Cambridge International AS & A Level Chemistry
3 Cut the ribbon up into 2 x 0.5 cm, 2 x 1.0 cm, 2 x 1.5 cm and 2 x 2.0 cm lengths. 4 From your mass for 10.0 cm of ribbon, estimate the mass of the 1.0 cm, 1.5 cm and 2.0 cm lengths.
Estimated mass of 1.0 cm lengths …………………g
Estimated mass of 1.5 cm lengths…………………g
Estimated mass of 2.0 cm lengths …………………g
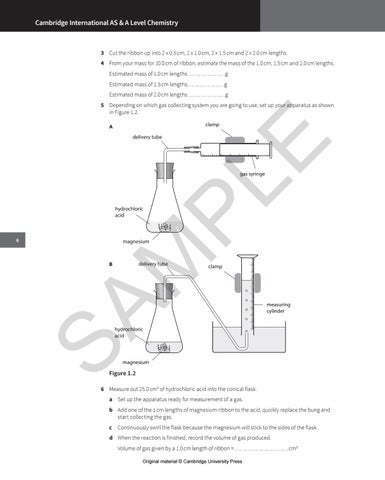
5 Depending on which gas collecting system you are going to use, set up your apparatus as shown in Figure 1.2.
delivery tube
E
clamp
A
M PL
gas syringe
hydrochloric acid
6
magnesium
delivery tube
clamp
SA
B
measuring cylinder
hydrochloric acid
magnesium
Figure 1.2
6 Measure out 25.0 cm3 of hydrochloric acid into the conical flask.
a Set up the apparatus ready for measurement of a gas.
b Add one of the 1 cm lengths of magnesium ribbon to the acid, quickly replace the bung and start collecting the gas.
c Continuously swirl the flask because the magnesium will stick to the sides of the flask.
d When the reaction is finished, record the volume of gas produced.
Volume of gas given by a 1.0 cm length of ribbon = ………………………… cm3 Original material © Cambridge University Press