Conference & Symposium
May 5-8, 2026
JW Marriott Las Vegas Resort & Spa l Las Vegas, Nevada













May 5-8, 2026
JW Marriott Las Vegas Resort & Spa l Las Vegas, Nevada












l American Board of Bioanalysis (ABB)
l AAB Board of Registry (ABOR)
l Associate Member Section (AMS)
l College of Reproductive Biology (CRB)
l Environmental Biology & Public Health Section (EBPH)
l National Independent Laboratory Association (NILA)
Association Management
Tyler Anderson
Marilyn Barbour, M.Ed., BSMT(ASCP)
Mark S. Birenbaum, Ph.D.
Sam Constantine
Robert Footlik, M.S., MLS(ASCP), HCLD(ABB-Ret.)
Josh Hampton
LeAnn Hampton
Kayla Harless
Vauna Harless
Lisa Huelsing
Tara Lambert
Kelli Lowry
Barbara Medina
Evan Meyers
AAB Program/MolDx Symposium...14.5 Contact Hours
CRB Workshop 8 Contact Hours
CRB Symposium 20 Contact Hours
CAP Inspector Training................. 5.5 Contact Hours
The Workshop, CRB Symposium, and AAB/AMS Technical Program have been approved by PEER (American Board of Bioanalysis).
In addition, contact hours earned can be used to satisfy CE requirements in the states of California, Florida, Louisiana, and Montana.
If you are licensed in the state of Florida please return the Florida CE form to the registration counter.






Welcome to Las Vegas and the 70th AAB and 30th CRB Anniversaries. We are excited to see you and look forward to sharing some history, creating new memories, and scoping out the future.
As we celebrate the special AAB and CRB anniversaries, take some time to learn about the history of these two groundbreaking groups. Follow the links for all the details: History of AAB; History of CRB.
You’ll have several chances to win cash, gift cards, and complimentary meeting registrations for AAB/CRB 2027: Hide-and-Seek AAB/CRB Anniversary Coins, Sponsor Passport, and attendance prize drawings for cash at the receptions. See the flyer in your registration packet or stop by the registration desk for more information.
The AAB and CRB program committees have organized outstanding educational programs featuring a diverse range of speakers, roundtable discussions on a variety of topics, and oral and poster abstracts covering new research and scientific developments. See the schedule of events for a complete list of educational sessions and activities (pages 16-21).
We want to thank the program committees for their hardwork organizing the educational programs and social activities.
Special thanks to our friends and colleagues at Nexpring Health for supporting the CRB Symposium attendance of two CRB members through the CRB Diarmaid Douglas-Hamilton Memorial Scholarship Awards. Nexpring Health created the ongoing scholarship program in honor of the remarkable legacy of the late Diarmaid Douglas-Hamilton, chief technology officer, senior vice president of research and development, and co-founder of Hamilton Thorne. Congratulations to the 2026 scholarship recipients – Morgan Orsolini, MS, PhD Candidate, and Claire Porter, junior embryologist.
Thank you to LifeAire Systems and Birenbaum & Associates for their continuing support of the educational component of the AAB Conference/CRB Symposium by providing four $500 best abstract awards – two for ART/IVF abstracts and two for traditional laboratory abstracts.
Thank you to all the sponsors supporting AAB/CRB 2026. We very much appreciate your participation. Help us thank the companies who help make the AAB/CRB educational programs possible. Visit the representatives of all the sponsor companies this week to see the latest science and technology and thank them for supporting this year ’s educational programs.
AAB and CRB are using a digital format for materials again this year, including the Official Program Book, CRB Symposium Syllabus, and the AAB Conference Syllabus. In addition to reducing waste, the new digital formats allow the most up-to-date material and easier interaction with the presenters’ slide decks. Download the free “AAB Events” app through the iOS or Google Play store and login with your credentials from the AAB website. If you do not have the mobile app, login through the event portal at www.aab.org to access the materials. Check in at the registration desk if you have any questions.
See the schedule of events on pages 16-21 for dates/times for all educational programming and group activities.
We look forward to visiting with you. If there is anything we can do to make your stay more enjoyable, please let us know.
Best regards,
Annette Iacono AAB President
Matthew Hudnall, M.Sc., TS(ABB) CRB President
Start Building Your Lab.
You already know you need lab equipment. You just need the right partner to deliver it.

One partner. Total accountability.
Planning. Procurement. Installation. Training.
All handled by Nexpring Health.
One vendor. One quote. One point of contact
Flexible timelines that adapt as plans evolve
Access to the right equipment for your lab
Built by teams who understand real embryology workflows
Planning a lab build or expansion?
Let’s talk.
Donald P. Evenson, 84, of Brookings, SD, passed away in March 2025. Dr. Evenson was an internationally renowned scientist for pioneering the assessment of sperm DNA damage as related to human and animal male factor infertility and toxicology. He and his colleagues published a couple hundred manuscripts and book chapters on this topic.
Dr. Evenson was an AAB member for over 25 years and was certified by ABB for over 23 years. He regularly attended the AAB educational conferences and CRB symposia and his company, SCSA® Diagnostics, supported the annual AAB/CRB educational programs for many years.
In 1964, Dr. Evenson received his B.A. degree in biology/ chemistry from Augustana College in Sioux Falls, SD. In 1968, he received his Ph.D. in cell and molecular biology at the University of Colorado. This was followed by a twoyear postdoctoral study of mammalian DNA structure at the Institute of Molecular Biophysics, Florida State University, then two years at the Union Carbide Research Institute, Tarrytown, New York, studying the DNA structure of a cancer virus. He then served a dual faculty appointment for eleven years at the Memorial Sloan Kettering Cancer Center and Cornell Graduate School of Medical Sciences in New York City. During this time, he studied the effects of

chemotherapy on sperm quality. His invention of the Sperm Chromatin Structure Assay (SCSA®) is now used around the world in numerous infertility clinics.
In 1983 Dr. Evenson joined the faculty at South Dakota State University as professor of biochemistry, where over the course of more than 25 years he continued to further develop the SCSA® protocol and study its significance in relation to male factor infertility in humans, pigs, horses, cattle, and zoo animals.
Dr. Evenson received, continuously over 25 years, NIH Research Project Grants and grants from other national agencies. His SCSA® test is internationally recognized as the first proven commercial test of sperm DNA integrity.
Dr. Evenson was president and director of SCSA® Diagnostics in Brookings, South Dakota. He was an adjunct professor in the Department of Obstetrics and
Gynecology, Sanford Medical School, University of South Dakota, Sioux Falls, South Dakota, up until the time of his death.
Dr. Evenson authored more than 200 peer-reviewed publications that have been cited in other publications more than 14,000 times. He was a member of various professional societies and served as an expert peer reviewer for various scientific journals. Dr. Evenson received multiple awards during his tenure at South Dakota State University, including the title of distinguished professor, and he received lifetime achievement awards from Augustana College and the International Animal Andrology Association.
He also carried out one-year sabbaticals together with his wife, Carol, at the Norwegian National Hospital in Oslo, Norway, and the Department of Animal Science, University of Sydney, Australia.
Dr. Evenson is survived by his wife, Carol, three children, Kay (Rick) Kasperson, Paul (Jen) Evenson, Mark (Meredith) Evenson, three grandchildren, Shamus (Cassie) Evenson, Grace Evenson, Zoe Evenson (John Mosher) and two great grandchildren, Soren Evenson, Freya Mosher, sister Bernice (Al) Teixeira and many nieces and nephews.





































PRESIDENT
Annette Iacono Brookside Clinical Laboratory Aston, PA
FIRST VICE PRESIDENT
Robert Bowen, M.D. Physicians Laboratory Services, Inc. Omaha, NE
SECOND VICE PRESIDENT
Zahra Kashi, Ph.D., HCLD/CC(ABB) Kashi Clinical Laboratories, Inc. Portland, OR
THIRD VICE PRESIDENT
Estrella Rosemberg, M.Sc., ELD/TS(ABB) Palm Beach Fertility Center Hollywood, FL
SECRETARY/TREASURER
John J. Weiss, Ph.D. Allermetrix, Inc. Franklin, TN
AAB-MLE PROFICIENCY TESTING CHAIRMAN
Warren Eric Vanderslice, Ph.D. AAB-MLE Proficiency Testing Houston, TX

ASSOCIATE MEMBER SECTION (AMS) LIAISON REPRESENTATIVE
Salu Ribeiro, M.Sc., TS(ABB), ELS/ ALS(AAB) Bay Area PL Services San Francisco, CA
REGIONAL ADMINISTRATIVE SECRETARIES
Southern Region
Michael Stout, Ph.D., HCLD/ELD/TS/ CC/ALD(ABB) Aspire Fertility Dallas Dallas, TX
Midwestern Region
Melanie Clemmer, Ph.D., HCLD(ABB) WVU Center for Reproductive Medicine Morgantown, WV
Eastern Region
Robert Middleberg, Ph.D. NMS Labs Horsham, PA
Western Region
Marlane Angle, Ph.D., HCLD/CC(ABB) Port Hadlock, WA
MANAGER/ SUPERVISOR REPRESENTATIVE
Daniel Dasig, M.Sc., TS(ABB) Kaiser Permanente Los Gatos, CA

COLLEGE OF REPRODUCTIVE BIOLOGY (CRB)
PRESIDENT
Matthew Hudnall, M.Sc., TS(ABB) Naperville, IL
NATIONAL INDEPENDENT LABORATORY ASSOCIATION (NILA)
CHAIR
John J. Weiss, Ph.D (Allermetrix, Inc.)
ENVIRONMENTAL BIOLOGY & PUBLIC HEALTH SECTION (EBPH)
CHAIR
Charles Edward Gilbert, Ph.D., M.Sc. East Moriches, NY




Sperm Test offers fast, visual results on samples, in addition to key parameters by a clinically validated, at-home screening solution that empowers patients with accurate, preliminary semen quality insights.
+ Vu is a compact and efficient CLIA-Waived semen analyzer built for streamlined labratory environments. Reliable, automated results without complex setup or extended training.
Edition reporting.
Tuesday, May 5, 2026
7:00 a.m. – 5:30 p.m. Registration Valencia Ballroom
7:00 a.m. – 5:00 p.m. American Board of Bioanalysis Meeting Murcia Room
8:00 a.m. – 4:00 p.m. ABB Certification Examinations Grand Ballroom
8:00 a.m. – 4:00 p.m. ABOR Certification Examinations Cataluna Room
8:00 a.m. – 12:00 p.m. CRB Workshop: Beyond the Standard Marquis Room 1:00 p.m. – 5:30 p.m. CRB Workshop (continued) Marquis Room
Wednesday, May 6, 2026
7:00 a.m. – 7:30 p.m. Registration Valencia Ballroom
7:00 a.m. – 8:00 a.m. Networking Breakfast Valencia Ballroom
8:00 a.m. – 10:15 a.m. CRB Symposium Grand Ballroom CDE
8:00 a.m. – 10:15 a.m. MolDx Symposium Cataluna Room
10:15 a.m. – 10:45 a.m. CRB and MolDx Break Valencia Ballroom
10:45 a.m. – 11:45 a.m. CRB Symposium Grand Ballroom CDE
10:45 a.m. – 11:45 a.m. MolDx Symposium Cataluna Room
11:45 a.m. – 1:15 p.m. Lunch and Scientific Roundtable Presentations Marquis Room
1:15 p.m. – 2:45 p.m. MolDx Symposium Cataluna Room
1:15 p.m. – 3:00 p.m. CRB Symposium Grand Ballroom CDE
2:45 p.m. – 3:30 p.m. MolDx Break Valencia Ballroom
3:00 p.m. – 3:30 p.m. CRB Break Valencia Ballroom
3:30 p.m. – 6:00 p.m. CRB Symposium Grand Ballroom CDE
3:30 p.m. – 6:00 p.m. MolDx Symposium Cataluna Room
6:00 p.m. – 7:30 p.m. “Seis de Mayo” Reception for All Conference/Symposium Attendees Valencia Ballroom
Thursday, May 7, 2026
7:00 a.m. – 7:30 p.m. Registration Valencia Ballroom
7:15 a.m. – 7:45 a.m. AAB Business Meeting Cataluna Room
7:30 a.m. – 8:30 a.m. Networking Breakfast Valencia Ballroom
8:30 a.m. – 10:20 a.m. CRB Symposium Grand Ballroom CDE
8:30 a.m. – 10:20 a.m. MolDx Symposium Cataluna Room
10:20 a.m. – 10:50 a.m. CRB and MolDx Break Valencia Ballroom
10:50 a.m. – 11:45 a.m. CRB Symposium Grand Ballroom CDE
10:50 a.m. – 11:45 a.m. MolDx Symposium Cataluna Room
11:45 a.m. – 1:15 p.m. Lunch and Sponsor-associated Roundtables Marquis Room
1:15 p.m. – 2:15 p.m. CRB Symposium Grand Ballroom CDE
1:15 p.m. – 2:15 p.m. MolDx Symposium Cataluna Room
2:15 p.m. – 2:45 p.m. CRB Business Meeting/Awards Valencia Ballroom
2:45 p.m. – 3:30 p.m. CRB and MolDx Break Valencia Ballroom
3:30 p.m. – 5:00 p.m. MolDx Symposium Cataluna Room
3:30 p.m. – 6:00 p.m. CRB Symposium Grand Ballroom CDE
6:00 p.m. – 7:00 p.m. Networking Reception Valencia Ballroom
Friday, May 2, 2025
7:00 a.m. – 12:30 p.m. Registration Valencia Ballroom
7:45 a.m. – 9:00 a.m. Networking Breakfast Valencia Ballroom
8:30 a.m. – 10:00 a.m. CRB Symposium Grand Ballroom CDE
10:00 a.m. – 10:30 a.m. CRB Break Valencia Ballroom
10:30 a.m. – 12:00 p.m. CRB Symposium Grand Ballroom CDE
Key insights from Reprotech’s From Legacy Processes to Leading Performance whitepaper.
Clinic A is a high-volume fertility clinic with 30+ years of experience and 1,300+ annual retrievals, managing a large legacy cryostorage inventory through fragmented systems that created growing inefficiencies in billing, consent, and inventory management.
Clinic A implemented Reprotech’s Cryologix (CLX) platform to centralize cryostorage operations across billing, consent management, inventory tracking, and off-site storage.
Cryologix delivered measurable results across the areas that matter most: patient engagement, revenue performance, and operational efficiency.
full tanks of nonrevenue specimens
WHY IT MATTERS
Clinic A’s experience demonstrates that when cryostorage is treated as strategic infrastructure not an afterthought clinics see measurable gains in: - Patient trust and engagement - Regulatory and documentation compliance - Space utilization and staff efficiency - Long-term, scalable revenue
LEARN MORE
For detailed data, phased implementation insights, and full financial analysis, read the complete whitepaper at cryo-logix.com/for-clinics Or scan the QR code.


Email Danika Batastini, Vice President of Partner Operations, at dlb@reprotech.com to learn how Cryologix can support your clinic’s growth.
Tuesday, May 5, 2026
Valencia Ballroom 7:00 a.m.-5:30 p.m.
Wednesday, May 6, 2026
Valencia Ballroom
7:00 a.m.-7:30 p.m.
Thursday, May 7, 2026
Valencia Ballroom
7:00 a.m.-7:30 p.m.
Friday, May 8, 2026
Valencia Ballroom 7:00 a.m.-12:30 p.m.
Tuesday, May 5, 2026
7:00 a.m.-5:00 p.m.
American Board of Bioanalysis Murcia Room
Wednesday, May 6, 2026 6:00 p.m.-7:30 p.m.
Seis de Mayo Reception and Poster Session Valencia Ballroom
Thursday, May 7, 2026
6:00 p.m.-7:00 p.m.
Networking Reception Valencia Ballroom
Thursday, May 7, 2026 2:15 p.m.-2:45 p.m.
College of Reproductive Biology Grand Ballroom CDE

Thursday, May 7, 2026 7:15 a.m.-7:45 a.m.
AAB Business Meeting Cataluna Room




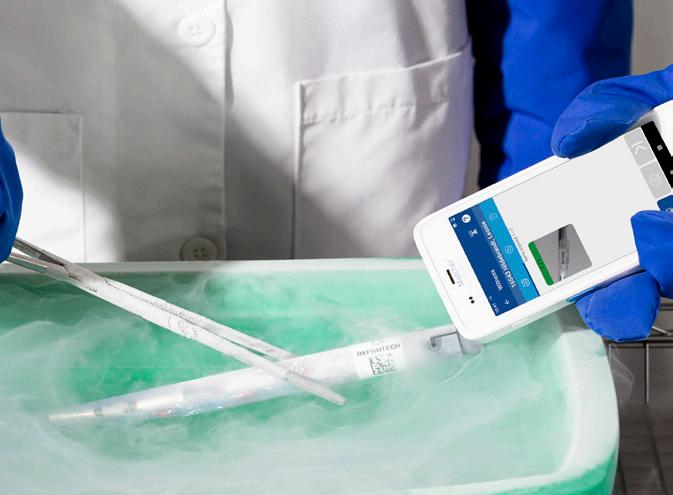
A seamless, end-to-end digital chain of custody, from lab to long-term storage and back.

Matcher and Reprotech bring together complementary technologies to deliver a fully connected, end-to-end digital chain of custody for IVF centers. Powered by Reprotech’s SmartSleeve™ and IMT Matcher™’s barcodebased witnessing and traceability system, the solution enhances safety, strengthens identification, and integrates seamlessly into existing clinic workflows, from on-site processes to secure, off-site cryostorage.
From clinic to cryostorage and back again, every step is traceable and transparent. The result is a safer, fully integrated workflow that supports clinics and patients with greater confidence in every stage of the cryostorage journey.
Reprotech is a trusted leader in long-term cryostorage innovation.

IMT Matcher is a barcode-based electronic witnessing system.

Stop by our AAB booth to learn more about Matcher and Reprotech!
Together with Reprotech’s Smartsleeve, we create an end-to-end digital chain of custody solution.
imtmatcher.com reprotech.com
May 5, 2026 ▪ JW Marriott Las Vegas Resort & Spa
TIME PRESENTATION/ACTIVITY
7:45-8:00 am Welcome and Introductions
8:00-9:00 am Overview of Current Clinical Strategies, Processing and Cryopreservation for Surgically Extracted Sperm
9:00-9:15 am Questions and Answers
9:15-10:15 am Experimental Methods to Preserve and Restore Fertility with Testicular and Ovarian Tissues
10:15-10:30 am Questions and Answers / Break
10:30-11:30 am Overview of Current Techniques for Ovarian and Testicular Tissue Freezing
11:30-12:00 pm Alternatives to TESE for Non-Obstructive Azoospermia and Advances in Sperm Vitrification Devices
12:00-12:15 pm Questions and Answers
12:15-1:00 pm Lunch On Your Own
SPEAKER
Eva Schenkman, Ph.D., M.Sc., HCLD/ELD/TS/CC/ALD(ABB)
Wayne Caswell, M.S., CLS(NCA)
Kyle Orwig, Ph.D.
Kyle Orwig, Ph.D.
Eva Schenkman, Ph.D., M.Sc., HCLD/ELD/TS/CC/ALD(ABB)
1:00-1:30 pm Ovarian Tissue Dissection Demonstration Byron Chuan, M.S.
1:30-2:00 pm TESE Tissue Biopsy Processing Demonstration
2:00-2:30 pm Processing of Testicular Tissue from Prepubertal Boys Demonstration
2:30-5:30 pm Hands-On Practice Stations
Wayne Caswell, M.S., CLS(NCA)
Byron Chuan, M.S.
Anthony Anderson, D.Sc., M.S., M.Sc., ELD(ABB)
Wayne Caswell, M.S., CLS(NCA)
Byron Chuan, M.S.
Kyle Orwig, Ph.D.
Eva Schenkman, Ph.D., M.Sc., HCLD/ELD/TS/CC/ALD(ABB)
Debbie Venier M.Sc., TS(ABB)
William Venier, M.Sc., ELD/TS(ABB)





EXPERT-LED ADVANCED A.R.T.
L AB CERTIFICATE COURSES IN:
Andrology
Embryology
ICSI
PG T-A/Embryo Biopsy
Vitrification
INVOcell
OUR STUDENTS LEARN
The latest IVF Lab techniques and procedures
Didactic knowledge behind procedural training
Current IVF lab operations, management, quality control, and safety measures
Advanced procedures using donated human oocytes, sperm, and embryos

ABB-APPROVED




of
As of April 29 , 202 6 v1.4 Tuesday, May 5 , 202 6 Time Pre –Conference Events
7:00 a.m. –5:30 p.m. Conference Registration Valencia Ballroom 7:00 a.m. –5:00 p.m. American Board of Bioanalysis (ABB) Meeting
8:00 a.m. –4:00 p.m. American Board of Bioanalysis (ABB) Certification Examination s Grand Ballroom AB CDE 8:00 a.m. –4:00 p.m. AAB Board of Registry (ABOR) Certification Examination s Cataluna Room
8:00 a.m. –5 : 3 0 p.m. CRB Workshop : Beyond the Standard: Advances in Testicular and Ovarian Tissue Preservation and Cryobiology –A HandsOn Workshop
Wednesday, May 6 , 202 6
AAB Conference –MolDx Symposium Cataluna Room CRB Symposium –“Looking Back and Moving Forward” Grand Ballroom CDE 7:00 a.m. –7:30 p.m. Conference Registration Valencia Ballroom 7:00 a.m. –8:00 a.m. Continental Breakfast
Grand Ballroom CDE
Valencia Ballroom 8:00 a.m. –8:15 a.m. Welcome and Celebration of 30 Years of CRB and 70 Years of AAB Melanie Clemmer, Ph.D., HCLD(ABB)
Grand Ballroom CDE
Cataluna Room IVF: The Way It Was Michael Vernon, Ph.D., HCLD(ABBRet)
Cataluna Room History of CRB Brooks Keel, Ph.D., HCLD(ABBRet)
8:15 a.m. –9:15 a.m. Currently Available MolDx Technologies Roya Pedram, Ph.D., HCLD/TS(ABB )
9:15 a.m. –10:15 a.m. MolDx Testing for Infectious Diseases Mark Pandori, Ph.D., HCLD(ABB)
Grand Ballroom CDE 10:15 a.m. –10:45 a.m. Break sponsored by Noblegen Cryogenics
Valencia Ballroom 10:45 a.m. –11:15 a.m. MolDx Testing Challenges in Public Health Laboratories Leah Gillis, Ph.D., HCLD(ABB)
Grand Ballroom CDE 11:15 a.m. –11:45 a.m.
Grand Ballroom CDE
Cataluna Room Progress, Partnership and Purpose: The Road Ahead for CRB Matthew Hudnall, M.Sc., TS(ABB)
Getting it Right: ART Laboratory Coding Melanie Clemmer, Ph.D., HCLD(ABB)
Wednesday, May 6, 2026 (continued)
CRB Symposium –“Looking Back and Moving Forward” Grand Ballroom CDE
AAB Conference –MolDx Symposium
Cataluna Room
Time
11:45 a.m. –1:15 p.m. Lunch and Scientific Roundtable Presentations Marquis 1:15 p.m. –2:00 p.m. “He Said, She Said”Female and Male Reproductive Physiology
Donor Gamete Use in an Era of Rapidly Developing Technology J. Wes Edmonds, Ph.D., HCLD/ELD/TS(ABB)
Grand Ballroom CDE
Cataluna Room
Cataluna Room (2:00 p.m. –2:45 p.m.) What Can We Learn from the Collection Dish?
Sharon Anderson, Ph.D., HCLD/ELD/CC(ABB) Grand Ballroom CDE
Break sponsored by Cryohexagon
Cataluna Room
Joseph Conaghan, Ph.D., HCLD(ABB) Dolores Lamb, Ph.D., HCLD(ABB )
2:00 p.m. –2:30 p.m. Male Infertility Genetic Testing
Dolores Lamb, Ph.D., HCLD(ABB )
Controversies in Evaluating Embryonic Aneuploidy
Richard Scott Jr., M.D. , HCLD/ALD(ABB)
2:30 p.m. –3:00 p.m.
Valencia Ballroom (2:45 p.m. –3:30 p.m.)
2:45 p.m. –3:30 p.m. (3:00 p.m. –3:30 p.m.) Break sponsored by Cryohexagon
Valencia Ballroom 3:30 p.m. –4:30 p.m.
Grand Ballroom CDE
Cataluna Room ( 3 : 30 p.m. –4 : 00 p.m.) Rapid Vitrification Update Christine Allen, Ph.D., M.Sc., TS(ABB)
Controversies in Evaluating Embryonic Aneuploidy (cont.)
Richard Scott Jr., M.D., HCLD/ALD(ABB)
Grand Ballroom CDE 4:30 p.m. –5 :00 p.m. Understanding Payors & Pathways to Coverage: MolDx ® a n d DEX ® ZCodes ® Jacq u eline Lekostaj , M.D., Ph.D., Medical Director at Palmetto GBA’s MolDx Program
Cataluna Room (4:00 p.m. –6:00 p.m.) Oral Abstracts
Valencia Ballroom Thursday, May 7, 2026
- 18 -
5:00 p.m. –6:00 p.m. Oral Abstracts
Cataluna Room 6:00 p.m. –7:30 p.m. Welcome to the AAB/CRB Seis de Mayo Celebration and Poster Session S ponsored by IVF Store and Biotech, Inc.
Time AAB Conference –MolDx Symposium Cataluna Room CRB Symposium“Looking Back and Moving Forward” Grand Ballroom CDE
Valencia Ballroom 7:30 a.m. –8:30 a.m. Networking Breakfast
7:00 a.m. –7:30 p.m. Conference Registration
Valencia Ballroom 7:15 a.m. –7:45 a.m. AAB Business Meeting (Open To All AAB Members)
Cataluna Room
Thursday, May 7, 2026 (continued)
Time AAB Conference –MolDx Symposium Cataluna Room CRB Symposium“Looking Back and Moving Forward” Grand Ballroom CDE
Grand Ballroom CDE
( 8 : 00 a .m. –8 : 30 a .m.) Legislative Update for ART Laboratories
Stefanie Rinehart, CRD Associates Erin Morton, CRD Associates
Cataluna Room
8:00 a.m. –8 : 3 0 a.m. Major AntiTrust Lawsuit: Health Care Providers vs. Health Care Insurance Companies Jeff Sherrin, O’Connell & Aronowitz
( 8 : 30 a .m. –9 : 30 a .m.) Beyond the R outine S emen A nalysis: The A dvanced D iagnostic S emen T ests D escribed in WHO 6
8:30 a.m. –9:00 a.m. Optimizing Certification Examinations: Question Creation, Result Analysis, and Insights Marilyn Barbour, M.Ed., BSMT(ASCP)
Dolores Lamb, Ph.D., HCLD(ABB )
Grand Ballroom CDE 9:00 a.m. –9:45 a.m. OIG Report On Spike in 2024 Medicare Spending for Genetic Tests and Importance of the RESULTS Act
Stefanie Rinehart, CRD Associates Erin Morton, CRD Associates
( 9 : 30 a .m. –10 : 20 a .m.)
Quality Control and Validation of CLIA Waived Andrology Testing E va Schenkman, Ph.D., M.Sc., HCLD/ELD/TS/CC/ALD(ABB)
Cataluna Room 9:45 a.m. –10:20 a.m. CLIA’s New “Alternate Pathway” for NonTraditional Doctoral Degrees and Other CLIA Changes (Virtual Presentation) Karen S. Sutterer, BS, MT(ASCP), Technical Advisor Survey Branch, Centers for Medicare & Medicaid Services, Division of Clinical Laboratory Improvement & Quality
Grand Ballroom CDE
Quality Matters: Publishing and Dissecting Literature
Cataluna Room
-
Grand Ballroom CDE
10:20 a.m. –10:50 a.m. Break sponsored by Sapyen
-
Christopher Barratt, Ph.D.
Valencia Ballroom 10:50 a.m. –11:45 a.m. Billing/Payment Challenges for MolDx and Toxicology Procedures Moderated by Stefanie Rinehart, CRD Associates
The Great Debate: Are Sperm Counts Declining? Dolores Lamb, Ph.D., HCLD(ABB ) Christopher Barratt, Ph.D.
Cataluna Room
• Medical Necessity/Clinical Utility Matthew Rutledge, President MD LABS
• Audits/Compliance Jeff Sherrin, O’Connell & Aronowitz
Marquis 1:15 p.m. –2:15 p.m. NextGeneration Sequencing (NGS) Moderated by Jeanne Ohrnberger, Ph.D., HCLD(ABB) 1:15 p.m. –2:00 p.m. NGS vs. Long Read Sequencing: What Fits Best Roya Pedram, Ph.D., HCLD(ABB) 2:00 p.m. –2:45 p.m. Operational Challenges and Practical Solutions in Routine Clinical NGS Testing Joy Nakitandwe, Ph.D., HCLD/CC(ABB)
Grand Ballroom CDE
11:45 a.m. –1:15 p.m. Lunch and Sponsorassociated Roundtables
Cataluna Room
Thursday, May 7, 2026 (continued)
AAB Conference –MolDx Symposium Cataluna Room CRB Symposium“Looking Back and Moving Forward” Grand Ballroom CDE
Time
Valencia Ballroom 2:45 p.m. –3:30 p.m. Break sponsored by Juno Genetics
2:15 p.m. –2:45 p.m. CRB Business Meeting/Awards
Valencia Ballroom 3:30 p.m. –Varying Times
Maria Katz, M.Sc., LCGC
Grand Ballroom CDE (4:30 p.m. –5:30 p.m.) Whole Genome Sequencing of Embryos Technical Challenges and Clinical Opportunities
NextGeneration Sequencing (NGS) (continued) Moderated by Jeanne Ohrnberger, Ph.D., HCLD(ABB) 3:30 p.m. –4:15 p.m. Making NGS Work in the Clinical Laboratory: Validation Considerations Pan Zhang, Ph.D., HCLD(ABB) 4:15 p.m. –5:00 p.m. Reporting: Case Studies and Dilemmas Pan Zhang, Ph.D., HCLD (ABB)
Grand Ballroom CDE (5:30 p.m. –6:00 p.m.) Question and Answer: Pandora's Box
Cataluna Room ( 3 : 30 p .m. –4 : 30 p .m.) Current and Emerging Genetic Testing Methodologies
Richard Scott Jr., M.D., HCLD/ALD(ABB)
Maria Katz, M.Sc., LCGC
Richard Scott Jr., M.D., HCLD/ALD(ABB)
Jenna Miller
Grand Ballroom CDE 6:00 p.m. –7:00 p.m. Networking Reception S ponsored by CooperSurgical Fertility Solutions
Valencia Ballroom Friday, May 8, 2026 Time CRB Symposium“Looking Back and Moving Forward” Grand Ballroom CDE
Valencia Ballroom 7:45 a.m. –9:00 a.m. Networking Breakfast
7:00 a.m. –12:30 p.m. Conference Registration
Valencia Ballroom 8:30 a.m. –9:30 a.m. Old IVF Practices –What Have We Learned? Kimball Pomeroy, Ph.D., HCLD(ABB)
Grand Ballroom CDE
Grand Ballroom CDE 9:30 a.m. –10:00 a.m. What to Do with Non2PN Embryos Jenna Miller
Valencia Ballroom 10:30 a.m. –11:30 a.m. Informed Consent: Whose “Job” Is It Anyway?
10:00 a.m. –10:30 a.m. Break
Grand Ballroom CDE Lisa Rinehart, RN, BSN, JD
Friday, May 8, 2026 Time CRB Symposium“Looking Back and Moving Forward” Grand Ballroom CDE 11:30 a.m. –12:00 p.m. Question and Answer: Ask the Experts All Remaining Presenters
Ballroom CDE 12:30 p.m. –6:30 p.m. Optional CAP Reproductive Laboratory Accreditation Program Inspector Training Seminar The CAP Workshop is not included in the CRB Symposium registration package. Separate registration for the CAP program is required.
The AAB Conference, CRB Workshop, Symposium, and CAP Reproductive Laboratory Accreditation Program Inspector Training Seminar have been approved by PEER (American Board of Bioanalysis). In addition, contact hours earned can be used to satisfy CE requirements in the states of California, Florida, Louisiana, and Montana.
Disclaimer: The content and views presented in this educational activity are those of the faculty/authors and do not necessar ily reflect those of the American Association of Bioanalysts (AAB) or the AAB College of Reproductive Biology (CRB). The presented materials and information are not necessarily exhaustive of the subject matter, and laboratory professionals and all other individuals should review and consider other relevant materials and professional opinions and advice on the subject matter before mak ing appropriate decisions.

CHAIR
David F. Carpenter, Ph.D. Ft. Myers, Florida
VICE CHAIR
Shirley L. Lovelace, MT(AAB) Shoreline, Washington
SECRETARY
Samuel Dunn, Ph.D., HCLD(ABB) Oklahoma State Department of Health Oklahoma City, Oklahoma BOARD MEMBERS
Sierra Dowling, MT(AAB) Sandys, Bermuda
Kristina Hsieh Dr. P.H., HCLD/PHLD(ABB)
California Department of Public Health Oakland, California
Alla Kazy, M.Sc., MDxT/MT(AAB) Biolife Social Circle, Georgia
Samuel H. Leggett III, MT(AAB) Children’s Hospital of the King’s Daughter Norfolk, Virginia
Mary Mayo, Ph.D., BCLD(ABB) Heartland Women’s Healthcare Mount Juliet, Tennessee
Joy Miller (Public Member) Long Beach, California
Ryan Brock Neil, Ph.D., BCLD(ABB) Eurofins Viracor Lenexa, Kansas
Salu Ribeiro, M.Sc., TS(ABB), ELS/ALS(AAB) Bay Area PL Services San Francisco, California
Anna Sokolova, ELS/ALS(AAB) Medtech For Solutions Pensacola, Florida
Nicola Winston, Ph.D., HCLD/CC/ALD(ABB) University of Illinios Chicago Chicago, Illinois
CHAIR EMERITUS
Richard R. Jones Sr., BA, MT(AAB) College Park, Georgia







Today’s male infertility clients demand a lot from their IVF specialists. You need a supplier you can count on for the advanced reproductive technology products you need, when you need them. Whether it’s Halosperm® DNA fragmentation kits, Nidacon PureSperm® isolation media, Leja® Standard Count disposable counting chambers, or any of our complete line of male infertility products and services, Andrology Store by Spectrum has the products you need, the service you expect for your Andrology Lab. For free samples call 800-358-7509, or email your request to order@AndrologyStore.com.

CHAIR
Dolores J. Lamb, Ph.D., HCLD(ABB) Children’s Mercy, Kansas City, Missouri
VICE CHAIR
Joseph Conaghan, Ph.D., HCLD/ELD(ABB) Half Moon Bay, California MEMBERS
SECRETARY/TREASURER
Michelle Altrich, Ph.D., HCLD(ABB) Eurofins Viracor, Lenexa, Kansas
Sharon Anderson, Ph.D., HCLD/ELD/CC(ABB) Myerstown, Pennsylvania
Melanie Clemmer, Ph.D., HCLD(ABB) WVU Center for Reproductive Medicine, Morgantown, WV
Marina O. Gvakharia, M.D., Ph.D., HCLD/ELD(ABB), MT(AAB) Cupertino, California

2025-2026
MEMBERS CONTINUED
Jeanne Ohrnberger, Ph.D., HCLD/CC(ABB) Genemarkers LLC Northville, Michigan
Erin Park, Ph.D., BCLD/HCLD(ABB) American Esoteric Laboratory Rossville, Tennessee
David L. Smalley, Ph.D., BCLD/PHLD(ABB) Memphis, Tennessee
Larry Smith Gilroy, California
Michael Alan Stout, Ph.D., HCLD/ELD/TS/CC/ALD(ABB) Aspire Fertility Dallas Dallas, Texas
Owatha Loye Tatum, Ph.D., HCLD/CC(ABB) Blackhawk Genomics, Concord, California
Aire~IRMU (In Room Modular Unit) redefines indoor air quality by leveraging the proven science of LifeAire's patented in-duct multi-stage air purification technology.

Susan Moore, Ph.D., M.Sc., HCLD(ABB) University of Missouri, Columbia, Missouri LifeAire Systems 2026 AAB Roundtable





Protection against airborne threats
Improved metrics of embryogenesis
Reduced contamination risk
Enhanced lab confidence
Negates a variable threatening the culture environment
Thursday May 7, 11:45 am - 1:15 pm

Speaker: Jason Russack, M.S. Air Quality Engineer
Title: The Invisible Intruders: Revealing Airborne Contaminants Jeopardizing IVF Success



CHAIR
Salu Ribeiro, M.Sc., TS(ABB), ELS/ALS(AAB) San Francisco, California
CHAIR ELECT Vacant
VICE CHAIR
Rhonda M. Coats, MT(AAB) Jackson, Louisiana
SECRETARY/TREASURER
Samuel H. Leggett III, MT(AAB) Virginia Beach, Virginia
AT LARGE BOARD MEMBERS
Juliana D. Delgado, MT(AAB) Homestead, Florida
Ellen Gibbons, MT(AAB) Lake Villa, Illinois
Elizabeth Beck, MT(AAB) Norwalk, Wisconsin
Kayla Parrish Charlottesville, Virginia
AAB LIAISON REPRESENTATIVE
David F. Carpenter, Ph.D. Ft. Myers, Florida
ADMINISTRATOR
Mark S. Birenbaum, Ph.D.
ASSOCIATE ADMINISTRATOR
Barbara K. Timm, M.Sc., ELD/TS(ABB)









CRB PRESIDENT
Matthew Hudnall, M.Sc., TS(ABB)
Reproductive Diagnostics Columbus, Ohio
CRB PRESIDENT ELECT
Alison Bartolucci, Ph.D., HCLD/ TS(ABB)
First Fertility Avon, Connecticut
CRB DIRECTOR MEMBER
Alicia Broussard, Ph.D., HCLD/ TS(ABB)
Generation Next Fertility Stamford, Connecticut
Medical Technologist Member
Salu Ribeiro, M.Sc., TS(ABB), ELS/ALS(AAB) San Franciso, California
CRB SECRETARY
Erin Scott, MD, Ph.D., HCLD(ABB)
University of Rochester Webster, New York
CRB SUPERVISOR MEMBER
Nanis Nassar, M.Sc., TS(ABB) East Meadow, New York
MEETING COMMITTEE CHAIR
Melanie Clemmer, Ph.D., HCLD(ABB)
WVU Center for Reproductive Medicine Morgantown, West Virginia
NATIONAL INDEPENDENT LABORATORY ASSOCIATION (NILA) OFFICERS & BOARD MEMBERS
VICE CHAIR
Robert Middleberg, Ph.D. NMS Labs Horsham, Pennsylvania CHAIR
John J. Weiss, Ph.D. Allermetrix, Inc. Franklin, Tennessee
SECRETARY
Eric Carbonneau, MS, MLS (ASCP)
TriCore Reference Laboratories Albuquerque, New Mexico
CHAIR
Charles E. Gilbert, Ph.D., M.Sc.
Epidemiology & Toxicology Institute Holbrook, New York
Chairperson Elect
Heinz R. Reiske, Ph.D., HCLD(ABB)
Eli Lilly and Company Avon, Indiana

1997 – Grace Centola, Ph.D., HCLD(ABB)
1998 – Richard Rawlins, Ph.D., HCLD(ABB)
1999 – Kristen Ivani, Ph.D., HCLD(ABB)
2000 – Thomas Pool, Ph.D., HCLD(ABB)
2001 – David McCulloh, Ph.D., HCLD(ABB)
2002 – Co-Chairs Barry Behr, Ph.D.,HCLD(ABB), and David McCulloh, Ph.D., HCLD(ABB)
2003 – Barry Behr, Ph.D., HCLD(ABB)
2004 – Gary Smith, Ph.D., HCLD(ABB)
2005 – David Battaglia, Ph.D., HCLD(ABB)
2006 – Kathleen Miller, TS(ABB)
2007 – Joseph Conaghan, Ph.D., HCLD(ABB)
2008 – Margaret Papadakis, Ph.D., HCLD(ABB)
2009 – Kimball Pomeroy, Ph.D., HCLD(ABB)
2010 – Dean Morbeck, Ph.D., HCLD(ABB)
2011 – William R. Boone, Ph.D., HCLD/ELD/CC(ABB)
2012 – Mark P.D. Dow, Ph.D., HCLD/ELD(ABB)
2013 – Marina O. Gvakharia, M.D., Ph.D., HCLD/ELD(ABB)
2014 – Carli Chapman, ELD/TS(ABB)
2015 – Stanley G. Harris, ELD/TS(ABB)
2016 – Gerard F. Celia, Ph.D, HCLD/TS(ABB)
2017 – Jean Popwell, Ph.D. HCLD/CC(ABB)
2018 – Anthony Anderson, D.Sc., ELD(ABB)
2019 – Dara Berger, Ph.D., HCLD/TS(ABB)
2020 – T. Arthur Chang, Ph.D., HCLD/ELD/CC(ABB)
2021 – Salu Ribeiro, M.Sc., TS(ABB), ELS/ALS(AAB)
2023 – Marlane J. Angle, Ph.D., HCLD/CC(ABB)
2024 – Charlene Alouf, Ph.D., HCLD/TS(ABB)
2025 – Melanie Clemmer, Ph.D., HCLD(ABB)
Members At Large
Kira Marquis National Labs Inc. Jeanne Ohrnberger Genemarkers, LLC
Sherry Dadgar, Ph.D. Personalized Medicine Care Diagnostics Laboratory
Rochelle Odenbrett Sanford Health Laboratories
FIRST VICE CHAIR
Jennifer Eastman, Ph.D., HCLD/TS(ABB)
State of Alaska Public Health Lab Wasilla, Alaska
SECOND VICE CHAIR
Sharon Massingale, Ph.D., HCLD/CC(ABB)
Alabama Department of Public Health Montgomery, Alabama
Wednesday,
AI: What’s Fake News, What’s to Stay?
Christine Allen, PhD, MSc, TS(ABB)
Clinical Air Quality Factors and Facts
Morgan Orsolini, MS, PhD Candidate
Current Landscape of Embryology Licensure in Different States
Nanis Nassar, MS, TS(ABB)
Designing IVF Labs for the Modern World
Eva Schenkman, Ph.D., M.Sc., HCLD/ELD/TS/CC/ALD(ABB)
Do No Harm: Basics of AI in IVF and Flaws to Watch For In Publications
Julianna Lamm, MS
Embryology in 2035: What Will Matter Most?
Lizeth Barrientos, MS, TS(ABB)
Genetic Evaluation of the Infertile Male
Dolores Lamb, PhD, HCLD(ABB)
How to Lead a New Team and Create a Culture No One Wants to Leave
Kristen Ivani, PhD, HCLD(ABB)
How Close Is A Non-Hormonal Male Contraceptive?
Christopher Barratt, PhD
Key Performance Indicators of Ovarian Stimulation Protocols on Number of Usable Blastocysts: What Should We Be Tracking?
Sharon Anderson, PhD, HCLD/ELD/CC(ABB)
Moving Frozen Specimens to Off-site, How to Implement?
Sung Tae Kim, PhD, HCLD(ABB)
Processing Suboptimal Sperm Specimens for ART
Melanie Clemmer, PhD, HCLD(ABB)
Seminal Oxidation Reduction Potential (ORP) and Its Clinical Implications
Manesh Kumar Panner Selvam, PhD, DVM, ALS(AAB)
Sperm Metabolism: The Missing Step Towards Better IVF Outcomes
Felipe Navarrete, PhD
The Threat of Politics in the IVF Lab
J. Wes Edmonds, PhD, HCLD/ELD/TS(ABB)
Ultra-Fast Vitrification: Donor Egg Perspective
Wayne Caswell, MS
Updating and Validating New Procedures
Alison Bartolucci, PhD, HCLD(ABB)
















Thursday, May 7, 2026
Beyond the Biopsy: Elevating PGT Through Collaboration
Kezia Emeny-Smith, TS(ABB), ELS(AAB) (Juno Genetics)
Blastocyst Cryopreservation in Practice: Clinical Evidence Behind Fast Vitrification and Rapid Warming
Brad Zavy, MS, TS(ABB) (Nexpring Health)
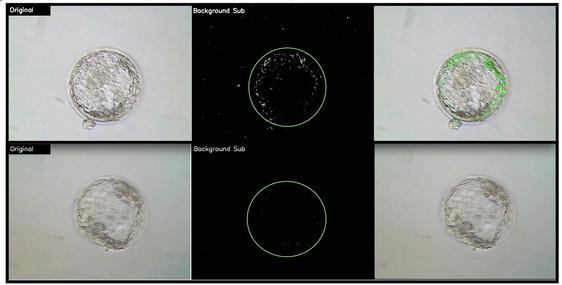
Blastocyst Real-Time Morphokinetics – Making the Invisible Visible
Cara Wessels Wells, Ph.D. (Viable Bioscience)
Closing the Gap: Extending Electronic Witnessing into Long-Term Cryostorage
Cristian Milevski (ReproTech)
Consistency Under Pressure: How IVF Lab Workflow Supports Clinical Outcomes
Amanda Hanson (Reproductive Solutions)
Discrepant PGT Cases – What You Need to Know
Amy Jordan, MS, LCGC (Luminary Genetics)
Environmental Stability in IVF: The Synergy Between Incubators and Media
Toni Di Berardino, Ph.D. (Astec Bio)
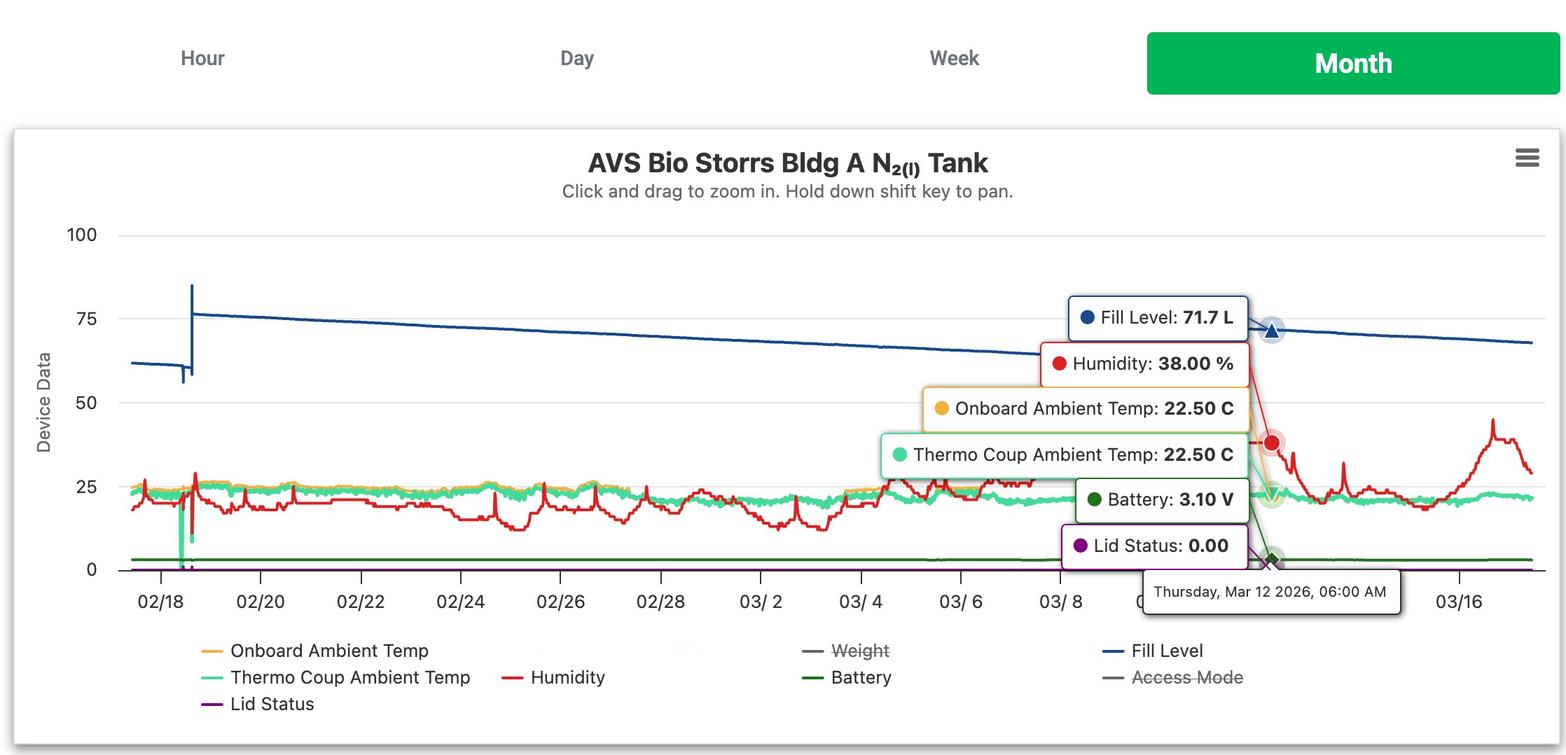
From Habit to Data-Driven Strategy: Rethinking LN2 Fill Optimization
Will Baird (Boreas Monitoring Solutions)
Good Survival, Now What? Troubleshooting and Optimizing Development After Oocyte Warming
Jennifer Hart, MS, TS(ABB) (Fairfax Eggbank)
Invisible Influencers: Case Studies in IVF Lab Air Quality & VOC Testing
Jason Russack (LifeAire Systems)
MIRI Evidence Witnessing and Traceability: Your Workflow, Our System – Perfectly in Sync (ESCO) by Fredrik Skoog, Application Specialist, ESCO; and Mariana Macedo, Embryologist/MS, ESCO
Optimizing Your PGT-A Outcomes
Angela Linville, Ph.D., and Shannon Kirkpatrick, M.Sc., TS/ELD(ABB) (Vitrolife Group)
PGT-SR: Unlocking the World of Structural Chromosomal Rearrangements
Brittany-Ann Delacruz, MS, LCGC (Luminary Genetics)
The Physics of Early Failure in Cryogenic Storage
Mark Langley (DeTech Solutions)
Protecting Every Specimen: Data-Driven Insights into Cryogenic Storage Safety
Catherine Welch (CryoFuture)
Reimagining Semen Analysis: Confronting Limitations and Embracing AI-Driven Solutions in Clinical Practice
Dr. Andreas Köthe, PhD, (MetaSystems International)
SB 729 and Its Ripple Effect: Operational Risks and Strategies for Fertility Centers Facing Rapid Growth
George Heywood (Matcher) and Brad Senstra (ReproTech)
Segmental Aneuploidies: Harmonizing Laboratory
Reporting and Clinical Management
Diego Marin, Ph.D., HCLD(ABB) (Centogene)
Strategies for Incorporating New Technologies into the Laboratory
Cynthia Hudson, M.Sc., TS(ABB), CLT(NYS), EMB(ACE), MT(ASCP), (Biotech/IVF Store)
Transported Semen Analysis: Considerations, Logistics, and Opportunities for Advanced Testing
Roxanna Abrines, BS, CLS Trainee/CPT I (CDPH), (Medical Electronic Systems)
Turning Data into Outcomes – Leveraging QAQC KPIs to Drive Patient Outcomes
Debra Mitchiner (eIVF)
Ultra-Fast Vitrification and Warming Protocols: Rethinking Cryopreservation Efficiency in the IVF Lab
Dori Dulaj (Kitazato)
Using AI for Sperm Selection
Anthony Anderson, D.H.Sc., ELD(ABB) (IVF Academy USA)
Email: info@AndrologyStore.com
Phone: (800)358-7509
Website: www.andrologystore.com
Andrology Store by Spectrum (formally Spectrum Technologies) has been a preferred supplier to the Advanced Reproductive Technology industry for more than 37 years. Beginning with Ovu-stick, an ovulation test kit sold exclusively to physician’s offices, Andrology Store by Spectrum has continued to expand its product offering with the singular aim of becoming the infertility specialists one-stop source for ART products. Peter Jensch, the founder of Spectrum Technologies and fondly known as “The PureSperm Guy,” brings extensive experience and knowledge of the industry and of the products he promotes. Recent achievements include the exclusive U.S. distributorship of both the Nidacon PureSperm isolation media and Halosperm DNA fragmentation technology. These products along with Leja’s Standard Count semen
analysis chambers and many other A.R.T. products Andrology Store by Spectrum offers are the industry standard in high quality andrology products. Andrology Store by Spectrum’s goal is to be the preferred supplier of “the products you need…the service you expect for your Andrology Lab.” Give us a call! We’d love to talk and see how we can help your Andrology Lab.
Email: don@astec-bio.com
Phone: (781)749-0734
Website: www.astecbiousa.com www.astec-bio.com/global
Astec is a manufacturer of precision incubators and workstations based in Fukuoka, Japan. The US subsidiary, Astec Bio USA, distributes Astec equipment throughout the Americas and is also the distributor for Astec SureGrip
continued on page 29

Plasticware, Oosafe plasticware, and Dewin micropipettes. Astec Bio USA can provide direct sales and service of Astec equipment with engineers based in the Boston and Toronto areas to cover North and South America.
Website: biotechinc.us
Biotech, Inc. is a leader in IVF, advancing the research, design, development, and commercialization of innovative reproductive technologies. Proudly manufacturing all products in the USA, Biotech supports both human and animal assisted reproduction markets with a commitment to quality and innovation.Our mission is simple: to deliver best-in-class, easy-to-use cryo-devices that IVF professionals can trust—every time. With a reputation built on reliability, performance, and family-centered values, Biotech is more than a manufacturer—we’re a partner in helping families begin. When it comes to IVF cryopreservation, excellence starts here.
Birenbaum and Associates
Website: www.birenbaum.org
Birenbaum and Associates, an association management company, has managed AAB since 1965, ABB since 1968, CRB since 1997, ABOR since 1999, and NILA since 2007.
Email: info@birr.nl
Phone: +31(0)294 230389
Website: www.birr.nl
BIRR is a family-owned Netherlands-based company and is highly committed to provide a broad portfolio of specifically customized IVF-disposables, while producing according to the highest industry quality and sustainability standards. Our portfolio contains a broad range of products,
continued on page 30



including a broad IVF Labware portfolio and gynecological products. BIRR has fertility expertise since 1971. IVF is in our DNA.
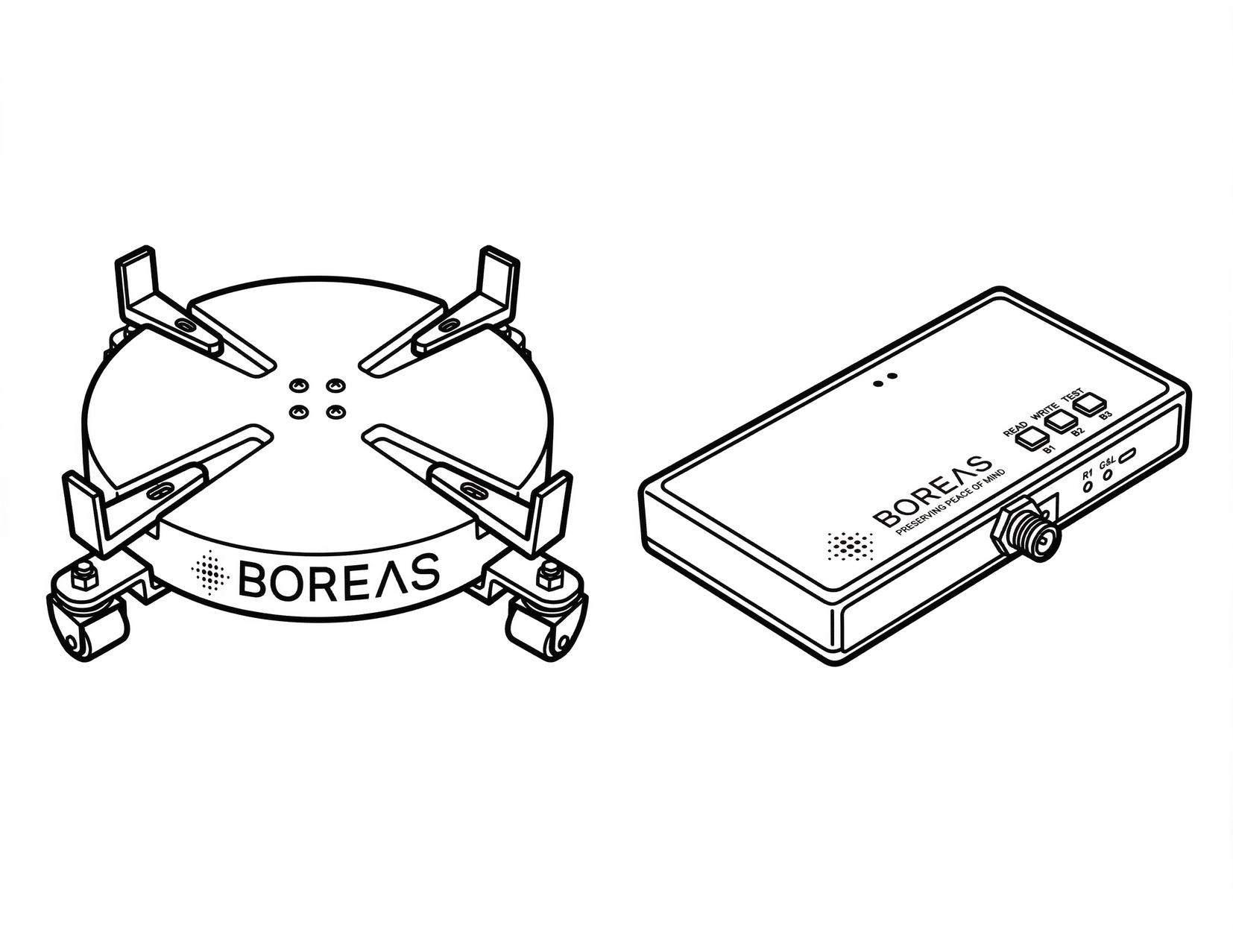
Website: www.boreasmonitoring.com
Boreas Monitoring Solutions, founded by fertility lab professionals with 80+ years of combined experience, developed the CryoScout® & CryoGuardian™ – patented weight-based cryotank monitoring systems that can detect tank failures days earlier than traditional temperature-based methods. Designed “by lab people, for lab people,” the system uses precision scales, cellular LTE connectivity, and a cloud-hosted portal to provide remote, real time 24/7 alerts and actionable tank health data, safeguarding priceless biological samples across fertility clinics, life sciences, animal sciences, and academia. Their deep knowledge of how lab professionals work allowed them to create the safest, most reliable monitoring system ever made. For more information, visit www.boreasmonitoring. com.
Website: www.centogene.com
Website: www.coopersurgical.com
By offering healthcare providers a comprehensive range of innovative medical solutions, we help them deliver rapid results, effective treatments, and more options at the right time, so that women, babies, and families can experience more possibilities, faster than ever. When the time is now, CooperSurgical leads the industry in next-generation fertility and birth solutions that support clinical efficiency and engaged and supported patients. All to help conceive, deliver, and protect healthy babies.
CRD Associates
Website: www.dc-crd.com
Website: www.cryofuture.com
CryoFuture is the largest network of fertility-dedicated cryopreservation facilities and the fastest-growing cryogenic transportation provider in the U.S. Its team combines deep reproductive medicine expertise with advanced technology solutions to ensure exceptional safety, precision, and service for clinics and their patients.
CryoFuture’s long-term cryostorage services feature tripleredundant monitoring and constant oversight to ensure embryos, eggs, and sperm remain safe. Its transportation services are white glove, with medically trained couriers handling every shipment using rigorously validated, GPS-tracked, temperature-monitored cryogenic systems throughout transport. Built by experienced embryologists, CryoFuture delivers the highest standards of safety, security, and transparency.
Email: office@cryohexagon.com
Phone: (914)336-4816
Website: www.cryohexagon.com
Cryohexagon provides advanced cryogenic storage solutions for IVF laboratories focused on capacity, control, and specimen safety. Our patented stainless steel racking system is designed to significantly increase storage within existing freezers while improving access for embryologists during sample retrieval and insertion. Combined with our digital ecosystem for inventory visibility, tracking, and chain of custody, CryoHexagon helps clinics optimize space, strengthen operational oversight, and modernize cryostorage workflows without compromising usability.
continued on page 32

Esco
Email: info@detechus.com
Phone: (860)228-1046
Website: www.Detechus.com
Cryogenic preservation is governed by physical laws that begin to shift at the moment of vacuum compromise. These inception-level failure signatures have historically remained unseen, as conventional approaches respond only after failure conditions are already in progress.
DeTech’s CryoCapture™ platform is grounded in the physics of vacuum behavior, recognizing deviation at its earliest point through external, non-invasive telemetry focused on performance parameters, including weightbased verification within a broader multi-parameter system. By identifying failure at inception, CryoCapture™ establishes a direct link to true system integrity. Designed for universal application across tank types and adjacent lab environments, including incubators, refrigerators, freezers, and controlled storage systems, the platform advances a new paradigm in asset awareness. CryoCapture™ defines a standard rooted in the physics of failure itself.
Website: www.eivf.org
eIVF, Inc. is a leading provider of healthcare software and a comprehensive EMR platform built for fertility practices. Supporting over 125 clinics worldwide, eIVF has been a technology leader in Reproductive Endocrinology and Infertility (REI) since 2002—helping clinics manage over one million cycles and streamline patient care. One of eIVF’s latest innovations, Fertility Vision, is a cloud-native analytics dashboard that delivers real-time, KPI-driven insights to improve clinic performance. It includes the enhanced QA/QC Embryology Dashboard, giving labs an elevated view of embryology KPIs to support quality and consistency. With a continued focus on innovation and integration, eIVF is a trusted technology partner in the fertility space. Learn more at www.eivf.org or email katie@ eivf.org to schedule a demo with our team.
Website: www.esco-medical.com
Esco Medical and Allwin Medical deliver an end-to-end IVF platform, integrated across the laboratory workflowequipment, traceability, and consumables in one cohesive ecosystem.
Esco Medical provides European-designed and manufactured multiroom and time-lapse incubators, IVF workstations, and witnessing and traceability systems. Allwin Medical complements this with a comprehensive portfolio of specialized IVF consumables, including embryo transfer catheters, oocyte pickup needles, insemination catheters, vitrification devices, denudation tips, and other critical laboratory disposables.
Together, the two brands help IVF clinics standardize workflows, strengthen traceability, reduce operational complexity, and focus on delivering clinical excellence. As part of the Esco Lifesciences Group, these brands reflect our commitment to advancing reproductive health through innovative solutions.
Website: www.eurofins.com/medical-device
Email: info@fairfaxeggbank.com
Phone: (888)352-5577
Website: fairfaxeggbank.com
Email: info@fairfaxcryobank.com
Phone: (800)338-8407
Website: fairfaxcryobank.com
Fairfax Cryobank and Fairfax EggBank are the trusted choice for donor sperm and donor eggs. Physicians worldwide rely on us for our decades of experience, rigorous donor screening standards, and commitment to exceptional service. Together, we provide a seamless, highquality solution to support clinics and the families they serve.
continued on page 34
At CryoFuture, your specimens are entrusted to experts who understand the significance behind every vial and straw. With over 300 years of combined IVF experience, CryoFuture’s embryologists have built a system centered on reliability, traceability, and compassion. Every process, from transport to long-term preservation, is designed to ensure your reproductive tissue remains safe, monitored, and ready for your next chapter.

White Glove Service
All transports are handled by trained medical couriers, never third-party carriers like FedEx or UPS

Safety-First Fleet
Local vehicles include cameras, driver alerts, speed monitoring, and fire- and crash-resistant safes
Tanks are GPS-tracked and remotely monitored for temperature, shock, and tilt
Comprehensive Coverage
Each transport includes up to $25,000 in specimen coverage
Managed by experienced embryologists with over 300 years of collective experience
Fortified Facilities
Seismically tested buildings with EF-5 rated vaults designed for maximum protection
Technology-Driven
Continuous 24/7 monitoring with weight, temperature, and thermal imaging
RFID Tracking
CryoFuture Signal™ provides accurate, realtime traceability for every specimen
Email: sales@meetfellow.com
Website: www.meetfellow.com/clinicians
Fellow is the nation’s largest dedicated, high-complexity semen analysis lab, with over 250,000 samples analyzed to date. Our peer-reviewed, diagnostic-grade mail-in process—published in Fertility and Sterility—accounts for sperm degradation over time and uniquely matches the accuracy of traditional testing performed within one hour. Trusted by over 3,000 clinics nationwide, Fellow removes barriers to testing, supports lab and clinic economics, improves patient conversions, and advances male reproductive health research. Fellow semen analysis is a covered benefit for eligible Progyny and Maven members. Visit the Fellow booth to learn how we can support your practice and patients.
Website: www.hblabinnovations.com
HB Lab Innovations Inc. is a California-based provider of premium equipment and consumables for IVF laboratories and fertility clinics. Our portfolio includes advanced vitrification devices and high-quality culture dishes, designed to meet the highest standards of performance and reliability.
In partnership with METIEC, HB Lab also offers comprehensive laboratory monitoring systems for incubators, tanks, and environmental conditions, helping clinics maintain optimal and consistent lab performance.
continued on page 35
Choose labware designed for IVF
IVF-dedicated design
Developed with IVF laboratories

Supporting consistency across IVF workflows

MEA/MEA+ tested labware portfolio
Manufactured in Europe under rigorous quality standards





Discover the BIRR difference!











Email: admission@ivfacademyusa.com
Phone: (210)988-6645
Website: www.ivfacademyusa.com
IVF Academy USA is a premier institution in San Antonio, Texas, dedicated to advancing the field of Assisted Reproductive Technology (ART) through specialized professional training. While the academy offers clinical and nursing pathways, its Lab School stands out for providing rigorous, hands-on programs designed for aspiring and current embryologists.
The Lab School curriculum focuses on high-demand skills, including Intracytoplasmic Sperm Injection (ICSI), embryo biopsy, and cryopreservation. Students benefit
graduates are prepared for the technical complexities of modern fertility care, bridging the gap between academic knowledge and clinical excellence in the laboratory.
Website: us.ivfstore.com
Your One-Stop Shop for High-Quality IVF Laboratory Supplies + Services.
At IVF Store, we are more than a supplier — we are your strategic IVF partner. We support IVF and andrology laboratories with high-quality supplies, consumables, equipment, and services, all from one reliable source. Our mission is to deliver choice, efficiency, and dependable service built around real scientific workflows.



of IVF labs. From andrology, culture, and vitrification solutions to micromanipulation equipment and essential labware, our comprehensive portfolio ensures your lab is always equipped with what it needs. Our intuitive website and fast, consistent shipping make ordering simple, while our knowledgeable team is always available to provide product guidance and support confident decision-making.
Email: george.carralejo @junogenetics.com
Website: www.junogenetics.com
Juno Genetics is a leading laboratory specializing in preimplantation genetic testing (PGT) for IVF clinics and patients. Our mission is to deliver rigorously validated, evidence-based testing of the highest clinical quality. We offer PGT-A, PGT-M, and PGT-SR using proprietary platforms developed and validated in-house. With over 30 years of research and more than 350 peerreviewed publications, our global teams are committed to advancing embryo genetics and improving patient outcomes. We prioritize close collaboration with IVF clinics, integrating workflows to ensure efficient, high-quality care. We also support education through continuing medical education and academic initiatives, promoting deeper understanding and innovation in reproductive genetics.
Kitazato
Email: info.us@kitazato-ivf.com
Phone: (856)702 40 41
Website: www.kitazato-ivf.com
Kitazato is a global Japanese company dedicated to ART. We conduct extensive research, develop, manufacture, and market high-quality medical devices and media to support diverse fertility treatments. One of our greatest contributions to the field has been
The Cryotop® Method, which revolutionized oocyte and embryo vitrification and secured its global implementation. Our team collaborates with scientists, physicians, and embryologists worldwide to ensure access to the latest knowledge and technology. In the U.S., Kitazato operates from its own East Coast facility, enhancing service and
continued on page 37

product availability. We are committed to delivering top-quality products and services to maximize IVF clinic success, helping patients fulfill their dream of having a baby. This mission drives us to support the entire IVF LifeCycle.
Email: info@LifeAire.com
Phone: (484)224-3042
Website: www.lifeaire.com
LifeAire Systems is a biomedical company that provides transformational air purification technology designed, and proven, to deliver the airborne metrics critical for optimal living cell environments. Protected by 26 patents, the technology specifically targets the cytotoxic levels of volatile organic compounds (VOCs), viable and nonviable particulates in air to bring levels within optimal ranges to protect the human embryo and living cells. Cells cultured in LifeAire-purified air have demonstrated statistically significant increases in cellular metrics and clinical
outcomes beyond any other technology in the market. From its Pennsylvania headquarters, LifeAire Systems supports global applications in in vitro fertilization, healthcare, life sciences and commercial markets.
Email: Support@luminarygenetics.com
Phone: (408) 503-6484
Website: www.LuminaryLifeSciences.com
Luminary Life Sciences illuminates the path to parenthood for individuals and families by pioneering a comprehensive suite of life science services tailored to meet the specialized needs of patients throughout their reproductive health journey. Our vision is to empower patients and healthcare professionals by offering innovative, customer-centered solutions that provide support from pre-conception to postbirth.
continued on page 38

Fairfax Cryobank and Fairfax EggBank are the trusted choice for donor sperm and donor eggs. Physicians worldwide rely on us for our decades of experience, rigorous donor screening standards, and commitment to exceptional service.
Together, we provide a seamless, high-quality solution to support clinics and the families they serve.

Extensive, diverse donor databases
Industry-leading screening & testing standards
Global distribution & trusted partnerships
Dedicated client support & service
Partner with us: fairfaxeggbank.com | fairfaxcryobank.com
The Luminary portfolio is setting new standards in reproductive health services by offering a wide range of specialized solutions including prenatal vitamins, cryogenic storage and genetic testing services.
Luminary Genetics is a leading provider of advanced genetic testing services for the IVF field. With a state-of-the-art Next Generation Sequencing platform and a committed team of scientists and geneticists, we deliver unparallel precision to give patients peace of mind about their future family’s health.
Website: www.imtmatcher.com
Matcher™ is a barcode-based electronic witnessing, labeling, workflow scheduling, traceability, and insights system designed to prevent errors caused by patient and specimen misidentification. It replaces manual double-checking by capturing the “who, what, where, and when” at every step, with photographic proof down to the embryo level.
Highly configurable and with no capital cost, Matcher supports the full clinic workflow—from patient registration through embryo transfer, PGT, and cryostorage. As part of Reprotech, Matcher integrates with Reprotech’s off-site cryostorage products to create a seamless, fully traceable digital chain of custody from clinic to long-term storage.
Used globally, Matcher helps clinics reduce risk, minimize the potential for mistakes, and streamline quality management—saving time while strengthening confidence in every step.
Email: info@mes-global.com
Phone: (818)591-3030
Website: www.mes-global.com
Medical Electronic Systems (MES) is a global provider of advanced semen analysis solutions and male fertility testing services. For over 25 years, MES has supported
continued on page 39


MetaSystems Group, Inc. (Medford, MA), a leading manufacturer of automated imaging slide scanning systems, and Fertility Solutions (Cleveland, OH) , the only FDA-cleared manufacturer of human semen analysis quality control has partnered to create an unparalleled, turn-key solution for the fertility market.
Utilizing MetaSystems cutting -edge AI technology combined with Fertility Solutions expertise in semen analysis procedures and human sperm quality control products, laboratories will have a premier solution for semen analysis that offers labor-savings and workflow efficiency. Through the incorporation of Deep Neural Networks (AI), MetaSystems delivers a powerful system offering sperm count, motility, viability, and morphology for a truly automated and walkaway experience. Individual sperm-level reporting delivers complete transparency and traceability, so you can trust every result.

www.metasystems.org
DeAna Paustian dpaustian@metasystems.org 617.924.9950
https://www.fertilitysolutions.com Anna Bort bortanna@fertilitysolutions.com (216) 491-0030
laboratories and clinics with innovative technologies designed to improve accuracy, efficiency, and access to male fertility diagnostics. MES offers in-lab solutions, including the SQA-Vision and SQA-iO automated semen analyzers, enabling rapid, standardized testing with advanced capabilities and integration potential. Additionally, MES provides at-home and transported testing options through MaleMan, a mail-in reference lab service for comprehensive analysis including DNA Fragmentation Index (DFI) testing, and YO, an at-home sperm test. By combining in-lab automation with flexible remote testing solutions, MES helps practices expand access, reduce operational burden, and deliver reliable, timely results to support patient care.
Website: www.metasystems-international.com
Nexpring
Email: info@nexpringhealth.com
Phone: (800)323-0503
Website: www.nexpringhealth.com
Nexpring Health is a global ART solutions partner uniting nine industry leaders to support the full spectrum of reproductive medicine.
We bring together Cook Reproductive Health, Hamilton Thorne, Irvine Scientific, Planer, IVFtech, Gynemed, Gynétics, Microptic, and Tek-Event combining more than 40 years of trusted expertise into one comprehensive partner dedicated to putting embryologists, clinicians, and fertility clinics at the center of everything we do.
Solutions that simplify. Support that never wavers.
From capital equipment and media to consumables and service, we deliver responsive, hands-on support at every critical moment.
Our integrated solutions simplify the complex, elevate daily workflows, and empower consistent, high-quality outcomes. Because when you can focus
page 38
on what matters most, the future of fertility care knows no limits.
Every embryo. Every procedure. Every cycle counts.
continued on page 40

NextGen LifeLabs
Email: info@nextgenlifelabs.com
Phone: (215)672-4838
Website: www.nextgenlifelabs.com
Empowering IVF laboratories for Successful Outcomes!
Unlock the Future of IVF! Since 2012, NextGen LifeLabs has been at the forefront of IVF innovation, delivering end-to-end solutions tailored for today’s advanced A.R.T. laboratories.
Why Choose Us?
• Expert Partnership: With nearly 50 years of combined experience, we collaborate with IVF programs to provide state-of-the-art equipment, premium consumables, and unparalleled support.
• Tailored Solutions: From single equipment installations to complete lab design and validation, we customize our offerings to fit your lab’s unique needs.
Our Cutting-Edge Portfolio Includes:
• Matcher Electronic Witnessing
• LabCapture Remote Alarm System
• LifeAire Air Purification
• Aethair VOC Monitoring
• Astec Equipment
• Micromanipulation Systems, Microscopes, Laser Systems
• Customized Equipment
What We Offer:
• Expert Installation & Preventive Maintenance
continued on page 41

Next-generation sequencing (NGS) technology is helping revolutionize reproductive research with fast, accurate, and comprehensive detection of genetic variants.
How can you make an impact? Learn more about expanded carrier screening (ECS) and preimplantation genetic testing (PGT) research with Ion Torrent™ NGS technology, including the Ion GeneStudio™ S5 and Genexus™ systems.
Kitted solutions are available for PGT-A, PGT-M, and carrier screening. Or, you can create a customized panel for your unique targets.
• Expansion & Relocation Consulting
• A Full Range of Laboratory Consumables
One Partner. One Solution. Everything your IVF lab needs to thrive in a competitive landscape.
Noblegen Cryogenics
Email: sales@noblegencryo.com
Contact number: (800)767-6532
Website: www.noblegencryogenics.com
Noblegen Cryogenics design, manufacture, and support high performance liquid nitrogen generators that deliver a reliable and cost effective alternative to traditional bulk supply. Their in-house generators serve a wide range of industries, including low-temperature research, IVF, dermatology, pharmaceuticals, and animal husbandry, helping organizations enhance efficiency, safety, and
control over their liquid nitrogen supply.
As part of the PEAK International Group, a global leader in laboratory gas generation, Noblegen benefits from a worldwide service network that provides fast, expert technical assistance on every continent. With a commitment to quality, innovation, and long term support, Noblegen empowers users across the globe to access liquid nitrogen seamlessly and sustainably.
O’Connell & Aronowitz
Website: www.oalaw.com
continued on page 42
Maximize



Email: customer.success@progenesis.com
Phone: (858)257-2122
Website: www.progenesis.com
Progenesis is a trusted partner in family-building, providing advanced genetic testing and integrated technologies that support physicians, embryologists, and IVF professionals. We equip clinical teams with the insights and tools needed to deliver exceptional patient care.
Committed to expanding access to reproductive genetics, Progenesis empowers the medical community through education, training, and collaboration—enhancing the patient experience along this deeply personal journey.
In 2026, we introduced Progenesis 360, a unified platform combining advanced genetic testing, AIpowered IVF software, and lab technology. From comprehensive screening services to intelligent EMR, real-time lab monitoring, and digital workflow solutions, Progenesis delivers a connected ecosystem designed to streamline operations and elevate standards across reproductive medicine.
Email: support@rsifertility.com
Phone: (800)465-0368
Website: www.rsifertility.com
At RSI, we are driven to improve the future of fertility through science, innovation, and compassion. Our mission is to develop cutting-edge, patient-focused technologies that increase the chances of successful pregnancies and reduce the stress of fertility treatment.


Enable rapid and reliable identification of disease-causing mutations, even at the early stages of pregnancy.
continued on page 43
centogene.com
Reprotech
Website: www.reprotech.com
Reprotech is a leading U.S. cryostorage partner for fertility clinics, with 35+ years of experience in secure, compliant long-term specimen management. We operate five disasterresistant biorepositories with redundant systems and 24/7 monitoring to reduce risk and protect patient specimens.
Our offsite model and Cryologix platform streamline inventory, consent, billing, and patient communication, improving visibility while reducing administrative burden. Through validated workflows, we manage the full specimen lifecycle from clinic integration and transport to intake and long-term storage, maintaining strict chain of custody.
Proprietary tools like Matcher® verification and Smartsleeve® identification add additional safeguards during handling and storage. The result: standardized processes, reduced liability, and more time for clinics to focus on patient care.
sapyen
Email: aimee@sapyen.co
Phone: (619)992-9050
Website: www.sapyen.co
Sapyen’s Advanced Home Semen Analysis provides the world’s most accurate, affordable, and convenient male fertility test. Our patent-pending SPX72 technology extends sperm viability, including motility, from 1 hour to 72 hours, enabling the safe transport of semen samples to certified laboratories for analysis. Patients send their semen sample in the Sapyen Test Kit to one of our partner labs for analysis. Test results are delivered back to the referring clinic. Sapyen is trusted and recommended by leading fertility clinics and specialists across the US.





from page
Email: Director@sperm1.com
Phone: (800)637-7776
Website: www.sperm1.com/sebnj/
SEBNJ provides the cryopreservation of Reproductive Cells & Tissue for pre-pubertal male and adult men by cryopreserving (freezing) their reproductive cell(s) & tissue, such as sperm and testicular tissue for future Assisted Reproductive procedures. SEBNJ offers short term and long term cryo- storage for transferred cryopreserved Embryos, Oocytes, and Sperm and Ovarian Tissue. SEBNJ offers Directed/Designated Donor screening services to individuals and couples seeking to achieve Assisted Reproduction while choosing their own Directed/Designated Donor. The Sperm & Embryo Bank of New Jersey, Inc. (SEBNJ) was established in 1989 and is a commercial, independent, and privately owned

A distinct approach to detecting cryogenic tank failure at the moment of vacuum compromise, identifying risk at inception before temperature, level, or weight-based systems respond
Designed for deployment across various tank types, manufacturers, and configurations
Applicable across cryogenic and adjacent lab equipment, including freezers refrigerators incubators and controlled storage conditions
Detechus com info@detechus.com 860-228-1046
Reproductive Cell & Tissue Bank located in New Jersey. SEBNJ is FDA registered as a Reproductive Cell & Tissue Bank. SEBNJ is licensed by: The New Jersey Department of Health as a Laboratory under CLIA, and the New York State Department of Health as a Reproductive Cell & Tissue Bank. For up-to-date information you may contact one of our Client Service Coordinators at: 800-637-7776 or email: Director@sperm1.com.
Thermo Fisher
Email: akeeth.pinto@thermofisher.com
Phone: (289)983-1380
Website: thermofisher.com/RHbyNGS
Thermo Fisher Scientific is the world leader in serving science. Our mission is to enable our customers to make the world healthier, cleaner and safer. We support reproductive health researchers with innovative technologies throughout
continued on page 45




the reproductive lifecycle – such as genetic analysis for carrier screening and preimplantation genetic testing.
Email: cara@emgenisys.com
Phone: (281)728-0491
Website: www.viablebio.com
Viable Biosciences, the human IVF division of EmGenisys, is developing a non-invasive, AI-enabled platform for blastocyst assessment based on quantitative analysis of short, 30-second real-time videos of human blastocysts. By capturing dynamic signals invisible to the human eye in short videos and integrating them with known clinical outcomes, our technology predicts embryo viability and ploidy status with objective, data-driven precision. This
patented approach is designed for seamless integration into routine laboratory workflows and does not require timelapse incubation systems or specialized optical hardware. We are actively expanding our clinical studies and looking to add more collaborators. If you are interested in helping shape the future of IVF, we invite you to join us to help make the invisible, visible!
Website: www.vitrolifegroup.com

In the ever-evolving realm of ART labs, NextGen LifeLabs stands out. We offer specialized equipment, expert support, preventative maintenance & service packages, and NEW lab consultations. We're the only company who offers truly unique, customizable solutions to meet your needs.
Let's fuel your innovation. Bring your lab to life!


Phone: Email: 215-672-4838 info@nextgenlifelabs.com continued on page 46

Vitrolife Group has been dedicated to the reproductive health market since 1994 and has grown by focusing on product development, groundbreaking research, and consistent quality control. Based on science and advanced research capabilities, our aim is to deliver products and services for the entire reproductive-health journey, providing consistent performance and guaranteed quality. We take a holistic approach to reproductive health where we provide training, support and a wide range of services for clinics and laboratories worldwide. We are recognized as a leading knowledge provider in the industry as we
from page 45
work with universities, research institutes, networks and communities to secure and improve successful treatment outcomes.
Vitromed
Website: www.vitromed.com/
you to all the Sponsors of the 2026 AAB Educational Conference and CRB Symposium
Looking Back at cr yotank moni toring

2026 MolDx Symposium Committee
Jeanne Ohrnberger, Ph.D., HCLD/CC(ABB), Chair
Barbara K. Timm, M.Sc., ELD/TS(ABB)
Marilyn Barbour, M.Ed., BSMT(ASCP)
Erin Morton
Stephanie Rinehart
Melanie Clemmer, Ph.D., HCLD(ABB), Chair
Christine Allen, Ph.D., M.Sc., TS(ABB)
Sharon Anderson, Ph.D., HCLD/ELD/CC(ABB)
Alison Bartolucci, Ph.D., HCLD/TS(ABB)
Wayne Caswell, M.Sc.
Matthew Hudnall, M.Sc., TS(ABB)
Eva Schenkman, Ph.D., M.Sc., HCLD/ELD/TS/ CC(ABB)
Richard Slifkin, M.Sc., TS(ABB)
1981 - Lucien Dean Hertert
1982 - Joseph A. Lazaroni, Ph.D.
1983 - Bernard I. Diamond, BLD(ABB)
1984 - Darwin S. Johnson, BLD (ABB)
1985 - David Birenbaum
1986 - Richard C. Paul, BLD(ABB)
1987 - Shirley Cresswell, HCLD(ABB)
1988 - Stanley C. Schaffer, BLD(ABB)
1989 - Nicholas T. Serafy, HCLD(ABB)
1990 - John A. Boffa, HCLD(ABB) and Alvin M. Salton, HCLD(ABB)
1991 - Everett S. Beneke, Ph.D., HCLD(ABB)
1994 - J. Douglas Hutchison, Ph.D., HCLD(ABB)
1996 - Melvin C. Bishop, HCLD(ABB)
2003 - Brooks A. Keel, Ph.D., HCLD(ABB)
2004 - Mark S. Birenbaum, Ph.D.
2007 - Nicholas T. Serafy Jr.
2009 - Richard G. Rawlins, Ph.D., HCLD/ELD(ABB)
2010 - Pat Lanza
2011 - David L. Smalley, Ph.D., BCLD/PHLD(ABB)
2012 - Annette Iacono
2016 - Robert I. Footlik, M.S., HCLD(ABB)
2024 - Tammie Schalue, Ph.D., HCLD/ELD/CC(ABB)

for the following ABB/ABOR examinations and PEER-approved events:
May 28-30, 2026, Thursday-Saturday
4th Annual GENES Conference
Loews Coronado Bay Resort Coronado, CA
Pre-Approved by ABB/PEER for up to 10 hours For more information contact: info@genesconference.com
June 18, 2026, Thursday
AAB Board of Registry Examination
Havert L. Fenn Center Fort Pierce, Florida
*
July 31-August 1, 2026, Friday-Saturday
CRB’s 2026 Laboratory Management and Leadership Workshop
Hilton Kansas City Airport
Kansas City, Missouri
Up to 12 Contact Hours Approved
October 24, 2026, Saturday
ABB Examination
Hilton Baltimore Inner Harbor
Baltimore, Maryland
Prior to ASRM Annual Meeting
Applicants from states other than California and Florida can take the AAB Board of Registry (ABOR) examination(s) at any accredited college or university that has a testing center.

Photographs & Memories, 2025 AAB Conference/CRB





















continued on page 53





































1980-81 - Richard A. Carr, BLD(ABB)
1981-82 - Elmer W. Hoyle, BLD(ABB)
1982-83 - James A. Elam, HCLD(ABB)
1983-84 - Clement W. Cochran, BLD(ABB)
1984-85 - Francis P. Monaco, BLD(ABB)
1985-86 - Alvin M. Salton, BA, MBA, HCLD(ABB)
1986-87 -
-
2004-05 -
2005-062006-072007-08 -
-
-

1996-97 - Nicholas T. Serafy Jr.
1997-98 - Robert J. Kokoski, Ph.D.
1998-99 - Brooks A. Keel, Ph.D., HCLD(ABB)
1999-00 - Helen Lewis, MCLD(ABB)
2000-01 - Pennell Painter, Ph.D., BCLD(ABB)
2001-02 - James E. Prier, Ph.D., HCLD(ABB)
2002-03 - Melvin C. Bishop, HCLD(ABB)
2003-04 - Patrick J. Quinn, Ph.D., HCLD/ELD(ABB)


David F. Carpenter, Ph.D.
Sperm Preserved for 3 Days NO PREDICTED MOTILITY
IVF-Grade Semen Analysis ACCURATE, AFFORDABLE, CONVENIENT
Only WHO-Compliant Reporting CLIA-CERTIFIED LABS
Avail able in all 50 US States INSURANCE ELIGIBLE






2013-14 - Joseph D. Musto, Ph.D., BCLD(ABB)
2014-15 - Kristen Zocco
2015-16 - Barry R. Behr, Ph.D., HCLD/ELD/CC(ABB)
2016-17 - Dolores J. Lamb, Ph.D., HCLD(ABB)
From page 50
2017-18 - Joseph D. Musto, Ph.D., BCLD(ABB)
2019-20 - Judy Kennedy
2020-21 - David Smalley, Ph.D., BCLD/PHLD(ABB)
2021-22 - Annette Iacono
2022-23 - Bonny Lewis Van, Ph.D., HCLD(ABB)
2023-24 - Joseph Conaghan, Ph.D., HCLD/ELD/CC(ABB)
2024-25 - John J. Weiss, Ph.D.

International Society For Clinical Laboratory Technology (ISCLT)
1963-65 - Darwin Kolle, RMT
1965-67 - Thomas Robinson, RMT
1967-69 - Stephen Bellack, RMT
1969-71 - Marvin France, RMT
1971-73 - Keith Knudson, RMT
1973-74 - Joseph P. Kelly, RMT
1974-75 - Robert Row, RMT
1975-76 - Marie Sherbell, RMT
1976-77 - Peter W. Lorey, MT(AAB)
1977-79 - Gordon Tolbert, RMT
1979 - Marlene Stemen, MT(AAB)
1979-80 - Gerard P. Boe, RMT
1980-81 - Peter W. Lorey, MT(AAB)
1981-83 - Anthony A. Metzcus, RMT
1983-85 - Michael J. Muzzillo, D.Sc., BLM(ABB), MT(AAB)
1985-87 - Nancy Matson, MT(AAB)
1987-89 - Richard J. Vidrine, MT(AAB)
1989-91 - Danny Yocom, MT(AAB)
1991-93 - Marilyn Moore, MT(AAB)
1993-95 - Michael J. Muzzillo, D.Sc., BLM(ABB), MT(AAB)
1995-97 - Jo Lynn Koehler, MT(AAB)
1997-98 - Richard R. Jones Sr., BA, MT(AAB)
AAB Associate Member Section (AMS)
1998-99 - Richard R. Jones Sr., BA, MT(AAB)
1999-01 - Sheryl Jensen, MT(AAB)
2001-03 - Shirley Lovelace, MT(AAB)
2003-05 - Margaret “Peg” Moran, MT(AAB)
2005-07 - Linda Jo Meierhans, MT(AAB)
2007-09 - Richard R. Jones Sr., BA, MT(AAB)
2009-11 - Susan M. Miller, MT(AAB)
2011-13 - Samuel H. Leggett III, MT(AAB)
2013-15 - Joan M. Fulk, MT(AAB)
2015-17 - Nancy M. Henderson, MT(AAB)
2017-19 - Kenneth Jensen, GS(ABB), MT(AAB)
2020-23 - Richard R. Jones Sr., BA, MT(AAB)
2023-25 - Samuel H. Leggett III, MT(AAB)









Photographs & Memories, 2025 AAB Conference/CRB Symposium from page 53












Andrology Store by Spectrum
www.andrologystore.com .............................pg. 22
Astec Bio
www.AstecBioUSA.com pg. 3
Birenbaum & Associates
www.aab.org pg. 2
BIRR
www.birr.nl ....................................................pg. 34
Boreas Monitoring
www.boreasmonitoring.com .........................pg. 46
Centogene
www.centogene.com ....................................pg. 42
CooperSurgical
www.coopersurgical.com pg. 12
CRD Associates
www.dc-crd.com ...........................................pg. 26
CryoFuture
www.cryofuture.co pg. 33
Cryohexagon
www.cryohexagon.com pg. 52
DeTech Solutions
www.detechus.com ......................................pg. 44
eIVF
www.eivf.org .................................................pg. 36
ESCO Medical
www.esco-medical.com pg. 47
Eurofins CellTx www.eurofins.com/medical-device pg. 29
Fairfax Cryobank/Fairfax Eggbank www.fairfaxcryobank.com, www.fairfaxeggbank.com pg. 37
Fellow
www.meetfellow.com ....................................pg. 43
HB Lab Innovations
www.hblabinnovations.com pg. 35
IMT Matcher
www.imtmatcher.com ...................................pg. 13
IVF Academy
www.ivfacademyusa.com pg. 15
IVF Store/Biotech
www.ivfqc.com .................. inside front cover/pg. 1
Juno Genetics
www.junogenetics.com .................................pg. 48
Kitazato
www.kitazato-ivf.com pg. 28
LifeAire Systems
www.LifeAire.com .........................................pg. 23
Luminary Genetics
www.luminarygenetics.com ..........................pg. 31
Medical Electronic Systems (MES)
www.mes-global.com pg. 9
MetaSystems
www.metasystems-international.com ...........pg. 38
NextGen LifeLabs www.nextgenlifelabs.com pg. 45
Nexpring Health www.nexpringhealth.com ...............................pg. 5
Noblegen Cryogenics www.noblegencryogenics.com .....................pg. 49
O’Connell & Aronowitz www.oalaw.com ............................................pg. 54
Progenesis
www.progenesis.com pg. 17
Reproductive Solutions (RSI) www.rsifertility.com .......................................pg. 17
Sapyen www.sapyen.co ............................................pg. 50
Sperm and Embryo Bank of New Jersey www.sperm1.com/sebnj/ outside back cover
Thermo Fisher Scientific www.thermofisher.com/pgt ...........................pg. 40
Viable Biosciences www.viablebio.com .......................................pg. 39
Vitrolife Group www.vitrolifegroup.com ................................pg. 41
Vitromed www.vitromed.com .......................................pg. 53

Peak lab hours = overlapping vitrification, warming, and retrievals. Every minute outside the incubator matters.
1-minute, 1-step warming (>90% time reduction)
Same trusted TS-NX formulation – just faster
Flexible packaging – micro vials or 20 mL packs
Lab Benefits
New protocols available in select regions
Same proven formulation (since 2019)
Flexible timing
1-10 minute equilibrium options
Seamless workflow integration
Save critical minutes during peak hours
No new validation required
Continuous CSCM system from culture to vitrification
Minimal workflow disruption
Ready to Go Faster?
Contact Nexpring Health Redefining the future of ART.
