
















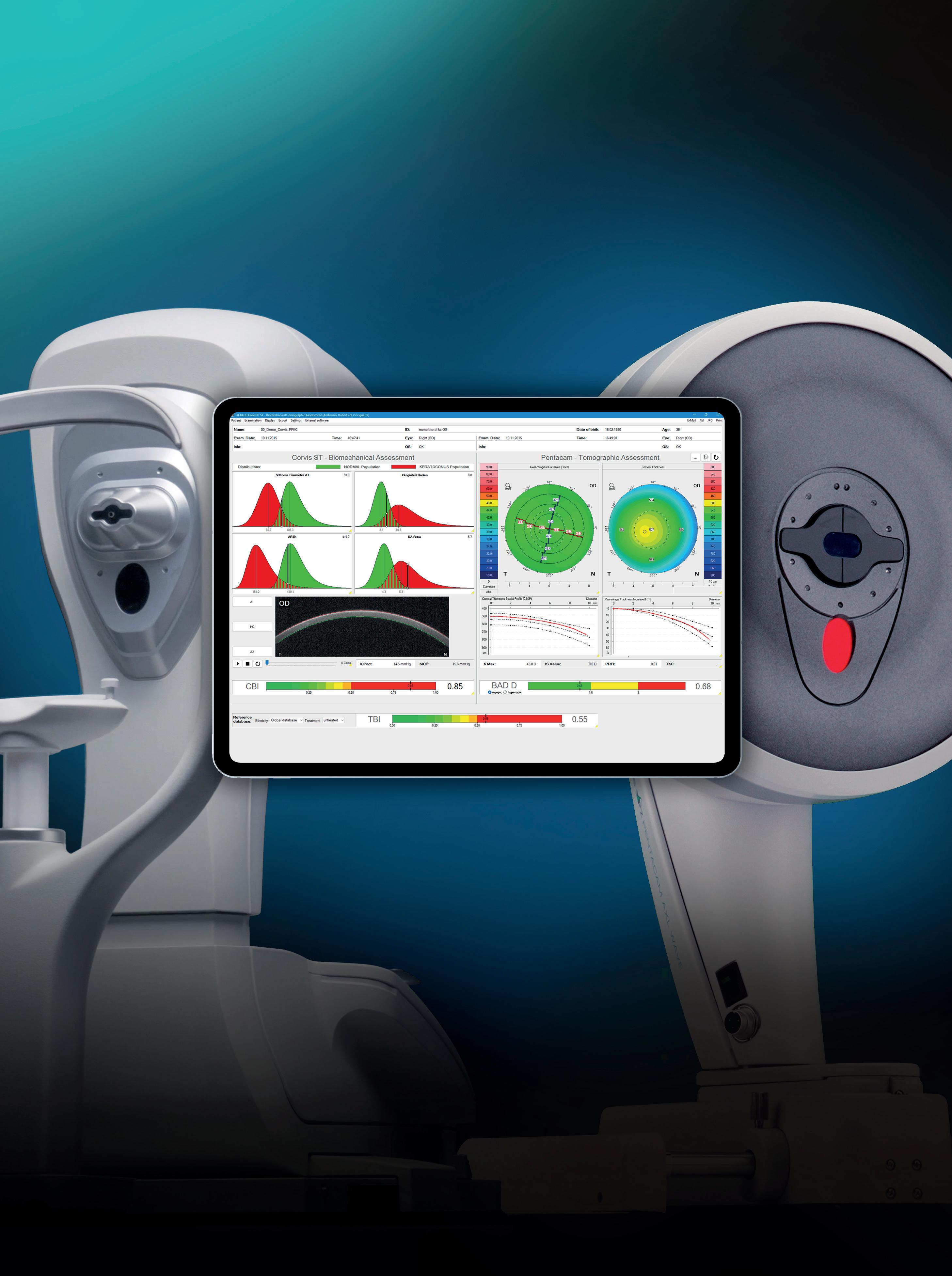
The Tomographic Biomechanical Index, or TBI, provides a unique combined expression of Corvis® ST und Pentacam® measurement data. It allows the risk of corneal ectasia to be assessed with greater reliability than ever before. The TBI assists you in selecting the optimal treatment based on sound reasoning.
More safety for you and your patients!


DR. HARVEY S. UY
University of the Philippines; Peregrine Eye and Laser Institute, Manila, Philippines harveyuy@gmail.com
DR. FRANCIS MAH
Scripps Clinic Medical Group La Jolla, California, USA Mah.Francis@scrippshealth.org

DR. WILLIAM B. TRATTLER
Center For Excellence In Eye Care Miami, Florida, USA wtrattler@gmail.com

DR. CATHLEEN MCCABE
The Eye Associates Sarasota, Florida, USA cmccabe13@hotmail.com
PROF. BURKHARD DICK
University Eye Hospital Bochum Bochum, Germany burkhard.Dick@kk-bochum.de

PROF. DR. SORCHA NÍ DHUBHGHAILL
Brussels University Hospital (UZ Brussel) Brussels, Belgium nidhubhs@gmail.com
Society Friends


We are looking for eye care professionals who can contribute articles to CAKE. Interested? Let's talk! Send us an email at editor@mediamice.com.
To place an advertisement, advertorial, symposium highlight, video, email blast, or other promotion in CAKE, contact sales@mediamice.com.
Matt Young CEO & Publisher
Gloria D. Gamat Chief Editor
Diana Truong Associate Editor
Nhat Vo
Raquel Jiménez
Marcelo Álvarez Graphic Designers
Andrés Pacheco Art Director
Chow Ee-Tan
Hazlin Hassan
Kendra Bruning
Marhiel Garrote
Michaela Perez Writers
Dr. Arun Gulani
Prateet Verma
Dr. Shilpa Ghosh Contributors
Matt Herman Head of Content Strategy
Hannah Nguyen COO
Travis Plage CFO
Ruchi Ranga
Society Relations & Conference Manager
Brandon Winkeler
Robert Anderson
Sven Mehlitz
International Business Development
A DISEASE FROM THE BRONZE AGE MEETS MODERN PUBLIC HEALTH, PROF. HUGH TAYLOR RECOUNTS THE LONG, MESSY AND SURPRISINGLY HUMAN CAMPAIGN TO WIPE TRACHOMA FROM AUSTRALIA IN OPENING EYES.

TWO TRACHOMA UNEXPECTED OPPORTUNITIES
At the end of my first year at Wilmer, I went to a Christmas party put on by the Immunology Laboratory people. Arthur (Art) Silverstein, head of the lab, chatted with me, asked about what I was doing, and what I had done before coming to Wilmer.
Art was one of the first immunologists to work full-time on eye issues, an unremarkable move now. He was interested in the work I had done with Fred and the NTEHP. He asked,
‘HOW EASY IS IT TO GET TRACHOMA?’
‘Oh, it’s so easy. All you have to do is be reinfected often.’
‘WOULD YOU LIKE TO SET UP AN ANIMAL MODEL OF TRACHOMA TO PROVE THAT?’
‘Why not? It would be easy.’
That easily-made agreement profoundly changed my work trajectory, my way ahead, my life.
The studies we did in this model led to the understanding of the importance of repeated
episodes of reinfection to lead to blinding trachoma. Later work would show maybe 200 episodes of reinfection were needed.
A few years later I was sitting quietly at a slit lamp in the dark counting microfilaria in the eye of a farm worker in the city of San Cristobal de las Casas, in Chiapas—the southernmost state of Mexico—just the two of us. Then, the voice of a third party, in English:
‘IS THERE SOMEONE HERE WHO KNOWS SOMETHING ABOUT TRACHOMA?’
What could I say?
‘Yes. That’d be me.’
The head of public health in Chiapas, Francisco Milan Velasco, had arrived in my life.
Thwarted in studying trachoma in Kenya, I was going ahead with studies on onchocerciasis. Between scheduled eye and skin examinations for that onchocerciasis study, we went to a community in the
highlands near San Cristobal and examined the whole community for trachoma.
One third of the younger children had active trachoma and about 12% of those over 40 had trichiasis, the in-turned lashes that rub on the front of the eye and lead to scuffing and blindness. A serious problem that needed to be addressed.
This led to showing the fundamental importance of facial cleanliness to stop the transmission of trachoma from one child to another. This has been a key component in the elimination of trachoma in many areas.

Prof. Hugh Taylor
President, Academia Ophthalmologica Internationalis (AOI).
Melbourne Laureate Professor Emeritus and Harold Mitchell Chair of Indigenous Eye Health, Melbourne School of Population and Global Health, at the University of Melbourne in Australia.
h.taylor@unimelb.edu.au


From static implants to responsive optics, cataract surgery is entering an era where vision doesn’t just get replaced—it gets refined.
By Dr. Shilpa Ghosh
For decades, cataract surgery has rested on a quiet gamble. Surgeons measure, calculate and plan with extraordinary precision. Patients trust that a few millimeters of acrylic placed inside the eye will restore clarity for a lifetime. And yet, after the incisions heal and the eye settles, there’s that uncomfortable question hovering in the background: Did we get it exactly right?
Sometimes, the answer is yes. Perfect distance vision, crisp contrast and no need for glasses. But other times, biology reminds us who’s really in charge. A minor refractive error. Residual astigmatism. A dependency on spectacles the patient hoped to leave behind.
For generations, these outcomes were accepted as inevitable. The eye, after all, is not a spreadsheet. It heals unpredictably, responds individually and rarely follows mathematical models to the decimal point. That assumption is now being challenged.
A new generation of “smart” intraocular lenses (IOLs) is transforming cataract surgery from a one-shot intervention into a dynamic process—one where vision can be adjusted, refined and optimized after surgery, based on real outcomes rather than predictions. In operating rooms and research labs around the world, the static lens is giving way to
something far more responsive. Not only is this better technology, it’s a fundamental shift in how we think about vision correction.
Traditional cataract surgery has always been about commitment. Once a lens is implanted, its power is locked in. Surgeons rely on biometry formulas, corneal measurements and experience to choose the “right” lens. But even with modern imaging and AI-enhanced calculations, refractive surprises remain part of the landscape. That’s where adjustable IOLs change the rules.
Rather than demanding perfection upfront, these lenses allow surgeons to fine-tune vision once the eye has healed. The most established example is the light adjustable lens (LAL), which uses photosensitive materials to modify lens power postoperatively.
After implantation and healing, patients undergo detailed refraction, just like a glasses test. If vision isn’t exactly where it needs to be, the surgeon applies a carefully calibrated ultraviolet (UV) light treatment in the clinic. The light triggers microscopic changes within the lens material, permanently altering its curvature and optical power. The result? Vision tailored not to theoretical models, but to the eye’s actual behavior.
Clinical outcomes have been impressive. A high percentage of patients achieve refractive accuracy within half a diopter of their target, often outperforming even premium toric and multifocal lenses.1,2 For patients with prior LASIK or PRK—eyes notorious for defying standard calculations—the LAL has been nothing short of revolutionary. For the first time, refractive accuracy is no longer a gamble. It’s a process.
WHEN HEAT BECOMES A TOOL
Light isn’t the only path to adjustability. Another branch of innovation has explored thermally responsive lenses, most notably through designs incorporating shape-memory alloys.
One such concept uses nitinol, a nickeltitanium alloy famous for its ability to “remember” a specific shape. When heated to a precise temperature, nitinol returns to its programmed configuration. Applied to IOLs, this property allows lens position, and therefore optical power, to change in response to controlled heating.
In theory, a surgeon could adjust vision in the clinic using a handheld device that delivers localized warmth. No incisions. No additional surgery. Just physics doing the heavy lifting. The appeal is obvious. Thermal
systems could potentially allow repeated adjustments over time, accommodating not only early postoperative changes but longterm shifts as patients age. Unlike light-based systems, they wouldn’t require strict UV protection protocols. However, translating elegant engineering into clinical reality is rarely straightforward. While early studies showed promise 3 , thermally adjustable lenses have faced developmental and regulatory hurdles. They remain largely investigational— an example of how innovation must survive not only scientific scrutiny but economic and logistical realities as well.
Still, the concept underscores the fact that adjustability is no longer a curiosity. It’s a design priority.
If light and heat represent today’s breakthroughs, fluidic and sensor-based lenses hint at tomorrow’s ambitions. Researchers are exploring lenses with microfluidic chambers that can subtly change shape by redistributing fluid, altering focus in response to external signals or potentially internal ones. Some designs envision lenses that could be adjusted multiple times over a patient’s lifetime. Others imagine truly adaptive optics that respond continuously rather than episodically. The challenges are immense. Any device inside the eye must remain crystal clear, biologically inert, mechanically stable and reliable for decades. Add moving parts or electronics, and the margin for error narrows dramatically. Yet progress in materials science, miniaturization and bioelectronics suggests these ideas aren’t fantasy. They’re simply early.
The findings even offer a timely Valentine’s Day takeaway. Gifting a plant benefits both home air quality and eye health, making it a thoughtful gesture that quite literally shows you care.
For surgeons, smart IOLs represent not only another premium option; they signal a philosophical shift. Historically, premium cataract surgery has demanded meticulous planning and extensive counselling about limitations. Even the best surgeons accept that a certain percentage of patients will need enhancements or compromises.
Adjustable lenses change that conversation. Instead of promising perfection, surgeons can promise optimization. Vision becomes something that can be measured, refined and finalized—rather than hoped for. This has practical consequences. Eyes once considered “high-risk”—post-refractive surgery patients, unusual corneas and extreme axial lengths—become more approachable. The pressure to achieve a flawless outcome on day one eases when refinement is built into the system. At the same time, practices must adapt. Adjustable IOLs introduce new workflows, equipment and patient education requirements. Cataract surgery becomes a staged journey rather than a single event. The economics shift as well, with premium pricing demanding clear communication of value. But for many surgeons, the trade-off is worth it: greater consistency, happier patients and fewer surgical revisions.
Despite their promise, smart IOLs aren’t magic. Most current adjustable lenses address refractive accuracy and not accommodation. Patients may still need reading glasses. Multifocal and extended-depth-of-focus designs are evolving, but they retain familiar trade-offs like halos or reduced contrast. Long-term data, while encouraging, is still maturing. Questions about how adjustable materials behave over decades remain under study. As with any innovation, time is the final examiner. And cost remains a barrier. Adjustable IOLs are premium technologies, accessible primarily to patients who can afford out-of-pocket expenses. Broader access will depend on competition, manufacturing scale and evolving reimbursement models.
Perhaps the most i mportant change smart IOLs bring is conceptual. Traditional lenses replace a cloudy structure with a fixed substitute. Adjustable lenses form a partnership with the eye—one that acknowledges biological variability and adapts to it. This mirrors a broader trend in medicine toward personalization. Devices are no longer static solutions; they’re evolving systems designed to work with individual biology rather than override it. The future “smart” IOL may not only adjust focus, but monitor eye health, track pressure or interface with diagnostic
tools. While those possibilities remain on the horizon, the direction is clear.
Smart IOLs won’t eliminate uncertainty entirely—eyes are too complex for absolutes. But they dramatically narrow the gap between intention and outcome. They turn refractive surprise into refinement. They replace resignation with responsiveness. They move cataract surgery from prediction to personalization.For surgeons, they offer control. For patients, confidence. For ophthalmology, a glimpse of a future where vision correction is no longer fixed but fluid, thoughtful and precise. The rise of smart IOLs isn’t just about seeing better. It’s about seeing differently—about what’s possible when technology finally adapts to the human eye, rather than asking the eye to adapt to technology. And in that clarity, the future looks remarkably bright.
1. Doane J, Newsom TH, Slade S, Thompson V, Bruns N, Vukich J. Clinical data registry comparing outcomes of two light adjustable lenses. J Cataract Refract Surg. 2025 Nov 1;51(11):948-954.
2. Yadav S, Emara B. Assessment of the LightAdjustable Intraocular Lens in cataract surgery: A systematic review. Presented at: 2025 ASCRS Annual Meeting; April 25-28, 2025; Los Angeles, CA.
3. Lapid-Gortzak R, Mitsel P, Shmukler R, et al. New nitinol-based thermomechanically adjustable intraocular lens (IOL) technology. J Refract Surg. 2023;39(10):662-667.


Dr. Shilpa Ghosh Cataract, cornea and anterior segment surgeon at Apeksha Hospital, Dibrugarh Assam University College of Medical Sciences in New Delhi, India.
ophtha.shilpaghosh.2020@gmail.com



As premium IOL options multiply, AECOS is betting that better language will lead to better choices and fewer postoperative surprises.
By Kendra Bruning
In 2019, during the 37th Congress of the European Society of Cataract and Refractive Surgeons (ESCRS) in Paris, Dr. Sheraz Daya (United Kingdom) was asked a question he kept hearing more often: which extendeddepth-of-focus (EDOF) lens he preferred.
What stuck with him was not the question itself, but how loosely the label was being used. As Dr. Daya recalled, “It was quite obvious that they did not fully understand the concept of EDOF and the fact that there were several mechanisms which achieved the same outcome of providing intermediate vision.” Since then, that corridor moment has only become more relevant. The premium intraocular lens (IOL) ecosystem is now crowded with expanded monofocal designs, non-diffractive optics, diffractive trifocals, small-aperture concepts and a steady stream of “reinventions” that arrive pre-labeled by marketing.
Surgeons are left translating a growing vocabulary into something patients can actually use when deciding how they want to see and what tradeoffs they are willing to tolerate.
Paul Dupeyre, an optometrist and research associate at Centre Ophtalmologique Iridis in France, sees beneath the product boom to identify a fundamental communication problem. “The main failure points stem from marketing-driven ambiguity in IOL terminology and the lack of standardized, patient-centered evaluation methods,” he said. The issue isn’t lack of data but its comparability. With protocols varying and performance dependent on individual factors, a single marketing label often substitutes for the nuanced conversation patients need. 1,2
The American-European Congress of Ophthalmic Surgery (AECOS) began building its categorization framework from first principles. The aim was to separate what an IOL does from how it does it, with language tested directly with patients to create not just a classification system but a communication philosophy.
“Good nomenclature should be simple, stable with a uniform set of rules with room for additions and changes and, most importantly, should be understandable by all stakeholders, principally patients,” Dr. Daya wrote.
That patient-facing emphasis is the framework’s differentiator. AECOS intentionally leans toward optimistic, functional language. Terms such as “range of focus” landed better than “range of vision” or “field,” which patients may interpret as peripheral vision.3 The goal is not to oversell, but to speak in the units patients actually live in, like dashboards, screens, books and nighttime roads.
Surgeons are not short on classification attempts. Optical design labels remain common but broad. 4 ESCRS has proposed an evidence-based functional classification built from literature analysis and defocuscurve patterns, dividing lenses into partial versus full range of field categories, with further subgrouping.5,6 AECOS aims for a shared language that works in clinic and in the real conversations surgeons have with patients and each other. 7 The “EDOF” label is the clearest example of why this matters. It has become a marketing umbrella that can mask distinct mechanisms, each with different visual phenomena. AECOS addresses this by making dysphotopsias a core descriptor alongside distinct performance and mechanism classifications. 4,7
AECOS organizes premium IOLs across three axes: performance, mechanism of action and dysphotopsia profile. 7
In practice, performance is the functional range patients care about, so AECOS favors plain range-of-focus language such as increased range of focus or full range of focus instead of assuming “EDOF” tells the whole story. 7 Mechanism matters because it is why two lenses with similar category labels can behave differently once they meet a real cornea and pupil.1 Dysphotopsias are treated as a defining feature, not a footnote, because they can drive satisfaction more than acuity
alone. 4 A surgeon might say: “This lens is built to broaden your range of focus, so you are more likely to handle screens and the dashboard without glasses. The design that creates that range can also bring some nighttime effects like halos or glare. If night driving is your top priority, we can choose an option with a quieter night profile, but you may have to trade a bit of near.” The conversation is familiar. The difference is that the tradeoffs are explicit.
Patients rarely ask whether an optic is diffractive, refractive or small-aperture. Surgeons still need to ask, because the mechanism is the part that actually meets the eye’s optics. Dr. Daya would not recommend the Rayner (Worthing, United Kingdom) EMV lens in an eye with a high level of positive corneal spherical aberration, because “the mechanism of action of this lens is by increasing positive spherical aberration.” When that effect stacks on top of a cornea that is already pushing peripheral rays out of alignment, quality can drop. In practice, the result can be more halos and less contrast sensitivity, especially in low light when pupils dilate.1,8
The point is not that one design is “bad.” It is that the mechanism is not neutral. A category framework does not replace biometry. It helps surgeons compare like with like, then choose with the patient’s anatomy and priorities in mind.4
AECOS argues that labels should reflect odds a patient can understand. Dr. Daya noted that the American National Standards Institute (ANSI) standard for EDOF labeling can be permissive, requiring only 50% of eyes to achieve 0.2 logMAR (20/32) at 67 cm when corrected for distance. AECOS sets a higher bar for increased range of focus, recommending 80% achieve 0.2 logMAR at 67 cm distance-corrected. That shift matters because it changes how confidently a surgeon can frame expected intermediate performance.

Dysphotopsias benefit from the same discipline. In Dr. Daya’s clinical experience and his own patient-reported outcomes tracking, approximately 10% of patients report significant halos early postoperatively, declining to roughly 5% by three months, and these symptoms are usually not severe enough to warrant exchange. Broader literature shows why definitions matter. In a 2025 metaanalysis of patient-reported outcomes with a trifocal IOL, 43.9% of patients reported halos of any severity, while 5.4% reported severe halos and 1.4% described halos as “very bothersome”.9 The numbers become clinically useful when surgeons and patients share definitions, including severity, timing and what “bothersome” means to the patient sitting in the chair.
Dupeyre and his colleague, Dr. Gilles Lesieur, have argued for an “IOL identity card,” a standardized profile that would bundle classification language with functional metrics. “The concept of an IOL identity card would allow surgeons to easily visualize standardized classification information together with key functional metrics,” they said.
This would create a standardized snapshot pairing AECOS classifications with functional metrics in a format supporting both clinical decision-making and patient education. 4,5 The promise is less hype and more comparability, especially as presbyopiacorrecting lenses expand beyond the traditional multifocal versus monofocal split. 2
In the premium IOL world, language is the scaffolding that holds expectations in place. AECOS treats nomenclature like a clinic tool. It’s simple enough to use between patients, structured enough to keep tradeoffs visible, and clear enough to support shared decision-making without turning consent into a lecture. The AECOS framework will not become the norm by committee. It will gain traction when it saves time, reduces misinterpretation and makes the tradeoffs easier to say out loud. The best categorization system is one that helps surgeons say what they mean, with fewer words and fewer surprises.
1. Miret JJ, Camps VJ, García C, et al. Analysis of the effect of pupil size and corneal aberration on the optical performance of premium intraocular lenses. J Clin Med. 2025;14(15):5336.
2. Park CY. Factors affecting postoperative satisfaction after presbyopia-correcting intraocular lens. J Clin Med. 2026;15(1):336.
3. Riaz KM, Wendelstein JA, Koch DD. Depth of field or depth of focus? J Cataract Refract Surg. 2024;50(12):1291-1292.
4. Lesieur G, Dupeyre P. Bringing clarity to IOL selection [Internet]. CRSToday. 2025 Oct. Available from: https://crstoday.com/articles/oct-2025/bringing-clarity-to-iol-selection
5. Gaspari L. Towards a unified IOL classification. EuroTimes (ESCRS). 2025 Jul 1. Available from: https://www.escrs.org/channels/eurotimes-articles/towards-a-unified-iolclassification.
6. Ribeiro F, Dick HB, Kohnen T, et al. Evidence-based functional classification of simultaneous vision intraocular lenses: seeking a global consensus by the ESCRS Functional Vision Working Group. J Cataract Refract Surg. 2024;50(8):794-798.
7. Daya SM. Let’s speak lenspeak: A discussion on modern IOL terminology. CRST Global. 2021 Feb. Available from: https://crstodayeurope.com/articles/feb-2021/lets-speaklenspeak/.
8. Kohnen T, Kasper T. At what pupillary size does an aspheric matter? CRST Global. 2007 Oct. Available from: https://crstodayeurope.com/articles/2007-oct/1007_15-php/.
9. Zhu D, Karki S, Dhariwal M, et al. Patient-reported outcomes of visual disturbances with a trifocal intraocular lens: a meta-analysis. Ophthalmol Ther. 2025;14(2):379-90.
CONTRIBUTOR

Dr. Paul Dupeyre
Clinical optometrist and Research Associate in ophthalmic optics at the Iridis Center in Albi, France. He is the co-founder of IOL Match, where he leverages his expertise in optics to help democratize clinical decision-support tools for lens selection.
p.dupeyre@iridis-robotics.fr
CONTRIBUTOR

Dr. Gilles Lesieur
Leading cataract and refractive surgeon based at the Iridis Center in Albi, France. He specializes in presbyopia surgery and is the cofounder of IOL Match, a free, non-commercial clinical decisionsupport application.
g.lesieur@iridis-robotics.fr
CONTRIBUTOR

Dr. Sheraz Daya
Internationally recognized leader in ophthalmology and the President of the American-European Congress of Ophthalmic Surgery (AECOS). He consistently ranks among the world’s top 100 most influential ophthalmologists.
sdaya@centreforsight.com





At the 41st Asia-Pacific Academy of Ophthalmology Congress (APAO 2026), Carl Zeiss Meditec (Jena, Germany) hosted a packed lunch symposium titled Presbyopia Today: Innovation and Patient Care.
With presbyopia numbers climbing and patient expectations rising in equal measure, the session offered a timely deep dive into contemporary management strategies, from laser vision correction to advanced intraocular lens (IOL) platforms and artificial intelligence (AI)assisted diagnostics.
For busy anterior segment surgeons, it was a concise tour of where the industry is and where it’s headed next.
PRESBYOPIA
Opening the session, Prof. Bai Ji (China) underscored the scale of the challenge. “By 2030, the global population with presbyopia will exceed 2.3 billion. This is not only a medical problem but also a social issue,” he said.

Presbyopia gets more precise with laser tweaks, AI checks and trifocals that behave.
Sponsored by ZEISS
Prof. Bai shared his experience with ZEISS PRESBYOND®, a binocular laser vision correction approach for presbyopes without cataract. Unlike earlier monovision strategies—often associated with reduced binocular quality when increasing anisometropia—ZEISS PRESBYOND® applies a blended vision concept.
“In the beginning, we used laser refractive surgery with minimonovision,” Prof. Bai explained. “Then we improved this skill and did not induce spherical aberration. Now we can give patients both eyes set up with blended vision.”
Clinical outcomes presented during the session demonstrated high patient satisfaction compared to other approaches. Prof. Bai cited a notable example, saying “Prof. Dan [Reinstein] performed ZEISS PRESBYOND® on 23 UK pilots who subsequently passed their Class 1 medical certification and returned to flying.”1
In the said study1, there’s three groups, reported Prof. Bai: “One is [EDoF without introduction]. The other one is the [EDoF with

monocular introduction]. And finally, we also chose the [EDoF with binocular introduction]. The safety outcome reported is good…ZEISS PRESBYOND®, compared with the other groups, is the best. The satisfaction is good.”
For surgeons considering adoption, Prof. Bai outlined several key success factors:
• Patient education: “After operation, there is an adaptation period. You must tell the patient what to expect.”
• Understanding visual demands: “You must know a patient’s work distance. Maybe some people just read books; maybe some people use computers and mobile phones.”
• Preoperative tolerance testing: “For ZEISS PRESBYOND®, you must check the patient’s tolerance for monovision and how much they can tolerate.”
A doctor who uses ZEISS PRESBYOND® (and has it in his own eyes) attending the symposium offered a practical perspective: “3D vision is good. I can play sports like normal. One of the things people complain about with monovision is loss of depth perception, especially when playing badminton or tennis. We don’t get that with ZEISS PRESBYOND®.” He added, “I can watch 3D movies, like Avatar, with no problem—that’s the biggest test for stereopsis.”
For surgeons wary of depth perception trade-offs, that may be the most practical endpoint of all.
SMARTER PREOPERATIVE SCREENING
Assoc. Prof. Gavin Tan (Singapore) turned the focus posteriorly, discussing AI integration into optical coherence tomography (OCT) imaging for preoperative assessment, focusing on the CIRRUS® PathFinder platform.
“We know there are demographic and technological changes that are changing how we need to manage eye care,” Prof. Tan said. “We have an increasing population, increasing prevalence of visual impairment and more workload for everyone.”
Relying solely on clinical examination is not sufficient. “Up to one in 10 subjects with seemingly normal maculae will have pathology found on OCT that was not identified in clinical examination—pathologies that can affect your choice of IOL,” Prof. Tan noted.
ZEISS CIRRUS PathFinder fully integrates AI-decision support into the device’s standard workflow, providing instant results at point of care. With 88% sensitivity and 93% specificity, it was validated and trained by leading retina specialists.
Prof. Tan outlined the workflow: “You acquire your normal macular cube. The device then does a quality check to ensure image quality is sufficient. If there’s poor image quality, those scans are labeled yellow. The system then detects whether there’s any pathology in each B-scan of interest, which is clearly labeled in red.”
The algorithm was trained on more than 75,000 B-scans across over 60 macular pathologies. Case examples demonstrated detection of subtle findings—including peripheral pigment epithelial detachments (PEDs), non-center-involving diabetic macular edema (DME), and epiretinal membranes—that could influence IOL selection.
“PathFinder can immediately provide accurate identification of peripheral B-scans with pathology, allowing physicians to focus on pathological scans. This improves physician efficiency, helps avoid missed pathology and optimizes patient outcomes,” Prof. Tan concluded.
TRIFOCAL ENTERS THE ARENA
Dr. Florian Kretz (Germany) presented three-month outcomes from a multicentric prospective study of the AT ELANA® 841P, a new hydrophobic trifocal IOL built on the CT LUCIA™ platform.
The lens incorporates “a full diffractive trifocal surface with an equal light distribution—47.5% for distance, 35% for near and 17.5% for intermediate,” said Dr. Kretz.
He detailed key design features. “The AT ELANA, like the CT LUCIA™ IOL, is a C-loop lens with a 360-degree sharp edge to minimize PCO,” he stated. “There is no real angulation of the haptics in regard to the optics, but they are slightly lifted, so the optical posterior surface is pressed to the posterior capsule to prevent PCO [posterior capsule opacification].”
At three months postoperatively, refractive and visual outcomes were strong. “All patients were within plus or minus half a diopter of sphere,” Dr. Kretz reported. “For monocular distance-corrected visual acuity, we reached 0.01 logMAR. Our intermediate at 66 cm was 0.07 logMAR, at 80 cm 0.08 and near was 0.14 logMAR.”2
The defocus curve demonstrated “a very smooth transition from intermediate to near, giving a large range of focus from +0.0 to -2.5, -3.0 diopters.”
Visual disturbances were reported at low rates. Over 90% of patients were satisfied with their vision at 3 months, and almost 90% reported seeing well without glasses or any other vision-correcting devices.2
“Most patients reported little or somewhat bothersome visual symptoms, but the percentages are very, very small,” Dr. Kretz noted. “Especially hazy vision and blurred vision—things that can be very bothersome for patients—were not really an issue in these patients.”
He concluded that “the AT ELANA hydrophobic trifocal IOL provides good distance, intermediate and near visual acuity with a defocus curve demonstrating VA better than 0.2 logMAR at the 3-month follow-up visit, achieving high patient satisfaction and spectacle independence.”
Prof. Zhu Xiangjia (China) addressed long-term stability of the AT LISA® tri family, with a focus on highly myopic eyes.
“Nowadays, refractive cataract surgery has become the mainstream procedure, and patients’ needs extend beyond vision recovery to encompass higher standards of visual quality,” Prof. Zhu said. “This has led to heightened demands for long-term stability of intraocular lenses.”
She noted that multifocal performance is particularly sensitive to lens position. “The greater complexity of the IOL design, the more susceptible its optical performance becomes to decentration.”3
Data from her team showed that “high myopic eyes have a larger capsular bag. The equatorial diameter is around 11.22 mm in myopic eyes, compared to smaller ones in emmetropic eyes.”⁴
Comparative analysis revealed that “with the increase of axial length, eyes implanted with C-loop IOLs showed increasing vertical and overall decentration. While in those implanted with plate-haptic IOLs, the vertical and overall decentration remained unchanged.”⁵
The AT LISA tri plate-haptic design demonstrated “better stability than C-loop multifocal IOLs in high myopia.” Optical quality metrics also favored the plate-haptic group. “The total and internal aberration in eyes implanted with plate-haptic IOLs was less than in C-loop eyes, and MTF was better in eyes implanted with plate-haptic IOLs.”
In a multicenter study, “plate-haptic multifocal IOL application in high myopia patients with axial length less than 28 mm showed good results with no significant difference compared to emmetropic eyes.”⁶
However, Prof. Zhu advised caution in extreme cases. “Application of these lenses in extreme myopia with axial length greater than 28 mm should be approached with caution.”
On patient selection, she emphasized comprehensive evaluation. “I usually check the cornea, astigmatism and fundus condition. Not only
the macula—we also have to care about the periphery for any laceration or tear. We need to treat fundus pathology before surgery.”
Prof. Zhu added that she generally does not recommend implanting trifocal IOLs for patients with AL of over 30 mm. For those with AL over 33 mm, she may even avoid implanting any IOL at all. “Highly myopic eyes are prone to capsular contraction after surgery, especially eyes with longer AL,” she noted. “They have micro-inflammation, so they are prone to contraction, which in the long term will cause a hyperopic shift.”
Session moderator Dr. Li Lian Foo (Singapore) summarized the integrated approach: “We started with what to do when patients have presbyopia—we need to do an OCT scan with PathFinder. For available solutions, we have ZEISS PRESBYOND® for young presbyopes, the C-loop trifocal lens AT ELANA for post-cataract presbyopia correction, and the AT LISA plate-haptic trifocal lens, especially useful for high myopes.”
Across all presentations, a consistent theme emerged: success in presbyopia management hinges on careful patient selection, meticulous preoperative assessment and alignment of technology with ocular anatomy and visual demands.
As the presbyopic population continues to grow, so too does the expectation for functional, spectacle-independent outcomes. The technology is here. The nuance, as always, lies in selection and execution.
The 41st Asia-Pacific Academy of Ophthalmology Congress (APAO 2026) was held from February 5-8, 2026 in Hong Kong. Reporting for this story took place during the event. This content is intended exclusively for healthcare professionals. A version of this article was first published on MediaMICE.com.
1. Reinstein DZ, Ivory E, Chorely A, et al. PRESBYOND laser blended vision LASIK in commercial and military pilots requiring Class 1 medical certification. J Refract Surg. 2023;39(1):6-14.
2. Auffarth GU, Kretz FTA, Tana-Rivero P. First clinical results of a multicentric prospective study on visual performance of patients implanted with a new trifocal aspheric hydrophobic intraocular lens. Clin Ophthalmol. 2026;20(0):1-15.
3. Meng J, He W, Rong X, et al. Decentration and tilt of plate-haptic multifocal intraocular lenses in myopic eyes. Eye Vis (Lond). 2020;7:17.
4. He W, Qiu X, Zhang S, et al. Comparison of long-term decentration and tilt in two types of multifocal intraocular lenses with OPD-Scan III aberrometer. Eye (Lond). 2018;32(7):1237-1243.
5. Zhu X, He W, Zhang Y, et al. Inferior decentration of multifocal intraocular lenses in myopia eyes. Am J Ophthalmol. 2018;188:1-8.
6. Meng J, Fang Y, Lian J, et al. Visual and patient-reported outcomes of a diffractive trifocal intraocular lens in highly myopic eyes: A prospective multicenter study. Eye Vis. 2023;10(1):19.


As cataract surgery becomes a refractive game, femtosecond platforms are evolving from precision tools into data-driven engines of accuracy. The question is no longer what femto can cut—but what it can calculate.
By Hazlin Hassan
For years, femtosecond laser–assisted cataract surgery (FLACS) has occupied an ambiguous place in ophthalmology. Celebrated for its precision yet questioned for its cost and necessity, femto technology was long framed as a “nice-to-have” adjunct rather than a core refractive tool. That perception is now shifting. As patient expectations rise and premium intraocular lenses (IOLs) become more common, cataract surgery is increasingly judged not by visual acuity alone, but by refractive precision, making femto technology more than just a supplementary tool.
Let’s take a look at some of the reasons why.
The earliest promise of femtosecond lasers lay in their mechanical accuracy, delivering perfectly round capsulotomies, reproducible incision geometry and reduced ultrasound energy through prefragmentation of the lens. While these benefits remain foundational, they now serve a broader refractive purpose.
Effective lens position (ELP) remains one of the most critical, and historically unpredictable, variables in refractive cataract surgery. Even small deviations in IOL position can result in clinically meaningful refractive error. As Prof. Dr. Burkhard Dick, professor of ophthalmology and director of the University Eye Clinic Bochum in Germany, explained, “We must keep in mind that a one mm deviation in IOL position can cause approximately 1.25 diopters of refractive error. Therefore, a laser that precisely centers the IOL can significantly reduce refractive differences.” Modern femtosecond systems contribute to improved ELP predictability.
According to Prof. Dick,
“One of the major advantages of femtosecond laser-assisted cataract surgery is that FLACS capsulotomies are highly accurate in size, circularity and centration, maximizing optic overlap and preventing IOL tilt or decentration compared with conventional surgical techniques.”
He cited studies showing that 91% of FLACS patients achieved a deviation of no more than 0.50 D from the desired refractive result, compared with 71% following manual surgery, illustrating the impact that predictable ELP positioning can have on refractive outcomes.
Hardware precision alone, however, does not guarantee refractive success. The true refractive value of femtosecond platforms emerges when laser accuracy is paired with digital alignment and image-guided planning.
“Digital alignment and image-guided planning are absolutely critical,” said Dr. Miswan Muiz Mahyudin, consultant ophthalmologist and refractive surgeon at Optimax Eye Specialist in Malaysia. “Each plane of a femtosecond laser operates at micron-level precision [1/1000 of a millimeter]. Without accurate imaging, registration and centration, that theoretical precision cannot be fully translated into clinical outcomes.” One of the most notable advances in this area is imageguided capsular marking for toric IOL alignment. Prof. Dick pointed to iris registration–based systems such as LENSAR (LENSAR, Inc.; Florida, United States) with IntelliAxis technology, which uses the patient’s iris “fingerprint” to align laser markings with the cornea’s steep axis. “The femtosecond laser markings on the capsule serve as an exact, permanent target site for toric IOL positioning, obviating the need for ink markings and enabling improved astigmatism correction,” he explained.
Early outcomes appear encouraging, he added. Prof. Dick referenced a study by Visco et al., reporting postoperative astigmatism of less than 0.5 D in 100% of patients treated with femtosecond-guided capsular marking.
Beyond capsular alignment, femtosecond platforms can also deliver precise astigmatic keratotomy incisions with software-controlled length and depth. Ongoing developments, including improved nomograms and advanced imaging technologies such as corneal topographic imaging, may further enhance the reliability of arcuate laser incisions for achieving targeted astigmatism correction, Prof. Dick noted.
As femtosecond cataract surgery increasingly overlaps with refractive principles, surgeon mindset is evolving, though not dramatically. Prof. Dick is careful to temper claims of disruption.
“We are not starting from scratch,” he noted. “We already have an excellent, well-established method: conventional cataract surgery. In the hands of a gifted and experienced surgeon, this method delivers excellent results.” Rather than replacing surgical skill, FLACS adds an additional layer of reassurance. “If ‘mind’ is involved, I would rather describe FLACS as a further step forward—one that gives both surgeon and patient peace of mind,” Prof. Dick said.
The payoff is tangible. “More of our patients than ever before are within 0.5 diopters, or even 0.25 diopters, of the target refraction,” said Prof. Dick. Dr. Muiz draws a clear distinction between cataract and corneal refractive surgery but acknowledges convergence at the level of planning and intent, even if the procedures themselves remain fundamentally different. “FLACS has encouraged cataract surgeons to think more deliberately about refractive targets and consistency, particularly in premium IOL cases,” he explained.
Perhaps the clearest signal that femtosecond platforms are becoming refractive engines lies in software. Next-generation systems are


beginning to leverage the vast imaging data they already collect, transforming diagnostics into actionable refractive intelligence. Prof. Dick highlighted integrated refractive planning with advanced imaging as a key frontier. Emerging systems such as the Femtosecond Laser Enhanced Refractive Outcome (FLERO) combine more than 1,800 imaging parameters with existing IOL formulas using artificial intelligence.
“This approach utilizes AI algorithms that combine femto imaging data with existing formulas, thereby improving refractive calculations,” he explained. The results are more than theoretical.
“We have just published a study in which we could show that FLERO significantly increased the proportion of eyes achieving the targeted postoperative refraction,”
Prof.
Future hardware advances may further reinforce this refractive role. Enhanced fragmentation, faster laser delivery and increased pulse energy could reduce or even eliminate the need for ultrasound energy altogether. According to Prof. Dick, this could lead to “truly ultrasound-free cataract surgery in the future, with unprecedented preservation of ocular tissues.” There may also be potential for expanding the role of femtosecond lasers in refractive surgery beyond traditional cataract indications, he added.
Despite these advances, femtosecond cataract surgery remains unevenly adopted. Experts agree that cost remains the most persistent barrier. This includes both the significant upfront investment required by surgeons or institutions and the additional per-case cost passed on to patients, said Dr. Muiz. Prof. Dick noted, “Currently, the operational and processing costs per case for femtosecond systems are one of the biggest barriers for many surgeons.” In markets where patients are unwilling or unable to pay additional fees, the economic equation remains challenging.
There is also ongoing debate around clinical differentiation. Large studies and meta-analyses have reportedly shown similar visual acuity and complication rates between FLACS and modern phacoemulsification in routine cases, Prof. Dick pointed out. For some surgeons, this raises questions about value and whether the benefits justify the expense. Dr. Muiz offered a pragmatic perspective. “While it may not guarantee superior refractive outcomes in every case, it can significantly reduce surgical variability in selected patients,” particularly in complex or high-risk cases.
For surgeons who still view femto as optional, Prof. Dick’s argument is firmly refractive. “The laser has indeed evolved into a ‘refractive engine’,” he said. “Today, it is a complex system that corrects refractive errors, eliminates astigmatism, and, in the case of toric lenses, ensures proper IOL alignment.”
As patient expectations continue to rise, refractive accuracy is no longer a luxury. It is the standard by which outcomes are judged.
Or, as Prof. Dick cautioned,
“As patients become ever more discerning and demanding, the 21st-century cataract surgeon will need to deliver the best possible functional and refractive outcomes. Otherwise? Watch your practice drown in unfavorable social media reviews.”
The evolution of the femtosecond laser is not about replacing conventional cataract surgery. It’s about redefining what cataract surgery can achieve. In that sense, next-generation femtosecond platforms are no longer accessories to surgery. They are increasingly the engines driving refractive intent, precision and performance forward.
CONTRIBUTOR
REFERENCE
*Van Der Donckt J, Young JA, Rademaker M, et al. Improved IOL power calculation with femtosecond laser enhanced refractive outcome prediction. Translational Vision Science & Technology. 2025;14(11):2811087.

Prof. Dr. Burkhard Dick Chairman and Head of the University Eye Clinic Bochum in Germany. He is one of the most influential surgeons in the world who pioneered bladeless laser cataract surgery in Europe.
burhard.dick@kk-bochum.de

CONTRIBUTOR m1muiz@gmail.com
Dr. Miswan Muiz Mahyudin
Consultant ophthalmologist and refractive surgeon at Optimax Eye Specialist in Malaysia. He specializes in presbyopia management and advanced laser vision correction.
COORDINATING THE FUTURE OF RLRL
Red light therapy has raced from clinical curiosity to a global contender. But with uptake outpacing consensus, ophthalmology now faces the inevitable next question: who sets the standards? From APAO 2026, the launch of IRLOS marks a turning point as the field begins to align data, safety and governance on a global scale.
By Marhiel Garrote

Red light therapy has moved fast in ophthalmology. What began as a regionally concentrated innovation is now a global conversation, complete with growing clinical data, expanding adoption and no shortage of opinions. Enter the International Red Light Ophthalmic Society (IRLOS).
Formally launched at the 41st Asia-Pacific Academy of Ophthalmology Congress (APAO 2026), IRLOS marks a clear pivot point for repeated low-level red light (RLRL) therapy. This was not just another symposium debut, but a signal that the field is ready to move from momentum to method, from rapid expansion to coordinated, evidencebased oversight.
Chaired by Prof. Mingguang He (Hong Kong) and Associate Professor Lisa Zhuoting Zhu (Australia), the session brought together leaders in myopia research, retinal imaging and laser safety to define the Society’s mission, governance and long-term roadmap.
The whole event was set out to answer a simple but pressing question: how do we bring structure, clarity and consistency to a therapy evolving in real time?
Opening the session, Prof. Ningli Wang (China, a leading authority in ophthalmology) framed myopia not as a simple refractive error, but
as an escalating global health challenge. “Myopia really has become a public health issue, not just in China, but the world,” he noted.
He reported that in parts of East Asia, myopia prevalence has exceeded 50%, with high and pathologic myopia increasingly linked to irreversible vision loss. The response, he emphasized, must be rooted in science rather than speed, saying, “We need the evidence…we need the research, we need the clinical trials.”
His remarks set the tone for IRLOS itself, underscoring that innovation must advance alongside rigorous oversight and long-term data.
Over the past few years, RLRL therapy has expanded rapidly across parts of Asia and is now drawing global attention. “Red light therapy is becoming available and also expanding use rapidly, not just in China, but also beyond,” noted Prof. He.
With more than 20 randomized controlled trials conducted in a relatively short time frame, clinical interest has accelerated quickly. In addition, the inclusion of the RLRL therapy in the clinical guidelines has further positioned it as a mainstream therapy. At the same time, variability in protocols and public messaging has created confusion.
“So that explains why we got so many confusing messages from the community, not just among the clinicians but also among the public,” said Prof. He.


To address these gaps, IRLOS was formally registered in Hong Kong as an independent, non-profit, ophthalmology-led body. Its purpose is straightforward yet ambitious: to provide independent, evidence based and clinically meaningful guidance on red light therapy in ophthalmology.
Prof. Jason Yam (Hong Kong) presented the Society’s proposed organizational framework, designed to promote transparency, accountability and international collaboration.
IRLOS will be overseen by a Board of Directors composed of senior ophthalmologists and researchers, supported by an Executive Committee and Advisory Board, alongside dedicated working groups focused on research, guideline development, education and policy.
“Having working groups and committees is important so that we can foster synergy among members working together,” Prof. Yam noted.
The Society plans to hold bi-annual meetings and establish regional chapters to encourage global participation. Structured CMEaccredited education and competency-based certification pathways are also in development, reflecting a commitment to standardized clinical practice rather than ad hoc adoption.
Turning to the evidence, Prof. Kyoko Ohno-Matsui (Japan) shared findings from high-myopia studies, including a randomized trial involving 192 children and adolescents. After one year, axial length increased by 0.34 mm in the control group but shortened by 0.06 mm in the red light group.¹
Instead of progressing by -0.75 D like the controls, treated patients demonstrated a slight +0.11 D shift toward hyperopia.
In a separate cohort of 198 highly myopic children, axial length shortened by 0.11 mm with a +0.18 D shift.²
She also presented unpublished institutional data from a prospective single-arm pediatric study in Japan, in which choroidal thickness also increased, by as much as 45.5 microns in one representative case, alongside a 0.3 mm axial shortening.
One particularly notable observation emerged from optical coherence tomography (OCT)-based structural modeling. “Flattening of scleral curvature never occurs in natural course,” Prof. Ohno-Matsui noted, describing it as “a remarkable finding.”
Adult data showed more modest but measurable effects, including axial shortening of 0.06 mm over two months and a 12-micron increase in choroidal thickness, with no retinal damage reported.
Laser safety was addressed by Prof. John Marshall (United Kingdom), who reviewed radiation exposure and device classification for the Eyerising Myopia Management Device as an example within established international frameworks.
Under international laser safety standards, emissions from the Eyerising Myopia Management Device only marginally exceeding Class 2 limits, with negligible risk both from laboratory measurements and human volunteer evidence.³ Exposure levels were contextualized against Maximum Permissible Exposure guidelines, which incorporate conservative safety factors and were not originally developed to define therapeutic safety thresholds.⁴
“It all looks exceedingly good,” Prof. Marshall said, emphasizing that when protocols are followed correctly, exposure remains well within accepted safety margins. He also stressed the importance of continued monitoring and structured implementation as clinical use expands.
The discussion reinforced that safety is not an adjunct consideration, but central to responsible clinical integration.
Prof. Junwen Zeng followed with large-scale real-world data from China, demonstrating effective refractive control and a reliable safety profile across multi-year cohorts.
He also highlighted the evolution of regulatory oversight in China, where device classification is transitioning toward higher regulatory standards. Rather than signaling retreat, the shift reflects increasing scrutiny and maturation, suggesting that red light therapy is entering a more formalized phase of evaluation.
Beyond Asia, Australian group data presented by Dr. Nellie Deen showed axial length shortening and sustained hyperopic shifts in multi-ethnic pediatric populations. “Short term outcomes do indicate, obviously, axial length and refraction benefits. It’s well tolerated,” she noted.
Clinical case discussions further suggested a potential role for RLRL as an adjunct in treatment-resistant patients within broader myopia management strategies.
Continuing the session, Dr. Loren Rose (Australia) shared real-world experience using red light therapy in treatment-resistant myopia. “I was excited but again, didn’t feel we knew enough at the early stages. So I was quite cautious,” she shared.

Dr. Rose described a rapidly progressing patient who continued to worsen despite atropine escalation and DIMS lenses, reaching 0.5 mm of axial elongation in one year.
After initiating RLRL therapy, following a structured atropine washout to ensure pupil diameter below 4 mm, axial length plateaued and refraction stabilized. At 18 months, axial length was reduced by 0.55 mm in one eye, with sustained stability at two years.
“It was very effective when DIMS and atropine combined were not,” she reported.
Dr. Rose now reserves RLRL for patients progressing more than 0.1 mm over six months despite maximal conventional therapy, with close OCT monitoring, choroidal thickness assessment and compliance tracking.
Dr. Juan Li (China) of Xi’an People’s Hospital presented preliminary data on 30 adults with high myopia (greater than -6.00 D) treated with RLRL for three months.
According to her report, axial length shortened by 0.1 mm at one month and 0.2 mm at three months, with a mean refractive improvement of 0.5 D. Subfoveal choroidal thickness increased early, macular pigment density improved and no ocular adverse events were reported. Compliance exceeded 90%.
“This is just our preliminary data,” she noted. “We [need to] extend both the treatment period and follow-up time.”
The findings suggest that structural remodeling associated with RLRL may extend beyond pediatric populations.
With the formal launch of the International Red Light Ophthalmic Society, red light therapy appears to be entering its next chapter. Not slower, but steadier. Not less ambitious, but more deliberate.
As data continue to emerge across pediatric and adult populations, and as regulatory and clinical frameworks evolve, IRLOS aims to provide the connective tissue: guidelines grounded in evidence, education rooted in experience, and collaboration that stretches well beyond regional borders. The message from APAO 2026? Red light therapy is no longer just expanding. It is organizing itself.
In a field that has moved quickly, IRLOS is betting that the next leap forward comes from doing things properly—together.
The 41st Asia-Pacific Academy of Ophthalmology Congress (APAO 2026) was held from February 5-8, 2026 in Hong Kong. Reporting for this story took place during the event. This content is supported by the International Red Light Ophthalmic Society (IRLOS). A version of the article was first published on MediaMICE.com.
1. Xu Y, Cui L, Kong M, et al. Repeated low-level red light therapy for myopia control in high myopia children and adolescents: A randomized clinical trial Ophthalmol. 2024;131(11):1314-1323.
2. Liu G, Rong H, Xuan L, et al. Axial Shortening Effects of Repeated Low-level Red-light Therapy in Children With High Myopia: A Multicenter Randomized Controlled Trial . Am J Ophthalmol. 2025;270:203-215.
3. CSA Group. Safety of Laser Products – Part 1: Equipment Classification and Requirements (IEC 60825-1 adoption notice). CSA Group Standards. Available at: https://scc-ccn.ca/standards/notices-of-intent/csa-group/safety-laserproducts-part-1-equipment-classification-and-0 Accessed on March 1, 2026.
4. American Academy of Ophthalmology. Managing retinal injuries from lasers. EyeNet Magazine. Available at: https://www.aao.org/eyenet/article/managingretinal-injuries-from-lasers Accessed on March 1, 2026.
APAO 2026 IRLOS LAUNCH: SETTING STANDARDS FOR RED LIGHT MYOPIA THERAPY



In an era of microns, formulas and AI-optimized outcomes, cataract refractive surgery has become exquisitely precise. Yet patients don’t live inside aberrometry printouts. They live in kitchens, cockpits and dimly lit streets. This era has revealed some uncomfortable truths: 20/20 measures clarity, but not necessarily capability. When optics change, the brain—and the lived world—must change with them.
By Diana Truong

In vision science, clarity has long been treated as synonymous with truth. If a patient sees 20/20, the assumption follows naturally: the world is being perceived as it truly is. The numbers line up. The optics are correct. The job is done… Or is it?
Because seeing clearly and seeing meaningfully are not identical experiences. And cataract refractive surgery— perhaps more than any other development in anterior segment surgery—has forced the profession to confront that difference. Replacing a cloudy crystalline lens with a precisely calculated intraocular lens (IOL) does more than sharpen retinal focus. It reorganizes a perceptual system. It recalibrates cortical interpretation. It changes how a human being encounters their lived world.
As cataract surgery has evolved from a sight-saving procedure into a refractive discipline, it has illuminated something both obvious and profound: vision is optical, but it is also experiential. And that realization complicates the very idea of “precision.”
Dr. Ruth Lapid-Gortzak (Netherlands), cataract and refractive surgeon at Amsterdam University Medical Center, recalls a writer who came for his day-after postop visit. He looked at her and said, “Doctor, I can see my wife again, and she is all wrinkled. She looks old.” For a moment, her heart sank, but then he continued, “And now I love her more.”
That brief exchange captures a paradox that no optical bench can measure. The patient gained acuity, but he also gained renewed perceptual intimacy. Details previously blurred into softness—the lines of age, the texture of lived time—emerged sharply. Yet instead of diminishing affection, the clarity intensified it.
What changed? Not simply the retinal image. A relationship changed. A perceptual world shifted. This is the paradox at the heart of modern refractive cataract surgery.
Surgeons can now deliver visual performance that exceeds pre-cataract function and, in some cases, even youthful baselines. Biometry is more exact. Formulas are refined. Astigmatism management is precise to fractions of a diopter. And yet technical perfection does not fully predict human experience.
What qualifies as “normal” vision? Is the goal perfect optical replication? Or is it something richer—something more adaptive?


For centuries, philosophy of mind evaluated perception primarily in terms of accuracy. A perceptual experience is successful if it corresponds to how the world actually is. If it does not, it fails.1 That framework is clean, intuitive and mathematically satisfying, but contemporary visual neuroscience suggests it is incomplete.
“The science of visual perception has a long history of developing quantitative, computational models to explain and predict visual performance on a variety of tasks. These models have typically been developed to account for objective visual performance, such as observer’s accuracy,” wrote Denison and colleagues. “Much less examined is how these models relate to the subjective appearance of a visual stimulus—that is, the observer’s phenomenal character of seeing the stimulus.”2
Objective performance is measurable. Subjective appearance is lived. And the refractive era exists precisely at the intersection of those two domains. When a new intraocular lens is implanted, surgeons are not simply restoring clarity. They are introducing a different optical system—one that interacts with neural architecture shaped by decades of visual history.
Consider contrast sensitivity. Contrast refers to “variation in luminance across space—the differences between the lightest and darkest regions of the image. Visual neurons tend to respond to luminance changes rather than absolute luminance.”2 The brain does
not register absolute luminance values. It emphasizes differences: edges, transitions, meaningful variation. Perception, therefore, is not a passive recording device. It is an interpretive process.
Premium IOLs acknowledge this implicitly. Rather than restoring the eye to a theoretical “normal,” they optimize for functional outcomes: near independence, extended range, reduced spectacle reliance. Tradeoffs are accepted—halos, contrast shifts, adaptation periods—in exchange for expanded capability.
In this light, refractive cataract surgery not only corrects the eye, but it also negotiates with the brain.
Patients frequently struggle to articulate what feels different after surgery. “Not many of them say things we don’t expect,” notes Dr. Lapid-Gortzak. “Most are quite happy, because they are well informed and now it needs healing time.”
But beneath that calm reassurance lies a complex neurobiological process. “I counsel patients actively on neuroadaptation,” she explains. “I tell them it exists, that it is a process. That with surgery we change the system and create a reset moment, which will be followed by a period of learning and adaptation.”
This concept—neuroadaptation—is central to understanding refractive surgery in the modern era. The moment a cataract
is removed, the optical system changes dramatically. Light distribution shifts. Aberration profiles change. Defocus patterns alter.
For patients receiving multifocal or extended depth of focus (EDOF) designs, the retinal image may now contain simultaneous focal planes or structured defocus. But the cortex has been interpreting input from a different optical system for years, sometimes decades. The brain must learn again.
Prettyman reframes perceptual success accordingly: “Perception is successful when it enables us to discriminate among those properties that are most useful for action, even if that involves inaccuracy.”1 In other words, usefulness may matter more than perfect fidelity.
Neuroadaptation underscores a foundational truth: vision is constructed. Incoming photons are only raw material. The brain integrates them with expectation, memory and attention. And that integration takes time.
The refractive era has ushered in extraordinary technological advancement. Optical coherence tomography provides micrometer resolution. Intraoperative aberrometry refines refractive targets. Artificial intelligence optimizes IOL calculations.
Precision is everywhere. And yet, “precision to me is a meaningless word,” says Dr. LapidGortzak. “The sum of our success is in the golden combination of a motivated patient,
an eye and brain that can handle the lenses, biometry and measurement carefully done, calculations executed according to protocol, and successful surgery and healing. There is no room for mistakes. But also no room for precision, because this negates the fact that we are dealing with a myriad of factors that are not all in our control.”
Her comment exposes a tension. Precision, in its narrow sense, suggests fine-grained measurement. Accuracy suggests correspondence with reality. But perceptual success depends on something broader.
Prettyman clarifies the distinction: “A perceptual experience is accurate if it specifies some way that the world is, and the world actually is that way; it is inaccurate otherwise. In contrast, a perceptual experience is precise if it enables the subject having that experience to make fine-grained discriminations among the actual properties of an object.”1
A patient can have a refractive outcome of plano and still feel dissatisfied. Another may have measurable residual error yet report excellent functional vision. Why? Because discrimination in lived context matters more than textbook exactness.
Research on spatial perception reinforces this idea. Systematic distortions of slant or distance may appear inaccurate on paper, yet they enhance sensitivity to environmentally relevant differences.1 The brain bends geometry to serve action. In functional terms, usefulness outweighs purity.
Neuroadaptation is not passive. It is guided by attention. “I usually also explain to the patients that hearing and sight are attention directed functions,” says Dr. Lapid-Gortzak. “Just like we don’t hear stuff that may distract us, we don’t look at things that distract us, and that is where visual side effects should go, to where our attention is not directed at.”
This observation aligns with philosophical accounts of perception. The Stanford Encyclopedia of Philosophy describes perception as, “Mature sensible experience (in
general) presents itself as, in Kantian phrase, an immediate consciousness of the existence of things outside us.” 3 Perception feels immediate, but immediacy is constructed.
For recipients of multifocal or EDOF lenses, new photic phenomena may initially demand attention. Halos are noticed. Glare is salient. But over time, attention recalibrates. The brain deprioritizes signals that are not behaviorally relevant. Dr. LapidGortzak references functional MRI studies exploring neuroadaptation, including work by Rosa et al. in the Journal of Cataract and Refractive Surgery, noting that adaptation is physiologically demonstrable.”⁴ Cortical plasticity is not theoretical. It is measurable. The perceptual system reorganizes itself around new optics.
Philosopher Martin Heidegger proposed that human beings do not encounter the world as detached observers. They inhabit a “lived world.”⁵ Objects are encountered in terms of use, purpose and capability. Vision, in this sense, is not about representation alone. It is about engagement.
“I had to do a fire and smoke drill,” recounted Dr. Lapid-Gortzak’s pilot patient postEDOF IOL implantation. “My copilot was wearing readers, but they did not fit over his gas mask. My colleague was unable to perform optimally under these conditions. I could handle everything, my tablet, the instruments, everything, wearing the gas mask. My colleague could not.”
The difference was not theoretical accuracy. It was operational independence. As Prettyman1 observes: “It is fairly uncontroversial that accurate representation of the world is not necessary for precise action.” Action defines success. The pilot with EDOF lenses did not simply see well. He functioned well.

“As refractive cataract surgery advances, ethical considerations deepen. If accuracy is not the sole metric of good vision, then outcome assessment must broaden.
“The choice of the IOL is guided by the wishes of the patients, and ocular situation, age, tear film, corneal regularity, retinal state etc.,” explains Dr. Lapid-Gortzak. “During the intake we map out all those points, and this usually leads to a choice that we think will benefit the patient most.” This individualized approach reflects a pluralistic understanding of perception.
There is no single ideal visual template. Different patients inhabit different worlds. A night driver. A pilot. A reader. A grandparent chasing toddlers. Each requires a visual system optimized for distinct tasks.
Refractive surgery, then, is not merely corrective. It is configurational. It shapes how a person moves through life.
The refractive revolution has restored sight to millions, but it has also revealed the layered architecture of perceptions: optics, cortex, attention and lived meaning intertwined.
“Clearly refractive lens surgery—be it cataract surgery or refractive lens exchange—is life changing, mostly for the better,” reflects Dr. LapidGortzak. “People gain sight without aids. The thing is, we are mostly concentrating on the unhappy few, because they are concerned and distressed, and this is the tone that prevails, also in public discourse. But basically, for most patients we increase their vision, their range of vision, their freedom to do things.” Freedom. That word reframes the discussion entirely. Not simply clarity. Not solely optical correctness. Not even just precision. But capability.
The most precise vision may not be the most exact representation of an abstract ideal. It may be the visual system that best enables human engagement. Refractive cataract surgery has not only sharpened sight. It has exposed the deeper truth that vision is not a static measurement but a dynamic partnership between optics and the brain. And in that partnership, success is not defined by numbers alone. It is defined by what a person can now do. By how they can now live. By the world they can now inhabit—fully, clearly and freely.

1. Prettyman A. Perceptual Precision. Minds Online. Available at: https://mindsonline.philosophyofbrains. com/2017/2017-session-3/perceptual-precision/. Accessed on February 1, 2026.
2. Denison RN, Block N, Samah J. What Do Models of Visual Perception Tell Us About Visual Phenomenology? In: De Brigard F, Sinnott-Armstrong W, editors. Neuroscience and Philosophy. Cambridge (MA): MIT Press; 2022.
3. Crane T, French C. The Problem of Perception. In: Zalta EN, editor. The Stanford Encyclopedia of Philosophy. Fall 2021 Edition. Available at: https://plato.stanford.edu/entries/perception-problem/. Access one February 1, 2026.
4. Rosa AM, Miranda AC, Patricio MM, et al. Functional magnetic resonance imaging to assess neuroadaptation to multifocal intraocular lenses. J Cataract Refract Surg. 2017;43(10):1287-1296.
5. Heidegger M. Being and Time. New York (NY): Harper & Row; 1962.

CONTRIBUTOR r.lapid@amsterdamumc.nl
Dr. Ruth Lapid-Gortzak Cataract and refractive surgeon at Amsterdam University Medical Center in the Netherlands. She is an expert in multifocal IOL implantation and complicated cataract surgery.


From refugee beginnings to the forefront of refractive and corneal innovation, Dr. Tess Huynh’s career in restoring sight is defined by empathy, purpose and commitment.
By Chow Ee-Tan
Restoring sight has always meant more to Dr. Tess Huynh (Australia) than technical mastery alone. “Vision underpins independence in ways people often don’t realize,” she says. “When you restore sight, you’re not just improving vision. You’re giving someone confidence, dignity and freedom.” For her, ophthalmology has never been just about sharper acuity, but about what clearer vision unlocks in everyday life.
Based in Sydney, Dr. Huynh has spent more than two decades working across cataract, corneal and refractive surgery, guided by a practice philosophy that gives equal weight to precision and care. Fluent in both English and Vietnamese—and closely attuned to Australia’s multicultural communities—she has built a career that extends well beyond the operating theater.
“My work has never been just about surgery,” she reflects. “It’s about communication, trust and understanding who the patient is beyond their eyes.”Her roles span private practice, teaching, examiner responsibilities and long-standing humanitarian missions across Southeast Asia, with each strand reinforcing her belief that vision care is as much relational as it is technical.

Dr. Huynh’s path into medicine and ophthalmology was not defined by a single epiphany, but by a series of formative experiences that steadily reinforced her sense of purpose. Her parents—VietnameseChinese—refugees, brought her and her three brothers to Australia in the late 1970s, instilling in her a deep appreciation for opportunity, resilience and education.
“We grew up knowing how much our parents had sacrificed,” she recalls. “Education wasn’t optional—it was a privilege.”
She would later become the first in her family to attend university, graduating in medicine from the University of Sydney in 1996. Along the way, encounters with inspiring figures left lasting impressions. Meeting Mother Teresa during one of her visits to Australia planted early seeds of service, while reading Gifted Hands, the autobiography of Dr. Ben Carson, provided reassurance and motivation during her teenage years.
“That book stayed with me,” she says. “It made me believe that background doesn’t limit what you can do if you remain committed to service.” During medical school, her interest
in charity work deepened when a classmate shared stories from Christian Blind Mission about ophthalmic surgeons traveling to underserved regions to restore sight.
“That was when ophthalmology really crystallized for me,” she explains. “It combined medicine, surgery and the ability to make an immediate, tangible difference.”
Dr. Huynh later secured a rare job-sharing ophthalmology training position at the Royal Victorian Eye and Ear Hospital, followed by a corneal fellowship at the Centre for Eye Research Australia. She credits strong peer support and exceptional mentorship for shaping her career.
“Mentorship matters enormously,” she says. “People like Prof. Hugh R. Taylor didn’t just teach skills. They encouraged vision, responsibility and courage.
For Dr. Huynh, reinventing the lens experience is not an abstract idea but a daily practice. Advances in ophthalmology have transformed what patients can expect from surgery, enabling procedures of remarkable

precision while delivering outcomes that meaningfully improve quality of life.
“As a refractive surgeon, I often see patients who’ve struggled with glasses or contact lenses for decades,” she explains. “When they regain visual freedom, the emotional impact can be profound.” She has also seen how presbyopia-correcting intraocular lenses have expanded possibilities in both cataract and refractive surgery, raising expectations while placing greater emphasis on patient selection and counselling. Dr. Huynh was among the first surgeons globally to use the Alcon (Geneva, Switzerland) Unity VCS phaco machine, a development she describes as “a genuine step-change in cataract surgery.” She is quick, however, to emphasize that technology alone is never the answer. “The best outcomes happen when innovation is paired with judgement,” she says. “Technology should enhance care, not replace thoughtful decision-making.”
Despite major advances, fear of vision loss remains one of the most common concerns among patients. Dr. Huynh addresses this directly through clear and honest communication.
“I always tell patients that in uncomplicated cases, success rates exceed 99%,” she says. “Modern techniques and safety measures have made these procedures remarkably safe.”
Decision-making, however, is never formulaic. Age, ocular anatomy, lifestyle, occupation and expectations all shape her recommendations. Comprehensive assessments, often lasting more than an hour, are central to her approach. One particularly meaningful case involved a young man with cerebral palsy and very high myopia. Concerned about her son’s long-term independence, his mother sought an alternative to heavy reliance on glasses.
“That case had always reminded me how vision intersects with dignity and autonomy,” she recalls. After careful evaluation, Dr. Huynh recommended phakic lens implantation as the safest and most effective option. The life-changing outcome for both patient
and family reinforced her belief in deeply personalized care.
Dr. Huynh’s clinical focus on corneal disease, particularly keratoconus, reflects her commitment to long-term outcomes. Techniques such as corneal collagen crosslinking have transformed management, while emerging approaches continue to expand therapeutic options.
“I’ve seen how early intervention can preserve vision for decades,” she says. “That’s incredibly rewarding.”
Her advocacy for sutureless pterygium surgery using conjunctival autografting with tissue glue reflects an emphasis on surgical elegance, patient comfort and faster recovery. At the same time, she is candid about the need to set realistic expectations.
“Visual phenomena like glare or halos can occur,” she explains. “Neuroadaptation takes time, and patients need reassurance and close follow-up.”
Dr. Huynh attributes much of her professional perspective to her cultural background. Fluent in Vietnamese, she connects easily with elderly migrants who may feel hesitant about Western medical interventions.
“Being able to speak their language changes everything,” she says. “It builds trust and allows patients to express fears they might otherwise keep silent.”
Rather than viewing identity as a challenge, she sees it as a strength that enhances empathy and deepens patient relationships. It is also a message she shares with young women considering the specialty.“Ophthalmology is a supportive field with generous mentors,” she says. “If you have passion and are willing to work hard, the rewards are immense. Giving back—through teaching or charity work— adds further meaning.”
Humanitarian service remains a defining thread throughout Dr. Huynh’s career. Beginning with Christian Blind Mission in the Philippines in 1999, her work has since expanded across Vietnam and Cambodia through Australian Health and Humanitarian Aid.
“Humanitarian work reminds you why you chose medicine in the first place,” she reflects.
One of her proudest achievements was establishing a retinopathy of prematurity screening program at the National Paediatric Hospital in Hanoi. “That program showed me how sustainable systems can change countless lives,” she says. Today, Dr. Huynh remains driven by innovation, teaching and service. She lectures at the University of Sydney and draws inspiration from colleagues increasingly drawn to humanitarian missions. Watching her son navigate medical school has added a new dimension to her understanding of mentorship and legacy. Looking ahead, she hopes to further elevate patient experience, mentor disadvantaged youth and contribute to outback ophthalmology.





























































































































































































































































































“I envision doing ophthalmology for as long as I can,” she reflects. “It transforms lives in many different ways—in many different places—and it gives hope to so many people.”






and humanitarian service.
tess.huynh@visioneyeinstitute.com.au











Lasers may be faster and implants smarter, but complex eyes are still being lost to reflexive surgery instead of deliberate strategy. In this provocative perspective, Dr. Arun Gulani argues that refractive failures are rarely about devices—and almost always about thinking—showing how disciplined staging, optical design and surgical restraint can turn even “impossible” eyes into predictable, spectacle-free successes.
By Dr. Arun Gulani













































Modern ophthalmology has never been more technologically advanced. Imaging is sharper, lasers are faster, implants more sophisticated and algorithms more refined than ever before. Yet visual dissatisfaction among complex refractive and cataract patients continues to rise. This paradox exposes an uncomfortable truth: technology has advanced faster than surgical thinking.
Radial keratotomy corneas, keratoconus, ectasia, corneal scars, postLASIK, PRK and SMILE interfaces, failed premium IOLs, transplant eyes and multi-operated patients are not technological failures. They are eyes that demand more than devices. They demand judgment.
The growing epidemic of unhappy premium lens patients underscores the need to understand visual optics deeply and to view lens implants as mere ingredients. The true “vision recipe” must be individually designed for each eye to maximize benefit.
After more than three decades of managing premium cataract, corneal and refractive complications from around the world, I have one philosophy that has remained constant: complex eyes are not solved by doing more surgery. They are solved by thinking better and applying the least interventional surgery to intelligently manipulate optics, even when delivered in carefully staged surgical “minuets.”
This mindset is embodied in the Gulani 5-S–based KLEAR™ (Kerato–Lenticulo–Refractive Extended Armamentarium) System, which treats surgical tools as individual “Lego pieces” that can be added, removed or rearranged forward, backward or around the entire visual system. Combined with an uncompromising pursuit of unaided visual potential and a meaningful patient experience, this framework forms the basis of the approach shared in this article.
KLEAR integrates corneal, lenticular and refractive techniques into a seamless, full spectrum system of techniques unified, or staged decision process, allowing complex eyes to be treated as complete optical systems rather than isolated anatomical problems. Refractive rehabilitation becomes orchestration, preservation and design—not reaction.
Let’s start with a case that illustrates this philosophy clearly. A pilot was referred after premium cataract surgery in a keratoconic eye left him with 20/200 vision, hyperopia, presbyopia, irregular astigmatism, high keratometry, a central anterior stromal scar and an already opened posterior capsule. He arrived angry and exhausted after consulting multiple skilled colleagues, all of whom focused on IOL exchange with scleral fixation, while differing in approach of suturing or gluing— none able to promise unaided 20/20 vision.
When he sat in my examination chair, surrounded by visiting surgeons expecting dramatic maneuvers, they instead observed hours of quiet strategy planning. The goals were uncompromising: painless, leastinterventional restoration of unaided emmetropia with a memorable patient experience.
Rather than reacting to the lens implant, I reframed the eye as a refractive system requiring orchestration. Guided by KLEAR, the eye was first staged into a controlled myopic platform using a piggyback lens. This positioning enabled LaZrPlastique® surface laser surgery to simultaneously remove the corneal scar, flatten keratometry, regularize astigmatism, expand the optical zone and design the optics to plano.
So in two brief, pain-free, topical procedures, the pilot achieved 20/15 unaided vision and regained his flying license. For the visiting surgeons, it was not the surgery that changed perspective—it was the thinking. The case reinforces that most surgical vision failures are not lens or
corneal problems alone, but refractive-pathway problems, and that disciplined staging and optical design can convert even “impossible” eyes into predictable successes.
In today’s era of digital visibility, complex cases are often approached as opportunities for dramatic surgical display. While impressive, these can distract from the patient’s real goal: to see as well as possible, as comfortably as possible, for as long as possible, ideally without glasses or contact lenses.
Many patients arrive disappointed that their primary visual request was never addressed. Hence my motto, “show me the patients,” as the only true measure of success. Mastery is not defined by how much tissue is moved or how complex a video appears, but by how little intervention is required to restore optical harmony.
Every surgical plan begins long before the first incision. When a surgeon accepts “better than before,” compromise enters the strategy. When the surgeon insists on emmetropia, optimal visual quality, comfort and dignity, the entire approach changes. Complex eyes do not require lowered expectations; they require elevated thinking.
I encourage every eye surgeon to be a vision-corrective surgeon. The eye is a single optical ecosystem, and surgeons must transcend traditional boundaries between refractive, corneal and lens-based specialties. While not every surgeon must be a retinal or glaucoma specialist, they should be capable of applying the KLEAR framework.
True refractive rehabilitation requires full-spectrum understanding— the ability to think across structures, consequences and time. This allows movement forward, backward or laterally through anatomy and optics while following a correct pathway, guided by the Gulani Planning System (GPS). Sometimes progress is not immediate improvement; it is positioning for staged improvement.


Sequencing is critical. In some complex eyes, I intentionally guide patients through a temporary, visually less favorable stage—not because the destination is uncertain, but because from that position the path becomes secure. I often describe this as traveling from Orlando to Paris via Iceland. The stop may be cold and dark, but the final destination remains non-negotiable. This strategy requires confidence: if the first stage fails, the next does not exist. An example of this is the first case we have described in this article (Lens first and corneal LazrPlastique later).
An “outside-in” is the other approach (making the cornea measurable and stable first, followed by lens based surgery). A 75-year-old nurse presented with counting-fingers vision in an eye with prior hexagonal keratotomy, 23.5 D of irregular astigmatism, keratometry nearing 89 D, ectasia, stable Fuchs dystrophy, high myopia and cataract. She had been advised scleral lenses, corneal transplantation and cataract surgery, always with low expectations for spectacle-free vision.
Instead, rehabilitation was staged with least intervention. INTACS were first used to stabilize ectasia, flatten keratometry and reduce astigmatism from 23.5 D to 1.4 D, making the cornea measurable. Months later, topical, sutureless cataract surgery with a toric implant
achieved 20/25 unaided vision, allowing her to return confidently to nursing.
Across countless similar referrals worldwide over decades, this mindset has allowed me to correct eyes deemed non-candidates or impossible to reach excellent unaided outcomes. In many unhappy premium lens cases referred to me, not a single patient required lens exchange. Instead, minimalistic, staged surgery delivered exactly what they originally sought: unaided vision.
Patients sense this confidence. They become partners in the process, and eventually its strongest advocates.
My relationship with industry as a key opinion leader has never been about celebrating machines, but about clarifying their purpose and elevating surgeons. Technology should evolve from diagnostic measurement into planning intelligence. When thought leads, technology amplifies it. When technology leads, thought recedes.
Staged rehabilitation reshapes the patient experience. Emotionally exhausted individuals often become engaged participants in their own journey, understanding their eyes and celebrating each step. Many ultimately return to their original surgeons with gratitude rather than resentment. Vision is restored, trust is restored and relationships are restored. To me, this is the highest form of surgical success.
LaZrPlastique, Corneoplastique® and LenzOplastique® are not merely techniques refined over decades. They represent a philosophy that treats tissue as precious and optics as art, refusing to discard what can be redesigned and committing to preserve what can be guided.
Complex refractive eyes are not problems to defeat. They are systems to understand. When surgery is guided by thought, restraint, staging and an uncompromising pursuit of excellence, even eyes labeled impossible can be led predictably to clarity, comfort and confidence.

CONTRIBUTOR gulanivision@gulani.com
Dr. Arun Gulani
World-renowned pioneer of next-gen LASIK and cataract surgery. He is the founding director of the Gulani Vision Institute and global CEO of Gulani Surgical Suites headquartered in Jacksonville (Florida, USA).

By Prateet Verma, Senior Director, R&D at BVI

In ophthalmic surgery, fatigue isn’t just about endurance; it directly impacts consistency and clinical outcomes, surgery after surgery. Ophthalmic procedures demand sustained visual concentration, micro-movements and constant coordination between surgeons, surgical technicians and the technologies. Over time, this intensity takes a toll. The physical strain of posture, mental load of decisionmaking, and the cognitive juggling of multiple instruments can lead to fatigue that affects both performance and outcomes.
When we talk about addressing fatigue, we are really talking about reimagining how people and technology work together in the operating room. That’s where innovation begins.
Ophthalmic surgery can broadly be classified into two categories: anterior segment (cataract, refractive and glaucoma) and posterior segment (retina).
In anterior segment procedures, surgeons are typically performing multiple cases per day, often with high cognitive load and minimal margin for error. The primary goal is to improve visual acuity that requires not only technical competence but constant decision-making across repetitive workflows. The mental focus demanded across those back-to-back cases is immense. Retinal surgery, on the other hand, is a marathon. Procedures are longer, many times more complicated and require intense focus for hours at a time. Here, the challenge is both physical and cognitive: maintaining steady posture, managing fine hand-eye control and interpreting micro-level visual cues without interruption.
Both types of surgery share a common thread: the cumulative impact of repetition, inefficiency and strain. Over time, these elements can lead to physical fatigue and mental overload, which results in a critical impact on performance and increased risk of error. Moreover, these inefficiencies can create ripple effects across the OR. The surgical technician, who plays a central role in managing instruments and interfacing with technology, also bears a significant cognitive load. Supporting both surgeons and technicians ergonomically and cognitively must, therefore, be part of the solution.

Technology can play a transformative role in addressing fatigue, but only when designed with the user in mind. Advanced visualization systems, intuitive interfaces and integrated workflows are helping to reduce physical strain and cognitive burden. Yet they also introduce new challenges with learning curves, interface complexity and potential for distraction and inefficiencies if not properly harmonized with clinical practice.
R&D begins with an understanding that innovation should support, not overshadow, the human experience of surgery. A device may be technologically advanced, but if it interrupts the surgeon’s natural rhythm, it risks becoming counterproductive.
That’s why our teams design with the surgeon, surgical technician and ORs in mind. Technology isn’t just about better performance but also to complement the surgical flow. It must feel intuitive, predictable and natural, allowing users to focus on the patient and not the technology.
No amount of lab innovation can substitute for real-world feedback. In our development process, we bring surgeons—our “development KOLs”—to our simulated wetlab environments early and often. They test, challenge and refine our designs. Most, if not all, of our most effective innovations originated from these collaborations.
We believe surgeons to OR are like pilots to the aircraft. In that analogy, our job is to design the cockpit and not just the engine. Every control, every interface, every data point must be placed with a purpose, helping them stay fully immersed in the procedure without unnecessary cognitive friction.
Two of our recent developments—Leos and Virtuoso—embody this design philosophy. Both were created with an explicit focus in reducing fatigue, improving ergonomics and enhancing control in the OR.
Leos, our laser endoscope system, helps reduce dependence on circulating technicians by simplifying workflow. Its wide HD display with autofocus and auto-illumination allows surgeons to visualize pathology in real-time prior to activating the laser, minimizing unnecessary movements and adjustments. If needed, the surgical technician can interface with the alternate draped monitor and adjust settings as intended by the user. The result: fewer steps, fewer distractions and smoother transitions from cataract to glaucoma procedure.
Virtuoso, our latest phaco-vitrectomy platform, takes ergonomic integration a step further. When we designed it, we paid as much attention to how the system “feels” as to how it “performs.” The user interface had to be intuitive and easy to visualize. The sound feedback had to be informative but not intrusive. Every sensory element was tuned to keep users, both surgeons and surgical technicians, focused and confident.
For the surgeon, ergonomics drives everything. We studied grip, weight distribution and wrist alignment for long procedures. The foot pedal, often underestimated, features ten customizable buttons that allow the surgeon to fully control the system. In a high-intensity environment or with a high-turnover surgical setting, that autonomy matters. It translates to precision, consistency and less fatigue over time.
The next frontier of fatigue reduction goes beyond physical ergonomics and into cognitive simplification through connectivity and intelligence.
Artificial intelligence and machine learning are already reshaping the way we think about surgical systems. Imagine a platform that anticipates steps, adjusts parameters dynamically or predicts maintenance needs before downtime occurs.
Connectivity plays a key role. When machines can communicate with each other and with diagnostic systems, the workflow becomes seamless. The goal isn’t to replace human judgment but to supplement data to make fast decisions, free cognitive bandwidth and allow surgeons to focus on what truly matters: the patient.
If we imagine the future, the operating room will look and feel different. We envision surgeon-centric ergonomics akin to an aircraft cockpit. Smart instrumentation that can make autonomous micro-adjustments, supporting rather than dictating surgeon action. Integrated data from clinic diagnostics to intraoperative imaging can converge in real time for decision-making and optimized outcomes.
Step by step, we’re building towards a connected, ergonomic and intelligent OR ecosystem that minimizes fatigue while maximizing precision, comfort and trust. Technology will always evolve, but the true progress in ophthalmic surgery comes from understanding the human experience behind the innovation. Fatigue, after all, isn’t just a physical limitation; it’s a design challenge.
By listening closely to surgeons, technicians and others involved, and by grounding our work in ergonomics, usability and empathy, we can ensure that every advancement serves a simple purpose: helping people see better and helping those who make that possible perform at their best.

pverma@bvimedical.com
Forget posters and podiums. In Hong Kong, eye health went political, financial and unapologetically measurable. What happened in one crowded room at APAO 2026 may end up shaping national eye care systems long after the congress lights went out.
By Michaela Perez

The energy should have been low. It was 9 AM on the last day of the 41st Asia-Pacific Academy of Ophthalmology Congress (APAO 2026) in Hong Kong. The venue— Eaton Club on Harbour Road—was a trek from the convention center. Yet when Brandon Ah Tong (Australia), chair of the Western Pacific region at the International Agency for the Prevention of Blindness (IAPB), stood up to welcome the crowd, nearly every seat was filled.
People had chosen to be there for a specific reason: the 2026 Global Summit for Eye Health, scheduled for November 2 in Antigua and Barbuda, needed commitments. Not the vague, aspirational kind. The measurable, accountable, put-your-name-on-it kind.
THE SUMMIT NOBODY SAW COMING
Amanda Davis (Australia), chair of IAPB’s Political & Institutional Engagement Committee, kicked things off by explaining what makes this summit different from the dozens of eye health conferences held every year.
“This is a political summit,” she said. “Its purpose is essentially to bring governments together to make commitments to what they will do to actually progress eye care in their own countries.”
Not an eye health conference. A political meeting. The distinction matters.
According to Ms. Davis, a United Nations (UN) resolution declaring that eye health
isn’t just a medical issue—it’s social, economic and developmental—shifted the conversation from we should do something to governments must act. The Global Summit is where those actions get codified.
The goal she outlined was ambitious but precise: at least 50 national government commitments; sectoral commitments from NGOs, private companies, academic institutions and professional bodies; an Eye Health Compact co-signed by governments and stakeholders; and, critically, an accountability portal jointly hosted by IAPB and the World Health Organization (WHO), where commitments are tracked publicly. “You want commitments and action,” Ms. Davis said. “There will be an accountability framework where those commitments will be deposited for all to see, for us all to use and monitor.” Translation: no more making pledges at conferences and quietly forgetting them six months later.
Before the room could start drafting commitments, Dr. Anthea Burnett (Australia), IAPB’s head of Data & Evidence, presented the Value of Vision investment case—the economic backbone of the summit.
The numbers are stark. Investing $7.1 billion globally in low- and middle-income countries across six targeted interventions would reduce global sight loss by 24% by 2030. That equates to 255 million cases averted—18 million fewer people with unoperated cataract, 39 million fewer with distance refractive error,
and nearly 200 million fewer with near vision impairment.¹
The real punchline came next. That $7.1 billion investment would generate $199 billion in economic benefits annually, a return of $28 for every dollar spent.¹
“That’s one of the highest treatment-based returns on investment in all of health,” Dr. Burnett said, noting that nutrition interventions deliver a 24:1 return, while cancer treatments sit closer to 10:1.
The case is grounded in real-world costing from 55 providers across 25 countries, with customized dashboards for each nation. Vietnam, for example: $73 million invested yields $1.6 billion in benefits. 2 Nepal, Indonesia and Sri Lanka each have similarly tailored figures ready for policymakers.
Then came the hard part. Drew Keys (Australia), director of Regional Engagement at IAPB, posed the central question: “What’s in your wheelhouse? Where does your society already have influence, legitimacy?” The room broke out into country groups for twenty minutes of concentrated discussion. When the reports came back, patterns quickly emerged.
A representative from Nepal spoke first. The Nepalese Ophthalmological Society, he explained, has helped develop national policy and strategic planning through a HighLevel Eye Health Body under government
leadership. Primary eye care centers now exist in all 77 districts, reaching 90% coverage.
The challenge? “The government’s priority is more on life-threatening diseases,” he said. Nepal employs 4,900 medical doctors in the public system—just five ophthalmologists, with no optometrists or ophthalmic assistants.
Their proposed commitment was pragmatic: advocate for government ownership of eye health using the 28:1 ROI as leverage, create public-sector positions for eye care professionals and establish quality assurance protocols.
Sri Lanka reported strong integration with its Ministry of Health but persistent funding gaps, particularly for children’s spectacles.
Indonesia’s issue was geography: 280 million people spread across thousands of islands, 2,000 ophthalmologists clustered in cities and eye health still not prioritized nationally. Vietnam focused on quality control amid the rapid expansion of private eye hospitals.
The commitments varied, but the throughline was clear: use the economic case to shift government priorities, strengthen data systems and expand access through existing networks.
At a mixed regional table spanning Thailand, China, Vietnam, New Zealand and Australia, one theme cut across all systems: collaborative care. Nitin Verma (Australia), speaking for the mixed regional group, described New Zealand as having reached what he called “utopia”—integrated networks where ophthalmology and optometry jointly advise the government on national programs. Australia’s Vision 2030 and Beyond, he added, brings ophthalmology, optometry, general
practitioners, rural providers and NGOs together for collaborative workshops focused on Indigenous health, diabetes and pediatrics, with clearly defined, non-duplicative roles.
Matt Young (United States), chief executive officer of Media MICE, raised a less comfortable truth: turf battles between ophthalmology and optometry still impede integrated care. His suggestions were practical rather than idealistic—colocated conferences, mutual invitations and structured agreements between professional societies.
The commitments that emerged centered on strengthening team-based care models, extending school screening programs and reducing organizational overlap.
By 11:30 AM, the commitment basket was full. Not finalized—this was only the starting line—but substantive, specific and anchored in national realities. Dr. Rohit Khanna (India), regional chair for outheast Asia at IAPB, closed with three words: partnership, collaboration, networking.
“We have buy-in from the societies,” he said. “Show a hand of commitment.” Hands went up across the room.
The commitment portal opens in the coming weeks, with a deadline of World Sight Day in October. Between now and then, drafts will be refined, tested against national priorities, verified and submitted ahead of the November announcement. Additional touchpoints are already scheduled, including a crossregional meeting in Thailand in April, the International Myopia Summit in Shanghai in May, and 2030 in Sight Live in Nairobi in June. But the real work begins on November 3—the day after the summit. Because
commitments without follow-through are simply better-documented broken promises. The accountability portal keeps pledges visible, the IAPB–WHO monitoring framework ensures progress is reported and the Global Compact provides a shared reference point for responsibility. As one participant put it: “Three things are required. Partnership, collaborations and networking. We have to look for good partners, collaborate with them and spread the network.”
And as Dr. Khanna noted, “This is not the first meeting or the last meeting. We’ll keep meeting more frequently this year.”
The commitment hour in Hong Kong was not an endpoint. It was an ignition point.
For the ophthalmologists who crossed Harbour Road that February morning instead of attending another surgical session, the message was unambiguous: the future of eye health will not be built in operating rooms alone. It will be shaped in rooms where people are willing to put their names on commitments—and then make sure they happen.

EDITOR’S NOTE
This article is based on the IAPB Cross-Regional Meeting held on February 6, 2026, at the Eaton Club, Hong Kong, in conjunction with APAO 2026. Reporting for this story took place during the event.
REFERENCES
1. Wong B, Hennessy J, Stern J, et al. The Value of Vision: An Investment Case for Eye Health. International Agency for the Prevention of Blindness. 2024. Available at: https://visionatlas. iapb.org/global-investment-case/. Accessed on February 25, 2026.
2. International Agency for the Prevention of Blindness. The Value of Vision: An Investment Case for Eye Health - Vietnam. 2024. Available at: https://visionatlas.iapb.org/countryinvestment-case/viet-nam/. Accessed on February 25, 2026.


















