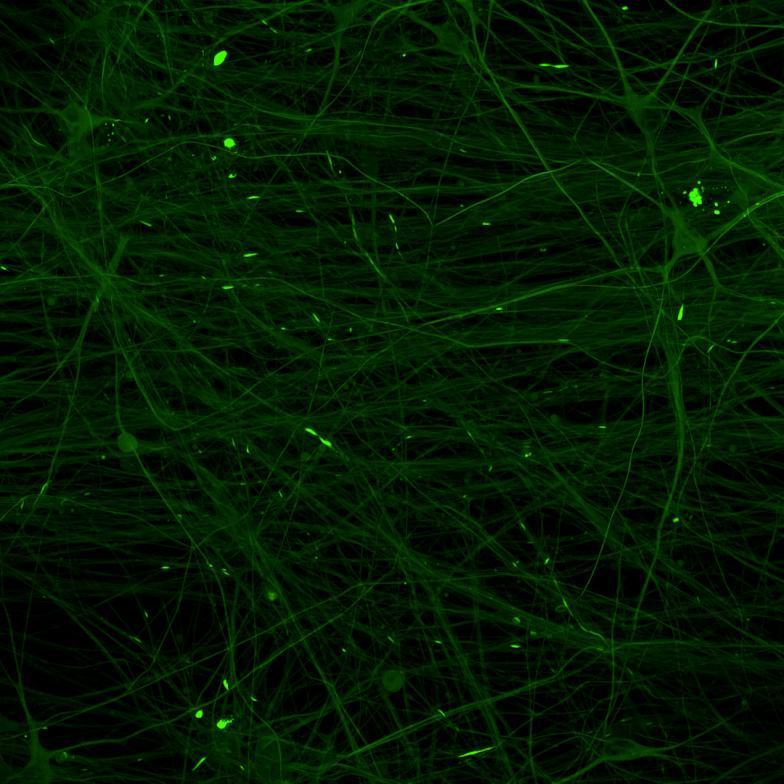
Projection of neurites through microchannels at 2 weeks
Conclusions



• Increasing over time, PFF exposure induced areas of bright EGFP-positive aggregates of full-length tau, and aggregation of the K18 fragment FRET pair as seen by positive normalised FRET (NFRET) signal
• K18 PFFs with P301L mutation induced aggregation more readily than full length 2N4R PFFs in both systems
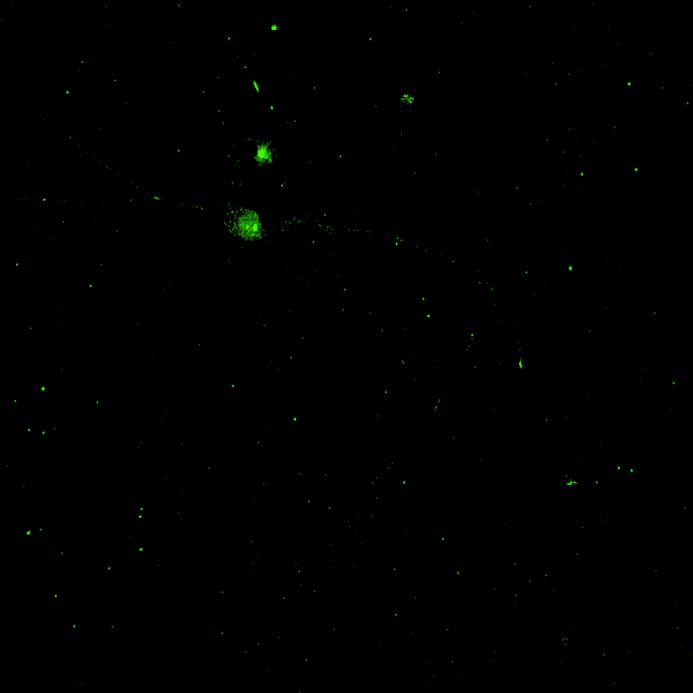
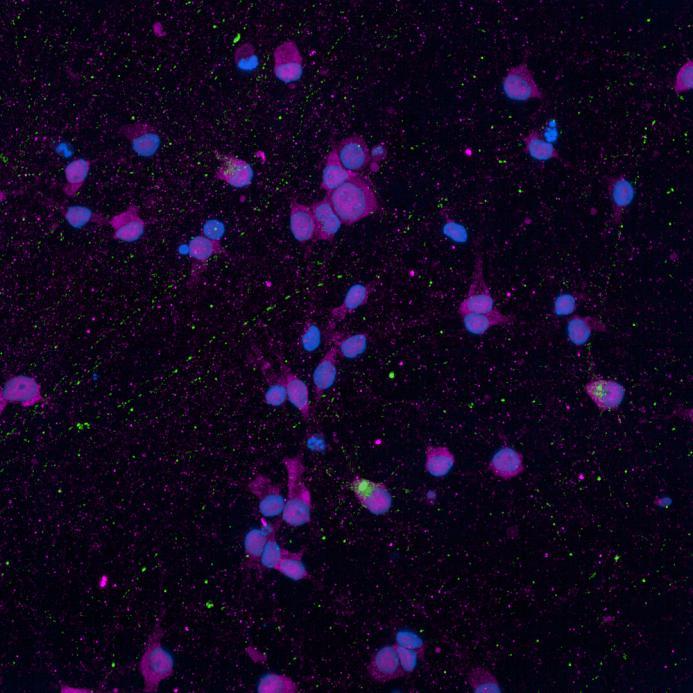
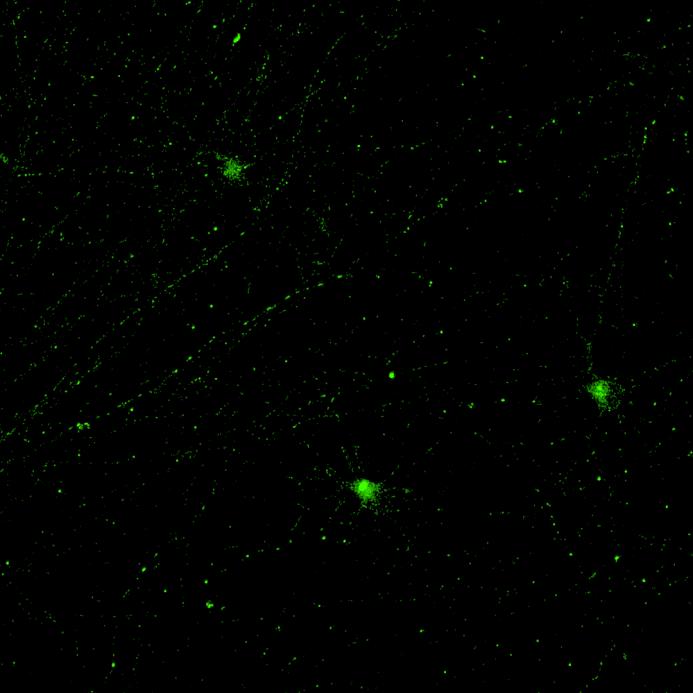
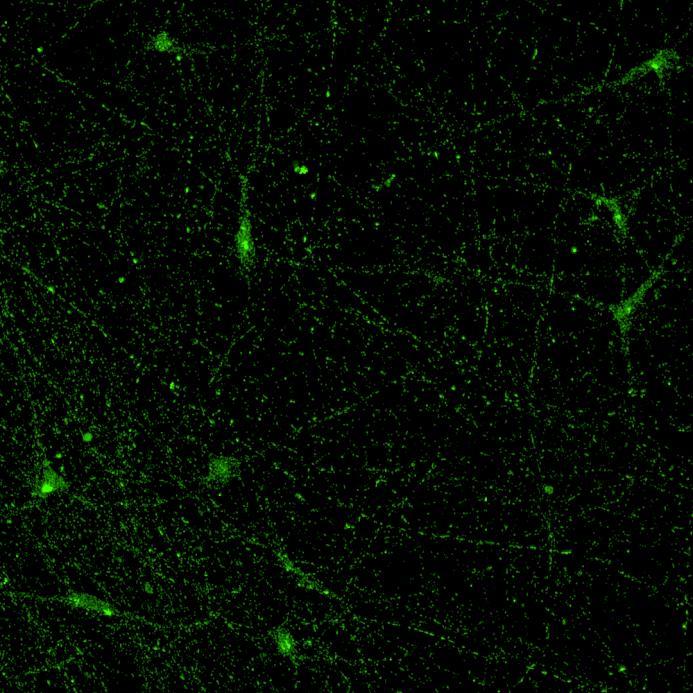
• A53T mutation increases the proportion of pS129 positive neurons after PFF exposure
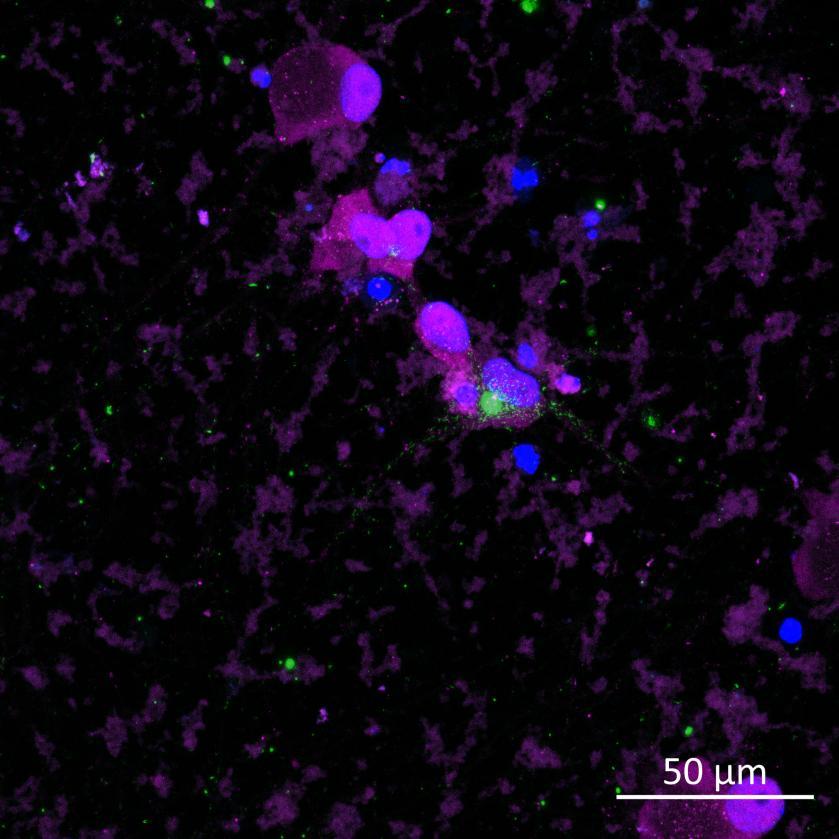
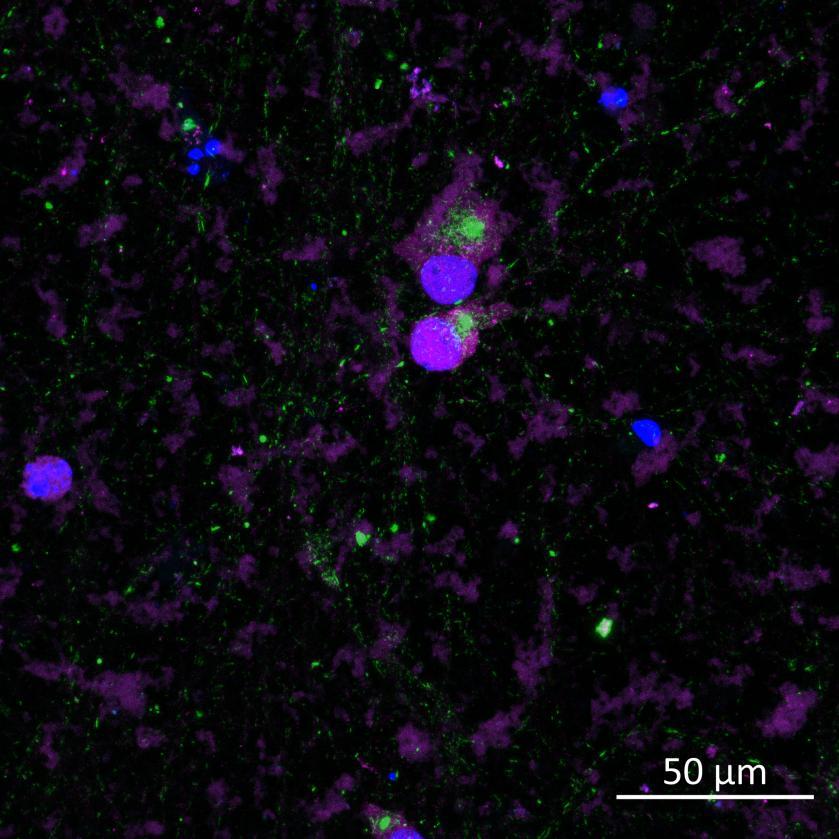
• High content imaging of human iPSC-derived neurons provides a platform to model the pathological seeded aggregation and phosphorylation
neurodegenerative proteins like tau and α-synuclein
• The ability to induce and detect disease-relevant protein
for therapeutic developers targeting protein misfolding, phosphorylation, and propagation
• In vitro assays quantifying tau and α-synuclein aggregation
pathology and support evaluation of mechanism-modifying interventions
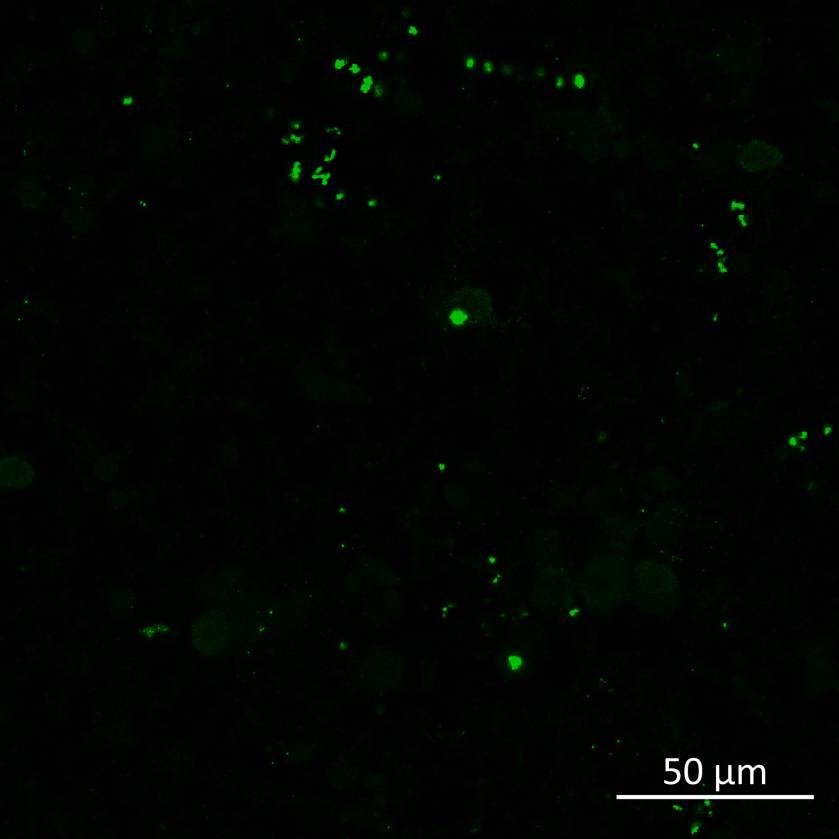
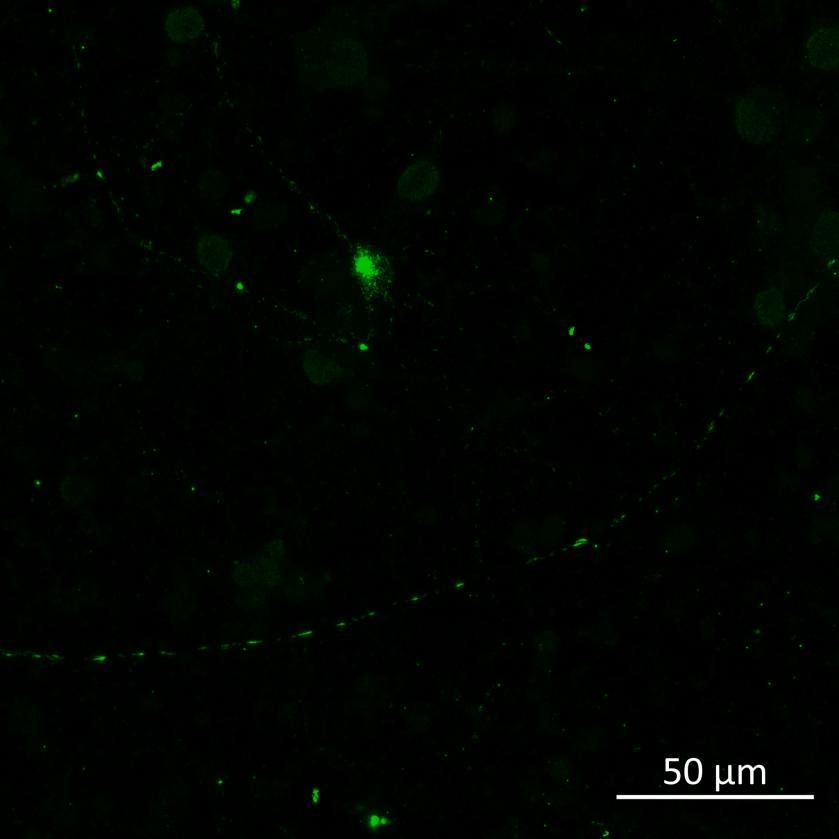
• A53T mutation increases pS129 pathology in both donor and acceptor chambers