Life Sciences and Industry Magazine
Interview
Duncan Lugton, Head of Policy and Impact at IChemE (UK), on scale-up investments in Engineering Biology


Life Sciences and Industry Magazine
Duncan Lugton, Head of Policy and Impact at IChemE (UK), on scale-up investments in Engineering Biology



With the new MALDI-8030 EasyCare you can do routine maintenance yourself. The compact instrument is easy to clean and quickly back in use again. Adding its outstanding performance parameters, it’s the ideal solution for high sample throughput, especially in a clinical environment.
Self-service maintenance
Less maintenance costs, increased uptime
Compact benchtop design
Allows installation almost anywhere in the lab
Outstanding performance
Offers comparable performance parameters to larger and more expensive MALDI-TOF instruments
www.shimadzu.eu/

A true multi-talent
Enables a wide range of applications, e.g. protein and polymer analysis or quality control for (bio)-pharmaceuticals
Available as linear positive MALDI-8020 EasyCare

SOFIE CARSTEN NIELSEN is the Director of the European Biosolutions Coalition and of the biosolutions industry community in Danish Industry since May 2023. She is a former political party leader and former Minister of Higher Education and Research, and was a member of the Danish Parliament for more than 10 years. She is a political scientist by education and has a Master’s in European Politics and Administration from the College of Europe, Bruges.
As the global race for biosolutions intensifies, Europe is at a critical crossroads, and I am deeply concerned about the future of our continent’s biotech industry. While countries like China and the United States rapidly advance their biotech industries, Europe’s slow and not up-to-date regulatory system poses a significant risk to maintaining our leadership. With recent global events and tariffs shaking global trade, immediate action is required to ensure our bio-companies thrive. We need a bold and broad EU Biotech Act to boost competitiveness, accelerate the green transition, and enhance European self-reliance.
Biotechnological solutions – we call them biosolutions – can meet global needs by boosting food production and enabling sustainable industrial processes. A McKinsey report highlights that advanced biotechnology could generate up to US$1trillion in global economic value over the next 15 years. The biosolutions industry supports over 700,000 jobs in Europe, strengthening sustainable supply chains and a circular economy.
However, biosolutions in Europe face fragmented regulatory regimes that hinder companies from scaling efficiently. They lack funding opportunities and encounter lengthy approval processes, which dishearten many firms. Addressing regulatory inefficiencies and enhancing strategic investments is necessary to secure our competitive edge – and there is no time to waste.
Three critical areas need immediate attention to help Europe’s biotech sector thrive:
1. Our regulatory processes are the slowest worldwide, adhering to outdated legislation, impairing our competitiveness. Reforming these frameworks is crucial to unleashing Europe’s innovative potential.
2. Second, strategic investment is vital to scaling biosolutions beyond pharmaceuticals. Investing in biomaterials can lead to groundbreaking products like biodegradable silk. We must ensure financing is accessible and avoid barriers that hinder investments.
3. Third, prioritising biosolutions within EU industrial policy will accelerate the shift to a bio-based economy and position Europe as a leader in sustainability. Advanced biotechnology can generate significant economic value, reduce CO2 emissions, and save water.
The need for action is undeniable. To solidify our role as a global leader in sustainability and innovation, Europe must embrace biosolutions by speeding up registration processes, ensuring accessible finance and infrastructure, and prioritising biosolutions in our industrial strategy. We must urge the European Commission, the European Parliament, and the EU Council to enact a bold and broad European Biotech Act to champion biosolutions as the cornerstone of our competitiveness and green transition. Let setbacks become comebacks and seize this opportunity to lead the biorevolution for a sustainable future in Europe. ■

Today, almost every workplace is a digital workplace, and we make them all work better across industries of every kind. In healthcare, TeamViewer is helping to transform outcomes. We enable doctors to give live virtual assistance to nursing staff, in medical settings and out in the community. Medical equipment can also be operated remotely and securely. The digital workplace company—connecting people with technology—enabling, improving, and automating digital processes. teamviewer.com/workbetter

Whatever your digital workplace, make work work better.

Many industrial biotech projects compete directly with established and optimised petro chemical processes, and most struggle to be as efficient on cost. Big outlays for new production plants can make price parity for greener processes a distant dream, choking off industry investment and further support from policymakers. Now a publicly funded French-German-Belgian initiative is showing how petroleum-based industrial advantages can shrink once a bioeconomy facility is up and running.
8 EC to speed up technology transfer in the EU by introducing a start-up and scale-up strategy
10 EMA to simplify EU market authorisation rules for biosimilars
12 Industry protests against Counciladopted EU pharma package
18 Analyst commentary
19 European Biotech Stocks
22 Update on clinical trials
25 Closing the gaps in quality management
28 Startup: ions.Bio
40 Focus: Techparks/Bioengineering
41 Bayer Berlin develops a Life Sciences Campus
43 Interview: Duncan Lugton, Head of Policy & Impact, IChemE, UK
46 It’s time to rethink biotech scaling
47 Unlocking Pichia for industry
48 Interview: Jeffrey Truong, Helmholtz Munich, Germany
50 Ensuring flexibility and resilience
52 Why life sciences leaders need more than science
60 Northern Europe: Sweden, Denmark, Norway and Finland
62 Western Europe: France, Belgium, The Netherlands and the UK
64 Central Europe: Germany, Switzerland and Austria
66 Southern Europe: Italy, Spain, and Portugal
68 Spanish biotech – its growth, impact and global role
69 Eastern Europe: Poland, Lithuania and the Czech Republic
FOCUS NEW MODALITIES
70 SLAS Europe 2025 in Hamburg: Innovation in lab automation tools drive discovery of new modalities
72 ThirdLaw Molecular: Spiroligomers –not antibodies, not small molecules
73 MabSwitch: Antibodies with allosteric ON/OFF switch
53 Biopeople
74 Science & Technology
76 News from associations
80 Company index
81 Events
82 Encore
IMPRINT European Biotechnology (ISSN 2364-2351) is published quarterly by: BIOCOM Interrelations GmbH, Jacobsenweg 61, D-13509 Berlin, Germany, Tel.: +49-30-264921-0, Fax: +49-30-264921-11, Email: service@european-biotechnology.com, Internet: www.european-biotechnology.com; Publisher: Andreas Mietzsch; Editorial Team: Thomas Gabrielczyk (Editor in Chief), Derrick Williams (Co-editor), Dr. Georg Kääb, Uta Mommert, Maren Kühr, Advertising: Oliver Schnell, +49-30-264921-45, Christian Böhm, +49-30-264921-49, Andreas Macht, +49-30-264921-54; Distribution: Nancy Weinert +4930-264921-40; Graphic Design: Michaela Reblin; Production: Martina Willnow; Printed at: Königsdruck, Berlin; European Biotechnology Life Sciences & Industry Magazine is only regularly available through subscription with a BIOCOM CARD. Annual subscription BIOCOM CARD Europe: €80 for private individuals (students €40) incl. VAT, €120 plus VAT for corporates. Prices includes postage & packaging. Ordered subscriptions can be cancelled within two weeks directly at BIOCOM AG. The subscription is initially valid for one calendar year and is automatically renewed every year after. The subscription can be cancelled at any time and is valid until the end of that calendar month. Failures of delivery, which BIOCOM AG is not responsible for, do not entitle the subscriber to delivery or reimbursement of pre-paid fees. Seat of court is Berlin, Germany. As regards contents: individually named articles are published within the sole responsibility of their respective authors. All material published is protected by copyright. No article or part thereof may be reproduced in any way or processed, copied, and proliferated by electronic means without the prior written consent of the publisher. This issue contains a supplement of mts med-tech supplies GmbH, Cover Photo: © G.Go - stock.adobe.com; ® BIOCOM is a registered trademark of BIOCOM AG, Berlin, Germany.
“Uncertainty” is the word most often used to describe President Donald Trump’s approach to trade, science and pharmaceutical pricing policy. His strategy seems to be to shock first and negotiate later – even with arch-rival China, which is challenging the United States’ decadeslong dominance in most biotech fields. Europ E an Biot E chnology takes a closer look at the situation as of June 2025.


Although ongoing trade disputes and protectionist policies dominate the headlines, regions and industry leaders that recognise the potential of bioengineering are already preparing for the future. EuropE an BiotEchnology spotlights emerging open innovation clusters and cutting-edge bioengineering platforms.
Science is not flawless. It’s a deeply human endeavour that’s often slow to accept breakthroughs. Just consider the HPV cancer vaccine, which took two decades to gain mainstream recognition. Prestige often goes to the well-connected, measured more by h-index than by true innovation. Yet one thing will never change: science depends on international cooperation. This cooperation doesn’t arise due to personal rapport. It’s born of necessity. Only by working together can we develop cures for untreatable diseases, build waste-free industries, secure food supplies despite threats like avian flu, or construct engines powered by CO2 and saltwater. These ideas and others are not dreams. They’re the foundation of a sustainable, competitive economy. Today, that foundation is cracking. The current US administration is undermining global science by banning foreign talent, replacing experts at the FDA and CDC with loyalists, promoting protectionism, and slashing NIH, NASA and NSF funding. These moves threaten to stall biomedical progress –and hand scientific leadership to authoritarian regimes like China.
The US is still a scientific powerhouse. But isolationism weakens global innovation. The democratic world must recover from its shock and act with unity, resolve and a renewed dedication to working together: Make Collaboration Great Again.

Thomas Gabrielczyk Editor-in-Chief

EUROPEAN COMMISSION With a new billion euro fund, the European Commission aims to bridge the valley of death for EU hightech SMEs. According to Reuters, a private-public €10bn scale-up fund is intended to be set up next year to generate more deeptech and biotech unicorns in the EU. However, no one has yet raised their hand to provide the necessary capital.
The European Commission wants to set up a huge public-private fund to bridge the financial ‘valley of death’ for technology companies that face scale up production. The fund is part of the new strategy ‘Choose Europe to start and scale’, published on 28 May 2025. It aims to reduce Europe’s lag in the number of biotech, AI and quantum computing unicorns compared to the US and China. The strategy could give a boost to Europe‘s leading food-tech and microbiology sector, which is ready to scale climateneutral, healthy and efficient alternatives to classical food, fuel and chemical production processes (see p. 3, 14). So far, most EU developers suffer from EU bureaucracy and a lack of public co-funding to set up industry-scale production plants in Europe. However, the market potential is high due to the fact that bio mass and precision fermentation create resilience against agricultural pathogens and problems in the chemical supply chain. Both new markets for biotech solutions together generated 13 times more revenue in 2024 than the pharmaceutical industry, where biotech drugs are among the top-selling products.
€10bn to build unicorns?
According to agency and media reports, the European Commission plans to launch a public-private scale-up fund worth at least €10bn to increase the number of European unicorns, i.e. hightech companies with a minimum turnover of €1bn. However, the official European Commission document titled ‘The
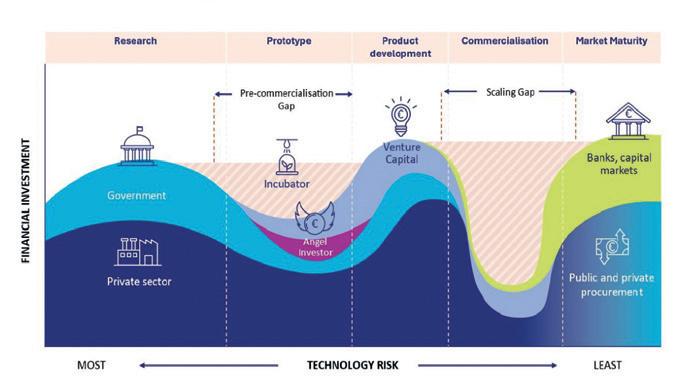
a new fund, the EC aims to bridgte the double valley of death for EU hightech SMEs.
EU Strategy for Start-ups and Scaleups – Choose Europe to start and scale’ (COM(2025) 270 final) does not contain any specific reference to a €10bn fund. Instead, the document refers to the creation of a ‘Scaleup Europe Fund’ with a public component and approximately four times as much private funding. The Reuters news agency quotes an ‘EU official’ who is said to have confirmed that a multi-billion euro amount is being targeted for the fund. The fund would acquire shares in promising companies and be managed by a private investment manager. The strategy document also does not provide any information on whether funds from existing programmes will be reallocated or new funds will be made available by the Commission. Just like the Clean Industrial Deal, the funding depends on the EU’s next multian -
nual financial framework (MFF) being increased. A decision on this, and thus also on the EU Commission’s ambitious goals, is expected this summer. Additional money would then start flowing in 2028.
Central to the new start-up and scaleup strategy is the ambition to improve access to finance. While private capital availability, including venture funding, has increased in recent years, according to the EU Commission, a notable funding gap remains for companies moving from early development to scale (see figure). This is especially true for biotech ventures, which often require substantial capital. “Although the EIC Fund has become one of Europe’s most important deep tech investors in just four years,

the demand for EIC support from excellent deep tech companies cannot currently be met,” commented Rafael Laguna de la Vera, head of the German innovation agency SPRIND. The Commission emphasised the importance of optimising existing EU-level instruments – such as InvestEU and the European Innovation Council (EIC) – to facilitate adequate funding for proof-of-concept, regulatory preparation, and market expansion.
Beyond financial support, the Commission seeks to create more favourable regulatory conditions. The current framework, though rigorous in consumer protection and ethics, is often complex and inconsistent across Member States. This fragmentation leads to delays in product approvals, complicated cross-border operations, and increases compliance costs, particularly for biotech companies subject to both national and EU health and safety regulations. Furthermore, many hightech companies struggle to recruit and retain staff with advanced expertise in science, regulation, or digital technologies. The strategy promotes greater mobility of highly skilled professionals within the EU and calls for enhanced coordination among research bodies, industry, and national authorities to better align skills development with sector-specific needs.
The Commission also highlights the need for a more interconnected innovation ecosystem like in the US biotech hub Boston, which is a one-stop-shop for investors, according to EY. However, the EC stresses the role of regional innovation hubs, university spinouts, and crossborder platforms in facilitating knowledge transfer, accelerating technology maturation, and supporting market entry. Frameworks such as Horizon Europe and Digital Innovation Hubs are key tools to support startups navigating complex development paths, especially in life sciences and health technologies.
■ t.gabrielczyk@biocom.eu
EU REGULATION The EU health watchdog EMA has asked biotech industry stakeholders to share their thoughts on its draft guidelines for developing and assessing biosimilars. The idea is to make it easier for patients in the EU to get access to biosimilars, while also keeping Europe an attractive place for developers.
The new approach for equivalence testing of biosimilars, which is outlined in a draft reflection paper, is designed to reduce the amount of clinical data required for the development and approval of biosimilar medicines. It builds on the 2024 Procedure for European Union guidelines and related documents within the pharmaceutical legislative framework. The EMA‘s consultation will end at the end of September and will be implemented in 2026.
While Europe has long been dominated with its homegrown biosimilar developers, foreign competition is gaining ground. Between 2022 and 2024, 11% of EMA-approved biosimilars came from India or South Korea (CAGR 30%), a 3% increase over the past three years. This trend is expected to accelerate as more non-European firms enter the space.
Biosimilars have been authorised based on studies comparing them to their reference medicines. This includes a comparability exercise on the quality aspects of
the active substances in both the biosimilar and the reference product, and the demonstration of clinical efficacy as well as safety of the biosimilar in confirmatory clinical trials. The EMA’s draft now suggests that demonstrated structural and functional comparability, together with comparative data on pharmacokinetics, may be sufficient to demonstrate similarity with the reference product. If adopted, the new rules are set to reduce the need for extensive clinical efficacy studies. Between now and 2030, 69 biologics in Europe and 118 in the US will lose exclusivity, creating an opportunity eight times larger than the 2012 to 2014 period.
Biosimilars have become important therapeutic options improving patient access to essential treatments such as costly biologics. By providing competition in the market, they can expand patient access to critical medicines. Today, biosimilars are available in six major therapeutic areas, contributing to 6.9 billion patient treatment days since the first EU approvals in 2006. However, more than 90% of biosimilar development is focused on oncology and immunology, leaving other therapeutic areas underserved. The EMA’s more streamlined approach would ultimately ensure wider availability of biosimilar medicines to patients in the EU. ■

Closely monitoring and analyzing samples at frequent timepoints is key to efficient bioprocessing. The Bioprocess Autosampler from Eppendorf provides this solution to you. Whether you are working in cell culture or fermentation, in early R&D or process development, with single-use or glass bioreactors, the Bioprocess Autosampler can handle sampling and bolus addition, helping you gain complete datasets and automate bioprocess control.
To learn more about our Bioprocess Autosampler, visit:

EUROPEAN COMMISSION Pharmaceutical associations have responded with sharp criticism to the adoption of the revised general pharmaceutical legislation by the EU Council of Ministers in the form of a directive and a regulation. They argue that the measures are not suitable for bringing Europe back to the forefront of pharmaceutical research and will make the EU an unattractive location for investment.

The criticism from the EU pharmaceutical association EFPIA and the German VFA is directed primarily at the restriction of intellectual property rights for pharmaceutical innovations, in particular for orphan drugs, which currently enjoy 20 years of patent protection plus a fiveyear supplementary protection certificate (SPC) and up to 11 years of data exclusivity protection (8 + 2 + 1) after approval.
The pharmaceutical industry lobby groups criticise the reduction of data exclusivity for original medicines from eight to six years – a measure intended to improve access to treatment even in resource-poor health systems – as an illtimed and misplaced experiment. They argue that it exposes original products to competition with cheaper generics and biosimilars too soon. While China and the US are sending clear signals to drug
innovators, the EU would forfeit its claim to leadership with this draft. Planned changes to market authorisation (simultaneously in all EU countries), the obligation to conduct comparative clinical trials and the permission to conduct investigator-driven clinical trials for indication expansion in areas with high unmet medical needs without the consent of the drug developer also make pharmaceutical lobbyists fear for future sales. The VFA is threatening the EU that this would lead to companies leaving in the long term.
However, US President Donald Trump’s threat of 80% price cuts in the world’s most expensive pharma market, the United States, makes this warning ring hollow. Last year, large pharmaceutical companies generated around 53% of their sales in the US, 20% in the EU and 7.5% in China, according to IQVIA. Nev-
ertheless, the EFPIA claims: “The decision to reduce intellectual property protection for pharmaceutical companies makes Europe less attractive, deters investment and jeopardises the development of innovative therapies in Europe without removing the underlying barriers and delays in access for patients.” VFA President Han Steutel goes one step further: “If you want innovation, you have to enable investment,” he said. He also said that the six-month advance notice period for supply shortages provided for in the Medicines Act is too bureaucratic and therefore impractical.
Furthermore, while the introduction of transferable exclusivity vouchers for antibiotics with a novel mechanism of action is a good incentive for the development of new antibiotics, the VFA criticises that the design is so cumbersome that the new instrument remains ineffective. Following the withdrawal of the pharmaceutical industry for reasons of profitability, antibiotic innovations are mainly taking place in biotechnological SMEs. In the context of the upcoming trilogue negotiations, the EU Parliament and the Council of Ministers must make changes to ensure the competitiveness of the EU as a location, the pharmaceutical associations demand. The EU medicines package is also intended to increase the number of clinical trials conducted in the EU. However, these lag significantly behind the declining figures in the US since 2015 and the rapidly rising figures in China since 2015.
■
t.gabrielczyk@biocom.eu





BIOECONOMY Many industrial biotech projects compete directly with traditional petro-based production processes. And many that start out in publicly funded industrial biotechnology start-up ecosystems in Europe fail because they’re simply not at first competitive when it comes to costs. The petrochemicals industry has had decades to optimise both production processes and return on investment in existing plants. Now a French-German-Belgian project is showing how those advantages dwindle when a bioeconomy plant is up and running.
To reach the goals set out in Europe’s Clean Industrial Deal, it will be essential to rapidly transform energy-intensive, fossil-based industries. So to kick things off, let’s take a look at the deal’s objectives and do a quick reality check.
Biotechnology has emerged as the central technology platform capable of supporting the planned transition. It offers a wide range of tools for doing so, among them energy-efficient biocatalysts for the chemical sector, climate-friendly production of food and animal feed proteins, microbial recovery of rare critical materials, manufacturing of pharmaceutically active substances and many others. These applications are closely aligned with objectives laid out in the EU Bioeconomy Strategy (2020). These include achieving food security, promoting the sustainable use of natural resources, reducing dependence on fossil inputs, mitigating climate change, and contributing to economic competitiveness and job creation within the European Union.
Despite its big ideas and huge potential, however, the EU has often lagged behind in the global race to ‘biologise’ industrial processes. Countries with more aggressive strategies like the US, China, Singapore and Brazil are more often viewed as benchmark setters. China for instance has invested heavily in fermentation capacity. Singapore is expediting regulatory approval for novel foods. Brazil is advancing with a clearly defined bioeconomy roadmap. The new government in the US, on the other hand, seems to have slammed the brakes on what largescale biomanufacturing has achieved there so far (s. page 54). Might that mean opportunities for Europe?
One of the biggest barriers to reaching clean industry goals within the EU is the high capital expenditure (CAPEX) associated with building demonstration or production facilities. Many biotech processes could be competitive with petrochemical alternatives in terms of operating costs. But when major new
infrastructure investments are required to get things up and running, that doesn’t mean much. Many a promising business model has struggled to survive the Valley of Death between innovation and industrial implementation. Many don’t.
The Circular Bio-based Europe Joint Undertaking (CBE JU) was set up to help more of these projects get the support they need. It’s a €2bn partnership between the European Union and the Biobased Industries Consortium (BIC) that funds projects advancing competitive circular bio-based industries in Europe. The partnership seeks to build on the success of its predecessor, the Bio-based Industries Joint Undertaking (BBI JU) while addressing the current challenges facing the industry. To reach clean industry goals and hit ambitious climate targets by shifting away from non-renewable fossil raw materials and minerals, the EU will have to strengthen its resource independence and support sustainable economic growth.
The stated role of CBE JU is to bring together various groups – ranging from farmers to scientists to large bio-based industries – to solve the technological, regulatory and market challenges facing the sector. Its public-private funding scheme is supposed to boost innovation and market deployment, and pave the way for future investment.
There are many EU-funded projects in circularity and biobased industries. A short list from the field of biomass utilisation alone reveals a very wide range in the field:
› Rural BioReFarmeries: Develops small-scale, decentralised green biorefineries to enhance farmer sustainability and competitiveness. Funding: €7.35m. Coordinator: Munster Technological University, Ireland.
› ONE EARTH: Establishing a circular bioeconomy by valorising residual animal biomass for terrestrial and marine
applications. Funding: €4m. Coordinator: University of Bologna, Italy.
› LUCRA: Demonstrating sustainable production of bio-succinic acid from organic waste using integrated electrochemical bioreactors. Coordinator: Bio Base Europe Pilot Plant, Belgium.
› NewWave: Develops sustainable, biobased manufacturing lines by converting biomass residues into valuable products. Funding: €8.5m.
› Bio-LUSH: Valorises underutilised European plant resources like forest residues or marine plants to produce high-quality fibres for sustainable products. Coordinator: Stockholm University, Sweden. When it comes to nuts and bolts, the BBI JU/CBE JU-funded AFTERBIOCHEM project is one of the very few projects that has managed to actually build a flagship biorefinery, now up and running in Carling Saint-Avold (France). There, sugar industry by-products and other non-food agricultural biomass are turned into seven high-added-value bio-based organic acids for fragrances and flavourings, personal care, food and feed, industrial chemicals and a potassium-rich fertiliser. Accounting for around half of global production, the EU is the world’s largest producer of sugar beets. The crop is important to Europe’s economy, but prices for beet-derived sugar have fallen, so the sector is being forced to capitalise on every potential income source. The industry supports 140,000 farmers in the bloc, as well as around 27,000 direct jobs in processing and further upstream employment. Until 2017, sugar-beet production in Europe was shored up by a quota system. However, since this system was scrapped, its end and other factors have led to a 30% fall in sugar prices. EU sugar-beet producers have responded by increasing production. But against a backdrop of worldwide overproduction of the commodity, that move has only increased price pressures.
Being competitive is key
AFTERBIOCHEM brought together 12 partners at different stages of the value chain: four SMEs, seven large companies
and one innovation cluster. The partners came from five European countries: Belgium, France, Germany, the Netherlands and Switzerland. They included German sugar producer Südzucker, Belgian nutritional products and services firm Kemin, French nail-polish manufacturer Fiabila and Dutch chemical company Celanese. The project’s core technology is based on ten years of R&D at its lead partner, French bio-based organic acid producer Afyren. It has developed an all-in-one, cutting-edge fermentation process that uses natural microorganisms coupled with separation and transformation steps.
The process releases no hazardous substances into the environment and produces just a fifth of the CO2 emitted in the production of fossil-based organic acids. It has also been optimised to cut waste. All fermentation outputs are turned into valuable products. “We don’t have any waste at the end of the process, we recycle water in the process, and we will massively reduce CO2 emissions. At the end of the process, we have a co-product – a high-value fertiliser with a very high potassium content –which goes back into the ground to start a new cycle of biomass production,” explains Afyren CEO and co-founder Nicolas Sordet. (read the complete in-depth interview with Nicolas Sordet online at european-biotechnology.com)
The technology has allowed the company to create multiple sustainable value chains at an industrial scale that connect numerous sectors, reducing dependence on non-renewable resources.
It took over a decade for Afyren to evolve from a start-up into the operator of an industrial-scale facility. Founded in 2012, the firm went public in Paris in 2021 and established its production site in the Moselle region, close to customers in Germany and the Benelux region. That move created 130 industrial jobs. Initially seen as a promising stock, Afyren’s share price peaked in late 2021, but by mid-2023 had declined sharply. Share price declining after the IPO is a
trend that many bio tech companies experience. The road to delivering promised products is often a long one.
In an interview with E urop E an B iot E chnology, Sordet doesn’t seek to hide the fact that people who have invested in his company might have been disappointed at how things have developed so far. But he also says Afyren’s challenges are typical for industrial biotech firms. “From the very beginning, the objective was to create a first plant whose model could be quickly replicated,” says the CEO. But building that plant required an investment of around €100m from partners who are willing to take a very long-term perspective when it comes to returns. “As an industrial biotech start-up, we’re stuck in the middle of Venture Capital and Private Equity. The public funds help until the proof-ofconcept, but afterwards there is hardly anything,” Sordet says. One of the venture firms that provided support early on was Sofinnova Partners, whose Industrial Biotech strategy invests in bioeconomy projects.
For its IPO in 2021, Afyren was able to leverage a kind of window of opportunity for companies deploying on the newly highlighted sustainability goals (SDGs). But that window closed shortly after, and the war between Russia and Ukraine shifted priorities in industries, governments and societies. The rising uncertainty in global trade conditions has made an already shaky economic environment worse.
With money from the IPO and public-private funding from the AFTERBIOCHEM project, Afryen managed to get its first plant up and running. That puts them in a position to commercialise at price levels comparable to the petroindustry, but with a much better carbon footprint. Analysts from Oddo BHF and Portzamparc (BNP Paribas) have therefore maintained a “buy” rating, with a price target of €5, citing financial results broadly in line with forecasts.
The company views its 16,000-tonnecapacity facility as the first of its kind. With €33m in reserves and €19m in new loans, Afyren is now aiming to optimise
production and reach profitability. While smaller listed firms have struggled in the current financial climate, Afyren’s plant and diversified product base have put it in a relatively strong position.
The company’s plans for the next steps don’t involve licensing its technology, but instead keeping it close and running more plants with partners in regions close to feedstock sources, Sordet says. Afyren sees its technology as a modular building block for chemical industries that want and have to reduce their carbon footprints. Preparations are now being made for more facilities, with two additional plants planned for construction in the coming years.
One major sign of success for a publicly supported biobased project is when industrial partners extend their collaboration beyond the funding period. Afyren and Südzucker, for instance, extended their strategic bioeconomy partnership in May.
In the last few years, the two companies have built a unique long-term feedstock partnership, creating a new value chain within Europe. “Our partnership with Südzucker, Europe’s leading sugar producer, has played a key role in securing long-term, competitive access to essential feedstocks and ensuring supply security,” says Nicolas Sordet. Marcus Reuter, Head of Co-Products at Südzucker, says that “working with Afyren enables us to upcycle sugar-beet byproducts into high-quality chemicals, thereby helping reduce dependence on petroleum-based derivatives.”
One of the partnership’s key strengths lies in its geographic proximity. Südzucker owns thousands of hectares of land near Afyren’s flagship biorefinery on the FrenchGerman border. That ensures access to locally sourced, bio-based raw materials.
The publicly propelled project has now turned into a solid business collaboration: a good example for industrial biotechnology in general – and a beacon of hope for the whole sector. ■ g.kaeaeb@biocom.eu
VENTURE CAPITAL European VC company Sofinnova Partners has been investing for more than a decade in early-stage companies that use biotechnology to have a positive environmental impact. A first Green Seed fund (at €22.5m) kicked off in 2012, and its successor funds Industrial Biotech-I (2017 with €125m) and IB-II (2021 with €175m) have only grown in popularity.
EuroBiotech _ Sofinnova has a decade of experience in industrial biotechnology investments. What has changed during this period?
Joško Bobanovi ć _Historically, we were among the early adopters in this field in Europe. Currently, there is a robust investment ecosystem with numerous entities investing in the sector and new ones emerging. These include funds dedicated to industrial biotechnology or those broadly investing in climate-tech and sustainability.
Additionally, the number of startups and entrepreneurs has significantly increased. Deal flow has doubled in less than four years, with a notable improvement in quality. In Europe, there is considerable scale-up demonstration capacity that attracts companies even from outside the continent, allowing startups to concentrate on execution rather than building demonstration facilities.
In end markets, there has been a substantial increase in demand for sustainably produced ingredients driven by strong consumer interest. Furthermore, startups are addressing issues related to supply chain resilience and local production, meeting current needs.
EuroBiotech SDG w as a driver of transformation in Europe . Has this changed?
Joško Bobanović_ We have regarded Sustainable Development Goals (SDGs) primarily as a universal language that facilitates the alignment of sustainability interests across various themes. The direction in Europe has remained consistent, as end markets continue to seek innovative

JOŠKO BOBANOVIC Partner Industrial Biotech Strategy at Sofinnova Partners, Paris (France)
solutions to address significant challenges, many of which are defined by the SDGs. Our fund investors are keen to support innovative sustainable solutions, and we have collaborated with some of them to develop precise methods for tracking the environmental impact of their initiatives.
EuroBiotech _ Some CEOs at industrial biotech startups say that a ‘pharma exit model‘ is missing. Do you agree?
Joško Bobanovi ć _Yes. To date, liquidity for investors has primarily been generated through IPOs. Although some of these public listings occurred relatively early and faced standard development challenges, there are still several publicly list-
ed companies whose stocks are performing well. However, the M&A market, which is prevalent in the biopharma sector, is largely absent in the industrial biotechnology space. In biopharma, innovation is developed at venture-backed companies and brought to market by large pharmaceutical firms. In contrast, large companies in industrial biotech are currently more focused on acquiring businesses, revenue and profits – sometimes with innovative products – but have not yet prioritised acquiring innovation.
We anticipate this shift will occur as industrial biotechnology startups create future -oriented products that are produced sustainably and enhance supply chain resilience in the current geopolitical climate. Large companies will need to respond to consumer demands for new performance, sustainability and clean label products, which can only be achieved by leveraging the innovation engines within the startup ecosystem.
EuroBiotech_What do you think can help us achieve a sustainable environment for sustainable companies?
Joško Bobanovi ć _It is essential to have exits to support investment in the sector and create successful examples. Success encourages further success, attracts new entrepreneurs to the sector, and expands opportunities. For this to occur, large companies should consider how to find external innovation beyond their internal labs, acknowledging the value small companies offer through their technology and innovative approaches. ■ g.kaeaeb@biocom.eu
JILL JORTNER, INVESTMENT ANALYST T. ROWE PRICE In addition to the uncertainty triggered by the US government with serious changes at the FDA and other healthcare institutions and an unclear stance of the US Secretary of Health and Human Services on various areas of innovation, the growing demand for health care services gives a positive momentum that can provide stability.
The US health care sector is going through a lot of changes right now, similar to when the Affordable Care Act was introduced. People are using health care services more than ever after COVID-19, with increased hospital visits, doctor appointments, and medication use, especially expensive specialty drugs.

This is good news for health care providers and the drug supply chain, but it’s challenging for insurance companies. They are struggling with slow government payments and new costs from recent amendments to laws, which means they might be more cautious with their Medicare plans in 2026.
Spending on specialty medicines is increasing because of new cancer treat-
ments and more uses for existing drugs. A recent law has made these medicines cheaper for patients, leading to increased demand. Companies that distribute drugs and manage pharmacy benefits are in a good position to profit, especially with new, cheaper versions of brands (i.e. the wave of newly approved and disseminated biosimilars) becoming available.
However, there are political and regulatory challenges. Budget talks, the possibility of new taxes on drugs, and policy changes under the current Health and Human Services Secretary are creating uncertainty. Proposed changes to Medicaid could reduce coverage, and the end of extra insurance subsidies might lower enrolment and usage.
German IMMUNIC INC. (Gräfelfing near Munich; Nasdaq: IMUX), a biotech company developing oral therapies for chronic inflammatory and autoimmune diseases, announced the closing of its public offering of (i) prefunded warrants, (ii) Series A warrants, and (iii) Series B warrants to purchase common stock or pre-funded warrants. Immunic received about US$65m upfront and could gain up to US$130m more if all Series A and B Warrants
are exercised. The financing was co-led by BVF Partners and Coastlands Capital, with participation from several institutional investors.
Swiss Pharma giant NOVARTIS AG announced an agreement to acquire Regulus Therapeutics (Nasdaq: RGLS), a San Diego-based clinical-stage biopharma company developing microRNA therapies for autosomal dominant polycystic kidney disease (ADPKD). Regulus’ lead
Despite these issues, the political situation might provide some stability. With narrow Republican majorities and past failures to repeal the Affordable Care Act, major changes to healthcare programs are unlikely. There is resistance to big Medicaid cuts, and recent laws show a more moderate approach. Investors should expect small policy changes rather than big ones. Although really every forecast must be evaluated with particular caution.
In summary, the health care sector in the US is experiencing both increased demand and uncertainty. Providers and pharmaceutical distributors are doing well, while the insurers and payers face challenges. Political gridlock might help stabilise things, but the sector remains dynamic and will and has to be closely watched by investors in 2025. ■
candidate, farabursen, targets miR-17 and recently completed a Phase 1b trial. Novartis will pay US$0.8bn upfront, with up to US$0.9bn in potential milestone payments. The deal aligns with Novartis’ focus on renal therapies and is expected to close in the second half of 2025, pending customary conditions. Regulus used to be a pioneer in microRNA research and development of drugs addressing the gene regulation machinery in humans
■
2cureX AB 0.34 6,740k
4SC AG 0.99 10,320k
A40WHL 0.63 19,600k
AB Science SA 1.14 68,160k
Abionyx Pharma SA 1.24 42,970k
Abivax SA 5.68 357,250k
AC Immune SA 1.66 164,050k
Active Biotech AB 0.02 19,440k
Adaptimmune Therapeutics plc 0.21 349,870k
ADC Therapeutics SA 2.94 291,618k
Addex Therapeutics Ltd 0.04 6,790k
ADL Bionatur Solutions SA 0.21 14,770k
Adocia SAS 3.99 70,630k
Advicenne SA 1.76 21,380k
Aelis Farma SAS 1.15 14,390k
Affimed NV 0.96 14,600k
Akari Therapeutics plc 0.93 62,740,000k
ALK-Abelló A/S 24.18 4,900,000k
Alkermes plc 25.20 4,190,000k
Allarity Therapeutics A/S 0.83 12,300k
Alligator Bioscience AB 0.35 6,000k
Altamira Therapeutics Ltd 0.09 427k
Alvotech SAS 8.04 2,130,000k
Alzinova AB 0.14 15,520k
Annexin Pharmaceuticals AB 2.63 20,976k
Aprea Therapeutics AB 1.54 8,531k
Aqua Bio Technology ASA 0.06 6,920k
Arctic Zymes Technologies ASA 1.43 72,270k
Arecor Therapeutics plc 0.32 11,780k
Argenx BV 475.40 29,190,000k
Arocell AB 0.04 8,340k
Arterra Bioscience SpA 2.32 15,500k
Ascelia Pharma AB 0.36 31,910k
Ascendis Pharma A/S 151.00 9,330,000k
Aton 0.01 6,720k
Autolus Therapeutics plc 1.76 468,390k
Avacta Group plc 0.35 145,780k
Avadel Pharmaceuticals plc 8.05 783,590k
Axichem AB 0.21 12,650k
Basilea Pharmaceutica AG 54.90 670,040k
Bavarian Nordic A/S 23.89 1,910,000k
BB Biotech 31.15 1,720,000k
Bergenbio ASA 0.10 6,550k
BICO Group B 3.22 221,870k
Bicycle Therapeutics plc 6.95 331,940k
Bioarctic AB 18.14 1,340,000k
Bioextrax AB 0.28 11,280k
Biofrontera AG 2.43 14,770k
Biogaia AB 9.30 912,700k
Bioinvent International AB 3.29 214,190k
Biomed-Lublin SA 1.17 83,370k
Biomérieux SA 120.40 14,250,000k
BioNTech SE 91.75 22,120,000k
Biophytis SA 0.13 2,870k
Bioporto Diagnostics A/S 0.19 84,200k
BioSenic SA 0.00 1,370k
Biosergen AB 0.05 11,980k
Biotage Sweden AB 12.98 1,040,000k
Bioventix plc 31.40 165,110k
Biovica International AB 0.05 3,210k Brain AG 2.01 45,440k
Camurus AB 54.85 3,230,000k
Cantargia AB 0.09 21,550k
Carbios
The unique and most complete list of share price developments of biotech companies listed in Europe – exclusively in European Biotechnology Magazine.
Cellectis SA 1.26 90,260k
Celon Pharma SA 4.99 290,290k
Celyad Oncology
Co.don AG 0.03 82,843k
Coegin Pharma AB 0.30 7,420k
CombiGene AB 0.23 4,670k
Cosmo Pharmaceuticals NV 63.00 1,010,000k
Therapeutics
DBV Technologies SA 1.68 223,500k
Diagonal Bio AB 0.00 360,000k
Diamyd Medical AB 0.89 121,120k
Diasorin SpA 93.54 5,050,000k
e-Therapeutics plc 0.09 53,170k
Elicera Therapeutics AB 0.48 21,400k Ellen AB 0.07 730k
Enzymatica AB 0.27 66,510k
Epigenomics AG 0.88 76,996k
Erytech Pharma SA 0.10 1,020k
Eurobio Scientific SA 24.95 258,270k
Eurofins Scientific SE 60.40 10,730,000k
Evaxion Biotech A/S 2.10 11,473k
Evgen Pharma plc 0.00 1,070k
Evolva N 1.19 8,620k
Evotec SE 7.11 1,260,000k
Expres2ion Biotech Holding AB 1.99 5,280k
Faron Pharmaceuticals Oy 2.62 294,440k
Fermentalg SA 0.50 44,920k
Flerie AB 4.22 329,050k
Fluicell 0.85 1,170k
Formycon AG 26.40 485,770k
Fusion Antibodies plc 0.07 824k
Gabather AB 0.00 4,724k
Galapagos NV 24.98 1,650,000k
Genedrive PLC 0.01 7,810k
Geneuro SA 0.07 2,110k
Genfit SA 3.56 177,690k
Genflow Biosciences plc 0.01 3,630k
GENinCode plc 0.02 5,020k
Genmab A/S 198.15 12,180,000k
Genovis AB 2.40 158,430k
Genoway SA 3.45 32,530k
Gensight Biologics SA 0.21 27,800k
Gentian Diagnostics AS 5.50 82,660k
Genus plc 22.80 1,520,000k
Global Bioenergies SA 0.12 2,310k
Glycorex Transplantation AB 0.19 14,360k
Guard Therapeutics International AB 1.58 32,770k
Hansa Biopharma AB 2.17 151,630k
HBM Healthcare Investments AG 184.20 1,290,000k
Heidelberg Pharma AG 4.24 204,580k
Hemogenyx Pharmaceuticals plc 1.95 8,960k
Herantis Pharma Oyj 1.28 31,200k
Hofseth Biocare ASA 0.15 71,900k
HOOKIPA Pharma 1.29 12,614k
Idorsia Ltd 2.29 467,840k
Immatics NV 5.32 643,000k
Immunic AG 0.69 65,160k
Immunovia AB 0.04 12,370k
ImmuPharma plc 0.03 12,490k
Infant Bacterial Therapeutics AB 4.84 63,070k
InflaRx NV 0.67 45,050k
Innate Pharma SA 1.64 150,980k
Integragen SA 0.52 3,430k
Intervacc AB 0.06 13,770k
Inventiva SA 2.78 402,150k
IO Biotech Inc. 1.15 75,699k
IRLAB Therapeutics AB 0.63 32,830k
Isofol Medical AB 0.09 12,950k
ISR Holding AB 0.19 6,760k
Kancera AB 0.10 9,490k
Kuros Biosciences AG 28.16 1,090,000k
Lipigon Pharmaceuticals AB 0.01 1,060k
Lipum AB
Lytix Biopharma AS
MaaT Pharma SA 5.18
Mabion Ltd 2.25
Magle Chemoswed
Mainz Biomed BV
Marinomed Biotech AG
MDxHealth SA
Medesis
Medigene
Medincell SA 15.26 507,340k
Medivir
Mendus AB
Merus BV
Modus Therapeutics Holding AB
Therapeutics
Newron Pharmaceuticals SpA 7.70 153,700k
Nextcell Pharma AB 0.08
Resources
NLS Pharmaceutics AG
Novacyt SA
Novozymes Biopharma DK A/S
NuCana ADR 0.12
Nykode Therapeutics ASA 0.13
ObsEva SA 9.22 1,080,000k
Okyo Pharma Ltd 2.16
Oncimmune Holdings plc 1.49 1,661k
Oncoarendi Therapeutics SA 1.39 28,800k
Oncopeptides AB 0.13 11,860k
OncoZenge AB 0.56 6,640k
Open Orphan plc 0.11 74,200k
Optibiotix Health plc 0.13 13,530k
Orphazyme A/S 135.00 5,610k
Oryzon Genomics SA 2.88 226,610k
OSE Immuno SA 5.11 117,640k
Ovoca Bio plc 0.01 1,280k
Oxford Biodynamics plc 0.00 2,940k
Oxford Biomedica plc 3.64 386,180k
Oxurion NV 0.02 127k
Paion AG 0.02 17,549k
Pangaea Oncology SA 1.70 58,500k
PCI Biotech Holding ASA 0.14 5,540k
Pentixapharm Holding AG 2.50 60,100k
Pharma Mar SA 80.60 1,480,000k
Pharming Group NV 0.98 664,320k
Photocure ASA 4.30 114,590k
Physiomics plc 0.00 60,642k
Plant Advanced Technologies SA 8.66 10,170k
PolyPeptide Group SF 21.25 706,120k
Poolbeg Pharma plc 0.03 15,750k
Poxel SA 0.64 32,850k
Predilife SA 3.51 13,060k
ProQR Therapeutics BV 1.59 169,390k
Prostatype Genomics AB 0.08 423k
Proteome Sciences plc 0.03 8,270k
Prothena Corporation plc 4.88 264,830k
Pure Biologics SA 0.98 3,910k
Q-Linea AB 0.00 19,290k
Qiagen NV 40.93 8,860,000k
Quantum Genomics SAS 0.07 4,740k
Redx Pharma plc 0.17 66,130k
Relief Therapeutics Holding AG 2.35 33,080k
Reneuron Group plc 0.03 1,940k
Ryvu Therapeutics SA 6.97 163,690k
Saniona AB 0.79 106,520k
Santhera Pharmaceuticals AG 12.54 161,530k
Sareum Holdings plc 0.17 29,570k
Sartorius AG 172.20 5,940,000k
Scancell Holdings plc 0.09 83,980k
Scandion Oncology A/S 0.00 37,562k
Selvita SA 6.34 116,010k
Sensorion SA 0.38 118,760k
SenzaGen AB 0.46 13,310k
Shield Therapeutics plc 0.03 28,650k
Silence Therapeutics plc 4.40 623,440k
Simris Group AB 0.01 7,170k
Skinbiotherapeutics plc 0.17 40,260k
Softox Solutions AS 0.01 6,720k
Sophia Genetics SA 2.50 167,080k
Spexis AG
Stayble
TME Pharma NV 0.08 4,940k
Transgene SA 0.95 96,930k
Trinity Biotech plc 0.57 212,410k
uniQure NV 13.04 706,800k
Nijmegen-based Byondis BV dosed the first cancer patient in a Phase I escalation and expansion trial with its novel SIRPalpha-directed monoclonal antibody (mAb) BYON4228 alone and in combination with MSD’s PD-1 immunocology blockbuster pembrolizumab. The trial is designed to assess the safety, pharmacokinetics, pharmacodynamics and efficacy of the blocker of the CD47-SIRPalpha axis, responsible for tumors’ ability to escape from recognition and destruction by the immune system.
Immunoncoly specialist Transgene SA and Tokyo-based NEC Corp. have presented promising data on Transgene’s personalised cancer neoantigene vaccine TG4050 at the American Society of Clinical Oncology (ASCO) Annual Meeting. In patients with resectable HPV-negative locally advanced head and neck squamous cell carcinoma, TG4050 proved to be safe and feasible in the adjuvant setting. Furthermore TG4050 induced immune responses to vaccine neoantigens that lasted for up to two years. All trial endpoints (NCT04183166) were met including safety, feasibility, immune activation and disease-free survival (100%).
In early June, Osivax SAS completed its Phase IIa clinical booster trial (NCT06582277) of its nucleoprotein-targeting broad-spectrum universal influenza vaccine OVX836 in 117 healthy subjects. Osivax expects final results in H2 2025.
NSCLC/SCLC
At ASCO conference, Tolremo Therapeutics AG (Basel) presented data from its ongoing Phase I study of TT125-802, an orally administered bromodomain inhibitor of CBP/p300, an epigenetic master regulator of transcriptional resistance, for patients with advanced solid tumours who have relapsed or are refractory to standard-of-care therapies. TT125-802 demonstrated an acceptable safety profile without thrombocytopenia and anti-tumour activity in advanced solid tumors, including five responses out of seven patients with non-small cell lung cancer (NSCLC).
Roche AG reported results from the Phase III IMforte study of its PD-L1 blocker atezolizumab in combination with PharmaMar SA‘s lurbinectedin as a first-line maintenance treatment for people with extensive-stage small cell lung cancer (ES-SCLC), following induction therapy with carboplatin, etoposide and atezolizumab. Patients in the IMforte study first completed four cycles of atezolizumab combined with chemotherapy, over the course of approximately three months, before being randomised into maintenance treatment. From the point of randomisation, the median overall survival (OS) for the Tecentriq plus lurbinectedin regimen was 13.2 months versus 10.6 months for atezolizumab alone. Median progression-free survival (PFS) by independent assessment was 5.4 months versus 2.1 months, respectively. No new safety signals were observed.
In the first week of June, Immunic AG (Martinsried, Germany) reported complete enrollment for two Phase III trials (ENSURE) with vidofludimus calcium (IMU-838) in patients with relapsing multiple sclerosis and Phase II study data in patients with progressive MS. In the patient population (n=467) of the Phase II trial, Immunic reported a clinically meaningful reduction of the haz-
ard ratio (HR) for the relative risk of 24week confirmed disability worsening (24wCDW) by 24% compared to placebo (HR 0.76). Further analyses by disease subtype showed that vidofludimus calcium was associated with a 33% reduction in 24wCDW in the primary progressive multiple sclerosis (PPMS) study population (n=152) compared to placebo (HR 0.67), a 19% reduction in the non-active secondary progressive multiple sclerosis (naSPMS) study population (n=268) compared to placebo (HR 0.81), and a 34% reduction in the active secondary progressive multiple sclerosis (aSPMS) study population (n=47) compared to placebo (HR 0.66).
Based on these results, 1,121 patients in ENSURE-1 and 1,100 patients in ENSURE-2 have been randomised to receive either 30 mg daily doses of vidofludimus calcium or placebo. The primary endpoint for both trials is time to first relapse up to 72 weeks. Secondary endpoints include time to confirmed disability worsening based on the Expanded Disability Status Scale (EDSS), volume of new T2-lesions, time to sustained clinically relevant changes in cognition, and magnetic resonance imaging (MRI)based endpoints. Top-line results are expected to be published in Q4/2026, according to CEO, Daniel Vitt.
In June, NodThera Ltd started dosing in its Phase II RESOLVE-1 (RESolution Of infLammation to treat obesity and cardioVascular disEase) trial investigating the potential of its oral NLRP3 inflammasome inhibitor NT-0796, in nondiabetic and diabetic patients with obesity. Within the randomised, double-blind, placebo-controlled trial, the British study sponsor will evaluate the efficacy and safety of NT-0796 in 160 patients over 24 weeks. There will be three treatment groups receiving either NT-0796 orally twice daily, NT-0796 orally once daily, or placebo. The trial’s primary endpoint is the change in
































































Fördergesellschaft IZB mbH Am Klopferspitz 19 82152 Planegg/Martinsried
Tel. + 49 (0)89.55 279 48-0
Fax + 49 (0)89.55 279 48-29 info@izb-online.de www.izb-online.de












Site: 26,000 m2, S1 laboratories
Real estate and facility management on site
Faculty Club and conference rooms for up to 100 people
Kindergarden (Bio Kids)
Hotel CAMPUS AT HOME
Restaurant SEVEN AND MORE, THE BOWL Food Lounge
IZB network with more than 40 start-ups
In the immediate vicinity are two Max Planck Institutes, faculties and facilities of the LMU, Munich University Hospital – Großhadern
body weight in participants from baseline to week 24. Secondary endpoints include the impact of NT-0796 on body composition, metabolic biomarkers, and HbA1c levels in participants with type 2 diabetes. Headline data from the study are anticipated in Q2 2026.
Cytovation ASA bagged an extra US$6m (NOK62m) through a second Series A extension to drive multi-centre Phase II testing of its beta-catentine blocking cancer vaccine CY-101 in patients with adrenocortical carcinoma and related cancers that are driven by WNT/beta-catenine signalling. The multi-centre Phase II trial will be conducted by Cancer Research UK as announced earlier this year in ACC patients. It will start in late 2025 and is expected to deliver first clinical readouts next year. According to updated preclinical data in adrenocortical carcinoma patients presented at the AACR meeting in Chicago (25-29 April, 2025), CY-101 triggered CD8+ T cell infiltration through its membranolytic activity and inhibited oncogenic Wnt/beta-catenin signaling by activating Axin2. CY-101 induced a dose-dependent cell death across mouse and human beta-catenindriven colorectal cancer (CRC), adrenocortical carcinoma, and melanoma cell lines in vitro , completely eradicating ACC tumours and significantly enhancing the efficacy of anti-PD-1 therapy in CRC and melanoma models. Furthermore, CY-101-treated tumours exhibited downregulation of Wnt/beta-catenin downstream target genes. These new preclinical results support Cytovation’s observations seen in the first-in-human CICILIA Phase I/IIa trial, which reported clinical activity in ACC and melanoma patients.
Spanish SpliceBio SA launched a Phase I/II study on dual-AAV-based therapy SB-007 for Stargardt Disease in midMarch. The company is using protein splicing to address rare familial disorders caused by mutations in large genes
such as retinal disorder Stargardt disease. So far, Stargardt disease has remained elusive to gene therapies due to the large size of the underlying defective ABCA4 gene. However, using split inteins – auto-processing domains –to carry out protein splicing, it has become possible to repair very large genes through protein splicing.
According to Vila-Perelló, Chief Executive Officer and co-founder of SpliceBio, “SB-007 is the first gene therapy in clinical development designed to restore expression of the full-length ABCA4 protein across all Stargardt disease patients, regardless of their mutations.”
In the Phase I arm of the study, cleared by the US Food & Drug Administration (FDA) in December 2024, SpliceBio will evaluate the safety and efficacy of a single dose of SB-007 administered subretinally.
NHL
British ADC maker Iksuda Therapeutics Ltd completed dosing of its first B cell non-Hodgkin lymphoma patient with CD19-directed IKS03 in early June. In the multicentre first-in-human study (NCT05365659), Iksuda Therapeutics will evaluate the safety, tolerability, preliminary antineoplastic activity, pharmacokinetics and pharmacodynamics of increasing dose levels of IKS03, and determine the recommended dose for dose-expansion. First hints to efficacy will be evaluated in disease-specific expansion cohorts.
At the end of May, ADC maker Heidelberg Pharma AG dosed the first patient in a Phase I study evaluating HDP-102 for the treatment of non-Hodgkin lymphoma (NHL). HDP-102 is Heidelberg Pharma’s second drug candidate from its proprietary Amanitin-payload ADC technology (ATAC technology) platform to enter clinical development. The ATAC targets CD37, a key antigen expressed on many B-cell lymphoma cells.
PYODERMA GANGRENOSUM
InflaRx NV has terminated Phase III development of its complement C5a
blocker vilobelimab in pyoderma gangrenosum (PG) following an interim analysis the Independent Data Monitoring Committee (IDMC) due to futility. This decision was based on data analyses of the first 30 patients enrolled in the study, with no unexpected adverse events noted by the IDMC.
Leuven-based Augustine Therapeutics
NV dosed the first patient in a Phase I clinical trial evaluating AGT-100216, the first peripherally-restricted, selective HDAC6 inhibitor (HDAC6i) for the treatment of Charcot-Marie-Tooth disease.
The first-in-human trial is designed to evaluate the safety, tolerability, pharmacokinetics and exploratory pharmacodynamics of oral AGT-100216 in healthy adult volunteers receiving either single ascending or multiple ascending doses of AGT-100216.
German Boehringer Ingelheim presented clinical data from two early-stage trials targeting the signal regulatory protein alpha (SIRPalpha) innate immune checkpoint at the 2025 American Society of Clinical Oncology (ASCO) Annual Meeting in Chicago. In a Phase Ib study conducted by Boehringer Ingelheim, the first-in-class SIRPalpha monoclonal antibody, BI 765063, which was licenced from French Ose Immunotherapeutics SA , demonstrated a manageable safety profile as well as preliminary signs of immune activation and additive antitumour activity when combined with PD-1 blocker ezabenlimab and cetuximab in patients with recurrent/metastatic (R/M) head and neck squamous cell carcinoma (HNSCC).
Additionally, in an open-label, Phase I trial, the second licenced SIRPalpha antibody, BI 770371, alone and in combination with ezabenlimab, was well tolerated in patients with advanced solid tumours. There were no dose-limiting toxicities in either treatment arm, and the maximum tolerated dose was not reached in either group. ■
QUALITY MANAGEMENT In the life sciences industry, Quality Management (QM) has long been synonymous with compliance: checking boxes to meet regulatory demands. But that definition no longer holds up. Today, QM must do much more than ensure compliance; it needs to enable speed, scalability, and sustained innovation across complex, global operations. To do that, prominent gaps need to be filled.
› Oliver Nürnberg, Director Product Management, Tenthpin Solutions AG
Life sciences companies are currently facing a growing list of pressures and challenges that are redefining the role of quality. This includes heightened compliance requirements and more frequent audits that are placing new demands on quality systems; fragmented data sources and nonharmonised processes that make it difficult to ensure consistent quality across regions or business units; transitioning from legacy or paper-based systems to modern digital platforms; and talent shortages in quality and compliance roles. It’s a lot.
Sector-specific pressures are adding complexity. For example, in the pharmaceuticals sector, companies face tighter batch release controls, greater traceability demands, and diverging global regulatory Oliver Nürnberg

standards. In medtech, the integration of device software and artificial intelligence (AI) introduces new validation and postmarket surveillance requirements.
Current market gaps
There are several critical gaps in today’s QM systems, thanks to insights gathered in collaboration with Tenthpin’s customers’ projects. These gaps can be allocated into two types; functional and technological.
Functional gaps include the likes of certificate handling, system integration, audit, and supplier management, as well as risk management. Verification of inbound certificates and generation of out-
Therapeutic antibody discovery & development



bound certificates remains largely manual or disconnected. There are still gaps between supply chain, manufacturing, and quality systems, limiting end-to-end traceability and response. Many solutions lack robust tools for audit preparation, supplier quality monitoring, and non-conformance tracking. Plus, few systems offer deeply embedded risk management features that can guide realtime decision-making. Technology gaps are also persistent. For example, AI tools, while increasingly available, remain difficult to validate and deploy in GxP-regulated environments. Smart automation is limited due to the essential need for human oversight in high-risk quality processes. There are still few truly interoperable platforms that offer an end-toend view of quality across the enterprise. These gaps impact both operational efficiency and the ability to maintain compliance at scale. They need to be closed, but how?
The answer is the range of Quality Management solutions designed to meet different needs across the value chain.
Enterprise Quality Management System (QMS) platforms remain a popular choice due to their configurability and scalability. Platforms that offer a wide range of capabilities, from document control and training management to deviation tracking and CAPA workflows. But the trade-off for customisation is complexity, which can impact heavily-regulated industries like life sciences. ERP-integrated QM modules present a streamlined alternative. By embedding quality processes within the same ecosystem as finance, procurement, and supply chain operations, these modules facilitate data flow and reduce duplication of effort. However, they may lack the depth and flexibility required for complex quality workflows.
Laboratory Information Management Systems (LIMS) also play a prominent
role for companies with heavy lab-based testing and sample management requirements. While LIMS solutions excel at managing test data and ensuring traceability at the sample level, they are not designed to handle broader quality system functions, such as supplier management or complaint handling. Custombuilt tools and workflows have emerged as a practical solution for many companies, especially those with unique business processes or legacy constraints. These solutions are often developed inhouse or co-created with partners to address specific gaps that off-the-shelf platforms cannot cover. But which approach is the right one?
To overcome the challenges in quality management (QM), the top 20 life sciences companies are deploying winnable strategies that are worth paying attention to.
One common trend among industry leaders is the move toward standardised, global platforms. By consolidating disparate systems and processes into unified solutions, life sciences companies are improving data consistency, enhancing traceability, and reducing the operational burden of maintaining multiple compliance frameworks. This standardisation also enables more effective governance, allowing organisations to monitor quality performance with greater clarity.
A stronger emphasis on strategic partnerships is proving to be a critical development. Leading firms are co-innovating with technology vendors and consulting partners to create solutions tailored to industry-specific needs while remaining scalable and compliant. These collaborations bring fruitful results with accelerators or industry templates that reduce implementation time and validation effort. Increasing their focus on establishing robust governance models is also a key trend. This includes clearly defined roles and responsibilities, integrated risk management practices, and performance monitoring mechanisms to ensure quality is maintained and continu -
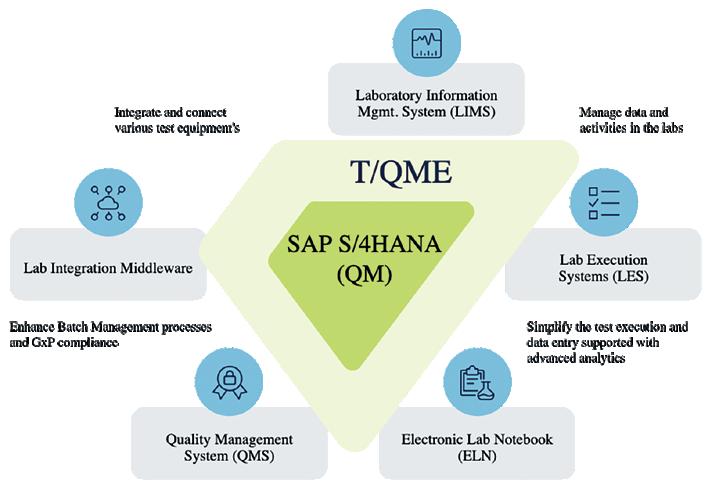
How Tenthpins’s T/QME improves quality management and insights developed by larger players can be adapted and scaled down effectively.
ously improved. By embedding quality into business planning and decision-making processes, they are making it an enabler of growth rather than a bottleneck. But not everyone is a top 20 company. So, what can smaller life sciences companies learn from it? By adopting prevalidated solutions and industry-standard frameworks, smaller firms can reduce implementation risk and accelerate time-to-value. Using proven technologies and best practices means they don’t have to start from scratch – many of the tools
Gaps remain in Quality Management; manual processes, outdated legacy systems, AI deployment in GxP environments, and talent shortages. But life sciences companies that make thoughtful, strategic investments in scalable, compliant, and digital-ready QM frameworks
will be best positioned to navigate future complexity. At Tenthpin, we’re addressing the challenges with co-innovations like SAP Batch Release Hub (BRH) and our own solutions within the Tenthpin Intelligent Quality Suite, including Tenthpin Quality Management Evolved. Right now, 16 of the top 20 life sciences companies trust our expertise. You can too. Explore Tenthpin’s industry standard solutions to see how we help close the Quality Management gaps and futureproof your approach. ■


Philipp Geyer, Ph.D., MBA, Founder & CSO, Managing Director
Sebastian Virreira Winter, Ph.D., Founder & CTO, Managing Director
Sophia Viellechner, Ph.D., Founder & CBO
About the company
ions.bio offers a comprehensive, all-inone solution for biomarker discovery. We design proprietary biomarker studies, partner with clinical experts to collect samples, and convert these samples into extensive multi-omics datasets. These datasets are rigorously analyzed using our advanced bioinformatics and AI-driven pipeline. The outcome is a new generation of innovative biomarkers. We actively collaborate with biotech and pharma companies, providing end-to-end support and services throughout the entire biomarker discovery and validation process.
The IZB is a vibrant hub for biotech innovation, enabling start-ups like ours to tackle pressing medical challenges and drive progress in biomarker discovery.
Our platform leverages cutting-edge mass spectrometry and ensures high-quality and reproducible results. Our clients benefit from our transparent project management, with regular updates and clear communication at every stage. We are committed to accelerating the transition from discovery to clinical application, helping clients unlock the full potential of their data.
At ions.bio, we are fueled by the desire to revolutionize healthcare through our expertise in mass spectrometry, advanced data analysis, and well-characterized bio-

FOUNDED: 2024
NUMBER OF EMPLOYEES AT THE IZB: 5
BUSINESS SEGMENT: Services, Biomarker Discovery
samples. Our goal is to make groundbreaking discoveries in clinical samples, enabling earlier disease detection and the discovery of novel therapeutic targets. By harnessing cutting-edge technology and collaborating across disciplines, our dynamic team is committed to bridging this gap. We aim to push the boundaries of biomarker discovery, advancing precision medicine and ultimately improving patient outcomes through innovative solutions that can detect diseases sooner and guide the development of new drugs.
CONTACT ions.bio Am Klopferspitz 19 82152 Planegg-Martinsried info@ions.bio www.ions.bio
2nd half-year 2025

BIOSPAIN From October 7 to 9, 2025, Barcelona will host a new edition of BIOSPAIN. Under the theme “Global crossroads, local vibes,” BIOSPAIN 2025 reaffirms its mission to connect Spanish biotechnology with the global stage.
BIOSPAIN returns in 2025 stronger than ever. Following a record-breaking 2023 edition – bringing together nearly 1,000 companies from 34 countries, over 2,200 attendees, and more than 5,000 partnering meetings – the upcoming event will take place from October 7 to 9 in Barcelona, reaffirming its status as a must-attend gathering for the biotech sector at both national and European levels.
Barcelona’s selection as host city is no coincidence. It is one of Southern Europe's leading life sciences innovation hubs, supported by a robust network of research centers, startups, universities, and pharmaceutical companies. This ecosystem makes the Catalan capital an ideal setting for fostering collaboration and advancing biotech development. BIOSPAIN stands as Spain’s flagship biotech-
nology event and one of the continent’s most influential. Its program tackles the industry’s key challenges and emerging trends, ranging from advanced therapies, personalised medicine, and industrial biotechnology to sustainability and digital transformation.
BIOSPAIN is a dynamic forum that brings together the entire ecosystem –investors, entrepreneurs, researchers, and institutional representatives. Its multidisciplinary approach encourages highvalue interactions and networking opportunities at every level.
In addition, its carefully curated social agenda fosters a relaxed atmosphere for building connections, exchanging ideas, and sparking new synergies. In 2025, BIOSPAIN is once again set to be the epicenter of biotech talent and innovation.


7 - 9 October, 2025
Barcelona, Spain

GREETING We are honoredtowelcomeyoutoa neweditionofBIOSPAIN, taking place from October 7 to 9, 2025, in Barcelona.Followingtheunprecedented success of the 2023 edition with a focus on internationalisationundertheclaim“Globalcrossroads, localvibes,”wereturnwithrenewedenthusiasm to further consolidate this gathering as theleadingbiotechnologyeventinSpainand one of the most significant across Europe.
AsoneofSouthernEurope’smainlifesciences hubs, Barcelona provides the perfect settingtodriveinnovation,fostercollaboration,andpromotesectorgrowth.Wewarmly invite you to be part of this unique experience that brings together all key players in the ecosystem: companies, researchers, investors, institutions, and more.
BIOSPAIN is a space for connection, knowledge, and forward thinking. We encourage you to take part, to share, to discover new opportunities—and to enjoy a social agenda designed to strengthen relationships. See you in Barcelona.
Ion Arocena CEO of AseBio
Registration/program
https://biospain2025.org/attend/ https://biospain2025.org/program/


DRUG DISCOVERY 2025 Drug Discovery 2025, an ELRIG conference, welcomes researchers from all scientific disciplines to learn, collaborate and explore the latest technologies to support the discovery of new therapeutics.
In October we welcome over 3,000 people to Liverpool, UK for Drug Discovery 2025, Europe’s largest conference dedicated to drug discovery in the life sciences community.
Why should
To learn! With two days of in-depth scientific content covering the full spectrum of the drug discovery and development process, there is something for everyone. Each day there are six parallel scientific tracks, poster sessions and specialist talks, so you can explore your interests
and perhaps look for new ideas and technologies to help your research.
To explore ! The exhibition hall is open throughout the conference, with over 200 companies, there is no better place to find out what tools and services are available to enable your research. The Tech Theatre hosts sessions on automation, robotics and AI, while the Breakthrough Zone is the space to find the newest innovations from the entrepreneurial exhibitors and tips to help you launch your own company.
To open doors! ELRIG is expecting over 3000 people to attend Drug Discovery

21–22 October, 2025
Liverpool Exhibition Centre, Kings Dock, Liverpool, UK
2025, there are dedicated sessions for those looking for careers advice, flash presentations from the Breakthrough Zone companies and networking opportunities. You never know where a conversation over a coffee might lead.
It’s free! Though you must register, all ELRIG events are free to attend, encouraging participation from all in the drug discovery community, enabling learning and collaboration across disciplines, academia, and industry.
ELRIG, is a UK-based not-for-profit volunteer-led organisation, dedicated to delivering inspiring events to the dynamic life science community. With a global community of over 22,000 life science professionals across all levels, ELRIG is committed to accessible and inclusive events.
21-22 October 2025
Liverpool Exhibition Centre, Kings Dock, Liverpool, L3 4FP
3,000+ delegates
>150 scientific talks
80+ speakers
200+ exhibitors

GREETING Welcome to Drug Discovery 2025! ELRIG is delighted to work with renowned organisations to bring the latest scientific research to our delegates. This year we are working with the Royal Society of Chemistry, British Pharmacological Society, SLAS, Cancer Re-
search Horizons, Alzheimer’s Research UK and The Protein Society. We also support the growth and development of the community by providing activities for our early career professionals in the Learning, Career & Breakthrough Zones. Join us and join in!
Del Trezise ELRIG’s Chair
400+ scientific posters www. elrig.org/portfolio/ drug-discovery-2025/
Programme Highlights
Plenary keynotes + 12 scientific tracks + Breakthrough Zone and Innovation Prize + Early Careers Professionals activities + Networking
21 – 22 October 2025 Exhibition Centre Liverpool
Drug Discovery 2025 is Europe’s largest free-to-attend life science conference, bringing together 3,000+ researchers to explore cutting-edge innovations and technologies in drug discovery.
80+ speakers
150 scientific talks
200+ exhibitors
400+ scientific posters
3,000+ delegates Breakthrough Zone and Innovation Prize Early Careers Professionals activities
12 scientific tracks

BIOTECH SUMMIT Join the 3rd BIOTECH SUMMIT AUSTRIA on October 23–24, 2025, in Graz – the heart of Austria’s Medical Science City. This highlevel conference connects biotech leaders, innovators, and investors to explore new trends, markets, and opportunities across Europe and beyond.
The BIOTECH SUMMIT AUSTRIA 2025 returns to Graz on October 23–24, bringing together leading minds from biotechnology, biopharma, medtech, research, and policy to exchange ideas, drive partnerships, and shape the future of health innovation.
As Austria’s flagship biotech conference, this two-day event offers a curated program designed specifically for executives, researchers, startups, investors, and decision-makers. Attendees will experience a dynamic mix of keynotes, best practice case studies, and panel dis-
cussions, focused on the most pressing trends and opportunities in the life science sector.
Key topics include:
› Innovation & translational research
› Internationalisation & global market access
› Funding & investment opportunities
› AI, digital health & emerging technologies
The summit also provides valuable networking formats, including pre-scheduled 1:1 meetings, informal sessions,

23–24 October, 2025 Graz, Austria
and a dedicated “Hike & Dine” pre-event (Oct 22) and “Dinner & Networking Party” on the first conference day.
Hosted by BIOTECH AUSTRIA, Human. technology Styria, and Health Hub Tirol, the summit builds on the success of previous editions in Graz (2023) and Innsbruck (2024). Past speakers include top voices from Roche, BioNTech, Novartis, MSD, AbbVie, and Helmholtz Centre for Infection Research.
Don’t miss your chance to connect, grow, and innovate with Europe’s biotech ecosystem.
Tickets available now!
› October 23–24, 2025 | Graz, Austria
› 250+ participants from 13+ countries expected
› Conference language: English
› www.biotech-summit-austria.com

















































BIO-EUROPE In 1994, BIO-Europe launched with the mission to drive biotech innovation and dealmaking forward through one-to-one partnering. Today, it proudly holds the mantle as Europe’s flagship partnering event. In its 31th edition in Vienna partnering will again become the science of connection.
Join us for the 31st BIO-Europe, the premier biopharma partnering event, in Vienna, Austria, November 3-5, 2025, one of the fastest growing life sciences hubs in Europe. BIO-Europe is set to bring together over 5,500 life science professionals from 60+ countries.
This flagship gathering offers unparalleled networking, innovative partnering programmes, and industry insights, making it essential for anyone in the biotechnology value chain. The more than
GREETING This year's BIO-Europe is heading to Vienna. Vienna's coffee houses have brewed ideas, debate, and innovations that have changed the world and BIO-Europe continues the tradition.

Over the last 31 years, BIO-Europe, has become Europe’s flagship partnering event. Its international reach
30,000 one-to-one meetings held onsite will connect leaders from academia, startups, and scaling-up biotech companies with pharma companies and forward-thinking investors.
Along with world-class workshops and panels, innovative company presentations, and an active exhibition, the variety of networking opportunities make this event an unrivalled forum for companies across the biotech value chain to meet and do business.
makes it a one-of-a-kind offering, with attendees from all parts of the biotechnology value chain gathering to efficiently identify, engage and form strategic relationships that drive their businesses successfully forward.
Come for the coffee. Stay for the connections.
Claire Macht Portfolio Director_EBD Group
EBD Group

Partnering at BIO-Europe is powered by partneringONE®, the gold-stand platform for licensing professionals to forge connections that lead to impactful collaborations. With a single login, delegates can schedule one-onone meetings, access on-demand content, and stay updated on conference activities.
Through three days of programme sessions, attendees will have the chance to learn about important trends and opportunities in life science from some of the brightest minds in the sector. The agenda will feature tracks focused on major industry trends including cell and gene therapy, artificial intelligence in drug development, and new technologies in drug discovery.
Together with the Biotechnology Innovation Organization (BIO), EBD Group is thrilled to welcome you to this “mustattend” partnering event for innovators and business leaders.
3–5 November, 2025 Vienna, Austria Registration https://informaconnect.com/ bioeurope/ Contact EBDcustomerservice@ ebdgroup.com
Phone +49 89 23121 7290 informaconnect.com/bioeurope
3–5, 2025 |VIENNA,

Join us for the 31st BIO-Europe
The premier biopharma partnering event, in Vienna, Austria, November 3-5, 2025. This flagship gathering is set to bring together over 5,500 life science professionals from 60+ countries, facilitating over 30,000 one-to-one meetings.
Register now before July 25, 2025 and save up to €1,500.
BIOEUROPE.COM

PHARMALAB 2025 In 2024, the conference track on potency assays was a great success and very well received. However, feedback from participants also revealed other areas of interest for exchanging experiences and opinions with colleagues and authorities’ representatives.
Currently, there is no European conference dedicated to Bioanalytical Control of Biological Drug Substances and Products. For many recent PharmaLab Congress participants, this represents a significant gap.
For this reason, the Call for Papers for PharmaLab 2025 specifically invited submissions on bioanalytical specifications, systems, and methods – such as residuals/ impurities, purity, identity (HPLC, CE, IEF, MS, etc.). As a result, the new conference track provides participants with the opportunity to explore and discuss advanced techniques for analyzing biological drug substances and products, as well as to exchange valuable experiences in this field.
Due to their origin and extraction from biological systems, biologically active substances and medicinal products are associated with a higher risk in terms of purity, identity and residues or impurities than many conventional medicinal products. Purity and identity testing, e.g. using methods such as HLPC, CE, IEF, MS etc., therefore plays an important role in the development,
authorisation and manufacture of biopharmaceutical products and therapeutics. This applies to protein-based products as well as to cell and gene therapies/ATMPs as well as modern vaccines. Their complexity often requires researchers to consider suitable analytical methods at an early stage of development that are also valid in later phases from a GMP perspective – which is also why this new conference track has been set up alongside the other more traditional analytical topics.
Another new area will be addressed with a pre-conference workshop: quality control of mRNA/LNP products. It will serve as an important platform for knowledge exchange and to promote discussions on the evolving landscape of mRNA therapeutics. PharmaLab is thus addressing another important topic that offers opportunities to redesign therapeutic approaches in various disciplines. Unlike viral vectors, mRNA/LNP technologies allow for precise and transient genetic modification without the risks associated with vi-
ral integration. Of course, PharmaLab 2025 will also cover the established topics. Overall, the congress includes the following conference tracks and pre-conference workshops:
on 25/26 November 2025
› GMP Compliance Trends in Analytical Laboratories
› Laboratory Optimisation, Automation and Digitalisation
› NEW! Artificial Intelligence in Laboratories
› Endotoxin and Pyrogen Testing
› Alternative and Rapid Microbiological Methods
› Cell and Gene Therapies/ ATMPsQuality and Safety
› Bioassays/Potency Assays – Regulatory Requirements, Development and Routine Use
› NEW! Bioanalytical Control of Biological Drug Substances and Products
on 24 November 2025
› 6th International Mycoplasma qPCR Testing User Day (Track QC Microbiology)
› NEW! Quality Control for mRNA/LNP Products ■
Early-bird rebate
Secure an early-bird rebate of €100 per conference day when booking before 31 August 2025!
Contact www.pharmalab-congress.com/trends
Programme Highlights
Eight parallel Conference Tracks
Two Pre-Conference Workshops
Exhibition
› More than 100 Presenters › Networking Event
› One-day tickets available
› Social event on 1st conference day
 NEW: Artificial Intelligence in Laboratories
 GMP Compliance Trends in Analytical Laboratories
 Laboratory Optimisation, Automation and Digitalisation
 Endotoxin and Pyrogen Testing
 Alternative and Rapid Microbiological Methods
 Cell and Gene Therapies/ATMPs - Quality and Safety
 Bioassays/Potency Assays
 NEW: Bioanalytical Control of Biological Drug Substances and Products
 Mycoplasma Detection
 NEW: Quality Control for mRNA/LNP Products
Premium Sponsor 2025

REAL ESTATE AND CAMPUS EXPANSION The EU Commission's call for evidence marks a positive step towards the proposed new European Biotech Act, signalling a significant prioritisation of the biotech sector in the EU. The direction of travel in the EU acknowledges the potential of biotechnology. In anticipation, the life sciences real estate sector grows – literally upwards.
Innovation patterns across key life sciences hubs can be seen from Boston's Center for Life Science to Europe. The US mentality to build up high in the sky pioneered also high-rise laboratory development at Kendall Square and in many other quarters in the life sciences hubs at the US east and west coast. The establishing principles now transform London's skyline through Euston Tower and One North Quay developments.
The European landscape reveals convergences with four innovation facilities across Limerick, Leeds, Leiden, and Harlow. The Biotech Campus Delft secures €500m for expansion. Meanwhile, Spain's emerging presence grows through Javier García Cogorro's SOKAI HUB España developments in San Sebastián (shown in photo), Madrid, and potentially Granada.
From Boston’s pioneering verticality to San Sebastián’s strategic positioning, these developments illustrate how global life sciences infrastructure responds to similar pressures whilst adapting to local contexts. Understanding these patterns provides essential intelligence for navigating an increasingly connected innovation landscape and makes the life sciences real estate an ever more attractive asset class for fund managers.
Two significant tower projects are highlighting London’s growing focus on highrise laboratory space to meet increasing demand. British Land is seeking investment partners for Euston Tower's com -
prehensive redevelopment into a life sciences hub. The redeveloped tower will provide approximately 570,000 sq ft of commercial space, including 215,000 sq ft of lab-enabled and flexible workspace, after securing planning consent in March.
Construction has commenced on One North Quay at Canary Wharf, which will become Europe’s tallest purpose-built commercial laboratory building. The 23-storey, 823,000 sq ft development is scheduled for completion in 2027 and will address London’s shortage of level 2 wet laboratory space.
Canary Wharf now houses over 30 specialist life sciences tenants, reflecting the area’s established cluster. Both developments demonstrate London's strategic approach to accommodating sector growth through vertical expansion rather

than traditional campus models. This approach maximises prime urban locations whilst delivering the specialist infrastructure that London's growing life sciences sector requires.
The life sciences real estate sector in Spain is gaining significant momentum, as evidenced by our latest newsletter featuring an above-average concentration of Spanish deals. SOKAI HUB España's launch stands out as a major development, with Quercus Investments and Columbus Venture Partners securing €100 million to develop life sciences facilities across Spain. Their flagship San Sebastian asset, requiring €80m investment, has already pre-leased 75% of its gross leasable area according to sources.
Other significant moves include i+Med’s 4,300 sqm research centre opening in Vitoria, Colonial’s €200m strategic investment in Deeplabs, and Euroespes’ sale-and-leaseback in Galicia. Notably, the Basque country features prominently in two of these four deals.
There is more on the way, also in Germany. In Mainz – the hometown of mRNA pioneer BioNTech – a new laboratory quarter is being developed in collaboration with Kadans and other technology partners. But also in Berlin, Cologne and Leipzig, huge areas are under construction or in planning, albeit rather broadly.
g.kaeaeb@biocom.eu
OPEN INNOVATION It all began with a view from the window of the 15th floor of Bayer’s site in Berlin, reminiscent of Boston before it became one of the most important biotech hotspots in the world. This inspired the vision of developing the Bayer Campus into “Boston an der Spree” – a vibrant healthcare campus that fosters collaboration and innovation.

Bayer is undergoing a major transformation of its research, production and administration site in Berlin. The goal: to create an internationally renowned life sciences campus that brings together diverse companies and ecosystem players. Through close partnerships with institutions such as Charité and the state of Berlin, the aim is to develop innovative therapies and digital healthcare solutions, making them available to patients more quickly.
A central component of this transformation is the Berlin Center for Gene and Cell Therapies (BC-GCT), which is being established in a public-private part-
nership with Charité - Universitätsmedizin Berlin. The centre will offer a unique combination of infrastructure in Europe, including an incubator and a GMP-certified manufacturing facility. The aim is to accelerate the development of innovative cell and gene therapies and represents a significant step forward in the field of regenerative medicine, addressing critical healthcare needs.
The centre will serve as a beacon for research and development, attracting top talent and fostering a culture of innovation. It will provide essential resources for startups and established companies alike, helping them navigate the complex regulatory landscape for cell and gene therapies. By facilitating collaboration between academia and industry, the
centre aims to accelerate the translation of scientific discoveries into clinical applications, ultimately benefiting patients who are in dire need of effective treatments.
Launched in November 2024, Bayer Co.Lab Berlin is a key element of Bayer’s external innovation strategy. As part of a pioneering global network of life science incubators – including Cambridge (US), Shanghai (China), Kobe (Japan) – Bayer Co.Lab connects entrepreneurs with world-class expertise, resources, and global networks. By identifying Europe’s top scientists and entrepreneurs, Bayer Co.Lab Berlin supports early-stage life science companies bringing their brilliant ideas to life. Startups remain independent through a no-strings-attached agreement while receiving invaluable mentoring from Bayer experts and industry leaders, along with access to state-of-the-art laboratory and office facilities. The community is designed to nurture entrepreneurial spirit and drive disruptive innovation.
Bayer Co.Lab focusses on breakthroughs in cutting-edge fields aligned to Bayer’s global research strategies. Two companies are already part of the Berlin community. MyoPax, a clinic-ready startup specialising in stem cell and gene editing technologies to develop muscle regeneration therapies, is the first resident company and was recently awarded a prestigious European Innovation Council Accelerator grant. Captain T Cell, developing potentially best-in-class autolo -
gous and first-in-class allogeneic T cell therapies to fight cancer, is the latest addition to the community.
In the future, Bayer Co.Lab Berlin will co-locate with the BC-GCT to empower more startups, expand the offering, and cultivate a strong entrepreneurial community all under one roof. By creating a vibrant ecosystem, Bayer supports the next generation of healthcare innovators, transforming groundbreaking ideas into viable products and services that can significantly impact patient care.
The Supply Center Berlin is Bayer’s excellence centre for manufacturing of parenteral dosage forms. The GMP manufacturing facility enables supply of high-quality medicinal products to patients around the globe, whether it is an aseptically manufactured biotechnological or terminally sterilised small molecule product. Being fully embedded in the Berlin Campus, the Supply Center offers integrated cutting-edge manufacturing solutions for the Bayer portfolio as well as contract and development manufacturing services to external partners. Bayer experts support all stages of the product life cycle from clinical to commercial, including complementary services such as supply chain management, procurement, quality and beyond. For further information please send an email to info@bayer.com.
The planned campus development is a clear commitment to Berlin as the global headquarters of Bayer’s Pharmaceuticals Division. This initiative aims to create new jobs and foster a robust network of employees from various sectors within the healthcare ecosystem. Additionally, the project may have a positive influence on the local labour market, potentially contributing to the economic vitality of the Berlin region.
The development will include stateof-the-art facilities that meet the highest standards of research and production. By investing in cutting-edge technology and

Myopax introduces themselves to Kai Wegner, the Governing Mayor of Berlin, and Stefan Oelrich, President of Bayer Pharmaceuticals, at the Bayer Co.Lab Berlin Grand Opening in November 2024
infrastructure, Bayer is positioning itself as a key player in the life sciences industry, ready to tackle the challenges of tomorrow's healthcare landscape. The site already offers rental options for chemical and biological laboratories of 300 square meters or more as well as office space of various sizes. For more information please contact info@bayer.com
The Life Science Campus will not only promote research and development but also contribute positively to urban development. The campus will be partially open to the surrounding urban space, support the development of neighboring districts, and align with Bayer's commitment to sustainable development. Extensive green spaces and meeting zones will be designed to ensure a high quality of stay and contribute to the overall well-being of the community. Additionally, sustainable energy supply and mobility concepts are being planned to promote environmentally friendly transportation.
Integrating green infrastructure will improve the campus's aesthetic, air quality, and biodiversity. Bayer's commitment to sustainability extends beyond the campus itself, as the company aims to set an example for responsible corporate prac-
tices in the life sciences sector. By prioritising eco-friendly practices, Bayer is working to minimise its environmental footprint while maximising its positive impact on society.
The Life Science Campus embodies a vision for the future of healthcare. By fostering collaboration among academic institutions, startups, established companies, policy makers and more, Bayer aims to create a dynamic environment for innovative ideas. This collaborative approach is vital to address the complex challenges faced by the healthcare industry today.
The campus will also serve as an educational hub, providing opportunities for professionals to enhance their skills and knowledge. By investing in human capital, Bayer is contributing to the growth of the entire healthcare ecosystem, ensuring a workforce ready to meet the demands of a rapidly evolving life sciences industry.
Bayer is committed to realising this vision, keeping Berlin at the forefront of the global life sciences landscape. By building a robust ecosystem that supports innovation and collaboration, Bayer is poised to make a lasting impact on the future of healthcare. ■
ENGINEERING BIOLOGY
Following the publication of recommendations for Engineering Biology in the UK from the House of Lords Science & Technology Committee, European Biotechnology Magazine spoke to Duncan Lugton, Head of Policy & Impact at the Institution of Chemical Engineers (IChemE), who submitted evidence to the committee, to find out more about the current landscape in the UK.
EuroBiotech_What were the primary motivations behind the £2bn investment in Engineering Biology announced in late 2023?
Lugton _This investment was primarily driven by the UK Government’s recognition of the field as a major opportunity for economic growth. This initiative aims to stimulate job creation, enhance industrial processes and support the transition to more sustainable practices. Additionally, the investment responds to recommendations from the House of Lords Science and Technology Committee, which included many of IChemE’s recommendations. The report highlighted the need to improve access to scaleup infrastructure and to develop the skills necessary to support innovation in this rapidly evolving sector – something
we believe is pivotal for the sector to thrive in the UK.
EuroBiotech _What progress has been made since the announcement, and how is the current Government enacting the strategy?
Lugton _Since the announcement, progress has been limited. Following its election in July last year, the Government has not specified its funding commitments in this area, with more detail only expected once the spending review process concludes in June 2025. While there has been positive rhetoric suggesting support for the strategy, detailed plans are still awaited. The UK Government has been working with the Engineering Biology Advisory Panel and others to understand how innovation infrastructure might be improved,

but this has not yet fed through to action. The Government’s Industrial Strategy is due to be published shortly, and we hope that some aspects of that can help support the sector, including funding but also the fundamentals including upskilling and reskilling our current workforce.
EuroBiotech _How would you assess the UK’s standing globally in engineering biology infrastructure?
Lugton _The UK has broad and deep strengths in engineering biology – including a world-class academic base, strong existing infrastructure and a skilled workforce – but it also faces several challenges that impact its global standing. One of the key issues is limited access to scale-up facilities, which significantly hinders the ability to translate innovation into com-
Advanced Pichia protein production strain & process development
Broad and versatile Pichia pastoris protein expression platform
Customized project setup suitable for every industry
































mercial success. This constraint contributes to a talent drain, as skilled professionals often move to countries like Belgium and the Netherlands, where better scaleup capacity exists – and they take their exciting new innovations with them. In comparison, economic heavyweights such as the United States and China offer attractive conditions for engineering biology, further highlighting the UK’s relative lag in infrastructure.





While the UK remains a strong player in foundational research and innovation, its position is undermined by infrastructural gaps that affect both talent retention and commercial competitiveness.
EuroBiotech_What are IChemE’s key recommendations to enhance the UK’s competitiveness?




















_IChemE feels that focus on strengthening innovation, skills and collaboration can help the UK become more competitive. This includes improved access to scale-up infrastructure through Government-subsidised innovation centres, brokering of access deals and the development of new facilities in underserved regions. Prioritising the training of chemical engineers is essential, alongside adjusting the academic syllabus for our scientists and engineers to include entrepreneurial skills. In addition, tax relief measures to support startups and a shift in the emphasis of Government research funding towards applied research could help.
DUNCAN LUGTON is Head of Policy & Impact at IChemE. He helps take the insights, skills and wisdom of chemical engineers and applies them to some of the biggest problems facing the world today. This includes producing materials and input to help policymakers make better decisions.
ports its members through networks, events, mentorship programs, and training resources that further contribute to its policy engagement and influence.
EuroBiotech _What steps are critical for commercialising sustainable tech (e.g., HutanBio’s algae biodiesel)?












Finally, we emphasise the importance of promoting stronger industry-academia collaboration through exchanges, mentoring initiatives and the creation of communities of practice - making sure that engineers have the business and communication acumen can help bring innovations to market.










EuroBiotech_How has IChemE communicated with the Government and influenced policy?
_We have a unique role as a Professional Engineering Institution, and we have served as a bridge between science, industry, and Government, for instance through submitting evidence to the House of Lords’ inquiry. In addition, IChemE sup-

Lugton_Several factors are crucial for this. First, access to scale-up facilities is essential to move innovations from lab to market. Collaboration with industry partners helps ensure that the technology meets real-world demands and has a clear path to adoption. Entrepreneurial training equips innovators with the skills needed to navigate commercial landscapes, while applied research ensures that practical challenges can be overcome and that technical development and refinement happens at pace. Timely funding from both Government and private sectors plays a key role in bridging the gap between innovation and implementation. Finally, mentorship and strong academic-industry connections provide invaluable
guidance and networks that can accelerate successful commercialisation.
EuroBiotech _Based on international models, what should the UK adopt, and what are its unique strengths?
Lugton_Fostering stronger integration between industry and academia (such as placement or exchange schemes), would be a good place to start. The UK should also prioritise developing a robust scale-up infrastructure that allows it to retain new innovations at risk of being lost to neighbouring European countries such as Belgium, the Netherlands, as well as the US. The Fraunhofer model is one that we think is particularly worth learning from in this area. At the same time, the UK benefits from unique strengths such as its world-class science base, well-established organisations, an emerging bioeconomy strategy and significant public policy focus. The previous Government committed to invest £2 billion in this area and it would be welcome if the current Government follows through on this in the forthcoming Industrial Strategy.
EuroBiotech_Which countries are serious competitors to the UK?
Lugton_Serious competitors to the UK include the United States, China, Belgium and the Netherlands. These countries are specifically mentioned because the UK has seen its talent and promising innovations move to these countries, given their attractive environments for scale-ups.
EuroBiotech_What are the next steps and principal challenges for UK scale-up efforts?
Lugton_The UK needs to make the most of the Industrial Strategy to put in place the infrastructure needed to support innovation, and to develop a robust plan to ensure that the country has the workforce needed to deliver it. More specifically, we look forward to the Government developing and implementing its plans in response to the House of Lords inquiry on engineering biology. The principal challenges include talent retention, bridging the gap between academic and commercial cultures, improving accessibility to infrastructure, increasing funding for applied research and addressing the limited commercial acumen among scientists.
■ t.gabrielczyk@biocom.eu
Engineering Biology or Synthetic Biology is the application of engineering principles to biology. It involves designing, building, and modifying biological systems – such as cells, enzymes, or genetic circuits – to perform specific tasks or produce useful products, like medicines, biofuels, or materials. Not all bioeconomy strategies place as much emphasis on engineering biology as the British approach. The EU’s current bio economy strategy has largely focused on enzymatic and microbial conversion of biomass from agricultural, forestry, and industrial waste streams, rather than on the photosynthetic fixation of excess atmospheric CO2eq. A strong example of engineering biology in ac-
tion comes from HutanBio Ltd, a spin-out from Cambridge University, UK. In May, the British/Malaysian start-up announced that the production process for its proprietary HBx microalgal bio fuel achieved net-negative carbon emissions. According to independent Life Cycle Analyses (LCAs), HutanBio’s salt-water microalgal closed bioreactor system removed up to 1.48 tonnes of CO2eq per tonne of biofuel produced across all three planned production sites: Morocco, the Middle East, and Western Australia. The LCA addressed all production stages – including raw material inputs, inbound transport, production and refinery processes, and waste handling.
■

DEEPTECH BIOECONOMY Biomanufacturing promises domestic, sustainable, sovereign and resilient production. Capacities are trapped in outdated scale-up processes. SPRIND urges investment and disruptive innovation to propel biomanufacturing. Demands are decentralised, non-sterile & continuous biomanufacturing capabilities.
› Dr Nicolas Krink, Senior Business & Research Analyst, German Federal Agency for Breakthrough Innovation SPRIND, Berlin, Germany
Biotechnology, empowered through advanced synthetic biology tools, has matured. And yet today’s capacity remains trapped in outdated scale-up processes. It still follows fine-chemical paradigms: sterile, low-volume, high-value production, locked into single feedstocks and products, with non-production chassis. This model fails to meet bulk industrial needs long promised but not yet delivered.
Fermentation processes require a rethink. Incremental innovations have sufficed for pharma but not for large-scale industrial biomanufacturing. The future lies in robust
non-sterile fermentation, Needs and Impacts of biotech scaling robust hosts, and continuous fermentation processes. Scaled biomanufacturing must be feedstock-flexible, resilient, and incorporate scale-down models to better predict large-scale outcomes. The only certainty ahead for markets and manufacturing is unpredictability. Bioprocesses must be designed with industrial end-use in mind to avoid the infamous ‘valley of death‘ so many are unable to bridge. Digitalisation and AI can dramatically accelerate optimisation. Most challenges are engineering, not scientific; they can be solved in years, not decades. Sustainability is just one benefit; sovereignty and resilience are now paramount. In a
shifting geopolitical landscape, domestic supply chains and production independence are critical for national security.
Decentralised, modular, rapidly adaptable production infrastructure will strengthen resilience against pandemics, trade conflicts, and geopolitical shocks. SPRIND’s deep-tech bioeconomy strategy (www. sprind.org/en/words/magazine/positionbioeconomy) calls for targeted scaling infrastructure. Biotech, green chemistry, and traditional chemical engineering must converge to create integrated, hybrid value chains built for resilience. Regulatory frameworks must evolve. Genetic engineering should be treated as a tool for chemical manufacturing, not subjected to pharma regulations. Industrial biotech represents the next phase of the chemical industry and needs streamlined approval, flexible hybrid processes, and differentiated risk assessments. Rapid, unbureaucratic public funding, venture capital, tax incentives are required. SPRIND uses its procurement driven tools to unlock economic potential, exemplified by the Curricular Biomanufacturing Challenge. One initiative is not sufficient to drive change. Many more such initiatives are urgently needed to tackle the real challenges in biomanufacturing. Investments in biotech scaling are investments in future security, economic independence, and technological sovereignty. ■
SPRIND
Pichia pastoris (syn. Komagataella phaffii), a methylotrophic yeast, is a proven platform for pharmaceutical protein production. However, its potential reaches far beyond. As a reliable, scalable, and cost-efficient platform for recombinant protein expression, Pichia is equally suited for industrial enzymes, food & feed, diagnostics, and biomaterials.
Key advantages include secretion of target proteins into the culture medium, and compatibility with chemically defined media. These streamline downstream processing and enable cost-effective scale-up. Pichia also supports efficient folding and disulfide bond formation, making it suitable for more complex proteins.
Tailored
While Pichia pastoris offers a strong foundation as an expression host, its true poten-

tial is realised through advanced engineering. VALIDOGEN’s proprietary UNLOCK PICHIA® system is one of the most flexible and powerful Pichia platforms available, designed to overcome expression bottlenecks across diverse applications. Importantly, its support for both methanol-based and methanol-free processes adds flexibility for varied safety and regulatory needs. Their molecular toolbox includes a novel library of strong promoters (methanol-induced and methanol-free), synthetic secre-
tion signals, native co-expression factors, and host strains optimised for robust performance. These modular components can be systematically combined and screened using high-throughput workflows to identify the best-performing expression strategies for each target protein.
By combining rational design with processlevel optimisation, VALIDOGEN transforms Pichia pastoris into a precision tool for recombinant protein production. Proven scalability to industrial volumes of up to 100,000 L underscores its readiness for commercial deployment. In an era demanding sustainability and efficiency Pichia offers a robust and scalable solution across industrial biotechnology.
■ Rosie Maddock, PhD, VALIDOGEN GmbH
MagStrep® Strep-Tactin®XT Beads close the gap between computational protein design and experimental proof. Optimized for automation and scalability, they transform protein purification from a bottleneck into a streamlined process.
A Munich-based team located at Helmholtz Munich and the Technical University of Munich recently introduced ENVLPE, a platform designed for efficient, safe, and versatile delivery of virtually all CRISPR-based therapeutics for somatic gene therapies.
Europ E an Biot Echnology spoke with Dong-Jiunn Jeffery Truong, lead inventor of ENVLPE, to discuss how this innovative approach is setting a new benchmark for gene editor delivery.

(3 rd from left) is currently a group leader at the Institute for Synthetic Biomedicine, headed by Prof. Gil Gregor Westmeyer (right), located in Helmholtz Munich. Together with Julian Geilenkeuser (left) and Niklas Armbrust (2 nd from left), they are actively pursuing opportunities to advance their technologies towards practical application and commercialisation.
EuroBiotech _What exactly is ENVLPE, and how does it compare to existing gene delivery platforms?
Dr Truong _ENVLPE is a novel, highly modular delivery platform based on virus-like particles (VLPs) derived and engineered from HIV-1. It inherits all the key strengths of lentiviral vectors, notably highly efficient delivery and proven low immunogenicity, without the drawbacks of integrating the genetic payload randomly into the genome, which poses a risk for insertional mutagenesis.
Lipid nanoparticles (LNPs), which excel in mRNA delivery, such as in vaccine applications, and liver targeting, remain limited in their ability to deliver ribonucleoproteins (RNPs) and suffer from poor programmability of tissue tropisms. Although we previously contributed to developing the first AAVbased split-Cas9 system, recognising that while adeno-associated viruses (AAVs) are excellent for long-term expression of intact gene copies, we realised that they are not optimal for gene
editing due to prolonged expression increasing immunogenicity risks and reducing editing precision with increased off-targets.
ENVLPE addresses these challenges effectively, offering a transient, precise delivery with a modular “hit-andrun” capability, i.e., the cell receives a load of pre-assembled gene editors (hit) and after having performed their job, they disappear quickly due to the cell’s natural protein turnover (run). By quickly changing ENVLPE’s surface proteins as easily as plug-and-play, we can reprogramme its tissue tropism without re-engineering the entire system or payload. For example, our recent study in Cell successfully demonstrated selective targeting and prime editing of the mutated Rpe65 gene in retinal pigment epithelium (RPE) cells in mouse models of retinitis pigmentosa.
EuroBiotech _CRISPR pioneers like David R. Liu, Jennifer Doudna, and Feng Zhang have also developed virus-like particles (eVLPs, EDVs, PNPs), with companies securing significant funding. What specific technological advantages does ENVLPE offer compared to these technologies?
Dr Truong _Virus-like particles themselves aren’t new, but previous implementations have struggled with efficiently delivering advanced, potent, yet unstable gene editors like prime editors. Any modification to the gene editor typically necessitates complete re-
optimisation of packaging components, creating significant development bottlenecks.
ENVLPE resolves these issues through its highly modular design by packaging gene editors via non-covalent interaction as opposed to protein-fusion design from other systems that strictly rely on release by viral proteases, encompassing cargo protection modules and logistical packaging improvements. Our approach delivers significantly higher editing efficiencies per particle in diverse in vitro and in vivo models, surpassing current leading platforms such as nChromaBio by more than 10-fold in in vivo settings. This means we achieve superior clinical outcomes with drastically reduced particle titers, minimising required dosages and optimising therapeutic efficacy.
EuroBiotech _What are your next steps to bring your technology closer to
translation? What are your next steps towards commercialisation?
Dr Truong _Having successfully established the core ENVLPE architecture, we will next expand our capabilities by building a comprehensive library of glycoproteins, including engineered variants, using traditional protein engineering and AI-assisted minibinder designs, enabling us to achieve selective targeting of various cell types relevant to therapeutic applications.
We are currently also integrating ENVLPE with our other complementary proprietary technologies to enhance efficient CAR expression cassette knockins. Leveraging our immunogene-free selection method that couples CAR integration with cell survival upon exposure to an otherwise lethal compound without using additional foreign selection markers, we can produce highly enriched and pure engineered CAR-T cell populations with uniform CAR ex-
pression. We actively seek partnerships with developers working on novel CARs or synthetic TCRs for high-value therapeutic indications who can benefit significantly from our delivery, knock-in, and selection technologies without relying on lentiviral random integrations.
Scaling is usually a major hurdle, but ENVLPE’s full compatibility with established industrial lentiviral vector manufacturing simplifies this challenge. We are currently exploring collaborations with partners with experience in lentiviral vector production to accelerate the scale-up and GMP-grade manufacturing processes.
Additionally, we are actively pursuing translational grants and industry partnerships to drive ENVLPE towards clinical application and commercialisation, ensuring that our gene editor delivery platform can rapidly benefit patients. ■ t.gabrielczyk@biocom.eu

Largest selection of enzymes for genomic research
Research-grade and GMP-grade production
Large to small scale lyophilization capabilities ISO 9001 and ISO 13485 certified
Formulations without components such as BSA, detergents or glycerol
Enzymes at user defined concentrations
Custom aliquoting, kitting, packaging & private label (OEM) Further information on Customized Products and Solutions from NEB
SUPPLY CHAIN In today’s rapidly evolving world, adapting to demand volatilities while ensuring uninterrupted supply of biological medicines is more critical than ever. At Boehringer Ingelheim, our global biopharmaceutical network delivers flexibility, resilience, and reliability. By leveraging harmonised procedures, standardised setups, and innovative logistics, we ensure the biologics we produce reach those in need – even during unexpected challenges.
› Dr. Rebekka Wuester, Communication Lead BioXcellence, Biopharmaceutical Contract Manufacturing Business, Boehringer Ingelheim Biopharmaceuticals GmbH
Boehringer Ingelheim BioXcellence™ operates large-scale cell culture facilities strategically located in the United States and Europe (Austria and Germany). This global footprint provides robust and reliable solutions to meet the evolving needs of the biopharmaceutical industry.
Our European facilities in Biberach, Germany and Vienna, Austria feature similar technical setups, ensuring positive technical comparability and thus consistent product quality. This harmo -
nisation is key to maintaining the highest standards across our network. Even slight differences between our U.S. facility in Fremont, California, and the European sites – such as stirrer or sparger configurations – are seamlessly managed thanks to our experience and operational excellence. This expertise in the network allows us to predict and address potential variations, ensuring smooth process transfers both for client-specific processes into our network and in case an additional supply node is requested by our customers also within our network.

One key idea is to develop robust and easily transferable production processes for large-scale manufacturing. This approach might offer the option to skip engineering runs, enabling us to immediately proceed with process performance qualification (PPQ) runs after completing small-scale runs, in vitro comparability studies, gap and risk assessments, as well as simulations and modeling.
By streamlining the manufacturing process, this method could save valuable time and resources. Not only does this approach enhance flexibility in supply for our customers, but it also accelerates the delivery of critical medicines to patients in need. This idea of ‘portable’ production processes exemplifies our commitment to innovation and efficiency.
Whether responding to demand fluctuations or timeline variability, our global manufacturing network setup enables interchangeability and a reliable supply without compromising quality. This consistency is achieved by a high level of process standardisation, thorough planning and execution, ensuring that every product meets the highest standards, regardless of the manufacturing site.
To further strengthen operational resilience for our customers, we offer access to our cryovessel pool and our newly
established Global Vessel Management Center – developed in collaboration with a trusted logistics partner to deliver seamless, reliable, and expert solutions. Designed for technical excellence, the Global Vessel Management Center offers validated cleaning processes, secure storage conditions, and comprehensive maintenance programs to support operational efficiency and regulatory adherence.
The COVID-19 pandemic and ongoing geopolitical uncertainties have underscored the importance of robust supply chains. That is why we have implemented a comprehensive Business Continuity Management (BCM) system designed to anticipate and mitigate risks, ensuring that our operations remain reliable under any circumstances.
Our business continuity management framework is built on three dimensions.
1. Enhancing production continuity for patient supplies: Our commitment to business continuity ensures uninterrupted operations, allowing us to reliably meet our customer’s needs and deliver on time, even in the face of unexpected challenges.
2. Managing contractual obligations: Customer requirements for BCM systems are integrated into our contracts, and regular audits ensure the effectiveness of our measures. This proactive approach builds trust and accountability with our partners.
3. Maintaining customer trust: Reliable delivery is essential to preserving our reputation and relationships with clients. Our BCM system mitigates the risk of disruptions that could impact trust, ensuring that our partners can rely on us.
Our global biopharmaceutical manufacturing network offers several key benefits that make Boehringer Ingelheim
Figure 2: The standardised setup of bioreactors and harmonised procedures across our large-scale cell culture facilities offer interchangeability, providing high flexibility and reliable supply security for our partners.
BioXcellence™ the partner of choice for large-scale cell culture contract manufacturing.
First, our network ensures resilient supply chains, even in times of geopolitical uncertainty. By leveraging our distributed facilities, we minimise risks and maintain a reliable delivery of critical medicines. This capability is particularly valuable for customers seeking stability and consistency in their supply strategies.
Second, we offer enhanced flexibility in responding to demand volatilities. With multiple sites registered for production, we can shift manufacturing runs between facilities to align with clients’ supply strategies. This adaptability allows us to meet changing market demands while maintaining high standards of quality and efficiency.
Third, our streamlined oversight processes reduce the complexity for clients. Customers with multiple sourcing supply security strategies can combine in-house production capabilities with various sites at one contract manufacturing organisation (CMO). This approach simplifies oversight efforts, allowing customers to focus on their core operations while trusting us to manage the complexities of manufacturing and logistics.
Finally, our harmonised setups and standardised processes ensure consistent product quality across all sites. Customers can trust that their products will meet the highest standards, regardless of where they are manufactured within our network.
Many of our customers have established their products at more than one site within our network, enabling us to discuss and adopt production schedules and jointly optimise supply strategies. This flexibility is a critical advantage in today’s dynamic market environment. By working closely with our partners, it is our ambition that their needs are met with precision and care, fostering longterm relationships built on trust and reliability.
At Boehringer Ingelheim BioXcellence™, we’re committed to leveraging our global biopharmaceutical manufacturing network to its fullest potential. By combining harmonised procedures, logistics systems, and business continuity management, we deliver for our partners and patients.
As the world continues to face challenges, our large-scale mammalian cell culture production network stands as a testament to the power of collaboration, innovation, and operational excellence.
EMBA In life sciences, breakthroughs are reshaping healthcare faster than ever. But true leadership means going beyond the lab – understanding the complex ecosystem where science, policy, and business collide. For those struggling to navigate this complexity, WU Vienna’s Executive MBA offers the insights and connections needed to stay ahead in an unpredictable industry.
› Paul Kospach and Jurgen Willems, WU Executive Academy
The biotech breakthroughs changing medicine are just the beginning. Real impact happens when you understand the bigger picture – healthcare economics, regulations, interdisciplinary teamwork, and the shifting political landscape. Life science leaders can no longer afford to operate in silos. Staying ahead requires a new kind of leadership toolkit.
That’s exactly what WU Vienna’s Executive MBA delivers: a deep dive into the complex healthcare ecosystem that brings together startups, pharma giants, IT innovators, regulators, and investors. Jurgen Willems, the program’s Academic Director, calls it a “playbook for navigating this dynamic and sometimes unpredictable industry.” For leaders eager to spot opportunities before anyone else, this programme is a game-changer.
This programme isn’t about theory alone. It’s designed for real professionals – scientists, clinicians, policymakers, and tech expert – who want to collaborate across disciplines and continents. The result? A vibrant network of forward-thinkers driving change at every level, from research labs to boardrooms.
With Austria emerging as a European life sciences hub, the programme offers unmatched access to one of the continent’s most exciting innovation ecosystems. Plus, the international curriculum combines high-level policy understand -

ing with practical leadership skills – arming you to lead teams, influence strategy, and implement solutions in a fast-moving environment.
What sets this executive MBA apart? It’s not just about managing change –it’s about shaping it. As Willems puts it, “People in life sciences are motivated by improving health and society. This programme gives you the tools to turn that motivation into real-world impact.” If you’re ready to lead healthcare innovation and steer your organisation through disruption, this is where you start.
The pandemic made one thing clear: life sciences are no longer a niche – they’re central to the health, resilience, and progress of our societies. But with rising rel-
evance come rising expectations. Scientific expertise alone isn’t enough. That’s why the Executive MBA Life Science & Health Care Management at WU Vienna was designed for professionals ready to lead across functions, sectors, and borders. Whether you're coming from biotech, medicine, IT, or policy, this program equips you with the strategic tools to lead confidently in a highly regulated, innovation-driven space.
Location: WU Executive Academy, Vienna
Start: October 13, 2025
Language: English
Format: Part-time, modular and hybrid
Audience: Professionals in life sciences, health, IT & policy
More info: executiveacademy.at/emba-lsh

CLEAN BIOLOGICS The French Clean Biologics Group, an international provider of services to the biopharmaceutical industry, has appointed Laurent Claisse as CEO. He succeeds Joseph Jammal, who held the position for five years. Claisse has extensive international management experience, particularly in North America, where the group intends to expand further. Recently they acquired Karyologic in North Carolina. The state is known for its strong presence in life science research and biotechnology. Before joining Clean Biologics, Claisse worked for Sartorius in the Washington DC - Baltimore region as Senior Vice-President, Operations. Prior to that, he held various leadership positions at Danaher companies, including Pall Corporation and Molecular Devices. ■
nesses, Jokela is well positioned to lead the foodtech company. Prior to joining Solar Foods, Jokela held senior positions at Körber Group and ABB Group. Since its launch in 2017, Solar Food has been led by co-founder Pasi Vainikka. He has joined the board and is handing over the baton to Jokela ■
TIRMED PHARMA Stockholm-based biotechnology company TIRmed Pharma has strengthened its regulatory expertise with the appointment of Elisabeth Augustsson, a leading regulatory affairs specialist. She will support the company as a consultant. Her appointment is an important step in the preparations for the clinical trials of TIR-C, the company’s novel immunomodulatory treatment for atopic dermatitis (eczema). Augustsson has extensive experience in working with the U.S. Food and Drug Administration (FDA) and European Medicinces Agency (EMA). ■

Elisabeth Augustsson

and automated sample storage, but also cryogenic capital equipment for the biopharmaceutical, biotechnology and academic research industries. Most recently, Smith led the global sales team at Azenta Life Sciences (formerly Brooks Life Sciences). Prior to that, his work focused on nanotechnology at Agilent/Keysight Technologies and RHK Technologies. Smith holds a B.S. in Human Biology with a minor in Business Administration from the University of Indianapolis ■ Ryan Smith


SOLAR FOODS The Finnish company Solar Foods OYJ is at a turning point on its journey from a technological innovator to a global commercial player. In April, Rami Jokela stepped in as the company’s new CEO to lead this next strategic phase. With his industrial expertise and experience in scaling and growing international busi -
Rami Jokela
ASTORIOM Ryan Smith has joined Astoriom in Rochdale (UK) as the new Global Head of Sales. He brings more than two decades of experience leading global life sciences sales teams to the global leader in R&D sample stability and biorepository storage. His experience as Senior Global Sales Leader includes not only outsourced biorepository services
NOVADIP Soumya Chandramouli, a seasoned executive with solid experience in senior international finance positions, is the new Chief Financial Officer of Belgiumbased Novadip. The late-stage clinical biotechnology company specialises in regenerative medicine. Chandramouli succeeds Virginie Cartage. She was instrumental in the development of Novadip during the company’s first ten years. She is now moving into the next phase of her career. Chandramouli has 25 years of experience in financial management, most recently as Chief Financial Officer of the IBA Group, a listed company in the medical technology and healthcare sectors. Novadip was founded in Belgium in 2013 and employs 45 people. Since its inception, the company has raised €88m in equity and nondilutive funds.
■ Soumya Chandramouli

In addition to implementing a 40% budget cut at the US Department of Health & Human Services in 2026, the Trump administration plans to eliminate all funding previously allocated by the Biden administration for industrial biotech expansion. However, in a move that has received little public attention, the administration is also quietly advancing sustainable industrial biotech production through the multibillion-dollar budget at the NSCEB (https://t1p.de/r4erg).
BIOTECH GEOPOLITICS The threat of high import tariffs and government-enforced price cuts in the world’s leading pharmaceutical market, the US, are beginning to have an impact on the global biopharmaceuticals sector. What exact geopolitical consequences the dismantling of US research funding and mass layoffs in regulatory, epidemic, and climate protection agencies will have remains to be seen. But the existing biotech world order is faltering and changing in unforeseen ways. Who stands to benefit?
Donald Trump entered his second term of office on January 20, but by mid-June most of the threats made by the US President still had yet to be fully implemented. Nor had his regime’s drastic cuts to the country’s research budget been upheld in court. But if announced plans for import tariffs, pharmaceutical price reductions, and cuts to research freedom really do go into full force, they could do serious damage to the fields of biomedicine and industrial biotechnology internationally.
The mere fact that 80% of planned NIH research funding has not been disbursed since February 2025 and 40% of the NIH budget plus 50% of the National Science Foundation (NSF) budget is to be cut in 2026 has been enough to incite panic in the biopharmaceutical ecosystem. NIH funding alone exceeds the R&D budget of the largest industry sponsors of clinical trials: 99% of the drugs approved by the FDA between 2010-2019 can be traced back to NIHfunded research (10.1001/jamahealthforum.2023.1921). Of the NIH’s annual US$47bn budget, US$18bn is used to fund international clinical trials in which US groups are involved.
Research and industry partners are suffering, of course, but also clinical service providers (CROs) and contract manufacturers (CDMOs). “Severe cuts to US re -
search funding, combined with deep uncertainty caused by unpredictable policies on tariffs and drug pricing, have led to significant investor hesitation in biotechnology and clinical research,” says Martin Krauss, President of the CRO association BVMA and CEO of FGK GmbH. “This is critically slowing the development of new therapies. Beyond the economic damage to innovation-driven industries, the longterm consequence will be fewer treatment options for patients,” he fears. “This troubling trend must be urgently addressed and reversed. At the same time, it presents a strategic opportunity for Europe to position biotechnology as a key sector and to strengthen the political and regulatory framework to attract increased investment and use its excellent basic research.“
There’s no question that the 157 executive orders issued during Trump’s first five months in office – just five fewer than Joe Biden signed in his entire term – are unsettling biopharmaceutical investors worldwide. Because they bear the hallmarks of protectionism and hostility to multilateralism, the biopharmaceutical financial engine that is fueled by international deals is sputtering. According to the EY Firepower Report, Big Pharma generates around 65% of its revenue from
products acquired through international M&A, agreements or joint ventures. Since 2023, cancer-related deals with Chinese biotech companies have taken centre stage. There and in other areas the impact of the new geopolitical approach that’s being nurtured by the White House – geared toward national interests – is already being felt. For May 2025, investment bank Jefferies reported a 57% decline in biotech financing (to US$2.7bn) compared to the same period last year. That followed an April that marked the worst figures since 2021. EY also reports record investment last year, along with its abrupt end in the first quarter of 2025. Jefferies analyst David Windley blames mass layoffs at the FDA, budget cuts at the NIH, and unclear statements from the White House about the structure of US drug prices. “Biotechs want clarity on FDA regulation, funding and drug pricing before committing to large investments,” Windley comments.
The US President is not taking a very systematic approach to his key objectives. To achieve his stated goal of bringing production back to the US, for example, he initially threatened to impose import tariffs on biopharmaceuticals, services and raw materials, that have now been suspended until the beginning of July due
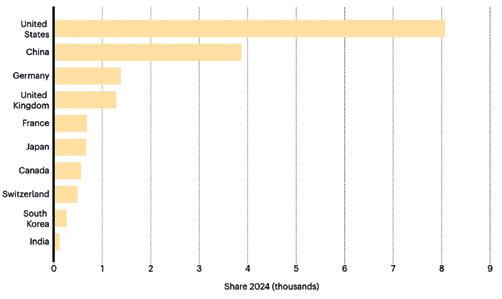
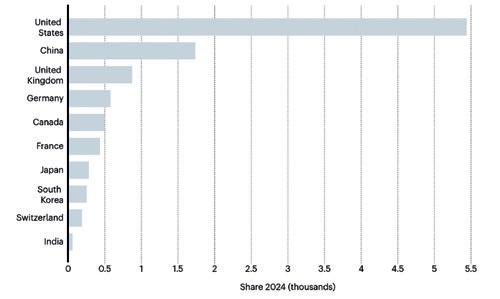
Fig. 1: In the Nature Index ranking of biological (left) and biomedical (right) publications, the US was the clear global leader ahead of China and the EU last year. However, the picture is different in general and in research areas such as cancer, where China overtook the US back in 2023.
to bilateral talks. At first the threat had the greatest impact on Big Pharma companies, which according to IQVIA data in 2023 generated around 53% of their sales in the US, 21% in the EU, and 7.5% in China. Novartis and Roche have since made commitments to invest US$50bn each in new US production centres and R&D. UCB and Sanofi followed suit with US$5bn and US$20bn respectively.
“I just don’t understand why the EU should give more money to innovative pharma companies, or monopoly extensions, when they’re basically transferring their production to the US.”
Medicines for Europe
But when Trump announced in early June via executive order that he wanted to reduce pharmaceutical prices by up to 90% if necessary in the largely unregulated US pharmaceutical market, the picture changed again. (According to the US Department of Health & Human Services, prices there are around 3.3 times higher than the global average). While Pfizer CEO Albert Bourla apparently calculated that Trump’s proposed increase in pharmaceutical prices in other markets could more than offset the planned price cuts in the US and publicly supported Trump’s de-
mand, a Roche spokesman expressed disenchantment with the plan: “Should the proposed EO go into effect, Roche’s ability to fund the significant investments previously announced in the US will be in question.” Dorothee Brakmann, CEO of Pharma Deutschland, clarified the basis for pharmaceutical investments: “A fair, sustainable price is a prerequisite for innovation, investment, and production sites in our own country.”
Dr. Jeremy Levin, CEO and Chairman of Ovid Therapeutics Inc and one of the most influential figures in the US biopharmaceutical scene, emphasised that “the US pharmaceutical market has long been a cornerstone of global drug revenues, often accounting for a significant share of profits that fuel research and development worldwide. These revenues are not abstract – they directly fund the discovery of therapies that extend and improve the lives of patients across the globe. When those funds shrink, so do the prospects for new treatments that offer hope to families facing serious and often incurable illnesses. Implementing MFN pricing could reduce US drug prices by up to 90%, potentially diminishing the revenue streams that support this innovation. This shift may prompt pharmaceutical companies to reevaluate their investment strategies, possibly diverting funds to markets with more favourable pricing structures.” Another executive order instructs the FDA to increase fees and inspections for foreign manufactur-
ing facilities, but critics complain that it is also unclear how that law is to be implemented.
The next top dog in biotech
Trump’s zigzag course to an unclear destination not only unsettles investors, but also fails to recognise long-standing import dependencies. For instance, in its current five-year plan, China is promoting four key technologies, – including bio technology – with US$1.25tn. Successful tariff negotiations with the Asian super power are therefore crucial for US bio pharmaceutical company development. A US survey of 124 biopharmaceutical firms found that 79% have at least one contract or product with a China-based or Chinese-owned CDMO. It’s estimated that 13% of the world’s manufacturing facilities that supply APIs to the US are in China, and it is a major global supplier of APIs for generic drugs. Additionally, China is behind approximately 42% of the global export value of antibiotics, and through strategic investments it overtook the US in the development of cancer drugs such as ADCs and bispecific antibodies in 2023.
And that’s not all. The number of clinical trials in the US has been in decline since 2015, but is rising dramatically in China (see Fig. 2). Xi Jinping, the country’s 61-year-old head of state, has set the strategic goal of making China biotech’s market leader by 2035. Based on two years of
research and consultation with private and public experts, a brand-new report from the US National Security Commission on Emerging Biotechnology (NSCEB) comes to a sobering conclusion: China is quickly ascending to biotech dominance after making the sector a strategic priority 20 years ago. To remain competitive, the US would have to take swift action in the next three years and invest US$15bn by 2031. “Otherwise, we risk falling behind, a setback from which we may never recover,” says the report. China is seizing the lead not only in pharmaceutical biotechnology, but also in industrial biotechnology, which is currently being strategically ignored by all but a few in the EU. But is that really the best course for a democratic Europe?
EU asks: ‘Where’s your money?’
Top EU politicians such as Teresa Ribera, Vice Executive President of the European Commission (EC) for Clean, Just and Competitive Transition, are clearly comfortable in the EU’s new role as the ‘guardian’ of multilateralism, the rule of law, and human rights. However, the energetic Spaniard faces fierce resistance from those in Brussels who, at meetings on the development of life sciences, scale-up, and bioeconomy strategies, pretend it’s not such a big deal strategically to drop the term ‘sustainability’ and replace it with ‘competitiveness’. “We must start acting now,” says Ribera, “even if it is difficult and expensive.”
Observers expect far-reaching consequences from the withdrawal of Joe Biden’s Executive Order (EO 14081) “Advancing Biotechnology and Biomanufacturing Innovation for a Sustainable, Safe, and Secure American Bioeconomy” from September 2022. The EO was linked to the prioritisation and extensive financing of climate-friendly biotech production processes – a red flag to Donald Trump, a self-confessed climate crisis denier.
However, though eliminating roughly a third of Biden’s EOs by mid-June, the new US government seems to have retained a committment to engineering biology. German innovation agency SPRIND (see p. 46) says pole position in the field remains open. “The consensus is clear:
the bio economy remains in transition, with no clear leaders paving the way,” SPRIND experts told EuropEan BiotEchnology on request, adding: “The ecosystem overpromises and underdelivers. Remember, BioMADE started during Trump’s first term; now the NSCEB report offers a clear roadmap – biotechnology as a disruptive force in the race against China and for national security. Europe must act fast with bold funding for the deep tech bioeconomy that embraces technology to stay in the
race. Waiting means losing.” Plant technology manufacturers such as Germany’s GEA, which will open its second Food Application and Technology Center (ATC) in the US in mid-July, remain unimpressed. “It is important for us to be close to the customer during the scaling step,” says Frederieke Reiners, Vice President of New Food at GEA. “Where the customer’s production plant will be built is a completely different question – since we have a global presence, the location that the custom-
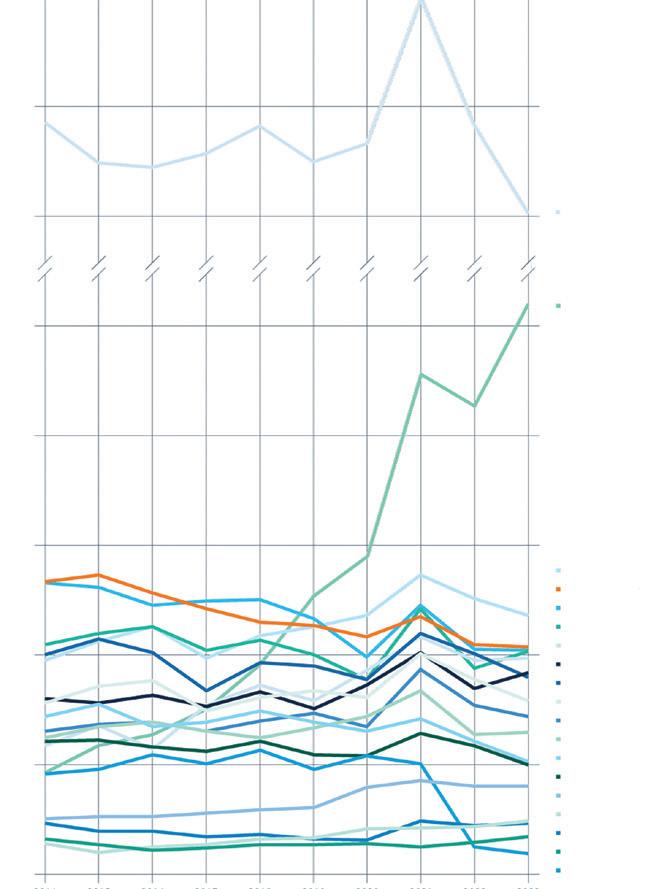
Fig. 2: When it comes to numbers of clinical trials, the US has been losing ground since 2015, while China is catching up thanks to massive government investment in biotech. As there are always a certain number of late registrations, reliable figures for 2024 and 2025 are not yet available, according to the VFA.
er chooses for their production plant is irrelevant to us, i.e. wherever the conditions are best for them.” With a market share of 40% and a thriving start-up scene, the US is ahead of both Europe (30%) and Asia (20%). “Scaling up to pilot scale is sufficient to calculate parameter-based upscaling to industrial-scale plants,” said Tatjana Krampitz, Head of Technology Management at GEA New Food. The German giant occupies a unique position in the field worldwide, as the company manufactures around 90% of the equipment required for scaling within the process chain itself. This expertise enables reliable calculations of reactor geometry on an industrial scale. The ATC supports a wide range of applications, including cultured meat, precision
fermentation, and alternative milk and egg products. Unlike in Europe, the approval process for novel foods in the US is fast and unbureaucratic.
Although EU members like the Netherlands, Denmark, and Italy have recently announced plans to invest large sums in the future markets of cellular agriculture, chemistry, and new materials, too little public money is still being spent in the EU on the expensive development of adequate infrastructure for scaling industrial biotech processes (see p. 3, 14, 43, 45). This is because the €200bn competitiveness superfund planned by EC President Ursula von der Leyen to finance the Clean Industrial Deal will not be available until 2028 at the earliest. And that’s provided it is included
as an additional pillar in the EU’s mediumterm financial plan (MFF). The first funds from a billion-euro IPCEI Biotechnology (Important Projects of Common European Interest) programme currently being prepared by Germany, Estonia and Finland and to be financed by EU countries, slated to invest exclusively in growth technologies, will not be available until 2028 at the earliest. The IPCEI Biotechnology also contains new initiatives, such as the promotion of biotech projects that use CO2 as a raw material. In the EU in general, the growth markets of pharmaceutical and industrial biotechnology are still not really a political priority. But developers want more, and the EU can’t afford to ignore them. ■
t.gabrielczyk@biocom.eu
Kristin Schreiber Director European Commission, DG for Internal Market, Industry, Entrepreneurship and SMEs
The evolving landscape of global bioeconomy policy marks a critical point in history. The European Union now faces a choice: to follow or to lead. I believe Europe must lead.
This is not about choosing between low-tech and high-tech. It is about recognising that Europe’s strength lies in its ability to combine both. The traditional bioeconomy – rooted in sustainable agriculture, forestry, and rural communities – has provided a foundation of a strong and resilient economy. But to remain competitive and to modernise our economy, we must now embrace the full potential of biotech and deep tech applied to life sciences to supplement conventional methods of manufacturing, agri-food processes, and more. This means increasingly developing and using AI and generative AI – for R&D, for optimising production processes or for discovering new value chains. It also means, facilitating market access of products produced with
the help of genetically modified microorganisms (GMMs), unlocking the potential of certain new genomic techniques (NGTs), and supporting biomanufacturing processes that can utilise CO2 as a feedstock. Just to name a few.

At a time when Europe is striving to strengthen competitiveness, reduce emissions, defossilise industry, reduce dependencies, and boost digital tech diffusion, we cannot afford to leave such strategic opportunities untapped. The metamorphosis toward a truly competitive, scaled, and resilient bioeconomy will only be possible through the synergistic integration of emerging technologies – from AI to synthetic biology – and by fostering partnerships that enable data sharing, industrial symbiosis, and effective teamwork.
Moreover, harnessing the full potential of biotech and deep tech can create new markets for biomass – and new opportunities for farmers and forest managers. The goal is a cohesive European bioeconomy – one that supports both business and primary producers.
The EU is actively supporting biotech through initiatives like Horizon Europe, the Strategic Technologies for Europe Platform (STEP), and the Circular Biobased Europe Joint Undertaking (CBE JU), de-risking investments and helping innovative biotech solutions to scale up. Over the past year, STEP has mobilised over €15bn to support Europe’s competitiveness, but we must do more – from skills to funding.
The European Commission is now working on the new EU Bioeconomy Strategy and the European Biotech Act, which will make it easier to bring biotech from the laboratory to factory and then onto the market, improve resource efficiency, and tap the significant growth potential of bio-based materials substituting fossil-based materials, and related industries. The Strategy for European Life Sciences will further drive innovation in biotech, tap into the full potential of data and AI, and boost deployment. This moment calls for action. For everyone. For Europe. ■
AVIATION Neste Oyj is scaling up its role in sustainable aviation with its largest deal yet at LAX: The Helsinki-based company will supply 8,800 metric tonnes (more than 11 million litres) of its blended Neste MY Sustainable Aviation Fuel (SAF) to FedEx. The renewable jet fuel will cover roughly 20% of FedEx’s annual jet fuel demand at the LA airport. Produced from used cooking oil, animal fat waste and other residues, Neste’s SAF is certified for commercial use and compatible with existing aircraft and fuelling systems when blended up to 50% with fossil jet fuel. For Neste, it’s another milestone in a global ramp-up that now sees 1.5 million tonnes of SAF production annually. ■
ONCOLOGY TILT Biotherapeutics
Ltd has secured US$25.6m in Series B financing, with the European Innovation Council Fund, Lifeline Ventures, Finnish Industry Investment (TESI), and Stephen Industries Inc Oy all returning to the table. The funding is earmarked to propel the company’s lead asset, TILT-123 – an oncolytic adenovirus armed with TNF and IL-2 – into a Phase II trial for platinum-resistant epithelial ovarian cancer. It will also support a Phase Ib melanoma combination study. Recent Phase Ia data, published in naturE communications (10.1038/s 41467-025-56482-w), painted a cautiously optimistic picture. ■
FINANCING Alvotech SA, a biosimilar specialist headquartered in Reykjavík, Iceland, has completed a private placement of 7.5 million Swedish Depositary Receipts (SDRs) and ordinary shares, raising gross proceeds of SEK750m (€66m). The offering, which drew interest from around 40 institutional investors, aims to diversify the shareholder base, increase free float on Nasdaq Stockholm, and bolster Alvotech’s standing in the biosimilars space. Alvotech has also announced a collaboration and licence agreement with Dr. Reddy’s to codevelop, manufacture and commercialise a biosimilar to the leading cancer immunotherapy Keytruda® (pembrolizumab). The partnership aims to accelerate development and expand global access to this high-value oncology market. ■
PARTNERSHIP H. Lundbeck A/S has teamed up with the Danish Centre for AI Innovation (DCAI) to tap into the power of Denmark’s national AI supercomputer Gefion. Lundbeck aims to speed up the discovery and development of new treatments for neurological and psychiatric disorders. The biopharmaceutical company focusing exclusively on brain health is among the first pharma companies to use Gefion, applying its advanced computing to explore molecules faster and fine-tune drug candidates more precisely. By combining science with smart technology, the company hopes to bring better treatments to people living with brain disorders, and to speed up the process at the same time. For DCAI, the partnership is a big step in showing how AI can help drive meaningful progress in healthcare. ■
CELL BIOLOGY Cancer cell movement during metastasis is a dynamic process regulated by multiple signals. However, how cells receive, process, and respond to these signals has been extremely difficult to detect. Finnish researchers at the University of Turku have developed a fluorescent probe that visualises signalling dynamics in moving cancer cells. In the microscopy image above, invasive breast cancer cells (labelled in yellow) can be seen degrading their underlying extracellular matrix (labelled gelatin in red; degraded areas appear as black holes). The scientists published their findings in n atur E c E ll B iology (10.1038/s 41556-025-01663-4). They are now exploring the therapeutic potential of these findings. ■

ALTERNATIVE PROTEIN Millow AB, a Gothenburg-based food-tech company, has flipped the switch on its first industrial-scale production facility – a repurposed 2,500m² former LEGO hall. The commissioning was supported by a €2.5m grant from the European Innovation Council, part of a €17.5m blendedfinance package. Later this year, the site is set to produce up to 500 kg of oatand mycelium-based protein per day. As interest in highly processed meat analogues cools, Millow’s dry-state fermentation process offers a new approach:
a clean label, binder free protein made from just two core ingredients, using a patented dry state fermentation process that allows for efficient scaling. “Our platform can swap grain substrates overnight, allowing any region to grow its own advanced protein with minimal resources,” points out co-founder Mohammad Taherzadeh. According to Millow, the product leaves no aftertaste like soyor pea-based competitors and the process uses significantly less energy. Product launches are expected by the end of 2025. ■
VC Novo Holdings A/S and the Export and Investment Fund of Denmark (EIFO) have jointly invested €48m in HealthCap IX, a Swedish life sciences venture fund focused on building innovative companies in areas of high unmet medical need. Following this investment, Nordic venture capital firm HealthCap will establish a local presence at Copenhagen’s BioInnovation Institute to boost its focus on Nordic opportunities, particularly supporting early-stage companies. This latest investment marks EIFO’s first collaboration with HealthCap, while
Novo Holdings is a returning supporter. Both investors aim to strengthen the region’s reputation as a global leader in life sciences and ensure that promising scientific projects have the support needed to grow into successful companies.
“The investments from Novo Holdings and EIFO not only enhance HealthCap’s ability to support transformative healthcare companies but also reinforce the Nordic region’s position as a global leader in life science innovation and entrepreneurship.” commented Mårten Steen, Managing Partner, HealthCap. ■
Cytovation ASA has raised NOK62m (around €5.4m), to advance its bifunctional peptide CY-101 into a Phase II trial in adrenocortical carcinoma, set to begin in late 2025 (see p. 24). CY-101, a membranolytic Wnt/β-catenin pathway inhibitor, has shown early antitumour activity in a Phase I trial and preclinical data demonstrated tumour elimination and immune activation in resistant models of ACC, CRC, and melanoma. The study is being conducted in partnership with Cancer Research UK and the Norwegian Cancer Society.
Avecom NV and Green Alchemy ApS have formed a strategic partnership to offer an integrated service for industrial waste-stream valorisation, combining Avecom’s expertise in biomass fermentation and microbial process development with Green Alchemy’s regulatory and policy advisory. The collaboration aims to accelerate adoption of bio-based solutions within the circular economy.
TIRmed Pharma AB, a Stockholm-based biotech, has secured €4.3m in funding to advance its topical oligonucleotide-based therapy for atopic dermatitis into clinical development. The capital will support completion of toxicology studies, GMP manufacturing, and initiation of a Phase Ib trial. The investment round was led by Sciety and its investment network, Sciety Venture Partners.
FINANCING French Massalia Therapeutics SA has been launched, backed by seed funding from Landmark Bioventures, the company founders and their network. Though the company did not disclose financial details of its seed round, the average cost for study registration is around €0.5m, and Phase I testing is estimated to cost between €1m and €5m. The company claimed to have an IND-ready, first-in-class drug candidate poised to target both cancer and fibrosis. Co-founded by Dr Marcel Blot-Chabaud and Professor Nathalie Bardin from the Cardiovascular and Nutrition Research Center at Aix-Marseille University, Massalia Therapeutics is set to develop SANF (Soluble Angiogenic Neoplastic and Fibrotic) factor. The goal is to address conditions that involve the interplay between fibrosis, neoplasm, and neovascularisation. Massalia expects to begin a first-inman dose escalation trial by November.■
DEAL Building on a US$415m ALS drug discovery collaboration with Alchemab Therapeutics Ltd inked in January, Eli Lilly & Company has secured rights to the first IND-ready programme. ATLX1282 is the first candidate from Alchemab’s AI-driven platform, which identifies antibodies linked to resilience in untreatable diseases. Alchemab will run the Phase I trial, after which Lilly will take over. ■
DIAGNOSTICS Leuven-based nanoelectronics hub Interuniversity Microelectronics Centre (Imec) has presented a highly miniaturised ingestible sensor at ITF World 2025 in Antwerp (May 20-21, 2025). The sensor prototype, developed at OnePlanet Research Center, is three times smaller than current capsule endoscopies and the first to provide redox balance measurements. This can provide insights into intestinal inflammation, the gut microbiome, and overall health, representing a significant leap forward in the non-invasive monitoring of gut health and management of gastrointestinal diseases.
Monitoring gut health has been challenging due to the complexity and inaccessibility of the GI tract. Traditional methods, such as endoscopy and colonoscopy, cannot provide a comprehensive view of the entire GI tract and are inherently unpleasant procedures. Capsule endoscopies, on the other hand, while currently used for diagnosing gastrointestinal disorders such as Crohn’s disease through visual inspection, also require unpleasant bowel preparation. Moreover, current solutions lack sensors to measure the chemical environment.
In contrast, Imec‘s miniaturised ingestible sensor offers a non-invasive solution for in vivo gut health monitoring. The sensor, 2.1 cm in length and 0.75cm in diameter, and thus three times smaller than existing capsule endoscopies, is designed to measure redox balance, pH, and temperature along the entire gastrointestinal tract. During a demonstration, Aniek Even swallowed the sensor, and showed real-time measurements on a screen saying “our small and easy-to-use device could revolutionise the way we diagnose and manage gastrointestinal diseases, including inflammatory bowel disease and gastrointestinal cancers”. The sensor can take measurements every 20 seconds over a period ranging from 24 hours to a week, depending on the individual’s gut motility.
The research team aims to evaluate the sensor in patient populations, with follow-up trials in collaboration with Radboud University Medical Center focusing on patients with cancer or chronic inflammation, as well as trials with other universities and clinical centers. The team welcomes opportunities from other institutions to incorporate the ingestible into diverse clinical trials, further advancing innovation in healthcare. ■

Imec’s miniaturised gut biosensor
Belgian lysosomal storage disease specialist Azafaros BV has added a new lead investor to its portfolio. In addition to €25m Series A leader Forbion, Jeito Capital co-led a €132m Series B round aimed at advancing two pivotal trials of Azafaros‘ lead compound nizabaglustat (AZ-3102). Azafaros BV’s oversubscribed Series B financing was led by Jeito Capital and Forbion Growth, with participation from Seroba, Pictet Group,
and existing investors Forbion Ventures, Schroders Capital, and BioGeneration Ventures. Proceeds will support two Phase III pivotal programmes involving Azafaros’s oral biphenyl-substituted Lido configured deoxynojirimycin derivative, nizubaglustat, in Niemann-Pick disease (Acid Sphingomyelinase Deficiency, ASMD) Type C and GM1/GM2 gangliosidoses. The trials are expected to commence at the end of this year. ■
Cancer Meds German BioNTech
SE has secured the largest funding awarded ever to a pharmaceutical company (£129m/ €153m) to open a research centre for cancer therapy in the United Kingdom. The funding is a payback of the British government to the reknowned mRNA corona vaccine specialist that announced in 2023 to invest up to £1bn over the next decade to broaden its R&D activities in the UK. BioNTech will particularly expand clinical testing of licenced cancer modalities such as bispecific cell engagers, ADCs and other promising drug formats. The agreement builds on an existing multi-year partnership between BioNTech and the UK government, aimed at accelerating clinical trials for personalised mRNA immunotherapies. It also fo -
cuses on establishing two new research and development (R&D) centres and a UK headquarters in London, which will house BioNTech’s artificial intelligence (AI) hub.
BioNTech, which acquired CureVac in June, plans to use the funding to support both R&D centres. The first will be located in Cambridge, with the second still in planning. The Cambridge facility will focus on genomics, oncology, structural biology, and regenerative medicine, and will accommodate more than 90 staff.
In addition, the company will merge its current London offices with its British AI subsidiary, InstaDeep, to form a new regional headquarters. The AI hub will continue to play a vital role in advancing BioNTech’s research capabilities through AI-powered medical innovation. ■
British investment firm SV Health Investors has secured US$269m for its second fund, Dementia Discovery Fund, or DDF-2. The capital will be used to invest in 10 to 15 companies developing or enabling novel therapeutics for dementia. It received backing from investors, including AARP, the British Business Bank, and Gates Frontier, as well as pharmaceutical companies Bristol Myers Squibb, Eli Lilly, and Pfizer, and the Alzheimer’s Association.
Paris-based Astraveus SAS has partnered with The Netherlands Center for the Clinical Advancement of Stem Cell and Gene Therapies (NecstGen) to evaluate a novel approach dubbed Lakhesys Benchtop Cell FactoryTM for cost-effective manufacturing of CAR-T cell therapies at NecstGen’s facility in Leiden. The partners want to demonstrate that the new system can significantly reduce costs and increase efficiency in CAR-T manufacturing in comparison to incumbent industry benchmarks. Additionally, NecstGen will produce lentiviral vectors for Astraveus’ internal research and development needs.
The Netherlands aims to take a leading role in pharmaceutical and industrial biotechnology by 2040, investing around €1.9 billion by 2029 under its ‘Cabinet Vision on Biotechnology’. The initiative has been welcomed by HollandBIO.
M&A Moonlake Therapeutics Inc., a Nasdaq-listed biotech firm based in the Swiss tax haven of Zug, is reportedly in advanced takeover discussions with US pharmaceutical giant Merck & Co (Merck, Sharp & Dohme, MSD). The offer is said to be around US$3bn, a premium to former market valuation of approximately US$ 2.6bn. ■
COST REDUCTION As research and development efforts and related programs are closed out, the Swiss company ADC Therapeutics SA (Lausanne; NYSE: ADCT) plans to shut down its UK facility and reduce the global workforce across functions by approximately 30%. Although gross proceeds from an actual PIPE financing are anticipated to be approximately US$100m, ADC Therapeutics intends to use the net proceeds from the PIPE to fund multiple catalysts primarily in support of ZYNLONTA® clinical development as well as ZYNLONTA commercialisation activities.
Also Zurich-based Molecular Partners AG (NASDAQ: MOLN), a company developing custom-built protein drugs known as DARPin therapeutics, has announced that it will reduce headcount by up to 40 positions, representing approximately 24% of its total workforce. ■

CANCER/MRNA First news first: BioNTech’s Biotheus acquisition paid off with a billion dollar BMS deal. German BioNTech SE from Mainz has struck a major global partnership with Bristol Myers Squibb (BMS) centred on BNT327, a bispecific PD-L1xVEGF-A antibody acquired through its 2023 takeover of Chinese biotech firm Biotheus. The deal includes a US$1.5bn upfront payment, up to US$2bn in further payments tied to ‘anniversaries of the agreement’ (original wording, still no clue why an annual celebration is a new kind of milestone), and potential milestone payments of up to US$7.6bn – making the original US$1bn Biotheus purchase a highly lucrative move. The whole deal around just one antibody comes with the enormous volume of roughly US$11bn!
BNT327, which combines immune checkpoint inhibition with anti-angiogenesis mechanisms, is currently in Phase 3 trials involving over 1,000 patients across several cancer types, including small-cell lung cancer and triple-negative breast cancer. Early results appear promising, although the class carries risk – as shown by a recent Phase III failure of a similar drug from Summit Therapeutics.
The BMS partnership is structured as a 50:50 cost and profit share, signalling
strong confidence in the antibody’s potential. BNT327 is now one of three key pillars in the company’s future oncology strategy, alongside mRNA-based cancer vaccines. With BMS onboard and further clinical data expected this year, BNT327 could become a new standard in immuno-oncology.
Additionally, BioNTech announced plans to acquire former mRNA-rival in pandemic times CureVac NV (Tübingen, Germany) via a public exchange offer to expand research, development, manufacturing, and commercialisation of mRNA-based cancer immunotherapies. CureVac’s mRNA design and formulation technologies are described as “complementary” to BioNTech’s existing expertise. In this deal, CureVac is valued at around US$1.25bn, after which it will become a wholly owned subsidiary. Its Tübingen site will be integrated into BioNTech’s global network.
Legal proceedings over mRNA patents and a share of the multibillions, which BioNTech bunkered together with Pfizer — initiated after CureVac waived protections during the COVID-19 pandemic — remain unresolved for the time being. But these newlyweds will find a solution. ■
BIOECONOMY AMSilk GmbH, Neuried (Germany) announced its expansion into the consumer care market with the launch of AMSilk4Shine and AMSilk4Care. AMSilk4Shine is an innovative ingredient made from silk proteins. It is designed to provide a more sustainable, high-performance alternative to traditional petroleum-based polymers, which are present in current rinse-aid formulations for automatic dishwashing and some screen and window cleaning products.
Their unique amino acid composition enables them to form ultra-thin, surfaceadhering coatings that deliver high-performance functionality while protecting the underlying surfaces. This makes them highly versatile for use in a range of cleaning and personal care applications. AMSilk proteins are biodegradable and reduce the carbon footprint of products by 53% and the chemical footprint by 57%. The cost competitive production was developed together with 21st. BIO from Copenhagen, Denmark. ■
INCUBATOR Vienna’s startup ecosystem is set to expand with the launch of Abundance Gate, a new private biotech and deep-tech incubator in the city’s southwest. Alongside it, a €50m VC fund is planned to support early-stage startups.
As reported by media, the incubator aims to bridge the gap between academic research and commercial success. Founded by Thomas Holzhuber and Timur Topalgökceli, the project addresses Vienna’s lack of lab infrastructure and strategic support for scaling biotech ventures.
The new Abundance Gate will offer up to 18,000 sqm of lab space and tailored “lab to market” support, backed by a network of 2,000 industry experts. The associated VC fund will invest €1–10 million in startups focused on biotech, life sciences, AI and interdisciplinary tech, with an emphasis on smart capital from technically knowledgeable investors.
Startups like Curie.bio and Resonate Bio are already active at the site. The model takes inspiration from Boston’s LabCentral, aiming to build a strong biotech cluster in Vienna, similar to those in Basel, Zurich or London. ■

Austrian Marinomed Biotech AG secures Swiss partner. Marinomed continues to navigate its way out of a challenging situation of self-managed insolvency proceedings. It has now signed an exclusive agreement with an undisclosed Swiss pharmaceutical partner for the development and commercialisation of Budesolv in Switzerland. Marinomed will receive milestone payments and royalties. Switzerland is intended to serve as a reference market for future international expansion of the formulation that supports hard-to-deliver drug-intake.
Molecular Health and Milan-based Axxam launch preclinical collaboration. The Heidelbergbased Molecular Health GmbH, founded by Friedrich von Bohlen – who has vast experience in consulting billionaire Dietmar Hopp’s biotech investments over two decades –, has entered into a strategic partnership with Italian company Axxam SpA. The aim is to accelerate the identification and validation of novel drug targets by combining Molecular Health’s AI-driven data analysis capabilities with Axxam’s biological and laboratory expertise.
“Understanding molecular causalities and variants in phenotypes, diseases and endotypes is essential for pharma and biotech companies to improve efficiency and success rates in drug development”, said Friedrich von Bohlen. “Axxam’s preclinical biology and chemistry assays and expertise will offer improved and accelerated outcomes in early drug discovery.”
FINANCING Barcelona-based epigenetics specialist Oryzon Genomics SA has cashed in €30m through an oversubscribed capital increase. At the end of April, the company issued 12,765,958 new shares, priced at €2.35 per share. An unnamed US-based institutional investor anchored the round with a €15m order, with the remaining demand filled by investors across the US, Europe, and Spain. The capital increase was capped at €30m by the company’s Board of Directors.
Oryzon Genomics announced it will use the proceeds to advance the clinical development of its oral LSD1 modulator vafidemstat for the treatment of aggression in CNS disorders such as Borderline Personality Disorder (BPD) and Autism Spectrum Disorder (ASD). Additionally, ongoing clinical programs in oncology and hematology will be expanded and the company will improve its legal and audit readiness for a potential dual listing on Nasdaq.
Oryzon has an advanced clinical portfolio with two LSD1 inhibitors, vafidemstat in CNS (Phase III-ready) and iadademstat in oncology (Phase II).
The company has other pipeline assets directed against other epigenetic targets like HDAC-6 where a clinical candidate, ORY-4001, has been nominated for its possible development in CMT and ALS. In addition, Oryzon has a platform for biomarker identification and target validation for a variety of malignant and neurological diseases.
mid-March, Porto-based startup PfxBiotech SA has achieved a major milestone by securing €9.5m in funding from the European Innovation Council (EIC) Accelerator. The company, based in Porto’s UPTEC innovation hub, was selected among 1,211 applicants, with only 6% of startups receiving funding.
Founded in 2022, PfxBiotech is one of the players pioneering precision fermentation to produce human milk proteins, with a particular focus on the iron-binding lactoferrin. The product is an ingredient in infant nutrition, reducing allergy risks, as well as in food supplements for seniors and athletes with specific dietary needs.
Besides human milk proteins, PfxBiotech’s platform allows developing oth -
er functional molecules. According to PfxBiotech, its approach represents a paradigm shift in food and biotech innovation. By using precision fermentation, the company can replicate nutritional compounds found in human milk, offering a sustainable and ethical alternative to traditional dairybased formulas. This method significantly reduces the environmental footprint of protein production while ensuring high bioavailability and safety for human consumption. The impact of this innovation extends beyond infant nutrition. The company is also addressing growing global demands for functional food ingredients that support immune health, gut health, and overall well-being. With an aging population, high-performance nutrition and next-generation bioactive ingredients are expected to become a future bull market. ■

DEAL Spanish pharma giant Ferrer Internacional SA has acquired the ex-US rights to commercialise Verge Genomics Inc’s small molecule treatment for amyotrophic lateral sclerosis (ALS), VRG50635. Ferrer will pay €112.5m in bio-dollars to market the Phase Ib 1-phosphatidylinositol-3-phosphate-5 kinase inhibitor across Central and South America, Southeast Asia, and Japan. The candidate, VRG50635, was developed using Verge’s artificial intelligence-driven Converge platform and is designed to
enhance neuronal survival in individuals with ALS. The agreement refills Ferrer’s development pipeline following the failure of its oral edaravone formulation in a Phase III clinical trial. Data from the ADORE trial indicated that oral edaravone (100mg once daily) did not achieve a statistically significant difference compared to placebo in slowing disease progression, as assessed by change from baseline in the ALS Functional Rating Scale – Revised (ALSFRS-R) score after 48 weeks of treatment. ■
PARTNERSHIP French preclinical CRO ERBC and Italian CDMO Menarini Biotech srl have partnered to improve the 5-15% success rate of preclinical-clinical transition of small molecules, monoclonal antibodies and ADCs. They describe their fully integrated service model as a “turnkey solution.” While the ERBC will contribute expertise in non-clinical safety and pharmacology studies, Menarini Biotech will
bring in its capabilities in Chemistry, Manufacturing and Controls (CMC), including scalable, platform-based manufacturing processes and early consideration of manufacturability, which are vital in de-risking and accelerating the path to clinical trials. The offer includes GLP and GMP-compliant services, regulatory-ready data packages, and expert guidance through IND submissions. ■
FINANCING At the beginning of May, Rome-based Angelini Ventures has marked its first investment in the cardiovascular and gene therapy space. The VC specialist co-led a US$45m Series B financing of Singapore’s Nuevocor. The oversubscribed round was coled by Kurma Partners; new investor Highlight Capital participated as well as existing investors Boehringer Ingelheim Venture Fund, ClavystBio, EDBI, SEEDS Capital and Xora Innovation. Nuevocor said it will use the proceeds to advance its programme in genetic dilated cardiomyopathy. The company addresses the condition by targeting the underlying biomechanical defects using its PrOSIA mechanobiology platform. Proceeds from the financing will be used to achieve clinical proof-of-concept for its lead candidate NVC-001 in patients with LMNA DCM, a rare genetic heart condition caused by mutations in the
LMNA gene. Dr Elia Stupka, Managing Director at Angelini Ventures, will join Nuevocor’s Board of Directors as Board member, together with Dr Lola Buono, Senior Associate, who will join as a Board Observer.
Nuevocor claims its approach surpasses the limitations of traditional gene therapy, which focuses on individual gene mutations, to treat defects within shared disease pathways across multiple cardiomyopathies. Its AAV gene therapies specifically target upstream biomechanical dysfunction in familiar cardiomyopathies. The approach centers on correcting cellular mechanotransduction pathways implicated in disease phenotypes across various genotypes. This is exemplified in their lead candidate NVC-001, which aims to treat LMNA-related DCM not by replacing the gene but by modulating the dysfunctional downstream pathways arising from LMNA mutations.
■
Back in February, Italian algae cracker specialist KelpEat srl (Savigliano) presented Solein ®powered protein crackers at the Pitti Taste event in Florence. Three months later, the company has secured an option to purchase 27 tons of Solein from Q3/2025 to the end of Q2/2028 from Finnish CO2-to-protein specialist Solar Foods Oy. KelpEat wants to introduce Solein-powered products to consumers in the US, one of the primary global markets for high-protein and healthy snacks and products. However, the company needs FDA approval for the dried tasteless protein powder made from Xanthobacter sp SoF1 bacteria before launch. The total value of the agreement for Solar Foods is up to €0.5m.
In mid-April, Spain became the first country in Europe to launch an AgriFoodtech Sandbox. It is designed to enable companies to rapidly test breakthrough technologies in close coordination with policymakers from the government, the regions of Navarra and La Rioja, and agrifood innovators, and under regulatory supervision. The sandbox is designed to speed up technology transfer to the emerging markets of precision agriculture, food ingredients, alternative protein production that drive the production of sustainable, and healthy food and feed. The Sandbox is being coordinated by the National Centre for Food Technology and Safety (CNTA), which is selecting candidates in response to a first call and will support the technology transfer process
EVENT Spain’s biotech sector drives innovation, economic growth, and scientific excellence. With strong R&D investment, global research impact, and a leading role in gender equality, it’s a key player in health, sustainability, and strategic autonomy. BIOSPAIN 2025 in Barcelona will showcase its potential and connect global biotech leaders.
› Ion Arocena, CEO of AseBio, Spain

Spain’s life sciences sector has experienced steady growth, positioning itself as a strategic driver of innovation and economic development. Backed by a strong scientific base, highly qualified talent, and a growing network of research centers and tech companies, it has become a key player in health, food, sustainability, and digitalisation. Within this ecosystem, biotechnology plays a central role as a transformative force in addressing global challenges.
The Spanish biotech sector stands out for its dynamism, knowledge generation, and impact on the productive fabric. From startups to established firms, biotech companies are leading innovation, R&D investment, and technology transfer. According to the AseBio Report 2024, the sector invested €1.282bn in R&D in 2023 – 68% from private funds –ranking fourth among Spain’s most R&Dintensive industrial sectors.
In 2024, biotech companies raised €181m through 53 funding operations. The sector remains the most researchintensive within Spanish industry, with 15.99% of its workforce in R&D roles –second only to dedicated R&D services. Overall, Spain hosts 4,411 companies engaged in biotech activities, over 1,000 of which are classified as biotech firms. Catalonia leads both in company numbers and average turnover.
Human health is the main area of activity (57%), followed by food (27%), agriculture and forestry (16%), animal health and aquaculture (15%), environment (12%), and industry (10%).
Spain also excels scientifically, ranking ninth worldwide in biotech scientific output, contributing 2.49% of global publications in 2023. Moreover, Span -
ish biotech research is cited 20% above the global average, underscoring the relevance and impact of the country’s scientific contributions.
The sector’s economic footprint is equally notable. Biotech activity generated €13.094bn in income (1.1% of GDP) and €5.229bn in tax revenue (0.4%). It also provides 131,214 jobs (0.67% of national employment), reinforcing its role as a generator of stable, high-quality jobs.
Additionally, the sector leads in gender equality. Since 2010, women have consistently made up over 50% of biotech workforces. Women represent 60% of biotech R&D staff and hold 29% of executive positions – an encouraging step toward more inclusive leadership.
This vibrant and strategic industry will be at the center of discussion at BIOSPAIN 2025, one of Europe’s leading biotech events, taking place in Barcelona from October 7-9, with participation from national and international experts.

AACR Poland’s Ryvu Therapeutics has given an update on its preclinical ADC RVU305 at AACR RVU305 and at ADC Payload Summit in early May. At the AACR Annual Meeting, Ryvu presented data on its brain-permeable, MTA-cooperative PRMT5 inhibitor RVU305 and ONCO Prime, novel synthetic lethal targets for colorectal cancer. At the ADC Payload Summit, CSO Krzysztof Brzózka presented Ryvu’s next-generation payloads. “We are rapidly advancing RVU305 with IND/CTAenabling studies on track for completion in H2/2025. In addition, our proprietary ONCO Prime platform has identified several novel synthetic lethal targets, including targets for KRAS-driven tumors, which offer immense potential to transform cancer treatment. We are also developing innovative next-generation ADCs with new payload classes,” Brzózka summarised. RVU305 demonstrated robust tumor growth inhibition (TGI) in MC38 tumors with MTAP loss, with confirmed target engagement, while the growth of the MC38 variant with an intact MTAP gene was unaffected at pharmacologically active doses. Furthermore, co-treatment with RVU305 and anti-PD-1 antibody was well tolerated and resulted in antitumor activity. RVU305 also demonstrated CNS penetration with predicted efficacious exposure in the brain in cynomolgus monkeys.
Through CRISPR screening of hIPSCs and patient tissue, Ryvu identified a new target for colorectal cancer (CRC) through synthetic lethal (SL) interactions and a conjugable small molecule drug candidate. ■

ADC Prague-based Sotio Biotech AS has reported promising preclinical data of its anti-cadherin-17 and antiLRRC15 antibody-drug conjugates (ADCs) SOT109 and SOT106, in solid tumours at the American Association for Cancer Research (AACR) Annual Meeting in Chicago in late April.
Sotio Biotech’s CDH17-targeting ADC SOT109, which carries Synaffix’ topoisomerase I payload Syntecan (DAR: 4) for the treatment of gastrointestinal cancers, was overexpressed in 98% of colorectal (CRC), 65% of gastric and 46% of gastroesophageal tumours in all disease stages and is a biomarker for CRC. Toxicity assessents demonstrated that SOT109 was well tolerated in cynomolgus monkeys and mice at dose levels of up to 25mg/kg and 150mg/kg, respectively, and showed no signs of target-mediated toxicity neither clinically nor at microscopic level. The pharmacokinetic profile observed in cynomolgus monkeys was sufficient to support Q2W or Q3W dosing in clinical settings. The drug candidate showed a preliminary therapeutic index favourable for SOT109 clinical development. According to Sotio Biotech, SOT109 exhibited potent efficacy, producing significant and sustained tumour regressions in cell and patient-derived xenografts.
Sotio Biotech’s LRRC15-targeting ADC SOT106 with four Ligachem-developed MMAE payloads linked to mAb light chains achieved statistically significant tumour regression in an LRRC15 low-expressing xenograft model of pediatric osteosarcoma. Complete responses and potent antitumour efficacy were also observed across
a range of models where LRRC15 is expressed directly on tumour cells, including multiple subtypes of soft tissue sarcoma, a therapy-resistant non-small-cell lung cancer model, and head and neck squamous cell carcinoma. SOT106 was well-tolerated in a dose of up to 10mg/kg and displayed a favourable pharmacokinetic and safety profile, good stability in vivo, and a high therapeutic index. Sotio Biotech expects to file for IND in the fourth quarter of 2026.
“SOT109 continues to show excellent tolerability and strong anti-tumour activity across multiple preclinical models of colorectal cancer, while SOT106 offers a novel precision approach with broad applicability in LRRC15-positive sarcomas and other solid tumours,” concluded Martin Steegmaier, CSO of Sotio Biotech. ■
VC Vilnius-based deeptech Venture
Capital investor Iron Wolf Capital announced in May it has reached one third of the target size of its second €100m fund. Approximately 40 business angels and the Lithuanian state bank ILTE have already made commitments. With the fund, Iron Wolf Capital aims at investing in sectors beyond telecommunications, IT, and semiconductors, namely AI, biotech, energy and space technologies. Typical investment tranches in the first fund ranged from €500,000 to €2bn, enough to bring 25 to 50 start-ups to life.
SLAS EUROPE With 25% more visitors (1,700), exhibitors (378) and new product launches (40) than last year, the 6th Annual Meeting of the Society for Laboratory Automation and Screening SLAS Europe 2025 broke all records. Highlights in Hamburg included advances in the automation of CRISPR, toxicity and phenotypic screening in human 3D cell aggregates and the presentation of a completely new protease-resistant and highly specific oral drug class.
Reducing the preclinical attrition rate, faster identification of new drug targets and specific binders with a high therapeutic index were the focus of international technology developers at the 6th SLAS Europe (20–22 May 2025) against a record backdrop. Almost 1,700 visitors (2024: 1,078) and 137 exhibitors (2024: 109) responded to the call of Hamburg-based SLAS President Philip Gribbon (see EuropEan BiotEchnology 4/2024) to find new partners, talent and technologies and “help shape the future of screening sciences”. The Hanseatic city is set to become the new European life sciences automation epicentre “with three further SLAS Europe conferences by 2031“,
Gribbon told EuropEan BiotEchnology. Strategically, the local LSN-cluster is expanding relations to the ambitous Scandinavian biotech ecosystem.
Inspired by Arthur C. Clarke’s Third Law “any sufficiently advanced technology is indistinguishable from magic”, ThirdLaw Molecular aims to replace antibodies with oral mid-size drugs that are non-immunogenic, not subject to protease degradation, but exactly match the shape of receptors and intracellular drug targets (see p. 72).
At SLAS, the US start-up presented its

ORYL Photonics SA’s co-founders Nathan Dupertuis (left) and Orly Tarun. The Lausanne-based start-up won the SLAS Ignite Award for OrylF1 (background), their laser-based light scattering instrument that allows ultrarapid, accurate and cheap high-throughput assessment of drug solubility and aggregation. It is 100 times faster than the previous standard HPLC and that needs 10 times less time than SLS/DLS methods, requires only a hundredth or tenth of the sample volume and preserves the sample material for subsequent kinetic or orthogonal measurements.
DNA-encoded library of 4.5 billion Spiroligomer™ molecules for the first time that yielded nanomolar binders in a screening. “We are eager to partner with companies interested in exploring this new modality for their existing targets,“ senior researcher DJ Bernsteel told EuropEan BiotEchnology in Hamburg.
A technology conceived in Germany and commercialised in the USA that promises to reduce the side effects of the currently fastest-growing therapy classes of antibody drug conjugates (ADCs), bispecific antibodies and CAR-T cell therapies is presented on p. 73. An allosteric conformational switch can be used to modulate the molecular geometry of (engineered) antibodies and eliminate T-cell hyperactivation independently of the paratope.
In addition, SLAS Europe focused on technologies that enable the automated cultivation of human cells and organoids and their cost-effective phenotypic screening. Chris Oliphant from AstraZeneca plc presented an automated platform for the culture of functional polarised macrophages from iPSCs (induced pluripotent stem cells) and immune-arrayed CRISPR screenings to identify drivers for chronic inflammation in autoimmune disorders. According to Oliphant, AZ investigates factors affecting efferocytosis in cells and organoids – the process where macrophages, engulf and clear apoptotic or dead cells to ensure healthy cell turnover and prevent the release of cellular contents that could trigger inflamma-
tion. As whole-genome CRISPR screening is currently too costly, due to limitations in reagent delivery, cell number limitations, and endpoint complexity, AZ carried out a case study across 186 genes. In the end AZ identified Gene35 as a relevant modulator upholding efferocytosis.
At the beginning of April, Jeffery Truong and colleagues had presented ENVLPE, a new approach (cEll, 10.1016/j. cell.2025.03.015, p. 50) that cuts the cost of CRISPR screens significantly by boosting efficiency. Instead of immunogenic and inefficient lentiviral/AAV vectors, the future company founders used virus-like particles (VLPs) equipped with glycoproteins for organ-specific delivery of CRISPR/Cas cargo. Additionally, only functional ribonucleoproteins (RNPs) consisting of Cas nuclease, guide and editing RNA strands are attached to an aptamer located at the inner VLP surface and are transiently expressed preventing immune reactions.
SLAS Europe also saw numerous new product launches for the expensive and, until now, poorly scalable processes required by drug developers such as AZ for the propagation, passage and differentiation of human iPSCs and organoids. The market is booming because organoids represent a significant step in the evolution to more representative disease modelling. This would significantly increase the preclinical success rate and thus the revenue per drug candidate.
In addition to British Wildcat Laboratory Solutions Ltd., which markets plastic-free 2D lab tube racks from the US company PulpFixin outside the US, Hamburg-based mo:re GmbH received the New Product Award, which SLAS presents for breakthrough innovations. Managing Director Lukas Gaats, who founded mo:re together with David Hackenberger (CTO), plans to make Mo:bot the “gold standard of scaled and automated organoid-based” drug development. Mo:bot integrates artificial intelligence to allow non-specialists to reproducibly mass-produce four organoid types – heart, brain, liver, and pancreas. Another one of the 12 automation start-

ups supported by SLAS as part of its AveNEW programme and portrayed on EuropEan-BiotEchnology com – Acoustofab Ltd (t1p.de/l3nny), Instromeda Ltd (t1p.de/ b2nfu), LifeTaq GmbH (t1p.de/trm88), Minos Biosciences SAS (t1p.de/1zwid ), MyxoTech GmbH (t1p.de/ku3xl ), Nanovery Ltd (t1p.de/9vvec), Nanoworx BV (t1p.de/ sivmj ), NestEgg Labs BV (t1p.de/3g0p5 ), ORYL Photonics SA (t1p.de/cc561), Phabioc GmbH (t1p.de/8ymey), Third Law Molecular Inc (t1p.de/z5y71), and Unicorn Biotechnologies Ltd (t1p.de/fd2hw) – also aims to enable the reproducible mass production of 3D organoids: Viennese LifeTaq-Analytics GmbH’s Oli-MAT cell cultivation unit, facilitates high-throughput production and scale-up of standardised 3D cultured tissue models for lung, kidney, the gastrointestinal tract, and skin under a controlled oxygen environment.
Another finalist for the SLAS Ignite Award for the most innovative business concept, presented a fully remote-controlled system for cell and organoid handling. EMMET, which was developed by Adam Glen and Jack Reid, automates every step in the cell culture process: from media exchanges and passaging to executing complex differentiation workflows. With onboard autonomous analytical systems and user-defined, machine-driven protocols, Emmet can automate the culture of any cell type, from cell lines to pluripotent stem cells (ESCs/iPSCs). Molecular De-
vices and Heartbeat.bio gave an update of their AI-powered organoid culture system CellExpress.ai.
Beyond the SLAS award-winning products, there were some fascinating technologies of the future on display: the acoustic levitation technology developed by British company Acoustofab Ltd, for example, replaces all of the pipetting and robotics hardware, tips and consumables that were previously required. A custom-built array of ultrasonic transducers that are precisely controlled to create so-called acoustic holograms levitates the components to be pipetted, sorted, mixed or printed completely contact-free. Another innovation launched by Instromeda Ltd at SLAS Europe is 1/3 the price while requiring 95% less human intervention compared to traditional SPR platforms.
German Ignite Award finalist Phabioc GmbH came up with two innovations reducing process and formulation development cost for complex molecules, such as antibodies or vaccines. Its SpecPlate allows for the dilution-free measurement of absorption spectra in highly concentrated samples while its PermeaPad is a biomimetic barrier for gastrointestinal, oral and mucosal drug permeability studies.
The next SLAS Europe conference will take place in Vienna from 19 to 21 May 2026. Hamburg will host SLAS Europe in 2027, 2029 and 2031. ■
t.gabrielczyk@biocom.eu
PLATFORM Thirdlaw Molecular Inc (Blue Bell, Pennsylvania) focuses on platforms that utilise novel Spiroligomer™ molecules, which possess fused-ring structures with multiple R-groups. Screening a DNA-encoded library consisting of 4.5 billion members has found nanomolar binders. The new platform has been presented at SLAS Europe 2025 in Hamburg.
In drug discovery, compounds are classified as either small molecules or biologics. Small molecules, defined by Lipinski’s “rule of 5,” weigh under 500 Daltons and are optimised for bioavailability. However, they mainly bind to small protein pockets, limiting their scope and increasing off-target effects. Biologics, such as antibodies, interact more specifically with large protein surfaces, avoiding many off-target interactions. However, they require injection and are prone to degradation.
At ThirdLaw Molecular, we have developed Spiroligomer™ molecules, a novel therapeutic class in this intermediate size range. They are very different from, and complementary to, the recently popular cyclic and stapled peptides (5003000 Da)–where both seek to combine the benefits of small molecules and antibodies.
Spiroligomer™ molecules are built from cyclic monomers connected into fused-ring, ladder-like frameworks with multiple stereocenters. Their rigid, protease-resistant backbones differ from the flexible structures of peptides. Embedded stereocenters allow precise 3D configurations and placement of functional groups. ThirdLaw Molecular’s synthetic platform also enables rapid incorporation of thousands of non-natural amino acids or R-groups.
As described above, the structure of Spiroligomer™ molecules makes them unique; however, a critical question re -
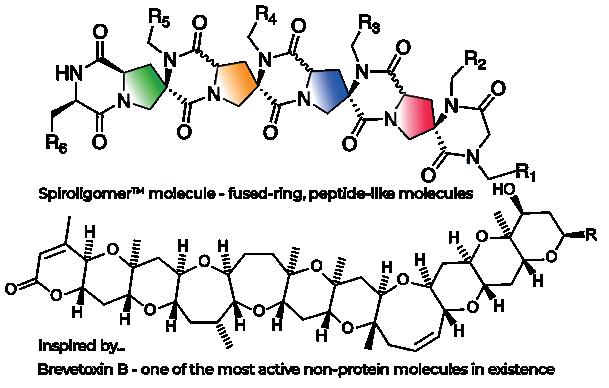
Spiroligmers drafted by Affinity Designer
mains: how do these structural changes result in superior pharmaceuticals? In a chloroalkane penetration assay, ten of 11 random Spiroligomer™ tetramers showed cell permeability; eight entered cells within 20 minutes via passive diffusion. This is significant: 85–90% of human proteins are intracellular, yet most approved small-molecule drugs target only a small subset (~850). Spiroligomer™ molecules bind to grooves found in all proteins, including “undruggable” intracellular targets. They also demonstrate oral bioavailability without special formulation – something peptides generally lack.
In collaboration with X-Chem Technologies, ThirdLaw Molecular has created a 4.5-billion-member DNA-Encoded Library (DEL) of Spiroligomer™ macromolecules. These can mimic monoclonal an-
tibodies in protein binding but are about 1/30 th the size, with the potential for oral delivery. Screens against extracellular targets (validated by antibodies but lacking small-molecule drugs) have already produced nanomolar binders. Functional validation is underway.
A second DEL of cell-permeable Spiroligomer™ molecules is in development for Q3/25. This library will focus on intracellular proteins currently inaccessible to traditional drug types.
In summary, ThirdLaw Molecular’s Spiroligomer™ molecules combine the size benefits of peptides with critical advantages: passive cell permeability, protease resistance, tunable 3D conformations, and R-group modularity. These features support a new generation of orally available, selective therapeutics for extracellular and intracellular targets. ■
ANTIBODY ENGINEERING Mabswitch Inc's platform technology enables the remote and repeated regulation of antibody affinity in various immunotherapeutic applications. This makes it possible to actively and individually mitigate adverse effects, increase specificity and maintain efficacy by improving immune fitness and persistence.
› Dr Yemi Onakunle, CEO, Mabswitch Inc., Los Angeles, USA
Mabswitch is developing ON/OFF control switches for antibodies using our Universal Allosteric linker and Switch Module for Antibodies (UNASMA™) technology that facilitates the regulation of virtually any antibody’s affinity for its target under physiological conditions. We achieve this by inserting a calmodulin-derived allosteric modulator domain as a linker between the variable domains of an antibody scFv, replacing the standard (G4S)n linker. This allosteric-linker switch is activated by a small molecular ligand, modulating antigen affinity through conformational changes in the antibody's binding site, which are triggered by the interaction of the ligand with the allosteric-linker. The fully human allosteric modulator domain ensures low immunogenicity and is compatible with virtually any scFv.
Many cancer immunotherapies face significant challenges, including triggering harmful side effects due to broad immune activation or losing effectiveness over time. Our innovative approach enables precise control over when and where immune cells are activated, enhancing the safety and efficacy of treatments, by:
› Activating only at the tumor site to avoid harming healthy tissues.
› Providing precise, on-demand control to limit side effects.
› Reducing immune system burnout by improving its fitness and persistence.
Our technology addresses key limitations of conventional immunotherapies, and leading pharmaceutical companies like AbbVie, BMS, and Amgen have taken notice, awarding us Golden Tickets that provided laboratory resources to validate the technology. Additionally, Mabswitch secured NIH grants to validate UNASMA™ in two groundbreaking applications: remote-controlled CAR-T therapy and mild elution immunoaffinity ligands.
But there is more: our award-winning remote-controlled antibody platform is not just for cancer, but broadly applicable to most antibody-based therapies, and could even enable first in class prod-
ucts in fields not yet accessible to therapeutic antibodies. It could revolutionise treatments for:
› Autoimmune diseases (like arthritis, multiple sclerosis, and lupus).
› Metabolic disorders (including diabetes and obesity).
› Blood and neurological diseases.
› Other next-generation therapies, such as CAR-T cell treatments and antibody-drug conjugates.
Mabswitch develops breakthrough cancer immunotherapies, but also offers to engineer our affinity switch into your antibody for application as remotely controllable ADC, CAR, TCE or other antibody-based products.
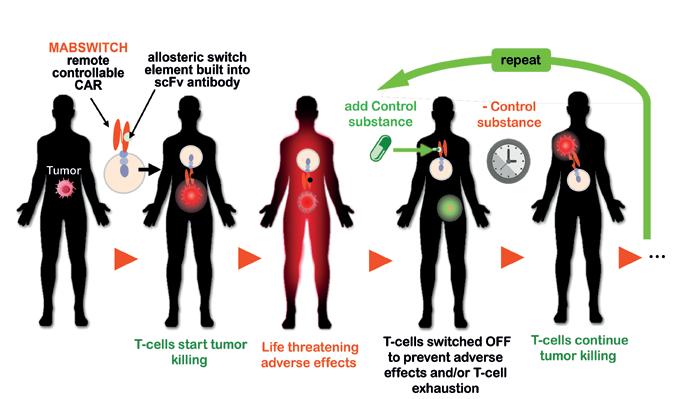
Mabswitch application example: “Time-resolved / repeatable ligand-controlled T-cell activation”
SEQUENCING BY EXPANSION For nearly two decades, Swiss pharma and diagnostic leader Roche has been searching for a next-generation sequencing method in personalised medicine to overcome the limitations of short read lengths in existing sequencing technologies. Following beta testing, the company plans to launch its new Sequencing by Expansion (SBX) technology commercially next year.
For 20 years, Illumina has commanded around 80% of the global next-generation sequencing (NGS) market with short-read technologies. But since February, its grip has been challenged. After closing its Applied Sciences division in April 2013 and long technology scouting, Switzerland’s pharma and diagnostics major Roche reported a breakthrough in long-read sequencing in February. Its nanopore-based sequencing-by-expansion (SBX) method is set to enter the global market in 2026 for Research Use Only (ROU). Due to a low signal-to-noise ratio and integration into an end-to-end sequencing ecosystem (see figure), it seems suitable for a wide range of applications in genome, exome,
and RNA sequencing. It is expected to sequence up to seven complete genomes per hour at 30-fold coverage, equivalent to five billion duplex reads per hour—sequencing both DNA strands simultaneously. In the medium term, however, Roche intends to establish SBX as the gold standard for diagnostics, prognosis and patient stratification in personalised medicine.
With the Broad Institute serving as a beta tester for all personalised med stuff, Roche starts exploring potential applications of its SBX platform, originally developed by Stratos Genomics Inc. Stratos

Nanopore sequencing was pioneered by the group of Hagan Bayley at the University of Oxford, UK, and commercialised first by 2005 University of Oxford spin-out Oxford Nanopore Technologies. It works by passing a single-stranded DNA or RNA molecule through a biological nanopore and detecting changes in ionic current to identify each nucleotide.
co-founders Mark Kokoris and Robert McRuer pioneered SBX chemistry, which involves converting DNA into a longer surrogate molecule called an Xpandomer to enhance sequencing accuracy and efficiency. This approach addresses the signal-to-noise challenges inherent in traditional nanopore sequencing methods which currently account for 5-10% of the total NGS market. Currently, the 0.7bn long-read sequencing market is growing by 31% annually and is set to reach a market share of up to 30% in the next seven years. This is due to the fact that applications requiring more comprehensive genomic information – such as structural variation analysis, epigenetics, and complex disease research – are increasingat high pace. SBX allows single-molecule sequencing with a significantly higher signal-to-noise ratio compared to the SMRT platform of PacBio and the nanopor e sequencing approach developed by Oxford Nanopore Technologies. “The solution to the signal-to-noise problem is a crucial efficiency factor for this technology. Thanks to this capability, we can operate flexibly across different throughput levels using the same sequencing system, which provides users with a significant advantage,” says Kokoris, since the 2020 acquisition of Stratos Head of SBX Technology at Roche.
The sequences for subsequent nanopore sequencing are first expanded into Xpandomers. These are based on expandable nucleoside triphosphates, known as X-NTPs, which contain signal-
enhancing reporter codes. A specially developed polymerase incorporates the X-NTP molecules into the growing Xpandomer, stabilised by polymerase enhancers (PEMs). After synthesis, the bonds in the Xpandomer are cleaved, resulting in an extended molecule approximately fifty times longer than the original sequence. This process complements the single strands during the reaction but, unlike PCR, does not amplify them. Although not entirely error-free, Roche reports a sequencing accuracy of at least 99.8% for single-nucleotide variants.
The second innovation concerns the reading of the sequences. After synthesis, the Xpandomer is passed through a nanopore, where voltage pulses control its movement and the reporter codes are read with high precision. This is achieved using a high-throughput sensor module developed by Genia Technologies, based on Complementary Metal Oxide Semiconductor (CMOS) technology, which enables ultra-fast and parallel processing.
Broad Clinical Labs, a subsidiary of the MIT and Harvard-affiliated Broad Institute, will assess SBX’s potential in theranostics. Although financial details of an agreement reported at the end of June remained undisclosed, Roche appears to be primarily interested in leveraging the Broad Institute’s extensive network of leading genomics researchers. An initial pilot project will focus on diagnosing rare genetic diseases through whole genome analysis.
Beyond diagnostics, SBX will also be used to select patients for clinical trials and to support research applications such as target screening via single-cell RNA sequencing. Current collaborations include projects with Edwin Cuppen from the Hartwig Medical Foundation to sequence cancer genomes, and with Aziz Al’Khafaji from Broad Clinical Labs to determine cancer therapy responses using multi-omics analyses. Unlike methods used by US market leader
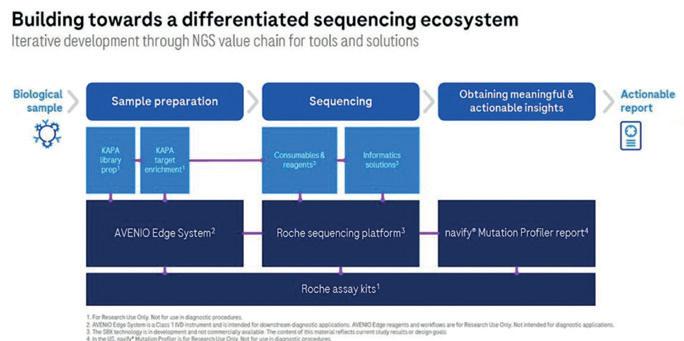
Roche is going to offer an integrated approach to SBX, integrating AI analysis, consumables and hardware.
Illumina, SBX enables the reading of entire genes with high speed, accuracy, and error-free performance. Its ability to generate read lengths exceeding 100 kilobase pairs offers new insights into disease mechanisms and therapeutic targets, driving forward the field of personalised medicine.
The scalability of SBX, developed by Stratos Genomics co-founders Mark Kokoris and Robert McRuer, is made possible by a high-throughput complementary metal-oxide-semiconductor (CMOS) sensor module. This enables fast, real-time base calling and supports
parallel processing of multiple samples, making the technology suitable for both small-scale research and high-throughput clinical diagnostics.
The collaboration with Broad Clinical Labs is intended to integrate SBX into clinical and translational workflows, ultimately leading to its adoption in routine diagnostics. A joint feasibility study will demonstrate the viability of family-wide whole genome sequencing in neonatal intensive care settings and as a standard diagnostic tool for diseases such as cystic fibrosis and sickle cell anaemia. ■ t.gabrielczyk@biocom.eu
RNA SILENCING Pharmacologists at the University of Göttingen, Germany, have reported a simple way to target and silence individual mitochondrial mRNAs in vivo, opening the avenue towards phenotypic screens (10.1126/ science.adr3498). At least 1.6 million patients worldwide suffer from mitochondrial diseases. At the end of May, Luis Cruz-Zaragoza and colleagues presented a way to target multiple mRNAs simultaneously in different cell types. Their approach involved using synthetic peptidemorpholino chimeras, that is chimeras composed of a mitochondrial targeting peptide and a polymorpholino oligonu -
cleotide, which were synthesised using click chemistry and subsequently imported into mitochondria to block translation. They tested their technique in cells including human cardiomyocytes and mouse liver cells – finding it could block mitochondrial protein production consistently. Applying this technique also revealed how mitochondrial gene expression affects cellular homeostasis. “The availability of this chimera-based mitochondrial mRNA silencing strategy provides a valuable tool for researchers to study mitochondrial gene expression, protein biogenesis, and their integration into cellular physiology,” he said. ■
NOVEL FOOD A recent Euroconsumers report underscores the potential of cultivated meat as a sustainable protein source to aid the EU in achieving its climate objectives and enhancing food security. A study by the consumer advocacy group found that 44-56% are open to trying cultivated meat, contingent upon EFSA approval. Despite this interest, the current EU regulatory framework imposes a lengthy approval process for novel foods, posing significant challenges for start-ups with limited funding. Euroconsumers advocates for integrating cultivated meat into the EU Biotech Act to streamline approval procedures, attract investment, and make these products more accessible to consumers. The report emphasises that, beyond ensuring safety, factors such as taste, price, and nutritional value are critical for consumer acceptance. By addressing these considerations, cultivated meat could play a pivotal role in diversifying Europe’s protein sources and reducing the environmental impact of food production. ■
COMPETITIVENESS Biotechnology continues to be a powerful economic engine for the EU, according to a WifOR Institute report commissioned by EuropaBio, Europe’s largest biotechnology industry group. The report analysed industry growth from 2008 to 2022. The sector’s gross value added (GVA) reached €38.1bn in 2022 - almost double that of 2008. Productivity stands out: biotech generated €160,000 GVA per employee, surpassing high-performing sectors like finance and ICT. Exports reinforce biotech’s strategic importance, with a €51.7bn trade surplus in 2022. The report captures the direct, indirect, and induced contributions of biotechnology across the EU27. It affirms the sector as not only research-intensive but also a core pillar of Europe’s economic resilience and global competitiveness.
co-operation with the following organisations:


















would like to subscribe, please refer to european-biotechnology.com

REGIONAL EVENTS SLAS is following up a record-breaking conference and exhibition in Hamburg, Germany, with two free-to-attend regional Meet-Ups this fall. We’ll be in Vienna on 23 September and Munich on 15-16 October.
The Society for Laboratory Automation and Screening is excited to gather members of the life sciences and lab automation communities together this fall. Join fellow life sciences researchers and lab automation professionals for two engaging afternoon scientific Meet-Ups in Europe. These informal gatherings foster collaboration and knowledge exchange, featuring a guest speaker sharing insights on cutting-edge developments in the field.
Regional Meet-Ups take place at local research facilities or innovation providers’ facilities, offering a behind-thescenes look at innovative technologies and workflows. Open discussions and networking opportunities encourage dialogue across disciplines. The day concludes with a relaxed apéro, allowing participants to connect over drinks and light refreshments.
in September
Save the date of 23 September from 14:00 - 17:30, when we’ll be at the Research Institute of Molecular Pathology (IMP), Campus-Vienna-BioCenter 1 in Vienna. We’re excited to host this MeetUp at the Vienna BioCenter, a top biology research center in Europe, comprising six research institutions, with IMP at its core. We invite students, academic and government researchers, technology providers and biotech entrepreneurs to join us at IMP, the leading molecular biology institute in Europe, to learn more about SLAS, share scientific insights and solve challenges.
Fun fact: In June 2020, SLAS was scheduled to hold its annual Europe Conference and Exhibition in Vienna, but COVID forced us to take that conference online. We still had some amazing keynote speakers – Johannes Grillari (Director of the Ludwig Boltzmann Institute for Traumatology) and Giulio Superti-Furga (Professor of Medical Systems Biology at the Medical University of Vienna).

We are excited to bring the educational and networking experience back to Vienna in person, 19-21 May 2026 with our Europe Conference and Exhibition at Messe Wien Exhibition & Congress Center. Find out how you can be a part of the #heimmannschaft or “home team” for SLAS as we get ready for SLAS Vienna.
On October 16, SLAS is teaming up with the Munich Lab Automation Community to host a Regional Community Meet-Up at the NanoTemper global headquarters. The afternoon will be comprised of a look into NanoTemper’s journey and its technological innova -

› September 23, 2025, Vienna
SLAS Regional Meet-Up
› October 15-16, 2025, Munich
SLAS Regional Meet-Up
› February 7-11, 2026, Boston
SLAS 2026 International Conference & Exhibition
tions and will be followed by a networking apéro. Speakers include Ali Ertürk, PhD, Erturk-Lab and Philipp Baaske, Co-founder and Executive Chairman of NanoTemper.
NanoTemper, a company providing easy-to-learn, high-quality biophysical tools that help researchers discover new therapies, is located at Tölzer Str. 1 in Munich. This event is also free to attend and a relaxed way to connect with others interested in life sciences research, laboratory automation and other scientific areas.
SLAS Technology guest editors Kalpesh Gupta, Ph.D., Moderna, and Mario Richter, Ph.D., AbbVie, are assembling a special edition on “NexusXp: The Connected Lab” in the field of lab automation. This edition aims to explore cutting-edge advancements, innovative technologies, and visionary concepts shaping the future of integrated laboratories. We invite researchers, scientists, and industry experts to submit their original research papers, case studies and review articles.
More information is available at https://www.slas.org/publications/callfor-papers/#Discovery.
■
BIOTECH COMMUNICATION MATTERS
BIOTECH AUSTRIA’s Communications & Public Relations taskforce provides expert guidance to help companies navigate complex messaging, enhance visibility and build strategic communication expertise.
Since its founding in December 2020, BIOTECH AUSTRIA has played a central role in advancing Austria’s life sciences sector. A cornerstone of its strategy is empowering member companies through access to specialist expertise. The Task Force Communications & PR (TACO) was initiated with founding member MC Services, an international life sciences investor relations and public relations firm contributing its longstanding expertise. The group offers practical guidance, peer exchange, and strategic insight to raise the visibility of Austrian biotech locally and in the international investment and biopharma community.
A coherent communication strategy is essential to every organization, regardless of its size or industry. Clear and consistent messaging supports various aspects, including corporate development, talent acquisition, investor engagement, partnerships and public trust. When communication is aligned with business goals, companies can maximize the impact of key milestones.
Life sciences companies face unique communication challenges. Transforming complex scientific ideas into accessible and compelling messages for scientific, financial or lay audiences requires both precision and strategic thinking. The stakes in this field are often high, as clinical trial results, financing milestones or regulatory updates can


› July 2, 2025, Vienna
CEO Lunch
› September 8, 2025, Vienna
Heurigen Meeting
› October 23-24, 2025, Graz
3. BIOTECH SUMMIT AUSTRIA
nication strategies, these events sharpen positioning and extend international reach.
To support members in navigating these demands, BIOTECH AUSTRIA’s Communications & PR Task Force shares best practices and encourages peer exchange. Regular workshops have addressed topics such as PR dos and don’ts, annual communication planning, leveraging conferences strategically, pitching to media and using social media to amplify key messages and milestones.
significantly impact a company’s trajectory. At the same time, biotech firms must meet diverse stakeholder expectations – from investors and regulators to patient groups – while standing out in an increasingly competitive landscape. International visibility is a key success factor: it enables biotech companies to raise capital, secure pharma partnerships, and expand globally. Conferences like BIO-Europe, hosted this autumn in Vienna, and BIOTECH AUSTRIA’s own Biotech Summit in October, are vital platforms for showcasing innovation, meeting potential partners, and spotlighting Austria’s life sciences ecosystem on the global stage. With professional preparation and targeted commu-
Building on early successes and member feedback, BIOTECH AUSTRIA has expanded its support framework to include working groups on finance, human resources and strategy, as well as a preclinical task force. The Communications & PR Task Force continues to evolve, with upcoming workshops planned on topics such as effective investor presentations, crisis communications, and engaging international trade media. As Austria’s biotech sector advances, the task force remains a trusted resource, helping companies communicate with clarity, confidence, and credibility. ■
For more information about MC Services, visit www.mc-services.eu
ANNUAL ASSEMBLY In Budapest, 11 associations of biotech students and young professionals from 9 countries came together at the Young European Biotech Network (YEBN) Assembly to elect a new board, reinforce their unity, and define a shared vision for the future of young biotechnologists across Europe.
On May 3, 2025, in the vibrant city of Budapest, the Delegate Assembly of the Young European Biotech Network (YEBN) was held, hosted by the Hungarian association MaBE – Magyar Biotechnológus-hallgatók Egyesülete. This significant event marked a pivotal moment for the network, bringing together representatives from member organizations across Europe for constructive dialogue, idea sharing, and strategic planning. During the assembly, a new Executive Board was elected, composed of seven motivated and skilled individuals, with Pau Esteve re-elected as President. This new leadership team is poised to guide YEBN through a phase of renewal, growth, and consolidation, aiming to strengthen the association’s role in the European biotechnology landscape.
The Delegate Assembly was more than a routine institutional meeting – it was a powerful opportunity to reinforce a sense of belonging and collectively define an ambitious and inclusive vision for the future. In a global context where collaboration is essential, YEBN reaffirms its core mission: to build a strong bridge connecting young biotechnologists, organizations, and talents from different countries, creating a transnational, dynamic, and opportunity-rich community. This community values intercultural dialogue and fosters an inclusive envi-
ronment where every voice is heard and every idea has the potential to become reality.
A continuously evolving network YEBN’s true strength lies in its networking capacity. Today, the association brings together eleven member organizations from nine different European countries, each contributing unique experiences, perspectives, and expertise. This growth reflects a gradual process built on openness, trust, and collaboration. Among the key players are: Biotecnologi Italiani, NGB from France, MaBE from Hungary, Biocatalyst Foundation from the Baltics, the Finnish Biotech Club, Biotech Academy in Rome, NEBIST from Portugal, Warsaw Society of Biotechnology “Symbioza”, ÖMG -

› June 30, 2025, Online YEBN Online Meet-up
BT from Austria, BtS from Germany, and Réseau Biotechno from France. This diversity enriches YEBN as a multifaceted platform, capable of flexibly and creatively addressing the challenges of a rapidly evolving sector.
Beyond institutional strengthening, YEBN is experiencing a period of renewed enthusiasm at the individual level. In recent months, 20 new volunteers have joined the network, bringing fresh energy, skills, and ideas. These include passionate students, emerging researchers, and young professionals, all eager to actively contribute to the community’s growth. Thanks to their involvement, YEBN is becoming increasingly vibrant, creative, and engaging, providing a stimulating environment to develop innovative projects and build lasting relationships. ■

21st. BIO (DK) 65
A
ABB Ltd (CH) 53
Abundance Gate (AT) 65
ADC Therapeutics (CH) 64
Afyren (F) 16, 82
Agilent Technologies (USA) 53
Alchemab Therapeutics (GB). . . . . . . . . . . 62
Alvotech SA (ISL) 60
Amplifybio LLC (USA) 82
Amsilk GmbH (DE) 65
Aphea.Bio NV (BE) 82
ASEBIO | BIOSPAIN 2025 (ES) 30, 31, 68
Astoriom (GB)...................... 53
Astraveus SAS (F) 63
AstraZeneca AB (SE/UK) 70
Augustine Therapeutics NV (BE) 24
Avecom NV (NL) 61
Axxam SpA (IT) 65
Azafaros BV (BE) 63
Azenta Life Sciences (USA) 53
B Bayer Co.Lab Berlin (DE) 13, 41
BioGeneration Ventures (NL) 63
BioNTech SE (DE) 63, 64, 82
BIOTECH AUSTRIA | BIOTECH Summit 34, 35
Boehringer Ingelheim (DE) 24, 50, CP4
Bristol Myers Squibb (USA) 63, 64
Brooks Life Sciences (USA) ............ 53
Byondis BV (NL) 22
C
Captain T Cell GmbH (DE) 41
Celanese (NL) ...................... 16
Clean Biologics Group (F) 53
Concept Heidelberg |
PharmaLabCongress 2025 (DE) 38, 39
Curevac N.V. (DE) 63, 64
Curie.bio (AT) 65
Cytovation ASA (NO) ............. 24, 61
D Danaher Group (USA) 53
Danish Centre for AI Innovation 60
DASGIP an Eppendorf Company (DE) .... 11
Dr. Reddy's Laboratories Ltd. (IND) 60
E
EBD Group | BIO-Europe 2025 (CH) 36, 37
Eli Lilly & Co Ltd (USA) ........... 62, 63
Elicit Plant (F) 82
ELRIG | Drug Discovery 2025 (GB) 32, 33
ERBC (F) 67
European Biotechnology Network (DE) 23
Export and Investment Fund of Denmark 61
F
Ferrer Internacional (ES) 66
FGK Clinical Research GmbH (DE) 27, 55
Fiabila (F) 16
Fördergesellschaft IZB (DE) ......... 23, 28
Forbion Capital Partners (NL) 63
G
GEA New Food (DE) 57, 58 Green Alchemy ApS (DK) 61
H
H. Lundbeck A/S (DK) 60
HealthCap IX (SE) 61
Heidelberg Pharma AG (DE) 24
Heura Foods (ES) 82
HutanBio Ltd. (GB) 45
I IBA Lifesciences GmbH (DE) 47
Iksuda Therapeutics Ltd. (GB) 24
Illumina Inc. (USA) 74
Immunic Therapeutics AG (DE) ...... 18, 22
Inflarx NV (DE) 24
InstaDeep (GB) 63
Institution of Chemical Engineers (GB) 43 ions.bio (DE) 28
Iron Wolf Captial (LT) 69
J/K
Jeito Capital (F) 63 KelpEat srl. (IT) 67
Kemin (BE) 16
Keysight Technologies (USA) ........... 53
Körber AG (DE) 53
Kurma Partners (F) 67
L/M
Landmark Bioventures AG (CH) 62
Lifeline Ventures (FI) 60
Mabswitch Inc. (USA) 73
Marinomed Biotechnologie GmbH (AT) 65
Massalia Therapeutics SA (F) 62
Menarini Biotech S.r.l. (IT) 67
Merck & Co (USA) 64
Millow AB (SE) 61
Molecular Devices Ltd. (USA) 53 Molecular Health (USA) .............. 65
Molecular Partners AG (CH) 64
MoonLake Therapeutics (CH) 64
mts med-tech supplies (DE) 45, Supplement
MyoPax ApS (DK) 41
MyxoTech GmbH (DE) 25
NEC Corp. (JP)
Richter BioLogics GmbH (DE) CP3 Roche AG (CH) ............... 22, 55, 74 Ryvu Therapeutics zoo (PL) 69
Schroder Investment S.A. (DE) ......... 63 Seroba Life Sciences (IRL) 63 Shimadzu Europa GmbH (DE) CP2
SLAS Society of Laboratory Automation and Screening (USA) 70-72, 77
Partners (F) 17
(CZ) 69
Oy (FI) 60
(DE)
(GB) .......... 63
(GB) 4-5
Tenthpin Solutions AG (CH) 9, 25-27
ThirdLaw Molecular (USA) 70, 72
TILT Biotherapeutics Ltd. (FI) 60 TIRmed Pharma (SE) .............. 53, 61 Tolremo Therapeutics AG (CH) 22 Transgene SA (F) 22
GmbH (AT) 43, 47
Wildcat Laboratory Solutions Ltd. (GB) 71 WU Wirtschaftsuniversität Wien (AT) 52, 59
25.09.2025 NUREMBERG The technology platform for the pharmaceutical and life science industry is the international trade fair for technologies for processing powders, solids and liquids in Europe. It is now also dedicated to the GxP-compliant production of liquid, semi-solid and solid pharmaceuticals, in which solid products play a major role. www.powtech-technopharm.com

8.-10.9.25
ECCE/ECAB – Engineering for a Sustainable World: Summit for Clean Technologies & Brighter Legacy, Lisbon (PT)
Info: Matthias Neumann, DECHEMA https://ecce-ecab2025.eu/
16.-18.9.25
ILMAC 2025, Basel (CH) Info: MCH Group https://www.ilmac.ch/de
17.-18.9.25
Life Sciences Baltics, Vilnius (LT) Info: Life Science Baltics https://lifesciencesbaltics.com/
24.-25.9.25
Future Food-Tech, London (UK) Info: rethink https://www.phacilitate.com/ advanced-therapies-europe/
30.9.-2.10.25
Festival of Biologics 2025, Basel (CH) Info: Terrapinn https://www.terrapinn.com
7.-9.10.25
BIOSPAIN 2025, Barcelona (ES)
Info: ASEBIO https://asebio.com
12.-14.10.25
World Health Summit 2025, Berlin (DE)
Info: WHS Foundation GmbH www.worldhealthsummit.org
13.-14.10.25
Nordic Life Science Days 2025, Gothenburg (SE) Info: Olivier Duchamp, SwedenBio www.nlsdays.com
17.-21.10.25
ESMO Congress 2025, Berlin (DE) Info: European Society for Medical Oncology – ESMO https://esmo.org
21.-22.10.25
Drug Discovery 2025, Liverpool (UK) Info: ELRIG https://elrig.org/
23.-25.10.25
3 rd BIOTECH SUMMIT AUSTRIA, Graz (AT) Info: BIOTECH AUSTRIA www.biotech-summit-austria.com
27.-29.10.25
World Orphan Drug Congress 2025, Amsterdam (NL) Info: Terrapinn https://www.terrapinn.com/
28.-30.10.25
CPHI Europe 2025, Frankfurt am Main (DE) Info: Informa https://www.cphi.com/
28.-29.10.25
Labotec 2025, Parma (IT) Info: Koeln Parma Exhibitions Srl www.labotec.one
3.-7.11.25
BIO-Europe 2025, Vienna (AT) Info: EBD Group https://informaconnect.com/bioeurope/
14.10.2025 VIENNA The renowned international aws Best of Biotech competition aims to turn groundbreaking ideas in the life sciences into successful innovations. This year, the award celebrates its 25th anniversary and honours the best business ideas in biotechnology and medtech once again.
https://www.b2match.com/ e/bestofbiotech

11.-13.11.25
PEGS Europe 2025, Lisbon (PT) + digital Info: Cambridge Innovation Institute CHI https://www.pegsummiteurope.com/
17.-20.11.25
MEDICA und COMPAMED 2025, Düsseldorf (DE)
Info: Messe Düsseldorf GmbH www.medica.de
05.–06.02.2026 MUNICH DxPx is one of the leading conferences for investors and industry partners who share a passion for diagnostics, digital health, precision medicine and life science tools. The international partnering conference offers an ideal networking platform.
https://dxpx-conference.com/

24.-26.11.25
PharmaLab-Kongress 2025, Düsseldorf/Neuss (DE)
Info: CONCEPT Heidelberg GmbH https://www.pharmalab-congress.com
2.12.25
Pharma Outsourcing – Find the right partner, Stockholm (SE)
Info: Maria Eriksson, Life Science Sweden http://www.pharmaoutsourcing.eu/
4.12.25
Redefining Every Stage of Investment (RESI) London 2025, London (UK)
Info: Life Science Nation https://resiconference.com/
7.-11.2.26
SLAS2026, Boston (USA)
Info: Society for Lab Automation and Screening – SLAS, https://www.slas.org
24.-27.3.26
Analytica 2026, Munich (DE)
Info: Messe München GmbH www.analytica.de
BIONTECH SE Instead of fighting in court, a company takeover can also ensure peace. This was the case with the acquisition of German competitor Curevac NV for around US$1.25bn (see p. 65), which had insisted on several mRNA patents. Now the patent attorneys have a little less to do.
AMPLIFYBIO LLC in Ohio has closed its doors after burning through US$200m in four years, with its speciality as CGT CDMO. Founded in 2021 with substantial backing from the likes of Viking Global and JD Vance’s Narya fund, AmplifyBio had secured partnerships until December and boasted a cutting-edge facility. However, none of this mattered when early-stage CGT funding dried up. ■
I’m in biotechnology because

NICOLAS SORDET, CEO, Afyren SA, Clermont-Ferrand/Lyon (F)
“... I wanted to build something with purpose – leveraging unique technology to create sustainable, circular solutions that cut fossil resource reliance for a healthier planet.”
According to a survey by GlobalData plc, the global cell and gene therapy (CGT) market is poised for exceptional growth at a compound annual growth rate of 44%, with sales projected to rise from US$8.7bn in 2024 to US$76.03bn in 2030, nearly tenfold in less than a decade. Significant advancements in treatments for gastrointestinal (GI) disorders are anticipated, driven by innovations from both large and small pharma companies, says GlobalData.

There is probably a phrase for “losing your way” in every language in the world. Setting a spatial goal and achieving it has probably been an archaic matter since cavemen. The linguistic transfer to idealistic goals only came later. Nowadays, in the age of anti-Social Media and Artificial Intelligence, this focus on survival still applies. In 2025, however, we unfortunately have to realise that almost all of
(media-covered) humankind has lost its way. A few old, stupid, religiously obsessed, power-greedy and money-hungry men have shifted the focus from the urgent repair of our spaceship Earth to atavistic wars that were long considered obsolete. Now the focus is no longer on protecting the biosphere, but on invest-
ing in deadly weapons. I’m sure there’s an expression for this in every language in the world: it sucks!
B ernado Glavo
CIRCULARITY With 16 investments across ten EU countries, the European Circular Bioeconomy Fund (ECBF) has successfully mobilised substantial private capital: the ratio of capital mobilised by the ECBF to the anchor investor EIB stands at 12.3x. This is the finding of a study by the Technopolis Group, commissioned to evaluate the fund. According to the impact evaluation report, the ECBF’s market share is estimated at 15%. The study highlights the fund’s central role in the EU venture capital landscape. As one of the first VC funds dedicated exclusively to growth-stage companies in the (circular) bioeconomy, the ECBF deploys its €300m fund to a portfolio including companies such as Protix, Heura Foods, Aphea.Bio, Elicit Plant, Nuritas and Paptic. The fund addresses a critical financing gap for bio-based innovations often seen as too complex for generalist VC. ■
AUTUMN EDITION In addition to the latest reports from science, business and politics, we will be focusing on CDMOs & CROs as well as Automation and Computational Biology/AI in the next issue of EuropEan BiotEchnology magazinE. Want to participate? Just contact Oliver Schnell (-45), Andreas Macht (-35), Christian Böhm (+49-30-264921-49), or please mail: marketing@biocom.de. Publishing date is 23 October 2025; deadline for ads is 10 October 2025. ■





YOUR PRODUCT — OUR COMPETENCE AND DEDICATION FOR MORE THAN 35 YEARS!
RichterBiologicsisyourprofessionaland experiencedpartnerofferingCDMOsolutions fromgenetoproductallfromonesource.
RichterBiologics:expertforlatestage andcommercialproduction.





RICHTERBIOLOGICS
Suhrenkamp59,22335Hamburg,Germany
Phone:+494055290-801
BusinessDevelopment@richterbiologics.eu

CONTACT US TO BRING YOUR PROJECT TO SUCCESS!






