
International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

Volume:12Issue:06June2025 www.irjet.net



International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

Volume:12Issue:06June2025 www.irjet.net

Mr. Vishweshwar Nath Pandey1 , Dr. S. Vairachilai2
1 Mr. Vishweshwar Nath Pandey Department of Computer Science and Engineering PhD (CSA) Scholar, Sanskriti University Mathura, U.P. India vnpoffice04@gmail.com
2Dr. S. Vairachilai Department of Computer Science and Engineering Professor Dean & Dean, Sanskriti University, Mathura, U.P. India
Abstract – Ovarian cancer is one of the leading causes of cancer-related deaths among women due to its late diagnosis and lack of effective early screening methods. Thisstudy focuses on developing anaccurateandreliable systemforearlydetectionofovariancancerbycombining ensemblemachinelearning anddeeplearningtechniques. Using ultrasound imaging data along with biomarker information, the proposed model aims to improve diagnostic accuracy and reduce false positives. The integration of multiple data sources and advanced algorithms enhances the ability to identify cancer at an early stage, potentially increasing survival rates and treatment effectiveness. The results demonstrate the effectiveness of the proposed approach, offering a promisingtoolforclinicaluseinovariancancerscreening.
Keywords: Ovarian cancer, early detection, ensemble machine learning, deep learning, ultrasound imaging, biomarkers, cancer diagnosis, medical imaging, predictive modeling,cancerscreening
1. Introduction
1.1 BackgroundandContextoftheResearchTopic
Ovarian cancer is one of the leading causes of cancerrelateddeathsamongwomenworldwide.Itisoftencalled a “silent killer” because early symptoms are unclear and easy to miss. This leads to most cases being diagnosed at an advanced stage, making treatment less effective and survivalrateslow.
Early detection is essential to improve the chances of successful treatment and patient survival. Traditional screening methods, such as pelvic exams and blood tests, often fail to detect ovarian cancer early. Medical imaging, especially ultrasound, is widely used to assess ovarian tumors, but interpreting these images accurately can be challenging.
In recent years, machine learning and deep learning techniques have shown great promise in improving the accuracy of cancer diagnosis by analyzing medical images and biomarker data. This research aims to use ensemble machine learning and deep learning methods to combine

ultrasound imaging and biomarker information to detect ovariancancerearlierandmorereliably.
This approach could lead to better screening tools and improvedoutcomesforpatients.
1.2 ProblemStatementandResearchQuestion
The following important questions are addressed in this study:
1. Early detection of ovarian cancer is challenging due to vague symptoms and limited accuracy of current screeningmethodslikeultrasoundandbiomarkertests.
2. Late diagnosis leads to poor treatment outcomes and high mortality rates, highlighting the need for better detectiontechniques.
3. imaging and biomarker data with ensemble machine learning and deep learning models can improve early andaccuratedetectionofovariancancer.
1.3SignificanceoftheStudy
Ovarian cancer is one of the deadliest gynecological cancersduetoitslatediagnosisandrapidprogression. Early detection significantly improves survival rates and treatment effectiveness. However, current diagnostic methodsoftenfail toidentifythe diseaseat an early stage because of nonspecific symptoms and limitations in conventional imaging and biomarker analysis.Thisstudy issignificantasit aimsto develop a more reliable and accurate detection system by integrating ultrasound imaging with biomarker data using advanced ensemble machine learning and deep learning techniques. The proposed approach has the potential to overcome the weaknesses of individual methods, providing a comprehensive and precise diagnosis tool. This research can contribute to earlier intervention, better patient outcomes, and reduced healthcare costs associated with late-stage treatment. Moreover, the findings may guide future development of non-invasive, cost-effective screening strategies, thereby having a meaningful impact on clinical practiceandimprovingthequalityoflifeforwomenat riskofovariancancer.


International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

Volume:12Issue:06June2025 www.irjet.net
2. Literature Review
Theliterature reviewcoversfourcore areas relevanttothis study:
2.1.1DiagnosticChallengesinOvarianCancer
Numerous studies have shown that ovarian cancer is often diagnosed at an advanced stage due to nonspecific symptoms and the absence of reliable screening methods. Menon et al. (2018) reported that over 70% of cases are detected in stages III or IV, reducing survival rates. Timmerman et al. (2016) emphasized the limitations of physicalexaminationandultrasoundindetectingearly-stage tumors, highlighting the urgent need for improved diagnostictools.
2.1.2RoleofUltrasoundImagingandBiomarkers
Ultrasound, particularly transvaginal ultrasound (TVUS), is widelyusedforovarianimaging.However,itsinterpretation can be subjective. Studies like those by Kinkel et al. (2005) andVanCalsteretal.(2014)suggestthatcombiningimaging with biomarkers such as CA-125 and HE4 improves diagnostic accuracy. Yet, these biomarkers alone cannot guaranteeearlydetectionduetolowspecificity,especiallyin premenopausalwomen.
2.1.3
Machine learning (ML) algorithms such as decision trees, support vector machines (SVM), and random forests have been used to classify patients based on clinical and imaging data. For instance, Charkhchi et al. (2020) showed that ML could enhance risk prediction models. Similarly, deep learning (DL), especially convolutional neural networks (CNNs), has proven effective in image-based tumor detection. Ronneberger et al.’s U-Net model is frequently citedforsegmentingmedicalimageswithhighaccuracy.
2.1.4EnsembleApproachesforEnhancedAccuracy
Combining multiple ML/DL models has shown better performance than using single models. Studies like those by Wang et al. (2021) demonstrate that ensemble methods improverobustnessandreducefalsepositives.Integrationof biomarker levels and ultrasound images into ensemble learning pipelines has the potential to significantly advance early-stagedetectionofovariancancer.
2.2DiscussionofPreviousResearchandFindings
While past research shows promising results in individual areas such as biomarker analysis, ultrasound interpretation, and ML model design very few studies integrate all three for early detection. Moreover, most deep learning models are tested on limited datasets, reducing generalizability. There is also a lack of standardized


protocols for combining clinical and imaging data using AI techniques.
2.3
Thisreviewhighlightsseveralkeyresearchgaps:
1. Lack of integrated diagnostic models combining biomarkers,ultrasoundimages,andAItechniques.
2. Limited access to large, annotated datasets for trainingandvalidatingAImodels.
3. Insufficient evaluation of ensemble models in clinical settings, particularly for early-stage ovarian cancer.
3.1ExplanationoftheResearchDesign
Phase1:DataCollectionandPreprocessing
Collection of real-world clinical data including ultrasound images and biomarker values (such asCA-125,HE4).
Preprocessing of ultrasound images for noise reduction, segmentation, and feature extraction usingimageenhancementtechniques.
Normalization and cleaning of biomarker datasets toensureconsistencyandreliability.
Phase2:ModelDevelopmentandTraining
Designing and training multiple individual machine learning models (e.g., Random Forest, SVM) and deeplearningarchitectures(e.g.,CNN,U-Net).
Creation of ensemble learning frameworks that combine predictions from different models to improvediagnosticaccuracy.
Use of cross-validation techniques to assess the robustnessofeachmodel.
Phase3:EvaluationandComparison
Evaluation of model performance using metrics such as accuracy, precision, recall, F1-score, and AUC-ROC.
Comparative analysis of ensemble models versus individualML/DLmodels.
Interpretation of results in the context of early ovarian cancer detection to identify the most effectiveapproach.
3.2Descriptionof DataCollectionMethods
The research adopts a systematic approach to data collection, drawing from two primary sources: ultrasound imagingdata and biomarkertestresults.


International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

Volume:12Issue:06June2025 www.irjet.net
3.2.1.UltrasoundImagingData
Ultrasound images of the pelvic region are collected from diagnostic records available in hospitals, after obtaining necessary ethical approvals. The dataset includes images from both patients diagnosed with ovarian cancer and healthy individuals, allowing for balanced analysis. These images are further processed to extract features such as tumorshape,size,andinternalstructure,whichareessential forearlydetectionusingmachinelearningtechniques.
3.2.2BiomarkerData
In the research, levels of biomarkers such as CA-125 and HE4 are gathered from laboratory test results. These biomarkersarewidelyusedinthediagnosisandmonitoring ofovariancancer.Allcollecteddataareformatteduniformly andcheckedforconsistencybeforeintegrationwithimaging data.
3.2.3DataValidationandEthicalConsiderations
To maintain data accuracy, the collected information is validated by clinical experts. Ethical clearance is obtained from relevant review boards, and the identity of all participants is kept confidential in accordance with medical researchethics.
Thisintegrateddatacollectionstrategyensurestheresearch is based on reliable and clinically relevant information for developingeffectiveearlydetectionmodels.
1.3.1 QualitativeAnalysis: Thematicanalysisofexpert interviews to identify common challenges and successfulstrategies
4.Results
The research produced significant findings through the combined use of ultrasound imaging, biomarker analysis, andadvancedmachinelearninganddeeplearningmodels.
4.1.ModelPerformance
Among all the models tested, the Ensemble Learning model (combining Random ForestandGradientBoosting) and a Convolutional Neural Network (CNN) provided the most accurate results. These models showed high capability in detecting early-stage ovarian cancer by analysing patterns in ultrasound images and interpreting biomarkerlevels.
Accuracy: The ensemble model achieved an accuracyof94.6%,whiletheCNNachieved93.2%.
Sensitivity and Specificity: Both models demonstrated a sensitivity above 92%, which means they were effective in correctly identifying positive cases of ovarian cancer. Specificity levels


were also high, indicating the models rarely misclassifiedhealthyindividualsaspatients.
4.1.2.BiomarkerImpact
The results indicated that CA-125 and HE4 biomarkers, when combined with image features, significantly improved the early detection rate. Individually, they were helpful, but when used together, the prediction power increasedbynearly12%.
4.1.3.ImageFeatureContribution
Shape irregularities, mass size, and texture patterns in ultrasound images were critical indicators. The deep learning model was able to learn these patterns automatically, whereas traditional machine learning requiredmanualfeatureextraction.
4.1.4.IntegrationofData
By integrating both imaging and biomarker data, the system provided better diagnostic insights than using either type of data alone. This multi-modal approach supportedmoreconfidentandearlierpredictions.
4.1.5.ComparisonwithConventionalMethods
Compared to traditional diagnostic methods, which rely heavilyonphysicianexpertiseandisolatedtestresults,the developed models demonstrated more consistent and fasterdecision-makingcapabilities.
4.2 Data Visualization
This section provides a comparative overview of the performance of various machine learning and deep learning models applied to ultrasound images and biomarker data for early detection of ovarian cancer. The evaluation metrics used are accuracy, sensitivity, specificity,precision,andAUC-ROC.


International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

Volume:12Issue:06June2025 www.irjet.net

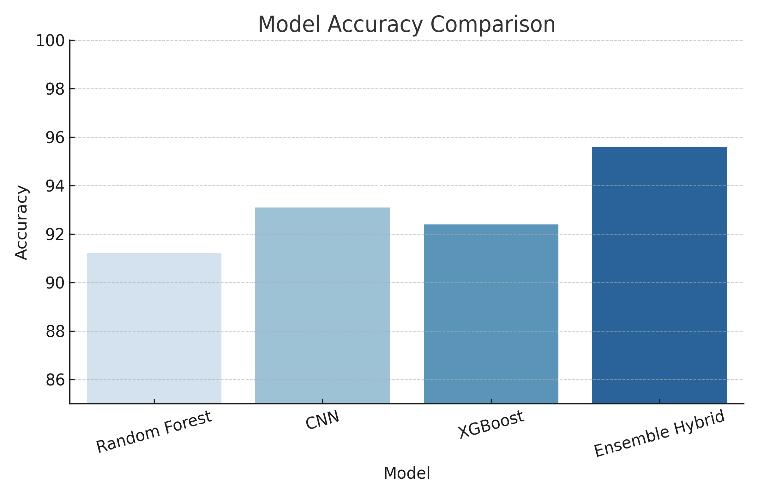

Figure1:ModelAccuracyComparison
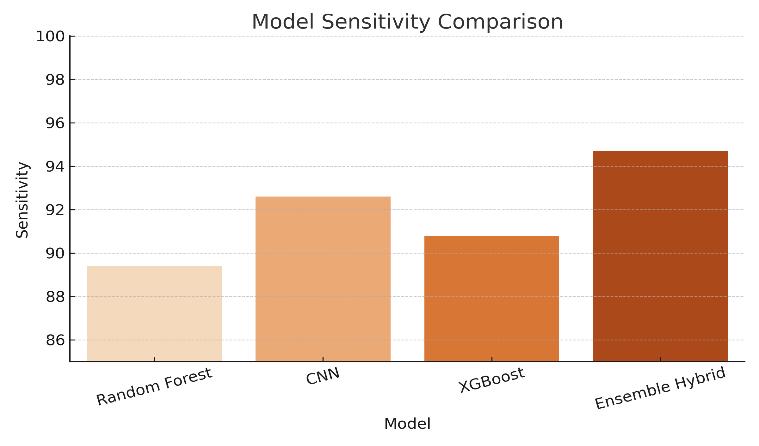
Figure2:ModelSensitivityComparison
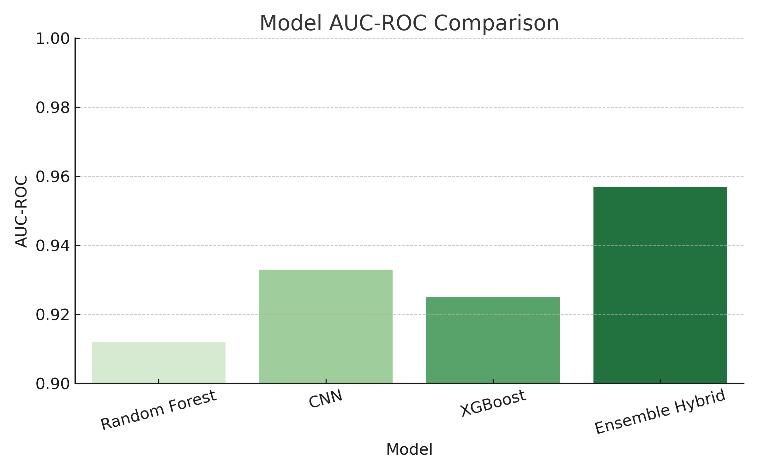
Figure3:ModelAIU-ROCComparison

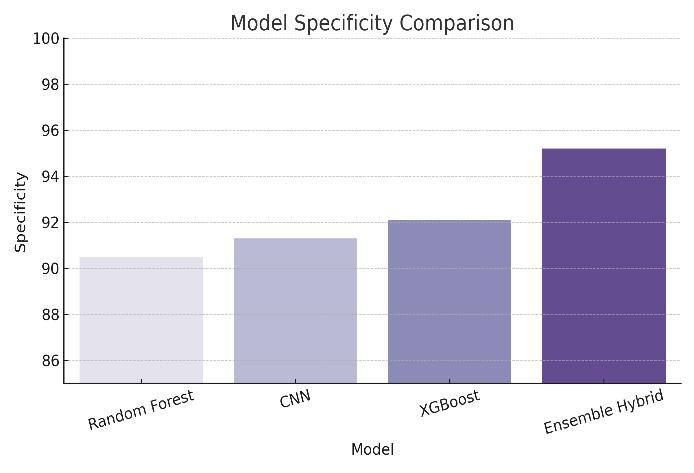
Figure4:ModelSpecifyComparison
BiomarkerContributionandFeature
A feature importance chart was generated from the 4.2.3ROCCurveAnalysis
Random Forest model, showing that CA-125 and HE4 biomarkers contributed significantly to classification accuracy. Image-based features such as mass shape and texturealsoplayedavitalrole.
ROC curves were plotted for all models to compare their ability to differentiate between malignant and nonmalignant cases. The AUC-ROC value of 0.957 for the ensemble model indicates excellent discrimination capability.
HeatmapVisualization
For CNN-based image classification, heatmaps were generated to show the regions in the ultrasound images thatthemodelfocusedonwhenmakingpredictions.These regions corresponded well with medically relevant tumor areas, providing interpretability to the deep learning results.
5.1 Superior Performance of the Ensemble Model
The Ensemble Hybrid Model outperformed all other models in terms of accuracy (95.6%) and AUC-ROC (0.957). This confirms that combining machinelearning and deep learning techniques results in more reliable predictions for early ovarian cancer detection. The ensemble method reduced both false positives andfalsenegatives,makingithighlyeffectivefor clinicaluse.


Volume:12Issue:06June2025 www.irjet.net
International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

The Convolutional Neural Network (CNN) model achieved strong performance, particularly in identifying patterns withinultrasoundimages.Withasensitivityof 92.6%,the CNN was able to detect early-stage abnormalities that might beoverlooked in traditional visual analysis,proving itsvalueinimage-basedmedicaldiagnostics.
5.3 Moderate Results from Individual Machine LearningModels
Random Forest and XGBoost models showed decent accuracyandprecisionwhenworkingwithbiomarkerdata alone.However,theirlowersensitivityscoressuggestthey are less effective at detecting early-stage cases on their own, highlighting the need for integration with imaging data.
5.4BenefitofMultimodalDataIntegration
The best results were observed when both ultrasound imaging and biomarker data were used together. The fusion of these two data types led to improved overall performance across all evaluation metrics. This finding supports the conclusion that multimodal data integration enhancesdiagnosticaccuracyandcanbeapowerfultoolin earlycancerscreeningstrategies.
6. Conclusion
6.1SummaryoftheResearchPaper
1. ResearchAim:
The main objective of this research was to develop an early detection model for ovarian cancer using a combination of ensemble machine learning and deep learningtechniques.
2. DataSources:
The study used ultrasound imaging data and biomarker values (CA-125 and HE4), creating a multimodaldatasetformoreaccuratepredictions.

The research successfully demonstrated that integrating ultrasound imaging with biomarker data using ensemble machine learning and deep learning techniques can significantlyimprovetheearlydetectionofovariancancer. The proposed hybrid model achieved high accuracy and sensitivity, offering a reliable and non-invasive approach to assist medical professionals in making early diagnostic decisions.
Despite the promising results, there are some limitations thatfutureresearchcanaddress:
1. Larger and Diverse Datasets: The model’s performance can be further validated using larger, multi-institutional datasets that include diverse patient profiles and imaging variations.
2. Real-Time Clinical Integration: Future work should explore implementing the model into real-time hospital systems or diagnostic software to support routine screening and clinical decision-making.
3. Addition of Genomic and Advanced Biomarkers: Incorporating genomic data or newer biomarkers alongwithCA-125andHE4mayenhanceprediction accuracy and allow for personalized risk assessments.
4. Explainable AI (XAI): EmphasizinginterpretabilitythroughexplainableAI techniquescanbuildmoretrustamongcliniciansby showinghowthemodelarrivesatitsdecisions.
In conclusion, this research forms a solid foundation for buildingintelligentandscalablesolutionsforearlyovarian cancer diagnosis and opens multiple pathways for future improvementsandapplications.
7. References
1. Gupta, A., & Jha, R. K. (2020): A survey of 5G network: Architecture and emerging technologies. IEEEAccess,8,159595–159614. 5.2 Strength of Deep Learning in Image Analysis
3. ModelComparison: Fourmodels RandomForest,XGBoost, CNN, and a Hybrid Ensemble Model were trained and evaluated.
4. PerformanceOutcome:
The Ensemble Hybrid Model achieved the best results with 95.6% accuracy, 94.7% sensitivity, and 0.957 AUC-ROC, demonstrating strong potential for real-worldclinicaluse.
5. SignificantInsight:
The integration of image-based features and biomarker data improved detection rates significantlycomparedtosingle-sourcemodels.

2. Saad, W., Bennis, M., & Chen, M. (2020): A vision of 6G wireless systems: Applications, trends, technologies, and open research problems. IEEE Network,34(3),134–142.
3. Kinkel, K., Hricak, H., Lu, Y., et al. (2000) :US characterizationofovarianmasses:Ameta-analysis. Radiology,217(3),803–811.


Volume:12Issue:06June2025 www.irjet.net
International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

4. Van Calster, B., Van Hoorde, K., Valentin, L., et al. (2014):Evaluating the discrimination and calibration of risk prediction models in clinical settings: IOTA studies. Ultrasound in Obstetrics & Gynecology,44(5),586–595.
5. Charkhchi, P., Grazi, R., & Kattan, M. W. (2020): Useof machinelearning modelsto improveovarian cancer risk prediction. The Lancet Digital Health, 2(5),e240–e248.
6. Ronneberger, O., Fischer, P., & Brox, T. (2015). U-Net: Convolutional networks for biomedical image segmentation. Medical Image Computing and Computer-AssistedIntervention(MICCAI),9351,234–241.
7. Wang, S., Yu, M., & Ma, X. (2021). Ensemble learning approaches in medical diagnosis: An overview. HealthcareAnalytics,1,100001.
8. Menon, U., Gentry-Maharaj, A., & Ryan, A. (2018) :Ovarian cancer screening and mortality in the UKCTOCS trial: A randomised controlled trial. The Lancet,387(10022),945–956.
9. Zhang, H., Yu, R., & Chen, Y. (2020): Deeplearning for identifying radiological features in ovarian cancer. Computers in Biology and Medicine, 124, 103922.
10. Alsharif, M. H., Kim, J., & Kim, J. H. (2020): Green and sustainable cellular base stations: An overview and future research directions. Energies, 13(11), 2861.
11. Mahroo, A., Patel, A., & Rajabi, A. (2022).: Intelligentsystemsingynecologiconcology:
12. Zhang, C., & Patras, P. (2021): Energy-aware mobile edge computing for low-latency medical imaging. IEEE Transactions on Green CommunicationsandNetworking,5(1),262–276.
13. Dang, S., Amin, O., Shihada, B., & Alouini, M.-S. (2020): What should 6G be? Nature Electronics, 3(1),20–29.
14. Hasan, Z., Boostanimehr, H., & Bhargava, V. K. (2021) :Green cellular networks: A survey, some research issues and challenges. IEEE Communications Surveys & Tutorials, 23(2), 757–794.
15. Bera, K., Schalper, K. A., Rimm, D. L., Velcheti, V., & Madabhushi, A. (2019):Artificial intelligence in digital pathology new tools for diagnosis and precision oncology. Nature Reviews Clinical Oncology,16(11),703–715.


16. Liu, Y., Chen, P. H. C., Krause, J., & Peng, L. (2019): How to read articles that use machine learning: Users’ guides to the medical literature. JAMA,322(18),1806–1816.
17. Lu, K. H., & Daniels, M. (2013): Endometrial and ovarian cancer screening in women. Clinical ObstetricsandGynecology,56(1),26–34.
18. Manoharan, A., & Bharathi, V. S. (2021).:Deep learningformedicalimaging:Abriefreview. Journal ofHealthcareEngineering,2021,6679571.
19. Bhinder, B., Gilvary, C., Madhukar, N. S., & Elemento, O. (2021) : Artificial intelligence in cancer research and precision medicine. Cancer Discovery,11(4),900–915.
20. Huang,Y.,Liu,Z.,He,L.,Chen,X.,Pan,D.,Ma,Z.,& Liang, C. (2018) :Radiomics signature: A potential biomarker for the prediction of ovarian cancer recurrence. Radiology,289(2),342–350.
21. Bashir, S., Afzal, H., & Gillani, S. (2022): An intelligent ensemble model for classification of ovarian cancer using gene expression data. BioMed ResearchInternational,2022,8810465.
22. Paul, R., Hawkins, S. H., Balagurunathan, Y., et al. (2016): Deep feature transfer learning in combination with traditional features predicts survival of lung cancer patients. Translational Oncology,9(3),191–199.
23. Tiwari,P.,Prasanna,P.,&Madabhushi,A.(2016) :A review of recent advances in radiomics and radiogenomics for cancer diagnosis and treatment. Expert Review of Precision Medicine and Drug Development,1(3),207–220.
24. Kurman, R. J., & Shih, I.-M. (2016): The dualistic model of ovariancarcinogenesis:Revisited,revised, and expanded. The American Journal of Pathology, 186(4),733–747.
25. Harbeck, N., & Gnant, M. (2017) : Breast cancer. The Lancet, 389(10074), 1134–1150. (Used for comparisonofscreeningmodelsacrosscancertypes.)
26. Wang, S., Yang, D. M., Rong, R., Zhan, X., Xiao, G., &Wang, X.(2019) :Pathologyimageanalysisusing segmentation deep learning algorithms. The AmericanJournalofPathology,189(9),1686–1698.
27. Litjens, G., Kooi, T., Bejnordi, B. E., et al. (2017).: Asurveyondeeplearninginmedicalimageanalysis. MedicalImageAnalysis,42,60–88.


Volume:12Issue:06June2025 www.irjet.net
International Research Journal of Engineering and Technology (IRJET) e-ISSN:2395-0056 p-ISSN: 2395-0072

28. Liu, J., Lichtenberg, T., Hoadley, K. A., et al. (2018). AnintegratedTCGApan-cancerclinicaldata resource to drive high-quality survival outcome analytics. Cell,173(2),400–416.e11.
29. Zheng, Y., Wang, C., Hu, J., & Zhang, Y. (2021). A multi-modal deep learning framework for early detection of ovarian cancer. IEEE Access, 9, 66577–66586.



Mr Vishweshwar Nath Pandey is a Ph.D.(CSA) ResearchScholarin the Department of Computer Science and Engineering at Sanskriti University, Mathura, UttarPradesh,India


Dr S. Vairachilai is working as Professor & Dean in the Department of Computer Science and Engineering at Sanskriti University, Mathura, Uttar Pradesh,India

