






































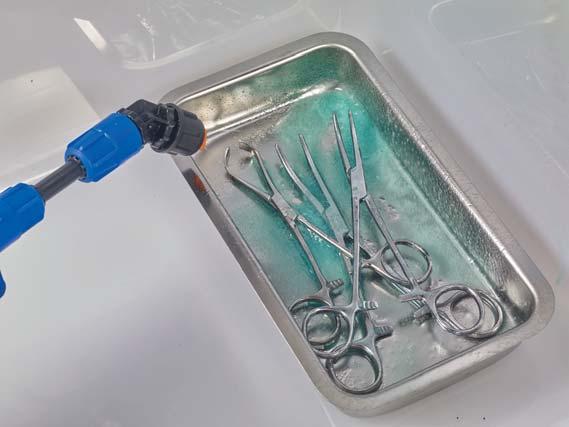
for use with Ruhof’s Prepzyme® Forever Wet Enzymatic Pre-Cleaner
The cleaning process begins at the point of use. When soils dry, they become harder to remove and increase the risk of corrosion, biofilm formation, and HAI’s. The Forever Wet Auto Sprayer delivers a consistent application of Prepzyme® Forever Wet in less than half the time—keeping instruments moist for up to 72 hours.


















FEBRUARY/MARCH 2026
Sourcing & Logistics
7 > Logistics Giants Accelerate Push into Healthcare
DANIEL BEAIRD
Infection Prevention
10 > Flu Season Sounds Alarm Bells in the Northern Hemisphere
MATT MACKENZIE
Sterile Processing
15 > OR/SPD Power Duos: Inside Two Children’s Hospitals Redefining Surgical Collaboration
KARA NADEAU
22 > Understanding the Key Features and Benefits of Mechanical Cleaning Tools: Ultrasonic Baths, Water Guns, and Washer-Disinfectors
WILLIAM LEIVA
28 > Tools of the Trade
ADAM OKADA
Departments
5 > Innovation trek
6 > What’s on the Web, Advertiser Index


Febuary/March 2026, Vol. 50, No. 2
Group Content Director Healthcare
Jennifer Breedlove
jbreedlove@endeavorb2b.com
Editor-in-Chief
Daniel Beaird
dbeaird@endeavorb2b.com
Associate Editor
Matt MacKenzie
mmackenzie@endeavorb2b.com
Senior Contributing Editor
Kara Nadeau
knadeau@hpnonline.com
ADVERTISING SALES
Director of Sales Healthcare
Jennifer Hazen
jhazen@endeavorb2b.com | 330-598-0308
East & West Coast
Kristen Hoffman
khoffman@endeavorb2b.com | 603-891-9122
Midwest & Central
Deborah Baron
dbaron@endeavorb2b.com | 917-763-7275
Advertising & Art Production
Production Manager | Ed Bartlett
Art Director | Kelli Mylchreest
Advertising Services
Karen Runion | krunion@endeavorb2b.com
Audience Development
Laura Moulton | lmoulton@endeavorb2b.com
Endeavor Business Media, LLC
CEO Chris Ferrell
COO Patrick Rains
CDO Jacquie Niemiec
CALO Tracy Kane
CMO Amanda Landsaw
EVP Infrastructure & Public Sector Group
Kylie Hirko
VP of Content Strategy, Infrastructure & Public Sector Group
Michelle Kopier
Healthcare Purchasing News USPS Permit 362710, ISSN 1098-3716 print, ISSN 2771-6716 online is published 7 times annually Feb/Mar, Apr/May, Jun/ Jul, Aug/Sep, Oct, Nov/Dec, Nov/Dec IBG, by Endeavor Business Media, LLC. 201 N Main St 5th Floor, Fort Atkinson, WI 53538. Periodicals postage paid at Fort Atkinson, WI, and additional mailing offices. POSTMASTER: Send address changes to Healthcare Purchasing News, PO Box 3257, Northbrook, IL 600653257. SUBSCRIPTIONS: Publisher reserves the right to reject non-qualified subscriptions. Subscription prices: U.S. $164.80 per year; Canada/Mexico $199.56 per year; All other countries $284.54 per year. All subscriptions are payable in U.S. funds. Send subscription inquiries to Healthcare Purchasing News, PO Box 3257, Northbrook, IL 60065-3257. Customer service can be reached toll-free at 877-382-9187 or at HPN@omeda.com for magazine subscription assistance or questions.
Printed in the USA. Copyright 2026 Endeavor Business Media, LLC. All rights reserved. No part of this publication should be reproduced or transmitted in any form or by any means, electronic or mechanical, including photocopies, recordings, or any information storage or retrieval system without permission from the publisher. Endeavor Business Media, LLC does not assume and hereby disclaims any liability to any person or company for any loss or damage caused by errors or omissions in the material herein, regardless of whether such errors result from negligence, accident, or any other cause whatsoever. The views and opinions in the articles herein are not to be taken as official expressions of the publishers, unless so stated. The publishers do not warrant either expressly or by implication, the factual accuracy of the articles herein, nor do they so warrant any views or opinions by the authors of said articles.
BY DANIEL BEAIRD
Healthcare organizations are facing a convergence of pressures across supply chains, clinical operations, and workforce capacity. From global logistics disruptions and increasingly complex therapies to persistent infection risks and seasonal surges, leaders are being forced to rethink resilience at every level of care delivery.
Large logistics providers accelerated their push into healthcare in 2025, responding to growing demand for temperature-controlled, time-critical, and highly regulated supply chain services. Ongoing vulnerabilities in healthcare logistics pushed hospitals, pharmaceutical manufacturers, and medical device companies to prioritize resilience, compliance, and end-to-end visibility, which are areas where global carriers hold significant scale advantages. UPS, DHL, and FedEx expanded healthcare as a core growth vertical, investing heavily in cold chain infrastructure, specialized facilities, digital tracking, and regulatory expertise to support fast-growing segments such as biologics, cell and gene therapies, specialty pharmaceuticals, and high-value medical devices.
Contributor Kara Nadeau explored how leaders at Ann & Robert H. Lurie Children’s Hospital and Nemours Children’s Hospital are improving patient safety and reliability through intentional collaboration between the operating room and sterile processing department. Rather than engaging only when issues arise, both
Connectwithus:

organizations established structured leadership partnerships spanning perioperative services and SPD, aligning teams around shared accountability and reinforcing sterile processing as foundational to safe surgery.
At Lurie Children’s, collaboration is embedded into daily practice through joint rounding, cross-department shadowing, and standardized reviews of upcoming cases to proactively address instrumentation and scheduling risks. These efforts have virtually eliminated immediate use steam sterilization and rapid cycles while delivering measurable cost savings. At Nemours, leaders rebuilt a once-strained OR/SPD relationship through mirrored leadership roles, daily huddles, a technician-led tray quality committee, and data-driven tracking of bioburden events, supported by strong education and recognition programs. Together, these approaches demonstrate how aligned leadership, routine collaboration, frontline engagement, and constructive use of data can drive meaningful gains in safety, quality, and culture.
Editor Matt MacKenzie reported that health officials were already on alert following an unusually severe and prolonged flu season in the Southern Hemisphere, often viewed as a predictor for the U.S. and Europe. Hospitals now face mounting clinical, operational, and supply chain pressures, underscoring the need for proactive planning, resilient supply chains, and adequate inventories of diagnostics and PPE.
Jimmy Chung, MD, MBA, FACS, FABQAURP, CMRP, Chief Medical Officer, Advantus Health Partners and Bon Secours Mercy Health, Cincinnati, OH
Joe Colonna, Chief Supply Chain and Project Management Officer, Piedmont Healthcare, Atlanta, GA; Karen Conway, Vice President, Healthcare Value, GHX, Louisville, CO
Dee Donatelli, RN, BSN, MBA, Senior Director Spend symplr and Principal Dee Donatelli Consulting LLC, Austin, TX
J. Hudson Garrett Jr., PhD, FNAP, FSHEA, FIDSA, Adjunct Assistant Professor of Medicine, Infectious Diseases, University of Louisville School of Medicine
Melanie Miller, RN, CVAHP, CNOR, CSPDM, Value Analysis Consultant, Healthcare Value Management Experts Inc. (HVME) Los Angeles, CA
Dennis Orthman, Consulting, Braintree, MA
Janet Pate, Nurse Consultant and Educator, Ruhof Corp.
Richard Perrin, CEO, Active Innovations LLC, Annapolis, MD
Jean Sargent, CMRP, FAHRMM, FCS, Principal, Sargent Healthcare Strategies, Port Charlotte, FL
Richard W. Schule, MBA, BS, FAST, CST, FCS, CRCST, CHMMC, CIS, CHL, AGTS, Senior Director Enterprise Reprocessing, Cleveland Clinic, Cleveland, OH
Barbara Strain, MA, CVAHP, Principal, Barbara Strain Consulting LLC, Charlottesville, VA
Deborah Petretich
Templeton, RPh, MHA,Chief Administrative Officer (Ret.), System Support Services, Geisinger Health, Danville, PA
Ray Taurasi, Principal, Healthcare CS Solutions, Washington, DC


Rates Continue to Rise as 2026 Begins
Pertussis cases are rising, and a spike in Arkansas is driving case numbers up across the nation. There were over 500 cases reported in 2025, which marks the “highest annual total since the state began collecting data on pertussis 15 years ago.” Children and adolescents make up the majority of the cases in the state, with infants younger than 1 year old accounting for over 60% of hospitalizations.
Read on: hpnonline.com/55341103
to Acquire Major
in GHX for AIDriven Healthcare Supply Chain Innovation
By digitizing and automating complex healthcare supply chain processes, GHX aims to reduce inefficiencies and help organizations be er leverage their data. Increased investment in AI is expected to enhance workflow simplification, actionable insights, automated decision-making, and ultimately, care delivery.
Read on: hpnonline.com/55342465
The AAP and more than 200 health groups “sent a le er to Congress urging lawmakers ‘to conduct swi and robust oversight
regarding the abrupt changes to the U.S. childhood vaccine schedule.’” Additionally, health officials across the country have banded together to “protect vaccine access and provide evidence-based health information to their residents.” Legislation has also been passed in some states aiming to protect access to vaccines.
Read on: hpnonline.com/55343188
The team “employed a novel intervention to prevent the blockage of a vital coronary artery, which is a very rare but o en lethal complication following a heart-valve replacement.” This suggests “a less traumatic alternative to open-heart surgery could become widely available for those at risk of coronary artery obstruction.”
Read on: hpnonline.com/55341679
States have been required to report the “percentage of patients covered by Medicaid and the Children’s Health Insurance Plan who are immunized” before now. Around 40% of children are covered by Medicaid, and CHIP “provides health insurance to children whose families do not qualify for Medicaid.”
Read on: hpnonline.com/55341293
Major carriers are expanding healthcare logistics capabilities to support temperature-sensitive products, specialty pharmaceuticals, and time-critical delivery.
BY DANIEL BEAIRD
Large logistics companies made a decisive push into healthcare in 2025.
Industry leaders UPS, DHL, and FedEx all expanded aggressively, drawn by healthcare logistics’ growth potential, customer stickiness, and higher margins. As hospitals, pharmaceutical manufacturers, and medical device companies rethink how products move from factory to point of care, healthcare logistics has shifted from a back-end function to a strategic pillar supporting supply chain resilience, cost control, and regulatory compliance. This evolution is being driven by a convergence of structural pressures reshaping the healthcare supply chain.
The pandemic exposed the fragility of healthcare supply chains, from PPE shortages to limited visibility into inventory levels and supplier capacity. In response, healthcare organizations have prioritized redundancy, traceability, and speed. Those are capabilities that large logistics providers are uniquely positioned to deliver at scale. To meet this demand, logistics companies have invested heavily in dedicated healthcare infrastructure, including temperature-controlled facilities, specialized handling processes, and technology
platforms designed to support regulated products.
Logistics giants UPS, DHL, FedEx make recent commitments
UPS has made healthcare a core strategic priority, investing heavily in cold chain infrastructure, precision delivery, and regulated logistics tailored to pharmaceuticals and biologics. In November 2025, it acquired Canada-based Andlauer Healthcare Group (AHG) for $1.6 billion to expand its temperature-controlled
healthcare transportation and distribution capabilities. AHG’s specialized cold chain network is expected to reduce transit times while enhancing end-to-end visibility, global reach, and quality assurance across UPS Healthcare operations.
Kate Gutmann, EVP and president of international, healthcare, and supply chain solutions for UPS, said Andlauer shares UPS’s focus on quality, precision, and speed. Michael Andlauer, founder and CEO of AHG, now leads UPS Canada Healthcare and AHG, with a mandate to expand specialized capabilities and support the distribution of high-value, temperature-sensitive therapies.
Earlier acquisitions of German cold chain logistics firms Frigo-Trans and BPL added European warehousing and time-critical freight forwarding capabilities, expanding UPS’s end-toend temperature-controlled services across key pharmaceutical markets. These investments reflect broader industry dynamics. Service quality,

real-time visibility, and regulatory compliance command premium pricing in healthcare logistics. Growth areas such as biologics, cell and gene therapies, specialty pharmaceuticals, and implantable devices require precise temperature control, tight delivery windows, and secure handling. Those are requirements that favor logistics providers with advanced technology, automation, and compliance expertise.
DHL has emerged as one of the most established global players in healthcare logistics, operating one of the largest dedicated networks for life sciences and healthcare products. Its offerings include GDP-compliant cold chain services, clinical trial logistics, and real-time monitoring across facilities worldwide. DHL’s global footprint and regulatory expertise have made it a preferred partner for multinational pharmaceutical and medical technology companies.
In September 2025, DHL acquired SDS Rx, a specialized provider of final-mile delivery and healthcare transportation services supporting long-term care and specialty pharmacies, radiopharmacies, and health system networks. SDS Rx operates more than 200 locations across the U.S. and specializes in same-day and expedited delivery services designed for the urgency and precision of clinical environments.
“The life sciences and healthcare sector is projected to grow at a compound annual growth rate of 11% through 2030,” said Mark Kunar, CEO of DHL Supply Chain North America, at the time of the acquisition. “Specialty pharmacy already accounts for approximately 50% of total prescription drug spending in the U.S., and the number of patients served by specialty pharmacies grew by 12% between 2018 and 2022.”
By integrating SDS Rx into its Life Sciences & Healthcare portfolio, DHL is extending its end-to-end supply
chain capabilities all the way to the point of care. The move aligns with DHL’s broader Strategy 2030 healthcare initiative and reflects growing recognition that final-mile delivery is a critical link between supply chain performance and patient outcomes.
Kunar said rising demand for specialty pharmaceutical and healthcare solutions presents significant opportunities for DHL to leverage its scale, expertise, and operational discipline.
“We are expanding our healthcare logistics capabilities, attracting a new segment of healthcare customers, and reinforcing our position as a trusted partner in building resilient and connected healthcare supply chains,” he said.
Drew Kronick, founder, CEO, and managing partner of SDS Rx, noted that demand for healthcare finalmile delivery continues to grow, and DHL’s acquisition is aimed at improving delivery precision, reliability, and speed for patients with the most urgent needs.
DHL also acquired CryoPDP in 2025, a specialty courier focused on clinical trials, biopharmaceuticals, and gene therapies. Together, these acquisitions strengthen DHL’s capabilities across critical segments of the healthcare supply chain, spanning pharmaceuticals, consumer health, animal health, and medical technology under the DHL Health Logistics umbrella.
FedEx is also expanding its healthcare footprint, with a focus on technology, compliance, and speed to support complex healthcare distribution requirements. The company has added specialized healthcare logistics services, including advanced tracking, value-added fulfillment, and cold chain solutions, while investing in dedicated life science centers designed to handle temperature-sensitive shipments such as biologics, vaccines, clinical samples, and investigational medicinal products.
FedEx operates life science centers in markets including Korea, Singapore, Japan, India, the U.S., and the Netherlands. It has also introduced healthcare-focused air cargo services, such as a direct route between Dublin, Ireland, and Indianapolis, to reduce transit times for pharmaceutical and other high-value healthcare products. These services integrate priority handling, real-time digital visibility, and intervention capabilities to maintain temperature and condition control throughout transport.
FedEx has earned IATA CEIV Pharma certification across multiple hubs and ground-handling operations, demonstrating compliance with stringent global standards for pharmaceutical logistics and providing healthcare customers with assurance around product safety and integrity.
Strategy 2030 and beyond DHL’s “Strategy 2030 – Accelerate Sustainable Growth” outlines its long-term plan to expand its logistics footprint by focusing on high-growth sectors and embedding sustainability into its core operations. Healthcare is a central pillar of this strategy, driven by growth rates that exceed the broader economy and by logistics requirements that extend beyond traditional freight or parcel delivery.
“We are ideally positioned to seize growth opportunities in a rapidly changing world thanks to our wellbalanced, diversified portfolio,” DHL Group CEO Tobias Meyer said in a statement. “We will fortify our market-leading position in global logistics with a strong focus on quality and on servicing the needs of fast-growing industry segments.”
DHL previously relied on its Strategy 2025 roadmap to navigate global disruptions, including the pandemic, supply chain volatility, and geopolitical tensions. Under Strategy 2030, the company aims
to grow revenue by 50% by 2030 through divisional and group-wide growth initiatives.
Recent FedEx earnings commentary shows healthcare emerging as a priority industry contributing to B2B revenue growth, alongside automotive and aerospace. FedEx is positioning healthcare as a core vertical within its logistics portfolio, recognizing that cold chain and time-critical healthcare shipping are high-value, technically demanding, and expected to grow rapidly.
As part of this focus, FedEx appointed Nick Gennari as president of global healthcare in 2024. In November 2025, Gennari also joined the board of GXP-Storage, a life sciences company specializing in compliant, visible, and scalable material management, signaling an effort to integrate storage and logistics for full lifecycle visibility.
“The life sciences industry has long treated storage and logistics as separate functions,” said Jeff Johnson, founder and CEO of GXP-Storage. “Nick’s appointment represents our commitment to uniting these worlds. By bridging the gap between ‘at rest’ and ‘in transit,’ we’re delivering the visibility, quality, and regulatory control our clients need across the full material lifecycle.”
UPS’s healthcare strategy similarly combines infrastructure expansion, targeted acquisitions, and technology investment. UPS Healthcare is rapidly scaling its global network, expanding its geographic footprint, enhancing cold chain and specialty capabilities, and embedding itself more deeply into life sciences and healthcare supply chains, positioning healthcare as a long-term growth engine beyond traditional parcel delivery.
While global integrators dominate large-scale healthcare distribution, a growing share of innovation and capacity expansion is coming from specialized and regional thirdparty logistics providers. These companies are targeting specific gaps in the healthcare supply chain, particularly where precision, customization, and regulatory expertise outweigh sheer network size.
Mid-tier 3PLs are differentiating by building deep capabilities in narrowly defined healthcare segments rather than broad, multi-industry networks. GXO Logistics’ acquisition of ColdChain Solutions, for example, strengthened its ability to support pharmaceutical manufacturers with temperature-controlled storage and handling across critical ranges.
Similarly, EVERSANA has expanded distribution infrastructure designed specifically for life sciences customers, integrating automation, robotics, and AI-driven fulfillment to support high-touch pharma commercialization models. Its focus extends beyond warehousing to services such as product launch support, specialty distribution, and patientcentric fulfillment.

39239738 © Wellesenterprises | Dreamstime.com
Regional providers such as Noatum Logistics are emphasizing digital platforms and compliance-ready processes to manage the complexity of life sciences shipments, particularly in international and clinical trial environments. Capabilities such as real-time temperature monitoring, serialization, chain-of-custody documentation, and audit readiness are increasingly table stakes for healthcare customers, and areas where specialized 3PLs can move faster than larger organizations.
These providers are also more willing to tailor systems and workflows to individual customer requirements, an approach that resonates with biotech firms and emerging pharma companies operating smaller but highly regulated supply chains.
Many specialized 3PLs are positioning themselves around high-growth segments that require bespoke logistics models like:
• Clinical trial logistics and investigational product distribution.
• Cell and gene therapy supply chains with ultra-low temperature requirements.
• Specialty pharmacy and direct-to-patient delivery.
• High-value medical devices and implantable products. In these areas, execution risk is high and tolerance for error is low, favoring providers with focused expertise and dedicated healthcare operations.
The expansion of specialized and regional 3PLs signals a shift toward a more segmented healthcare logistics market. Rather than relying solely on large, integrated carriers, healthcare manufacturers and providers are increasingly assembling hybrid networks, pairing global reach with niche partners that offer precision, flexibility, and deep regulatory knowledge.
For healthcare supply chains facing growing product complexity and cost pressure, these providers offer an alternative model: smaller scale, but purpose-built for the demands of modern healthcare logistics. HPN

BY MATT MACKENZIE
The Southern Hemisphere’s experience with flu, coming as it does directly before the Northern Hemisphere’s season, can act as a bellwether as to what the season in the U.S. and Europe will be like. This time, it didn’t look promising – a WHO report tracking the global situation regarding seasonal influenza stated that “in the Southern Hemisphere, some countries have had unusually long seasons compared to previous years, with virus activity remaining higher than usual in recent months.”1
One of the culprits of this increase in activity was a rise in detections of subclade K of influenza A(H3N2). This was the driver behind high levels in Australia and New Zealand in August 2025. A CIDRAP article in early January reported that “seasonal flu activity remains high across the [U.S.], and elevated levels are expected to continue for several more weeks. Hospital admissions for the flu rose this week, with 39,945 admitted, up from 33,301 patients last week. Eight flu-related pediatric deaths were reported this week, compared with one
last week, including Louisiana’s first child flu death of 2026, for a U.S. season total of 17. Today’s FluView report reflects eight new flu-related deaths in children nationwide, all in midDecember to early January. All were attributed to influenza A. Of the six deaths that had subtyping performed, all were caused by the H3N2 strain.”
Of course, flu is not the only respiratory illness to worry about. The same CIDRAP article pointed out that RSV levels are elevated across several regions, and while COVID activity “remains low at a national level,” activity is increasing.2
Hospitals and health systems face increased pressure during these months as more patients come through the doors. It places additional strain everywhere, from clinicians to supply chain to patients themselves. HPN spoke with Bridget Stovall, MSN, RN, from Cardinal Health to hear an expert’s opinion on how hospitals can brace themselves for what may prove to be a lengthy season with a late peak.
What lessons did clinicians learn during last year’s respiratory illness season?
Stovall: Every respiratory season is a reminder of how vital it is to be prepared for potential surges of sick patients. This time of year can get very chaotic. If a facility is caught off guard during a surge, the negative impacts can be significant. Patients may experience substantial delays in treatment, even while providers are working around the clock.
proactively collaborate with trusted distributors.

Each hospital department plays a critical role in supporting positive outcomes for patients. From intake, to diagnostics, to protecting the facility’s most vulnerable patients, it’s important that every function is prepared to handle what a surge can bring. With thoughtful planning and the right medical products and solutions, facilities can stay nimble and maintain strong momentum throughout the entire season.
Are there any supply chain vulnerabilities that concern during this respiratory illness?
Stovall: Healthcare provider demands for personal protective equipment (PPE) can surge during peaks, potentially leading to supply shortages or delays in care. For example, while working in a hospital during the height of the pandemic, I had to use the same N95 face mask for several days straight due to supply concerns.
The uncertainty surrounding access to necessary PPE like face masks is something I hope clinicians never experience again. Those events underscored the critical importance of preparedness and resilient supply chains. Today, healthcare systems are leveraging lessons learned from the pandemic to strengthen inventory management, diversify sourcing, and
To help maintain stable inventory levels, hospitals and healthcare facilities can work with distributors to leverage historical data and collaborate with supply chain teams to establish flexible ordering strategies. This shifts the burden away from providers. According to the Health Industry Distributors Association (HIDA), providers who work with a distributor gain 30% more time and resources to devote to patient care. By prioritizing proactive planning and collaboration, healthcare facilities can ensure that frontline teams will have the protection they need to deliver safe, uninterrupted care, even during peak season.
How can clinicians prepare for a potentially prolonged respiratory illness season?
Stovall: Every department within a hospital or healthcare facility should maintain sufficient equipment to support patient care throughout the respiratory illness season and establish contingency plans to address unexpected supply shortages. These measures can help clinicians stay focused on delivering safe, effective treatment throughout a prolonged season without the added stress of resource uncertainty.
A few examples:
• The lab within a hospital or healthcare facility is a vital department, as it works with clinicians to support diagnostics and monitor patient illnesses. An unexpected surge in demand can lead to limited supply of critical products like diagnostic tests, backlogs in testing and longer wait times for results. Delayed diagnostic results can be extremely frustrating for clinicians and patients. In the absence of a timely diagnosis, providers cannot
make informed treatment decisions, potentially leading to negative patient outcomes. Ensuring the facility is adequately stocked with critical diagnostic products can go a long way in supporting better patient outcomes and promoting lab efficiency. Rapid test kits help provide quick results, as they can be read by a clinician without having to go through a separate clinical lab. This speeds up diagnosis and treatment, particularly when conducted at the point of care, meaning at the patient’s bedside or where treatment is provided.
•Proper PPE for clinicians is essential for reducing flu transmission and the spread of germs. Healthcare facilities may consider prioritizing the supply and proper use of a variety of PPE products, including:
•N95 Respirators
•Procedural masks
•Exam gloves
•Infection control apparel including gowns, coveralls, footwear and more
•Face shields and eye protection
How can clinicians advise their patients for avoiding respiratory illness and what patients can do when they’re sick?
Stovall: Educating patients is a simple, yet effective way to help prevent respiratory illness. Providers can share seasonal reminders with patients, emphasizing critical habits such as covering coughs and sneezes, washing hands correctly and often, and staying home and away from others when sick.
Facilities and clinicians can further support these efforts by providing masks and hand sanitizer and cleaning frequently touched surfaces like counters, handrails and doorknobs to protect both patients and staff. Additionally, the Centers for Disease Control and Prevention (CDC) offers free, printable educational posters
that can be displayed in highly visible areas. Overall, creating and promoting a healthy environment for patients, visitors, and providers can help protect everyone in high-risk environments.
Sick patients should avoid others, prioritize rest and drink plenty of fluids. If symptoms persist, they should consider calling a healthcare provider or visiting urgent care. CDC recommends that sick patients should only go to the emergency room if they are experiencing severe symptoms or if
they are in a high-risk group, which includes young children, people 65 and older, pregnant women, and people with certain medical conditions. This will allow hospitals to better prioritize patients during a very busy time of year.
What can be done to mitigate burnout among clinicians as the season wears on?
Stovall: This time of year can take a toll on clinicians both mentally and
physically. It can often feel like a revolving door with so many patients. As soon as one is discharged, another is ready to be seen. Long hours, increased demand and critical decision-making can significantly add to workplace stress.
Clinicians should keep self-care top-of-mind during this busy time. When not working, it is helpful to de-stress and make time for enjoyable activities with family or friends. The American Nurse Association (ANA) suggests that periodically
BY DANIEL BEAIRD
As cold and flu season ramps up, healthcare facilities are entering one of the most operationally challenging periods of the year. Staffing shortages, rising costs, and increased scrutiny tied to infection-related readmissions are colliding with the continued threat of emerging pathogens. In hospitals and long-term care se ings, even minor lapses in hand hygiene can have an outsized effect on patient safety, staff well-being, and financial performance.
HPN recently spoke with Deborah Chung, regional marketing manager for healthcare at Essity, about how healthcare leaders can be er prepare their facilities for seasonal surges.
According to Chung, the pressures facing healthcare organizations during cold and flu season are intensifying. “Many facilities are dealing with staffing shortages, rising operational costs, and now stricter penalties tied to infection-related admissions,” she said. “In se ings like hospitals and long-term care facilities, even a small lapse in hygiene can create a trickle effect that impacts workflow and financial performance.”
Preparation, she noted, starts with a proactive approach to hygiene, one that enables organizations to operate more efficiently while protecting patients and staff. Nearly half of healthcare-associated infections (HAIs) are linked to poor hand hygiene, making it a critical area of focus. High-capacity dispensers, standardized placement, and science-based training programs can improve compliance and reduce unnecessary strain on staff.
Technology is also playing an increasingly important role. Chung pointed to growing adoption of digital tools, including AI-enabled and connected systems, that help address workforce gaps. “Eighty-eight percent of healthcare managers agree that investing in digital solutions can help offset workforce chal-
lenges,” she said. Digital training platforms designed for frontline environmental services teams can reinforce best practices while supporting consistency across shi s and departments.
Training, Chung emphasized, must go beyond one-time education. Engaging, ongoing training is essential for driving lasting behavior change. She recommends keeping content visual and concise, connecting hygiene practices to real-world outcomes, and incorporating hands-on learning with the products staff use every day. “When teams understand how their actions protect vulnerable patients, hygiene becomes a shared responsibility,” she said.
Another o en-overlooked consideration is inclusive hygiene, particularly in restrooms across healthcare facilities. Chung explained that inclusive hygiene goes beyond regulatory compliance to address usability, comfort, and dignity for a wide range of users. “Over half of visitors encounter some kind of challenge in public restrooms,” she said, citing issues such as loud hand dryers or dispensers that are difficult to use for people with limited dexterity or sensory sensitivities.
Inclusive hygiene strategies encourage healthcare organizations to ask broader questions: Does the space feel safe and welcoming? Is it easy to navigate? Does it support privacy and comfort? Chung pointed to inclusive hygiene playbooks, informed by feedback from users, cleaners, and facility managers, as a practical way to make incremental improvements that benefit everyone.
Ultimately, Chung said, preparing for cold and flu season is about building a resilient foundation. When hygiene is intuitive and accessible, staff can spend more time on patient care, patients feel safer, and facilities are be er positioned to weather the busiest, and riskiest, months of the year.
disconnecting from work when possible is important for preventing burnout. Engaging in pleasant experiences outside of work gives clinicians a chance to relax and recharge physical and emotional reserves.
According to the ANA, emotional support is also essential to manage the stress of workplace demands and the mental load of patient care. Establishing a strong support system and maintaining open communication with colleagues can be invaluable.
The Occupational Safety and Health Administration (OSHA) recommends clinicians prioritize their own physical health by eating nutritiously, resting as much as possible and exercising. Focusing on good habits and stress management can help clinicians stay healthy and ready to give their best during respiratory season.
Generally, what should supply chain leaders be thinking about to weather a potentially long respiratory illness season?
Stovall : Demands for supplies like PPE can surge during peak respiratory illness season. To help maintain stable inventory levels, supply chain leaders can work with distributors to leverage historical data and collaborate with supply chain teams to establish flexible ordering strategies. This shifts the burden away from providers.
Specialized handling capabilities within the healthcare supply chain, such as cold storage and chemical vaults, are essential to safeguarding the potency and efficacy of certain life-saving medicine and medical products. Thousands of medical products such as vaccines, penicillin, IV solutions, laboratory products and medical devices, must often travel through the supply chain with strict storage and shipping conditions to

maintain their integrity. Choosing a trusted distribution partner with robust specialized handling capabilities means that vaccines can be administered with confidence, lab diagnostic tests are reliable, and medical treatments are safe and effective.
The
A medical products team helps providers stay ready with essential solutions, including support for clinical labs, PPE, and safety-engineered devices for vaccine administration.
The clinical lab’s daily supply needs can shift quickly during sea-
CDC estimates up to 88% of sharps injuries can be prevented by choosing safer medical devices. Safetyengineered devices around sharps are designed to decrease the risk of needlestick injuries, minimize exposure to blood and bodily fluid, and comply with OSHA regulations.
else our readers should know?
Stovall: With thoughtful planning and a focus on prevention, facilities can be well-equipped to meet the challenges of flu season and maintain a safe, healthy environment. A comprehensive mitigation plan should consider the distinct needs of clinicians, patients and individual hospital departments.
sonal surges, making it challenging to predict how much to order before the season starts. To mitigate the risk of supply shortages, a reserved inventory program gives labs the option to secure their preferred diagnostic products when they need them, even during an unexpected rush in demand. Participating providers have inventory reserved exclusively for them, located close to their

facility for prompt delivery, helping to prevent unexpected stockouts.
Vaccination programs are also a key part of respiratory season preparedness, often managed by hospitals and/ or healthcare facilities. Having the right products and a plan in place helps ensure facilities are equipped to deliver vaccines and do so safely. The planning process should
include hands-on training for those administering vaccines to reduce the potential for accidents.
The CDC estimates up to 88% of sharps injuries can be prevented by choosing safer medical devices. Safety-engineered devices around sharps are designed to decrease the risk of needlestick injuries, minimize exposure to blood and bodily fluid, and comply with OSHA regulations.
Examples of this technology include:
• Safety needles with integrated and robust shields that cannot easily be removed and are activated with one hand to help prevent accidental needlestick exposures.
• Blunt-tip and cannula needles designed with a blunt point to reduce risks of injury.
• Disposable sharps containers with lids that allow for one handed disposal, forced horizontal drop and counterbalanced lids to prevent access to sharps once they’re disposed. HPN
REFERENCES:
1. “Seasonal influenza - Global situation.” World Health Organization. https://www.who.int/ emergencies/disease-outbreak-news/item/2025-DON586
2. “US respiratory virus activity reaches high levels as flu, RSV spread.” CIDRAP. Bergeson, Laine. https://www.cidrap.umn.edu/respiratory-syncytial-virus-rsv/us-respiratory-virus-activityreaches-high-levels-flu-rsv-spread




















hpnonline.com/subscribe

Leaders at Lurie Children’s and Nemours
Children’s share practical strategies to break silos and improve outcomes.
BY KARA NADEAU
Sterile processing and surgical services are inextricably linked. Yet in many organizations, the sterile processing department (SPD) and operating room (OR) still function at arm’s
length, interacting mainly when something goes wrong.
Two children’s hospitals are working differently. At Ann & Robert H. Lurie Children’s Hospital of Chicago and Nemours Children’s Hospital in


Wilmington, Del., leadership “power duos” spanning perioperative services and SPD are building intentional, structured collaboration that reaches from the C-suite to the frontline.
Their efforts demonstrate how organizations can move beyond blame, align around shared goals, and improve safety and efficiency for surgical patients.
Why collaboration matters: patient safety, reliability, and compliance
Both organizations describe collaboration between the SPD and OR as foundational to surgical operations and critical to patient safety.
“At the backbone of every hospital and every operating room is sterile processing. If the SPD shuts down, there will be chaos,” said Nemours Children’s Hospital Sterile Processing Department Manager Kwame Addomah Gyabaah, MHA, Mini MBA, BS, ACHE, CHL, CER, CIS, CRCST.

Nemours Children’s Hospital Assistant Vice President, Perioperative Services & SPD (Delaware Valley) Edna Gilliam, DNP, MBA, RN, CNOR, NEA-BC, emphasized the interdependence of perioperative workflows.
“Our processes are so interconnected that neither department can operate in a silo or succeed alone. We need transparent and collaborative conversations so we can work together to provide the best possible care for our patients.”
At Lurie Children’s Hospital, Senior Director of Surgical & Procedural Services John Olmstead, MSN, MBA, CNOR, FACHE, noted the clinical risk associated with reprocessing failures. “There’s nothing more dangerous in healthcare than reusing a reusable device that isn’t sterile and literally injecting a germ into the patient.”
Lurie Children’s Director of Procedural Care - Surgical Services, W. Zeh Wellington, DNP, MSN, RN, NE-BC, added that the stakes are inseparable from the hospital’s mission. “Every instrument we touch has a direct connection to a child on an operating table. Collaboration isn’t optional. It’s instrumental. It’s how we protect our patients with every tray, every instrument, every scope.”
Olmstead also pointed to the regulatory drivers for collaboration given that TJC (The Joint Commission) and state health agencies have intensified their focus on SPDs because of the high risks associated with reusable devices.
“Regulators are really getting into the finer details of sterile processing because of its complexity and criticality to the safe provision of care,” he stated. “Their goal is patient protection, not citation quotas. Therefore, it is incredibly important for OR and SPD leaders to frame compliance not as a burden but as a shared responsibility to protect vulnerable patients.”
While each hospital began from a different starting point, both
leadership pairs built collaboration through consistent structure, shared ownership, and a relentless focus on safety. Their approaches reflect two distinct but equally effective models.
Power Duo #1: Lurie Children’s – embedding collaboration in daily practice
When Wellington joined Lurie Children’s, he made “intentional collaboration” a priority from the outset. “Historically, SPD and OR departments have operated in silos,” he said. “But when children come to our hospital at their most vulnerable time, they don’t know this department or that department — they just know they are having surgery.”
“It’s very easy to become ‘us versus them’ on both sides – the SPD and the OR,” said Olmstead. “It’s not, ‘can you afford to take time to make a collaborative team?’ It’s more of “can
you afford to not take time to do it?’ Because it’s so critical that we get this right.”
Working closely together, Wellington and Olmstead have implemented structured practices that require the two teams to plan and problem-solve together.

Wellington rounds in both the OR and SPD each morning for team huddles to discuss patient safety, instrumentation, bioburden findings, and follow-up actions. The emphasis is on shared accountability, as he explained:

“I ask the teams, ‘What would happen if bioburden was discovered on an instrument in the OR? What if this was your child, parent, or other loved one?’ We view all patients as our family members. When they’re on that table in the operating room, we’re
Actionable insights from two children’s hospitals that transformed teamwork across sterile processing and surgical services.
1. Align Leaders Around Shared Goals
Begin with the OR and SPD leadership teams. Establish expectations for transparency, accountability, and shared ownership.
“It really must be a top-down strategy,” said Olmstead. “It takes physical, intentional steps.”
Gilliam added: “The first step is le ing go of defensiveness. This isn’t punitive — these are issues we need to fix together.”
2. Increase CrossDepartment Visibility
Cross-department visibility strengthens understanding and respect. Invite surgeons, nurses, and leaders to observe SPD workflows, and have SPD shadow staff in the OR to build appreciation for each team’s complexity.
“When we spend time in each other’s spaces, we really see what the other team is up against,” Gyabaah said. “That goes a long way toward building one team.”
“It’s a total eye-opener when surgeons see the SPD in action,” Olmstead said.
3. Make Collaboration Routine, Not Reactive
Institute daily touchpoints that occur regardless of issues: huddles, rounding, and case-review calls. Commi ees like tray-quality review groups help identify trends and drive improvements.
“That’s why I round in the OR and SPD every single morning,” Wellington said. “These daily touchpoints keep us aligned on safety. We’re not waiting for problems — we’re talking about what’s coming next and how to prevent issues before they happen.”
“Our teams meet every day to know what’s going on — what we did right today, what we can improve, and how to
be be er the next time,” Gyabaah said. “Keeping that daily dialogue going is a great thing for both departments.”
Positive reinforcement and proactive updates build trust.
“You can’t just show up when something breaks,” Olmstead said. “If the only messages people hear are negative, you lose them. You build a team by recognizing what’s going well and coaching from there.”
“It can’t only be about pointing out issues,” Gyabaah said. “We ask questions, we share what went well and where we can improve. That balance helps the team feel supported, not singled out.”
5. Engage Frontline Champions
Frontline champions serve as bridges between leadership and staff, helping translate expectations, reinforce culture, and maintain momentum during change.
“Champions are important because they are the informal leaders,” Gilliam explained. “Their enthusiasm spreads.”
6. Empower and Recognize the Frontline
Empowerment goes beyond communication — it means involving frontline staff directly in shaping workflows, surfacing risks, and designing solutions. Leaders at both hospitals stressed that meaningful improvement accelerates when those doing the work have a voice in how it evolves.
“If you want to solve a problem, ask the frontline,” said Wellington. “They know the innovations we need. My job is to listen, act on their ideas, and give them the credit.”
Gyabaah emphasized the value of inviting staff into continuous improvement conversations.
“We’re always asking our staff, ‘What could we be doing differently?’ Their input keeps the communication open and helps us move forward as one team.”
7. Start Small and Build on Successes
Choose a few high-impact issues first, demonstrate progress, then expand efforts.
“The journey of a thousand steps starts with the first one,” said Olmstead.
Gilliam recommended: “Take three burning issues, achieve success, then move to another three.”
8. Use Transparent Data to Reframe ‘Always’ and ‘Never’ Replace frustration with facts.
“You show the data — 20,000 trays processed and only five issues — and that changes the conversation,” Gilliam said.
9. Keep the Focus on the Patient Shared purpose strengthens collaboration more than any metric.
“I frame discussions around safety and quality — around what we’re doing for the kids,” Wellington said. “These could be our children.”
10. Celebrate Wins Early and Often
Consistent, visible recognition reinforces progress and pride. Mark bioburden-free days, reporting improvements and certification milestones with meaningful celebrations that honor SPD as clinical partners.
“We want them to feel valued,” Gilliam said, describing Nemours Children’s white-coat ceremonies and ongoing recognition.
“The excitement in SPD and the OR is magnetic,” Wellington said. “When you celebrate what’s going well, that energy becomes contagious across the whole team.”
committed to taking care of them. Whether you’re an SPD technician working in decontam, sterilization or prep and pack, an OR scrub technologist, or a nurse circulating in the operating room – you’re all part of that surgery.”
“We perform a lot of audits to ensure we are catching bioburden, and ensure our tools and supplies are top notch,” he continued. “When things sometimes happen, we want to identify them, have a corrective plan, rectify them, and make sure things are done properly going forward. Regular rounding and team huddles help us continue this constant focus on success.”
Wellington and Olmstead provide their team members with cross training opportunities. OR team members spend “a day in the life of the SPD” to understand what happens to instruments after a case, and in turn, SPD team members will spend “a day in the life of the OR” to understand how the instruments they reprocess are used.
Visits to the SPD also extend to the surgeons and the C-suite.
“Zeh spends a lot of time with our surgeons and one of our favorite things is when he takes a surgeon up to see our



SPD because they don’t know what to expect,” said Olmstead. “It’s a total eye opener for them.”
“As a result of a lot of hard work and dedication on John’s part, our C-suite has always been very supportive of the SPD,” said Wellington. “Every time I have invited them for a visit, they come down from their offices to the ninth floor where our department is located.”
The DRILL - daily multidisciplinary case review
Wellington developed a standardized process called “Daily Review of NextDay Cases” (DRILL) where SPD and OR professionals participate in daily calls to review upcoming cases and instrument needs.
During the DRILL process, they identify potential backto-back tray conflicts, workflow constraints, or other issues that could necessitate use of immediate use steam sterilization (IUSS) or accelerated cycle/rapid sterilization, practices that Wellington and Olmstead have committed to eliminating.
DRILL
1. D aily review of next day cases and turnover needs of trays/ instrumentation
2. R earranging schedule to eliminate need for quick turnover
3. Input from surgeon of specific needs for the case
4. List of one-of-a-kind trays – reorder additional trays if need persists
5. Look over and review all preference cards
Wellington’s early identification of pressures allows the team to adjust schedules, reallocate trays, or procure additional inventory to ensure reprocessing windows are safe and compliant.
improvements
Wellington and Olmstead emphasized the importance of engaging SPD professionals directly in problem-solving.
“If you want to know how to solve a problem, ask the frontline,” Wellington said. “They know the innovations we need — our job is to act on them and give them credit.”
One recent improvement came from a long-tenured SPD technician who identified a persistent workflow risk. During routine rounding, Wellington and Olmstead asked her what issue she would address if she could change one thing in the department.
“She explained that nurses often came into SPD and walked straight into prep and pack,” Wellington said.

“That created a risk that someone might inadvertently pull a tray that wasn’t fully processed or hadn’t undergone the final check required before it could be transported to the OR.”
The technician proposed a simple, high-impact solution: a clearly defined boundary marking where staff must pause before entering the prep-and-pack area.
“She said, ‘I wish we had a red line on the floor so people know where to stop,’” Wellington recalled. “John was like, ‘Let’s do it, Wellington.’”
Wellington and Olmstead acted immediately, calling for installation of a line of red floor tiles to distinguish the general-access area from the restricted prep-and-pack zone. Anyone entering now must stop and request permission, ensuring an SPD technician can escort them and verify trays are safe, complete, and ready for use.
The change reduced unnecessary traffic, protected workflows, and reinforced SPD’s role in maintaining sterile integrity.
“When frontline staff bring forward ideas like that, it’s our responsibility to implement them,” Wellington said. “Those insights are part of the secret sauce of SPD success.”
The collaboration between the SPD and OR team led and fostered by Wellington and Olmstead, and the new practices implemented have contributed to greater patient safety and cost savings.
Clear operational and quality outcomes include:
• IUSS (aka flash) sterilization virtually eliminated
• IUSS sterilizers removed from service, saving more than $20,000 annually in maintenance costs
• Use of an accelerated cycle/rapid sterilization method reduced to zero
“We have worked collaboratively – the SPD and OR teams – to essentially eliminate flash (IUSS) sterilization,” said Olmstead. “I can’t remember the last time somebody had to flash something. And over the past year, there has been zero use of accelerated cycle/rapid sterilization, which is almost unheard of.”
Mutual respect between the SPD and OR teams, ongoing collaboration, and shared problem-solving have helped shift the narrative at Lurie Children’s from “SPD as a problem source” to “SPD as a partner.”
Olmstead said that a critical component of this shift has been how leaders respond when issues arise.
“You can’t punish a service into excellence,” Olmstead said. “You can’t shrink yourself to greatness. You can’t cut yourself into high quality. And ‘throwing darts’ — firing people, threatening them, blaming them — has never improved a single process. You can create turnover, but you can’t threaten people into excellence.”
Instead, the Lurie Children’s team uses coaching, structured accountability, education, and recognition to build both competence and trust. This approach, combined with
the elimination of IUSS and rapid sterilization, has allowed the infection prevention team to evaluate cases holistically rather than defaulting to SPD as the presumed source of problems.
Power Duo #2: Nemours Children’s — building ‘one team’ through structure and culture
When Gilliam arrived at Nemours six years ago, she noted how there was a “clear divide between OR and SPD” and described the relationship between the two teams as “tense” and “negative.”
“When SPD became a part of my oversight, we worked really hard to create a ‘one team’ kind of perspective,” she stated.
Gyabaah joined Nemours four years later. Having been employed by other hospitals that lacked alignment between SPD and OR, it came as no surprise to him that he and Gilliam had to work on relationship building between the teams to strengthen this ‘one team’ approach.
“Once the relationship is built, you should get the feeling that they’re here to help you and not to point fingers and point out your faults,” said Gyabaah.
They partnered to deliberately align structures, workflows, and communication across departments.
Mirrored leadership roles
They created lead SPD technician roles designed to mirror the OR’s lead surgical technicians. The OR’s lead surgical technicians, who serve as liaisons between surgeons and perioperative staff, are paired with lead technicians in SPD assigned by specialty or service. Each specialty now has a designated partner on both sides, establishing direct, reliable communication channels for instrument questions, specialty set needs, and emergent issues.
“The whole goal of that was to give each clinical service a clear point of contact in both departments they could communicate with,” said Gilliam. “So, if the OR is missing instruments or ordering instruments, they know the person to call in SPD for input on these matters. That has really opened the doors for communication and collaboration.”
Daily huddles and transparent communication
SPD and OR teams meet daily to discuss what worked well and what requires follow-up. Lead OR technicians regularly spend time in the SPD and lead SPD technicians in the OR.
“Transparency is the number one word that I use in terms of benefit,” said Gyabaah. “We try to be transparent with each other. We are curious and ask questions of each other such as, ‘What could we be doing differently?’
Conversations like that keep the communication open and reinforce that we are one team.”

After identifying some trending tray quality issues in the SPD, Gilliam and Gyabaah formed a tray quality committee led by SPD technicians, OR representatives, and a perioperative quality and safety specialist. The members meet every morning to review issues in real time, identify trends, and drive corrective action.
“One thing I love about this committee is we can identify when an issue is repeating itself, dig in, and get to the root cause of it – where it’s coming from and what is causing it,” said Gyabaah. “It’s helped us a lot to pinpoint what we need to change in our department and in the OR as well.”
“Sometimes, if something happens that same day, we tackle it immediately,” he added. “We record all identified issues, and for those that aren’t urgent, we review them at the end of each month to uncover where they originated and develop corrective actions.”
Gyabaah has also implemented a highly visible, data-driven approach to tracking bioburden events. He uses what he has defined as the “360 rule” — a tray that leaves the OR, passes through the full SPD reprocessing cycle, and returns
to the OR still containing tissue, blood, or other soil, where it is then identified by the surgical team.
The team records each 360 event and maintains a running count of days without such an occurrence on a board in the department. Any new event resets the count to zero. Recently, Nemours reached 97 days with no bioburden; the team plans to formally recognize staff when they hit 100 days.
“To date this year, we have only had five 360 events,” said Gyabaah. “When something goes wrong, we circle back, meet as a team, brainstorm, and move forward from there. That’s gone a long way to help us in sterile processing and periop – and the benefits are passed along as great outcomes for our patients.”
Supporting technicians with education and recognition
To support technicians, Nemours uses a point-based algorithm tied closely to education. For example, if an SPD technician forgets to add a filter to a tray, the event is logged, and the SPD educator becomes involved at each escalation step. Corrective action is reserved for cases where coaching and education have been provided over time.
Gilliam says this approach, along with the presence of a perioperative quality and safety specialist, has led to an increase in event reporting — a positive sign.
“When they don’t report, we don’t know there’s an issue to be fixed,” she said. “Now we’re getting the incident reports, and we understand our opportunities. We’ve seen our bioburden drop, and staff can see the success of their work.”
Recognition is equally emphasized and intentionally built into the culture. Nemours supports a clinical ladder for SPD, shares the costs for sterile processing certifications, and encourages SPD staff to pursue infection prevention certification to further strengthen their influence on quality and safety.
Gilliam, Gyabaah, and the department of surgery host a white coat ceremony for SPD professionals who earn all four sterile processing certifications. When an individual attains this level of certification, they are presented a personalized white lab coat during a breakfast attended by members of the hospital’s executive leadership team who celebrate their achievement.
Commenting on this practice, Gilliam stated, “We want them to feel valued as clinical partners.”
Gilliam and Gyabaah’s collaborative work – and successes – are grounded in transparent data and reporting.
“Communication is the start of collaborative SPD/ OR improvements but it cannot be the only factor,” said

Gyabaah. “To snap out of the ‘blame game,’ you need reliable and consistent metrics that both teams acknowledge as the source of truth.”
“Let’s say you have a surgeon who claims every tray he receives from the SPD has an issue,” Gilliam added. “When you can show them the data to prove tray issues do not happen ‘all the time’ – metrics that prove out of the 20,000 trays the SPD processed in the past month, only five had issues – that reframes the conversation and interaction.”
The “power duos” at Lurie Children’s and Nemours Children’s show that strong SPD/OR collaboration is achievable and sustainable. By aligning leadership, hardwiring communication into daily workflow, using data constructively, and elevating the clinical role of SPD, these organizations have improved reliability, quality, and culture across the surgical continuum.
Their experiences offer a practical roadmap for any facility seeking to strengthen SPD/OR partnerships and enhance surgical safety. HPN
BY WILLIAM LEIVA
Mechanical cleaning is a critical step in the decontamination process of reusable medical devices.1,2 Standards, guidelines, and standard operating procedures require proper cleaning to ensure the removal of visible debris and to prepare instruments for effective disinfection and sterilization.3-5 Mechanical cleaning tools are designed to make this process safer, faster, and more consistent. Understanding the features, benefits, and correct use of
1.Identify key types of mechanical cleaning tools and describe their main features and functions.
2.Explain the benefits and limitations of each mechanical cleaning method.
3.Demonstrate the safe and effective use of mechanical cleaning tools.
Sponsored by:

each tool is essential for maintaining safety and compliance in healthcare and laboratory settings.
LEARNING OBJECTIVE #1:
Identify key types of mechanical cleaning tools and describe their main features and functions. Mechanical cleaning tools are designed to remove contaminants efficiently and consistently and to reduce the strain that sterile processing professionals face during the decontamination process. These tools aim to reduce reliance on manual cleaning processes, which can be time-consuming and inconsistent.
The three main types of mechanical cleaning tools are ultrasonic baths, water guns, and washer-disinfectors, also known as mechanical washers. Each serves a unique function and has specific features suited to different cleaning needs.
Water guns, sometimes called highpressure spray systems, use pressurized water to remove visible debris and soil (Figure 1A). They are often used for pre-cleaning before a more thorough mechanical or chemical cleaning step.5,6 Key features include: • Adjustable water pressure to avoid damaging delicate instruments.
Table1. Overview of mechanical cleaning tools with main features and usage.
GunsDifferent tip adaptors and spray nozzles Before ultrasonic or washerdisinfector cleaning
Portable and easy to useWhen devices remain contaminated a er further steps
Ultrasonic BathsUses bubbles (cavitation)When instruments require ultrasonic cleaning
Common frequencies: 35-40 KHz
Timers and set cyclesOn short cycles for general instruments
Uses cleaning detergents
Some models include irrigation ports
Effective for hard-to-reach areas
Washer-Disinfectors Fully automated cyclesInstruments either required washerdisinfector
Specific cycles based on loadIn sterile processing departments required to deliver large throughput Require specific load carriers

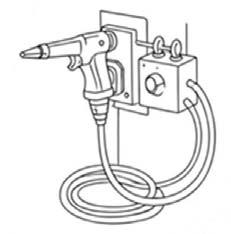

1: A. Rendering of a water gun used in sterile processing and B. water gun tip adaptors and spray nozzles for targeting specific areas. This figure was created using ChatGPT. All content was reviewed by the author for accuracy and appropriateness.
• Spray nozzles (Figure 1B) designed for targeting specific areas, such as tapered and luer-slip adapters.
•Portable units suitable for small setups or sink-side cleaning stations.
•Some water guns use steam to flush debris out.
Water guns are primarily used to flush debris from surgical instruments before placing them into ultrasonic baths or washer-disinfectors. They provide rapid, hands-on cleaning with moderate efficiency.
Ultrasonic Baths
Ultrasonic baths use high-frequency sound waves to create tiny bubbles in a liquid solution, a process called cavitation (Figure 2A). These bubbles collapse when they contact instrument surfaces, helping to dislodge debris from hard-to-reach areas.
Ultrasonic bath systems are especially useful for cleaning complex instruments with lumens, hinges, or other design features that are difficult
to clean manually.5,7 Key features of this technology include:
• Bath size: Ranges from small tabletop units used in dental settings, to large baths with multi-level load carriers for a higher throughput.
• Frequency and power settings: The frequency is measured in kHz, with most models using frequencies between 35 and 40 kHz. 1 kHz is equivalent to 1000 sound waves per second. Units with 35 kHz provide slightly larger bubbles than units using 40 kHz, yet both are effective in dislodging soil.
• Pre-programmed cycles : Customized for different instrument types.
• Timers: Allow standardized cleaning cycles, with some specifically designed for devices such as robotic arms or laparoscopic devices.
• Chemical agents: Different cleaning chemistries are used on ultrasonic baths including enzymatic, neutral, and mild-alkaline detergents.
• Irrigation system : Some units include multiple ports with special adaptors for instruments that require irrigated ultrasonic cycles; in this case, the adaptors must be fully inserted into the instrument flushing port (Figure 2B).
Functionally, ultrasonic baths provide a consistent, thorough cleaning that reduces manual labor and increases safety by minimizing direct contact with contaminated instruments.
Washer-disinfectors, also known as mechanical washers or auto washers, are automated units that combine pre-cleaning, washing, and rinsing, and may include thermal disinfection or lubrication features.8-10 They are designed for larger instrument loads, including multiple-level and specific load carriers for anesthesia, ophthalmology, and lumened or channeled devices. Washer-disinfectors
This lesson was developed by Solventum. Lessons are administered by Endeavor Business Media.
A er careful study of the lesson, complete the examination online at educationhub.hpnonline.com. You must have a passing score of 80% or higher to receive a certificate of completion.
The Certification Board for Sterile Processing and Distribution has preapproved this in-service unit for one (1) contact hour for a period of five (5) years from the date of original publication. Successful completion of the lesson and post-test must be documented by facility management and those records maintained by the individual until recertification is required. DO NOT SEND LESSON OR TEST TO CBSPD. www.cbspd.net.


Healthcare Sterile Processing Association, myhspa.org, has pre-approved this in-service for 1.0 Continuing Education Credits for a period of three years, until January 12, 2028.
For more information, direct any questions to Healthcare Purchasing News editor@hpnonline.com.

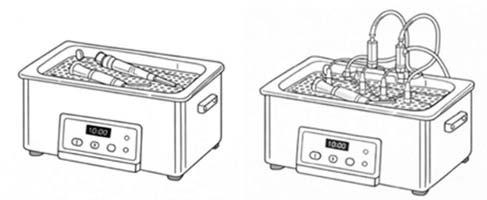
facilitate standardized cycles that manual methods cannot achieve. Key features include:
•Built-in rinsing, washing, and drying stages.
•Monitoring systems for temperature, time, and detergent concentration.
• Reduced water, energy, and chemical consumption, especially in newer models.
These units assist with cleaning and disinfection, reduce human error, and minimize staff exposure to contaminated instruments. Instruments emerge clean, disinfected, and ready for inspection, packaging, and sterilization, improving workflow and safety.
Explain the benefits and limitations of each mechanical cleaning method.
Mechanical cleaning methods play an essential role in the safe and effective reprocessing of reusable medical instruments. While all mechanical cleaning tools aim to remove debris and reduce contamination, each method offers unique benefits and presents specific limitations. Understanding these strengths and weaknesses helps healthcare and laboratory staff select the most appropriate tool for each stage of the cleaning process and apply it safely and effectively.
Water guns are commonly used as a first step in mechanical cleaning. Their primary benefit is the rapid removal of gross, visible debris such as blood, tissue, or other soil from instruments. By using pressurized water, water guns can quickly flush contaminants from surfaces and internal channels, making them especially useful immediately after instrument use. They are flexible, easy to operate, and can be used in sink-side cleaning areas with minimal setup. Compared to more complex systems, water guns are also cost-effective, making them accessible for facilities of many sizes.
Despite these advantages, water guns have clear limitations. They are designed only for pre-cleaning and cannot
replace thorough cleaning steps. While they remove visible debris, they do not reliably clean microscopic contaminants or microorganisms. Additionally, improper use may create splashing, which increases the risk of staff exposure to contaminated fluids if personal protective equipment (PPE) or splash-control systems are not used.3 Water pressure must also be carefully adjusted, as excessive force can damage delicate instruments or force debris deeper into lumens. For these reasons, water guns should be viewed as a supportive tool rather than a complete cleaning solution.
Ultrasonic baths offer a more advanced level of cleaning by using high-frequency sound waves to create cavitation bubbles in a liquid solution. When these bubbles collapse, they release energy that loosens debris from instrument surfaces, including areas that manual scrubbing cannot reach. This makes ultrasonic baths particularly effective for instruments with hinges, joints, lumens, or intricate designs. Another major benefit is uniform cleaning, as the cavitation process reaches surfaces consistently. By reducing the need for manual scrubbing, ultrasonic baths also reduce staff handling and decrease exposure to contaminated instruments. However, ultrasonic baths also have limitations that must be carefully managed. Instruments usually require pre-cleaning before ultrasonic processing, and some may need soaking to achieve optimal results. Certain instruments can be sensitive to cavitation, particularly at lower frequencies, which may cause surface damage if manufacturer instructions for use (IFUs) are not followed. Additionally, there is no standardized method to measure the strength or quality of cavitation, as current indicators only confirm whether cavitation is occurring, not how effective it is at a specific frequency. This makes adherence to validated cycles and manufacturer guidance especially important.
“Mechanical cleaning methods play an essential role in the safe and effective reprocessing of reusable medical instruments.”
Washer-disinfectors represent the most automated mechanical cleaning option. Their greatest benefit is the ability to deliver standardized, repeatable cleaning cycles that may also include thermal disinfection. These systems can process large volumes of instruments

efficiently, reducing variability and human error associated with manual cleaning. Monitoring systems track critical parameters such as time, temperature, and detergent concentration, supporting regulatory compliance and quality assurance. Newer washer-disinfectors often include self-loading carriers, which reduce physical labor and improve workflow efficiency.

The main limitations of washerdisinfectors relate to cost and flexibility. They require a significant initial investment and ongoing maintenance, which may be challenging for smaller facilities. Washer-disinfectors may also be less suitable for very delicate or irregularly shaped instruments unless specialized load carriers or customized cycles are available. Implementing new load carriers or cycles can require additional resources and validation. Furthermore, staff must be properly trained to load instruments correctly, select appropriate cycles, and monitor performance to ensure safe and effective operation.
Water guns, ultrasonic baths, and washer-disinfectors each serve an important role in mechanical cleaning workflows (Table 1). By understanding their benefits and limitations, staff can combine these methods effectively to improve cleaning outcomes, protect sterile processing professionals, and maintain compliance with standards and manufacturer IFUs. By weighing benefits against limitations, staff can develop workflows that maximize cleaning efficiency and safety. For instance, a workflow may combine instrument soaking, water guns for precleaning, ultrasonic bath systems, and washer-disinfectors for full sets of instruments.
Despite these three technologies, the decision about which system to use depends on the IFU and its companion documents, such as wall charts. Currently, there is no universal cleaning process appropriate for use across all instruments, including those within the same instrument group (e.g., laparoscopic devices). Following IFUs is critical for sterile processing professionals.
Demonstrate the safe and effective use of mechanical cleaning tools.
Demonstrating safe and effective use of mechanical cleaning tools relies on following manufacturer instructions and applying best practices. While general safety protocols are standardized across healthcare facilities and reinforced through accreditation, each sterile processing department must consider the nuances of different technologies to ensure they meet their specific needs.
For water guns, staff should always wear PPE to prevent exposure to splashes and aerosolized droplets. Aerosolization occurs when small liquid droplets are broken into tiny particles that can remain suspended in the air. Droplets smaller than five micrometers can be inhaled, potentially reaching the respiratory tract and carrying contaminants. This can happen during cleaning with water guns or ultrasonic baths, so the use of splash guards, proper ventilation, and PPE is essential to reduce inhalation risk. When flushing or rinsing instruments, the water gun and instruments should be pointed downward to prevent splashes and contamination. Water pressure must be adjusted carefully to avoid damaging instruments, and instruments should be held securely while flushing. Prerinsing instruments before transferring them to ultrasonic baths or washer-disinfectors further enhances cleaning effectiveness.10
For ultrasonic baths, the tank should be filled with the recommended solution at the correct concentration, following both equipment and chemical manufacturer guidelines. Instruments must be fully submerged without overcrowding, which can be challenging in facilities with limited ultrasonic capacity or high instrument throughput. The appropriate cycle time and frequency should be set according to manufacturer instructions. Regularly changing the cleaning solution and maintaining the tank ensures consistent performance and prevents contamination.
Washer-disinfectors require proper instrument loading according to manufacturer instructions to ensure full exposure to all cleaning and disinfection stages.11 The correct cycle should be selected based on instrument type, and load carriers and irrigation ports must be properly positioned. Staff should monitor cycle parameters, including time, temperature, and detergent concentration, to verify effectiveness. Routine maintenance and validation checks are necessary to maintain compliance and operational safety.
Following these instructions ensures instruments are cleaned thoroughly while protecting both staff and equipment from harm. By adhering to best practices, sterile processing personnel can maintain safe, efficient, and reliable cleaning workflows.
Mechanical cleaning, both manual and semi-automated, is a critical step in instrument decontamination, ensuring
that instruments are free from debris and ready for further reprocessing steps. Water guns, ultrasonic baths, and washer-disinfectors each offer unique features that make cleaning safer, faster, and more reliable than manual methods. Ultrasonic baths excel at reaching intricate areas, water guns provide rapid gross-debris removal, and washer-disinfectors offer standardized, large-scale cleaning and disinfection. These systems are used in a sequential fashion, with some instruments requiring
or recommending all of them, while others only require some of these steps.
Understanding the benefits and limitations of each method is essential. Ultrasonic baths are highly effective but require careful handling and maintenance; water guns are flexible but limited in depth of cleaning; and washer-disinfectors are efficient yet require a significant capital investment. By knowing when and how to use each tool, sterile processing professionals can optimize
1.Why is mechanical cleaning important in the decontamination of medical devices?
A.It makes instruments look newer
B.It replaces sterilization
C.It removes visible debris and prepares instruments for further reprocessing steps
D.It is optional if instruments look clean
E.It only improves speed, not safety
2.Which of the following is NOT a main type of mechanical cleaning tool discussed in the text?
A.Water guns
B.Ultrasonic baths
C.Washer-disinfector
D.Mechanical washers
E.Lint free cloth
3.What is the main purpose of water guns in mechanical cleaning?
A.Flushing visible debris before further cleaning steps
B.Sterilizing instruments
C.Drying instruments
D.Lubricating instruments
E.Inspecting instruments
4.Which feature of water guns helps prevent damage to delicate instruments?
A.Steam-only operation
B.Adjustable water pressure
C.Automatic timers
D.Chemical detergents
E.Thermal disinfection
5.Ultrasonic baths remove debris using which process?
A.Heat sterilization
B.Manual scrubbing
C.High-pressure water spray
D.Cavitation created by sound waves
E.Dry air circulation
All CEU quizzes must be taken online at: educationhub.hpnonline.com The cost to take the quiz is $10.
6.What does “1 kHz” mean when discussing ultrasonic bath cleaning systems?
A.1,000 bubbles per second
B.1,000 sound waves per second
C.1,000 cleaning cycles per minute
D.1,000 liters of water per hour
E.1,000 instruments per load
7.Why are ultrasonic baths especially useful for complex instruments?
A.They use higher temperatures
B.They are faster than all other methods
C.They reach hard-to-clean areas like hinges and lumens
D.They require no chemicals
E.They disinfect instruments completely
8.Which is a limitation of ultrasonic baths?
A.They cannot clean visible debris
B.They replace washer-disinfectors
C.Some instruments may be damaged by cavitation
D.They do not use water
E.They cannot be standardized
9.What is a key benefit of washer-disinfectors?
A.Low purchase cost
B.Manual control of every step
C.Standardized cleaning and possible thermal disinfection
D.Use only for single instruments
E.No training required
10.Which action helps ensure safe use of mechanical cleaning tools?
A.Skipping manufacturer instructions
B.Overcrowding instruments during cleaning
C.Wearing PPE and following proper loading guidelines
D.Using the highest pressure at all times
E.Reusing cleaning solutions indefinitely


workflows, improve patient safety, and comply with regulatory standards.
Equally important is using these tools safely and effectively by following manufacturers’ IFUs. Proper training, PPE use, solution concentration, adequate maintenance, and adherence to recommended cycles help prevent damage to instruments and reduce the risk to staff. Combining these mechanical cleaning methods into an appropriate workflow ensures that every instrument is prepared for the next step in decontamination. By mastering the features, benefits, limitations, and proper use of these tools, healthcare and laboratory personnel can confidently maintain high standards of cleanliness, safety, and efficiency in their daily work, ultimately protecting both staff and patients. HPN
REFERENCES:
1.World Health Organization. Decontamination and reprocessing of medical devices for healthcare facilities. Geneva, Switzerland: WHO; 2016. Accessed January 4, 2026. https://www.who. int/publications/i/item/WHO-FWC-WSH-15.04
2. Rutala WA, Weber DJ. Disinfection, sterilization, and antisepsis: An overview. Am J Infect Control. 2019;47(suppl):A3-A9. doi:10.1016/j.ajic.2019.01.018
3. Centers for Disease Control and Prevention. Guideline for disinfection and sterilization in healthcare facilities. Atlanta, GA: US Department of Health and Human Services; 2019. Accessed January 4, 2026. https://www.cdc.gov/infectioncontrol/guidelines/disinfection
4. US Food and Drug Administration. Reprocessing medical devices in health care settings: Validation methods and labeling guidance for industry and Food and Drug Administration
staff. Silver Spring, MD: FDA; 2015. Accessed January 4, 2026. https://www.fda.gov/regulatoryinformation/search-fda-guidance-documents
5.Association for the Advancement of Medical Instrumentation. ANSI/AAMI ST79: Comprehensive guide to steam sterilization and sterility assurance in health care facilities. Arlington, VA: AAMI; 2023.
6. Association of periOperative Registered Nurses. Guidelines for perioperative practice. 2023 ed. Denver, CO: AORN; 2023.
7.Association for the Advancement of Medical Instrumentation. ANSI/AAMI ST91: Flexible and semi-rigid endoscope processing in health care facilities. Arlington, VA: AAMI; 2022.
8. International Organization for Standardization. ISO 15883-1:2017 Washer-disinfectors—Part 1: General requirements, terms and definitions and tests. Geneva, Switzerland: ISO; 2017.
9. International Organization for Standardization. ISO 15883-2:2018 Washer-disinfectors—Part 2: Requirements and tests for washer-disinfectors employing thermal disinfection. Geneva, Switzerland: ISO; 2018.
10. International Organization for Standardization. ISO 15883-5:2016 Washer-disinfectors—Part 5: Test soils and methods for demonstrating cleaning efficacy. Geneva, Switzerland: ISO; 2016.
11. US Food and Drug Administration. Biological indicator (BI) premarket notification [510(k)] submissions. Silver Spring, MD: FDA; 2021. Accessed January 4, 2026. https://www.fda.gov/ regulatory-information/search-fda-guidance-documents/biological-indicator-bi-premarketnotification-510k-submissions
William Leiva MBA - MPH is currently a Subject Matter Expert on Medical Devices Sterilization and Reprocessing and is currently the Senior Program Manager for Sterile Reprocessing with Medtronic. He holds undergraduate degrees in physics and engineering and master’s degrees in business administration and public health, where he focused his research on infection prevention strategies involving medical device reprocessing. He is currently a candidate for a Ph.D. in Cybernetics and Econometrics, researching around the economic impact of the burden of premature mortality. He has published peer review articles on the application of 3D printing technologies for medical devices and for austere environments.







BY ADAM OKADA
Q: “I am a new facility IP, and I am unfamiliar with the needs of the endoscopy area. What equipment do you recommend that they need/ should have?”
A: At an educational seminar in September 2025, I had the pleasure of watching the great Michael Matthews1 give a presentation on cleaning in the decontamination area. In his presentation, he gave a wonderful example of why we need the right tools to do the required tasks in sterile processing (SP) and endoscopy. A painter needs a variety of tools to do different painting
projects. For example, painting a wall requires very different tools than painting a portrait. A carpenter may need dozens of tools for a woodworking project: hammer, saw, chisel, drill, sander, etc.
Reprocessing medical devices is complex and requires very specialized tools and equipment. Having only one size brush available at the SP sink for flexible scopes can be challenging if there are multiple models of endoscopes in use. Consider how each scope model’s instructions for use (IFU) recommend a specific size brush.
Returning to your question, let’s take a look at the most essential

specialized tools that you might need to reprocess flexible endoscopes properly.
After point-of-use treatment is performed at the point-of-use, the scope will need to make its way to the decontamination and reprocessing area. When transporting contaminated flexible scopes from the patient room, you’ll need to make sure you have a transport container that is Occupational Safety and Health Administration (OSHA) compliant.2 This means your container should be puncture-resistant, leak-proof, and large enough to hold your endoscope
without having to tightly coil it (which can damage the delicate components inside).
There are several container options on the market. Do some research and find an option that best fits your facilities’ specific needs. And no, buying bins at your local department store does not make you a smart shopper. Where are the IFUs for those bins? Are they leak-proof? Surveyors will ask those questions when they see those $5 bins.
Transporting this expensive and delicate piece of equipment is an often-overlooked step, and one that could cost your facility dearly if it’s not performed properly.
(and possibly a leak tester TESTER)
Once the scope has safely made its way to our decontamination area, the first step in the cleaning process is to make sure the scope is safe to submerge in water/solutions by performing a leak test. There are several internal and external seals on a flexible endoscope to ensure that fluid cannot invade the electronic internal components of the scope. If fluid invasion occurs, the result will be a very expensive repair, if not a complete replacement. This step is critical to perform so that subsequent steps can be successful.
A dry leak test is performed first, followed by a wet (submerged) leak test. Always make sure to refer to your scope’s specific IFU.
Did you know that your leak tester will need to be tested? Most automated leak testers have a specific pound per square inch (psi) requirement; so, how do you know if yours is meeting this requirement? Some newer leak testers have a self-testing function, but all of the older machines, such as a very common leak tester from a scope manufacturer that holds 90% of the market, do not. There are leak tester testers

(Fig. 1) that can test your leak tester to ensure that the tester is testing as it should be.
There will be a test later. Don’t get testy about it.
If cleaning is the most important step in the process, then brushing becomes the single most important component in the entire process. If it’s not clean, it can’t be sterile, and it’s essential that you have the right tools for this
will inactivate one or both solutions. Instead, use a non-solution-soaked sponge, which is both effective and less costly. You’re welcome.
When ANSI/AAMI ST91:20215 for endoscope reprocessing came out in early 2022, it contained the strongest recommendation possible for moving away from manual disinfection soaking and strongly recommended the use of the automated endoscope reprocessor (AER). Manual soaking was such a highly variable process that a more automated, repeatable one in the AER became a preferred option. However, errors can still occur with the AER, so this process will need to be monitored. I would pay special
Based on a 2018 water study performed by Ofstead and Associates, residual water left in the channels of endoscopes was proven to foster microbial growth and provided a breeding ground for biofilm.
vital step. Classical Kovachian proverb says, “Keep it clean.”4
Previously mentioned, you shouldn’t just have one brush at the sink. Some flexible scopes have two lumen ports requiring two different size brushes. Some have a special brush required just for the larger entry port, and then a long lumen brush for the internal channel. Do you have a brush or sponge for the external surfaces of your scope? You should! Your manufacturer’s IFU will give you the sizes needed, so you’ll always need to have those brushes on hand.
Pro tip: Do not use enzymatic sponges at the sink with a different brand and dilution of enzymatic solution inside. The manufacturers have never tested the mixture on each product. We don’t know if it will work, if the enzymes will react, or if they
attention to the water inlet line into your AER. Outbreaks have occurred from contaminated water lines, so review ANSI/AAMI ST108:20236 on water quality for more information about testing here.
If your facility does not have an AER, and is still soaking scopes manually, I will align with ST91 and give my strongest recommendation possible for sunsetting this practice and acquiring an AER for your facility.
Another important takeaway from ST91 was the concept of 10 minutes of forced-air drying for scopes. Based on a 2018 water study performed by Ofstead and Associates7, residual water left in the channels of endoscopes was proven to foster microbial growth and provided a breeding ground for biofilm.
The rationale here is simple: microorganisms love dark, damp places, and endoscope channels provide a narrow, dark, damp chamber ideal for microbial growth.
In the past, we assumed that vertical hanging was sufficient. Gravity was supposed to do the drying job for us; however, because of the narrow channels and slight bends to flexible endoscopes, water droplets were shown to be present in vertically hung scopes for up to two weeks later.
Modern endoscopy departments may have continuous forced-air drying cabinets (Fig. 2) that utilize high-efficiency particulate air (HEPA) filtered air pushed through the scope(s). The turbulent airflow forces water from the channels and creates a hostile environment for microbes. I would certainly recommend these cabinets.
In August 2025, I wrote an article here in HPN titled “Visual Communication”9 about the importance of communication using


images, graphics, or signage to convey clear visual indications between users who may never have an opportunity to see each other or speak directly. Nowhere is the need for clear interpretation of messaging more important than in the endoscopy area!
Reprocessing technicians need to be able to clearly label scopes as having been processed. I can imagine few things more problematic in the endo suite than a contaminated scope being used on multiple patients due to improper labeling.
There are many options for labeling, and they will be dependent on the unique processes present at your facility. Blank tags can do the job, but you probably want to look at options that are less open to human error.
As far as clear visual indicators go, it’s tough to beat the convenience of a push-button tag (Fig. 3) that clearly indicates when your facility policy hangtime has expired, and the scope is due for reprocessing.
There are certainly more tools (e.g., chemistries, dosing machines, ergonomic sinks, etc.) that we can use in the endoscopy suite, but these are some of the major focus areas for surveyors that you may want to start with for compliance purposes. Flexible
endoscopes aren’t getting less complex as technology advances, so it’s vital that we ensure our departments are reprocessing these devices safely and responsibly. To achieve this, we’ll need the proper tools and equipment to do the job. HPN
1.Matthews, M. (2025, September 27). Cleaning in the decontamination area. PWSPA Conference - Valley Presbyterian Hospital, Los Angeles, CA, United States of America.
2.Occupational Safety and Health Administration. Code of Federal Regulations: Occupational Exposure to Bloodborne Pathogens. Title 29 [Bloodborne pathogens], Standard No. 1910.1030(d)(2)(viii). United States Dept of Labor/OSHA. https://www.osha.gov/laws-regs/regulations/standardnumber/1910/1910.1030
3.Healthmark, A Getinge company. (2020, August). Instrument Care - Healthmark Leak Tester Tester. https://www. hmark.com/product/leak-tester-tester/
4.Kovach, Stephen M. (n.d.). “Keep it clean.” [Quote].
5.AAMI (2021). ANSI/AAMI ST91:2021 Flexible and semi-rigid endoscope processing in health care facilities. Arlington, VA: Association for the Advancement of Medical Instrumentation.
6.AAMI (2023). ANSI/AAMI ST108:2023 Water for the processing of medical devices. Arlington, VA: Association for the Advancement of Medical Instrumentation.
7.Ofstead, C. L., Heymann, O. L., Quick, M. R., Eiland, J. E., & Wetzler, H. P. (2018). Residual moisture and waterborne pathogens inside flexible endoscopes: Evidence from a multisite study of endoscope drying effectiveness. American Journal of Infection Control, 46(6), 689–696. https://doi. org/10.1016/j.ajic.2018.03.002
8.Getinge. (2025). EDS8 Drying and Storage Cabinet for endoscopes. https://www.getinge.com/int/products/getingeeds8-drying-and-storage-cabinet/
9.Okada, A. (2025, August 26). Visual Communication. Healthcare Purchasing News (HPN) Online. https://www. hpnonline.com/sterile-processing/article/55306340/visualcommunication
10.Healthmark, A Getinge company. (2020, August). Labeling and Signage – 7-Day Indicator Hangtime Label. https://www. hmark.com/product/7-day-indicator-hangtime-label/
Adam Okada has 18+ years of experience in Sterile Processing and is passionate about helping improve the quality of patient care by giving SPD professionals and their partners greater access to education and information. He has worked in just about every position in the Sterile Processing Department, including Case Cart Builder, SPD Tech I, II, and III, Lead Tech, Tracking System Analyst, Supervisor of both SPD and HLD, Manager, and now as an Educator. Adam is the owner of Sterile Education, the world’s first mobile application dedicated to sterile processing education, and a former Clinical Manager at Beyond Clean. He has published articles for HSPA’s Process magazine, is a co-chair on AAMI WG45 as well as co-project manager for the KiiP “Last 100 Yards” group, and is the former President for the Central California Chapter of HSPA. Adam is currently a Clinical Education Specialist at Healthmark, A Getinge company, where he works on Healthmark webinars, hybrid events, and educational videos, as well as the “Ask the Educator” Podcast with Kevin Anderson.








Better tools build better outcomes. Let’s strengthen endo together.
Endoscope processing is built on a series of critical steps, and each one calls for tools that support clarity and compliance. Our endo-focused solutions and educational support are designed to help your team build a strong foundation for success.
Manual Cleaning
Labeling
Protective Equipment
Inspection Tools
Transport & Storage
Educational Resources




Let’s build a stronger endo foundation for your team.
Scan to get started.