
International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net p-ISSN: 2395-0072


International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net p-ISSN: 2395-0072
Amra Odobašić1 , Indira Šestan1 , Amna Karić2, Melisa Ahmetović1 , Husejin Keran1
1University of Tuzla, Faculty of Technology , Department of Physical Chemistry and Electrochemistry, Urfeta Vejzagića 8, Tuzla, Bosnia and Herzegovina
2University of Zenica, Faculty of Engineering and Natural sciences, Department of Chemical Engineering, Travnička cesta 1, Zenica, Bosnia and Herzegovina
Abstract - Theconceptofcirculareconomycametolifeas anideaamongeconomistsin1990,andforthechemicaland otherindustrialbranchesitisonlygainingimportance.The countriesoftheWesternBalkanscommittedthemselvesto theimplementationoftheGreenDealwhensigningtheSofia Agreement,whichincludesagradualtransitiontoacircular economymodel.InOctober2020,theEuropeanCommission announced an economic investment plan for the Western Balkans,withinwhichitwillprovideupto30billioneuros for the purpose of supporting sustainable connectivity, human resources, energy, green transition and implementation of the Green Agenda, and therefore initiativesinthefieldofcirculareconomy[1].Bymeansof the 9R principles of the circular economy (according to English, refuse, rethink, reduce, reuse, repair, refurbish, remanufacture,repurpose,recycle,recover),thedegreeof realizationofthecirculareconomycanbemonitored[2].
Bentonite,duetoitsuniqueadsorptionproperties,iswidely usedintheindustryforwaterpurificationandremoval of impuritiesinoils.However,afteruse,itsfurthertreatment and recycling become key issues for the sustainability of theseprocesses.Theresultsoftheadsorptionexperiments show that bentonite has a high adsorption capacity to remove Pb and Cu ions (the efficiency for both Pb and Cu wasover98%).Researchresultsindicatethatbentonitecan beregeneratedandreusedinvariousindustrialprocesses, which contributes to reducing waste and improving sustainability in industrial applications. HCL acid with a concentration of 0.2 mol/l was used for bentonite regeneration after Pb adsorption, and HCL with a concentration of 0.01 mol/l was used for bentonite regeneration after Cu adsorption. The regeneration efficiencyforPbionsinthefirstcyclewas74.5807%,inthe second 44.5775%, and in the third cycle 42.7355%. The regeneration efficiency for Cu ions in the first cycle was 100%, in the second cycle 98.843% and in the third cycle 98.837%.
Key Words: bentonite, adsorption, lead, copper, adsorption capacity, regeneration
HeavymetalssuchasPbandCuareaseriousenvironmental problem,becausetheirpresenceinwatercanhavenegative consequences for human health and ecosystems. The increased concentration of heavy metals in water is the resultofindustrialactivities,agriculture,urbanizationand other human activities. Lead and copper are particularly significantbecauseoftheirtoxicityandlong-termeffectson organisms.
Bentonite is a type of clay that is widely used in the adsorptionprocessduetoitsfavorablecharacteristics,such ashighsurfacearea,theabilitytoretainvariousions,anda favorableprice-efficiencyratio.Duetoitsphysico-chemical properties,bentoniteisusedtoremoveheavymetalsfrom polluted water and soil. Many studies have shown the efficiencyofbentoniteinremoving PbandCuions,which makesitapotentialmaterialforthetreatmentofpolluted waters. According to literature data, the efficiency of adsorptionofPbandCdfromwastewaterusingbentonite ranges between 90-98%, and for Cu between 85-92%[3]. Fastadsorptionhasgreatpracticalsignificance,asitenables water purification in a short period of time, which contributestogreatereconomyandefficiencyoftheprocess. Bentoniteregenerationafteradsorptionofheavymetalsisof key importance for economically and environmentally sustainablecleaningtechnologies.Testingthepossibilityof regenerationenablesthereuseofbentonite,whichreduces the need for its constant replacement and optimizes economicprofitability.Differentregenerationmethods(such asacidandbasewashing,electrokineticcleaning,etc.)canbe applied,butitshouldbeinvestigatedwhichoneisthemost effectiveandenvironmentallyfriendly.
The aim of this work was to characterize bentonite as an adsorbentforPbandCuionsandexaminetheefficiencyof theadsorptionprocessaswellasthepossibilityofbentonite regenerationafteradsorption.
As a material in this paper used natural bentonite from BosniaandHerzegovina,productofcompany“Bentoproduct” Šipovo,BosniaandHerzegovina.

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net p-ISSN: 2395-0072
ThepHvalueofthebentonitesuspensionwasdeterminedby contactingthebentonitewithdistilledwaterandmeasuring thepHvalueafter72h.Thepointofzerocharge(pHpzc)was determinedusing 0.1 M NaNO3 as a electrolyte. A series ofthis electrolyte ranging from 2 to 10 wasprepared. The pH value was adjusted with HClandNaOH(0.1/0.01 M).Volumeof50mLoftheprepared solutions was put in contact with 0.2 gbentonite.ThepHvaluewasmeasured again. The pH value that has notchanged after 24 hour was represented as the point of zero charge. The ion exchange capacity of bentonite was determined complexometrically,bytreatingbentonitewithasaturated solution Ca(OH)2, and the Ca ion content was determined beforeandafterexchange.Ascanningelectronmicroscope with energy dispersive X-ray spectroscopy (JEOL JSM –6460LV) was used to analyze the shape and size of the particles, as well as the morphology of the bentonite particles before and after adsorption at an accelerating voltage of 20 kV while X-ray fluorescence analysis of bentonite samples before and after adsorption was performedusingaBrukerS8TigerXRFspectrometer.
3. RESULTS AND DISCCUTION
3.1. Physical and chemical characterization of bentonite
Adsorption of pollutants on a suitable adsorbent depends both on the properties of the adsorbent such as crystal structure,totalchargeandchargedistribution,sorbentpore size,surfaceavailability,etc.[4]andonthepropertiesofthe pollutant itself. The pH of zero charge is the pH value at whichthetotalchargeonthebentonitesurfaceis equalto zero,thatis,thestateofthebentonitesurfacewhenthesum of the negative charges is equal to the sum of the positive charges.
ThepHoftheadsorbentsuspensioninwater isanindicator oftheoveralldominanceofacidicorbasicfunctionalgroups onthesurfaceofthesolidphase.Fortheexaminedbentonite sample, the pH of zero charge was 9.5, and the pH of the suspension was 9.7, so based on these values, it can be concluded that bentonite as an dsorbent is richer in basic functionalgroups(eg–OH).
The main mineral component of bentonite is montmorillonite,thereforethepropertiesofmontmorillonite dominatethecharacteristicsofbentonite.Cationexchangeis oneoftheimportantcharacteristicsofmontmorillonite.To determine the cation exchange capacity (CEC), bentonite changes to the Ca 2+ form by treatment with a saturated solution of Ca(OH)2 . Namely, there is an exchange of ions frombentonitewithCa2+ ions.Theamountofcalciuminthe solution, before and after the change, was determined complexometrically.
ThedeterminedCECfor1gramofbentonitewas0.85mmol Ca 2+ ,andfor100gramsofbentoniteitwas85mmolCa 2+ ThisCECvalueconfirmsthattheusednaturalbentoniteisa type of clay that mostly consists of the mineral montmorillonite, given that the CEC value for montmorilloniterangesfrom40to150mmol/100g[5,6,7].
SEM characterization of the pure bentonite sample was performed after adsorption of Pb(II) and Cu(II) ions and showninthefigure1.

Fig-1:SEMimageofapurebentonitesample
OntheFig.1ofapurebentonitesample,“finepowder”can be seen on the surface, which indicates that there is a developedporousstructureinthepowderstructure.Gapsin thematerialcanbeseeninsomepartsofthevideo,which indicatethatthestructureoftheshownparticlesischaotic and irregular, which indicates that there is a lot of empty spaceinthestructure.

a)
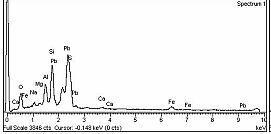
b)
Fig-2:a)SEMimageandb)EDSspectrogramofa bentonitesampleafteradsorptionofPb(II)300mg/l:in selectedpoints
The pictures clearly show the places where the compositionandstructureofthebentonitehaschanged(Fig 3.)

a)
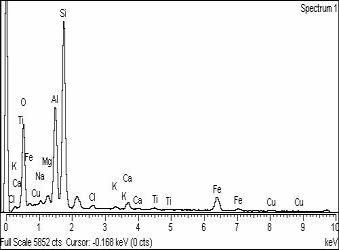
b)
Fig-3:a)SEMimageandb)EDSspectrogramofa bentonitesampleafteradsorptionofCu(II)10000mg/l

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net
The results of X-ray diffraction analysis (XRD) of a pure bentonitesampleconfirmedthepresenceofmontmorillonite (M),quartz(Q)andcristobalite(C).Fig.4showsthatthere was no visible change in the mineralogical composition of bentonite.
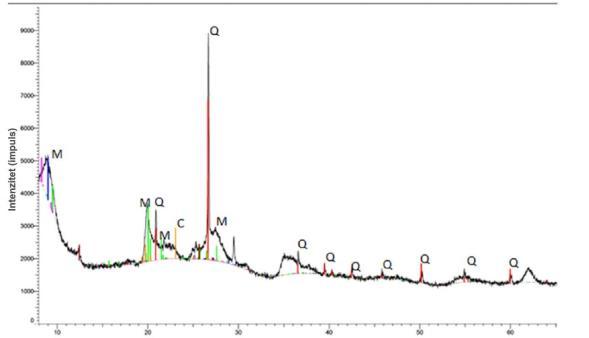
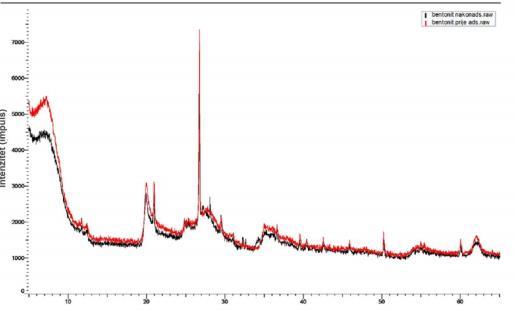
Fig-4.a)Diffractogramofapurebentonitesample; b) diffractogramsofbentonitebefore(red)andafter(black) adsorption
3.2. Results of adsorption isotherms
Tables1.and2.showthevaluesoftheFreundlichisotherm parametersforPb(II)andCu(II).Table1. showstheresults obtainedbynonlinearandlinearregressionforFreundlich isothermparameters,fordifferentprocessparameters,with theindicatedvalueofthecorrelationcoefficientR2forPb(II) ion while Table 2. shows the results obtained by linear regressionforFreundlichisothermparametersatdifferent concentrations with the indicated value of the correlation coefficientR2 forCu(II)ion.
Table-1:Valueofadsorptionisothermparameters
Parameters
2395-0072
Themeanvalueofthecorrelationcoefficientfornon-linear regressionisR2 =0.9464,whileforlinearregressionitisR2 = 0.9558. A good agreement of the results with the FreundlichisothermfortheadsorptionofPb(II)ionsusing bentonitewaspreviouslydemonstratedbyD.JohnBabuet al.[8].Theresultofthecorrelationcoefficientforthelinear regressionisslightlyhigher.Thevalueoftheconstantnfrom the Freundlich model characterizes the intensity of the adsorptionprocess.Whenn>1,adsorptionofadsorbateon the adsorbent is favored, and when n <1, the sorption

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net
processisofachemicalnature[9].Thevalueobtainedinthis test is in the range of 0.3-2.8, which implies that the adsorptionprocessofPb(II)ionsaccordingtothismodelon theusedadsorbentisveryfavorableforasorbentmassof 0.5g(n˃of1).
Table-2:Valueofadsorptionisothermparameters
74.5807% was achieved with the HCl acid solution with a concentrationof0.4mol/l.
AscanbeseeninFig.5,thehighestefficiencyofadsorption (99.8917%)anddesorption(74.5807%)isachievedinthe firstcycle.Inthesecondcycle,theadsorptionefficiencyis the lowest and amounts to 40%, while the desorption efficiencyisslightlyhigherandamountsto44.5775%.Inthe thirdcycle,anadsorptionefficiencyof72.45%isachieved, whilethedesorptionefficiencyisevenlowerandamountsto 42.7355%. The justification for using bentonite as an adsorbentofPbionsforonlyonecycleisitsselectivityfor Pb(II)ions,highremovalefficiency,andaveryfavorablepH value for the treatment of wastewater loaded with this analyte.
ThevalueoftheKF constantforalltestedconcentrationsis high, which indicates a large adsorption capacity of the adsorbenttowardstheadsorbate.Inthecaseofadsorption of Cu (II) ions, the values of this parameter are below 0, whichindicatestheheterogeneityofthebentonitesurface. The coefficient of linearity R2 for concentrations less than 100mg/lislessthan0.9(R 2 <0.9),whichindicatesthatat lower concentrations this isotherm model does not agree well.Because bentonite is widely present in nature, inexpensive,andhasalargespecificsurfaceareaandhigh exchange capacity, it can be used as a cost-effective adsorbentfortheremovalofheavymetalionssuchasPb 2+ , Fe2+ ,Zn 2+ ,andCr 3+ fromindustrialwastewatertoreduce costs and solve disposal problems in an environmentally sustainablemanner.
3.3. Results of bentonite regeneration tests after adsorption Cu (II) and Pb (II)
Chemical regeneration, for the extraction of adsorbate, different solvents (acidic, basic or salt) are used. The efficiencyofchemicalregenerationbasicallydependsonthe solubility of adsorbate in solvents and can change the structureoftheadsorbent[10].Thedesorptionprocesswas followed through three cycles. When examining the possibility of desorption of Pb(II) ions and the reuse of bentonite, three adsorption experiments were performed under the same conditions (initial concentration of Pb(II) ions 200 mg/l, solution volume 50 ml, pH value of the solution 5, amount of bentonite 2 g, mixing speed 50 rpm andcontacttime10min.)250C,mixingspeed50rpmand mixing time 10 min) in HCl acid solutions of different concentrations(0.2,0.4and0.6mol/l),volume50ml.The results showed that the highest desorption efficiency of
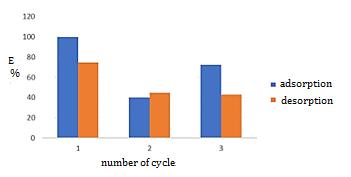
Fig-5:Resultsofadsorption-desorptionprocessesforPb (II)ions
The Cu (II) ion regeneration process was also followed through three cycles. Three adsorption experiments were performedunderthesameconditions(initialconcentration ofCu(II)ions10,000mg/l,solutionvolume50ml,pHvalue ofthesolution5,amountofbentonite1g,mixingspeed300 rpmandcontacttime60min.)witha0.1MHClsolutionasa desorptionagentthatshowedthebestresults.
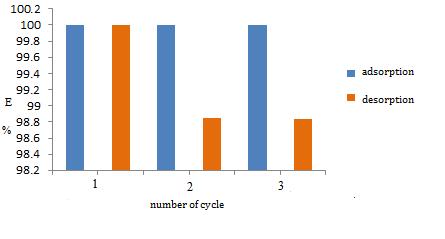
Fig-6:Resultsofadsorption-desorptionprocessesforCu (II)ions
AscanbeseenfromFig.6,theefficiencyoftheCu(II)ion adsorptionprocessdecreaseswitheachsubsequentcycle. Thismeansthatthereisacertainnumberofactivesitesafter thefirstandeverysubsequentadsorption,thatis,thatthe sitesremainpermanentlyoccupiedbyionsoftheanalyzed

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net p-ISSN: 2395-0072
metals,sotheamountofionsinsubsequentcyclesissmaller. Thedesorptionefficiencywasnotnegligibleandamounted to98.843%forCu(II)ionsintheIIcycle,or98.837%forCu (II) ions in the III cycle, which further indicates that the efficiency decreases during the cycle, but still has a value above90%.
3.4. The possibility of using bentonite after adsorption as a starting raw material
Maximum permitted concentrations of heavy metals in building materials such as cement and brick are not universally regulated at the level of the European Union. However,thereareEuropeanstandardsandguidelinesthat deal with the content of hazardous substances in constructionproducts:
•REACHregulation(Registration,Evaluation,Authorization andRestrictionofChemicals):
•Europeanstandardsforcement(EN197-1):
•Europeanstandardsforbricks(EN771-1):
Inadditiontothesestandards,someEUmemberstatesmay havetheirownregulationsorguidelinesthatdeterminethe maximum permitted concentration of heavy metals in buildingmaterials.Therefore,itisimportanttoconsultthe national regulations or standards relevant to the specific country.
Oneofthemostpromisingapplicationsofwastebentonite afteritsuseasanadsorbentinwastewatertreatmentisin the construction industry. Utilization of such waste materialsreduceslandfillspacerequirementsandcanhelp preventdepletionofnaturalresources[18].Immobilization of heavy metals occurs during the hydration process that takesplaceinsidethecementmaterial[20].Namely,during thehydrationofthestructure,cementmineralsgothrough certain changes that enable the immobilization of heavy metalionswithinthenewlycreatedcrystalnetworkwhere theyremainboundwithinthecrystalstructureofthenewly formed cement minerals [21]. Mineral additives - such as spent bentonite can be considered pozzolanic materials, because they influence and participate in the hydration process.Thiscanreducecementconsumption,reduceCO₂ emissionsandimprovethemechanicalpropertiesofthefinal product [11]. However, the composition of the spent bentonite must be analyzed to ensure that it does not adverselyaffectthepropertiesoftheconcrete.
It can be used as an additive to cement and concrete to improve mechanical properties and reduce permeability. Duetoitsstructure,itimprovesthestrengthandstabilityof the material. Studies have shown that bentonite that has adsorbedlead(Pb),cadmium(Cd)andcopper(Cu)canbe incorporated into cement mixtures, with the metals remainingstablyboundwithinthematerial[12].Inaddition,
due to its physical properties, waste bentonite can be included intheproductionofbricksandceramics.Forthis purpose,bentoniteusedcan bemixed withclay,sandand otherbindersintheproductionoffiredbricksandceramics. It can increase plasticity during shaping and thermal stabilityduringfiring[19].Athighfiringtemperatures(9001100°C)heavymetalsareincorporatedintothestructureof the material, thereby reducing their potential ecotoxicity. Theplasticityofbentonitecontributestobetterprocessing ofthematerial,whiletheboundorganicmattercanimprove theenergycharacteristicsoffiringandcancontributetoa controlledporestructure,therebyimprovingtheinsulating propertiesofthebrick.
Its ability to retain water and nutrients can increase soil fertility,whichisbeneficialindryareas.Bentonitesaturated with heavy metals can be used to improve soil conditions undercontrolledconditions,ensuringthatthemetalsarenot bioavailable to plants. It also improves the water and nutrientholdingcapacityofdegradedsoils[13,14].Available literaturedatashowthatbentonitewithadsorbedZnandCu can be used to stabilize soils in mining regions, reducing metalmobilityby75-85%.Itcanalsobeusedasacarrierfor fertilizersandpesticides,improvingtheireffectivenessand reducingleachingintogroundwater[15].
CharacterizationofbentoniteusingSEMandXRDtechniques indicates preservation of structural and adsorption properties, which suggests its suitability for reuse in industrial applications. Adsorption analysis using the Freundlichisothermconfirmsthehighcapacityofbentonite fortheadsorptionofheavymetals,evenafterregeneration.
The research results confirm that bentonite, after regenerationusingHCl,iseffectiveinthere-adsorptionof lead (Pb) and copper (Cu), whereby extremely high adsorption rates (over 90%) were achieved in all regeneration cycles. Regeneration of bentonite under conditionsoflowconcentrationofHCl (0.01mol/L for Cu and 0.2 mol/L for Pb) shows significant efficiency in the removal of heavy metals, and the results of cyclic regenerations indicate the stability and long-term sustainability of the process. Considering the achieved results,regeneratedbentonitecansignificantlycontributeto reducingindustrialwasteandimprovingthesustainabilityof the process, which opens up them possibility of its wider application in various industrial branches, such as water purificationandwastewatertreatment.
[1] VonderLeyenU.StatementbyPresidentvonderLeyen at the joint press conference with President Costa following the EU-Western Balkans Summit, 2024.

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net p-ISSN: 2395-0072
https://ec.europa.eu/commission/presscorner/api/file s/document/print/en/statement_24_6528/
[2] Potting J, Hekkert MP, Worrell E, Hanemaaijer A. CircularEconomy:Measuringinnovationintheproduct chain. Policy Report. The Hague, Netherlands: PBL NetherlandsEnvironmentalAssessmentAgency,2017.
[3] Mina B. Jovanović, “Uklanjanje jona metala iz vodenih rastvora korišćenjem zeolita: mehanizam, kinetika i primenaufluidizovanomsloju”.Doktorskadisertacija, Univerzitet Beogradu, Tehnološko-metalurški fakultet,2016.
[4] Kosmulski M., “Surface Charging and Points of Zero Charge.Fromtheseries,SurfactantScienceSeries”,Vol. 145,CRCPress,TaylorandFrancisGroup,2009.
[5] JockA.Alexander,MuhammadA.AhmadZ.Surajudeen Abdulsalam, Usman A. El-Nafaty & Umar O. Aroke “Isotherm studies of lead(II), manganese(II), and cadmium(II) adsorption by Nigerian bentonite clay in singleandmultimetalsolutions”,ParticulateScienceand Technology,2018
[6] Stathi P., Papadas I.T., Tselepidou A., Deligiannakis Y. “Heavy-metaluptakebyahighcation-exchange-capacity montmorillonite: The role of permanent charge sites” GlobalNESTJournal,12(3),248-255,2010
[7] Lorenz P. Meier, Rolf Nuesch “The Lower Cation ExchangeCapacityLimitofMontmorillonite.Journalof ColloidandInterfaceScience217,77–85,1999.
[8] JohnBabuD.,KalyaniG.,JogaH.Rao,PrasannaKumarY., P.King ”Biosorption characteristics of Lead onto Bentonite Clay, as Low-cost natural biosorbent. International Journal of Scientific & Engineering Research,3(8),2012
[9] Momčilović M. Z. “Kinetički i ravnoteţni adsorpcioni procesi pri uklanjanju pojedinih štetnih katjonskih sastojaka iz vodenih rastvora akticnim ugljevima dobijenih hemijsko-termičkom obradom srţi ploda divljegkestenaišišarkecrnogbora”.Niš:Univerzitetu Nišu,Prirodno-matematičkifakultet,2012.
[10] Momina,ShahadatMohammad,SuzylawatiIsamil“Study oftheadsorption/desorptionofMBdyesolutionusing bentoniteadsorbentcoating”,JournalofWaterProcess Engineering, 2020. https://doi.org/10.1016/j.jwpe.2020.101155
[11] VitaH.,InnaT.,AlinaN.,MargaritaS.,IaroslavR.,Iryna D.,ViсtoriaV.,InnaV.,LudmilaS.”SpentBiosorbentsas AdditivesinCementProduction”,Volume21,Issue2,p. 131–138,2020. https://doi.org/10.12911/22998993/116328
[12] TrusI.M.,FleisherH.Y.,TokarchukV.V.,GomelyaM.D., Vorobyova V.I. “Utilization of the residues obtained duringtheprocessofpurificationofmineralminewater asacomponentofbindingmaterials”.VoprosyKhimiii KhimicheskoiTekhnologii,(6),104–109,2017.
[13] YuC.;KangminC.;XianghaoR.;MeilingL.;YingyingK.; Moon-Hyun H.; Kyu-Jung C. “Modified bentonite as a conditioning agent for stabilizing heavy metals and retaining nutrients in sewage sludge for agricultural uses ”,WaterSci Technol84(9):2252–2264,2021. https://doi.org/10.2166/wst.2021.450
[14] Kumar M.,Bolan N.,Jasemizad T., Padhye L.P.,Sridharan S. ,Singh L.,Bolan S.,J.O’Conn or,H.Zhao,S.M.Shaheen,Song H.,Siddique K.HM.., Wang H. ,Kirkham M.B., Rinklebe J. “Mobilization of contaminants: Potential for soil remediation and unintended consequences,Sci Total Environ,839,Article156373,2022.
[15] ZahidH.,TangC.,MuhammadI.,RiazA.K.,ChenY.,Di S.,MuhammadM.”Bentoniteclaywithdifferentnitrogen sources can effectively reduce nitrate leaching from sandysoil,PLoSONE17(12),2022. https://doi.org/10.1371/journal.pone.0278824
[16] Wisdom C.U.,Ruth O.E.U.,Emmanuel O.E.,Michael C.E.,”Statisticaloptimizationofbiodieselsynthesisfrom wastecookingoilusingNaOH/bentoniteimpregnated catalyst”,CleanerWasteSystems,2022. https://doi.org/10.1016/j.clwas.2022.100049
[17] Gihoon K., Dong-Wan C., Eilhann E. K., Jörg Rinklebe ”Beneficial Use of Fe-impregnated Bentonite as a CatalystforPyrolysisofGrassCutinto Syngas,Bio-oil and Biochar”, Chemical Engineering Journal448(2),137502,2022. DOI:10.1016/j.cej.2022.137502
[18] MuhammadA.,MuhammadF.I.,MominaR.,Muhammad U.A.,AmanU.,HamidM.,Qing-fengL. “Developing a sustainableconcreteincorporating bentoniteclayand silica fume: Mechanical and durability performance” , JournalofCleanerProduction,2022. https://doi.org/10.1016/j.jclepro.2021.130315
[19] MuhammadY.,MuhammadS.,AazibS.,MuhammadN.A. K.,InayatU.K.”Utilizingbentoniteclayasapozzolanic material to Reduce Cement Consumption and Cost in ConstrutionIndustry,JOJMaterialSci.7(5),2023. DOI:10.19080/JOJMS.2023.07.555722
[20] Hemalatha T., Ramaswamy A.”A review on fly ash characteristics – towards promoting high volume utilizationindevelopingsustainableconcrete”,J.Clean. Prod.,147,546-559,2017.

International Research Journal of Engineering and Technology (IRJET) e-ISSN: 2395-0056
Volume: 12 Issue: 08 | Aug 2025 www.irjet.net p-ISSN: 2395-0072
[21] Tashiro C.,Ikeda K.,InoueY.”Evaluationofpozzolanic activity by the electric resistance measurement method”,Cem.Concr.Res.,24,1133-1139,1994.
© 2025, IRJET | Impact Factor value: 8.315 | ISO 9001:2008 Certified Journal | Page436