
IMMpress Magazine Magazine of the Department of Immunology, University of Toronto 2023 | vol.11 no.1 9 772291 239001 ISSN 2291-2398 public perception of science Science and Faith: A Case for Compatibility From hoax to hyperbole: The truth behind the Alzheimer’s Disease scandals Popular science - who gets to have an opinion?
Empowering Your Immunology Research For 20 Years
For over two decades, our scientists have focused on creating unique products for the scientific community—empowering you with antibodies and reagents to find answers for your immunology research. We are constantly innovating to ensure you have the tools needed to make legendary discoveries.
Discover how our 29,000 reagents can make a difference in your lab:
• Fluorophore-antibody conjugates: dyes for conventional and spectral flow cytometry
• TotalSeq™: reagents for multiomic applications
• LEGENDplex™: bead-based immunoassays for cytokine detection
• MojoSort™: magnetic cell separation system for cell isolation and enrichment
• Ultra-LEAF™ & GoInVivo™ antibodies: ideal for functional assays like blocking or stimulation assays
• GMP recombinant proteins: for research and ex vivo cell bioprocessing applications

biolegend.com BioLegend products are manufactured in an ISO 13485:2016-certified facility to ensure the highest quality standards. BioLegend is a registered trademark of BioLegend, Inc. All other trademarks are the property of their respective owners. World-Class Quality | Superior Customer Support | Outstanding Value
the difference at biolegend.com/en-us/immunology
Discover
When one imagines a scientist, we typically turn to an image of someone in a white lab coat surrounded by an array of test tubes. Popularized by children’s cartoons of scientists and overused stock footage of a scientist in the lab, public perception of a scientist is often an oversimplified representation and clouded by success stories. Behind these images, however, are a much more complex identity with ups and downs.
By the same token, popular science knowledge is more than it presents itself to be. Concepts are simplified to accommodate the limited scientific vocabulary. This often begs the question of if the science is effectively and appropriately communicated. Conveying science is difficult and can influence public decisions. A recent example of such is the divided opinions about vaccines. This challenge can be further complicated by misconduct in science. Such blunders and misunderstandings can be rapidly amplified. To counteract miscommunication, increased efforts on both ends are being made to improve the gap between science and the public. New technologies and targeted research allow for more initiatives and increased science accessibility and literacy.
In this issue, public perception of science is represented on the cover as a minimalistic illustration of an eye. From afar, it looks like a simple eye. Upon closer inspection, however, one can see the multiple elements that make up the eye, suggesting that things are not as simple as they seem. Behind the shadow are more layers of knowledge unbeknownst to the public eye. As such, this cover invites you to explore what is behind the shadow as there is more than meets the eye

Design notes
As important as the words on the pages, the visuals form a great part of the public perception of science. In this issue, our talented team of designers have utilized multiple elements of design to effectively communicate the words of our writers, from the use of illustrations to graphs that scientists are familiar with. As you read through this issue, we hope you can appreciate the power of design in how information is conveyed and received. We would like to thank all of our new and returning designers for their beautiful creations and for their creative contribution in making science more accessible.
- Kitty Liu-
EDITORS-IN-CHIEF
Karen Yeung
Philip Barbulescu
DESIGN DIRECTOR
Kitty Liu
SOCIAL MEDIA COORDINATOR

Salma Sheikh-Mohamed
SENIOR EDITORS
Baweleta Isho
Steve Lee
Salma Sheikh-Mohamed
Ammarah Naseer
Siu Ling Tai
Stephanie Wong
Karen Yeung
Evey Zheng
DESIGN ASSISTANTS
Jennifer Ahn
Philip Barbulescu
Baweleta Isho
Manjula Kamath
Meggie Kuypers
Erika McCartney
Louis Ngai
Stephanie Wong
Karen Yeung
Evey Zheng
CONTENT CONTRIBUTORS
Jennifer Ahn
Johanne Audouze
Philip Barbulescu
Melanie Bercovitch
Matthew Bianca
Maye Cheng
Ash Hagerman
Manjula Kamath
Meghan Kates
Erika McCartney
James Pollock
Salma Sheikh-Mohamed
Alara Tuncer
Deeva Uthayakumar
Matthew Wong
Tianning Yu
Evey Zheng
FOUNDING EDITORS
Yuriy Baglaenko
Charles Tran
Copyright © 2013 IMMpress Magazine. All rights reserved. Reproduction without permission is prohibited. IMMpress Magazine is a student-run initiative. Any opinions expressed by the author(s) do not necessarily reflect the opinions, views or policies of the Department of Immunology or the University of Toronto.
About the Cover
IMMpress
P LEASERE-READ, RE-GIFTAND I F Y UO ,TSUMYLLAER ELCYCER
Science and Faith
A Case for Compatibility

From hoax to hyperbole
The truth behind the Alzheimer’s Disease scandals

Popular science

Who gets to have an opinion?
Paths of a PhD in biomedical science

Science fiction or reality: where do genetically modified organisms belong?

Ideas worth spreading - The Power of accessible scientific outreach Initiatives

Social Media - The shiny new tool for science reporting
When life gives you lemons - An interview with alumni Dr. Derek Clouthier
Being a scientist in industry vs academia
The slow decay of our reliance on science


When scientific terminology becomes jargon
A disgraced scientist and his retracted papers

Big corruption in big pharma: a glance into the pharmaceutical industry


Advances in gene therapy
From digital art to PHDOODLESHOP: how a side hustle tunred into a small business
The road less traveled: Why some PhD graduates choose public sector careers
Make it make cents: The inadequacies of research funding in Canada A

@immpressmag facebook.com/IMMpressMagazine immpressmagazine.com
› CONNECT WITH US!
SUBMIT FEEDBACK
CONTENTS VOL 11. NO 1. [2023]
›
on
articles
We would love to hear your thoughts! Comment
our
online or send us an e-mail at editor@immpressmagazine.com
Book Review of Awakenings by Oliver Sacks, 1990 edition 8 14 15 16 18 20 22 24 25 26 28 30 32 34 35
› FEATURE ON [ISSUE] 7 10 12 14 16 22 25 26 28 32 34
With this issue of IMMpress Magazine, we shine the spotlight on how modern media and scientific communication has shaped the public perception of science. It is impossible to discuss any aspect of modern society without acknowledging the role that science and technology have played in enhancing our quality of life. In this digital age with information at our fingertips, there is a growing interest in understanding the science that permeates our daily lives. Simultaneously, it is now easier than ever for scientists to communicate with the public audience through social media—however we have seen that like a game of broken telephone, the spread of information can take on a mind of its own in the face of innocent and purposeful misinterpretation. Given the speed at which misinformation can spread, scientists have an important responsibility to engage the general audience in open and effective dialogue to communicate information in a meaningful way. We continue this dialogue with our fellow Department of Immunology alumnus, Dr. Derek Clouthier, who speaks on importance of creating meaningful discussion between scientists and the public (p18).
We open this issue with a commentary on the age-old discussion regarding the compatibility between science and faith (p7). We also explore how technology has accelerated and broadened the spread of scientific ideas through social media (p16), the rise of popular science (p12), the success of science outreach initiatives (p15). On the flip side, we examine how the spread of misinformation can contribute to the erosion of public trust in science (p22) and the importance of clear communication in science (p24). Next, we highlight cases of academic paper retractions (p25) and how media interpretation of scientific misconduct has rocked the perception of Alzheimer’s disease research (p10). With increasing conversations over science powering the ever-swaying nature of public opinion, we revisit historically controversial topics in the media and give an updated view on genetically modified organisms (GMOs) (p14), corruption in “Big Pharma” (p26), and advances in gene therapy (p28). Lastly, we take a look at what careers await prospective PhD graduates, breaking down the paths of a PhD in biomedical science (p2), comparing jobs in academia versus industry (p20) and the public sector (p32), as well as tracking trends in government support for research funding in Canada (p34). We close with a new segment on student IMMtrepreneurship, featuring recent Department of Immunology alumnus Dr. Melanie Girard, the founder of PHDOODLESHOP, a small business creating science-themed illustrations (p30).
We would like to thank our wonderful team of writers, editors, and designers who have given their time and voices to this issue of IMMpress Magazine, especially as we enter our 11th year of publication! It is with deep gratitude that we say goodbye to our Co-Editor in Chief Philip Barbulescu as he steps down from his position. We are thankful for his years of dedication and contribution to the IMMpress team as writer, designer, editor, and leader. At the same time, we would like to extend a warm welcome to James Pollock as the new Co-Editor-in-Chief. We are also saying goodbye to our Social Media Coordinator Salma Sheikh-Mohamed and welcoming Tianning Yu as her replacement. We hope our readers enjoy this issue, and as always stay safe and healthy!
 Philip Barbulescu Karen Yeung
Philip Barbulescu Karen Yeung


LETTER FROM THE
EDITORS
IMMpress Vol. 11 No. 1 2023 5
Philip Barbulescu (Co-Editor-in-Chief; left), Karen Yeung (Co-Editor-in-Chief; center), Kitty Liu (Design Director, right)
FROM THE CHAIR
These go to 11.
That line from This is Spinal Tap says it all, and highlights how IMMpress Magazine did not just go up to 10 Volumes, but when you need highest level of output from our outstanding UofT Immunology graduate students, well, they do not stop at 10, they go to 11.
This issue of IMMpress Magazine examines the timely notion of how the public’s perception of science has changed over the years, and how the work of scientists is appreciated, questioned, and understood by the public. Given how some sectors of the public responded to the remarkable advances made by scientists to address the need for vaccines and treatments during the pandemic, it is clear that better engagement by the scientific community is needed.
The interview with one of our alumni, Dr. Derek Clouthier, offers remarkable insights as to the non-linear way careers in Immunology can develop, and how seemingly unfortunate events can lead to wonderful new opportunities. IMMpress also connects with Mélanie Girard, who is a recent alumna of UofT Immunology, and her entrepreneurial successes further emphasizes the breadth of accomplishments achieved by our alumni. With these stories in mind, I look forward to the upcoming American Association of Immunologist (AAI) meeting, where I will be hosting our first international UofT Immunology alumni event, as we celebrate the many achievements of our Alumni community.
Finally, I would like to acknowledge and thank Philip Barbulescu’s leadership as Co-Editor in Chief for several issues, as he will be stepping down from this position, plus his many years as writer, designer, and editor. I welcome, and look forward to working with, James Pollock, who will take over as the new Co-Editor-in-Chief, serving alongside with Karen Yeung, who continues in her role as a phenomenal Co-Editor in Chief of IMMpress Magazine.

I wish all a wonderful Summer, as you enjoy this issue, and importantly whether you are an alumnus of UofT Immunology, or a prospective student, I really look forward to hearing from you.
 Juan Carlos Zúñiga-Pflücker, PhD Professor and Chair Department of Immunology
Juan Carlos Zúñiga-Pflücker, PhD Professor and Chair Department of Immunology
LETTER
&cience aith A Case for Compatibility S F
Faith and religion are pillars of human society that have existed since the beginning of mankind. Different forms of spirituality have appeared and evolved over time, with thousands of religions being formally recognized around the globe today. Despite the differences between the beliefs of each faith, one common thread amongst many of them is their apparent incongruence with science.

Like oil and water, science and religion are often treated as immiscible and incompatible. This is usually attributed to their differing explanations on how the universe and life came to be. Science predominantly holds to the Big Bang and biological evolution as explanations on the origin and development of life as we know it. Meanwhile, many religions believe in a variety of concepts, including intelligent design by a creator and reincarnation. However, this perceived incompatibility may be overblown for a number of reasons.
Firstly, many religious groups are partially or entirely onboard with scientific explanations on the origins of life. Some groups in monotheistic religions, like Judaism, Christianity, and Islam, believe in theistic evolution, which holds to the idea that a creator used natural laws and processes to bring about life and the universe. Many Buddhists see no conflict in biological evolution with their beliefs, as they understand organisms to be constantly changing over time. Hinduism similarly has no qualms with these scientific theories, as their religious texts lack a central creation event that would oppose the Big Bang or evolution.
Nevertheless, there are many creation-focused religious groups that deny or argue against the existence of these scientific processes, arguing for a pure creation event backed by alternative interpretations of scientific concepts like homology. An argument does arise on whether the denial of scientific theory may invalidate the veracity of what each religion purports to be true: if a religion makes a false or inaccurate claim, how can we trust the validity of its other beliefs? This leads to an important point regarding differences in the core purposes of religion and science.
Regardless of each religion’s position on scientific theory, these stances are not the central doctrines of these faiths. Science at its core is focused on discovering truths based on empirical evidence. Religion, however, focuses on finding and interacting with a higher being or achieving an enlightened state of existence. Herein lies the discrepancy. Science is expected to explain the origin of life, as uncovering the truth is one of its primary goals. This is not the case for religion,
which may attempt to explain universal origins but ultimately strives for other objectives, such as the worship of a creator or the betterment of a person’s life. Invalidating religion due to an inaccurate explanation on biological processes or the origin of life is attempting to use science as a lens to view faith – an incompatible proposition that also acts as a disservice to the purpose of religion altogether.

Now, one may argue that the purpose of religion is outdated in modern society as it was originally used as an attempt to explain why phenomena like lightning and rain were occurring. With improved scientific knowledge and advancement over the centuries, surely science should have supplanted religion in society. And yet, religion is thriving around the world in even the most scientifically advanced nations. Religion clearly holds a more significant role in society than just explaining natural phenomena. For many, faith can provide a community to join and integrate with. For others, faith may bring about peace and tranquility or provide a deeper meaning to life. The importance of religion in society is multi-faceted, making it unlikely to ever be replaced by scientific knowledge or explanations.
Overall, there is fundamentally no reason a faith follower cannot be a scientist or vice versa. In fact, faith and science can actually be quite synergistic, with interest in one leading to the other. For example, a scientist may look to religion after learning about the complexities and intricacies of the universe, while a faith follower may seek to understand more about the cycle of life and creation through studying science. Thus, the incompatibility between science and faith lies not in their core focuses or beliefs, but rather when trying to force one into the other. One does not (and should not) preclude the other in society, with both providing tremendous benefit to how humankind progresses and functions.
- Matthew Wong
IMMpress Vol. 11 No. 1 2023 7
Paths of a PhD in Biomedical Science
Public confidence in scientists and medical scientists has remained high globally but has seen declines post-pandemic. In comparison to various groups including those in news media, business leaders, and even elected officials, trust in those with scientific training is high. In this infographic, we explore data surrounding the rigorous training scientists require to obtain a biomedical PhD, the challenges that graduate students might face, and the variety of doors that it opens.
Overall, the PhD experience varies by large margins depending on the country and the institution selected. Program structure, average length to completion, availability of funding, research focus, and opportunities available to students are areas that vary the most between programs and have large effects on the overall experience of a graduate student—both MSc and PhD.
The data presented in this infographic was obtained from published literature surveying graduate and post-graduate students across the world.
Lack of affordable housing
Learning about other career options
30% 20% 10% 0% <11 21-30 41-50 61-70 Average weekly hours spent on graduate degree >80 Hours per week North America Asia Europe Average PhD length 5-7 years 5 years 3-4 years
1 Career path 60% 45% 30% 15% 0% Maintaining a work-life balance Financial issues Funding difficulties Availibility of research jobs available Uncertainty about value of PhD
after starting a PhD Mental health Political landscape Imposter syndrome Other Competition / Lack of job opportunities 80% 60% 40% 20% 0% Discouraging funding climate Work-life balance Salary Too demanding Don’t enjoy research culture in academia
outside
Concerns
Prefer a research career
academia
Australia Asia & Middle East Africa Europe North / Central America South America Do you expect your graduate degree to improve your job prospects? Overall 6 25 48 15 6 31 39 17 13 60 19 5 27 47 15 20 48 25 9 13 54 21 5 24 48 18 Not at all Barely Somewhat Unsure Substantially Dramatically Are you studying in the country you grew up in? 37% 63% No Yes Somewhat satisfied 40% 30% 20% 10% 0% Very satisfied Undecided Somewhat dissatisfied Very dissatisfied
Working with
40% 30% 20% 10% 0% Intellectual Challenge University / Academic Environment Creativity Chance to consider professional options What did you enjoy most as a PhD student Social life Chance to use skills in future career 4 1 1 Finding a permanent job after graduation 60% 40% 20% 0% Overall cost of living
Why are PhDs not pursuing academic careers Requires too much administration work Political climate is hostile towards academia
How satisfied are you with your decision to pursue a PhD?
bright and interesting people
Work-life balance
most difficult
1 3 3 3 8 IMMpress Vol. 11 No. 1 2023
Living as an international student in a different country Future student debt The
challenges for graduate students
Which of the following would you most like to work in ultimately?
40% 30% 20% 10% 0% Lack of support / unawareness Conflicting interests with supervisor / employer Personal or family issues Lack of planning on own behalf Transition to working in a foreign country Uncertain
Career advice recieved Focus on particular career directions
Career challenges
ACADEMIA
INDUSTRY RESEARCH
HEALTHCARE & CLINICAL SCIENCE
GOVERNMENT
SCIENTIFIC WRITING & COMMUNICATION ADMINISTRATION
OUTREACH & SCIENTIFIC EDUCATION
BUSINESS & CONSULTING
LAW, POLICY & REGULATORY AFFAIRS
NON-PROFIT
DATA SCIENCE
5 4 3 2 1 low Rank: high Industry Academia Government Non-profit Medical 10 10 12 20 48% 11 16 19 25 28 18 22 27 23 9 30 28 20 14 7 31 23 21 17 8 Networking events 80% 60% 40% 20% 0% Attending career workshops and seminars
Lecturer / Teaching professor, Professor, Postdoctoral scholar, Research associate, Lab Manager, Staff scientist 80% 60% 40% 20% 0% Academia Medical Sector None Non-profit Government Startup
Discussing with a supervisor / PI Discussing career future with a mentor
Individualized
Scientist / investigator, Director of research, Discovery researcher, Researcy Associate, Study director, Postdoctoral scholar, Specialist lab lead (Imaging, FACS, mass spectrometry)
Laboratory manager, Grant manager, Program associate, Program specialist, Study director, Program manager / director, Scientific review officer, Policy analyst, FDA Reviewer, Scientist / Investigator, Research associate Industry
Science / Medical writer, Science Career Specialist, Events coordinator, Curriculum developer, Outreach coordinator / director, Education specialist, Community relations manager, Museum educator / exhibit developer, Teacher / educator
Product development scientist, Application scientist, Business development, Analyst, Scientific sales, Market researcher, Strategy consultant, Accounts manager, Venture capital, Investment-related consulting, C-suite position Medical Science Liaison, Scientific marketing, Journal editor, Animator, Illustrator, Communications officer, Journalism, Skill-focus instructor, Public relations, Medical affairs, Broadcasting (science-related), Press officer, Technical support
Legislative policy analyst, Intellectual property law, Public affairs officer, Advocacy, Health program specialist, Patent agent, Program / legislative analyst, Policy advisor, Policy Analyst, Public health analyst, Health policy, Radiation safety specialist Program manager / director, Grants manager, Policy analyst, Scientific Review Officer, Animal subjects specialist, Advocacy associate, Science Education / Outreach, Scientist / Investigator, Research associate
Biostatistician, Bioinformatician, Quality control specialist, Research Software Engineer, Genomics / Proteomics specialist, Data steward
- Philip Barbulescu
NON-PROFIT
Clinical data analyst / manager, Clinical trials coordinator / director, Genetic counselor, Epidemiologist, Clinical laboratory associate, Public health coordinator, Translational research director, Clinical research coordinator Technology licensing agent, Development officer, Technology transfer agent, Career development officer / advisor, Core coordinator / administrator, Senior research officer, Biocurator, IRB (internal review board) administrator, Program coordinator
Developing social media (LinkedIn / Twitter)
development plan
Discussions with career counsellor PhD Masters
36 18 41 24 50% 19 Yes Have asked, but have not recieved yet No Wanted to, but have not sought it Prefer not to say Career options with a PhD and/or MSc Work Cited: 1 Woolston C. Nature Career Feature. (2022) 2 Gehr S., Garner C.C., Kleinhans K.N. Nature Career Feature (2020) 3 Shift learning. PhD survey - Nature collaboration. (2019) 4 Woolston C. Nature. (2017) 5 Petrie K. et al. Vanderbilt School of Medicine. (2020) 1 1 3 2 2 5 IMMpress Vol. 11 No. 1 2023 9
Which of the following steps were taken to help advance your career?
Have you ever recieved help for anxiety or depression linked to your studies?
From HOAX
The truth behind the Alzheimer’s Disease scandals to HYPERBOLE :
InAugust 2021, a citizen’s petition was filed against the company Cassava Science, accusing them of egregious misconduct and data falsification. The topic of this misconduct: an Alzheimer’s disease drug that was poised to continue to phase 3 clinical trials. The petition called for a pause in trial progression to prevent patients from being treated with a drug based on potentially fraudulent research.
Alzheimer’s disease is a devastating progressive form of dementia that is steadily rising in prevalence in our society. Despite decades of research on this disease, scientists have still had little to no success in finding an effective treatment. Much of the research in the field has been focused on abnormal aggregations of the protein amyloid beta (B) that forms plaques in the brains of patients and are thought by many to cause disease formation. However, Cassava Science claimed that they had found a novel target for Alzheimer’s disease, a protein called Filamin A, which they stated was involved in amyloid B signaling. They developed their drug, Simufilam, in order to stabilize this protein and prevent disease progression.
Shortly after the original petition, several whistleblowers came forward regarding false data published by Cassava Science. They state that “Cassava scientists repeatedly claim to have conducted seemingly undoable experiments.” On top of the methodology concerns, the preclinical data has come under scrutiny for potential falsification of images, particularly Western blot data.

Western blot is a technique used by scientists that can detect the presence of a particular protein. Bands of proteins are identified both by how far they travel down a gel and if they are recognized by an antibody that can detect the presence of that protein. Although incredibly useful when used correctly, as most scientists do, it is also one of the most common techniques where data falsification has been discovered in research. Manipulation of a gel can often be detected by scientific sleuths. In a paper examining the prevalence of ‘problematic figures’ in biomedical research, investigators found 782/20,261 (3.8%) papers with Western blot data examined had at least one inappropriate duplication in the image. At least half of the images seem to show purposeful signs of data manipulation.
This is an incredibly alarming finding in the scientific literature. Researchers depend on the trail of knowledge from those that came before them in order to guide them down their own new avenues of research. How can scientists trust the preceding literature if the possibility of fraud is prevalent and growing?
Fortunately, scientific misconduct and hoaxes are receiving growing attention from journals. And with this new awareness, hopefully there will be more stringent review of papers before publishing to keep the literature to high standards. In line with this, the number of retracted articles
10 IMMpress Vol. 11 No. 1 2023
has increased in recent years along with increasing transparency about the process from editors. Although retracted papers do not always result from scientific misconduct, fraud does account for about 60% of these incidences. Additionally, relatively few authors are the culprit for a large number of retractions related to misconduct. Data published by Cassava Science has led to several paper retractions, most of which are from just two authors affiliated with the company.
Unfortunately, when it rains, it pours, and the hits just kept coming in the Alzheimer’s disease research field. During the investigation into Cassava Science, expert examiners were led to a 2006 Alzheimer’s disease paper published by Sylvain Lesné in the lab of Karen Ashe. This paper claimed to have identified a novel oligomer (variant) of amyloid B that when injected into mice can lead to memory decline even in the absence of neurodegeneration. However, the examiners found bands in the Western blot that appear to have been duplicated, meaning the band identifying the oligomer of interest could have been falsified. Subsequent investigation has found 20 of Lesné’s published papers from his time as a principal investigator seem to contain suspect images, half of which concern his identified oligomer.
Unfortunately, scandals such as these can undermine the public’s trust in research and the health care system. Information gets picked up by the media and stretched well beyond the original context. This can completely mislead even the savviest of readers.

Perhaps, all of this came at the perfect time for Lesné’s amyloid B misconduct to be used as a scapegoat for mounting frustrations due to negative data in Alzheimer’s disease clinical trials. Recently, several monoclonal antibody drugs targeting amyloid B plaques have gone through phase 3 clinical trials. Despite robust ability to clear plaques, evidence for slowed cognitive decline has been minimal. One of these drugs, lecanemab, had a phase 3 trial that ran for 18 months and enrolled nearly 1800 patients with early stage Alzheimer’s disease. This drug decreased amyloid plaques in the brain and led to a statistically significant slowing in cognitive decline. This sounds like groundbreaking news; however, the decline was only a 0.45 improvement out of an 18-point scale. Therefore, despite statistical significance, there remain many questions regarding the clinical meaningfulness of these data. Additionally, there were safety concerns due to brain swelling and small brain bleeds. This has left many scientists, patients, families and caregivers wondering if the risks are worth the rewards and where the field should go from here.
Although this incidence of misconduct is egregious and evident, the magazine article that widely publicized this finding (‘Blots on a Gel?’ written by Charles Piller in Science magazine) opted for sensationalism over fact. Piller claimed that this likely erroneous find has “misdirected Alzheimer’s research for 16 years.” He makes it seem as if the entire field of amyloid B research is problematic due to this one paper.
However, the amyloid B hypothesis predates this one paper by over a decade, with the original seminal papers published in 1991-1992. Several of the most distinguished scientists in the field have fought back over this slander of the prevalent amyloid B hypothesis, stating that this one instance of misconduct does not eliminate decades of work and properly conducted research. Although individual scientists may have wasted time and money trying to replicate a result generated from probable misconduct, there are no clinical trials targeting this oligomer and it has not been propagated by the literature due to lack of confirmation. Therefore, the amyloid B hypothesis is not under any serious threat. Despite this, numerous media outlets have picked up the story and misconstrued it with clickbait headlines such as “Leading Alzheimer’s theory undermined: Did tampering waste 16 years of research?”
Where does that leave us readers, scientists, and science enthusiasts? It can be hard to reconcile egregious misconduct in the biomedical sciences. In a world of publish or perish, one can perhaps understand where a desperate scientist could get the idea to falsify their data. However, we as a scientific field have to demand better of ourselves and those around us. Research integrity is not only important, but vital to the safety of patients, the advancement of treatments and the validity of the scientific field. We must pressure journals to better validate articles before publishing. And as always, we must be cautious of the information given to us and be thorough in our critique of any media we consume. Despite the scandals, scientists remain strong in their conviction to continue advancing treatments and knowledge in the Alzheimer’s disease field. These instances of misconduct can and will be overcome.
`
“Cassava scientists repeatedly claim to have conducted seemingly undoable experiments.”
IMMpress Vol. 11 No. 1 2023 11
- Meghan Kates
POPULAR SCIENCE who gets to have an opinion?
During the COVID-19 pandemic, terms like “public engagement” and “accessible science” became buzzwords in conversations surrounding the way health officials communicated with the public. It’s a difficult spot to be in. We recognize that communication with the public and the ability to summarize complicated scientific jargon for a lay audience is an important part of scientific communication. However, what happens when this is taken advantage of and scientists’ own words are used against them to fuel mistrust and false information? Are scientists causing more harm than good by attempting to communicate with their audience so readily? Who gets to know what’s going on, and should the public be included?
Issues of mistrust between the public and scientific community have always been an issue, but the COVID-19 pandemic really brought them to light. While it was great for the scientific community that new COVID-19 research was being conducted and published so readily, it also meant that from the public’s point of view, there was a constant barrage of new information, some of which did not align with each other. Perhaps one of the largest-scale examples of this was the issue of hydroxychloroquine as a COVID-19 therapeutic. In 2020, a group in France published a small study suggesting that hydroxychloroquine had potentially therapeutic benefits for COVID-19 treatment. Many scientists immediately spoke out against
using this drug as an anti-viral, citing the potential adverse effects. However, it was too late. There were already enough prominent figureheads who had picked it up for it to gain traction. As hydroxychloroquine gained popularity, pharmaceutical companies and private practitioners took advantage of this, increasing the drug’s production. Some studies also showed an increase in prescriptions for hydroxychloroquine from private healthcare practitioners after the article was published. Not long after, multiple follow-up studies were published to show that this drug had little to no positive impact in the context of COVID-19, with one study even indicating that it had potentially very harmful effects. However, by this point, the damage had been done. There was a shortage of hydroxychloroquine in 2020, directly impacting the health of patients with arthritis and other inflammatory immune disorders who relied on this drug. There have also been many people who took advantage of the hype and subsequent retraction of this drug to begin conspiracy theories or say that the scientific community cannot be trusted.
Issues surrounding miscommunication in science are not new. For example, concerns surrounding genetically modified organisms (GMOs) as food have been a global conversation piece for decades. On the one hand, scientists and those in the food production industry are avid proponents of using biotechnology to help meet
12 IMMpress Vol. 11 No. 1 2023
the growing food demands of an ever-growing global population. On the other hand, a sizeable proportion of the general public believes that these genetically modified foods are unsafe to eat. In fact, a 2020 survey revealed that 67% of adults believe that scientists do not fully understand the impacts of GMOs on our health.

Another topic which often brings forth issues of trust in science is climate change. The interesting thing about climate change is that we are able to see it. Unlike other sciences, people all over the world can see with their own eyes the negative impacts of a rapidly changing atmosphere. However, when it comes to supporting policies that will help stop or reverse the impacts of climate change, a large proportion of people do not support such endeavours. One study conducted by the World Economic Forum found that there has been a steady increase in the amount of research paid for by private industries in the last decade. However, people are already skeptical of research paid for by the private sector. This puts scientists in a position where they need to work even harder at creating transparency and developing trust. There is also the issue of political and social identity. Particularly in more heterogenous countries such as Canada and the United States of America where identity politics is pervasive, it is difficult for people to reconcile science that disagrees with the political alliance they have set themselves up with.
One would think that the most logical way to create trust would be increased openness and transparency by sharing information. However, scientists are human, and when inevitably, an error is made, this may only harm the mission of creating trust. There is a Catch-22 scientists find themselves in. Information and important scientific breakthroughs must be shared in order to influence political mandates that will aid in rectifying issues such as climate change and the transmission of a deadly virus. However, the more information that the public is given, the more ammunition they have to throw at scientists as evidence of their untrustworthiness when inevitably there is a change in the literature. Perhaps the answer is not to bombard the
public with information but rather to encourage the public’s engagement in the scientific process. Public engagement in science differs from science communication in that it encourages the public to participate from the beginning. Rather than conducting the research and disseminating the results, public engagement encourages the communities and individuals that are impacted by the issue at hand to form meaningful relationships with the scientists that are studying them. This gives the public a sense that they are involved in the scientific process and encourages conversation about the benefits and risks involved in research. It also ensures that the public and scientists are on the same page, and that people are aware that research may not necessarily answer the question at hand, but that it will provide more information. The public’s view of scientists will not change overnight, and it definitely will not change if scientists are not willing to adjust the way they interact with the public. This is a relationship that is crucial to the health and safety of us all. The COVID-19 pandemic was an opportunity for a reset, and we must be willing to learn from our previous mistakes if we hope to change anything in the future.
IMMpress Vol. 11 No. 1 2023 13
- Salma Sheikh-Mohamed
Science Fiction or Reality
Where do genetically modified organisms belong?

Evolving agricultural practices to meet food demand can be traced as far back as 12,000 years ago, when the agricultural revolution resulted in the transition of human populations from nomadic hunter-gatherer societies to food-producing settlements. Beginning with the domestication of cereals and legumes, and then animals, humans during this neolithic era were no longer constrained by the carrying capacity of the land but could now produce seemingly endless supplies of food to support a growing population. Around 8000 BCE, the first instances of humans genetically modifying crops and animals were recorded, through traditional methods like selective breeding of these species with desirable traits and crossbreeding two different crops or animals to produce hybrid varieties with increased vigour. Fast-forward to the nineteenth and twentieth centuries, following Gregor Mendel’s pioneering discoveries in the field of genetics, and James Watson and Francis Crick’s structural determination of DNA, genetic engineering emerged to manipulate the DNA of organisms, to not only improve our understanding of biology, but also to drive efficient, large-scale production of food and medicine.
Natural methods of crossbreeding can take many years and relies on less-controlled genetic manipulation that could have unwanted effects on other traits in crops. The introduction of genetically modified organisms (GMOs), however, allows for specific insertion of useful genes into a crop or animal without disrupting other desirable traits that had been carefully selected for over millennia. With the advent of GMOs, we can now combine the genes of two completely unrelated organisms to create completely new hybrids that would not occur naturally. For example, the insertion of bacterial genes into corn, soybean, cotton, and tobacco allows these plants to produce their own pesticide, reducing the cost of farming these crops by improving yield and elimi-
nating pesticide use. In addition to improving crop yields, these genes can enhance the nutrition of crops; a notable example is “Golden Rice,” which produces 20-fold more beta-carotene than other rice varieties.
Cost reduction, improved yield, increased nutrition, and more sustainable farming practices all sound enticing, especially to developing countries where the increased prevalence of poverty coincides with starvation and malnourishment. According to the World Health Organization, 90% of the scientific community believes that GMOs are safe to use, yet only approximately one third of consumers share this sentiment. Why the disparity?
The concept of the GMO is shrouded in hesitancy, generating concerns about causing allergic reactions, antibiotic resistance, toxic effects on the body, mutations, effects on pregnancy, and potential gene transfer to the consumer. While these are valid concerns, to date, no negative effects on human health or the environment have been reported for GMOs on the market. As with many new biotechnologies, much of the hesitancy is fueled by misinformation and mistrust in large corporations, and a reluctance to “play God”. To curb these doubts, governments must reduce the legal and administrative hurdles that farmers face when adopting GMO practices, and ensure consumers have transparent access to relevant health- and environment-related information.
The truth of the matter is that nearly 811 million people are suffering from malnutrition, and humanity holds in its hands a powerful technology that can tackle these oncoming challenges of food insecurity to transform the way we live off the land. As GMOs take flight, we are entering an age where the line between science fiction and reality becomes increasingly blurry.
- Deeva Uthayakumar
As the human population explodes and extreme weather conditions surge due to climate change, we place an increasing burden on our agricultural industry, which is in dire need of innovation.
14 IMMpress Vol. 11 No. 1 2023
Ideas Worth Spreading –
The Power of Accessible Scientific Outreach Initiatives

Technology. Entertainment. Design.
The convergence of these three themes into short, influential talks forms the basis of TED conferences – one of the most successful and widespread scientific outreach initiatives in recent decades. As a nonpartisan, non-profit company formed in 1984, TED is dedicated to communicating transformative ideas in highly engaging and accessible formats.
So, what exactly makes a TED Talk? Hosted at TED conferences worldwide, these talks are unique in their design. Invited speakers must formulate a talk which effectively communicates complex ideas and concepts to a broad audience. With a strictly enforced 18-minute window to disseminate one’s best ideas on the TED stage, breaking down these talks into digestible pieces that are intuitive and easy for non-specialists to follow is no easy task. The successful result is an informed and inspired audience with a motivation to further spread those ideas and engage with the world in new, previously unimagined ways.
TED breaks down traditional barriers to accessing information and has transformed the way that knowledge is disseminated. Widely available and free of cost online, these talks can be accessed by anyone regardless of geographic location. To promote global accessibility, a community of TED Translators volunteer to translate and subtitle these talks in up to 115 languages. The creative expansion in the form of TEDx, independently organized events hosted in a similar format to traditional TED conferences, has expanded the influence of these talks to cities around the world. As research has shown that accessible information and effective scientific communication is associated with greater public participation in policymaking, TED plays a large role in creating a more knowledgeable and engaged society.
With an early emphasis on technology and design, the 1984 inaugural TED conference was hosted in Monterey, California and featured one of the first presentations of Apple’s Macintosh computer. TED has since widely diversified its reach, often hosting speakers who are experts in almost all fields –from science to business to global issues. Featuring recipients of the Nobel, Fields, Pulitzer, Oscar, and Grammy
awards, the TED stage has attracted some of the world’s best and brightest.
Since the first TED Talks were posted online and shared with the world in 2006, popularity of this initiative has soared to new heights. One of the earliest posted talks titled “Do Schools KillCreativity” delivered by Sir Ken Robinson, remains the most watched TED Talk to date with over 74 million views. Robinson, previously a British Professor of Arts Education at the University of Warwick, proposed a carefully crafted case for an education system that cultivates creativity through acknowledging multiple types of intelligence. In equal parts entertaining and moving, this talk captured the interest and enthusiasm of a global audience.
What does the power of a successful scientific outreach initiative like TED do? It creates and empowers a community of people who are passionate about scientific discovery and the positive impact it can have on the world. Cultivating a culture of curiosity and innovation will be crucial for advancements in our understanding of the world while addressing the challenges society will face in years to come.
IMMpress Vol. 11 No. 1 2023 15
- Matthew Bianca
Social media
The shiny new tool for science reporting
Social media has revolutionized the way we consume information, and science is no exception. With just a few clicks, we can access the latest scientific breakthroughs, research findings, and discussions from experts in the field. According to a 2021 survey by the Canadian Internet Registration Authority (CIRA), 69% of Canadians are active on social media platforms, making it an excellent avenue for science communication.
Social media is particularly appealing to younger generationsaccording to a survey conducted by the Ontario Science Centre, young

Canadians between the ages 1834 have a higher tendency to use social media for staying informed about science, with 56% of them saying they follow science-related accounts. In contrast, only 44% of those over the age of 35 use social media for science news. These statistics highlight the importance of social media in science communication, particularly in engaging younger audiences.
In a recent study analyzing the use of Twitter in Canada to promote scientific research, the authors found that Twitter has become a critical platform for sharing scien-
16 IMMpress Vol. 11 No. 1 2023
69% of Canadians are active on social media platforms, making it an excellent avenue for science communication.
tific research and engaging with the public. The hashtag #scicomm is frequently used, particularly in academic circles, to promote science communication and engage with a broader audience. The use of social media for science communication has also been successful in many ways, for example by increasing the visibility of underrepresented groups in science, like women. A 2019 study published in the journal PLOS ONE analyzed the Twitter activity of over 1,600 scientists in Canada and found that women who tweeted about their research were mentioned in scientific papers more often than men who were equally qualified but tweeted less or not at all. It has also allowed for the rapid dissemination of information about emerging scientific findings and research, increasing public awareness, and promoting public engagement with science.
Another significant advantage of social media is the ability to reach beyond the academic circle. However, it also comes with its own set of challenges, particularly when it comes to the trustworthiness of information sources. According to the 2021 CIRA survey, 53% of Canadians trust universities as a source of reliable scientific information, while only 23% trust social media platforms.
Unfortunately, the ease of reporting with social media has allowed misinformation to spread faster than wildfires. Social media platforms use algorithms to personalize the content that users see on their feeds. These algorithms consider users’ behavior, including what they like, comment on, and share, to determine what content to show them. While this can create a more engaging and personalized experience for users, it also has some downsides, creating echo chambers where engagement in misinformation exposes individuals to further misinformation that confirms their existing beliefs.
Social media’s role in spreading misinformation and conspir-
acy theories became significantly prominent during the COVID-19 pandemic and has contributed to the vaccine hesitancy and public distrust of government and public health institutions. According to the Canadian Science Policy Centre’s survey, 34% of Canadians believe that the government is hiding information about the pandemic, and 37% believe it is a hoax, which may be linked to the spread of misinformation on social media. These statistics highlight the risks of using social media for science communication and the need to counter misinformation.
The vast amount of information available on social media can make it difficult to distinguish credible sources from unreliable ones. Thus, when encountering science news on social media, it is crucial to critically evaluate the source of the information. Non-scientists should take the time to consider the credentials of the individual presenting the information. If the author is not a trained scientist or expert in the field, their claims may not be accurate or reliable. Secondly, non-scientists should be cautious of sensational or exaggerated headlines that may be designed to grab attention but are not necessarily supported by underlying science. It is important to read beyond the headline and examine the content of the article or post to determine whether the claims made are supported by evidence from credible sources.
Unfortunately, according to the 2018 Canadian Science Literacy Survey, 43% of Canadians have a low level of science literacy, while 25% have a high level of science literacy. This underscores the need for clear and accurate science reporting that is accessible to all Canadians, regardless of their level of scientific knowledge.
Social media has become a valuable tool for science communication. However, it is important to recognize the potential risks of using social media, including the
spread of misinformation. Non-scientists should critically seek out credible sources that are written in plain language and that provide context and background information. Scientists and science communicators should continue to leverage these platforms to engage with the public, increase awareness and understanding of science, and foster a more informed and scientifically literate society.
From Twitter activity of over 1,600 scientists in Canada, women who tweeted about their research were mentioned in scientific papers more often than men who were equally qualified but tweeted less or not at all
43% of Canadians have a low level of science literacy, while 25% have a high level of science literacy
Young Canadians have a higher tendency to use social media for staying informed about science
23% of Canadians trust social media platforms as a source of reliable scientific information
IMMpress Vol. 11 No. 1 2023 17
- Alara Tuncer
Dr. Derek Clouthier is currently working at AstraZeneca as a Global Medical Affairs Lead in Oncology. After completing his PhD in 2015 under the supervision of Dr. Tania Watts, Derek pursued various unexpected opportunities that led him to successful positions in major pharmaceutical companies. In this interview, we had the privilege to discuss with Derek his career, his views on the public perception of science and advice he has for current graduate students.
clinicians gave Derek the tools for his future positions as a Field Medical Advisor at Pfizer and Medical Science Liaison (MSL) at AstraZeneca. Derek now holds the position of Global Medical Affairs Lead (Oncology) at AstraZeneca where he is responsible for the development and execution of AstraZeneca’s global medical affairs strategy and launch plans. Derek ensures integration across key regions and markets with cross-functional stakeholders in areas including research and development, regulatory, commercial, and market access. He is also tasked with organizing the global medical team through delivery of current publications, training, and external resources.
For three years, Derek focused on genomic and immune biomarker discovery for early phase trials with immuneoncology agents. His main responsibility was to strategically gather different stakeholders, such as oncologists, pathologists, nurses, and scientists, and promote an environment where the team would be motivated to work together. This experience of communicating data and clinically relevant information between basic scientists and
Improving the public perception of science as a scientist
Working in the field of medical affairs, Derek speaks on being trained to gather insights into how physicians


“All of a sudden, I was in a career that I never would have expected.”
“My time in the pharmaceutical industry has been sufficient to give me a holistic view of how medicines are commercialized in Canada and around the world. In the end, I think that cycling accident led me down an unplanned, but very exciting, fortunate, and rewarding path!”
18 IMMpress Vol. 11 No. 1 2023
interpret clinical data and what evidence gaps exist for addressing unmet clinical needs. This process is repeated until stakeholders have the evidence they need to make an informed assessment. Derek believes this process should be applied to the general public by actively exploring the questions they have regarding the scientific research we are conducting. It is important to bridge the gap by understanding how we can serve the public and bring their perspective into the work that we do. This discussion is built on Derek’s experiences with two main groups of skeptics in the face of science: “those who seek to understand, learn, and challenge scientific findings in a genuine and balanced way and those who challenge the evidence because it contradicts what they want to believe is true”.
Derek reflects that although the second group has always existed to challenge ideas even before COVID-19, they have gained an unlimited access to a self-validating newsfeed to entrench their beliefs in recent years. This group of skeptics can now “organize themselves to amplify their voice and influence in the community”, which would also include the media and political leaders. Issues may arise as this group of people begin misinforming the moderate members of the public who may be uncertain about who or what to trust. Involving the general public in the journey towards scientific discovery is one way to ensure the evidence we generate can answer questions in a way that is meaningful to them and create opportunities for public-centric scientific discussions.
The importance of being “agile” in your studies and career
Undoubtedly, an integral part to Derek’s success was his PhD education. Derek reflects on the various aspects of obtaining a PhD that will help students grow as a life-long learner. These include the translatable skills gained indirectly such as project management, storytelling, and distilling complex information to identify actionable insights. One crucial skill that Derek wants to highlight is the ability to have “agile” ways of working. In the pharmaceutical industry, many workshops are held to explore this idea of being agile. Essentially, this concept is an approach used in project management to deliver results continuously while incorporating changes from each iteration along the way. Fortunately, this is a fundamental mindset that many PhDs have already developed during their time in graduate school.
Derek first heard the term “agile” in 2018, but he has incorporated the idea since the beginning of his PhD with Dr. Watts. At the end of his first year in 2011, Derek and Dr. Watts sketched out a plan of what his thesis chapters would look like with clear expectations for experiments, models, and publications. Despite surprises along the way, Derek never lost sight of his main goals and made steadfast iterative updates to the body of evidence that would later become the

story and foundation of his thesis. Regardless of the project Derek is working on now, he would always have a clear vision on the bigger picture and knew how to prioritize his efforts to maximize the impact of the work produced. This way of thinking and approaching big projects is a major contributing factor to Derek excelling as a leader in all his roles in the industry.
Advice as a successful alumnus to current graduate students
Although Derek’s trajectory has been unplanned and “messier” than expected, he has gained a valuable outlook on how to maximize the graduate school experience. Derek speaks on learning the difference between pushing yourself and pressuring yourself after graduating. In retrospect, Derek wishes he learned to “take his foot off the gas” to make more time for the people in his life and to fully enjoy the freedom available in in an academic work environment. He also warns against letting your assumptions or doubts get in the way of trying things. Derek comments on how scientists are hard-wired to feel the need of overcoming some deficiency to survive in the world, but it’s simply untrue. It’s important to get the perspectives from people in positions you desire, however, you should also take time to reflect on yourself and assess the types of environments you will do well in.
“Without even knowing you’re doing it; your PhD is one of the best times to master agile ways of working. Stay focused and refer to this framework to hold yourself accountable to your deliverables and make sure that your time and efforts are aligned to what you need to graduate.”
“When you’re choosing your trajectory, think about what you want to achieve with the job rather than what you need to be good at it. At the end of the day, the job you end up with is only as meaningful as what you want to do with it.”
IMMpress Vol. 11 No. 1 2023 19
- Maye Cheng
Working as a Scientist
As budding researchers, students of science and technology are met with the same old question regarding their choice of career– “Academia or industry? Where would you like to work?”
teaching courses. The teaching component of your work would depend on your position and your institution. But contributing as a teacher at the institution in some capacity is required by most institutions when evaluating tenure or promotion, which are highly competitive processes. Taking up administrative roles is more on a voluntary basis than a requirement, but it certainly helps your position in the institute as you get more involved in the behind-the-scenes of the management. Mentoring your students is an essential investment made for the well-being of your research group and helps increase the overall productive research output. You need to give attention to detail and provide constructive feedback and guidance on various aspects of your student’s research work – from experimental design to analysis of results. This also requires a good amount of reading the literature and keeping up with the current knowledge in your field.
While these two choices are not the only available career options anymore, they are indeed the more conventional paths that most young scientists tend to take. One may argue that, irrespective of the system you are in, at the end of the day, you are still a scientist – a person in pursuit of objective scientific truth. While this is not false, it is a distorted sense of the truth. One scientist in an academic institution and another scientist working for a company may be trying to answer the same broad fundamental question – but the lens through which they look at the question is heavily influenced by the system that houses them.
Working for an academic institution as a principal investigator (PI) involves taking care of a group of scientists. It is terribly similar to running your own company. You have employees that can be students, research associates, lab technicians and post-doctoral researchers. One of the main tasks as a scientist at an academic institution is to fund this company of researchers. This includes applying for grants, communicating, and propagating your group’s work to the masses, budgeting and managing your team.
Apart from this, you are involved in mentoring your students, taking up administrative tasks for the institution and
As a scientist, working in industry is similar to working in academia in many ways. You still need the same attention to detail and scientific prowess. However, depending on your position, it is likely that you will be working on the bench and doing experiments first-hand. As a scientist in industry, you will be part of a group of scientists which is led by a manager. You will be directly answerable to the manager who is responsible for reporting your team’s research progress to higher-ups. This setting might sound similar to working as a research associate in a research group in academia where your manager may be your PI. However, there are still several key differences between the two settings.
As a research associate or a PI in an academic institution, you are likely to be working majorly, if not entirely, in an independent manner. In contrast, there is more of a hierarchical structure to your position in industry where your manager is closely monitoring your progress. This may work better for people who like working towards a short-term goal within a limited time limit. In industry, you are more likely to be closely collaborating on the research project that is assigned to your whole team rather than you as an individual.
The second significant difference is the level of flexibility in these two jobs. This may be both in terms of working hours as well as the research questions you pursue. The underlying factor that gives rise to this difference is the motivation behind the research being conducted in academic and industrial settings. Industrial units are first and foremost, businesses, and their primary motive is to make profits. If the research being conducted by your team is not directly converting into profits, your question may be changed, and you may have little to no control over this. Moreover, research is largely


20 IMMpress Vol. 11 No. 1 2023
in Academia vs. Industry
driven by the company’s corporate goals and there are set milestones. Achieving these milestones is important for positive performance reviews and salary hikes.
In academic institutions, the foremost goal is to publish peer-reviewed scientific articles. This helps improve the world ranking of the institution and helps get government and non-government funding to further improve research and teaching at the institute. Money is important in both settings but in academia, it is more of an indirect outcome of the primary goal, which is helping push the boundaries of our current knowledge. Thus, research projects in academia can be more obscure and long-term as they encompass a larger scope of exploration. One of the most important benefits of being a scientist in academia is being able to pursue research questions primarily, if not entirely, based on your interest. This is unfortunately not easily possible in an industrial setting.
This ties into the flexibility of working hours. In academia, the emphasis is on exploration of the scientific question which realistically, may take an indefinite amount of time on a given day. So, it is up to the scientist to decide their own working hours to best answer the research question. In industry, the emphasis is on efficiency, i.e., how can you get the most amount of useful information in the least amount of time within the limited time framework of your assigned project. Thus, most companies have set working hours – typically from 9am to 5pm. This ensures that each day is accounted for, efficiency can be measured and hence, improved. Money earned in profits is also often reinvested into buying latest high-throughput technology and equipment. Such fast technology upgradation in academia is harder because the return on this investment is accumulated over long periods of time.
In industry, fixed working hours and a focus on efficiency means that you are more likely to get immediate rewards for your hard work. Promotions and bonuses are given based on your efficiency which is regularly monitored through performance reviews. Given the relatively narrower and streamlined scope of the projects, this is more achievable than publishing peer-reviewed articles in broader self-directed projects. There are additional benefits to working for a company such as vacation days, more health benefits and paid leave, especially paid maternity leave.
The flexibility in working hours and self-direction of your research questions in academia come at a cost – your salary. Academia is infamous for underpaying its researchers at
all levels, especially younger scientists. In Canada, research associates and post-doctoral fellows get paid an average salary that is at least thirty percent lower than an equivalent scientist position in a company. Your pay and other benefits increase as you go to higher positions in academia and as you gain experience. However, this is largely based on the quality of the research articles you publish and not necessarily on the number of hours you put into your work. One may argue that these two factors would likely correlate. However, scientific discoveries are not an outcome of a linear process and hence, it would be unfair to assume this.
All in all, industry and academia are both wonderful places to further your pursuit of scientific knowledge. They are completely different but a whole lot similar. You might be a better fit for one over the other and that would depend on your priorities and who you are as a person.
- Manjula Kamath
IMMpress Vol. 11 No. 1 2023 21
The Slow decay ofour reliance Inscience

Y
ourfriends,yourfamily,yourco-workers.Yournext-doorneighbourof10 years,arandompasserbyonthestreet.Throughoutthepandemicwehave livedwiththerealitythatanyonecouldspreadCOVIDtous,eveniftheyare notshowingsymptoms.Toprotectourselvesfrominfection,wehavealso neededtorelyontheexpertiseofstrangersintheircapacityasscientists.Itcanbehard totellwhomtotrustthesedays,andthesteadyreleaseofmisinformationmakesthis evenmoredifficult.ButhowmuchofaneffecthasthishadonCanadians’trust?Has pandemicfatiguecausedthepublictolosetheirfaithinscience?
EarlierthisyearCanadiancommunicationsagencyProofStrategiesconductedand releasedtheiryearlyCanTrustIndex,ameasureofCanadians’trustinvariousgroups overtime.Whenaround1,500Canadianswereaskedaboutwhichcategoriesofpeopletheyconsideredtobesourcesofreliableinformation,themosttrusted groupsoverallweremedicaldoctors,scientistsandfriends&family. Celebrities,influencersandpoliticianswereamongtheleasttrusted groupsin2023.However,thepublic’strustinthesegroupshaschanged significantlyoverthepastthreeyears.Trustindoctorsfelleachyear(81% ofrespondentstrusteddoctorsin2021;78%in2022;73%in2023)as wellastrustinscientists(77%in2021,75%in2022;69%in2023), whereastrustinfriends&familyactuallyincreased(64%in 2021,65%in2022,68%in2023).Thereportalsosuggests thatCanadianstrustmajorpublichealthfigureslikeCanada’s ChiefMedicalOfficerofHealthandtheWHOlessthanindividualdoctorsandscientistsforCOVID-19information.
Thisreporttellsusthatpublictrustinscienceremains stronginCanadabutiserodingeachyear,atrendthatbegan beforethepandemic.Itisaconcernthatisbeingfeltinseveralcountriesaroundtheworld aswell.AstudyconductedinGermanyfound signsofwaveringtrustinscienceamongGermansandthatrespondentshadthreekeyreasonsfortrustingordistrustingscientists. Thosewhotrustedscientistscitedtheirexpertise(i.e.scientistsarehighlyskilledand knowledgeableintheirfield),integrity(i.e. scientistsholdhighstandardsandaregenerallymoralpeople), andbenevolence (i.e.scientistsconductresearchfor thegoodofhumankind).Alternatively,thosewho didnottrustscientistscitedtheirexpertise(i.e.scientistsmakemistakes andoftendisagree witheachother), integrity(i.e.scientistschangetheir resultsastheysee fit),andbenevolence(i.e.scientists areonlyafter moneyandfame).
22 IMMpress Vol. 11 No. 1 2023
Oneexplanationforthislackoftrustistherecentparadigmshiftinhowwereceiveandjudgeinformation.The numberofpossiblesourcesofnewsandinformationhasdramaticallyincreasedfortheaveragepersonassocialmediausagebecomesmorecommonplace,anditisbecomingharder totelllegitimateinformationfromfakenews.Thepolitical climateinmanycountriesisalsobecomingincreasinglypolarizedinrecentyears,leadingmanypeopletodistrust sourcesofinformationiftheysuspectitinvolvesanunwelcomepoliticalagenda.Evenreputablenewsoutletshavebeen calledoutforreleasingmisinformationduetothefinancial incentivizationofreleasingsensationalarticles,oftenatthe expenseofjournalisticstandards.
Thecostofgrowingmistrusthasalreadycausedincredibleharmtoindividualsandcommunities,whichwilllikely onlyworsenifmistrustcontinuestoincreaseovertime.The CouncilofCanadianAcademiesestimatesthatmisinformationcontributedtovaccinehesitancyin2.35millionCanadiansin2021,costingthehealthcaresystem$300millionin hospitalstaysandleadingto3,000preventabledeaths.Some vaccine-preventablediseaseslikemeasleshavere-emergedin CanadaandtheUSlargelyduetomisinformationcampaigns onsocialmedia.Otherareasofsciencealsosufferfromthe consequencesofpublicmistrust.Climatechangeskepticism issowidespreadthatithascontributedtostalledclimateactioninmanycountriesandattheinternationallevel.The adoptionofgenetically-modifiedfoods,windenergy,andnuclearenergyhasbeenopposedbypublicandpoliticalgroups withmisleadingclaimsaboutthesetechnologies.
Misinformationisnottheonlycauseofmistrustinscience,however,asthehistoryandcontemporarycultureofthe scientificestablishmenthavealsocontributedtopublicmistrust.Thepastexploitationofwomen,raciallydisenfranchisedgroups,andLGBTQ+people(tonameafewgroups) inresearchhasdamagedmanypopulations’relationship withinstitutionalscience.Theequityissuesthatstillexistin STEMtodaykeepthesecommunitiesontheoutsideorthe peripheryofthesystem.Sowhilemisinformationplaysasignificantroleingrowingpublicmistrust,fundamentalchanges needtobemadefromwithinthesystemtofullyaddressthe problem.
Scientistshavetraditionallyusedaninformationdeficit modeltocombatpublicmistrustinscience,whichassumes thatpeopledistrustsciencebecausetheydonotunderstand it,thereforethesolutionistomakethemunderstand.Unsurprisingly,throwingfactsatpeopleisnotthebestwaytoget themtoagreewithyou,andthismodelhaslikelycontributed tothepatronizingviewofscientiststhatmanymembersof thepublichold.Whileitisimportantforthepublictounderstandthetechnicalaspectsofscience,itisarguablyeven moreimportantforscientiststounderstandtheemotional perspectivesofpeoplewhohaveturnedawayfromthesystem.Compassionatescientificcommunicationwillbecome essentialfornavigatingoutofthisongoingcredibilitycrisis. Byengagingwithskepticsonahumanlevelandaddressing therootcausesofmistrust,thescientificcommunitymaybe abletostoporreversethepublic’scrumblingconfidencein science.
-JamesPollock

M e d i ca l d o cto rs 8 1 78 73 77 75 6 9 6 4 6 5 6 8 Sci e n ti sts F r i e n ds / FA M I LY 2 0 2 1 2 0 2 3 2 0 2 2
vaccinehesitancy
$300millioninhospitalstays leadingto 3,000preventabledeaths.
The public’s trust in these e categories of people for reliable information
Misinformationcontributedto
In2.35millionCanadians in2021, costingthehealthcaresystem
IMMpress Vol. 11 No. 1 2023 23
Whenscientific terminology becomesjargon

Scientistsareusedtocommunicatingwiththeirlab,classmates,andmorebroadlywith ascientificaudience.Buthave youevertriedtoexplainyour project,oranyspecificscientificconcept tofriendsandrelativesfromanon-scientificbackground?Ifyes,youwillprobablyhavenoticedthatsomeconceptsthat seemeasytounderstandforyoumight bemorechallengingforthisnon-scientificaudience.Oneofthemostimportantreasonsistheuseofscientificjargon,i.e.“thespecializedlanguageofa trade,profession,orsimilargroup”,or inthiscasethespecificterminologyused inthescientificcommunity.
Themoreweworkonatopic,the morewebecomeexpertsaboutit.And themorewebecomeexpertsaboutit,the harderitgetstorememberwhatwe knewbeforewestartedandtoadaptour communicationaccordingly.Thisiseven moredifficultasthescientificcommunityisusedtoanextremelypreciseand specificwaytocommunicate,whichdifferssignificantlyfromeverydaycommunication.
Whiletheimpactoftheuseofterminologymaynotbetooimportantata personalscale,itisofutmostimportance thatscientistsaremindfulofnotusing jargonwhenaddressingabroaderaudience.Byimprovingourcommunication, weimprovethepublicunderstandingof science,andencourageresponsesthat aremorescientificallyfavourableand reasonable.Thishasshowntobeofparticularimportanceforvaccinationamid theSARS-CoV2pandemics:effective communicationsurroundingthemechanismofactionofthevaccineandthe
proofofitsefficacyhadtobedelivered tothepopulationinordertoachievea higherrateofvaccination.
Onanotherlevel,communicationis essentialinthecontextofhealthcare whendeliveringadiagnosis,orcounsellingapatient,thatinvolvesinmany casesmakinglife-changingdecisions. Severalstudieshaveshownthatpatients areverylikelytomisunderstandtheir medicalsituationandtheiroptionsdue totheuseofinsufficientlyexplainedscientificjargon.AstudybyCharpentier et al. classifiedmedicaljargoninsevendifferentforms:technicalterminology(the useofhighlytechnicaltermssuchas “upperendoscopy”),acronymsandabbreviations,medicalvernacular(words suchasanemia,thatmaybefamiliarbut notalwayswellunderstood),medicalizedEnglish(wordslikeprogressive,impressive,positive/negative,thatdon’t havethesamemeaninginthemedical fieldthanineverydaylife),unnecessary synonyms(suchasambulate,oferythema,thatcouldeasilybereplacedby morecommonwords),euphemisms,and judgmentaljargon(suchasfailedtreatment).
So,howcanscientistsreducetheuse ofscientificterminologyandpromote moreeffectivecommunication?Firstof all,consideringtheaudienceweareaddressing:whatistheirbackground knowledge?Second,limitingtheuseof specificterms,inparticularabbreviations,andexplainingtheminfulldetails whennecessary.Finally,makingsurethe audienceunderstandsandhasachance toaskanyclarificationquestionsthat mayberequired.
-JohanneAudouze
24 IMMpress Vol. 11 No. 1 2023
ADisgracedScientist andhis RetractedPapers

Dr.PieroAnversahadbuilta dazzlingreputationasarevolutionaryscientist.Asa professoratHarvardMedicalSchool, hedirectedtheCenterforRegenerativeMedicineatBrighamand Women’sHospital.Healsoservedon theBoardofScientificCounselors,a peer-reviewcommitteethatevaluates thequalityofresearchconductedat NationalInstituteofHealth(NIH).
Aboveall,Dr.Anversawasthepioneerofcardiacstemcells.Hisresearch claimedthatstemcells–animmature celltype–canregeneratetheheartby producingcardiomyocytes(heart cells)alongwithcellsthatmakeup themuscleandbloodvessels.In2001, hislabreportedin Nature thattransplantedstemcellscouldrestoredamagedhearttissueinmicethatunderwentmyocardialinfarction,amedical termforheartattack.Then,theAnversalabshowcasedin NewEngland JournalofMedicine astudyconductedwithhumanpatientsamples. Thisstudyultimatelyshowedthatcells inthehumanadultheartcandivide andgrowuponinjury.Withthesetwo landmarkpapers,Anversashattered theprevailingparadigmthattheadult heartremainsstaticandincapableof regeneration,inspiringclinicaltrials forstemcelltherapyandrousinghope amongpatientswithcardiacfailure.
Howeverin2018,aseriesofpaper retractionsderailedhiscareer,turning himfromesteemedscientisttoascandalousfraud.Anversa’sdownfallhad startedyearsprior,whenscientistsreportedtheirinabilitytoreproduceAnversa’sfindings.Forinstancein2014, Dr.JefferyMolkentin’slabpublished in Nature astudythatdirectlychallengedAnversa’sclaims.Molkentin andcolleaguesshowedthatcardiac “stemcells”areunabletogeneratea significantnumberofcardiomyocytes uponagingorcardiacinjury.Infact, Molkentinandmanyothersquestionedtheexistenceof“cardiacstem

cells”.Suchdoubttriggeredinternal investigationsbyHarvardandrevelationsthatmuchofAnversa’sdata–mostlymicroscopicimages–were heavilymanipulatedoroutrightfabricated.Thistriggeredaseriesofevents thatledtothecollapseofhiscareer. Anversawasforcedtoresign,aclinicaltrialforstemcelltherapywas halted,anda10-million-dollarsettlementwasreachedbetweentheU.S. governmentandBrighamand Women’sHospital(BWH).Withthe costlysettlement,BWHresolvedallegationsthatAnversaandhislabfalsifieddatatoreceivefederalNIH grants.Despitethisresolution, New EnglandJournalofMedicine retractedaformerstudybyAnversaand expressedconcernabouthisother publicationsinOctober2018.HarvardMedicalSchoolandBWHthen calledvariousacademicjournalsto retract31paperspublishedbytheAnversalab.Thescandalthusconcluded witharejectionof“cardiacstem cells,”whichhadonceawedthescientificcommunity.Itimportantlytolda cautionarytaleaboutaresearchcareer builtonfalsifieddata,andthehefty punishmentthatcomesalongwithit.
-JenniferAhn
IMMpress Vol. 11 No. 1 2023 25
BIG
Pharmaceutical drugs have been revolutionary in modern society, saving or extending the lives of billions of people around the world. They have lengthened the average life expectancy in developed countries to over 70 years of age (an increase of 20-30 years) over the past century. However, this gift to humanity has not come without its share of issues. The average price of pharmaceutical drugs has increased by 500% since the 1980s, a rate that is three times greater than increases in prices due to inflation in the same time period. Additionally, thousands of drugs are recalled each year due to safety or efficacy concerns, indicating that faulty products are regularly being approved for public consumption. The culprit of these issues lies no further than the entity creating these products: the pharmaceutical industry.
Pharmaceutical companies, colloquially known as “Big Pharma”, have been known to increase prices of their drugs drastically and unreasonably (a practice known as “price gouging”) to widen their profit margins, resulting in their inaccessibility to many who depend on them. A recent example of this is the ongoing fiasco concerning insulin prices in the United States (US). Insulin is a daily medication required by people with diabetes to control their blood glucose levels. Shockingly, a vial of insulin in the US in 2018 costed six times more than it did in 2000 and was almost five times more expensive than the nearest country’s average

26 IMMpress Vol. 11 No. 1 2023
CORRUPTION IN BIG PHARMA
a glance into the pharmaceutical industry

price. Pharmaceutical companies, like Eli Lilly, have recently come under public scrutiny for these unfair drug prices. As a response to this public outrage, Eli Lilly announced price cuts to insulin of up to 70% in March 2023, a long overdue discount that should help Americans afford their medication. Nevertheless, price gouging still exists for countless drugs driven by a desire to increase earnings rather than care for patient’s health.
Perhaps more egregious than overpriced drugs are how pharmaceutical companies can slip ineffective or harmful products past regulations. The US Food and Drug Administration (FDA) has a rigorous process to approve drugs, which involves ensuring the product is effective and not overtly harmful throughout the preclinical and clinical stages of drug development. However, in the clinical stage, results from manufacturer sponsored trials have historically been biased by either downplaying adverse reactions or overstating therapeutic responses. This misreporting of safety and efficacy makes it easier for a drug to receive FDA approval, allowing for companies to quickly recoup research and development costs at the detriment of consumers. It’s no wonder most lawsuits against pharmaceutical companies have revolved around their drugs being less effective or more dangerous than advertised, leading to unintended side effects or even death of patients.
The largest US pharmaceutical lawsuit was a case against GlaxoSmithKline (GSK) in 2012. GSK was found guilty of marketing 10 drugs for nonFDA approved uses, compensating doctors for prescribing these products, and understating side effects of their type 2 diabetes drug, Avandia. This antidiabetic currently holds a boxed warning, indicating dangerous side effects, due to its high risk for those who have heart disease – information that was withheld from consumers initially. GSK was forced to pay $3 billion in total settlements for their fraudulent business and research practices.
Despite the negative reputation
Big Pharma has garnered, the industry remains incredibly necessary for its contribution to modern healthcare. Big Pharma should be seen as a saving grace with the products they provide; instead, many see them as uncaring and corrupt. Strategies to improve sentiment and integrity in the industry could include creating better legislation on fair drug pricing, increasing external scientific review and regulation over drug development, and separating research funding companies from drug testing to reduce bias. Reaching this point may be difficult, but will be an important shift back to the core purpose of the industry: to make people’s lives healthier and better.
The average price of pharmaceutical drugs has increased by 500% since the 1980s
IMMpress Vol. 11 No. 1 2023 27
- Matthew Wong
advances in
Every cell in your body has a genome, a collection of over 20,000 genes that instruct the body to function properly. Genes are made up of smaller building blocks called DNA bases. With over three billion bases in the human genome, a natural outcome is mutations, when incorrect bases occur. Although many mutations are harmless, some impact important genes and cellular pathways, causing dysfunction and debilitating disease. Gene therapies target the root cause of these previously thought “incurable” diseases by fixing mutations in the affected cells.

Each gene therapy is tailored to the nature of the genetic disease, in terms of how best the genetic mutation can be corrected and which tissues are primarily affected. To reach the target cells, gene therapies can be delivered in vivo (inside the body), through injection into the bloodstream, or at a specific site, such as the muscle, retina, or cerebrospinal fluid. An example of an in vivo gene therapy is LUXTURNA. It delivers a functional version of the RPE65 gene to the eye. RPE65 is mutated in individuals that suffer from inherited retinal diseases, such as Leber congenital amaurosis and retinitis pigmentosa, causing progressive vision loss. This is an example of gene addition therapy which introduces new copies of genes to replace the function of the mutated ones. Other gene therapies can prevent the disease-causing genes from functioning, called gene silencing therapies, such as ONPATTRO. This is a treatment for patients with hereditary transthyretin-mediated amyloidosis, a disease caused by a mutation in the transthyretin gene, leading to ner-

vous system dysfunction and heart failure. Transthyretin is primarily expressed in the liver, and the mutant produces misfolded proteins that clump together, forming aggregates that can build up in the heart and nerves. ONPATTRO is delivered into the bloodstream, where it makes it to the liver and blocks the mutated transthyretin gene from forming disease-causing protein aggregates.
However, in vivo gene therapies cannot feasibly treat all affected cells to restore their normal function, especially if the genetic disease affects multiple organs or the blood system. For blood cell disorders, it’s easiest to administer the treatment ex vivo (outside the body), before transferring the modified cells back to the patients. All blood cells come from bone marrow stem cells, and by isolating and modifying these stem cells directly, essentially all the derived blood cells will also possess the gene therapy. HEMGENIX is a gene therapy for patients with beta-thalassemia. Affected individuals have a mutation in their beta-globin gene and cannot produce functional hemoglobin, which is the component of red blood cells required to transport oxygen throughout the body. HEMGENIX inserts a functional copy of the beta-globin gene into the genome of the patient’s bone marrow stem cells. New red blood cells emerged from these modified stem cells will have a copy of the correct beta-globin gene, capable of producing functional hemoglobin. In addition, other ex vivo therapies can also engineer a patient’s immune cells to become better at recognizing and killing cancer cells.
28 IMMpress Vol. 11 No. 1 2023
gene therapy

After almost 50 years of active research, we’re approaching the golden age of gene therapy, where thousands have been developed, and several are passing rigorous clinical trials to prove their safety and lasting therapeutic effects. Gene therapy has come a long way since its start in the 1970s, when scientists first began developing and optimizing ways to transfer functioning genes into cells as a therapeutic option. Unfortunately, initial attempts at human gene therapy in the 1990s were not completely effective and led to cancer or death in some participants. A clinical trial in 1999 recruited five infants with severe combined immune deficiency-X1 to participate, who would otherwise die from unresolved infections due to a dysfunctional immune system. The gene therapy was delivered into their blood stem cells ex vivo. However, nearly three years after the treatment, two patients developed leukemia, or cancer of the immune system. Upon analysis of their genomes, scientists found that the location where the therapeutic gene was inserted into the patients’ genomes activated a cancer-causing gene.
In 1999, a patient named Jesse Gelsinger participated in a gene therapy trial to treat his rare metabolic disorder. Within a day, Jesse had an intense inflammatory response followed by multi-organ failure, eventually being taken off life support five days later. The ensuing court cases revealed that 691 volunteers in various FDA gene therapy experiments had died or fallen ill in the seven years leading to Jesse’s death. In effect, clinical testing for gene therapies came to a halt.

These events made it apparent that gene therapies would not truly be safe and effective for decades to come. Yet, through these trials and errors, scientists developed a better understanding of how our complex genomes are regulated and the relationships between genetic mutations and disease. Progress was also made on safer and more accurate methods of gene delivery, allowing clinical trials to resume using improved gene therapies. Notably, CRISPR-based gene editing tools were developed, which can change DNA bases at precise locations in the genome to correct genetic mutations. As some CRISPR-based clinical trials are currently undergoing clinical testing, the field should take caution from its past and be precautious when testing new therapies on patients.
The current landscape of gene therapy is a rapidly advancing field that stands to help millions of people with rare and debilitating monogenic diseases. Gene therapies, when implemented safely, alleviate their suffering and allow them to lead fulfilling lives. For many that would benefit from approved therapies, however, receiving treatment is still a distant reality. With gene therapy companies charging upwards of 1 million dollars for a single treatment, and bureaucratic delays in obtaining public healthcare coverage, patients with degenerative genetic diseases are still suffering. Further gene therapy research must reconcile safety above all else. Research into cost reduction and accessibility are critical next steps forward to implementing life-improving gene therapies for all those affected.
IMMpress Vol. 11 No. 1 2023 29
- Ash Hagerman
From digital art to PHDOODLESHOP

How a side hustle turned into a small business
Indulging in personal activities and hobbies outside of research during grad school can be for personal enjoyment, but for some dedicated students, it can also be a way to earn a little money on the side. After all, grad school can be financially challenging, and side hustles can help subsidize assistantship or fellowship stipends. Dr. Melanie Girard, a recent PhD graduate from the Department of Immunology (Class of 2022; the Watts Lab), started PHDOODLSHOP that sells science-themed stickers, accessories, and clothing during her final year of PhD. Melanie shared with us how she turned her side hustle while in grad school to a booming small business.
What inspired you to start a side hustle? How did you find out that science-themed illustrations are right for you?
https://phdoodleshop.com/


When I was younger, I enjoyed painting and drawing, but I lost that hobby when I started grad school. I changed cities and didn’t bring my art supplies with me. After 4 years of focusing on my PhD research I had a sudden urge to be creative, so I started doing digital art. I was also using illustrator to make figures for my papers, so I suppose that is what inspired me to make science-themed illustrations. I didn’t really plan to make my hobby into a business at first, but at some point, I thought it would be fun to open an Etsy shop and share my illustrations in the form of stickers. When I saw that people were actually buying them, I decided to invest more time into growing a small business.
What are the skills you learned in your PhD that helped you with your business?

Being able to manage various aspects of my business requires organizational skills that I developed during my PhD.
Another skill I find useful is the ability to learn new things on my own. For example, I taught myself about product development, branding, how to build and maintain an e-commerce website and how to market my products on social media.



It is also important to not be afraid to try new things even though they might fail. During my PhD, for every experiment that succeeded, I had done countless number of experiments that had failed. I learned that you need to try different things to figure out what works and what doesn’t.

✦ ✦
Dr. Melanie Girard
30 IMMpress Vol. 11 No. 1 2023
@phdoodleshop
As a small business owner/ entrepreneur, what are the main challenges/ hardships you have encountered so far?
I find the marketing aspect of my business quite challenging. I reach my audience through social media, mainly through Instagram. Posting on social media every other day is time consuming and requires a lot of creativity. I find it difficult to post consistently.
You started PHDOODLESHOP during the final year of your PhD. How did you balance your business with your PhD work?
I was initially doing this as a hobby so I would be happy to work on this during the evenings and weekends. I find that it helped me disconnect from my research.






Where do you see your business in 3 years? Do you see yourself running your business as a full time in the next few years?
I would like my brand identity to be polished so that my business really stands out amongst other science-themed retailers. I also hope to have a warehouse and to outsource most of the manufacturing. Finally, I hope to broaden my audience to STEM graduate students in general rather than only life sciences. As for my next career move, I am not sure at the moment. I think my business stems from a need to be creative that I didn’t fulfill during my years in grad school. At some point, I think I will want to use my scientific background to find a job in industry. I would still like to run my business on the side because I really enjoy it.
Please share some advice for graduate students who are looking to start a side hustle.
I think it would be a good idea to ask yourself what skill(s) you have and see if that skill can be useful to others. For example, if you are a good piano player, perhaps you could teach piano. For my business, I started selling stickers because they do not cost much to produce and to ship. I opened an Etsy shop. The advantage selling through Etsy is that the platform will direct relevant customers to your page for you, so you don’t necessarily have to invest time and money into marketing and advertisement. I then used the profits and reinvested them into more expensive equipment and products, and to build my own website. I find that this strategy is particularly good for graduate students because we don’t have a lot of money to invest, and we are not in a position to take financial risks.
Do you see yourself as successful? In what ways?
I am proud that I completed my PhD. It was intellectually and emotionally challenging, but I persevered. I am also proud of my business because I feel like there is value in the products that I have to offer. Products that reflect the day-to-day life and experiences of research lab scientists are hard to find, so people get really excited when they discover my shop!
Sides hustles not only can help developing personal skills outside of research but also can help you to make extra money. If you want to learn more about Dr. Girard’s business and her story, check out her business website (https://phdoodleshop. com/) or follow her on Instagram (@phdoodleshop)!
✦ ✦
- Tianning Yu IMMpress Vol. 11 No. 1 2023 31
The Road Less Traveled:
Why Some PhD Graduates Choose Public Sector Careers
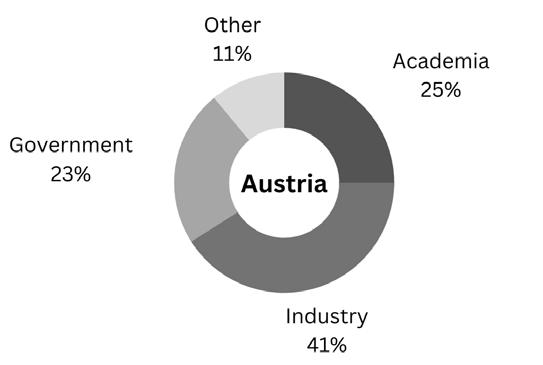

Inrecent years, PhD graduates have seen a shift in employment opportunities, with a growing number of non-traditional roles available beyond teaching and research. The specialized knowledge and expertise gained through rigorous research training make PhD professionals highly valuable to government agencies, non-profits, and private companies. In Canada, “law and social, community, and
government services” ranked as the third most common career route for PhD graduates in 2016. And around 12% of University of Toronto PhD graduates between 2000 and 2015 ended up working in the public sector. In this article, we will explore some factors influencing PhD holders to choose a career path in the public sector.




Where are the PhD graduates now?

GOVERNMENT
32 IMMpress Vol. 11 No. 1 2023
A PhD’s career choice is primarily influenced by their personal preferences, including their desire for intellectual challenges, independence, and level of responsibility. Those who work in academia may have more research autonomy, but they often face greater job insecurity due to the scarcity of permanent positions. In contrast, those who choose to work in the public sector typically have less independence, but enjoy greater job security and a more stable career path. Ultimately, the choice of career path will depend on the individual’s priorities and values.
However, putting individuality aside, other external factors also shape personal choices in an impactful way. The objective supply and demand relationships between PhDs and the individual employment sector also determine the position availability, as in how many PhD holders there are vs. how many PhD holders are needed. A 2015 study revealed that due to the limited availability of academic positions, regions with a higher number of PhDs awarded tended to have a greater influx of individuals seeking employment outside academia, including the public and private sectors. Furthermore, supply and demand relationships for PhDs can vary significantly across different disciplines. A 2022 study found that PhD graduates from social sciences and humanities are more likely to be employed in the public sector compared to their peers from STEM subjects and economics.
In addition, external encouragement can also impact a PhD’s motivation and confidence in pursuing specific employment sectors. The 2022 study also found that if a PhD graduate is encouraged by their supervisor to pursue a public sector career, then they are more likely to seek employment there.
The conventional perception of government jobs in North America has been that they offer more stability, better pay, and greater benefits than academia, making them an attractive option for PhD holders, particularly when tenure positions in academia are scarce. However, the COVID-19 pandemic has significantly impacted this view. According to a recent report, the US government currently has 1.1 million unfilled positions - almost double compared to five years ago. The study also revealed high dissatisfaction with wages in the government sector, and pandemic-related layoffs had undermined job stability in government roles.
During the pandemic, research funding was prioritized towards COVID-19-related research, with the aim of accelerating the process of understanding and producing a solution to this newly evolved disease. Many countries, including Canada, even allocated additional public funding
towards such research despite economic contraction. As a result, funding for non-COVID-related research was reduced. The funding priorities undoubtedly altered the employment landscape for PhD holders significantly, albeit to varying degrees across different fields. For instance, PhD holders in infectious disease and public health may have experienced different employment prospects in the public sector than those in cancer research or geography studies. However, the exact outcomes are yet to be revealed.
When it comes to overall career choices, individuals weigh and balance different factors. In China, government jobs are typically perceived unfavorably. Yet, many PhD holders have recently opted to work in the public sector due to a mismatch in supply and demand. With many new graduates and an economic downturn caused by the pandemic, many PhD holders have chosen to pursue government positions despite the negative perception of these jobs. In one local report from authorities in Chaoyang District in Beijing, 95% of new government employees held a master’s or PhD degree, but were employed in routine government positions that did not require advanced degrees.
At the end of the day, the career decisions of PhD holders come down to their subjective assessment of career prospects. Graduates tend to choose career paths they believe offer the best opportunities, which depend on the available labour market information, job descriptions, and their own abilities. To make informed decisions, PhD holders need to objectively evaluate their skills through peer feedback, supervisor input or leveraging tools like the Stanford Biosciences Individual Development Plan.
It is worth noting that while European PhD career outcomes have been extensively analyzed, there is a lack of recent systematic surveys on PhD graduate employment in North America, let alone specifically in the public sector. As such, connecting with graduates already working in your desired field can provide a more comprehensive understanding of the job description and daily work responsibilities, leading to better-informed decisions.
- Evey Zheng
IMMpress Vol. 11 No. 1 2023 33
Make it make cents:
the inadequacies of research funding in Canada
Regardless of what Copernicus says, our world revolves around money; so, it’s not surprising that money is the biggest roadblock of modern day scientific discoveries. The majority of funding for research in Canada comes directly from the federal government. The Government of Canada distributes the allotted research budget to the Big Three funding agencies: CIHR (Canadian Institutes of Health Research), NSERC (Natural Sciences and Engineering Research Council of Canada), and (Social Sciences and Humanities Research Council of Canada) SSHRC, known together as the Tri-Council Agencies. The Tri-Councils, much like the Olympians, reign over the land, sky, and sea of research funding. It is their role to determine which of the many applicants get the glory of a much-coveted grant to fund their research projects. And while the cost of absolutely everything has increased in the past 20 years, the available funding through the Tri-Council has remained stagnant.
While the federal budget for research has not increased, the success rate for applicants of the tri-council grants has steadily declined. In 2005 about 31% of CIHR applicants were awarded grants, yet by 2018 less than 15% of applicants received funding. The inflation-adjusted average funding through in the past 20 years has remained more or less the same despite a 3.5x increase in applicants. In an attempt to counteract this, the federal government created more available awards, not by increasing the budget for research, but by decreasing the award value by 25%. The average award has decreased from $950,000 to $725,000 between 2018 and 2020. This means that the painstaking effort that goes into writing a grant application is more often than not unrewarded and many important research projects go unfunded. And
while sharing the wealth does benefit the few extra awardees, a band-aid solution is not sufficient to maintain the increasing financial demands of the research industry.
The types of research that does get funded by the Tri-Council has also changed to favour studies that align with the goals of the Canadian government. This resulted in a general decrease in funding for basic science research and better odds for translational and applied research projects such as pandemic reduction strategies. In addition to this shift, the process of accepting grants has also changed – in addition to peer reviewers who rank the applications, a second round of review is what makes the final funding decisions. This committee, primarily made of civil servants, often doesn’t have the background to understand the nuances of the proposed research. As immunologists, this may have a smaller impact on our funding success than seen in other fields, as many if not all projects in our field can be described through a public health lens, but it certainly impedes learning for knowledge’s sake. Basic science provides the foundation for all other research, without it we would see little advancement in the fields of medicine or technology.
How does Canada expect to hold any ground in global science and innovation without providing the financial contributions needed to do the actual work? It is worth arguing that scientific progress can only be successful when the people involved have the tools they need to succeed, and when you can afford to hire those people in the first place.
- Erika McCartney
GRANT F U N D I N G G R A N T T
GRANT
“IN 2005 ABOUT 31% OF CIHR APPLICANTS WERE AWARDED GRANTS, YET BY 2018 LESS THAN 15% OF APPLICANTS RECEIVED FUNDING.”
34 IMMpress Vol. 11 No. 1 2023
A BOOK REVIEW OF AWAKENINGS BY
OLIVER SACKS,
1990 EDITION
Sacks takes us back to the early days of defining neurological disease when sleeping sickness (Encephalitis lethargica) abruptly washed over the world shortly after the first world war and influenza pandemic wrapped the globe around their staunch fingers. Ending a decade later in 1927, those who survived were met with post-encephalitic syndromes years later with Parkinsonism as the common tardy assailant.

Parkinsonism is a term used to group neurological disorders that result in movement disabilities similar to Parkinson’s disease (PD), including traditional symptoms of loss of motor control in forms of tremor, rigidity and slowed movement. I stress the importance of not remaining satisfied with my reduced definition of Parkinsonism. This leaky umbrella-like term hides the many, many disorders grouped under this label including atypical Parkinson’s disease and ones that have yet to be identified. Sacks’ patients affected with sleeping sickness would have most likely developed a form of atypical Parkison’s disease, which not only tends to progress faster than PD, but often does not respond well to the traditional levodopa therapy prescribed for PD.
PD and Parkinsonism were in its early, superficial days of categorization and thus when the drug L-dopa (levodopa) was naively introduced as a ‘cure’ for “Parkinsonism” in 1969, there was no hesitation in broadly adopting this treatment with all and any types of the disease. L-dopa is used to make dopamine in the body, and as dopamine is reduced within these groups of disease, L-dopa works to replace dopamine and control bradykinesia or slowness of speed.
Sacks too looked at L-dopa with wide eyes and began to document the ‘awakenings’ of his patients who have been experiencing post-sleeping sickness-Parkinsonism as early as 1930. After a short 90 days of treatment, Sacks saw the quick detrimental effects of this ‘holy grail’ but could not communicate this within the Neuroscience community without his peers becoming enraged by his questioning of such a monumental ‘cure’. The weakness of L-dopa was not seen yet within general PD patients and thus his peers were ignoring his concerns of the unpredictable side effects his patients were experiencing such as becoming overly sensitive to small doses, developing motor ‘restlessness’, or having no response
to L-dopa. The patients were waking up and this was enough for the irresistible excitement swarming L-dopa to continue. The pedestal L-dopa was thrown on top of was growing taller and more and more impervious until its poor consequences shot up as thick vines and tightly tangled itself around and around the therapeutic statue, eventually reaching and contaminating the simple beauty of L-dopa.
And this all comes down to how we tend to see and subsequently define complex disease. If we start off describing the disease in reductive ways then the treatments can only be reductive.
In Awakenings, Sacks intersects “biology and biography” through a strong focus on the patients’ identities. He spends time observing and listening his patients, using their words and metaphors to explain their experiences – he explores “what is was like to be human, to stay human, in the face of unimaginable adversities and threats.” It is his case histories, carefully documenting the stories of his patients, his experiences with them before, during, and after L-dopa, that define Parkinsonism. We cannot reduce Parkinsonism to a few lines on a medical record. Parkinsonism is a condition that people – bodies and identities – are experiencing, and the tangible, documented symptoms are only the tip of the deep, cloudy iceberg that we are attempting to visualize. Yet we continue to be uncharitable to disease we do not understand by focusing and treating what’s right in front of us. Sacks states we need to be co-explorers with our patients to help define, treat, and as his book focuses on, communicate complex diseases such as Parkinsonism. Along with this holistic view of disease, he emphasized that we need to work to push ourselves to see the intolerable – that the treatment of symptoms does not address the whole issue. The same drug can result in a variety of responses that are both hopeful and eventually disastrous as L-dopa has shown. Here, Sacks and his case histories pushes us to step into the world of the resilient individuals who are faced with such calamity and see them as not only a patient affected with a disease but a person struggling to adapt and survive in a world where treatments often come with poor consequences that must be balanced.
- Melanie Bercovitch
“These things are so bizarre that I cannot bear to contemplate them.”
- Poincaré
IMMpress Vol. 11 No. 1 2023 35
Department of Immunology

University of Toronto
1 King’s College Circle
Toronto, ON M5S 1A8 Canada
Old Medical Building - Graduate Laboratory in Biochemistry (c1966)
Photo Credits: University of Toronto Archives.
















 Philip Barbulescu Karen Yeung
Philip Barbulescu Karen Yeung




 Juan Carlos Zúñiga-Pflücker, PhD Professor and Chair Department of Immunology
Juan Carlos Zúñiga-Pflücker, PhD Professor and Chair Department of Immunology












































