SSRIs and Storks: The Complicated Relationship Between SSRIs and Pregnancy
Dinosaur Cognition: Not Set in Stone
All Senses to Center Stage: Synesthesia in Action



SSRIs and Storks: The Complicated Relationship Between SSRIs and Pregnancy
Dinosaur Cognition: Not Set in Stone
All Senses to Center Stage: Synesthesia in Action
FEATURED ARTICLE
13 18 23 28 8 34 40 50
SSRIS AND STORKS: THE COMPLICATED RELATIONSHIP BETWEEN SSRIS AND PREGNANCY
by Talia Mohideen | art by Maisy Richardson
TWISTING THE KNIFE: THE OPEN CASE OF MENSTRUATION, THE BRAIN, AND THE MISSED DISEASE OF ENDOMETRIOSIS
by Zoë Shelley | art by Abigail Schoenecker
A LITERARY CLASSIC: OXYTOCIN AND DOPAMINE IN AUTISM SPECTRUM DISORDER
by Sydney Keenan | art by JD Jarolimek
PRETEND PLAY, REAL BENEFITS: HOW PLAY WIRES THE BRAIN IN CHILDHOOD
by Leo Mahlke | art by Leo Mahlke
FEATURED ARTICLE
DINOSAUR COGNITION: NOT SET IN STONE by Kate Billow | art by Anna McFadden
F(OCUS) 1: THE SCIENCE OF FLOW STATE by Julian Cardenas-Moncada | art by Nancy Duer
DRINKS ON ME: HOW ALCOHOL CONSUMPTION IMPACTS YOUR BRAIN by Daniel Bader | art by Ruimei Chau
IPADS, PLAY, AND PLASTICITY: HOW DIGITAL MEDIA AFFECTS YOUNG BRAINS
by Samuel Jacobs | art by Zoe Stiefel
LEQEMBI: A NEW ALZHEIMER’S DRUG BRINGS HOPE AND SPARKS CONTROVERSY
by Charlotte Tobin | art by Querida Alleyne
FEATURED ARTICLE
ALL SENSES TO CENTER STAGE: SYNESTHESIA IN ACTION by Tara Dacey | art by Alexandra Tapia 55 45
Art by Nancy Duer
If you have any questions or comments regarding this Issue 12, please write a letter to the editor at brainstorm.vassar@gmail.com.
Check out our website to read our articles, find out how to get involved, and more at greymattersjournalvc.org.

EVELYNN BAGADE Editor-in-Chief


OBERMUELLER Senior Managing Editor
Senior Managing Editor



Executive

EDEN LANHAM Senior Editor, Scientific Editing


Senior Editor, Scientific Editing

BILGER Production Manager

Coordinator
ALYSSA GU Senior Editor, Lay Review

BILLOW Senior Editor, Lay Review

KYLE BENSON Senior Editor, General Editing

FALLON Senior Editor, General Editing


Layout Executive & Website Manager


Assistant Layout Executive
Graduate Student Executive
Maisy Richardson
Abigail Schoenecker
JD Jarolimek
Leo Mahlke
Anna McFadden
Nancy Duer
Ruimei Chau
Zoe Stiefel
Querida Alleyne
Alexandra Tapia
Alexandra Adsit
Abigail Tramell
Anna Insua
Ashley Hong
Caleb Joyce
Caroline Martin
Elyssa Ulip
Freddy Pulver
George Hosein
Giana Rizzo
Grace Buckles
Jannessa Ya
Lou Chicatelli
Matthew Rawson
Max Tuz
Paige King
Sonya Inkelas
Sydney Goldsmith
Talia Mohideen
Talia Mohideen
Zoë Shelley
Sydney Keenan
Leo Mahlke
Kate Billow
Julian Cardenas-Moncada
Daniel Bader
Samuel Jacobs
Charlotte Tobin
Tara Dacey
Bojana Zupan PhD
Hadley Bergstrom PhD
Lori Newman PhD
Stephanie Jackvony PhD
Brooke Berbeco — Social Media
Summer Stern Layout
Catherine Zhang Layout
Alex Astalos
Eli Kanetsky
Lucy Gaffneyboro
Ren Nicolau
Bertha Shipper
Bailey Mann
Joseph Lippman
Emily Nothdurft
Stella Petersen
Alex Orellana Rico
Evan Seker
Olivia Del Tatto
Erica Langlais
Hadley Snell
Nathaniel Dozier
Ben Weiner
Ben Salamon
Sydney Wong
Mercedes Fippinger
Quincey Dern
Rachel Kang
Susanna Osborne
Amaya Gab
Aleena Abdelhady
Mercer Colby
Lily Paine
Lena Lynch
Alaina Desautels
John Gao
Minh Tâm Ong
Anna Cohen
Shayni Richter
Chloe Bilger
Mihika Hete
Grace Cabasco
Sydney Jones
Kaitlin Raskin
Tien Le
Lea Repovic
Cooper Jaffe
Zachary Garfinkle
Jacqueline Rosenblum
Kamiliya Aumiller
Lila Horberg Decter
When you think about neuroscience, what is the first thing that comes to mind? You might picture sterile laboratories, mysterious brain scans, rats running in mazes, and scientists in white coats speaking in ten-syllable words you can barely understand. However, a closer look reveals that neuroscience is not just a complex scientific discipline, but a unique perspective through which we can gain a deeper understanding of ourselves and the world around us.
The articles in the latest Issue 12 of GMJ demonstrate that neuroscience overlaps and intersects with various fields of scientific inquiry and can be used to answer a wide range of questions. I encourage readers to learn about how neuroscience can deepen our knowledge of women’s health through ‘SSRIs and Storks: The Complicated Relationship Between SSRIs and Pregnancy’ and ‘Twisting the Knife: The Open Case of Menstruation, the Brain, and the Missed Disease of Endometriosis.’ Neuroscience can also give us insights into childhood development, as captured in ‘Pretend Play, Real Benefits: How Play Wires the Brain in Childhood,’ and ‘iPads, Play, and Plasticity: How Digital Media Affects Young Brains.’ Further, readers will be delightedly surprised to learn that neuroscience can even help us understand the dinosaurs in ‘Dinosaur Cognition: Not Set in Stone.’ Truly, neuroscience can be found in almost any corner you look.
As Issue 12 goes to print, I feel incredibly proud of the diverse set of articles our team has crafted this semester, and also feel a sense of sadness as my graduation date nears and my time with GMJ comes to an end. GMJ has been one of the most meaningful parts of my college experience, and has been integral in helping me find my voice and reminding me that I, just as much as everyone, have the right to participate in scientific conversations.
I am excited to welcome Julia Fallon into her new role as Editor-in-Chief. I have long maintained a deep respect for Julia’s curiosity, dedication, endless energy, and natural composure as a leader. I trust that Julia and our rising executive board will continue to carry out our mission of scientific accessibility with poise and precision, and I look forward to cheering on GMJ from the sidelines.
I extend great thanks to the 2025–2026 executive board for putting their trust in me over the past year, and I would also like to thank the wonderful faculty of Vassar’s Neuroscience & Behavior Program for their continued support of our initiative. Of course, all of us at GMJ thank our readers for continuing to read and engage with the world of neuroscience.
If our readers take away anything from this issue, I hope it is this: neuroscience is everywhere, and it belongs to everyone.
Sincerely,
Evelynn Bagade

Editor-in-Chief
by Talia Mohideen | art by Maisy Richardson
*Note: This article uses female-gendered language to refer to pregnant and postpartum people due to the vast majority of cited literature being focused on female-identifying subjects. The editors wish to acknowledge that pregnancy is independent of gender identity.
For many expecting mothers, pregnancy is filled with excitement about this new stage in life — starting a family and raising a child. However, pregnancy also brings new challenges, including new responsibilities, hormonal changes, and risks of various complications [1, 2, 3]. Women who take medication to manage depression and anxiety have the additional challenge of deciding whether to continue during pregnancy. Depression is a mood disorder characterized by persistent sadness, fatigue, changes in sleep or appetite, and a loss of interest or pleasure in hobbies; anxiety is a disorder characterized by fear, nervousness, and worry, as well as physical symptoms such as heart palpitations, shortness of breath, dizziness, and muscle tension [4, 5]. To cope with these conditions, more than thirty million Americans use antidepressants, with selective serotonin reuptake inhibitors (SSRIs) being the most common [6]. Initially introduced in the early 1980s, SSRIs such as Lexapro,
Prozac, and Zoloft have become the standard pharmaceuticals used to treat depression over the past few decades [7]. This is largely due to their shorter list of associated side effects when compared with medications used earlier in psychiatric history, which carried a higher risk for cardiovascular issues, among other complications [8, 9]. However, as is common with most medications, SSRIs have their downsides. Common side effects of SSRIs include emotional blunting and sexual dysfunction [10, 11]. When women who take SSRIs become pregnant, they may have reason to be concerned about how this medication will impact their fetus — for example, potentially increasing the risk of miscarriage, preterm birth, and cardiac defects [12]. However, for women with depression, anxiety, or both, there are also risks of not using SSRIs during pregnancy [13, 14]. Women can become particularly vulnerable to depression during pregnancy if they are also coping with life stress and a lack of support [15]. Left untreated, depression jeopardizes maternal well-being by increasing the risk of suicidal ideation and postpartum depression (PPD) — conditions that pose a serious threat to both mother and fetus [12]. As a result, women are often faced with the difficult decision of whether to continue SSRI use during pregnancy.
An expectant mother experiencing depression may feel as though she is not living, but simply surviving [16]. When she gets out of bed in the morning, she may find it difficult to take her pregnancy vitamins or feel as though she has no purpose [16]. Watching her comfort TV show or chatting with friends, which used to bring her joy, makes her feel as though she is going through the motions without any emotion [17, 18]. Anxiety disorders are similarly incapacitating: for a mother with anxiety, her thoughts can be consuming [19]. She might worry about everything — things she did or said, the health of her fetus, and her family’s future. It feels impossible to stop these thoughts from occupying her mind, making it nearly impossible to focus [19]. With millions enduring the reality of depression and anxiety every day, it begs the question of how and why the disorders develop [20, 21]. What are the neurological mechanisms behind these conditions? And why do SSRIs provide a way to mitigate such difficult thoughts and feelings?
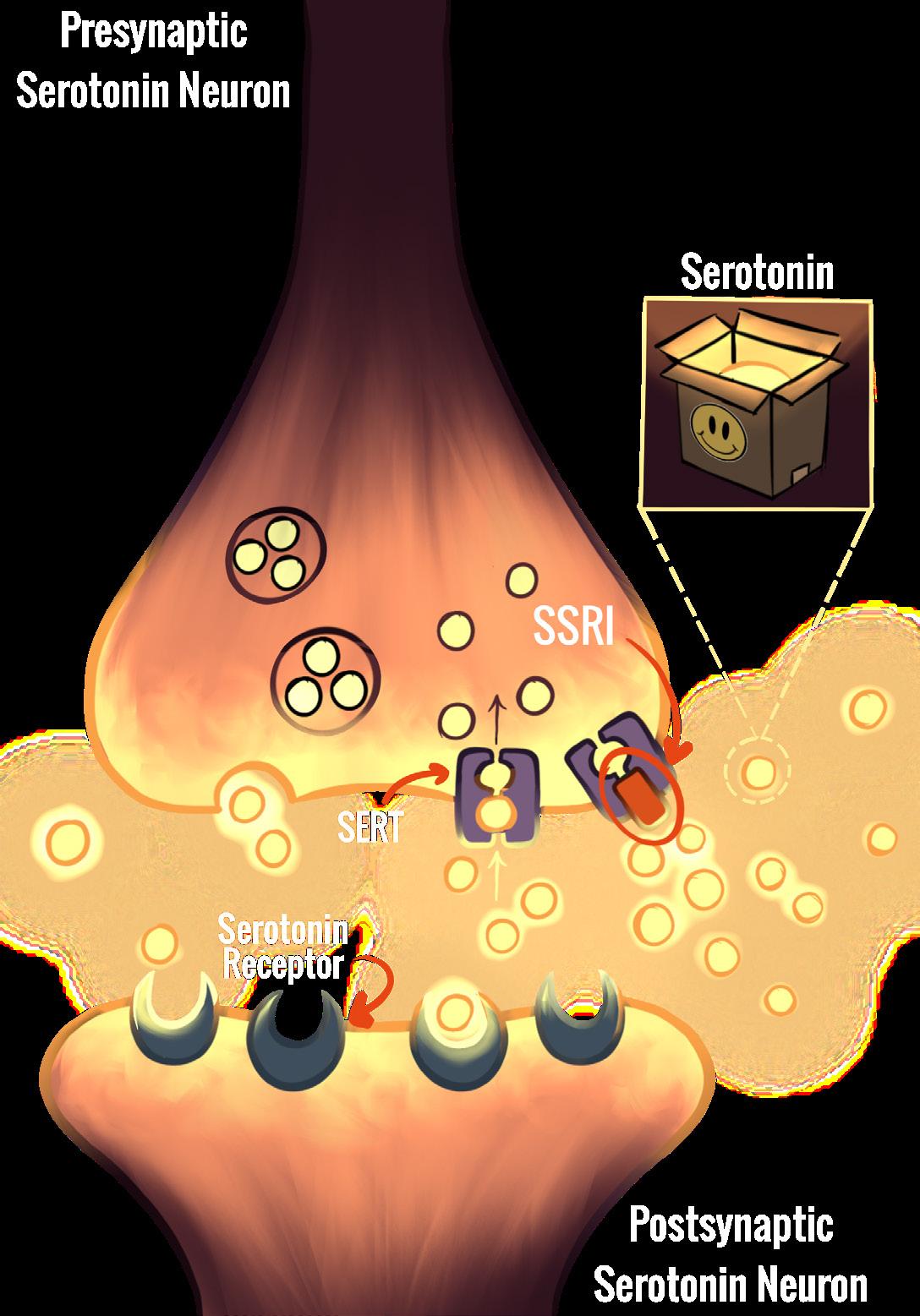
Imagine that the main communication cells of your brain, called neurons, are houses in a neighborhood expecting a package delivery. These packages, or neurotransmitters, are molecules that carry messages from one neuron to the next [22, 23]. In order to do this, neurotransmitters must cross the synapse, the gap between two neurons, and bind to receptors that specifically recognize that neurotransmitter [22, 23]. Afterwards, the process of reuptake allows neurotransmitters to be reabsorbed into the neuron that released them, just as when mail goes back to the sender [24, 25]. Serotonin is a neurotransmitter involved in regulating mood, sleep, and appetite [26, 27]. Increased serotonin in the synapse, which makes receptor binding more likely, is linked to improved emotional states — when serotonin ‘letters’ enter someone’s synaptic ‘mailbox’ regularly, they may experience more positive emotions [26]. Sometimes, however, the mailbox is completely empty or overflowing with packages, both of which can be harmful, just as both deficient and excessive synaptic serotonin can be problematic [28, 29]. An empty mailbox may be the result of excessive reuptake. The flooding of serotonin into the synapse and its subsequent reuptake must therefore be balanced [24, 25].
Serotonin deficiency in the synapse, which is associated with depression and anxiety, may be due to dysregulation of the serotonin transporter (SERT), which results in excessive reuptake [30, 31]. In this case, dysregulated SERT is analogous to a mailman forgetting to deliver any packages to the houses and simply bringing them all back to the post office. SSRIs bind to and inhibit SERT, reducing serotonin reuptake, so the packages are delivered and stay in the mailbox for longer until they can be received or taken back to the post office [32, 33, 34]. The serotonin molecules stay in the synapse longer, resulting in increased and prolonged signaling [32, 33, 34]. In other words, SSRIs allow the ‘positive packages’ to continue to their destination rather than being taken up by SERT.
Serotonin and SSRIs may cross the placenta — an organ that provides the fetus with water and nutrients crucial to its survival and development [35, 36, 37].
Serotonin, which can act as both a neurotransmitter and a hormone, is crucial for supporting embryonic development [38]. Hormones are molecules that carry chemical signals through the blood, similar to how neurotransmitters carry messages between
neurons [39]. During pregnancy, serotonin is involved in the positioning of organs, eye development, and the production of neurons in the fetus. SSRIs may block serotonin absorption in the placenta, thereby affecting aspects of fetal development that are dependent on the concentration of serotonin. As a result, taking SSRIs during pregnancy increases the risk of birth defects such as heart, brain, and skull malformations. In summary, SSRI use by the mother can have a myriad of effects on the child, both during development and after birth, contributing to controversy about whether women should continue their regular SSRI treatment while pregnant [14, 36].
While SSRIs can be helpful to people with depression and anxiety, there is also cause for an individual deciding to discontinue medication [40, 41]. Emotional blunting, a decrease in emotional responsiveness that is often described as feeling ‘numb,’ is a common side effect of SSRIs that causes many people to consider stopping or changing their medication regimen [40]. When SSRIs increase the amount of serotonin
in the synapse, the brain physically adapts. Specifically, it is believed that the SSRIs cause serotonin receptors to become less sensitive and decrease in number, leading to less serotonin binding overall [42]. When people discontinue SSRIs, reuptake is no longer inhibited, and the concentration of serotonin at the synapse decreases. As a result, emotional blunting ceases, and the reversion to stronger emotions can be difficult to manage [43]. This decrease in serotonin and in receptor sensitivity is what underlies SSRI withdrawal, a phenomenon characterized by sleep disruption, irritability, dizziness, nausea, anxiety, and depression [42]. Other withdrawal effects include struggles with concentration and maintaining an active social life [43]. Withdrawal symptoms will continue until the brain returns to its original level of serotonin sensitivity, which can take as short as two weeks or as long as several months after stopping SSRI usage [41, 44].
SSRI withdrawal can be even more challenging as a result of the significant changes to expectant mothers’ bodies and brains as hormones fluctuate [12, 45, 46]. The increased levels of the hormones oxytocin and estrogen during pregnancy regulate the release of other hormones and neurotransmitters that are involved in motherhood [47, 48]. For example, oxytocin is involved in breastfeeding, which is important in facilitating attachment between mother and child [49]. Additionally, the hormonal fluctuations that occur during pregnancy can have a major impact on a pregnant person’s mental state — for example, increased estrogen is associated with increased vulnerability to depression [50, 51]. The decision to discontinue SSRI treatment alone can have a major impact on mood, and discontinuation during pregnancy can have even more profound effects [12, 45, 46]. Depression symptoms, such as poor sleep and nutrition, which may re-emerge during SSRI withdrawal, can increase the risk of other health issues for both the mother and fetus [52, 53]. Thus, pregnant individuals who decide to discontinue SSRIs may benefit from other forms of treatment for depression or anxiety, such as cognitive or psychotherapy, or tapering off these drugs [12].
Mothers in the postpartum period are at risk for developing depression due to major hormonal changes [54]. Thus, not taking SSRIs can also be dangerous for mothers who previously used them, as it could lead to postpartum depression (PPD), a specific type of depression affecting mothers after giving birth [12]. Nearly 13-19% of new mothers experience PPD, which is characterized by depressed mood, sleep and appetite disturbance, fatigue, and feeling guilty, worthless,
and overwhelmed [55, 56, 57]. Individuals struggling with this condition may be overly worried or preoccupied about their infant’s health, feeding, and bathing safety, to the point of dysfunction. In extreme cases, they may even experience suicidal thoughts and thoughts of harming their child. PPD is a very complex disorder, hence the seemingly contradictory symptoms [57]. Children of mothers suffering from PPD can also be affected, as they may experience impairments in cognitive development, as well as emotional and behavioral issues [58]. SSRI treatment for mothers with PPD can mitigate some of their symptoms [58]. Coupled with unclear information on depression and SSRI treatment options, further research is needed to address the mental health concerns affecting the pregnant and postpartum population.
There is a lack of consensus regarding whether SSRIs increase or decrease the risk of certain health conditions, prompting discussions regarding whether it is best to continue or discontinue SSRIs [6]. For example, pre-eclampsia, a condition characterized by high blood pressure in expectant mothers, is a leading cause of maternal morbidity and mortality [2, 59, 60]. Mothers diagnosed with this condition may experience headaches, visual disturbances, chest pain, or confusion [61]. Pre-eclampsia increases the risk of fetal death and preterm birth [62]. Additionally, children born to mothers with pre-eclampsia tend to be underweight due to preterm birth, and thus may have a higher risk of cardiovascular and metabolic complications later in life [62, 63]. If left untreated, pre-eclampsia can progress into eclampsia, a life-threatening condition [64]. A mother with eclampsia will experience one or more seizures, which involve strong and sustained convulsions of several muscles, inability to breathe, and increased heart rate [65, 66]. The connection between SSRIs and pre-eclampsia is complicated. There is a potential bidirectional relationship between depression and pre-eclampsia — pregnant women diagnosed with pre-eclampsia are more than twice as likely to develop mood and anxiety disorders [67]. Depression symptoms themselves may contribute to a lack of self-care, which can then increase the risk of pre-eclampsia development due to poor maternal health overall. There may be a greater risk of pre-eclampsia associated with SSRI use; however, the validity of this relationship is contested [6]. Since there are shared determinants between depression and pre-eclampsia, individuals
taking SSRIs may be at higher risk [6]. It is also plausible that SSRIs prevent pre-eclampsia [68]. Increased serotonin is associated with decreased arginine vasopressin (AVP), a hormone associated with pre-eclampsia. By decreasing AVP, SSRIs may decrease the risk of pre-eclampsia [68]. The case of pre-eclampsia is not an isolated example, and it illustrates the myriad challenges associated with choosing to continue SSRIs during pregnancy [67].
Approximately one third of infants exposed to SSRIs during the third trimester show signs of Poor Neonatal Adaptation Syndrome (PNAS) — a condition that, like SSRI withdrawal, is characterized by irritability, muscle tone irregularities, jitteriness, and feeding difficulties [69, 70]. These symptoms have been observed in infants several hours after birth; however, these are typically mild and often resolve naturally over several days or weeks [71]. In acute cases, PNAS may involve more severe symptoms such as respiratory issues and seizures [72]. The effects of PNAS can be reduced through breastfeeding, skin-to-skin contact, and pharmacotherapy. Since SSRIs can be passed into breastmilk, breastfeeding can also help the infant gradually taper off the medication and reduce withdrawal symptoms [14]. Some mothers may choose to discontinue SSRI treatment during pregnancy due to fear of their child experiencing PNAS; however, in most cases, the risks associated with this condition seem to be minor [71].
In recent years, there has been an increase in the popularity of political movements against psychiatric medications, as well as greater misinformation and weaponization of maternal and women’s health [73, 74]. While skepticism is important in science, we as a society must also recognize the benefits modern medicine provides. Millions of people benefit from SSRIs, and for most, the small risks associated with continuing them during pregnancy are minute compared to those of discontinuation [6, 12, 52]. Of course, each individual is different and must make their own decision in consultation with professionals [12]. Future research will likely give us a better understanding of how continued and discontinued SSRI use during pregnancy affects maternal and fetal well-being. The mother’s and fetus’s health are deeply intertwined — a healthy mother ensures a healthy environment for the fetus to develop. But, above all, the mother’s own health must be prioritized, as an improved quality of life allows for a more fulfilling experience both as a parent and as an individual.
References on page 61.


by Zoë Shelley | art by Abigail Schoenecker

Last night, you went to bed on time. But when you wake up today, you feel exhausted. Everywhere. Normally, you spring out of bed, throw on your favorite workout clothes, and are out and back from a run in no time. Instead, your muscles ache incessantly as you press yourself out of bed; every movement is in slow motion. And although your body feels stalled, your thoughts are speeding through your mind. An anxiety you rarely experience is now wreaking havoc, and a tightness forms in your shoulders and neck. Both are signs that an insistent buzz of stress has begun to settle in. On top of the hum of your racing thoughts, there is a relentless ache in your stomach. Even as you stand up and start to get ready for the day, the pain persists, cutting into every moment of
your morning. The discomfort keeps you in bed for most of the day, a heating pad pressed onto your stomach. As you fall asleep that night, you think that maybe you are being hit with the worst case of the flu you have ever had. Awoken at 3:00 a.m. by the pang of your bladder calling you to the bathroom, you sit down and find your pajama pants have been stained with bright red blood. How could you have forgotten? Your period was due. This vicious cycle of events is a common occurrence for women everywhere. But now imagine that exhaustion, that irritability, that incessant, pulsing, never-ending pain, as not just a monthly factor of your life, but a near constant presence marking every day. This ceaseless pain is a reality for women living with endometriosis.
There are 1.8 billion people worldwide who menstruate every month [1]. In essence, menstruation is the shedding of the endometrium — the inner lining of the uterus, which is the central organ of the female reproductive system [2, 3]. During the menstrual cycle, the endometrium prepares for possible egg implantation, supports the uterus for pregnancy, and sheds in the absence of implantation [3]. All of these functions are regulated by chemical messengers that travel through the bloodstream called hormones [3]. A central hormone-regulating pathway is the hypothalamic-pituitary-ovarian (HPO) axis, which facilitates communication between the brain and the ovaries to orchestrate the menstrual cycle [4, 5]. The HPO axis begins with the hypothalamus, a brain region that controls a plethora of functions such as sleep, body temperature, and hunger — all of which keep our bodies in a balanced state [6]. The hypothalamus kicks off these processes by producing gonadotropin-releasing hormone (GnRH), which signals the pituitary gland — a major regulator of other hormone-secreting tissues — to secrete its own regulatory hormones called gonadotropins into the bloodstream [5]. These pituitary regulatory hormones are what truly drive the reproductive cycle [7]. The two gonadotropins secreted are luteinizing hormone (LH) and follicle-stimulating hormone (FSH) [7, 8]. A high enough concentration of LH and FSH cues the ovaries to secrete the hormones estrogen and progesterone [9, 10]. Estrogen and progesterone facilitate the growth of the endometrium and biological processes such as fertility and pregnancy [11]. These hormones signal back to the hypothalamus and the pituitary gland as needed, completing the cycle by
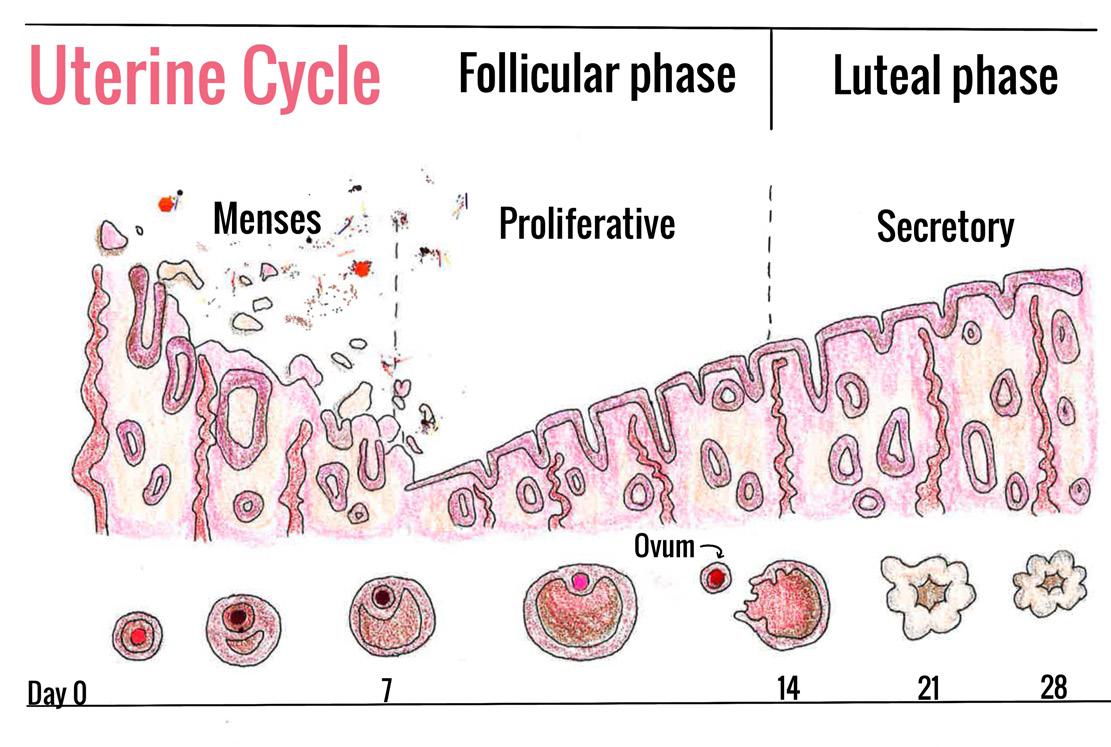
releasing more GnRH, LH, or FSH [12]. Throughout the menstrual cycle, LH and FSH are released by the pituitary gland in an alternating pattern, dictating the phase of the cycle [7].
The menstrual cycle involves changes to the endometrium regulated by hormone fluctuations, and it is characterized by four phases: the follicular, ovulatory, luteal, and menstrual phases [13]. During the follicular phase, low levels of estrogen and progesterone prompt the pituitary gland to increase FSH secretion [14, 15]. As the pituitary gland releases more FSH, an immature egg starts to grow in the ovaries and releases estrogen, which stimulates the endometrium to thicken [14, 15 16]. In the ovulatory phase, estrogen and LH increase rapidly until they reach their peak [14]. Due to the surge in LH levels, the ovaries release the now mature egg into the fallopian tubes, part of the female reproductive system that connects the ovaries to the uterus [14, 17]. Progesterone levels rise in the middle of ovulation as LH surges [14, 18]. During the luteal phase, progesterone reaches its peak concentration before decreasing along with estrogen, and LH and FSH levels return to baseline. These hormonal changes trigger the menstrual phase, during which the thickened endometrium sheds and produces the bleeding we know as a period [14, 18]. By shedding resource-hungry tissue rather than maintaining it, the uterus can preserve a healthy environment, save energy, and rebuild when a new fertilized egg is present [19]. Simultaneously, the oscillating hormones throughout the menstrual cycle also create a myriad of physical symptoms.
Throbbing pain in the lower abdomen, also known as cramps, is the most prevalent symptom of the menstrual cycle, experienced by most people who menstruate [20]. Cramps are the result of the uterus contracting to shed the endometrium and begin with immune cells recognizing the damaged tissue of the endometrium, prompting an inflammatory response [21]. During inflammation, immune cells remove damaged cells and initiate the process of repairing tissue [22, 23]. Immune cells control inflammation by releasing small proteins called cytokines [24]. Cytokines can be either pro-inflammatory, meaning they increase inflammation, or anti-inflammatory, meaning they decrease inflammation [25]. Pro-inflammatory cytokines trigger the release of prostaglandins, which in turn amplify the effects of the cytokines [26]. Prostaglandins are hormone-like substances produced at
sites of tissue damage, and ultimately play a key role in the commonly resulting pain and inflammation [27, 28]. Just before the luteal phase ends, progesterone rapidly decreases, which triggers the production of prostaglandins from the endometrium to facilitate its shedding [29]. Concurrently, prostaglandins prompt uterine contractions and significantly increase the sensitivity of nerve endings, leading to the pain associated with menstrual cramps [30]. The higher the amount of prostaglandins produced, the more severe the menstrual cramps [29]. However, cramps are not the only physical symptoms that occur during the menstrual cycle — significant changes are also happening in the brain as a result of hormonal activity.
As previously mentioned, hormones play a critical role in dictating the menstrual cycle. Estrogen and progesterone can enter the brain and bind to their respective hormone receptor proteins to cause a cellular response [31, 32]. When estrogen interacts with its receptor on neurons — a type of brain cell — the hormone alters the cells’ critical method of communication: neurotransmission [33]. Neurons release small molecules called neurotransmitters that act on other neurons and allow for communication within the brain [34, 35]. The feelings of anxiety, exhaustion, and lack of motivation in the days leading up to a period may be symptoms resulting from fluctuating estrogen and progesterone levels as hormones modify concentrations of certain neurotransmitters [14]. Dopamine, serotonin, glutamate, and GABA are key neurotransmitters that bring about changes associated with respective phases of the menstrual cycle [36]. Dopamine modulates reinforcement and assists in the regulation of mood [37, 38]. When estradiol increases, it leads to higher dopamine concentrations, resulting in enhanced motivation, boosted mood, and increased reward perception [39]. Increased levels of estradiol also stimulate an increase in serotonin levels, which are associated with reduced anxiety, improved mood, and enhanced cognitive functioning [40, 41]. Estradiol also inhibits the protein that breaks down serotonin and increases the sensitivity of serotonin receptors [42]. When estradiol rises in the follicular and ovulatory phases, dopamine and serotonin levels simultaneously increase [36, 37, 43]. Following this initial rise in dopamine and serotonin, estradiol decreases and progesterone increases during the luteal and menstrual phases, leading to reduced dopamine and serotonin synthesis and subsequent
negative feelings [43]. Estradiol also directly modulates glutamate, the primary excitatory neurotransmitter which makes a neuron more likely to release neurotransmitters [44, 45, 46]. Increased progesterone, however, is associated with decreased glutamate signaling and lower neuronal excitability and thus lower chances for a neuron to release neurotransmitters [47]. As such, progesterone increases concentrations of the primary inhibitory neurotransmitter, GABA, which works to balance neurotransmission in the brain by reducing neuronal activity [47, 48].
In individuals with endometriosis, the mechanisms of a typical menstrual cycle are dysregulated. Endometriosis is a chronic inflammatory condition characterized by the growth of endometrial-like tissue outside the uterus, typically in the lower abdomen [37, 49]. To have a better sense of the disruption seen in endometriosis, imagine how painful it would be if a segment of your stomach tissue with the role of performing stomach-specific tasks began to grow in your eye. In its diagnosis, endometriosis is categorized by location and severity: superficial endometriosis, deep infiltrating endometriosis, and ovarian endometriotic cysts [37]. Superficial endometriosis appears on the surface of the tissue in the lower abdominal regions; deep infiltrating endometriosis involves scar tissue that extends into the muscle layer of the intestines, bladder, or other critical organs; and in ovarian endometrial cysts, sac-like structures filled with fluid or masses of skin form inside the ovaries [37, 50].
Endometrial-like tissue developing in areas external to the uterus functions as it would inside the uterus: growing, thickening, and shedding each month [51]. Therefore, individuals with endometriosis still experience the monthly renovation of the uterine lining, but this drastic transformation of endometrial-like tissue also occurs in various other regions of their body simultaneously [52]. Outside the uterus, broken-down tissue has no way to exit the body, often causing immense or even paralyzing pain, as well as inflammation and adhesions [53]. The proliferation of endometrial-like tissue in regions where it should not be found also creates heavy menstrual bleeding, painful sex, and, in some cases, infertility [54]. There is no clear explanation for the exact cause of this dysfunction in tissue growth [55]. A leading theory is retrograde menstruation — when menstrual blood containing endometrial cells flows backwards through the fallopian tubes into the peritoneal cavity,
fluid-filled areas between abdominal organs [56, 57]. However, retrograde menstruation occurs in most people who menstruate, so it cannot be the only cause of endometriosis [58]. Factors such as immune system dysfunction, endometrial cells spreading via the bloodstream, or tissues transforming themselves into endometrial tissue are some other theories regarding the causes of endometriosis [57]. The endometrial-like tissue that migrates to various regions of the body contains estrogen receptors, as well as the ability to produce its own estrogen [59]. This means that the survival and growth of endometrial-like tissues, just like the endometrium, the brain, and other tissues, are heavily influenced by estrogen produced during the menstrual cycle [60]. In a person who menstruates without endometriosis, estrogen will rise during the follicular phase and peak during ovulation, eventually decreasing when progesterone is produced in the ovaries [7]. However, individuals with endometriosis experience progesterone resistance, meaning progesterone cannot play its regulatory role [61]. As a result, estrogen will overpower all other hormones and cause inflammation [61, 62].
Women with endometriosis also have higher levels of prostaglandins and cytokines as a result of this estrogen-caused inflammation, which are known to contribute to the survival and growth of endometrial-like scar tissue [63, 64]. Prostaglandins also promote the expression of proteins that synthesize estrogen around the tissue, which further stimulates its growth and the progression of the condition [65]. Endometrial-like scar tissue itself has a high concentration of inflammatory cells and cytokines, creating an inherently inflammatory environment [66]. This high concentration is due to the constant production of pro-inflammatory cytokines, which are a hallmark of the scar tissue present in endometriosis. Due to the cycle of pro-inflammatory cytokines releasing prostaglandins and prostaglandins enhancing cytokines, the tissue is chronically self-inflamed. This chronic self-inflammation further contributes to the growth and sustainment of the endometrial-like scar tissue in the body. Endometriosis is effectively a self-growing, self-sustaining, and self-progressive disorder [66]. Additionally, prostaglandins affect sensory neurons, increasing their sensitivity and contributing to chronic inflammatory pain symptoms [63]. This
increase in inflammation and neuronal sensitivity leads to non-painful stimuli becoming painful [67]. Something as simple as a papercut can be excruciating when there is enough external inflammation and sensitivity. Thus, when the inflammation and sensitivity turn internal, any simple biological process — all of which involve internal stimuli — turns all the more excruciating [67].
Typically, inflammation is associated with reduced gray matter; however, this is not seen in endometriosis [68]. Gray matter is brain tissue made up of cell bodies, the ‘processing’ segment of the neuron [69]. Throughout a typical menstrual cycle, gray matter thickness fluctuates in response to estradiol and progesterone, although there is no conclusive research on the specific changes associated with each phase [70]. Ovarian hormones have been shown to cause gray matter increase or decrease depending on the region of the brain [70]. These standard gray matter volume changes that occur in all people who menstruate are also seen in individuals with endometriosis [70, 71]. But in comparison to people who menstruate without endometriosis, women who suffer from endometriosis may experience increased gray matter volume specifically in the cerebellum, a brain region critical to processing and anticipation of pain as well as pain-related emotional responses, which suggests that their pain processing is amplified [71, 72]. For people with endometriosis, the combined effects of changes to function and structure in the brain give rise to pain levels higher than the norm in the menstrual cycle.
Endometriosis was first identified as a disease more than a hundred years ago, but progress on finding treatments or even accessible diagnoses has since been tediously slow [73]. It was only in 1993 that the National Institute of Health passed a law allowing women to be studied in clinical trials, and it was only a year later that the Food and Drug Administration established the Office of Women’s Health, dedicated to ensuring the inclusion of women in clinical trials and the advancement of women’s health care [74]. Knowing this, it doesn’t come as a surprise that women will spend an average 25% more years in poor health than men [74]. Today, 10% of women worldwide have been diagnosed with endometriosis [54, 75]. Despite this condition's prevalence, it can take women 7–10 years to get a diagnosis, with women of color experiencing even longer delays [75, 76]. Considering the array of painful symptoms associated with endometriosis, waiting for a diagnosis is extremely detrimental to quality of life [71]. Additionally, delays in diagnosis make it difficult for individuals to find effective treatments [77]. This situation is exacerbated by the fact that treatment options are already limited due to not yet truly understanding the precise cause of endometriosis, which can be attributed to the lack of funding for research. Subsequently, the absence of funding results in less incentive for researchers to conduct further studies to fill knowledge gaps about the condition, preventing further advancements in treatment and diagnosis methods. In combination, these factors create a cycle of ignorance that further prolongs and worsens an already painful process of seeking relief for those with the disease [77]. Doctors and researchers must continue delving into the fascinating role estrogen and progesterone play in catalyzing changes in neurotransmitter concentrations, gray matter volumes, and the structure of brain regions. An improved comprehension of these mechanisms, spurred by adequate research funding, could ensure that all people who menstruate — especially those with endometriosis — are one step closer to understanding the full extent to which their menstrual cycle affects their bodies.
References on page 64.


by Sydney Keenan | art by JD Jarolimek

*Note: This article uses person-first language to refertoAutism Spectrum Disorder.
From the moment we’re born, social interactions begin to shape our lives [1]. Whether it be hearing our parents’ voices or seeing their facial expressions, these early interactions serve as the foundation for more complex social behaviors, including conversations and relationships [2]. As we develop, we learn how to interpret social cues and respond to others’ emotions [2]. Nevertheless, for some individuals, navigating the social world is not as intuitive as it may be for others. While many children relish in the chaos of their school cafeteria, eager to exchange jokes and stories with their peers, some may find this environment challenging. For individuals with autism spectrum disorder (ASD), interpreting facial expressions or maintaining reciprocal conversations with their peers may be difficult, leading to social interactions that can feel confusing, unpredictable, or overwhelming [3, 4, 5]. ASD is a neurodevelopmental disorder associated with various social deficits, alongside restrictive and repetitive behaviors or fixation of interests [6, 7, 8]. Certain individuals with ASD may seek treatment to improve their quality of life and alleviate the challenges of socializing with others. Therefore, research into the possible mechanisms underlying ASD can guide avenues for therapy development. Nevertheless, for many individuals, ASD is not something to be ‘treated,’ but rather a different way of experiencing the world that must be better understood.
A lifelong social journey begins when a newborn baby catches their parent’s gaze and beams back. Humans are biologically wired to form connections with others [1]. From infancy, we possess specialized cognitive processes for navigating the social world, including an innate ability to detect and recognize faces [1, 9]. As we develop, we expand our repertoire of social skills — such as verbal communication, empathy, cooperation, and conflict resolution — through experience and interactions with others [1]. In turn, these social skills guide two broad categories of social behavior: affiliative and agonistic [10, 11]. Affiliative behaviors, like a child happily sharing their toys with other kids, are interactions between individuals that develop, maintain, and strengthen social bonds [10]. In contrast, agonistic behaviors are often associated with conflict and can encompass actions like a child withdrawing from conversation or avoiding peers [11]. Both types of behaviors begin to manifest as individuals develop social complexity, usually occurring during the early stages of childhood [12]. These trends continue into adulthood, influencing how individuals form romantic partnerships, collaborate with coworkers, and learn conflict resolution [12]. Despite similar patterns in how people interact with one another, sociability can vary greatly from person to person [13]. Some people are more introverted and require periods of solitude to recharge their social battery, while others are more extroverted and can feel recharged by social interactions and group settings [14]. However, in some cases, differences in social behavior may be more prominent and can interfere with an individual’s daily functioning, leading to the classification of a disorder [15, 16, 17]. Understanding these differences can provide critical information for developing personalized therapies for behavioral and cognitive disorders, as well as establishing a stronger understanding of the background of disorders such as ASD [13].
ASD is characterized by differences in social cognition, repetitive behaviors, fixation of interests, and atypical perception, including heightened sensitivity to sound, light, and texture [6, 7, 8]. Symptoms are typically categorized into two domains: deficits in social communication and interaction, and restricted, repetitive patterns of behavior [6]. A child with autism may struggle to make friends because they have
difficulties initiating or maintaining conversation across a loud cafeteria table. Additionally, environments like cafeterias can be overstimulating, with distracting sensory inputs such as the stickiness of the table and the strong smell of lunch meat. Furthermore, many individuals with ASD also prefer a strict and predictable routine, so someone else sitting in their usual spot in the cafeteria can feel overly stressful. Nevertheless, ASD symptoms can vary tremendously person-to-person, which made prior attempts to categorize ASD under a single, universal set of parameters difficult [18, 19]. It wasn’t until 2013 that the concept of autism as a spectrum was first presented, leading to an expansion of diagnostic criteria that considered the variability between the number and severity of ASD symptoms [18, 19]. Subsequently, this broadening of diagnostic criteria has likely contributed to the increased number of ASD diagnoses observed in recent decades [18, 20, 21].
Despite the increased prevalence of ASD in recent years, the mechanisms and possible causes behind the disorder remain relatively unclear [22]. ASD may arise from a combination of genetic and environmental factors, such as gene mutations, maternal infection, higher parental age, and hormonal imbalance that affect the developing brain [18, 23]. As there is still so much to learn about ASD, there are many different elements being explored. One promising area of ASD research focuses on neuromodulators — molecules released by brain cells to transmit signals to other cells — that are believed to be involved in both the onset of ASD and the persistence of symptoms [24 25]. While several mechanisms are being studied in relation to ASD, two neuromodulators in particular have stood out due to their role in social behaviors: dopamine and oxytocin [26].
Dopamine, primarily produced in the ventral tegmental area (VTA) — a brain region known for regulating reward processing, learning, and memory — is most commonly known for its role in reinforcing behavior [27, 28, 29]. Neuromodulators like dopamine are released between neurons, brain cells that process and communicate information throughout the brain and body [30]. Dopamine release from the VTA has been theorized to facilitate reinforcement-learning, a process that shapes future behaviors by encouraging or discouraging actions through a reward or punishment
system [27, 31]. The process of reinforcement learning is believed to occur via a phenomenon known as ‘reward prediction error,’ which hypothesizes that the activity of dopamine-releasing neurons helps encode the difference between a predicted and received reward [27, 31, 32] When a reward is better than expected, dopamine release increases relative to an individual's baseline, and when a reward is worse than expected, dopamine release drops below their baseline [27, 32]. To incentivize you to do your chores, your parents might decide to reward you with chocolate at dinner instead of vegetables. If you received double the amount of chocolate you were expecting, neurons in the VTA would release more dopamine to encode an outcome that was better than expected. Conversely, if you completed all your chores and hoped to receive chocolate with dinner but your parents still served you vegetables, VTA neurons would release less dopamine than normal to encode that this outcome was worse than expected.
Subsequently, these changes in the firing of VTA neurons can influence behaviors through the process of reinforcement learning by forming new reward associations and updating previously learned associations [27, 33]. During the first week, when you do your chores and get twice the chocolate you expected, dopamine release increases, helping your brain form the association and incentivizing you to complete your work in the future. Yet, in the second week, when you don’t receive chocolate despite doing your chores, the relative decrease in dopamine can update your previously formed association, telling your brain that chores don’t always equal chocolate. The updated association can discourage you from completing chores in the future, because the outcome you want is not always guaranteed. A similar pattern occurs in
social behavior where a positive reward prediction error, caused by a reward that was better than expected and increased dopamine release from the VTA, often leads to affiliative behaviors [33]. Conversely, a negative reward prediction error, or decreased dopamine release, can contribute to agonistic behaviors [33]. Adolescents with ASD may experience dysregulated reward prediction errors in response to social stimuli, but not non-social stimuli [34]. Dysfunction of this ability to assign values to social stimuli likely contributes to atypical social reinforcement learning and decreased motivation to engage in social interactions, commonly seen in individuals with ASD [34, 35].
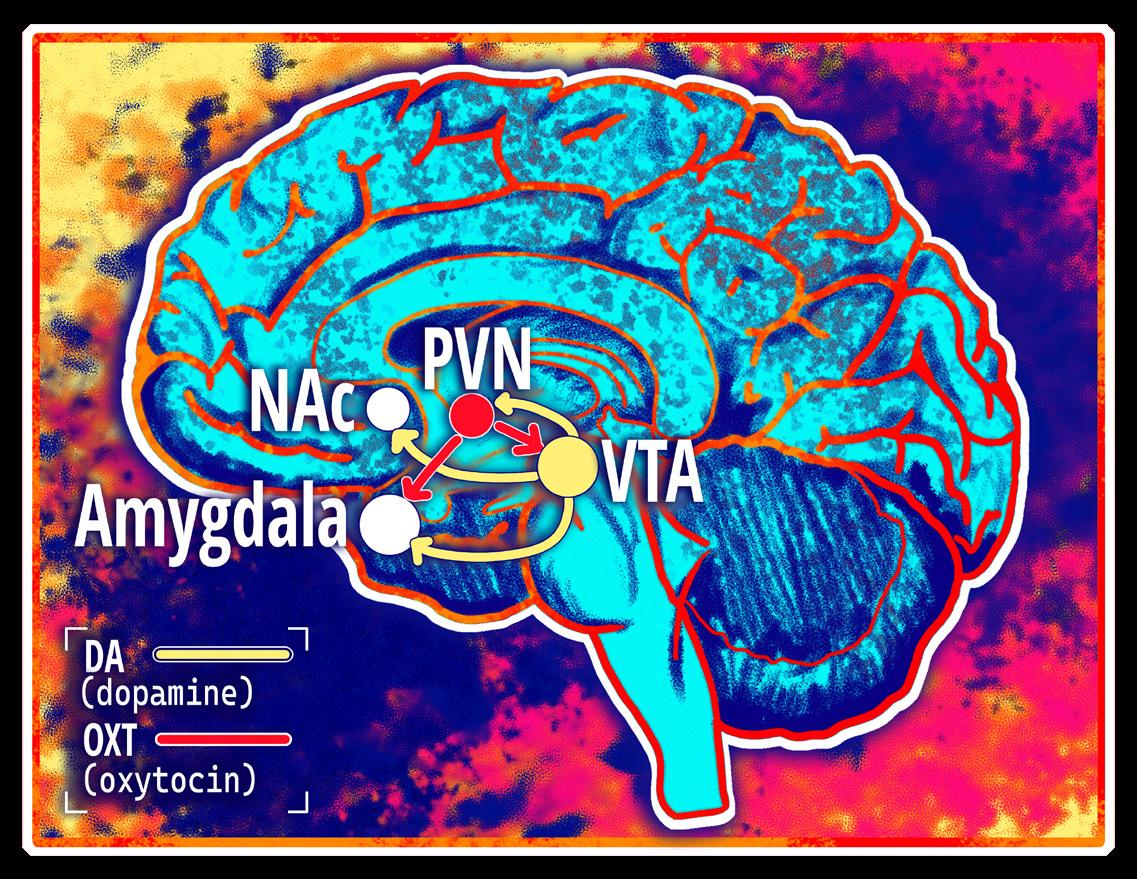
In addition to dopamine, oxytocin plays a key role in many social behaviors, and both neuromodulators regulate one another’s release through multiple mechanisms [36, 37, 38]. Oxytocin influences a multitude of social behaviors, including social recognition and forming close relationships with other individuals [39, 40]. It is primarily produced in the paraventricular nucleus of the hypothalamus (PVN), a region involved in controlling diverse survival behaviors such as appetite, alertness, social and defensive behavior, and pain [41]. The PVN delivers oxytocin to numerous regions of the brain, including the VTA, where it binds onto dopamine neurons [36, 42]. Oxytocin increases dopamine release from the VTA into brain regions such as the nucleus accumbens (NAc), helping the brain encode positive social value via reward prediction error [36, 43, 44]. Therefore, oxytocin can influence how rewarding a behavior ‘feels’ by modulating dopamine release from the VTA to the NAc [36, 45].
Oxytocin release from the PVN also serves a key role in enhancing the salience, or noticeability, of social stimuli, which help guide social behavior [46, 47, 48]. By directing the brain’s focus toward social stimuli instead of background distractions, individuals can focus on signals significant to the social environment [47, 48]. Oxytocin elicits this effect by regulating various sensory pathways, notably through its interaction with the brain’s dopamine system [46, 47, 48]. Similarly to how oxytocin regulates reward-encoding dopamine neurons, oxytocin is also believed to modulate the salience of social stimuli by regulating dopamine firing [46]. Unlike the dopamine neurons involved in reward prediction error, which only increase activity in response to an unexpected positive reward, these dopamine neurons increase firing in response to any
salient stimuli, positive or negative [43]. Oxytocin, therefore, functions like a biological DJ turntable by selectively amplifying social signals — such as visual and auditory cues — while dampening non-social signals to help the brain prioritize signals most important to social interaction [49, 50].
Oxytocin and dopamine signaling also regulate social behavior by modulating the activity of the amygdala [51]. The amygdala is a brain region mostly known for its role in processing emotions and regulating fear and anxiety responses, which in turn contribute to various social behaviors [52, 53]. Increased oxytocin release can help encour age social engagement by decreasing anxi ety-related signaling from the amygdala [51, 54]. However, it is be lieved that oxytocin does not elicit these effects on its own; in stead, it interacts with other neuromodula tors such as dopamine to produce them [51]. Oxytocin and dopamine receptors are found to gether throughout the amygdala; therefore, it is likely that both oxy tocin and dopamine signaling contribute to the regulation of anx iety-related behaviors and, in turn, promote affiliative behaviors [51, 55, 56].
Dopamine signaling between the VTA and NAc helps encode reward and reinforce behavior, and thus, reductions in dopamine release can disrupt social reward processing [27, 33, 43]. Therefore, individuals with ASD may not get as much reinforcement from positive social interactions, causing them to feel less motivated to engage or initiate them in the future [33].

Since oxytocin and dopamine mutually influence one another, dysfunction in both neuromodulators has been implicated in ASD [36]. Dysregulated oxytocin and dopamine signaling can cause atypical social reward processing and social salience encoding [57, 58]. This dysfunction likely contributes to social deficits associated with ASD, like avoiding eye contact or becoming overstimulated by certain environments [36, 59, 60]. Deviations in oxytocin levels can disrupt the regulation of dopamine neurons in the VTA, often resulting in decreased dopamine release [36, 42, 45].
Dysfunction in oxytocin-dopamine signaling has a similar effect on social salience, as dysregulated release can impair the brain's ability to differentiate the importance of social stimuli relative to unrelated ‘background noise’ [46]. As a result, individuals with ASD experience difficulty socializing, as too much sensory input and background noise can make it challenging for the brain to filter and process social cues [37, 38, 61]. In a noisy environment such as a playground, a child with ASD might struggle to notice a friend waving or calling their name, as dysregulated oxytocin and dopamine signaling make it hard for their brain to mark these social cues as important amidst all the other sensory stimuli. Furthermore, this may lead to a feedback loop of feeling left out or having difficulty making new friends. Dysfunction in oxytocin-dopamine signaling can also contribute to emotional challenges associated with ASD, such as atypical regulation of the amygdala [57]. When oxytocin and dopamine bind to receptors on the amygdala, they can help regulate amygdala activity and decrease anxiety-related signals, subsequently promoting affiliative behaviors [51, 55, 56]. When this regulation is disrupted, individuals may exhibit heightened anxiety and reduced motivation to engage in social interactions — core features associated with ASD [57, 62].
Research into ASD all shares the same primary goal: to increase knowledge to improve the quality of life and long-term outcomes for individuals with autism and their families [63, 64]. Interventions such as pharmacological and behavioral therapies do not aim to fix or eliminate autism, but rather strive to manage symptoms [65, 66]. Importantly, therapies for autism are often used in conjunction with one another to holistically address symptoms [67]. Despite oxytocin and dopamine dysfunction in autism still being a relatively new area of research, some clinical trials targeting these neuromodulators have shown promise [68, 69, 70]. In both children and adults with ASD, oxytocin administration in children and adolescents with ASD has been shown to improve various symptoms [68, 71, 72]. Adults with ASD showed a significant reduction in repetitive behaviors for up to four hours after administration of oxytocin and increased attention to social cues, such as others' eyes or mouths [68, 72]. In children and adolescents with ASD, oxytocin has been shown to improve social abilities such as communication, motivation, and awareness [71].

Much like oxytocin, clinical trials testing medications targeting dopamine deficits have also shown efficacy in treating associated ASD symptoms [69, 70]. There are only two medications currently approved by the Food and Drug Administration (FDA) for symptoms associated with ASD: risperidone and aripirazole, both of which target dysregulated dopamine signaling [73, 74]. Children treated with risperidone exhibited decreased sensory processing abnormalities, decreased hypersensitivity, and decreased
irritability [69]. Similarly, aripiprazole was shown to improve symptoms associated with ASD, such as irritability, mood swings, and severe tantrums [75]. Though not developed for ASD treatment, clozapine, believed to increase dopamine release, yielded similar results to aripiprazole when taken for management of ASD symptoms [70]. Specifically, clozapine administration was found to decrease severe disruptive behaviors such as self-injury and tantrums [70, 76]. But due to severe side effects including seizures, decreased immune response, and heart issues, it is only used as a last resort intervention for people with severe, treatment-resistant symptoms [70]. Despite encouraging results after treatment, research into the efficacy and safety of these interventions remains limited [77, 78]. Continued research into oxytocin and dopamine dysfunction in ASD is critical for guiding future clinical trials aiming to discover new pharmacological therapies [24, 79, 80].
ASD reflects a complex interaction between neurobiology and behavior, particularly in neural systems that regulate social reward, perception, and motivation [81]. Research into the neuromodulators oxytocin and dopamine has begun to clarify how dysfunction in social reward processing and social salience encoding contributes to social challenges experienced by individuals with ASD [62]. While these findings do not insinuate that autism is something that needs to be ‘fixed,’ they offer valuable insight for understanding how individuals with ASD experience and navigate the social world [65, 66, 82]. Rather than seeking out a single cause or universal treatment, ASD research aims to develop a more nuanced and individualized understanding that emphasizes the variability of the disorder and the need for personalized interventions [83, 84]. Of course, the decision to seek treatment is dependent on the individual, and many people with ASD do not wish to seek therapeutic intervention. As research into ASD continues to evolve, it holds promise for developing targeted pharmacological and behavioral interventions that work to alleviate symptoms and improve quality of life — ensuring individuals with ASD have access to tools and support that can help them better navigate social environments [24, 65, 66].
References on page 67.

by Leo Mahlke | art by Leo Mahlke
Why is it that babies love playing peek-a-boo with their parents, or building block structures and pretending to cook meals? Play is an integral part of childhood that is not only fun for children, but also important in building foundational skills across development [1, 2, 3]. A baby begins to mimic actions, socially interact, and develop physical abilities through a variety of seemingly simple activities, including peek-a-boo, crawling, or shaking a toy rattle [3, 4]. Once they have a foundation, children can learn to plan and make goal-directed decisions while playing with building blocks [5, 6, 7]. In their toy kitchen, children think creatively and representatively to regard a piece of wood as a hot stove, and a piece of red plastic as an apple [3, 8]. As they play pretend with their friends, they take on a new role, such as pretending to be a mother, and this facilitates the development of social understanding and empathy skills [9]. Throughout childhood, play is a critical, integrative driver of social, cognitive, and sensorimotor development, shaping brain function and even acting as a powerful therapeutic tool for enhancing communication skills [1, 2, 3].
As a child begins to scribble with crayons on a blank sheet, unsure of what the final image will become, they choose certain lines to retrace. Gradually, their scribbles form into a sketch resembling a kitten. As they continue adding more scribbles, they draw a new connection and think their drawing actually looks more like a dragon. The child adds a castle and repurposes fainter sketch marks to create fire bursting from the dragon’s mouth. A similar, though longer, process is occurring at the same time in the child’s brain [10, 11, 12]. The brain undergoes structural and functional changes in response to the environment, a process referred to as neuroplasticity [10]. Neuroplasticity is strongest in childhood as many neurological connections between brain cells form, strengthen, and shift depending on frequency of use [10, 11, 12]. With repetitive use, these connections are reinforced and reassembled into stronger, more

efficient circuits, while those that are seldom activated are reorganized to support other relevant cognitive functions [10, 11, 12]. Therefore, during the window of childhood, play functions as an essential contributor to the development of a unified brain — physically connecting regions associated with emotion, cognition, language, and physical movement [1, 3].
As a child plays, a protein called brain-derived neurotrophic factor (BDNF) increases in abundance [3, 13, 14]. BDNF is a vital protein that supports the brain by aiding in the survival, growth, communication, and specialization of the brain cells called neurons, which respond to stimuli [5]. During play, BDNF production increases in brain regions that process memory, decision-making, self-regulation, attention, fear and aggression, visual integration, and motor skills [5, 6, 15]. Picture two children constructing a block city with long, car-filled roads connecting the buildings. Arranging the blocks engages motor and judgment skills, which stimulate BDNF production in the prefrontal cortex (PFC), a brain region responsible for decision-making, planning, logic, and memory [5, 16].
The increase of BDNF enhances communication between neurons in the PFC such that signals are easier to pass between neurons [5, 17]. Just as the block city has roads that carry toy cars from one building to another, neurons send chemical signals to one another [18]. The toy cars parked outside each building resemble when the chemical signals arrive at the receiving end of an adjacent neuron, and bind to its receptors [18, 19]. When BDNF production is amplified in the PFC, neurons grow more branches that strengthen and stabilize the connections between brain cells, increasing receptor density and enhancing signaling efficiency [5, 17, 18]. These molecular changes support learning, flexibility, and memory, allowing children to build more complex block cities [16, 20].

Think back to a baby playing peek-a-boo with a parent. The baby is smiling, giggling, and wanting the play to continue because they are incentivized by their own enjoyment [4, 21]. They feel rewarded by play because of the release of dopamine, a chemical messenger that reinforces behavior by regulating processes such as incentivization [4, 21]. Similarly, dopamine reinforces social play and supports learning, neuroplasticity, and stress mitigation [3, 4, 5]. Play continues to be rewarding even as children get older due to the development of the hippocampus, a brain region that consolidates learning by turning new experiences into long-term memories [22, 23]. As a result, dopamine flow in this region allows playful activities to capture a child’s curiosity and attention,
promoting incentivized, self-motivated learning, and memory consolidation [22, 23]. While dopamine and neuroplastic changes highlight how play shapes the brain at a cellular level, these effects do not occur in isolation [22, 23]. Instead, they influence how entire brain regions interact, revealing how play engages the brain as an integrated system [22, 23].
The seemingly simple play that children engage in often involves a variety of different skills and senses, requiring the brain to use multiple regions simultaneously [1, 2, 5]. As these regions are repeatedly co-activated over time, the connections between them gradually strengthen [1, 2, 5]. Consider a child reaching into a dollhouse, picking up a doll, and role-playing as the character by exclaiming, ‘I’m hungry!’ [1]. The child moves the doll through the house in search of something to eat, accidentally knocking over a toy chair in the process [1, 24, 25]. After picking up the chair, the child also resolves the character’s hunger by placing the doll in the chair at the kitchen table with some plastic food [5, 24]. Within this small and seemingly insignificant moment, the child has simultaneously used language, emotion, creativity, problem-solving, and focused attention [1, 2, 5].
One vital brain network impacted by play is the cortico-cerebellar system [24]. The system comprises the interaction between the premotor cortex, which aids in motor planning, and the cerebellum, a region that executes coordinated motor responses [24, 26]. The cortico-cerebellar system also supports the prediction of actions and the ability to make spontaneous adjustments [24]. The moment the child realized they had knocked over the chair, the cortico-cerebellar system detected a mismatch between the predicted actions and those actually executed [24]. After the child fixes the chair and decides to place the doll in it, they may also predict that a limp doll in a chair may fall out of the seat; therefore, they will make sure it is propped up [24]. As the cortico-cerebellar system becomes more efficient through repeated use, actions become faster and smoother [5, 24]. By playing, the child can also practice calmly devising and executing solutions, demonstrating how role-playing scenarios may help children learn to apply these emotional-regulation skills in real life [1, 5, 27]. This is because pretend play promotes connectivity between the PFC and the amygdala, a brain region associated
with fight-or-flight tendencies [5]. Over time, an interactive relationship forms, allowing the PFC to regulate the amygdala’s emotional extremes by filtering them through logical reasoning [5].
As babies grow, their increasing mobility leads them to more perceptual exploration and learning opportunities, strengthening the interdependence between physical, sensory, and cognitive skills [1, 2, 3]. Imagine an infant crawling to a bin of plastic food and dumping it out to search for a toy apple. When playing pretend kitchen, the baby sees the shapes and colors of their toys and hears the crashing sound they make as they tumble from the overturned bin. The baby may even feel the strong contrast between the rough, bumpy texture of the carpet they are crawling on and the smooth surface of their plastic apple, which they might even attempt to bite. Here, the infant is engaging in sensorimotor play, a style of play in which babies actively explore the perceptual properties of objects in their environment [2]. Sensorimotor play is typically initiated through imitation, engaging mirror neurons [1, 2, 28]. Whether we are watching an action or executing it ourselves, mirror neurons respond the same way [1, 2, 28]. Imagine a mom play-

ing with toy kitchenware alongside her baby. She shows them how she stirs her empty bowl with a spoon, and the infant observes before attempting to mirror this motion with their own bowl and spoon. The baby became engaged in play by observing their mom stir a toy bowl, and the activation of their mirror neurons supported them in mimicking her motion [1].
As the infant crawls and dumps out a large bin of food, they use gross motor skills involving wholelimb movement, which support later milestones such as running and jumping [3, 8]. However, when the infant uses their hand to grasp the spoon and drop the plastic apple into their bowl, they use their fine motor skills — precise movements executed by smaller muscles which can be improved with play [3, 8]. Repeated muscular engagement promotes neuromotor development, thus supporting children’s emerging independence and exploratory behavior [2]. Even something as simple as an infant sitting on the floor and reaching for a toy to play with may help refine their range of motion and spatial awareness [2].
In addition to motor development, pretend play encourages children to think both creatively and logically [29, 30]. In this sense, children must flexibly engage in different pathways of thinking and idea-generating, such as divergent and convergent thinking [29, 30]. Divergent thinking is characterized by flexible, open-ended brainstorming, while convergent thinking leads the mind to a single, concrete, and logical solution to a problem [29, 30]. When a four-year-old child cooks a make-believe meal of spaghetti, they recruit their executive control network (ECN), which manages decision-making, problem-solving, and memory [2,31]. Therefore, the ECN allows the child to remember and execute the steps of cooking the meal in a methodical manner [5, 32, 33]. However, the child notices that there is no meatball toy [3, 33]. Now, they must think flexibly to make a logical plan that replaces meatballs with another object [3, 33]. The child must employ divergent thinking, prompting them to creatively devise an alternative path [29, 30]. They scan their environment, see round pom-poms, and decide that the pom-poms will represent meatballs. However, when the child finally settled on the pompoms and completed the order, they used convergent thinking skills to make and execute a final decision [29, 30]. Both divergent and convergent thinking are enhanced through childhood play because the child is constantly generating a mix of fantastical, emotional, and imaginative ideas and using logic to contextualize them [29, 34, 35].
Producing free-flowing thoughts to generate ideas
engages a large network known as the default mode network (DMN), which is active during imaginative and unprompted thinking, such as daydreaming [34, 35, 36]. The DMN and ECN function in parallel to integrate creative thought with goal-directed evaluation, allowing children to develop ideas and identify solutions [30, 34, 35]. More efficient communication between the DMN and ECN is associated with creative abilities, such as switching between spontaneous idea generation and analysis, thereby allowing for the refinement of ideas [29, 34, 35]. As we grow, even seemingly simple interactions such as playing pretend kitchen can influence the ways in which the brain builds relationships between physical, perceptual, and cognitive processes, leading to the development of increasingly complex skills throughout life [1, 2, 3].
Engaging in play gives children the chance to not only improve their cognitive skills, but also to develop social skills like perspective-taking and empathy [37]. Imagine a group of children playing pretend family, with each child acting out how each family member is portrayed in their own life. ‘Mom’ argues with ‘Sister’ over bedtime. ‘Dog’ barks to get ‘Brother’s’ attention, and he gives his pet some pretend table scraps. ‘Dad’ announces that the baby doll is crying because it got left alone in its cradle. Here, the children are assigning feelings and motives to their own fictional characters [9]. At the same time, they also understand that the other children’s behavior reflects the character they are pretending to be [9]. Therefore, when they play pretend-family, the children are beginning to exercise their emerging empathy, the cognitive ability to anticipate the thoughts and feelings of others while also understanding that they are all pretending [9, 38].
By practicing empathy during play, children can also rehearse social situations ranging from conflict resolution to caregiving [9]. For example, when the child pretending to be ‘Dad’ anticipates that the baby will cry when left alone, they are taking on the baby’s perspective [9, 39]. The child uses both creative thinking and empathy when they decide to stay with the baby doll and care for it [9, 39]. Pretend play recruits a part of the brain known as the posterior superior temporal sulcus (pSTS), which supports a wide variety of processes in social cognition, such as perspective-taking and empathy [39]. Therefore, even when

the child playing ‘Mom’ tries to get the ‘Sister’ to go to bed, and the ‘Dog’ must bark to ask for food, the children work on developing an understanding of the emotions and perspectives of others who are different from themselves [9, 39]. Engaging in these activities helps children better understand others’ internal thoughts and feelings, setting up critical lifelong empathy skills [9, 39].
Because pretend play can help children develop perspective-taking skills and empathy, it can be a powerful tool to implement into therapies for children with neurodevelopmental disorders such as autism spectrum disorder (ASD) [3, 37]. To varying degrees, people with ASD typically engage in restricted, repetitive behaviors and face challenges with sensory integration, social skills, nonverbal communication, and staying in sync with others during conversation [3]. When two people have a conversation, they have several backand-forth exchanges, maintain eye contact, use appropriate intonation, and adjust their expressive body language as the conversation shifts from topic to topic [40, 41]. As the pair navigates this conversation, they demonstrate interpersonal synchrony — the ability to maintain shared rhythm, coordination, and attention while speaking [40, 41]. People with ASD are more likely to struggle with interpersonal synchrony and often have irregular pausing and timing, lack of
eye contact, and responses that may appear emotionally disengaged [40, 41]. Challenges with interpersonal synchrony are associated with atypical activity in the mirror neural network [42]. Some people with ASD may also show reduced activity in brain regions involved in tracking eye gaze and joint attention, a process in which two people each intentionally look at and pay attention to the same object [43, 44].
The integration of pretend play into therapy — or play therapy — for children with ASD helps activate brain regions that facilitate social interaction [3]. Play therapy is not an attempt to make people with ASD act and communicate in ways considered more ‘typical’ [3]. Instead, this intervention aims to help people with ASD practice skills to convey their wants, needs, and thoughts [3]. Since play therapy supports the development of the PFC, engaging in this activity could help people with ASD navigate changes in conversational rhythm, turn-taking, and understanding of shared goals [40, 41]. Play therapy gives people with ASD a chance to practice different social situations, providing them with tools to apply in real-life conversations [45]. As mentioned earlier, the pSTS is activated during social interactions and processing in children, strengthening the region as it is repeatedly activated, which can also help support communication skills for people with ASD over time [45]. Play also strengthens children’s observation-execution matching systems and shared attention on an object or activity, making it an effective tool for therapy [45, 46]. By engaging in an activity such as playing kitchen alongside a therapist, children with ASD develop skills by mirroring their therapist’s actions [3, 46]. Prominent forms of therapy for people with ASD have frequently been reported as highly physically and mentally abusive, viewing ASD as a pathology in need of curing rather than a neurological difference [47]. Play therapy, on the other hand, is intended to be non-disruptive and enjoyable while also being neurologically beneficial [3].
Pretend play can support children throughout development, both in its free and spontaneous forms and when it is structured, such as in therapeutic settings [3]. Though crucial to their developmental milestones, the children’s pretend kitchens, blocks, and dollhouses might be left to collect dust. With the increasing domination of technology, children are interacting more and more with electronic devices rather than real toys [3, 48]. As mobile games progressively replace in-person play, children are becoming de-
prived of the interactions that are crucial to childhood development (See the article on screen time on page X for more information on this topic!) [39, 48]. Given the benefits that play offers to childhood development, ensuring unstructured play is a part of the childhood education experience, such as recess, gives children the space to interact with their environments in free and unprompted ways [49, 50]. While structured play can be beneficial in a therapeutic environment, unguided play allows children to express themselves freely, be curious, and learn from mistakes [49, 50]. Even though play may be seen as a casual pastime, or even a break from learning, it is actually an essential part of childhood and should be taken seriously when it comes to raising children and building education systems [49, 50, 51]. It is vital that we evaluate the tools used to support children’s learning.
References on page 71.

by


Dinosaurs have been brought back to life. The prehistoric creatures roam free across a small strip of islands to which travel is strictly prohibited. The scientific disasters that transpired at the Jurassic Park and World theme parks have left Chris Pratt and Bryce Dallas Howard with an impossible task: to venture to one of the islands to save the dinosaurs from a volcanic eruption. This dangerous mission drives the plot of Jurassic World: Fallen Kingdom (2018), the fifth installment in the Jurassic Park film franchise, which has enraptured audiences since 1993 [1, 2]. A central challenge of the film is the cleverness of Chris Pratt’s favorite Velociraptor, Blue, who learns complex tasks and demonstrates emotional intelligence like a toothy, lethal golden retriever. Blue is touted as ‘potentially the second most intelligent piece of life on this planet,’ inflating the Velociraptor’s intelligence to near-human levels [2, 3]. While the Jurassic Park films suggest the possibility of highly intelligent dinosaurs, another widely held belief depicts them as mindless hunting machines mercilessly swept from the planet 66 million years ago [4, 5]. How have these two vastly different views formed about dinosaur intelligence? As the largest, most diverse, and most abundant creatures on the planet, dinosaurs dominated the planet for a remarkable 160 million years,
such as multi-component tool use, social group organization and cooperation, or complex language [8, 9]. So, how intelligent were the dinosaurs? The field of paleoneurology has emerged to tackle this question, relying on techniques similar to those of anthropologists, archaeologists, and neuroscientists to investigate dinosaurs’ cognitive capabilities [10]. Granted, these methods primarily come from fields focused on human life and inevitably color our understanding of the prehistoric creatures at the expense of the unique contexts in which they actually lived [5, 10,].
The term ‘dinosaurs’ is a colloquial catch-all for the scaly creatures that ruled the planet long before humans, but describes a very diverse group of organisms [6, 11]. Dinosaurs were reptiles who lived from roughly 230 to 66 million years ago, when an asteroid struck the Yucatán Peninsula in modern-day Mexico, wiping out three-quarters of all animal and plant species [8, 12, 13]. Birds are direct descendants of the only dinosaurs to survive this extinction event, so your pet chicken or the hummingbird at your bird feeder is technically a living dinosaur [14, 15, 16]! Since we do not live in the fictional world of Jurassic Park and cannot directly evaluate dinosaurs, birds are useful proxies for inferring dinosaur brain anatomy and function [8, 10, 17].

Before attempting to compare the brains of birds and dinosaurs, it is important to emphasize the diversity of dinosaurs across three primary groups: ornithischians, sauropods, and theropods [8]. Famous ornithischian dinosaurs include the threehorned Triceratops and the spiny Stegosaurus [17]. ‘Ornithischian’ in Greek can be translated to ‘birdhipped,’ as ornithischians’ pelvic structures resemble those of modern-day birds, even though birds did not evolve from the ornithischian lineage [18]. Sauropods were similarly misnamed, since ‘sauropod’ means ‘lizard-footed’ in Greek, even though their gigantic feet actually bear
little resemblance to those of modern-day lizards. The Brachiosaurus, Brontosaurus, and Titanosaurus make sauropods the lovable, gentle giants of the Jurassic Park franchise [1, 18, 19]. Finally, theropods are likely the most recognizable dinosaurs, including the famous Tyrannosaurus and Blue the Velociraptor, and are the most common subjects of paleoneurological study [8, 20]. Translating to ‘beast-footed’ in Greek, theropods primarily walked on two feet, ate mostly meat, had three toes or claws on each of their four limbs, and many were likely covered in feathers, much like birds [21]. Fittingly, modern-day birds evolved directly from theropods [7, 17].
With at least 66 million years of evolution differentiating them, modern-day birds and dinosaurs are functionally and anatomically distinct from one another [7, 14]. Still, birds share some key similarities with Avialae dinosaurs — the theropods most closely related to birds — that make them apt comparisons [21]. For example, the fossil record suggests that late avialan dinosaurs like Archaeopteryx and Anchiornis likely had wing and tail feathers, also implying the presence of the associated neurological mechanisms [10, 21]. Birds’ brain activity while completing cognitive tasks like solving puzzles can also be used to hypothesize which neurological capabilities may have been shared by dinosaurs with similar morphological characteristics [10]. Another avenue of inquiry comes from dinosaurs’ and birds’ next closest living relatives: crocodilians, a group of reptiles that includes alligators and crocodiles [17]. Crocodilians share a range of morphological similarities with many dinosaurs, including their large, pointed teeth and scaly exteriors [22]. Since birds and crocodilians are still alive, the features they share with dinosaurs provide clues about the behavior, lifestyles, and cognitive abilities of these extinct beasts [17].
Picture the classic image of a brain: a gelatinous, pinkish-gray mass. A soft and fleshy organ, the brain does not fossilize — only hard tissues like bone do [17]. So, when we use the fossil record to approximate dinosaur morphology and anatomy, we are limited to biological information that does not include the brain itself [10]. Using what evidence we do have, such as tooth shape or fossilized tracks, we can elucidate aspects of dinosaur behavior, including the general diet of a specific species, nesting sites and mating behavior, and preferred climate [22, 23]. For example, analysis of a site in Colorado suggests that theropods
engaged in ceremonial courtship rituals similar to those of modern-day birds. This implies that theropods may have exhibited such flamboyant bird-like behaviors as flapping, dancing, and vocalizing [23, 24]. In the absence of brain tissue, paleoneurologists make cognitive inferences using fragments of dinosaur skulls, called endocasts, which are fossilized cranial cavities with indentations left by the brain [10]. Endocasts can provide neuroanatomical reference points that indicate the relative size of both the brain and its specialized regions using computed tomography (CT) scanning to digitally reconstruct the brain [17, 25, 26]. Imagine a T. rex being shuttled into a doughnut-shaped CT scanner — that’s what this technique attempts to simulate! Endocasts can reveal the relative sizes of the olfactory bulb, responsible for processing smell, or the flocculus, which plays a role in stabilizing vision and is generally larger in flying creatures than in terrestrial animals [27, 28, 29]. A larger olfactory bulb may indicate a well-developed sense of smell, an important trait for crocodilian hunting strategies [30]. Meanwhile, a larger flocculus points to greater agility or even flight, as in modern-day birds [27].
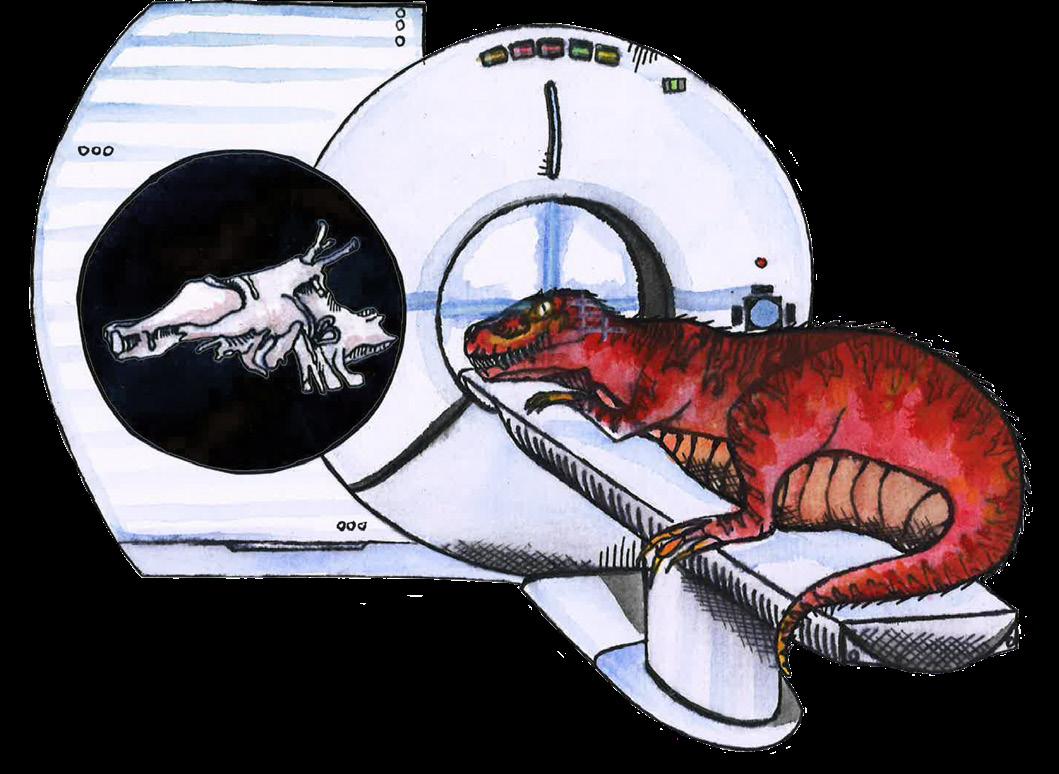
Endocasts are extremely useful for revealing sensory or anatomical traits, but discerning ‘intelligent’-type behaviors from endocasts is more challenging [31]. Indeed, it is difficult to identify levels of intelligence even in living animals, as defining what qualifies as an intelligent behavior or trait is in itself, quite a challenge [32, 33]. Debates surrounding the emotional intelligence, tool-use capacity, and social dynamics of, for instance, great apes — humans’ closest living relatives — have stumped comparative biologists for decades [34]. Tool use is widely regarded as a measure of intelligence, a trait once believed to
singularly occur in humans, but has since been observed in various animals, including birds and crocodilians [32, 35, 36]. An additional marker of intelligence is problem-solving skills; innovation is associated with reduced extinction risk and larger brain size [37]. These metrics of intelligence can be associated with physical traits that endocasts and fossil evidence might reveal, such as opposable digits, a large brain cavity, bipedalism, and language centers [8]. Ultimately, intelligence is largely immeasurable and subjective, especially when applied to an extinct species [38]. Nevertheless, endocasts and fossil evidence can help uncover pieces of a larger cognitive puzzle [10].
The most straightforward method of endocast analysis is to assess total brain size [3]. An early hypothesis posits that brain size directly correlates with cognitive ability [33]. However, if this were true, whales, which have the largest recorded brains, would be the most intelligent animals on the planet [39]. Alternatively, relative brain size — the comparative size of an animal’s brain to its body — has a stronger correlation with intelligence and is quantified by the Encephalization Quotient (EQ) [3, 40]. EQ tends to be mammalian-centric, so other metrics have emerged for birds (BEQ) and reptiles (REQ) to more accurately compare EQ across species [36, 40]. Large theropods like T. rex tend to have normal-to-high BEQs and REQs comparable to those of living birds and reptiles, suggesting that theropods could have been as intelligent, or perhaps even more intelligent, than modern-day birds
heavily on smell and therefore have proportionally larger olfactory bulbs than humans, who depend less on smell [43]. Similarly, comparing the relative sizes of brain regions from dinosaur endocasts with those of birds and crocodilians can reveal functional differences that point to lifestyle and cognitive traits in dinosaurs [27]. The boundaries between brain regions can be roughly identified by surface details left on the endocast that indicate the peaks and valleys of the outer layer of the brain [31]. The volumes of these brain regions can also be calculated with CT scanning and compared across species [31]. Ultimately, a broad analysis of all available data — fossil evidence, evolutionary relationships, and endocasts — can help reveal a comprehensive picture of dinosaurs’ lives [8]. Only then can we start to discern if they were brilliant strategic animals, brainless hunting machines, or somewhere in between [8].
Most dinosaur brains likely resembled bird brains, and no, this is not an insult [3, 8, 10]. Birds have wonderfully intricate brains that support many cognitive functions, such as complex vocal language — a trait otherwise only discovered in mammals — robust memory, innovative problem-solving, and tool use [32, 44, 45]. You are probably most familiar with the mammalian picture of a brain, with its wrinkly grayish-pink exterior. However, the mammalian brain has had over 300 million years to differentiate itself
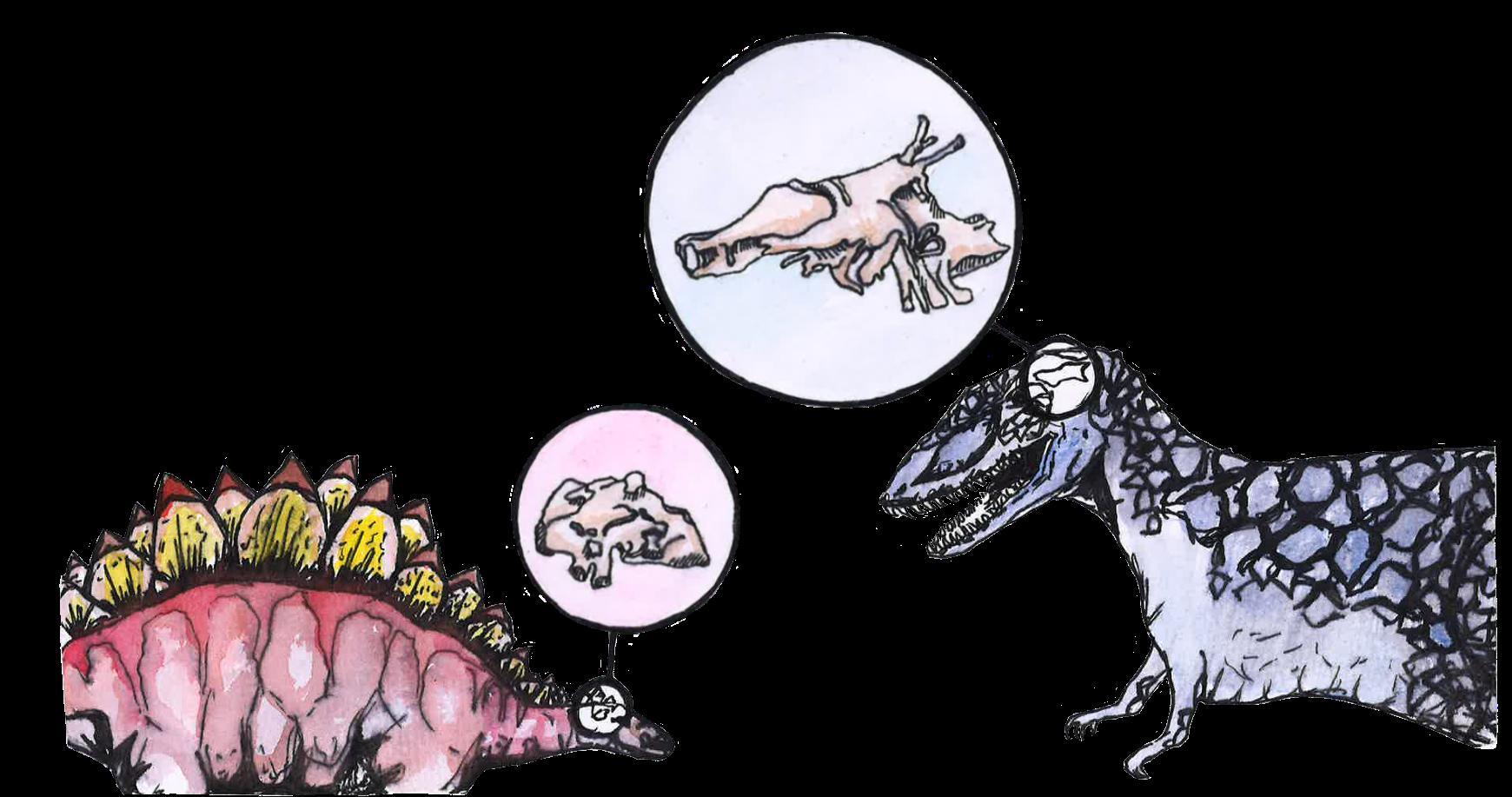
functions, and conscious mental processes [49]. Instead of a neocortex, birds and reptiles have a functionally equivalent structure called a pallium [50]. A major difference between the pallium and the neocortex lies in the optimization of surface area and volume [50]. The wrinkles of the neocortex allow for greater cellular connectivity, whereas the smoother avian-reptilian pallium hypothetically limits functional connectivity [8]. Because birds, reptiles, and dinosaurs all share a common ancestor, it is almost certain that dinosaur brains followed their pallial level of organization — a conclusion reinforced by endocast analysis [8, 47]. The avian pallium has an important substructure, the nidopallium, which is the seat of sophisticated decision-making in birds and has been linked to emotional, motivational, and perceptual processes [50, 51]. Since reptiles lack a nidopallium, evolutionary evidence alone cannot tell us whether dinosaurs had one [52]. Some dinosaur endocasts, however, reveal physical markings consistent with the presence of a nidopallium, though smaller in volume than those of modern-day birds [53]. If the presence of the nidopallium were confirmed, we could further infer the cognitive capabilities of dinosaurs, such as memory, decision-making, and innovation [53].
Collectively, evolutionary and endocast evidence support the argument that dinosaurs’ brains were both organizationally and functionally comparable to those of birds [3, 8]. Therefore, we can speculate that dinosaurs may have possessed some of the sophisticated cognitive abilities of birds [3]. For instance, many bird species have complex vocal language that rivals humans’ and may even use grammatical structure [44]. It is possible that dinosaurs were capable of vocalizations similar to those made by modern-day birds — a theory supported by fossil evidence of a similar voice box in both birds and dinosaurs [54]. Vocal learning in birds has been associated with larger brain size and greater problem-solving abilities, so bird-like vocalizations in dinosaurs may indicate enhanced cognitive abilities [37]. Many birds, such as crows, have demonstrated robust memories, innovation, and tool use [45]. Likewise, it has been speculated that dinosaurs could have used tools, based on what fossil evidence suggests about the dexterity of their limbs and digits [36]. Essentially, dinosaurs were likely not as cognitively complex as primates, as some have suggested, solely because of the organization of their brains [3, 8]. However, birds — which represent in brain structure what dinosaur brains probably looked like — are very intelligent animals [37]. So, dinosaurs, too, could have been as intelligent as their feathered descendants, except much, much toothier [8].
In addition to differences in the structural organization of mammalian and avian brains, they are also distinct in their organization at the cellular level [8, 50, 55]. The mammalian neocortex is highly organized and densely packed with neurons, or brain cells, that form six distinct layers and many functional columns, like an apartment building with hallways connecting units and an elevator between floors [56]. In contrast, the avian-reptilian pallium is more nuclear and radial in its neural organization, forming neural clusters, like a neighborhood with numerous cul-de-sacs off which houses radiate [52]. You can see how it might be easier to deliver a letter from one apartment to another than to drive all the way from one house on a cul-de-sac to a second house on a different culde-sac. The cul-de-sac model of organization theoretically limits the extent to which the avian-reptilian brain can expand and adapt, especially compared to the efficiency of the human brain [8, 52]. For instance, neuroplasticity — the capacity for change between connections in the brain — is a key process behind learning (57). In a nuclear system, neuroplasticity may be restricted because neurons are less connected to one another than in the layers of the mammalian neocortex [8]. The means of communication between neurons, like the houses, are restricted to travel along a central road, whereas in the apartment building, you could yell your message through the vents to reach the floor above you or climb the fire escape. Essentially, mammalian layered organization theoretically provides more avenues for connection and speed of transmission. While we can only hypothesize about the consequences of pallial organization on dinosaur intelligence, one theorist has used a comparative approach towards endocasts to defend the notion that dinosaurs could not have reached mammalian levels of sophistication [8]. However, these conclusions are based on research that is focused predominantly on mammalian learning and brains rather than on birds — an example of mammalian-centric bias in cognitive research [50, 55].
The vast size difference between dinosaurs and birds makes it even harder to know the degree to which the neural organization of bird brains can actually apply to dinosaur brains [3, 8]. The effect of scaling up bird brains to dinosaur proportions would impact both neuron density and neural length; in some cases, neural density is even more indicative of intelligence than EQ [8, 58]. Based on a generous scaling technique, one estimate suggests that dinosaurs’
neuron abundance was equal to that of primates, a conclusion that is highly debated [3, 17]. A separate endocast technique assumes that the T. rex cranial cavity was occupied less by brain tissue itself and more by fluid, characteristically more like crocodilians and less like birds. This endocast approach yields a lower brain tissue volume estimate and suggests that T. rex’s neural density was more similar to that of crocodilians than primates [17]. Together, these estimates highlight both the importance of evolutionary relationships and the challenges of relying on them to make inferences [10].
The tools that have been developed to piece together dinosaur cognitive abilities, namely endocasts and evolutionary comparisons, are helpful but imperfect, as they are subject to human bias and interpretation [5, 10]. A trend in comparative biology is the tendency to privilege our own brand of human intelligence, a line of reasoning that often places humans at the top of an evolutionary pyramid [59, 60]. The qualities and behaviors used to define intelligence, such as tool use, language, or problem-solving, are classic benchmarks of human cognition, and quantifying these traits in nonhuman animals may not be realistic [5, 8]. Take vision, for example. Humans’ vision is restricted to a narrow band of light on the electromagnetic spectrum we call ‘visible light’ — a term that reflects our anthropocentrism [61, 62]. However, many birds’ eyes are equipped to see not only visible light but also ultraviolet (UV) light, allowing them to detect natural patterns on prey, food, and mates that we cannot [61]. Based on eye structure evidence, it is possible that some bird-like theropods were also capable of UV vision [63]. These dinosaurs would have seen the Earth in quite literally a whole different light, illustrating how narrow our experiences are when compared across the animal kingdom [63]. To bridge this knowledge gap, some researchers have proposed a context-dependent approach that frames cognitive success as a species’ ability to adapt and thrive under its unique environmental pressures [5]. In this sense, T. rex’s tiny arms, razor-sharp teeth, and bloodhound-like nose reflect its brain’s ability to coordinate the motor and hunting skills required to become an apex predator in its ecological niche [64, 65, 66]. To understand how intelligent dinosaurs were, we must first recognize that being a dinosaur meant having an entirely different brain, body, and environment than our own [5].
The evidence for dinosaur intelligence remains subject to debate, and we may never know how these magnificent creatures ruled their version of Earth [8, 10, 17]. Still, we can respect the longevity of their mighty 160-million-year reign [6]. While humans may possess more ‘intelligence’ than our reptilian predecessors, we have inflicted permanent damage onto the planet in just 300,000 years: one million animal and plant species are currently threatened with extinction, and countless terrestrial and marine environments have been significantly altered over the course of humans’ reign [67, 68]. The unprecedented destruction of the human era may be the very reason that the ‘dinosaur age’ seems so idyllic [4]. However, modern technology may be able to repair some human-inflicted harm, with the new possibility of resurrecting extinct species like the woolly mammoth, the passenger pigeon, and, yes, the dinosaurs [69]. The existence of a real Jurassic Park may not be such a far-off fantasy, but would this reality be as picturesque as the screen portrays? The beloved franchise poses the question: Do we have the right to interfere with the laws of nature, and what are the potential pitfalls of our intelligence? Perhaps human-like intelligence is not to be coveted at all, but rather something that will destroy the world as we know it, like a self-made asteroid strike.
References on page 73.

by Julian Cardenas-Moncada | art by Nancy Duer

Sitting in a carbon-fiber cockpit while driving at approximately 200 miles per hour, British Formula 1 driver Lewis Hamilton crossed the finish line in a flash. Despite the waving checkered flag and roaring crowd, Hamilton kept driving at full speed, unaware he had just won the 2020 Spanish Grand Prix. During the race, Hamilton’s attention had remained focused on one goal: making each lap better than the last. Speaking to reporters after the race, he described being ‘in a different zone,’ so focused that he ‘didn't even know it was the last lap’ [1]. In high-stakes Formula 1 racing, drivers make countless micro-decisions at extreme speed, where the difference between winning and losing can be measured in millimeters or milliseconds [2]. The phenomenon that Hamilton experienced during the Grand Prix illustrates flow state.
First conceptualized in 1975, flow is a psychological state of deep task absorption characterized by intense concentration and reduced self-awareness, often emerging when skill and challenge are matched [3]. Flow leads to sharper attention, increased motivation, reduced self-consciousness, and a feeling of time distortion [4]. These traits have framed flow as a highly sought-after mental state desired among athletes, academics, and others striving to achieve peak productivity [4, 5]. Flow has recently gained popularity in online media through the TikTok trend ‘genuinely achieving flow state.’ These representations are inaccurate as flow is not a switch that can be flipped on instantaneously, but rather a complex mental state that, when given the right conditions, can be achieved by almost anyone [6, 7].
In day-to-day life, there are times when it feels as though stress overrides our choices, making already daunting tasks even more difficult. Stress is a physiological and psychological state that arises from a perceived threat and may even prevent a stressed individual from achieving their goals [8]. However, stress is not always negative; counterintuitively, it can positively impact an individual. Eustress is defined as a positive response to challenges that arise from successful goal-directed actions, meaningful momentary experiences, and stable personality qualities like resilience and optimism [9]. Stress, specifically while one is actively engaged with a challenging task, may become motivational when the task is perceived as both manageable and personally meaningful. [9, 10]. When motivational stress arises, individuals are more likely to enter and sustain flow [5, 11, 12]. This characteristic of flow can be observed across different high-performance domains, such as professional athletes or highly skilled musicians [13, 14]. Sustaining focus during a performance involves not only technical expertise but also the ability to manage the stress that comes from performing. When highly skilled musicians perceive a stress-inducing situation as a manageable challenge — viewing a concert as a creative opportunity rather than a threat — the resulting eustress can actually enhance performance [13, 14]. This positive stress response is most effective when the demands of the task are balanced with the performer's mastery [3]. By aligning high-level skills with these situational demands, musicians can sustain the deep engagement necessary to enter a state of flow [15]. While skill and stress management set the stage for entering flow, motivation can also propel an individual toward flow. When an individual is highly motivated to complete a task, they tend to invest more energy into achieving their goal [16]. An individual’s motivation is often multifaceted, comprising both extrinsic and intrinsic components. However, not all forms of motivation can lead to deep engagement [16]. Extrinsic motivation may come from recognition or rewards, such as winning a prize or a medal, whereas intrinsic motivation comes from within oneself, such as participating for the pure enjoyment of playing a sport [17, 18, 19]. These forms of motivation can coexist and may shift over time, depending on the individual and the task. At the beginning of their careers, some athletes may experience largely extrinsic motivation, including the glory of winning, gaining popularity, or making their family proud. As their skills and passion for the sport develop, though, they may
become more intrinsically motivated [19]. Ongoing research suggests that flow and intrinsic motivation are closely associated with one another, and it can ultimately be difficult to determine whether one necessarily leads to the other [17]. The combination of internal enjoyment and sense of accomplishment can make the activity itself rewarding, creating a self-sustaining cycle of motivation [20]. In other words, engaging in intrinsically rewarding activities increases the likelihood of entering flow, and experiences with flow in turn reinforce continued participation [18].
The relationship between intrinsic motivation and sustained engagement is closely tied to the brain’s reward systems — interconnected brain structures that rely on dopamine, a chemical that is involved in signaling reward prediction, motivation, and learning [21, 22]. During a task, a continuous loop of action and feedback facilitated by dopamine-releasing brain cells in the reward pathways helps sustain engagement and motivation [11, 23]. The striatum, a brain region that plays a critical role in decision-making, motor control, and reward processing, mediates a portion of this process [24]. The striatum is also involved in the interpretation of reward prediction error (RPE), a part of the reward system that differentiates between an expected outcome and reality. When an outcome turns out to be better than expected, the behavior responsible for the outcome is reinforced, and future similar actions will become more efficient and automatic [24]. Although RPE is a distinct neural process that occurs independently from flow, the increased automaticity it can produce may serve to significantly increase the likelihood of transitioning into flow [4, 25]. Activities that provide rapid, continuous feedback are particularly effective in supporting reward prediction, encouraging an environment optimal for flow [25]. Surgery is a prime example of how uninterrupted environmental feedback can enhance performance. While operating, a surgeon receives continuous visual and tactile feedback that informs their next excision or retraction, helping maintain focus [4, 6, 26]. The putamen, a structure integral to the brain's dopaminergic reward system and responsible for motor control and self-regulation, is also implicated in flow [27, 28]. The putamen responds to performance-related feedback during task engagement, helping to reinforce and automate the precise motor responses required for flow [28]. Repeated reinforcement supports perception-action coupling — the process by which sensory information is continuously integrated with motor responses — allowing
individuals to respond more efficiently with less conscious effort [29, 30]. This intrinsic reward system ensures that the flow state is not just an event happening at peak performance, but rather a self-reinforcing experience that individuals are neurobiologically wired to seek out [12, 31].
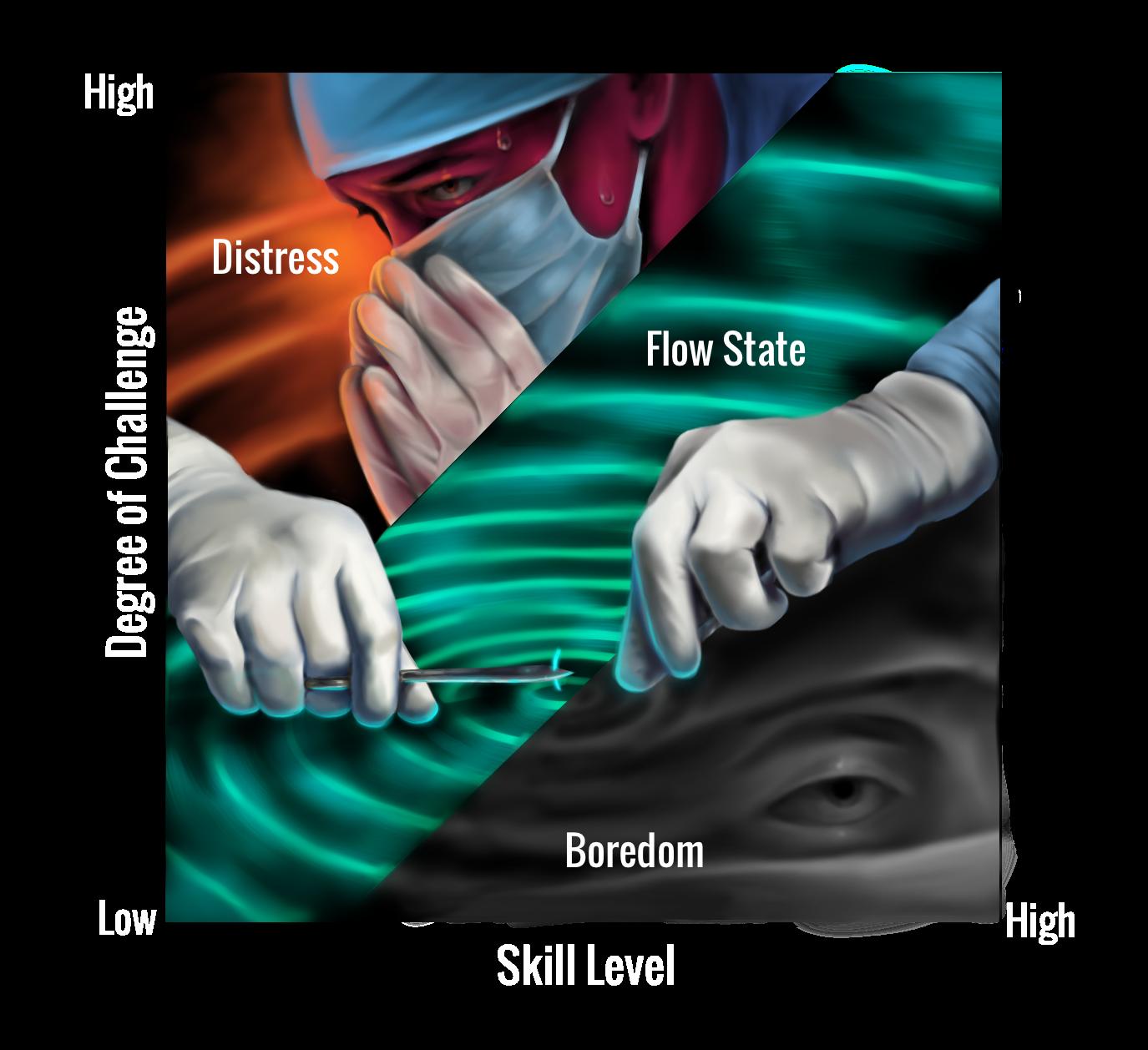
The trance-like concentration that led Lewis Hamilton to keep driving even after he had officially won the 2020 Spanish Grand Prix illustrates how it feels to experience flow [1]. As opposed to conscious monitoring of each movement, actions unfold automatically, awareness of the passage of time fades, and peak performance can be achieved [4, 12, 32]. Given the complex nature of flow, no single theory can fully explain how the state occurs in the brain [11]. Nonetheless, current models provide a useful framework for understanding flow, suggesting that it is the result of shifting activity between large-scale brain networks [11, 20, 32]. The task-positive network (TPN) is a broad term used to describe brain networks that increase in activity during externally directed attention and are thought to support focus, problem-solving, and goal-oriented action [33, 34]. In contrast, the default mode network (DMN) is associated with self-referential thoughts, which include mind-wandering and evaluation of past or future events [34]. Additionally, the Salience Network (SN) filters
internal and external stimuli and allocates resources to either the TPN or DMN depending on which network is perceived as relevant [25]. During flow, the SN allocates resources towards the TPN to focus attention on external stimuli [11]. Think of a cellist performing in an orchestra: while on stage waiting for their cue, they are flooded with internal and external stimuli. As their mind wanders internally between various self-referential thoughts, their external focus remains on the director. However, once their cue approaches and the moment at hand becomes more important, the SN begins to shift activity from the DMN, the cellist’s internal thoughts, to the TPN, their focus on the director’s movement. During flow, the SN is theorized to heavily prioritize the TPN over the DMN’s noisy chatter, leading to attention being shifted externally rather than internally [11, 33].
A complementary model for the reduced self-awareness and externalized attention characteristic of flow is the transient hypofrontality hypothesis (THH), which proposes that intense focus temporarily reduces activity in parts of the prefrontal cortex [35]. The prefrontal cortex is a brain region associated with many important cognitive processes, such as self-monitoring and conscious control, and is thought to be a key part of the DMN [12, 35]. As reduced prefrontal activity may limit self-referential thought through reduced DMN activation, the THH could help explain the diminished self-awareness experienced during flow [11, 12, 38]. Furthermore, as expertise increases, sustaining flow may require new challenges in which decreased DMN activity allows for creative adaptability [36]. As one becomes highly skilled in their field, the simple act of performing a task they've already mastered is no longer a challenge, and challenge-toskill matching becomes dependent on adapting to spontaneous ideas that are shaped in real time. In creative performance contexts, such as jazz improvisation, highly experienced musicians show decreased DMN activity and increased creativity during high-flow performances. When DMN activity decreases, creative ideation increases, presenting a new challenge that the performer must adapt to. As such, the decreased DMN activity both encourages flow through challenge-to-skill matching and allows the musician to perform more fluidly and creatively [37].
Similarly, digital environments such as video games are likely to lead to flow because they are associated with an increased activation in the TPN and decreased activity in regions of the prefrontal cortex [39]. They are often designed to maintain a close balance between skill and challenge, while also
providing clear goals and immediate feedback [40]. These features help sustain focused, externally directed attention and minimize boredom, frustration, and excessive self-monitoring, making games a particularly useful model for understanding the cognitive conditions that support flow [40]. Moderate levels of challenge produce the highest levels of engagement, while tasks that are too easy or too difficult result in lower activation [39]. Hence, the relationship between challenge and engagement in interactive tasks supports the idea that flow occurs when attention is optimally engaged and self-referential processing is suppressed [11, 41]. Together, these mechanisms suggest that flow may arise from a coordinated shift in brain activity [32]. This dynamic balance reduces self-awareness and enables efficient, automatic performance, producing the immersive, ‘being in the zone’ experience typical of flow [11].
When in flow, people can experience a sense of time distortion — the minutes may fly by or slow down [42]. We all rely on internal timing systems that help regulate both long-term biological rhythms and moment-to-moment time perception [43, 44]. Brain regions such as the basal ganglia and prefrontal cortex help keep track of incremental time changes by integrating rhythmic signals to estimate how much time has passed [45]. Under typical conditions, these signals are continuously monitored, enabling us to judge how much time has elapsed [46]. However, during flow, the combination of reduced activity in the DMN and prefrontal cortex, heightened attentional
engagement, and efficient motor execution alters how this timing information is processed [4]. For a professional driver like Lewis Hamilton, maintaining this level of concentration is vital; the brain prioritizes rapid motor execution and environmental feedback, and the typical signals used to track the passage of time are deprioritized. The time distortion could make the hours of high-speed racing feel like mere minutes, or, conversely, a single split-second maneuver feel long.
Notably, time distortion is not merely a subjective feeling — it is one of the few aspects of flow that can be represented quantitatively [42]. While many features of flow, such as enjoyment or a sense of control, rely on personal experience, shifts in time perception can actually be measured through differences between perceived and actual time lapses. During flow, reduced self-referential thinking and sustained attention lead to fewer internal thoughts and distractions. Conceptually, this translates to reduced temporal awareness, which contributes to a compressed sense of time [11]. Simultaneously, continuous engagement in perception-action coupling — where what you see is immediately translated into action — keeps your attention fully occupied with the task, reducing opportunities for conscious monitoring and contributing to a diminished awareness of time [29, 30, 45]. For instance, when driving a car, you are constantly reacting to changes in the road by slightly adjusting the wheel [47]. Since your attention is fully focused on these ongoing adjustments, you are less aware of how much time has passed [48]. The consistent measurement of the difference between perceived and actual time following certain tasks provides a useful measure of deep attentional engagement without requiring individuals to refocus attention on themselves during the task. Consequently, by bridging the gap between a person’s inner experience and their measurable brain activity, temporal time distortion offers useful evidence that someone is experiencing flow. Although this observation presents a useful framework to understand flow, it also highlights a key challenge: directly assessing the experience can disrupt it [42].
Even without specific craft mastery or professional expertise, individuals can increase their likelihood of entering a state of flow through practices such as meditation and temporally structured learning [39, 42]. Rooted in ancient cultural traditions, meditation has long been used to quiet the mind and deepen

one’s connection to the surrounding environment [49]. In modern contexts, it has gained recognition for its mental health benefits, including reducing stress and anxiety while promoting present-moment awareness [50]. Beyond these benefits, research suggests that meditation may also support some of the neural pathways associated with flow states [51]. Engaging in focused breathing and sustained attention during meditation can temporarily alter brain activity and has been associated with changes in brain connectivity and plasticity [52, 53]. In particular, meditation appears to influence connectivity across the TPN, the DMN, and the SN, supporting sustained focus and efficient performance. These neuronal changes enable executive control regions, such as the frontal lobes, to redirect attention away from internal distractions toward what is happening in the present moment [37, 54, 55]. As a result, meditation functions not only as a tool for relaxation but also as a form of cognitive training that may help to create an environment in which the brain can more readily enter flow [50, 52, 56].
Structuring tasks to facilitate sustained focus can also help induce flow. When studying, breaking material into manageable chunks while gradually tackling more challenging concepts can help maintain engagement without becoming overwhelming [7, 11]. The Pomodoro method, which alternates 25 minutes of focused work with 5-minute breaks, is one example of how to set clear goals and support the kind of sustained, task-oriented concentration associated with flow [57]. Additionally, activities that provide rapid and continuous feedback, such as using quick succession flashcards, are particularly effective in supporting reward prediction and fostering an ideal environment for flow [58]. Taken together, these study methods draw on key components of deep engagement and significantly increase the chances of entering flow [7].
Like many other self-evaluating reports, Lewis Hamilton’s account exemplifies the continuously evolving theory that demonstrates the benefits that flow can have in performance and action. As we have seen, flow is a state of deep focus in which attention becomes fully absorbed in an activity, self-referential thoughts fade, and the passage of time may feel altered. Moreover, flow typically arises when skill and challenge level are matched, motivation is sustained, and attention is directed towards the task at hand [9, 11, 20]. Although current research suggests that flow
may involve shifts across large-scale brain networks and reduced self-referential processing, these findings are not consistent, and no single neuroscientific model can completely explain the phenomenon [32]. Instead, existing studies provide a useful framework for understanding how flow may arise [11, 12, 20]. Even without a complete neurological account, however, flow remains an extremely valuable concept both scientifically and practically. Through clear goals, balanced challenges, reduced distractions, and trained attention, flow cultivates deeper engagement. In doing so, flow provides insight into the regions and mechanisms of the brain that direct focus while also generating practical strategies for enhancing performance, strengthening motivation, and making activities more intrinsically rewarding across both professional and everyday contexts [39, 42, 50].
References on page 76.

by Daniel Bader | art by Ruimei Chau and Alexandra Adsit
After barely scraping through his Econ midterm, Joe decides to go out with his friends to distract himself by getting wildly drunk. At the frat house, he is immediately enveloped by the loud music, bright neon lights, and throngs of people dancing. His buddy Brock hands him a shot, quickly followed by a cold beer. A couple of drinks in, Joe is relaxed and has completely forgotten about how he mixed up the concepts of profit and revenue. He quickly loses count of his drinks and fades away, only to get up the next morning feeling sick and unable to remember any details from his epic night. Although Joe wakes up hungover, he successfully achieved what he wanted. As it did for Joe, alcohol intoxication affects the way we perceive the world around us [1, 2].
Alcohol is a psychoactive drug that alters perception, cognition, behavior, and consciousness [3]. Between the potential short-term consequences of intoxication, like interpersonal violence or car accidents, and the long-term effects of chronic consumption, like liver and cardiovascular disease, alcohol is responsible for over 3.3 million deaths annually worldwide [1]. Still, over 40% of the global population consumed alcohol in the last year, since drinking carries tremendous cultural and social value [4]. Alcohol is frequently consumed at parties or used as a social tool, as it facilitates socialization by ‘taking the edge off’ [5]. However, drinking in excess can cause immediate health emergencies and predispose individuals to addiction later in life [5, 6, 7]. Since alcohol is such a common, yet potentially dangerous, element of our society, it is important to understand how drinking alters your physical and mental state if you do choose to indulge [3].
Alcohol is a depressant, meaning that each drink Joe has reduces communication between neurons, cells in the nervous system that process and transmit information [1, 8]. Neural communication is the result of a series of electrical and chemical signals between neurons that relay information in the brain [1, 2, 9]. Signaling is moderated by chemical

messengers, called neurotransmitters, which are released by neurons into the space between two neurons, called the synapse [1, 10]. Once neurotransmitters are released into the synapse, they are either absorbed by the adjacent neuron, enabling neuron-to-neuron communication, or broken down and removed [1]. Alcohol significantly changes the concentrations of the two main neurotransmitters that modulate brain activity: GABA and glutamate [1, 4, 9]. GABA is the body’s primary inhibitory neurotransmitter, meaning that its release prevents neural signaling [1, 4]. Conversely, glutamate is an excitatory neurotransmitter that induces neural signaling. As Joe downs his shot, the alcohol enters the bloodstream and flows to the brain, causing an increase in GABA production and a decrease in glutamate release [1, 4]. The combination of increased GABA and decreased glutamate slows neuronal communication, preventing specific brain regions from properly performing their functions [1, 11]. Compare the rate of neuronal communication to the speed of a car: if GABA is the brake pedal and glutamate is the gas pedal, then drinking alcohol pushes the brake while releasing the gas, effectively slowing the car down [1, 4].
Reduced communication within and between brain regions negatively affects motor control and memory until alcohol is fully processed and removed from the body [11, 12]. As Joe goes to get his next drink, he
staggers towards his friends, stumbling over his own feet as he dramatically tries to stop himself from falling over. These changes in Joe’s movement and coordination are due to impaired neuronal signalling in the cerebellum, a brain region that is responsible for balance and motor coordination [13, 14]. Similarly, excessive drinking can decrease communication within the hippocampus, a different brain region that manages learning and memory [11]. When Joe got carried away drinking heavily with his friends and woke up the next morning with significant gaps in his memory, he experienced a decrease in hippocampal communication that may lead to the phenomenon colloquially known as ‘blacking out’ [11, 15]. Thankfully for Joe, GABA and glutamate levels return to baseline as alcohol leaves the body, and full motor and memory functionality will return as Joe sobers up [1, 16].
Beyond affecting motor and memory control, intoxication also impacts the brain’s reward system, a group of structures that reinforce behaviors using a neurotransmitter called dopamine [17, 19]. As the last of Joe's sixth beer trickles down his throat, the alcohol increases dopamine secretion into the synapse [1, 17, 18]. Alcohol reduces the efficacy of synaptic proteins that degrade neurotransmitters, leading to unregulated dopamine concentration in the synapse [1]. The prolonged presence of dopamine in the synapse amplifies the pleasure often felt when drinking [1, 17, 18. Any associations Joe makes between drinking and positive feelings can incentivize future drinking behavior [1, 20]. Positive sensations brought on by drinking may outweigh the negative experience that Joe has when he learns about all the embarrassing things he did while intoxicated last Saturday.
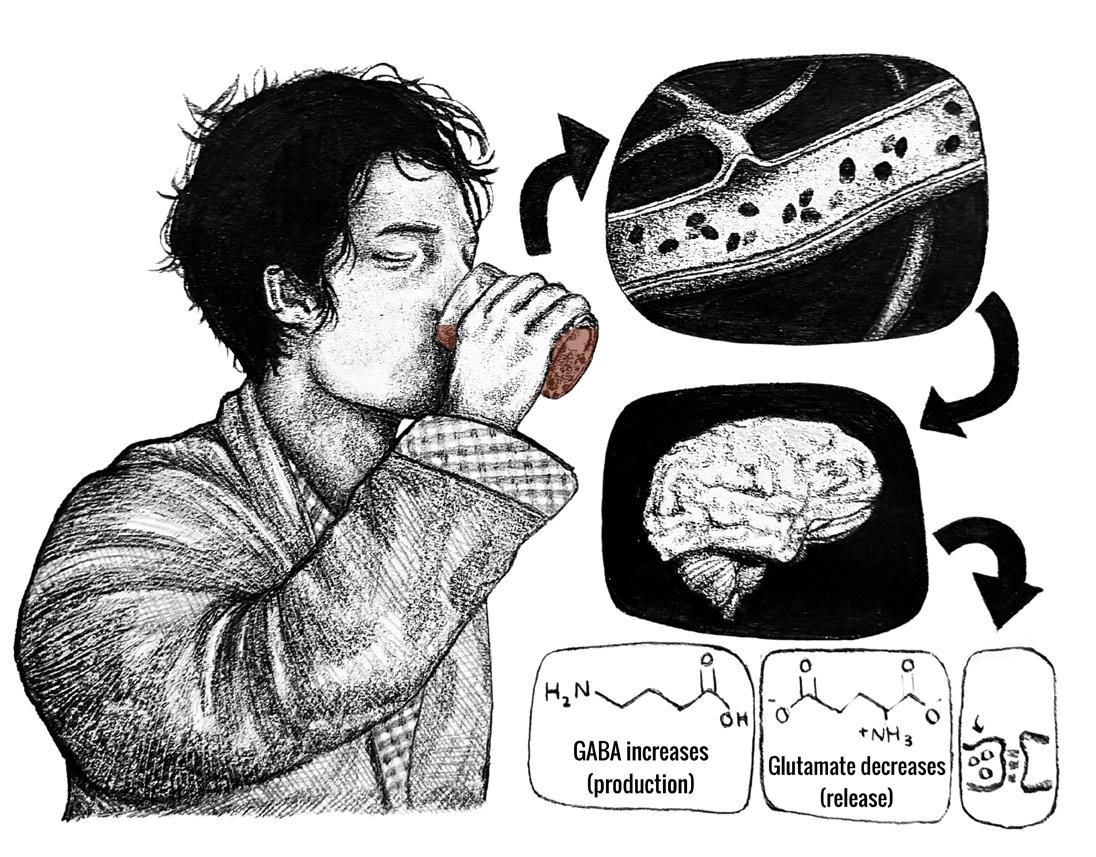
Hours after his last drink, Joe starts to sober up, but not until his body has cleared most of the alcohol from his system. Although variable, it generally takes the body about one hour to metabolize a standard drink, such as a single can of beer or glass of wine, so that all the alcohol in the bloodstream is degraded [21]. Alcohol clearance is the rate at which alcohol is removed from the bloodstream — a process directly associated with an individual’s alcohol tolerance, or how much a person can drink before becoming intoxicated [21, 22]. Proteins called alcohol dehydrogenases (ADHs) initially break down alcohol into acetaldehyde, a molecule that is toxic at high concentrations and often causes nausea, vomiting, and a rapid pulse [23, 24]. The pounding headache, fatigue, and vertigo ruining Joe’s Sunday morning are likely due to residual acetaldehyde driving the severity of his hangover [25]. Fortunately, hangovers do not last forever. Acetaldehyde is further metabolized into harmless acids by another group of molecules called aldehyde dehydrogenases (ALDH) [24, 26]. Once acetaldehyde is fully broken down and cleared from the bloodstream, most hangover symptoms subside [25].
The amount of time it takes for a person to metabolize alcohol depends on their Blood Alcohol Content (BAC), or the proportion of total body water that is composed of alcohol during periods of drinking [21]. Body water is composed of body fluids and liquids found in tissues, constituting around 50–60% of total body mass. The consumption of any kind of liquid — including your favorite juice, soda, coffee, tea, or 21+ drink — becomes part of the body’s total water content. Joe, who is smaller than his massive, bodybuilder friend Brock, knows that Brock can drink significantly more than he does on nights out without feeling as drunk. Heavier people tend to have more body water than lighter individuals, so long as most of their body weight consists of body fluids and water-dense tissues rather than fat. Imagine adding a single drop of red dye to a small glass of water; the liquid would immediately turn bright red. However, adding a single drop of dye to a large bucket of water would have a more subtle effect. Similarly, since muscle contains more water than body fat, leaner individuals will have a lower BAC after the same number of drinks because they have more relative body water than people of the same weight with a higher body fat percentage. The same logic explains
why men typically have higher alcohol tolerance than women, as the latter tend to have higher body fat percentages than men of the same weight [21]. But why can Joe’s smaller friend, Lola, who drinks three times per week, every week, outdrink him every Saturday night? Repeated exposure can also impact alcohol tolerance, as clearance rate increases with habitual drinking, and self-perceptions of intoxication also decrease with subsequent use [27].
While the short-term effects of intoxication fade away as individuals return to sobriety, alcohol use disorder (AUD) can have long-lasting negative impacts, including decreasing the body’s ability to fight off diseases [28]. AUD is characterized by loss of control over alcohol intake, compulsive alcohol use, and a negative emotional state when not drinking [29, 30]. Alcohol abuse can negatively affect the immune system's ability to fight off pathogens, which are microorganisms that cause disease within a host [28, 31]. The immune system is composed of a complex network of specialized cells, tissues, and organs that defend the body against infection [32]. T cells are specialized cells within the immune system that recognize and eliminate pathogens, recruit other immune cells, and generate long-term disease recognition [32, 33]. At first, T cells are naïve, or unspecialized, meaning they have not yet been activated to respond to a specific pathogen [33, 34]. Some activate to fight infection, and the rest develop to specialize in memory, which supports the immune system in responding more effectively to pathogens it has previously encountered [33, 34]. Unfortunately, AUD reduces the number of
naïve T cells, which may increase the risk of developing cancer and infection by limiting the body’s amount of available adaptive cells [34]. Chronic alcohol exposure also acts as a harmful toxin to DNA at the site of T cell maturation, significantly decreasing T cell production and leading to a deficiency [34]. Additionally, alcohol depletes natural killer cells (NKs), a part of the immune system that complements T cells by providing rapid surveillance against a wide range of threats, including virally infected cells and tumors [28, 35]. Therefore, decreases in NKs may explain why AUD is connected to increased susceptibility to cancer of the liver, breast, mouth, pancreas, and colon [28, 36]. Chronic alcohol consumption depletes key immune components, raising the likelihood of infection and disease [28, 34].
While AUD can weaken the immune system and increase vulnerability to pathogens, drinking itself causes further harm since consumption of alcohol directly triggers an immune reaction [37]. In the brain, alcohol activates immune proteins called toll-like receptors (TLRs) [37]. Activation of TLRs triggers the production of proinflammatory cytokines, another class of proteins that generate inflammation at sites of injury and infection [37, 38, 39]. Inflammation plays a crucial role in the immune response by recruiting immune system components to resolve damage, often manifesting as heat and swelling in the affected area [37, 38, 39]. However, prolonged inflammation in the brain, known as neuroinflammation, can cause inflammatory immune cells to target healthy neurons [37, 38]. Picture neuroinflammation as a fire alarm. Stimulated TLRs create commotion and signal for help [40]. Cytokines are like firefighters: they are necessary for putting out fires, but if constantly and unnecessarily involved, they can cause excessive water damage and even more destruction than the fire itself. Hence, while TLRs and the subsequent cytokine response are crucial for immune function, habitual drinking can cause uncontrolled immune-generated neuroinflammation, leading to sustained neuronal toxicity and eventual death [37, 41]. In addition to AUD, chronic TLR activation and subsequent neuroinflammation are hallmarks of many other diseases, including Alzheimer’s disease and Parkinson’s disease [37]. Alzheimer’s is characterized by cognitive and memory impairment, and Parkinson’s often manifests as tremors and walking abnormalities [37, 42]. While these diseases are very different from each other and AUD, they demonstrate how chronic TLR-mediated immune activation can lead to extremely serious neurological complications [37, 42].
Beyond long-term immune consequences, chronic alcohol abuse can lead to similarly persistent effects on brain structure [43]. One major consequence is atrophy, or shrinking, of brain tissue, a gradual structural change usually associated with aging [43]. Gray matter is the brain tissue that makes up the outer layer of the brain and spinal cord, processing signals between adjacent brain regions [44]. Typical brain aging involves the gradual loss of gray matter volume, which is associated with decreased cognitive efficacy [43, 45]. However, habitual drinkers often experience dramatic gray matter reductions at a rate much faster than natural brain aging [43]. The brain age paradigm, which uses images and scans of brain structures to predict the age of tested individuals, has found that alcohol abuse structurally ages the brain past its biological age by several years, in some cases by over a decade [46, 47]. In essence, habitual drinking acts like a fast-forward button for the brain’s biological clock [48]. Alcohol-induced gray matter reductions are commonly seen in the brain structure primarily responsible for impulse control, called the anterior cingulate cortex (ACC) [49, 50, 51]. These changes in the ACC may lead to impulsivity and failure to consider long-term consequences, such as driving home after drinking despite altered reflexes and judgment [52]. Additionally, alcohol abuse can cause gray matter volume in the hippocampus to shrink, leading to long-term learning and memory issues [53]. AUD specifically impairs recollection of episodic memory, which includes the conscious recollection of specific personal events from the past and the imagination of anticipated events in the future. These impairments occur because alcohol abuse decreases individuals’ ability to organize events chronologically and
remember the spatial context of memory, like the layout of the room or who was next to them when watching the finale of their favorite show [53].
Similar to AUD-induced gray matter reductions, drinking is associated with decreases in white matter, which can negatively affect one’s ability to think critically and solve complex problems [54, 55]. White matter is tissue primarily composed of axons, the components of neurons that span long distances in the brain and send signals to the rest of the nervous system [54, 56]. White matter axons can transfer information between hemispheres, a process necessary to ensure executive functioning, a set of cognitive skills that allows individuals to work through novel challenges and maintain focus, such as when solving a puzzle or building a Lego set [57, 58]. Natural aging typically involves a slight decrease in white matter volume, leading to certain cognitive deficits [54]. Sharper rates of deterioration are indicative of memory disorders like dementia, characterized by a decline in cognitive functioning that interferes with daily life without compromising consciousness or perception [54, 59]. While the volume of gray and white matter has been shown to partially recover with abstinence from alcohol, prolonged drinking can cause permanent structural changes that may have lasting detrimental effects on a person's physical and mental health [48].
Structural changes in the brain as a result of alcohol abuse can predispose individuals to a variety of neurological disorders [59, 60]. Excessive drinking is connected to cognitive decline, and alcohol abuse doubles the risk of dementia in elderly populations [59]. Cognitive decline in AUD populations may manifest as issues with sensory processing, memory, social cognition, walking, and balance. Although underlying structural causes are distinct, AUD-induced dementia can often mimic the symptomology of Alzheimer’s disease [59]. Individuals with AUD can also develop dementia as a by-product of other AUD-induced diseases [1, 60]. For example, Marchiafava-Bignami disease (MBD) is a neurological disease common in men over 45 with a history of alcohol abuse [1, 60]. MBD is caused by neuronal damage and cell death in the corpus callosum, a large white matter tract that connects the two hemispheres of the brain [1, 60, 61]. As the connection between
brain hemispheres is impeded, individuals may experience issues with muscle control, mood disorders, or dementia, and may even enter comatose states [1, 60]. Thus, AUD-induced dementia can also manifest through the development of other neurological diseases [1].
Additionally, AUD can cause nutritional deficiencies that may lead to further devastating neurological conditions [1]. People suffering from alcohol abuse often get a significant part of their caloric intake from nutritionally deficient alcoholic beverages, which may lead to shortages in essential dietary vitamins like thiamine (Vitamin B1) [1, 62]. Thiamine is necessary for nerve function, so a shortage of this crucial vitamin can result in neuron damage [62, 63]. AUD can also contribute to thiamine deficiency since habitual drinking increases stomach acidity, which harms mucous layers in the gut [48]. Pumps in the mucous layer that transport thiamine into the bloodstream require a specific pH level to function and are ineffective with increased acidity [64]. Eventually, untreated thiamine deficiency can develop into Wernicke encephalopathy (WE) — a neurological disease characterized by extreme confusion, visual disturbances, and poor muscular control [1]. WE is reversible, but if left untreated, it can lead to permanent memory and behavioral impairments [65]. Hence, chronic drinking acts as a catalyst for severe and often irreversible neurodegenerative conditions [1, 65].
While drinking has been and will remain an important cultural and societal practice, it is important to recognize the potential risk that alcohol poses toward brain health [6]. To practice safety when drinking, it is crucial to understand how inebriation immediately impacts the brain; by altering neurotransmission, intoxication innately leads to memory and cognitive impairments, physical changes, and impulsivity [11, 52]. While the altered mental and physical state brought on by drinking may be sought after in social scenarios, it can be extremely dangerous in the wrong setting, like behind the wheel of a car or in the deep end of a swimming pool. Furthermore, as drinking is popularly practiced across cultures, continents, and demographics, it is important to navigate potential risk factors for developing AUD, like drinking during adolescence [6, 66]. Additionally, alcohol abuse can both lead to depressive or anxious symptoms, while also possibly being misused as an emotional crutch for those already suffering from depression or anxiety [67]. Learning to tread the fine line is important; drinking can be fun and sometimes helpful in social settings, but the innately addictive nature of alcohol combined with the catastrophic neurological effects of AUD cannot be overstated [17, 59].
References on page 79.

by Samuel Jacobs | art by Zoe Stiefel

It's Friday night, and the Parents are eating dinner at the local Italian restaurant with their toddler, Elizabeth. The waiter is taking a long time to arrive, and Parent notices Elizabeth’s lip starts to quiver. To avoid the imminent and disruptive temper tantrum, Parent quickly gives Elizabeth the iPad. Elizabeth opens Netflix and soon forgets her prior frustration. When the server appears, Parent asks Elizabeth several times what she wants for dinner before Elizabeth looks up at the waiter, back down at her screen, and finally mumbles, ‘Mac and cheese.’ The Parents’ story is not unique: overreliance on screen-based entertainment for emotional regulation is becoming increasingly common [1]. In the U.S., children aged three to five generally spend over two hours per day in front of screens [2]. Like all children, Elizabeth is especially sensitive to feedback from her environment [3]. Her behavioral, social, and academic skills rely on prior experiences to shape future responses [4]. When digital media dominates a large portion of her early years, she will have fewer opportunities to play outside, use her imagination, and engage with friends [5]. These activities support mental and physical growth, and their absence can result in detrimental consequences later in life [5]. As Elizabeth enters middle school, her craving for screen time intensifies. Her
screen use sets her apart from her peers, leaving her feeling anxious and isolated [6]. Increased technology use in childhood and adolescence is highly associated with hyperactivity, impulsivity, and aggression, as well as worry, anxiety, and depression [7, 8]. While occasional access to screens at home and at school is not inherently harmful to early childhood development, screen time that is left unsupervised, particularly through age eight, can have lifelong impacts [9, 10].
Moments such as waiting for food, listening to a story, or recovering from disappointment are crucial experiences for a child’s development [10]. Every child’s brain is composed of neurons, the specialized cells that serve as the fundamental building blocks of the brain and nervous system [11]. These neurons communicate with one another by sending electrical and chemical signals, forming intricate neural systems that coordinate everything from basic functions to complex thoughts and behaviors [12]. Importantly, these systems are not fixed; they are constantly reshaped by experience [13]. This adaptability reflects neuroplasticity, the brain’s ability to reorganize and refine its structure by creating, adjusting, and reinforcing connections in response to both internal and external influences. Through this dynamic process, neural systems evolve, allowing children’s brains to learn, develop, and respond to their environments [13].
Each time Elizabeth engages with the world around her, she strengthens neural connections that support executive function [14]. Cognitive flexibility, working memory, and inhibitory control are all processes under the umbrella of executive function, enabling children to guide behaviors towards goals [15]. When Elizabeth shifts from coloring on the kids' menu to playing peek-a-boo with her parents, she practices cognitive flexibility [16]. When Parent tells Elizabeth to get ready for dinner after playing outside, she must follow multistep instructions: ‘Take off your shoes and coat, wash your hands, then come sit down.’ Here, she relies on her working memory, which
assists in the retention and manipulation of information to complete a task [17, 18]. When she wants to throw her fork on the floor but refrains, she exercises inhibitory control by disregarding inappropriate actions that may get her in trouble [19]. Children rapidly develop executive function skills on their own, and adults can support the strengthening of these capacities through co-regulation — a process that refers to the way parents and children regulate each other’s actions and emotions [14, 17, 20].
Zooming into Elizabeth’s mind, development continues where we cannot see: the brain’s tissue and its wiring [21]. One type of brain tissue is gray matter, which processes and sends information throughout the brain and body [22]. The other type of tissue in the brain is white matter, which consists of bundles of axons, the part of the neuron that transmits signals [23]. White matter becomes increasingly organized during early childhood as its pathways become increasingly myelinated, or insulated by a fatty sheath, which speeds up signal transmission [5, 23, 24]. When these pathways are well organized, neural signals travel more efficiently across the brain regions, supporting the development of language and executive function [25]. These pathways are highly sensitive to environmental input during early childhood [5]. When preschoolers spend more time on screens, their white matter tends to be more disorganized and less efficient at communicating information between brain regions [5].
After dinner, the impaired communication in Elizabeth’s brain results in less conversation with her parents on the drive home. Elizabeth is still completely engrossed in her iPad as she makes her way from the car to the couch. Instead of spending quality time with her parents, the screen occupies her entire focus. Precious moments that could have been filled with stories or games that otherwise engage Elizabeth’s mind are instead devoid of human interaction. Because of frequent screen usage, children are missing healthy social interactions and conversations with their parents and the people around them, which are critical to a child’s development of emotional regulation and language [26, 27].
Healthy development is influenced both by children's interactions with their caregivers and their engagement with their environments [28]. Outdoor exploration and play in early childhood are tied to improvements in attention, working memory, emotional
well-being, and social competence [28]. For instance, at the park, Elizabeth can chase ants, invent games, and play with others, guided by her curiosity to explore the world around her. By engaging in varied, low-stimulation environments, Elizabeth strengthens neural pathways that support executive function skills [29]. On the other hand, high-stimulation activities such as iPad games lead Elizabeth to tune out external stimuli, disobey instructions, and self-isolate [30]. Healthy development in early childhood is therefore supported not by constant stimulation but by cycles of interaction, challenge, failure, recovery, and support — all of which lay the foundation for behavior in later life [26, 31].
Screen exposure can lead to changes in the prefrontal cortex, a brain region responsible for executive functions such as planning, attention regulation, working memory, and inhibitory control [32]. The prefrontal cortex depends on white matter pathways that connect it with other brain regions involved in attention, emotional regulation, and decision-making [24]. Longer screen time correlates with reduced white and gray matter thickness in prefrontal regions


responsible for impulse control, sustaining focus, and steering behavior toward a goal [33]. Greater screen exposure is also linked to reduced prefrontal activity and poorer inhibitory control, suggesting that the neural systems supporting self-regulation are less efficiently activated as screen usage increases [34]. For example, watching more videos on the iPad may make Elizabeth more likely to get upset if her mac and cheese is not to her liking. Additionally, watching fast-paced videos, like short-form content on YouTube or TikTok, can increase inhibitory control errors immediately after viewing, meaning that high-stimulation media further impairs impulse control and attention [35]. Together, frequent screen use influences brain development by changing how often children practice executive function [36].
After school one day, Elizabeth is playing with her LEGOs when one of her spaceships breaks. The frustration builds quickly; her parents know that she’s about to melt down. Before the crying can really begin, the familiar solution appears. The iPad lights up, Elmo starts singing, and the problem seems to vanish as Elizabeth is entranced by the screen. But a crucial learning opportunity has been lost [28]. When a child calms themselves down after something goes wrong, they are practicing how to move through feelings of frustration, boredom, and disappointment alone [37]. This is also a moment where co-regulation is
beneficial, as parents can help their children work through challenging emotions and prepare them for similar experiences in the future [14]. So, in Elizabeth’s case, when screens are used as a shortcut to self-regulation, she is less able to minimize stress on her own, which can have widespread effects on her behavior and mood [38].
After Elizabeth’s almost-tantrum with the LEGOs, the sun sets, and it’s bedtime. Parent brings Elizabeth to her bedroom, hoping for an easy night, but Elizabeth refuses to sleep. She jumps out of bed and starts running around the room, screaming that she has to watch the newest Blippi episode. It has been a long day, and Parent is exhausted. It is hard for Parent to resist the simple, convenient solution of the iPad, and what could have been a screen-free bedtime routine becomes filled with digital media, altering the conditions under which Elizabeth’s brain prepares for sleep and recovery [39]. Beyond negative behavioral outcomes, screen time damages sleep quality [40]. Healthy sleep quality plays a central role in child development, supporting cognitive functioning, emotional well-being, and behavioral development [41]. The brain forms long-term memories during sleep by coordinating neuronal activity across brain regions to integrate information from the day, altering neural system connectivity accordingly [42]. Since children are still learning to self-regulate in early childhood, consistent healthy sleep is crucial for the development of sustained attention and managing frustration
[43]. When sleep is shorter or more fragmented, children show greater difficulty with emotional regulation and cognitive functioning [44]. They may also become more irritable and struggle to recover from everyday challenges [44]. Access to devices at bedtime is a major culprit for difficulty falling asleep, more nighttime awakenings, and poorer overall sleep quality [45]. Over time, as evening media use becomes routine, differences in sleep duration and quality may shape how a child’s neurons develop to support language, attention, and self-control [46, 47].
By the time children enter elementary school, the developmental systems shaped during early childhood begin to influence academic learning [5]. Elizabeth starts kindergarten and becomes one of many students struggling to pay attention during story time or follow instructions for class activities [5]. Academic performance depends heavily on attention regulation, language development, sleep quality, and emotional self-control [43]. When these systems develop less effectively, as is often the case with high screen usage, the impacts appear in weaker literacy skills and classroom engagement [5]. At the same time, not all screen use affects learning in the same way. Well-designed, age-appropriate educational programming and interactive media can support early language and literacy by introducing new vocabulary, reinforcing phonics and word recognition, and encouraging cognitive skills such as imaginative play (for more on this, check out this article on page 82!) [2]. When these tools involve responsive interaction — especially with caregiver participation — they can help young children retain new information and strengthen learning [2].
When Elizabeth starts middle school, her heavy screen exposure can starkly affect her behaviors in the most ordinary of places: family dinner, car rides, and bedtime [33, 48]. Elizabeth’s attempts to focus fail as her attention continually slips; her body seems to move before her brain has approved it [33, 48]. Her inability to concentrate can be indicative of further conditions that may require diagnosis and treatment [48]. Continued recreational screen use in childhood may contribute to her having more Attention-Deficit/ Hyperactivity Disorder (ADHD) related behaviors like trouble concentrating on tasks, excessive movement/ restlessness, impulsivity, and distractibility [48, 49, 50]. Attention challenges do not always look dramatic; sometimes, they look like constant almosts: almost done, almost listening, almost calm [48]. Because ADHD-like symptoms are closely tied to disruptions in attention-regulation networks, structural
differences in both the prefrontal cortex and its connecting pathways may help explain why inattention and impulsivity sometimes appear alongside increases in early media exposure [25, 33].
For children who show ADHD-like symptoms, doctors often recommend physical activity to help them regulate their energy [51]. In all children, physical activity supports healthier brain development and stronger cognitive functioning [25, 52]. In the brain, these benefits are reflected in greater white matter integrity, particularly in pathways linked to attention, suggesting that physical activity helps maintain the connections between brain regions [25]. Greater screen exposure often reduces time spent exercising, subsequently thinning the white matter in the brain [25]. When screens replace playing soccer, hiking with friends, or walking around the mall, the brain has fewer opportunities to strengthen attention-supporting neural circuits, and this relationship may explain why attention and behavior shift over time with excessive screen use [25, 50].
In high school, Elizabeth’s friends meet new people through sports, arts, and clubs, while she is stuck doomscrolling on Instagram. The lack of socialization leads to Elizabeth feeling less fulfilled in her life, a common consequence since greater daily screen exposure is associated with a higher prevalence of depressive symptoms [50]. Particularly among adolescents, extensive time on social media can have serious detrimental consequences on mental health [53, 54]. Unhealthy comparisons to others on social media can harm self-esteem, and a hyperfixation with public image can also result in burnout, emotional dysregulation, and social anxiety [54]. Elizabeth begins to feel a vortex of confining distress as her interactions with friends decrease [54]. Thus, over time, these everyday patterns of increased screen usage shape how adolescents like Elizabeth manage stress, self-regulate, and interact with others as they grow into adults [50, 55].
However, Elizabeth need not become a cautionary tale about technology as a whole. Screens themselves are not inherently harmful; their developmental impact depends largely on how they are used and what experiences they replace [56]. Pediatric guidelines encourage co-viewing, where caregivers watch digital media with children and actively discuss what appears on the screen [57]. When adults ask questions, explain unfamiliar words, or connect
the digital story to real-world experiences, children demonstrate stronger vocabulary and comprehension outcomes [57]. In these moments, the screen becomes a tool rather than a replacement for conversation [58]. Research also suggests that parental involvement during screen use may influence how children engage cognitive control systems in the brain [59]. When caregivers guide children through digital content, children demonstrate stronger activation in brain regions aligning with inhibitory control and regulatory processing [59]. Pediatric organizations also recommend limiting recreational screen use for preschool children to roughly one hour per day while prioritizing physical activity, sleep, and caregiver interaction [2]. When screens are used sparingly and with caregiver engagement, their influence can actually support healthy brain development in children [56]. But for many kids, the iPad continues to glow.
References on page 82.


by Charlotte Tobin | art by Querida Alleyne

Most of us take for granted that we will always recognize the people we love, and few things are as difficult as being forgotten by someone who knew you well. Unfortunately, for millions of Americans, Alzheimer’s disease makes this a painful reality of everyday life. Alzheimer’s is a progressive brain disorder that not only erases memory but erodes cognition and independence more broadly — and it is extremely common. One in 10 Americans over the age of 65 lives with the disease, and as the U.S. population ages, this number is expected to grow [1]. Despite its severity and prevalence, the disease remains poorly understood [2]. Because linking its observed biological features to cognitive decline is difficult, the root cause of Alzheimer’s has several competing explanations [2]. Consequently, while existing medications treat symptoms such as irritability, inattention,
and language impairment, no medication had been widely approved to address any single potential root cause of Alzheimer’s — until lecanemab (brand name Leqembi) in 2023 [3]. Approved in more than twenty countries, lecanemab represents hope for a new generation of therapies that slow the progression of Alzheimer’s rather than just manage its symptoms [4, 5]. At the same time, lecanemab’s approval has stirred debate. Uncertainty about whether its clinical benefits justify its costs and risks has raised deeper questions about how we research, evaluate, and approve treatments for complex and devastating diseases.
Alzheimer’s disease is a progressive neurodegenerative disorder, meaning it is characterized by the gradual death of neurons [6]. Neurons are specialized brain cells responsible for processing information and transmitting signals to other neurons [7]. More specifically, these cells send and receive information across small gaps between them called synapses [8]. It is the organization of these neurons and synapses into larger networks that enables core processes such as memory, learning, and behavior [9]. In the case of Alzheimer’s, synapses deteriorate and neurons die over time, progressively disrupting entire neural networks and driving the cognitive decline typically seen in individuals with the disease [10]. In early stages, symptoms manifest as minor behavioral changes, including mild confusion, difficulty concentrating, and wandering [4]. In later stages, cognitive impairment compromises the body’s most involuntary functions such as speech, movement, and swallowing [10]. Ultimately, people with Alzheimer’s can no longer control basic bodily functions and lose the ability to manage daily life on their own [4].
Despite the prevalence and severity of Alzheimer’s, research on the many hallmark features observed in the brains of affected individuals has yet to reveal a
‘smoking gun’ — the single biological mechanism responsible for the neuronal damage that characterizes the disease [11]. As a result, several competing hypotheses identify various proteins as the culprits of cognitive decline. One prominent explanation is the tau hypothesis, which links the irregular buildup of tau proteins — structural proteins found in neurons — to disruptions in neuronal function that can result in cell death [12]. In healthy neurons, tau stabilizes structures that support nutrient and signal transport within the cell [13]. In Alzheimer’s, tau forms tangled clumps that interfere with this transport system, damaging neurons and impairing brain function [14]. The tau hypothesis posits that these tangles are the main cause of cognitive decline [14]. Despite being a strong theory, no developed tau-targeting medication has demonstrated robust or consistent enough cognitive benefit in clinical trials to be approved [15, 16]. Another leading theory, the inflammation hypothesis, attributes neuronal damage and disease progression to the overactivation of microglia, a type of immune cell [15]. Under normal conditions, the primary function of microglia is to protect neurons by pruning inactive synapses and removing harmful debris [17]. The inflammation hypothesis maintains that the primary source of cognitive decline in Alzheimer’s is not simply the buildup of proteins like tau, but the subsequent and potentially destructive immune response these proteins elicit [18]. Within this framework, when microglia are overactive, they release inflammatory molecules that can damage surrounding neurons and exacerbate disease progression [15]. However, there is disagreement over whether this inflammation is a cause of Alzheimer’s or rather a reaction to existing damage [18]. This uncertainty, coupled with inconsistent results in clinical trials, has so far prevented any microglia-targeting therapy from reaching approval [11].
Finally, and most relevant to lecanemab, is the amyloid beta (Aß) hypothesis. During normal neuronal maintenance, the breakdown of large proteins produces Aß, a type of protein fragment [19]. While a moderate amount of Aß is healthy, an excess can accumulate into harmful clumps called plaques [20]. When these plaques form between neurons, they can contribute to the structural damage of synapses and interfere with neuron-to-neuron signaling [21]. As this damage worsens, synapses become inactive and are removed by the immune system [22]. This decrease in synapses and the resulting compromised communication are strongly linked to the cognition and memory deficits present in individuals with Alzheimer’s disease [23]. Since Aß plaques are considered a
leading cause of Alzheimer’s, they are a common focus for medication aimed at addressing the root cause of the disease [24]. Among currently prescribed therapies, lecanemab was the first FDA-approved treatment shown to target this underlying mechanism of Alzheimer’s [25, 26].
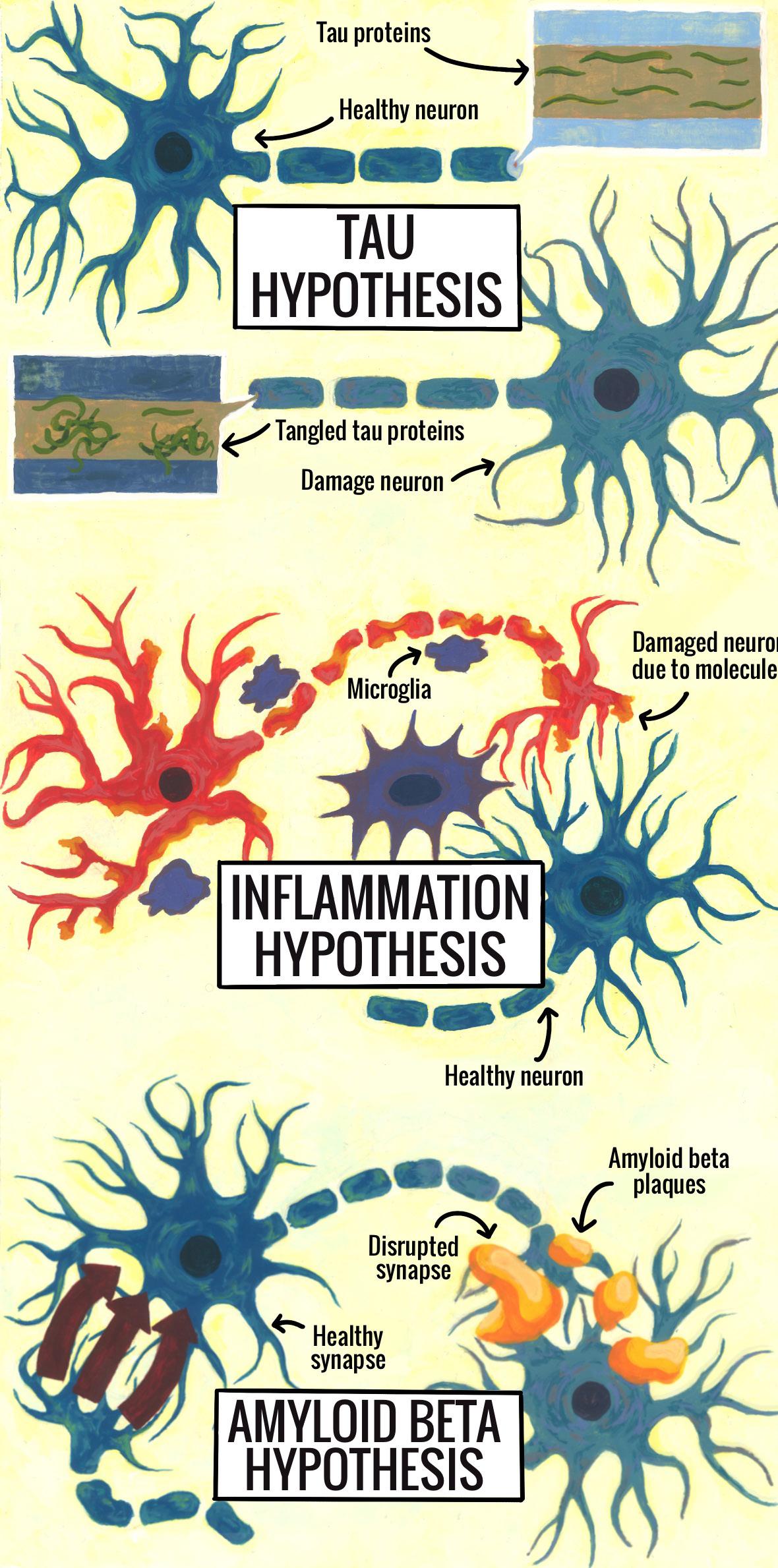
After decades of symptom-focused drug development, lecanemab belongs to a new generation of therapies designed to target a potential underlying cause of Alzheimer’s. More specifically, lecanemab primarily targets protofibrils –– smaller, earlier clumps of Aß that form as precursors to larger plaques [3]. Crucially, protofibrils are considered more toxic to neurons than plaques, and impair synaptic function early in the disease process [27]. Think of this process as mold growing in a house: By the time mold accumulates into large, visible patches, the fungus has already been silently causing irreversible structural damage to the home for much longer. Those large patches are similar to Aß plaques weakening neural networks in the brain [28]. The earlier, more inconspicuous damage is similar to the damage caused by protofibrils, which interfere with synaptic connections between neurons before any visible accumulation occurs [2,29]. Lecanemab is designed to target these early clusters and clear the larger plaques, intervening at both stages of Aß buildup and theoretically improving its effectiveness at slowing disease progression [3, 29].
This intervention works through lecanemab’s design as an antibody with the ability to target harmful amyloid structures in patients diagnosed with early-stage Alzheimer’s [30]. Antibodies are proteins that recognize and bind to specific targets in the body, effectively flagging them for removal [31]. In this case, lecanemab antibodies bind to Aß species of varying sizes and have a high affinity for the smaller toxic protofibrils [29]. By attaching to these structures, lecanemab recruits immune cells to clear the flagged amyloid from neural tissue [32]. Targeting Aß protein buildup in its earlier, more toxic form may help limit initial synapse damage, giving lecanemab the
potential to slow cognitive decline at its source [33]. Because Aß accumulation is one of the most characteristic features of Alzheimer’s, targeting the clumps has long been an area of interest for researchers [33]. To translate lecanemab from a theory to tangible benefit for patients, the drug was evaluated in a series of clinical trials [29]. The results told an interesting story.
Lecanemab was tested in a clinical study of several hundred people with early-stage Alzheimer’s during which each participant received either lecanemab or a placebo for a year and a half [29]. In order to be considered successful, lecanemab could not just perform better than the placebo; it had to work significantly better as measured by a sensitive composite cognitive scale [34]. At one year, though the treatment group experienced less cognitive decline than the placebo, the trial failed to meet the exact requirements for the sensitive measurement [34, 35]. The study continued for an additional six months, at which point the researchers performed a traditionally used though less precise analysis and found that lecanemab reduced cognitive decline by 27% compared to the placebo [26]. On top of this reduction, the treatment group showed less Aß buildup by the end of the study, meaning the drug worked as intended in the brain [26]. Because lecanemab slowed cognitive decline and reduced amyloid levels with relatively few adverse reactions, it was uniquely successful compared to past clinical trials of similar therapies at the time [26, 36]. The benefits of lecanemab led to its full Food and Drug Administration (FDA) approval in July 2023 [36].
To understand this approval, it is important to examine how the FDA evaluates new therapies. In the United States, all drug approval decisions are made by the FDA, often with input from independent advisory committees consisting of various experts in relevant fields alongside patient and consumer representatives [37]. The FDA follows the advisory committee’s recommendation the vast majority of the time [38]. One notable exception was aducanumab (Aduhelm), an earlier Aß-targeting therapy that was approved despite near-unanimous advisory committee opposition; it was discontinued shortly after amid widespread controversy, becoming one of the most contentious decisions in recent FDA history [38]. Evaluating Alzheimer’s medications is notoriously difficult, which is part of what made lecanemab’s
approval so notable and prior decisions so fraught. Because the underlying biological processes behind Alzheimer’s remain uncertain, it is hard to reach an agreement on whether any given biological improvement, like amyloid reduction, is sufficient to produce meaningful clinical benefit [26]. In situations of unmet need, where a disease is severe and treatment options are limited, regulators are willing to tolerate more uncertainty about a drug’s long-term benefits even if a therapy’s success is merely plausible and not proven [39]. These approvals reflect the challenge of defining a tolerable level of uncertainty and set a precedent for future medications. Researchers may disproportionately pursue Aβ-targeting treatments because doing so seems more likely to succeed given past approvals. As a result, there is concern that alternative approaches that could prove even more valuable will be ignored [40].
An additional complication to the scientific debate surrounding the Aβ hypothesis is that the levels of Aβ observed in the brain do not consistently align with the severity of cognitive symptoms [41]. While the accumulation of Aβ is considered to be a major contributor to cognitive decline, some individuals exhibit levels of buildup typically associated with latestage impairment while remaining at their cognitive baseline. This apparent ‘resistance’ to neurodegeneration suggests that plaques themselves may not cause the decline in symptoms but may rather be a tightly linked result of the true underlying mechanism of Alzheimer’s [41]. This complexity further calls into question whether removing Aβ alone is sufficient to meaningfully alter disease progression and also helps explain why proven reductions in Aβ accumulation do not consistently translate to substantial clinical improvement.
A specific complicating factor in evaluating lecanemab’s effectiveness is the widely cited ‘27%’ reduction in cognitive decline, which was obtained using a less precise though more conventional overall cognitive scale [26, 42]. At first glance, a 27% reduction seems like a substantial effect. However, this figure is misleading because it reflects a relative difference between the treatment and placebo group, rather than the actual magnitude of change experienced by patients [26]. The difference between the amount the treatment group declined relative to the other group was 27%, but relative to the overall cognitive scale
was just 2.5%. It’s like saying losing $7 in the second round of poker is a 30% improvement compared to losing $10 in the first round — technically true but not necessarily meaningful in practice. Furthermore, in order for there to continue to be a 27% difference, the gap in cognitive decline between the placebo and treatment groups must continue to grow over time. If both groups decline at similar rates after the trial period, the relative benefit of lecanemab compared to placebo will not hold, suggesting the reported effect may be an overestimate of long-term impact [36, 42].
The uncertainties surrounding lecanemab’s effectiveness become even more important when considering the realities of the treatment, as it is an intervention that requires continual monitoring, carries nontrivial risk, and may be massively burdensome for both caregivers and increasingly incapacitated individuals [36]. Lecanemab is administered twice-monthly and comes with a black box warning, the FDA’s strongest safety designation, signaling a risk of potentially life-threatening side effects [43]. One of the most notable side effects is an increased risk of amyloid-related imaging abnormality (ARIA), a finding on brain imaging that is associated with swelling or bleeding [26, 36]. Though ARIAs are often mild or asymptomatic, patients may experience headaches, confusion, vomiting, vision loss, difficulty walking, and even death [44]. The association of lecanemab with the potentially severe risks of ARIAs is the reason for the drug’s black box warning [45]. While ARIAs occurred in both treatment and placebo groups, they occurred at higher rates in the treatment group and represent a distinct risk that patients should weigh when considering lecanemab [26].
ARIA prevalence is not an isolated burden; it also necessitates frequent imaging, adding to the overall hardship of treatment [36]. Magnetic resonance imaging (MRI) frequencies vary from patient to patient depending on risk factors, notably increased genetic risk for serious adverse events [46]. Regardless of results from genetic pre-screening, at least four MRIs are required in the first six months of lecanemab injections. Patients may need additional MRIs if they develop ARIA, which was the case in a significant portion of the clinical trial patients [36]. The cost of this imaging is not included in the drug’s $26,500 annual cost [47]. For patients and caregivers already navigating a difficult diagnosis, this adds both
financial strain and the burden of frequent medical appointments [48]. And, without infrastructure to reduce these practical burdens, sustained treatment will remain out of reach for many, limiting how much lecanemab can meaningfully improve lives beyond a clinical setting [49].
The evidence surrounding lecanemab is difficult to evaluate. On the one hand, treatment reduces the amount of Aβ plaque buildup [26, 50]. On the other hand, the translation of that neurophysical reduction to a distinct cognitive benefit is modest [49]. Furthermore, the drug’s mechanism rests on the Aβ hypothesis, which remains scientifically contested [51]. Though complicated, the successes and shortcomings of lecanemab contribute to our understanding of how the FDA recalibrates evidence thresholds in the face of unmet need — a topic undoubtedly important to understand, with many implications for future therapies and other diseases [52]. The case also spurs questions about acceptable levels of treatment burden, a question that is perhaps not as commonly asked but is nonetheless an important consideration [52].
Importantly, the story of lecanemab speaks to the limitations of relying on a single mechanism to explain and treat a disease as complex as Alzheimer’s [36]. The body and brain are a complex system, reflecting constant interactions between many biological mechanisms. Prioritizing one explanation for a condition over another risks narrowing our understanding and curtailing meaningful progress [51]. Lecanemab should be seen as a component of a broader therapeutic strategy that addresses more than just a single mechanism. Single-target approaches are inherently limited, and lecanemab should not be considered the ‘final boss’ of Alzheimer's therapies [36]. Instead, a combination of therapies must be investigated alongside broader considerations of lifestyle and environment to fully and effectively address the onset and progression of the disease [36, 53]. Ultimately, Alzheimer’s may not demand a single answer, but rather a willingness to accept complexity both in how it is studied and how it is treated.
References on page 85.


by Tara Dacey | art by Alexandra Tapia
Most people have a favorite song and a favorite color, but for a person with synesthesia, these could be one and the same. Synesthesia is the involuntary perception of additional sensations that occurs when a stimulus, such as an image or sound, simultaneously activates two or more typically unrelated senses [1, 2]. Synesthetic experiences manifest as combinations of sight, hearing, touch, taste, or smell [3,4]. For a sensory experience to be considered synesthesia, it must clearly be the result of a stimulus, like automatically perceiving the color pink at the sound of a flute [1, 5]. Such instances are distinct from hallucinations, which are not triggered by external stimuli [6]. While synesthesia may seem unusual or almost psychedelic, it should not be mistaken for an indication of other mental conditions or a cause of distress. Rather, synesthesia is a benign and unique variation of sensory perception [6].
About 4% of the global population experiences synesthesia, meaning that at a concert with 10,000 people in attendance, roughly 400 people might see colors or visualize words in response to the music, lighting, or other stimuli present [4]. Manifestations vary widely among individuals with synesthesia; different peo ple experience unique sensations in response to identical stimuli [2, 7]. At the strum of a guitar, one person with synesthesia might taste a mango, whereas another might physically feel the smooth tex ture of the music gliding across their hands. However, synes thetic experiences remain relatively stable for each person: the same stimulus consistently elicits the same response throughout their life [4, 8]. For example, if a person with synesthesia hears the music note ‘A’ at age seven and perceives the color magenta, they will still visualize magenta upon hearing the same note at age seventy-five. The direction of synesthetic perception also tends to remain stable [4]. In most cases, perception is unidirectional: the inducer, the
triggering stimulus, leads to the concurrent, the additional perceptual event, and not the other way around.. For the person who visualizes magenta alongside the music note ‘A,’ sound induces the experience of color, but a magenta-colored stimulus would be unlikely to induce the sound of the music note ‘A’. The unidirectionality and lifetime consistency of synesthesia make it easier to categorize and understand [4].

While synesthesia is considered a single condition, each specific combination of inducer and concurrent is its own subtype, typically named based on the stimulus and the additional sensory experience elicited [1,2,4]. Although each subtype of synesthesia has unique characteristics and manifests differently in every person, subtypes can be grouped into clusters based on a shared concurrent experience [2,9]. For example, the cluster created for colored sequences includes the number-color and weekday-color synesthesia subtypes [2]. Clusters are helpful tools for understanding differences in synesthetic experiences, but there is currently no overarching theory explaining how the condition develops [2]. However, the existence of shared clusters among people supports the theory that some subtypes may stem from a common cause [1, 2]. Multiple subtypes of synesthesia are associated with global hyperconnectivity, which refers to abnormally heightened communication between brain regions [10, 11]. Networks between regions are formed through neuroplasticity, the ability of brain cells called neurons to create or strengthen connections with other neurons [4]. Brain-derived neurotrophic factor (BDNF), a protein involved in the development of neuron structure and function, plays a key role in facilitating neuroplasticity [4]. Concentrations of the protein are
generally higher in people with synesthesia than in those without, which could support the theory that global hyperconnectivity and increased neuroplasticity are associated with the development of synesthesia [1,4, 12].
People with synesthesia display heightened connectivity between regions of the brain responsible for sensory processing, including parietal, visual, and auditory regions [11, 13]. The parietal cortex is crucial for integrating information from multiple senses to create a cohesive perception of a stimulus [14]. The disinhibited feedback model provides one theory of how and why synesthesia manifests, explaining it through the involvement of the parietal cortex [13, 15]. In most people, information is sent from brain areas involved in initial sensory processing to the parietal cortex, not the other way around. By contrast, in people with synesthesia, the parietal cortex may transfer sensory information back to the areas involved in initial sensory processing, causing concurrent experiences [13, 15]. It is hypothesized that the feedback connections from the parietal cortex are disinhibited, which is similar to releasing the brakes of a car [13]. Pressing the brakes inhibits the car’s movement, causing it to come to a stop. Likewise, by inhibiting the parietal

cortex, feedback connections are seemingly blocked, and synesthetic experiences may lessen as a result [13]. Disinhibition occurs when you lift your foot off the brake, taking the car out of an inhibited state and allowing it to move forward. If the parietal cortex is disinhibited in synesthesia, the feedback connections might be abnormally powerful and thereby activate additional sensory modalities: visual, auditory, tactile, gustatory (taste), and olfactory (smell) [13, 16]. The activation of sensory areas consequently elicits a concurrent [13, 16].
In addition to the possible structural and functional mechanisms underlying synesthetic experiences, genetics may also play a role in the development of the condition [9, 17]. Close relatives of people with synesthesia are more likely to also have the condition [9, 17]. Members of the same family may even develop different subtypes of synesthesia, further indicating that the condition has a common origin regardless of which synesthetic experiences manifest [9]. However, no single gene is responsible for the occurrence of synesthesia; instead, multiple genes may act as predisposing factors [9,17]. Even if someone does not have synesthesia but has relatives with the condition, they may have greater connectivity and BDNF concentrations than a person who does not have relatives with synesthesia [18]. While BDNF levels differ between people with and without synesthesia, differences in concentration alone cannot explain why people with synesthesia have such vastly distinct experiences [1]. Relatives of people with synesthesia may also display increased creativity and improved episodic memory, traits frequently observed in people with synesthesia, without necessarily experiencing synesthesia themselves [18]. The presence of shared cognitive traits between those with synesthesia and their relatives without synesthesia supports the possibility of a heritable factor of synesthesia [2]. If a specific genetic component of synesthesia is identified, it could explain why certain people are prone to developing synesthesia [19].
In combination with genetics, atypical brain structures seen in early childhood development may lead to the emergence of synesthesia [1, 20]. Environmental influences could be crucial in determining whether someone who is predisposed to synesthesia through brain structure develops the condition, as well as how it manifests [2, 18]. People who
develop synesthesia might observe an object that involves multiple senses, such as a blue toy that plays music, and then internalize and enhance the association [2]. The mechanism behind this internalization is based on the adaptability of infants’ brains, which contain over 100 billion neurons. As learning occurs, the pathways the brain uses most often grow stronger, while the less useful ones fade away through a process known as ‘pruning’ [21]. In people with developmental synesthesia, a lack of pruning leads to excessive connections after early phase development, causing hyperconnectivity [1]. As a result, developmental synesthesia emerges in early childhood and continues through adulthood [1]. In contrast, post-accidental synesthesia is a subtype that typically arises due to permanent nerve damage caused by brain injury [20, 22]. For example, in people who have experienced damage to vision-processing regions, a form of visual synesthesia might occur as an attempt by the brain to overcompensate for the loss of sight and better interpret sensory stimuli [20]. Regardless of whether changes occur during development or after injury, structural abnormalities are implicated in the onset of synesthesia [1, 20].
There are seemingly endless ways for the brain to alter its processing of stimuli; more than 80 subtypes of synesthesia have been discovered thus far [4]. One of synesthesia’s most common subtypes is grapheme-color (GC) synesthesia, named after graphemes — letters or numbers — that trigger the perception of color [8, 23]. For the 1–2% of the population living with GC synesthesia, atypically strong associations between graphemes and colors may form during childhood [4, 23]. People with GC synesthesia exhibit increased brain activity in the color-processing region in response to grapheme stimuli, possibly leading them to perceive certain characters as colors [4]. The brain region that responds to graphemes, known as the visual word form area (VWFA), and the brain region responsible for color processing are adjacent [1, 11]. When a grapheme is processed in the VWFA of someone with synesthesia, the color-processing region may also be activated: this is an example of the cross-activation model [11]. Similarly, it has been hypothesized that enhanced multisensory integration — the brain’s process of unifying sensory information from multiple modalities to create a single perception — may also be rooted in the hyperconnectivity of the VWFA and color-processing area [11, 24, 25]. Those with GC synesthesia sometimes display enhanced recall of facts through long-term memory
and a more vivid recall of personal details, known as episodic memory [2, 23]. The basis of this enhanced memory may lie in the tendency to think using mental images, as well as in the hyperconnectivity seen in those with synesthesia [2].
Another subtype involving the VWFA is ticker-tape synesthesia (TTS), in which hearing speech results in seeing the words on a banner in the mind’s eye, separate from the external world [13, 26]. A person with TTS could hear someone say, ‘Let’s get brunch on Saturday!’ and automatically visualize the sentence as if it were torn from a magazine. TTS is the only known subtype in which the inducer and concurrent are both language-based; language-processing centers in the brain overactivate the VWFA, which transforms heard speech into internally visualized letters [13, 26, 27]. As with other synesthesia subtypes, individuals with TTS may have an enhanced working memory, a form of short-term memory that allows information to be stored and manipulated, similar to a mental workspace [27]. An enhanced working memory could mean a person with synesthesia exhibits advanced strategic thinking while planning moves during a game of chess or possesses an above-average ability to remember names [27].
While some subtypes of synesthesia, like TTS, have a single predominant theory, auditory-visual (AV) synesthesia has many proposed mechanisms [11]. AV synesthesia is a subtype characterized by an auditory stimulus that causes a visual, typically colorful, experience. The neurological processes that lead a person with AV synesthesia to, say, hear a cat’s meow and perceive the color turquoise may be similar to those underlying GC synesthesia. Specifically, cross-activation between parietal, auditory, and color-processing areas may play a role in AV synesthesia. However, the directionality of signal transmission between the regions activated by the inducer, concurrent areas, and the parietal cortex remains undetermined. Two models currently predominate: the two-stage model and the disinhibited feedback model. The two-stage model suggests that signal transmission travels from the inducer areas to concurrent areas and finally to the parietal cortex. Alternatively, the disinhibited feedback model posits that transmission stems from the inducer areas to the parietal cortex and then to concurrent areas. Recall how the rate of communication from the parietal cortex can be like the movement of a car when you lift your foot off the brakes. Understanding the abnormal pathway taken by stimuli to higher processing areas in AV synesthesia is vital in determining whether the two-step or disinhibited
feedback model is correct, though both theories suggest that this subtype results from abnormal parietal cortex involvement during sensory transmission [11].
Many explanations exist for how synesthesia develops in individuals, but some theories additionally suggest that the condition may be evolutionarily advantageous for the human species in general [18, 29]. Indeed, synesthesia may enhance individuals’ abilities to understand stimuli and unconsciously seek additional clues for identification through other senses [18, 29]. Individuals with synesthesia can exhibit cognitive abnormalities, including heightened memory, intellectual curiosity, and creativity [1, 2]. The ability to interpret and create requires cross-sensory mapping, a process closely related to synesthesia [30]. Cross-sensory mapping is the correspondence between sensory modalities — like matching higher pitches to increased brightness — which is distinct from combining information from multiple senses through multisensory integration [14, 31]. Making associations between senses requires abstract thinking, a process that also supports the understanding of figurative language, such as metaphors [30]. Metaphors begin with sensory perception, often combining multiple sensory modalities. For example, the meaning of the term ‘sour face’ is understood through the connection of the gustatory and visual modalities. Individuals with heightened creativity have a greater ability to establish metaphorical connections and to link seemingly unrelated concepts [30]. During creative processes, correspondence between the senses is increased for all people, but in people with synesthesia, hyperconnectivity and cross-activation between modalities connect the senses even further
[11, 30, 32]. People with synesthesia may also have more vivid mental imagery than those without, further aiding creativity and allowing them to derive more enjoyment from imaginative activities [30]. Picture a potter with synesthesia wishing to craft a tea set, with a tea cup resembling a pink lotus in full bloom and a saucer like a lily pad. The potter may be able to visualize the lotus cup and lily pad saucer in such vivid detail that the mental image serves as the perfect reference for crafting the set. While synesthesia does not guarantee greater creativity, it prompts unique sensory experiences that can fuel artistic and creative endeavors.

Just as no two pieces of original art are identical, no two individuals have the same brain. Unique individual patterns of brain development may be responsible for certain subtypes of synesthesia, similar to other neurological conditions [4]. People with autism spectrum disorder (ASD) display a higher prevalence of synesthesia than the general population, and people with synesthesia tend to exhibit more common characteristics of autism than the general population, supporting a potential link between the conditions [33]. ASD and synesthesia share many cognitive attributes, including hypersensitivity to stimuli, enhanced perceptual functioning, increased attention to detail, and superior memory [17, 34]. Evidence for the connection includes global hyperconnectivity, which is often present in both people with ASD and people with synesthesia [34, 35]. Additionally, for people with ASD, synesthesia has been suggested to contribute to the development of savant abilities, such as perfect pitch or extraordinary mathematical capacity, which are present in about 10% of people with ASD [36, 37]. There is a positive correlation between genes associated
with ASD and those associated with synesthesia, but because each condition is influenced by many genes, it is difficult to pinpoint which ones may influence either condition [17].
Synesthesia may also be related to autonomous sensory meridian responses (ASMR), pleasant tingling sensations that initiate at the scalp and are typically triggered by whispering and crisp sounds [32]. Inducers for ASMR responders are also common inducers for people with synesthesia [32]. The crisp sound of a snare drum may cause an ASMR responder to feel tingling sensations and a person with sound-taste synesthesia to taste black licorice [34]. Synesthesia tends to be more prevalent in ASMR responders than in the general population, and people with synesthesia are more likely to be ASMR responders [32]. The high frequency of overlap between people who respond to ASMR and those with synesthesia may be explained by similar mechanisms. Both conditions involve highly individualized experiences regarding inducers and concurrents, as well as multisensory experiences. Additionally, ASMR responses might result from the cross-activation of the primary cortex and regions related to touch, which parallels the cross-activation model of synesthesia. Both people with synesthesia and ASMR responders exhibit abnormal patterns of functional connectivity, supporting the theory of shared developmental origins. Furthermore, the argument has been made that ASMR could be classified as its own subtype of synesthesia [32]!
As a form of sensory perception, synesthesia could provide valuable insight into how the brain works. Determining whether the structure of specific brain regions predisposes a person to synesthesia is not about identifying the ‘magic’ key that makes color out of music, but understanding how slightly different brains lead to incredibly different states of awareness [18]. Perhaps what is special about being human is that, though we generally share similar internal machinery, there are countless minute differences that define our individual selves and experiences. For those with the condition, synesthetic perception impacts not just baseline sensory information, but may also continuously influence how one understands and connects to the world. The highly individualized expressions of synesthesia make large-scale studies challenging, but further research could expand the existing understandings of the connections between sensory modalities and neuroplasticity, in both synesthesia itself and other conditions [4]. Our brains are inherently flexible, and synesthesia is just one of many intriguing manifestations.
References on page 87.

FEATURED SSRIS AND STORKS: THE COMPLICATED RELATIONSHIP BETWEEN SSRIS AND PREGNANCY
1. Jee, S. B., & Sawal, A. (2024). Physiological changes in pregnant women due to hormonal changes. Cureus, 16(3), e55544. doi: 10.7759/cureus.55544
2. Álvarez-Fernández, I., Prieto, B., & Álvarez, F. V. (2016). Preeclampsia. Revista del Laboratorio Clínico, 9(2), 81-89. doi: 10.1016/j.labcli.2016.04.002
3. Stewart, D. E., & Vigod, S. N. (2019). Postpartum depression: pathophysiology, treatment, and emerging Therapeutics. Annual Review of Medicine, 70, 183196. doi: 10.1146/annurev-med-041217-011106
4. Sarno, E., Moeser, A. J., & Robison, A. J. (2021). Neuroimmunology of depression. Advances in Pharmacology 91, 259–292. doi: 10.1016/bs.apha.2021.03.004
5. Szuhany, K.L., & Simon, N. M. (2022). Anxiety disorders: A review. JAMA 328(24), 2431–2445. doi: 10.1001/ jama.2022.22744
6. Gumusoglu, S. B., Schickling, B. M., Vignato, J. A., Santillan, D. A., & Santillan, M. K. (2022). Selective serotonin reuptake inhibitors and preeclampsia: A quality assessment and meta-analysis. Pregnancy Hypertension, 30, 36-43. doi: 10.1016/j.preghy.2022.08.001
7. Fagiolini, A., Cuomo, A., & McIntyre, R. S. (2024). SSRIs. In Pocket guide to practical psychopharmacology: SSRIs and SNRIs in clinical practice 7–75. Springer Nature. doi: 10.1007/978-3-031-80490-8_2
8. Cowen, P. J. (2023). SSRIs in the treatment of depression: A pharmacological cul-de-sac? In M. Browning, P. J. Cowen, & T. Sharp (Eds.), Emerging neurobiology of antidepressant treatments, 66, 1–19. Springer Nature. doi: 10.1007/7854_2023_447
9. Wang, S.-M., Han, C., Bahk, W.-M., Lee, S.-J., Patkar, A. A., Masand, P. S., & Pae, C.-U. (2018). Addressing the side effects of contemporary antidepressant drugs: A comprehensive review. Chonnam Medical Journal, 54(2), 101–112. doi: 10.4068/cmj.2018.54.2.101
10. Camino, S., Strejilevich, S. A., Godoy, A., Smith, J., & Szmulewicz, A. (2023). Are all antidepressants the same? The consumer has a point. Psychological Medicine, 53(9), 4004–4011. doi: 10.1017/ S0033291722000678
11. Jing, E. & Straw-Wilson, K. (2016). Sexual dysfunction in selective serotonin reuptake inhibitors (SSRIs) and potential solutions: A narrative literature review. Mental Health Clinician, 6(4), 191–196. doi: 0.9740/mhc.2016.07.191
12. Alwan, S., Friedman, J.M. & Chambers, C. (2016). Safety of selective serotonin reuptake inhibitors in pregnancy: A review of current evidence. CNS Drugs 30, 499–515 doi: 10.1007/s40263-016-0338-3
13. Bérard, A., Sheehy, O., Zhao, J. P., Vinet, É., Bernatsky, S., & Abrahamowicz, M. (2017). SSRI and SNRI use during pregnancy and the risk of persistent pulmonary hypertension of the newborn. British Journal of Clinical Pharmacology, 83(5), 1126-1133. doi: 10.1111/ bcp.13194
14. Dubovicky, M., Belovicova, K., Csatlosova, K. & Bogi, E. (2017). Risks of using SSRI / SNRI antidepressants during pregnancy and lactation. Interdisciplinary Toxicology, 10(1), 30-34. doi: 10.1515/intox-2017-0004
15. Jahan, N., Went, T. R., Sultan, W., Sapkota, A., Khurshid, H., Qureshi, I. A., & Alfonso, M. (2021). Untreated depression during pregnancy and its effect on pregnancy outcomes: A systematic review. Cureus, 13(8), e17251. doi: 10.7759/cureus.17251
16. Watson, R., Harvey, K., McCabe, C., & Reynolds, S. (2019). Understanding anhedonia: A qualitative study exploring loss of interest and pleasure in adolescent depression. European Child & Adolescent Psychiatry, 29(4), 489–499. doi: 10.1007/s00787-019-01364-y
17. Serretti, A. (2023). Anhedonia and depressive disorders. Clinical Psychopharmacology and Neuroscience, 21(3), 401. doi: 10.9758/cpn.23.1086
18. Tolentino, J. C., & Schmidt, S. L. (2018). DSM-5 criteria and depression severity: Implications for clinical practice. Frontiers in Psychiatry, 9, 450. doi: 10.3389/ fpsyt.2018.00450
19. Adwas, A. A., Jbireal, J. M., & Azab, A. E. (2019). Anxiety: Insights into signs, symptoms, etiology, pathophysiology, and treatment. East African Scholars Journal of Medical Sciences, 2(10), 580–591. doi: 10.36349/EASMS.2019.v02i10.006
20. Remes, O., Brayne, C., & Lafortune, L. (2016). A systematic review of reviews on the prevalence of anxiety disorders in adult populations. Brain and Behavior, 6(7), e00497. doi: 10.1002/brb3.497
21. Stringaris, A. (2017). Editorial: What is depression? Journal of Child Psychology and Psychiatry, 58(12), 1287-1289. doi: 10.1111/jcpp.12844
22. Südhof, T. C. (2018). Towards an understanding of synapse formation. Neuron, 100(6), 276–293. doi: 10.1016/j.neuron.2018.08.042
23. Hampel, L., Lau, T. (2022). Neurobiological principles: Neurotransmitters. In: Riederer, P., Laux, G., Nagatsu, T., Le, W., Riederer, C. (Eds.), NeuroPsychopharmacotherapy. Springer Nature. doi: 10.1007/978-3-03062059-2_365
24. Özçete, Ö. D., Banerjee, A., & Kaeser, P. S. (2024). Mechanisms of neuromodulatory volume transmission. Molecular Psychiatry, 29(11), 3680-3693. doi: 10.1038/s41380-024-02608-3
25. Zeppelin, T., Ladefoged, L. K., Sinning, S., Periole, X., & Schiøtt, B. (2018). A direct interaction of cholesterol with the dopamine transporter prevents its outto-inward transition. PLOS Computational Biology, 14(1), e1005907. doi: 10.1371/journal.pcbi.1005907
26. Jones, L. A., Sun, E. W., Martin, A. M., & Keating, D. J. (2020). The ever-changing roles of serotonin. The International Journal of Biochemistry & Cell Biology, 125, 105776. doi: 10.1016/j.biocel.2020.105776
27. Pourhamzeh, M., Moravej, F. G., Arabi, M., Shahriari, E., Mehrabi, S., Ward, R., Ahadi, R., & Joghataei, M. T. (2022). The roles of serotonin in neuropsychiatric disorders. Cellular and Molecular Neurobiology, 42(6), 1671–1692. doi: 10.1007/s10571-021-01064-9
28. Kayabaşı, Y., Güneş, B., Erbaş, O. (2021). Serotonin Receptors and Depression. Journal of Experimental and Basic Medical Sciences, 2(2), 240-246. doi: 10.5606/jebms.2021.75662
29. Mikkelsen, N., Damkier, P., & Pedersen, S. A. (2023). Serotonin syndrome—A focused review. Basic & Clinical Pharmacology & Toxicology, 133(2), 124-129. doi: 10.1111/bcpt.13912
30. Carhart-Harris, R., & Nutt, D. (2017). Serotonin and brain function: A tale of two receptors. Journal of Psychopharmacology 31(9), 1091. doi: 10.1177/0269881117725915
31. Ye, X., Ghosh, S., Shin, B. C., Ganguly, A., Maggiotto, L., Jacobs, J. P., & Devaskar, S. U. (2024). Brain serotonin and serotonin transporter expression in male and female postnatal rat offspring in response to perturbed early life dietary exposures. Frontiers in Neuroscience, 18, 1363094. doi: 10.3389/ fnins.2024.1363094
32. Murthy, M. K. (2025). Molecular pathways linking the serotonin transporters (SERT) to depressive disorder: From mechanisms to treatments. Neuroscience, 584, 2-31. doi: 10.1016/j.neuroscience.2025.08.009
33. Sharp, T., Collins, H. (2023). Mechanisms of SSRI therapy and discontinuation. In: Browning, M., Cowen, P.J., Sharp, T. (Eds.) Emerging Neurobiology of Antidepressant Treatments. Current Topics in Behavioral Neurosciences, 66. Springer Nature. doi: 10.1007/7854_2023_452
34. Xue, W., Wang, P., Li, B., Li, Y., Xu, X., Yang, F., Yao, X., Chen, Y. Z., Xu, F., & Zhu, F. (2016). Identification of the inhibitory mechanism of FDA approved selective serotonin reuptake inhibitors: An insight from molecular dynamics simulation study. Physical Chemistry Chemical Physics, 18(5), 3260–3271. doi: 10.1039/ C5CP05771J
35. Aengenheister, L., Keevend, K., Muoth, C., Schönenberger, R., Diener, L., Wick, P., & Buerki-Thurnherr, T. (2018). An advanced human in vitro co-culture model for translocation studies across the placental barrier. Scientific Reports, 8(1), 5388. doi: 10.1038/ s41598-018-23410-6
36. Bérard, A., Levin, M., Sadler, T., & Healy, D. (2019). Selective Serotonin Reuptake Inhibitor Use During Pregnancy and Major Malformations: The Importance of Serotonin for Embryonic Development and the Effect of Serotonin Inhibition on the Occurrence of Malformations. Bioelectricity, 1(1), 18-29. doi: 10.1089_bioe.2018.0003
37. Roberts, R. M., Green, J. A., & Schulz, L. C. (2016). The evolution of the placenta. Reproduction, 152(5), R179-R189. doi: 10.1530/REP-16-0325
38. Lv, J., & Liu, F. (2017). The role of serotonin beyond the central nervous system during embryogenesis. Frontiers in Cellular Neuroscience, 11, 259363. doi: 10.3389/fncel.2017.00074
39. Stárka, L., & Dušková, M. (2020). What Is a Hormone? Physiological Research, 69(Suppl 2), S183. doi: 10.33549/physiolres.934509
40. Masdrakis, V. G., Markianos, M., & Baldwin, D. S. (2023). Apathy associated with antidepressant drugs: a systematic review. Acta Neuropsychiatrica, 35(4), 189–204. doi: 10.1017/neu.2023.6
41. Palmer, E. G., Sornalingam, S., Page, L., & Cooper, M. (2023). Withdrawing from SSRI antidepressants: Advice for primary care. The British Journal of General Practice, 73(728), 138. doi: 10.3399/bjgp23X732273
42. Horowitz, M. A., & Taylor, D. (2019). Tapering of SSRI treatment to mitigate withdrawal symptoms. The Lancet Psychiatry, 6(6), 538–546. doi: 10.1016/S22150366(19)30032-X
43. Mahmood, R., Wallace, V., Wiles, N., Kessler, D., Button, K. S., & Fairchild, G. (2024). The lived experience of withdrawal from selective serotonin reuptake inhibitor (SSRI) antidepressants: A qualitative interview study. Health Expectations, 27(1), e13966. doi: 10.1111/hex.13966
44. Davies, J., & Read, J. (2019). A systematic review into the incidence, severity and duration of antidepressant withdrawal effects: Are guidelines evidence-based? Addictive Behaviors, 97, 111-121. doi: 10.1016/j.addbeh.2018.08.027
45. Servin-Barthet, C., Martínez-García, M., Pretus, C., Paternina-Die, M., Soler, A., Khymenets, O., Pozo, Ó. J., Leuner, B., Vilarroya, O., & Carmona, S. (2023). The transition to motherhood: Linking hormones, brain and behaviour. Nature Reviews Neuroscience, 24(10), 605-619. doi: 10.1038/s41583-023-00733-6
46. Jee, S. B., & Sawal, A. (2024). Physiological changes in pregnant women due to hormonal changes. Cureus, 16(3), e55544. doi: 10.7759/cureus.55544
47. Liu, N., Yang, H., Han, L., & Ma, M. (2022). Oxytocin in women’s health and disease. Frontiers in Endocrinology, 13, 786271. doi: 10.3389/fendo.2022.786271
48. Berkane, N., Liere, P., Oudinet, J. P., Hertig, A., Lefèvre, G., Pluchino, N., Schumacher, M., & Chabbert-Buffet, N. (2017). From pregnancy to preeclampsia: A key role for estrogens. Endocrine Reviews, 38(2), 123144. doi: 10.1210/er.2016-1065
49. Kim, S., & Strathearn, L. (2016). Oxytocin and maternal brain plasticity. New Directions for Child and Adolescent Development, 2016(153), 59-72. doi: 10.1002/cad.20170
50. Trifu, S., Vladuți, A., & Popescu, A. (2019). Neuroendocrine aspects of pregnancy and postpartum depression. Acta Endocrinologica (Bucharest), 15(3), 410–415. doi: 10.4183/aeb.2019.410
51. Albert, K. M., & Newhouse, P. A. (2019). Estrogen, stress, and depression: Cognitive and biological interactions. Annual Review of Clinical Psychology, 15, 399-423. doi: 10.1146/annurev-clinpsy-050718-095557
52. Liu, X., Molenaar, N., Agerbo, E., Momen, N. C., Rommel, A.-S., Lupattelli, A., Bergink, V., & Munk-Olsen, T. (2022). Antidepressant discontinuation before or during pregnancy and risk of psychiatric emergency in Denmark: A population-based propensity score–matched cohort study. PLOS Medicine, 19(1), e1003895. doi: 10.1371/journal.pmed.1003895
53. Henssler, J., Schmidt, Y., Schmidt, U., Schwarzer, G., Bschor, T., & Baethge, C. (2024). Incidence of antidepressant discontinuation symptoms: A systematic review and meta-analysis. The Lancet Psychiatry, 11(7), 526-535. doi: 10.1016/S2215-0366(24)00133-0
54. Wu, X., & Jin, R. (2025). Effects of postpartum hormonal changes on the immune system and their role in recovery. Acta Biochimica Polonica, 72, 14241. doi: 10.3389/abp.2025.14241
55. Suryawanshi, O. I. V., & Pajai, S. (2022). A comprehensive review on postpartum depression. Cureus, 14(12), e32745. doi: 10.7759/cureus.32745
56. Emerson, M. R., Mathews, T. L., Struwe, L. (2018). Postpartum Depression Screening for New Mothers at Well Child Visits. The American Journal of Maternal/Child Nursing, 43(3), 139-145, doi: 10.1097/ NMC.0000000000000426
57. Stewart, D. E., & Vigod, S. N. (2019). Postpartum depression: Pathophysiology, treatment, and emerging therapeutics. Annual Review of Medicine, 70, 183196. doi: 10.1146/annurev-med-041217-011106
58. Liu C, Ystrom E, McAdams TA. (2023). Long-term maternal and child outcomes following postnatal SSRI treatment. JAMA Network Open, 6(8), e2331270. doi: 10.1001/jamanetworkopen.2023.31270
59. Kattah, A. (2020). Preeclampsia and kidney disease: Deciphering cause and effect. Current Hypertension Reports, 22(11), 91. doi: 10.1007/s11906-020-01099-1
60. Dines, V., Šuvakov, S., Kattah, A., Vermunt, J., Narang, K., Jayachandran, M., Garovic, V. D., & others. (2023). Preeclampsia and the kidney: Pathophysiology and clinical implications. Comprehensive Physiology, 13(1), 4231–4267. doi: 10.1002/j.2040-4603.2023. tb00249.x
61. Vigil-De Gracia, P., Vargas, C., Sánchez, J., Collantes-Cubas, J. (2023). Preeclampsia: Narrative review for clinical use. Heliyon, 9(3), e14187. doi: 10.1016/j. heliyon.2023.e14187
62. Bokslag, A., Van Weissenbruch, M., Mol, B. W., & De Groot, C. J. (2016). Preeclampsia: Short and longterm consequences for mother and neonate. Early Human Development, 102, 47-50. doi: 10.1016/j.earlhumdev.2016.09.007
63. Zhu, W., Wang, Q., & Chen, M. (2025). Identifying genes and traits associated with pre-eclampsia using summary statistics. PLOS One, 20(5), e0323683. doi: 10.1371/journal.pone.0323683
64. Ahmed, A., Rezai, H., & Broadway-Stringer, S. (2017). Evidence-based revised view of the pathophysiology of preeclampsia. In M. S. Islam (Ed.), Hypertension: From basic research to clinical practice 956, 355–374. Springer International Publishing. doi: 10.1007/5584_2016_168
65. Fishel Bartal, M., & Sibai, B. M. (2020). Eclampsia in the 21st century. American Journal of Obstetrics and Gynecology, 226(2 Supp.), S1239–S1247. doi: 10.1016/j. ajog.2020.09.037
66. Nass, R. D., Zur, B., Elger, C. E., Holdenrieder, S., & Surges, R. (2019). Acute metabolic effects of tonic-clonic seizures. Epilepsia Open, 4(4), 599-608. doi: 10.1002/epi4.12364
67. Zhao, C., Chen, F., Li, Q., Peng, L., & Yue, C. (2025). Bidirectional Mendelian randomization analysis of mood disorders and risk of preeclampsia-eclampsia. International Journal of Women’s Health, 17, 3557–3565. doi: 10.2147/IJWH.S539218
68. Vignato, J. A., Gumusoglu, S. B., Davis, H. A., Scroggins, S. M., Hamilton, W. S., Brandt, D. S., Pierce, G. L., Knosp, B. A., Santillan, D. A., & Santillan, M. K. (2023). Selective serotonin reuptake inhibitor use in pregnancy and protective mechanisms in preeclampsia. Reproductive Sciences, 30(2), 701–712. doi: 10.1007/ s43032-022-01065-z
69. Evans, S. H., Cameron, M. W., & Burton, J.. (2017). Hypertonia. Current Problems in Pediatric and Adolescent Health Care, 47(7), 161-166. doi: 10.1016/j. cppeds.2017.06.005
70. Viguera, A. C., McElheny, S. A., Caplin, P. S., Kobylski, L. A., Rossa, E. T., Young, A. V., Gaccione, P., Góez-Mogollón, L., Freeman, M. P., & Cohen, L. S. (2023). Risk of poor neonatal adaptation syndrome among infants exposed to second-generation atypical antipsychotics compared to antidepressants: Results from the National Pregnancy Registry for Psychiatric Medications. Journal of Clinical Psychiatry, 84(1), 22m14492. doi: 10.4088/JCP.22m14492
71. Cantin, C., Snelgrove-Clarke, E., Gaudet, L., & RossWhite, A. (2024). Characteristics of breastfeeding newborns in the first month of life with in utero selective serotonin reuptake inhibitor medication exposure: A scoping review protocol. JBI Evidence Synthesis, 22(1), 144–152. doi: 10.11124/JBIES-23-00048
72. Convertino, I., Sansone, A. C., Marino, A., Galiulo, M. T., Mantarro, S., Antonioli, L., Fornai, M., Blandizzi, C., & Tuccori, M. (2016). Neonatal adaptation issues after maternal exposure to prescription drugs: Withdrawal syndromes and residual pharmacological effects. Drug Safety, 39(10), 903–924. doi: 10.1007/s40264016-0435-8
73. Öngür D. (2025). Psychiatry and the Make America Healthy Again Commission. JAMA Network, 333(24), 2145–2146. doi: 10.1001/jama.2025.6255
74. Gostin, L. O., Finch, A., & Lurie, P. (2025). Making America healthy again: Remedies for Robert F. Kennedy Jr.'s campaign against chronic disease. Hastings Center Report, 55(4), 2-8. doi: 10.1002/hast.5020
TWISTING THE KNIFE: THE OPEN CASE OF MENSTRUATION, THE BRAIN, AND THE MISSED DISEASE OF ENDOMETRIOSIS
1. Rohatgi, A., & Dash, S. (2023). Period poverty and mental health of menstruators during COVID-19 pandemic: Lessons and implications for the future. Frontiers in Global Women’s Health, 4. doi: 10.3389/ fgwh.2023.1128169
2. Watters, M., Martínez-Aguilar, R., & Maybin, J. A. (2022). The menstrual endometrium: From physiology to future treatments. Frontiers in Reproductive Health, 3. doi: 10.3389/frph.2021.794352
3. Critchley, H. O., Maybin, J. A., Armstrong, G. M., & Williams, A. R. (2020). Physiology of the endometrium and regulation of menstruation. Physiological Reviews, 100(3), 1149–1179. doi: 10.1152/physrev.00031.2019
4. Janes, L. (2020). Hypothalamic-pituitary-gonadal (HPG) axis. Encyclopedia of Personality and Individual Differences, 2112–2114. doi: 10.1007/978-3-31924612-3_766
5. Zheng, C.-Y., Yu, Y.-X., Cao, S.-Y., & Bai, X. (2024). Epigenetics of inflammation in hypothalamus pituitary gonadal and neuroendocrine disorders. Seminars in Cell & Developmental Biology, 154, 340–345. doi: 10.1016/j.semcdb.2023.04.001
6. Xie, Y., & Dorsky, R. I. (2017). Development of the hypothalamus: Conservation, modification and Innovation. Development, 144(9), 1588–1599. doi: 10.1242/ dev.139055
7. Herbison, A. E. (2020). A simple model of estrous cycle negative and positive feedback regulation of GnRH secretion. Frontiers in Neuroendocrinology, 57, 100837. doi: 10.1016/j.yfrne.2020.100837
8. Lambalk, C. B. (2023). The enigma of the gonadotropin-releasing hormone pulse frequency governing individual secretion of luteinizing hormone and follicle-stimulating hormone. F&S Reports, 4(2), 27–32. doi: 10.1016/j.xfre.2023.02.010
9. Oktem, O., Akin, N., Bildik, G., Yakin, K., Alper, E., Balaban, B., & Urman, B. (2017). FSH stimulation promotes progesterone synthesis and output from human granulosa cells without luteinization. Human Reproduction, 32(3), 643–652. doi: 10.1093/humrep/ dex010
10. Allen, J. J., Herrick, S. L., & Fortune, J. E. (2016). Regulation of steroidogenesis in fetal bovine ovaries: Differential effects of LH and FSH. Journal of Molecular Endocrinology, 57(4), 275–286. doi: 10.1530/ jme-16-0152
11. Dias Da Silva, I., Wuidar, V., Zielonka, M., & Pequeux, C. (2024). Unraveling the dynamics of estrogen and progesterone signaling in the endometrium: An overview. Cells, 13(15), 1236. doi: 10.3390/cells13151236
12. Kauffman, A. S. (2022). Neuroendocrine mechanisms underlying estrogen positive feedback and the LH surge. Frontiers in Neuroscience, 16. doi: 10.3389/ fnins.2022.953252
13. Schmalenberger, K. M., Tauseef, H. A., Barone, J. C., Owens, S. A., Lieberman, L., Jarczok, M. N., Girdler, S. S., Kiesner, J., Ditzen, B., & Eisenlohr-Moul, T. A. (2021). How to study the menstrual cycle: Practical tools and recommendations. Psychoneuroendocrinology, 123, 104895. doi: 10.1016/j.psyneuen.2020.104895
14. Draper, C. F., Duisters, K., Weger, B., Chakrabarti, A., Harms, A. C., Brennan, L., Hankemeier, T., Goulet, L., Konz, T., Martin, F. P., Moco, S., & van der Greef, J. (2018). Menstrual cycle rhythmicity: Metabolic Patterns in healthy women. Scientific Reports, 8(1). doi: 10.1038/s41598-018-32647-0
15. Avila-Varela, D. S., Hidalgo-Lopez, E., Dagnino, P. C., Acero-Pousa, I., del Agua, E., Deco, G., Pletzer, B., & Escrichs, A. (2024). Whole-brain dynamics across the menstrual cycle: The role of hormonal fluctuations and age in healthy women. Npj Women’s Health, 2(1). doi: 10.1038/s44294-024-00012-4
16. Zhang, W. Y., McCracken, M., Dominguez, L. V., Zhang, A., Johal, J., & Aghajanova, L. (2024). The impact of estradiol supplementation on endometrial thickness and intrauterine insemination outcomes. Reproductive Biology, 24(2), 100886. doi: 10.1016/j.repbio.2024.100886
17. Makhanova, A., Maner, J. K., Joslin, M. D. M., Phimmasene, K. E. N., Jasienska, G., & Marcinkowska, U. M. (2025). Women’s affiliation across the menstrual cycle: Cycle phase variation and associations with progesterone and estradiol. Hormones and Behavior, 170, 105709. doi: 10.1016/j.yhbeh.2025.105709
18. Practice Committees of the American Society for Reproductive Medicine and the Society for Reproductive Endocrinology and Infertility. (2021). Diagnosis and treatment of luteal phase deficiency: A committee opinion. Fertility and Sterility, 115(6), 1416–1423. doi: 10.1016/j.fertnstert.2021.02.010
19. Okada, H., Tsuzuki, T., & Murata, H. (2018). Decidualization of the human endometrium. Reproductive Medicine and Biology, 17(3), 220–227. doi: 10.1002/ rmb2.12088
20. Serrahima, C., & Martínez, M. (2023). The experience of dysmenorrhea. Synthese, 201(5). doi: 10.1007/ s11229-023-04148-9
21. Kyathanahalli, C. N., Tu, F. F., Ashenafi, G., Schroer, M. S., & Hellman, K. M. (2025). Seeking the root causes of menstrual pain: A systematic review of biomarkers in menstrual effluent. Molecular Pain, 21. doi: 10.1177/17448069251360092
22. Chavda, V. P., Feehan, J., & Apostolopoulos, V. (2024). Inflammation: The cause of all diseases. Cells, 13(22), 1906. doi: 10.3390/cells13221906
23. Fang, X.-X., Zhai, M.-N., Zhu, M., He, C., Wang, H., Wang, J., & Zhang, Z.-J. (2023). Inflammation in pathogenesis of chronic pain: Foe and friend. Molecular Pain, 19. doi: 10.1177/17448069231178176
24. Liu, C., Chu, D., Kalantar‐Zadeh, K., George, J., Young, H. A., & Liu, G. (2021). Cytokines: From clinical significance to quantification. Advanced Science, 8(15). doi: 10.1002/advs.202004433
25. Liu, M., Saredy, J., Zhang, R., Shao, Y., Sun, Y., Yang, W. Y., Wang, J., Liu, L., Drummer, C., Johnson, C., Saaoud, F., Lu, Y., Xu, K., Li, L., Wang, X., Jiang, X., Wang, H., & Yang, X. (2020). Approaching inflammation paradoxes—proinflammatory cytokine blockages induce inflammatory regulators. Frontiers in Immunology, 11. doi: 10.3389/fimmu.2020.554301
26. Yao, C., & Narumiya, S. (2018). Prostaglandin‐cytokine crosstalk in chronic inflammation. British Journal of Pharmacology, 176(3), 337–354. doi: 10.1111/bph.14530
27. Jin, D., & Zhong, T. P. (2021). Prostaglandin signaling in ciliogenesis and development. Journal of Cellular Physiology, 237(6), 2632–2643. doi: 10.1002/jcp.30659
28. Tallima, H., & El Ridi, R. (2018). Arachidonic acid: Physiological roles and potential health benefits – A Review. Journal of Advanced Research, 11, 33–41. doi: 10.1016/j.jare.2017.11.004
29. Fajrin, I., Alam, G., & Usman, A. N. (2020). Prostaglandin level of primary dysmenorrhea pain sufferers. Enfermería Clínica, 30, 5–9. doi: 10.1016/j.enfcli.2019.07.016
30. Barcikowska, Z., Rajkowska-Labon, E., Grzybowska, M. E., Hansdorfer-Korzon, R., & Zorena, K. (2020). Inflammatory markers in dysmenorrhea and therapeutic options. International Journal of Environmental Research and Public Health, 17(4), 1191. doi: 10.3390/ ijerph17041191
31. Pillerová, M., Borbélyová, V., Hodosy, J., Riljak, V., Renczés, E., Frick, K. M., & Tóthová, Ľ. (2021). On the role of sex steroids in biological functions by classical and non-classical pathways. an update. Frontiers in Neuroendocrinology, 62, 100926. doi: 10.1016/j. yfrne.2021.100926
32. Prior, J. C. (2020). Women’s reproductive system as balanced estradiol and progesterone actions—a revolutionary, paradigm-shifting concept in women’s health. Drug Discovery Today: Disease Models, 32, 31–40. doi: 10.1016/j.ddmod.2020.11.005
33. Lewitus, V. J., & Blackwell, K. T. (2023). Estradiol receptors inhibit long-term potentiation in the dorsomedial striatum. Eneuro, 10(8). doi: 10.1523/eneuro.0071-23.2023
34. Teleanu, R. I., Niculescu, A.-G., Roza, E., Vladâcenco, O., Grumezescu, A. M., & Teleanu, D. M. (2022). Neurotransmitters—key factors in neurological and neurodegenerative disorders of the Central Nervous System. International Journal of Molecular Sciences, 23(11), 5954. doi: 10.3390/ijms23115954
35. Rizo, J. (2018). Mechanism of neurotransmitter release coming into focus. Protein Science, 27(8), 1364–1391. doi: 10.1002/pro.3445
36. Del Río, J. P., Alliende, M. I., Molina, N., Serrano, F. G., Molina, S., & Vigil, P. (2018). Steroid hormones and their action in women’s brains: The importance of hormonal balance. Frontiers in Public Health, 6. doi: 10.3389/fpubh.2018.00141
37. Wang, J.-X., Zhuang, J.-Y., Fu, L., Lei, Q., Fan, M., & Zhang, W. (2020). How ovarian hormones influence the behavioral activation and inhibition system through the dopamine pathway. PLOS ONE, 15(8). doi: 10.1371/journal.pone.0237032
38. Pourhamzeh, M., Moravej, F. G., Arabi, M., Shahriari, E., Mehrabi, S., Ward, R., Ahadi, R., & Joghataei, M. T. (2021). The roles of serotonin in neuropsychiatric disorders. Cellular and Molecular Neurobiology, 42(6), 1671–1692. doi: 10.1007/s10571-021-01064-9
39. Diekhof, E. K. (2018). Estradiol and the reward system in humans. Current Opinion in Behavioral Sciences, 23, 58–64. doi: 10.1016/j.cobeha.2018.03.010
40. Azargoonjahromi, A., & Alzheimer’s Disease Neuroimaging Initiative. (2024). Serotonin enhances neurogenesis biomarkers, hippocampal volumes, and cognitive functions in alzheimer’s disease. Molecular Brain, 17(1). doi: 10.1186/s13041-024-01169-4
41. Quah, S. K. L., McIver, L., Roberts, A. C., & Santangelo, A. M. (2020). Trait anxiety mediated by amygdala serotonin transporter in the common marmoset. The Journal of Neuroscience, 40(24), 4739–4749. doi: 10.1523/jneurosci.2930-19.2020
42. Hernández-Hernández, O. T., Martínez-Mota, L., Herrera-Pérez, J. J., & Jiménez-Rubio, G. (2019). Role of estradiol in the expression of genes involved in serotonin neurotransmission: Implications for female depression. Current Neuropharmacology, 17(5), 459–471. doi: 10.2174/1570159x16666180628165107
43. E. Yoest, K., A. Cummings, J., & B. Becker, J. (2015). Estradiol, dopamine and motivation. Central Nervous System Agents in Medicinal Chemistry, 14(2), 83–89. doi: 10.2174/1871524914666141226103135
44. Rodríguez-Campuzano, A. G., & Ortega, A. (2021). Glutamate transporters: Critical components of glutamatergic transmission. Neuropharmacology, 192, 108602. doi: 10.1016/j.neuropharm.2021.108602
45. Mutluay, S. U., & Karataş, H. (2022). A review of glutamate and its receptors: Their roles in Brain Physiology and pathology. Acta Medica, 53(2), 99–109. doi: 10.32552/2022.actamedica.650
46. Teleanu, R. I., Niculescu, A.-G., Roza, E., Vladâcenco, O., Grumezescu, A. M., & Teleanu, D. M. (2022a). Neurotransmitters—key factors in neurological and neurodegenerative disorders of the Central Nervous System. International Journal of Molecular Sciences, 23(11), 5954. doi: 10.3390/ijms23115954
47. Bencker, C., Gschwandtner, L., Nayman, S., Grikšienė, R., Nguyen, B., Nater, U. M., Guennoun, R., Sundström-Poromaa, I., Pletzer, B., Bixo, M., & Comasco, E. (2025). Progestagens and progesterone receptor modulation: Effects on the brain, mood, stress, and cognition in females. Frontiers in Neuroendocrinology, 76, 101160. doi: 10.1016/j.yfrne.2024.101160
48. Jung, J., Williams, S., & Ralph, M. A. (2025). The Role of GABA in Semantic Memory and Its Neuroplasticity. doi: 10.7554/elife.91771.3
49. Charatsi, D., Koukoura, O., Gkorezi Ntavela, I., Chintziou, F., Gkorila, G., Tsagkoulis, M., Mikos, T., Pistofidis, G., Hajiioannou, J., Daponte, A. (2018). Gastrointestinal and Urinary Tract Endometriosis: A review on the Commonest Locations of Extrapelvic Endometriosis. Advances in Medicine, 2018(1). doi: 10.1155/2018/3461209
50. Rocha, T.P., Andres, M.P., Carmona, F., Baracat E.C., Abrão M.S. (2022). Deep Endometriosis: the Involvement of Multiple Pelvic Compartments Is Associated with More Severe Pain Symptoms and Infertility. Reproductive Sciences, 30, 1668–1675. doi: 10.1007/ s43032-022-01104-9
51. Allaire, C., Bedaiwy, M.A., Yong, P.J. (2023). Diagnosis and management of endometriosis. Canadian Medical Association Journal, 195(10). doi: 10.1503/ cmaj.220637
52. Saunders, P.T.K., Horne, A.W. (2021). Endometriosis: Etiology, pathobiology, and therapeutic prospects. Cell, 184(11), 2807-2824. doi: 10.1016/j.cell.2021.04.041
53. Gruber, T. M., Mechsner, S. (2021). Pathogenesis of Endometriosis: The Origin of Pain and Subfertility. Cells, 10(6), 1381. doi: 10.3390/cells10061381’
54. Hudson, N. (2021). The missed disease? Endometriosis as an example of 'undone science'. Reproductive Biomedicine & Society Online, 14, 20-27. doi: 10.1016/j.rbms.2021.07.003.
55. Cousins, F.L., McKinnon, B.D., Mortlock, S., Fitzgerald, H.C., Zhang, C., Montgomery, G.W., Gargett, C.E. (2023). New concepts on the etiology of endometriosis. Journal of Obstetrics and Gynaecology Research, 49(4). doi: 10.1111/jog.15549
56. Bermo, M.S., Koppula, B., Kumar, M., Leblond, A., Matesan, M.C. (2020). The Peritoneum: What Nuclear Radiologists Need to Know. Seminars in Nuclear Medicine, 50(5), 405-418. doi: 10.1053/j.semnuclmed.2020.04.005
57. Hegazy, A.A. (2024). International Journal of Reproductive BioMedicine. doi: 10.18502/ijrm.v22i5.16433
58. Viganò, P., Caprara, F., Giola, F., Di Stefano, G., Somigliana, E., Vercellini, P. (2024). Is retrograde menstruation a universal, recurrent, physiological phenomenon? A systematic review of the evidence in humans and non-human primates. Human Reproduction Open, 2024(3). doi: 10.1093/hropen/hoae045
59. Nie, J., Yi, Y., Liu, X., Guo, S.W. (2025). Progressively diminished estrogen signaling concordant with increased fibrosis in ectopic endometrium. Human Reproduction Open, 2025(3). doi: 10.1093/hropen/ hoaf028
60. Garvey, M. (2024). Endometriosis: Future Biological Perspectives for Diagnosis and Treatment. International Journal of Molecular Sciences, 25(22). doi: 10.3390/ijms252212242.
61. Wang, Y., Nicholes, K., Shih, L.M. (2020). The Origin and Pathogenesis of Endometriosis. Annual Review Pathology: Mechanisms Disease, 15, 71-95. doi: 10.1146/annurev-pathmechdis-012419-032654
62. Oală, I.E., Mitranovici, M.I., Chiorean, D.M., Irimia, T., Crișan, A.I., Melinte, I.M., Cotruș, T., Tudorache, V., Moraru, L., Moraru, R., Caravia, L., Morariu, M., Pușcașiu, L. (2024). Endometriosis and the Role of Pro-Inflammatory and Anti-Inflammatory Cytokines in Pathophysiology: A Narrative Review of the Literature. Diagnostics,14(3), 312. doi: 10.3390/diagnostics14030312
63. Machairiotis, N., Vasilakaki, S., Thomakos, N. (2021). Inflammatory Mediators and Pain in Endometriosis: A Systematic Review. Biomedicines, 9(1), 54. doi: 10.3390/biomedicines9010054
64. Itani, R., Soubra, L., Karout, S., Rahme, D., Karout, L., Khojah, H.M.J. (2022). Primary Dysmenorrhea: Pathophysiology, Diagnosis, and Treatment Updates. Korean Journal of Family Medicine, 43(2), 101-108. doi: 10.4082/kjfm.21.0103.
65. Tamura, K., Yoshie, M., Kusama, K., Tsuru, A. (2025). Mechanisms of Decidual Dysfunction and Infertility in Endometriosis: Roles of Prostaglandins and SASP. Reproductive Medicine and Biology, 24(1). doi: 10.1002/rmb2.12663
66. Wang, L., Xing, Y. (2026). Exploring the therapeutic potential of disulfiram in endometriosis: mechanisms targeting inflammation, oxidative stress, pyroptosis, and angiogenesis. Gynecology and Obstetrics, 313. doi: 10.1007/s00404-026-08355-w
67. Jang, Y., Kim, M. & Hwang, S.W. (2020). Molecular mechanisms underlying the actions of arachidonic acid-derived prostaglandins on peripheral nociception. Journal of Neuroinflammation, 17. doi: 10.1186/ s12974-020-1703-1
68. Zhang, Y., Catts, V., Sheedy, D., McCrossin, T., Kril, J.J., Weickert, C.S. (2016). Cortical grey matter volume reduction in people with schizophrenia is associated with neuro-inflammation. Translational Psychiatry, 6. doi: 10.1038/tp.2016.238
69. Aird-Rossiter, C., Zhang. H., Alexander, D.C., Jones, D.K., Palombo, M. (2026). Decoding gray matter, large-scale analysis of brain cell morphometry to inform microstructural modeling of diffusion MR signals. Communications Biology, 9(1), 138. doi: 10.1038/ s42003-025-09353-5.
70. Rizor, E.J., Babenko, V., Dundon, N.M., Beverly-Aylwin, R., Stump, A., Hayes, M., Herschenfeld-Catalan. L., Jacobs, E.G., Grafton, S.T. (2024). Menstrual cycle-driven hormone concentrations co-fluctuate with white and gray matter architecture changes across the whole brain. Human Brain Mapping, 45(11). doi: 10.1002/hbm.26785.
71. Maulitz, L., Nehls, S., Stickeler, E., Ignatov, A., Kupec, T., Henn, A.T., Chechko, N., Tchaikovski, S.N. (2024). Psychological characteristics and structural brain changes in women with endometriosis and endometriosis-independent chronic pelvic pain. Human Reproduction, 39(11), 2473-2484. doi: 10.1093/humrep/deae207.
72. Manda, O., Hadjivassiliou, M., Varrassi, G., Zavridis, P., Zis, P. (2025). Exploring the Role of the Cerebellum in Pain Perception: A Narrative Review. Pain and Therapy, 14(3), 803-816. doi: 10.1007/s40122-025-00724-8.
73. Roychoudhury, S., Buza, N. (2025). Endometriosis Then and Now: A 100-Year Journey Around Pathogenesis and Clinicopathologic Associations. Archives of pathology & laboratory medicine, 150(1), 12-18. doi: 10.5858/arpa.2025-0309-RA.
74. Warren, A., Garrett, K., Frame, L.A. (2025). Disparities in women’s health and clinical considerations from a translational science perspective: A narrative review and framework for future directions. Women’s Health, 21. doi: 10.1177/17455057251399009
75. Bougie, O., Nwosu, I., Warshafsky, C. (2022). Revisiting the impact of race/ethnicity in endometriosis. Reproduction and Fertility, 3(2), R34–R41. doi: 10.1530/RAF-21-0106
76. De Corte, P., Klinghardt, M., von Stockum, S. and Heinemann, K. (2025). Time to Diagnose Endometriosis: Current Status, Challenges and Regional Characteristics—A Systematic Literature Review. BJOG: An International Journal of Obstetrics & Gynaecology, 132, 118-130. doi: 10.1111/1471-0528.17973
77. Ellis, K., Munro, D., Clarke, J. (2022). Endometriosis Is Undervalued: A Call to Action. Frontiers in Global Women's Health, 3. doi: 10.3389/fgwh.2022.902371.
1. Ilyka, D., Johnson, M. H., & Lloyd-Fox, S. (2021). Infant social interactions and brain development: A systematic review. Neuroscience & Biobehavioral Reviews, 130, 448–469. doi: 10.1016/j.neubiorev.2021.09.001
2. Leitzke, B. T., Cochrane, A., Stein, A. G., DeLap, G. A., Green, C. S., & Pollak, S. D. (2024). Children’s and adolescent’s use of context in judgments of emotion intensity. Affective Science, 6(1), 117–127. doi: 10.1007/ s42761-024-00279-5
3. Keating, C. T., & Cook, J. L. (2021). Facial expression production and recognition in autism spectrum disorders. Psychiatric Clinics of North America, 44(1), 125–139. doi: 10.1016/j.psc.2020.11.010
4. Kern Koegel, L., Ashbaugh, K., Navab, A., & Koegel, R. L. (2016). Improving empathic communication skills in adults with autism spectrum disorder. Journal of Autism and Developmental Disorders, 46(3), 921–933. doi: 10.1007/s10803-015-2633-0
5. Boldsen, S. (2022). Autism and the sensory disruption of Social Experience. Frontiers in Psychology, 13. doi: 10.3389/fpsyg.2022.874268
6. Neurodevelopmental disorders. (2022). Diagnostic and Statistical Manual of Mental Disorders. doi: 10.1176/appi.books.9780890425787.x01_neurodevelopmental_disorders
7. Frye, R. E. (2018). Social skills deficits in autism spectrum disorder: Potential biological origins and progress in developing therapeutic agents. CNS Drugs, 32(8), 713–734. doi: 10.1007/s40263-018-0556-y
8. Wang, L., Wang, B., Wu, C., Wang, J., & Sun, M. (2023). Autism spectrum disorder: Neurodevelopmental risk factors, biological mechanism, and precision therapy. International Journal of Molecular Sciences, 24(3), 1819. doi: 10.3390/ijms24031819
9. Talaat, F. M. (2023). Real-time facial emotion recognition system among children with autism based on Deep Learning and IOT. Neural Computing and Applications, 35(17), 12717–12728. doi: 10.1007/s00521023-08372-9
10. Jasso del Toro, C., & Nekaris, K. A.-I. (2022). Affiliative behaviors. Encyclopedia of Animal Cognition and Behavior, 106–111. doi: 10.1007/978-3-319-55065-7_1040
11. Young, C. (2019). Agonistic behavior. Encyclopedia of Animal Cognition and Behavior, 1–6. doi: 10.1007/9783-319-47829-6_320-1
12. Hay, D. F., Paine, A. L., Perra, O., Cook, K. V., Hashmi, S., Robinson, C., Kairis, V., & Slade, R. (2021). Prosocial and aggressive behavior: A longitudinal study. Monographs of the Society for Research in Child Development, 86(2), 7–103. doi: 10.1111/mono.12427
13. Dickerson, B. C. (2020). Dysfunction of social cognition and behavior. Continuum, 21(3), 660–677. doi: 10.1212/01.con.0000466659.05156.1d
14. Tuovinen, S., Tang, X., & Salmela-Aro, K. (2020). Introversion and social engagement: Scale validation, their interaction, and positive association with self-esteem. Frontiers in Psychology, 11. doi: 10.3389/ fpsyg.2020.590748
15. Golya, N., & McIntyre, L. L. (2017). Variability in adaptive behaviour in young children with autism spectrum disorder. Journal of Intellectual & Developmental Disability, 43(1), 102–111. doi: 10.3109/13668250.2017.1287886
16. Hur, J., DeYoung, K. A., Islam, S., Anderson, A. S., Barstead, M. G., & Shackman, A. J. (2019). Social context and the real-world consequences of social anxiety. Psychological Medicine, 50(12), 1989–2000. doi: 10.1017/s0033291719002022
17. Moraleda Sepúlveda, E., & López Resa, P. (2021). Evaluating quality of life in families with Williams Syndrome patients. Health and Quality of Life Outcomes, 19(1). doi: 10.1186/s12955-021-01704-0
18. Hodges, H., Fealko, C., & Soares, N. (2020). Autism spectrum disorder: Definition, epidemiology, causes, and clinical evaluation. Translational Pediatrics, 9(S1). doi: 10.21037/tp.2019.09.09
19. Rosen, N. E., Lord, C., & Volkmar, F. R. (2021). The diagnosis of autism: From kanner to DSM-III to DSM-5 and beyond. Journal of Autism and Developmental Disorders, 51(12), 4253–4270. doi: 10.1007/s10803021-04904-1
20. Grosvenor, L. P., Croen, L. A., Lynch, F. L., Marafino, B. J., Maye, M., Penfold, R. B., Simon, G. E., & Ames, J. L. (2024). Autism diagnosis among US children and adults, 2011-2022. JAMA Network Open, 7(10). doi: 10.1001/jamanetworkopen.2024.42218
21. Andrade, C. (2025). Autism spectrum disorder, 1. The Journal of Clinical Psychiatry, 86(2). doi: 10.4088/ jcp.25f15878
22. Grabrucker, A. M. (Ed.). (2021). Autism spectrum disorders. Exon Publications. doi: 10.36255/exonpublications.autismspectrumdisorders.2021
23. Wang, M., Zhang, X., Zhong, L., Zeng, L., Li, L., & Yao, P. (2025). Understanding autism: Causes, diagnosis, and advancing therapies. Brain Research Bulletin, 227, 111411. doi: 10.1016/j.brainresbull.2025.111411
24. Marotta, R., Risoleo, M. C., Messina, G., Parisi, L., Carotenuto, M., Vetri, L., & Roccella, M. (2020). The neurochemistry of autism. Brain Sciences, 10(3), 163. doi: 10.3390/brainsci10030163
25. Teleanu, R. I., Niculescu, A.-G., Roza, E., Vladâcenco, O., Grumezescu, A. M., & Teleanu, D. M. (2022). Neurotransmitters—key factors in neurological and neurodegenerative disorders of the Central Nervous System. International Journal of Molecular Sciences, 23(11), 5954. doi: 10.3390/ijms23115954
26. Guidolin, D., Tortorella, C., Cervetto, C., Marcoli, M., Maura, G., & Agnati, L. F. (2025). Interaction between oxytocin and dopamine signaling: Focus on the striatum. International Journal of Molecular Sciences, 26(17), 8711. doi: 10.3390/ijms26178711
27. Wang, A. R., Groome, A., Taniguchi, L., Eshel, N., & Bentzley, B. S. (2020). The role of dopamine in reward-related behavior: Shining New Light on an old debate. Journal of Neurophysiology, 124(2), 309–311. doi: 10.1152/jn.00323.2020
28. Padilla-Coreano, N., & Martínez-Rivera, F. J. (2025). How dopamine guides our social world. Pharmacological Reviews, 77(5), 100085. doi: 10.1016/j. pharmr.2025.100085
29. Cai, J., & Tong, Q. (2022). Anatomy and function of ventral tegmental area glutamate neurons. Frontiers in Neural Circuits, 16. doi: 10.3389/fncir.2022.867053
30. Azarfar, A., Calcini, N., Huang, C., Zeldenrust, F., & Celikel, T. (2018). Neural coding: A single neuron’s perspective. Neuroscience & Biobehavioral Reviews, 94, 238–247. doi: 10.1016/j.neubiorev.2018.09.007
31. Palminteri, S. (2025). Human reinforcement learning processes and biases: Computational characterization and possible applications to behavioral public policy. Mind & Society, 24(2), 249–273. doi: 10.1007/ s11299-025-00329-w
32. Schultz, W. (2016). Dopamine reward prediction error coding. Dialogues in Clinical Neuroscience, 18(1), 23–32. doi: 10.31887/dcns.2016.18.1/wschultz
33. Solié, C., Girard, B., Righetti, B., Tapparel, M., & Bellone, C. (2021). VTA dopamine neuron activity encodes social interaction and promotes reinforcement learning through social prediction error. Nature Neuroscience, 25(1), 86–97. doi: 10.1038/s41593-02100972-9
34. Kinard, J. L., Mosner, M. G., Greene, R. K., Addicott, M., Bizzell, J., Petty, C., Cernasov, P., Walsh, E., Eisenlohr‐Moul, T., Carter, R. M., McLamb, M., Hopper, A., Sukhu, R., & Dichter, G. S. (2020). Neural mechanisms of social and nonsocial reward prediction errors in adolescents with autism spectrum disorder. Autism Research, 13(5), 715–728. doi: 10.1002/aur.2273
35. Schuetze, M., Rohr, C. S., Dewey, D., McCrimmon, A., & Bray, S. (2017). Reinforcement learning in autism spectrum disorder. Frontiers in Psychology, 8. doi: 10.3389/fpsyg.2017.02035
36. Petersson, M., & Uvnäs-Moberg, K. (2024). Interactions of oxytocin and dopamine—effects on behavior in health and disease. Biomedicines, 12(11), 2440. doi: 10.3390/biomedicines12112440
37. Orchard, E. R., Dakin, S. C., & van Boxtel, J. J. (2022). Internal noise measures in coarse and fine motion direction discrimination tasks and the correlation with autism traits. Journal of Vision, 22(10), 19. doi: 10.1167/jov.22.10.19
38. Limon, A., & Corona-Moreno, M. (2025). Hyperconnectivity and disrupted signal-to-noise processing in autism. The Palgrave Encyclopedia of Disability, 1–9. doi: 10.1007/978-3-031-40858-8_179-1
39. Jones, C., Barrera, I., Brothers, S., Ring, R., & Wahlestedt, C. (2017). Oxytocin and social functioning. Dialogues in Clinical Neuroscience, 19(2), 193–201. doi: 10.31887/dcns.2017.19.2/cjones
40. Havranek, T., Bacova, Z., & Bakos, J. (2024). Oxytocin, GABA, and dopamine interplay in autism. Endocrine Regulations, 58(1), 105–114. doi: 10.2478/enr-20240012
41. Iremonger, K. J., & Power, E. M. (2025). The paraventricular nucleus of the hypothalamus: A key node in the control of behavioural states. The Journal of Physiology, 603(8), 2231–2243. doi: 10.1113/jp288366
42. Hou, G., Hao, M., Duan, J., & Han, M.-H. (2024). The formation and function of the VTA dopamine system. International Journal of Molecular Sciences, 25(7), 3875. doi: 10.3390/ijms25073875
43. Cross, E. A., Borland, J. M., Shaughnessy, E. K., Lee, S. D., Vu, V., Sambor, E. A., Meisel, R. L., Huhman, K. L., & Albers, H. E. (2025). Distinct subcircuits within the mesolimbic dopamine system encode the salience and valence of social stimuli. Psychopharmacology, 242(10), 2219–2232. doi: 10.1007/s00213-025-06793-z
44. Rappeneau, V., & Castillo Díaz, F. (2024). Convergence of oxytocin and dopamine signalling in neuronal circuits: Insights into the neurobiology of social interactions across species. Neuroscience & Biobehavioral Reviews, 161, 105675. doi: 10.1016/j.neubiorev.2024.105675
45. Xiao, L., Priest, M. F., & Kozorovitskiy, Y. (2018). Oxytocin functions as a spatiotemporal filter for excitatory synaptic inputs to VTA dopamine neurons. eLife, 7. doi: 10.7554/elife.33892
46. Shamay-Tsoory, S. G., & Abu-Akel, A. (2016). The social salience hypothesis of oxytocin. Biological Psychiatry, 79(3), 194–202. doi: 10.1016/j.biopsych.2015.07.020
47. Froemke, R. C., & Young, L. J. (2021). Oxytocin, neural plasticity, and social behavior. Annual Review of Neuroscience, 44(1), 359–381. doi: 10.1146/annurev-neuro-102320-102847
48. Triana-Del Rio, R., Ranade, S., Guardado, J., LeDoux, J., Klann, E., & Shrestha, P. (2022). The modulation of emotional and social behaviors by oxytocin signaling in limbic network. Frontiers in Molecular Neuroscience, 15. doi: 10.3389/fnmol.2022.1002846
49. Fineberg, S. K., & Ross, D. A. (2017). Oxytocin and the social brain. Biological Psychiatry, 81(3). doi: 10.1016/j. biopsych.2016.11.004
50. Sun, Y., Wang, X., Chen, Y., Luan, Z., & Hao, R. (2025). The impact of exogenous oxytocin on visual cortex plasticity across different stages of visual development. Scientific Reports, 15(1). doi: 10.1038/s41598025-96573-8
51. de la Mora, M. P., Pérez-Carrera, D., Crespo-Ramírez, M., Tarakanov, A., Fuxe, K., & Borroto-Escuela, D. O. (2016). Signaling in dopamine D2 receptor-oxytocin receptor heterocomplexes and its relevance for the anxiolytic effects of dopamine and oxytocin interactions in the amygdala of the rat. Biochimica et Biophysica Acta (BBA) - Molecular Basis of Disease, 1862(11), 2075–2085. doi: 10.1016/j.bbadis.2016.07.004
52. Šimić, G., Tkalčić, M., Vukić, V., Mulc, D., Španić, E., Šagud, M., Olucha-Bordonau, F. E., Vukšić, M., & R. Hof, P. (2021). Understanding emotions: Origins and roles of the amygdala. Biomolecules, 11(6), 823. doi: 10.3390/biom11060823
53. Meisner, O. C., Dal Monte, O., Fagan, N., Nandy, A. S., & Chang, S. W. (2025). Oxytocin in the amygdala sustains prosocial behavior via state-dependent amygdala–prefrontal modulation. The Journal of Neuroscience, 45(36). doi: 10.1523/jneurosci.2416-24.2025
54. Nerio-Morales, L. K., Boender, A. J., Young, L. J., Lamprea, M. R., & Smith, A. S. (2024). Limbic oxytocin receptor expression alters molecular signaling and social avoidance behavior in female prairie voles (microtus ochrogaster). Frontiers in Neuroscience, 18. doi: 10.3389/fnins.2024.1409316
55. An, X., Yu, P., & Chang, G. (2025). Dopamine type II receptors in amygdala along with oxytocin in hypothalamus regulate social behavior in male Mandarin voles. Pharmacology Biochemistry and Behavior, 250, 174002. doi: 10.1016/j.pbb.2025.174002
56. Hernández-Mondragón, J. C., Hernández-Hernández, D. A., Crespo-Ramírez, M., Prospero-García, O., Rocha-Arrieta, L., Fuxe, K., Borroto-Escuela, D. O., & Perez de la Mora, M. (2023). Evidence for the existence of facilitatory interactions between the dopamine D2 receptor and the oxytocin receptor in the amygdala of the rat. relevance for anxiolytic actions. Frontiers in Pharmacology, 14. doi: 10.3389/fphar.2023.1251922
57. Gordon, I., Jack, A., Pretzsch, C. M., Vander Wyk, B., Leckman, J. F., Feldman, R., & Pelphrey, K. A. (2016). Intranasal oxytocin enhances connectivity in the neural circuitry supporting social motivation and social perception in children with autism. Scientific Reports, 6(1). doi: 10.1038/srep35054
58. Kruppa, J. A., Gossen, A., Oberwelland Weiß, E., Kohls, G., Großheinrich, N., Cholemkery, H., Freitag, C. M., Karges, W., Wölfle, E., Sinzig, J., Fink, G. R., Herpertz-Dahlmann, B., Konrad, K., & Schulte-Rüther, M. (2018). Neural modulation of social reinforcement learning by intranasal oxytocin in male adults with high-functioning autism spectrum disorder: A randomized trial. Neuropsychopharmacology, 44(4), 749–756. doi: 10.1038/s41386-018-0258-7
59. Madipakkam, A. R., Rothkirch, M., Dziobek, I., & Sterzer, P. (2017). Unconscious avoidance of eye contact in autism spectrum disorder. Scientific Reports, 7(1). doi: 10.1038/s41598-017-13945-5
60. Millington, E. (2024). Sensorily stressed: an exploration of the relationship between anxiety and sensory reactivity in autistic people. University of Glasgow. doi: doi: 10.5525/gla.thesis.84280
61. Zhang, J., Eaton, M., Chen, X., Zhao, Y., Kant, S., Deming, B. A., Harish, K., Nguyen, H. P., Shu, Y., Lai, S., Wu, J., Que, Z., Wettschurack, K. W., Zhang, Z., Xiao, T., Halurkar, M. S., Olivero-Acosta, M. I., Yoo, Y.-E., Lanman, N. A., … Yang, Y. (2025). Restoration of excitation/inhibition balance enhances neuronal signal-to-noise ratio and rescues social deficits in autism-associated scn2a -deficiency. bioRxiv. doi: 10.1101/2025.03.04.641498
62. Mayer, A. V., Preckel, K., Ihle, K., Piecha, F. A., Junghanns, K., Reiche, S., Rademacher, L., Müller-Pinzler, L., Stolz, D. S., Kamp-Becker, I., Stroth, S., Roepke, S., Küpper, C., Engert, V., Singer, T., Kanske, P., Paulus, F. M., & Krach, S. (2022). Assessment of reward-related brain function after a single dose of oxytocin in autism: A randomized controlled trial. Biological Psychiatry Global Open Science, 2(2), 136–146. doi: 10.1016/j.bpsgos.2021.10.004
63. Pfeiffer, B., Piller, A., Giazzoni-Fialko, T., & Chainani, A. (2016). Meaningful outcomes for enhancing quality of life for individuals with autism spectrum disorder. Journal of Intellectual & Developmental Disability, 42(1), 90–100. doi: 10.3109/13668250.2016.1197893
64. Dai, Y., Chen, M., Deng, T., Huang, B., Ji, Y., Feng, Y., Liu, S., & Zhang, L. (2023). The importance of parenting self‐efficacy and social support for family quality of life in children newly diagnosed with autism spectrum disorder: A one‐year follow‐up study. Autism Research, 17(1), 148–161. doi: 10.1002/aur.3061
65. Maniram, J., Karrim, S. B., Oosthuizen, F., & Wiafe, E. (2022). Pharmacological management of core symptoms and comorbidities of autism spectrum disorder in children and adolescents: A systematic review. Neuropsychiatric Disease and Treatment, Volume 18, 1629–1644. doi: 10.2147/ndt.s371013
66. Abbeduto, L., & Sahin, M. (2021). Developing and evaluating treatments for the challenges of autism and related neurodevelopmental disabilities. Journal of Neurodevelopmental Disorders, 13(1). doi: 10.1186/ s11689-021-09404-y
67. Alsayouf, H. A., Talo, H., Biddappa, M. L., Qasaymeh, M., Qasem, S., & De Los Reyes, E. (2020). Pharmacological intervention in children with autism spectrum disorder with standard supportive therapies significantly improves core signs and symptoms: A single-center, retrospective case series. Neuropsychiatric Disease and Treatment, Volume 16, 2779–2794. doi: 10.2147/ndt.s277294
68. Griffiths, J. L., Mishaal, R. A., Nabetani, M., & Goldman, R. D. (2022). Oxytocin for the treatment of autism spectrum disorder in children. Canadian Family Physician, 68(2), 103–105. doi: 10.46747/cfp.6802103
69. Panda, P. K., Sharawat, I. K., Gupta, D., Palayullakandi, A., Sopanam, S., & Saha, S. (2025). Efficacy and safety of risperidone and aripiprazole in reducing severity of irritability in children with autism spectrum disorder: A randomized controlled trial. Brain and Development, 47(5), 104454. doi: 10.1016/j.braindev.2025.104454
70. da Rosa, A. L., Bezerra, O. S., Rohde, L. A., & Graeff-Martins, A. S. (2024). Exploring clozapine use in severe psychiatric symptoms associated with autism spectrum disorder: A scoping review. Journal of Psychopharmacology, 38(4), 324–343. doi: 10.1177/02698811241241384
71. Parker, K. J., Oztan, O., Libove, R. A., Sumiyoshi, R. D., Jackson, L. P., Karhson, D. S., Summers, J. E., Hinman, K. E., Motonaga, K. S., Phillips, J. M., Carson, D. S., Garner, J. P., & Hardan, A. Y. (2017). Intranasal oxytocin treatment for social deficits and biomarkers of response in children with autism. Proceedings of the National Academy of Sciences, 114(30), 8119–8124. doi: 10.1073/pnas.1705521114
72. Yamasue, H., Okada, T., Munesue, T., Kuroda, M., Fujioka, T., Uno, Y., Matsumoto, K., Kuwabara, H., Mori, D., Okamoto, Y., Yoshimura, Y., Kawakubo, Y., Arioka, Y., Kojima, M., Yuhi, T., Owada, K., Yassin, W., Kushima, I., Benner, S., … Kosaka, H. (2018). Effect of intranasal oxytocin on the core social symptoms of autism spectrum disorder: A randomized clinical trial. Molecular Psychiatry, 25(8), 1849–1858. doi: 10.1038/ s41380-018-0097-2
73. Davico, C., Secci, I., Vendrametto, V., & Vitiello, B. (2023). Pharmacological treatments in autism spectrum disorder: a narrative review. Journal of Psychopathology, 29(1/2). doi: doi: 10.36148/2284-0249N251
74. Osuna-Luque, J., Rodríguez-Ramos, Á., Gámez-del-Estal, M. del, & Ruiz-Rubio, M. (2018). Behavioral mechanisms that depend on dopamine and serotonin in caenorhabditis elegans interact with the antipsychotics risperidone and Aripiprazole. Journal of Experimental Neuroscience, 12. doi: 10.1177/1179069518798628
75. Bartram, L. A., Lozano, J., & Coury, D. L. (2019). Aripiprazole for treating irritability associated with autism spectrum disorders. Expert Opinion on Pharmacotherapy, 20(12), 1421–1427. doi: 10.1080/14656566.2019.1626825
76. da Rosa, A. L., da Costa, M. R., Sorato, G. B., Manjabosco, F. de, de Bem, É. B., Dellazari, L., Falcão, A. B., Cia, L. de, Bezerra, O. S., Borges, R. B., Rohde, L. A., & Graeff-Martins, A. S. (2025). Clozapine for treatment-resistant disruptive behaviors in youths with autism spectrum disorder aged 10-17 years: Protocol for an open-label trial. JMIR Research Protocols, 14. doi: 10.2196/58031
77. Huang, Y., Huang, X., Ebstein, R. P., & Yu, R. (2021). Intranasal oxytocin in the treatment of autism spectrum disorders: A multilevel meta-analysis. Neuroscience & Biobehavioral Reviews, 122, 18–27. doi: 10.1016/j.neubiorev.2020.12.028
78. Choi, H., Kim, J. H., Yang, H. S., Kim, J. Y., Cortese, S., Smith, L., Koyanagi, A., Dragioti, E., Radua, J., Fusar-Poli, P., Shin, J. I., Cheon, K.-A., & Solmi, M. (2024). Pharmacological and non-pharmacological interventions for irritability in autism spectrum disorder: A systematic review and meta-analysis with the grade assessment. Molecular Autism, 15(1). doi: 10.1186/s13229-024-00585-6
79. Gora, C., Dudas, A., Vaugrente, O., Drobecq, L., Pecnard, E., Lefort, G., & Pellissier, L. P. (2024). Deciphering autism heterogeneity: A molecular stratification approach in four mouse models. Translational Psychiatry, 14(1). doi: 10.1038/s41398-024-03113-5
80. Annamneedi, A., Gora, C., Dudas, A., Leray, X., Bozon, V., Crépieux, P., & Pellissier, L. P. (2023). Towards the convergent therapeutic potential of G protein‐coupled receptors in autism spectrum disorders. British Journal of Pharmacology, 182(14), 3044–3067. doi: 10.1111/bph.16216
81. Lawrence, K. E., Hernandez, L. M., Eilbott, J., Jack, A., Aylward, E., Gaab, N., Van Horn, J. D., Bernier, R. A., Geschwind, D. H., McPartland, J. C., Nelson, C. A., Webb, S. J., Pelphrey, K. A., Bookheimer, S. Y., Dapretto, M., Aylward, E., Bernier, R. A., Bookheimer, S. Y., Dapretto, M., … Wolf, J. (2020). Neural responsivity to social rewards in autistic female youth. Translational Psychiatry, 10(1). doi: 10.1038/s41398-020-0824-8
82. Kimber, L., Verrier, D., & Connolly, S. (2024). Autistic people’s experience of empathy and the autistic empathy deficit narrative. Autism in Adulthood, 6(3), 321–330. doi: 10.1089/aut.2023.0001
83. Litman, A., Sauerwald, N., Green Snyder, L., FossFeig, J., Park, C. Y., Hao, Y., Dinstein, I., Theesfeld, C. L., & Troyanskaya, O. G. (2025). Decomposition of phenotypic heterogeneity in autism reveals underlying genetic programs. Nature Genetics, 57(7), 1611–1619. doi: 10.1038/s41588-025-02224-z
84. Manzo, J., Hernández-Aguilar, M. E., Toledo-Cárdenas, M. R., Herrera-Covarrubias, D., Coria-Avila, G. A., Libreros-Jiménez, H. M., Fernández-Cañedo, L., & Ortega-Pineda, L. A. (2025). The long and winding road to understanding autism. NeuroSci, 6(3), 84. doi: 10.3390/neurosci6030084
1. Ezmeci, F. (2023). Brain development as 21st-century skills: What is the play doing to my brain? Journal of Pedagogical Sociology and Psychology, 5(3), 21-27. doi: 10.33902/jpsp.202323257.
2. Canepa, M. E., & Ramenghi, L. A. (2026). The neurobiology of play: a narrative review of evidence from mice and humans for advancing neurorehabilitation. Frontiers in Neuroscience, 19. doi: 10.3389/ fnins.2025.1729411.
3. Elbeltagi, R., Al-Beltagi, M., Saeed, N. K., & Alhawamdeh, R. (2023). Play therapy in children with autism: Its role, implications, and limitations. World Journal of Clinical Pediatrics, 12(1), 1–22. doi: 10.5409/wjcp. v12.i1.1.
4. Achterberg, E. J., van Kerkhof, L. W., Servadio, M., van Swieten, M. M., Houwing, D. J., Aalderink, M., Driel, N. V., Trezza, V., & Vanderschuren, L. J. (2015). Contrasting roles of dopamine and noradrenaline in the motivational properties of social play behavior in rats. Neuropsychopharmacology, 41, 858–868. doi: 10.1038/npp.2015.212.
5. Yogman, M., Garner, A., Hutchinson, J., Hirsh-Pasek, K., & Golinkoff, R. M. (2018). The power of play: A pediatric role in enhancing development in young children. Pediatrics, 142(3). doi: 10.1542/peds.2018-2058.
6. Chu, W. T., Mitchell, T., Foote, K. D., Coombes, S. A., & Vaillancourt, D. E. (2021). Functional imaging of the brainstem during visually-guided motor control reveals visuomotor regions in the Pons and midbrain. NeuroImage, 226. doi: 10.1016/j.neuroimage.2020.117627.
7. Eisenreich, B. R., Akaishi, R., & Hayden, B. Y. (2017). Control without controllers: Toward a distributed neuroscience of executive control. Journal of Cognitive Neuroscience, 29(10), 1684–1698. doi: 10.1162/ jocn_a_01139.
8. Matheis, M., & Estabillo, J. A. (2018). Assessment of fine and gross motor skills in children. In Handbook of Childhood Psychopathology and Developmental Disabilities Assessment (pp. 467–484). Springer, Cham. doi: 10.1007/978-3-319-93542-3_25.
9. Semeijn, M. (2018). Interacting with fictions: The role of pretend play in theory of mind acquisition. Review of Philosophy and Psychology, 10, 113–132. doi: 10.1007/s13164-018-0387-2.
10. Innocenti, G. M. (2022). Defining neuroplasticity. Handbook of Clinical Neurology, 184, 3–18. doi: 10.1016/B978-0-12-819410-2.00001-1.
11. Vieira, M. S., Santos, A. K., Vasconcellos, R., Goulart, V. A. M., Parreira, R. C., Kihara, A. H., Ulrich, H., & Resende, R. R. (2018). Neural stem cell differentiation into mature neurons: Mechanisms of regulation and biotechnological applications. Biotechnology Advances, 36(7), 1946–1970. doi: 10.1016/j. biotechadv.2018.08.002.
12. Chuma, S., Kiyosue, K., Akiyama, T., Kinoshita, M., Shimazaki, Y., Uchiyama, S., Sotoma, S., Okabe, K., & Harada, Y. (2024). Implication of thermal signaling in neuronal differentiation revealed by manipulation and measurement of intracellular temperature. Nature Communications, 15. doi: 10.1038/s41467-02447542-8.
13. Passos, A. P. D. (2023). Integration of motor, cognitive, nutritional, metabolic, and epigenetic influence variables related to early childhood as a tool to promote child development at kindergarten schools. In Recent Perspectives on Preschool Education and Care. IntechOpen. doi: 10.5772/intechopen.113145.
14. Sgro, M., & Mychasiuk, R. (2020). Playful genes: What do we know about the epigenetics of play behaviour? International Journal of Play, 9(1), 25–38. doi: 10.1080/21594937.2020.1720139.
15. Miranda, M., Morici, J. F., Zanoni, M. B., & Bekinschtein, P. (2019). Brain-derived neurotrophic factor: A key molecule for memory in the healthy and the pathological brain. Frontiers in Cellular Neuroscience, 13. doi: 10.3389/fncel.2019.00363.
16. Bein, O., & Niv, Y. (2025). Schemas, reinforcement learning and the medial prefrontal cortex. Nature Reviews Neuroscience, 26(3), 141–157. doi: 10.1038/ s41583-024-00893-z.
17. von Bohlen und Halbach, O., & von Bohlen und Halbach, V. (2018). BDNF effects on dendritic spine morphology and hippocampal function. Cell and Tissue Research, 373, 729–741. doi: 10.1007/s00441-0172782-x.
18. Prigge, C. L., & Kay, J. N. (2018). Dendrite morphogenesis from birth to adulthood. Current Opinion in Neurobiology, 53, 139–145. doi: 10.1016/j. conb.2018.07.007.
19. Gipson, C. D., & Olive, M. F. (2016). Structural and functional plasticity of dendritic spines – root or result of behavior? Genes, Brain and Behavior, 16(1), 101–117. doi: 10.1111/gbb.12324.
20. Amodeo, D. A., Grospe, G., Zang, H., Dwivedi, Y., & Ragozzino, M. E. (2017). Cognitive flexibility impairment and reduced frontal cortex BDNF expression in the Ouabain model of Mania. Neuroscience, 345, 229–242. doi: 10.1016/j.neuroscience.2016.05.058.
21. Manduca, A., Servadio, M., Damsteegt, R., Campolongo, P., Vanderschuren, L. J., & Trezza, V. (2016). Dopaminergic neurotransmission in the nucleus accumbens modulates social play behavior in rats. Neuropsychopharmacology, 41, 2215–2223. doi: 10.1038/npp.2016.22.
22. Biderman, N., Bakkour, A., & Shohamy, D. (2020). What are memories for? The hippocampus bridges past experience with future decisions. Trends in Cognitive Sciences, 24(7), 542–556. doi: 10.1016/j. tics.2020.04.004.
23. Gruber, M. J., & Ranganath, C. (2019). How curiosity enhances hippocampus-dependent memory: The prediction, appraisal, curiosity, and exploration (PACE) framework. Trends in Cognitive Sciences, 23(12), 1014–1025. doi: 10.1016/j.tics.2019.10.003.
24. Kerney, M., Smaers, J. B., Schoenemann, P. T., & Dunn, J. C. (2017). The coevolution of play and the cortico-cerebellar system in primates. Primates, 58, 485–491. doi: 10.1007/s10329-017-0615-x.
25. Camarata, S., Miller, L. J., & Wallace, M. T. (2020). Evaluating sensory integration/sensory processing treatment: Issues and analysis. Frontiers in Integrative Neuroscience, 14. doi: 10.3389/fnint.2020.556660.
26. Fernández, V., Llinares-Benadero, C., & Borrell, V. (2016). Cerebral cortex expansion and folding: What have we learned? The EMBO Journal, 35(10), 1021–1044. doi: 10.15252/embj.201593701.
27. Bredikyte, M., & Brandisauskiene, A. (2023). Pretend play as the space for development of self-regulation: Cultural-historical perspective. Frontiers in Psychology, 14. doi: 10.3389/fpsyg.2023.1186512.
28. Henschke, J. U., & Pakan, J. M. P. (2023). Engaging distributed cortical and cerebellar networks through motor execution, observation, and imagery. Frontiers in systems neuroscience, 17, 1165307. doi: 10.3389/ fnsys.2023.1165307
29. Eon Duval, P., Frick, A., & Denervaud, S. (2022). Divergent and convergent thinking across the schoolyears: A dynamic perspective on creativity development. The Journal of Creative Behavior, 57(2), 186–198. doi: 10.1002/jocb.569
30. Rawlings, B., S., Chetwynd-Talbot, D., Husband, E., Nuttall, A., Quinn, E., Taggart, R., & Roome, H. E. (2025). Divergent thinking is linked with convergent thinking; implications for models of creativity. Thinking & Reasoning, 31(4), 586–608. doi: 10.1080/13546783.2025.2485059
31. Shen, K., Welton, T., Lyon, M., McCorkindale, A. N., Sutherland, G. T., Burnham, S., Fripp, J., Martins, R., & Grieve, S. M. (2019). Structural core of the Executive Control Network: A high angular resolution diffusion MRI study. Human Brain Mapping, 41(5), 1226–1236. doi: 10.1002/hbm.24870
32. Buss, A. T., & Spencer, J. P. (2017). Changes in frontal and posterior cortical activity underlie the early emergence of executive function. Developmental Science, 21(4). doi: 10.1111/desc.12602
33. Clark, C. A., Chevalier, N., Nelson, J. M., James, T. D., Garza, J. P., Choi, H.-J., & Espy, K. A. (2016). I. Executive Control in early childhood. Monographs of the Society for Research in Child Development, 81(4), 7–29. doi: 10.1111/mono.12268
34. Kucyi, A., Esterman, M., Riley, C. S., & Valera, E. M. (2016). Spontaneous default network activity reflects behavioral variability independent of mind-wandering. Proceedings of the National Academy of Sciences, 113(48), 13899–13904. doi: 10.1073/pnas.1611743113
35. Zhang, W., Sjoerds, Z., & Hommel, B. (2020). Metacontrol of human creativity: The neurocognitive mechanisms of convergent and divergent thinking. NeuroImage, 210, 116572. doi: 10.1016/j.neuroimage.2020.116572
36. Sun, J., He, L., Chen, Q., Yang, W., Wei, D., & Qiu, J. (2021). The bright side and dark side of daydreaming predict creativity together through brain functional connectivity. Human Brain Mapping, 43(3), 902–914. doi: 10.1002/hbm.25693
37. Arda Tuncdemir, T. B. (2025). Integrating social-emotional learning through play: Perspectives from early childhood educators. Journal of Research in Childhood Education, 1–19. doi: 10.1080/02568543.2025.2567504
38. Shvo, M., Klassen, T. Q., & McIlraith, S. A. (2020). Towards the role of theory of mind in explanation. Lecture Notes in Computer Science, 75–93. doi: 10.1007/978-3-030-51924-7_5
39. Hashmi, S., Vanderwert, R. E., Price, H. A., & Gerson, S. A. (2020). Exploring the benefits of doll play through neuroscience. Frontiers in Human Neuroscience, 14. doi: 10.3389/fnhum.2020.560176
40. McNaughton, K. A., & Redcay, E. (2020). Interpersonal Synchrony in autism. Current Psychiatry Reports, 22(3). doi: 10.1007/s11920-020-1135-8
41. Koehne, S., Hatri, A., Cacioppo, J. T., & Dziobek, I. (2016). Perceived interpersonal synchrony increases empathy: Insights from autism spectrum disorder. Cognition, 146, 8–15. doi: 10.1016/j.cognition.2015.09.007
42. Bhat, A. N., Hoffman, M. D., Trost, S. L., Culotta, M. L., Eilbott, J., Tsuzuki, D., & Pelphrey, K. A. (2017). Cortical activation during action observation, action execution, and interpersonal synchrony in adults: A functional near-infrared spectroscopy (FNIRS) study. Frontiers in Human Neuroscience, 11. doi: 10.3389/ fnhum.2017.00431
43. Bolis, D., & Schilbach, L. (2018a). Observing and participating in social interactions: Action perception and action control across the autistic spectrum. Developmental Cognitive Neuroscience, 29, 168–175. doi: 10.1016/j.dcn.2017.01.009
44. Zhang, K., Chen, J., Yang, Z. et al. Investigating joint attention in children with autism spectrum disorder through virtual reality and eye-tracking: a comparative study. Educ Inf Technol 30, 18779–18798 (2025). doi: 10.1007/s10639-025-13554-0
45. Keating, J., Hashmi, S., Vanderwert, R. E., Davies, R. M., Jones, C. R., & Gerson, S. A. (2023). Embracing neurodiversity in doll play: Investigating neural and language correlates of doll play in a neurodiverse sample. European Journal of Neuroscience, 60(3), 4097–4114. doi: 10.1111/ejn.16144
46. Schottelkorb, A. A., Swan, K. L., & Ogawa, Y. (2020). Intensive child‐centered play therapy for children on the autism spectrum: A pilot study. Journal of Counseling & Development, 98(1), 63–73. doi: 10.1002/jcad.12300
47. Kirkham, P. (2017). ‘the line between intervention and abuse’ – autism and applied behaviour analysis. History of the Human Sciences, 30(2), 107–126. doi: 10.1177/0952695117702571
48. Hosokawa, R., & Katsura, T. (2018). Association between Mobile Technology use and child adjustment in early elementary school age. PLOS ONE, 13(7). doi: 10.1371/journal.pone.0199959
49. Russ, S. W. (2016). Pretend play: Antecedent of adult creativity. New Directions for Child and Adolescent Development, 2016(151), 21–32. doi: 10.1002/ cad.20154
50. Houser, N. E., Roach, L., Stone, M. R., Turner, J., & Kirk, S. F. L. (2016). Let the children play: Scoping review on the implementation and use of loose parts for promoting physical activity participation. AIMS Public Health, 3(4), 781–799. doi: 10.3934/publichealth.2016.4.781
51. Prompona, S., Papoudi, D., & Papadopoulou, K. (2019). Play during recess: Primary School Children’s perspectives and Agency. Education 3-13, 48(7), 765–778. doi: 10.1080/03004279.2019.1648534
1. Spielberg, S. (Director). (1993). Jurassic Park [Film]. Universal Pictures; Amblin Entertainment.
2. Bayona, J.A. (Director). (2018). Jurassic World: Fallen Kingdom [Film]. Universal Pictures; Amblin Entertainment; Legendary Pictures; The Kennedy/Marshal Company; Perfect World Pictures.
3. Herculano-Houzel, S. (2023). Theropod dinosaurs had primate-like numbers of telencephalic neurons. Journal of Comparative Neurology, 531(9), 962–974. doi: 10.1002/cne.25453
4. Reiner, A. (2023). How smart could dinosaurs get? The Journal of Comparative Neurology, 531(9), 956–958. doi: 10.1002/cne.25471
5. Jensen, T. R., Jacobs, I., Kverková, K., Lalić, L., Polonyiová, A., Stehlík, P., Reber, S. A., & Osvath, M. (2025). T. rex cognition was T. rex-like—A critical outlook on diverging views of the neurocognitive evolution in dinosaurs. The Anatomical Record. doi: 10.1002/ar.70074
6. Chiarenza, A. A. (2024). The macroecology of Mesozoic dinosaurs. Biology Letters, 20(11), 20240392. doi: 10.1098/rsbl.2024.0392
7. Benson, R. B. J. (2018). Dinosaur macroevolution and macroecology. Annual Review of Ecology, Evolution, and Systematics, 49(Volume 49), 379–408. doi: 10.1146/annurev-ecolsys-110617-062231
8. Reiner, A. (2023). Could theropod dinosaurs have evolved to a human level of intelligence? Journal of Comparative Neurology, 531(9), 975–1006. doi: 10.1002/cne.25458
9. Thibault, S., Py, R., Gervasi, A. M., Salemme, R., Koun, E., Lövden, M., Boulenger, V., Roy, A. C., & Brozzoli, C. (2021). Tool use and language share syntactic processes and neural patterns in the basal ganglia. Science, 374(6569), eabe0874. doi: 10.1126/science. abe0874
10. Balanoff, A. M. (2024). Dinosaur palaeoneurology: An evolving science. Biology Letters, 20(12), 20240472. doi: 10.1098/rsbl.2024.0472
11. Mannion, P. D. (2024). The spatiotemporal distribution of Mesozoic dinosaur diversity. Biology Letters, 20(12), 20240443. doi: 10.1098/rsbl.2024.0443
12. Chiarenza, A. A., Farnsworth, A., Mannion, P. D., Lunt, D. J., Valdes, P. J., Morgan, J. V., & Allison, P. A. (2020). Asteroid impact, not volcanism, caused the end-Cretaceous dinosaur extinction. Proceedings of the National Academy of Sciences, 117(29), 17084–17093. doi: 10.1073/pnas.2006087117
13. Condamine, F. L., Guinot, G., Benton, M. J., & Currie, P. J. (2021). Dinosaur biodiversity declined well before the asteroid impact, influenced by ecological and environmental pressures. Nature Communications, 12. doi: 10.1038/s41467-021-23754-0
14. Brusatte, S. L., O’Connor, J. K., & Jarvis, E. D. (2015). The origin and diversification of birds. Current Biology, 25(19), R888–R898. doi: 10.1016/j.cub.2015.08.003
15. James, F. C. (2021). How many dinosaurs are birds? BioScience, 71(9), 991–994. doi: 10.1093/biosci/ biab060
16. Field, D. J., Burton, M. G., Benito, J., Plateau, O., & Navalón, G. (2025). Whence the birds: 200 years of dinosaurs, avian antecedents. Biology Letters, 21(1), 20240500. doi: 10.1098/rsbl.2024.0500
17. Caspar, K. R., Gutiérrez-Ibáñez, C., Bertrand, O. C., Carr, T., Colbourne, J. A. D., Erb, A., George, H., Holtz Jr, T. R., Naish, D., Wylie, D. R., & Hurlburt, G. R. (2024). How smart was T. rex? Testing claims of exceptional cognition in dinosaurs and the application of neuron count estimates in palaeontological research. The Anatomical Record, 307, 3685–3716. doi: 10.1002/ar.25459
18. Sander, P. M. (2023). Sauropods. Current Biology, 33(2), R52–R58. doi: 10.1016/j.cub.2022.12.024
19. Edwards, G. (Director). (2025). Jurassic World Rebirth [Film]. Universal Pictures; Amblin Entertainment.
20. Rowe, A. J., & Rayfield, E. J. (2025). Carnivorous dinosaur lineages adopt different skull performances at gigantic size. Current Biology, 35(15), 3664–3673. doi: 10.1016/j.cub.2025.06.051
21. Agnolin, F. L., Motta, M. J., Brissón Egli, F., Lo Coco, G., & Novas, F. E. (2019). Paravian phylogeny and the dinosaur-bird transition: An overview. Frontiers in Earth Science. doi: 10.3389/feart.2018.00252
22. Kundanati, L., D’Incau, M., Bernardi, M., Scardi, P., & Pugno, N. M. (2019). A comparative study of the mechanical properties of a dinosaur and crocodile fossil teeth. Journal of the Mechanical Behavior of Biomedical Materials, 97, 365–374. doi: 10.1016/j. jmbbm.2019.05.025
23. Lockley, M. G., McCrea, R. T., Buckley, L. G., Deock Lim, J., Matthews, N. A., Breithaupt, B. H., Houck, K. J., Gierliński, G. D., Surmik, D., Soo Kim, K., Xing, L., Yong Kong, D., Cart, K., Martin, J., & Hadden, G. (2016). Theropod courtship: Large scale physical evidence of display arenas and avian-like scrape ceremony behaviour by Cretaceous dinosaurs. Scientific Reports, 6. doi: 10.1038/srep18952
24. Soma, M., & Iwama, M. (2017). Mating success follows duet dancing in the Java sparrow. PLOS ONE, 12(3), e0172655. doi: 10.1371/journal.pone.0172655
25. Hu, J., Xu, X., Zhao, Q., He, Y., Forster, C. A., & Han, F. (2024). Endocranial morphology of three early-diverging ceratopsians and implications for the behavior and the evolution of the endocast in ceratopsians. Paleobiology, 50(3), 490–502. doi: 10.1017/ pab.2024.25
26. Bruner, E. (2018). The brain, the braincase, and the morphospace. In E. Bruner, N. Ogihara, & H. Tanabe (Eds.), Digital Endocasts: From Skulls to Brains (pp. 93–114). Springer Japan. doi: 10.1007/978-4-43156582-6_7
27. Belén von Baczko, M., Cardillo, A. F., Ulloa-Guaiquin, K., Desojo, J. B., & Paulina-Carabajal, A. (2024). Paleoneurology and neuroanatomical notes on the South American “rauisuchian” Saurosuchus galilei from the Ischigualasto formation, San Juan, Argentina. The Anatomical Record, 307, 1239–1253. doi: 10.1002/ar.25362
28. Paulina-Carabajal, A., & Currie, P. J. (2017). The braincase of the theropod dinosaur Murusraptor: Osteology, neuroanatomy and comments on the paleobiological implications of certain endocranial features. Ameghiniana, 54(5), 617–640. doi: 10.5710/ AMGH.25.03.2017.3062
29. Zaidi, S., Yasir, H., & Jaka, A. (2024). Floccular syndrome—An atypical presentation of paraneoplastic cerebellar degeneration. Journal of Ayub Medical College Abbottabad, 36(3). doi: 10.55519/JAMC-0312493
30. Dumont Jr, M. V., Santucci, R. M., de Andrade, M. B., & de Oliveira, C. E. M. (2020). Paleoneurology of Baurusuchus (Crocodyliformes: Baurusuchidae), ontogenetic variation, brain size, and sensorial implications. The Anatomical Record, 305(10), 2670–2694. doi: 10.1002/ar.24567
31. Labra, N., Mounier, A., Leprince, Y., Rivière, D., Didier, M., Bardinet, E., Santin, M. D., Mangin, J. F., Filippo, A., Albessard-Ball, L., Beaudet, A., Broadfield, D., Bruner, E., Carlson, K. J., Cofran, Z., Falk, D., Gilissen, E., Gómez-Robles, A., Neubauer, S., … Balzeau, A. (2024). What do brain endocasts tell us? A comparative analysis of the accuracy of sulcal identification by experts and perspectives in palaeoanthropology. Journal of Anatomy, 244(2), 274–296. doi: 10.1111/ joa.13966
32. Kabadayi, C., & Osvath, M. (2017). Ravens parallel great apes in flexible planning for tool-use and bartering. Science, 357(6347), 202–204. doi: 10.1126/science.aam8138
33. van Schaik, C. P., Triki, Z., Bshary, R., & Heldstab, S. A. (2021). A farewell to the encephalization quotient: A new brain size measure for comparative primate cognition. Brain, Behavior and Evolution, 96(1), 1–12. doi: 10.1159/000517013
34. Leavens, D. A., Bard, K. A., & Hopkins, W. D. (2019). The mismeasure of ape social cognition. Animal Cognition, 22, 487–504. doi: 10.1007/s10071-017-1119-1
35. Wink, M. (2022). Intelligence in the animal kingdom. In R. M. Holm-Hadulla, J. Funke, & M. Wink (Eds.), Intelligence—Theories and Applications (pp. 87–103). Springer. doi: 10.1007/978-3-031-04198-3_4
36. Varricchio, D., Hogan, J., & Freimuth, W. (2021). Revisiting Russell’s troodontid: Autecology, physiology, and speculative tool use. Canadian Journal of Earth Sciences. doi: 10.1139/cjes-2020-0184
37. Audet, J.-N., Couture, M., Lefebvre, L., & Jarvis, E. D. (2024). Problem-solving skills are predicted by technical innovations in the wild and brain size in passerines. Nature Ecology & Evolution, 8, 806–816. doi: 10.1038/s41559-024-02342-7
38. Glock, H.-J. (2019). Agency, intelligence and reasons in animals. Philosophy, 94(4), 645–671. doi: 10.1017/ S0031819119000275
39. Burger, J. R., George Jr., M. A., Leadbetter, C., & Shaikh, F. (2019). The allometry of brain size in mammals. Journal of Mammalogy, 100(2), 276–283. doi: 10.1093/jmammal/gyz043
40. Lauters, P., Vercauteren, M., & Godefroit, P. (2023). Endocasts of ornithopod dinosaurs: Comparative anatomy. In Progress in Brain Research (Vol. 275, pp. 1–23). Elsevier. doi: 10.1016/bs.pbr.2022.10.002
41. Felice, R. N., & Goswami, A. (2018). Developmental origins of mosaic evolution in the avian cranium. Proceedings of the National Academy of Sciences, 115(3), 555–560 doi: 10.1073/pnas.1716437115
42. Fong, S., Rogell, B., Amcoff, M., Kotrschal, A., van der Bijl, W., Buechel, S. D., & Kolm, N. (2021). Rapid mosaic brain evolution under artificial selection for relative telencephalon size in the guppy (Poecilia reticulata). Science Advances, 7(46). doi: 10.1126/sciadv. abj4314
43. McGann, J. P. (2017). Poor human olfaction is a 19th-century myth. Science, 356(6338), eaam7263. doi: 10.1126/science.aam7263
44. Spierings, M. J., & ten Cate, C. (2016). Budgerigars and zebra finches differ in how they generalize in an artificial grammar learning experiment. Proceedings of the National Academy of Sciences, 113(27), E3977–E3984. doi: 10.1073/pnas.1600483113
45. Balakhonov, D., & Rose, J. (2017). Crows rival monkeys in cognitive capacity. Scientific Reports, 7(1), 8809. doi: 10.1038/s41598-017-09400-0
46. Zou, Y., Zhang, Z., Zeng, Y., Hu, H., Hao, Y., Huang, S., & Li, B. (2024). Common methods for phylogenetic tree construction and their implementation in R. Bioengineering, 11(5), 480. doi: 10.3390/bioengineering11050480
47. Shimizu, T., Shinozuka, K., Uysal, A. K., & Leilani, S. (2017). The origins of the bird brain: Multiple pulses of cerebral expansion in evolution. In S. Watanabe, M. A. Hofman, & T. Shimizu (Eds.), Evolution of the Brain, Cognition, and Emotion in Vertebrates (pp. 35–57). Springer Japan. doi: 10.1007/978-4-43156559-8_2
48. Ksepka, D. T., Balanoff, A. M., Smith, N. A., Bever, G. S., Bhullar, B.-A. S., Bourdon, E., Braun, E. L., Burleigh, J. G., Clarke, J. A., Colbert, M. W., Corfield, J. R., Degrange, F. J., De Pietri, V. L., Early, C. M., Field, D. J., Gignac, P. M., Gold, M. E. L., Kimball, R. T., Kawabe, S., Lefebvre, L., Marugán-Lobón, J., Mongle, C.S., Morhadt, A., Norell, M.A., Ridgely, R.C., Rothman, R.S., Scofield, R.P., Tambussi, C.P., Toress, C.R., van Tuinen, M., Walsh, S.A., Watanabe, A., Witmer, L.M., Wright, A.K., Zanno, L.E., Jarvis, E.D., Smaers, J. B. (2020). Tempo and pattern of avian brain size evolution. Current Biology, 30(11), 2026-2036.e3. doi: 10.1016/j.cub.2020.03.060
49. Kass, J. H., Qi, H.-X., Reed, J. L., & Krubitzer, L. A. (2026). The evolution of neocortex. In J. H. Kaas & S. Herculano-Houzel (Eds.), Evolution of Nervous Systems (3rd ed., Vol. 2, pp. 163–182). Academic Press. doi: 10.1016/B978-0-443-27380-3.00034-8
50. Güntürkün, O., von Eugen, K., Packheiser, J., & Pusch, R. (2021). Avian pallial circuits and cognition: A comparison to mammals. Current Opinion in Neurobiology, Evolution of Brains and Computation, 71, 29–36. doi: 10.1016/j.conb.2021.08.007
51. Carlén, M. (2017). What constitutes the prefrontal cortex? Science, 358(6362), 478–482. doi: 10.1126/ science.aan8868
52. Puelles, L., Sandoval, J. E., Ayad, A., del Corral, R., Alonso, A., Ferran, J. L., & Martínez-de-la-Torre, M. (2017). The pallium in reptiles and birds in the light of the updated tetrapartite pallium model. In Evolution of Nervous Systems: Second Edition (Vol. 1, pp. 519–555). doi: 10.1016/B978-0-12-804042-3.00014-2
53. Gaetano, T. M., Yacobucci, M. M., & Bingman, V. P. (2017). On the paleontology of animal cognition: Using the brain dimensions of modern birds to characterize maniraptor cognition. Journal of Advanced Neuroscience Research, 12–19. doi: 10.15379/24093564.2017.03
54. Yoshida, J., Kobayashi, Y., & Norell, M. A. (2023). An ankylosaur larynx provides insights for bird-like vocalization in non-avian dinosaurs. Communications Biology, 6, 152. doi: 10.1038/s42003-023-04513-x
55. Rose, J. (2022). The avian brain. Current Biology, 32(20), R1076–R1079. doi: 10.1016/j.cub.2022.07.072
56. Hawkins, J., Lewis, M., Klukas, M., Purdy, S., & Ahmad, S. (2019). A framework for intelligence and cortical function based on grid cells in the neocortex. Frontiers in Neural Circuits, 12. doi: 10.3389/fncir.2018.00121
57. Mateos-Aparicio, P., & Rodríguez-Moreno, A. (2019). The impact of studying brain plasticity. Frontiers in Cellular Neuroscience, 13. doi: 10.3389/fncel.2019.00066
58. Olkowicz, S., Kocourek, M., Lučan, R. K., Porteš, M., Fitch, W. T., Herculano-Houzel, S., & Němec, P. (2016). Birds have primate-like numbers of neurons in the forebrain. Proceedings of the National Academy of Sciences, 113(26), 7255–7260. doi: 10.1073/ pnas.1517131113
59. Font, E., Burghardt, G. M., & Leal, M. (2023). Brains, behaviour, and cognition: Multiple misconceptions. In C. Warwick, P. C. Arena, & G. M. Burghardt (Eds.), Health and Welfare of Captive Reptiles. Springer. doi: 10.1007/978-3-030-86012-7_7
60. Wiest, G. (2025). Hierarchical concepts in neuroethology. In Hierarchies in the Brain, Mind and Behaviour: A Principle Of Neural and Mental Function (pp. 31–93). Springer Nature Switzerland. doi: 10.1007/978-3-031-78030-1_3
61. Cronin, T. W., & Bok, M. J. (2016). Photoreception and vision in the ultraviolet. Journal of Experimental Biology, 219(18), 2790–2801. doi: 10.1242/jeb.128769
62. Kopnina, H., Washington, H., Taylor, B., & Piccolo, J. J. (2018). Anthropocentrism: More than just a misunderstood problem. Journal of Agricultural and Environmental Ethics, 31(1), 109–127. doi: 10.1007/s10806018-9711-1
63. Woodruff, D. C., Naish, D., & Dunning, J. (2021). Photoluminescent visual displays: An additional function of integumentary structures in extinct archosaurs? Historical Biology, 33(9), 1718–1725. doi: 10.1080/08912963.2020.1731806
64. Gignac, P. M., & Erickson, G. M. (2017). The Biomechanics Behind Extreme Osteophagy in Tyrannosaurus rex. Scientific Reports, 7, 2012. doi: 10.1038/ s41598-017-02161-w
65. Hughes, G. M., & Finarelli, J. A. (2019). Olfactory receptor repertoire size in dinosaurs. Proceedings of the Royal Society B: Biological Sciences, 286(1904). doi: 10.1098/rspb.2019.0909
66. Holtz Jr., T. R. (2021). Theropod guild structure and the tyrannosaurid niche assimilation hypothesis: Implications for predatory dinosaur macroecology and ontogeny in later Late Cretaceous Asiamerica. Canadian Journal of Earth Sciences, 58(9), 778–795. doi: 10.1139/cjes-2020-0174
67. Galway-Witham, J., & Stringer, C. (2018). How did Homo sapiens evolve? Science, 360(6395), 1296–1298. doi: 10.1126/science.aat6659
68. Turvey, S. T., & Crees, J. J. (2019). Extinction in the Anthropocene. Current Biology, 29(19), R982–R986. doi: 10.1016/j.cub.2019.07.040
69. Seddon, P. J. (2017). The ecology of de-extinction. Functional Ecology, 31(5), 992–995. doi: 10.1111/13652435.12856
1. YouTube. (2020). Lewis Hamilton’s Record, Valtteri Bottas Gets Hot And The Best Team Radio | 2020 Spanish Grand Prix. YouTube. https://www.youtube. com/watch?si=zOa-V0ciVKVW4l6R&v=JvYiin5gClQ&feature=youtu.be
2. Ziv, G. (2024). An ecological and embodied approach for training the racecar driver. Frontiers in Sports and Active Living, 6. doi: 10.3389/fspor.2024.1415406
3. Kotler, S., Mannino, M., Kelso, S., & Huskey, R. (2022). First few seconds for flow: A comprehensive proposal of the neurobiology and neurodynamics of state onset. Neuroscience & Biobehavioral Reviews, 143(104956), 104956. doi: 10.1016/j.neubiorev.2022.104956
4. Harris, D. J., Vine, S. J., & Wilson, M. R. (2017). Neurocognitive mechanisms of the flow state. Progress in Brain Research, 234, 221–243. doi: 10.1016/ bs.pbr.2017.06.012
5. Antonini Philippe, R., Singer, S. M., Jaeger, J. E. E., Biasutti, M., & Sinnett, S. (2022). Achieving Flow: An Exploratory Investigation of Elite College Athletes and Musicians. Frontiers in Psychology, 13. doi: 10.3389/fpsyg.2022.831508
6. Gale, M. K., Baker-Matsuoka, K., Nisky, I., & Okamura, A. M. (2025). Effect of Performance Feedback Timing on Motor Learning for a Surgical Training Task. IEEE Transactions on Biomedical Engineering, 1–10. doi: 10.1109/tbme.2025.3621106
7. Peifer, C., Wolters, G., Harmat, L., Heutte, J., Tan, J., Freire, T., Tavares, D., Fonte, C., Andersen, F. O., van den Hout, J., Šimleša, M., Pola, L., Ceja, L., & Triberti, S. (2022). A Scoping Review of Flow Research. Frontiers in Psychology, 13. doi: 10.3389/fpsyg.2022.815665
8. James, K. A., Stromin, J. I., Steenkamp, N., & Combrinck, M. I. (2023). Understanding the relationships between physiological and psychosocial stress, cortisol and cognition. Frontiers in Endocrinology, 14(1). doi: 10.3389/fendo.2023.1085950
9. Kloidt, J., & Barsalou, L. W. (2024). Establishing a Comprehensive Hierarchical construct of Eustress (CHE). Current Psychology, 43. doi: 10.1007/s12144024-06750-7
10. Uphill, M. A., Rossato, C. J. L., Swain, J., & O’Driscoll, J. (2019). Challenge and Threat: A Critical Review of the Literature and an Alternative Conceptualization. Frontiers in Psychology, 10(1). doi: 10.3389/ fpsyg.2019.01255
11. Lu, H., Van der Linden, D., & Bakker, A. B. (2025). The neuroscientific basis of flow: Learning progress guides task engagement and cognitive control. NeuroImage, 308, 121076. doi: 10.1016/j.neuroimage.2025.121076
12. Gold, J., & Ciorciari, J. (2020). A Review on the Role of the Neuroscience of Flow States in the Modern World. Behavioral Sciences, 10(9), 137. doi: 10.3390/ bs10090137
13. Irie, N., Morijiri, Y., & Yoshie, M. (2023). Symptoms of and coping strategies for music performance anxiety through different time periods. Frontiers in Psychology, 14. doi: 10.3389/fpsyg.2023.1138922
14. Rowland, D. L., Moyle, G., & Cooper, S. E. (2021). Remediation Strategies for Performance Anxiety across Sex, Sport and Stage: Identifying Common Approaches and a Unified Cognitive Model. International Journal of Environmental Research and Public Health, 18(19), 10160. doi: 10.3390/ijerph181910160
15. Aust, F., Beneke, T., Peifer, C., & Wekenborg, M. (2022). The Relationship between Flow Experience and Burnout Symptoms: A Systematic Review. International Journal of Environmental Research and Public Health, 19(7), 3865. doi: 10.3390/ijerph19073865
16. Huskey, R., Craighead, B., Miller, M. B., & Weber, R. (2018). Does intrinsic reward motivate cognitive control? a naturalistic-fMRI study based on the synchronization theory of flow. Cognitive, Affective, & Behavioral Neuroscience, 18(5), 902–924. doi: 10.3758/ s13415-018-0612-6
17. Akyol, P., & İmamoglu, O. (2019). The Relationship between Motivation and Flow States in Sports Faculty Students. Asian Journal of Education and Training, 5(3), 440–446. doi: 10.20448/journal.522.2019.53.440.446
18. Morris, L. S., Grehl, M. M., Rutter, S. B., Mehta, M., & Westwater, M. L. (2022). On What Motivates us: a Detailed Review of Intrinsic v. Extrinsic Motivation. Psychological Medicine, 52(10), 1–16. doi: 10.1017/ S0033291722001611
19. Deci, E. L., & Ryan, R. M. (2017). Self-Determination theory in work organizations: The state of a science. Annual Review of Organizational Psychology and Organizational Behavior, 4(1), 19–43. doi: 10.1146/annurev-orgpsych-032516-113108
20. Alameda, C., Sanabria, D., & Ciria, L. F. (2022). The brain in flow: A systematic review on the neural basis of the flow state. Cortex, 154, 348–364. doi: 10.1016/j.cortex.2022.06.005
21. Di Domenico, S. I., & Ryan, R. M. (2017). The emerging neuroscience of intrinsic motivation: A new frontier in self-determination research. Frontiers in Human Neuroscience, 11(145). doi: 10.3389/fnhum.2017.00145
22. Wang, A. R., Groome, A., Taniguchi, L., Eshel, N., & Bentzley, B. S. (2020). The role of dopamine in reward-related behavior: Shining new light on an old debate. Journal of Neurophysiology, 124(2), 309–311. doi: 10.1152/jn.00323.2020
23. Zhou, Z., Yi, L., Wang, Q., Lim, T. M., & Tan, E. K. (2023). Role of dopamine in the pathophysiology of Parkinson’s disease. Translational Neurodegeneration, 12(1). doi: 10.1186/s40035-023-00378-6
24. Bamford, I. J., & Bamford, N. S. (2019). The Striatum’s Role in Executing Rational and Irrational Economic Behaviors. The Neuroscientist, 25(5), 475–490. doi: 10.1177/1073858418824256
25. Linden, D., Tops, M., & Bakker, A. B. (2020). Go with the flow: A neuroscientific view on being fully engaged. European Journal of Neuroscience, 53(4). doi: 10.1111/ejn.15014
26. Trute, R. J., Alijani, A., & Erden, M. S. (2024). Visual cues of soft-tissue behaviour in minimal-invasive and robotic surgery. Journal of Robotic Surgery, 18(1). doi: 10.1007/s11701-024-02150-y
27. Drori, E., Cohen, L., Arkadir, D., Yahalom, G., & Mezer, A. A. (2025). Multiparametric quantitative MRI uncovers putamen microstructural changes in Parkinson’s disease. Npj Parkinson’s Disease, 11(1). doi: 10.1038/s41531-025-01020-0
28. Pascucci, D., Hickey, C., Jovicich, J., & Turatto, M. (2017). Independent circuits in basal ganglia and cortex for the processing of reward and precision feedback. NeuroImage, 162, 56–64. doi: 10.1016/j.neuroimage.2017.08.067
29. Zhao, S., Uono, S., Li, C., Yoshimura, S., & Toichi, M. (2018). The Influence of Self-Referential Processing on Attentional Orienting in Frontoparietal Networks. Frontiers in Human Neuroscience, 12. doi: 10.3389/ fnhum.2018.00199
30. Hutchison, R. M., & Gallivan, J. P. (2018). Functional coupling between frontoparietal and occipitotemporal pathways during action and perception. Cortex, 98, 8–27. doi: 10.1016/j.cortex.2016.10.020
31. Peifer, C., & Tan, J. (2021). The Psychophysiology of Flow Experience. Advances in Flow Research, 191–230. doi: 10.1007/978-3-030-53468-4_8
32. Barnett, K., & Vasiu, F. (2026). Enhanced functional connectivity between the default mode network and executive control network during flow states may facilitate creativity and emotional regulation, and may improve health outcomes. Frontiers in Behavioral Neuroscience, 19. doi: 10.3389/fnbeh.2025.1690499
33. Knyazev, G. G., Savostyanov, A. N., Bocharov, A. V., Levin, E. A., & Rudych, P. D. (2020). Intrinsic Connectivity Networks in the Self- and Other-Referential Processing. Frontiers in Human Neuroscience, 14, 579703. doi: 10.3389/fnhum.2020.579703
34. Chou, T., Dougherty, D. D., Nierenberg, A. A., & Deckersbach, T. (2022). Restoration of default mode network and task positive network anti-correlation associated with mindfulness-based cognitive therapy for bipolar disorder. Psychiatry Research: Neuroimaging, 319, 111419. doi: 10.1016/j.pscychresns.2021.111419
35. Dietrich, A., & Al-Shawaf, L. (2018). The Transient Hypofrontality Theory of Altered States of Consciousness. Journal of Consciousness Studies, 25(11-12), 226–247. https://www.ingentaconnect.com/content/ imp/jcs/2018/00000025/f0020011/art00012#
36. Fulmer, S. M., & Tulis, M. (2016). Adding nuance to the challenge-skill relationship: The interaction of perceived and actual skill. International Journal of Educational Research, 77, 143–154. doi: 10.1016/j. ijer.2016.04.002
37. Rosen, D., Oh, Y., Chesebrough, C., Zhang, F. (Zoe), & Kounios, J. (2024). Creative flow as optimized processing: Evidence from brain oscillations during jazz improvisations by expert and non-expert musicians. Neuropsychologia, 196, 108824. doi: 10.1016/j.neuropsychologia.2024.108824
38. Xuan, B., Mackie, M.-A., Spagna, A., Wu, T., Tian, Y., Hof, P. R., & Fan, J. (2016). The activation of interactive attentional networks. NeuroImage, 129, 308–319. doi: 10.1016/j.neuroimage.2016.01.017
39. de Sampaio Barros, M. F., Araújo-Moreira, F. M., Trevelin, L. C., & Radel, R. (2018). Flow experience and the mobilization of attentional resources. Cognitive, Affective, & Behavioral Neuroscience, 18(4), 810–823. doi: 10.3758/s13415-018-0606-4
40. Gabriel, O. P., D'amelio, T. A., Manuel, C. F., & María Luz, B. G. (2024). Flow state as a performance measure in esports. [Estado de flow como medida de rendimiento en Esports] Interdisciplinaria, 41(1), 1415. doi: doi: 10.16888/interd.2024.41.1.14
41. Tse, D. C. K., Nakamura, J., & Csikszentmihalyi, M. (2019). Beyond challenge-seeking and skill-building: Toward the lifespan developmental perspective on flow theory. The Journal of Positive Psychology, 15(2), 1–12. doi: 10.1080/17439760.2019.1579362
42. Hancock, P. A., Kaplan, A. D., Cruit, J. K., Hancock, G. M., MacArthur, K. R., & Szalma, J. L. (2019). A meta-analysis of flow effects and the perception of time. Acta Psychologica, 198, 102836. doi: 10.1016/j. actpsy.2019.04.007
43. Desai, D., Momin, A., Hirpara, P., Jha, H., Thaker, R., & Patel, J. (2024). Exploring the Role of Circadian Rhythms in Sleep and Recovery: A Review Article. Cureus, 16(6), e61568. doi: 10.7759/cureus.61568
44. Mendoza, J. (2025). Brain circadian clocks timing the 24h rhythms of behavior. Deleted Journal, 2(1). doi: 10.1038/s44323-025-00030-8
45. Paton, J. J., & Buonomano, D. V. (2018). The Neural Basis of Timing: Distributed Mechanisms for Diverse Functions. Neuron, 98(4), 687–705. doi: 10.1016/j. neuron.2018.03.045
46. Teghil, A., & Wittmann, M. (2025). How the body and brain process time. Neuroscience & Biobehavioral Reviews, 179, 106416. doi: 10.1016/j.neubiorev.2025.106416
47. Hua, A., Westlake, K. P., Bonnet, C. T., & Wang, J. (2025). Sensorimotor mismatch disrupts motor automaticity and increases anxiety during a goal-directed balance task. Frontiers in Human Neuroscience, 19. doi: 10.3389/fnhum.2025.1632265
48. Tomassini, A., & Morrone, M. C. (2016). Perceived visual time depends on motor preparation and direction of hand movements. Scientific Reports, 6(1). doi: 10.1038/srep27947
49. Calderone, A., Latella, D., Impellizzeri, F., Pasquale, P. de, Famà, F., Quartarone, A., & Calabrò, R. S. (2024). Neurobiological Changes Induced by Mindfulness and Meditation: A Systematic Review. Biomedicines, 12(11), 2613–2613. doi: 10.3390/biomedicines12112613
50. Chen, H., Liu, C., Zhou, F., Chiang, C.-H., Chen, Y.-L., Wu, K., Huang, D.-H., Liu, C.-Y., & Chiou, W.-K. (2022). The Effect of Animation-Guided Mindfulness Meditation on the Promotion of Creativity, Flow and
Affect. Frontiers in Psychology, 13. doi: 10.3389/ fpsyg.2022.894337
51. Prakash, R. S., Shankar, A., Tripathi, V., Yang, W. F. Z., Fisher, M., Bauer, C. C. C., Betzel, R., & Sacchet, M. D. (2024). Mindfulness Meditation and Network Neuroscience: Review, Synthesis, and Future Directions. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging. doi: 10.1016/j.bpsc.2024.11.005
52. De Filippi, E., Escrichs, A., Càmara, E., Garrido, C., Marins, T., Sánchez-Fibla, M., Gilson, M., & Deco, G. (2022). Meditation-induced effects on whole-brain structural and effective connectivity. Brain Structure and Function, 227(6), 2087–2102. doi: 10.1007/ s00429-022-02496-9
53. Venditti, S. (2025). Remodeling the Epigenome Through Meditation: Effects on Brain, Body, and Well-being. Sub-Cellular Biochemistry/Subcellular Biochemistry, 231–260. doi: 10.1007/978-3-03175980-2_7
54. Zhang, Y., Chen, S., Zhang, Z., Duan, W., Zhao, L., Weinschenk, G., Luh, W.-M., Anderson, A. K., & Dai, W. (2023). Effect of Meditation on Brain Activity during an Attention Task: A Comparison Study of ASL and BOLD Task fMRI. Brain Sciences, 13(12), 1653. doi: 10.3390/brainsci13121653
55. Sorella, S., Crescentini, C., Matiz, A., Chang, M., & Alessandro Grecucci. (2025). Resting-state BOLD temporal variability of the default mode network predicts spontaneous mind wandering, which is negatively associated with mindfulness skills. Frontiers in Human Neuroscience, 19. doi: 10.3389/fnhum.2025.1515902
56. Prakash, R. S., Shankar, A., Tripathi, V., Yang, W. F. Z., Fisher, M., Bauer, C. C. C., Betzel, R., & Sacchet, M. D. (2024). Mindfulness Meditation and Network Neuroscience: Review, Synthesis, and Future Directions. Biological Psychiatry: Cognitive Neuroscience and Neuroimaging. doi: 10.1016/j.bpsc.2024.11.005
57. Ogut, E. (2025). Assessing the efficacy of the Pomodoro technique in enhancing anatomy lesson retention during study sessions: a scoping review. BMC Medical Education, 25(1). doi: 10.1186/s12909-02508001-0
58. Bjurström, M. F., Lundkvist, E., Sturesson, L. W., Borgquist, O., Lundén, R., Fagerlund, M. J., Lipcsey, M., & Kander, T. (2025). Digital learning resource use among Swedish medical students: insights from a nationwide survey. BMC Medical Education, 25(1). doi: 10.1186/s12909-025-07446-7
1. Sahu, P., Verma, H. K., & Bhaskar, L. (2025). Alcohol and alcoholism associated neurological disorders: Current updates in a global perspective and recent recommendations. World Journal of Experimental Medicine, 15(1). doi: 10.5493/wjem.v15.i1.100402
2. Garrisson, H., Scholey, A., Ogden, E., & Benson, S. (2021). The effects of alcohol intoxication on cognitive functions critical for driving: A systematic review. Accident Analysis & Prevention, 154.. doi: 10.1016/j.aap.2021.106052
3. Hamed, M. A., Ekundina, V. O., & Akhigbe, R. E. (2023). Psychoactive drugs and male fertility: Impacts and mechanisms. Reproductive Biology and Endocrinology, 21(1), 69. doi: 10.1186/s12958-023-01098-2
4. Wiegmann, C., Mick, I., Brandl, E. J., Heinz, A., & Gutwinski, S. (2020). Alcohol and Dementia – What is the Link? A Systematic Review. Neuropsychiatric Disease and Treatment, 16, 87–99. doi: 10.2147/NDT. S198772
5. Chung, T., Creswell, K. G., Bachrach, R., Clark, D. B., & Martin, C. S. (2018). Adolescent Binge Drinking. Alcohol Research : Current Reviews, 39(1), 5–15. doi: 10.35946/arcr.v39.1.02
6. Sudhinaraset, M., Wigglesworth, C., & Takeuchi, D. T. (2016). Social and Cultural Contexts of Alcohol Use. Alcohol Research : Current Reviews, 38(1), 35–45. doi: 10.35946/arcr.v38.1.05
7. Orio, L., Alen, F., Pavón, F. J., Serrano, A., & García-Bueno, B. (2019). Oleoylethanolamide, Neuroinflammation, and Alcohol Abuse. Frontiers in Molecular Neuroscience, 11. doi: 10.3389/fnmol.2018.00490
8. Azarfar, A., Calcini, N., Huang, C., Zeldenrust, F., & Celikel, T. (2018). Neural coding: A single neuron’s perspective. Neuroscience & Biobehavioral Reviews, 94, 238–247. doi: 10.1016/j.neubiorev.2018.09.007
9. Wu, Z., Lin, D., & Li, Y. (2022). Pushing the frontiers: Tools for monitoring neurotransmitters and neuromodulators. Nature Reviews Neuroscience, 23(5), 257–274. doi: 10.1038/s41583-022-00577-6
10. Burkhardt, P., & Sprecher, S. G. (2017). Evolutionary origin of synapses and neurons – Bridging the gap. BioEssays, 39(10). doi: 10.1002/bies.201700024
11. Harrison, N. L., Skelly, M. J., Grosserode, E. K., Lowes, D. C., Zeric, T., Phister, S., & Salling, M. C. (2017). Effects of acute alcohol on excitability in the CNS. Neuropharmacology, Alcoholism, 122, 36–45. doi: 10.1016/j.neuropharm.2017.04.007
12. Maestú, F., de Haan, W., Busche, M. A., & DeFelipe, J. (2021). Neuronal excitation/inhibition imbalance: Core element of a translational perspective on Alzheimer's pathophysiology. Ageing Research Reviews, 69. doi: 10.1016/j.arr.2021.101372
13. De la Monte, S. M., & Tong, M. (2025). Molecular and biochemical pathologies in human alcohol-related cerebellar white matter degeneration. Advances in Drug and Alcohol Research, 5. doi: 10.3389/ adar.2025.15342
14. Schmahmann, J. D. (2019). The cerebellum and cognition. Neuroscience Letters, The Cerebellum in Health and Disease, 688, 62–75. doi: 10.1016/j. neulet.2018.07.005
15. Hermens, D. F., & Lagopoulos, J. (2018). Binge Drinking and the Young Brain: A Mini Review of the Neurobiological Underpinnings of Alcohol-Induced Blackout. Frontiers in Psychology, 9. doi: 10.3389/ fpsyg.2018.00012
16. Urbanik, A., Kozub, J., Karcz, P., & Ostrogórska, M. (2021). Changes in the brain directly following alcohol consumption—A study of healthy male individuals, with the use of proton magnetic resonance spectroscopy (1HMRS) and diffusion (DWI). Alcohol and Alcoholism, 56(4), 415–424. doi: 10.1093/alcalc/ agaa119
17. Dahchour, A., & Ward, R. J. (2022). Changes in Brain Dopamine Extracellular Concentration after Ethanol Administration; Rat Microdialysis Studies. Alcohol and Alcoholism, 57(2), 165–175. doi: 10.1093/alcalc/ agab072
18. Chau, P., Lidö, H. H., Söderpalm, B., & Ericson, M. (2018). Acamprosate’s ethanol intake-reducing effect is associated with its ability to increase dopamine. Pharmacology Biochemistry and Behavior, 175, 101–107. doi: 10.1016/j.pbb.2018.09.009
19. Costa, K. M., & Schoenbaum, G. (2022). Dopamine. Current Biology, 32(15), 817–824. doi: 10.1016/j. cub.2022.06.060
20. Telzer, E. H. (2016). Dopaminergic reward sensitivity can promote adolescent health: A new perspective on the mechanism of ventral striatum activation. Developmental Cognitive Neuroscience, Special Section: The Developmental Neuroscience of Adolescence: Revisiting, Refining, and Extending Seminal Models, 17, 57–67. doi: 10.1016/j.dcn.2015.10.010
21. Jones, A. W. (2019). Alcohol, its absorption, distribution, metabolism, and excretion in the body and pharmacokinetic calculations. WIREs Forensic Science, 1(5). doi: 10.1002/wfs2.1340
22. Sarala, M., Miettunen, J., Koskela, J., Mustonen, A., Rose, R. J., Hurtig, T., Veijola, J., & Niemelä, S. (2020). Frequent intoxication and alcohol tolerance in adolescence: Associations with psychiatric disorders in young adulthood. Addiction, 115(5), 888–900. doi: 10.1111/add.14889
23. Devi, A., Levin, M., & Waterhouse, A. L. (2023). Inhibition of ALDH2 by quercetin glucuronide suggests a new hypothesis to explain red wine headaches. Scientific Reports, 13(1). doi: 10.1038/s41598-02346203-y
24. Zhou, H., & Gelernter, J. (2024). Human genetics and epigenetics of alcohol use disorder. The Journal of Clinical Investigation, 134(16). doi: 10.1172/JCI172885
25. Palmer, E., Tyacke, R., Sastre, M., Lingford-Hughes, A., Nutt, D., & Ward, R. J. (2019). Alcohol Hangover: Underlying Biochemical, Inflammatory and Neurochemical Mechanisms. Alcohol and Alcoholism, 54(3), 196–203. doi: 10.1093/alcalc/agz016
26. Bereda, G. (2022). Mental and Physical Symptoms of Alcohol Hangover. Neurons and Neurological Disorders, 1(1), 1–4. doi: 10.58489/2836-8851/004
27. Haass-Koffler, C. L., & Perciballi, R. (2020). Alcohol Tolerance in Human Laboratory Studies for Development of Medications to treat Alcohol Use Disorder. Alcohol and Alcoholism, 55(2), 129–135. doi: 10.1093/ alcalc/agz103
28. Obad, A., Peeran, A., Little, J. I., Haddad, G. E., & Tarzami, S. T. (2018). Alcohol-Mediated Organ Damages: Heart and Brain. Frontiers in Pharmacology, 9. doi: 10.3389/fphar.2018.00081
29. Carvalho, A. F., Heilig, M., Perez, A., Probst, C., & Rehm, J. (2019). Alcohol use disorders. The Lancet, 394(10200), 781–792. doi: 10.1016/S01406736(19)31775-1
30. Witkiewitz, K., Litten, R. Z., & Leggio, L. (2019). Advances in the science and treatment of alcohol use disorder. Science Advances, 5(9). doi: 10.1126/sciadv. aax4043
31. Balloux, F., & van Dorp, L. (2017). Q&A: What are pathogens, and what have they done to and for us? BMC Biology, 15(1), 91. doi: 10.1186/s12915-017-0433-z
32. Poon, M. M. L., & Farber, D. L. (2020). The Whole Body as the System in Systems Immunology. iScience, 23(9). doi: 10.1016/j.isci.2020.101509
33. Sun, L., Su, Y., Jiao, A., Wang, X., & Zhang, B. (2023). T cells in health and disease. Signal Transduction and Targeted Therapy, 8(1), 235. doi: 10.1038/s41392-02301471-y
34. Zuluaga, P., Sanvisens, A., Teniente-Serra, A., El Ars, O., Fuster, D., Quirant-Sánchez, B., Martínez-Cáceres, E., & Muga, R. (2020). Loss of naive T lymphocytes is associated with advanced liver fibrosis in alcohol use disorder. Drug and Alcohol Dependence, 213. doi: 10.1016/j.drugalcdep.2020.108046
35. Mace, E. M. (2023). Human natural killer cells: Form, function, and development. Journal of Allergy and Clinical Immunology, 151(2), 371–385. doi: 10.1016/j. jaci.2022.09.022
36. Bottos, A., Gotthardt, D., Gill, J. W., Gattelli, A., Frei, A., Tzankov, A., Sexl, V., Wodnar-Filipowicz, A., & Hynes, N. E. (2016). Decreased NK-cell tumour immunosurveillance consequent to JAK inhibition enhances metastasis in breast cancer models. Nature Communications, 7(1). doi: 10.1038/ncomms12258
37. Pascual, M., Calvo-Rodriguez, M., Núñez, L., Villalobos, C., Ureña, J., & Guerri, C. (2021). Toll-like receptors in neuroinflammation, neurodegeneration, and alcohol-induced brain damage. IUBMB Life, 73(7), 900–915. doi: 10.1002/iub.2510
38. Caslin, B., Mohler, K., Thiagarajan, S., & Melamed, E. (2020). Alcohol as friend or foe in autoimmune diseases: A role for gut microbiome? Gut Microbes, 13(1). doi: 10.1080/19490976.2021.1916278
39. Liu, M., Saredy, J., Zhang, R., Shao, Y., Sun, Y., Yang, W. Y., Wang, J., Liu, L., Drummer, C., Johnson, C., Saaoud, F., Lu, Y., Xu, K., Li, L., Wang, X., Jiang, X., Wang, H., & Yang, X. (2020). Approaching Inflammation Paradoxes—Proinflammatory Cytokine Blockages Induce Inflammatory Regulators. Frontiers in Immunology, 11. doi: 10.3389/fimmu.2020.554301
40. El-Zayat, S. R., Sibaii, H., & Mannaa, F. A. (2019). Tolllike receptors activation, signaling, and targeting: An overview. Bulletin of the National Research Centre, 43(1), 187. doi: 10.1186/s42269-019-0227-2
41. Gupta, S. C., Kunnumakkara, A. B., Aggarwal, S., & Aggarwal, B. B. (2018). Inflammation, a Double-Edge Sword for Cancer and Other Age-Related Diseases. Frontiers in Immunology, 9, 2160. doi: 10.3389/fimmu.2018.02160
42. Chopade, P., Chopade, N., Zhao, Z., Mitragotri, S., Liao, R., & Chandran Suja, V. (2023). Alzheimer’s and Parkinson’s disease therapies in the clinic. Bioengineering & Translational Medicine, 8(1). doi: 10.1002/ btm2.10367
43. Çınaroğlu, M., Yılmazer, E., Ülker, S. V., Taçyıldız, K., & Tarlacı, S. (2025). Volumetric and cortical thickness alterations in alcohol dependence: Evidence of accelerated brain aging and clinical correlations. Frontiers in Psychiatry, 16. doi: 10.3389/fpsyt.2025.1662842
44. Henmar, S., Simonsen, E. B., & Berg, R. W. (2020). What are the gray and white matter volumes of the human spinal cord? Journal of Neurophysiology, 124(6), 1792–1797. doi: 10.1152/jn.00413.2020
45. Pereira-Rufino, L. da S., Gobbo, D. R., Conte, R., Sino, R. M. de, Oliveira, N. N. de, Fidalgo, T. M., Sato, J. R., Carrete Junior, H., Souza-Formigoni, M. L. O., Shi, Z., Vissoci, J. R. N., Wiers, C. E., & Céspedes, I. C. (2025). Reduced gray matter volume in limbic and cortical areas is associated with anxiety and depression in alcohol use disorder patients. Psychiatry Research: Neuroimaging, 347. doi: 10.1016/j.pscychresns.2025.111946
46. Guggenmos, M., Schmack, K., Sekutowicz, M., Garbusow, M., Sebold, M., Sommer, C., Smolka, M. N., Wittchen, H.-U., Zimmermann, U. S., Heinz, A., & Sterzer, P. (2017). Quantitative neurobiological evidence for accelerated brain aging in alcohol dependence. Translational Psychiatry, 7(12), 1279. doi: 10.1038/s41398-017-0037-y
47. Cole, J. H., & Franke, K. (2017). Predicting Age Using Neuroimaging: Innovative Brain Ageing Biomarkers. Trends in Neurosciences, 40(12), 681–690. doi: 10.1016/j.tins.2017.10.001
48. Nutt, D., Hayes, A., Fonville, L., Zafar, R., Palmer, E. O. C., Paterson, L., & Lingford-Hughes, A. (2021). Alcohol and the Brain. Nutrients, 13(11), 3938. doi: 10.3390/ nu13113938
49. Herman, A. M., & Duka, T. (2019). Facets of impulsivity and alcohol use: What role do emotions play? Neuroscience & Biobehavioral Reviews, Addiction: A Neurobiological and Cognitive Brain Disorder, 106, 202–216. doi: 10.1016/j.neubiorev.2018.08.011
50. Denier, N., Zahnd, K., Stein, M., Moggi, F., Kupper, Z., Federspiel, A., Wiest, R., Grieder, M., Soravia, L. M., & Bracht, T. (2025). Brain Connectivity of the Cingulate Cortex in Alcohol Use Disorder: Exploring Its Association With Mindfulness. Addiction Biology, 30(10). doi: 10.1111/adb.70036
51. Stevens, B. W., Morris, J. K., Diazgranados, N., & Ramchandani, V. A. (2021). Common Gray Matter Reductions in Alcohol Use and Obsessive-Compulsive Disorders: A Meta-analysis. Biological Psychiatry Global Open Science, 2(4), 421–431. doi: 10.1016/j. bpsgos.2021.11.010
52. Gröpper, S., Spengler, S., Stuke, H., Gawron, C. K., Parnack, J., Gutwinski, S., Wiers, C. E., & Bermpohl, F. (2016). Behavioral impulsivity mediates the relationship between decreased frontal gray matter volume and harmful alcohol drinking: A voxel-based morphometry study. Journal of Psychiatric Research, 83, 16–23. doi: 10.1016/j.jpsychires.2016.08.006
53. Le Berre, A.-P., Fama, R., & Sullivan, E. V. (2017). Executive Functions, Memory, and Social Cognitive Deficits and Recovery in Chronic Alcoholism: A Critical Review to Inform Future Research. Alcoholism: Clinical and Experimental Research, 41(8), 1432–1443. doi: 10.1111/acer.13431
54. Groh, J., & Simons, M. (2025). White matter aging and its impact on brain function. Neuron, 113(1), 127–139. doi: 10.1016/j.neuron.2024.10.019
55. Daviet, R., Aydogan, G., Jagannathan, K., Spilka, N., Koellinger, P. D., Kranzler, H. R., Nave, G., & Wetherill, R. R. (2022). Associations between alcohol consumption and gray and white matter volumes in the UK Biobank. Nature Communications, 13(1), 1175. doi: 10.1038/s41467-022-28735-5
56. Luo, L. (2021). Architectures of neuronal circuits. Science, 373(6559). doi: 10.1126/science.abg7285
57. Pihlaja, R., Öhman, S., Ollila, H., Tuulio-Henriksson, A., Koskinen, S., Salmela, V., Tiainen, M., Hästbacka, J., & Hokkanen, L. (2026). Executive functioning after COVID-19: Associations between self-report, informant-report, objective cognition, and psychological factors. A prospective cohort study. Journal of Psychosomatic Research, 205. doi: 10.1016/j.jpsychores.2026.112616
58. Peer, M., Nitzan, M., Bick, A. S., Levin, N., & Arzy, S. (2017). Evidence for Functional Networks within the Human Brain’s White Matter. Journal of Neuroscience, 37(27), 6394–6407. doi: 10.1523/JNEUROSCI.3872-16.2017
59. Zahr, N. M. (2024). Alcohol Use Disorder and Dementia: A Review. Alcohol Research : Current Reviews, 44(1), 03. doi: 10.35946/arcr.v44.1.03
60. Singer, E., Bhatt, K., Prashad, A., Rudman, L., Gadelmoula, I., & Michel, G. (2023). Diagnosis and Management of Marchiafava-Bignami Disease, a Rare Neurological Complication of Long-term Alcohol Abuse. Discoveries, 11(2). doi: 10.15190/d.2023.7
61. De León Reyes, N. S., Bragg-Gonzalo, L., & Nieto, M. (2020). Development and plasticity of the corpus callosum. Development, 147(18). doi: 10.1242/ dev.189738
62. Teixeira, J., Pereira, I., Castanho, M., & Simões do Couto, F. (2025). What is the impact of thiamine deficiency on cognitive function in patients with alcohol use disorder? – A systematic review. European Journal of Internal Medicine, 134, 59–65. doi: 10.1016/j.ejim.2025.01.008
63. Smith, T. J., Johnson, C. R., Koshy, R., Hess, S. Y., Qureshi, U. A., Mynak, M. L., & Fischer, P. R. (2021). Thiamine deficiency disorders: A clinical perspective. Annals of the New York Academy of Sciences, 1498(1), 9–28. doi: 10.1111/nyas.14536
64. Li, P., Zhu, Z., Wang, Y., Zhang, X., Yang, C., Zhu, Y., Zhou, Z., Chao, Y., Long, Y., Gao, Y., Liu, S., Zhang, L., Gao, P., & Qu, Q. (2024). Substrate transport and drug interaction of human thiamine transporters SLC19A2/A3. Nature Communications, 15(1). doi: 10.1038/s41467-024-55359-8
65. Bagash, H., Marwat, A., Marwat, A., Kraus, B., Bagash, H., Marwat, A., Marwat, A., & Kraus, B. (2021). A Case of Chronic Wernicke Encephalopathy (WE): An Underdiagnosed Phenomena. Cureus, 13. doi: 10.7759/ cureus.19100
66. Aloi, J., Crum, K. I., Blair, K. S., Zhang, R., Bashford-Largo, J., Bajaj, S., Schwartz, A., Carollo, E., Hwang, S., Leiker, E., Filbey, F. M., Averbeck, B. B., Dobbertin, M., & Blair, R. J. R. (2021). Individual associations of adolescent alcohol use disorder versus cannabis use disorder symptoms in neural prediction error signaling and the response to novelty. Developmental Cognitive Neuroscience, 48. doi: 10.1016/j. dcn.2021.100944
67. Blandino, K., DiLeo, A., Antonoudiou, P., Weiss, G. L., & Maguire, J. (2025). Stress and alcohol impact network states involved in emotional processing: Relevance of comorbid AUD and psychiatric illnesses. Psychopharmacology. doi: 10.1007/s00213-02506928-2
1. Twenge, J. M., Martin, G. N., & Spitzberg, B. H. (2019). Trends in U.S. Adolescents’ media use, 1976–2016: The rise of digital media, the decline of TV, and the (near) demise of print. Psychology of Popular Media Culture, 8(4), 329–345. doi: 10.1037/ppm0000203
2. 2. Canadian Paediatric Society, Ponti, M., Bélanger, S., Grimes, R., Heard, J., Johnson, M., Moreau, E., Norris, M., Shaw, A., Stanwick, R., Van Lankveld, J., & Williams, R. (2017). Screen time and young children: Promoting health and development in a digital world. Paediatrics & Child Health, 22(8), 461–468. doi: 10.1093/pch/pxx123
3. 3. Luby, J. L., Rogers, C., & McLaughlin, K. A. (2021). Environmental conditions to promote Healthy childhood Brain/Behavioral development: Informing early preventive interventions for delivery in routine care. Biological Psychiatry Global Open Science, 2(3), 233–241. doi: 10.1016/j.bpsgos.2021.10.003
4. 4. Bick, J., & Nelson, C. A. (2017). Early experience and brain development. Wiley Interdisciplinary Reviews Cognitive Science, 8(1–2). doi: 10.1002/wcs.1387
5. 5. Hutton, J. S., Dudley, J., Horowitz-Kraus, T., DeWitt, T., & Holland, S. K. (2019). Associations between Screen-Based Media Use and Brain White Matter Integrity in Preschool-Aged Children. JAMA Pediatrics, 174(1), e193869. doi: 10.1001/jamapediatrics.2019.3869
6. 6. Zablotsky, B., Ng, A. E., Black, L. I., Haile, G., Bose, J., Jones, J. R., & Blumberg, S. J. (2025). Associations between screen time use and health outcomes among US teenagers. Preventing Chronic Disease, 22, E38. doi: 10.5888/pcd22.240537
7. McDaniel, B. T., & Radesky, J. S. (2017). Technoference: parent distraction with technology and associations with child behavior problems. Child Development, 89(1), 100–109. doi: 10.1111/cdev.12822
8. Vergunst, F., Commisso, M., Geoffroy, M., Temcheff, C., Poirier, M., Park, J., Vitaro, F., Tremblay, R., Côté, S., & Orri, M. (2023). Association of childhood externalizing, internalizing, and comorbid symptoms with long-term economic and social outcomes. JAMA Network Open, 6(1), e2249568. doi: 10.1001/jamanetworkopen.2022.49568
9. Likhar, A., Baghel, P., & Patil, M. (2022). Early childhood development and social determinants. Cureus, 14(9), e29500. doi: 10.7759/cureus.29500
10. 10. Muppalla, S. K., Vuppalapati, S., Pulliahgaru, A. R., & Sreenivasulu, H. (2023). Effects of Excessive screen time on Child Development: An Updated review and Strategies for Management. Cureus, 15(6), e40608. doi: 10.7759/cureus.40608
11. Prieto, N., & Wrobleski, J. (2022). Neuron. In Encyclopedia of Animal Cognition and Behavior (pp. 4655–4659). doi: 10.1007/978-3-319-55065-7_1224
12. Zhao, W., Johnston, K. G., Ren, H., Xu, X., & Nie, Q. (2023). Inferring neuron-neuron communications from single-cell transcriptomics through NeuronChat. Nature Communications, 14(1), 1128. doi: 10.1038/s41467-023-36800-w
13. Gazerani, P. (2025). The neuroplastic brain: current breakthroughs and emerging frontiers. Brain Research, 1858, 149643. doi: 10.1016/j.brainres.2025.149643
14. Paley, B., & Hajal, N. J. (2022). Conceptualizing emotion regulation and coregulation as Family-Level phenomena. Clinical Child and Family Psychology Review, 25(1), 19–43. doi: 10.1007/s10567-022-003784
15. Schäfer, J., Reuter, T., Leuchter, M., & Karbach, J. (2024). Executive functions and problem-solving— The contribution of inhibition, working memory, and cognitive flexibility to science problem-solving performance in elementary school students. Journal of Experimental Child Psychology, 244, 105962. doi: 10.1016/j.jecp.2024.105962
16. Egner, T., & Siqi-Liu, A. (2024). Insights into control over cognitive flexibility from studies of task-switching. Current Opinion in Behavioral Sciences, 55, 101342. doi: 10.1016/j.cobeha.2023.101342
17. Blair, C. (2016). Executive function and early childhood education. Current Opinion in Behavioral Sciences, 10, 102–107. doi: 10.1016/j.cobeha.2016.05.009
18. Chai, W. J., Hamid, A. I. A., & Abdullah, J. M. (2018). Working Memory From the Psychological and Neurosciences Perspectives: A review. Frontiers in Psychology, 9, 401. doi: 10.3389/fpsyg.2018.00401
19. Kang, W., Hernández, S. P., Rahman, M. S., Voigt, K., & Malvaso, A. (2022). Inhibitory Control Development: a Network Neuroscience perspective. Frontiers in Psychology, 13, 651547. doi: 10.3389/fpsyg.2022.651547
20. Lobo, F. M., & Lunkenheimer, E. (2020). Understanding the parent-child coregulation patterns shaping child self-regulation. Developmental Psychology, 56(6), 1121–1134. doi: 10.1037/dev0000926
21. Cao, M., Huang, H., & He, Y. (2017). Developmental Connectomics from Infancy through Early Childhood. Trends in Neurosciences, 40(8), 494–506. doi: 10.1016/j.tins.2017.06.003
22. Aird-Rossiter, C., Zhang, H., Alexander, D. C., Jones, D. K., & Palombo, M. (2026). Decoding gray matter, large-scale analysis of brain cell morphometry to inform microstructural modeling of diffusion MR signals. Communications Biology, 9(1), 138. doi: 10.1038/ s42003-025-09353-5
23. Stassart, R. M., Möbius, W., Nave, K., & Edgar, J. M. (2018). The Axon-Myelin unit in Development and Degenerative disease. Frontiers in Neuroscience, 12, 467. doi: 10.3389/fnins.2018.00467
24. Neves, K. (2016). White Matter Expansion. In Evolution of Nervous Systems (pp. 291–308). doi: 10.1016/ b978-0-12-804042-3.00047-6
25. Rodriguez-Ayllon, M., Derks, I. P., Van Den Dries, M. A., Esteban-Cornejo, I., Labrecque, J. A., Yang-Huang, J., Raat, H., Vernooij, M. W., White, T., Ortega, F. B., Tiemeier, H., & Muetzel, R. L. (2019). Associations of physical activity and screen time with white matter microstructure in children from the general population. NeuroImage, 205, 116258. doi: 10.1016/j.neuroimage.2019.116258
26. Van Barreveld, M., Scheper, A., Vissers, C., Duinmeijer, I., & Hakvoort, B. (2025). Social–Emotional Functioning and Quality of Life in Language Disorders: A Systematic Review of Development from Childhood to Adolescence. International Journal of Language & Communication Disorders, 60(3), e70039. doi: 10.1111/1460-6984.70039
27. Bell, C., Bierstedt, L., Hu, T., Ogren, M., Reider, L. B., & LoBue, V. (2024). Learning through language: The importance of emotion and mental state language for children’s social and emotional learning. Social and Emotional Learning Research Practice and Policy, 4, 100061. doi: 10.1016/j.sel.2024.100061
28. Amicone, G., Petruccelli, I., De Dominicis, S., Gherardini, A., Costantino, V., Perucchini, P., & Bonaiuto, M. (2018). Green Breaks: The restorative effect of the school environment’s green areas on children’s cognitive performance. Frontiers in Psychology, 9, 1579. doi: 10.3389/fpsyg.2018.01579
29. Ernst, J., Sobel, D., & Neil, A. (2022). Executive function in early childhood: Harnessing the potential of nature-based practices to elevate and equalize outcomes. Frontiers in Education, 7. doi: 10.3389/feduc.2022.1011912
30. Tekeci, Y., Torpil, B., & Altuntaş, O. (2024). The impact of screen exposure on screen addiction and sensory processing in typically developing children aged 6–10 years. Children, 11(4), 464. doi: 10.3390/ children11040464
31. Britto, P. R., Lye, S. J., Proulx, K., Yousafzai, A. K., Matthews, S. G., Vaivada, T., Perez-Escamilla, R., Rao, N., Ip, P., Fernald, L. C. H., MacMillan, H., Hanson, M., Wachs, T. D., Yao, H., Yoshikawa, H., Cerezo, A., Leckman, J. F., & Bhutta, Z. A. (2017). Nurturing care: promoting early childhood development. The Lancet, 389(10064), 91–102. doi: 10.1016/s01406736(16)31390-3
32. Friedman, N. P., & Robbins, T. W. (2021). The role of prefrontal cortex in cognitive control and executive function. Neuropsychopharmacology, 47(1), 72–89. doi: 10.1038/s41386-021-01132-0
33. Shou, Q., Yamashita, M., & Mizuno, Y. (2025). Association of screen time with attention-deficit/hyperactivity disorder symptoms and their development: the mediating role of brain structure. Translational Psychiatry, 15(1), 447. doi: 10.1038/s41398-025-03672-1
34. Lewin, K. M., Meshi, D., Aladé, F., Lescht, E., Herring, C., Devaraju, D. S., & Wray, A. H. (2023). Children’s screentime is associated with reduced brain activation during an inhibitory control task: A pilot EEG study. Frontiers in Cognition, 2. doi: 10.3389/ fcogn.2023.1018096
35. Kostyrka-Allchorne, K., Cooper, N. R., Kennett, S., Nestler, S., & Simpson, A. (2019). The Short-Term Effect of Video Editing Pace on Children’s Inhibition and N2 and P3 ERP Components during Visual Go/ No-Go Task. Developmental Neuropsychology, 44(4), 385–396. doi: 10.1080/87565641.2019.1630628
36. Kar, S. S., Dube, R., Goud, B. K. M., Gibrata, Q. S., El-Balbissi, A. A., Salim, T. A., & Fatayerji, R. N. M. a. K. (2025). Impact of screen time on development of children. Children, 12(10), 1297. doi: 10.3390/children12101297
37. Perry, N. B., Dollar, J. M., Calkins, S. D., Keane, S. P., & Shanahan, L. (2018). Childhood self-regulation as a mechanism through which early overcontrolling parenting is associated with adjustment in preadolescence. Developmental Psychology, 54(8), 1542–1554. doi: 10.1037/dev0000536
38. Jusienė, R., Breidokienė, R., Baukienė, E., & Rakickienė, L. (2025). Emotional Reactivity and Behavioral problems in preschoolers: the interplay of parental stress, Media-Related Coping, and child screen time. Children, 12(2), 188. doi: 10.3390/children12020188
39. Small, G. W., Lee, J., Kaufman, A., Jalil, J., Siddarth, P., Gaddipati, H., Moody, T. D., & Bookheimer, S. Y. (2020). Brain health consequences of digital technology use. Dialogues in Clinical Neuroscience, 22(2), 179–187. doi: 10.31887/dcns.2020.22.2/gsmall
40. Arshad, D., Joyia, U. M., Fatima, S., Khalid, N., Rishi, A. I., Rahim, N. U. A., Bukhari, S. F., Shairwani, G. K., & Salmaan, A. (2021). The adverse impact of excessive smartphone screen-time on sleep quality among young adults: A prospective cohort. Sleep Science, 14(04), 337–341. doi: 10.5935/1984-0063.20200114
41. LeBourgeois, M. K., Hale, L., Chang, A., Akacem, L. D., Montgomery-Downs, H. E., & Buxton, O. M. (2017). Digital media and sleep in childhood and adolescence. PEDIATRICS, 140(Supplement_2), S92–S96. doi: 10.1542/peds.2016-1758j
42. Brodt, S., Inostroza, M., Niethard, N., & Born, J. (2023). Sleep—A brain-state serving systems memory consolidation. Neuron, 111(7), 1050–1075. doi: 10.1016/j. neuron.2023.03.005
43. Toth, C., Osser, B., Bondar, L. I., Fazakas, R., Marcu, F. M., Pascalau, N. A., Suciu, R. N., & Dogaru, B. G. (2025). Associations between screen time, sleep, and Executive Function in School-Aged Children and Adolescents: The Moderating Role of Digital Content and Age. Journal of Clinical Medicine, 14(24), 8842. doi: 10.3390/jcm14248842
44. Ordway, M. R., Logan, S., & Sutton, E. H. (2022). Sleep deficiency in young children. Clinics in Chest Medicine, 43(2), 229–237. doi: 10.1016/j.ccm.2022.02.007
45. Carter, B., Rees, P., Hale, L., Bhattacharjee, D., & Paradkar, M. S. (2016). Association between Portable Screen-Based Media Device access or use and sleep outcomes. JAMA Pediatrics, 170(12), 1202. doi: 10.1001/jamapediatrics.2016.2341
46. Félix, A., & Candeias, A. (2025). Sleep as a Developmental Process: A systematic review of cognitive, emotional, and behavioral outcomes in children aged 6–12 years. Clocks & Sleep, 7(4), 66. doi: 10.3390/ clockssleep7040066
47. Woods, A. D., Jiao, J. L., Morgan, P. L., & Buxton, O. M. (2023). Is sleep longitudinally related to children’s achievement, executive function and classroom behaviour? Infant and Child Development, 33(2). doi: 10.1002/icd.2426
48. Tamana, S. K., Ezeugwu, V., Chikuma, J., Lefebvre, D. L., Azad, M. B., Moraes, T. J., Subbarao, P., Becker, A. B., Turvey, S. E., Sears, M. R., Dick, B. D., Carson, V., Rasmussen, C., Investigators, C. S., Pei, J., & Mandhane, P. J. (2019). Screen-time is associated with inattention problems in preschoolers: Results from the CHILD birth cohort study. PLoS ONE, 14(4), e0213995. doi: 10.1371/journal.pone.0213995
49. Nivins, S., Mooney, M. A., Nigg, J., & Klingberg, T. (2025). Digital Media, Genetics, and Risk for ADHD symptoms in Children: a longitudinal study. Pediatrics Open Science, 2(1), 1–10. doi: 10.1542/pedsos.2025-000922
50. Dai, Y., & Ouyang, N. (2026). Excessive screen time is associated with mental health problems in US children and adolescents: physical activity and sleep as parallel mediators. Humanities and Social Sciences Communications, 13(1). doi: 10.1057/s41599-02606609-1
51. Chan, Y., Jang, J., & Ho, C. (2021). Effects of physical exercise on children with attention deficit hyperactivity disorder. Biomedical Journal, 45(2), 265–270. doi: 10.1016/j.bj.2021.11.011
52. Chaddock-Heyman, L., Erickson, K. I., Kienzler, C., Drollette, E. S., Raine, L. B., Kao, S., Bensken, J., Weisshappel, R., Castelli, D. M., Hillman, C. H., & Kramer, A. F. (2018). Physical activity increases white matter microstructure in children. Frontiers in Neuroscience, 12, 950. doi: 10.3389/fnins.2018.00950
53. Stiglic, N., & Viner, R. M. (2019). Effects of screentime on the health and well-being of children and adolescents: a systematic review of reviews. BMJ Open, 9(1), e023191. doi: 10.1136/bmjopen-2018-023191
54. Zubair, U., Khan, M. K., & Albashari, M. (2023). Link between excessive social media use and psychiatric disorders. Annals of Medicine and Surgery, 85(4), 875–878. doi: 10.1097/ms9.0000000000000112
55. Hahnefeld, A., Fink, M., Beherec, S. L., Baur, M. A., Bernhardt, K., & Mall, V. (2024). Correlation of screen exposure to stress, learning, cognitive and language performance in children. European Child & Adolescent Psychiatry, 34(5), 1615–1624. doi: 10.1007/ s00787-024-02593-6
56. Eirich, R., McArthur, B. A., Anhorn, C., McGuinness, C., Christakis, D. A., & Madigan, S. (2022). Association of screen time with internalizing and externalizing behavior problems in children 12 years or younger. JAMA Psychiatry, 79(5), 393. doi: 10.1001/jamapsychiatry.2022.0155
57. Ponti, M. (2023). Screen time and preschool children: Promoting health and development in a digital world. Paediatrics & Child Health, 28(3), 184–192. doi: 10.1093/pch/pxac125
58. Brushe, M. E., Haag, D. G., Melhuish, E. C., Reilly, S., & Gregory, T. (2024). Screen time and Parent-Child talk when children are aged 12 to 36 months. JAMA Pediatrics, 178(4), 369. doi: 10.1001/jamapediatrics.2023.6790
59. Meng, X., Liang, X., Liu, C., Cheng, N., Lu, S., Zhang, K., Yin, Y., Cheng, T., Lu, C., & Wang, Z. (2023). Associations between screen media use and young children’s inhibitory control: Evidence from behavioral and fNIRS study. Computers in Human Behavior, 152, 108041. doi: 10.1016/j.chb.2023.108041
LEQEMBI: A NEW ALZHEIMER’S DRUG BRINGS HOPE AND SPARKS CONTROVERSY
1. ALZHEIMER'S ASSOCIATION REPORT. (2025). 2025 Alzheimer's disease facts and figures. Alzheimer’s & Dementia, 21(4). doi: 10.1002/alz.70235.
2. Hampel, J., Hardy, J., Belnnow, K., Chen, C., Perry, G., Hyun Kim, S., Villemagne, V. L., Aisen, P., Vendruscolo, M., Iwatsubo, T., Masters, C. L., Cho, M., Lannfelt, L., Cummings, J. L., & Vergallo, A. (2021). The amyloid- pathway in Alzheimer’s disease. Molecular Psychiatry, 26, 5481–5503. doi: 10.1038/s41380-02101249-0.
3. Cummings, J., Leisgang O., Amanda M., Cammann, D., Powell, J., & Chen, J. (2024). Anti-amyloid monoclonal antibodies for the treatment of Alzheimer's disease. BioDrugs, 38, 5-22. doi: 10.1007/s40259023-00633-2.
4. Alzheimer's Association Report. (2024). 2024 Alzheimer's disease facts and figures. Alzheimer's and Dementia, 20(5), 3708-3821. doi: 10.1002/alz.13809.
5. Yakushev, I., Verger, A., Brendel, M., Cecchin, D., Fernandez, P. A., Fraioli, F., Grimmer, T., Tolboom, N., Traub-Weidinger, T., Guedji, E., & Van Weehaeghe, D. (2025). Lecanemab approval in the EU: What should we be ready for? The EANM perspective. Eur J Nucl Mol Imaging, 52(5), 1607-1610. doi: 10.1007/s00259025-07066-9.
6. Breijyeh, Z., & Karaman, R. (2020). Comprehensive review on Alzheimer’s disease: Causes and treatments. Molecules, 25(24), 5789. doi: 10.3390/molecules25245789.
7. Deco, G., & Kringelbach M. L. (2017). Hierarchy of information processing in the brain. Neuron, 94(5), 961-968. doi: 10.1016/j.neuron.2017.03028
8. Von Bartheld, C. S. (2018). Myths and truths about the cellular composition of the human brain: A review of influential concepts. Journal of Chemical Neuroanatomy, 93, 2-15. doi: 10.1016/j.jchemneu.2017.08.004.
9. Dupret, D., Fusi, S., & Panzeri, S. (2026). Neural population activity for memory: Properties, computations, and codes. Neuron, 114(3), 390-407. doi: 10.1016/j. neuron.2025.11.007.
10. Kamatham, P. T., Shukla, R., Khatri, D. K., & Vora, L. K. (2024). Pathogenesis, diagnostics, and therapeutics for Alzheimer's disease: Breaking the memory barrier. Ageing Research Reviews, 101. doi: 10.1016/j. arr.2024.102481.
11. Zhang, J., Zhang, Y., Wang, J., Xia, Y., Zhang, J., & Chen, L. (2024). Recent advances in Alzheimer’s disease: mechanisms, clinical trials and new drug development strategies. Signal Transduction and Targeted Therapy, 9. doi: 10.1038/s41392-024-01911-3.
12. Kametani, F., & Hasegawa, M. (2018). Reconsideration of amyloid hypothesis and tau hypothesis in Alzheimer's disease. Frontiers in Neuroscience, 12. doi: 10.3389/fnins.2018.00025.
13. Lippens, G., & Gigant, B. (2019). Elucidating tau function and dysfunction in the era of cryo-EM. Journal of Biological Chemistry, 294(24), 9316-9325. doi: 10.1074/jbc.REV119.008031.
14. Cáceres, C., Heusser, B., Garnham, A., & Moczko, E. (2023). The major hypotheses of Alzheimer’s disease: related nanotechnology-based approaches for its diagnosis and treatment. Cells, 12(23), 2669. doi: 10.3390/cells12232669.
15. Du, X., Wang, X., & Geng, M. (2018). Alzheimer’s disease hypothesis and related therapies. Translational Neurodegeneration, 7(2). doi: 10.1186/s40035-0180107-y.
16. Bakota, L., & Brandt, R. (2026). Towards mechanism-based tau-targeted therapies. Neural Regeneration Research, 21(2), 687-688. doi: 10.4103/NRR. NRR-D-24-01240.
17. Kierdorf, K., & Prinz, M. (2017). Microglia in steady state. Journal of Clinical Investigation, 127(9), 32013209. doi: 10.1172/JCI90602.
18. Kinney, J.W., Bemiller, S.M., Murtishaw, A.S., Leisgang, A.M., Salazar, A.M. and Lamb, B.T. (2018). Inflammation as a central mechanism in Alzheimer's disease. Alzheimer's & Dementia: Translational Research & Clinical Interventions, 4, 575-590. doi: 10.1016/j. trci.2018.06.014.
19. Sehar, U., Rawat, P., Reddy, A. P., Kopel, J., & Reddy, P. H. (2022). Amyloid beta in aging and Alzheimer’s disease. International Journal of Molecular Sciences, 23(21), 12924. doi: 10.3390/ijms232112924.
20. Takahashi, R.H., Nagao, T. & Gouras, G.K. (2017). Plaque formation and the intraneuronal accumulation of ß-amyloid in Alzheimer's disease. Pathology International, 67, 185-193. doi: 10.1111/pin.12520.
21. Zhang, H., Jiang, X., Ma, L., Wei, W., Zehui, L., Chang, S., Wen, J., Sun, J., & Li, H. (2022). Role of Aß in Alzheimer’s-related synaptic dysfunction. Frontiers in Cell and Developmental Biology, 10. doi: 10.3389/ fcell.2022.964075.
22. Rajendran, L., & Paolicelli, R. C. (2018). Microglia-mediated synapse loss in Alzheimer's disease. Journal of Neuroscience, 38(12), 2911-2919. doi: 10.1523/ JNEUROSCI.1136-17.2017.
23. Tönnies E., & Trushina E. (2017). Oxidative stress, synaptic dysfunction, and Alzheimer’s disease. Journal of Alzheimer’s Disease, 57(4), 1105-1121. doi: 10.3233/ JAD-161088.
24. Yadollahikhales, G., & Rojas, J. C. (2023). Anti-amyloid immunotherapies for Alzheimer's disease: A 2023 clinical update. Neurotherapeutics, 20(4), 914–931. doi: 10.1007/s13311-023-01405-0.
25. Papaliagkas, V. (2025). Anti-amyloid therapies for Alzheimer’s disease: progress, pitfalls, and the path ahead. International Journal of Molecular Sciences, 26(19), 9529. doi: 10.3390/ijms26199529.
26. van Dyck, C. H., Swanson, C. J., Aisen, P., Bateman, R. J., Chen, C., Gee, M., Kanekiyo, M., Li, D., Reyderman, L., Cohen, S., Froelich, L., Katayama, S., Sabbagh, M., Vellas, B., Watson, D., Dhadda, S., Irizarry, M., Kramer, L. D., & Iwatsubo, T. (2022). Lecanemab in Early Alzheimer’s disease. The New England Journal of Medicine, 388, 9-21. doi: 10.1056/NEJMoa2212948.
27. He, Y., Wei, M., Wu, Y., Qin, H., Li, W., Ma, X., Cheng, J., Ren, J., Shen, Y., Chen, Z., Sun, B., Huang, F.-D., Shen, Y., & Zhou, Y.-D. (2019). Amyloid oligomers suppress excitatory transmitter release via presynaptic depletion of phosphatidylinositol-4,5-bisphosphate. Nature Communications, 10, 1193. doi: 10.1038/s41467019-09114-z.
28. Marin, M. A., Ziburkus, J., Jankowsky, J., & Rasband, M. N. (2016). Amyloid- plaques disrupt axon initial segments. Experimental Neurology, 281, 93–98. doi: 10.1016/j.expneurol.2016.04.018.
29. Vitek, G. E., Decourt, B., & Sabbagh, M. N. (2023). Lecanemab (BAN2401): an anti–beta-amyloid monoclonal antibody for the treatment of Alzheimer disease. Expert Opinion on Investigational Drugs, 32(2), 89–94. doi: 10.1080/13543784.2023.2178414.
30. Chowdhury S., & Chowdhury N.S. (2023). Novel Anti-amyloid-beta (Aß) Monoclonal antibody lecanemab for Alzheimer’s disease: A systematic review. International Journal of Immunopathology and Pharmacology, 37. doi: 10.1177/03946320231209839.
31. Aboul-Ella, H., Gohar, A., Ahmed Ali, A., Ismail, L.M., El-Regal Mahmoud, A.E., Elkhatib, W.F., & Aboul-Ella, H. (2024). Monoclonal antibodies: From magic bullet to precision weapon. Molecular Biomedicine, 5(47). doi: 10.1186/s43556-024-00210-1.
32. Barrera-Ocampo, A., & Lopera, F. (2016). Amyloid-beta immunotherapy: The hope for Alzheimer’s disease? Colombia Médica, 47(4), 203-212.
33. Ono, K., & Tsuji, M. (2020). Protofibrils of amyloid-ß are important targets of a disease-modifying approach for Alzheimer’s disease. International Journal of Molecular Sciences, 21(3), 952. doi: 10.3390/ ijms21030952.
34. Berry, D.A., Dhadda, S., Kanekiyo, M., Li, D., Swanson, C.J., Irizarry, M., Kramer, L.D., & Berry, S.M. (2023). Lecanemab for patients with early Alzheimer’s disease: Bayesian analysis of a phase 2b dose-finding randomized clinical trial. JAMA Network Open, 6(4). doi: 10.1001/jamanetworkopen.2023.7230.
35. Swanson, C.J., Zhang, Y., Dhadda, S., Wang, J., Kaplow, J., Lai, R.Y.K., Lannfelt, L., Bradley, H., Rabe, M., Koyama, A., Reyderman, L., Berry, D.A., Berry, S., Gordon, R., Kramer, L.D., & Cummings J.L. (2021). A randomized, double-blind, phase 2b proof-of-concept clinical trial in early Alzheimer’s disease with lecanemab, anti-A protofibril antibody. Alzheimer’s Research & Therapy, 13(80). doi: 10.1186/s13195-02100813-8.
36. Cummings, J., Apostolova, L., Rabinovici, G.D., Atri, A., Aisen, P., Greenberg, S., Hendrix, S., Selkoe, D., Weiner, M., Petersen, R.C., & Salloway, S. (2023). Lecanemab: appropriate use recommendations. The Journal of Prevention of Alzheimer’s Disease, 10(3), 362-377. doi: 10.14283/jpad.2023.30.
37. Corbin, J., & Walker, A.J. (2023). Chapter 75 - FDA overview. Translational Radiation Oncology, 453-457. doi: 10.1016/B978-0-323-88423-5.00019-4.
38. Daval, J.R., Teng, T.W., & Russo, M. (2023). Association of advisory committee votes with US Food and Drug Administration decision making on prescription drugs, 2010-2021. JAMA Health Forum, 4(7). doi: 10.1001/jamahealthforum.2023.1718.
39. Alexander, G.C., Knopman, D.S., Emerson, S.S., Ovbiagele, B., Kryscio, R.J., Perlmutter, J.S., & Kesselheim, A.S. (2021). Revisiting FDA approval of aducanumab. The New England Journal of Medicine, 385(9), 769-771. doi: 10.1056/NEJMp2110468.
40. van Bokhoven, P., de Wilde, A., Vermunt, L., Leferink, P.S., Heetveld, S., Cummings, J., Scheltens, P., & Vijverberg, E.G.B. (2021). The Alzheimer’s disease drug development landscape. Alzheimer’s Research and Therapy, 13(186). doi: 10.1186/s13195-021-00927-z.
41. Gómez-Isla, T., & Frosch, M.P. (2022). Lesions without symptoms: understanding resilience to Alzheimer’s disease neuropathological changes. Nature Reviews Neurology, 18, 323-332. doi: 10.1038/s41582022-00642-9.
42. Espay, A.J., Kepp, K.P., & Herrup K. (2024). Lecanemab and donanemab as therapies for Alzheimer’s disease: An illustrated perspective on the data. eNeuro, 11(7). doi: 10.1523/ENEURO.0319-23.2024.
43. Rajendran, Y., Kondampati, N., Eerike M., Mali, K., & C, L.F. (2024). A longitudinal analysis of black box warnings: Trends and implications for drug safety. Cureus, 16(4). doi: 10.7759/cureus.57597.
44. Doran, S.J., & Sawyer, R.P. (2024). Risk factors in developing amyloid related imaging abnormalities (ARIA) and clinical implications. Frontiers in Neuroscience, 18. doi: 10.3389/fnins.2024.1326784.
45. Xing, X., Zhang, X., Wang, K., Wang, Z., Feng, Y., Li, X., Hua, Y., Zhang, L., & Dong, X. (2025). Post-marketing safety concerns with lecanemab: A pharmacovigilance study based on the FDA adverse event reporting system database. Alzheimer’s Research and Therapy, 17(15). doi: 10.1186/s13195-024-01669-4.
46. Villain, N., Planche, V., Lilamand, M., Cordonnier, C., Soto-Martin, M., Mollion, H., Bombois, S., & Delrieu, J. (2025). Lecanemab for early Alzheimer’s disease: Appropriate use recommendations from the French federation of memory clinics. The Journal of Prevention of Alzheimer’s Disease, 12(4). doi: 10.1016/j. tjpad.2025.100094.
47. Jiang, X., Lv, G., Mendes, M.E., Wu, J., Yuan, J., & Lu, Z.K. (2026). Lifetime cost-effectiveness of Lecanemab for early Alzheimer’s disease. Frontiers in Public Health, 14. doi: 10.3389/fpubh.2026.1692508.
48. Anil, A.A., Krishnan, A.L., Chandra, P., & Unnikrishnan, M.K. (2025). Lecanemab: The advent of biologicals in Alzheimer’s disease, affordability, and clinical relevance. Journal of Applied Pharmaceutical Science, 15(3), 30-38. doi: 10.7324/JAPS.2025.204843.
49. van Veluw, S.J., & Young, M.J. (2024). Ethical considerations for the use of anti-amyloid immunotherapies in patients with early Alzheimer’s disease. Alzheimer’s and Dementia, 20(5), 3664-3665. doi: 10.1002/alz.13795.
50. Albertini, G., Zielonka, M., Cuypers, M., Snellinx, A., Xu, C., Poovathingal, S., Wojno, M., Davie, K., van Lieshout, V., Craessaerts, K., Wolfs, S., Pasciuto, E., Jaspers, T., Horré, K., Serneels, L., Fiers, M., Dewilde, M., & De Strooper, B. (2025). The Alzheimer’s therapeutic Lecanemab attenuates A pathology by inducing an amyloid-clearing program in microglia. Nature Neuroscience, 29, 100-110. doi: 10.1038/ s41593-025-02125-8
51. Behl, C. (2024). In 2024, the amyloid-cascade-hypothesis still remains a working hypothesis, no less but certainly no more. Frontiers in Aging Neuroscience, 16. doi: 10.3389/fnagi.2024.1459224.
52. Largent, E.A., Peterson, A., & Fernandez, H. (2021). FDA drug approval and the ethics of desperation. JAMA Internal Medicine, 181(12), 1555-1556. doi: 10.1001/jamainternmed.2021.6045
53. Livingston, G., Huntley, J., Sommerlad, A., Ames, D., Ballard, C., Banerjee, S., Brayne, C., Burns, A., Cohen-Mansfield, J., Cooper, C., Costafreda, S. G., Dias, A., Fox, N., Gitlin, L. N., Howard, R., Kales, H. C., Kivimaki, M., Larson, E. B., Ogunniyi, A., … & Mukadam, N. (2020). Dementia prevention, intervention, and care: 2020 report of the Lancet Commission. The Lancet, 396(10248), 413-446. doi: 10.1016/S01406736(20)30367-6.
1. Ward, J., Simner, J., Simpson, I., Rae, C., del Rio, M., Eccles, J. A., & Racey, C. (2024). Synesthesia is linked to large and extensive differences in brain structure and function as determined by whole-brain biomarkers derived from the HCP (human connectome project) cortical parcellation approach. Cerebral Cortex, 34(11). doi: 10.1093/cercor/bhae446
2. Ward, J., & Simner, J. (2020). Chapter 13 - Synesthesia: the current state of the field. Multisensory Perception, 283–300. doi: 10.1016/b978-0-12-8124925.00013-9
3. Bianchi, I., Paradis, C., & van de Weijer, J. (2025). Perceptual structure of opposites across sensory modalities. Language and Cognition, 17. doi: 10.1017/ langcog.2025.10016
4. Eckardt, N., Sinke, C., Bleich, S., Lichtinghagen, R., & Zedler, M. (2024). Investigation of the relationship between neuroplasticity and grapheme-color synesthesia. Frontiers in Neuroscience, 18. doi: 10.3389/ fnins.2024.1434309
5. Sun, Y., Yang, H., Zhu, D., Wang, Y., & Suen, R. (2026). Harnessing synesthesia to unlock new pathways in learning for students with disabilities. Learning and Instruction, 102, 102292. doi: 10.1016/j.learninstruc.2025.102292
6. Collerton, D., Barnes, J., Diederich, N. J., Dudley, R., ffytche, D., Friston, K., Goetz, C. G., Goldman, J. G., Jardri, R., Kulisevsky, J., Lewis, S. J. G., Nara, S., O’Callaghan, C., Onofrj, M., Pagonabarraga, J., Parr, T., Shine, J. M., Stebbins, G., Taylor, J.-P., … Weil, R. S. (2023). Understanding visual hallucinations: A new synthesis. Neuroscience & Biobehavioral Reviews, 150, 105208. doi: 10.1016/j.neubiorev.2023.105208
7. Berger, J. J., Harris, I. M., Whittingham, K. M., Terpening, Z., & Watson, J. D. (2019). Substantiating synesthesia: A novel aid in a case of grapheme-colour synesthesia and concomitant dyscalculia. Neurocase, 26(1), 29–35. doi: 10.1080/13554794.2019.1695846
8. Chu, I., Kokash, A., Li, S. P., & Vendrame, M. (2025). Grapheme-color synesthesia in patients with epilepsy: A pilot study. Epilepsy & Behavior, 166, 110378. doi: 10.1016/j.yebeh.2025.110378
9. Tilot, A. K., Kucera, K. S., Vino, A., Asher, J. E., Baron-Cohen, S., & Fisher, S. E. (2018). Rare variants in axonogenesis genes connect three families with sound–color synesthesia. Proceedings of the National Academy of Sciences, 115(12), 3168–3173. doi: 10.1073/pnas.1715492115
10. Hillary, F. G., & Grafman, J. H. (2017). Injured brains and adaptive networks: The benefits and costs of hyperconnectivity. Trends in Cognitive Sciences, 21(5), 385–401. doi: 10.1016/j.tics.2017.03.003
11. Brauchli, C., Elmer, S., Rogenmoser, L., Burkhard, A., & Jäncke, L. (2017). Top–down signal transmission and global hyperconnectivity in auditory-visual synesthesia: Evidence from a functional EEG resting-state study. Human Brain Mapping, 39(1), 522–531. doi: 10.1002/hbm.23861
12. Mueller, K., Arelin, K., Möller, H. E., Sacher, J., Kratzsch, J., Luck, T., Riedel-Heller, S., Villringer, A., & Schroeter, M. L. (2016). Serum BDNF correlates with connectivity in the (pre)motor hub in the aging human brain—a resting-state fmri pilot study. Neurobiology of Aging, 38, 181–187. doi: 10.1016/j.neurobiolaging.2015.11.003
13. Hauw, F., Béranger, B., & Cohen, L. (2024). Subtitled speech: The neural mechanisms of ticker-tape synaesthesia. Brain, 147(7), 2530–2541. doi: 10.1093/ brain/awae114
14. Coslett, B. H., & Schwartz, F. M. (2018). Chapter 18 - The parietal lobe and language. (2018). Handbook of Clinical Neurology, 365–375. doi: 10.1016/b978-0444-63622-5.00018-8
15. Lalwani, P., & Brang, D. (2019). Stochastic resonance model of synaesthesia. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1787), 20190029. doi: 10.1098/rstb.2019.0029
16. Yeo, B. B., & Yu, J. (2025). Atypical cortical regional homogeneity observed in grapheme-colour synaesthesia and its subtypes. Cortex, 192, 135–151. doi: 10.1016/j.cortex.2025.09.003
17. Bouvet, L., Amsellem, F., Maruani, A., Tonus-Vic Dupont, A., Mathieu, A., Bourgeron, T., Delorme, R., & Mottron, L. (2019). Synesthesia & autistic features in a large family: Evidence for spatial imagery as a common factor. Behavioural Brain Research, 362, 266–272. doi: 10.1016/j.bbr.2019.01.014
18. Ward, J., & Filiz, G. (2020). Synaesthesia is linked to a distinctive and heritable cognitive profile. Cortex, 126, 134–140. doi: 10.1016/j.cortex.2020.01.002
19. Speed, L. J., Croijmans, I., Dolscheid, S., & Majid, A. (2021). Crossmodal associations with olfactory, auditory, and tactile stimuli in children and adults. I-Perception, 12(6). doi: 10.1177/20416695211048513
20. Yong, Z., Hsieh, PJ. & Milea, D. Seeing the sound after visual loss: functional MRI in acquired auditory-visual synesthesia. Exp Brain Res 235, 415–420 (2017). doi: 10.1007/s00221-016-4802-6
21. Sakai, J. (2020). How synaptic pruning shapes neural wiring during development and, possibly, in disease. Proceedings of the National Academy of Sciences, 117(28), 16096–16099. doi: 10.1073/pnas.2010281117
22. Schwartzman, D. J., Bor, D., Rothen, N., & Seth, A. K. (2019). Neurophenomenology of induced and natural synaesthesia. Philosophical Transactions of the Royal Society B: Biological Sciences, 374(1787), 20190030. doi: 10.1098/rstb.2019.0030
23. Anash, S., & Boileau, A. (2024). Grapheme-color synesthesia and its connection to memory. Cureus. doi: 10.7759/cureus.67524
24. Choi, I., & Lee, S.-H. (2025). Locomotion-dependent auditory gating to the parietal cortex guides multisensory decisions. Nature Communications, 16(1). doi: 10.1038/s41467-025-57347-y
25. Newell, F. N., & Mitchell, K. J. (2016). Multisensory integration and cross-modal learning in synaesthesia: A unifying model. Neuropsychologia, 88, 140–150. doi: 10.1016/j.neuropsychologia.2015.07.026
26. Delsanti, R., Hauw, F., Lahbari, R., Bouhali, F., & Cohen, L. (2025). Bridging speech and Sight: White matter anatomy in ticker-tape synaesthesia. Brain Communications, 7(5). doi: 10.1093/braincomms/fcaf316
27. Hauw, F., El Soudany, M., Rosso, C., Daunizeau, J., & Cohen, L. (2023). A single case neuroimaging study of tickertape synesthesia. Scientific Reports, 13(1). doi: 10.1038/s41598-023-39276-2
28. Hauw, F., El Soudany, M., & Cohen, L. (2023). The advantage of being a synesthete: The behavioral benefits of ticker-tape synesthesia. Cortex, 168, 226–234. doi: 10.1016/j.cortex.2023.08.011
29. Chun, C. A., & Hupé, J. (2015). Are synesthetes exceptional beyond their synesthetic associations? A systematic comparison of creativity, personality, cognition, and mental imagery in synesthetes and controls. British Journal of Psychology, 107(3), 397–418. doi: 10.1111/bjop.12146
30. Merter, S. (2017). Synesthetic approach in the design process for enhanced creativity and multisensory experiences. The Design Journal, 20(sup1), S4519-S4528. doi: 10.1080/14606925.2017.1352948
31. Liu, Q., & Lupyan, G. (2026). The unity of sense and mind: A review of cross-domain mapping. Psychonomic Bulletin & Review, 33(1). doi: 10.3758/s13423025-02805-3
32. Poerio, G. L., Ueda, M., & Kondo, H. M. (2022). Similar but different: High prevalence of synesthesia in autonomous sensory meridian response (ASMR). Frontiers in Psychology, 13. doi: 10.3389/ fpsyg.2022.990565
33. Hughes, J. E. A., Simner, J., Baron-Cohen, S., Treffert, D. A., & Ward, J. (2017). Is synaesthesia more prevalent in autism spectrum conditions? Only where there is prodigious talent. Multisensory Research, 30(3–5), 391–408. doi: 10.1163/22134808-00002558
34. van Leeuwen, T. M., Neufeld, J., Hughes, J., & Ward, J. (2020). Synaesthesia and autism: Different developmental outcomes from overlapping mechanisms? Cognitive Neuropsychology, 37(7–8), 433–449. doi: 10.1080/02643294.2020.1808455
35. Limon, A., & Corona-Moreno, M. (2025). Hyperconnectivity and disrupted signal-to-noise processing in autism. The Palgrave Encyclopedia of Disability, 1–9. doi: 10.1007/978-3-031-40858-8_179-1
36. van Leeuwen, T. M., Wilsson, L., Norrman, H. N., Dingemanse, M., Bölte, S., & Neufeld, J. (2021). Perceptual processing links autism and synesthesia: A co-twin control study. Cortex, 145, 236–249. doi: 10.1016/j. cortex.2021.09.016
37. Rudziński, G., Pożarowska, K., Brzuszkiewicz, K., & Soroka, E. (2024). An outline of savant syndrome. Psychiatria Polska, 58(4), 681–691. doi: 10.12740/pp/ onlinefirst/157104