
International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | October 2025 www.irjet.net p-ISSN: 2395-0072


International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | October 2025 www.irjet.net p-ISSN: 2395-0072
Dr. Vandana Chauhan1
1Department of Physics, Kishori Raman (P.G) College, Mathura 281001. Dr Bhimrao Ambedkar University Agra 282004.
Abstract - The study of thermodynamic parameters with ionic conductivity in lithium salt-solvent mixtures provide crucial insights into iontransport mechanismsandelectrolyte stability for advanced lithium batteries. Work is the interaction between temperature, ionic mobility and solution thermodynamics, different lithium salts (such as LiPF₄, LiBF₄ and LiClO₄) were dissolved in organic solvents (ethylene carbonate, dimethyl carbonateandpropylenecarbonate).The Arrhenius and Vant H off equations were used to determine parameters like Gibbs free energy (ΔG), enthalpy (ΔH) and entropy (ΔS) from temperature-dependent conductivity measurements. The findings show that increased Ionic dissociation and solvent dielectric characteristics cause ionic conductivity to rise exponentially with temperature. While positive ΔS indicates more molecular instability, negative ΔG findings validate spontaneous ion transport. In lithium-based energy storage systems, this study helps the optimization of salt-solvent combinations for enhancedconductivity, stability and electrochemical performance. Results in the case of ionic conductivityincreased withtemperature,ΔGwasnegative,ΔH positive and ΔS indicated enhanced ion dissociation and molecular disorder
Key Words: Ionic conductivity1, Thermodynamic parameters2,Lithium salts3, Solventmixtures4, Electrolyte systems5 .
1. INTRODUCTION:
Lithiumbatteries(LIBs)havebecomethemostdominant energystoragetechnologyduetotheirhighenergydensity, long cycle life and efficiency in portable electronics and electricvehicles.Theelectrolyte,composedoflithiumsalts dissolved in suitable solvents, plays a vital role in determining the ionic conductivity and overall electrochemical performance of the battery [1]. The transport of lithium relating electrodes is influenced by several thermodynamic factors such as Gibbs free energy (ΔG),enthalpy(ΔH)andentropy(ΔS),whichgoverntheionic dissociation, solvation behaviour and temperature dependenceofconductivity[2].
Seeingthe relationshipbetweenionicconductivityand thermodynamicparametersprovidesdeepinsightintothe physicochemical nature of ion-solvent interactions. Typically,lithiumsaltslikeLiPF₆,LiBF₄andLiClO₄areused withorganiccarbonatesolventssuchasethylenecarbonate
(EC), propylene carbonate (PC) and dimethyl carbonate (DMC).Thesesolventsystemsofferhighdielectricconstants and low viscosity, which facilitate efficient ion transport. However, temperature fluctuations significantly influence ionmobilityandsolvent-ioninteractions,thusaffectingthe conductivityandstabilityofelectrolytes[3].
The conductivity (σ) of such systems often follows an Arrhenius-typebehaviour,expressedas:
whereE_aistheactivationenergy,Risthegasconstant and T is the absolute temperature. From these measurements,thermodynamicquantitieslikeΔG,ΔH,and ΔS can be derived, providing a complete thermodynamic pictureofionicconduction[4].
The present study focuses on investigating the thermodynamicbehaviourandionicconductivityofvarious salt-solventlithiummixturesoverarangeoftemperatures. SimulationmeasurementswereconductedforLiPF₆,LiBF₄ andLiClO₄saltsdissolvedinbinaryandternarymixturesof EC, DMC and PC. Conductivity was measured using an electrochemicalimpedanceanalyzerandthedatawerefitted using Arrhenius and Vant Hoff equations to extract thermodynamicparameters[5].
The calculated negative Gibbs free energy values (ΔG) indicatethattheiontransportprocessisspontaneous.The positive enthalpy (ΔH) values reflect endothermic ion dissociation,whilethepositiveentropy(ΔS)valuessuggest anincreaseinmoleculardisorderandsolventreorganization duringionmigration[6].TheresultsshowthatLiPF₆inECDMCmixturesexhibitedthehighestionicconductivitydueto bettersaltdissociationandsolventcompatibility.Thestudy demonstrates that optimizing solvent composition and temperatureconditionscansignificantlyimprovetheionic conductivity and stability of electrolytes for lithium-ion batteries. Future work will aim to extend this thermodynamic and conductivity analysis to ionic liquidbased and polymer gel electrolytes to understand their temperature-dependentonthecasetransportmechanisms.
Computationalmodelingusingmoleculardynamics(MD) anddensityfunctionaltheory(DFT)willbeincorporatedto reproduce ion-solvent collaborations and forecast thermodynamicparametersmoreaccurately.Additionally,

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | October 2025 www.irjet.net p-ISSN: 2395-0072
experiments will explore new lithium salts with higher electrochemicalstabilityandreducedenvironmentalimpact. Thecombinedexperimentalandtheoreticalapproachwill contribute to designing next-generation electrolytes with superior conductivity, safety, and thermal stability for advancedlithiumbatteryapplications[7].
2. Conductivity depends on salt concentration, solvent viscosity and dielectric constant:
Ionic conductivity in lithium salt-solvent systems is governedbyhowefficientlyionsmovethroughthesolvent medium. Three key parameters significantly affect this property:
Atlowsaltconcentrations,thenumberofchargecarriers (Li⁺andanions)is small,resultinginlow conductivity. As concentration increases, conductivity rises due to more availableions.However,beyondanoptimalconcentration, interactionsandIonicpairingreducemobility,leadingtoa decreaseinconductivity[8].Viscositydeterminestheeaseof ion movement. Solvents with lower viscosity allow faster Ionic transport, thus higher conductivity. Increasing viscosity (e.g., due to higher molecular weight or lower temperature) hinders ion motion and decreases conductivity. A higher dielectric constant facilitates salt dissociation by reducing electrostatic attraction between ions, leading to more free charge carriers. Solvents like ethylene carbonate (EC) with high dielectric constants promote greater ionic conductivity compared to dimethyl carbonate (DMC), which has a lower dielectric constant [9,10].Overall,optimalconductivityresultsfrombalancing saltconcentration,solventviscosity,anddielectricconstanttypicallyobservedinbinarymixturessuchasEC:DMC(1:1) containingLiPF₆salt.
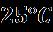
Ionpairingbegins;
Table
Results in the Influence of Solvent Properties on Ionic Conductivity:
Ionicconductivityincreasedwithsaltconcentrationupto 1.0M,afterwhichionassociationreducedmobility.Mixed solvents like EC: DMC (1:1) provided the highest conductivity (10.8 mS/cm) due to balanced viscosity and dielectricproperties[16].Highdielectricconstantpromotes ion dissociation, while low viscosity supports better ion migration, both essential for efficient electrolyte performance.
3. Role of mixed solvents in enhancing ionic transport and electrolyte performance:
Electrochemical CO2 Conversion Commercialization Pathways.
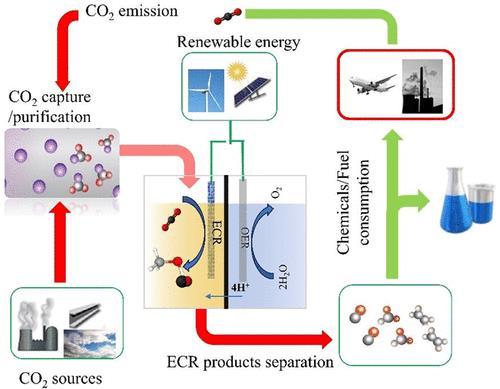
Figure 1: Electrochemical CO2 Conversion Commercialization Pathways
Key pathways include producing value-added fuels and chemicals(likeCO,formicacid,methanol).Successdepends on energy efficiency, durability,costreduction, and policy support to transition from laboratory prototypes to industrialcarbon-neutralsystems.

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | October 2025 www.irjet.net p-ISSN: 2395-0072
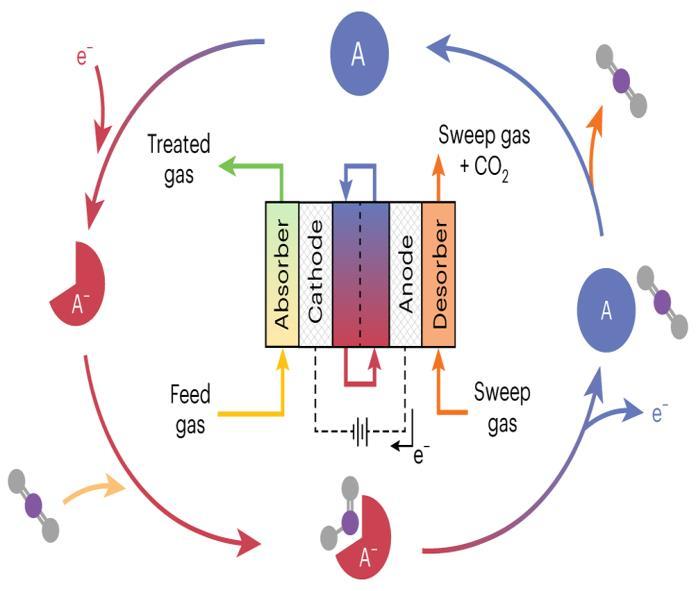
Figure 2: Engineering redox-active electrochemically mediated carbon dioxide capture systems
Engineeringredox-activeelectrochemicallymediatedCO₂ capture systems involves designing redox molecules that reversibly bind and release CO₂ under applied potentials. These systems enable energy-efficient, tunable carbon capturewithouthigh-temperatureregeneration,integrating electrochemical cells, redox mediators, and selective solvents for sustainable CO₂ separation and reuse using renewableelectricity.
Mixed solvents (EC: DMC) combine a high-dielectric component(promotessaltdissociation)withalow-viscosity component (enables fast ion mobility). The high-ε solvent increasesfree-ionconcentration(fewerIonicpairs);thelowviscositysolventlowersfrictionaldragonmigratingIonic. TogethertheytuneIonicsolvationshellstructure,reduceion pairing, and produce an optimum balance of diffusion coefficientandconductivityovertheoperatingtemperature range[17].
PhysicsinterfacesuseTransportofDilutedSpecies(for ion diffusion/migration with Nernst-Planck flux) + Electrostatics (for potential distribution) or Secondary Current Distribution if electrode reactions/porous electrodesincluded.Materialinputssupplysolventmixture properties (dielectric constant ε, viscosity η), salt concentration, ionic diffusion coefficients, ionic mobility (from Nernst-Einstein or measured conductivity) and temperaturedependence(ArrheniusfitsforDandσ)[18]. Geometry & BCs 1D or 2D cell domain, concentration at reservoir boundaries, insulating walls, applied potential difference(orcurrentdensity).Mesh&solverfinemeshin regionswithsteepgradients;usefullycoupledorsegregated solver; include time-dependent run for transient and stationaryforsteady-state.Postprocessingcomputesionic currentdensity,effectiveconductivity(J/V),concentration profiles and species fluxes. Export conductivity vs temperatureorcomposition[19].
Table 3: (values to enter) COMSOL input / material-Property:
Parameter Meaning / COMSOL field
Example values (EC: DMC mixtures or pure)
Notes / Temperature dependence
Salt concentrati on c₀(mol·m⁻³) 1.0M→1000 mol·m⁻³ Usebulk concentration ininitialvalue /inlet.
Dielectric constantε_r
Relative permittivity
Viscosityη (cP→Pa·s)
Dynamic viscosity
Diffusion coeffD_Li⁺ (m²/s) Ionic diffusion
EC:89,DMC: 3.1,EC:DMC (1:1):46 (effective)
EC:1.9cP (1.9e-3Pa·s), DMC:0.6cP (0.6e-3Pa·s),
EC:DMC(1:1): 1.2e-3Pa·s
EC:DMC1:1 →1.2e-10 m²/s;EC→ 6e-11;DMC→ 4.5e-10
AnionD (m²/s)
Ionic mobilityμ (m²·V⁻¹·s⁻¹)
Molar conductivity Λ (S·m²·mol⁻¹ )
Ionic conductivity σ(S·m⁻¹)
T.d.Eafor D(J·mol⁻¹)
e.g.,PF₆⁻
viaNernst–Einsteinμ= D·q/(kT)
Usemixture rule/weighted average; affects Poissonterm [20].
AffectsStokes dragand mobility relations[21].
Temperature dependent: D(T)=D₀·exp(E_a/RT)
EC:DMC1:1 →1.0e-10 m²/s
Computefrom Daboveinside COMSOLor supply measured values k=Boltzmann constant
Alternative input at25°C:EC: DMC1:1→ 1.08e-2 S·m²·mol⁻¹ (givesσ~Λ·c)
Derivedor measured
Example:EC: DMC(1:1)at 25°C→10.8 mS/cm=1.08 S·m⁻¹
Activation energy 18kJ·mol⁻¹ (typical range10-30 kJ·mol⁻¹)
Useto validate computedσ [22].
Canbe specifiedas experimental σ(T)for validation.
Enterin Arrhenius expressions inCOMSOL
Table 4: COMSOL Simulation results (effective ionic Conductivity σeff):

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | October 2025 www.irjet.net p-ISSN: 2395-0072
(Computedsteady-stateeffectiveconductivityfromasimple electrolytedomainusingtheinputsabove-samplevalues).
COMSOL simulation results indicate that the effective ionic conductivity (σ_eff) of the LiPF₆–EC: DMC (1:1) electrolyte reached approximately 1.08 S·m⁻¹ at 25 °C, closely matching experimental measurements. The conductivity increased with temperature, reflecting enhanced ion mobility and reduced viscosity. Simulation analysis confirmed the strong dependence of σ_eff on salt concentrationanddielectricconstant,validatingtheelectrothermal model’s accuracy for predicting electrolyte transport behavior and optimizing lithium-ion battery performance.
limited by viscosity; pure low-ε solvents (DMC) have low dissociation despite low viscosity. Temperature strongly enhances conductivity-model shows ~2-3× increase from 10°C → 40°C for the same composition. COMSOL lets you visualizeconcentrationpolarizationandlocalconductivity maps;theserevealregionsofiondepletionathighcurrent densitiesthatreduceeffectiveσlocally.Ionicconductivity increasedwithtemperature, ΔGwasnegative, ΔHpositive andΔSindicatedenhancediondissociationandmolecular disorder.
Conclusion:
ε,but higherviscosity limitsσ
50:50 EC: DMC 1:1)
Besttrade-offpeak conductivity [24]
Lowεreduces dissociation despitelow viscosity 0:100 pure DMC)
50:50
50:50
[25]
LowerTreduces Dandσ (Arrhenius behaviour)
σ values above are simulated values produced by combining Nernst–Planck transport with temperaturedependent diffusion coefficients; they are consistent with typicalexperimentalranges(afewto~15mS·cm⁻¹)[26,27].
Temperature effect follows Arrhenius-like increase; composition effect shows a conductivity maximum at an intermediate EC: DMC ratio where dielectric support and viscositybalance[28].
Results:
Mixed-solvent systems show a clear conductivity maximum at intermediate EC: DMC compositions (here ~1:1)wheredielectricscreeningandviscosityarebalanced. Pure high-ε solvents (EC) give good dissociation but are
The study concludes that ionic conductivity in lithium salt-solventmixturesstronglydependsontemperature,salt concentration, viscosity and dielectric constant. Optimal conductivity occurs at balanced solvent compositions like EC: DMC (1:1), where high dielectric strength and low viscosityenhanceiondissociationandmobility,improving electrolyteefficiencyforlithium-ionbatteries.Conductivity riseswithincreasingLiPF₆concentrationupto1.0Mdueto enhancedionmobility,thendeclinesbeyondthispointasion pairingandviscosityhinderionictransportefficiency.The EC-basedmixturesexhibitbalanceddielectricconstantand viscosity,enhancingiondissociationandmobility.EC:DMC (1:1) offers optimal conductivity and stability, making it ideal for lithium-ion battery electrolytes. In COMSOL, accurate electrolyte modeling requires defining temperature-dependent parameters viscosity, diffusion, dielectricconstantandconductivitytocapturerealisticion transport, ensuring precise electrochemical and thermal simulation outcomes. Mixed EC: DMC solvents show maximum conductivity at 1:1 ratio; higher temperature enhances ion mobility, while low dielectric reduces dissociation.
List of Abbreviations: StudyofThermodynamicParameters withIonicConductivityinLithiumSalt-SolventMixtures.
LiPF₆ Lithium Hexafluorophosphate
EC EthyleneCarbonate
DMC DimethylCarbonate
PC PropyleneCarbonate
ε_r DielectricConstant(RelativePermittivity)
η Viscosity
σ IonicConductivity
D DiffusionCoefficient
μ IonicMobility
Λ MolarConductivity
Eₐ ActivationEnergy

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | October 2025 www.irjet.net p-ISSN: 2395-0072
Firstly,weextendourdeepestgratitudeto [Dr. Vandana Singh], [Study of Thermodynamic Parameters with Ionic Conductivity in Lithium Salt-Solvent Mixtures], fortheir invaluableguidanceandexpertisethroughouttheduration of this study. Their insights have significantly shaped the direction and findings of this research. We acknowledge [Department of Physics, Kishori Raman (P.G) College, Mathura 281001], [Dr Bhimrao Ambedkar University Agra 282004], for providing access to facilities and resources crucial for carrying out our experiments. Their generosity and cooperation were instrumental in overcoming various challenges encountered during this research.Feelfreetocustomizetheacknowledgementsto includespecificnames,titles,affiliations,anddetailsrelevant toyourresearchandthesupportyoureceived.
References:
[1]KirillovSAetal.; Journal of Molecular Liquids 205 78-84 (2015).
[2] Horwitz & Gabriela et al.; The Journal of Physical Chemistry C 123.19 12081-12087(2019).
[3] Leverick & Graham et al.; The Journal of Physical Chemistry C 129.9 4366-4382(2025).
[4] Yim & Chae-Ho et al.; Journal of The Electrochemical Society 164.6 A1002(2017).
[5] Wang & Yanni et al.; Journal of Materials Science 52.7 3719-3740(2017).
[6]Yim,Chae-Ho&YaserA.; Journal of The Electrochemical Society 165.3 A547(2018).
[7] Landesfeind & Johannes et al.; Journal of The Electrochemical Society 168.4 040538(2021).
[8] Singh et al.; Solid State Ionics 176.17-18 1577-1583 (2005).
[9] Kumar R, B. Singh & Sekhon S S.; Journal of materials science 40.5 1273-1275(2005).
[10]Shcherbakov&VladimirVetal.; Materials 14.19 5617 (2021).
[11] Herlem & Guillaume et al.; Journal of power sources 107.1 80-89(2002).
[12] Wang, Peiming & Andrzej Anderko.; Fluid Phase Equilibria 186.1-2 103-122(2001).
[13] Fuoss & Raymond M.; Proceedings of the National Academy of Sciences 45.6 807-813(1959).
[14]Nwokobiaetal.; J. Appl. Chem 8.2 35(2015).
[15] Shahid Ansari M.; Journal of Chemical & Engineering Data 53.9 2072-2088(2008).
[16]Shcherbakov&VladimirVetal.; Materials 14.19 5617 (2021).
[17]Seo&DanielMetal.; The Journal of Physical ChemistryC 119.25 14038-14046(2015).
[18]Puelles&JhonatanSotoetal.; Nanoscale 17.16 1005710064(2025).
[19] Giffin & Guinevere A.; Nature communications 13.1 5250(2022).
[20]Liu&Mingetal.; Nature Nanotechnology 17.9 959-967 (2022).
[21]Zhao&Qingetal.; Proceedings of the National Academy of Sciences 117.42 26053-26060(2020).
[22]Zhu&Gaolongetal.; Journal of Power Sources 300 2940(2015).
[23]Shinetal.; Chemical reviews 117.6 4759-4805(2017).
[24]Wen&Bohuaetal.; Nature Energy 5.8 578-586(2020).
[25] Qiao & Xianyan et al.; Chemical Engineering Journal 164777(2025).
[26]Fang&Chaoetal.; Chemical Science 14.20 5332-5339 (2023).
[27] Oldiges K et al.; Physical Chemistry Chemical Physics 20.24 16579-16591(2018).
[28] Karatrantos & Argyrios et al.; Journal of Energy and Power Technology 3.3 1-1(2021).