

Welcome to the data and digital health edition of INSPIRE
In this edition of INSPIRE, we journey through the rapidly evolving landscape of data and digital health — where artificial intelligence interprets complex patterns, robotics extends human capability, and digital tools connect patients and clinicians in new ways.
We also explore the deeper questions that come with progress: how to build ethical, trustworthy systems; how to harness data for population health; and how strong national infrastructure can turn information into insight. At this pivotal moment, technology, compassion, and evidence are converging to redefine what health and medical research can achieve for all Australians.
Across the pages that follow, you’ll find diverse stories that reflect the breadth of this transformation.
Initiatives such as the Zero Childhood Cancer Data Lakehouse, MERLIN, Trial Link, and ADAPt are redefining how we collect, connect, and apply health data to improve lives. From AI powered lung mapping advancing respiratory disease research, innovations in Electron Microscopy transforming rural healthcare, and wearable “pacemakers for gait” enhancing quality of life for Australians living with Parkinson’s, these projects demonstrate how innovation is harnessing advanced technology to create more equitable, life-changing health solutions.
Yet, as we embrace innovation, we must do so with caution and care. As the World Health Organization’s Director-General recently observed, “as AI becomes more sophisticated and its health applications expand, so must our efforts to make them safe, effective, ethical and equitable”. This balance between progress and protection is at the heart of this edition. Articles on privacy legislation and medico-legal complexities — including the Avant Foundation’s perspective — highlight the importance of secure systems and informed consent. Others examine how we can ensure that consumers retain agency amid the growing volumes of data collected about them.
Crucially, several contributions explore the intersection of data and equity. We are reminded that data gaps can
create blind spots: when certain groups or conditions are underrepresented in datasets, it leads to inequitable access to care and limits research. As Hepatitis Australia and Rare Voices Australia demonstrate, a nationally coordinated approach could help overcome these inequities — empowering research, improving diagnosis, and ultimately saving lives.
At Research Australia, we are working to help realise this vision. We are calling for a unified National Health and Medical Data Infrastructure Strategy — one that maps existing assets, harmonises governance, and supports coordinated investment across jurisdictions. This blueprint would enable seamless integration between research, clinical care, and system performance, ensuring Australia’s data is treated as a true national asset.
Coordinated infrastructure, underpinned by longterm, multi-party investment and harmonised governance, would strengthen productivity, reduce duplication, and accelerate research discovery and translation. It would also provide the foundation for a safe, ethical, and equitable digital health future — one that protects privacy, builds trust, and ensures that the benefits of innovation reach every community.
The stories in this issue of INSPIRE capture the energy, complexity, and promise of this moment in Australian health and medical research and innovation. Together, they remind us that the data and digital revolution is not just about technology — it’s about people, partnerships, and purpose.
And with collaboration, courage, and care, Australia can lead the world in building a health system that is both digitally advanced and deeply human.
Best wishes, Nadia and your Research Australia Team






Australian Health & Medical Research & Innovation


Targeted & Harmed

Advancing Health Equity Through Health Technology

Harnessing AI to unlock clinical insights for childhood cancer research

Publisher Research Australia Ltd
Art Direction Matthew Ware p +61 403 844 763 e matt@objktive.com
For Advertising enquiries please contact the Research Australia office on p 02 9295 8546 or e admin@researchaustralia.org researchaustralia.org


INSPIRE ONLINE issuu.com/researchaustralia
INSPIRE is a publication of Research Australia Ltd ABN 28 095 324 379 384 Victoria Street Darlinghurst NSW 2010
Who can submit articles?
Any current member of Research Australia who would like to share a relevant story that affects their organisation including, philanthropic donations and their outcomes, research findings, and any other related health and medical research topic that affects the Australian population.
Submission guidelines & deadlines For information regarding how to submit and publishing deadlines visit the Research Australia website
Disclaimer
The opinions expressed in INSPIRE do not necessarily represent the views of Research Australia. Whilst every effort has been made to ensure accuracy, no responsibility can be accepted by Research Australia for omissions, typographical or inaccuracies that may have taken place after publication. All rights reserved.
The editorial material published in INSPIRE is copyright. No part of the editorial contents may be reproduced or copied in any form without the prior permission from Research Australia. © Research Australia 2025.

CHRISTOPHE KEREBEL
My Twitter : @chriskere CHRISTOPHE KEREBEL CHRISTOPHE KEREBEL My Twitter : @chriskere
HARRY PERKINS INSTITUTE OF MEDICAL RESEARCH
Australian Health & Medical Research & Innovation










CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
CURTIN UNIVERSITY
Australian Health & Medical Research & Innovation

58
CURE Asthma
ASTHMA AUSTRALIA

60 Mathematical modelling shapes global policy to eliminate malaria THE KIDS RESEARCH INSTITUTE AUSTRALIA

62 From Data to Action
DIGITAL HEALTH CRC

70
Australia risks falling behind in the synthetic health data race
DIGITAL HEALTH CRC
68 How healthcare data is improving cancer diagnosis and outcomes
VCCC ALLIANCE

Zero Childhood Cancer Data Lakehouse: Enabling Scalable, Timely Insights
LUMINESCE ALLIANCE




64
Unlocking the potential of linked data for clinical trials

72
CROHN'S COLITIS CURE

New members


66
“It’s not just electronic paper” – digital health research to transform health translation
JAMES COOK UNIVERSITY

IBD-perfect It’s not magic!


CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
THE UNIVERSITY OF WESTERN AUSTRALIA
THE UNIVERSITY OF WESTERN AUSTRALIA
Events wrap-up
2025 FUTURE HEALTH LEADERS PROGRAM
In August, AbbVie and Research Australia commenced the second annual Future Health Leaders Program. Bringing together 12 incredible female Year 11 students from across Australia— finalists from an application pool of over 100 students—in Canberra for a day of inspiration, insights, and immersion into the world of health and medical research and innovation.
The day began with an inspiring panel discussion featuring Professor Rosemary Korda, Dr Kylie Jones and Professor Emily Lancsar who shared their career journeys and insights on leadership. Participants then headed to Parliament House, where they engaged with the Hon Rebecca White MP, Hon Emma McBride MP and Dr Sophie Scamps MP, and had the opportunity to meet their local members. The day wrapped up with an exciting visit to CSIRO Black Mountain, which included hands-on lab activities with CSIRO researchers.
Over October and November, the students participated in a series of virtual sessions with even more inspiring sector leaders, with the sessions focusing on clinical and research careers, global challenges in HMR, and AI and digital health.

* images used with permission
PHILANTHROPY ROUNDTABLE
In September, Research Australia held a Philanthropy Roundtable focusing on Advancing Sustainable Funding. The event explored how the sector can unlock new opportunities and approaches to secure stronger, more sustainable philanthropic funding for health and medical research, and explored current giving trends and financial stability across the charity sector.
We heard from Cate Bennett, standing in for Sue Woodward AM as Acting Commissioner of the Australian
Charities and Not-for-Profits Commission (ACNC), who shared the latest ACNC data and insights into emerging trends shaping strategic decision-making. Kathryn Borkovic, CEO of SpinalCure Australia, reflected on her organisation’s achievements and lessons learned on the challenging path to creating national impact. Marcel Svatos, General Manager of Business Operations and Philanthropy at ANZUP Cancer Trials Group, also spoke about their transformative journey in redefining the organisation’s fundraising culture.
Students at CSIRO Black Mountain
L-R Dr Sophie Scamps MP and Hon Rebecca White MP with the Future Health Leaders*
NATIONAL STRATEGY WORKSHOP
Research Australia held a virtual workshop to discuss the draft National Health and Medical Research Strategy in preparing for our submission as part of the consultation process. Attendees shared their insights and reflections on the draft Strategy, and identified gaps and shortcomings, contributing to Research Australia’s submission to the government.
Key Recommendations include:
• Clearer roles for state and territory governments and industry in funding and supporting health and medical research and innovation.
• Strengthening discovery science, backed by increased investment through the NHMRC, MRFF and ARC, while also investing in bridging gaps in translation and commercialisation.
• Stronger focus on prevention policy and investment, and a systems-wide approach to equity.
• Targeted focus on workforces such as early- and mid-career researchers, clinician researchers, lived experience researchers and priority populations as part of a National Health and Medical Research Innovation Workforce Plan.
• Greater support for “emerging technologies” beyond AI to include innovations such as quantum technologies, robotics, and synthetic biology.
• Creating an Australian equivalent to the US BARDA, the UK’s ARIA, and the EU’s HERA tasked with financing late-stage medical countermeasure development to support local manufacturing and development and build national sovereign capability.
Our full submission is available here

UNIVERSITY ROUNDTABLE
On 28 October, Research Australia and Flinders University co-hosted a University Roundtable in Adelaide, South Australia, bringing together 49 representatives from 26 universities. The session, chaired by Guest Chair Professor Billie Bonevski, explored the theme “Workforce – Looking Ahead: Shaping the Research Workforce for Tomorrow.”
The Department of Defence provided valuable insights into its research workforce and discussed opportunities for greater collaboration with the health and medical research sector. Professor Barney Glover AO, Commissioner of Jobs and Skills Australia, spoke about ongoing work in future workforce planning and shared lessons that could be applied across the research landscape.
A draft discussion paper, “Workforce – Looking Ahead: Shaping the Research Workforce for Tomorrow,” was tabled for feedback during the session. This will be shared with members once finalised.
University
NATIONAL STRATEGY DATA ENABLER WORKSHOP
Research Australia, in partnership with the National Health and Medical Research Strategy Secretariat and Digital Health CRC convened a workshop to identify practical and implementable changes the national strategy can action to build capacity and capability in data assets and digital technologies that are accessible, interoperable and linked. Participants from across the health and medical research and innovation ecosystem discussed a range of ideas focusing on data infrastructure, workforce, governance and regulatory systems as well as community engagement - from ethics, social license and trust.
Attendees at Research Australia’s University Roundtable hosted at Flinders
DATA AND DIGITAL HEALTH
The Backbone of Australia’s Health and Medical Research and Innovation Future
In 2025, the importance of digital health and health data is no longer a future aspiration – it is an urgent national imperative. Across the health and medical research and innovation sector, data underpins every discovery, every innovation, and every patient outcome.
It drives precision medicine, fuels preventive care, and enables the translation of research into realworld solutions. It is also the gateway to transformative advanced technologies which will set the trajectory of the sector for generations to come – not only the adoption of generative artificial intelligence, which has seen a lot of attention just this year but innovations such as quantum computing and genomics, to name just a few.
Yet, while Australia’s researchers and clinicians produce world-leading science, they continue to be hampered by fragmented systems, inconsistent data access, and a lack
of coordinated national leadership. As the national peak for health and medical research and innovation, Research Australia has consistently asserted that health data is one of the nation’s most valuable assets, and digital health is a critical aspect of the systems and infrastructure that collates, stores and transforms it into impact.
This position has been powerfully reinforced by our members and stakeholders across various channels, in all our webinars, symposia and workshops, including those we have hosted in collaboration with the Digital Health CRC throughout 2025. We have been proud to bring together representatives from government, academia and industry to articulate, ideate and progress what is needed to ensure this critical national asset can be utilised for the future health and wealth of all Australians.
2025: CENTRING DATA AND DIGITAL HEALTH IN ALL REFORMS
Few years have seen such opportunity to reimagine data and digital health policy as 2025. Anchored by the draft National Health and Medical Research Strategy, the Strategic Examination of Research & Development (SERD)
and the development of the 2026 National Research Infrastructure Roadmap, the year marked an important opportunity to realise the potential of Australia’s health and medical research and innovation landscape. Within the National Strategy, Data [& Advanced Technology] formed 1 of 4 key ‘Enabling Initiatives’ – a move which was strongly endorsed by Research Australia and reflects the critical role of data and health technologies in catalysing discoveries and advancing healthcare delivery. The SERD process amplified this further, calling for improved access to research to generate new growth opportunities, including through AI, enhanced databases, and better researcher access. Equally, the 2026 National Research Infrastructure Roadmap issues paper sets out suggestions for boosting Australia’s data capabilities, particularly within the Aboriginal and Torres Strait Islander knowledge systems, Humanities and New research infrastructure topics.
Furthermore, this year has seen a strong national focus on productivity – with the Productivity Commission undertaking 5 inquiries, including a dedicated pillar on Harnessing data and digital technology. The advent of a

permanent Australian Centre for Disease Control (CDC) has further embedded the momentous role of robust data collection and analysis to protect the nation from future health threats and ensure health system resilience. Across our policy submissions to the SERD,
Research Australia has maintained a clear line of sight on the imperative role of data and digital health, which must be supported through:

• Coordinated national stewardship
• Sustainable, smarter investment
• Interoperable infrastructure
• Person-centred governance and cohesive regulatory frameworks
• Prioritisation of ethics and social license
• Workforce and skills development
Around 30% of the world’s data is healthcare data, with some estimates indicating up to 97% of this data is going unused1. By taking bold action, Australia can unlock the vast and currently untapped potential of this critical national asset to effect transformative change for the nation’s health and prosperity.
LOOKING AHEAD: THE ROLE OF RESEARCH AUSTRALIA
Data, supported by digital health systems and technologies are the connective tissue of Australia’s future health and medical research and innovation system. They are enablers of innovation, collaboration, and equity. But realising their potential requires more than statements of intent; it demands leadership, coordination and investment.
As 2025 draws to a close, Research Australia continues to play its convening role –bringing together government, industry, academia, and communities to advance a shared national vision. As we approach the finalisation of the National Strategy and SERD before year-end, we stand at a precipice to put data and digital health at the heart of Australia’s health and medical research and innovation agenda. Our message has and will always remain clear: data is not just a by-product of research – it is its foundation – an asset that directly contributes to a healthy nation, and a healthy economy.
1 Forbes. (2023). ‘What To Do About Healthcare’s ‘Messy Desk’ Data Dilemma’. Accessed at: https://www.forbes.com/councils/ forbestechcouncil/2023/12/12/what-to-do-about-healthcaresmessy-desk-data-dilemma/
Authors: Max Chester, Policy Officer and Dr Talia Avrahamzon, Head of Policy, Projects & Advocacy at Research Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

ADVANCING HEALTH EQUITY THROUGH HEALTH TECHNOLOGY: INGHAM INSTITUTE’S PERICH CENTRE
Ingham Institute for Applied Medical Research in Liverpool NSW has launched its new Centre for Robotics & Health Technology – the Perich Centre – a multidisciplinary hub dedicated to ensuring health innovation is developed and deployed with equity at its core.
The Centre will serve as a flagship site for research and development, teaching, and advanced training in healthcare automation, with a focus on technologies that can transform care delivery while addressing health disparities.
Launched in September at a ceremony in Liverpool by Minister for Medical Research the Hon. David Harris MP, the hub was described as an ‘automation imaginarium’ — a place where robotics, digital tools, and artificial intelligence are critically evaluated and adapted to serve diverse populations.
Minister David Harris MP said, ‘Access to life-changing innovation should be for everyone, regardless of their background or circumstances. With its focus on impact,



inclusion, and collaboration, this latest development by Ingham Institute is set to become a national and global leader in healthcare automation.’
EQUITY AS A DESIGN PRINCIPLE
For Dr Glen Schlaphoff, clinical leader of the Centre, its work is guided by a clear principle: equity. ‘Without ethical engagement, community design and strong clinical leadership, healthcare technologies can actually widen health gaps, rather than reducing them,’ he noted. ‘At Ingham Institute, “tech-quity” drives everything we do — from early-stage innovation to large-scale adoption.’This perspective informs a wide-ranging research agenda. Current projects include wearable devices to support maternal and women’s health, rehabilitation robotics
The launch of the new Petrich Centre

for acquired brain injury, AI-driven tools for cancer and stroke treatment, and digital solutions for chronic disease prevention and management. Each initiative is developed with a strong emphasis on inclusion, ethics, and clinical oversight to ensure technologies improve access and outcomes for the populations who need them most.
LOCAL RELEVANCE, NATIONAL SIGNIFICANCE, GLOBAL IMPACT
The Ingham Institute’s location in South Western Sydney is significant. This region is one of the most culturally diverse in Australia and faces considerable health inequities. For the Perich family, who have long supported the Ingham Institute, this makes the area an ideal place to pioneer change.
‘If we develop solutions that work here — that are inclusive, ethical, and evidence-based — we can make a difference globally,’ said Mark Perich.
By focusing on technologies that are responsive to the realities of local communities, the Centre seeks to generate evidence and approaches that can inform both national and international health systems.
MULTIDISCIPLINARY RESEARCH AND TRAINING
The Perich Centre brings together more than 60 clinicianscientists across nursing, midwifery, allied health, and surgical and medical specialties. The breadth of expertise enables a deeply multidisciplinary approach to innovation, where technologies are evaluated not only for technical feasibility but also for clinical relevance, ethical considerations, and system-level integration.
AREAS OF FOCUS INCLUDE:
• Remote diagnostics, prognostics, therapeutics and monitoring to improve access for people in rural and regional areas
• Smarter hospital systems to streamline workflows and support frontline staff
• Allied health–led rehabilitation robotics and gamified therapies
• AI and machine learning applications in cardiology, oncology, neurology, and stroke
• Cost-effectiveness studies of surgical robotics
• Advanced immersive training for future health professionals.

SUSTAINING HEALTH SYSTEMS THROUGH INNOVATION
The establishment of Ingham Institute’s Centre for Robotics & Health Technology is the culmination of sustained vision and advocacy by the Ingham Institute Board and Institute Director Professor Les Bokey. For Professor Bokey, the responsible adoption of automation is essential to the sustainability of healthcare systems.

‘The considered and correct adoption of automation in health is not just a moral imperative,’ he said. ‘It is how we create sustainable health systems, improve outcomes across diverse populations, and unlock the full potential of technology for global health impact.’
‘Equitable introduction, assessment and acceleration of automation in health is as diverse as it is critical,’ emphasises Ingham Institute CEO Simone Proft. ‘Our work pioneering “tech-quity” means:
• Wearable devices that monitor your health from home – wherever home is;
• Advocating for personalised cancer care for refugee and migrant populations;
• Eradicating gender referral bias in access to robotic surgery;
• Applying proven technologies from industries like mining and manufacturing to advance health outcomes for priority populations;
• World first MRI-guided therapies, performed in a public hospital.’
A NEW ERA FOR HEALTH TECHNOLOGY
The launch of the Perich Centre marks a significant milestone in the evolution of health technology research in Australia. By combining strong community roots with a global outlook, the Centre is positioned to lead in the development of technologies that are both cutting-edge and equitable. Its work demonstrates that innovation and equity are not competing priorities but interdependent goals that, together, can redefine the future of healthcare.
Author: Ingham Institute for Applied Medical Research was established to ‘Inspire Health and Transform Care’, starting in south-western Sydney. Its new Perich Centre opened in September 2025.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

SILVER LININGS IN THE DIGITAL CLOUD

How artificial intelligence can make healthcare more human, not less.
THE INFORMATION PARADOX
Our world is generating more data than ever – yet, the everyday person’s understanding of health and medical information remains low. A previous survey by the Australian Government reports that 60% of Australians do not understand their own healthcare, contributing to patient frustration, poorer co-management and health outcomes, widened equity gaps, and higher health system costs. Take a few steps in the shoes of a person living with chronic disease: you receive a new diagnosis in hospital, explained to you on a ward round; get discharged from hospital with a discharge summary and results written in jargon; attend a fifteen-minute follow-up appointment with a new doctor in three months’ time; leave with a script for two new medications and a referral back to your GP, who is booked out for the next six weeks. How do you make sense of it all? Most people choose between Google and giving up – but it doesn’t have to be that way. The space between points of contact with our health system is a window of opportunity to bridge patient care. People already have health documents in their hands – now, they need the tools to understand them.
SHIFTING TOWARD COLLABORATIVE CARE
People are not passive recipients of care – they are active participants in the understanding, decision-making, and co-management of their health. Shared care models should prioritise being informed and educated as an endpoint for successful healthcare delivery. Naturally, these endpoints will translate over time into traditional outcomes – such as the uptake of preventative health measures (e.g. immunisation and cancer screening), adherence to treatment plans and medications, reduced preventable re-hospitalisation, and slowed disease progression – in a way that empowers patients to be at the centre of their health journey. Moreover, we need to prioritise equity: general and health literacy, cognitive load, stress, language barriers, cultural context, and socioeconomic determinants all shape health communication and outcomes. It is vital that our interventions are designed to reach people who need them most.
REIMAGINING THE USE-CASE FOR AI
What if artificial intelligence wasn’t used to accelerate speed and efficiency, but for its slowness, patience, and depth of care? In our busy health system – which already fares better than many countries – clinicians are timepoor, juggling high patient loads, administrative burden, billing models, and siloed teams. Activities such as quality patient counselling, literacy- and relationship-building, and health promotion are all but invisible in our performance indicators. While there has been an explosion of interest in clinician-facing AI technologies, particularly in the

realm of diagnostics, there is an untapped goldmine of applications in patient-facing tools. We should not view AI as a replacement for clinicians; rather, the health system of the future should utilise digital and AI technologies to complement and strengthen clinician-led care. This means taking ownership of AI development in a regulated and ethical manner, developing policies and guardrails for patient-centred AI use, and designing AI as a companion to optimise the value of patient-clinician interactions. In this way, we harness AI’s natural strengths to provide continuously accessible, reiterative, empathetic and personalised care.
CO-DESIGNING TRUST IN DIGITAL TOOLS
In big data, what we build reflects history: an amalgamation of what we know – or rather, what we have measured – to date, propelled forward into new predictions and permutations. It is up to human designers to bring intention, governance, and oversight to this process. One way to build trust into digital tools is to adopt a co-design approach with health consumers. This circular process brings lived experience to digital products, and uses them to inform interface design, functionality, safety, algorithmic testing/outputs, and value-add. It is crucial that gaps in inputs and outputs are identified by an overarching framework, and that our tools address – and do not reinforce – health disparities. The process should be repeated with a focus on each target community; for instance, rural or specific cultural groups. Furthermore, co-design needs to occur between scientists, policymakers, and industry. As AI evolves, clinical experts should stand alongside health consumers in advocating for its responsible use to advance health outcomes.
FUTURE ROADS
We are at a turning point – not just in how we treat disease, but how we communicate about it. Embracing these principles takes us one step further toward designing a patient-centred health system in the era of artificial intelligence – one where healthcare delivery is not a once-off consult, but a lifelong conversation.
Author: Dr Sara Sukudom is a Basic Physician Trainee and Project Lead at Orva, an initiative to develop patient-facing AI that provides reliable, on-demand personalised health education and counselling. Her research team includes Dr Julien Flack and Professor Girish Dwivedi Team affiliations include Harry Perkins Institute of Medical Research and the University of Western Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

TARGETED & HARMED
AI Reveals the True Cost of Online Marketing to our children
DIGITAL MARKETING IN THE AI ERA
Children and youth are increasingly immersed in digital environments. In 2024, 97% of Australian adolescents owned a personal screen-based device, and 84% of children aged 8–12 engaged with social media or messaging platforms. According to the Australian Bureau of Statistics, children and youth spend up to 20 hours online weekly. Each digital interaction contributes to extensive data profiles, with estimates suggesting that by age 13, platforms may have collected up to 72 million data points per child. Our research show that Artificial intelligence (AI) facilitates the rapid analysis of these profiles, enabling highly personalised `eting strategies that exploit individual vulnerabilities to maximise engagement and profit. An Australian study found that teenagers encounter an average of 168 online food promotions weekly, with 99.5% advertising unhealthy products. Evidence links such exposure to increased consumption and sustained use into adulthood. However, the personalised nature of digital marketing presents significant challenges for monitoring and regulating the content young people are exposed to.
FLIPPING THE SCRIPT – AI FOR GOOD
Using the power of AI, our team has developed SCANNER, the world’s first AI-enabled system designed to detect harmful online marketing practices. SCANNER can detect more than 500 brands across unhealthy foods and beverages, alcohol, gambling, vaping and tobacco products and infant formula at an accuracy greater than 85% in videos and images. The benefit of SCANNER is that it reduces human involvement in data processing and increases the speed of analysis by more than 20 times compared to manual methods. SCANNER is the result of a unique interdisciplinary collaborative project between the Global Centre for Preventive Health and Nutrition (GLOBE) and the Institute for Intelligent Systems Research and Innovation (IISRI) at Deakin University. This innovative partnership combines the expertise of public health policy researchers from GLOBE with machine learning researchers from IISRI in the development and application of a system that facilitates governments to monitor accountability and compliance of existing regulations related to the marketing of harmful products in a low-resource, novel manner.
We used SCANNER in our #DigitalYouth project, the first study to comprehensively measure children and youth’s (aged 8 to 25 years old) ‘actual’ exposure to harmful digital marketing globally. In the #DigitalYouth project, we collected online screen recordings from more than 300 children and youth in Australia and used SCANNER to analyse the data to understand their exposure to harmful online marketing. Findings from the pilot study show that, on average, children aged 8–13 were exposed to about 13 junk food marketing daily. Teenagers (14–17) saw at least one gambling ad, six alcohol ads, and 24 junk food ads per day. Young adults (18–25) encountered two instances of gambling marketing, seven alcohol ads, and 23 junk food ads each day online. These advertisements often include prompts such as ‘Learn

more’, ‘Install now’, ‘Buy now’, encouraging engagement from children and youth and enabling a seamless process of exposure to purchase without needing to move away from the screen. Initial findings from the #DigitalYouth study are concerning, showing that more than 95% of paid food advertisements seen by children and teenagers promote unhealthy products.
PROTECTION FROM ONLINE MARKETING HARMS
Children have the right to participate online while being protected from harm. The UN Convention on the Rights of the Child (CRC) affirms their rights to access information (Article 17), health (Article 24), privacy (Article 16), and protection from economic exploitation (Article 32). General Comment No. 25 (2021) confirms these rights apply in digital contexts, requiring safe access and protection from online harms. Data-driven marketing of unhealthy products undermines these rights by exploiting developmental vulnerabilities and promoting harmful norms. As a CRC signatory, Australia is legally obligated to protect children from such practices. Our research shows parents are aware of these risks but feel powerless to intervene, especially as children age. They strongly support government regulation to prevent online targeting of children.

FOR A HEALTHIER AND SAFER DIGITAL FUTURE
Governments in Australia and globally are beginning to recognise the urgent need to regulate harmful digital marketing and protect children from online harms. Recent actions include new vape and tobacco laws and restrictions on online unhealthy food advertising in the UK and Norway. While promising, these measures must go further to balance protection with participation. The World Health Organization advocates for comprehensive global regulation of digital marketing of unhealthy products across all digital platforms, urging legal accountability, transparency, and enforcement. Comprehensive policies must protect children’s rights, ensure safe digital spaces, and include cross-border cooperation and independent monitoring systems.
Our research shows that AI can be harnessed for public good. Our AI tool SCANNER offer governments and regulators the ability to detect and track harmful marketing practices in real time, helping to create healthier online environments that uphold the rights, safety, and wellbeing of children and young people.
of Health and Social Development/Institute for Health Transformation, Deakin University.
Authors: Dr Navoda Liyana Pathirana, Christine Driessen, Dr Florentine Martino, Dr Ana Paula Cardoso Richter and Professor Kathryn Backholer, Faculty of Health/School
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

HARNESSING AI TO UNLOCK CLINICAL INSIGHTS FOR CHILDHOOD CANCER RESEARCH
Automatically extracting structured data from clinical reports is vital but challenging, given unstructured text, inconsistent formats and complex medical language.



At Children’s Cancer Institute, the Computational Biology team , supported by Luminesce Alliance , is using Artificial Intelligence to semi-automate data extraction and verify cliniciancurated information, improving research quality and accelerating progress in paediatric precision medicine.
THE PROBLEM: FRAGMENTED CLINICAL DATA
Clinical reports often contain vital information about a patient’s diagnosis, treatment, and response to therapy. However, these reports are typically stored as scanned PDFs, vary widely in format across institutions, and include handwritten notes, checkboxes, and other non-standard elements. For researchers, accessing and interpreting this data is a time-consuming and error-prone process that requires scarce medical expertise and meticulous manual effort. Typos, misinterpretations, and inconsistencies can undermine data quality, limiting its usefulness for research and precision medicine.
THE SOLUTION: AI-POWERED AUTOMATION
To overcome these barriers, the Computational Biology team at Children’s Cancer Institute (CCI) is developing an AI workflow that leverages Large Language Models (LLMs) to extract structured, research-ready data (such as diagnoses, treatments, outcomes) from free-text clinical reports. This initiative is part of the ZERO Childhood Cancer Program, which aims to improve outcomes for children with cancer through precision medicine.
Supported by Luminesce Alliance, the team has built a secure, cloud-based environment and defined compliant use cases for processing de-identified patient reports. The proof-of-concept workflow includes several key steps: redaction of sensitive information, secure storage in Azure Data Lake, image-to-text conversion using Azure Document Intelligence, and AI-based information retrieval via OpenAI’s LLM services.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
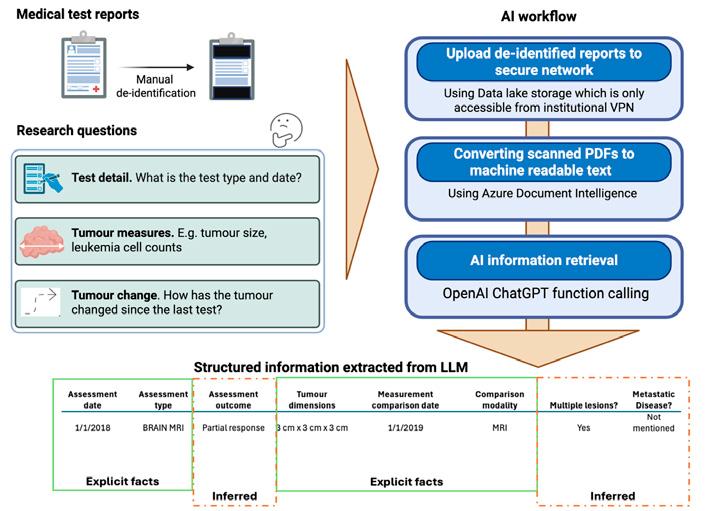
MAKING THE PATIENT JOURNEY VISIBLE TO RESEARCHERS
A key impact of this work is enabling researchers to interpret and analyse the disease journey of each cancer patient. By converting unstructured clinical notes into structured data tables, the AI system enables researchers to quickly understand treatment protocols, disease progression, and patient responses. This visibility is crucial for identifying patterns, evaluating treatment efficacy, and developing new therapeutic strategies.
LLMs are particularly well-suited to this task because they can process complex medical terminology, adapt to diverse report formats, and generate accurate summaries. By automating the extraction of critical details, such as tumour size, presence of metastases, and treatments, LLMs remove a major bottleneck in paediatric oncology research.
PILOT STUDY: DESIGN, SAFEGUARDS, AND EARLY RESULTS
To evaluate the feasibility of this approach, the team conducted a pilot study using a benchmark dataset of 11 representative clinical reports from a cohort of 168 patients. These reports were selected for their complexity and potential to challenge AI-based extraction, including low-resolution scans, misoriented pages, handwritten annotations, and diverse formatting styles.
Each report was manually de-identified by two reviewers to ensure patient privacy. The redacted reports were then uploaded to a secure Azure Data Lake, accessible only within the CCI network and protected against external threats. Using Azure Document Intelligence, the scanned PDFs were converted into machine-readable text. This text was then analysed by OpenAI’s LLM to extract both explicit facts and inferred clinical information, which was organised into structured tables for evaluation.
CCI pilot study uses Large Language Model Artificial Intelligence (LLM AI) to automate data extraction
To validate the AI’s performance, the extracted data was compared against a “ground truth” established by expert scientists through manual review. The results were promising: the PDF-to-text conversion was highly accurate, even for handwritten content, and the LLM reliably extracted explicit facts such as test names, dates, and numerical results.
ADDRESSING CYBERSECURITY AND COMPLIANCE
Given the sensitive nature of medical data, cybersecurity and legal compliance were central to the project’s design. The CCI Cybersecurity and Legal teams conducted a thorough review of the workflow, including the Azure Data Lake environment and the integration of AI services. Their assessment confirmed that the system met institutional standards for data protection and legal compliance, leading to formal approval for use with redacted clinical reports.
CHALLENGES AND AREAS FOR IMPROVEMENT
While the pilot study demonstrated accurate extraction of explicit facts, the AI system faced challenges with some clinical interpretation and cross-document synthesis. For example, the LLM occasionally failed to infer whether a patient had locally spread tumours or metastatic tumours.
To address these limitations, the team plans to enhance the AI’s capabilities using clinician-labelled training data and refined prompt engineering. Additionally, the redaction process will be streamlined using a combination of AI and human review, and larger validation datasets will be used to improve data quality assurance.
LOOKING AHEAD: SCALING FOR IMPACT
The next phase of the project involves obtaining full institutional approval to apply the workflow to future patient reports. By incorporating feedback from clinicians and expanding the dataset, the team aims to further improve the accuracy and reliability of AI-based data extraction. This will enable faster, more scalable access to high-quality clinical data, accelerating research and supporting the development of personalised treatment strategies.
CONCLUSION
This pilot study marks a significant step forward in the use of AI for clinical data extraction in paediatric oncology. By demonstrating the feasibility of LLM-based automation in a secure and compliant environment, the Computational Biology team at CCI has laid the groundwork for a scalable solution that can transform how researchers access and use clinical information. Ultimately, this innovation has the potential to remove a major barrier to high-quality research, paving the way for more effective and personalised treatments for children with cancer.




Authors: Dr Wenhan Chen is a Senior Bioinformatician at Children’s Cancer Institute and Adjunct Associate Lecturer at The University of New South Wales. His research focuses on improving childhood cancer care by developing liquid biopsy, a minimally invasive molecular test to track cancer over time to guide precision treatment. Associate Professor Mark Cowley is Deputy Director (Enabling Platforms and Collaboration) at Children’s Cancer Institute. He holds several leadership positions, including Head of the Luminesce Alliance Data Enabling Platform, co-Head of the ACRF Childhood Cancer Liquid Biopsy Program, and President of Australasian Genomic Technologies Association (AGTA). Article submitted by Luminesce Alliance.
Dr Wenhan Chen
Associate Professor Mark Cowley
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
PRECISION HEALTHCARE
INGHAM INSTITUTE FOR APPLIED MEDICAL RESEARCH AND LIVERPOOL HOSPITAL PIONEER RESEARCH IN REMOTE ROBOTIC THERAPEUTICS
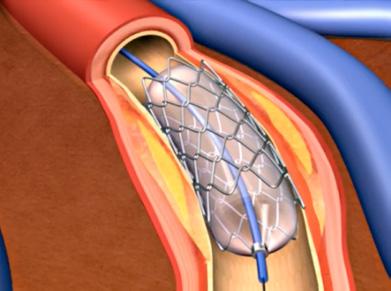
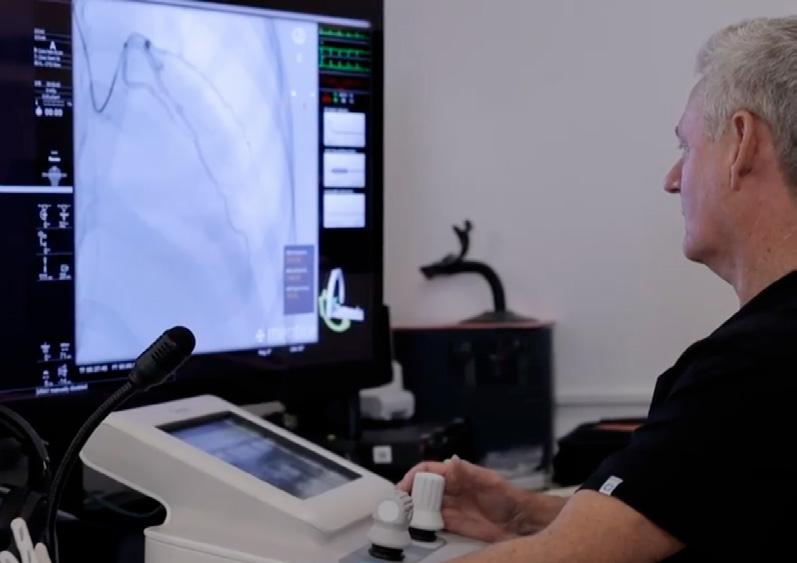

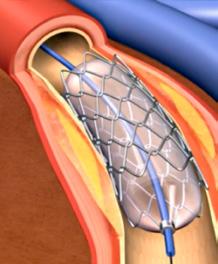
A world-first clinical trial of robotic cardiac technology
In a medical emergency like a heart attack, every second counts. The faster a patient receives treatment, the better their chance of survival and recovery.
A groundbreaking NSW-first clinical trial at Liverpool Hospital has tested the effectiveness of robot-assisted stent procedures compared to traditional manual methods. The randomised controlled clinical trial was run in partnership with the Ingham Institute’s Centre for Robotics and Health Technology (Perich Centre).
Stents, or Percutaneous Coronary Intervention (PCI), is a common, lifesaving procedure for people experiencing coronary heart disease, a condition that causes more than one in ten deaths across Australia. Using small wires, balloons and stents, PCI clears blockages in the arteries and restores blood flow to the heart.
The PARTY Trial (Percutaneous coronary intervention using Assisted Robotic Technology) was led by Professor Craig Juergens and Dr James Leung, from the Ingham Institute and Liverpool Hospital, and compared traditional PCI methods with robotic interventions. The 148-patient trial was conducted under strict clinical guidelines, with every robotic procedure supervised by experienced doctors.
‘Robotic technology is paving the way for a new standard in cardiac procedures, offering the opportunity to enhance patient care through robotic precision and advanced tools for these life-saving procedures,’ says Prof Craig Juergens.
Ingham Institute for Applied Medical Research was established to ‘Inspire Health and Transform Care’, starting in south-western Sydney.
Ingham Institute’s research is shaped by the unique health needs of its local population which is diverse and rapidly growing. It’s also home to many priority populations, which encounter systemic barriers that exacerbate disparities in access to healthcare services, nutritious food, and health living environments resulting in poorer health outcomes.

While robot-assisted PCI procedures are still in their infancy, trials like PARTY are helping clinicians understand the potential long-term benefits including:
• Improved procedural precision and outcomes: Robotic systems offer enhanced dexterity, vision, and precision, which can reduce complications and promote faster recovery. This is particularly valuable in settings with limited post-operative support.
• Decreased risk of clinical complication and potential to reduce logistical delays associated with patient transfer, as well as reduction in strain on ambulance services when and where remote access is feasible.
• Reduction in radiation exposure for patients and clinicians.
• Remote robotic-assisted procedures for rural and regional patients to receive treatment where they live via enhanced telemedicine.
The cardiac robot used in the trial was generously funded through a philanthropic donation from the Perich family.
INGHAM INSTITUTE’S REMOTE ROBOTICS RESEARCH
Remote cardiac intervention is one facet of the Ingham Institute’s research and clinical trials in robotically-enabled remote therapeutics.
Ingham Institute’s Centre for Robotics and Health Technology, the Perich Centre, opened in September. The Centre is dedicated to TECH-QUITY – pioneering global health equity through health technology.
In cancer, cardiovascular, diabetes, neurology, paediatrics and stroke, research is directed to improving barriers to access and levelling disparity in health outcomes.
Like Prof Juergens and Dr Leung who led this trial, the majority of the Institute’s affiliates are clinicians practising in a major tertiary hospital right across the road, so research is set up to rapidly translate into real life impact. And because south western Sydney is, demographically speaking, a microcosm of the world, findings offer the potential of global application and impact.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
AI-POWERED LUNG MAP DETECTS PULMONARY
FIBROSIS
BEFORE DAMAGE APPEARS
Scientists have created the first detailed map of individual cells in the adult human lung, revealing their location, identity, and gene activity.


Visualisation of the cellular atlas developed by Associate Professor McCarthy’s team
Associate Professor Davis McCarthy, head of SVI’s Bioinformatics and Cellular Genomics Lab

Using spatial transcriptomics, the atlas includes both healthy and diseased tissue, offering groundbreaking insights into lung function and chronic diseases like idiopathic pulmonary fibrosis, marking a major advance in respiratory disease research.
An international team of researchers has, for the first time, created a highly detailed map of individual cells in the adult human lung – using spatial transcriptomics to not only get the location and identity of individual cells but also the detailed gene expression, or gene activity, profile of each of those cells. This comprehensive cellular atlas includes both healthy lung tissue and lungs from individuals affected by chronic lung diseases, such as idiopathic pulmonary fibrosis (IPF). This breakthrough represents a leap forward in respiratory disease research and offers a new window into understanding the cellular architecture of the lungs.
DETECTING DISEASE BEFORE IT BECOMES VISIBLE
Using cutting-edge spatial transcriptomics – an advanced technique that maps gene expression in the context of tissue structure – the team examined over 1.6 million cells. Their analysis of lung tissue from people with IPF revealed a surprising and important discovery: signs of disease were present at the molecular level even in areas of lung tissue that appeared structurally intact under traditional methods of examination. In other words, changes in gene expression can be detected before visible scarring or structural damage occurs.
AI-DRIVEN INSIGHTS FOR EARLY INTERVENTION
These early cellular signatures could prove crucial in developing new therapeutic strategies. Artificial intelligence (AI) was used to analyse the massive dataset, enabling the team to identify patterns and changes not easily seen through conventional methods. This AI-driven insight opens the door to precision medicine approaches that tailor treatments to the patient’s specific disease stage – intervening earlier in the disease process, potentially before significant tissue damage and symptoms emerge.
IPF is a progressive and currently incurable lung disease that causes scarring (fibrosis) of lung tissue, making it increasingly difficult for patients to breathe. It initially presents as shortness of breath and can quickly advance, leading to respiratory failure. Without effective treatment, IPF is typically fatal. In Australia alone, over 1,250 people are diagnosed with the disease each year, most commonly between the ages of 50 and 70.
GLOBAL COLLABORATION REVEALS
CELLULAR NICHES
St Vincent’s Institute of Medical Research (SVI) Associate
Professor Davis McCarthy, one of the lead researchers on the project, explained the impact of their findings with a powerful analogy: ‘If you imagine a city map in an atlas, we’ve been able to create a “Google Street View” to identify individual neighbourhoods and buildings – it’s that revolutionary.’
Published in international collaboration and relied on image-based spatial transcriptomics, integrated with AI-powered computational methods. The researchers profiled the expression of 343 genes across lung tissue samples from 26 individuals who had undergone lung transplantation due to pulmonary fibrosis, alongside samples from nine individuals without fibrosis.

‘While there are some new pharmacological treatments that can slow the progress of IPF, the only current effective therapy is lung transplantation,’ said Associate Professor McCarthy. ‘Unfortunately, transplants are not widely accessible, and they bring their own set of serious medical challenges.’ He co-led the study with Associate Professor Nicholas Banovich from the Translational Genomics Research Institute and Associate Professor Jon Kropski at Vanderbilt University Medical Center in the United States.
The team’s analysis revealed where fibrotic changes occur at the cellular level and provided new insight into the cellular and molecular mechanisms that drive the disease. They identified 12 distinct, molecularly defined spatial niches in both healthy and fibrotic lungs—offering a roadmap for future studies to explore how these niches evolve and contribute to disease.
‘In recent years, it has become clear that genetic factors contribute substantially to a person’s risk of developing IPF,’ said McCarthy. ‘But until now, the precise influence of those genetic variants on the structure and function of lung cells in real tissue environments has remained unclear.’This study marks an important step toward resolving that gap in understanding, and it lays the groundwork for future research into not only IPF but potentially other chronic lung conditions as well.
Author: Research in SVI’s Bioinformatics and Cellular Genomics Lab, headed by Associate Professor Davis McCarthy, has a robust foundation in statistics and machine learning. The software tools Davis has developed have been downloaded over a million times. His team is working closely with colleagues at St Vincent’s Breast Screen Clinic, the University of Melbourne, and the University of Adelaide to identify breast cancer in mammogram images using artificial intelligence, with the aim of transforming population breast cancer screening.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
SEEING THE FUTURE
How explainable AI could potentially transform the diagnosis and management of Inherited Retinal Diseases

Inherited retinal diseases (IRDs) are a group of uncommon eye disorders of genetic origin that often impair vision slowly, sometimes leading to blindness.
Affecting around ten thousand Australians, these conditions pose diagnostic and therapeutic challenges due to their clinical and genetic heterogeneity. Our research team is pioneering the use of explainable artificial intelligence (XAI) to revolutionise how IRDs are detected, classified, and monitored over time.
THE COMPLEXITY OF INHERITED RETINAL DISEASES
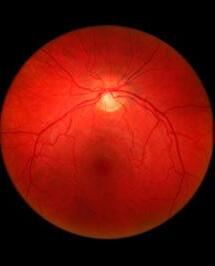
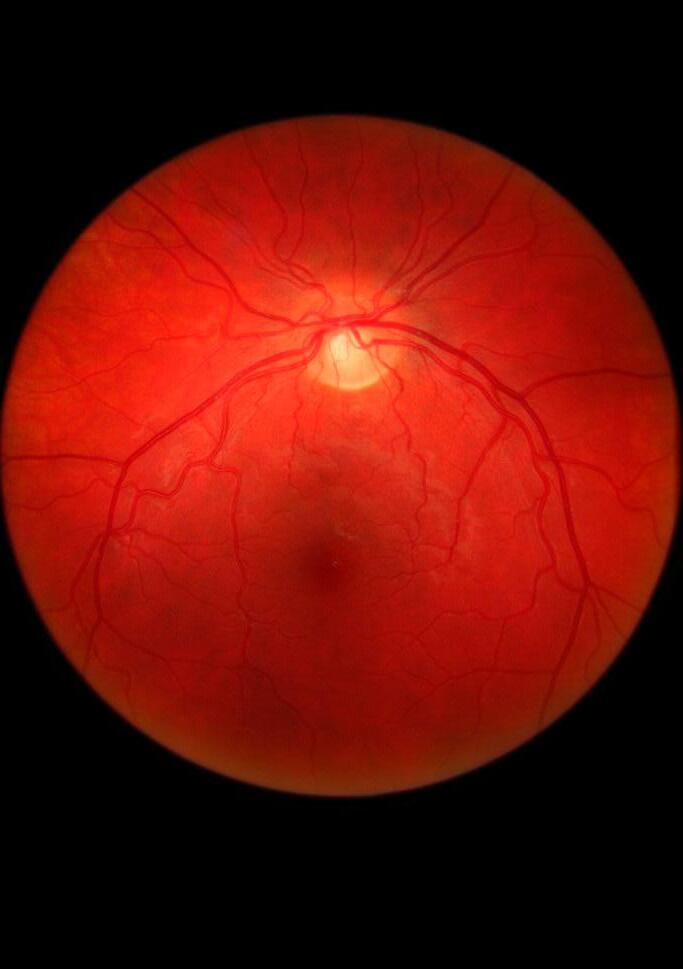
The retina, a light-sensitive layer at the back of the eye, is responsible for processing light signals crucial for vision. With multiple sublayers and cell types, it is one of the most intricate structures in the human body. Eye care health professionals use a variety of imaging techniques in order to assess its structure and function. Central (Figure 1A)
and wide-field (Figure 1C) photographs capture en face views of the retina, while cross-sectional scans, such as optical coherence tomography (OCT), reveal retinal sublayers (Figure 1B). Different wavelengths of light can also be used to infer retinal health and metabolism (Figures 1D and 1E).

Because the retina is crucial to vision, any retinal damage or degeneration can cause significant visual impairment. IRD management is complex. There are currently over 330 genes associated with IRD. Clinical presentations can vary significantly even within the same genetic mutation, and patients with different genetic mutations can sometimes show very similar clinical presentations. This heterogeneity makes it difficult to predict individual disease progression and what a patient’s vision will be like in the future.
AI OFFERS A WAY FORWARD
By learning patterns from large datasets, AI can make predictions even on new, unseen cases. In IRD research,
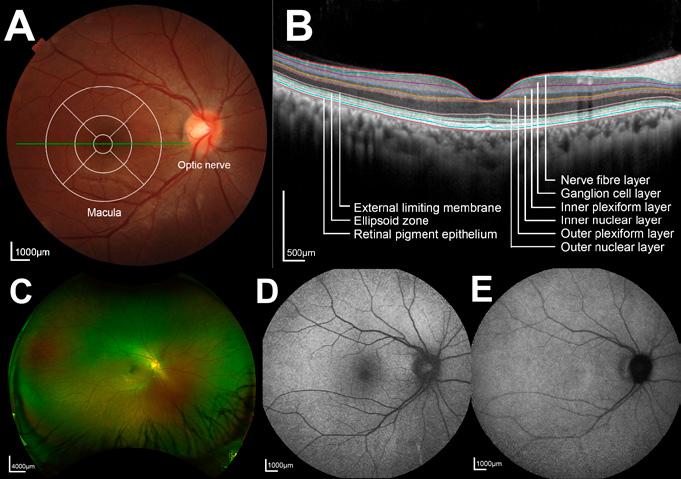
Figure 1: A healthy retina imaged by different modalities. (A) Photograph of the central retina. (B) Optical coherence tomography, demonstrating various retinal sublayers. (C) Widefield pseudocolour photograph. (D) Fundus autofluorescence image of the central retina using short-wavelength light. (E) Fundus autofluorescence image of the central retina using near-infrared light.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
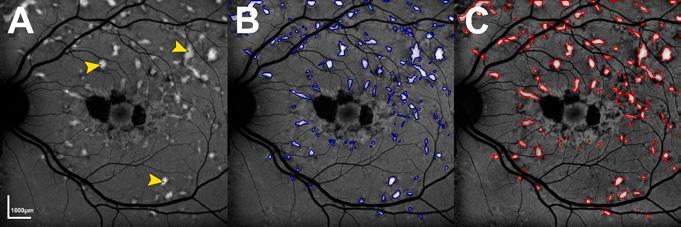
AI has already shown promise – for example, in Stargardt disease, where bright hyperautofluorescent flecks are a key clinical feature. AI algorithms have been utilised to automatically detect and outline these flecks, with accuracy approaching that of expert clinicians. This demonstrates not only the feasibility of AI in IRD, but also its potential to accelerate and scale diagnostic processes. There has also been a rise in XAI, particularly in healthcare, where transparency and trust are as important as accuracy. By opening up the ;black box’ of AI, XAI helps clinicians to understand why an AI model makes particular predictions. This allows better translation of complex data into meaningful patient care.
With high-performance AI models being available for image analysis in general and for retinal image analysis in particular, the time is right to harness their power and repurpose them for specific diagnostic applications. Our team has access to large datasets in IRDs as well as clinical and AI expertise which places us in a unique position in shaping the future of IRD early diagnosis and treatment planning.
This work is a collaborative effort between Professor Ajmal Mian and his team from Computer Science, and
clinician scientists Associate Professor Khyber Alam, Dr Jason Charng, Dr Han Trinh, and Dr Zahra Tajbakhsh from the Department of Optometry and Vision Sciences at the School of Health and Clinical Sciences at The University of Western Australia (UWA). Together, we are leveraging data from the Western Australian Retinal Degeneration (WARD) study to build robust AI models that:
• Detect and classify IRDs using high-resolution retinal imaging and genetic data.
• Predict individual disease progression, both structurally and functionally, enabling personalised care pathways.
• Utilise XAI techniques to identify interpretable markers of disease, offering clinicians transparent insights into model decisions.
An overview of this workflow is illustrated in Figure 3, where multimodal retinal images and patient data are processed through XAI models to generate predictions of a patient’s future visual function, while also highlighting the retinal regions driving these predictions.
At the heart of this innovation is The University of Western Australia, one of the world’s leading research institutions. UWA’s commitment to interdisciplinary collaboration,
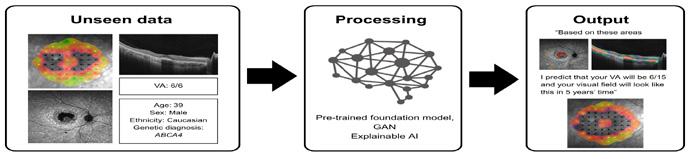
researchers’ explainable artificial intelligence (AI) workflow for inherited retinal disease. This research sits at the intersection of technological innovation and medical advancement. The rapid evolution of AI, imaging technologies, and genomic analysis is transforming how we understand and treat complex diseases. By harnessing these tools, we are not only improving diagnostic accuracy but also enabling earlier interventions and more tailored treatment strategies.
Figure 2: Example of artificial intelligence (AI)-facilitated segmentation of hyperautofluorescent flecks in Stargardt disease. (A) Short-wave autofluorescence image of an eye with Stargardt disease. Pointers indicate examples of hyperautofluorescent flecks. (B) Flecks segmented by an AI algorithm. (C) Manually segmented flecks.
Figure 3: Example of the



cutting-edge infrastructure, and societal impact places its researchers in a unique position to address some of the most pressing health challenges of our time. This project exemplifies how academic excellence can be translated into real-world solutions, bridging the gap between data science and clinical care.
Importantly, our work is also about equity and access. Many patients with IRDs live in remote or underserved communities, where specialist care is limited. By embedding XAI tools into teleophthalmology platforms, we aim to democratise access to expert-level diagnostics, reducing delays and improving outcomes.
As we move toward real-world deployment, we envision a future where explainable AI becomes a trusted partner in eye care beyond IRD, illuminating the path from uncertainty to clarity, and ensuring that all patients have access to high standard eye care in the age of digital medicine.
Authors: Professor Ajmal Mian, Associate Professor Khyber Alam, Dr Han Trinh, Dr Jason Charng, Dr Zahra Tajbakhsh, and Associate Professor Fred Chen. School of Health and Clinical Sciences, Optometry and Vision Science, The University of Western Australia.
L to R: Dr Han Trinh, Dr Zahra Tajbakhsh, Assoc Prof Khyber Alam, Assoc Prof Fred K Chen, Dr Jason Charng
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
BREAKING BARRIERS WITH NANOTECHNOLOGY
How a New Generation Electron Microscope
Can Transform Rural Healthcare

Figure 1: The Phenom Pharos G2 Desktop Electron Microscope, size of a coffee machine.

Dramatically reducing the size, complexity and cost traditionally associated with Electron Microscopy, this system is about the size of a home coffee machine, making access in remote regions a possibility for the first time.
It is not just a technological advancement; it is a beacon of hope for a healthier, more connected future for all Australians.

of resolving single proteins, viruses, and key cellular changes indicative of renal disease, cancer, and rare diseases. This level of detail is critical for precise diagnosis and ultimately, for guiding effective treatment.
FAR-REACHING IMPACTS
LEVERAGING DIGITAL IMAGING TECHNOLOGY AND NANOTECHNOLOGY
Electron microscopy (EM) plays a key role in the diagnosis of renal, skin, complex and rare diseases, but the increasing cost of the technology restricts its availability to only a handful of metropolitan locations. This can add time to critical disease diagnosis for patients and clinicians in rural and remote NSW and Indigenous communities. It also limits their use for broader educational purposes such as high school and undergraduate teaching.
Over the past five years, researchers at the Correlative Microscopy Facility (CMF) at Ingham Institute for Applied Medical Research (IIAMR), Dr Tzipi Cohen Hyams and Professor Murray Killingsworth, have collaborated with Thermo Fisher Scientific and its Australian representative, ATA Scientific, to adapt the Phenom Pharos system for medical research and cell biology applications. Initially designed for geological and engineering use in remote locations, the Phenom Pharos G2 has now been optimised for biomaterials and life science studies.
SMALL SIZE, IMMENSE POTENTIAL
The microscope dramatically reduces the size, complexity, and costs traditionally associated with EM. Its compact and movable design is about the size of a desktop computer or personal coffee machine. The team has refined imaging acquisition parameters, enhanced software functionality, and developed simplified sample preparation protocols.
Recent performance evaluations of the desktop electron microscope have demonstrated the system’s ability to deliver fast, high-resolution images with minimal training, service, and maintenance requirements. Its innovative design and application have already been recognised, with the Desktop Electron Microscope Initiative (DEMI) winning the 2023 NSW Health Pathology (NSWHP) Innovation Award and named as a Finalist in the 2023 NSW Health Awards. These achievements highlight its potential for broader implementation in medical research and education.
The newly developed desktop electron microscope is no ordinary instrument. It boasts the extraordinary capability

‘The impact of this innovation extends far beyond the laboratory bench,’ says Dr Tzipi Cohen Hyams. ‘The “desktop” size and significantly lower cost of this new EM system make its rollout to smaller, regional, and rural laboratories a tangible reality. This means that for the first time, communities outside of major metropolitan centres will have direct, timely access to world-class diagnostic technology.’
This is particularly significant for Aboriginal and Torres Strait Islander communities, for whom renal disease presents a serious health challenge. Faster, more accurate diagnoses will directly improve patient outcomes, alleviate anxiety for patients and their carers, and empower local healthcare staff with advanced tools.
The DEMI project respects the fundamental right of every patient to access world-class technology for serious illness diagnosis, irrespective of their geographical location. It empowers scientific staff, clinicians, and decisionmakers, fostering pride and a ‘can-do’ mentality in rural and remote regions. Ultimately, this pioneering desktop electron microscope will ensure that EM technology can support research and diagnosis to improve outcomes that truly matter, without the usual barriers to access.
Professor Murray Killingsworth notes that ‘the DEMI project is not just a technological advancement; it is a beacon of hope for a healthier, more connected future for all Australians, particularly those in our most remote and regional communities.
‘We believe that this microscope will play a key role in current and future educational outreach programs, providing hands-on experience to students and researchers in rural and regional areas. It will be a cornerstone of future STEM initiatives at Ingham Institute’s Perich Centre for Robotics & Health Technology, encouraging student participation in science and technology fields.’
Authors: Dr Tzipi Cohen Hyams is the Correlative Microscopy Manager at the Ingham Institute for Applied Medical Research.
Professor Murray Killingsworth is the Clinical Sciences Research and Head, Correlative Microscopy Facility Lead at the Ingham Institute of Applied Medical Research.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

NO DRAMAS
BRIDGING THE HEALTH INFORMATION GAP FOR YOUNG PEOPLE
Social Media Solutions for Regional Health Organisations to Communicate with Young People

Young people in regional Central Queensland face health challenges such as vaping and bullying, but struggle to access relevant information through traditional channels.
Using participatory action research with applied theatre methods, CQUniversity researchers worked directly with young people to understand their health priorities and communication preferences. The study, No Dramas, revealed that short-form social media videos are the most effective channel for youth health communication, but only when content aligns with existing platform formats and trends. Young people trust peer-created content more than traditional authority figures or printed materials. The research demonstrates that health organisations must meet young people where they are digitally, empowering
them to create authentic, experience-based content rather than expecting engagement with conventional health education approaches.
BROCHURES DON’T WORK FOR REGIONAL YOUTH
When they face serious health issues regional youth participants preferred getting information from friends and social media. So, health organisations need to rethink how they communicate with young people – forget brochures and posters! If the message doesn’t feel relevant or relatable, young people won’t engage with it. To be effective, health communication must meet youth where they already are—online—and speak in ways that feel familiar and trustworthy.
Dr Linda Lorenza, Lead Chief Investigator
Popcorn lung, Anyone else heard of this?
“There is no reason why people living in Regional, Rural, Remote or very Remote (RRRvR) communities shouldn’t have the same health outcomes as those in metropolitan areas.” INSPIRE, 2025, 036
YOUTH PARTICIPATION HELPS IDENTIFY WHAT WORKS FOR YOUNG PEOPLE
Participatory action research using drama methods with nearly 70 regional young people as co-researchers, successfully uncovered what health matters concern young people, and how they tend to find health information. TikTok-style short-form videos are the most effective option, but the content must match the style and trends with which young people already engage. No Dramas demonstrated that when young people are involved in creating the content, it feels more authentic and young people are more likely to trust and share it. The digital format of this content type offers inclusivity benefits, where young people who struggle with literacy are able to use voice commands to seek information on their mobile devices.
TIKTOK IS POPULAR, BUT USE VARIES
TikTok was a key platform in the study. Female-identifying youth often used it for dance challenges, while maleidentifying youth preferred gaming-related and comic content. Both groups understood features like “duets,” where users respond to others’ videos. But this feature can be used to mock or bully others. So, while TikTok is a powerful tool, it also has risks. Understanding how different groups of young people use the platform helps health practitioners and organisations to tailor health messages more effectively.
For example, the clip
Anyone else heard of this? juxtaposed a popcorn seller with popcorn lung, a condition developed through vaping. The comic clip with #popcornlung inspired young people to google popcorn lung, a health reality and potential deterrent to vaping.
SOCIAL MEDIA IS FAST, ACCESSIBLE AND RISKY
Young people can access health information quickly through social media. Short videos that combine visuals and text shared on platforms like TikTok, communicate more in less time than it takes to read a brochure. Trends change fast. TikToks are quickly outdated. Timely, effective social media attracts young people. Messages need to

keep up with current trends to interest youth. But there are downsides, like fast-spreading of misinformation and censorship – LGBTQI+ censorship on TikTok the risks.

Young people are more likely to talk about health concerns with friends or trusted adults than with health professionals. Health messages must reflect the unique needs of young people in language that feels natural to them. Marginalised groups of young people often face discrimination in healthcare settings. When youth feel heard and respected, they’re more likely to engage.
No Dramas found that young people want to share their own experiences and help others. They create real and relatable content based on their lives. This kind of storytelling can lead to positive behaviours. Health organisations should engage with youth to co-create health messages, empowering young people to take control of their health.
STAYING RELEVANT REQUIRES RESOURCES
As trends change, social media quickly loses ‘social’ relevance. What’s popular today is forgotten tomorrow. Keeping up with what’s trending takes time, effort, and money. By engaging with youth in social media development, health organisations can keep up, and even keep ahead. Changing government policies—like banning TikTok on official devices—can make it harder for health organisations to use these platforms. Despite these challenges, No Dramas shows that social media maintains strong potential for youth health communication.
Since the No Dramas study, a national ban on social media for young people aged under 16 has come into place in Australia. This poses a new challenge to health practitioners and organisations – how will youth aged under 16 find information without social media? No Dramas suggests health practitioners and organisations ask young people themselves.
Author: Dr Linda Lorenza, Lead Chief Investigator for the No Dramas Youth Participatory Action Research. She co-leads the Applied Arts and Regional Communities (AARC) research cluster of the Centre for Research in Equity and Advancement of Teaching and Education (CREATE).
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
TRANSFORMING AGED CARE THROUGH DIGITAL INNOVATION
How SMART on FHIR tools and dashboards can unlock the power of quality indicator data for personalised care and continuous improvement.
In aged care, every data point reflects a real life –each resident’s dignity and wellbeing depends on timely, informed care decisions.
Yet, the data collected through Australia’s National Aged Care Mandatory Quality Indicator Program (QI Program) remains siloed, its full potential untapped. Designed to drive continuous quality improvement, the QI Program data remains underutilised due to several systemic reasons:
1. Data collection is cumbersome and time-consuming, involving multiple personnel and manual processes. Providers must gather individual resident data using government-issued spreadsheet templates, which are then aggregated for submission.
2. This data is typically sourced from disparate IT sources, including clinical information systems (CIS), risk management, and medication software. Even within these sources, data can be spread across several forms and, in some cases, clinical notes.
3. The sector’s relatively low digital maturity means there is a paucity of digital tools to effectively leverage the QI Program data. Most importantly, it is unclear whether this data informs direct care decisions that could benefit individual residents.

COLLABORATING TO BREAK DOWN BARRIERS
To address these issues, the Aged Care Data Compare (ACDC) project was launched to design, develop, and evaluate digital tools that integrate QI Program data into clinical workflows. Led by the University of Queensland (UQ) in collaboration with Regis Aged Care, AutumnCare, the Australian eHealth Research Centre (AEHRC), the Digital Health Cooperative Research Centre (DHCRC), and the Department of Health, Disability & Ageing (DHDA), the project exemplifies the kind of academic-industry partnership championed by the Royal Commission into aged care quality and safety.
As a foundational step, researchers conducted interviews and workshops to assess existing workflows, organisational capacity, and contextual factors influencing QI Program data collection and reporting. The goal was to identify potential barriers and facilitators for effective implementation of the solutions.
This study uncovered fragmented, manual and resourceheavy processes stretched across multiple IT systems. Barriers included limited integration of IT systems supplying the required data, complex data definitions within the QI Program, misalignment of resident assessments for clinical care and for QI reporting, and workforce challenges such as high turnover and inadequate digital literacy.
However, strong governance structures, strategic alignment, and a sector-wide commitment to quality improvement emerged as key enablers. While participants noted a sense of change fatigue, they also voiced optimism that digital innovation could re-energise care delivery and streamline reporting.
SOLUTION DESIGNED TO BETTER CAPTURE DATA
The first digital tool developed was a SMART on FHIR-based application (app) designed to consolidate resident-level QI data from several sources within the CIS into a single, real-time view. Integrated with AutumnCare’s CIS, the app securely accesses resident-level data from the CIS database using a FHIR API and provides a real-time summary of the resident’s status in the quality indicators of hospitalisations, pressure injuries, unplanned weight loss, activities of daily living, incontinence, and restrictive practice.
The visual summary immediately flags critical indicators, for example, the resident has experienced significant weight loss - giving clinicians actionable insights at a glance. Clinicians can use these insights to initiate targeted care planning protocols, personalising care based on each resident’s needs. The app also allows bedside data capture, enhancing the richness of clinical records. Post-implementation interviews and a handson workshop with Regis staff revealed the app’s strong
potential to transform QI Program data into actionable insights for care. Staff reported it streamlined handovers, case reviews, and onboarding.
The app proves how interoperable digital tools can embed personalised care into workflows and accelerate aged care’s long overdue digital transformation.
A SINGLE SOURCE OF TRUTH
The second tool is a dashboard designed to transform QI Program data into actionable insights for strategic oversight and resident-centred care. Co-designed with Regis staff using participatory methods, the dashboard underwent iterative development and pilot testing with 30 end-users. It enables multi-level exploration of QI Program data: comparing groups of facilities, viewing individual facilities, resident groups, and individual residents.

By consolidating disparate data sources into a single, intuitive interface, the dashboard empowers aged providers to identify and prioritise quality indicators where they are underperforming or trending downwards and monitor progress over time. Pilot results confirmed strong usability and high user satisfaction; more than 85% of users endorsed its potential for personalised care, and over 90% agreed it would strengthen quality monitoring.
A HUGE BOOST FOR DIGITAL TRANSFORMATION IN AGED CARE
Together, these innovations represent a leap forward in aged care’s digital transformation journey. By standardising data, streamlining collection, integrating fragmented sources, and embedding actionable insights into clinical workflows, the ACDC project empowers providers to deliver more personalised, efficient, and high-quality care.
Clinical and Quality Operations Manager at Regis Aged Care, Sara Mayfield, said, ‘The ACDC project gave us the tools to streamline resident assessments and gain real-time insights through the SMART on FHIR app and interactive QI dashboard. Our teams were astonished at how quickly complex data became actionable insights that elevated care across every level’Building upon the experience and outputs of ACDC, the DHCRC has invested further in scaling the solutions, with the aim of delivering lasting benefits to residents, empowering care staff, and strengthening the aged care sector as a whole.
Authors: Ronald Dendere & Murray Hargrave, The University of Queensland. Meagan Snewin, DHCRC. Filomena Ciavarella & Imtiaz Bhayat, Regis Aged Care. Samantha Scholte, DHCRC. This project is supported by the Digital Health CRC Limited (DHCRC). DHCRC is funded under the Australian Commonwealth's Cooperative Research Centres CRC Program.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
PAIN ROADMAP
A non-pharmacological route to opioid reduction
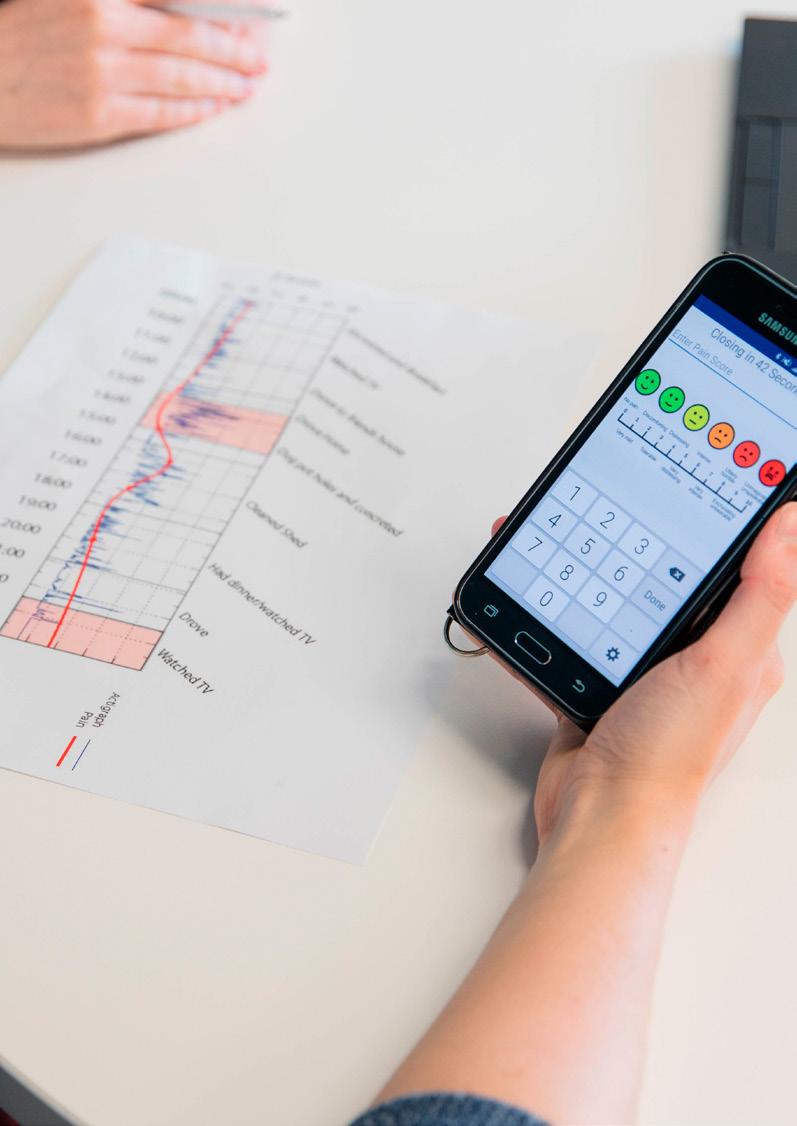

Clinicians and patients review the results from Pain ROADMAP’s analysis together to identify and modify the activities that lead to flare-ups.
Dr Nicole Andrews

Anew pilot trial is testing mobile health technology that helps people reduce pain flareups and support gradual opioid reduction.
Developed at The University of Queensland’s RECOVER Injury Research Centre, Pain ROADMAP is showing promise as a practical, non-pharmacological tool for safer, more sustainable pain management.
More than 3.6 million Australians live with chronic pain. Although the causes and intensity vary, chronic pain is defined as pain that persists for most days over a period of three months or longer. For many, opioid medication is a mainstay of daily management. It is estimated that nearly 40% of people prescribed opioids for chronic pain use high doses, placing them at greater risk of dependence and accidental overdose.
MANAGING FLARE-UPS
‘For people with chronic pain, physical activity is like a double-edged sword. We encourage people to exercise and be active, but doing too much in one go can significantly exacerbate pain levels, resulting in flareups,’ explains Dr Nicole Andrews from The University of Queensland’s RECOVER Injury Research Centre.
When pain flare-ups make it challenging to work, care for family, or enjoy life, many people rely on extra doses of opioid medications to cope. This type of medication is referred to as PRN or pain relief as needed. Research by Dr Andrews and colleagues has shown that people who frequently push through pain are more likely to be prescribed opioids and to take higher or more frequent doses than recommended.
‘People often want to decrease their opioid use, but tapering off is challenging without a viable alternative treatment to manage severe flare-ups,’ said Dr Andrews.
WHAT IS ACTIVITY PACING?
One promising non-pharmaceutical strategy for managing flare-ups is activity pacing. By breaking daily tasks into smaller, manageable segments, you can stay active without triggering a flare-up.
‘Activity pacing is great in theory, but people find it really difficult to master,’ said Dr Andrews.
‘It can be hard to recognise the triggers if the onset of pain is delayed, and that disconnect makes it very hard to change our habits.’One research participant explained:
‘The barriers I have are with myself more than anything. Being a person like I am, you just can’t sit and watch the dust pile up... it frustrates me quite badly, to the point where I will harm myself by going the extra. It’s very hard to change yourself.’
PAIN ROADMAP
Pain ROADMAP was created to bridge this gap. The mHealth (mobile health) solution helps people identify the
specific activities that trigger pain flare-ups and modify them with guidance from a clinician.
Users wear an activity monitor (Actigraph) and record their pain levels, medication use, and activity in a mobile app. The data are uploaded to a secure online portal that generates weekly summaries of average pain, activity patterns, and opioid use. The system uses an algorithm to visually map the associations between activity and pain flare-ups, giving clinicians clear insights to help patients adjust their routines.

Through these insights, patients can learn to stay active while avoiding the behaviours that worsen pain, often with transformative results.
After training with Pain ROADMAP, one participant shared:
‘I learnt to pace myself, to do things in a different way. Maybe it took a little longer, but it didn’t matter. I still was able to do things that I used to do before the injuries… You never lose the pain, but you learn to deal with it in a way that doesn’t affect your life as much. I’ve stopped virtually all my medications, become more active, healthier, and lost weight. It’s like a new life. Finally, a nonpharmaceutical option for opioid tapering
Following the Pain ROADMAP pilot intervention, participants experienced a significant reduction or elimination of the pain flare-ups. They also reported fewer depressive symptoms, less activity avoidance, and improved productivity. While opioid reduction wasn’t the study’s main goal, the results were encouraging. Almost 80% of participants who were taking ‘as needed’ opioid medication at the start had stopped by the program’s end, and overall opioid doses decreased slightly.
Pain ROADMAP’s initial success suggests a new path forward, where technology, clinical expertise, and patient insight come together to make pain management safer and more sustainable. A randomised controlled trial is now underway to evaluate the feasibility and effectiveness of Pain ROADMAP in supporting opioid tapering. The project brings together researchers and clinicians from the Tess Cramond Pain Research Centre (Royal Brisbane and Women’s Hospital), the Australian e-Health Research Centre (CSIRO), and UQ’s RECOVER Injury Research Centre.
Author: Dr Nicole Andrews is a Senior Research Fellow at The University of Queensland’s RECOVER Injury Research Centre and an Occupational Therapist at the Tess Crammond Pain Research Centre at the Royal Brisbane and Women’s Hospital. She is internationally recognised for her work on activity pacing and developing innovative digital tools that promote safer, more active lives for people with pain.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
DIABHQ
Harnessing the Power of Data to Transform Diabetes Care

Imagine living with a condition that requires you to make ~180 health-related decisions every day for the rest of your life.
This is the reality for approximately 11,000 Australian children and young people currently living with type 1 diabetes and facing not only the burden of day-to-day management, but the risk of complications and reduced life expectancy.
The Rio Tinto Children’s Diabetes Centre, based at The Kids Research Institute Australia (The Kids) and Perth Children’s Hospital (PCH), is developing an innovative digital platform that aims to improve the outlook for children living with diabetes by utilising data more effectively. DiabHQ integrates data from a range of different sources and makes it available to patients and families, clinicians and researchers through three customised, interconnected portals.
Management of diabetes creates an enormous volume of data – including device data from continuous glucose monitors, insulin pumps and activity trackers. Currently this information remains siloed across incompatible devices and platforms making it difficult to access and limiting its use.
Extensive consultation has identified a range of challenges and opportunities for the three groups of users who stand to benefit most from better access to diabetes data and information. Patients and families want a ‘one-stop-shop’ where they can access trusted resources, data and tools to support self-management in one place. For clinicians, access to information that helps them deliver care where it is needed is key, as well as maximising outcomes within current funding constraints. Frustrations faced by researchers include recruitment of participants to trials and the time it takes to translate research findings into practice.
DiabHQ app in use.

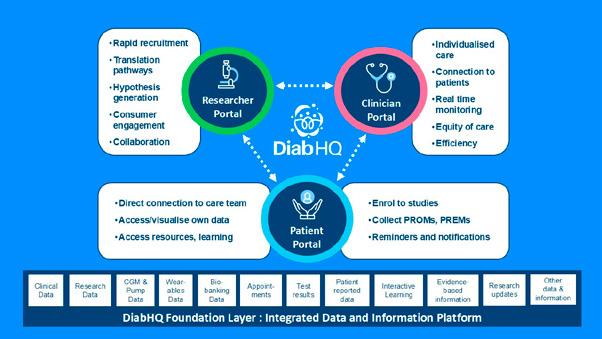
The DiabHQ project, a collaboration between the Kids and PCH, was funded by Stan Perron Charitable Foundation in 2023. It aims to create a comprehensive, scalable digital ecosystem that connects patients and families, clinicians and researchers and reduces barriers to effective selfmanagement, optimal care and translational research.
The project team has worked closely with consumers, clinicians, researchers, data experts and software developers to deliver the foundation data layer and the Patient Portal to date, with plans for the other two portals well advanced. Iterative development and co-design methodology allow regular input and feedback from users, ensuring that the final solution meets user requirements.
The Patient Portal, co-designed with a dedicated Consumer Reference Group, is available as an app for mobile devices, offering functionality that includes data visualisation (HbA1c trend, growth charts, pathology results), access to educational resources, appointment management, triage support and collection of patientreported measures. The DiabHQ app has been used for the last four months by a pilot group of more than 50 users and is scheduled for broader rollout from early November.
The Clinician Portal will support more equitable and efficient clinical care through remote monitoring, risk stratification and tailored care planning and the Researcher Portal will enable accelerated research and translation through rapid recruitment, secure access to de-identified data for approved studies, collaboration support and advanced digital capabilities.
DiabHQ has been designed to be scaled and adapted for implementation at other diabetes centres (including adult centres) and potentially also for other chronic diseases. The platform uses FHIR (Fast Healthcare Interoperability Resources) standards to maximise interoperability, API access for seamless integration, and a modular architecture that allows flexibility for future use-cases. A focus on documentation and process standardisation will reduce the effort required to implement the platform in other settings.

DiabHQ is hosted in the AWS cloud and includes state-ofthe-art security controls to ensure the privacy of sensitive patient data. Rigorous penetration testing of all current components has been undertaken by external cyber security teams and this will be repeated on a regular basis as the platform evolves.
Critical factors in the success of the DiabHQ project date include involvement of clinicians in the design and development process, ensuring their support for the project and advocacy for use of the Patient Portal. It has also been important to select technology partners with not only the requisite skills and experience, but also a shared vision to improve the lives of children living with diabetes. The formation of a Steering Committee including senior decision-makers across both The Kids and PCH as well as two consumer representatives has helped to quickly resolve roadblocks and to ensure that the needs and priorities of patients and families are considered at all levels of the project.
The project team expects the DiabHQ app to quickly become part of standard care following its official launch on November 13, the day before World Diabetes Day. Pilot users have provided consistently positive feedback about using the app, and clinicians are excited about its potential to help families prepare for their scheduled appointments and provide patient-reported outcome data to guide clinical care. This major milestone for the DiabHQ platform development marks the first step towards the project vision to harness the power of data to transform diabetes care.
Authors: Ms Helen Clapin, DiabHQ Program Manager and Data Theme Lead, at the Rio Tinto Children’s Diabetes Centre, The Kids Research Institute Australia, and Ms Erica Rojas Wood, Data Project Officer at the Rio Tinto Children’s Diabetes Centre, The Kids Research Institute Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
ImmsonTIME



A digital innovation transforming the approach to childhood vaccination.
Coordinated action is needed to improve childhood vaccination coverage and address the disparities in coverage occurring in certain groups.
Curtin University and the Boorloo (Perth) Public Health Unit (PHU) are working collaboratively to develop a datadriven tool, using a machine learning approach, to predict delayed vaccination in children and identify families for inclusion in a proactive vaccination support program in Perth.
Immunisation is considered one of the most cost effective, equitable and successful public health interventions globally and has significantly contributed to increased life expectancy and the reduction of infant mortality. Immunisation protects vulnerable populations, reduces burden on healthcare systems, and prevents outbreaks of vaccine-preventable diseases (VPDs). Timely and complete vaccination in early childhood is essential for protecting young children from potentially life-threatening VPDs, at a time when these diseases can be more severe. The global resurgence of measles is a stark reminder of the critical need to highlight childhood vaccination as a public health priority.
In Australia, a 95% benchmark for childhood vaccination coverage is set to maintain public health. Historically, Australia has been successful in achieving and sustaining high rates of childhood vaccination, however, over the last 5 years, childhood vaccination rates have been falling. Inequities exist in certain population groups with lower uptake, including Aboriginal 1 children, and children of parents experiencing social disadvantage, living in rural and remote areas, or born overseas. In Western Australia (WA) in 2024, 88.9% of all children were fully immunised at 2 years of age, with some areas with coverage as low as 73.73%, underscoring the magnitude of the issue.
1 The use of the word Aboriginal within this article is inclusive of all Aboriginal and Torres Strait Islander people. The term Aboriginal is used in preference to Aboriginal and Torres Strait Islander people, in recognition that Aboriginal people are the original inhabitants of Western Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
BARRIERS TO VACCINATION
Barriers to childhood vaccination can be broadly categorised into access and acceptance barriers. Access barriers include practical difficulties related to vaccination appointments, such as the booking of appointments or transportation to appointments, concerns with affordability and the costs associated with vaccination, and other issues related to social and structural inequities. Acceptance barriers can include parental concerns or beliefs about vaccination, and this may be related to social influences including exposure to misinformation or a lack of information.
To address barriers for families of Aboriginal children in Perth, a successful vaccination support program is currently provided by the Boorloo PHU, however, the program cannot be expanded to all children due to resource limitations and the large population served. There is critical need for an innovative solution to identify families with children at higher risk of delayed vaccination for inclusion in a support program.
LEVERAGING DIGITAL HEALTH TECHNOLOGIES
Machine learning and artificial intelligence are reshaping the healthcare landscape and driving innovations in areas including predictive modelling, early disease detection and individualised care. Curtin University and the Boorloo PHU are utilising digital health technologies to develop a predictive model to assess the risk of children missing scheduled vaccinations and identify families in need of proactive support. Multiple data sources which are routinely used for public health immunisation followup have been integrated to comprehensively capture predictors of delayed vaccination. Predictors may include maternal socio-demographic factors (such as Index of Relative Socioeconomic Advantage and Disadvantage, whether an interpreter is required, mother’s age at child’s birth) and pregnancy-related information (such as pregnancy vaccines received, whether mother smoked during pregnancy, number of previous pregnancies). The use of advanced machine learning algorithms, including regularised logistic regression, decision trees and random forests, will enable superior predictive performance compared to conventional methods, enhancing accuracy and reliability. Machine learning algorithms will uncover critical patterns, anticipate the specific needs of families and support the development of a tailored support program.
SUPPORT FOR VACCINATION
Once the predictive model has been reliably validated, the potential for its use in public health programs will be explored. Other successful public health support programs for immunisation include regular reminders and engagement with families prior to scheduled vaccination dates, the provision of information and discussion of any concerns, the booking of vaccination appointments, the provision of transport to appointments, and a home visiting vaccination service for families at highest risk. An appropriate public health program will be tailored to the final model. Addressing the specific challenges faced by certain groups, such as those with logistical or practical barriers to vaccination, reduces health inequities related to vaccination coverage and has the potential to improve vaccination coverage.
IMPACT AND FUTURE DIRECTION
This digital innovation represents a significant advancement in childhood vaccination and public health. The program addresses the challenge of predicting delayed vaccination in time to provide critical early intervention, allowing for proactive intervention and support from birth prior to the child becoming overdue for vaccinations. It reaches at-risk families and ensures resources are directed where they are most needed. The innovation provides opportunities to replicate successful models in other regions facing similar challenges, particularly those with limited access to healthcare infrastructure. It is essential to identify those most in need of support not only in WA but at a national and global level to ensure optimal vaccination coverage, promote health equity and protect the health of communities.
Authors: Teya Duggleby, Project Coordinator, Health Sciences Research at Curtin University, on behalf of the project team. The project team includes Professor Gavin Pereira, Project CoLead, Professor of Epidemiology and Dean of Research at Curtin University. Dr Ben Scalley, Project Co-Lead, Medical Director at Boorloo PHU. Dr Anastasia Phillips, Project Medical Lead, Public Health Physician at Boorloo PHU. Dr Kingsley Wong, Biostatistician and Health Data Scientist at Curtin University. Ellie Darcey, Epidemiologist at Boorloo PHU. Haylee Hawkins, Lead Consumer Representative, Healthy Pregnancies Consumer Reference Group.



CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
WALKING TALL
How Digital Tools are Helping People with Parkinson’s Take Back Their Steps

From smartphone apps to wearable ‘pacemakers for gait,’ new digital innovations are transforming the way Parkinson’s disease is managed.
Researchers at UNSW Sydney and Neuroscience Research Australia, supported by Shake It Up Australia Foundation, are harnessing patient co-design and realworld data to improve walking, reduce falls, and enhance quality of life.
RETHINKING PARKINSON’S CARE THROUGH TECHNOLOGY
Walking is something most of us take for granted. But for the 200,000 Australians living with Parkinson’s, every step
can demand enormous concentration. Gait dysfunction— shorter steps, freezing, and loss of balance—affects independence, increases falls, and undermines confidence. In fact, up to 70% of people with Parkinson’s experience falls each year.
To meet this challenge, Associate Professor Matthew Brodie and his team from UNSW School of Biomedical Engineer have been developing digital tools that directly respond to the needs of people with Parkinson’s through UNSW spinout Walking Tall Health . Their flagship innovation, the Walking Tall app, was co-designed with patients and launched in 2023 following a clinical trial funded by Shake It Up Australia Foundation and The
Michael J. Fox Foundation for Parkinson’s Research. The app is freely available for community use on Android Google Play and App Store and is being used by thousands of people worldwide.
The app delivers a simple but powerful intervention: a rhythmic metronomic beat at different walking speeds to help users pace their steps. It combines this with coaching prompts such as ‘walk tall’ or ‘focus on big steps.’ Crucially, it was shaped by direct feedback from people with Parkinson’s themselves, ensuring usability and impact in daily life. Positive feedback from app users includes ‘Love it,’ ‘Very helpful’, and ‘Great encouragement to walk’.
CLINICAL TRIALS AND REAL-WORLD IMPACT
The Walking Tall app was tested in a 13-week clinical trial involving 62 people with Parkinson’s. Half the participants used the app’s gait training function, while the others followed a traditional program known as the Otago Exercise Program (OEP).
The difference was stark. Those using OEP exercised for about 60 minutes per week, while app users averaged 150 minutes per week—more than double the activity. For a progressive condition like Parkinson’s, consistency of exercise is one of the most important predictors of mobility and independence.
Participants also reported that the app made walking less mentally taxing. ‘Those who live with Parkinson’s told us it can take 100% of their concentration just to walk across a room,’ says A/Prof Brodie. ‘The metronome beat helps replace that need to think about each step, so walking becomes more natural again.’The app lowers barriers to daily training by keeping things simple. Just six minutes a day, three days a week, is enough to make a measurable difference. Audio encouragement, progress markers, and even playful rewards like ‘gold cups’ add motivation. Importantly, the app can be used in any setting—around the block, in a park, or even just up and down the hallway at home.
BEYOND THE CLINIC: THE ROLE OF DATA AND PASSIVE SENSING
Digital tools like Walking Tall don’t just support rehabilitation—they also generate valuable data about movement patterns. This can provide clinicians with objective measures of how patients are walking in their everyday environments, not just during short clinic visits. Over time, such data could reveal early signs of worsening gait, guide timely interventions, and personalise therapies.
The emphasis on co-design has also been central. People living with Parkinson’s were involved from the very beginning, shaping everything from the timing of


the metronome beats to the app’s interface. Walking Tall Health’s Chief Science Officer, Dr Martin Ostrowski, who has Parkinson’s himself, played a pivotal role in translating lived experience into practical solutions. ‘It’s all about keeping it simple and building trust,’ he explains. ‘When I use the app, it gives me that rhythm so I can walk without using all my mental effort.’This model of patientdriven design, combined with data-rich feedback loops, is setting a new standard for how digital health tools can evolve. Each update to the app builds on user feedback, ensuring the technology adapts to real-world needs.
A ‘PACEMAKER FOR GAIT’: THE NEXT STEP
The success of the Walking Tall app has paved the way for a wearable band prototype—strapped above the ankles—that acts as a ‘pacemaker for gait.’ The device synchronises stimulation across limbs to restore walking rhythm and stability.
In early testing, results have been remarkable. One participant, using the bands, was able to walk 42 kilometres in a single day—an unimaginable feat beforehand. Stories like this underscore the potential of digital devices to transform daily life, not just manage symptoms.
In December 2023, the project received a National Health and Medical Research Council (NHMRC) Development Grant of $1 million to accelerate the device toward clinical approval and widespread use. The goal is to manufacture the technology sustainably in Australia and make it affordable for patients worldwide. With broad application across conditions including stroke, diabetes, and dementia, its impact could extend far beyond Parkinson’s alone.
Author: A neuroscientist and engineer, Associate Professor Matthew Andrew Brodie has over ten years of experience conceiving new research ideas, developing technology-based interventions, and leading clinical trials. His key areas of expertise include peripheral nerve stimulation and neurorehabilitation, health app development and telemedicine, the biomechanics of human movement, wearable technology and smart textiles, and more.
Associate Professor Brodie leads a multidisciplinary team passionate about advancing the intersection of neuroscience, engineering, and human health. His group is currently welcoming motivated research students for a range of innovative projects.
Article submitted by Shake It Up Australia Foundation.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
HEPLINK AUSTRALIA
The
national link to hepatitis care and treatment

THE CHALLENGE OF VIRAL HEPATITIS IN AUSTRALIA
Almost 300,000 people in Australia are currently living with hepatitis B or hepatitis C. These viruses can cause serious liver damage, increasing the risk of liver disease and liver cancer. Sadly, nearly 1,000 Australians die each year from complications related to hepatitis B and hepatitis C, despite hepatitis C being curable and hepatitis B preventable through vaccination and manageable with the right care.
Often, these conditions show no symptoms until significant liver damage has occurred. Stigma, discrimination, and persistent myths about viral hepatitis continue to prevent many people from seeking testing and treatment.
HEPLINK AUSTRALIA
HepLink Australia is the national ‘front door’ to viral hepatitis support, treatment, and care.
The service provides a confidential and free pathway to essential information and services, ensuring that everyone
in Australia, whether they’re living with viral hepatitis, newly diagnosed, a healthcare professional, or a community worker, can access the guidance, support, and care needed.
It is an innovative model that offers phone and in-person services, as well as establishing Australia’s first national community hepatitis minimum data set. This initiative helps identify gaps in care and informs targeted outreach efforts, ensuring people affected by viral hepatitis are linked to appropriate services.
THERE ARE FIVE KEY PARTS TO THE SERVICE:
1. Service Delivery
HepLink Australia provides free, confidential support by phone and in person, offering nurse navigation, trusted information, practical advice, brief interventions, and referrals to specialised services.
Healthcare professionals can call HepLink during a consultation to receive expert guidance on clinical decision-making, navigating complex client needs, and improving patient outcomes. The Nurse Navigator can assist with treatment pathways, referrals, and practical advice.
2. Network Development
HepLink Australia identifies areas of highest unmet need for people affected by viral hepatitis and seeks to grow a network of safe services to support access for anyone, anywhere in Australia
HepLink Australia actively builds strong partnerships with local health and community organisations to expand access to hepatitis support and care. By working together, we strengthen referral networks, share knowledge, and ensure communities receive coordinated, culturally safe, and timely services.
HepLink Australia is building a safe, stigma-free national support network. We work with communities and partners to ensure everyone feels respected, supported, and empowered to seek the care they need without fear or judgment.
3. Referral and Navigation
HepLink Australia actively builds and strengthens referral pathways so people affected by viral hepatitis can get the care they need quickly and without delay.
HepLink Australia works in partnership with specialised support services, including cancer care, mental health, alcohol and other drug (AOD), and social services, to ensure clear and effective referral protocols.

4. Data Collection and Management
We use anonymous information to better understand community needs and improve hepatitis services. This helps us reach more people, offer better support, and make sure care is tailored to what matters most.

We use secure systems to manage anonymous information responsibly, helping us improve hepatitis services while protecting our clients privacy. Everything we do is designed to support communities safely and respectfully.
5. Targeted Outreach Activities
HepLink Australia identifies gaps in hepatitis services and responds with tailored outreach and small-scale initiatives designed to meet local and emerging needs.
These responsive projects are developed in collaboration with communities and partners. They ensure support is relevant, timely, and grounded in real-world challenges.
LOCAL CONNECTIONS AND PARTNERSHIPS
HepLink is a partnership between Hepatitis Australia and community hepatitis organisations in every state and territory. It is funded by the Australian Government Department of Health, Disability and Ageing.
The service connects callers with local hepatitis organisations and services across Australia. This ensures that people receive support that is relevant to their needs and location.
HepLink collaborates with government, researchers, peak bodies and other service providers to support the national response to viral hepatitis in Australia.
BRIDGING GAPS IN HEPATITIS CARE
HepLink Australia is more than a helpline, it is a vital connection point in the national effort to eliminate hepatitis B and hepatitis C. By offering accessible, culturally sensitive, and locally relevant support, HepLink enables people and communities to take control of their liver health. With early intervention and ongoing care, we can reduce preventable deaths and improve the lives of thousands of Australians.
HepLink is available Monday to Friday, 9am–5pm, by calling 1800 437 222. The service is free, and no Medicare card is required.
Author: Hepatitis Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
ADDRESSING THE POLICY VACUUM OF CONSUMERGRADE WEARABLE DEVICES: TOWARDS EQUITY IN DIGITAL CARDIAC CARE
The promise of wearable cardiac devices in nonmetropolitan Australia

In recent years, consumer-grade wearable device technology has progressed beyond heart rate and oxygen saturation measurement.
Devices, including smart watches and rings, now have the ability to conduct advanced analysis of physiological parameters, allowing for detection of arrhythmia, blood pressure measurement, body composition analysis, and sleep architecture characterisation. Some are also equipped with advanced risk stratification capabilities allowing for vascular age assessment, detection of heart failure decompensation, and algorithmic prediction of cardiac events. Where geographic isolation has hindered access to cardiovascular care, these devices offer unrealised opportunities for remote monitoring and intervention in non-metropolitan (i.e. rural, regional, or
remote) communities (Figure 1). For example, a farmer in non-metropolitan Australia could share weeks of continuous ECG data with their general practitioner without leaving the property, enabling timely arrhythmia diagnosis. A young Aboriginal person in a remote community, for whom cardiovascular risk scores are known to chronically underestimate risk, could have premature hypertension detected by a wearable, allowing for early intervention without long-distance travel. A visiting specialist could use longitudinal wearable device data to triage patient reviews, enabling increased visit efficiency. Rather than simply wellness tools or accessories, wearable cardiac devices have the potential to bridge structural gaps in non-metropolitan communities, allowing for personalised and comprehensive cardiovascular care

THE POLICY VACUUM
Despite the growing potential of wearable cardiac devices, technological advancement has created a policy vacuum whereby innovation has outpaced clinical integration. For non-metropolitan clinicians, this results in the dilemma of acting on unvalidated consumer device-obtained data without clear medico-legal protections or ignoring a wealth of potentially urgent and clinically relevant information arising from these devices. Further, if the clinician does interpret device data, for example weeks of ECG data from a smartwatch, how are they to be reimbursed? These questions highlight the urgent need for policy reform and clinical practice standards to address the rapid proliferation of wearable cardiac device technology.
In Australia, the Medicare Benefits Schedule (MBS) was established in 1984 to provide government rebates for medical services. Whilst the MBS has codes for ambulatory cardiac devices ( item 11716) for indications including syncope, pre-syncope, palpitations, and surveillance following cardiac procedure, there are no codes in place for clinical use of consumer-grade wearable devices. Regulatory frameworks, including the Therapeutic Goods Administration, continue to adapt to the fast-changing landscape of wearable device innovation. Whilst individuals purchase consumergrade wearables for prices set by the commercial market, downstream costs of data interpretation and contextualisation are effectively externalised to individual clinicians. Furthermore, although some consumer-grade devices have been validated in clinical practice, others lack rigorous clinical evaluation, resulting in uncertainty regarding data reliability, validity, and accuracy. Therefore, several questions arise: who owns the data, who pays for the data, and who is ultimately responsible for the data? These questions raise broader concerns from an ethical and data sovereignty perspective: what is stopping these data from being used to train proprietary algorithms, generating profit through multinational data monetisation, potentially exposing users to cybersecurity breaches? This calls for the implementation of robust policy frameworks aligning technological innovation with ethical clinical practice, both in metropolitan and non-metropolitan regions.
THE DIGITAL HEALTH DIVIDE: CHALLENGES AND PATHWAYS TO SOLUTIONS
Unlike clinical devices, consumer-grade devices are subject to significant autobiographical symbolism. Devices become components of self-expression, representing agency and empowerment associated with a consumer’s

decision to purchase and wear a product. As symbols of wellness, athleticism, technological sophistication, and affluence, consumer-grade wearables are typically advertised to higher income groups; especially those with existing brand loyalty. However, when confounded by policy constraints and lack of public healthcare reimbursement for the integration of wearables into clinical practice, this results in a digital health divide whereby socioeconomic status determines access to consumer-grade devices and their potential concomitant cardiovascular health benefits. Consequently, the digital health divide poses a significant barrier to the integration of consumer-grade wearable devices in non-metropolitan cardiovascular care.
One solution to the digital health divide lies in the reform of relevant policy across the Australian healthcare system. The creation of MBS codes would legitimise the use of wearables in clinical practice, which could subsequently lead to the development of policy measures to democratise the availability of devices. This includes targeted implementation through Aboriginal Community Controlled Health Organisations and non-metropolitan health networks. Therefore, this could facilitate the integration of consumer-grade devices in routine clinical practice in non-metropolitan areas, in an equitable, sustainable, and practicable manner.
CONCLUSION
Modern consumer-grade wearable cardiac devices offer unprecedented opportunities to improve cardiovascular care. However, the lack of healthcare policy and reimbursement results in ethical and financial challenges for clinicians, as well as equity gaps for non-metropolitan communities. Ultimately, unfunded care is unequal care, and the future of these devices depends largely on the ability of healthcare policy to keep up with technological innovation, rather than the technology itself.
Authors: Professor Girish Dwivedi is a Professor of Cardiology at the University of Western Australia (Harry Perkins Institute of Medical Research) and Clinical Lead for Innovation and Consultant Cardiologist at Fiona Stanley Hospital. Dr Annora A Kumar is a physician at Fiona Stanley Hospital and a Master of Philosophy candidate at the University of Western Australia, where her research focuses on artificial intelligence and cardiology.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

PRIVACY, ARTIFICIAL INTELLIGENCE AND PATIENT DATA
Realising public benefits while protecting privacy
As AI transforms health research, understanding privacy law and AI regulation is critical. From identifying confidential data to navigating ethical requirements and planning for commercialisation, here’s what researchers need to know about protecting patient privacy.
When a California patient discovered his cells had been turned into a patented, commercially valuable cell line without his knowledge or consent, it became a cautionary tale that researchers working with AI and health data cannot afford to ignore.
The 1990 Moore v Regents of University of California case established that patient trust depends on transparency — especially when commercial interests are at play.
As Big data and AI move increasingly to the heart of health and medical research projects, whether in academic investigations, clinical translation or commercialisation, this lesson becomes more urgent. While many researchers are aware of the ethical requirements for human research, few grasp how privacy legislation applies to AI driven research, and the consequences of getting it wrong.
IDENTIFYING CONFIDENTIAL DATA
Much of the data used in research is de-identified, but if it’s not, strict privacy requirements apply. The Commonwealth Privacy Act applies to private sector organisations and Commonwealth agencies and defines personal information as information about an individual that is reasonably identifiable. Small datasets with
identifiable characteristics, rare conditions, genomic data, and longitudinal patterns can all make individuals reasonably identifiable, even when names are removed.
Health and genetic information are classified as sensitive information under the Privacy Act and has a higher level of privacy protection.
THE LEGAL FRAMEWORK
As a member-owed mutual organisation and Australia’s largest medical indemnity provider, representing more than 95,000 doctors, Avant sees first-hand the medicolegal complexities that can arise when managing health information.
To collect, use or disclose health information for research, organisations must either obtain patient consent or rely on research exceptions in the Privacy Act. If it’s impracticable to obtain consent, health information can be used in research relevant to public health or public safety, if the National Health and Medical Research Council research privacy guidelines are followed.
Aligned with the National Statement on Ethical Conduct in Human Research, these guidelines require a Human Research Ethics Committee (HREC) to weigh whether the public interest in the proposed research substantially outweighs the public interest in protecting privacy. HRECs assess research merit, risks to participants, privacy protections, and whether consent is truly impracticable.
THE COMMERCIALISATION CHALLENGE
Here’s where transparency becomes critical. The secondary use of data and research for commercial purposes poses challenges and can impact the decision to participate in research. Research exemptions don’t apply to use of data for commercial purposes, and the boundary between commercial use and public health benefit is often blurred.
In Moore v Regents of University of California, a doctor developed a cell line from patient, patented it, and entered commercial agreements without the patient’s consent. The patient claimed that the tissue was his property and that the doctor failed to disclose the extent of his research and economic interests. The court confirmed that potential commercial interests must be disclosed to patients. While this case focused on the use of human tissue for commercial purposes, its principles apply equally to patient data.
For researchers whose work may lead to commercialisation, it’s important to plan for this from the start including consideration of any privacy issues and ethical and legal requirements. This means providing participants with information about future use of research outcomes and recognising their right to decide whether to be involved.
AI-SPECIFIC CONSIDERATIONS
AI raises distinctive privacy challenges. Models may enable re-identification of training data, infer sensitive

attributes not in the dataset, or repurpose the data beyond original intentions.
A recent example considering the interplay between privacy and AI is the I-MED Radiology case. In 2024, the Australian Information Commissioner made preliminary inquiries of I-MED after it learnt via media reports that I-MED had disclosed patient data to train a diagnostic AI model. The Commissioner was satisfied the data had been sufficiently de-identified, publishing about the case as a “beneficial example of good privacy practices”.

De-identification for AI projects requires robust methodology and documentation to reduce the risk of a privacy breach. Even if de-identified data is shared with an AI company, community concerns around the ethics of AI mean that, at a minimum, organisations should be transparent about their use of AI.
THE REGULATORY LANDSCAPE
Several regulatory developments will reshape the landscape.
AI tools developed in research may be regulated products under the Therapeutic Goods Administration’s software as a medical device regime , particularly where they involve clinical decision support software and diagnostic algorithms. Researchers should ensure they are aware of TGA requirements and consider regulatory pathways early.
Australia’s Voluntary AI Safety Standard establishes ten guardrails for safe and responsible use of AI. While currently voluntary, the government is considering mandatory requirements for high-risk AI - likely including health applications.
Amendments to the Privacy Act in 2024 significantly increased the Australian Information Commissioner’s enforcement powers for breaches of the Act. The Commissioner can now issue fines and infringement notices for less serious breaches, including non-compliant privacy policies. Maximum penalties have increased to $50 million, meaning greater regulatory scrutiny.
MAINTAINING TRUST
Ensuring privacy compliance maintains community trust in the value of research. With proper planning and understanding of the legal framework, researchers can advance innovation while protecting privacy. As AI transforms health research, proactive privacy compliance isn’t just about avoiding penalties - it’s about maintaining the trust that allows research to benefit all.
Author: Georgie Haysom, BSc LLB(Hons) LLM(Bioethics) GAICD GCPysBM, General Manager, Advocacy Education and Research, Avant Foundation.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
INVISIBILITY IS NO SUPERPOWER
It’s Time for a Nationally Coordinated Approach to the Collection and Use of Rare Disease Data.
Shining a light on the unseen—every Australian with a rare disease deserves to be counted and cared for.

We can’t care for what we don’t count. Rare diseases impact everything—from people’s daily lives to the complex systems and services they depend on.
Rare diseases are the leading cause of death in children, responsible for six in 10 deaths, with a hospital mortality rate 13 times higher than common conditions. In Australia, rare diseases are estimated to contribute direct and indirect mortality costs exceeding $100b each year—and this only accounts for the financial impact of some of the 7000 known rare diseases. It’s easy to see why rare diseases are now recognised internationally as a global health emergency by the United Nations and the World Health Organization.
Collectively common, an estimated 2 million Australians live with rare disease. To have the same chance at a
good quality of life as other Australians, all systems— health, disability, aged care, housing, education and employment—must work together. Yet, Australians living with rare diseases remain largely invisible across every system.
UNDERSTANDING AND ADDRESSING DATA AND DATA GAPS IN RARE DISEASE
Addressing data gaps is an urgent priority for the rare disease sector in Australia and globally. As identified in the Australian Government’s National Strategic Action Plan for Rare Diseases (the Action Plan), rare diseases are not routinely counted or recorded. A nationally coordinated approach to the collection and use of rare disease data, including registries, is a key Action Plan priority.
Reliance on existing proxies, such as the International Classification of Diseases (ICD) codes, leaves more than
40% of rare diseases invisible in our health systems, leading to:
• Delays in diagnosis and treatment access, and progressive decline
• Inability to measure prevalence and burden of disease
• Gaps in understanding socio-economic costs
• Limited evidence to support investment in rare disease therapies and clinical trials
BUILDING THE FOUNDATIONS FOR VISIBILITY
Since the launch of the Action Plan in 2020, Rare Voices Australia (RVA)—the national peak body for Australians living with rare disease—has consistently advocated for the recognition of rare disease data challenges. In 2023, RVA and registry experts at Monash University conducted an audit of Australian registries and databases collecting rare disease data. This research informed the report, Recommendations for a National Approach to Rare Disease Data, outlining an aspirational vision for a single national person-centred approach to rare disease data that is:
• Flexible and responsive, to enable changes and additions to the dataset
• Modular, to enable interoperability and linkage with individual rare disease registries and other relevant data collections
• Mandated, to ensure all Australians living with rare disease are counted, so no one is left behind
In Australia’s Top 10 Rare Disease Research Priorities report, published in 2025, parents, carers, health professionals and rare disease community representatives ranked ‘Data Collection and Use’ as number 5. This highlights the urgency and shared commitment to change.
Globally, the United Nations Resolution on ‘Addressing the Challenges of Persons Living with a Rare Disease and their Families’ calls on Member States, including Australia, to:
Collect, analyse and disseminate disaggregated data on persons living with a rare disease… to identify discrimination and to assess progress towards the improvement of the status of persons living with a rare disease.”
A CLEAR PATH FORWARD BASED ON INTERNATIONAL PRECEDENTS
The gaps are clear, and the consequences of inaction are profound. We cannot continue to guess local statistics based on international datasets as these extrapolations



cannot make Australians living with rare disease visible. Data invisibility denies Australians living with rare disease the timely care and treatments that could change or save lives.
Australia must implement an appropriate coding system to make rare diseases count. International precedents provide a clear path forward. Germany and France have successfully implemented ORPHAcodes —a comprehensive, internationally recognised classification system for rare diseases. ORPHAcodes are being mapped to ICD and SNOMED systems, and they uniquely link to the Orphanet Functioning Thesaurus, which describes the functional daily impacts of rare diseases. This dual capability could transform the understanding of rare disease within the National Disability Insurance Agency, especially as most Australians living with rare disease meet the government’s definition of disability.
Nationally networked Centres of Expertise (CoE) for rare diseases are an essential vehicle for achieving visibility and improving care, ranking fourth in Australia’s Top 10 Rare Disease Research Priorities.
Encouragingly, several CoE are emerging across Australia. The Rare Care Centre at Perth Children’s Hospital is internationally recognised for best practice, and actively implementing ORPHAcodes—an important step towards systemic visibility. However, isolated progress is not enough. National leadership and coordination are essential to ensure equity.
The time is now. A nationally coordinated approach to rare disease data will enable equitable access to care, empower research and ultimately save lives. Australia has both the evidence and the momentum—now we need the leadership and commitment to act.
Author: Dr Falak Helwani is the Research and Evaluation Manager at Rare Voices Australia. She is a former cell biologist and mother to two children living with a rare disease.
Dr Falak Helwani
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

SIMPLIFYING CONSENT FOR HEALTH AND MEDICAL RESEARCH
A new, simplified participant information and consent form (PICF) template is improving how participants engage with health and medical research studies in Australia, and enabling data sharing to improve Australians’ health through research.
For ethical health research, including clinical trials, participants need information to help them make an informed decision about participating in a study.
The global shift towards sharing clinical trial data promises to improve efficiency and reduce the costs of research, accelerating better health outcomes. Obtaining participant consent to share data at the outset of a trial is critical to ensuring transparency and respecting autonomy. However, most of the sector uses PICF templates created over a decade ago that are often long, complex, and difficult to understand.
CT:IQ (Clinical Trials: Impact & Quality), in partnership with the Australian Research Data Commons (ARDC)
HeSANDA partners at the launch of Health Data
at SAHMRI. © ARDC / Andy Steven

through the People Research Data Commons and 72 national partners contributing to Health Data Australia, has created a simpler, shorter, and more engaging PICF template for Australian health and medical research. The new participant-centric template was designed by a multidisciplinary project team, including consumers, researchers, contract research organisations and human research ethics committees, with input from over 700 survey respondents and a suite of consumer workshops.
The simplified PICF template is free for researchers to access and modify and is accompanied by a comprehensive user guide. It helps potential participants make better-informed decisions about research participation and future sharing of their research data. For researchers, the template facilitates clarity and certainty about data sharing, enabling them to confidently share data via Health Data Australia to improve lives through accelerated health research.
AN ACCESSIBLE AND CONSUMERFOCUSED APPROACH
The template has received support from state governments, highlighting its potential to inform standards nationally. James Cokayne, head of Research Ethics and Governance in the NSW Ministry of Health, has emphasised the significance of the PICF template and user guide, as it fills an important gap in replacing the existing templates with more consumer-focused materials.
‘CT:IQ has broken down consent into its component parts to create a practical, next-generation template that encourages researchers to think about the design, audience and language of the consent form.’‘It’s a groundbreaking way of thinking about how participant consent is presented in human research, ensuring that it is both meaningful and informative.’Simon Windsor, Manager of Research Governance and Ethics in SA Health’s Southern Adelaide Local Health Network (SALHN), is positive about the new approach.
‘If you’ve got a 30-page document, it’s a genuine barrier to being involved in clinical trials and health research. This project will have a beneficial impact on the community through better communication, which is crucial to healthcare in the modern era.’SALHN incorporated a barcode in the PICF template, allowing the consent forms to be readily scanned into participants’ electronic medical records.
‘We were the first site in Australia to use the PICF template during beta-testing. We deployed the template at SALHN as one of the early adopters,’ said Simon.
Following the successful launch phase, the project is seeking endorsement from the National Health and
Medical Research Council to replace the existing PICF templates. Simplifying PICFs is a promising shift towards more accessible, participant-friendly approaches in health and medical research, reducing barriers to informed consent and appropriate data reuse.

REMOVING BARRIERS TO DATA SHARING FOR BETTER HEALTH OUTCOMES
The simplified PICF template is a key initiative within ARDC’s broader program to support ethical and secure data sharing, enabling collaborative research and fostering innovation to improve healthcare outcomes nationally.
The Health Studies Australian National Data Asset (HeSANDA) program provides national infrastructure for researchers to share and access data from health studies and clinical trials. Coordinated by the ARDC, HeSANDA clinical trials connects 72 health and medical research organisations, health service providers, and clinical trial networks through 9 nodes. Together, these partners codesigned the framework for sharing clinical trial data via the Health Data Australia platform.
A key component of HeSANDA is Health Data Australia, an online catalogue launched in 2023 that contains descriptive metadata for hundreds of Australian clinical trial datasets and enables researchers to request access to this data. It is now being expanded to include cohort datasets in partnership with 40 health organisations.
Alongside building digital infrastructure, HeSANDA is also addressing culture and policy to enable health data sharing. The simplified PICF template is a significant development in this space. Additional developments include a methodological guide for the secondary use of clinical trial data in research in partnership with the Cochrane Prospective Meta-Analysis Group (facilitated through the University of Sydney NHMRC Clinical Trials Centre), and practical guidance to better support ethics committees when considering requests for sharing of clinical research data in partnership with CT:IQ, which will be released in late 2025.
By addressing the technical, cultural, and policy dimensions of health data sharing, the HeSANDA is helping researchers access and reuse health research data safely and efficiently, maximising the impact of publicly funded research and strengthening Australia’s contribution to global health research.
Author: Mark Mclean is the Program Manager of HeSANDA at ARDC. Jo Savill is a Senior Science Communicator at ARDC.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

CURE ASTHMA
Australia’s Unique Opportunity
Asthma is invariably described as an incurable condition, yet there is no scientific basis to believe that asthma is fundamentally incurable.
We would not accept this proposition for most other serious diseases. Lung cancer research seeks to cure lung cancer, Cystic Fibrosis research seeks to cure CF. Yet despite great progress in management approaches over the past decades, where ‘on-treatment remission’ has become a possibility, there is a widely and deeply held view by many that asthma is, in some inextricably tangled way, a ‘treatable but incurable disease’.
We cannot accept this - the burden of asthma in Australia, which remains amongst the highest in the world, is too great, and the need for solutions is too compelling. The catalytic spark of the CURE Asthma Initiative- now detailed in a full supplement of the Medical Journal of Australia- is to formally name the ambition to CURE Asthma and bring together our best clinical, epidemiological and scientific talent nationally, to discover transformative solutions.
WHY A FOCUS ON CURE ?
The scope of the asthma problem is enormous and growing. Asthma affects approximately 2.8 million people (11% of the population)—one of the highest prevalence rates globally. Children, First Nations communities,
and socioeconomically disadvantaged groups are disproportionately impacted. Nearly 400,000 children are affected, making asthma the leading cause of disease burden in the 0–14 age group, causing more hospitalisations than any other chronic condition.
A diagnosis of asthma in childhood is a curse that is associated with significant risk of premature all-cause morbidity and mortality in later life. Hundreds die burden at such scale and with such unpredictability as asthma. Inhaled corticosteroids , the usual standard of care, suppress inflammation and are effective for many, but they do not alter disease trajectory - we lack disease-modifying or curative therapies. Far from being ‘under control’, the worsening statistics call for a focused response. Moreover, when asked directly, consumers do not want incremental improvement in treatment, they want and deserve cures.
BUT ARE CURES FEASIBLE?
Can asthma be cured? The emerging answer is: yes!in principle. Asthma is not one disease. It is a set of molecularly distinct endotypes (molecular subgroups that can be used to find and match personalised treatments). A single universal cure is not plausible; instead, we should expect multiple cures - each endotype will require a different mechanism-directed solution.


Asthma is acquired and this means we can focus on key transitions from health to asthma that have been identified in large scale epidemiology studies. Environment plus genetic predisposition switch the disease ‘on’. Crucially, spontaneous complete remission also sometimes occurs turning the disease ‘off’- and durable on-treatment remission is now achievable in subsets of patients. These findings point to the way forward to find the deeper molecular causes of asthma (and to exploit our natural protections and resolution processes that protect most from asthma). The challenge is to convert these insights into curative treatments and make off-treatment remission predictable, repeatable, and scalable.
The key science is to define the specific pathways, cell states, and tissue programs that distinguish “disease switched off” from “disease maintained.” There are already important clues in the empirical evidence. GWAS (Genome wide association screening) has identified variants associated with spontaneous complete remission (in tissue repair, protein folding, immune tone). Epigenetic studies show persistent methylation (scars) from unresolved injury - precisely the molecular signatures that discriminate resolution from persistence, and which can also mis-shape the immune system and redirect lung cell lineages to dysfunctional phenotypes.
Moreover, we are in an unusually favourable era. Mechanistic discovery biology, endotype-level clinical phenotyping, new drug design technologies, and precision trial methodologies are accelerating - driving rapid advances in computational biology including profound advances in AI/machine learning, impossible

even five years ago. These mechanistic insights now support a serious scientific proposition: curing asthma is feasible and the earliest examples may emerge within a decade.
WHY AUSTRALIA IS UNIQUELY ADVANTAGED
The CURE Asthma initiative is uniquely positioned to drive transformative progress because it builds on decades of Australia’s world-leading clinical research, longitudinal cohort epidemiology and fundamental basic and discovery research.
Our CURE-ADIRE (Asthma Data Integration Research Engine) will bring together data and biosamples from around 75,000 patients from 11 major cohort studies. These cohorts span more than 60 years across the entire lifespan. They are ongoing and unparalleled globally in both scale and detail, a national resource of international significance. Working in four complementary streams (Definitions, Mechanisms, Treatments and Trials), CURE Asthma bridges these exceptional national resources with cutting-edge advances in molecular science, computational biology and innovative AI methods like ‘digital twins’ to accelerate discovery with clinical, trials bringing curative therapies within reach.
Authors: Anthony Flynn, Asthma Australia and Professor Gary Anderson, University of Melbourne.
CURE Asthma Symposium 2024
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
MATHEMATICAL MODELLING SHAPES GLOBAL POLICY TO ELIMINATE MALARIA
Researchers based at The Kids Research Institute Australia are using mathematical models of malaria dynamics to eliminate one of the world’s deadliest diseases.
Global malaria cases surged to an estimated 263 million in 2023, with more than 597,000 deaths, according to the World Health Organization (WHO). The vast majority – approximately 95 per cent – occurred in Africa, where vulnerable groups such as children and pregnant women are particularly affected.
The Kids Research Institute Australia’s Global Diseases Modelling team is tackling this issue head on by conducting world-leading mathematical modelling research designed to guide key interventions in the fight against malaria.
Fiona Stanley Chair in Child Health Research and The University of Western Australia Professor Melissa Penny, has spent more than eight years shaping global malaria
strategies through the WHO and says modelling research of this nature had a long history in the fight against malaria.
“We use data and modelling to better understand the transmission and biology of malaria, and to identify effective interventions to curb the rising number of cases of this life-threatening disease,” Professor Penny said.
“The models provide vital evidence to support research and development, investment, and policy decisions on novel malaria interventions like new vaccines and drugs.
“This work has directly informed impact and economic assessments of malaria vaccines rolled out in African countries through the Gavi, The Vaccine Alliance, which led to the decision to administer 24 million doses of the vaccines across 20 African countries.

Professor Melissa Penny and the Global Disease Modelling team.

“But we still have a long way to go – malaria cases rose by 11 million in the space of just 12 months between 2022 – 2023, and it hits hardest in regions where many people lack services to prevent, detect and treat the disease.”
GATES FOUNDATION GRANT SUPERCHARGES RESEARCH EFFORTS
Professor Penny and her team recently received a USD $4.7 million grant from the Gates Foundation to accelerate their work and develop two new linked programs.
The first will focus on updating the team’s flagship OpenMalaria platform, an open-source mathematical model of malaria dynamics co-developed with Professor Nakul Chitnis (Swiss Tropical and Public Health Institute, Switzerland). The program simulates how Plasmodium falciparum – the parasite that causes malaria – infects people, predicts whether cases will be mild or severe, and estimates how infections spread from individuals to mosquitoes and then through the wider community.
“By updating OpenMalaria with the latest data – including how interventions have shifted disease patterns by age and setting over the past 20 years – our team will gain deeper insights into how to better use different existing and novel malaria interventions to support continued reduction of cases,” Professor Penny said.
The second program will apply mathematical modelling, including the use of OpenMalaria, to support the development and testing of new malaria interventions –such as vaccines, medications, and long-acting injectable drugs – by predicting their potential to reduce malaria cases and deaths.
“Together, these programs aim to improve decisionmaking and accelerate progress toward malaria elimination now and in the future,” she said.
“Updates to OpenMalaria will help us to understand the potential impact of new drugs, long-acting preventative treatment, and vaccines, even before they enter clinical trials.
“The models will estimate how many lives these new tools might save and in which African settings.
“The models will also help product developers understand how optimising characteristics of the new tools will lead to greater impact, thus supporting research and development decisions along the clinical pathway.
“This funding also provides a vital opportunity to

collaborate with many partners world-wide from academia, industry and policy who are all dedicated to solving one of the world’s biggest health challenges.”
UTILISING MODELLING APPROACHES TO TACKLE WIDE-SPREAD INFECTIOUS DISEASES
In addition to using modelling research to support WHO policy decisions on new and existing malaria vaccines, funding from Open Philanthropy has allowed the team to utilise the same approach to tackle infectious diseases such as COVID-19, Strep A and respiratory syncytial virus (RSV).
“The work we are doing in mathematical modelling at The Kids Research Institute Australia is helping to change global attitudes about the use of mathematical modelling to control, treat and cure infectious diseases,” Professor Penny said.
“Modelling connects key stakeholders that can bring actionable solutions to global problems in child and adolescent health.”
MALARIA STATISTICS
• 2.2 billion cases of malaria and 12.7 million deaths have been averted since 2000
• In 2023 there were 263 million cases and 597,000 deaths from malaria
• The WHO African region is home to 95% of malaria deaths
• Children under 5 years account for 76% of all malaria deaths in the Africa region WHO aims to reduce malaria deaths by 90% by 2030
Author: Professor Melissa Penny is the inaugural Fiona Stanley Chair in Child Health Research at The University of Western Australia and The Kids Research Institute Australia. Professor Penny currently leads the Global Disease Modelling research team at the Wesfarmers Centre of Vaccines and Infectious Diseases, based at The Kids, and recently moved to Western Australia from Switzerland, where she was Professor at the University of Basel (Switzerland), Swiss Tropical and Public Health Institute (Swiss TPH), and Head of the Disease Modelling Research unit.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
FROM DATA TO ACTION
Turning performance monitoring into actionable dashboards
Harnessing administrative and clinical information stored in hospital electronic medical records, near-real-time dashboards can transform performance monitoring data to relevant, timely and accessible information that drives quality improvement.
Health services use performance monitoring to ensure quality care and patient safety. Measures for this process are defined by Australia’s National Safety and Quality Health Service Standards (NSQHS 2nd Ed). Data-driven performance monitoring at an organisational level often relies on burdensome auditing, data validation, analysis and report curation. Each step consumes valuable human and other organisational resources. Additionally, performance data typically reported in organisational ‘scorecards’ are often based on small samplings, tracking aggregate values that limit exploration, and are produced in timeframes and timelines that don’t align to the everyday workings of busy hospitals and clinicians. This lack of timely access to relevant data can limit those who are best placed to take remedial action and address gaps.













THE ADAPT INITIATIVE
ADAPt, the Actionable Dashboards Accreditation Project, is a collaborative project delivering near-realtime performance monitoring dashboards aligned to the NSQHS standards, against which hospital organisations are mandatorily and regularly assessed. Through the Digital Health Cooperative Research Centre’s collaborative research model, a partnership with Monash University’s Faculties of Information Technology, and Medicine, Nursing and Health Sciences, Eastern Health (EH) a public health service, the Australian Council on Healthcare Standards (ACHS), and the Victorian Department of Health was established. This collaboration of researchers, clinicians, eHealth specialists, accreditation assessors and health policy makers took an iterative co-design approach (figure 1) in contributing, producing and implementing dashboards that improve accreditation readiness and provide a tool for continuous quality improvement. Driven by a user-centred approach, the project actively engaged end-users from the outset to ensure the platform was purpose built to meet real-world needs and deliver meaningful, measurable impact.
TURNING EMR DATA INTO INSIGHTS
Continuous quality and safety assurance requires comprehensive data from across the entire patient population to support accurate, near real-time insights throughout the health service. In more digitally mature organisations, much of this is captured in the Electronic Medical Record (EMR). The ADAPt platform captures and analyses this data at scale (over 3 million clinical events in the first two years), in alignment with the NSQHS standards, including blood product administration, medication prescribing, risk assessments, pathology results, discharge summaries, and clinical handovers. With over 150,000 records per day, the sheer volume of clinical care data captured demonstrates the depth of the dashboards.
FROM ACCREDITATION TO EVERYDAY PRACTICE
The recent shift to health services being provided short notice (24-48 hours) of upcoming accreditation assessments, replacing planned assessments scheduled several months in advance, has highlighted the need for a more agile and effective approach to preparing for the accreditation assessment process. ADAPt addresses this challenge by providing a dynamic interface to visualise and interpret complex data, enabling continuous support for NSQHS assessment activities in a streamlined and efficient manner.
The dashboards provide clear visibility of performance against set targets across specific areas of the hospital network (e.g. wards, clinical teams) enabling early identification of issues and timely implementation of improvement strategies. This continuous visibility of performance data allows for ongoing evaluation of the effectiveness of these strategies, fostering a proactive culture of continuous improvement. This ultimately drives the development of safer, more responsive clinical environments.
The ADAPt dashboards have enabled proactive performance monitoring of clinical standards for undertaking a Best Possible Medication History (BPMH) and Medication Reconciliation (Standard 4 – Medication Safety Standard) continually prior to a short-notice assessment at Eastern Health where the ADAPt platform has been trialled. This early visibility allowed specific areas of the hospital network, such as wards and clinical teams,


to identify proactively in near-real-time opportunities to improve. This shifted momentum towards identifying areas in need of continuous improvement, with teams developing and implementing targeted strategies that lead safer clinical environments. The daily refresh of performance data ensured that the impact of these strategies could be easily evaluated.
Key benefits and value identified by health service staff included:
• Less time spent on manual data collection and preparation
• Data confidence and near real-time access
• Empowering pre-emptive action before formal accreditation and governance processes
• Increased cohort size, data collection points and frequency paints a more comprehensive clinical picture
• Clinical and quality staff shared thoughts on aspects of the dashboards, consistent with the wider experience across all standards.
‘We can observe results 4–5 months earlier now. Previously we would need to wait for point of care audits every quarter, identify improvements and implement these a month later, then wait for another 3–4 months to observe the outcomes. We made changes to VTE [venous thromboembolism] risk assessment forms and could see increased compliance of the number of VTE assessments made across the organisation.’ The project’s success is a result of interdisciplinary and multi-disciplinary collaboration. With the right tools, the right data, and the right partnerships, hospitals can shift from reactive compliance to proactive, patient-centred improvement.
Authors: Cathy Ngo, Agnes Haryanto, Gregory Duncan and Michael Wybrow on behalf of the project team. The project team includes researchers and clinicians from Monash University Faculty of Information Technology and Faculty of Medicine, Nursing and Health Sciences, and Eastern Health. The ADAPt project is supported by Digital Health CRC Limited (DHCRC). DHCRC is funded under the Australian Commonwealth’s Cooperative Research Centres (CRC) program.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL



UNLOCKING THE POTENTIAL OF LINKED DATA FOR CLINICAL TRIALS
MEETING THE NEEDS OF THE CLINICAL TRIALS SECTOR
Australia’s rich population health data—spanning hospitals, registries and health services—is the envy of the world. The Population Health Research Network (PHRN) is now collaborating with industry to transform the way clinical trials are designed and conducted. Strengthening this infrastructure will boost the potential for therapeutic development.
For many working in the clinical trials sector, the benefits of linking health data are clear. It can improve trial efficiency, reduce costs, and strengthen the evidence base. Yet in practice, access in Australia has been challenging. Through national consultations with industry, trialists spoke candidly about the obstacles: long and unpredictable timelines, unclear costs, and fragmented application processes have all acted as barriers to incorporating linked data into trial


design. The consultations also revealed opportunities to enhance PHRN products and services to better meet the needs of the clinical trial sector.
INTRODUCING TRIAL LINK
In 2023, the PHRN received targeted funding to simplify access and build the tools and infrastructure clinical trialists require to use real-world linked data.
Trial Link—a national gateway for discovering, linking, accessing, and learning about linked data—is an important step forward for clinical trials. The project provides clarity on eligibility, costs, and governance, helping to reduce barriers and offering training to researchers new to linked data in both academia and industry.
Trial Link is built around five key pillars: Discover, Data, Link, Access, and Learn. This framework ensures that


from early planning through to governance and analysis, researchers can navigate the process with greater transparency and efficiency. Consumer input helps guide the PHRN’s understanding of community concerns and the factors that underpin public confidence in using data linkage in trials.

Insights from sector consultations sparked a range of initiatives that are now underway. These include the routine linkage of clinical registries, tools to estimate sample size and costs, pilot access to aggregate data for rapid feasibility checks, and new online training resources. Together, these initiatives are designed to provide researchers with the right tools to plan clinical trials.
BOOSTING TRIALS WITH LINKED DATA
Clinical trials stakeholders report that connecting trial data with routinely collected sources— including hospital


admissions, emergency records, Medicare and PBS data, and mortality data across a trial’s lifecycle— can significantly enhance their research.
These benefits span the entire trial journey: from study justification and feasibility assessments, to recruitment, conduct, and data collection; from long-term follow-up and monitoring to validation of results. Augmenting trial data with linked administrative records can add critical insights beyond those available within the trial itself and offer benefits to both patients and investigators.
For example, a cardiovascular trial may only have funding to follow patients up for two years posttreatment, whereas having access to linked hospital and mortality data could allow researchers to track outcomes for more than a decade—without the cost and burden of re-contacting participants. We’ve seen examples where linked data has revealed important harms or benefits that were not evident within the original trial timeframe. Such insights provide critical data for establishing the evidence base for new therapies and informing health service planning.
LOOKING AHEAD
Trial Link represents an industry-first in unlocking the potential of Australia’s linked health data. By making data more discoverable, accessible, and usable, PHRN is supporting faster, more efficient clinical trials that deliver better evidence and ultimately better health outcomes for Australians. These developments also position Australia as a world leader in clinical trial data infrastructure—helping to attract investment, strengthen research capability, and accelerate the translation of discoveries into real-world therapies.
Authors: Dr Kate Miller (Senior Advisor, Science and Partnerships) and Dr Felicity Flack (Senior Manager, Strategy and Services), are part of the Population Health Research Network, funded through the National Collaborative Research Infrastructure Strategy. Article submitted by the University of Western Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
“IT’S NOT JUST ELECTRONIC PAPER”
– DIGITAL
HEALTH
RESEARCH TO TRANSFORM
HEALTH
TRANSLATION

Digitalised medical record systems are driving new ways to undertake research within hospitals and health systems with new opportunities to rapidly translate in regional Queensland.
The transformation of health systems to leverage Electronic Medical Records (EMRs) is delivering a new era of health research equity and enquiry.
While these digital systems hold immense, largely untapped potential, the reality for many healthcare institutions is that they often function as little more than ‘electronic paper’—static digital filing cabinets rather than dynamic tools for discovery. For time-poor clinicians on the front lines, the valuable aggregated data captured during every patient interaction frequently remains inaccessible, locked away behind fragmented governance and complex data extraction procedures. This failure to release aggregated health data creates a critical gap between the data we collect and our ability to translate it into practice, representing a lost opportunity to accelerate clinical excellence.

ENABLING DIGITAL HEALTH RESEARCH: THE ESTABLISHMENT OF THE RESEARCH DATA LABORATORY
In response to this challenge, the Townsville Hospital and Health Service (THHS) has pioneered a model that treats the EMR not as ‘electronic paper’, but as the foundation for a dynamic research capability. Through the establishment of the Research Data Laboratory (RDL), THHS has built an integrated service that provides the essential infrastructure, governance, and data extraction expertise clinicians need to translate their frontline insights into evidence. Crucially, the RDL was designed not as a static data repository, but as an end-to-end service built to overcome the key barriers clinicians face. It achieves this by combining three critical elements: a clear and secure pathway for data access that navigates the complex governance landscape; powerful, analysisready infrastructure and tools; and dedicated expert support throughout the project lifecycle. The impact of this model is tangible. In its first three years of operation, the RDL has facilitated over 772 requests for research and quality improvement projects, with a median data delivery time of 5 business days. This responsive turnaround not only accelerates the pace of inquiry but fosters a culture where data-driven questions are actively encouraged, not hindered by delay.
By removing these fundamental barriers, the RDL empowers clinicians to investigate local healthcare challenges, turning their insights into evidence-based improvements in patient care. Take, for example, a recent study on hyperkalaemia, a common but serious condition. Using the RDL, clinicians were able to rapidly analyse five years of data from nearly 100,000 hospital admissions. The study revealed that treatment for the condition was often inconsistent and identified specific high-risk groups, such as patients with kidney failure, who experienced more severe episodes. Armed with this evidence, the health service can now implement more robust monitoring and standardised treatment protocols, directly translating research findings into improved patient safety at the bedside.
SCALING TO MEET DEMAND: PARTNERING FOR STATEWIDE IMPACT
While the RDL model proved highly effective locally, the true potential for transformation lies in collaboration and scale. A successful local innovation achieves far greater impact when integrated into a broader network. The key to this expansion has been a unique partnership with the Queensland Digital Health Centre (QDHeC), a state-wide collaborative established by The University of Queensland and Queensland Health to build a true learning health system. This partnership creates a powerful synergy:

the RDL acts as the specialised, on-the-ground engine, deeply embedded within the health service, while QDHeC provides the overarching network and strategic platform to connect multiple entities across the state. This is achieved through tangible initiatives, such as QDHeC’s SMART Hub, which provides a framework for streamlining data extraction from the state’s electronic medical record system for research, and its KeyPoint platform, which offers secure infrastructure for sharing sensitive data. By aligning the RDL’s local data expertise with QDHeC’s state-wide infrastructure and governance, this partnership elevates the model from a site-specific solution to a statewide asset. It enables research questions to be answered at a much larger scale by comparing data and outcomes across different regions and diverse patient populations—a crucial step in building a truly learning health system for Queensland.
THE FUTURE IS COLLABORATIVE: A NEW ERA FOR HEALTH TRANSLATION
This integrated, multi-level approach represents a new blueprint for embedding research directly into the fabric of daily healthcare. The journey from a passive EMR system to an active and localised data laboratory, and now to a powerful, collaborative statewide network, demonstrates a clear pathway to unlocking the full potential of digital health. By prioritising streamlined governance and building supportive infrastructure, this model is accelerating clinical excellence and delivering research translation into practice every day. It offers a robust and replicable framework for other health services in Australia to transform their own digital systems from simple records into powerful tools for discovery and, most importantly, for delivering better health outcomes for all Australians.
Authors: Professor Andrew Mallett is a leader in kidney medicine, clinical genomics and translation across those spaces. He is a Professor of Medicine and Head of Discipline for Internal Medicine at James Cook University. Professor Clair Sullivan is an internationally recognised leading practising and academic clinical informatician. She is Director of the University of Queensland’s Queensland Digital Health Centre, and the University’s inaugural Professor of Digital Health. Rudolf Schnetler is the Research Data Laboratory Lead and a Research Fellow in Townsville, Australia. He heads the Research Data Laboratory (RDL) at the Townsville Hospital and Health Service.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
HOW HEALTHCARE DATA IS IMPROVING CANCER DIAGNOSIS AND OUTCOMES
By VCCC Alliance and University of Melbourne staff


Natalie Diepenhorst, a lived experience advocate at VCCC Alliance
Victorian Health Minister, the Hon Mary-Anne Thomas MP, officially launched Data Connect in 2023. L-R: VCCC Alliance CEO Prof Grant McArthur AO, the Hon Mary-Anne Thomas MP, Senior Research Fellow, Data Connect, Fanny Franchini; and former Data Connect Chair Prof Jon Emery.

Natalie Diepenhorst was diagnosed with colorectal cancer at 32, following a prolonged period of unexplained anaemia that began after the birth of her first child. Despite persistent symptoms including fatigue and frequent headaches—common during pregnancy—Natalie continued to feel unwell for months after giving birth.
‘Everyone told me I was supposed to feel tired because I was a new mum,’ says Natalie, who is a lived experience advocate with the VCCC Alliance.
Concerned that her symptoms persisted nine months after the birth, Natalie consulted her doctor. Blood tests confirmed severe anaemia, and she underwent three iron infusions, with no improvement. After a further nine months without resolution, her care team decided to investigate more deeply with a colonoscopy.
The colonoscopy revealed colorectal cancer—an outcome that shocked even her gastroenterologist. ‘He was stunned by what he found and went above and beyond to ensure I received urgent and appropriate care.’
Natalie’s diagnosis came nearly 18 months after her symptoms first appeared. Her experience highlights the challenge of distinguishing cancer symptoms from those commonly experienced during and after pregnancy, especially in younger adults, and underscores the need for greater awareness and earlier investigation of persistent symptoms.
THE IMPORTANCE OF CONNECTED HEALTH DATA
The VCCC Alliance-led Data Connect program, spearheaded by the University of Melbourne Cancer in Primary Care team, has accessed anonymous information on two million patients and their 78 million medical visits.
This is the first and only example of using linked data from general practice for cancer research in Australia, addressing a major gap in Australian health and medical research.
Access to linked datasets is allowing researchers to examine cancer across the whole journey for more than 600,000 people in Victoria who have had cancer – from pre-diagnosis to diagnosis and treatment, through to treatment, post-treatment, and palliative care.
The type of data collected includes reasons for visiting the GP, prescribed medicines, current and long-term illnesses, test results, height, weight, blood pressure, and lifestyle factors, like smoking. Data from hospitals and clinical registries includes information about treatments, cancer stage and survival.
OPENING THE WINDOW TO EARLIER CANCER DETECTION
One of the most significant findings from the Data Connect program to date is the identification of early detection windows for some of Australia’s most common cancers.
Dr Meena Rafiq is looking at patterns in general practice activity – such as frequency of visits, test ordering, and test results – to figure out how far diagnosis could potentially be brought forward. She found that lung and colorectal (large intestine and bowel) cancer could be diagnosed earlier
Diagnosis of sarcoma—a rare and deadly bone cancer mostly affecting young people— advanced and multiple imaging requests up to six months before diagnosis.

Based on these findings, Dr Rafiq developed a clinical tool supporting GPs to further investigate markers including bone pain and soft tissue lumps.
Another team member, Dr Jianrong Zhang, interrogated the data to find that the lung cancer diagnosis process typically takes more than 100 days.
This is at odds with optimal care guidelines for lung cancer, which recommend a maximum of 35 days from the first visit to the GP to diagnosis.
Dr Zhang also found that patients who wait longer than 70 days for a lung cancer diagnosis have worse survival outcomes, highlighting the crucial need to support GPs with more efficient diagnostic processes.
And, interestingly, a study led by Dr Allison Drosdowsky has revealed a ‘waiting time paradox’ for colorectal cancer. This is also known as the ‘sick quick’ phenomenon, where those with more advanced cancer present with more severe symptoms and are fast-tracked through the health system but have poorer outcomes despite faster diagnosis. Dr Drosdowsky’s work suggests 120 days should be the maximum waiting time for colonoscopy for people with less severe symptoms.
FROM DATA TO BEST PRACTICE
The Data Connect program is enabling researchers to reshape cancer care.
With a detailed look at this data, researchers have identified gaps in care and developed strategies to address these, improving outcomes for patients and supporting healthcare teams on the ground to provide best-practice care in line with the optimal care guidelines.
Data Connect is producing evidence-based tools, enabling GPs to identify potential cancer cases earlier with pointof-care digital health interventions like Future Health Today
Author: The VCCC Alliance Data Connect program is spearheaded by The Cancer in Primary Care team – based at the Melbourne Medical School and Collaborative Centre for Genomic Cancer Medicine, a joint venture of the University of Melbourne and Peter MacCallum Cancer Centre.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
AUSTRALIA RISKS FALLING BEHIND IN THE SYNTHETIC HEALTH DATA RACE

While global competitors invest millions in privacy-preserving synthetic electronic medical records, Australia’s fragmented state-by-state approach threatens to undermine our research capabilities and economic competitiveness. The technology exists, but without national coordination, we are missing a critical opportunity.
Imagine creating medical datasets that resemble real patient records but contain no actual patient information. Data that researchers can use to develop AI models, test hypotheses, and accelerate discoveries without waiting months or years for ethics and governance approvals.
That is the promise of synthetic health data: artificially generated information that mimics the statistical properties of real data while protecting individual privacy.
South Australian researchers recently achieved a remarkable feat: they recreated the results of complex pancreatitis studies using purely synthetic patient data in just days, rather than the months or years typically required for ethics and governance approvals. This breakthrough, enabled by SA Health’s partnership with Gretel AI, demonstrates how synthetic electronic medical records (EMRs) can unlock health and care innovation while protecting patient privacy. Yet this success story highlights a troubling reality. While SA Health pioneers ahead and WA Health experiments with synthetic data

hackathons, Australia lacks the unified national strategy that our international competitors have embraced, and they are racing ahead.
THE GLOBAL INVESTMENT SURGE AUSTRALIA IS MISSING
The US is spending substantial federal resources across multiple agencies. The FDA’s medical imaging AI program develops regulatory frameworks for synthetic image data, while AHRQ released a nationally representative synthetic claims database covering Medicare, Medicaid, and commercial insurance. The 2022 FDA Modernization Act 2.0 permits the use of synthetic data for drug development, signalling regulatory acceptance.
The EU launched SYNTHIA , a €22.4 million initiative involving partners from academia, the pharmaceutical sector, and the healthcare sector. Germany’s BfArM Health Data Lab examines the use of synthetic data for health insurance billing.
In Canada, Health Cities Edmonton validated a synthetic health dataset using records from 100,000 Albertans, while CanPath launched a synthetic dataset with over 40,000 observations for research and education in 2024. Israel’s MDClone platform, serving major international health systems, launched with over 50 million synthetic patient records in 2022.
Some universities and research institutions now waive the ethical review requirements for studies that use synthetic rather than human data. This regulatory change accelerates research timelines but also highlights the need for robust governance frameworks. As noted in recent commentary, while synthetic data supports hypothesis generation and preliminary testing, risks such as patient re-identification and model collapse require rigorous validation standards and transparent reporting protocols.
THE AUSTRALIAN PARADOX: CAPABILITY WITHOUT COORDINATION
Australia has the technical capability and institutional knowledge to be a leader in synthetic health data. SA Health supports 27,000 practitioners across a network of 1.8 million people, with ethics committees now approving synthetic data projects within days. South Australia is also the first state to complete statewide EMR implementation, generating over 1.5 million electronic medical records since 2019, which provides a strong foundation for highquality synthetic data.
However, this capability highlights our deeper problem: Australia’s healthcare data system remains hindered by fragmentation. Researchers face data access delays of up to two years despite policy rhetoric about open data. Hospital datasets sit under state custody while Medicare and PBS data remain with the Commonwealth. When patients cross state borders for care, their records
don’t follow. No single source holds sufficient data for comprehensive policy analysis.
This fragmentation has real costs. Researchers must navigate 20 distinct data collections for a single project. The extreme complexity means only time-rich, well-funded teams can access the data needed for breakthrough research, leaving smaller institutions and innovative startups locked out.

A COORDINATED PATH FORWARD IS EMERGING
The Synthetic Data (SynD) community of practice, coordinated by the Digital Health CRC, represents Australia’s first coordinated national response to synthetic data. With founding members and collaborators spanning all Australian states and territories, including representation from health departments, universities, policy experts and consumers, SynD has developed the governance framework, evaluation standards, and knowledge infrastructure Australia desperately needs.
The framework addresses legal and ethical assessments, technical validation checks, privacy risk management using the Five Safes framework, and guidance for data use agreements. It provides a blueprint for safe and effective synthetic data implementation across jurisdictions.
Yet, a framework alone is not enough. We urgently need policymakers and data custodians to act. State health departments must adopt synthetic data generation and sharing capabilities and implement governance frameworks. Federal leadership must establish national coordination mechanisms and legislative clarity. Researchers must advocate for balanced perspectives. Synthetic data presents legitimate privacy risks, bias concerns, and validation challenges that demand rigorous oversight, not passive acceptance.
The technology exists. The governance framework has been developed. The time is right for policymakers, researchers, and healthcare leaders to mobilise, prioritise national coordination, and ensure Australia leads in synthetic health data innovation. We need to act today to secure our place at the forefront of healthcare transformation.
Authors: Dr Amir Marashi, Data scientist, Digital Health CRC, Professor James Boyd, Chair of Digital Health and Innovation, La Trobe University, Dr Melanie Haines, Education and Workforce Director, Digital Health CRC, and Dr Ben Hachey, AI Strategic advisor. This project is supported by the Digital Health CRC Limited (DHCRC). DHCRC is funded under the Australian Commonwealth's Cooperative Research Centres CRC Program.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
IBD-PERFECT
Turning Data into Better Care

Aof care are measured. Built on CCCare — a worldfirst point-of-care digital platform and linked Clinical Quality Registry — IBD-PERFECT is fasttracking national standards and delivering on the recommendations of the State of the Nation in IBD report.

FROM REPORT TO REFORM
The first State of the Nation in IBD report revealed a stark reality: despite world-class clinicians and therapies, Australians living with Crohn’s disease and ulcerative colitis face inequitable and inconsistent care. Too often, care depends on where a person lives rather than what they need. The report called for a coordinated national response with clear standards.
Crohn’s Colitis Cure (CCCure) responded with IBDPERFECT, Australia’s first national benchmarking exercise designed to improve the quality and consistency of IBD care.
Remarkably, this program was developed without government funding, driven instead by clinicians, researchers and consumers, and funded by CCCure and partner pharmaceutical companies, who recognised that innovation could not wait.
This leadership contrasts with the structural bias in federal funding, which continues to prioritise clinical quality registries for acute and high-mortality conditions, even though chronic diseases now account for more than 80% of Australia’s total health expenditure. Meanwhile, IBD prevalence continues to rise, and is projected to soon to affect 1% of Australians, consistent with trends seen across western nations.
THE POWER OF POINT-OF-CARE DATA
At the heart of IBD-PERFECT is Crohn’s Colitis Care (CCCare), CCCure’s world-leading, disease-specific digital health platform. Used daily by clinicians and patients at the point of care, CCCare captures comprehensive clinical data, treatment decisions, and patient-reported outcomes during routine IBD consultations.
This information flows securely through a linked clinical quality registry in near real-time, eliminating double data entry and the delays and costs of traditional registries. Clinicians receive timely feedback, and consumers can be confident that trends in care are identified and addressed in real-time, improving safety, consistency, treatment decisions, progress monitoring, and care delivery.
By embedding CCCare directly in the clinical workflow,
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
everyday practice becomes continuous learning, closing the gap between data collection and care improvement
‘Traditional registries look backward. CCCare and IBDPERFECT let us look forward. We can improve care now,’ says Professor Jane Andrews, an internationally respected gastroenterologist and Co-Chair of the Global IBD Registry (GLIDE ).
BENCHMARKING WHAT MATTERS
IBD-PERFECT compares performance across participating sites using indicators that reflect care quality and are actionable now. Initial indicators include steroid & opiate use, anaemia, smoking, clinically active disease, and colorectal cancer screening practices. As the program expands, it will add modules to report against quality of life and life participation issues, healthcare utilisation, paediatric & transition care, along with quality in IBD surgery.
Rather than a top-down audit, it is a collaborative learning system. Each site receives a performance dashboard, enabling clinicians to identify variation, share best practice, and co-design targeted quality-improvements.
Because the data are collected and reported in real time, improvements can be made now, not in the next audit cycle. This dynamic feedback loop — measure, compare, improve — is what makes IBD-PERFECT more than a benchmarking project, it is an engine for clinical excellence, designed around the proven principle of a plan-do-study-act cycle.
The program’s first national report is due for release in early 2026 and will provide Australia’s most comprehensive picture of IBD care quality to date, laying the groundwork for national standards.
ACCELERATING THE IMPLEMENTATION OF NATIONAL STANDARDS
One of the most powerful aspects of IBD-PERFECT is that it operationalises the State of the Nation recommendations. By embedding standardised measures and quality indicators within CCCare, the program provides a ready-made pathway to implement national IBD standards rapidly and consistently across the country.
IBD-PERFECT is also a mechanism for reform. It allows the health system to test, refine, and scale standards in a real-world setting, using data already being collected in routine care.
This approach aligns directly with national initiatives including the Australian Commission on Safety and Quality in Health Care’s CQR Framework and the Australian Digital Health Strategy.
DRIVING MEASURABLE IMPACT
Economic modelling shows that IBD-PERFECT delivers significant value to both the health system and the community. Across the first 20 CCCare sites, the program has improved adherence to evidence-based care, a 12% reduction in avoidable hospital admissions and surgeries and proves that real-time benchmarking translates directly into better outcomes.
When scaled nationally, IBD-PERFECT is projected to deliver more than $100 million in annual savings, reflecting a 20:1 direct return on investment (ROI) and a total ROI exceeding 40:1 once indirect economic benefits such as productivity gains, reduced carer burden, and improved quality of life are included.From a broader social perspective, the social return on investment surpasses $500 for every $1 invested. This is clear evidence that improving access, consistency, and quality of care delivers both economic and human dividends.
CONNECTING AUSTRALIA TO GLOBAL RESEARCH
CCCure’s participation in the Global IBD Registry (GLIDE ) ensures that Australia’s IBD data can also influence knowledge via inclusion into a truly global learning ecosystem.
GLIDE connects national and regional registries across Europe, India, Australasia, and the Americas, enabling privacy-preserving, cross-border collaboration to compare outcomes, identify best practice, and drive innovation.
Through GLIDE, CCCare and IBD-PERFECT ensure that Australian insights both shape and are shaped by the world’s best IBD care.
REDEFINING RESEARCH: FROM UNMET NEED TO INNOVATION
IBD-PERFECT represents what the future of research can be. By analysing de-identified, real-world, real-time data from across Australia, researchers can pinpoint unmet patient needs, uncover variation, and rapidly test new interventions.
The integrated model enables learning while doing, where every clinical encounter contributes to discovery and improvement. It bridges the bench, bedside, and community, creating a living evidence base for innovation.
For partners across academia, industry, and policy, it offers a ready platform for co-creation, accelerating translational research, validating digital biomarkers, and advancing precision care. In doing so, CCCare and IBDPERFECT demonstrate that real-time data can power better care and better science.
A VISION FOR THE FUTURE
For patients, IBD-PERFECT means greater transparency, accountability, and confidence that their care meets national best practice. For clinicians, it provides actionable feedback. For researchers, a living laboratory for discovery. And for health systems, a scalable, standardsbased model combining clinical quality, digital innovation, and economic sustainability.
Author: Bill Petch, CEO, Crohn’s Colitis Cure. With insights from Professor Jane Andrews, Chair & Medical Director Crohn’s Colitis Cure and Co-Chair, Global IBD Registry (GLIDE).
Visualisation of medical professionals using the IBD-PERFECT platform



CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
IT’S NOT MAGIC!
The MERLIN project accelerates child health research

Child health datasets can be uniquely sensitive. At the University of Western Australia, a multidisciplinary team, led by Professor Britta Regli-von Ungern-Sternberg, is addressing this challenge.
SMALL PEOPLE, BIG DATA
Born into a computer age, with increasingly digital and connected health systems, the children of today will collect huge volumes of health data throughout their lives in a manner that no previous generation has done.
Both the quantity and the quality of this data – often preserved in its source format rather than simply summarised as a report – mean that the health
The MERLIN project received a Merit Award at the 2025 INCITE Awards, WA’s longest-running and premiere technology awards
‘informationome’ is expanding in ways that surpass the ability of even most diligent and invested health professionals to grapple with.
Digital medical records and integrated medical devices also multiply the potential data sources that can be drawn upon for research. Studies that previously captured data at intervals of minutes can now include sub-second resolution across all parameters. Previous tomes of impenetrable doctors’ scrawl locked away in a basement compactus are now replaced with digital text that can be searched, indexed, and retrieved nearly instantly.
However, faced with swelling data banks and medical records teeming with a variety and multitude of documents, there are new challenges. The quantity of data for a single
patient will exceed the ‘human context window’ – the amount that any person could hold command over while contemplating a decision or course of action.
LISTENING TO CHILDREN & FAMILIES
A changing information landscape may appear to call, ipso facto, for a pivot to a big data model – demanding the slurping in great swathes of child health data to generate insights and guide care. However, this landscape is accompanied by well-founded concerns for the well-being of children and the protection of their data by families and by society at large. These concerns extend to emerging technologies, including many kinds of artificial intelligence that are providing new ways to tackle data challenges.
Engaging with young people and families and addressing these concerns is key. Exploring these new technologies together is crucial.
A NOVEL COMPUTING MODEL
In order to begin this journey, the MERLIN project provides a unique platform, including a world-first embedded supercomputer and a suite of tailored computing tools, specifically for child health research. The highperformance computing capabilities of MERLIN enable new approaches to data analysis, including artificial intelligence, to be applied in a secure and readily explainable setting.
By being physically located within Perth Children’s Hospital, the MERLIN computing infrastructure allows researchers to perform cutting-edge artificial intelligence and machine learning techniques without data ever leaving the hospital walls. This represents a revolution – rather than an evolutionary step – in the approach to child health information research, providing the flexibility to explore data with a wide range of tools that were previously inaccessible.
The MERLIN model is ‘bring the supercomputer to the data’ and aligns with consumer expectations, health sector strategies for data-driven care and innovation, and the practical requirements of acute paediatric care. This model has been facilitated by the transformative nature of current-generation GPU accelerators, including the NVIDIA H100 GPUs that power MERLIN.
New capabilities, however, don’t invalidate the learnings of ‘big iron’ supercomputing. Maintaining a rigorous approach to computing governance, data safety, and optimal resource utilisation is critical to ensure value is achieved. MERLIN takes these learnings, using the same software systems used in major national supercomputers such as Pawsey’s Setonix, adapted for our specific security requirements.

CRITICAL CAPABILITIES
Ongoing advances in child health research will increasingly rely on large datasets and artificial intelligence methods. Involving children and families on this journey is requisite to ensure that the methods and objectives serve their needs.

Developing and maintaining the capabilities – both in human skills and infrastructure – to perform this research and to innovate in research methodology will determine if consumer needs can be genuinely embraced and reflected in how child health research is conducted. MERLIN provides this flexibility and also supports a pipeline to develop capability and experience.
MERLIN embraces both new advances in computing technology and the needs of children and families to advance child health and wellbeing. Continuing this innovative journey, in partnership with consumers, will help researchers to give every child the best chance to a happy and healthy start to life.
ACKNOWLEDGEMENTS
The MERLIN project is kindly funded by a project grant from the Stan Perron Charitable Foundation.
Authors: Dr Harry Smallbone leads the technical implementation of MERLIN at Perth Children’s Hospital as a medical AI expert. Harry is a physician trainee at Fiona Stanley Hospital and is completing his PhD studies in medical AI and robotics at the University of Western Australia.
Dr Thomas Drake-Brockman is a rural generalist and GP trainee from country WA, with a dual background in computer science and medicine. Thomas has appointments at Perth Children’s Hospital, The Kids Research Institute Australia, and in Medical School at the University of Western Australia, where they are also completing a PhD.
Professor Britta Regli-von Ungern-Sternberg AM FAHMS is an internationally recognised authority in paediatric anaesthesia and a national leader in consumer engagement in research. She holds the Foundation Chair of Paediatric Anaesthesia at the University of Western Australia and supervises the MERLIN project as part of an extensive program of clinical and translational research. Article submitted by the University of Western Australia.
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
ZERO CHILDHOOD CANCER DATA LAKEHOUSE: ENABLING SCALABLE,
TIMELY INSIGHTS
The volume of biomedical data is growing at an exponential pace, challenging even the most advanced research institutions.

Kelly, Ryder, Alan and Charlese
At just 10 months old Ryder was diagnosed with a serious and difficult to treat brain tumour. In 2023 he was enrolled in the Zero Childhood Cancer Program (ZERO) which has significantly increased his chances of survival. Today, Ryder is a normal four-year-old boy who loves life and has no idea how sick he has been.
L-R


Nowhere is this more apparent than in precision oncology, where clinicians and scientists need to combine huge amounts of molecular and clinical data so they can be interrogated to find lifesaving discoveries for cancer patients today.
Supported by Biology research group at Children’s Cancer Institute (CCI) is at the forefront of data-driven precision oncology innovations. The team currently manages more than two petabytes of cancer patient data (equivalent to 2000 terabytes) from over 2800 patients participating in the Zero Childhood Cancer Program (ZERO), a world leading precision medicine program. Thanks to recent federal funding, the team needs to scale their data systems to help ~4000 more young Australians in the coming three years, likely 5 petabytes of biomedical Big Data.

FROM WAREHOUSES TO LAKEHOUSES: WHY CHANGE WAS NEEDED
For several years CCI relied on a combination of a Data Lake and a Data Warehouse to support finding answers for patients enrolled in ZERO. The process began with raw and processed genomic data being stored as large files in a Data Lake (a system that functions like a giant digital filing cabinet). From there, selected data was tidied, transformed, and loaded into a MySQL Data Warehouse, which organises information into structured tables (think giant spreadsheets), like cataloguing books neatly onto library shelves.
This hybrid model worked reasonably well for a restricted set of predefined genes, allowing the system to remain reliable and responsive for clinical reporting. However, the trade-off was significant: most of the molecular information remained locked away, out of reach for research and discovery. Keeping public reference data current was also challenging, as these datasets are both large and rapidly changing.
As research expanded beyond this limited gene panel, the cracks became clear:
• Data remained locked in unwieldy files in the Data Lake, limiting discoverability and making cohort-scale queries impractical.
• Cohort-wide analysis depended on brittle custom data pipelines, akin to searching the entire filing cabinet by hand.
• Keeping reference datasets in sync locally was cumbersome and required substantial storage and compute resources.
The result was a split system: a polished, efficient warehouse for a narrow slice of the data, and a cumbersome, file-based approach for everything else. What researchers needed was a single platform that could combine the scale of the Data Lake with the reliability of
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
the warehouse, while providing the flexibility to explore and analyse the entire genome.
THE LAKEHOUSE SOLUTION
Growing from an honours project, over the course of two years the Lakehouse architecture was designed and implemented on the Microsoft Azure Databricks platform. At its core, the Lakehouse combines two key technologies:
• Delta Lake/Delta Tables provide reliable storage, with molecular and clinical data kept in compressed files that preserve integrity while allowing fast queries. These are represented as ‘Delta Tables,’ which can be treated like database tables but at cloud scale.
• Apache Spark delivers distributed computing by breaking large workloads into parallel tasks across thousands of cores. Analyses that once took days can now be completed in minutes or even seconds.
A key advantage of this architecture is that storage and compute are decoupled. This means we can use the virtually unlimited scalability of cloud storage to hold vast molecular datasets, while Spark provides the flexibility to run far more complex computations (e.g. machine learning algorithms) than a traditional Data Warehouse can support. By separating these two layers, the Lakehouse delivers both cost efficiency and analytical power, unlike traditional warehouses where storage and compute are tightly bound, driving up costs and limiting scalability.
PROCESSING BIOMEDICAL DATA AT SCALE
To manage Delta Tables effectively, CCI applies the medallion architecture, refining data through three stages:
1. Bronze tables (Raw Tier) capture ‘raw’ outputs and clinical metadata as a complete, auditable record.
2. Silver tables (Processing Tier) clean, harmonize, and annotate bronze data, enabling consistent crosscohort analysis and reuse.
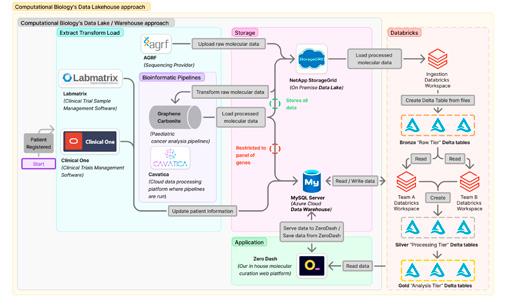
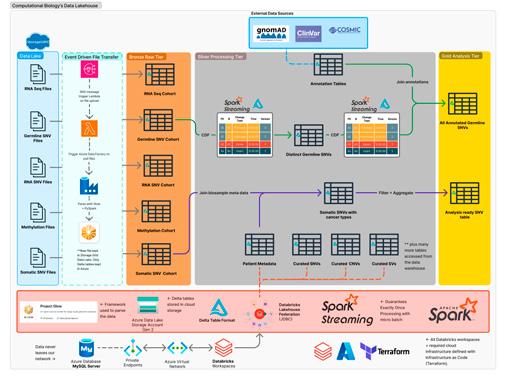
Figure 1. In the Data Lake+Warehouse model, only a subset of processed molecular results can be stored in the MySQL warehouse for fast access, with full datasets left in the Data Lake. Our Data Lakehouse approach overcomes this by storing all processed data as Delta Tables, which can be enriched with warehouse data and either written back to the warehouse or queried directly at scale with Spark.
Figure 2. Files are ingested through an event-driven pipeline into cohort tables organised by molecular data type (RNA, SNV, methylation). These bronze-tier tables are enriched with patient information and may include additional mutation types from the Data Warehouse. Using Delta Tables’ change data feed (CDF) with Spark Streaming, we can process only new or updated records, making annotation of large cohorts far more efficient.
3. Gold tables (Analysis Tier) provide trusted, analyticsready datasets for advanced research and machine learning, building from silver tables.
This layered approach ensures the data is trustworthy, reusable, and easy to build on. It also means different teams can work on different layers at the same time, like chefs in a kitchen each handling different steps of a recipe.
By organising data this way, we can train and test machine learning models much faster and with greater reliability. Delta Tables let us keep track of exactly which version of the data was used, ensuring results can be reproduced. And because Delta integrates directly with Spark, we can train models on massive datasets in parallel, cutting down on time and cost.
Together, the medallion architecture and Delta Tables turn an overwhelming ocean of biomedical data into an organised foundation for building scalable, reliable, and clinically useful artificial intelligence.



IMPACT ON RESEARCH AND CARE
The Lakehouse is more than a technical upgrade; it is a strategic enabler for cancer research and clinical care. By transitioning to a Lakehouse, CCI has unlocked the ability to run queries across more than 12 billion genomic variants. Importantly, this capability extends beyond DNA data to RNA, epigenetics and other clinical information. And this data infrastructure will underpin an ambitious project to share ~5PB of federated, harmonised genomic and clinical data from 6000 high-risk childhood cancers with European and Canadian partners.

For clinicians, this translates into faster identification of patient-specific mutations and biomarkers. For researchers, it means the ability to test hypotheses quickly and reliably across massive datasets. And for patients and families, it brings the promise of more precise, timely, and effective treatments.
CONCLUSION: BUILDING THE FUTURE OF PRECISION MEDICINE
The Lakehouse architecture has transformed how biomedical data is managed, processed, and used within the Zero Childhood Cancer Program, providing a platform that is scalable, reliable, and ready for the future of oncology research. In the era of precision medicine, where data is as critical as diagnostics or therapies, this implementation ensures ZERO can keep pace with both scientific discovery and patient need. Most importantly, it shows how the Lakehouse can turn overwhelming molecular and clinical data into actionable insights. This successful use case demonstrates that the same approach can be applied to other clinical and research settings that face the challenges of large, complex, and diverse datasets.
Authors: James Bradley is a Junior Bioinformatics
Data Engineer at Children’s Cancer Institute. James specialises in big data processing, leveraging dual degrees in software engineering and bioinformatics. He architects Databricks-based lakehouse strategies that transform petabytes of molecular data into queryable, AI/ML-ready resources. With a passion for scalable systems, James bridges biology and technology to unlock insights that drive discovery and innovation. Associate Professor Mark Cowley is Deputy Director (Enabling Platforms and Collaboration) at Children’s Cancer Institute. In 2018, Mark joined the Institute to establish the Computational Biology Group. He now holds several leadership positions, including Head of the Luminesce Alliance Data Enabling Platform, co-Head of the ACRF Childhood Cancer Liquid Biopsy Program, and President of Australasian Genomic Technologies Association (AGTA), the peak body in the region. Article submitted by Luminesce Alliance.
James Bradley
Associate Professor Mark Cowley
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

NEW MEMBER PROFILES
Research





AVANT AVANT IS A MEMBER-OWNED ORGANISATION FOUNDED BY

A GROUP OF DOCTORS IN 1893.
We are Australia’s largest medical indemnity insurer, protecting over half of all doctors, and we offer an extensive range of products and services to support them in their professional and personal lives.
Our focus is on delivering high-quality insurance products, financial and legal services specifically for doctors, as well as solutions for medical practices. This is done in the context of our commitment to a sustainable health system and societal wellbeing.
We have three specific objectives to:
1. Support research that will improve quality, safety, sustainability and professionalism in the practice of medicine
2. Fund programs working with medical practitioners to improve patient care and health outcomes
3. Help doctors overcome their experience of disadvantage in the pursuit of their medical or healthcare studies.
Today, Avant represents over 90,000 medical practitioners and students, across every state and territory. No one knows better than us, the unique context and challenges doctors face.
AVANT FOUNDATION

Avant Foundation’s purpose is to create transformational change in healthcare through medical research, medical education and health programs.
Avant Foundation has four annual grant programs to help us fulfil our impact objectives: Member Grants, Research Grants, Bursaries and Transformation Grant. To find out more visit our website www.avant. org.au/foundation
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL


HEPATITIS AUSTRALIA
LEADING THE NATIONAL RESPONSE TO VIRAL HEPATITIS
Hepatitis Australia is the national peak community organisation for people affected by viral hepatitis and Australia’s state and territory community hepatitis organisations.
A VISION FOR ELIMINATION
In Australia, almost 300,000 people are living with hepatitis B and hepatitis C. These are preventable and manageable conditions, yet every year 1,000 people die with viral hepatitis. Viral hepatitis is the leading cause of liver cancer, Australia’s fastest growing cause of cancer death.
All Australian governments have committed to eliminating viral hepatitis as a public health threat by 2030. No one should be dying from hepatitis anymore.
Our vision is to eliminate viral hepatitis for all. Our purpose is to enable everyone to live free from the impact of viral hepatitis.
This is achieved through evidence-based advocacy, strategic partnerships, and community-led initiatives that prioritise the voices and needs of those most affected.
STRATEGIC LEADERSHIP AND NATIONAL IMPACT
Hepatitis Australia plays a central role in shaping Australia’s public health response to viral hepatitis. Working with governments, health services, researchers, and community partners, the organisation leads coordinated action to increase testing, improve access to care and treatment, and eliminate stigma and discrimination.
Our work is evidence-led and supported by our expert Research and Clinical Advisory Committee. The group consists of experts in hepatitis B, hepatitis C, research, evaluation, public health, and related fields, who provide strategic guidance and advice on Hepatitis Australia’s programs and policy work and the work of its members.
National awareness initiatives such as World Hepatitis Day have helped to mobilise communities and raise visibility of viral hepatitis as a pressing public health issue.
OUR MEMBERS
Hepatitis Australia’s member organisations are the eight state and territory community hepatitis organisations. They are an important part of achieving hepatitis elimination in Australia, providing education, outreach, and treatment support in local communities. Being on the ground means they can connect directly with people, understand local needs, and give clear, practical help so people can get the care and information they need.
EQUITY, INCLUSION AND COLLABORATION
Elimination cannot be achieved without addressing health inequities. Hepatitis Australia and its members work alongside people with lived experience from all walks of life - including Aboriginal and Torres Strait Islander communities, people born overseas, people who inject drugs, and people in custodial settings, ensuring that every voice is heard and every community is supported.
LOOKING AHEAD
Hepatitis Australia remains focused on driving coordinated national action and strengthening the collective capacity of its members to achieve lasting change for people and communities affected by viral hepatitis.
We are committed to working in partnerships and would be pleased to discuss opportunities to partner with researchers to achieve both equity and impact, ensuring that no-one is left behind as we eliminate viral hepatitis for everyone.



SOUTHERN CROSS UNIVERSITY
SOUTHERN CROSS UNIVERSITY (SCU) IS A YOUNG AND AMBITIOUS UNIVERSITY WHOSE RESEARCH IS POWERED BY COLLABORATION AND DRIVEN BY IMPACT.


With campuses stretching from Coffs Harbour to the Gold Coast, our researchers combine regional insight with global reach to address some of the world’s most pressing challenges. We change lives through revolutionary learning and research that delivers real outcomes for people and communities.
While our research spans many disciplines, health and medical research is among our most connected and fastest-growing strengths. We bridge research and practice to create healthier people, stronger communities and more sustainable care – from Indigenous Australian women’s health to the science of functional foods and natural medicines.
HARVEST TO HEALTH IMPACT CLUSTER
Bringing together expertise in plant science, functional foods and health, the cluster develops sustainable natural products that promote wellbeing and reduce disease risk. From beekeeping for sustainable livelihoods in Indigenous communities across the Indo-Pacific, to clinical trials exploring the medicinal benefits of cannabis, our research translates local innovation into advances in global health.
CLINICAL RESEARCH LEADERSHIP
Located within the National Centre for Naturopathic Medicine – Australia’s first national centre dedicated to advancing evidence-based natural medicines and non-pharmacological interventions – the Clinical Trials Centre delivers Phase I–IV trials, pharmacokinetic studies and telehealth-enabled research. The
Centre’s strong links with rural, regional and remote communities position it as a national leader in expanding equitable access to clinical research. Strengthening health through research and practice
Our Faculty of Health brings together excellence across nursing, midwifery, osteopathy, psychology and sport and exercise science, supported by purpose-built facilities for simulation, clinical training and applied research.
Within our Ageing in Supportive Environments theme, researchers are pioneering a world-first Delirium Toolkit to help carers and clinicians better identify, prevent and manage delirium, one of the most common and costly conditions affecting older adults. This evidence-based innovation is already improving recovery outcomes and reducing hospitalisations in aged-care and community settings.
Projects such as SISTASCREEN , SISTABIRTH and ISISTAQUIT are transforming outcomes for First Nations women through culturally grounded, community-led programs focused on cervical cancer screening, maternal health and smoking cessation. Our Physical Activity, Sport and Exercise Research theme explores the future of health through sport, from traditional participation and elite performance to the emerging field of eSports health and wellbeing. Together, our researchers, clinicians and communities are shaping a healthier future within and beyond the regions we serve.
Students, Sophie Cotton and Val Slesarenko in the Biomedical Science lab
Building B Gold Coast campus. © Elise Derwin
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL
THE LAST WORD

AT THE TECHNOLOGYPOLICY NEXUS
Using health data to improve Australian health outcomes.
The use of health data has the potential to improve outcomes for millions of Australians.
And its application is varied, from identifying and responding to emerging health threats, through to preventing and managing chronic conditions. Yet researchers, innovators and policy makers all face challenges in using health data effectively, including determining if data already exist, and if so – who holds it, and how to access it.
Health data is generated in diverse settings and can include clinical, administrative, genomic, phenomic, therapeutic and imaging data types. Distributed across institutes and sectors, health data is often subject to different regulations.
To address these challenges, national data infrastructure initiatives strategically curate and mobilise datasets. In partnership with the research community and industry, the Australian Research Data Commons (ARDC) delivers digital research infrastructure – facilitating the creation, analysis and retention of high-quality data assets. By providing researchers with a competitive advantage through data, we are accelerating Australian research and innovation.
The ARDC is part of a network of national research infrastructure facilities, funded through the Australian Government’s National Collaborative Research Infrastructure Strategy (NCRIS). Collectively, we provide the technology, services and expertise that underpin
Australian research, translation and innovation – including the use of health data.
Through the ARDC’s Health Studies Australian National Data Asset (HeSANDA) program, resources such as Health Data Australia – a national catalogue of Australian clinical trials data – we are improving health research data access and collaboration. The resources, frameworks and governance delivered through these initiatives contribute to a more coherent overview of data for health research, but there is still work to be done.
KNOWING WHERE TO LOOK IS CRUCIAL –SO IS BEING ABLE TO BRING TOGETHER DIFFERENT TYPES OF DATA.
Research infrastructure is critical to the productivity of the health system, to health research and to medical innovation. Australia has led the world in data linkage units and national integrated data assets. The Population Health Research Network is a collaboration that securely links data across Australia to support population health research and improve health outcomes. With the Australian Institute of Health and Welfare and the Australian Bureau of Statistics, Australia has the necessary building blocks to advance health and medical research.
Just as important as bringing together data is making sure that it is interoperable. That is, the data can be understood and analysed together. As the volume and types of healthrelated data increase, sustained investment, strategic direction, and effective data governance are necessary to harmonise Australian health data.
Common data models, for example, make it feasible to ask important questions about the effectiveness of our

treatments, medicines and interventions. One example is OMOP, the Observational Medical Outcomes Partnership. To meet these needs, the ARDC is convening a national initiative – the Australian Health Data Evidence Network –distributed across ACT, NSW, QLD, SA and VIC, to adopt the globally recognised OMOP common data model for hospital electronic medical records (EMR) data.
INVESTMENT IN RESEARCH
INFRASTRUCTURE AND COLLABORATIVE PARTNERSHIPS MEANS AUSTRALIA IS WELL PLACED TO CAPITALISE ON TECHNOLOGICAL ADVANCES SUCH AS ARTIFICIAL INTELLIGENCE (AI).
High-quality, machine-actionable data – appropriately diverse and securely managed – is necessary to fully realise the benefits of AI. The application of AI to health data presents tremendous opportunities; from drug discovery, to clinical imaging, to personalised and even predictive medicine.
These technological shifts are changing infrastructure needs, creating new challenges requiring nationally coordinated governance models and resources. Through the Advanced Analytics and AI Resource Hub, the ARDC, in partnership with the Australian Data Science Network, is providing researchers with access to AI tools, training and infrastructure to support responsible, advanced analytics in health research.
TECHNOLOGICAL LEAPS CAN ALSO REVEAL INFRASTRUCTURE OR REGULATORY GAPS.
Around the world, governments, researchers and clinicians are exploring the application of AI to health



data assets. In parallel however, increasing cybersecurity threats and a volatile geopolitical landscape underscore the need for sophisticated data security approaches.
The privacy of patients and research subjects is not negotiable. The analysis of sensitive health data must be done securely, and in compliance with relevant regulations. For this reason, data custodians often require researchers to use trusted or secure environments for sensitive data analysis. Incorporating technical, governance and other safeguards, these environments enable secure collaborative analysis in ways that meet regulatory and ethical requirements.
The collaborative analysis of sensitive health data relies on fit-for-purpose infrastructure. Since 2012, the ARDC Nectar Research Cloud has provided national cloud computing resources to Australian researchers. As AI and other technologies rapidly evolve, so too do computing needs. A lack of sovereign and secure compute environments that meet regulatory and security standards limits health research and translation outcomes. To truly maximise the impact of Australian health data research, continued investment in underpinning and enabling infrastructure is vital.
Perhaps more than any other data type, the use of health data relies on trust; from patients whose information is used, to clinicians relying on tools, and across different jurisdictions. Governance of data and digital infrastructure are fundamentally linked. As technologies underpinning the generation and use of health data rapidly evolve, it is imperative that Australia’s data governance capability is continually strengthened to support the use of health data in research – ultimately ensuring the use of this data benefits the health and wellbeing of all Australians.
Author:
Rosie Hicks, CEO Australian Research Data Commons.
Rosie Hicks
CHRISTOPHE KEREBEL
CHRISTOPHE KEREBEL

