








Progress against cancer requires a united effort. Researchers, clinicians, patients, and the broad community all play vital roles in advancing our understanding and discovering new ways to prevent, diagnose, and treat this disease. When we work together, our collective strength knows no bounds, and our achievements become limitless.
This annual report highlights some of our most significant breakthroughs—landmarks made possible through collaboration. These stories demonstrate the transformative power of partnership and shared purpose.
I am deeply proud of the impact our team has made in 2025. The unified effort of every contributor continues to inspire hope and propel us toward our goal: helping people live healthier, longer lives. Together, we are making a difference in the fight against cancer.
Warm regards,
Kunle Odunsi, MD, PhD
Director, University of Chicago Medicine Comprehensive Cancer Center
Dean for Oncology, Biological Sciences Division
The Abbvie Foundation Distinguished Service Professor University of Chicago

LISA'S STORY
“I knew that I had to do this.”
Raised in a Christian home by a single mother, Lisa Howard always helped others in her South Side community. Even when she was diagnosed with multiple myeloma, she continued to help by enrolling in a clinical trial as part of her treatment at the University of Chicago Medicine.
She participated in a clinical trial to test ways to help newly diagnosed patients achieve long-term remission. Her treatment would be long and intense: four months of lowdose chemotherapy, a higher dose of chemotherapy, a stem cell transplant, and 15 months of low-dose chemotherapy.
“Participating in this clinical trial was something I knew was lifesaving and life-changing for me, especially knowing that clinical trials get a really bad rap in the African-American culture,” she said. “I knew that I had to do this.”
Now in remission, Lisa helps educate the community about multiple myeloma and clinical trials. She’s become the go-to person whenever friends, family members or neighbors are newly diagnosed with cancer.
“I had to show others that, yes, the past has mistreated us, and there's a lot of medical mistrust that has taken place. However, we're moving forward with new options and new treatments.”
In 2025, our advances in clinical care were all about expansion—more opportunities to collaborate, an expanded network, and more programs that specialize in providing personalized care to our patients.
The University of Chicago Medicine has announced an extended collaboration agreement with AbbVie through 2027 to support innovative solutions in cancer research and clinical trials. Building on a partnership that began in 2016, this extension marks more than a decade of collaboration focused on translational and clinical research to improve and accelerate positive outcomes for patients with cancer.
Despite significant advances in cancer treatment, the disease remains a leading cause of death worldwide, underscoring the critical need for continued innovation in therapeutic approaches and clinical trial methodologies. This extended partnership positions both organizations to address these challenges through collaborative research and the accelerated translation of discoveries into patient care.
The extension of the collaboration is designed to further advance medical research in oncology and streamline clinical trials operations to provide greater connectivity between clinicians and researchers at both organizations.
The University of Chicago Medicine is launching its Cancer Network’s new national initiative to extend its academic medicine expertise in cancer care and research to communities across the country. AdventHealth Cancer Institute Shawnee Mission, located in Kansas, will serve as the network’s first national affiliate.
“This affiliation gives patients in the Kansas City area better access to the latest advances in cancer diagnosis and treatment, as well as opportunities to participate in clinical trials informed by our research,” said Mark Anderson, MD, PhD, Executive Vice President for Medical Affairs at the University of Chicago.
“By affiliating with a strong community-focused healthcare provider like AdventHealth Shawnee Mission, we can improve cancer outcomes in Kansas City and advance the science and practice of academic medicine nationwide.”
“This affiliation gives patients in the Kansas City area better access to the latest advances in cancer diagnosis and treatment, as well as opportunities to participate in clinical trials informed by our research.”
Mark Anderson, MD, PhD, Executive Vice President for Medical Affairs at the University of Chicago
UChicago Medicine will collaborate with AdventHealth Shawnee Mission in Merriam, to enhance cancer care in the Kansas City region by expanding access to advanced treatments, leading-edge clinical trials, and expert second opinions at a new facility that opened in May 2024. Called AdventHealth Cancer Institute, the state-of-the-art building includes chemotherapy infusion centers, pharmacy, imaging, lab services, and physical therapy – all in one location. It also features the region’s only MRI-guided radiation therapy. This new facility builds off more than a decade of care that was provided at the former AdventHealth Cancer Center Shawnee Mission.
The UChicago Medicine Cancer Network allows affiliates to remain independent while aligning with UChicago Medicine’s rigorous standards in clinical care, medical education, and biomedical research. Affiliates also support onsite research operations, offering their patients seamless access to UChicago Medicine-sponsored clinical trials and select industry trials.
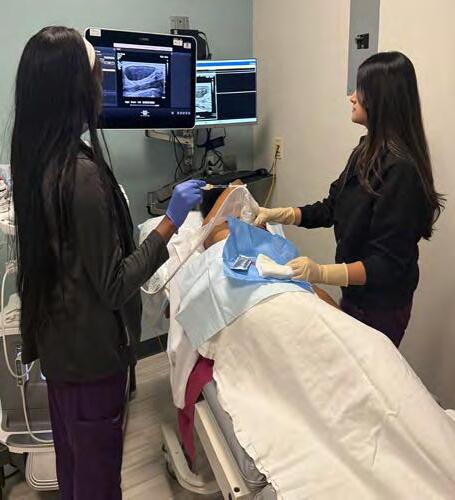
When a mammogram or ultrasound detects a highly suspicious lesion in the breast, patients usually have to wait up to three weeks for a biopsy to determine if they have breast cancer. To eliminate that agonizing wait, the University of Chicago Medicine launched BIOPSY NOW, a new initiative that provides same-day breast and lymph node biopsies for patients whose mammograms and ultrasounds suggest a very high likelihood of cancer. While the biopsy results still take a few days, the BIOPSY NOW program allows a patient to get all of their tests done in one visit. That means a faster diagnosis, and if necessary, a head start on treatment. UChicago Medicine is the only hospital system in the Chicago area to offer this patient-focused program. BIOPSY NOW, which was launched Sept. 15, also immediately schedules a patient to meet with a breast cancer surgeon and the surgical team, sometimes on the same day.
The University of Chicago Medicine’s focal therapy program for prostate cancer has completed more than 300 procedures — placing it among the highest-volume programs in the U.S. Focal therapies are new, minimally invasive procedures that precisely target tumors within the prostate. They mark a significant advancement from just a few years ago, when most prostate cancer cases were treated by removing or radiating the entire prostate gland.
The past approaches were “overtreatment,” in some cases, and often came with unwanted side effects, such as urinary incontinence or erectile dysfunction. With focal therapies, the prostate remains intact, minimizing or eliminating side effects and allowing for faster recovery times.
These treatments are provided through UChicago Medicine’s unique, multidisciplinary focal therapy program that offers each patient a nurse navigator who educates them on the therapy and the process, along with a collaborative team of radiologists, radiation oncologists and urologic oncologists. Additionally, UChicago Medicine researchers are currently running three clinical trials on focal therapies, one of which involves a new treatment that could soon be available to patients upon FDA approval.
UChicago Medicine is the only hospital in Chicago—and one of just a handful nationwide—to offer the full spectrum of focal therapies for prostate cancer.

A first-of-its-kind center for rare blood cancers has launched at the University of Chicago Medicine, thanks to a generous gift from philanthropists Mark and Maureen Miller. For the couple and their family, the gift is personal: A routine blood test two years ago revealed that Mark Miller’s white blood cell count was elevated. Soon after, doctors diagnosed him with chronic myeloid leukemia, or CML. Miller, a Colorado-based businessman with an illustrious career in companies related to medicine, began reaching out to experts and researching the disease. He turned to UChicago Medicine hematologist/ oncologist Michael Drazer, MD, PhD, who ultimately guided his care journey. During this time, he and Miller spoke about the understudied and underfunded field of rare blood cancers. Drazer shared his vision to lead a collaborative team that could focus on research and new therapies for these diseases — and to establish a hub for sharing data globally (a data commons) and multidisciplinary approach to support research across institutions. On the path to remission, Miller felt compelled to help. The Miller Family Center for Rare Blood Cancers, which began its work in July, builds on UChicago’s history in the study and treatment of CML. Miller and his family hope the center’s efforts will greatly expand knowledge in the field and help close gaps in research and care for patients with rare blood cancers.
Community Outreach and Engagement (COE) is central to the mission of the University of Chicago Medicine Comprehensive Cancer Center (UCCCC). It serves as a bridge between UCCCC and partners to facilitate community responsive research, education and policies that promote optimal care along the cancer continuum. Our Office of Community Engagement and Cancer Health Equity (OCECHE) is the driving force behind our COE efforts. In 2025, OCECHE held 175 community events.
OCECHE worked with leadership of the Cancer Prevention and Control Research Program and the Cancer Service Line network to create a shared governance model that provides an infrastructure for partnership with our Northwest Indiana catchment area partners. Through this model, OCECHE is pivoting its strong track record of health equity work and leveraging the clinical footprint of UChicago Medicine’s expanding cancer care network. In 2025, OCECHE invited representatives from multiple sectors including faith-based communities, community-based organizations, minority-serving institutions, health insurers, patient advocates, local government, public health agencies and safety net centers, to come to the UChicago Medicine Crown Point facility for a meet-and-greet. At the meeting, the group shared concerns about cancer-related issues in Lake County, Indiana, and decided that priority areas to focus on include cancer screening access and navigation and environmental exposures and cancer risk.


Through a joint grant with Northwestern Medicine and the Black Cancer Collaborative, OCECHE engaged both researchers and community members in research. The project was on sustainable, patient-centered strategies to engage the Black community in clinical trials. Researchers presented publications and research to advocates, community research advocates gave feedback on presentations, and together, the groups co-designed community events and created a video library of survivor stories.
The University of Chicago College campus and medical center are co-located in one physical space. Current campus policy only bans smoking within University buildings but permits smoking in designated spaces on campus. With 25-30% of the medical center’s staff living on the South Side, where about 60% of residents report using tobacco, there is an opportunity to make an impact on the health of the UChicago campus. Marcia Tan, PhD, MPH, received a two-year grant from the Truth Initiative to establish a 100% nicotine/ tobacco-free policy on UChicago’s campus and promote tobacco education and cessation.
Food insecurity is a growing concern for many, especially cancer patients. As a way to engage patients, promote clinical trials and also respond to the current food insecurity issues many patients are facing, the UCCCC Clinical Trials Support Office (CTSO) transformed Infusion Center lobbies into farmers markets at the UChicago Ingalls/Harvey and UChicago Flossmoor locations. Held during the week of Thanksgiving, the market provided patients, patient advocates, family, friends, and staff with almost 800 pounds of free fresh produce, including potatoes, carrots, onions, cranberries, apples, oranges and bananas. OCECHE provided cancer patients and their families with fresh laid eggs from their Urban Farm as well as sweet treats for Thanksgiving meals. The effort was funded in part by a Gilead grant and UCCCC COE.
Urban Farm, which launched April 2025 in West Pullman on Chicago's far South Side, is the result of an OCECHE community-academic partnership development mini grant (Academic Co-PI, Marcia Tan, PhD, MPH). The farm has a variety of fruit and vegetables as well as 12 hens that lay fresh eggs daily. The UChicago Medicine Urban Health Initiative nominated the Urban Farm as one of the sites for the Day of Service and Reflection and the farm was awarded two fruit trees by the Wood St. Collective environmental justice group. During the summer, farm labor is provided by community members and university faculty and staff. Produce and eggs are provided free of charge to those that provide farm labor, as well as cancer patients. Cancer patients were provided fresh laid eggs at the CTSO Farmers Market hosted at the UChicago Ingalls and UChicago Flossmoor Infusion Centers the week of Thanksgiving (See story above). Farm labor nor cancer status is mandatory to obtain items free of charge; however, a small donation is encouraged. In-kind and monetary donations for the farm can be provided by contacting communitycancer@uchicago.edu


During the summer, farm labor is provided by community members and university faculty and staff.
The UCCCC prioritizes science enrichment, research training and personalized mentorship for students as young as high school, giving them a jump-start on careers in cancer research and care. Through the Office of Education and Career Development, we train the next generation of cancer researchers and clinicians by providing robust educational and professional development opportunities for learners at all levels.
This year, UChicago joined the Emerging Cancer Scholars Exchange, a collaboration among the nation’s top cancer centers to provide early-career faculty speaking engagements at prestigious peer institutions. Phoenix Miao, PhD, Associate Professor in the Ben May Department for Cancer Research, was selected by the University of California San Francisco Helen Diller Family Comprehensive Cancer Center to present his work on dissecting stemnessinduced mechanisms of cancer immune resistance. Heather Whitney, PhD, Assistant Professor of Radiology, was selected to present her work on artificial intelligence in medical imaging at the University of Pennsylvania Abramson Cancer Center.




In April, the UCCCC partnered with the American Association for Cancer Research (AACR) to nurture the cancer-related career ambitions of young people from both local communities and countries across the globe. In coordination with the 2025 AACR annual conference, held in Chicago, the UCCCC co-led the Special Program for High School Students, exposing more than 300 Chicago youth to cancer science and careers. The UCCCC also hosted 13 AACR Global Scholars in Training for a half-day program featuring faculty talks, facilities tours, a poster roundtable session, and a networking reception to promote international collaborations.
On April 9, the UCCCC hosted its inaugural Trainee Associate Member Research Symposium to celebrate the scientific accomplishments of our graduate students, medical students, postdoctoral fellows, and clinical residents and fellows. Twenty-six trainees presented their work. Leah Ziolkowski (graduate student, Metabolism), Gia Jadick (graduate student, Medical Physics), and Samantha Yee, PhD (postdoctoral scholar, Department of Obstetrics & Gynecology), earned top honors. Professor of Computer Sciences Rick Stevens, PhD, delivered a keynote address highlighting novel drug discovery approaches through collaborations between the University of Chicago and Argonne National Laboratory. Trainee Associates will share new scientific insights at the 2026 symposium on April 10.




On May 7, UCCCC trainees joined the American Cancer Society Cancer Action Network (ACS CAN) for Illinois Cancer Action Day in Springfield. The group urged state legislators to support two cancer-focused policies: 1) Taxation of all tobacco products at a rate equal to that of regular cigarettes, and 2) $1M in state funds for colorectal cancer screenings and prevention. Their efforts were rewarded: The Illinois General Assembly included increased tax on tobacco products, including e-cigarettes, in the revenue bill for FY2026 and approved state funding for the screening and prevention of colorectal cancer across Illinois.




On August 8, the UCCCC celebrated the 10th anniversary of researcHStart, an immersive cancer research training program for Illinois high school students. The program was initiated and is led by the University of Chicago with partner sites at the University of Illinois Chicago, the University of Illinois UrbanaChampaign, Northwestern University, and Rush University. Among the 257 researcHStart alumni, nearly 70% remain committed to careers in biomedicine including cancer research and care. The celebration featured a keynote address by Eileen Dolan, PhD, researcHStart co-founder and UCCCC Deputy Director. Justin Banks, a secondyear at Pritzker School of Medicine, received the inaugural Ira R. Cohen ResearcHStart Alumni Award in recognition of his achievements in science, medicine, and service.



On October 4, the University of Chicago welcomed over 4,000 guests for the annual South Side Science Festival. There, UCCCC faculty, staff, and trainees hosted the popular “Cancer Corner,” which featured activities across the cancer research continuum. The main attraction was "Coco the Colon," an inflatable, walk-through colon to teach about gastro-intestinal diseases and encourage cancer screening. Other highlights included a "glow dough" sunscreen and UV demonstration, LEGO DNA mutation modeling, pipette watercolor painting, and pedal-powered smoothie blenders to promote a fiber-rich diet for colon health.


JOHN'S STORY
“I'm really happy this trial was available.”
After treatment for prostate cancer, semi-retired toy designer John Balogh is back to his favorite routine of taking long walks with his wife Linda along the shores of Lake Michigan near their home in Gary, Indiana. His outlook is bright after participating in a clinical trial called LO-RADS, which was developed by UChicago Medicine researchers to determine whether focusing higher doses of radiation directly at the cancer within the prostate — while lowering the dose to nearby healthy prostate tissue — can cure the disease with fewer side effects.
It’s a different approach to prostate cancer treatment, which has traditionally involved removing or radiating the entire prostate gland and can sometimes cause urinary urgency, bowel changes or sexual dysfunction.
As part of the trial, John had five radiation treatments per week for four weeks. Other than a brief episode of constipation, he experienced no complications — and more than a year later, he remains symptom- and cancer-free.
“I have other health issues, but as far as my prostate goes, I’m good,” John said. “I’m really happy this trial was available, and I was excited to be part of it.”
3D illustration of a T cell surface engineered with chimeric antigen receptors (CARs) (red and green structures), enabling the immune cell to recognize and bind specific target antigens.
Our more than 200 physician and scientists work closely together to make major advances in cancer research. Here are some highlights of discoveries from 2025.
In recent years, CAR-T cell therapy—where a patient’s own immune cells are harvested and genetically engineered to recognize a specific tumor antigen and attack cancer cells—has shown remarkable outcomes in the treatment of various cancer types. However, major challenges with this therapy include poor tumor penetration, toxic side effects, development of resistance mechanisms and the complex, patient-specific engineering process for each treatment. To address these limitations and improve cell therapy products, UChicago researchers, Anthony Kossiakoff, PhD, Otho S.A. Sprague Distinguished Service Professor of Biochemistry and Molecular Biology, and Ainhoa Arina, PhD, Research Associate Professor in the laboratory of Ralph Weichselbaum, MD, Daniel K. Ludwig Distinguished Service Professor and Chair of Radiation and Cellular Oncology, developed a new platform called GA1CAR. In this system, an engineered protein G variant (GA1) is fused to the T-cell receptor signaling machinery, and the part that recognizes tumor targets is delivered in the form of short-lived antibody pieces known as Fab fragments. This modular “split” design separates
the antigen-recognition element from the signaling machinery within the CAR-T cell, giving clinicians precise control over how, when, and where the engineered T cells attack cancer. This flexibility is particularly valuable in solid tumors, where the presence of multiple antigens within the same tumor has limited the effectiveness of singletarget therapies. In animal studies, GA1CAR-T cells performed the same as or better than conventional CAR-T cells, and while both therapies reduced tumor burden, GA1CAR-T cells showed greater activation and produced more inflammatory cytokines in response to the same antigen dose. Importantly, GA1CAR-T cells maintained their function over extended periods and could be reactivated weeks later with a fresh dose of Fab. This capability opens the door for repeatable therapy where the dose can be adjusted as needed, without having to create new T cells each time. The research team is now exploring ways to integrate radiation therapy with the GA1CAR platform and to develop next-generation Fab fragments that stay in the body longer and reach tumors more effectively.
High-grade serous ovarian cancer is the most common and deadliest form of ovarian cancer. It is often diagnosed at advanced stages and remains notoriously difficult to treat. Although surgery and chemotherapy can be effective initially, the cancer frequently recurs and shows strong resistance to immunotherapy. A research team led by Ernst Lengyel, MD, PhD, Arthur L. and Lee G. Herbst Distinguished Service Professor and Chair of Obstetrics and Gynecology, focused not on the cancer cells but on the tumor-supporting fibroblasts, known as cancer-associated fibroblasts (CAFs). These CAFs help tumors evade immune detection and promote metastasis. The team identified a key enzyme, nicotinamide N-methyl transferase (NNMT), which is highly active in CAFs. NNMT drives epigenetic and metabolic changes that convert normal fibroblasts into immunesuppressing CAFs. These altered CAFs release signals that transform monocytes into myeloidderived suppressor cells (MDSCs), which further
dampen the immune response. By inhibiting NNMT, researchers were able to reverse these effects, effectively reprogramming CAFs and reducing their tumor-supportive behavior.
In collaboration with the National Center for Advancing Translational Sciences and the NCI Experimental Therapeutics (NExT) program, the team screened over 150,000 compounds and developed a highly targeted NNMT inhibitor. In animal models, the inhibitor significantly reduced tumor burden. Most importantly, when combined with immune checkpoint inhibitors, the drug successfully halted tumor growth — a first in ovarian cancer research. This innovative approach marks a shift from traditional cancer therapies, focusing not on the tumor cells but on altering the tumor microenvironment. The findings offer a promising new strategy to overcome therapy resistance and could reshape the future of ovarian cancer treatment.
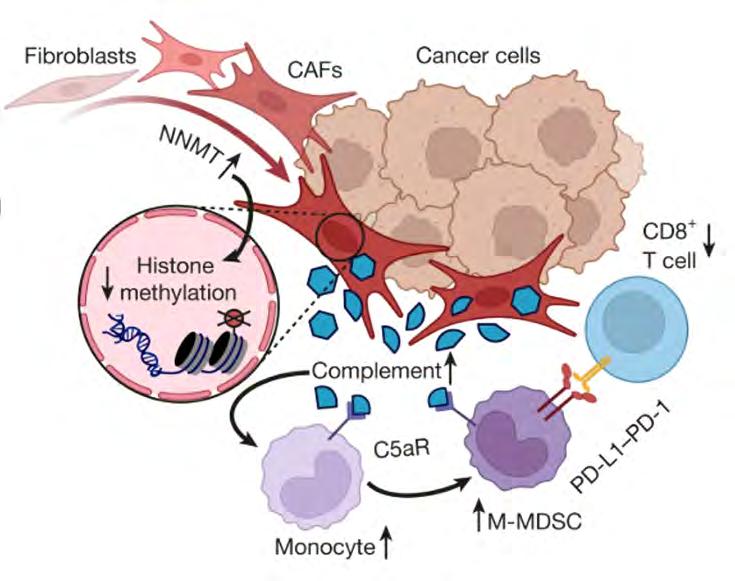
Image above: Schematic showing nicotinamide N-methyl transferase (NNMT)-expressing cancer-associated fibroblasts (CAFs) recruiting monocytic myeloid-derived suppressor cells (M-MDSCs) via complement factors to suppress T cells; NNMT inhibitors reduce this effect and restore CD8+ T cell function.
Liquid biopsies are non-invasive tests that identify signs of cancer through blood samples. They typically rely on detecting fragments of DNA shed by tumor cells. Although current DNA-based methods work well in later stages, they often fail to detect early-stage cancer because there is not enough circulating tumor DNA (ctDNA) present in the blood.
Chuan He, PhD, the John T. Wilson Distinguished Service Professor of Chemistry and Professor of Biochemistry and Molecular Biology, and his team have focused instead on circulating cell-free RNA (cfRNA), which better accurately reflects realtime cellular activity. RNA abundance alone can vary greatly based on timing and sample prep, making it less dependable; however, the team specifically studied RNA modifications, which are chemical changes that remain stable regardless of RNA abundance. This made them a more reliable biomarker for cancer detection. When tested on blood samples from colorectal cancer patients,
the new method detected cancer with 95% accuracy, even in its earliest stages.
Standard tests ignore signals from the gut microbiome, despite growing evidence of its role in cancer. The UChicago team not only measured RNA modifications from human cells but also discovered unique RNA changes in gut microbes near tumors. The microbiome turns over more rapidly than human cells, releasing modified RNA into the bloodstream earlier. By analyzing RNA modifications from both human and microbial sources, this test offers a highly sensitive, stable, and non-invasive approach to early cancer detection. It significantly outperforms current stool- or blood-based tests, which drop below 50% accuracy in early stages. He is now working to commercialize the test through UChicago’s Polsky Center for Entrepreneurship and Innovation, offering hope for earlier intervention and better patient outcomes.
Pancreatic ductal adenocarcinoma (PDAC) is one of the deadliest cancers, known for its aggressive nature and resistance to treatment. Part of the challenge lies in the complex makeup of the tumors, which consist of diverse cell types with varying gene expression patterns and behaviors. Understanding this transcriptional heterogeneity is critical for improving diagnosis and treatment. Researchers at the University of Chicago, led by Matthew Stephens, PhD, Professor of Human Genetics, and including Marc Bissonette, MD, developed a statistical method called Generalized Binary Covariance Decomposition (GBCD) to tackle this complexity. Using singlecell RNA sequencing (scRNA-seq) data from over 35,000 cancer cells across 59 PDAC patients, they uncovered not just known tumor subtypes (“classical” and “basal”), but also a previously hidden stress-response gene expression program that is strongly linked to poor survival outcomes.
GBCD allows researchers to identify patterns of gene activity across individual tumor cells, and
it detects both known and novel transcriptional programs that were previously missed in bulk sequencing data. But tumors are extremely variable between patients, making it difficult to detect shared patterns, even with modern tools. GBCD is specifically designed to cut through that noise, revealing meaningful signals hidden in large, complex datasets. Most notably, the team found that many genes in the stress-response program are regulated by ATF4, a key transcription factor in the integrated stress response pathway that helps cancer cells survive harsh environments. This program predicted worse outcomes independent of tumor stage or subtype, highlighting a powerful new biomarker. This study not only demonstrates the value of analyzing single-cell gene expression in PDAC but also introduces a method that could apply to many other cancers. GBCD provides a path forward for better identifying high-risk patients and developing more personalized, targeted therapies based on gene expression programs—not just genetic mutations.
A groundbreaking study led by Ralph Weichselbaum, MD, Daniel K. Ludwig Distinguished Service Professor and Chair of Radiation and Cellular Oncology, has revealed an unexpected side effect of high-dose radiation therapy in the treatment of metastatic cancer. Radiation is typically used to destroy tumors but sometimes it triggers the “abscopal effect” — where distant, untreated tumors shrink due to immune system activation. The researchers have now identified the opposite response where in some cases, tumors that weren’t directly treated with radiation actually grow following treatment, a phenomenon the researchers have coined the “badscopal effect.”
The study found that this effect is driven by a protein called amphiregulin, which is produced in higher amounts by tumors treated with high-dose radiation. Amphiregulin appears to suppress the immune system’s ability to fight cancer and makes tumor cells more resilient. It specifically promotes the accumulation of immunosuppressive myeloid cells and increases expression of CD47, a molecule that acts as a “don’t eat me” signal to prevent immune cells from destroying cancer cells. This not only allows existing metastases to
grow but also reduces the effectiveness of immunotherapy in combination with radiation.
Experiments in both patient samples and animal models confirmed that radiation increased amphiregulin levels, leading to tumor growth at untreated sites. However, blocking amphiregulin either with antibodies or through gene editing reduced tumor size and slowed metastatic progression. Even more promising, combining radiation therapy with blockers of amphiregulin and CD47 led to significant control of metastases in animal models.
These findings mark a potential paradigm shift in how radiotherapy is used, particularly for patients with advanced or metastatic cancer. Rather than seeing radiation solely as a local treatment, the study highlights its systemic effects and the possibility of counteracting its unintended consequences by targeting molecules like amphiregulin. The research team now plans to launch clinical trials to validate these results and explore personalized approaches to radiotherapy based on a patient’s biological response to treatment.
Image above: Illustration of a patient receiving targeted radiation therapy.
In the tumor microenvironment, cancer cells consume the majority of available nutrients to survive under low-oxygen and high-stress conditions, especially in aggressive cancers like pancreatic cancer. This creates a hostile environment for T cells, depriving them of essential nutrients and leading to dysfunction and exhaustion. Previous assumptions focused on nutrient shortages as the primary challenge for T cells in tumors, but a new study revealed a more surprising culprit.
Using a tool that measures hundreds of nutrient concentrations in tumors, the research team led by Alexander Muir, PhD, Assistant Professor in the Ben May Department of Cancer Research at UChicago at the University of Chicago, in collaboration with the researchers at the University of Pittsburgh, analyzed 118 key metabolites and discovered an unexpected buildup of an oncometabolite, phosphoethanolamine, rather than a deficiency in nutrients. This metabolite was found in abnormally high levels across both human and mouse tumors and was shown to suppress T cell function, limiting their ability to recognize and attack cancer cells. This suggests tumors may actively accumulate phosphoethanolamine as a strategy to evade immune detection. The study sheds light on how the metabolic environment
within tumors can hinder the function of T cells, which are crucial for targeting and eliminating cancer. This discovery opens the door to improving cancer immunotherapy by targeting the metabolic conditions that impair immune responses.
The study findings have important implications for cancer immunotherapy, which often relies on reactivating T cells to attack tumors. Many patients don’t respond to these treatments due to T cell suppression, and this research points to a new explanation which is the presence of immunosuppressive metabolites like phosphoethanolamine. Measuring such metabolites could serve as a diagnostic tool and guide the development of next-generation therapies aimed at restoring immune function. The team is now focused on understanding why phosphoethanolamine accumulates in tumors and developing strategies to block its immunosuppressive effects. They hope this metabolite could serve as a biomarker for tumor burden or immune suppression, helping identify which patients might benefit most from immunebased therapies. Ultimately, the goal is to create therapies that reduce phosphoethanolamine levels, enhancing T cell performance and boosting the success of immunotherapy in cancer treatment.
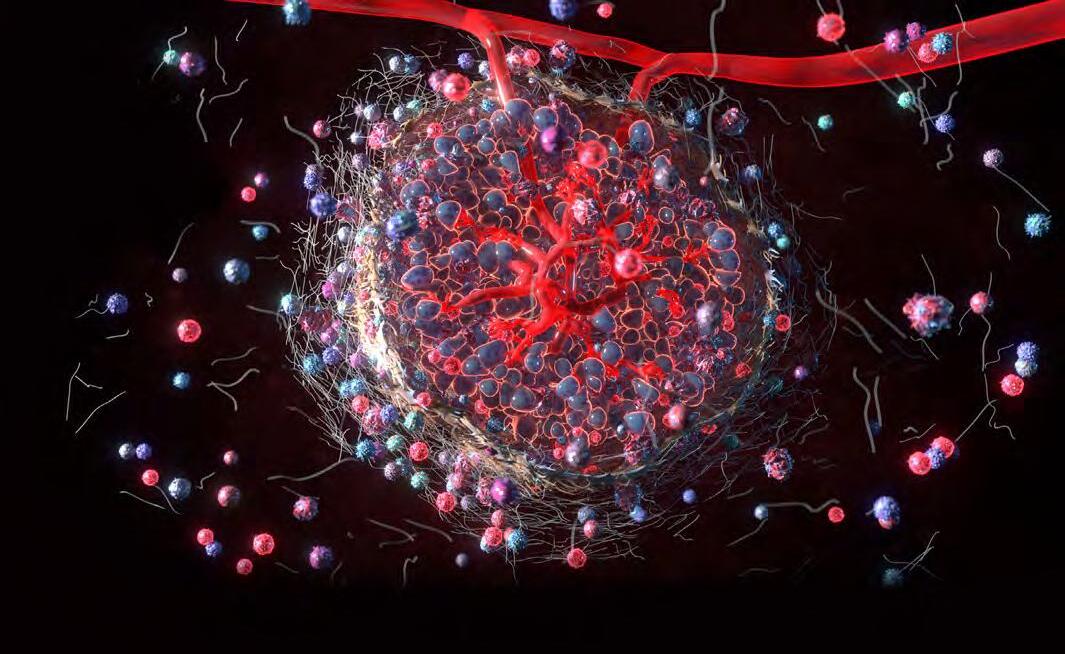
3D illustration of the tumor microenvironment, showing complex cellular interactions among immune cells, stromal components, and extracellular matrix, highlighting support for tumor growth and immune evasion.
Conceptual illustration of AI-driven cancer drug discovery.

A research team led by Evgeny Izumchenko, PhD, Assistant Professor of Medicine at the University of Chicago Medicine, in collaboration with InSilico Medicine and Prelude Therapeutics, have identified a promising new therapeutic target for adenoid cystic carcinoma (ACC), a rare and difficult-totreat cancer of the salivary glands. Using artificial intelligence to analyze tumor gene expression, the team pinpointed PRMT5, an enzyme involved in epigenetic regulation, as a key driver of ACC and a potential treatment target.
Testing a selective PRMT5 inhibitor (PRT543) developed by Prelude Therapeutics, the researchers found that blocking PRMT5 significantly reduced tumor growth in various preclinical models, including cell lines, organoids, and patient-derived xenografts. The treatment downregulated critical cancer-related genes like
MYB and MYC and showed effectiveness regardless of the tumor's NOTCH1 mutation status, a marker of aggressive disease.
Although PRT543 showed tumor-suppressing effects, it was not curative on its own. To enhance efficacy, the researchers tested lenvatinib, an existing multi-kinase inhibitor, in combination with PRT543. The combo showed stronger tumor inhibition in vitro. Importantly, some patients with high levels of PRMT5, MYC, and MYB — along with elevated lenvatinib target genes — may benefit most from this combination therapy, while others could respond to PRMT5 inhibitor alone. This study not only identifies a viable new target for ACC but also highlights the move toward personalized, targeted treatment strategies that reduce side effects while improving patient outcomes.
The UChicago Cancer Registry collects, maintains and reports detailed information for pediatrics and adult patients diagnosed with, and treated for, cancer at the University of Chicago Medicine in Hyde Park, the University of Chicago Comer Children’s Hospital and the University of Chicago Medicine Comprehensive Cancer Center at Silver Cross Hospital. A dedicated team of data management specialists provides important data services to advance basic, translational, and clinical research at our nationally recognized, American College of Surgeons-approved clinical cancer program.
Registry data are essential for not only advancing research to improve patient care, but also for cancer program development, community outreach activities and decision-making in oncology and public healthcare policies.
Please note: the data presented here are from 2024. Data from 2025 will be available later in 2026.
In 2024, 5,671 patients were diagnosed and/or treated at the University of Chicago Medicine. This included both malignancies and benign central nervous system neoplasms. Of these, the majority of patients (4,763, 84%) were newly diagnosed, and the remaining (908, 16%) are documented in the registry for recurrent or progressive disease or those that obtain a diagnostic confirmation at UChicago Medicine. Of those seen, 5,497 (97%) were adults (26-100+ years) at diagnosis and (174, 3%) were 0-25 years.
The most frequently seen cancers for adult patients representing 21%, (1,172), were of the genitourinary (GU) system. For pediatric and adolescent and young adult patients, endocrine (40 patients, 23%) were the most frequently seen cancers.
Notes: Endocrine includes benign pituitary adenomas. Neuro Oncology includes benign central nervous system neoplasms. Other includes blood dyscrasias, myelodysplastic/myeloproliferative syndromes, other skin, mesotheliomas, and cancers with other histology/primary site designations.
Distribution by race and gender shows that our patient population is predominately White (3,533 patients, 62%), followed by Black (1,457 patients, 26%) and Hispanic (398 patients, 7%). There were 2,720 males and 2,951 females.
5,671
Patient Geographics
The majority of patients (83%) seen in 2024 were Illinois residents. Nearly 17% (976 patients) of the total number of patients first seen in 2024 lived in other states, primarily in the neighboring states of Indiana (14%), Michigan (1%) and Wisconsin (1%). We also served five international patients. 625726 7+26 5 62
The following UCCCC members were recognized by receiving distinguished honors and awards in 2025:
Mark Applebaum, MD, Associate Professor of Pediatrics, was honored with the The Fellow of the American Society of Clinical Oncology (FASCO) designation for extraordinary volunteer service, engagement, and dedication to ASCO.
Joyce Chen, PharmD, PhD, Assistant Professor in the Pritzker School of Molecular Engineering and Ben May Department for Cancer Research, was named to the Scientific Advisory Board of the Lung Cancer Research Foundation.
The Lymphoma Research Foundation honored Adam DuVall, MD, MPH, Assistant Professor of Medicine and Pediatrics, with the Hope Award for his work treating and supporting patients.


Thomas F. Gajewski, MD, PhD, AbbVie Foundation Professor of Pathology, received a Champion of SITC Award by the Society for Immunotherapy of Cancer (SITC). He was also honored with The Distinguished Scientist Award from the Association of American Cancer Institutes (AACI).
Kunle Odunsi, MD, PhD, AbbVie Foundation Distinguished Service Professor of Obstetrics and Gynecology, Director, UCCCC and Dean for Oncology, Biological Sciences Division, was the 539th speaker for the University of Chicago’s Convocation ceremony.
Sonia Kupfer, MD, Professor of Medicine; Megan McNerney, MD, PhD, Associate Professor of Pathology; and Akash Patnaik, MD, PhD, MMSc, Associate Professor of Medicine, were elected to the American Society for Clinical Investigation, one of the oldest and most esteemed nonprofit honor societies of physician-scientists.
Mitchell C. Posner, MD, Thomas D. Jones Distinguished Service Professor of Surgery, was honored by the National Pancreas Foundation with the prestigious Legacy Award for his outstanding clinical excellence, leadership and lasting impact in the field of surgery.

Jeffrey Rathmell, PhD, Chair of the Ben May Department for Cancer Research and Director of the Ludwig Center at the University of Chicago, has been named the recipient of the Cancer Research Institute (CRI) 2025 Frederick W. Alt Award for New Discoveries in Immunology. The Alt Award honors a former CRI postdoctoral fellow whose work has had a significant impact on the field of immunology and who has achieved excellence in academic or industry leadership.
Sonali M. Smith, MD, FASCO, Elwood V. Jensen Professor of Medicine and Section Chief of Hematology/Oncology at UChicago Medicine, was elected to the American Society of Clinical Oncology (ASCO) Board of Directors, with her term beginning in June 2025.
Wendy Stock, MD, Anjuli Seth Nayak Professor of Medicine, was named a "Giant in Cancer Care" by OncLive for her work in leukemia.
Kal Tsanov, PhD, Assistant Professor of Medicine, Alex Muir, PhD, Assistant Professor in the Ben May Department for Cancer Research, and Ari Rosenberg, MD, Assistant Professor of Medicine, were named "40 Under 40 in Cancer" winners.
The following individuals were named one of Clarivate’s Highly Cited Researchers of 2025 (in the top 1% of cited researchers):
• Ralph Weichselbaum, MD
• Chuan He, PhD
• Thomas Gajewski, MD, PhD
• Jeffrey Rathmell, PhD
• Tao Pan, PhD
• Wenbin Lin, PhD
• Matthew Stephens, PhD
• Eugene Chang, PhD
Frederick Howard, MD, received a Rising Stars Award at the San Antonio Breast Cancer Symposium.

CRYSTAL'S STORY
By the time Crystal Hendricks-Kretzer came to University of Chicago Medicine in June 2023, a tumor in her left breast had spread to her lymph nodes. She decided to enroll in the I-SPY2 clinical trial, a long-running, nationwide study aimed at developing targeted, personalized breast cancer treatments. The study uses gene expression data to identify biomarkers that can predict which tumors are most likely to respond to specific treatments.
“The patient voice is at the center of all cancer research.”
Crystal received a novel antibody-drug conjugate treatment followed by 12 weeks of standard chemotherapy. After that, doctors failed to detect any cancer in her MRI scans. Her doctor commented that Crystal had an amazing response to her therapy and her disease melted away. While many targeted drugs are now available to cancer patients, clinical trials offer access to cutting-edge treatments that may offer even more precision.
“It’s important that people know about their options when it comes to clinical trials, and it’s critical that the patient voice is at the center of all cancer research,” Crystal said. “I realized that the only reason I had access to the amazing treatments I did was because of the women who participated in trials a decade ago.”
CREDITS
Editor: Jane Kollmer
Writers: Jamie Bartosch
Chandrika Abburi, PhD
Matt Wood, PhD
Editorial Advisors: Kunle Odunsi, MD, PhD
M. Eileen Dolan, PhD
Kathleen Goss, PhD
Designer: SBDWorks, Inc.



