
International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 11 | Nov 2025 www.irjet.net p-ISSN: 2395-0072


International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 11 | Nov 2025 www.irjet.net p-ISSN: 2395-0072
Prof. Shobha S Biradar1 , Mamta2
1 Professor, Master of Computer Application, VTU, Kalaburagi , Karnataka ,India
2 Student, Master of Computer Application, VTU, Kalaburagi , Karnataka ,India
ABSTRACT- The accurate and timely diagnosis of bloodrelated disorders is a critical component of clinical hematology, as abnormalities in blood cells often serve as early indicators of disease progression. Manual examination of peripheral blood smears by pathologists, although considered the gold standard, is time-consuming, labor-intensive, and prone to inter-observer variability. In particular, the detection and classification of different blood cell types including red blood cells (RBCs), white blood cells (WBCs), platelets, and immature cells such as reticulocytes - are essential for diagnosing conditions such as anemia, leukemia, infections, and bone marrow dysfunctions.
Keywords: A small, standardized microscopic-blood benchmarkthatprovidestiny,labeledsingle-cellimages for rapid prototyping and reproducible comparisons. Computer vision, object detection.
A small, standardized microscopic-blood benchmark that providestiny,labeledsingle-cellimagesforrapidprototyping andreproduciblecomparisons.It’sidealforsanity-checking preprocessing,augmentation,andbaselineCNNarchitectures beforemovingtolargerclinicalsets.Becausetheimagesare normalizedandcompact,experimentsrunquicklyandhyper parametersweepsarecheap.Manyteamsusethisdatasetas afirst-steptovalidatetrainingpipelinesandmetricreporting conventions. Treat results on this benchmark as initial proofs-of-conceptratherthandefinitiveclinicalevidence.Use ittoiteratefastandtoestablishreproduciblebaselines.[1]
A largemulti- centerwhite-blood- cellcollectionwithtens of thousands of images and multi- expert labels that supportsbothclassificationandsegmentationwork.Itsscale and diversity across microscopes and labs make it well suitedfortrainingrobustdeepclassifiersandsegmentation backbones.Annotatedmasksenableexperimentationwith detection→segmentation→classificationpipelinesandwith
Mask-basedfeatureextraction.Thedatasetisparticularly useful for transfer-learning experiments and domainadaptation research. Because it reflects real-world variability, it’s a strong choice for pushing models toward clinical resilience. Use it to train models expected to generalizeacrosslabsanddevices.[2]
The primary challenge lies in designing a system that can accurately detect and classify multiple objects in a video streamwhilemaintainingreal-timeperformance.Achieving thisrequiresbalancingspeed,accuracy,andcomputational efficiency. Furthermore, real-world environments often presentadditionaldifficultiessuchasvariationsinlighting, object occlusion, background clutter, and camera noise, whichmakedetectiontasksmorecomplex.
Theprimaryobjectiveofthisstudyistodesignanddevelop an intelligent deep learning–based framework for the automated classification of blood cells and the accurate detectionofreticulocytesfrommicroscopicsmearimages. The study aims to address the limitations of manual diagnosticmethodsbyintroducingascalable,interpretable, and reliable system that can be deployed across diverse clinicalenvironments.Toachievethisoverarchinggoal,the followingspecificobjectivesareoutlined:
Themethodologyofthisstudyisstructuredtosystematically design, implement, and evaluate a deep learning–based framework for blood cell classification and reticulocyte detection. It consists of multiple stages, beginning with dataset collection and preprocessing, followed by model development, training, evaluation, and deployment. Each stage has been carefully chosen to ensure accuracy, robustness,andclinicalapplicabilityoftheproposedsystem.
Thefirststepinvolvesdatacollectionandannotation.Highresolutionmicroscopicimagesofperipheralbloodsmears areobtainedfrompubliclyavailablehematologicaldatasets such as BCCD, HemaCell, and other institutional sources. Theseimagescontaindifferenttypesofbloodcellsincluding redbloodcells,whitebloodcells,platelets,andreticulocytes. Expertannotationisusedto label eachcell type, ensuring that the dataset is reliable and suitable for supervised learning.

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 11 | Nov 2025 www.irjet.net p-ISSN: 2395-0072
Zhangetal. (AMLcGAN,2023) -Demonstratedaconditional GAN approach to myoblast segmentation in AML cytology slides, addressing texture similarity and small object boundaries. Generative augmentation improved segmentationrobustnessandhelpedindata-scarcesettings by synthesizing realistic variants. The cGAN pipeline reportedstrongermaskfidelitycomparedtovanillaU-Net baselines in their cohort. This work illustrates how generativemodelscansupplementscarcenotationsforrare cell types like reticulocytes/blasts. Consider generative augmentationwhenlabeledexamplesarelimitedorcostlyto obtain. Use cGAN outputs cautiously validate on held-out clinicalscans[1]
Applied classification studies (Res Net/Efficient Net pipelines,2021–2024) multipleempiricalpapersadapt modern backbones for multi-class leukocyte typing and reticulocytedetection,showingstrongaccuracygainswith transfer learning. These studies typically combine classimbalance strategies (focal loss, oversampling) with testtime augmentation and assembling. They report high performanceoncurateddatasetsbutcautionaboutcross-lab generalization. Adopt their feature-engineering and loss strategies,andalwaysvalidateexternally.Theseworksshow practicalstepstomovefromtoydatasetstolargerclinical collections[2]
This improves throughput and reduces false positives in dense smears compared to single-shot whole-slide classification. Segmentation masks enable morphological featureextractions(nucleus/cytoplasmratios)thatsupport clinicalexplainability.Thesepipelinesalsoeasepatch-based scaling to whole-slide images (WSI). Use detection→ segmentation→classifierarchitectureforproduction-grade workflows[3]
The proposed AI-based blood cell and reticulocyte classificationsystemisdesignedasanintelligentdiagnostic aid that integrates seamlessly into existing clinical workflows.Itactsasanextensionofdigitalpathologytools and laboratory information systems, bridging the gap between manual microscopy and fully automated hematologyanalyzers.
Overall Architecture Perspective Input Layer: Accepts digitalbloodsmearimages(capturedviamicroscopeorslide scanner). Processing Layer: Handles image preprocessing (noiseremoval,normalization,augmentation).
Relationship with Existing Systems Traditional Microscopy: The system reduces reliance on timeconsumingmanualcountsbyautomatingclassification.Flow Cytometry:Providesacost-effectivealternative,particularly
forreticulocyteestimation,withouttheneedforexpensive instrumentation.
System Context Users: Laboratory technicians, pathologists, researchers, and system administrators (as defined in Section 3.1). Environment: Runs on local laboratoryPCs,hospitalservers,orcloudplatforms.
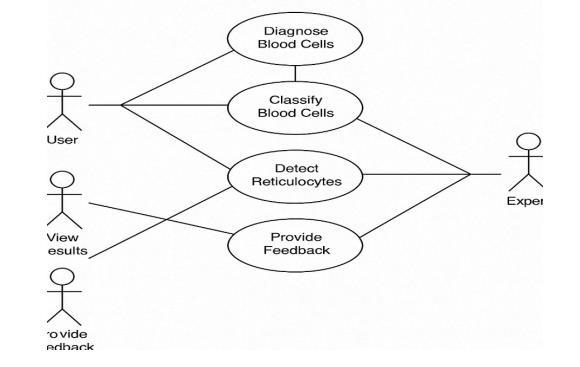
7.2: Use case Diagrams
8. SCREENSHOTS
Figure 7.1: Home page
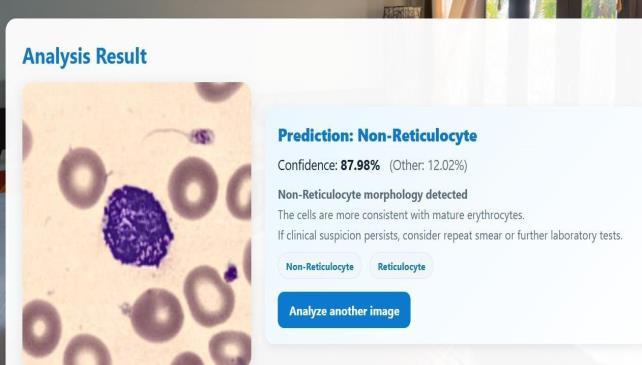
Figure 7.2: Prediction Page

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 11 | Nov 2025 www.irjet.net p-ISSN: 2395-0072
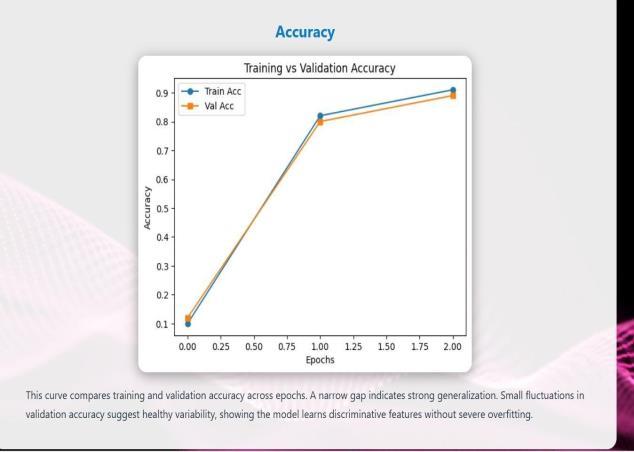
9. SOFTWARE TESTING
The testing strategy adopted ensures that the system is evaluated at every stage of development, from individual modulestocompleteend-to-endworkflows.Bothwhite-box testing(unitandintegration)andblack-boxtesting(system and validation) are applied. Testing also includes performance, usability, and output verification to ensure reliabilityinrealclinicalenvironments.
Level of Testing
Unit Testing: Objective: Totestindividualmodulesofthe system.
Scope: Preprocessing,Segmentation,Classification,Report Generationmodules.
Example:Ensuringthatthepreprocessingmodulecorrectly normalizesimagebrightnessandcontrastbeforesendingit tothesegmentationmodel.
Integration Testing
Objective: Toensuresmoothdataflowandcommunication betweenmodules.
Scope: Preprocessing→ Segmentation→ Classification → ReportGeneration.
Example: Verifying that segmented cells are properly passedintotheclassificationmodelwithnodataloss.
System Testing
Objective: Tovalidatethe completesysteminreal-world conditions.
Scope: Fromimageuploadtofinalreportgeneration.
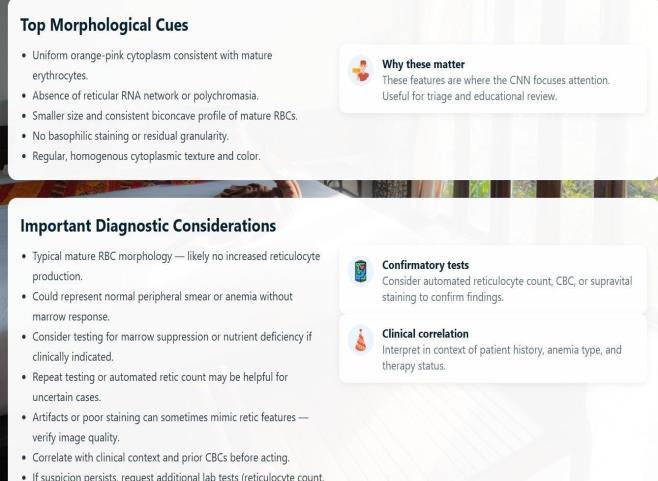
Example: Upload image → preprocess → detect cells → classify → calculate reticulocyte % → generate report → validate.
Output Testing
Objective: To verify correctness, clarity, and usability of reports.
Scope: DiagnosticreportsgeneratedinPDF/CSVformat.
Example: Checking that reports contain cell counts, reticulocyte percentage, and model confidence scores in a clinicallyinterpretableformat.
The implementation of an AI-based Blood Cell and Reticulocyte Classification System demonstrates the effectiveness of deep learning in assisting hematological diagnosis. Traditional manual microscopy is timeconsuming, error-prone, and highly dependent on expert availability.Theproposedsystemautomatestheworkflow by preprocessing images, segmenting cells, classifying different blood components, and calculating reticulocyte percentageswithhighaccuracy.
Thesolutionalsoaddressesscalabilityandadaptability,with deploymentoptionsrangingfromlocalGPUserversinlabs to cloud-based environments integrated with Hospital InformationSystems(HIS/LIS).Thisensuresaccessibilityin bothadvancedhealthcarefacilitiesandresource-constrained regions.
AlthoughtheproposedAI-basedBloodCellandReticulocyte Classification System performs efficiently and has been validated in clinical settings, there is significant scope for futureimprovementandexpansion:
Support for More Blood Disorders: Extendclassification beyond RBC, WBC, Platelets, and Reticulocytes to include

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 11 | Nov 2025 www.irjet.net p-ISSN: 2395-0072
abnormal cells (e.g., sickle cells, blast cells in leukemia, malaria-infectedRBCs).Automatedetectionofhematological disordersforfasterdiagnosis.
Integration with Mobile and Point-of-Care Devices: Developamobileapplicationlinkedwithsmartphone-based microscopesforruralorlow-resourceclinics. Enablerealtimescreeninginfieldsettings.
[1] MedMNIST / BloodMNIST standardized microscopicblood-cellimagebenchmark(MedMNISTv2).
[2] Raabin-WBC large white-blood-cell image dataset (~40kimages)withmulti-expertlabels.
[3]ALL-IDB(ALL-IDB1/ALL-IDB2) acutelymphoblastic leukemiaimagedatasetsforblast/normalclassification.
[4] BCCD (Blood Cell Count and Detection) objectdetectiondatasetforbloodcells(boundingboxes).
[5]CommunityWBCcollections(Kaggle/GitHubcurated white-blood-celldatasets).
[6] AMLcGAN / myeloblast segmentation (2023) conditional-GAN model and dataset paper for myeloblast segmentation.
[7]Reviews&surveysonWBCclassificationandmedicalimageDL(2022–2025).
[8] Large-scale classification & benchmark papers (ResNet/EfficientNettransfer-learningforleukocytetyping).
[9]Detection+segmentationpipelinepapers(YOLO/U-Net /MaskR-CNNappliedtoblood-smearimages).
[10]Papersonevaluation&bestpractices(classimbalance, patient-wisesplits,label-noisehandling).
[11]End-to-endblastdetection/automationstudiesforALL &AML(2019–2024appliedwork).
[12]MILandWSIapproachesforslide-levelpredictionand weaksupervision(2024–2025advances).
[13] Preprocessing and augmentation studies (stainnormalization,artifactfiltering,patchextraction).
[14] Reproducible toolkits & code repositories (PyTorch/TensorFlownotebooksforBloodMNIST/ALL-IDB /BCCD).
[15] Empirical comparisons of detection+classification stacksforbloodcells(2024–2025benchmarkstudies).