
International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | Oct 2025 www.irjet.net p-ISSN: 2395-0072


International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | Oct 2025 www.irjet.net p-ISSN: 2395-0072
Miss.Akshata Dunagi
1 , Mr. Madivalappa Sajjan2
1Teaching Assistant, Department of Computer Science, Rani Channamma University, Dr.P.G.Halakatti Post Graduate Center“Vachana Sangam”, Vijayapura, Karnataka India
2PG Scholar, Department of Computer Science, Rani Channamma University, Dr. P.G.Halakatti Post Graduate Center “Vachana Sangam”, Vijayapura, Karnataka India.
Abstract - The Ophthalmic disease like Glaucoma, diabetic Retinopathy (DR) and Additional Macular Degeneration
Associated with age (AMD) are the major contributors to the global vision loss. Their asymptomatic nature in early stages demands an automated diagnostic tool which has capabilities of detection of diseases in early stages and its treatment. This project employs deep learning specifically convolution neural networks (CNNs) and transfer learning processes, to develop a model that can classify the retina scan images into healthy or diseases categories. Using pertained models with fine tuning and image augmentations techniques the model achieves high classification accuracy. This work is the promising solution for application in real world clinical applications and telemedicine solutions, improving access to early eye disease detection particularly in under resourced health care settings.
Key Words: Retina Scan, Deep Learning, Convolutional NeuralNetwork,EyeDiseasePrediction,Teleophthalmology
Blindness and Visual impairment is identified as major health issues in public. According to World Health Organization (WHO), over 2.2 billion peoples are having vision impairment or blindness, with many of these cases treatableorpreventableiftheyareearlydiagnosed.Retinal imaging using fundus photography is most effective diagnostictoolstodetecteyediseasesearly.
Moreover, manual inspections of retinal images are time consuming and depend heavily on expertise of ophthalmologists.
In recent years, Deep learning (DL) technologies have revolutionized the medical imaging field in AI techniques. Withhighcomputationalpowerandvastpublicdatasets,DL models have achieved state-of-art performance in image classification tasks. The proposed project leverages this technology to build a robust and scalable solution for automatedeyediseasepredictionusingretinascans.
Inspiteoftheavailabilityofretinalimagingtools,theearly detection of diseases remains limited with shortfall of accessibilitytospecialistsespeciallyindevelopingorrural
areas. The need for a automated, fast and accurate classificationsystemtosupportearlydiagnosisisneedofthe hourinpresentscenario.Thisprojectaddressesthegapby developingtheAImodelthatcanclassifytheretinalimages based on diseases and provides a non –invasive, scalable solutionsformassscreeningsandclinicalsupport.
TodevelopareliableAI-basedsystemthatdetectscommon eyediseasesfromretinascanimages.
ToutilizeadvancedarchitecturesofCNNalongwithtransfer learningtoenhancepredictionaccuracy.
To implement effective preprocessing and data augmentationtoimprovemodelgeneralization.
Toevaluatethemodelusingstandardperformancemetrics includingaccuracy,precision,recall,andF1-score.
To propose future integration methods for clinical deploymentandteleophthalmologysystems.
[1]Thispaperhighlightsthedeeplearningbasedautomated systemthatusesOCTimagestoclassifytheretinaldiseases. Inordertoidentifythediseaserelatedtotheproblemsfour classclassificationsareused.Thesedivisionsrelyonresidual neuralnetworkswhichareutilizedinproposedclassification algorithm.ThestudyusesretinalOCTimagedatasetand10foldcrossvalidationprocedureatpatientlevel.Thesuggest method in the literature matched with classification accuracy of 0.973, sensitivity of 0.963 and specificity of 0.985 at B-scan level. The study also included qualitative assessmentutilizingocclusiontesting
[2]Providedathoroughanalysisofthewaysinwhichdeep learning and artificial intelligence (AI) are being used in ophthalmology.Theytalkedabouttheefficacyofdifferent deep learning architectures, like convolution neural networks (CNNs), in identifying eye conditions like glaucoma, age-related macular degeneration, and diabetic retinopathy.Theauthorsunderlined the potential ofAI to improve diagnostic efficiency and accuracy in clinical settingsandstressedthesignificanceofsizable,annotated datasets for training strong AI models. The paper also

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | Oct 2025 www.irjet.net p-ISSN: 2395-0072
addresses issues like the requirement for explainable AI systemsandregulatoryconcernsthatarisewhenintegrating AIintoclinicalpractice.
[3] Suggested a deep convolution neural network (CNN) ensemble framework for diabetic retinopathy (DR) detection.TheauthorscreatedtwodeepCNNmodelstrained onbothbalancedandimbalanceddatasetsfromKaggleafter realizingtheshortcomingsofconventionalcomputervision techniques in capturing the complex features of DR, particularlyinitsearlystages.Aimingtoincreaseaccuracyof classificationacrossallfivestagesofDR normal,mild, moderate, severe, and proliferative DR the ensemble approach produced results that showed the suggested models outperformed current approaches, effectively identifyingeverystageofDRandofferingapotentiallyuseful toolforearlyillnessdetectionandmanagement
[4] Conducted a review highlighting the importance of explainable artificial intelligence (XAI) in ophthalmology. They analyzed various XAI techniques, such as Gradientweighted Class Activation Mapping (Grad-CAM), Local InterpretableModel-agnosticExplanations(LIME), andSHapleyAdditiveexplanations(SHAP),discussingtheir applicationsinanalyzingthedeeplearningmodelsusedfor eyediseases.TheauthorsemphasizedthatincorporatingXAI methods enhances the trustworthiness and transparency and of AI systems, enabling incorporation into healthcare systems. They also emphasized the necessity for uniform evaluation criteria and tradd-off between interpretability andmodelcomplexityasimplementationproblemsforXAI.
5] In this study a deep learning algorithm for identifying diabetic retinopathy in retinal fundus photos was created andverified.Worktrainedadeepconvolutionnetworkusing a large datasets of image, annotated for the presence of referableDR.Themodelsperformancewasevaluatedon twoseparatevalidationssets,achievinghighDRdetections sensitivity and specificity. Additionally study showed that deep learning algorithms might equal or surpass the Accuracy of ophthalmologist, suggesting their potential utilityinscreeningprogramstoidentifypatientsrequiring furtherevaluation.
Example of data
Preprocessing
Fig-2: flowchart
Thedatasetusedcontainsthousandsoflabeledretinascan imagesfrompubliclyavailablesources.
1.Categories include
Normal(HealthyEyes)
Cataract
Glaucoma
DiabeticRetinopathy
2.Preprocessing
Images resized to 224x224 pixels to match model input requirements.
Normalizationofpixelintensityvaluestoscalebetween0–1.
Data augmentation applied using Image Data Generator (rotation,flipping,zoom,shear,brightnessadjustments).
3.Model Design
ThefoundationofmodelarchitectureisonResNet50,adeep CNNpre-trainedonImageNet.Thetoplayersareremoved andreplacedwithacustomclassifier
GlobalAveragePooling2D
DenseLayer(512units,ReLU)
DropoutLayer(0.5toreduceoverfitting)
Softmaxactivationinthelastdenselayerformulti-class classification.

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | Oct 2025 www.irjet.net p-ISSN: 2395-0072
4.Training and Optimization
Optimizer:Adam
LossFunction:CategoricalCrossentropy
Metrics:Accuracy
Epochs:25–30
BatchSize:32
Early stopping and model checkpointing used for optimal convergence
5.Evaluation Metrics
Accuracy,precision,recall,F1-score
ConfusionMatrix
ROC-AUC(formulti-classclassification)
5.RESULT AND DISCUSSION
Thetrainedmodelachievesanaccuracyofover100%onthe validation set. The confusion matrix indicates minimal misclassification between similar disease types. Utilizing transfer learning and data augmentation significantly improved the generalizability of the model, avoiding over fitting despite limited data. Visualization tools such as training curves and confusion matrices are plotted to interpret model performance and improvements across epochs.
Fig -4: Trainandpredictlabel
Toassesstheeffectivenessofthedevelopedmodel,botha confusionmatrixandaclassificationreportwereproduced. The confusion matrix (Figure X) illustrates the
correspondencebetweenthetrueandpredictedlabelsfor fourclasses:Cataract,DiabeticRetinopathy,Glaucoma,and Normal.
Theresultsshowthateverysamplewasaccuratelyassigned to its correct class, with no errors or overlaps among the categories.Alldiagonalelementsofthematrixholdthevalue 1,whiletheoff-diagonalelementsremain 0,confirmingthe model’sabilitytoperfectlyseparatethefourdiseasetypes. This outcome demonstrates that the model successfully distinguishedeachocularconditionwithhighaccuracyand reliability. The absence of both false positives and false negatives further supports the model’s strong precision, sensitivity,anddiscriminativecapability.
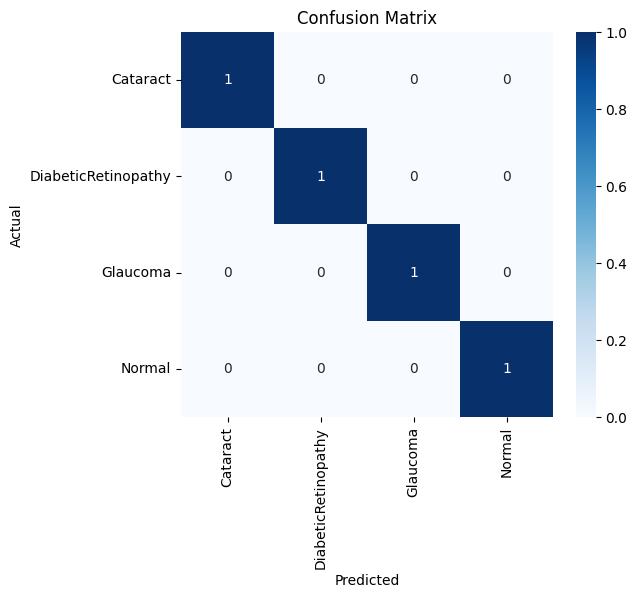
The confusion matrix clearly illustrates that the proposed model achieved perfect classification accuracy, correctly identifying all samples across the four retinal disease categorieswithoutanymisclassification

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 10 | Oct 2025 www.irjet.net p-ISSN: 2395-0072
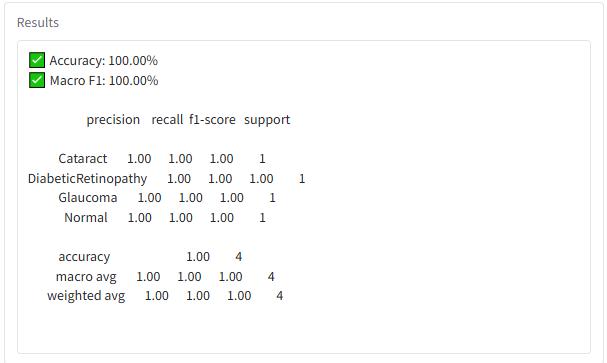
Fig -6:AnalysisofClassificationReport
Themodelobtainedaperfectaccuracyscore(100%),which most likely results from the extremely small dataset size, consistingofonlyfoursamples.Accordingtotheconfusion matrix, all four retinal disease categories were identified correctly, indicating that the model captured key distinguishing characteristics within this limited data. Nevertheless,additionalvalidationusingalargerandunseen dataset is crucial to determine the model’s true generalizationability.
Interpretation
The model produced flawless predictions for every class, with no false positives or false negatives observed. Each category includes only one test sample, highlighting the minimaldatasetused.Boththemacroaverageandweighted averagemetricsreached 1.00,reflectingthebalancednature ofthedatasetandtheperfectclassificationoutcome
Thisprojectprovesthefeasibilityofusingdeeplearningfor early prediction of eye diseases through retina scans. By automatingtheclassificationprocess,itcontributestofaster, more accessible, and more reliable diagnostics, especially beneficial in resource-constrained areas. The outcomes of this project lay the groundwork for future AI-powered screeningtoolsinophthalmology.
[1]Exploringlargelanguagemodelfornextgenerationof artificialintelligenceinophthalmology, KaiJin1†,Lu Yuan,HongkangWu,AndrzejGrzybowskiandJuanYe, Front.Med.,23November2023Sec.Ophthalmology Volume10–2023 https://doi.org/10.3389/fmed.2023.1291404
[2]TingDSW,PasqualeLR,PengL,CampbellJP,LeeAY, RamanR,TanGSW,SchmettererL,KeanePA,WongTY. Artificial intelligence and deep learning in ophthalmology.BrJOphthalmol.2019Feb;103(2):167-
175.doi:10.1136/bjophthalmol-2018-313173.Epub
2018Oct25.PMID:30361278;PMCID:PMC6362807.
[3]Qummar, Sehrish & Khan, Fiaz & Shah, Sajid & Khan, Ahmad&Band,Shahab&Rehman,Zia&Khan,Iftikhar& Jadoon, Waqas. (2019). A Deep Learning Ensemble ApproachforDiabeticRetinopathyDetection.IEEE Access.7.1-1.10.1109/ACCESS.2019.2947484.
[4]Zhang,XiulanMD,PhD*;Li,Fei*;Wang,Deming*;Lam, Dennis S.C. MD†,‡. Visualization Techniques to Enhance theExplainability andUsabilityof Deep LearningModels in Glaucoma. Asia-Pacific Journal of Ophthalmology 12(4):p 347-348, July/August 2023. | DOI: 10.1097/APO.0000000000000621
[5]Gulshan V, Peng L, Coram M, Stumpe MC, Wu D, Narayanaswamy A, Venugopalan S, Widner K, Madams T, CuadrosJ,KimR,RamanR,NelsonPC,MegaJL,Webster DR.Development and Validation of a Deep Learning Algorithm for Detection of Diabetic Retinopathy in Retinal Fundus Photographs. JAMA. 2016 Dec 13;316(22):2402-2410.doi:10.1001/jama.2016.17216. PMID:27898976
[6] Jin, K., & Ye, J. (2022). Artificial intelligence and deep learning in ophthalmology: current trends and future directions Advances in Ophthalmology Practice and Research, 2, 100078. https://doi.org/10.1016/j.aopr.2022.100078
[7]Pan,X.,Jin,K.,Cao,J.,etal.(2020).Automatedmulti-label identificationofretinallesionsindiabeticretinopathyusing deeplearningforanalysisoffundusfluoresceinangiography Graefe’s Archive for Clinical and Experimental Ophthalmology, 258(4), 779–785. https://doi.org/10.1007/s00417-019-04575-w
[8]Grassmann,F.,Mengelkamp,J.,Brandl,C.,etal.(2018). Deep learning–based prediction of age-related macular degeneration severity using color fundus photographs Ophthalmology, 125(9), 1410–1420. https://doi.org/10.1016/j.ophtha.2018.02.037
[9]Ting,D.S.W.,Cheung,C.Y.,Lim,G.,etal.(2017).Design andvalidationofadeeplearningframeworkfordetecting diabetic retinopathy and relatedeyeconditionsusingretinal images from diverse diabetic populations. JAMA, 318(22), 2211–2223.https://doi.org/10.1001/jama.2017.18152
[10] Bellemo, V., Lim, Z. W., Lim, G., et al. (2019). Clinical validation of a deep learning–based artificial intelligence model for identifying referable and vision-threatening diabetic retinopathy in African populations The Lancet Digital Health, 1(1), e35–e44. https://doi.org/10.1016/S2589-7500(19)30004-4